Abstract
The synthesis and characterization of new enantiopure cyclopentadienyl titanium oximato compounds (S,R)-[(η5-C5H5)Ti{κ2NO,(R)NH·HCl}Cl2] (R = Ph (phenyl) 1a·HCl, Bn (benzyl) 1b·HCl, 2-pic (2-picolyl) 1c·HCl), (S,R)-[(η5-C5H5)TiCl2{κ2NO,(Ph)NH}] (1a) and (S,R)-[(η5-C5H5)2TiCl{κ2NO,(R)NH}] (R = Ph 2a, Bn 2b, 2-pic 2c), along with studies on their behaviour in D2O at different pD values are reported. The structure of previously described ammonium-oxime (2S,5R)-{NOH,(Bn)NH·HCl} (b·HCl) and novel titanium derivative 1a have been determined by single crystal X-ray crystallography. The effect of the compounds on cytotoxicity, cell adhesion and migration of the androgen-independent prostate cancer PC-3 cells has been assessed. Compounds 2b and 2c are more cytotoxic than additive doses of titanocene dichloride and free oxime proligand, probing the synergistic effect of these novel compounds. The cytotoxicity of 2b and 2c has been further evaluated against human renal Caki-1, colon DLD-1 and triple negative breast MDA-MB-231 cancer cell lines. The activity found for 2c on PC-3 and Caki-1 is higher than that of highly active Titanocene Y (bis-[(p-methoxybenzyl)cyclopentadienyl]titanium(IV) dichloride), while showing selectivity against renal cancer when compared to a non-tumorigenic human renal (HEK-293T) cell line. Compounds 2b and especially 2c are apoptotic in Caki-1 cancer cell lines. Cell adhesion and wound-healing assays confirmed that derivatives 1c·HCl, 2b and 2c affect the adhesion and migration patterns of the PC-3 cell line. Interactions of the novel compounds with plasmid (pBR322) DNA have also been studied, showing that the oximato Ti(IV) derivatives have a weak or no interaction with DNA at physiological pH.
Keywords: Cyclopentadienyl titanium compounds, chirality, PC-3 and Caki-1 cell lines, DNA, cell adhesion, cell migration
Graphical abstract
Novel easily accessible enantiopure cyclopentadienyl titanium oximato compounds are reported. Two of them are strongly cytotoxic against PC-3 and Caki-1 cancer cells while inducing apoptosis and showing selectivity when compared to a non-tumorigenic renal cell line. They also affect the adhesion and migration ability of human prostate cancer cells.

Introduction
Although cisplatin and follow-on derivatives are still widely used in the clinic, their side effects, general low solubility and intrinsic or acquired resistance in some cancer types have encouraged research for the discovery of new metal compounds with improved properties [1–6]. Within this context, titanium derivatives play a central role. Budotitane [cis-diethoxybis(1-phenylbutane-1,3-dionato)titanium(IV)] [7–9] and titanocene dichloride ([(η5-C5H5)2TiCl2], TDC) [10,11] were the first reported Ti(IV) compounds which showed promising anticancer properties. Budotitane was the first non-platinum complex to enter clinical trials for its activity towards colon tumour cells, but the compound did not progress beyond phase I due to formulation problems [7,8,12–15]. TDC exhibited significant antitumor activity, in vitro and in vivo, against cisplatin-resistant cells and tumours generally difficult to treat [10,11,13–16]. Based on these encouraging results, TDC entered clinical trials as well [17]. Although the clinical response was not significant enough to justify continuing trials, the absence of bone marrow toxicity was thought to be promising and the interest in the potential of superior titanium compounds as anticancer agents remains since then [5,18]. The instability and insolubility of TDC in water and lack of standard formulation were contributing factors to discontinuing from clinical trials. Thus, a plethora of modified titanium-based compounds have been synthesized and studied as potential antitumor agents, especially during the last decade [5,6,14,15,18–32]. Substituted titanocenes such as Titanocene Y [33,34] (Ti-Y, Fig. 1), Titanocene Y* [35,36] (Ti-Y*, bis-[(p-diethylaminobenzyl)cyclopentadienyl]titanium(IV) dichloride, Fig. 1) and Titanocene T [37] (Ti-T, bis-[{(1-methyl-3-diethylaminomethyl)indol-4-yl}methylcyclopentadienyl]titanium(IV) dichloride, Fig. 1), developed by the group of M. Tacke, or titanium compounds with salan ligands (Ti-salan, Fig. 1) described by the group of E. Tshuva [14,38–40] have shown great therapeutic promise and are undergoing further development [36,40,41]. Heterometallic Ti-Au derivatives have also demonstrated to be promising drug candidates for the treatment of renal cancer [30]. A recently published comparison between the biological behaviour of Ti-Y and Ti-salan derivatives demonstrate that they do not follow analogous bio-distribution patterns, showing that the ligands bound to the Ti(IV) centre assume an important relevance for the activity of these metal-based drugs [41].
Fig. 1.
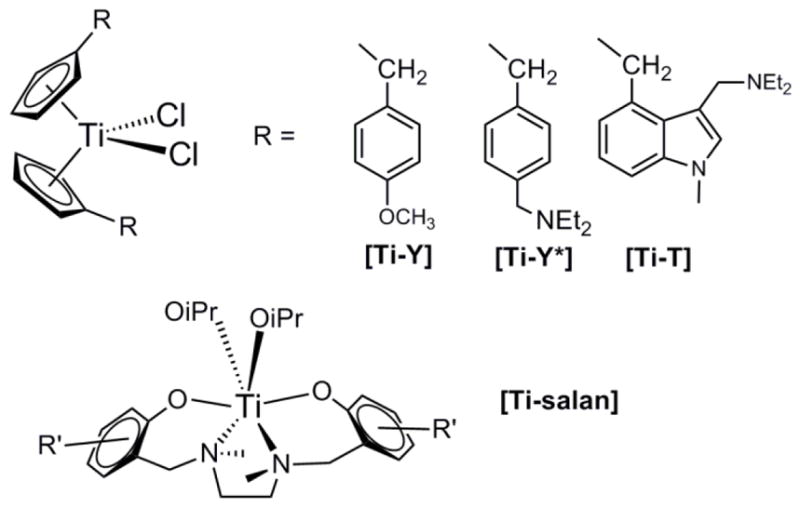
Selected titanium compounds with relevant antitumor properties
In this context, oxime-containing ligands offer significant advantages for biological applications. Some oxime organic derivatives have been reported to have biological effects such as endothelium-independent relaxation in blood-vessels, an increase in the targeting of specific nuclear bases of DNA and oxidative DNA cleavage [42,43]. Oxime-containing Pt(II) compounds have been reported as a novel class of nonclassic platinum-based complexes with interesting antitumor properties, different DNA binding behaviour and a different pattern of protein interaction from those found in classical cisplatin [44,45]. The study on a variety of Rh(III) and Ir(III) compounds with oximato ligands show a strong cytotoxic effect toward HeLa and HL60 cancer lines, whereas the compounds do not modify DNA in a similar way to that of cisplatin [46]. DNA-binding studies of oxime-containing ruthenium compounds have been previously investigated, but only a weak, non-intercalative in nature interaction was found [47]. We reported recently a new family of chiral, water soluble and stable p-cymene ruthenium(II) compounds with amino-oxime ligands. The compounds show significant effects on cytotoxicity, cell adhesion to collagen and migration of androgen-independent prostate cancer cells while they do not exhibit strong interactions with plasmid (pBR322) DNA [48].
Although oxime and oximato metal derivatives have been extensively studied [49–51], oximato titanium compounds have received little attention. The first titanocene oximato derivatives, [(η5-C5H5)2Ti(H2O)(κ2O=NCRR′)]+, were reported by Thewalt et al [52] and described as surprisingly stable against air and water. More recently, the synthesis of a novel family of titanocene compounds of general formula [(η5-C5H5)2Ti(κ2O=NCRR′)Cl] [53–55] and several oximato titanium derivatives [Ti(OR1)4-n(κ2O=NCR2R3)n] (where n = 1–4) have also been published [56–61]. Oxime groups are presented as excellent chemical modifiers with a wide versatility of coordination modes going from mono- κN, κO to di-hapto κ2N,O, either with side on or bridging coordination, depending on the nature of the ligand and the metal centre [49–51,62]. Some titanium oximato compounds have shown interesting antibacterial properties [60,61], however, their anticarcinogenic effects have not been reported before.
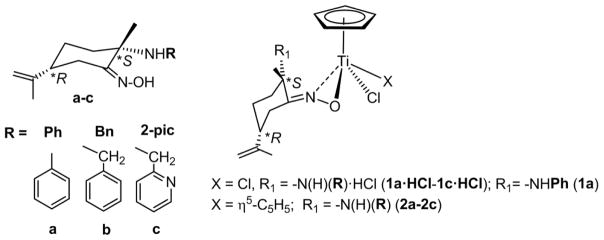
Encouraged by our previous results on the enantiomerically pure amino-oxime ruthenium compounds [48], we hypothesized that oximato ligands could increase the water solubility and stability of the resulting titanium derivatives [52]. Thus, we explored reactions of [(η5-C5H5)TiCl2] and [(η5-C5H5)2TiCl2] with the previously described enantiopure amino-oxime derivatives (2S,R)-{NOH,(R)NH} (R = phenyl a, benzyl b, 2-picolyl c, see Fig. 2) [48,63,64], derived from R-limonene. This naturally occurring terpene is an inexpensive starting reagent, commercially available in optically pure form and easily tailored by stereoselective functionalization [63–65].
Fig. 2.
Enantiomerically pure amino-oxime ligands and related Ti(IV) compounds
The effect of stereochemistry on biological activity is of great importance in medicinal chemistry, as many of the biological targets are chiral [66–69]. Although various examples of chiral anticancer titanium compounds have been reported [39,70–72], examples of enantiopure titanium anticancer compounds are scarce [67]. Manna et al [67] have shown that stereochemistry plays an important role in determining cytotoxicity and should be considered in the design, modification and improvement of active compounds. In this work we report on the synthesis and characterization of a novel family of chiral cyclopentadienyl ammonium- or amino-oximato titanium derivatives (Fig 2). We describe their in vitro activity against human androgen-independent prostate cancer PC-3 cell line as well as the study of their interactions with plasmid (pBR322) DNA. The cytotoxicity of selected compounds has been evaluated on renal cancer Caki-1, colon cancer DLD-1, triple negative breast cancer MDA-MB-231 and non-tumorigenic human embryonic kidney HEK-293T cell lines. The cell death type as well as the effects on cell migration of selected compounds is also described.
2. Experimental Section
2.1. Chemicals and synthesis
Synthesis of titanium complexes were performed at an argon/vacuum manifold using standard Schlenk techniques or in a glove-box MBraun MOD System. Solvents were dried by known procedures and used freshly distilled. (2S,5R)-{NOH,(R)NH}, (R = Ph a, Bn b, 2-pic c) [63,64], corresponding adducts (2S,5R)-{NOH,(R)NH·HCl} (R = Ph a·HCl, Bn b·HCl, 2-Pic c·HCl) [63,64], [(η5-C5H5)TiCl3] [73] and Titanocene Y [74] were prepared according to previous reports. NEt3, HCl (1M in ether), DCl (35% wt in D2O), NaOD (40% wt in D2O), NaCl, KCl, Na2HPO4, KHPO4 and [(η5-C5H5)2TiCl2] were purchased from Sigma-Aldrich. NEt3 was stored under molecular sieves prior use. Commercially available reagents were used without further purification. NMR spectra were recorded on a Bruker 400 Ultrashield. 1H and 13C chemical shifts are reported relative to tetramethylsilane. 15N chemical shifts are reported relative to liquid ammonia (25 °C). 31P chemical shifts are reported relative to phosphoric acid (85% wt in D2O, 25 °C). Coupling constants J are given in Hertz. Elemental analyses were performed in our laboratories (UAH) on a Perkin Elmer 2400 CHNS/O Analyzer, Series II.
2.1.1. (S,R)-[(η5-C5H5)Ti{κ2NO,(Ph)NH·HCl}Cl2] (1a·HCl)
A diethylether (10 mL) solution of (2S,5R)-{NOH,(Ph)NH} (0.52 g, 2.01 mmol) and [(η5-C5H5)TiCl3] (0.44 g, 2.01 mmol) was warmed up in a J. Young valved ampule for 3 h at 55 °C. The resulting yellow suspension was filtered to isolate the yellow solid which precipitates from the mixture and washed twice (2×5 mL) with hexane. The solid was dried under vacuum and identified as derivative 1a·HCl. Yield: 0.64 g, 1.34 mmol, 66%. Solubility in H2O at 24 °C (mM): 2.1 ± 0.2. Value of pH ([2.0 mM]) in H2O at 24 °C: 2.40. Anal. Calcd for C21H27N2OTiCl3: C, 52.80; H, 5.70; N, 5.86%; Found: C, 53.29; H, 5.45; N, 6.09%. FTIR (KBr): ū 3090–3120 (br, NH·HCl); 1640, 1570 (w-m, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 11.62 (br, 2H, -NH·HCl), 7.21 (m, 5H, -C6H5), 6.63 (s, 5H, C5H5), 4.86, 4.80 (both s, each 1H, =CH2), 3.19, 3.18 (both m, overlapped, 2H, -CH26), 3.07 (m, 1H, -CH23), 2.45 (m, 1H, -CH24), 2.16 (m, 1H, -CH-C=), 1.82 (m, 1H, -CH23), 1.44 (m, 1H, -CH24), 1.77, 1.76 (both s, each 3H, NC-CH3 + CH3C=). 13C-NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 154.1 (−, Cq=N), 147.0 (−, =Cq-Me), 132.1 (−, CipsoPh), 129.6, 129.5, 126.1 (+, C6H5), 121.7 (+, C5H5), 111.7 (−, =CH2), 66.7 (−, Cq-NH), 46.3 (+, -CH5), 38.9 (−, -CH23), 32.9 (−, -CH26), 25.5 (−, -CH24), 21.9, 21.0 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 333.6 (C=N), 79.5 (NH·HCl).
2.1.2. (S,R)-[(η5-C5H5)Ti{κ2NO,(Bn)NH·HCl}Cl2] (1b·HCl)
A diethylether (10 mL) solution of (2S,5R)-{NOH,(Bn)NH} (0.42 g, 1.54 mmol) and [(η5-C5H5)TiCl3] (0.34 g, 1.54 mmol) was stirred for 30–45 min at room temperature. The resulting yellow suspension was filtered to isolate the yellow solid which precipitates from the mixture and washed twice (2×5 mL) with hexane. The solid was dried under vacuum and identified as derivative 1b·HCl. Yield: 0.58 g, 1.18 mmol, 77%. Solubility in H2O at 24 °C (mM): 8.1 ± 0.4. Value of pH ([2.0 mM]) in H2O at 24 °C: 2.16. Anal. Calcd for C22H29N2OTiCl3: C, 53.74; H, 5.94; N, 5.70; Found: C, 53.85; H, 5.81; N, 5.62. FTIR (KBr): ū 3100–3200 (br, NH·HCl); 1638, 1572 (both m, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 10.35, 9.84 (br, each 1H, -NH·HCl), 7.66 (m, 2H, -C6H5), 7.19 (m, 3H, -C6H5), 6.84 (s, 5H, C5H5), 4.84, 4.78 (both m, each 1H, CH2-Ph), 4.84, 4.78 (both s, each 1H, =CH2), 2.80, 2.44 (both m, each 1H, -CH26), 2.91 (m, 1H, -CH23), 2.64 (m, 1H, -CH24), 2.05 (m, 1H, -CH-C=), 1.75 (m, 1H, -CH23), 1.58 (m, 1H, -CH24), 2.08, 1.77 (both s, each 3H, NC-CH3 + CH3C=). 13C- NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCL3): δ 155.1 (−, Cq=N), 146.9 (−, =Cq-Me), 130.9 (−, CipsoPh), 131.7, 129.3, 128.7 (+, C6H5), 122.1 (+, C5H5), 111.7 (−, =CH2), 63.5 (−, Cq-NH), 47.6 (−, -CH2Ph), 46.2 (+, -CH5), 39.0 (−, -CH23), 32.4 (−, -CH26), 25.8 (−, -CH24), 21.4, 21.0 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCL3): δ 331.8 (C=N), 67.3 (NH·HCl).
2.1.3. (S,R)-[(η5-C5H5)Ti{κ2NO,(2-plc)NH·HCl}Cl2] (1c·HCl)
A diethylether (10 mL) solution of (2S,5R)-{NOH,(2-pic)NH} (0.40 g, 1.46 mmol) and [(η5-C5H5)TiCl3] (0.32 g, 1.46 mmol) was stirred for 5 min at room temperature. The resulting yellow suspension was filtered to isolate the yellow solid which precipitates from the mixture and washed twice (2×5 mL) with hexane. The solid was dried under vacuum and identified as derivative 1c·HCl. Yield: 0.49 g, 1.00 mmol, 70%. Solubility in H2O at 24 °C (mM): 8.6 ± 0.4. Value of pH ([2.0 mM]) in H2O at 24 °C: 2.21. FTIR (KBr): ū 3150–3200 (br, NH·HCl); 1643, 1572 (both m, C=N). Anal. Calcd for C21H28N3OTiCl3: C, 51.19; H, 5.73; N, 8.53; Found: C, 51.16; H, 5.43; N, 9.03. 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 10.43 (br, 2H, -NH·HCl), 8.40, 7.70, 7.25, 7.31 (all m, each 1H, -C5NH4), 6.76 (s, 5H, C5H5), 4.77, 4.72 (both s, each 1H, =CH2), 4.40 (m, 2H, CH2pic), 2.97, 2.56 (both m, each 1H, -CH26), 2.76 (m, 1H, -CH23), 2.18 (m, 1H, -CH-C=), 2.08 (m, 1H, -CH24), 1.88 (overlapped, 5H, -CH23 +-CH24 + NC-CH3), 1.73 (s, 3H, CH3C=). 13C-NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 152.8 (−, Cq=N), 150.4 (−, CipsoC5NH4), 147.3, 138.9, 124.1, 122.9 (all +, -NC5H4), 146.3 (−, =Cq-Me), 122.0 (+, C5H5), 111.0 (−, =CH2), 61.6 (−, Cq-NH), 44.5 (−, -CH2-NC5H4), 44.2 (+, -CH5), 37.9 (−, -CH23), 31.5 (−, -CH26), 25.5 (−, -CH24), 21.2, 20.3 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 335.0 (C=N), 284.9 (NC5H4), 56.3 (NH·HCl).
2.1.4. (S,R)-[(η5-C5H5)Ti{κ2NO,(Ph)NH}Cl2] (1a)
A toluene (5 mL) solution of 1a·HCl (0.20 g, 0.42 mmol) and NEt3 (0.058 mL, 0.42 mmol) was stirred for 30 min at room temperature. The resulting orange suspension was filtered to separate the solution from insoluble NEt3·HCl. The orange solution was evaporated to dryness and the resulting yellow solid washed twice with hexane (2×5 mL) and dried under vacuum. Yield: 0.13 g, 0.29 mmol, 69%. CHN analysis data found did not agree with theoretical data calculated for C21H26N2OTiCl2, due to impurification of the solid with NEt3·HCl. Recrystallization of the solid from toluene at −20 ºC affords crystals suitable for X-ray analysis. FTIR (KBr): ū 3374 (m, NH); 1641, 1601 (both m, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 7.10 (m, 2H, -C6H5), 6.72 (m, 3H, -C6H5), 6.82 (s, 5H, C5H5), 4.77, 4.74 (both s, each 1H, =CH2), 3.71 (br, 1H, NH), 2.95, 2.49 (m, 1H, -CH26 ), 2.22 (m, 1H, -CH-C=), 2.12, 1.86 (m, 1H, -CH23), 1.87, 1.75 (m, 1H, -CH24), 1.85, 1.73 (both s, each 3H, NC-CH3 + CH3C=). 13C- NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 162.8 (−, Cq=N), 147.4 (−, =Cq-Me), 145.5 (CipsoPh), 129.2, 118.6, 115.2 (+, C6H5), 121.2 (+, C5H5), 110.2 (−, =CH2), 57.7 (−, Cq-NH), 46.1 (+, -CH5), 43.3 (−, -CH23), 31.2 (−, -CH26), 26.3 (−, -CH24), 23.2, 20.7 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 319.0 (C=N), 85.1 (NHPh).
2.1.5. (S,R)-[(η5-C5H5)2Ti{κ2NO,(Ph)NH}Cl] (2a)
A toluene (5 mL) solution of [(η5-C5H5)2TiCl2] (0.25 g, 1.00 mmol), (2S,5R)-{NOH,(Ph)NH} (0.26 g, 1.00 mmol) and NEt3 (0.12 mL, 0.95 mmol) was warmed up to 50 ºC and stirred overnight. The resulting orange suspension was filtered, to eliminate insoluble NEt3·HCl, and the final orange solution evaporated to dryness to afford a yellow solid which was washed twice with hexane (2×5 mL) and dried under vacuum. Yield: 0.28 g, 0.59 mmol, 59%. Solubility in H2O at 24 ºC (mM): 2.7 ± 0.6. Value of pH ([2.0 mM]) in H2O at 24 ºC: 4.34. Anal. Calcd for C26H31N2OTiCl: C, 66.32; H, 6.64; N, 5.95; Found: C, 66.62; H, 6.89; N, 5.99. FTIR (KBr): ū 3370 (mr, NH); 1644, 1602 (both m, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 7.15 (m, 2H, -C6H5), 6.73 (m, 3H, -C6H5), 6.40, 6.41 (both s, each 5H, C5H5), 4.75, 4.72 (both s, each 1H, =CH2), 3.53 (br, 1H, NH), 2.83 (m, 1H, -CH26), 2.15 (m, 1H, -CH-C=), 2.01 (m, 1H, -CH23), 1.83 (m, 2H, overlapped -CH24+6), 1.73 (m, 1H, -CH23), 1.63 (m, 1H, -CH24), 1.76, 1.56 (both s, each 3H, NC-CH3 + CH3C=). 13C- NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 160.5 (−, Cq=N), 149.2 (−, =Cq-Me), 147.0 (CipsoPh), 129.4, 118.3, 115.8 (+, C6H5), 117.2, 117.1 (+, C5H5), 109.5 (−, =CH2), 57.4 (−, Cq-NH), 46.3 (+, -CH5), 44.0 (−, -CH23), 28.0 (−, -CH26), 26.6 (−, -CH24), 23.7, 21.2 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 400.2 (C=N), 86.1 (NHPh).
2.1.6. (S,R)-[(η5-C5H5)2Ti{κ2NO,(Bn)NH}Cl] (2b)
An analogous procedure to that described for 2a was followed, starting from [(η5-C5H5)2TiCl2] (0.20 g, 0.80 mmol), (2S,5R)-{NOH,(Bn)NH} (0.22 g, 0.80 mmol) and NEt3 (0.11 mL, 0.80 mmol). Compound 2b was obtained as a yellow solid. Yield: 0.35 g, 0.72 mmol, 88%. Solubility in H2O at 24 ºC (mM): 6.6 ± 0.2. Value of pH ([2.0 mM]) in H2O at 24 ºC: 5.54. Anal. Calcd for C27H33N2OTiCl: C, 66.88; H, 6.86; N, 5.78; Found: C, 67.29; H, 6.62; N, 5.85. FTIR (KBr): ū 3370 (m, NH); 1646, 1601 (both m, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 7.32 (m, 5H, -C6H5), 6.39, 6.39 (both s, each 5H, C5H5), 4.76, 4.74 (both s, each 1H, =CH2), 3.76, 3.55 (both m, each 1H, -CH2Ph), 2.92 (m, 1H, -CH26), 2.05 (m, 1H, -CH-C=), 1.90 (m, 1H, -CH23), 1.72 (m, 1H, -CH26), 1.68 (m, 2H, -CH24), 1.59 (m, 1H, -CH23), 1.56 (m, 1H, -CH24), 1.25 (br, 1H, NH), 1.47, 1.25 (both s, each 3H, NC-CH3 + CH3C=). 13C-NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 159.2 (−, Cq=N), 149.3 (−, =Cq-Me), 141.6 (CipsoPh), 128.7, 128.7, 127.2 (+, C6H5), 117.1, 117.1 (+, C5H5), 109.4 (−, =CH2), 57.1 (−, Cq-NH), 47.3 (−, -CH2Ph), 45.6 (+, -CH5), 41.2 (−, -CH23), 27.8 (−, -CH26), 26.2 (−, -CH24), 23.9, 21.3 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 398.9 (C=N), 60.0 (NHBn).
2.1.7. (S,R)-[(η5-C5H5)2Ti{κ2NO,(2-pic)NH}Cl] (2c)
An analogous procedure to that described for 2a was followed, starting from [(η5-C5H5)2TiCl2] (0.30 g, 1.20 mmol), (2S,5R)-{NOH,(2-pic)NH} (0.33 g, 1.20 mmol) and NEt3 (0.11 mL, 1.20 mmol). Compound 2c was obtained as a yellow-orange solid. Yield: 0.28 g, 0.61 mmol, 61%. Solubility in H2O at 24 ºC (mM): 15.7 ± 1.7. Value of pH ([2.0 mM]) in H2O at 24 ºC: 5.22. Anal. Calcd for C26H32N3OTiCl: C, 64.27; H, 6.64; N, 8.65; Found: C, 64.41; H, 6.47; N, 8.89. FTIR (KBr): ū 3304 (m, NH); 1640, 1591, 1569 (both s, C=N). 1H NMR (plus HSQC, plus HMBC, plus COSY, 400.1 MHz, 293 K, CDCl3): δ 8.50, 7.60, 7.30, 7.12 (all m, each 1H, -C5NH4), 6.38, 6.38 (both s, each 5H, C5H5), 4.77, 4.74 (both s, each 1H, =CH2), 3.91, 3.70 (both m, each 1H, CH2-C5NH4), 2.84 (m, 1H, -CH26), 2.07 (m, 1H, -CH-C=), 1.98 (m, 2H, overlapped -CH23+6), 1.78 (s, 3H, CH3C=), 1.64 (m, 1H, -CH23), 1.62 (m, 2H, -CH24), 1.60 (m, 1H, -CH24), 1.48 (br, 4H, NC-CH3 + NH). 13C-NMR (plus APT, plus gHSQC, plus HMBC, 100.6 MHz, 293 K, CDCl3): δ 157.6 (−, Cq=N), 148.1 (−, =Cq-Me), 160.2 (CipsoC5NH4), 149.3, 136.7, 122.9, 122.9 (+, C5NH4), 117.1, 117.1 (+, C5H5), 109.6 (−, =CH2), 48.5 (−, -CH2-C5NH4), 45.3 (+, -CH5), 41.1 (−, -CH23), 27.7 (−, -CH26), 26.2 (−, -CH24), 23.9, 21.3 (both +, CH3-CNH + CH3-C=). 15N NMR (gHMBC, 40.5 MHz, 293 K, CDCl3): δ 402.1 (C=N), 312.5 (C5NH4), 52.6 (NHpic).
2.1.8. 1H NMR experiments at various pH
Titanium compounds were dissolved in 2000 μL of D2O, 1,4-dioxane ([Ti]:[1,4-dioxane] = 1:3) was then added as internal reference and time-dependent 1H NMR spectra of 500 μL aliquots of final solutions were carried out (no changes on measured pD values were observed before and after 1,4-dioxane addition). Phosphate buffer saline (PBS) was prepared according to Cold Spring Harbor Protocols (http://cshprotocols.cshlp.org/content/2006/1/pdb.rec8247) using NaCl, KCl, Na2HPO4 and KHPO4 in D2O. Adjustment of pD (pD = pH* + 0.4, where pH* = pHmeter reading in D2O) was carried out using a solution of DCl (0.01M) or NaOD (0.01M) in D2O, with the help of a HANNA HI208 pHmeter. Titanium compounds were then dissolved in 2000 μL of the freshly prepared PBS, final pD measured (7.30–7.38) and time-dependent 1H NMR spectra of 500 μL aliquots of final solutions were carried out at 25 ºC. Alternatively, titanium compounds were dissolved in 2000 μL of D2O, 1,4-dioxane ([Ti]:[1,4-dioxane] = 1:3) was then added as internal reference, final pD adjusted to 7.4 using a solution of NaOD (0.01M) in D2O, and time-dependent 1H NMR spectra of 500 μL aliquots of final solutions were carried out. The percentage of metal bound cyclopentadienyl was estimated by integration of the resonance due to 1,4 dioxane (δ 3.75 in D2O) relative to the observed metal bound C5H5 singlets. Alternatively, the percentage of metal bound cyclopentadienyl was estimated by integration of the signals arising from the aromatic rings of the Ph, Bn or 2-pic substituent of the free ammonium-oxime a·HCl-c·HCl that is formed in D2O right after dilution of the compounds versus the observed metal bound C5H5 singlets.
2.1.9 Single-crystal X-ray structure determination
Suitable single crystals of 1a and b·HCl for the X-ray diffraction study were selected. Data collection was performed at 200(2) K, with the crystals covered with perfluorinatedether oil. The crystals were mounted on a Bruker-Nonius Kappa CCD single crystal diffractometer equipped with a graphite-monochromated Mo-Kα radiation (λ = 0.71073 Å). Multiscan [75] absorption correction procedures were applied to the data. The structures were solved, using the WINGX package [76], by direct methods (SHELXS-97) and refined using full-matrix least-squares against F2 (SHELXL-97) [77]. All non-hydrogen atoms were anisotropically refined. Hydrogen atoms were geometrically placed and left riding on their parent atoms except for the hydrogen atoms H1, H2 and H10 in compound b·HCl and H2, H3, H81 and H82 in 1a that were found in the Fourier map and refined. Full-matrix least-squares refinements were carried out by minimizing Σw(Fo2-Fc2)2 with the SHELXL-97 weighting scheme and stopped at shift/err < 0.001. The final residual electron density maps showed no remarkable features. Crystal data for b·HCl: (C17H25ClN2O), FW = 308.84, Orthorhombic, space group P212121, crystal dimensions (mm3) 0.50 × 0.48 × 0.47, a = 9.0817(6), b = 9.2452(5), c = 19.9983(9) Å, V = 1679.10(16) Å3, Z = 4, ρcalcd = 1.222 g cm−3, μ = 0.229 mm−1, F(000) = 664, θ range = 3.001 to 27.498, deg, no. of rflns collected = 13323, no. of indep rflns / Rint = 3847 / 0.0653, no. of data/restraints/params = 3847 / 0 / 202, R1/wR2 (I>2σ(I)) = 0.0409 / 0.0961, R1/wR2 (all data) = 0.0599 / 0.1047, GOF (on F2) = 0.967, Absolute structure parameter = −0.05(3). Final difference Fourier maps did not show peaks higher than 0.303 nor deeper than −0.275 eÅ−3. Crystal data for 1a: (C21H26Cl2N2OTi), FW = 441.24, Orthorhombic, space group P212121, crystal dimensions (mm3) 0.42 × 0.29 × 0.28, a = 10.4465(16), b = 12.885(2), c = 16.065(3) Å, V = 2162.4(6) Å3, Z = 4, ρcalcd = 1.355 g cm−3, μ= 0.656 mm−1, F(000) = 920, θ range = 3.16–27.51 deg, no. of rflns collected = 17759, no. of indep rflns / Rint = 4956 / 0.0700, no. of data/restraints/params = 4956 / 0 / 260, R1/wR2 (I>2σ(I)) = 0.0394 / 0.0813, R1/wR2 (all data) = 0.0767 / 0.0949, GOF (on F2) = 1.059, Absolute structure parameter = −0.03(3). Final difference Fourier maps did not show peaks higher than 0.315 nor deeper than −0.347 eÅ−3. CCDC-1413283 (1a), -1413284 (b·HCl).
CCDC-1413283 and 1413284 contain the supplementary crystallographic data for this paper. Copies of these data can be obtained free of charge on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (fax: (+44)1223-336-033; e-mail: deposit@ccdc.cam.ac.uk)
2.2. Cell culture, inhibition of cell growth and cell death analysis
2.2.1. Cell culture and MTT assays for PC-3 cells
The androgen-unresponsive cell line PC-3 was obtained from the American Type Culture Collection (Manassas, VA) and may be related to recurrent prostate cancers that have achieved androgen independence [78]. All culture media were supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin/amphoterycin B (Life Technologies, Barcelona, Spain). The culture was performed in a humidified 5% CO2 environment at 37 °C. After the cells reached 70–80% confluence, they were washed with phosphate buffered saline (PBS), detached with 0.25% trypsin/0.2% EDTA and seeded at 30,000–40,000 cells/cm2. The culture medium was changed every 3 days.
PC-3 (1 × 105) cells were grown in 96-well plates. After 24 h, cells with RPMI-1640 medium (Roswell Park Memorial Institute) (Mediatech Inc., Manassas, VA) containing 10% FBS and 1% antibiotic/antimycotic (penicillin/streptomycin/amphotericin B) were treated with the compounds at 24 and/or 72 h. Stock solutions of [(η5-C3H5)TiCl3], [(η5-C5H5)2TiCl2] and ammonium-oxime proligands were freshly prepared in 1% of DMSO in water, while oximate titanium compounds were dissolved in water. The stock solutions were then diluted in complete medium and used for sequential dilutions to desired concentrations. The final concentration of DMSO in the cell culture medium did not exceed 0.1%. Control groups with and without DMSO (0.1%) were included in the assays.
The cytotoxic activity of the chemical compounds was screened against PC-3 cell line within a wide concentration range. Cell growth was determined by a tetrazolium assay, which measures the reduction of substrate MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] to a dark blue formazan product by mitochondrial dehydrogenases in living cells. Isopropanol was added to each well to dissolve the formazan precipitate and absorbance values were read at 570 nm using the plate reader Multiskan FC (ThermoScientific, Walthan, Massachusetts, USA) with a reference wavelength at 620 nm [79,80]. Each experiment was repeated at least three times and each concentration was tested in at least six replicates. Results are expressed as a percentage of survival with respect to control cells in the absence of the compound. IC50 values (half-inhibitory concentration) were calculated from curves constructed by plotting cell survival (%) versus compound concentration (M). The IC50 values were calculated with the GraphPad Prism software.
2.2.2. Cell culture and XTT assays for Caki-1, DLD-1, MDA-MB 231 and HEK-293T cells
Human renal carcinoma cells Caki-1, human breast adenocarcinoma cells MDA-MB-231, as well as the human colorectal adenocarcinoma cells DLD-1 in comparison with healthy human embryonic kidney cells HEK-293T, were used to study 2b and 2c’s cytotoxic activity. The cells were all obtained from the American Type Culture Collection (ATCC) (Manassas, Virginia, USA). All the cells were grown adherently. The Caki-1 and DLD-1 cells were cultured in RPMI-1640 media containing 10% FBS, Life Technologies, Grand Island, NY), 1% Minimum Essential Media (MEM) nonessential amino acids (NEAA, Mediatech) and 1% penicillin-streptomycin (Pen Strep, Mediatech). The HEK-293T and MDA-MB-231 cells were maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Mediatech) supplemented with 10% FBS, 1% Minimum Essential Media (MEM) nonessential amino acids (NEAA, Mediatech) and 1 % penicillin-streptomycin (Pen Strep, Mediatech). All cells were cultured at 37° C and 5% CO2 and 95% air in a humidified incubator.
For evaluation of cell viability, cells were seeded at a concentration of 5×10^3 cells/well in 90 μl of DMEM or RPMI-1640 without phenol red and without antibiotics, supplemented with 10% FBS and 2 mM L-glutamine into tissue culture grade 96-well flat bottom microplates (BioLite Microwell Plate, Fisher Scientific, Waltham, Massachusetts, USA) and grown for 24h at 37° C under 5% CO2 and 95% air in a humidified incubator. Cisplatin was dissolved in H2O. All titanium compounds were dissolved in 1% of DMSO in water before addition to cell culture. The final concentration of DMSO in the cell culture medium did not exceed 0.1%. The intermediate dilutions of the compounds were added to the wells to obtain concentrations of 1 μM, 10 μM and 100 μM, and the cells were incubated for 72 h. Control groups with and without DMSO (0.1%) were included in the assays. Following 72 h drug exposure, 50 μL per well of 2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2Htetrazolium-5-carboxanilide (XTT) (Roche Diagnostics Indianapolis, Indiana, USA) labelling mixture was added to the cells at a final concentration of 0.3 mg mL−1 and incubated for 4 h at 37° C under 5% CO2 and 95% air in a humidified incubator. The optical absorbance of each well in a 96-well plate was quantified using BioTek ELx808 absorbance microplate reader (BioTek Winooski, VT) set at 450 nm wavelength. The percentage of surviving cells was calculated from the ratio of absorbance of treated to untreated cells. The IC50 value was calculated as the concentration reducing the proliferation of the cells by 50% and is presented as a mean (± S.E.M) of at least two independent experiments each with triplicate measurements.
2.2.3. Cell viability for Compounds 2b and 2c, cisplatin and Tltanocene Y. ApoTox-Glo Triplex Assay
For apoptosis, viability, and cytotoxicity assays, the Caki-1 cells were seeded in 96-well opaque-walled tissue culture plates with clear bottoms (Thermo Scientific Nunc; Somerset, NJ, USA) at an initial density of 5 × 104 cells/mL in DMEM without phenol red and without antibiotics, supplemented with 10% FBS and 2 mM Lglutamine. Following 24 h incubation, cells were treated with 5 μM of compounds 2b, 2c or 30 μM of cisplatin, Titanocene Y for 72 h. The cells were then assayed using the ApoTox-Glo Triplex Assay (Promega GmbH, High-Tech-Park, Mannheim, Germany). 20 μl of viability/cytotoxicity reagent containing both glycylphenylalanyl-aminofluorocoumarin (GF-AFC) and bis-alanylalanyl-phenylalanyl-rhodamine 110 (bis-AAF-R110) substrates were added to each well, and they were briefly mixed by orbital shaking at 200 rpm for 30 seconds and then incubated at 37 °C for 2 h. Fluorescence was measured at a 400 nm for excitation/505 nm for emission (viability) and 485 nm for excitation/520 nm for emission (cytotoxicity/necrosis) using a BioTek Fluorescence Microplate Reader (BioTek U.S., Winooski, VT)). Next, 100 μl of Caspase-Glo 3/7 reagent was added to each well, and the samples were briefly mixed by orbital shaking at 200 rpm for 30 seconds and then incubated at room temperature for 1 hour. Luminescence was measured for 1 second and is proportional to the amount of caspase activity present (BioTek U.S., Winooski, VT). The results for each treatment were expressed as fold change between non-treated (0.1% DMSO) and treated samples. ApoTox-Glo Triplex assays were repeated twice (n = 2), and each repetition was run in quadruplicate. The average of the four values was used for statistical calculations. The data are presented as the mean value.
2.3. Cell adhesion assay
Concentrated type-I collagen solution (8 μg/cm2) was diluted in 10 mM glacial acetic acid and coated onto 96-well plates for 1 h at 37 °C. Plates were washed twice with PBS (pH 7.4). PC-3 cells were harvested with 0.25% trypsin/0.2% EDTA and collected by centrifugation. They were suspended in RPMI-1640 medium/ 0.1% (w/v) bovine serum albumin (BSA) (pH 7.4) and treated with the complexes for 30 min. Then, cells plated at 2.5 × 104 cells per 100 μl. The assay was terminated at 40 min by aspiration of the wells. Cell adhesion was quantified by MTT colorimetric assay as described above.
2.4. Wound-healing assay
PC-3 cells were seeded in 24-well plates at a density of 1.5×l05 cells in complete media (10% FBS and 1% penicillin/streptomycin/amphoterycin b), and then grown to confluence for 24 h. A small wound area was made with a scraper in the confluent monolayer. The cells were then incubated in either the absence or presence of the complexes. Four representative fields of each wound were captured using a Nikon Dlaphot 300 inverted microscopy at different times (0–6 h). Wound areas of samples at time 0 were averaged and assigned a value of 100%.
2.5. Data analysis
Data were subjected to one-way anova and differences were determined by bonferronl’s multiple comparison test. Each experiment was repeated at least three times. Data are shown as the means of individual experiments and presented as the mean ± S.E.M; P < 0.05 was considered statistically significant.
2.6. Mobility shift assay
2 μl aliquots of pBR322 plasmid DNA (100 μg/mL) in buffer (5 mm Tris/HCl, 50 mM NaClO4, pH = 7.49) were incubated with different concentrations (0.3 mM stock solutions, within the range 0.25–2.0 metal complex:DNAbp) of the ligands a·HCl-c·HCl, and compounds 1a·HCl-1c·HCl, 2a–2c at 37 °C for 20 h in the dark in a total volume of 12 μL. Samples of cisplatin-, TDC-, and (η5-C5H5)TiCl3-DNA were prepared as controls. After the incubation period, the samples were loaded onto the 1 % agarose gel. The samples were separated by electrophoresis for 1.5 h at 83 V in Tris-acetate/EDTA buffer (TAE). Afterwards, the gel was stained for 30 min with a solution of GelRed Nucleic Acid stain.
3. Results and Discussion
3.1. Syntheses and characterization of titanium compounds
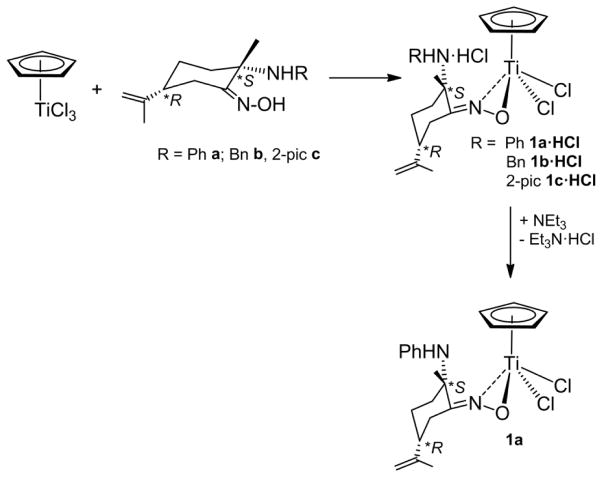
Reactions of [(η5-C5H5)TiCl3] with enantiopure amino-oxime derivatives (2S,5R)-{NOH,(R)NH} (R = Ph a, Bn b, 2-pic c) [63,64] affords new chiral-at-ligand ammonium-oximato titanium compounds (S,R)-[(η5-C5H5)Ti(κ2NO,(R)NH·HCl)Cl2] (R = Ph 1a·HCl, Bn 1b·HCl, 2-pic 1c·HCl) respectively (Fig. 3). The elimination of HCl takes place with subsequent formation of ammonium RNH·HCl fragments within the ligand. Milder reactions conditions are required depending on the nature of the amine substituent, following the order 2-pic > Bn > Ph. Thus, the reaction proceeds in min at room temperature to afford 1c·HCl while warming of the solutions at 50 °C is needed for quantitative formation of 1a·HCl.
Fig. 3.
Synthesis of monocyclopentadienyl oximato titanium compounds
Compounds 1a·HCl-1c·HCl are slightly soluble in toluene, benzene or diethylether, and soluble in dichloromethane or chloroform, which agree well with the presence of the ammonium moiety. In order to obtain crystals suitable for an X-ray diffraction study, synthesis of more soluble amino-oximato titanium compound [(η5-C5H5)Ti(κ2NO,(Ph)NH)Cl2], was attempted. Treatment of 1a·HCl with NEt3, in a stoichiometric ratio 1:1, leads to isolation of pure (S,R)-[(η5-C5H5)Ti(κ2NO,(Ph)NH)Cl2], la, which is formed in solution with the concomitant precipitation of Et3N·HCl. X-ray quality crystals of enantiomerically pure 1a were grown from a saturated toluene solution at −20 °C; an ORTEP diagram is shown in Fig. 4. The X-ray diffraction structure confirms a dihapto κ2NO coordination of the oximato unit to the titanium centre, which is the usual coordination mode found for other titanium(IV) oximato derivatives [52,55–57,59,62,81]. N, O and C atoms of the ON=C unit are coplanar, and the plane that contained them bisects the angle Cl(2)-Ti-Cl(3) along the Ti-Cp centroid vector. Ti-N and Ti-O bond distances are similar to those observed in oximato titanium alkoxide [56,57,59,62] or cyclopentadienyl titanium(IV) hydroxylaminato compounds [82].
Fig. 4.

ORTEP drawing of compound 1a with 30% probability ellipsoids. Hydrogen bonded to carbon atoms have been omitted for clarity. Representative bond lengths (Å) and angles (deg): Ti(1)-O(1) 1.8860(19); Ti(1)-N(1) 2.102(2); N(1)-O(1) 1.401(3); N(1)-C(1) 1.281(3); O(1)-Ti(1)-N(1) 40.71(9); N(1)-O(1)-Ti(1) 77.91(14); O(1)-N(1)-Ti(1) 61.34(11); C(1)-N(1)-Ti(1) 178.9(2); C(6)-N(2)-C(21) 124.3(3).
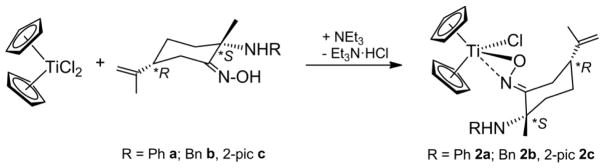
Synthesis of titanocene oximato compounds required the addition of an external base. Thus, reactions of [(η5-C5H5)2TiCl2] with derivatives a–c leads to detection of unreacted starting reagents by 1HNMR. Treatment of [(η5-C5H5)2TiCl2] and amino-oxime derivatives a–c in the presence of NEt3 allows isolation of compounds (S, R)-[(η5-C5H5)2Ti(κ2NO,(R)NH)Cl] (R = Ph 2a, Bn 2b, 2-pic 2c) (Fig. 5), which are formed together with Et3N·HCl.
Fig. 5.
Synthesis of titanocene amino-oximato compounds
Full NMR spectroscopy characterization of the titanium compounds was achieved with the help of bidimensional 1H-1H COSY, 13C-1H HSQC, HMBC and 15N-1H HMBC experiments.
Disappearance of the downfield =NOH proton signal observed in the 1H NMR spectra of free ligands a–c confirm oxime deprotonation and oximato coordination to the metal center. Thus, 1H NMR spectra of 1a·HCl·1c·HCl show broad resonances at δ 11.62 (1a·HCl), 9.84, 10.35 (1b·HCl) and 10.43 (1c·HCl) assigned to NH·HCl hydrogen atoms. The association between the resonances at δ 9.84, 10.35 with those due to the –CH2Ph substituent observed in 1H-1H COSY NMR of compound 1b·HCl, unambiguously confirms the assignation made (see Supplementary data). In contrast, 1H NMR spectra of 1a, 2a–2c show broad resonances at δ 3.71 (1a), 3.53 (2a), 1.25 (2b) and 1.48 (2c), assigned to -NHR protons. Further confirmation of the oximato formation was provided from 13C and 15N-1H HMBC NMR data. Oximato coordination shifts the 13C NMR resonance due to the CqNOTi group (δ 152.8–154.5 (1a·HCl-1c·HCl), 160.5–157.6 (2a–2c)) to higher field relative to the signal assigned to CqNOH in the amino-oxime free ligands (δ 164.9 (a), 163.0 (b), 162.4 (c)). The difference between the chemical shifts of the nitrogen signals arising from the oxime =NOH fragment in proligands a–c (δ 343.5 (a), 340.0 (b), 343.2 (c)) and those found in compounds 1a·HCl-1c·HCl, 1a and 2a–2c (δ 331.8–335.0, 319.0 and δ 398.9–402.1, respectively) also supports the proposed structure for these Ti(IV) compounds. Amino nitrogen resonances found in 15N-1H HMBC NMR spectra of amino-oximato 1a, 2a–2c (δ 85.1 (1a), 86.1 (2a), 60.0 (2b), 52.6 (2c)) keep almost unaltered relative to those found for free derivatives a–c (δ 84.1 (a), 60.0 (b), 51.8 (c)), while signals at δ 79.5 (1a·HCl), 67.3 (1b·HCl) and 56.3 (1c·HCl) are assigned to ammonium nitrogen atoms.
IR spectra of ammonium-oximato 1a·HCl-1c·HCl exhibit strong, broad v(-NH, -NH·HCl) bands at ū 3090–3200 cm−1. In contrast, IR spectra of 1a, 2a–2c show sharp absorptions at ū 3374 (1a), 3370 (2a), 3370 (2b) and 3304 (2c) cm−1, assigned to amino ν(-NH) bands. C=N stretching frequencies of oximato group [54,56,59] are observed in the 1638–1646, 1570–1602 cm−1 regions, with similar wavenumbers relative to those bands found in the spectra of organic derivatives a–c [48].
We attempted quaternization of the amino group present in compounds 2a–2c by reaction of the oximato titanium derivatives with HCl (1M solution in diethylether) or iodomethane. Addition of HCl to tetrahydrofurane, chloroform or toluene solutions of 2a–2c in a Ti:HCl ratio of 1:1 affords corresponding [(η5-C5H5)2TiCl2] and organic compounds a–c, respectively. Reactions of 2a–2c with iodomethane were monitored by 1H NMR spectroscopy (CDCl3 or C6D6 in a J. Young valve NMR tube). Starting reactants were detected after 24 h of reaction at room temperature, with no resonances assignable to any new organometallic compound. An increase of the temperature to 50–90°C gave rise to signals that indicated minor to significant decomposition of compounds 2a–c, while the singlet observed for iodomethane (δ 2.15 (CDCl3) or 1.43 (C6D6)) remained unchanged.
3.2. Hydrolysis behaviour of titanium compounds
In comparison to the limited solubility in water of the starting materials [(η5-C5H5)nTiCl4-n] (n = 1, 2), new compounds 1b·HCl, 1c·HCl, 2b and 2c formed clear yellow solutions (6.6–15.7 mM) after water addition, while derivatives 1a·HCl or 2a are only slightly soluble in water after sonication.
The hydrolytic behaviour of the novel oximate titanium compounds was evaluated by 1H NMR in D2O and D2O:[D8]THF (1:9). Upon dissolution in D2O (1,4-dioxane was used as internal reference [83]), monocyclopentadienyl compounds 1a·HCl-1c·HCl give acidic yellow solutions (2 mM, pD = 2.80-2.56, pD = pH* + 0.4, where pH* = pHmeter reading in D2O [84]), while compounds 2a–2c afford pD values of 4.74 (2a), 5.95 (2b) and 5.62 (2c), respectively, at similar concentrations of ca. 2 mM. Formation of free proligands (2S,5R)-{NOH,(R)NH·HCl} (R = Ph a·HCl, Bn b·HCl, 2-pic c·HCl) was observed right after dilution of mono- or dicyclopentadienyl compounds, and no resonances due to water soluble species with metal bound oximato ligands were apparent from the recorded 1H NMR spectra of any of the novel derivatives. In this regard, we expected that titanocene oximato hydrolysis would afford free amino-oxime compounds a–c, which are insoluble in water. However, resonances due to soluble a·HCl-c·HCl are observed within the first 5 min of dilution, which confirm that both, halide and oximato ligands are rapidly hydrolyzed in aqueous medium. White, X-ray quality crystals of b·HCl were formed from a solution of compound 2b in D2O at room temperature; an ORTEP diagram is shown in Fig. 6. The X-ray molecular structure found confirms the formation of the ammonium-oxime derivative.
Fig. 6.
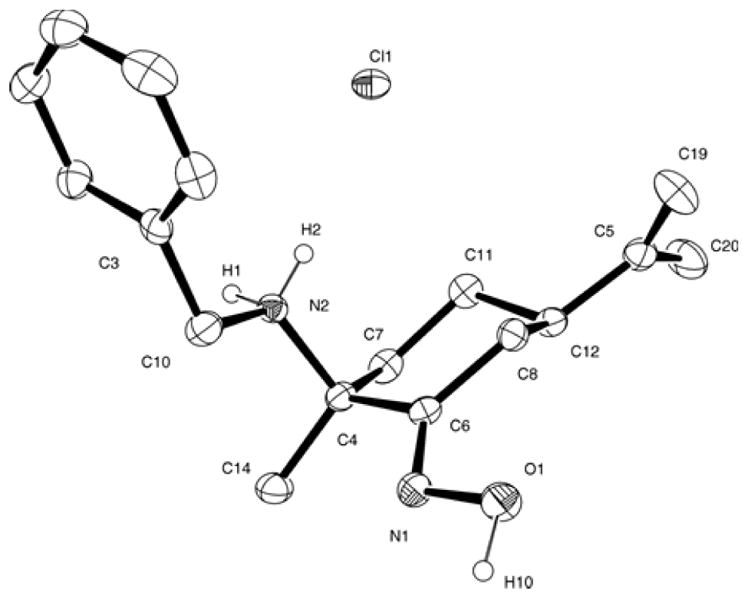
ORTEP drawing of compound b·HCl with 30% probability ellipsoids. Hydrogen bonded to carbon atoms has been omitted for clarity. Representative bond lengths (Å) and angles (deg): N(1)-O(1) 1.400(3); N(1)-C(6) 1.271(4); O(1)-N(1)-C(6) 112.9(2); C(10)-N(2)-C(4) 114.9(2).
The aqueous solution behaviour of [(η5-C5H5)nTiCl4-n] (n = 1, 2) has been well documented [83–89]. Rapid hydrolysis of halide ligands occurs through a series of equilibrium reactions in which cyclopentadienyl rings (Cp) remain metal bound at pH < 3.0–3.5. At higher pH, protonolysis of Cp initiates to give cyclopentadiene as well as insoluble, uncharacterized titanium hydrolysis products [83,84,87,89]. Accordingly, resonances due to cyclopentadiene (multiplets at δ 6.61, 6.55 and 2.98, see Supplementary data) were observed in the recorded 1H NMR spectra of more acidic 1a·HCl-1c·HCl or 2a only after 24 h, while more basic solutions of 2b and 2c exhibit, within the first min after dilution, minor multiplets assigned to free cyclopentadiene. Besides those resonances, the 1H NMR spectra of titanocene derivatives 2a–2c show, during the first 24 h, metal bound Cp species (ca. 80–90% of metal bound Cp relative to 1,4-dioxane after 1 h and less than 10% after 24 h for 2b and 2c) similar to those detected before in D2O solutions of [(η5-C5H5)2TiCl2] at pD = 4.1 (singlets at δ 6.60 and 6.50 [83,87], see Supplementary data). Evaluation of cytotoxic compounds (see Cytotoxicity studies, section 3.3.1) 1c·HCl, 2b or 2c in a 1:9 D2O:[D8]THF mixture by 1H NMR lead to similar results. The first spectrum taken following D2O addition reveal a rapid reaction for all derivatives, with indication of free oxime proligand formation and complete disappearance of cyclopentadienyl resonances corresponding to compounds 1c·HCl, 2b or 2c.
Titanocene compounds 2b and 2c were also evaluated in phosphate buffered saline (PBS) solutions in D2O by 1H and 31P NMR. Their 1H NMR spectra showed minor metal bound Cp species only within the first hour (ca. 2% of metal bound Cp relative to free oxime ligand after 1 h, see Supplementary data), observed always with free proligands and minor resonances due to cyclopentadiene. 31P NMR spectra of the 2b or 2c solutions in PBS do not revealed important changes compared with that of PBS solution in D2O while the spectra recorded within 10 min of adding the titanium compounds were the same as those recorded subsequently (only one broaden signal detectable at δ 2.07, assigned to inorganic phosphates present in the PBS solution [90,91]). Similar results were observed when physiological pH was achieved by dissolving the organometallic derivatives in D2O and adjusting the pD to 7.4 by NaOD addition. Aromatic region of the 1H NMR spectra obtained following the two different methods were analogous.
The results described herein indicate that the novel oximate titanium compounds are rapidly hydrolysed in water to afford free ammonium-oxime proligands. As already described, cyclopentadienyl ring loss rate depends on pH, but no important differences between our systems and [(η5-C5H5)nTiCl4-n] (n = 1, 2) have been found so far at similar pH values.
3.3. In vitro cell studies
Titanocene Y has already shown an encouraging activity in PC-3 tumour-bearing mice [92]. Recently, we published a family of cytotoxic ruthenium oxime compounds able to modulate both adhesion and migratory capabilities of PC-3 cells using concentrations of only 5 μm [48]. Thus, we evaluated the effect of the novel chiral cyclopentadienyl oximato Ti(IV) compounds on cytotoxicity, cell adhesion and migration of the human androgen-independent prostate cancer PC-3. In addition and since some titanocene compounds (such as titanocene Y, titanocene Y*, titanocene T and heterometallic titanocene-gold compounds [14,20–30,33,34,36,37]) have shown to be very active against renal cancer both in vitro and in vivo, we evaluated the cytotoxicity of selected compounds in another three cell lines (human renal Caki-1, human colon DLD-1 and human triple negative breast MDA-MB-231 cancer cell lines). In order to assess the compounds’ selectivity for cancerous cells with respect to normal cell lines, they were also screened for their antiproliferative effects on the non-tumorigenic human embryonic kidney cells HEK-293T. Effects of selected compounds on cell death in vitro were also evaluated and the results are discussed in the next sections.
3.3.1. Cytotoxicity studies
The cytotoxicity of the complexes 1a·HCl-1c·HCl, 2a–2c, a·HCl-c·HCl and [(η5-C5H5)TiCl3] on the human prostate cancer (PC-3) cell line was assessed as the IC50 values after 72 h of incubation. The results are summarized in Table 1.
Table 1.
IC50 (μM) ± S.E.M.a for compounds 1a·HCl-1c·HCl, 2a–2c and a·HCl-c·HCl in the androgen-independent prostate cancer PC-3 cells after 72 h of incubation.
| Compound | IC50 values |
|---|---|
| 1a·HCl | 121.3 ± 5.7 |
| 1b·HCl | 113 ± 6.5 |
| 1c·HCl | 25.7 ± 8.6 |
| 2a | 139.2 ± 8.4 |
| 2b | 59.3 ± 6.8 |
| 2c | 17.3 ± 5.2 |
| a·HCl | 201.7 ± 9.4 |
| b·HCl | 177 ± 5.5 |
| c·HCl | 204.7 ± 15.6 |
| [(η5-C5H5)TiCl3] | >300 |
Each value represents the mean ± S.E.M. of at least three independent experiments.
Although starting organometallic systems [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2] [27] and water soluble proligands a·HCl-c·HCl are poorly cytotoxic (IC50 > 170 μM), oximato titanium compounds 1c·HCl and 2c are more effective than Titanocene Y [27,92], under similar experimental conditions, with an inhibitory activity of 75% at concentrations of 50 μm. Other titanium compounds which have proved their antitumor activity against PC-3 cells line are Schiff-base titanium (IV) derivatives [93] (IC50 values within the range 5–18 μM) or heterometallic titanocene-gold compounds (IC50 values ranged from 25–40 μM) [27].
The antiproliferative effect of the most active derivatives 1c·HCl, 2b and 2c were also assessed after an exposure time of 24 h, showing inhibitory activities of 20–30% at concentrations of 50 μM (see Supplementary data).
We also studied the effect of the combination of organometallic derivatives [(η5-C5H5)2TiCl2] or [(η5-C5H5)TiCl3] with proligands b·HCl or c-HCl on the cellular viability. The additive dose of [(η5-C5H5)TiCl2] and b·HCl or c·HCl produced lower antiproliferative effects than those observed after treatment with only 2b or 2c. This fact indicates that there is indeed a synergistic effect of these novel compounds 2b and 2c in their in vitro activity on PC-3 cell lines. In contrast, addition of a combined dose consisting of [(η5-C5H5)TiCl3] and proligands b·HCl or c·HCl did not cause any significant difference in cell viability compared to that found for 1b·HCl or 1c·HCl, respectively (see Supplementary data). These findings suggest that the activity of compounds 2b and 2c does not result from that of free ligands and titanocene cations following ligand dissociation, as could be inferred from our results on their hydrolytic behaviour, but rather involves an active titanium species whose formation is positively influenced by the presence of the oximato ligand within pro-drugs 2b or 2c.
Thus, compounds 2b and 2c were selected for further studies in vitro on renal cancer (Caki-1), colon cancer (DLD-1), breast cancer (MDA-MB-23) and non-tumorigenic human renal (HEK-293T) cell lines. The cytotoxicity of 2b and 2c was assayed by monitoring their ability to inhibit cell growth using the XTT assay (see Experimental Section) after 72 h of incubation with the drug (since we had already seen that the IC50 values greatly decreased overtime for PC-3 cell lines). The results are collected in Table 2. In this table we have included IC50 values already reported with the same cell lines and incubation times for control compounds TDC, titanocene Y and cisplatin [27,30].
Table 2.
IC50 values (μM) ± S.E.M.a in human cell lines were determined with 2b and 2c. Reported values for cisplatin, [(η5-C5H5)2TiCl2], and Titanocene Y are indicated for some cell lines.
The IC50 values obtained show clearly that there is a selectivity of the new titanium compound 2c for the human prostate and the renal cancer cell lines over the triple negative breast and the colon cancer cell lines. The values for 2b and 2c in Caki-1 cell lines are lower than those reported for cisplatin, TDC and even for titanocene Y, which is a benchmark for titanocene derivatives in this cell line. Very importantly, compounds 2b and especially 2c are considerably less toxic to the non-tumorigenic human embryonic kidney cell line (HEK-293T) than to Caki-1 renal cells (2c is a 15-fold less toxic). Both compounds display low IC50 values in the range of other improved-titanocene derivatives described so far in Caki-1 cancer cell lines [35,36]. While heterometallic titanocene derivatives have been described recently with IC50 in the low micromolar or sub-micromolar range [27,30], 2b and especially 2c, represent good examples of discrete, easily accessible organometallic compounds containing solely a titanium centre. Their cytotoxicity in PC-3 and Caki-1 cancer cell lines, their selectivity and solubility properties in water warrant further exploration of these compounds as potential prostate and renal anticancer agents.
3.3.2. Mechanism of Cell Death
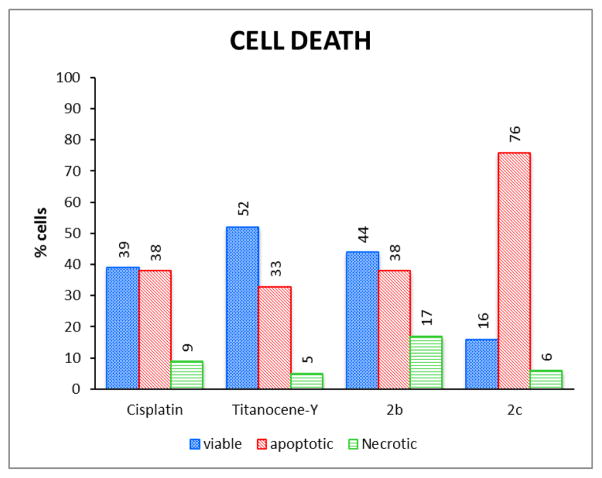
We selected cytotoxic compounds 2b and 2c as models to evaluate the mechanism of cell death in renal Caki-1 cancer cells (Fig. 7). We also evaluated the mechanism of cell death in Caki-1 (under the same conditions and using the approximate IC50 concentration) of cisplatin and Titanocene Y. Caki-1 viability, necrosis, and apoptotic activity were analysed by the ApoTox-Glo Triplex (see Experimental Section) which combines three chemical assays to assess viability, cytotoxicity, and caspase activation events within a single well. Viability and cytotoxicity are inversely proportional, such that if the viability of cells is reduced, cytotoxicity is usually stimulated. In general, the cytotoxicity may be due to programmed cell death (apoptosis) or primary necrosis. The treatment of Caki-1 cancer cells with 5 μM of 2b or 2c induces apoptosis in 38% and 76% of cells respectively. The treatment of these cells with 30 μM (ca. IC50 value for both cisplatin and Titanocene Y [27]) induced apoptosis in 38% and 33% respectively. Those results clearly point to apoptosis as the principal mode of cell death in Caki-1 cells treated with oximato titanocene compounds 2b and 2c. Other titanocenes like TDC, Titanocene C (bis-[N,N-dimethylamino-2(N-methylpyrrolyl)-methyl-cyclopentadienyl]titanium(IV) dichloride), X ([1,2-di(cyclopentadienyl)-1,2-di-(4-N,N-dimethylaminophenyl)ethanediyl]titanium(IV) dichloride) and Y, and heterometallic complexes containing titanocene fragments are also known to induce apoptosis in different cancer cell lines [16,21,27,30,94,95]. Moreover, as shown in Figure 7, compound 2c in a concentration 6 times lower than that of cisplatin and Titanocene Y (one of the benchmarks for activity in Caki-1 cancer cells and tumours) reduces the viability of cells in a much higher percentage and is more apoptotic.
Fig. 7.
Compounds 2b, 2c, cisplatin and Titanocene Y impaired the viability of Caki-1 cells by inducing apoptosis. Following a 72 h incubation period, the compounds (5 μM) 2b or 2c or (30 μM) Cisplatin or Titanocene Y reduced Caki-1 viability by 61% (cisplatin), 48% (Titanocene Y), 56% (2b) and 84% (2c); induced necrosis in 9% (cisplatin), 5% (Titanocene Y), 17% (2b) and 6% (2c), and induced apoptosis in 38% (cisplatin), 33% (Titanocene Y), 38 % (2b) and 76 % (2c) of treated cells. The effect of cisplatin and Titanocene Y, 2b and 2c on viability, necrosis and apoptosis was assessed by measuring protease activity using the non-cell-permeable substrates, cell-permeable substrates and by measuring the total caspase-3 and -7 activities with the ApoTox-Glo Triplex Assay. The effect of each treatment was determined by comparing treated and untreated cells. From the Apotox assay a cell population that was neither necrotic nor apoptotic was detected and their mode of death could not be assigned. Those cells were not included in the graph.
3.3.3. Cell adhesion
The oximato titanium complexes 1c·HCl, 2b and 2c, which showed the highest antiproliferative effects on the PC-3 cell line, were further evaluated on cell adhesion to type-I collagen in order to determine their effect on metastatic capability of human androgen-independent prostate PC-3 cancer cells. Tumour metastasis is the process by which a tumour cell leaves the primary tumour, travels to a distinct site and establishes a secondary tumour. The disruption of intercellular structures and cell-cell local interaction (cell adhesion) can lead to the dissociation of the primary tumour and an enhanced potential for dissemination and metastatic spread of cancer cells to secondary location [96–100]. To investigate the effect of our compounds on the cell adhesion in vitro, we incubated PC-3 cells in the absence or presence of 50 μM of 1c·HCl, 2b or 2c on a collagen plate. In order to establish a comparison, we also performed the experiment on the starting materials [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2] and c·HCl. PC-3 cells rapidly adhered to collagen basement in 40 min. The adhesion pattern exhibited by organometallic derivative [(η5-C5H5)2TiCl2] (32%) was similar to that of cyclopentadienyl oximato titanium compounds (1c·HCl (30%), 2b (32%), 2c (40%), Fig. 8), all of them showing a very significant increase of the adhesion ability relative to that observed for control cells. Monocyclopentadienyl and organic ammonium-oxime derivatives, [(η5-C5H5)TiCl3] (17%) and c·HCl (14%), displayed a lower adhesion ability than that of oximato titanium systems [48].
Fig. 8.
Effect of [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2], c·HCl, 1c·HCl, 2b and 2c derivatives on adhesion of PC-3 cells to type-I collagen was studied after treatment with organic and organometallic compounds for 40 min. Data are the mean ± S.E.M of at least four experiments. *, P < 0.05; ***, P < 0.001 versus Control.
3.3.4. Cell migration
To the best of our knowledge, the effect of organometallic titanium compounds on the migratory capability of cancer cells has been scarcely studied. In this regard, the treatment of human 518A2 melanoma cells with schisandroxymethyltitanocene for 24 h revealed a similar improvement on the cell migration as [(η5-C5H5)2TiCl2]. Both titanocene derivatives were able to prevent wound healing in the cancer cells colonies, with dose concentrations of 0.1 μM [101]. Very recently, a heterometallic Ti-Au compound described by the group of Contel and co-workers has demonstrated anti-migratory effects on Caki-1 cells with concentrations of 5 μM [30].
The wound-healing assay is a useful method for gauging the anti-migratory activity of drug candidates. In order to evaluate cell migration we performed wound-healing assays in which a small wound area was made on the plate with a confluent monolayer of cells.
After 6 h of incubation, the wound in the control wells was almost fully repopulated with migrated cells (Fig. 9). The cells treated with 50 μM of complexes 1c·HCl, 2b and 2c showed a significant lower migration capability (62–72% of wound healing) than that of control cells, while compounds [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2] and c·HCl treated cells exhibited a non-significant lower migration ability than that of oximato titanium systems (Fig. 9).
Fig. 9.
Effect of [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2] c·HCl, 1c·HCl, 2b and 2c on cell migration was studied in human androgen-independent prostate cancer PC-3 cell line. Microscopic analysis of the cell-free area was carried out at the indicated time (6 h) after the addition of the complexes and the width of the area invaded by prostate cells was estimated.
3.4. Interactions with plasmid DNA
DNA is one of the critically important targets in cancer chemotherapy. Although the nature of the active species of titanium anticancer drugs and their mechanism of action remained unresolved, the notion of DNA being their probable target was supported by early works, which found that Ti localizes to the chromatin and inhibits DNA synthesis [1]. However, later investigations indicated that titanium interacts only weakly with DNA at neutral pH. Thus, below pH 5, titanium compounds such as [(η5-C5H5)2TiCl2] or Ti(IV) hydrolysis species bind to nucleotides, notably to the phosphate units, but binding is generally very weak near pH 7 [15,16,19,84,89,102,103]. Weak DNA interactions between Titanocene Y and DNA double helix have been recently found [36,104].
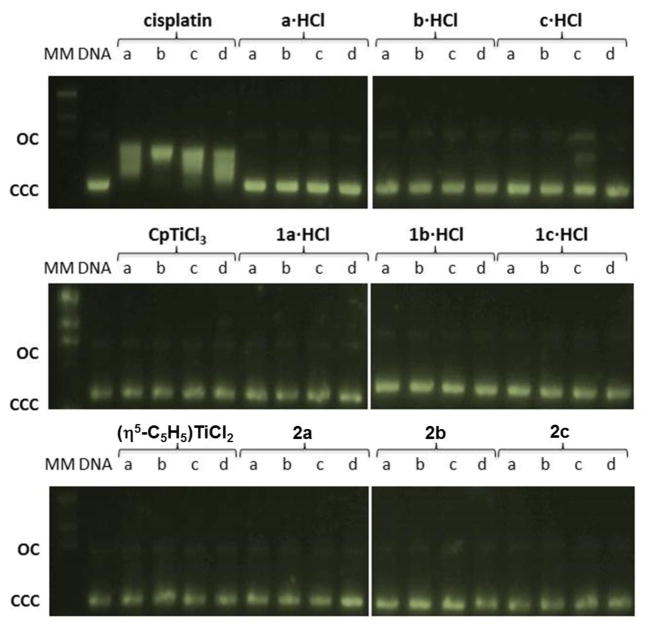
We performed agarose gel electrophoresis studies to evaluate the effects of the compounds 1a·HCl-1c·HCl, 2a–2c, a·HCl-c·HCl, [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2] and cisplatin on plasmid (pBR322) DNA. The plasmid has two main forms; form I (supercoiled or covalently closed, CCC) and II (relaxed or open circular, OC). The electrophoretic patterns of plasmid DNA incubated with increasing amounts of cisplatin, organic derivatives a·HCl-c·HCl and titanium complexes [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2], 1a·HCl-1c·HCl and 2a–2c, are shown in Fig. 10. Changes in electrophoretic mobility of both forms are usually taken as evidence of metal-DNA binding. Unwinding of the DNA causes a retardation of the fast running band of the CCC form, whereas condensation and cross-linking of DNA accelerates the migration of the OC form. Binding of cisplatin to plasmid DNA results in a decrease in mobility of the CCC form and an increase in mobility of the OC form (see lanes a–d for cisplatin in Figure 10) [27,30,44,105–107]. At physiological pH, none of the tested ammonium-oxime or Ti(IV) derivatives showed any important impact on the DNA mobility. This fact suggests either that the compounds do not interact with DNA or that interaction is too weak to be detected by this technique.
Fig. 10.
Electrophoresis mobility shift assays for cisplatin and compounds [(η5-C5H5)TiCl3], [(η5-C5H5)2TiCl2], a·HCl-c·HCl, 1a·HCl-1c·HCl and 2a–2c (see Experimental Section for details). DNA refers to untreated plasmid pBR322. Lanes a, b, c and d correspond to metal/DNAbp ratios of 0.25, 0.5, 1.0 and 2.0 respectively. OC open circular or relaxed form (Form II), CCC covalently closed or supercoiled form, (Form I).
Conclusions
The results presented here and those reported before by us on ruthenium derivatives, support the idea that the use of the enantiopure amino-oxime ligands a–c is a useful strategy to design easily accessible chiral and water soluble metal compounds with potential as anticancer agents. The novel oximato Ti(IV) compounds exhibit a different DNA binding behaviour from that found in cisplatin. The studies on the hydrolytic behaviour of the new titanium derivatives did not permit the detection of water soluble titanium oximato-containing hydrolysis product. However, titanocene derivatives 2b and 2c have demonstrated to have synergistic effects relative to additive doses of TDC and free proligands. This fact suggests the existence of titanium active species whose formation is positively influenced by the presence of the oximato ligands. Compound 2c displays some of the lower IC50 values described so far in PC-3 and Caki-1 cancer cell lines for titanocene derivatives containing solely a titanium centre, while showing selectivity against renal cancer when compared to the non-tumorigenic human renal HEK-293T cell line. The cell death type for compounds 2b and specially 2c is mainly through apoptosis. Furthermore, 2b and 2c show interesting in vitro results on cell adhesion and migration of the androgen-independent prostate cancer PC-3.
Supplementary Material
Highlights.
We report the synthesis of new enantiopure cyclopentadienyl oximato Ti(IV) compounds.
The novel compounds affect the adhesion and migration patterns of PC-3 cancer cells.
2b and 2c are more cytotoxic against Caki-1 cancer cells than cisplatin or titanocene Y.
2b and specially 2c preferentially induce apoptosis in Caki-1 cancer cells.
2b and 2c show selectivity against Caki-1 compared to non-tumorigenic HEK-293T cells.
Acknowledgments
Financial support from the Comunidad Autónoma de Madrid (CAM, I3 Program, Ref. 3090XF067) and the Universidad de Alcalá(UAH, Project CCG2014/EXP-025) is acknowledged. Y.B. acknowledges the Agencia Española de Cooperación Internacional (AECI) for fellowship. Brooklyn College (The City University of New York) authors thank the National Cancer Institute (NCI) for grant 1S1CA182844 (to M.C.). Authors would like to thank Dr. Jacob Fernandez-Gallardo for performing the mobility shift assay with pBR322 plasmid DNA.
Abbreviations
- ANOVA
Analysis of Variance
- Budotitane
cis-diethoxybis(1-phenylbutane-1,3-dionato)titanium(IV)
- Caki-1
Human renal cancer cell line
- Cisplatin
(SP-4-2)-diamminedichloroplatinum(II)
- COSY
Correlation Spectroscopy
- DLD
Human promyelocytic leukemia cell line
- DLD-1
Human colon cancer cell line
- DMEM
Dulbecco’s Modified Eagle’s Medium
- DMSO
dimethyl sulfoxide
- DNA
deoxyribonucleic acid
- EDTA
ethylendiaminetetraacetic acid
- Et
ethyl
- FBS
Fetal Bovine Serum
- FTIR
Fourier Transformed Infrared spectroscopy
- HEK-293T
Non-tumorigenic human embryonic kidney cell line
- HeLa
Human cervical cancer cell line
- HL60
Human promyelocytic leukemia cell line
- HSQC
Heteronuclear Single Quantum Coherence spectroscopy
- HMBC
Heteronuclear Multiple Bond Correlation spectroscopy
- MDA-MB-231
Triple negative human breast cancer cell line
- MEM
Minimum Essential Media
- MTT
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NMR
Nuclear Magnetic Resonance
- NEAA
Nonessential Amino Acids
- PBS
Phosphate Buffered Saline
- PC-3
Human androgen-independent prostate cancer cell line
- RPMI
Roswell Park Memorial Institute medium
- TDC
Titanocene dichloride
- THF
tetrahydrofuran
- SEM
Standard Error of Mean
- Titanocene C
bis-[N,N-dimethylamino-2-(N-methylpyrrolyl)-methyl-cyclopentadienyl] titanium(IV) dichloride
- Titanocene T
bis-[{(1-methyl-3-diethylaminomethyl)indol-4-yl}methyl cyclopentadienyl]titanium(IV) dichloride
- Titanocene X
[1,2-di(cyclopentadienyl)-1,2-di-(4-N,N-dimethylaminophenyl)ethanediyl]titanium(IV) dichloride
- Titanocene Y
bis-[(p-methoxybenzyl)cyclopentadienyl]titanium(IV) dichloride
- Titanocene Y*
bis-[(p-diethylaminobenzyl)cyclopentadienyl]titanium(IV) dichloride
- XTT
2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)-2Htetrazolium-5-carboxanilide
Appendix A. Supplementary data
Supplementary data associated with this article can be found in the online version, at http://. These data include: Representative NMR spectra of compounds a–c, a·HCl-c·HCl, 1a·HCl-1c·HCl, 2a–2c, representative time dependent 1H NMR spectra in D2O and PBS solutions, cell viability of representative compound 2c after 24 and 72 h, comparison between the cytotoxicity of compounds 1b·HCl, 1c·HCl, 2b, 2c and [(η5-C5H5)TiCl3]+b·HCl, +c·HCl or [(η5-C5H5)2TiCl2]+b·HCl, +c·HCl on cell viability of PC-3 cells; bond lengths and angles for X-ray molecular structures of 1a and b·HC1.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Keppler BK. Metal Complexes in Cancer Chemotherapy. Wiley VCH; Weinheim: 1993. [Google Scholar]
- 2.Barnes KR, Lippard SJ. In: Metal Complexes in Tumor Diagnosis and as Anticancer Agents. Sigel A, Sigel H, editors. Vol. 42. Marcel Dekker Inc; 2004. pp. 179–208. [Google Scholar]
- 3.van Rijt SH, Sadler PJ. Drug Discov Today. 2009;14:1089–1097. doi: 10.1016/j.drudis.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garbutcheon-Singh KB, Grant MP, Harper BW, Krause-Heuer AM, Manohar M, Orkey N, Aldrich-Wright JR. Curr Top Med Chem. 2011;11:521–542. doi: 10.2174/156802611794785226. [DOI] [PubMed] [Google Scholar]
- 5.Gasser G, Ott I, Metzler-Nolte N. J Med Chem. 2011;54:3–25. doi: 10.1021/jm100020w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muhammad N, Guo ZJ. Curr Opin Chem Biol. 2014;19:144–153. doi: 10.1016/j.cbpa.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 7.Hartmann M, Keppler BK. Comments Inorganic Chem. 1995;16:339–372. doi: 10.1080/02603599508035776. [DOI] [Google Scholar]
- 8.Keppler BK, Schmahl D. Arzneimittelforschung. 1986;36-2:1822–1828. [PubMed] [Google Scholar]
- 9.Keller HJ, Keppler B, Schmahl D. Arzneimittelforschung. 1982;32-2:806–807. [PubMed] [Google Scholar]
- 10.Kopf H, Kopfmaier P. Angew Chem-Int Edit Engl. 1979;18:477–478. doi: 10.1002/anie.197904771. [DOI] [PubMed] [Google Scholar]
- 11.Kopfmaier P, Grabowski S, Kopf H. Eur J Med Chem. 1984;19:347–352. [Google Scholar]
- 12.Keppler BK, Friesen C, Moritz HG, Vongerichten H, Vogel E. Struct Bond. 1991;78:97–127. [Google Scholar]
- 13.Caruso F, Rossi M. Mini-Rev Med Chem. 2004;4:49–60. doi: 10.2174/1389557043487565. [DOI] [PubMed] [Google Scholar]
- 14.Tshuva EY, Peri D. Coord Chem Rev. 2009;253:2098–2115. doi: 10.1016/j.ccr.2008.11.015. [DOI] [Google Scholar]
- 15.Buettner KM, Valentine AM. Chem Rev. 2012;112:1863–1881. doi: 10.1021/cr1002886. [DOI] [PubMed] [Google Scholar]
- 16.Olszewski U, Hamilton G. Anti-Cancer Agents Med Chem. 2010;10:302–311. doi: 10.2174/187152010791162261. [DOI] [PubMed] [Google Scholar]
- 17.Korfel A, Scheulen ME, Schmoll HJ, Grundel O, Harstrick A, Knoche M, Fels LM, Skorzec M, Bach F, Baumgart J, Sass G, Seeber S, Thiel E, Berdel WE. Clin Cancer Res. 1998;4:2701–2708. [PubMed] [Google Scholar]
- 18.Hartinger CG, Dyson PJ. Chem Soc Rev. 2009;38:391–401. doi: 10.1039/b707077m. [DOI] [PubMed] [Google Scholar]
- 19.Abeysinghe PM, Harding MM. Dalton Trans. 2007:3474–3482. doi: 10.1039/b707440a. [DOI] [PubMed] [Google Scholar]
- 20.Strohfeldt K, Tacke M. Chem Soc Rev. 2008;37:1174–1187. doi: 10.1039/b707310k. [DOI] [PubMed] [Google Scholar]
- 21.Hogan M, Tacke M. In: Titanocenes: Cytotoxic and Anti-angiogenic Chemotherapy Against Advanced Renal-Cell Cancer. Jaouen G, MetzlerNolte N, editors. Vol. 32. Springer-Verlag Berlin; Berlin: 2010. pp. 119–140. [Google Scholar]
- 22.Pelletier F, Comte V, Massard A, Wenzel M, Toulot S, Richard P, Picquet M, Le Gendre P, Zava O, Edafe F, Casini A, Dyson PJ. J Med Chem. 2010;53:6923–6933. doi: 10.1021/jm1004804. [DOI] [PubMed] [Google Scholar]
- 23.Wenzel M, Bertrand B, Eymin MJ, Comte V, Harvey JA, Richard P, Groessl M, Zava O, Amrouche H, Harvey PD, Le Gendre P, Picquet M, Casini A. Inorg Chem. 2011;50:9472–9480. doi: 10.1021/ic201155y. [DOI] [PubMed] [Google Scholar]
- 24.Gonzalez-Pantoja JF, Stern M, Jarzecki AA, Royo E, Robles-Escajeda E, Varela-Ramirez A, Aguilera RJ, Contel M. Inorg Chem. 2011;50:11099–11110. doi: 10.1021/ic201647h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hartinger CG, Metzler-Nolte N, Dyson PJ. Organometallics. 2012;31:5677–5685. [Google Scholar]
- 26.Ceballos-Torres J, Virag P, Cenariu M, Prashar S, Fajardo M, Fischer-Fodor E, Gomez-Ruiz S. Chem-Eur J. 2014;20:10811–10828. doi: 10.1002/chem.201400300. [DOI] [PubMed] [Google Scholar]
- 27.Fernandez-Gallardo J, Elie BT, Sulzmaier FJ, Sanau M, Ramos JW, Contel M. Organometallics. 2014;33:6669–6681. doi: 10.1021/om500965k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Deally A, Hackenberg F, Lally G, Muller-Bunz H, Tacke M. Organometallics. 2012;31:5782–5790. doi: 10.1021/om300227h. [DOI] [Google Scholar]
- 29.Glasner H, Meker S, Tshuva EY. J Organomet Chem. 2015;788:33–35. doi: 10.1016/j.jorganchem.2015.04.023. [DOI] [Google Scholar]
- 30.Fernandez-Gallardo J, Elie BT, Sadhukha T, Prabha S, Sanau M, Rotenberg SA, Ramos JW, Contel M. Chem Sci. 2015 doi: 10.1039/c5sc01753j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gao LM, Vera JL, Matta J, Melendez E. J Biol Inorg Chem. 2010;15:851–859. doi: 10.1007/s00775-010-0649-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hernandez R, Lamboy J, Gao LM, Matta J, Roman FR, Melendez E. J Biol Inorg Chem. 2008;13:685–692. doi: 10.1007/s00775-008-0353-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fichtner I, Pampillon C, Sweeney NJ, Strohfeldt K, Tacke M. Anti-Cancer Drugs. 2006;17:333–336. doi: 10.1097/00001813-200603000-00012. [DOI] [PubMed] [Google Scholar]
- 34.Hilger RA, Alex D, Deally A, Gleeson B, Tacke M. Lett Drug Des Discov. 2011;8:904–910. [Google Scholar]
- 35.Claffey J, Muller-Bunz H, Tacke M. J Organomet Chem. 2010;695:2105–2117. doi: 10.1016/j.jorganchem.2010.05.025. [DOI] [Google Scholar]
- 36.Fichtner I, Behrens D, Claffey J, Deally A, Gleeson B, Patil S, Weber H, Tacke M. Lett Drug Des Discov. 2011;8:302–307. [Google Scholar]
- 37.Walther W, Fichtner I, Deally A, Hogan M, Tacke M. Lett Drug Des Discov. 2013;10:375–381. [Google Scholar]
- 38.Meker S, Margulis-Goshen K, Weiss E, Magdassi S, Tshuva EY. 2012;51:10515–10517. doi: 10.1002/anie.201205973. [DOI] [PubMed] [Google Scholar]
- 39.Glasner H, Tshuva EY. Inorg Chem. 2014;53:3170–3176. doi: 10.1021/ic500001j. [DOI] [PubMed] [Google Scholar]
- 40.Ganot N, Redko B, Gellerman G, Tshuva EY. RSC Adv. 2015;5:7874–7879. doi: 10.1039/c4ra13484b. [DOI] [Google Scholar]
- 41.Schur J, Manna CM, Deally A, Koster RW, Tacke M, Tshuva EY, Ott I. Chem Commun. 2013;49:4785–4787. doi: 10.1039/c3cc38604j. [DOI] [PubMed] [Google Scholar]
- 42.Scaffidi-Domianello YY, Legin AA, Jakupec MA, Roller A, Kukushkin VY, Galanski M, Keppler BK. Inorg Chem. 2012;51:7153–7163. doi: 10.1021/ic300148e. [DOI] [PubMed] [Google Scholar]
- 43.Scaffidi-Domianello YY, Meelich K, Jakupec MA, Arion VB, Kukushkin VY, Galanski M, Keppler BK. Inorg Chem. 2010;49:5669–5678. doi: 10.1021/ic100584b. [DOI] [PubMed] [Google Scholar]
- 44.Bartel C, Bytzek AK, Scaffidi-Domianello YY, Grabmann G, Jakupec MA, Hartinger CG, Galanski M, Keppler BK. J Biol Inorg Chem. 2012;17:465–474. doi: 10.1007/s00775-011-0869-5. [DOI] [PubMed] [Google Scholar]
- 45.Ossipov K, Scaffidi-Domianello YY, Seregina IF, Galanski M, Keppler BK, Timerbaev AR, Bolshov MA. J Inorg Biochem. 2014;137:40–45. doi: 10.1016/j.jinorgbio.2014.04.008. [DOI] [PubMed] [Google Scholar]
- 46.Wirth S, Rohbogner CJ, Cieslak M, Kazmierczak-Baranska J, Donevski S, Nawrot B, Lorenz IP. J Biol Inorg Chem. 2010;15:429–440. doi: 10.1007/s00775-009-0615-4. [DOI] [PubMed] [Google Scholar]
- 47.Chitrapriya N, Mahalingam V, Zeller M, Lee H, Natarajan K. J Mol Struct. 2010;984:30–38. doi: 10.1016/j.molstruc.2010.09.004. [DOI] [Google Scholar]
- 48.Benabdelouahab Y, Munoz-Moreno L, Frik M, de la Cueva-Alique I, El Amrani MA, Contel M, Bajo AM, Cuenca T, Royo E. Eur J Inorg Chem. 2015:2295–2307. doi: 10.1002/ejic.201500097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kukushkin VY, Pombeiro AJL. Coord Chem Rev. 1999;181:147–175. doi: 10.1016/s0010-8545(98)00215-x. and references therein. [DOI] [Google Scholar]
- 50.Milios CJ, Stamatatos TC, Perlepes SP. Polyhedron. 2006;25:134–194. doi: 10.1016/j.poly.2005.07.022. [DOI] [Google Scholar]
- 51.Smith AG, Tasker PA, White DJ. Coord Chem Rev. 2003;241:61–85. doi: 10.1016/s0010-8545(02)00310-7. [DOI] [Google Scholar]
- 52.Thewalt U, Friedrich R. ZNaturforsch(B) 1991;46:475–482. [Google Scholar]
- 53.Pandey OP, Sengupta SK, Tripathi CM. Molecules. 2005;10:653–658. doi: 10.3390/10060653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chaudhary A, Sharma N, Dhayal V, Saxena A, Nagar M, Bohra R. Appl Organomet Chem. 2011;25:198–206. doi: 10.1002/aoc.1742. [DOI] [Google Scholar]
- 55.Carvalho M, Galvao AM, Kredatusova J, Merna J, Pinheiro PF, Salema MM. Inorg Chim Acta. 2012;383:244–249. doi: 10.1016/j.ica.2011.11.019. [DOI] [Google Scholar]
- 56.Baumann SO, Bendova M, Fric H, Puchberger M, Visinescu C, Schubert U. Eur J Inorg Chem. 2009:3333–3340. doi: 10.1002/ejic.200900381. [DOI] [Google Scholar]
- 57.Baumann SO, Bendova M, Puchberger M, Schubert U. Eur J Inorg Chem. 2011:573–580. doi: 10.1002/ejic.201000881. [DOI] [Google Scholar]
- 58.Mirzaee M, Norouzi M, Faghani M, Amini MM, Khavasi HR. Transit Met Chem. 2014;39:55–62. doi: 10.1007/s11243-013-9773-x. [DOI] [Google Scholar]
- 59.Chaudhary A, Dhayal V, Nagar M, Bohra R, Mobin SM, Mathur P. Polyhedron. 2011;30:821–831. doi: 10.1016/j.poly.2010.12.025. [DOI] [Google Scholar]
- 60.Samuel B, Tummalapalli K, Giri PV, Pathak M. Med Chem Res. 2014;23:699–707. doi: 10.1007/s00044-013-0660-y. [DOI] [Google Scholar]
- 61.Samuel B, Ethiraj KR, Pathak M. Med Chem Res. 2015;24:1504–1513. doi: 10.1007/s00044-014-1234-3. [DOI] [Google Scholar]
- 62.Davidson MG, Johnson AL, Jones MD, Lunn MD, Mahon MF. Polyhedron. 2007;26:975–980. doi: 10.1016/j.poly.2006.09.055. [DOI] [Google Scholar]
- 63.Brecknell DJ, Carman RM, Singaram B, Verghese J. Aust J Chem. 1977;30:195–203. doi: 10.1071/ch9770195. [DOI] [Google Scholar]
- 64.Tkachev AV, Rukavishnikov AV, Chibiryaev AM, Denisov AY, Gatilov YV, Bagryanskaya IY. Aust J Chem. 1992;45:1077–1086. [Google Scholar]
- 65.Larionov SV. Russ J Coord Chem. 2012;38:1–23. doi: 10.1134/s1070328412010058. and references therein. [DOI] [Google Scholar]
- 66.Kilpin KJ, Cammack SM, Clavel CM, Dyson PJ. Dalton Trans. 2013;42:2008–2014. doi: 10.1039/c2dt32333h. [DOI] [PubMed] [Google Scholar]
- 67.Manna CM, Armony G, Tshuva EY. Inorg Chem. 2011;50:10284–10291. doi: 10.1021/ic201340m. [DOI] [PubMed] [Google Scholar]
- 68.Fu Y, Soni R, Romero MJ, Pizarro AM, Salassa L, Clarkson GJ, Hearn JM, Habtemariam A, Wills M, Sadler PJ. Chem-Eur J. 2013;19:15199–15209. doi: 10.1002/chem.201302183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Blanck S, Maksimoska J, Baumeister J, Harms K, Marmorstein R, Meggers E. Angew Chem-Int Edit. 2012;51:5244–5246. doi: 10.1002/anie.201108865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Gomez-Ruiz S, Kaluderovic GN, Polo-Ceron D, Prashar S, Fajardo M, Zizak Z, Juranic ZD, Sabo TJ. Inorg Chem Commun. 2007;10:748–752. doi: 10.1016/j.inoche.2007.03.016. [DOI] [PubMed] [Google Scholar]
- 71.Gomez-Ruiz S, Kaluderovic GN, Prashar S, Polo-Ceron D, Fajardo M, Zizak Z, Sabo TJ, Juranic ZD. J Inorg Biochem. 2008;102:1558–1570. doi: 10.1016/j.jinorgbio.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 72.Miller M, Tshuva EY. Eur J Inorg Chem. 2014;2014:1485–1491. doi: 10.1002/ejic.201301463. [DOI] [Google Scholar]
- 73.Herrmann WA. Synthetic Methods of Organometallic and Inorganic Chemistry. Vol. 1. Thieme Verlag; 1996. p. 51. [Google Scholar]
- 74.Sweeney NJ, Mendoza O, Muller-Bunz H, Pampillon C, Rehmann FJK, Strohfeldt K, Tacke M. J Organomet Chem. 2005;690:4537–4544. doi: 10.1016/j.jorganchem.2005.06.039. [DOI] [Google Scholar]
- 75.Blessing RH. Acta Crystallogr Sect A. 1995;51:33–38. doi: 10.1107/s0108767394005726. [DOI] [PubMed] [Google Scholar]
- 76.Farrugia LJ. J Appl Crystallogr. 2012;45:849–854. doi: 10.1107/s0021889812029111. [DOI] [Google Scholar]
- 77.Sheldrick GM. Acta Crystallogr Sect A. 2008;64:112–122. doi: 10.1107/s0108767307043930. [DOI] [PubMed] [Google Scholar]
- 78.Kaighn ME, Narayan KS, Ohnuki Y, Lechner JF, Jones LW. 1979;17:16–23. [PubMed] [Google Scholar]
- 79.Mosmann T. J Immunol Methods. 1983;65:55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 80.Zhang YS, Cai X, Yao JJ, Xing WX, Wang LHV, Xia YN. Angew Chem-Int Edit. 2014;53:184–188. doi: 10.1002/anie.201306282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Maurer C, Pittenauer E, Puchberger M, Allmaier G, Schubert U. ChemPlusChem. 2013;78:343–351. doi: 10.1002/cplu.201300014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Dove AP, Kiesewetter ET, Ottenwaelder X, Wayrnouth RM. Organometallics. 2009;28:405–412. doi: 10.1021/om800571j. [DOI] [Google Scholar]
- 83.Erxleben A, Claffey J, Tacke M. J Inorg Biochem. 2010;104:390–396. doi: 10.1016/j.jinorgbio.2009.11.010. [DOI] [PubMed] [Google Scholar]
- 84.Guo ML, Guo ZJ, Sadler PJ. J Biol Inorg Chem. 2001;6:698–707. doi: 10.1007/s007750100248. [DOI] [PubMed] [Google Scholar]
- 85.Kopf H, Grabowski S, Voigtlander R. J Organomet Chem. 1981;216:185–190. doi: 10.1016/s0022-328x(00)85759-9. [DOI] [Google Scholar]
- 86.Toney JH, Marks TJ. J Am Chem Soc. 1985;107:947–953. doi: 10.1021/ja00290a033. [DOI] [Google Scholar]
- 87.Doppert K. Naturwissenschaften. 1990;77:19–24. doi: 10.1007/bf01131789. [DOI] [Google Scholar]
- 88.Carofiglio T, Floriani C, Sgamellotti A, Rosi M, Chiesivilla A, Rizzoli C. J Chem Soc-Dalton Trans. 1992:1081–1087. doi: 10.1039/dt9920001081. [DOI] [Google Scholar]
- 89.Mokdsi G, Harding MM. J Organomet Chem. 1998;565:29–35. doi: 10.1016/s0022-328x(98)00441-0. [DOI] [Google Scholar]
- 90.Qu Y, Kipping RG, Farrell NP. Dalton Trans. 2015;44:3563–3572. doi: 10.1039/c4dt03237c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Turner BL, Mahieu N, Condron LM. Soil Sci Soc Am J. 2003;67:497–510. [Google Scholar]
- 92.Dowling CM, Claffey J, Cuffe S, Fichtner I, Pampillon C, Sweeney NJ, Strohfeldt K, Watson RWG, Tacke M. Lett Drug Des Discov. 2008;5:141–144. doi: 10.2174/157018008783928463. [DOI] [Google Scholar]
- 93.Obeid A, El-Shekeil A, Al-Aghbari S, Al-Shabi J. J Coord Chem. 2012;65:2762–2770. doi: 10.1080/00958972.2012.703780. [DOI] [Google Scholar]
- 94.Cuffe S, Dowling CM, Claffey J, Pampillon C, Hogan M, Fitzpatrick JM, Carty MP, Tacke M, Watson RWG. Prostate. 2011;71:111–124. doi: 10.1002/pros.21227. [DOI] [PubMed] [Google Scholar]
- 95.Kater L, Claffey J, Hogan M, Jesse P, Kater B, Strauss S, Tacke M, Prokop A. Toxicol Vitro. 2012;26:119–124. doi: 10.1016/j.tiv.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 96.Hanahan D, Weinberg RA. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 97.Jiang WG, Sanders AJ, Katoh M, Ungefroren H, Gieseler F, Prince M, Thompson SK. Semin Cancer Biol. 2015 doi: 10.1016/j.semcancer.2015.03.008. in press. [DOI] [PubMed] [Google Scholar]
- 98.Harlozinska A. Anticancer Res. 2004;24:3508–3508. [PubMed] [Google Scholar]
- 99.Mareel M, Leroy A. Physiol Rev. 2003;83:337–376. doi: 10.1152/physrev.00024.2002. [DOI] [PubMed] [Google Scholar]
- 100.Woodhouse EC, Chuaqui RF, Liotta LA. Cancer. 1997;80:1529–1537. doi: 10.1002/(sici)1097-0142(19971015)80:8+<1529::aid-cncr2>3.3.co;2-#. [DOI] [PubMed] [Google Scholar]
- 101.Gmeiner A, Effenberger-Neidnicht K, Zoldakova M, Schobert R. Appl Organomet Chem. 2011;25:117–120. doi: 10.1002/aoc.1725. [DOI] [Google Scholar]
- 102.McLaughlin ML, Cronan JM, Schaller TR, Snelling RD. J Am Chem Soc. 1990;112:8949–8952. doi: 10.1021/ja00180a046. [DOI] [Google Scholar]
- 103.Yang P, Guo ML. Coord Chem Rev. 1999;185–6:189–211. doi: 10.1016/s0010-8545(98)00268-9. [DOI] [Google Scholar]
- 104.Lally G, Deally A, Hackenberg F, Quinn SJ, Tacke M. Lett Drug Des Discov. 2013;10:675–682. [Google Scholar]
- 105.Keck MV, Lippard SJ. J Am Chem Soc. 1992;114:3386–3390. doi: 10.1021/ja00035a033. [DOI] [Google Scholar]
- 106.Perrin LC, Prenzler PD, Cullinane C, Phillips DR, Denny WA, McFadyen WD. J Inorg Biochem. 2000;81:111–117. doi: 10.1016/s0162-0134(00)00092-1. [DOI] [PubMed] [Google Scholar]
- 107.Xu ZH, Zhang YM, Xue ZQ, Yang XL, Wu ZY, Guo ZJ. Inorg Chim Acta. 2009;362:2347–2352. doi: 10.1016/j.ica.2008.10.021. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.