Abstract
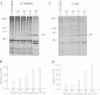
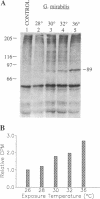
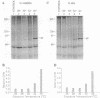
Two extremely eurythermal goby fishes, Gillichthys mirabilis and Gillichthys seta, which encounter habitat temperature variations of approximately 30 degrees C, showed seasonal acclimatization of endogenous levels and of onset temperatures for enhanced synthesis of a 90-kDa-class heat shock protein (HSP90). Summer-acclimatized fishes had higher levels of HSP90 in brain tissue than winter-acclimatized specimens, as shown by Western blot analysis. For winter-acclimatized fishes, increased synthesis of HSP90 was observed when the temperature was raised from a control temperature (18 degrees C) to 28 degrees C. For summer-acclimatized fish, no significantly increased synthesis of HSP90 occurred until the experimental temperature was raised to 32 degrees C. These data suggest that the threshold temperature at which enhanced expression of HSP-encoding genes occurs is not hard-wired genetically but may be subject to acclimatization. A causal relationship between seasonal changes in steady-state levels of HSP90 and the threshold temperature for enhanced HSP90 synthesis is discussed in terms of existing models for the regulation of HSP gene expression.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blake M. J., Fargnoli J., Gershon D., Holbrook N. J. Concomitant decline in heat-induced hyperthermia and HSP70 mRNA expression in aged rats. Am J Physiol. 1991 Apr;260(4 Pt 2):R663–R667. doi: 10.1152/ajpregu.1991.260.4.R663. [DOI] [PubMed] [Google Scholar]
- Craig E. A., Gross C. A. Is hsp70 the cellular thermometer? Trends Biochem Sci. 1991 Apr;16(4):135–140. doi: 10.1016/0968-0004(91)90055-z. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lindquist S. The heat-shock response. Annu Rev Biochem. 1986;55:1151–1191. doi: 10.1146/annurev.bi.55.070186.005443. [DOI] [PubMed] [Google Scholar]
- Perisic O., Xiao H., Lis J. T. Stable binding of Drosophila heat shock factor to head-to-head and tail-to-tail repeats of a conserved 5 bp recognition unit. Cell. 1989 Dec 1;59(5):797–806. doi: 10.1016/0092-8674(89)90603-x. [DOI] [PubMed] [Google Scholar]
- Sorger P. K., Nelson H. C. Trimerization of a yeast transcriptional activator via a coiled-coil motif. Cell. 1989 Dec 1;59(5):807–813. doi: 10.1016/0092-8674(89)90604-1. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]