Abstract
The potato (Solanum tuberosum) has been cultivated globally for food for millenia. Potato contains proteinase inhibitor II, which catalyzes the release of cholecystokinin (CCK), leading to delayed gastric emptying in humans. The present study investigated the anti-obesity effects of Slendesta™ Potato Extract (SLD), a standardized potato extract containing 5% proteinase inhibitor II, in the ob/ob obese mice. Three doses of SLD (50, 150 or 300 mg/kg) were orally administered to ob/ob mice once a day for 28 days, whereas control mice were administered distilled water. Four weeks after SLD treatment, the changes in body weight, food consumption, epididymal fat weight, serum chemistry, insulin, leptin and adiponectin contents, and fat histopathology were determined and compared with ob/ob mice treated with 750 mg/kg conjugate linoleic acid. As a result of SLD treatment in the obese mice, body weight, food consumption, epididymal fat, serum biochemistry, histomorphological changes of fat and pancreas were significantly and dose-dependently decreased compared with ob/ob control mice. These obesity and type 2 diabetes associated alterations were significantly inhibited after SLD treatment for 28 days. Thus, the present results indicate that SLD has potential as an alternative therapeutic agent for obesity.
Keywords: ob/ob mouse, obesity, potato extract, potato proteinase inhibitor II
Introduction
The worldwide incidence of obesity associated with metabolic syndrome continues to escalate, despite the increased awareness and global efforts to understand its pathogenesis and develop treatments (1). Dysregulated energy homeostasis may result from a reduction in physical activity, increased energy-dense food availability and overconsumption, combined with genetic, social and economic complicating factors (2). Among the critical determinants for the development of obesity may be an increase in the regional distribution of body fat (i.e., abdominal obesity). Abdominal obesity may present with a number of atherogenic risk factors, including hypertension, dyslipidemia, alterations in coagulation and inflammatory cytokine profiles, and hyperinsulinemic insulin resistance (3). As an expected consequence, there has been an increase in morbidity and mortality due to cardiovascular disease (4).
Prior studies have been conducted to elucidate the association between increased adiposity and insulin resistance. Adipokines (2) including leptin and adiponectin, which are secreted by adipocytes, may modulate the sensitivity of insulin, whose action activates multiple signaling events following phosphorylation of insulin receptor, among other molecules, in patients with type 2 diabetes (5–7).
A number of oral anti-diabetic medicines including thiazolidinediones and metformin are currently used for improving insulin resistance (8). However, currently available medications for metabolic syndrome exhibit various adverse effects and high rates of secondary failure (9). Thus, there has been a growing interest and focus on complementary and alternative approaches to metabolic syndrome (10).
Conjugated linoleic acid (CLA) refers to a group of isomers of linoleic acid (cis-9, cis-12 octadecadienoic acid). CLA has been reported to possess certain anti-carcinogenic (11), anti-atherogenic (12) and immunomodulatory (13) activities, in addition to exhibiting anti-obesity and anti-diabetic effects (14,15). In addition, CLA treatment during adipocyte differentiation reduces lipid accumulation and inhibits the expression of peroxisome proliferator-activated receptor gamma, a nuclear receptor that activates genes involved in lipid storage and metabolism (16,17). A CLA dosage of 750 mg/kg has been shown to confer anti-obesity and anti-diabetic effects on ob/ob obese mice (18).
The potato tuber (Solanum tuberosum) is the source of potato proteinase inhibitor (PPI) II, which elicits a satiety response (19) and delays gastric emptying in humans (20). While several methods to isolate and purify PPI II have been developed for use in a laboratory setting, they are all laborious and expensive (21–23). Considering the low yield and complexity of PPI II isolation, crude PPI concentrate containing a number of thermostable proteinase inhibitors, including PPI II, has previously been developed (24). This crude concentrate exhibited satiety-promoting activity in vivo and enhanced the cholecystokinin (CCK) release in a similar manner to PPI II (25). Slendesta® Potato Extract 5% Powder (SLD) is a standardized potato extract that contains 5% PPI II. The present study was conducted to determine the anti-obesity and anti-diabetic effects of SLD on ob/ob mice, which are leptin-deficient and which is an established mouse model of obesity. The highest dosage of SLD used was 300 mg/kg. In previous clinical and in vivo studies (25,26), which employed ~15 mg/kg PPI II, satiety response and anti-obesity effects were observed.
The present study investigated the anti-obesity and anti-diabetic effects of SLD, with the aim of determining its potential as a complementary or alternative agent for the management of metabolic syndrome. SLD dosages of 50, 150 and 300 mg/kg were administered orally once a day for 28 days to ob/ob mice. Body weight, food consumption, organ weight, serum chemistry and cytokines associated with obesity and diabetes were measured.
Materials and methods
Materials
SLD, a light beige-colored powder, was provided by Kemin Food L.C. (Des Moines, IA, USA). SLD was composed of 78.9% carbohydrates, 15.3% protein and ~20 mg/g Na. CLA capsules containing a light pink solution were purchased from RexGene Biotech Co., Ltd., (Chungwon-gun, South Korea). SLD was stored at 4°C in a refrigerator, and CLA capsules were stored in a desiccator to protect them from light and humidity until use. SLD and CLA were dissolved or suspended in distilled water on the day of first administration.
Animals
Five normoglycemic wild-type C57BL/6JJms mice (5-weeks-old; Japan SLC, Inc., Shizuoka, Japan), and 25 male genetically obese mice (C57BL/6JHam-ob/ob 5-weeks-old; SLC) were used following acclimatization for 28 days. The mice (n=5 per polycarbonate cage) were housed in a room controlled for temperature (20–25°C), humidity (40–45%) and alternating 12-h light/dark cycles. Standard rodent chow (Samyang Foods, Wonju, Korea) and water were supplied ad libitum. SLD (50, 150 or 300 mg/kg) or CLA (750 mg/kg) solutions were administered by gastric gavage once a day for 28 days. Equal volumes of distilled water were orally administered in the wild-type and ob/ob control mice. All mice were sacrificed on Day 28 post-administration by exsanguination following intramuscular injection of Zoletil 50 (0.05 ml/kg; Virbac Korea Co. Ltd., Seoul, Korea). The present study was approved by the Institutional Animal Care and Use Committee of Daegu Hanny University (IRB approval no. DHU2011-016; Gyeongsan, Republic of Korea).
Body weight
Body weight was measured once a day for 28 days, from one day prior to the initiation of administration (Day −1) using an automatic electronic balance (Precisa Gravimetrics AG, Dietikon, Switzerland). At the initiation of administration (Day 0) and at sacrifice (Day 28), all experimental animals were fasted overnight (~12 h, with no restriction of water intake) to reduce the differences from feeding.
Mean daily food consumption
An equal quantity of food (150 g) was supplied to each cage, and the remaining food was weighed after 24 h using the aforementioned automatic electronic balance. These measurements were conducted on Days 1, 7, 14, 21, 25 and 27. The difference between the supplied and remaining diet represented was the individual mean daily food consumption of mice (g/day/mouse).
Organ weight
Following sacrifice, the weights of the pancreas and epididymal fat pad were measured. To reduce the differences from individual body weights, relative weights (%) were calculated using the body weight at sacrifice.
Blood glucose levels
At sacrifice, blood was collected from the caudal vena cava under anesthesia. The collected blood was deposited into a sodium fluoride glucose vacuum tube (BD Biosciences, Franklin Lakes, NJ, USA), and plasma was separated by centrifugation at 1,100 × g for 10 min at room temperature. Blood glucose levels were detected using an automated blood analyzer (TBA-200FR; Toshiba, Tokyo, Japan).
Plasma insulin levels
To detect plasma insulin levels, blood was collected at sacrifice via the caudal vena cava under anesthesia, and the collected blood was deposited in heparin pretreated vacuum tubes (BD Biosciences) for plasma separation. Plasma insulin levels were assayed with enzyme-linked immunosorbent assay kit (ELISA; Boehringer Mannheim GmbH, Mannheim, Germany) as previously detailed (27).
Serum biochemical parameters
At sacrifice, ~1 ml blood was collected from the caudal vena cava under anesthesia. All blood samples were centrifuged at 21,130 × g for 10 min at 4°C using a clotting activated serum tube (Vacutte®; Greiner Bio-One GmbH, Kremsmünster, Austria). Next, levels of serum total cholesterol (TC), free fatty acid (FFA) and triglyceride were detected using an Olympus AU400 automated blood analyzer (Olympus Corporation, Tokyo, Japan).
Serum leptin and adiponectin levels
To quantify serum levels of leptin and adiponectin, serum was separated from the collected blood using a standard method and ELISAs were performed as previously described (28). Briefly, serum leptin levels were determined using a Mouse Leptin ELISA (cat. no. EZML-82K; EMD Millipore, Billerica, MA, USA) and serum adiponectin levels were detected using a Mouse/Rat adiponectin ELISA kit (Otsuka Pharmaceutical Co., Ltd., Tokyo, Japan), according to the manufacturer's protocols.
Histopathology
After measuring the organ weight, the splenic lobes of pancreas, epididymal fat pad and dorsal abdominal fat pad attached on the muscularis quadratus lumborum were sampled. Sampled tissues were fixed in 10% neutral buffered formalin (Sigma-Aldrich, St. Louis, MO, USA). After paraffin embedding (Leica Microsystems GmbH, Wetzlar, Germany), 3–4 µm serial sections were prepared. Representative sections were stained with hematoxylin and eosin (H&E; Sigma-Aldrich) for light microscopy (Eclipse 80i; Nikon Corporation, Tokyo, Japan) examination. The histological profiles of individual organs were observed.
Histomorphometry
Based on the previous histomorphometrical analysis of metabolic mice (29) or diabetic rats (27,30), adipocyte deposition and hypertrophy, percentages of pancreatic zymogen granule occupied regions, pancreatic islet numbers, and occupied percentages were determined. The mean diameters of epididymal and dorsal abdominal white adipocytes were calculated as an indicator of adipocyte hypertrophy in restricted view fields on a computer monitor using an automated image analysis system (iSolution FL ver. 9.1; IMT iSolution Inc., Quebec, Canada). The mean diameter was expressed in µm and a minimum of ten white adipocytes per each fat pad was measured. Thickness of dorsal abdominal fat pad (mm) was measured using an automated image analysis process. Mean area occupied by zymogen granules were calculated as percentages between one field of liver (%/mm2 of pancreatic parenchyma) using an automated image analysis process. Mean number of pancreatic islets were counted using an automated image analysis process located in appropriate regions of pancreas (n/10 mm2 of pancreatic parenchyma). Mean areas occupied by the pancreatic islets was calculated as percentages between the appropriate pancreas field (%/10 mm2 of pancreatic parenchyma) using an automated image analysis process. The samples were analyzed by the same histopathologist who was blinded to sample identity.
Immunohistochemistry
Additional prepared serial sectioned tissues were immunostained using an immunohistochemical avidin-biotin-peroxidase kit (Vectastain Elite ABC kit; cat. no. PK-6200; Vector Laboratories, Inc., Burlingame, CA, USA), according to a previously described method (31). Briefly, the tissue sections were incubated with guinea pig anti-insulin (1:100; cat. no. ab7842; Abcam, Cambridge, UK) or rabbit anti-glucagon (1:50; cat. no. ab8055; Abcam) polyclonal antibodies. Based on previous histomorphometrical analyses of endocrine cells of normal ddN mice (32) and diabetic rats (27,30), cells displaying >10% immunoreactive density of insulin and glucagon were regarded as positive immunoreactive (IR). The number of insulin- and glucagon-IR cells and the ratio of insulin-IR to glucagon-IR cells were determined. Mean numbers of insulin-IR and glucagon-IR cells were counted in restricted view fields (mm2 of pancreatic parenchyma) on a computer monitor using an automated image analysis process. The aforementioned ratio was determined based on these values. The samples were analyzed by the same histopathologist who was blinded to sample identity.
Statistical analysis
All values are expressed as the mean ± standard deviation. Statistical analyses were conducted using SPSS for Windows, version 14.0K (SPSS, Inc., Chicago, IL, USA). Multiple comparison tests for different dose groups were conducted. Variance homogeneity was assessed using the Levene test. If the Levene test indicated no significant deviations from variance homogeneity, the obtain data were analyzed by one-way analysis of variance test followed by the least-significant differences (LSD) multi-comparison test. In cases where significant deviations from variance homogeneity were observed with the Levene test, the Kruskal-Wallis H test was conducted. When a significant difference was observed in the Kruskal-Wallis H test, the Mann-Whitney U-Wilcoxon Rank Sum W test was conducted to determine the specific pairs of group comparison. The criterion for statistical significance was set at either P<0.05 or P<0.0.1.
Results
Body weight change
Prior to the initiation of administration of the study products, all ob/ob mice displayed significantly (P<0.01) increased body weight compared with the wild-type mice. Significantly decreased body weight was detected in mice treated with CLA and SLD (150 and 300 mg/kg dose) from days 20, 22 and 23 compared with ob/ob control. Furthermore, the body weight gain during the 28-day administration period was decreased in these treatment groups compared with ob/ob control, and this reduction was significant in the 150 and 300 mg/kg SLD groups (Table I).
Table I.
Body weight detected during 28 days of oral treatment of SLD and CLA in ob/ob mice.
| SLD groups (mg/kg) | ||||||
|---|---|---|---|---|---|---|
| Time point | Control | ob/ob control | CLA | 50 | 150 | 300 |
| Day −1 | 24.40±1.03 | 42.94±4.71a | 42.58±2.91 | 42.74±4.00 | 42.96±2.28 | 42.60±4.36 |
| Day 0 | 21.22±0.94 | 39.60±4.50a | 39.90±2.73 | 40.10±4.63 | 39.50±2.56 | 39.70±4.65 |
| Day 7 | 25.38±1.06 | 46.30±4.16a | 43.42±2.98 | 44.16±3.13 | 43.70±2.98 | 41.88±4.33 |
| Day 14 | 25.44±1.34 | 46.88±4.07a | 41.56±2.93 | 44.82±1.37 | 43.24±3.30 | 41.14±4.45 |
| Day 20 | 26.20±1.16 | 50.22±4.52a | 42.72±2.94b | 48.50±1.51 | 43.82±2.73 | 42.40±4.53 |
| Day 21 | 25.66±1.20 | 49.84±4.78a | 42.08±2.47b | 48.98±1.00 | 43.46±2.69 | 42.06±4.59 |
| Day 22 | 26.04±1.25 | 50.74±5.01a | 43.12±3.27b | 50.06±0.92 | 44.16±2.44 | 41.88±5.17b |
| Day 23 | 25.86±1.26 | 50.26±4.63a | 42.42±2.90b | 49.58±1.47 | 43.58±2.64b | 41.52±5.16b |
| Day 28 | 23.82±1.24 | 48.46±4.03a | 40.08±2.75c | 47.96±1.38 | 41.82±2.29b | 39.50±5.23c |
| Gaind | 2.60±0.40 | 8.50±1.48a | 0.18±1.74c | 7.86±4.52 | 2.32±0.72c | −0.20±2.31c |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. control
P<0.05
P<0.01 vs. ob/ob control
Body weight gain (g) throughout Days 0–28; SLD, Slendesta™; CLA, conjugated linoleic acid; MW, Mann-Whitney U-Wilcoxon Rank Sum W test.
Mean daily food consumption
Significant (P<0.01) increases of mean daily food consumption were detected in all ob/ob mice compared with wild-type control. However, mice treated with CLA and 150 or 300 mg/kg SLD exhibited significant (P<0.01) decreases in mean daily food consumption from Day 14 compared to the ob/ob control (Table II).
Table II.
Mean daily food consumption during 27 days of oral treatment with SLD or CLA in ob/ob mice.
| Food consumption (g/day/mouse) | ||||||
|---|---|---|---|---|---|---|
| Group | Day 1 | Day 7 | Day 14 | Day 21 | Day 25 | Day 27 |
| Controls | ||||||
| Intact | 5.12±0.59 | 4.68±0.48 | 4.62±0.54 | 4.84±0.54 | 4.74±0.69 | 4.60±0.53 |
| ob/ob | 7.36±0.60a | 6.84±0.40a | 6.94±0.50a | 7.18±0.60a | 6.88±0.47a | 6.66±0.46a |
| CLA | 7.32±0.41a | 6.62±0.52a | 5.20±1.04b | 5.64±0.53b | 4.46±0.23b | 4.32±0.97b |
| SLD (mg/kg) | ||||||
| 50 | 7.14±0.26a | 6.86±0.29a | 6.78±0.76a | 7.18±0.91a | 6.74±0.55a | 6.48±0.28a |
| 150 | 7.06±0.59a | 6.50±0.55a | 5.74±0.47b,c | 5.80±0.73b,c | 5.54±0.38b,c | 5.16±0.11b |
| 300 | 7.06±0.47a | 6.42±0.44a | 5.62±0.35b,c | 5.18±0.84b | 4.36±0.79b | 4.00±0.70b |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test)
P<0.01 vs. ob/ob control (LSD test)
P<0.05 vs. intact control (LSD test). SLD, Slendesta™; CLA, conjugated linoleic acid.
Epididymal fat and pancreatic weight change
Significant (P<0.01) increases in epididymal fat pad weight were detected in ob/ob control mice compared with the wild-type control at sacrifice. However, these increases were significantly (P<0.01) decreased by treatment of CLA and 150 or 300 mg/kg SLD. Mice treated with 50 mg/kg SLD showed similar epididymal fat pad weights as compared with ob/ob control mice (Table III). Significant (P<0.01) decreases in relative pancreas weight were detected in all ob/ob mice regardless of treatment, compared with the wild-type control. However, no meaningful changes in absolute pancreas weight were evident in the CLA and SLD treated-mice compared with the ob/ob control mice (Table III).
Table III.
Organ weight in ob/ob mice orally administered SLD or CLA.
| Absolute organ weight (g) | Relative organ weight (% of body weight) | |||
|---|---|---|---|---|
| Group | Epididymal fats | Pancreas | Epididymal fats | Pancreas |
| Controls | ||||
| Intact | 0.078±0.024 | 0.118±0.014 | 0.323±0.079 | 0.494±0.054 |
| ob/ob | 1.145±0.287a | 0.138±0.042 | 2.353±0.477a | 0.287±0.095a |
| CLA | 0.634±0.121a,b | 0.113±0.028 | 1.573±0.205a,b | 0.280±0.063a |
| SLD (mg/kg) | ||||
| 50 | 1.083±0.176a | 0.139±0.036 | 2.260±0.376a | 0.289±0.070a |
| 150 | 0.637±0.117a,b | 0.132±0.024 | 1.520±0.248a,b | 0.314±0.042a |
| 300 | 0.545±0.105a,b | 0.126±0.011 | 1.391±0.265a,b | 0.324±0.059a |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test).
P<0.01 vs. ob/ob control (LSD test). SLD, Slendesta™; CLA, conjugated linoleic acid.
Blood glucose
Significantly (P<0.01) increased blood glucose levels were detected in ob/ob control mice compared to the wild-type control. However, the blood glucose levels were significantly (P<0.01) reduced by the treatment with CLA and SLD (150 and 300 mg/kg) compared to the ob/ob control. Mice treated with 50 mg/kg SLD displayed similar blood glucose levels as the ob/ob control (Table IV).
Table IV.
Serum biochemical parameters and adiponectin levels in ob/ob mice orally administered SLD or CLA.
| Group | Glucose (mg/dl) | TC (mg/dl) | FFA (mmol/l) | Triglyceride (mg/dl) | Adiponectin (µg/ml) | Leptin (ng/ml) | Insulin (pM) |
|---|---|---|---|---|---|---|---|
| Controls | |||||||
| Intact | 138.20±10.06 | 106.20±6.61 | 0.62±0.08 | 87.60±12.84 | 22.06±3.37 | 8.64±1.22 | 368.20±40.14 |
| ob/ob | 598.00±48.98a | 289.00±18.22a | 1.77±0.17a | 131.40±8.44a | 8.98±1.46b | 1.30±0.17a | 5,043.00±350.75b |
| CLA | 484.80±43.75a,c | 247.60±8.99a,c | 1.42±0.09a,c | 106.80±10.33a,c | 8.70±0.33b | 1.36±0.35a | 4,000.60±217.20b |
| SLD (mg/kg) | |||||||
| 50 | 602.40±67.01a | 280.60±14.01a | 1.73±0.16a | 128.60±6.66a | 9.39±1.03b | 1.23±0.32a | 5,168.00±265.60b |
| 150 | 428.20±43.42a,c | 247.80±10.18a,c | 1.35±0.11a,c | 101.00±9.96c,d | 12.22±1.44b,e | 1.26±0.31a | 3,651.80±836.52b,f |
| 300 | 383.00±54.30a,c | 232.20±21.02a,c | 1.18±0.15a,c | 94.60±9.13c | 13.53±1.12b,f | 1.26±0.17a | 3,082.60±122.49b,f |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test)
P<0.01 vs. intact control (MW test)
P<0.01 vs. ob/ob control (LSD test)
P<0.05 vs. intact control (LSD test)
P<0.05
P<0.01 vs. ob/ob control (MW test). SLD, Slendesta™; CLA, conjugated linoleic acid; TC, total cholesterol; FFA, free fatty acid.
Plasma insulin levels
Significantly (P<0.01) increased plasma insulin levels were detected in ob/ob control compared with the wild-type control. However, these increases were significantly (P<0.01) inhibited by treatment with SLD (150 and 300 mg/kg) compared to the ob/ob control. In mice treated with 50 mg/kg SLD, the plasma insulin levels were similar to the levels in the ob/ob control mice (Table IV).
Serum TC, FFA and triglyceride levels
Significant (P<0.01) increases of serum TC levels were detected in ob/ob control compared to the wild-type control. However, serum TC levels were significantly (P<0.01) decreased in mice treated with CLA and 150 or 300 mg/kg SLD compared to the ob/ob control (Table IV). Significant (P<0.01) increases in serum FFA and triglyceride levels were detected in the ob/ob control compared with the wild-type control. However, these increases in serum FFA and triglyceride levels were significantly (P<0.01) decreased by treatment with CLA and 150 or 300 mg/kg SLD compared with the ob/ob control (Table IV).
Serum leptin and adiponectin levels
Significant (P<0.01) decreases of serum leptin levels were detected in ob/ob control compared to the wild-type control. However, similar serum leptin levels were detected in mice treated with the three doses of SLD and CLA compared with the ob/ob control (data not shown). Significant (P<0.01) decreases of the serum adiponectin levels were detected in ob/ob control mice compared to the wild-type control. However, the serum adiponectin levels were significantly (P<0.01) increased in mice treated with 150 and 300 mg/kg SLD compared to the ob/ob control. In mice treated with CLA and 50 mg/kg SLD, the serum adiponectin levels were similar to the levels in ob/ob control mice (Table IV).
Adipocyte diameters and the deposited abdominal fat pad thickness
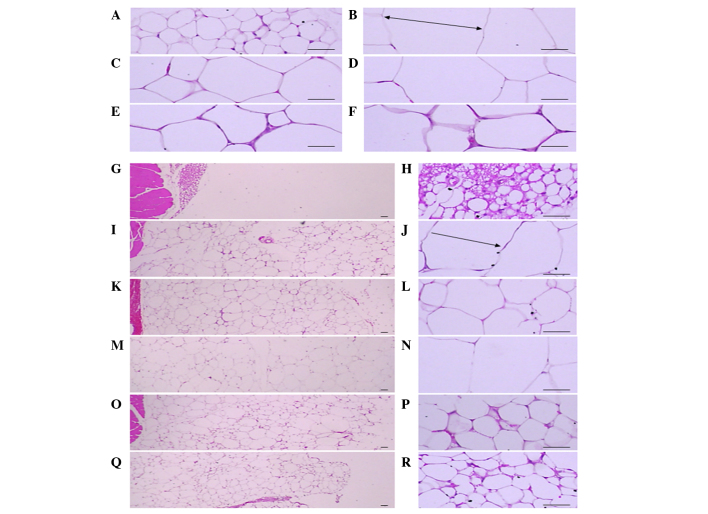
Significant (P<0.01) increases of epididymal and abdominal adipocyte diameters were detected in the ob/ob control compared to the wild-type control. However, diameters of both adipocyte types were significantly (P<0.01) decreased in mice treated with CLA and SLD (150 and 300 mg/kg) compared to the ob/ob control. Mice treated with 50 mg/kg SLD showed a similar trend compared to the ob/ob control (Table V and Fig. 1). Significant (P<0.01) increases of the deposited abdominal fat pad thickness were detected in the ob/ob control mice compared to the wild-type control. However, the abdominal fat pad thickness was significantly (P<0.01) decreased in mice with CLA and SLD (150 and 300 mg/kg) compared to the ob/ob control. In mice treated with 50 mg/kg SLD, a similar trend as the ob/ob control mice was observed (Table V and Fig. 1).
Table V.
Adipocytes in ob/ob mice orally administered SLD or CLA.
| Group | Epididymal adipocytes diameter (µm) | Abdominal adipocyte diameter (µm) | Abdominal fat pad thickness (mm; mean fat pads from muscle) |
|---|---|---|---|
| Controls | |||
| Intact | 88.59±13.03 | 46.27±8.53 | 1.25±0.28 |
| ob/ob | 322.31±27.88a | 215.89±14.24a | 6.03±0.45a |
| CLA | 218.25±37.98a,b | 168.30±23.87a,b | 4.73±0.59a,b |
| SLD (mg/kg) | |||
| 50 | 298.87±18.55a | 217.65±20.14a | 6.13±0.16a |
| 150 | 215.34±37.79a,b | 145.27±15.34a,b | 4.77±0.49a,b |
| 300 | 173.65±33.84a,b | 101.73±23.92a,b | 3.71±0.41a,b |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test).
P<0.01 vs. ob/ob control (LSD test). SLD, Slendesta™; CLA, conjugated linoleic acid.
Figure 1.
Histological profiles of the epididymal fat pad and abdominal fat pad (magnification, ×100 in the left panels and ×400 in the right panels). Note that significant increases of epididymal and abdominal adipocytes were detected in the ob/ob control compared to wild-type control. However, diameters of both adipocyte types were significantly (P<0.01) decreased in treated with CLA and SLD (150 and 300 mg/kg) compared to ob/ob control. (A, G and H) Wild-type and (B, I and J) ob/ob control, (C, K and L) CLA, (D, M and N) 50, (E, O and P) 150 and (F, Q and R) 300 mg/kg SLD-treated ob/ob mice (stain, hematoxylin and eosin; scale bars, 80 µm). Arrows indicates the adipocyte diameter measured. SLD, Slendesta™; CLA, conjugated linoleic acid (750 mg/kg) treated group.
Exocrine pancreas zymogen granule contents
Significant (P<0.01) decreases in the exocrine pancreas zymogen granule contents (the percentages of exocrine pancreas occupied by zymogen granules) were detected in ob/ob control mice compared to the wild-type control mice. However, the exocrine pancreas zymogen granule contents were significantly (P<0.01 or P<0.05) increased in mice treated with CLA and SLD compared to the ob/ob control, except for mice treated with 50 mg/kg SLD (Table VI).
Table VI.
Histomorphology of pancreas in ob/ob mice orally administered SLD or CLA.
| Group | Zymogen granules (%/mm2) | Pancreatic islets (n/10 mm2) | Islet occupied regions (%/10 mm2) |
|---|---|---|---|
| Controls | |||
| Intact | 66.63±8.47 | 7.60±1.82 | 3.58±1.46 |
| ob/ob | 31.90±5.85a | 27.20±1.92a | 24.34±4.55a |
| CLA | 42.40±2.53a,b | 19.60±2.07a,b | 17.79±2.10a,b |
| SLD (mg/kg) | |||
| 50 | 32.56±5.94a | 26.40±3.36a | 23.00±2.65a |
| 150 | 42.19±4.46a,b | 22.40±1.67a,b | 18.09±2.07a,b |
| 300 | 59.67±3.90b | 19.20±1.92b | 13.31±1.45b |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test).
P<0.01 vs. ob/ob control (LSD test). SLD, Slendesta™; CLA, conjugated linoleic acid.
Pancreatic islet hyperplasia and expansions
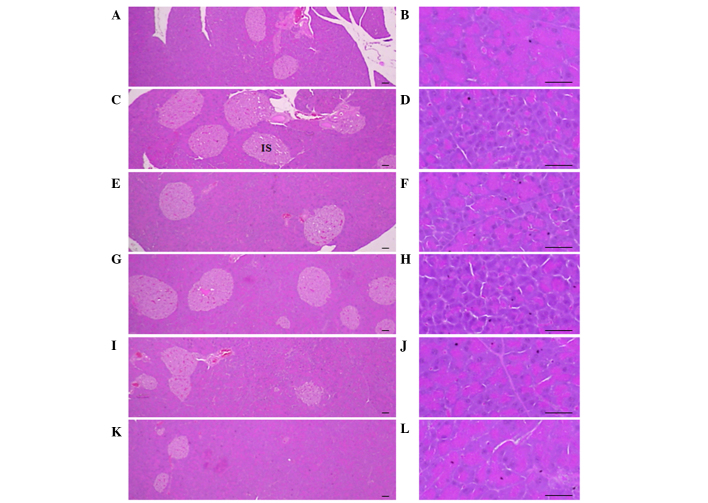
Significant (P<0.01) increases of pancreatic islet numbers and percentages of islet occupied regions were detected in the ob/ob control compared to the wild-type control, resulting from marked hyperplasia of the pancreatic islet itself or component endocrine cells. However, these events were significantly (P<0.01) reduced by treatment with CLA and SLD (150 and 300 mg/kg), compared to the ob/ob control. Mice treated with 50 mg/kg SLD displayed similar pancreatic islet numbers and percentages of islet occupied regions compared with ob/ob control (Table VI and Fig. 2).
Figure 2.
Histological profiles of the pancreas (magnification, ×100 in the left panels and ×400 in the right panels). Marked decreases in exocrine pancreas zymogen granule contents (the percentages of exocrine pancreas occupied by zymogen granules) were detected in the ob/ob control compared to wild-type control. However, the exocrine pancreas zymogen granule contents were increased in mice treated with CLA (750 mg/kg) and SLD (150 or 300 mg/kg), compared to the ob/ob control. In addition, increases in pancreatic islet numbers and percentages of islet occupied regions were detected in the ob/ob control compared to wild-type control. However, these events were reduced by treatment with CLA (750 mg/kg) and SLD (150 and 300 mg/kg) compared to the ob/ob control. (A and B) Wild-type mice, (C and D) ob/ob control mice, (E and F) CLA-treated ob/ob mice and (G and H) 50, (I and J) 150 and (K and L) 300 mg/kg SLD-treated ob/ob mice (stain, hematoxylin and eosin; scale bars, 80 µm). SLD, Slendesta™; CLA, conjugated linoleic acid; IS, pancreatic islets.
Pancreatic islet insulin-IR and glucagon-IR cells
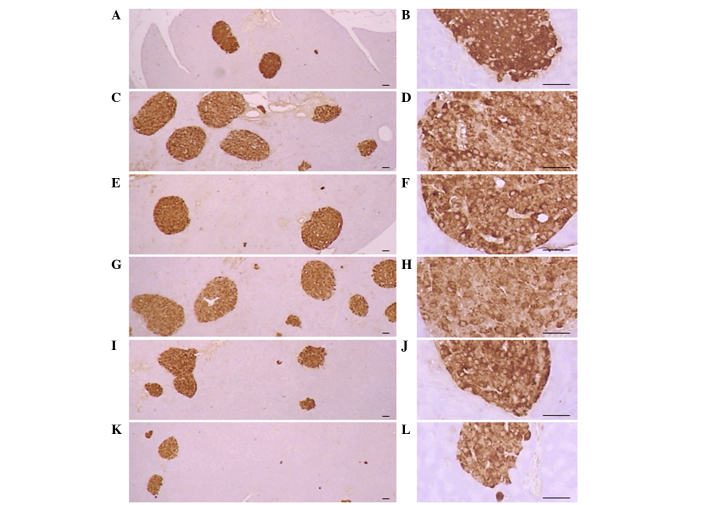
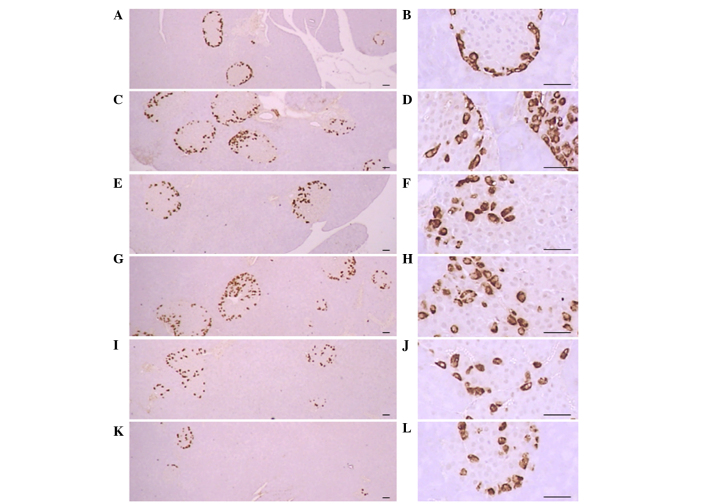
Significant (P<0.01) increases of insulin-IR and glucagon-IR cells were detected in the ob/ob control compared to the wild-type control, resulting from marked hyperplasia of endocrine cells. However, these increases were significantly (P<0.01) normalized by treatment of CLA and SLD compared to the ob/ob control mice, except for mice treated with 50 mg/kg SLD. No meaningful changes in insulin/glucagon-IR cells were demonstrated in all ob/ob mice compared to the wild-type control due to the simultaneous increases of the both insulin-IR and glucagon-IR cells, regardless of treatment (Table VII and Figs. 3 and 4).
Table VII.
Insulin- and glucagon-producing cells in ob/ob mice orally administered SLD or CLA.
| Group | Insulin-producing cells (cells/mm2) | Glucagon-producing cells (cells/mm2) | Insulin/glucagon-producing cell ratio |
|---|---|---|---|
| Controls | |||
| Intact | 610.40±95.00 | 247.40±59.33 | 2.51±0.30 |
| ob/ob | 1,931.60±253.53a | 826.00±111.88a | 2.35±0.29 |
| CLA | 1,242.40±133.11a,b | 522.80±65.76a,b | 2.39±0.29 |
| SLD (mg/kg) | |||
| 50 | 2,046.00±304.41a | 829.40±133.74a | 2.49±0.29 |
| 150 | 1,533.00±169.27a,b | 628.80±84.59a,c | 2.45±0.24 |
| 300 | 975.80±85.90b | 414.60±34.05a,b | 2.36±0.20 |
Values are expressed as the mean ± standard deviation (n=5).
P<0.01 vs. intact control (LSD test).
P<0.01
P<0.01 vs. ob/ob control (LSD test). SLD, Slendesta™; CLA, conjugated linoleic acid.
Figure 3.
Histological profiles of the insulin-producing cells in the pancreas (magnification, ×100 in the left panels and ×400 in the right panels). Increases in the number of insulin-IR cells were detected in the ob/ob control mice compared to wild-type mice. These increases were attenuated by treatment with CLA (750 mg/kg) and SLD (150 and 300 mg/kg) compared to the ob/ob control. (A and B) Wild-type mice, (C and D) ob/ob control mice, (E and F) CLA-treated ob/ob mice and (G and H) 50, (I and J) 150 and (K and L) 300 mg/kg SLD-treated ob/ob mice (stain, ABC; scale bars, 80 µm). SLD, Slendesta™; CLA, conjugated linoleic acid; IR, immunoreactive cells.
Figure 4.
Histological profiles of the glucagon-producing cells in the pancreas (magnification, ×100 in the left panels and ×400 in the right panels). Increases in the numbers of glucagon-IR cells were detected in the ob/ob control mice compared to wild-type mice. These increases were attenuated following treatment with CLA (750 mg/kg) and SLD (150 and 300 mg/kg) compared to the ob/ob control. (A and B) Wild-type mice, (C and D) ob/ob control mice, (E and F) CLA-treated ob/ob mice and (G and H) 50, (I and J) 150 and (K and L) 300 mg/kg SLD-treated ob/ob mice (stain, ABC; scale bars, 80 µm). SLD, Slendesta™; CLA, conjugated linoleic acid; IR, immunoreactive cells.
Discussion
Obesity has been a major public health concern for decades, particularly in developed countries. Obesity is an expensive burden on society, increasing medical care costs and affecting the available labor pool (33). At the individual level, obesity is a growing health issue; obesity-related health problems include hyperlipidemia, diabetes mellitus and cardiovascular diseases (34). Efforts to reduce obesity have included research that has increased understanding of the association between increased adiposity and insulin resistance (2,7). Metabolic syndrome refers to the simultaneous combination of obesity and related diseases, including diabetes, hypertension, hypercholesterolemia and hypertriglyceridemia (35). Therefore, a complex disease animal model is required in order to discover or develop novel drugs and/or alternative/complementary therapies against metabolic syndrome.
In the present study, to understand the anti-obese and anti-diabetic effects of SLD in genetically induced obese-ob/ob mice, SLD was orally administered once a day for 28 days. The effects were compared to untreated ob/ob and normoglycemic wild-type mice. CLA was used as a reference drug, which affects obesity and diabetes.
Increased mean daily food consumption was detected in the ob/ob mice at each time point compared with the wild-type mice. CLA presently decreased the daily food consumption when compared to the ob/ob mice, and showed anti-diabetic and anti-obesity effects, which is consistent with a previous study (18). Similar results were observed with SLD at 150 and 300 mg/kg dose, and lead by the end of the study to a normalization of food consumption to a level similar to that observed in wild-type mice. It is possible that SLD operates by a similar mechanism of action as CLA. SLD is a standardized potato extract containing 5% PPI II, which elicits a satiety response (19) and delays gastric emptying in humans (20). This is peculated to occur via the increased release of CCK, one of the most extensively studied peptides involved in the regulation of food intake (25). However, the possibility that other mechanisms may be involved cannot be entirely excluded, as SLD also influences serum adiponectin levels, while CLA does not affect serum adipokine. Further investigations are therefore warranted.
In addition, an increased accumulation of adipose tissues is a common feature in obesity, and adipocyte hypertrophy has been observed in a previous histological examination (36). Adipose tissues are considered to be an organ for energy storage, and also an endocrine and secretory organ (37). Adipose tissues secrete adipokines and changes in the expression, secretion and activity of these adipokines as a result of obesity may be implicated in the development of various diseases including insulin resistance (2,37). Among these, leptin was initially investigated as a satiety signal regulating food intake and energy expenditure and was found to effectively reduce appetite and feed intake (38). Therefore, deficiencies in leptin signaling or functioning in the hypothalamus are speculated to contribute to the development of obesity (38). Adiponectin (also known as Acrp30) is a novel adipokine that has been recently identified (39). It is exclusively expressed in adipose tissue (40) and is abundantly released into the circulating blood (41). More recently, obesity-related decreases in plasma adiponectin levels have been reported in humans (42) and experimental animals (43). Furthermore, it has been shown that hypoadiponectinemia is closely related to insulin resistance (2). In the present study, 150 and 300 mg/kg SLD directly inhibited the deposition of adipose tissues and the hypertrophy of adipocytes, while improving serum adiponectin abnormal changes in ob/ob mice, similar to CLA. These results are indicate that the favorable effects on diabetes-related obesity may be induced by SLD concentrations as low as 150 mg/kg. However, neither CLA nor SLD had an influence on the serum leptin levels in ob/ob mice, which are deficient in leptin.
Obesity leads to the development of pancreatic steatosis, acinar cell atrophy and a diminution in the number of zymogen granules (44,45). The increase of zymogen granules in exocrine pancreatic acinar cells indicates the production of digestive enzymes, particularly for lipid and protein digestion (46). In the present study, the decline in pancreatic zymogen granules was histopathologically detected in ob/ob control mice compared to wild-type control mice, which indicated a large release of pancreatic juice to remove the lipids. Consequently, the contents of pancreatic zymogen granules were markedly decreased. However, this decrease of zymogen granules in the exocrine pancreas was markedly inhibited by treatment with 150 and 300 mg/kg SLD, which was similar to the effects of CLA. Therefore, the anti-obese effects of SLD and CLA appear to mediate the inhibition of lipid digestion by decreasing the pancreatic enzyme production. PPI II is active in eliciting a satiety response (19) and delayed gastric emptying in humans (41), elevating the circulation of cholecystokinin (CCK) (25). We propose that the anti-obesity effects of SLD may include the inhibition of lipid and protein digestion by decreasing the release of pancreatic enzymes. However, further investigation is warranted to elucidate a mechanism underlying this phenomenon.
Hyperglycemia is the primary symptom of diabetes, and it must be controlled to effectively treat diabetes (47). All ob/ob mice used in the present study also showed hyperglycemia. Hyperglycemia was markedly and significantly decreased by treatment with 150 and 300 mg/kg SLD, but not 50 mg/kg SLD, providing direct evidence that >50 mg/kg SLD is necessary for a potent anti-diabetic effect. Increased insulin secretion is in part related to pancreatic islet hyperplasia in the progression of insulin-resistance in type-2 diabetes (48). Pancreatic islet insulin-producing cells are increased in area and in number, leading to increased insulin secretion to maintain glucose homeostasis. Glucagon-producing cells also increase in similar proportions to the increase in insulin cells (48). In the present study, a marked increase in islet-occupied regions and insulin- and glucagon-producing cells were observed in ob/ob control mice with normal insulin/glucagon cell ratios. However, treatment with CLA and SLD (150 and 300 mg/kg) brought the endocrine pancreas secretion of insulin in the ob/ob mice to normal levels. These results suggest that SLD exerts a protective effect against insulin resistance. Progression of chronic diabetes in ob/ob mice is associated with the development of hyperlipemia (18). The critical problem in hyperlipemia is elevated levels of serum FFA, triglyceride and TC (49). Similar favorable hypolipidemic, anti-obesity and anti-diabetic effects were observed in mice treated with 150 mg/kg SLD, compared with 750 mg/kg CLA in the present study.
The presents results suggest that 28 days of continuous oral treatment of SLD effectively improved or normalized the parameters associated with obesity and diabetes and their related complications in ob/ob mice. Thus, SLD may be a promising alternative therapeutic agent for the treatment of metabolic syndrome, mediated by an already known mechanism, through the food intake regulatory peptide CCK. However, further investigations are required to identify the specific mechanism by which the observed effects of SLD are mediated.
Acknowledgements
This study was supported by the National Research Foundation of Korea grant funded by the Korea government (no. 2011-0030124). The authors would like to thank Ms. Samanta Maci (Kemin) for providing assistance in reviewing the manuscript.
Glossary
Abbreviations
- SLD
Slendesta
- CLA
conjugated linoleic acid
- ob/ob
C57BLKS/J-ob/ob
- TC
total cholesterol
- FFA
free fatty acid
- PPI
potato proteinase inhibitor
- CCK
cholecystokinin
- H&E
hematoxylin and eosin
- IR
immunoreactive
- LSD
least-significant differences
- MW test
Mann-Whitney U-Wilcoxon Rank Sum W test
References
- 1.Yun JW. Possible anti-obesity therapeutics from nature-a review. Phytochemistry. 2010;71:1625–1641. doi: 10.1016/j.phytochem.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 2.Mitchell M, Armstrong DT, Robker RL, Norman RJ. Adipokines: Implications for female fertility and obesity. Reproduction. 2005;130:583–597. doi: 10.1530/rep.1.00521. [DOI] [PubMed] [Google Scholar]
- 3.Kunitomi M, Wada J, Takahashi K, Tsuchiyama Y, Mimura Y, Hida K, Miyatake N, Fujii M, Kira S, Shikata K, Maknio H. Relationship between reduced serum IGF-I levels and accumulation of visceral fat in Japanese men. Int J Obes Relat Metab Disord. 2002;26:361–369. doi: 10.1038/sj.ijo.0801899. [DOI] [PubMed] [Google Scholar]
- 4.Hida K, Wada J, Eguchi J, Zhang H, Baba M, Seida A, Hashimoto I, Okada T, Yasuhara A, Nakatsuka A, et al. Visceral adipose tissue-derived serine protease inhibitor: A unique insulin-sensitizing adipocytokine in obesity. Proc Natl Acad Sci USA. 2005;102:10610–10615. doi: 10.1073/pnas.0504703102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wolf G, Chen S, Han DC, Ziyadeh FN. Leptin and renal disease. Am J Kidney Dis. 2002;39:1–11. doi: 10.1053/ajkd.2002.29865. [DOI] [PubMed] [Google Scholar]
- 6.Yamauchi T, Kamon J, Waki H, Imai Y, Shimozawa N, Hioki K, Uchida S, Ito Y, Takakuwa K, Matsui J, et al. Globular adiponectin protected ob/ob mice from diabetes and ApoE-deficient mice from atherosclerosis. J Biol Chem. 2003;278:2461–2468. doi: 10.1074/jbc.M209033200. [DOI] [PubMed] [Google Scholar]
- 7.Sakaue H, Nishizawa A, Ogawa W, Teshigawara K, Mori T, Takashima Y, Noda T, Kasuga M. Requirement for 3-phosphoinositide-kependent dinase-1 (PDK-1) in insulin-induced glucose uptake in immortalized brown adipocytes. J Biol Chem. 2003;278:38870–38874. doi: 10.1074/jbc.M306151200. [DOI] [PubMed] [Google Scholar]
- 8.Seufert J, Lübben G, Dietrich K, Bates PC. A comparison of the effects of thiazolidinediones and metformin on metabolic control in patients with type 2 diabetes mellitus. Clin Ther. 2004;26:805–818. doi: 10.1016/S0149-2918(04)90125-7. [DOI] [PubMed] [Google Scholar]
- 9.Inzucchi SE. Oral antihyperglycemic therapy for type 2 diabetes: Scientific review. JAMA. 2002;287:360–372. doi: 10.1001/jama.287.3.360. [DOI] [PubMed] [Google Scholar]
- 10.Sicińska P, Pytel E, Maćczak A, Koter-Michalak M. The use of various diet supplements in metabolic syndrome. Postepy Hig Med Dosw (Online) 2015;69:25–33. doi: 10.5604/17322693.1135416. (In Polish) [DOI] [PubMed] [Google Scholar]
- 11.Parodi PW. Cows' milk fat components as potential anticarcinogenic agents. J Nutr. 1997;127:1055–1060. doi: 10.1093/jn/127.6.1055. [DOI] [PubMed] [Google Scholar]
- 12.Lee KN, Kritchevsky D, Pariza MW. Conjugated linoleic acid and atherosclerosis in rabbits. Atherosclerosis. 1994;108:19–25. doi: 10.1016/0021-9150(94)90034-5. [DOI] [PubMed] [Google Scholar]
- 13.Parker J, Daniel LW, Waite M. Evidence of protein kinase C involvement in phorbol diester-stimulated arachidonic acid release and prostaglandin synthesis. J Biol Chem. 1987;262:5385–5393. [PubMed] [Google Scholar]
- 14.Banni S, Carta G, Angioni E, Murru E, Scanu P, Melis MP, Bauman DE, Fischer SM, Ip C. Distribution of conjugated linoleic acid and metabolites in different lipid fractions in the rat liver. J Lipid Res. 2001;42:1056–1061. [PubMed] [Google Scholar]
- 15.Pariza MW, Park Y, Cook M. The biologically active isomers of conjugated linoleic acid. Prog Lipid Res. 2001;40:283–298. doi: 10.1016/S0163-7827(01)00008-X. [DOI] [PubMed] [Google Scholar]
- 16.Bocher V, Pineda-Torra I, Fruchart JC, Staels B. PPARs: Transcription factors controlling lipid and lipoprotein metabolism. Ann N Y Acad Sci. 2002;967:7–18. doi: 10.1111/j.1749-6632.2002.tb04258.x. [DOI] [PubMed] [Google Scholar]
- 17.Granlund L, Pedersen JI, Nebb HI. Impaired lipid accumulation by trans10, cis12 CLA during adipocyte differentiation is dependent on timing and length of treatment. Biochim Biophys Acta. 2005;1687:11–22. doi: 10.1016/j.bbalip.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 18.Hue JJ, Lee KN, Jeong JH, Lee SH, Lee YH, Jeong SW, Nam SY, Yun YW, Lee BJ. Anti-obesity activity of diglyceride containing conjugated linoleic acid in C57BL/6J ob/ob mice. J Vet Sci. 2009;10:189–195. doi: 10.4142/jvs.2009.10.3.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hill AJ, Peikin SR, Ryan CA, Blundell JE. Oral administration of proteinase inhibitor II from potatoes reduces energy intake in man. Physiol Behav. 1990;48:241–246. doi: 10.1016/0031-9384(90)90307-P. [DOI] [PubMed] [Google Scholar]
- 20.Schwartz JG, Guan D, Green GM, Phillips WT. Treatment with an oral proteinase inhibitor slows gastric emptying and acutely reduces glucose and insulin levels after a liquid meal in type II diabetic patients. Diabetes Care. 1994;17:255–262. doi: 10.2337/diacare.17.4.255. [DOI] [PubMed] [Google Scholar]
- 21.Levitt J. The isolation and preliminary fractionation of proteins from dormant and growing potato tubers. Plant Physiol. 1951;26:59–65. doi: 10.1104/pp.26.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bryant J, Green TR, Gurusaddaiah T, Ryan CA. Proteinase inhibitor II from potatoes: Isolation and characterization of its protomer components. Biochemistry. 1976;15:3418–3424. doi: 10.1021/bi00661a004. [DOI] [PubMed] [Google Scholar]
- 23.Kim MH, Park SC, Kim JY, Lee SY, Lim HT, Cheong H, Hahm KS, Park Y. Purification and characterization of a heat-stable serine protease inhibitor from the tubers of new potato variety ‘Golden Valley’. Biochem Biophys Res Commun. 2006;346:681–686. doi: 10.1016/j.bbrc.2006.05.186. [DOI] [PubMed] [Google Scholar]
- 24.Vlachojannis JE, Cameron M, Chrubasik S. Medicinal use of potato-derived products: A systematic review. Phytother Res. 2010;24:159–162. doi: 10.1002/ptr.2829. [DOI] [PubMed] [Google Scholar]
- 25.Komarnytsky S, Cook A, Raskin I. Potato protease inhibitors inhibit food intake and increase circulating cholecystokinin levels by a trypsin-dependent mechanism. Int J Obes (Lond) 2011;35:236–243. doi: 10.1038/ijo.2010.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Peters HP, Foltz M, Kovacs EM, Mela DJ, Schuring EA, Wiseman SA. The effect of protease inhibitors derived from potato formulated in a minidrink on appetite, food intake and plasma cholecystokinin levels in humans. Int J Obes (Lond) 2011;35:244–250. doi: 10.1038/ijo.2010.136. [DOI] [PubMed] [Google Scholar]
- 27.Kim JD, Kang SM, Park MY, Jung TY, Choi HY, Ku SK. Ameliorative anti-diabetic activity of dangnyosoko, a Chinese herbal medicine, in diabetic rats. Biosci Biotechnol Biochem. 2007;71:1527–1534. doi: 10.1271/bbb.70058. [DOI] [PubMed] [Google Scholar]
- 28.Sahai A, Malladi P, Pan X, Paul R, Melin-Aldana H, Green RM, Whitington PF. Obese and diabetic db/db mice develop marked liver fibrosis in a model of nonalcoholic steatohepatitis: Role of short-form leptin receptors and osteopontin. Am J Physiol Gastrointest Liver Physiol. 2004;287:G1035–G1043. doi: 10.1152/ajpgi.00199.2004. [DOI] [PubMed] [Google Scholar]
- 29.Jung YM, Lee SH, Lee DS, You MJ, Chung IK, Cheon WH, Kwon YS, Lee YJ, Ku SK. Fermented garlic protects diabetic, obese mice when fed a high-fat diet by antioxidant effects. Nutr Res. 2011;31:387–396. doi: 10.1016/j.nutres.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 30.Kim JD, Kang SM, Seo BI, Choi HY, Choi HS, Ku SK. Anti-diabetic activity of SMK001, a poly herbal formula in streptozotocin induced diabetic rats: Therapeutic study. Biol Pharm Bull. 2006;29:477–482. doi: 10.1248/bpb.29.477. [DOI] [PubMed] [Google Scholar]
- 31.Edwin N, Leigh CM. Immunocytochemical identification of islet cells containing calcitonin gene-related peptide-like immunoreactivity in the plains rat pancreas (Pseudomys australis) Singapore Med J. 1999;40:528–530. [PubMed] [Google Scholar]
- 32.Lee HS, Chang JH, Ku SK. An immunohistochemical study of the pancreatic endocrine cells of the ddN mouse. Folia Histochem Cytobiol. 2010;48:387–393. doi: 10.2478/v10042-010-0026-y. [DOI] [PubMed] [Google Scholar]
- 33.Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC, Biryukov S, Abbafati C, Abera SF, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–781. doi: 10.1016/S0140-6736(14)60460-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liese AD, Mayer-Davis EJ, Haffner SM. Development of the multiple metabolic syndrome: An epidemiologic perspective. Epidemiol Rev. 1998;20:157–172. doi: 10.1093/oxfordjournals.epirev.a017978. [DOI] [PubMed] [Google Scholar]
- 35.Deen D. Metabolic syndrome: Time for action. Am Fam Physician. 2004;69:2875–2882. [PubMed] [Google Scholar]
- 36.Morange PE, Lijnen HR, Alessi MC, Kopp F, Collen D, Juhan-Vague I. Influence of PAI-1 on adipose tissue growth and metabolic parameters in a murine model of diet-induced obesity. Arterioscler ThrombVasc Biol. 2000;20:1150–1154. doi: 10.1161/01.ATV.20.4.1150. [DOI] [PubMed] [Google Scholar]
- 37.Fujita H, Fujishima H, Koshimura J, Hosoba M, Yoshioka N, Shimotomai T, Morii T, Narita T, Kakei M, Ito S. Effects of antidiabetic treatment with metformin and insulin on serum and adipose tissue adiponectin levels in db/db mice. Endocr J. 2005;52:427–433. doi: 10.1507/endocrj.52.427. [DOI] [PubMed] [Google Scholar]
- 38.Bjørbaek C, Kahn BB. Leptin signaling in the central nervous system and the periphery. Recent Prog Horm Res. 2004;59:305–331. doi: 10.1210/rp.59.1.305. [DOI] [PubMed] [Google Scholar]
- 39.Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF. A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem. 1995;270:26746–26749. doi: 10.1074/jbc.270.45.26746. [DOI] [PubMed] [Google Scholar]
- 40.Maeda K, Okubo K, Shimomura I, Funahashi T, Matsuzawa Y, Matsubara K. CDNA cloning and expression of a novel adipose specific collagen-like factor, apM1 (AdiPose Most abundant Gene transcript 1) Biochem Biophys Res Commun. 1996;221:286–289. doi: 10.1006/bbrc.1996.0587. [DOI] [PubMed] [Google Scholar]
- 41.Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J, Hotta K, Shimomura I, Nakamura T, Miyaoka K, et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun. 1999;257:79–83. doi: 10.1006/bbrc.1999.0255. [DOI] [PubMed] [Google Scholar]
- 42.Matsubara M. Plasma adiponectin decrease in women with nonalcoholic Fatty liver. Endocr J. 2004;51:587–593. doi: 10.1507/endocrj.51.587. [DOI] [PubMed] [Google Scholar]
- 43.Maebuchi M, Machidori M, Urade R, Ogawa T, Moriyama T. Low resistin levels in adipose tissues and serum in high-fat fed mice and genetically obese mice: Development of an ELISA system for quantification of resistin. Arch Biochem Biophys. 2003;416:164–170. doi: 10.1016/S0003-9861(03)00279-0. [DOI] [PubMed] [Google Scholar]
- 44.Tasso F, Clop J, Sarles H. The interaction of ethanol, dietary lipids and proteins on the rat pancreas. II. Ultrastructural study. Digestion. 1971;4:23–34. doi: 10.1159/000197093. [DOI] [PubMed] [Google Scholar]
- 45.Wilson JS, Korsten MA, Leo MA, Lieber CS. Combined effects of protein deficiency and chronic ethanol consumption on rat pancreas. Dig Dis Sci. 1988;33:1250–1259. doi: 10.1007/BF01536675. [DOI] [PubMed] [Google Scholar]
- 46.Hiatt JL, Gartner LP. Color textbook of histology. WB Saunders Co.; Philadelphia USA: 2007. pp. 1–592. [Google Scholar]
- 47.Sathishsekar D, Subramanian S. Beneficial effects of Momordica charantia seeds in the treatment of STZ-induced diabetes in experimental rats. Biol Pharm Bull. 2005;28:978–983. doi: 10.1248/bpb.28.978. [DOI] [PubMed] [Google Scholar]
- 48.Terauchi Y, Takamoto I, Kubota N, Matsui J, Suzuki R, Komeda K, Hara A, Toyoda Y, Miwa I, Aizawa S, et al. Glucokinase and IRS-2 are required for compensatory beta cell hyperplasia in response to high-fat diet-induced insulin resistance. J Clin Invest. 2007;117:246–257. doi: 10.1172/JCI17645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kamada T, Hata J, Kusunoki H, Ito M, Tanaka S, Kawamura Y, Chayama K, Haruma K. Eradication of Helicobacter pylori increases the incidence of hyperlipidaemia and obesity in peptic ulcer patients. Dig Liver Dis. 2005;37:39–43. doi: 10.1016/j.dld.2004.07.017. [DOI] [PubMed] [Google Scholar]