Abstract
Both CB1 receptor antagonism and agonism, in particular by 2-arachidonyl glycerol (2-AG), have been shown to reduce somatic symptoms of morphine withdrawal (MWD). Here we evaluated the effects of both systemic pretreatment with the monoacylglycerol lipase (MAGL) inhibitor MJN110 (which selectively elevates 2-AG) and central administration of both MJN110 and the CB1 antagonist (AM251) on the affective properties of MWD. Acute MWD induced place aversion occurs when naloxone is administered 24 h following a single exposure to a high dose of morphine. Systemic pretreatment with the MAGL inhibitor, MJN110, prevented the aversive effects of acute MWD by a CB1 receptor-dependent mechanism. Furthermore, in a double dissociation, AM251 infusions into the central amygdala, but MJN110 infusions into the basolateral amygdala, interfered with the naloxone-precipitated MWD induced place aversion. As well, MJN110, but not AM251, infusions into the interoceptive insular cortex (a region known to be activated in acute MWD) also prevented the establishment of the place aversion by a CB1 mechanism of action. These findings reveal the respective sites of action of systemically administered MJN110 and AM251 in regulating the aversive effects of MWD.
INTRODUCTION
Withdrawal from opiates has been shown to be a driving force in the maintenance of opiate addiction (eg, Koob, 2009a). In animal models, morphine withdrawal (MWD) can be produced by terminating chronic exposure to morphine or by administering an opiate antagonist to morphine pretreated animals. Indeed, MWD symptoms can be observed in both humans (Heishman et al, 1990; June et al, 1995) and other animals (Eisenberg, 1982; Martin and Eades, 1964) when naloxone is administered several hours following a single exposure to a high dose of morphine. The withdrawal is apparent not only by behavioral symptoms of abstinence, but also by the ability of such withdrawal to serve as an aversive motivational stimulus. Parker et al (2002) demonstrated that the aversive properties of naloxone-precipitated MWD were evident up to 48 h after a single injection of morphine, but not saline, in a conditioned place aversion (CPA) paradigm.
There is growing evidence that activation of the endocannabinoid system (eCB) may ameliorate symptoms of opiate addiction. The eCB system consists of two receptors (CB1 and CB2), the eCBs, anandamide (AEA; Devane et al, 1992) and 2-arachidonyl glycerol (2-AG; Sugiura et al, 1995) and the enzymes that regulate their synthesis and degradation (Ahn et al, 2008). CB1 antagonism interferes with a naloxone-precipitated MWD CPA (Wills et al, 2014) and with somatic symptoms of MWD in rats (Mas-Nieto et al, 2001; Rubino et al, 2000). However, somewhat paradoxically THC (Bhargava, 1976), AEA (Vela et al, 1995), and 2-AG (Yamaguchi et al, 2001) have also been reported to reduce the intensity of MWD somatic symptoms in mice. When systemically administered, AEA and 2-AG are rapidly degraded by fatty acid amide hydrolase (FAAH; Cravatt et al, 1996) and monoacylglycerol lipase (MAGL; Dinh et al, 2002), respectively. Blocking these catabolic enzymes produces a prolonged elevation of the respective eCB. Ramesh et al (2011) reported that both FAAH (PF-3845) and MAGL (JZL184) inhibitors were effective in attenuating precipitated MWD somatic symptoms in mice; although the MAGL inhibitor was more effective than was the FAAH inhibitor. Although FAAH inhibition does not significantly modify the establishment of a naloxone-precipitated MWD CPA (Wills et al, 2014), the potential of MAGL inhibition to interfere with the affective component of acute naloxone-precipitated MWD has not been evaluated in rats. Recently, a potent and selective MAGL inhibitor, MJN110 (Niphakis et al, 2013), has been developed that selectively elevates 2-AG by 10-fold, but not AEA. In rat brains, doses of 5 mg/kg (i.p.) and greater, produced maximal inhibition of MAGL for up to 12 h following administration. MJN110 also inhibits the alternative 2-AG hydrolase, ABHD6, but with ~300-fold lower potency than MAGL. MJN110 has been shown to inhibit both acute and anticipatory nausea in rat gaping models (Parker et al, 2014) and to alleviate mechanical allodynia in a rat model of diabetic neuropathy (Niphakis et al, 2013).
The experiments reported here evaluated the potential of systemic administration of MJN110 and central administration of both MJN110 and the CB1 antagonist, AM251, to interfere with the affective properties of MWD in rats. The aim was to determine the site of action of agonism and antagonism of the eCB system in regulating a naloxone-precipitated MWD CPA. Considerable evidence implicates the amygdala in the neurocircuitry of the negative reinforcement associated with MWD (eg, Koob, 2009a). Therefore, we evaluated the potential of intracranial administration of MJN110 and AM251 into both the basolateral amygdala (BLA) and the central nucleus of the amygdala (CeA) to interfere with the aversive affective effects of MWD. As well, both the BLA and the CeA receive input from the interoceptive insular cortex (IC; McDonald, 1998; McDonald et al, 1999), which also has been implicated in both addiction and nausea processes (Contreras et al, 2007). As CB1 agonism in the BLA (Ganon-Elazar and Akirav, 2009), but CB1 antagonism in the CeA (Zarrindast et al, 2008), produces stress-relieving and anxiolytic effects in rats in aversive environments, we predicted that MJN110 in the BLA, but AM251 into the CeA, would reduce the aversive properties of MWD. Inactivation of the interoceptive IC has recently been demonstrated to prevent the acquisition of a naloxone-precipitated MWD CPA in rats (Li et al, 2013). Therefore, we also predicted that elevation of the regulatory neurotransmitter 2-AG by intracranial administration of MJN110 into the interoceptive IC (Allen et al, 1991; Cechetto and Saper, 1987; Contreras et al, 2007) would also interfere with the establishment of a naloxone-precipitated MWD CPA.
MATERIALS AND METHODS
Subjects
Subjects were male Sprague–Dawley rats weighing between 350 and 450 g. Animals were housed individually as described in Wills et al (2014). All animal procedures were approved by the Animal Care Committee of the University of Guelph and adhere to the guidelines of the Canadian Council of Animal Care.
Drugs
Morphine, naloxone, and vehicle (VEH) were prepared as previously described in Wills et al (2014). For the systemic experiments (experiments 1 and 2), the MAGL inhibitor, MJN110, and CB1 antagonist, AM251, were prepared in VEH at a final concentration of 10 mg ml−1 and 1 mg ml−1, respectively. The concentration of MJN110 was selected on the basis of its ability to maximally inhibit MAGL activity in rats in vivo (Niphakis et al, 2013), whereas the concentration of AM251 has been previously found to have no effect on the establishment of a naloxone-precipitated CPA (Wills et al, 2014). For the intracranial experiments (experiments 3 and 4), MJN110 and AM251 were prepared in the same VEH at a concentration of 5 and 0.25 μg/μl, respectively; for experiment 5, MJN110 and AM251 were prepared in the same VEH at a concentration of 2 and 0.1 μg/μl, respectively.
Surgery
For experiments 3–5, rats were anesthetized with isoflurane gas and prepared for intracranial surgery as described in detail in Limebeer et al (2012). Once rats were stabilized in the stereotaxic frame in the flat skull position (Paxinos and Watson, 1998), small bilateral holes were drilled into the exposed skull and stainless steel guide cannulas (22 G, 6 or 8 mm below pedestal) were lowered into the CeA (experiment 3), BLA (experiment 4) or the interoceptive IC (experiment 5) using the following coordinates relative to Bregma, CeA: −2.2 mm anteroposterior (AP), +4.3 mm mediolateral (ML), and –6.0 mm dorsoventral (DV) from the skull surface; BLA: −2.3 mm AP, +5.0 mm ML, −6.5 mm DV from the skull; interoceptive IC (10° divergent angle): −0.5 mm AP;+ 5.0 mm ML; –4.5 mm DV from the skull. The guide cannulas were stabilized to the skull using six screws and dental cement. Once the dental cement hardened, stainless steel dummies were inserted into the guide cannulas to prevent obstruction.
Histology
Guide cannula placements in experiments 3–5 were determined through the histological examination of brain tissue as described in detail in Limebeer et al (2012). Before perfusion, rats were microinfused with Chicago blue dye to verify diffusion of the drug was localized to the CeA, BLA, and interoceptive IC; analyses revealed an average spread of 0.35 mm in the CeA and BLA, and 0.75 mm in the interoceptive IC in each of the AP, ML, DV coordinate planes. Following, rats were deeply anesthetized with a lethal dose of Euthansol (85 mg/kg i.p.) and transcardially perfused with PBS buffer (0.1 M) followed by 4% formalin. Brains were removed and stored in a 20% sucrose and 4% formalin solution overnight at room temperature, after which they were preserved at a temperature of 4 °C until sectioned. Brains were frozen and sliced into 60 μm sections using a CM1850 Leica cryostat and relevant slices were mounted onto gelatin-subbed glass microscope slides. Slide tissue sections were then stained with thionin, cover-slipped, and examined using a Leica MZ6 Stereomicroscope. Rats with improper cannula placement were excluded from the study. All n's reported in the manuscript reflect the post-histology numbers per group.
Apparatus
The conditioning apparatus was a plain black rectangular box as previously described in Wills et al (2014) with chambers differentiated solely by the floor texture. Removable floors were used to transition the boxes from conditioning cycles to pretest/test trials. During conditioning, single black metal floors made of a grid or hole pattern were used as contextual cues. During pretest and test trials, split black metal floors equally divided into a half grid/half hole pattern were used. Ethovision software was used to define box perimeters and assign a neutral floor zone between the two floors for pretest and test trials.
Procedure
All procedures are as described in detail in Wills et al (2014). Before all experiments, rats received a 15-min pretest. Rats were assigned to a pretreatment drug group and drug floor matched on the basis of initial pretest preferences. Rats with a bias of more than 200 s for either floor were removed from the experiment. In all experiments, Ethovision software tracked activity and measured the time spent on each floor.
Experiment 1: Potential of systemic MJN110 to produce a CPP or a CPA
The rats (n=8) were injected with 10 mg/kg MJN110 on one day and VEH on another day (24 h apart; counterbalanced order) 2 h before placement in the conditioning box with the grid or the hole floor (counterbalanced) for 45 min. Twenty-four hours after the final conditioning day of the single conditioning trial, the rats were given daily 15 min test trials with the split grid/hole floor for 3 days. On each test, rats received an i.p. injection of VEH 2 h before being placed in the box.
Experiment 2: Effect of systemic MJN110 and the interaction of systemic MJN110 and AM251 on the establishment of a MWD CPA
A three-day conditioning cycle was used to obtain the MWD CPA. On the first day, the floor opposite the assigned drug floor was paired with a s.c. saline injection (saline floor). The rats were administered an i.p. injection of VEH (n=12), 10 mg/kg MJN110 (n=11) or 10 mg/kg MJN110+1 mg/kg AM251 (n=12), 2 h (MJN110 or VEH) or 30 min (AM251) before the saline injection. The inclusion of a group treated with AM251 alone was omitted based on our previous findings reporting no significant effect of 1 mg/kg AM251 on the establishment of a MWD CPA (Wills et al, 2014). Ten minutes after the saline injection, the rats were placed in the conditioning chamber for 45 min. On the second day, 24 h post saline injection, all rats received a high dose of morphine (20 mg/kg, s.c.) in an empty shoebox cage. The rats were monitored for signs of respiratory depression and stimulated when required to prevent decease (<5% mortality rate). Finally, on the third day, 24 h post morphine, the assigned drug floor was paired with an injection of naloxone (MWD floor). As on the saline trial, the rats received VEH, MJN110, or MJN110+AM251, 2 h (MJN110 or VEH) or 30 min (AM251) before the naloxone injection (1 mg/kg), which occurred in the conditioning room 10 min before being placed in the conditioning box for 45 min. The final groups were: VEH (n= 12), MJN110 (n=11), and MJN110+AM251 (n=12). Five days later, rats were given daily 15 min tests as described above, with the exception that rats received a s.c. injection of saline 10 min before being placed in the box.
Experiment 3: Effect of MJN110 and AM251 on the establishment of a MWD CPA when delivered to the CeA
As in experiment 2, a three-day conditioning cycle was used to obtain the MWD CPA. One hour before both the saline conditioning trial (day 1) and the MWD conditioning trial (day 3), rats received bilateral microinfusions of VEH (n=8), 2 μg of MJN110 (n=10), or 0.1 μg of AM251 (n=6) into the CeA at a rate of 0.2 μl/min for 2 min. Injector tips were inserted to extend 2 mm beyond the length of the guide cannula. Following the two-min infusion period, the injectors were left in place for an additional minute to ensure full diffusion of the drug from the injector. Rats were injected with saline (day 1) or naloxone (day 3) 10 min before being placed in the conditioning boxes for 45 min. Beginning 5 days after conditioning, rats received daily 15 min tests as described above.
Experiment 4: Effect of MJN110, AM251, and MJN110–AM251 on the establishment of a MWD CPA when delivered to the BLA
The procedures were identical to those of experiment 3 except that VEH (n=8), 2 μg MJN110 (n=10), 0.1 μg AM251 (n= 7), and 2 μg MJN110–0.1 μg AM251 (n=8) were bilaterally infused into the BLA.
Experiment 5: Effect of MJN110, AM251, and MJN110–AM251 on the establishment of a MWD CPA when delivered to the interoceptive IC
The procedures were identical to those of experiment 3 except that VEH (n=10), 2 μg MJN110 (n=8), 0.1 μg AM251, and 2 μg MJN110–0.1 μg AM251 were bilaterally infused into the interoceptive IC at a rate of 0.5 μl/min for 2 min.
Data Analysis
In each experiment, the time (s) spent on each floor during each of the three test trials was entered into a three-factor mixed design, with the between group factor of pretreatment drug and the within group factors of floor (saline-paired or MWD-paired) and test trial. As well, the activity measures during conditioning trials were entered into a mixed factors analysis of variance (ANOVA) with the between groups factor of pretreatment drug and the within-groups factor of conditioning trial (saline or MWD) for each experiment. Significance was set at p<0.05.
RESULTS
Experiment 1: MJN110 Does Not Produce a CPP or CPA
Single trial place conditioning with MJN110 did not produce a significant preference or aversion for the drug paired floor, F (1, 7)=0.21, NS. Pooled across test trials, rats spent an equal amount of time on the VEH paired floor (M=411.32 s±42.19 SEM) and the MJN110 paired floor (M=373.96 s,±39.98 SEM), and the order of conditioning (VEH first vs MJN110 first) did not significantly alter this effect. An evaluation of activity during both conditioning trials revealed no motoric effects of MJN110 compared with VEH.
Experiment 2: Systemic MJN110 Interferes With the Establishment of a MWD CPA, Which is Reversed by AM251
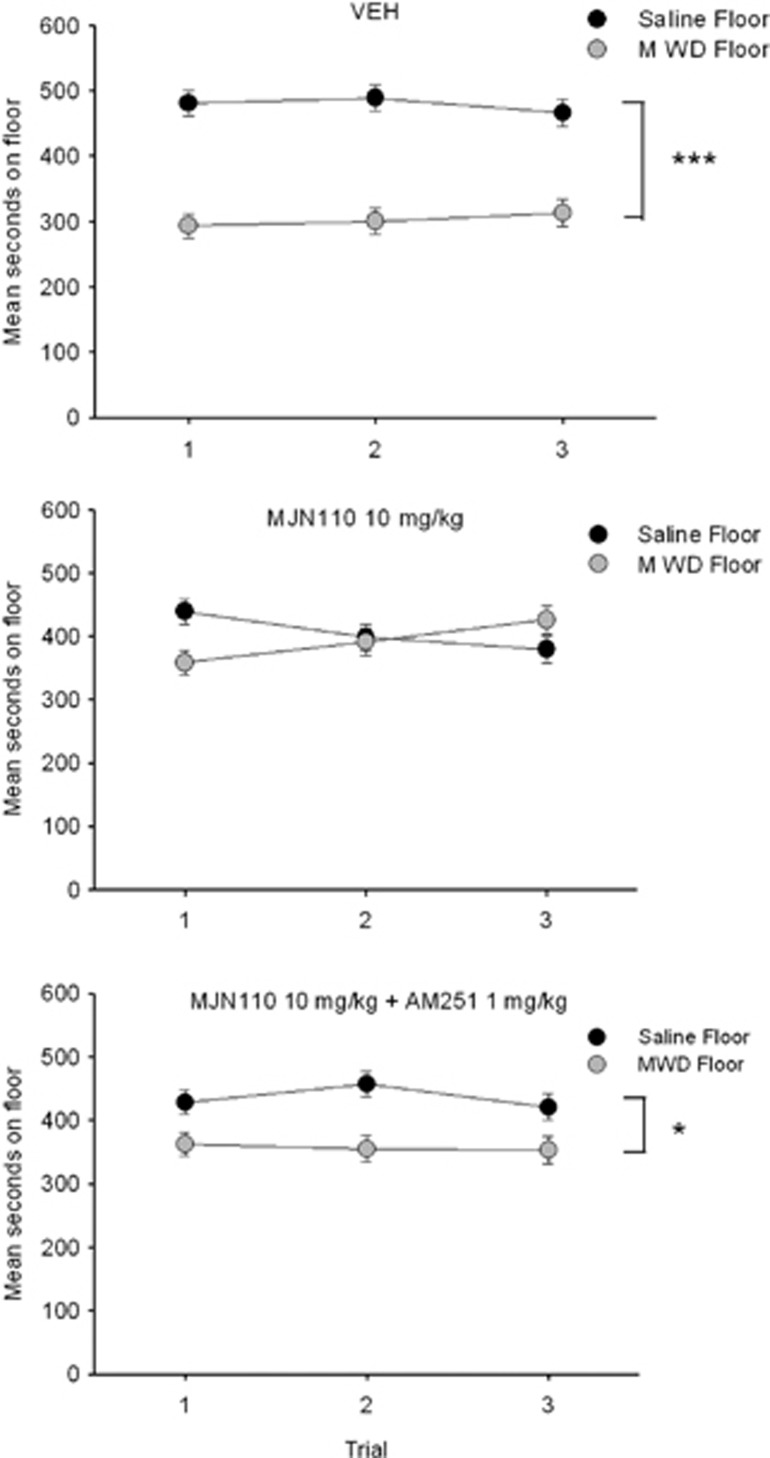
As seen in Figure 1, MJN110 significantly interfered with the establishment of the naloxone-precipitated MWD CPA, an effect that was reversed by the CB1 antagonist AM251.The ANOVA revealed a significant floor by drug interaction, F (2, 32)=7.4; p=0.002. Rats pretreated with VEH (p<0.001), or MJN110+AM251 (p=0.017), but not MJN110 alone, spent significantly less time on the MWD-paired floor than the saline-paired floor. Evaluation of activity during the conditioning trial revealed significant effects of trial, F (1, 32)=273.3, p<0.001; drug, F (2, 32)=11.4, p<0.001; and a significant trial by drug interaction, F (2, 32)=8.0, p<0.01. Overall, rats were significantly less active during MWD conditioning than saline conditioning. In addition, pretreatment with MJN110+AM251 significantly reduced activity compared with VEH and MJN110 on the saline trial and compared with VEH on the MWD trial. Group MJN110 did not differ from VEH on any trial.
Figure 1.
Mean (±SEM) time spent in seconds on the saline-paired floor and the MWD-paired floor for each pretreatment drug group (VEH, 10 mg/kg MJN110, 10 mg/kg MJN110+1 mg/kg AM251 during conditioning) during each 15 min test trial. Asterisks indicate a significant difference between the saline and morphine withdrawal paired floors. *p<0.05, ***p<0.001.
Experiment 3: AM251 Delivered to the CeA Interferes with the Establishment of a MWD CPA
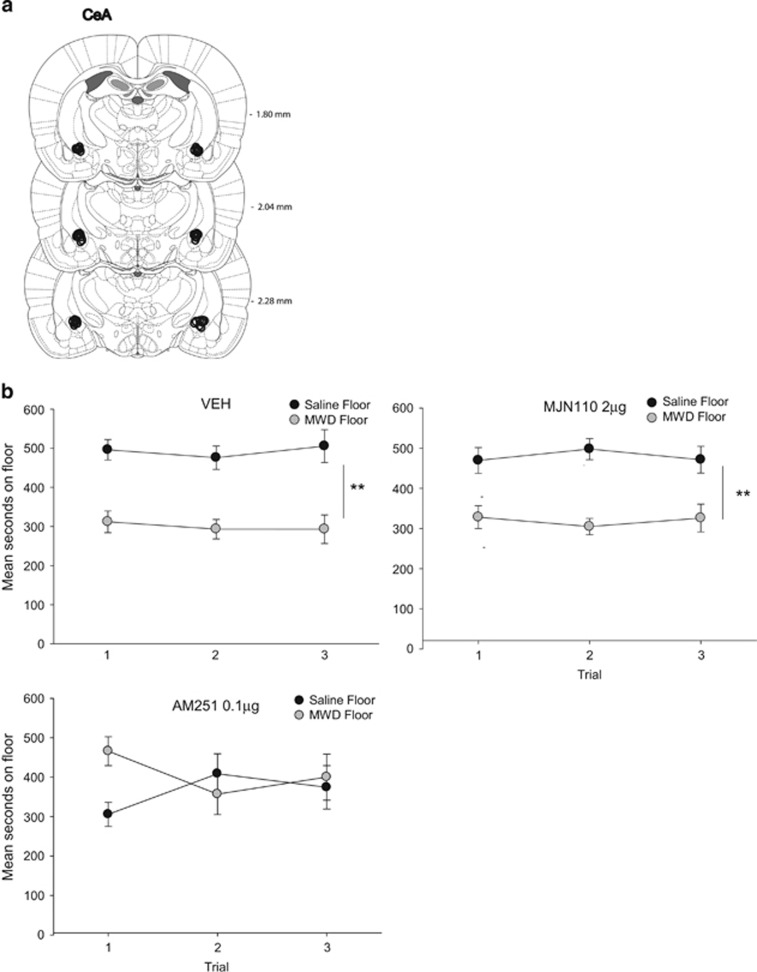
As seen in Figure 2, central administration of AM251 into the CeA interfered with the establishment of a naloxone-precipitated MWD CPA. The ANOVA revealed a significant floor × drug interaction, F (2, 21)=4.6, p=0.02. VEH- and MJN110-treated rats (p's<0.01), but not the AM251-treated rats, spent significantly less time on the MWD floor than the saline floor, pooled across tests. Analysis of the mean distance traveled during conditioning revealed only a significant effect of trial, F (1, 21)=116.8, p<0.001; rats were significantly less active during the MWD trial than the saline trial. There was no effect of MJN110 or AM251 on activity on either the saline or MWD conditioning trial.
Figure 2.
(a) CeA cannula placements for all rats included in the experiment. (b) Mean (±SEM) time spent in seconds on the saline-paired floor and the MWD-paired floor for each pretreatment drug group (VEH, 2 μg MJN110, 0.1 μg AM251 into the CeA during conditioning) during each 15 min test trial. Asterisks indicate a significant difference between the saline and morphine withdrawal paired floors. **p<0.01.
Experiment 4: MJN110 Delivered to the BLA Interferes with the Establishment of a MWD CPA
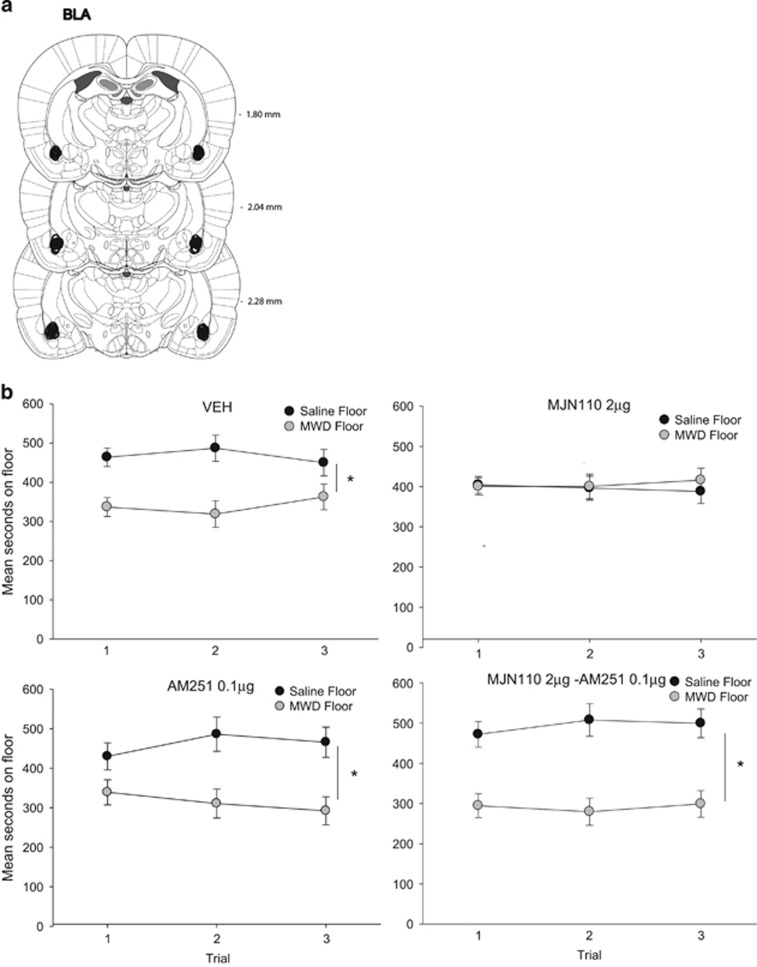
As seen in Figure 3, central administration of MJN110 into the BLA interfered with the establishment of a naloxone-precipitated MWD CPA and this effect was reversed by co-administration with AM251. The ANOVA revealed a significant floor by drug interaction, F (3, 29)=3.1, p=0.04. Rats administered VEH, AM251, and MJN110–AM251 but not MJN110, bilaterally to the BLA 1 h before conditioning spent significantly less time on the MWD paired floor than the saline floor (p's<0.05). Evaluation of conditioning activity, revealed a significant effect of trial, F (1, 29)=56.4, p<0.001. Overall, rats were significantly less active on the MWD conditioning trial compared with the saline conditioning trial and drug pretreatment did not alter this on either trial.
Figure 3.
(a) BLA cannula placements for all rats included in the experiment. (b) Mean (±SEM) time spent in seconds on the saline-paired floor and the MWD-paired floor for each pretreatment drug group (VEH, 2 μg MJN110, 0.1 μg AM251, 2 μg MJN110–0.1 μg AM251 into the BLA during conditioning) during each 15 min test trial. Asterisks indicate a significant difference between the saline and morphine withdrawal paired floors. *p<0.05.
Experiment 5: MJN110 Delivered to the Interoceptive IC Interferes with the Establishment of a MWD CPA
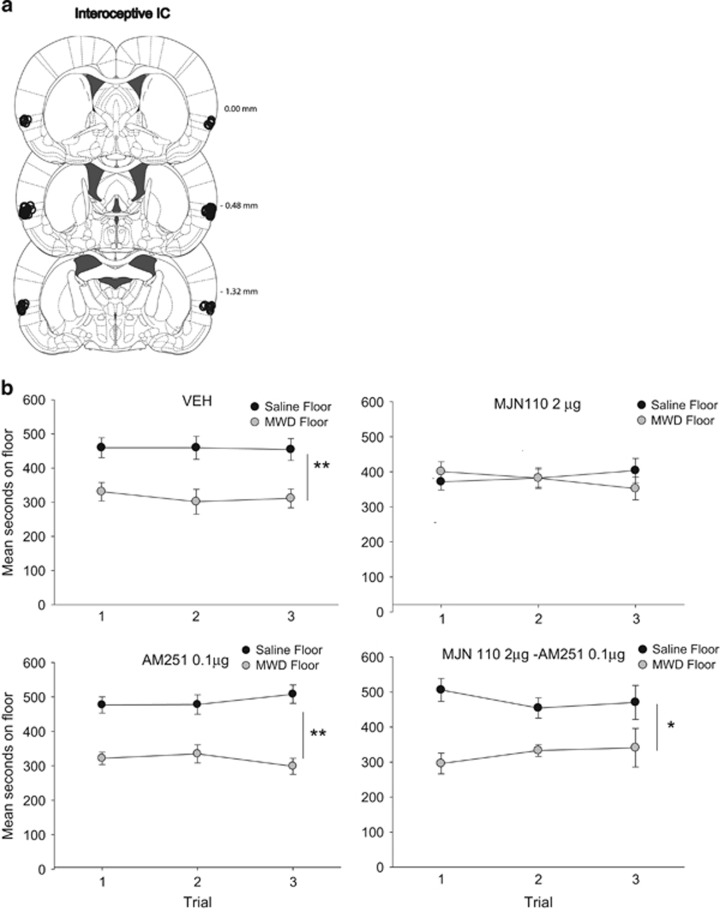
As seen in Figure 4, MJN110 administered to the interoceptive IC interfered with the naloxone-precipitated MWD CPA and this effect was reversed by co-administration with AM251. The ANOVA revealed a significant floor × drug interaction, F (3, 29)=3.6, p=0.03. Pooled across trials, VEH, AM251, and MJN110–AM251 displayed a significant aversion for the MWD paired floor (p's<0.05), but group MJN110 did not show an aversion. Evaluation of activity during conditioning revealed a significant effect of trial, F (1, 29)=156.1, p<0.001; drug, F (3, 29)=4.1, p=0.02; and a significant trial by drug interaction, F (3, 29)=4.5, p=0.01. Overall, rats were significantly less active on the MWD trial than the saline trial. Furthermore, rats treated with AM251 or MJN110–AM251 were significantly less active on the saline trial, but not the MWD trial, than rats that were treated with VEH or MJN110.
Figure 4.
(a) Interoceptive IC cannula placements for all rats included in the experiment. (b) Mean (±SEM) time spent in seconds on the saline-paired floor and the MWD-paired floor for each pretreatment drug group (VEH, 2 μg MJN110, 0.1 μg AM251, 2 μg MJN110–0.1 μg AM251 into the interoceptive IC during conditioning) during each 15 min test trial. Asterisks indicate a significant difference between the saline and morphine withdrawal paired floors. *p<0.05, **p<0.01.
DISCUSSION
Systemic administration of the MAGL inhibitor, MJN110, prevented the establishment of a naloxone-precipitated MWD CPA, an effect that was reversed by the CB1 antagonist AM251. The ability of MJN110 to interfere with the CPA is not due to rewarding properties of the inhibitor per se, as MJN110 produced neither a place preference nor aversion. As well, intracranial administration of MJN110 (2 μg, bilaterally) into the BLA and into the interoceptive IC, central areas implicated in the negative effects of dependence (Contreras et al, 2007; Koob, 2009b; Li et al, 2009, 2013), also prevented the naloxone-precipitated MWD CPA. Although rats were less active on the MWD conditioning trial than on the saline conditioning trial in each experiment, MJN110 did not modify their activity relative to VEH controls. Consequently, the elevation of 2-AG by MAGL inhibition (Niphakis et al, 2013) appears to counteract the aversive properties of MWD. The ability of MAGL inhibition to alleviate aversive effects of MWD is in agreement with previous studies investigating the ability of MAGL inhibition to reduce somatic symptoms of MWD in morphine-dependent mice, whether spontaneous or precipitated by naloxone (Ramesh et al, 2011, 2013).
Contrary to the BLA and interoceptive IC, intra-CeA administration of the CB1 receptor antagonist AM251, and not the MAGL inhibitor MJN110, interfered with the naloxone-precipitated MWD CPA. Although rats were significantly less active during MWD conditioning, intra-CeA AM251 did not significantly modify activity during conditioning relative to VEH rats. The ability of AM251 to attenuate the aversive properties of MWD are in agreement with systemic studies conducted by Wills et al (2014), and identifies the CeA as a neural substrate mediating the effect of AM251 on MWD. Indeed, the role of the CeA in contributing to the aversive state of MWD has been demonstrated in lesion (Watanabe et al, 2002; Xu et al, 2012), and c-fos studies (Frenois et al, 2002; Ishida et al, 2008; Jin et al, 2004, 2005). As intra-CeA AM251 has been shown to antagonize the anxiogenic effects produced by systemic naloxone in the elevated-plus maze (Zarrindast et al, 2008), it is likely that it also reduced the anxiety associated with MWD in the present study.
Within the BLA, MJN110 reduced the aversive properties of MWD through a CB1-dependent mechanism; likely by elevating 2-AG. These findings are in agreement with a previous study reporting the ability of CB1 agonism within the BLA to prevent stress-enhanced aversive learning (Ganon-Elazar and Akirav, 2009). Considering the modulatory role of the eCB in regulating the hypothalamic–pituitary–adrenal axis in this region (Hill and Tasker, 2012), it is possible that BLA 2-AG reduces the aversive properties of MWD by acting to constrain hypothalamic–pituitary–adrenal axis activity.
The double dissociation between the ability of CB1 receptor agonism (elevated 2-AG through MAGL inhibition) in the BLA and CB1 receptor antagonism in the CeA to interfere with a MWD CPA is consistent with the opposite pattern of c-fos expression in these regions during naloxone-precipitated MWD in rats; specifically, c-fos expression was significantly enhanced in the CeA, whereas it was slightly decreased in the BLA (Frenois et al, 2002). Given that cannabinoids are produced pre-synaptically and act retrogradely to inhibit neurotransmitter release (GABA or glutamate), it is feasible that the CB1 receptor antagonist reduces MWD by disinhibition of GABA release in the CeA, whereas 2-AG reduces MWD by inhibition of GABA release in the BLA—ultimately restoring neuronal activation to baseline levels in both regions. Indeed, CB1 receptors have been found to modulate GABA transmission in both regions (Azad et al, 2003; Katona et al, 2001; Roberto et al, 2010), and the complex interplay between cannabinoid-mediated inhibition and excitation of the amygdala plays an important role in the regulation of anxiety states which is largely disrupted in MWD (see Ruehle et al, 2012 for a review). Alternatively, given the more essential role of the CeA in mediating aversive withdrawal and the connectivity between the CeA and BLA (Jin et al, 2005; Wenzel et al, 2011), it is also possible that intra-BLA MJN110 may interfere with MWD by indirectly inactivating the CeA as would be the direct effect of AM251 in the CeA. Indeed, inhibition of GABA release in the BLA would elevate excitation of pyramidal neurons projecting to the intercalated cells, which would lead to greater GABA release within the CeA (as described in detail in Katona et al, 2001).
Unlike the amygdala, the IC has only recently been implicated in addiction processes. Naqvi et al (2007) were the first to propose that the insula may be critical to nicotine addiction in human stroke patients. The insula had been implicated in the representation of interoceptive states (Craig, 2002). In pre-clinical animal models, Contreras et al (2007) reported that inactivation of the interoceptive IC blocked the expression of both LiCl-induced malaise in rats and of amphetamine-induced conditioned place preference, suggesting that the interoceptive IC is involved in the modulation of a ‘state of well being'. Inactivation of the interoceptive IC has recently been demonstrated to prevent the acquisition of a naloxone-precipitated MWD CPA in rats (Li et al, 2013). As naloxone-precipitated MWD also produces malaise in rats as indicated by conditioned gaping (Mcdonald et al, 1997), it is conceivable that elevation of 2-AG in this region reduced the aversive properties of MWD thereby interfering with the CPA. Indeed, 2-AG and MJN110 (but not AEA or FAAH inhibition) exogenously delivered to the interoceptive IC have been reported to reduce the establishment of LiCl-induced conditioned gaping reactions, a measure of nausea in rats, by a CB1-dependent mechanism of action (Sticht et al, 2015a, 2015b).
The current findings revealed the respective sites of action of systemically administered MJN110 (BLA and interoceptive IC) and AM251 (CeA) to interfere with the establishment of a one-trial acute naloxone-precipitated MWD CPA. Given the role of the eCB and these brain regions in emotional processing and interoceptive deviations from normal, it is suggested that these effects are mediated by the ability of these compounds to counteract the aversive properties of MWD. Ultimately, these findings reveal region specific complexities of the eCB in the regulation of opiate withdrawal that warrant further investigation.
Funding and Disclosure
The authors declare no conflict of interest.
Acknowledgments
The research described here was funded by research grants to Linda A Parker from the Natural Sciences and Engineering Council of Canada (NSERC: 92057), and the Canadian Institutes of Health Research (CIHR:137122) and by an NSERC postgraduate doctoral scholarship to Kiri L Wills. Additional funding was provided by research grants from the National Institutes of Health (DAO32933, DAO33760) to Benjamin F Cravatt. Kiri Wills, Gavin Petrie, Cheryl Limebeer and Erin Rock are trainees in the laboratory funded by Linda Parker's acknowledged grants and Micah Niphakis is a trainee in Benjamin Cravatt's laboratory funded by his acknowledged grants.
References
- Ahn K, McKinney MK, Cravatt BF (2008). Enzymatic pathways that regulate endocannabinoid signaling in the nervous system. Chem Rev 108: 1687–1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen GV, Saper CB, Hurley KM, Cechetto DF (1991). Organization of visceral and limbic connections in the insular cortex of the rat. J Comp Neurol 311: 1–16. [DOI] [PubMed] [Google Scholar]
- Azad SC, Eder M, Marsicano G, Lutz B, Zieglgansberger W, Rammes G (2003). Activation of the cannabinoid receptor type 1 decreases glutamatergic and GABAergic synaptic transmission in the lateral amygdala of the mouse. Learn Mem 10: 116–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhargava HN (1976). Inhibition of naloxone-induced withdrawal in morphine dependent mice by 1-trans-Δ9-tetrahydrocannabinol. Eur J Pharmacol 36: 259–262. [DOI] [PubMed] [Google Scholar]
- Cechetto DF, Saper CB (1987). Evidence for a viscerotopic sensory representation in the cortex and thalamus in the rat. J Comp Neurol 262: 27–45. [DOI] [PubMed] [Google Scholar]
- Contreras M, Ceric F, Torrealba F (2007). Inactivation of the interoceptive insula disrupts drug craving and malaise induced by lithium. Science 318: 655–658. [DOI] [PubMed] [Google Scholar]
- Craig AD (2002). How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 3: 655–666. [DOI] [PubMed] [Google Scholar]
- Cravatt BF, Giang DK, Mayfield SP, Boger DL, Lerner RA, Gilula NB (1996). Molecular characterization of an enzyme that degrades neuromodulatory fatty-acid amides. Nature 384: 83–87. [DOI] [PubMed] [Google Scholar]
- Devane WA, Hanuš L, Breuer A, Pertwee RG, Lesley A, Griffin G et al (1992). Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258: 1946–1949. [DOI] [PubMed] [Google Scholar]
- Dinh TP, Carpenter D, Leslie FM, Freund TF, Katona I, Sensi SL et al (2002). Brain monoglyceride lipase participating in endocannabinoid inactivation. Proc Natl Acad Sci USA 99: 10819–10824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg RM (1982). Further studies on the acute dependence produced by morphine in opiate naive rats. Life Sci 31: 1531–1540. [DOI] [PubMed] [Google Scholar]
- Frenois F, Cador M, Caille S, Stinus L, Le Moine C (2002). Neural correlates of the motivational and somatic components of naloxone-precipitated morphine withdrawal. Eur J Neurosci 16: 1377–1389. [DOI] [PubMed] [Google Scholar]
- Ganon-Elazar E, Akirav I (2009). Cannabinoid receptor activation in the basolateral amygdala blocks the effects of stress on the conditioning and extinction of inhibitory avoidance. J Neurosci 29: 11078–11088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heishman SJ, Stitzer ML, Bigelow GE, Liebson IA (1990). Acute opioid physical dependence in humans: effect of naloxone at 6 and 24 hours postmorphine. Pharmacol Biochem Behav 36: 393–399. [DOI] [PubMed] [Google Scholar]
- Hill MN, Tasker JG (2012). Endocannabinoid signaling, glucocorticoid-mediated negative feedback, and regulation of the hypothalamic-pituitary-adrenal axis. Neuroscience 204: 4–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishida S, Shimosaka R, Kawasaki Y, Jin C, Kitamura Y, Araki H et al (2008). Involvement of the amygdala on place aversion induced by naloxone in single-dose morphine-treated rats. Yakugaku Zasshi 128: 395–403. [DOI] [PubMed] [Google Scholar]
- Jin C, Araki H, Nagata M, Shimosaka R, Shibata K, Suemaru K et al (2005). Expression of c-Fos in the rat central amygdala accompanies the acquisition but not expression of conditioned place aversion induced by withdrawal from acute morphine dependence. Behav Brain Res 161: 107–112. [DOI] [PubMed] [Google Scholar]
- Jin C, Araki H, Nagata M, Suemaru K, Shibata K, Kawasaki H et al (2004). Withdrawal-induced c-Fos expression in the rat centromedial amygdala 24h following a single morphine exposure. Psychopharmacology (Berl) 175: 428–435. [DOI] [PubMed] [Google Scholar]
- June HL, Stitzer ML, Cone E (1995). Acute physical dependence: time course and relation to human plasma morphine concentrations. Clin Pharmacol Ther 57: 270–280. [DOI] [PubMed] [Google Scholar]
- Katona I, Rancz EA, Acsady L, Ledent C, Mackie K, Hajos N et al (2001). Distribution of CB1 cannabinoid receptors in the amygdala and their role in the control of GABAergic transmission. J Neurosci 21: 9506–9518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koob GF (2009. a). Brain stress systems in the amygdala and addiction. Brain Res 1293: 61–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koob GF (2009. b). Dynamics of neuronal circuits in addiction: reward, antireward, and emotional memory. Pharmacopsychiatry 42(Suppl 1): S32–S41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li CL, Zhu N, Meng X-L, Li Y-H, Sui N (2013). Effects of inactivating the agranular or granular insular cortex on the acquisition of the morphine-induced conditioned place preference and naloxone-precipitated conditioned place aversion in rats. J Psychopharmacol 27: 837–844. [DOI] [PubMed] [Google Scholar]
- Li M, Hou YY, Lu B, Chen J, Chi ZQ, Liu JG (2009). Expression pattern of neural synaptic plasticity marker-Arc in different brain regions induced by conditioned drug withdrawal from acute morphine-dependent rats. Acta Pharmacol Sinica 30: 282–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Limebeer CL, Rock EM, Mechoulam R, Parker LA (2012). The anti-nausea effects of CB1 agonists are mediated by an action at the visceral insular cortex. Br J Pharmacol 167: 1126–1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin WR, Eades CG (1964). A comparison between acute and chronic physical dependence in the chronic spinal dog. J Pharmacol Exp Ther 146: 385–394. [PubMed] [Google Scholar]
- Mas-Nieto M, Pommier B, Tzavara ET, Caneparo A, Da Nascimento S, Le Fur G et al (2001). Reduction of opioid dependence by the CB(1) antagonist SR141716A in mice: evaluation of the interest in pharmacotherapy of opioid addiction. Br J Pharmacol 132: 1809–1816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDonald AJ (1998). Cortical pathways to the mammalian amygdala. Prog Neurobiol 55: 257–332. [DOI] [PubMed] [Google Scholar]
- McDonald AJ, Shammah-lagnado SJ, Shi C, Davis M (1999). Cortical afferents to the extended amygdala. Ann N Y Acad Sci 877: 309–338. [DOI] [PubMed] [Google Scholar]
- Mcdonald R, Parker L, Siegel S (1997). Conditioned sucrose aversions produced by naloxone-precipitated withdrawal from acutely administered morphine. Pharmacol Biochem Behav 58: 1003–1008. [DOI] [PubMed] [Google Scholar]
- Naqvi NH, Rudrauf D, Damasio H, Bechara A (2007). Damage to the insula disrupts addiction to cigarette smoking. Science 315: 531–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niphakis MJ, Cognetta AB, Chang JW, Buczynski MW, Parsons LH, Byrne F et al (2013). Evaluation of NHS carbamates as a potent and selective class of endocannabinoid hydrolase inhibitors. ACS Chem Neurosci 4: 1322–1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker LA, Cyr JA, Santi AN, Burton PD (2002). The aversive properties of acute morphine dependence persist 48h after a single exposure to morphine evaluation by taste and place conditioning. Pharmacol Biochem Behav 72: 87–92. [DOI] [PubMed] [Google Scholar]
- Parker La, Niphakis MJ, Downey R, Limebeer CL, Rock EM, Sticht MA et al (2014). Effect of selective inhibition of monoacylglycerol lipase (MAGL) on acute nausea, anticipatory nausea, and vomiting in rats and Suncus murinus. Psychopharmacology (Berl) 232: 583–593. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C (1998) The Rat Brain in Stereotaxic Coordinates, 6th edn. Acad Press: New York. [Google Scholar]
- Ramesh D, Gamage TF, Vanuytsel T, Owens RA, Abdullah RA, Niphakis MJ et al (2013). Dual inhibition of endocannabinoid catabolic enzymes produces enhanced antiwithdrawal effects in morphine-dependent mice. Neuropsychopharmacology 38: 1039–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramesh D, Ross GR, Schlosburg JE, Owens RA, Abdullah RA, Kinsey SG et al (2011). Blockade of endocannabinoid hydrolytic enzymes attenuates precipitated opioid withdrawal symptoms in mice. J Pharmacol Exp Ther 339: 173–185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberto M, Cruz M, Bajo M, Siggins GR, Parsons LH, Schweitzer P (2010). The endocannabinoid system tonically regulates inhibitory transmission and depresses the effect of ethanol in central amygdala. Neuropsychopharmacology 35: 1962–1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubino T, Massi P, Vigano D, Fuzio D, Parolaro D (2000). Long-term treatment with SR141716A, the CB1 receptor antagonist, influences morphine withdrawal syndrome. Life Sci 66: 2213–2219. [DOI] [PubMed] [Google Scholar]
- Ruehle S, Rey AA, Remmers F, Lutz B (2012). The endocannabinoid system in anxiety, fear memory and habituation. J Psychopharmacol 26: 23–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sticht MA, Limebeer CL, Rafla BR, Parker LA (2015. a). Intra-visceral insular cortex 2-arachidonoylglycerol, but not N-arachidonoylethanolamide, suppresses acute nausea-induced conditioned gaping in rats. Neuroscience 286: 338–344. [DOI] [PubMed] [Google Scholar]
- Sticht MA, Limebeer CL, Rafla BR, Abdullah RA, Poklis JL, Ho W et al (2015. b). Endocannabinoid regulation of nausea is mediated by 2-arachidonoylglycerol (2-AG) in the rat visceral insular cortex. Neuropharmacology 102: 92–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura T, Kondo S, Sukagawa A, Nakane S, Shinoda A, Itoh K et al (1995). 2-Arachidonoylgylcerol: a possible endogenous cannabinoid receptor ligand in brain. Biochem Biophys Res Commun 215: 89–97. [DOI] [PubMed] [Google Scholar]
- Vela G, Ruiz-gayo M, Fuentes JA (1995). Anandamide decreases naloxone-precipitated withdrawal signs in mice chronically treated with morphine. Neuropharmacology 34: 665–668. [DOI] [PubMed] [Google Scholar]
- Watanabe T, Yamamoto R, Maeda A, Nakagawa T, Minami M, Satoh M (2002). Effects of excitotoxic lesions of the central or basolateral nucleus of the amygdala on naloxone-precipitated withdrawal-induced conditioned place aversion in morphine-dependent rats. Brain 958: 423–428. [DOI] [PubMed] [Google Scholar]
- Wenzel JM, Waldroup SA, Haber ZM, Su ZI, Ben-Shahar O, Ettenberg A (2011). Effects of lidocaine-induced inactivation of the bed nucleus of the stria terminalis, the central or the basolateral nucleus of the amygdala on the opponent-process actions of self-administered cocaine in rats. Psychopharmacology 217: 221–230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wills KL, Vemuri K, Kalmar A, Lee A, Limebeer CL, Makriyannis A et al (2014). CB1 antagonism: interference with affective properties of acute naloxone-precipitated morphine withdrawal in rats. Psychopharmacology (Berl) 231: 291–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu W, Li YH, Tan BP, Luo XJ, Xiao L, Zheng XG et al (2012). Inhibition of the acquisition of conditioned place aversion by dopaminergic lesions of the central nucleus of the amygdala in morphine-treated rats. Physiol Res 8408: 437–442. [DOI] [PubMed] [Google Scholar]
- Yamaguchi T, Hagiwara Y, Tanaka H, Sugiura T, Waku K, Shoyama Y et al (2001). Endogenous cannabinoid, 2-arachidonoylglycerol, attenuates naloxone-precipitated withdrawal signs in morphine-dependent mice. Brain Res 909: 121–126. [DOI] [PubMed] [Google Scholar]
- Zarrindast M-R, Sarahroodi S, Arzi A, Khodayar MJ, Taheri-Shalmani S, Rezayof A (2008). Cannabinoid CB1 receptors of the rat central amygdala mediate anxiety-like behavior: interaction with the opioid system. Behav Pharmacol 19: 716–723. [DOI] [PubMed] [Google Scholar]