ABSTRACT
During development of the peripheral nervous system, excess neurons are generated, most of which will be lost by programmed cell death due to a limited supply of neurotrophic factors from their targets. Other environmental factors, such as ‘competition factors' produced by neurons themselves, and axon guidance molecules have also been implicated in developmental cell death. Semaphorin 3A (Sema3A), in addition to its function as a chemorepulsive guidance cue, can also induce death of sensory neurons in vitro. The extent to which Sema3A regulates developmental cell death in vivo, however, is debated. We show that in compartmentalized cultures of rat sympathetic neurons, a Sema3A-initiated apoptosis signal is retrogradely transported from axon terminals to cell bodies to induce cell death. Sema3A-mediated apoptosis utilizes the extrinsic pathway and requires both neuropilin 1 and plexin A3. Sema3A is not retrogradely transported in older, survival factor-independent sympathetic neurons, and is much less effective at inducing apoptosis in these neurons. Importantly, deletion of either neuropilin 1 or plexin A3 significantly reduces developmental cell death in the superior cervical ganglia. Taken together, a Sema3A-initiated apoptotic signaling complex regulates the apoptosis of sympathetic neurons during the period of naturally occurring cell death.
KEY WORDS: Sympathetic neurons, Cell death, Sema3A, Rat, Mouse
Summary: A Sema3A-initiated apoptotic signaling complex regulates the apoptosis of sympathetic neurons during their period of naturally occurring cell death.
INTRODUCTION
In the developing vertebrate nervous system, neurons are generated in excess. Many of these neurons are non-essential or inappropriately connected and are eliminated by programmed cell death (Oppenheim, 1991). In peripheral neurons, apoptosis is in large part due to the limited supply of neurotrophic factors produced by their targets of innervation. Neurons that make appropriate connections and successfully compete for neurotrophic factors survive, whereas unsuccessful neurons are eliminated by developmental apoptosis (Levi-Montalcini, 1987; Oppenheim, 1991). Some neurons undergo apoptosis at early developmental stages while extending their axons and before reaching their final targets (Coggeshall et al., 1994; White and Behar, 2000). It is therefore conceivable that the environment contains cues that not only guide developing axons, but also actively induce neuronal apoptosis. Moreover, it has been speculated that apoptosis at early stages of development might be related to axon pathfinding errors, possibly as a mechanism to eliminate those neurons with axons that have wandered astray and innervate non-target tissues (Oppenheim, 1991).
It has recently become apparent that neuronal apoptosis can be induced in response to axon guidance molecules, including ephrin A5, semaphorin 4D, semaphorin 3A (Sema3A) and Drosophila NetA (Gagliardini and Fankhauser, 1999; Yue et al., 1999; Giraudon et al., 2004; Newquist et al., 2013). Sema3A, a secreted glycoprotein that belongs to the semaphorin family of axon guidance molecules, plays a crucial role in axonal pathfinding and neuronal patterning of numerous populations of peripheral nervous system (PNS) and central nervous system (CNS) neurons (Luo et al., 1993; Messersmith et al., 1995; Behar et al., 1996; Varela-Echavarria et al., 1997). In vitro, Sema3A induces apoptosis of CNS neurons, such as cerebellar granule neurons (CGNs) (Shirvan et al., 1999) and retinal ganglion cells (RGCs) (Shirvan et al., 2002), as well as PNS neurons such as paravertebral sympathetic ganglion neurons (Shirvan et al., 1999) and sensory dorsal root ganglion (DRG) neurons (Gagliardini and Fankhauser, 1999; Ben-Zvi et al., 2006). Neuropilin 1 (Npn1) supports high-affinity binding of Sema3A (He and Tessier-Lavigne, 1997; Kolodkin et al., 1997) and functions as a co-receptor in a complex with plexin A family members (Takahashi et al., 1999; Tamagnone et al., 1999; Yaron et al., 2005). The plexin A cytoplasmic domain is essential for Sema3A signaling (Takahashi et al., 1999; Tamagnone et al., 1999). Although Sema3A can induce apoptosis when applied to cultured neurons in vitro, to what extent Sema3A directly induces apoptosis in vivo via Npn1 and plexin A proteins is debated (Ben-Zvi et al., 2008; Haupt et al., 2010).
In the context of target-derived survival mediated by neurotrophic factors, nerve growth factor (NGF), together with its receptor tyrosine kinase TrkA (also known as Ntrk1), is internalized and retrogradely transported as a complex from the axon terminal to the cell body to support the survival of sympathetic and sensory neurons (Cosker et al., 2008). Using compartmentalized cultures of rat superior cervical ganglia (SCG) sympathetic neurons, we report here that Sema3A is retrogradely transported from distal axons to cell bodies to induce apoptosis. Importantly, this retrograde Sema3A death signal triggers the extrinsic apoptosis pathway, requires both Npn1 and plexin A3, and contributes significantly to programmed cell death of SCG neurons in vitro and in vivo.
RESULTS
Sema3A is retrogradely transported from the distal axons to the cell bodies of sympathetic neurons
In the developing PNS, Sema3A plays a crucial role as a chemorepulsive axon guidance molecule for postganglionic sympathetic neurons and repels developing axons from non-target tissues (Nakamura et al., 2000; Kolodkin and Tessier-Lavigne, 2011). Upon receptor binding on the neuronal growth cone, the Sema3A ligand-receptor complex undergoes rapid endocytosis (Fournier et al., 2000). The fate of the internalized complex, however, has not yet been investigated. To determine whether Sema3A internalized at the axon terminal is retrogradely transported to the cell body, compartmentalized cultures of sympathetic neurons were used (Fig. 1A). Campenot chambers were prepared and dissociated SCG neurons were plated in the central compartment and left to grow their axons into fluidically isolated side compartments. To visualize Sema3A, a fluorescent tandem dimerized tomato (tdT)-Sema3A fusion protein, as well as tdT alone, were generated (Fig. S1A-C). The bioactivity of tdT-Sema3A was tested by examining its binding to Npn1 (Fig. S1D) and axon repulsion of NGF-responsive DRG neurons (Fig. S1E,F). Unlike tdT alone, tdT-Sema3A was able to bind to Npn1 (Fig. S1D) and induced axon repulsion of DRG neurons (Fig. S1E,F).
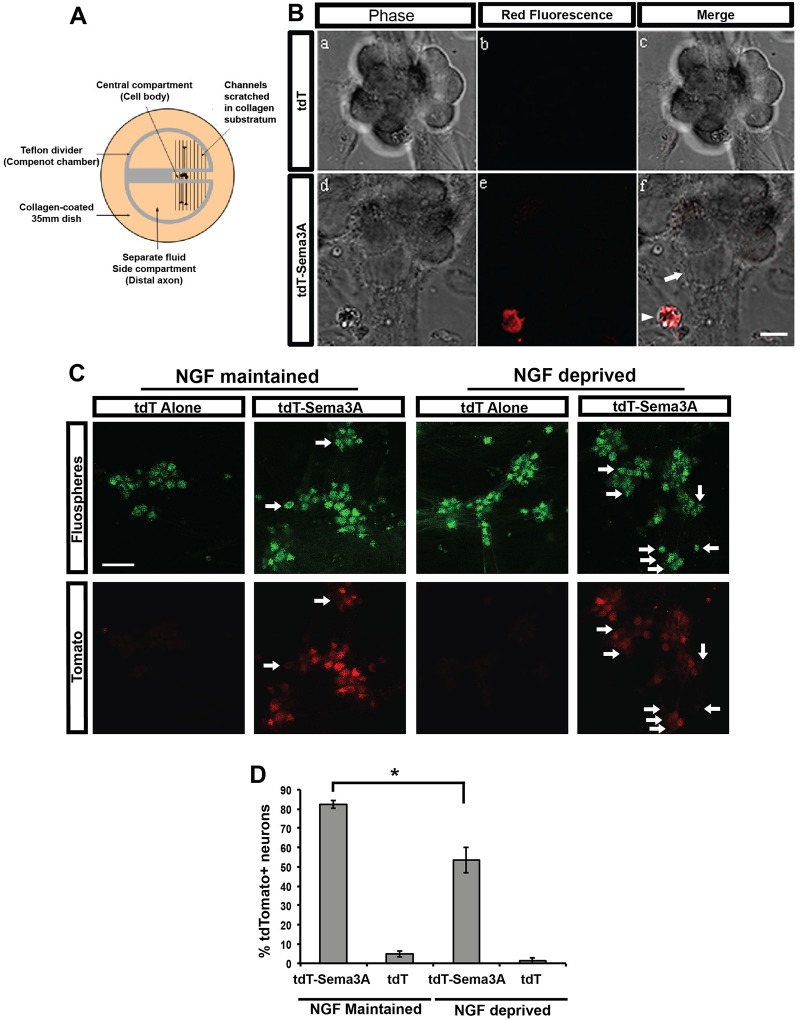
Fig. 1.
Sema3A is retrogradely transported from the distal axons to the cell bodies of sympathetic neurons. (A) Diagram of a compartmentalized culture containing neuronal cell bodies and growing distal axons in separate fluid compartments. (B) Soluble tdT or tdT-Sema3A were added to the SCG medium bathing the distal axons in compartmentalized cultures. Unlike tdT alone (a-c), tdT-Sema3A (d-f) was detected by direct fluorescence imaging in cell bodies after being retrogradely transported from distal axons to cell bodies (arrowhead in f). Arrow (f) shows a cell body from a neuron not retrogradely transporting tdT-Sema3A. (C) Compartmentalized cultures of P5 SCG neurons provided with tdT-Sema3A, or tdT alone, along with FluoSpheres indicates cell bodies from neurons projecting axons into the side compartment (green fluorescence, top panels) and retrogradely transporting tdT-Sema3A (red fluorescence, bottom panels). These transport studies were performed on neurons that were either maintained or deprived of NGF (as indicated). Arrows indicate neurons that did not retrogradely transport tdT-Sema3A. (D) Quantification of the data in C, with the bars representing tdT-Sema3A+ neurons/FluoSphere+ neurons. Results are presented as mean±s.e.m. from three independent experiments. *P<0.05, Student's t-test. Scale bars: 20 μm in B; 50 μm in C.
To determine whether Sema3A can be retrogradely transported, the distal axons of SCG neurons grown in compartmentalized cultures for 7-14 days, during which there is robust axon growth into the side compartments, were exposed to tdT-Sema3A, or tdT alone, for 24 h. Interestingly, some of the cell bodies in the central compartment displayed tdT fluorescence (Fig. 1B), but only in cultures in which tdT-Sema3A, and not tdT, was added to the axon compartments (Fig. 1B). The neurons that displayed accumulation of tdT-Sema3A appeared to be atrophic and were possibly undergoing apoptosis (Fig. 1B). Because the induction of apoptosis could potentially cause an underestimation of the number of neurons that were retrogradely transporting tdT-Sema3A, these experiments were repeated in neurons maintained in a pan-caspase inhibitor (BAF) to eliminate cell death. Immunofluorescence labeling of tdT with tomato antibodies was also performed on these cultures to better detect the retrogradely transported tdT-Sema3A. Interestingly, the majority of NGF-maintained SCG neurons appeared to retrogradely transport tdT-Sema3A under these conditions (Fig. 1C). To quantify the percentage of SCG neurons capable of retrogradely transporting tdT-Sema3A from distal axons to the cell body, we determined the percentage of neurons that projected into the side compartments where the tdT-Sema3A was applied. This was accomplished by applying fluorescently labeled carboxylate beads that are internalized and retrogradely transported (FluoSpheres, 515 nm emission) to the distal axons along with tdT-Sema3A, or tdT alone. When compared with the total number of neurons that were FluoSphere labeled, and thus projecting into the side compartments (Fig. 1C), the cell bodies of 82.2±2.0% of these NGF-maintained neurons were tdT-Sema3A+. This was a significant increase compared with neurons treated with tdT alone (P<0.01), in which only 4.7±1.5% of cell bodies were tdT+ (Fig. 1D). Thus, most SCG neurons (82.2%) were able to internalize and retrogradely transport Sema3A from distal axons to the cell body in compartmentalized cultures.
To determine whether the retrograde transport of Sema3A was affected by the trophic status of the neurons, Sema3A retrograde transport studies were performed on neurons that were deprived of NGF at the same time that tdT-Sema3A was applied to the distal axons. tdT-Sema3A was also retrogradely transported in NGF-deprived sympathetic neurons, but to a lesser extent (Fig. 1C,D). Quantification of the number of NGF-deprived neurons that retrogradely transported tdT-Sema3A revealed that 53.4±6.7% were tdT-Sema3A+, as compared with only 1.3±1.4% of NGF-deprived neurons that transported tdT alone (significantly different, P<0.01). When tdT-Sema3A transport was compared between NGF-maintained neurons (82.2%) and NGF-deprived neurons (53.4%), this difference was statistically significant (P<0.05), indicating that neurons that were atrophic and beginning to undergo apoptosis transported less Sema3A. This is likely to be due to a generalized reduction in axonal integrity and, therefore, retrograde transport in the NGF-deprived neurons.
While analyzing the morphology of tdT-Sema3A+ neurons in the central compartment in the absence of caspase inhibitors, we observed that their cell bodies were small and exhibited an atrophic, phase dark morphology characteristic of sympathetic neurons undergoing apoptosis (Fig. 1B, arrowhead). This suggested that Sema3A might function as a retrograde death signal. To determine whether Sema3A could induce cell death in sympathetic neurons, mass cultures of SCG neurons were exposed to tdT-Sema3A for 24 h. Interestingly, tdT-Sema3A+ SCG neurons had pyknotic nuclei (Fig. 2B-D), indicative of apoptosis. To examine whether apoptotic-appearing neurons expressed Npn1 on their cell surface, immunofluorescence labeling of Npn1 was performed without cell permeabilization. Surprisingly, Npn1 was observed on the surface of healthy, tdT-Sema3A− neurons (Fig. 2A) but not on dying neurons with cell bodies containing tdT-Sema3A (Fig. 2B). These data suggest that Npn1 might be internalized upon Sema3A binding in neurons, as has been reported previously (Fournier et al., 2000), and retrogradely transported to the cell body.
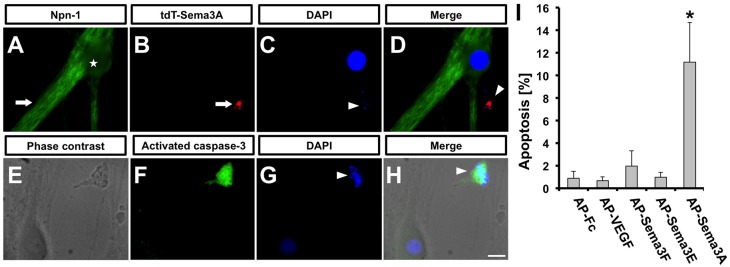
Fig. 2.
Sema3A induces apoptosis of primary SCG neurons. (A-D) Mass cultures of SCG neurons were treated with tdT-Sema3A for 24 h and then immunofluorescently labeled for Npn1 and with DAPI. tdT-Sema3A was found in cell bodies (arrow in B), where it colocalizes with pyknotic nuclei (arrowhead in C,D). Npn1 was expressed on the cell surface of both cell bodies (asterisk in A) and axons (arrow in A) of healthy neurons, but was not found in apoptotic cell bodies (arrowhead in D). (E-H) SCG neurons treated with AP-Sema3A for 24 h and then immunolabeled with antibodies to activated caspase 3. (F) Image of a cell body undergoing apoptosis that was positive for activated caspase 3 and containing a pyknotic nucleus (arrowhead in G,H). (I) Quantification of the percentage of activated caspase 3+ neurons after exposure to class 3 semaphorins. Error bars indicate s.e.m.; *P<0.05, Student's t-test. Scale bar: 20 μm.
Sema3A, but not Sema3E or Sema3F, mediates SCG cell death via caspase 3 activation
To determine whether Sema3A induces cell death of SCG neurons via apoptosis, caspase activation was examined in mass cultures of SCG neurons treated with alkaline phosphatase-tagged Sema3A (AP-Sema3A), or AP-Fc alone (Giger et al., 2000). Apoptosis was measured by counting activated caspase 3+ neurons 24 h after Sema3A exposure. 11.1±3.5% of SCG neurons were positive for activated caspase 3 when exposed to AP-Sema3A (Fig. 2F,I) and had nuclear pyknotic bodies (Fig. 2G), as compared with the AP-Fc control that had only 0.8±0.6% apoptosis (Fig. 2I). An unrelated Npn1 ligand, VEGF165, did not induce apoptosis of SCG neurons (Fig. 2I). Other class 3 semaphorin family members, including Sema3E and Sema3F, failed to induce apoptosis comparable to Sema3A (Fig. 2I). The low level of apoptosis observed with AP-Fc (0.8±0.6%), AP-VEGF165 (0.6±0.3%), AP-Sema3F (1.9±1.3%) and AP-Sema3E (0.9±0.4%) was not significantly different from that of untreated controls. Taken together, Sema3A, but not Sema3E or Sema3F, induced apoptosis of sympathetic neurons via caspase 3 activation.
Retrograde Sema3A-induced cell death of sympathetic neurons requires microtubule-based axonal transport
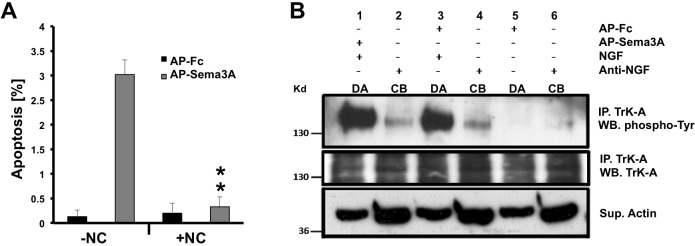
In compartmentalized cultures of sympathetic neurons, selective exposure of distal axons to Sema3A induced apoptosis of the cell bodies within 24 h (Fig. 1A). In fact, within 6 h of tdT-Sema3A application to distal axons, tdT-Sema3A+ cell bodies were observed to be undergoing cell death (Fig. 3A). We speculated that the retrograde transport of Sema3A may be necessary to induce neuronal apoptosis. To address this possibility, microtubules were pharmacologically depolymerized by exposing the distal axons in compartmentalized cultures to nocodazole (NC) along with AP-Sema3A, or AP-Fc as a negative control. Six hours after these treatments, activated caspase 3+ cell bodies were quantified. Sema3A-induced apoptosis was almost completely blocked in SCG neurons treated with NC, as compared with SCG neurons exposed to AP-Sema3A alone (3±0.2% without NC versus 0.3±0.2% with NC, P<0.01; Fig. 3A). These data suggest that microtubule-based transport is required for Sema3A-induced retrograde cell death.
Fig. 3.
Retrograde Sema3A-induced cell death requires microtubule-based axonal transport in sympathetic neurons. (A) In compartmentalized cultures of sympathetic neurons, distal axons were exposed to either AP-Sema3A or AP-Fc, with or without nocodazole (NC). Six hours after stimulation, immunofluorescent labeling of activated caspase 3 was performed, followed by quantification of activated caspase 3+ cell bodies. The short 6 h treatment was used, even though this resulted in a lower percentage of Sema3A-mediated apoptosis, because NC alone inhibits retrograde transport of NGF-TrkA complexes, which will result in apoptosis of the neurons within 24 h. Mean±s.e.m. from three independent experiments. **P<0.01, Student's t-test. (B) In three separate SCG compartmentalized cultures (lanes 1 and 2, 3 and 4, 5 and 6), distal axons (DA) were treated with control AP-Fc (lanes 3 and 5) or AP-Sema3A (lane 1) with or without NGF. In some conditions the cell bodies (CB) were treated with anti-NGF (lanes 2, 4 and 6). After 24 h, the neurons were subjected to TrkA immunoprecipitation followed by western blotting with the indicated antibodies. Sema3A treatment of axons in the presence of NGF does not affect TrkA activation or its retrograde transport to cell bodies (lanes 1 and 2), when compared with neurons treated with AP-Fc and NGF (lanes 3 and 4). Supernatants from the immunoprecipitations were analyzed for actin as a loading control. Similar results were obtained from three independent experiments.
Sema3A induces apoptosis of sympathetic neurons via the extrinsic pathway
To determine the cellular mechanisms by which Sema3A induces apoptosis, we first examined whether Sema3A inhibits NGF-mediated retrograde survival. NGF, upon activation of its receptor tyrosine kinase TrkA, is internalized and retrogradely transported from the distal axons to the cell bodies as a stable NGF-TrkA complex to signal cell survival (Cosker et al., 2008). To determine whether TrkA activation, or retrograde transport of activated TrkA, is affected by Sema3A, we stimulated distal axons of compartmentalized cultures with Sema3A and NGF while the cell bodies were deprived of NGF. Under these conditions, autophosphorylated TrkA in the cell bodies is exclusively derived from the distal axons. Sema3A did not inhibit TrkA activation in the distal axons, nor did it alter the retrograde transport of phosphorylated TrkA (Fig. 3B). Thus, Sema3A did not appear to significantly reduce retrograde NGF-TrkA signaling in sympathetic neurons to induce cell death.
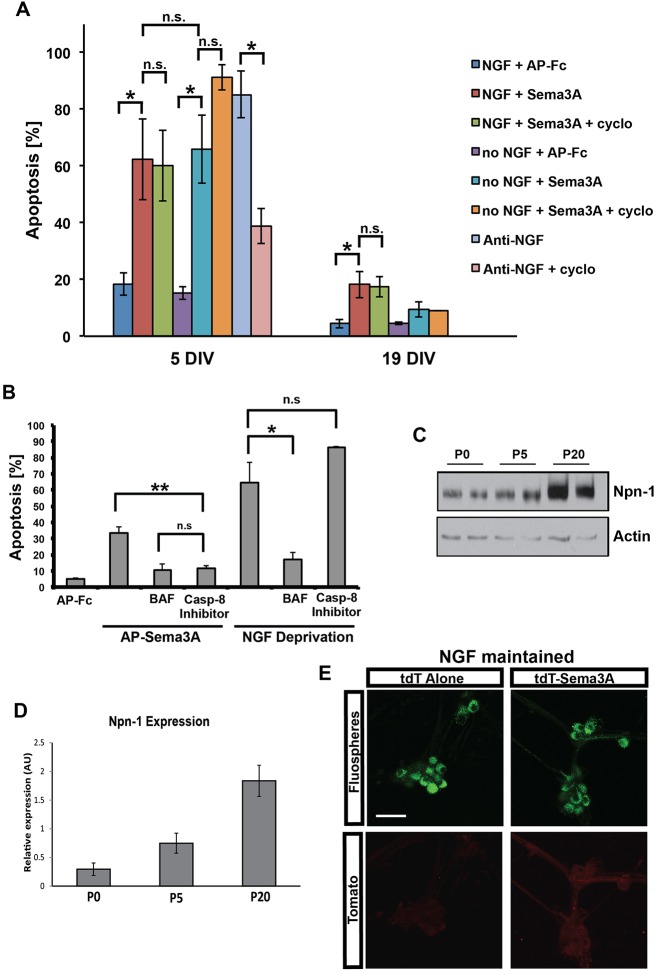
NGF deprivation of sympathetic neurons triggers apoptosis via the intrinsic pathway, which requires gene expression that induces cytochrome c release from mitochondria, leading to the activation of caspases (Chang et al., 2002; Kirkland and Franklin, 2003; Freeman et al., 2004; Jacobs et al., 2006). The extrinsic pathway, by contrast, is typically initiated by death receptors such as the tumor necrosis factor receptors (TNFRs) that initiate caspase 8 activation at the plasma membrane (Benn and Woolf, 2004; Kantari and Walczak, 2011). Apoptosis-induced gene expression that occurs via the intrinsic pathway upregulates several proapoptotic mediators, such as cyclin D1, c-Jun, BIM (also known as Bcl2l11) and SM-20 (also known as Egln1) (Freeman et al., 2004), and NGF deprivation-induced apoptosis of sympathetic neurons is inhibited by cycloheximide (Deckwerth and Johnson, 1993; Estus et al., 1994; Freeman et al., 2004). To determine whether inhibition of protein synthesis also protected neurons from Sema3A-induced cell death, SCG neurons were treated with AP-Sema3A, or AP-Fc, in the presence or absence of cycloheximide. Nuclear condensation and apoptotic morphology (axon degeneration, loss of phase-bright appearance of cell bodies) were measured from images of the same fields of neurons before and after the stimulations in order to determine the total percentage of neurons that die in the presence Sema3A (Fig. 4A). There was no significant difference in the number of neurons that underwent Sema3A-induced apoptosis in the presence or absence of cycloheximide, suggesting that protein synthesis is not required for Sema3A-induced death (Fig. 4A). Consistent with previous reports, cycloheximide did rescue a large percentage of neurons from undergoing apoptosis upon NGF withdrawal (Fig. 4A).
Fig. 4.
Sema3A induces apoptosis via the extrinsic pathway. (A) SCG neurons were maintained for 5 DIV or 19 DIV, at which time they are fully mature. The neurons were then deprived of NGF, deprived of NGF in the presence of cycloheximide, given Sema3A in the presence or absence of NGF, or given Sema3A in the presence of cycloheximide for 24 h. The percentage of neurons that underwent apoptosis was quantified by measuring both cellular morphology and nuclear morphology. Mean±s.e.m. from three independent experiments. (B) SCG neurons were maintained in vitro for 5 DIV and then exposed to AP-Fc, AP-Sema3A, or deprived of NGF in the presence or absence of the pan-caspase inhibitor BAF (50 µM) or caspase 8 inhibitor II (5 µM). Apoptosis was ascertained 24 h later as described for A. Mean±s.e.m. from three independent experiments, except for the AP-FC treatment (n=2). (A,B) n.s., not significant; *P<0.05, **P<0.01, Student's t-test. (C) Sympathetic neurons maintained in culture to the equivalent of P0, P5 or P20 were detergent extracted and subjected to Npn1 immunoblotting after immunoprecipitation of Npn1. Note that Npn1 expression increased by P20 relative to actin, which was used as a loading control. (D) The experiments in C were quantified from three independent cultures; mean±s.e.m. (E) Retrograde Sema3A transport studies of compartmentalized cultures of P18 SCG neurons. Note that at this age little or no tdT-Sema3A was retrogradely transported, as compared with the transport of tdT alone. Scale bar: 50 μm.
As further support for Sema3A acting via the extrinsic pathway to induce apoptosis, we examined whether Sema3A-induced apoptosis could be blocked by a selective inhibitor of caspase 8. As expected, a pan-caspase inhibitor blocked cell death of sympathetic neurons that were either deprived of NGF or exposed to Sema3A (Fig. 4B). Specific inhibition of caspase 8, however, selectively blocked Sema3A-mediated apoptosis but did not inhibit NGF deprivation-induced apoptosis (Fig. 4B), indicating that caspase 8 is required for Sema3A-induced, but not NGF deprivation-induced, cell death. Taken together, the data presented in Fig. S2, Figs 3 and 4 support the hypothesis that Sema3A-induced apoptosis occurs via the extrinsic pathway.
It is possible that Sema3A could function to enhance, or accelerate, apoptosis in sympathetic neurons that are not receiving sufficient NGF. To test this hypothesis, NGF was removed from sympathetic cultures, but we did not include an anti-NGF antibody in the medium during this deprivation (which is used to block NGF that remains on the collagen and cell surface). This NGF deprivation method allows the neurons to survive in this very low amount of NGF for 24-48 h before apoptosis is initiated. AP-Sema3A, or AP-Fc alone, was added to these NGF-deprived neurons, and the extent of cell death measured after 24 h. As before, Sema3A induced robust and significant apoptosis in NGF-deprived sympathetic neurons, as compared with AP-Fc alone (Fig. 4A, P<0.05). Interestingly, there was no difference in the level of apoptosis induced by Sema3A in NGF-maintained neurons versus NGF-deprived neurons (Fig. 4A), indicating that Sema3A does not act to enhance or trigger apoptosis induced by NGF withdrawal.
Activation of the c-Jun transcriptional pathway is an important mediator of several different forms of neuronal cell death, including developmental apoptosis as well as injury-induced cell death (Karin and Gallagher, 2005; Borsello and Forloni, 2007). c-Jun phosphorylation in SCG neurons was examined upon Sema3A exposure. The number of phospho-c-Jun-immunolabeled neurons (Fig. S2A,B) was significantly higher in neurons exposed to AP-Sema3A than in those exposed to AP-Fc alone or to VEGF (Fig. S2B). These data suggest that Sema3A triggers c-Jun activation in sympathetic neurons, and that the Sema3A-triggered apoptotic pathway is largely extrinsic in nature.
Mature sympathetic neurons are resistant to Sema3A-induced apoptosis
In addition to examining the effect of protein synthesis inhibitors, we investigated whether the age of the sympathetic neurons influenced their sensitivity to Sema3A-induced apoptosis. Sympathetic neurons undergo a transition from acutely requiring NGF for survival to becoming largely insensitive to its removal (Easton et al., 1997). This transition, known as maturation, occurs in vivo as well as in primary SCG neurons in vitro (Angeletti et al., 1971; Goedert et al., 1978; Easton et al., 1997). In Figs 2 and 3, our experiments were performed on neurons maintained for 10-14 days in vitro (DIV), a period during which sympathetic neurons are in the process of becoming insensitive to apoptosis induced by NGF withdrawal. Sympathetic neurons at 5 DIV (immature) or 19 DIV (fully mature) were exposed to Sema3A and cell death was assessed. AP-Sema3A induced death of a significantly higher percentage of immature neurons than mature neurons (Fig. 4A). However, as much as 15% of mature SCG neurons remained Sema3A sensitive and still underwent apoptosis (Fig. 4A).
To determine whether the loss of sensitivity to Sema3A with age was due to a loss of expression of the receptor for Sema3A, Npn1, mass cultures of sympathetic neurons were matured in culture to the equivalent of postnatal day (P) 0, 5 and 20. The neurons were then detergent extracted, Npn1 was immunoprecipitated from these extracts and subjected to immunoblotting to determine the expression levels of Npn1 protein. The level of Npn1 did not decline with age and, in fact, increased by P20 (Fig. 4C,D). It is unlikely, therefore, that the loss of Sema3A sensitivity in mature neurons (Fig. 4A) is due to the downregulation of Npn1.
Interestingly, the retrograde transport of tdT-Sema3A declined in compartmentalized cultures of mature neurons (Fig. 4E). In fact, mature neurons were not observed to retrogradely transport tdT-Sema3A to the extent observed in young, NGF-dependent neurons (Fig. 1C,D). Although there was some specific, but faint, labeling in tdT-Sema3A-treated neurons as compared with tdT-treated neurons (Fig. 4E), this amount of labeling could not be accurately quantified. It is possible that this low amount of retrograde transport accounts for the 15% of apoptosis that occurs in mature neurons exposed to Sema3A. Taken together, mature sympathetic neurons are significantly less sensitive to Sema3A-induced cell death, which might be due to their inability to internalize and retrogradely transport Sema3A.
Npn1 and plexin A3 are required for Sema3A-induced cell death in sympathetic neurons
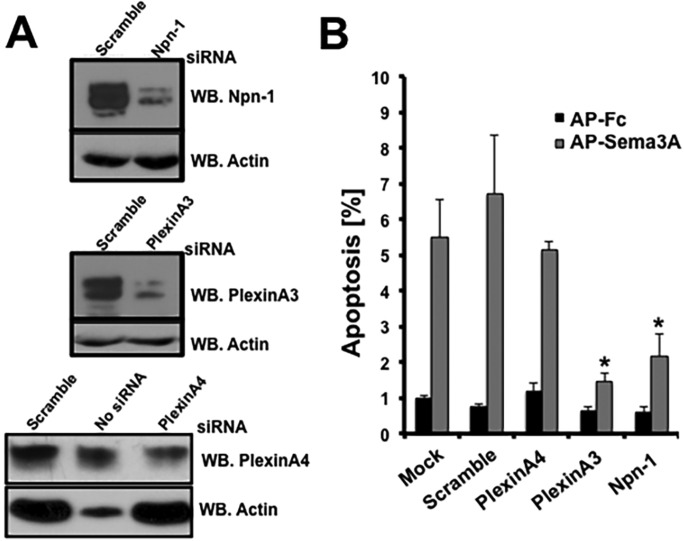
Sema3A mediates axon growth collapse in vitro and axon guidance in sympathetic and sensory neurons in vivo via Npn1 and plexin A4 (Yaron and Zheng, 2007; Pasterkamp and Giger, 2009). The observation that the Npn2 ligand Sema3F does not induce apoptosis of SCG neurons (Fig. 2) suggests that binding of Sema3F to Npn2 does not signal apoptosis (Giger et al., 1998). To determine which receptor components are necessary for Sema3A-mediated apoptosis of sympathetic neurons, siRNA silencing of Npn1, plexin A3 and plexin A4 was performed. Protein knockdown in primary SCG neurons was confirmed by immunoblotting (Fig. 5A). Activated caspase 3 immunolabeling was performed to quantify apoptosis in Sema3A-treated SCG neurons that were transfected with siRNAs to Npn1, plexin A3 (Plxna3), plexin A4 (Plxna4) or non-targeting RNA. As observed previously, apoptosis was induced by AP-Sema3A exposure, as compared with AP-Fc, in neurons transfected with noncoding siRNA (6.7±1.6% versus 0.7±0.1%; Fig. 5B). In marked contrast, Sema3A largely failed to induce apoptosis in neurons that underwent Npn1 or plexin A3 silencing, and there was no significant difference between neurons treated with AP-Sema3A or AP-Fc control (2.1±0.6% versus 0.6±0.1% for Npn1; 1.4±0.2% versus 0.6±0.1% for plexin A3; Fig. 5B). Knockdown of plexin A4 expression, however, did not significantly inhibit Sema3A-induced cell death (Sema3A 5.1±0.2% versus AP-Fc 1.2±0.2%; Fig. 5B). These results suggest that Sema3A-induced apoptosis of sympathetic neurons is mediated by an Npn1-plexin A3 receptor complex that is distinct from the Npn1-plexin A4 receptor complex that mediates Sema3A-induced axon repulsion (Yaron et al., 2005).
Fig. 5.
Npn1 and plexin A3 are required for Sema3A-induced apoptosis. (A) Cell lysates derived from primary sympathetic neurons that were not transfected or transfected with Npn1, Plxna3, Plxna4 or non-targeting control siRNAs were subject to western blotting to confirm effective knockdown of these proteins. Actin immunoblotting served as a loading control. (B) Activated caspase 3 immunofluorescence labeling of primary SCG neurons transfected with the siRNAs described in A was performed after their exposure to AP-Fc or AP-Sema3A for 24 h. Activated caspase 3+ cells were quantified as percentage of apoptosis. Mean±s.e.m. from three independent experiments. *P<0.05, Student's t-test.
Plexin A3 and Npn1 are required for developmental cell death of sympathetic neurons in vivo
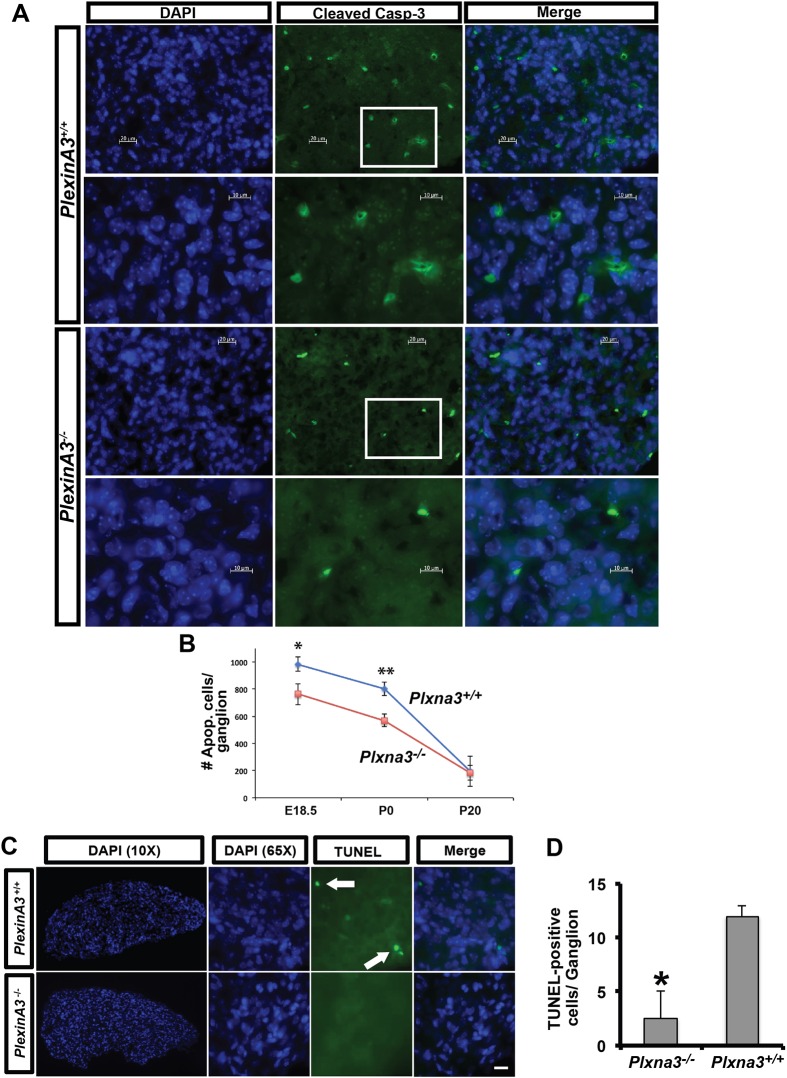
To determine whether plexin A3 and Npn1 are involved in developmental cell death of sympathetic neurons in vivo, the extent of apoptosis in the SCG was examined in Plxna3−/− knockout mice and in Npn1f/f;synapsin-Cre (Syn-Cre) mice. Apoptotic cells were detected in SCG sections of embryonic day (E) 18.5, P0 and P20 Plxna3−/− mice by immunofluorescence labeling for activated caspase 3 (Fig. 6A,B).
Fig. 6.
Programmed cell death is reduced in the SCG of Plxna3 knockout mice. (A) Activated caspase 3 immunofluorescence labeling (green) revealed a decrease in the number of apoptotic neurons in the P0 SCG of Plxna3−/− compared with Plxna3+/+ mice. Nuclei are stained with DAPI (blue). Boxed regions are shown at higher magnification in the lower row for each genotype. (B) The analysis in A was performed on E18.5 (n=5 for each genotype), P0 (n=4 for each genotype) and P20 (n=4 for Plxna3+/+ and n=5 for Plxna3−/−) SCG and activated caspase 3+ cells per ganglion quantified; mean±s.e.m. (C) TUNEL staining of Plxna3+/+ and Plxna3−/− SCG was performed on P1-P2 mice (n=3 for both genotypes). Arrows indicate TUNEL+ cell bodies. (D) Quantification of the number of TUNEL+ cells per ganglion from C; mean±s.e.m. (B,D) *P<0.05, **P<0.01, Student's t-test. Scale bars: 20 μm in C, and in A rows 1 and 3; 10 μm in A rows 2 and 4.
Interestingly, the number of activated caspase 3+ SCG neurons per ganglion was reduced significantly in E18.5 Plxna3−/− as compared with Plxna3+/+ mice (763±76 versus 985±55 activated caspase 3+ cells/ganglion, respectively). There were also significantly fewer activated caspase 3+ neurons in the SCG of P0 Plxna3−/− compared with Plxna3+/+ mice (571±46 versus 804±50 activated caspase 3+ cells/ganglion, respectively). By P20, an age when programmed cell death is largely complete in the SCG, there was no difference in the level of apoptosis between Plxna3−/− and Plxna3+/+ mice (184±53 versus 195±110, respectively). A similar reduction in apoptosis was observed by analyzing TUNEL staining, with fewer TUNEL+ cells observed in P1-P2 Plxna3−/− than in Plxna3+/+ SCG (2.5±2.5 versus 12±1 TUNEL+ cells/ganglion, respectively; Fig. 6C,D). The reduction of both activated caspase 3+ (E18.5 and P0) and TUNEL+ (P1-2) neurons in Plxna3−/− mice was significantly different than in Plxna3+/+ mice (P<0.05).
Npn1 was deleted from sympathetic neurons using Npn1f/f;Syn-Cre in order to avoid the embryonic lethality of Npn1−/− mice due to vascular defects, which would have precluded analysis of programmed cell death in the SCG (Kitsukawa et al., 1997). The loss of Npn1 was confirmed in the SCG of Npn1f/f;Syn-Cre mice by the absence of AP-Sema3A binding to sections of SCG from Npn1f/f;Syn-Cre as compared with Npn1f/f mice (Fig. 7A). Similar to Plxna3−/− mice, there were fewer apoptotic neurons in the SCG of P1-P2 Npn1f/f;Syn-Cre as compared with Npn1f/f mice (3±2.5 versus 51±10.8 caspase 3+ neurons/ganglion, P<0.05; Fig. 7B). Taken together, these data indicate that both plexin A3 and Npn1 are involved in developmental cell death of SCG neurons, most likely via their interaction with Sema3A.
Fig. 7.
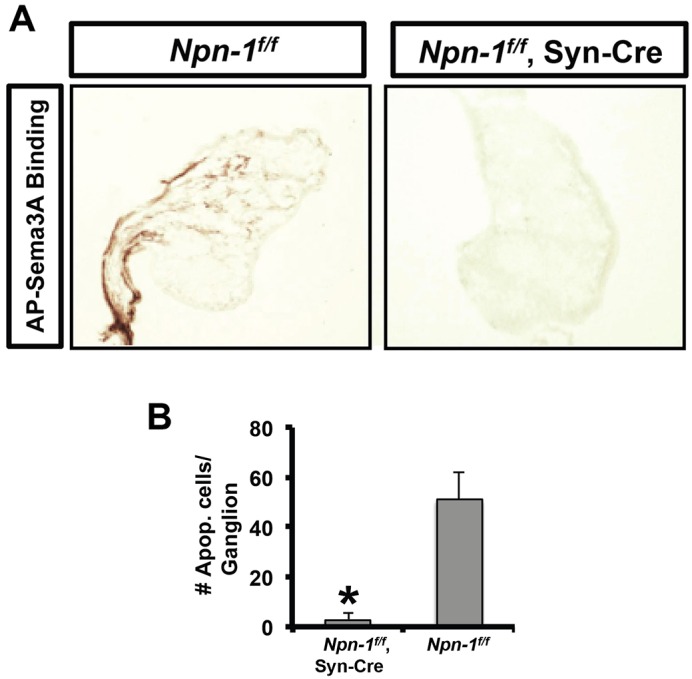
Neural deletion of Npn1 rescues neurons from apoptosis. (A) Confirmation of Npn1 deletion from sympathetic neurons was achieved using a Sema3A binding assay. Assays were performed on sections of SCG isolated from either Npn1f/f or Npn1f/f;Syn-Cre mice at P1-P2. Npn1f/f;Syn-Cre SCG did not bind Sema3A (right), in contrast to Npn1f/f SCG (left), functionally confirming the deletion of Npn1. (B) SCG from Npn1f/f (n=3) or Npn1f/f;Syn-Cre (n=3) mice were serially sectioned and the number of activated caspase 3+ cells per ganglion quantified as in Fig. 6. Mean±s.e.m. *P<0.05, Student's t-test.
Based on these observations, we sought to determine whether there was a sustained increase in the number of neurons in the SCG after the period of programmed cell death. In some knockout mice, the deletion of a molecule involved in apoptosis results in an initial protection from cell death but the eventual elimination of these neurons, such as in p75−/− (p75 is also known as Ngfr) mice (Deppmann et al., 2008). In Bax knockout mice, by contrast, all neurons are rescued from apoptosis permanently (Deckwerth et al., 1996), indicating that Bax is absolutely required for the apoptosis pathway in SCG neurons. To distinguish between these two possibilities, the numbers of neurons in the SCG of P20 Plxna3−/− and Plxna3+/+ mice were compared. Because a delay in apoptosis would be difficult to detect by activated caspase 3 staining if it occurred over the course of days to weeks, tyrosine hydroxylase (TH) immunolabeling was analyzed at P20. TH immunostaining, which labels greater than 95% of SCG neurons, was performed on serial sections of SCG, and the total number of TH+ neurons in each ganglion was counted (Fig. 8A,B). There was no significant difference in the number of TH+ neurons between Plxna3−/− and Plxna3+/+ SCG (6244±924 neurons versus 7072±97 neurons, respectively). Therefore, the neurons that are protected from apoptosis in Plxna3−/− mice between E18.5 and P2 eventually go on to die and do not persist beyond P20.
Fig. 8.
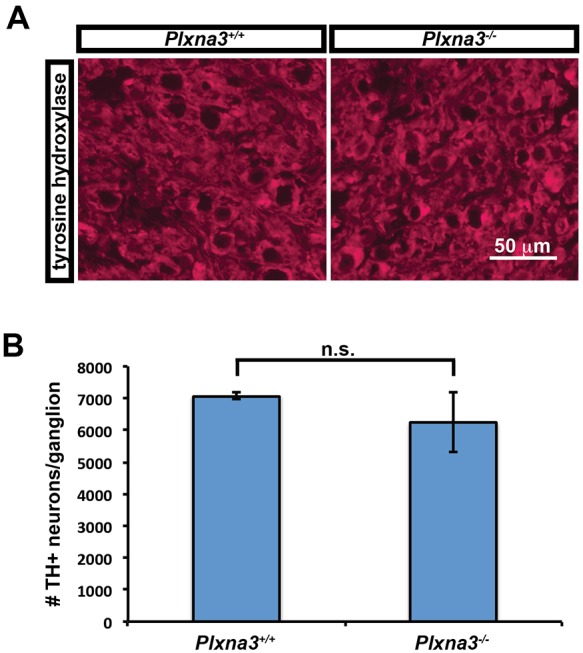
Programmed cell death in the SCG is eventually complete by P20 in Plxna3−/− mice. (A) Tyrosine hydroxylase (TH) immunolabeling was performed on serial sections of SCG from P20 Plxna3+/+ (left) and Plxna3−/− (right) mice. (B) The total number of TH+ neurons in each SCG was ascertained by counting the number of TH+ cells in every third section throughout the entire ganglia (n=3 for both genotypes). There was no significant difference by t-test. Mean±s.e.m.
DISCUSSION
Sema3A is best known for its role as a repulsive axon guidance cue for developing PNS and CNS neurons (Luo et al., 1993; Messersmith et al., 1995; Behar et al., 1996; Varela-Echavarria et al., 1997). Besides its role in regulating neuronal morphology, accumulating evidence suggests that Sema3A induces neuronal apoptosis (Gagliardini and Fankhauser, 1999; Shirvan et al., 1999, 2002; Ben-Zvi et al., 2006). Despite recent progress, whether Sema3A, Npn1 and plexins are physiologically relevant in developmental cell death in vivo, and the mechanisms underlying semaphorin-induced apoptosis, remain poorly understood (Ben-Zvi et al., 2006, 2008; Jiang et al., 2007, 2010; Haupt et al., 2010). Here we identify Sema3A as a retrograde death signal for developing sympathetic neurons. In compartmentalized cultures of SCG neurons, Sema3A applied to distal axons is retrogradely transported to the cell soma and, upon its arrival, triggers caspase 3 and caspase 8-dependent cell death. Mechanistic studies identified Npn1 and plexin A3 as obligatory receptor components for Sema3A-induced cell death. Sema3A-initiated apoptosis is not NGF sensitive and progresses via the extrinsic cell death pathway. Importantly, deletion of either Plxna3 or Npn1 protects SCG neurons from naturally occurring cell death during early postnatal development, indicating that retrograde Sema3A signals the programmed cell death of sympathetic neurons by a plexin A3-Npn1 receptor complex.
Eliminating excessively produced neurons by programmed cell death is an important step during nervous system development and patterning. In the PNS, it is well established that remote signals at nerve terminals influence the final number of neurons through the regulation of cell death and survival (Levi-Montalcini, 1987; Oppenheim, 1991). Pro-survival neurotrophic factors are produced in limiting amounts by their peripheral targets and neurons compete for access to neurotrophic factors. Mounting evidence suggests the existence of pro-apoptotic signals, such as precursor forms of neurotrophins, that trigger cell death through activation of p75 (Lee et al., 2001; Teng et al., 2010). Evidence for semaphorin-induced cell death stems predominantly from experiments with neural tumor cells and cultured neurons (Gagliardini and Fankhauser, 1999; Shirvan et al., 1999; Bagnard et al., 2001; Ben-Zvi et al., 2006). The extent to which Sema3A is involved in developmental cell death of embryonic sensory and motor neurons is debated (Ben-Zvi et al., 2008, 2013; Haupt et al., 2010). During embryonic development, Sema3A is broadly expressed in the PNS and is often found along the trajectory of sensory, motor and sympathetic fibers (Giger et al., 1996). Whether Sema3A primarily functions in axon guidance of sympathetic neurons or also contributes to their programmed cell death, however, has remained unknown. Here we show that Sema3A induces the developmental stage-dependent cell death of sympathetic neurons. We propose two possible models of how Sema3A may regulate the developmental cell death of sympathetic neurons. First, Sema3A released from non-target areas is endocytosed at the growth cone and retrogradely transported to the cell body to trigger cell death. Prolonged exposure to Sema3A may trigger cell death and eliminate neurons with axons that have wandered astray and fail to reach their appropriate target. Consistent with this idea, the PNS of Sema3a−/− mice shows profound defasciculation of sensory, motor and sympathetic projections. Second, sympathetic neurons themselves that have innervated their correct targets and receive sufficient NGF, i.e. ‘winning neurons’, express Sema3A as a competition factor and release it into target areas or the ganglion itself. This Sema3A then induces the apoptosis of neurons that are not obtaining sufficient amounts of NGF, i.e. ‘losing neurons’. The observations that NGF-deprived neurons are not more susceptible to Sema3A, and that Sema3A induces the apoptosis of neurons even in the presence of NGF, provide some evidence contrary to this competition model. An important future direction will be to identify the cell types that express Sema3A, and when during development they do so, to aid in the prediction of whether Sema3A acts to eliminate off-target neurons or acts as a competition factor.
The observation that retrograde Sema3A-induced apoptosis is inhibited by microtubule depolymerization indicates that the death-inducing signal is retrogradely transported from distal axons to cell bodies. When coupled with the previous observation that Sema3A binding to Npn1 triggers rapid endocytosis at the neuronal growth cone (Fournier et al., 2000), the retrograde apoptosis signal might be composed of a Sema3A-Npn1-plexin A3 ligand-receptor complex, rather than Sema3A alone. Similar to Sema3A, growth inhibitory molecules belonging to the ephrin family (Jurney et al., 2002; Zimmer et al., 2003), or the reticulon family member Nogo-A (also known as Rtn4) (Joset et al., 2010), have been shown to undergo rapid endocytosis, furthering the likelihood that axon guidance complexes may also affect cellular events remotely from their initial location of receptor binding. Additional studies are needed to determine the molecular composition of the retrogradely transported death complex, as well as the mechanism by which it is internalized and trafficked to cell bodies. The signaling events subsequent to Sema3A-Npn1-plexin A3 receptor assembly that induce apoptosis are not well understood. Although we did not observe alterations in TrkA retrograde signaling upon Sema3A treatment, semaphorins have been reported to inhibit NGF-mediated Akt and Erk signaling (Atwal et al., 2003), suggesting that they could potentially antagonize NGF signaling and induce apoptosis via the intrinsic pathway. The observations, however, that Sema3A-mediated apoptosis did not require new protein synthesis and required caspase 8 activation, suggest that death occurred via an extrinsic pathway. There were some similarities, however, between NGF deprivation-induced death and Sema3A-induced death, such as the activation of c-Jun and the loss of sensitivity in mature neurons. Both the intrinsic pathway and the extrinsic pathway can induce JNK/c-Jun signaling (Benn and Woolf, 2004; Kantari and Walczak, 2011). Our observation that mature neurons are less sensitive to both NGF deprivation and Sema3A suggests that there might be common components between these two death pathways, such as Apaf1 (Wright et al., 2007). Alternatively, Sema3A receptor components or downstream components of the Sema3A-initiated apoptotic signaling cascade could be downregulated as sympathetic neurons mature. We did not observe a decrease in Npn1 expression, but the retrograde transport of Sema3A was reduced dramatically in mature sympathetic neurons, suggesting that the loss of sensitivity to Sema3A might be at the level of receptor internalization and transport. Interestingly, the pro-apoptotic serine/threonine kinase NDR (also known as Stk38) was recently identified as a signaling target of MICAL, which associates with plexins and is required for Sema3A-mediated axon repulsion (Terman et al., 2002; Zhou et al., 2011). It will be important to determine whether any of these signaling molecules are altered during the maturation of sympathetic neurons.
Lastly, it is well established that Sema3A and other members of the semaphorin family are upregulated following injury to the PNS and CNS (Pasterkamp and Giger, 2009). Axonal injury to the mammalian optic nerve, for example, triggers death of the majority of RGCs (Dickendesher et al., 2012) and correlates with an upregulation of Sema3A. Remarkably, intraocular injection of blocking antibodies to Sema3A inhibits injury-induced death of RGCs (Shirvan et al., 2002), suggesting that a detailed understanding of the Sema3A cell death mechanisms might aid the development of therapeutic strategies to block neuronal cell death following nervous system injury or disease.
MATERIALS AND METHODS
Culture of SCG neurons
The production of primary sympathetic neurons from rat Sprague-Dawley embryos has been described previously (Tsui-Pierchala and Ginty, 1999). Glial cells were eliminated from the cultures by including aphidicolin (5 µg/ml, Sigma) to the culture medium. Compartmentalized cultures of sympathetic neurons were produced as described previously (Tsui-Pierchala and Ginty, 1999). In both mass and compartmentalized cultures, neurons were maintained for 5-21 DIV before treatment. The experimental treatments of these neurons, including siRNA silencing, are described in the supplementary Materials and Methods. All procedures on mice and rats were performed in accordance with National Institutes of Health guidelines and were approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Michigan.
Sema3A fusion proteins
The construction and bioactivity analysis of Sema3A fusion proteins, including tdT-Sema3A and AP-Sema3A, are described in the supplementary Materials and Methods.
Immunofluorescence and TUNEL labeling
Some of the Plxna3−/− mice used in this study were generously provided by Alex Kolodkin (Cheng et al., 2001). For conditional Npn1 knockout mice, neuron-specific Npn1f/f;Syn-Cre P1 mice were generated from heterozygous matings (Gu et al., 2003). For the analysis of whole SCG, P1 mice were collected and the neck regions were fixed with 4% paraformaldehyde (PFA) in PBS overnight at 4°C, washed in PBS and cryoprotected. Tissue cryosections (10 µm) were used for immunofluorescence labeling of activated caspase 3. SCG sections were incubated in PBS containing 1% BSA (Sigma), 0.1% Triton X-100 (Sigma), MOM (Vector Labs) and 5% goat serum for 1 h at room temperature (RT), followed by incubation with primary antibody against activated caspase 3 (1:250; Cell Signaling Technology, 9661) or TH (1:250; Millipore, AB1542). For immunofluorescence labeling of primary SCG neurons, the cells were washed in cold PBS and fixed in 4% PFA for 20 min at RT. Neurons were permeabilized in PBS containing 0.3% Triton X-100 (5 min, RT), incubated in PBS containing 1% BSA and 5% normal goat serum (NGS; 30 min, RT) and then incubated with activated caspase 3 antibody (1:500) or RFP antibody for tdT-Sema3A (1:250; Rockland, 600-401-379) in the blocking solution overnight. The labeling was detected using Alexa 488-conjugated donkey anti-rabbit and Alexa 633-conjugated donkey anti-sheep antibodies (both 1:250; Biotium, 20015 and 20134; 2 h, RT).
The terminal deoxynucleotidyl transferase-mediated dUTP nicked end labeling (TUNEL) assay was performed on 10 µm sections of SCG using the ApopTag Fluorescein In Situ Apoptosis Detection Kit (Millipore) following the manufacturer's protocol. Microscopy and the quantification of immunolabeling/apoptosis are described in the supplementary Materials and Methods.
Immunoblotting
Western blotting was performed as described previously (Tsui-Pierchala and Ginty, 1999). Antibodies (1:1000, unless stated otherwise) and dilutions used in immunoblotting were: anti-phosphotyrosine (Millipore, 05-321), anti-TrkA (Santa Cruz, SC-11), anti-activated caspase 3 (Cell Signaling, 9661), anti-Npn1 (Cell Signaling, 3725), anti-plexin A3 (Cell Signaling, 5512), anti-plexin A4 (Cell Signaling, 3816), anti-actin (Santa Cruz, SC-1616), anti-DsRed (Clontech, 632496) and anti-β-actin (Sigma, A2066; 1:2000). Immunoblots were visualized using enhanced chemiluminescence substrates (SuperSignal, Thermo Fisher).
Immunoprecipitations
After stimulation of compartmentalized sympathetic neurons, the dishes were placed on ice, gently washed twice with PBS, and the cell bodies and distal axons were lysed separately with immunoprecipitation (IP) buffer as described previously (Tsui-Pierchala and Ginty, 1999). TrkA was immunoprecipitated with Trk antibody (C-14; Santa Cruz, SC-11) along with protein A and protein G (Roche). After 24 h gentle agitation at 4°C, the immunoprecipitates were washed three times with IP buffer and the complexes were prepared for SDS-PAGE by adding 2× sample buffer (125 mM Tris pH 6.8, 20% glycerol, 4% SDS, 0.016% Bromophenol Blue) and heating for 6 min at 100°C. These extracts were subjected to SDS-PAGE followed by electroblotting onto PVDF membranes (Millipore) and western blot analysis as above.
Acknowledgements
We thank Fumikazu Suto and Alan Halim or expert technical assistance.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
H.A., A.B.W., B.A.P. and R.J.G. conceived and designed the experiments. A.B.W., H.A. and T.L.D. carried out the experiments. F.I. and Y.Y. produced and provided tissues of the appropriate ages and provided live Plxna3−/− mice. A.B.W., H.A., T.L.D., B.A.P. and R.J.G. analyzed the data. A.B.W., H.A. and B.A.P. wrote the manuscript. A.B.W., H.A., T.L.D., B.A.P. and R.J.G. reviewed and edited the manuscript.
Funding
This research was funded by the National Institutes of Health [grants R01 NS058510 to B.A.P. and R01 NS081281 to R.J.G.]; the Dr Miriam and Sheldon Adelson Medical Research Foundation (R.J.G.); and by The University of Michigan Regents Fellowships (T.L.D. and A.B.W.). Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.134627/-/DC1
References
- Angeletti P. U., Levi-Montalcini R. and Caramia F. (1971). Analysis of the effects of the antiserum to the nerve growth factor in adult mice. Brain Res. 27, 343-355. 10.1016/0006-8993(71)90259-9 [DOI] [PubMed] [Google Scholar]
- Atwal J. K., Singh K. K., Tessier-Lavigne M., Miller F. D. and Kaplan D. R. (2003). Semaphorin 3F antagonizes neurotrophin-induced phosphatidylinositol 3-kinase and mitogen-activated protein kinase kinase signaling: a mechanism for growth cone collapse. J. Neurosci. 23, 7602-7609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagnard D., Vaillant C., Khuth S. T., Dufay N., Lohrum M., Puschel A. W., Belin M. F., Bolz J. and Thomasset N. (2001). Semaphorin 3A-vascular endothelial growth factor-165 balance mediates migration and apoptosis of neural progenitor cells by the recruitment of shared receptor. J. Neurosci. 21, 3332-3341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behar O., Golden J. A., Mashimo H., Schoen F. J. and Fishman M. C. (1996). Semaphorin III is needed for normal patterning and growth of nerves, bones and heart. Nature 383, 525-528. 10.1038/383525a0 [DOI] [PubMed] [Google Scholar]
- Ben-Zvi A., Yagil Z., Hagalili Y., Klein H., Lerman O. and Behar O. (2006). Semaphorin 3A and neurotrophins: a balance between apoptosis and survival signaling in embryonic DRG neurons. J. Neurochem. 96, 585-597. 10.1111/j.1471-4159.2005.03580.x [DOI] [PubMed] [Google Scholar]
- Ben-Zvi A., Manor O., Schachner M., Yaron A., Tessier-Lavigne M. and Behar O. (2008). The Semaphorin receptor PlexinA3 mediates neuronal apoptosis during dorsal root ganglia development. J. Neurosci. 28, 12427-12432. 10.1523/JNEUROSCI.3573-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Zvi A., Sweetat S. and Behar O. (2013). Elimination of aberrant DRG circuitries in Sema3A mutant mice leads to extensive neuronal deficits. PLoS ONE 8, e70085 10.1371/journal.pone.0070085 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benn S. C. and Woolf C. J. (2004). Adult neuron survival strategies-slamming on the brakes. Nat. Rev. Neurosci. 5, 686-700. 10.1038/nrn1477 [DOI] [PubMed] [Google Scholar]
- Borsello T. and Forloni G. (2007). JNK signalling: a possible target to prevent neurodegeneration. Curr. Pharm. Des. 13, 1875-1886. 10.2174/138161207780858384 [DOI] [PubMed] [Google Scholar]
- Chang L. K., Putcha G. V., Deshmukh M. and Johnson E. M. Jr (2002). Mitochondrial involvement in the point of no return in neuronal apoptosis. Biochemie 84, 223-231. 10.1016/S0300-9084(02)01372-X [DOI] [PubMed] [Google Scholar]
- Cheng H.-J., Bagri A., Yaron A., Stein E., Pleasure S. J. and Tessier-Lavigne M. (2001). Plexin-A3 mediates semaphorin signaling and regulates the development of hippocampal axonal projections. Neuron 32, 249-263. 10.1016/S0896-6273(01)00478-0 [DOI] [PubMed] [Google Scholar]
- Coggeshall R. E., Pover C. M. and Fitzgerald M. (1994). Dorsal root ganglion cell death and surviving cell numbers in relation to the development of sensory innervation in the rat hindlimb. Brain Res. Dev. Brain Res. 82, 193-212. 10.1016/0165-3806(94)90163-5 [DOI] [PubMed] [Google Scholar]
- Cosker K. E., Courchesne S. L. and Segal R. A. (2008). Action in the axon: generation and transport of signaling endosomes. Curr. Opin. Neurobiol. 18, 270-275. 10.1016/j.conb.2008.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deckwerth T. L. and Johnson E. M. Jr (1993). Temporal analysis of events associated with programmed cell death (apoptosis) of sympathetic neurons deprived of nerve growth factor. J. Cell Biol. 123, 1207-1222. 10.1083/jcb.123.5.1207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deckwerth T. L., Elliott J. L., Knudson C. M., Johnson E. M. Jr, Snider W. D. and Korsmeyer S. J. (1996). BAX is required for neuronal death after trophic factor deprivation and during development. Neuron 17, 401-411. 10.1016/S0896-6273(00)80173-7 [DOI] [PubMed] [Google Scholar]
- Deppmann C. D., Mihalas S., Sharma N., Lonze B. E., Niebur E. and Ginty D. D. (2008). A model for neuronal competition during development. Science 320, 369-373. 10.1126/science.1152677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickendesher T. L., Baldwin K. T., Mironova Y. A., Koriyama Y., Raiker S. J., Askew K. L., Wood A., Geoffroy C. G., Zheng B., Liepmann C. D. et al. (2012). NgR1 and NgR3 are receptors for chondroitin sulfate proteoglycans. Nat. Neurosci. 15, 703-712. 10.1038/nn.3070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Easton R. M., Deckwerth T. L., Parsadanian A. S. and Johnson E. M. (1997). Analysis of the mechanism of loss of trophic factor dependence associated with neuronal maturation: a phenotype indistinguishable from Bax deletion. J. Neurosci. 17, 9656-9666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estus S., Zaks W. J., Freeman R. S., Gruda M., Bravo R. and Johnson E. M. Jr (1994). Altered gene expression in neurons during programmed cell death: identification of c-jun as necessary for neuronal apoptosis. J. Cell Biol. 127, 1717-1727. 10.1083/jcb.127.6.1717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fournier A. E., Nakamura F., Kawamoto S., Goshima Y., Kalb R. G. and Strittmatter S. M. (2000). Semaphorin3A enhances endocytosis at sites of receptor-F-actin colocalization during growth cone collapse. J. Cell Biol. 149, 411-422. 10.1083/jcb.149.2.411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freeman R. S., Burch R. L., Crowder R. J., Lomb D. J., Schoell M. C., Straub J. A. and Xie L. (2004). NGF deprivation-induced gene expression: after ten years, where do we stand? Prog. Brain Res. 146, 111-126. 10.1016/S0079-6123(03)46008-1 [DOI] [PubMed] [Google Scholar]
- Gagliardini V. and Fankhauser C. (1999). Semaphorin III can induce death in sensory neurons. Mol. Cell. Neurosci. 14, 301-316. 10.1006/mcne.1999.0787 [DOI] [PubMed] [Google Scholar]
- Giger R. J., Wolfer D. P., De Wit G. M. J. and Verhaagen J. (1996). Anatomy of rat semaphorin III/collapsin-1 mRNA expression and relationship to developing nerve tracts during neuroembryogenesis. J. Comp. Neurol. 375, 378-392. [DOI] [PubMed] [Google Scholar]
- Giger R. J., Urquhart E. R., Gillespie S. K. H., Levengood D. V., Ginty D. D. and Kolodkin A. L. (1998). Neuropilin-2 is a receptor for semaphorin IV: insight into the structural basis of receptor function and specificity. Neuron 21, 1079-1092. 10.1016/S0896-6273(00)80625-X [DOI] [PubMed] [Google Scholar]
- Giger R. J., Cloutier J.-F., Sahay A., Prinjha R. K., Levengood D. V., Moore S. E., Pickering S., Simmons D., Rastan S., Walsh F. S. et al. (2000). Neuropilin-2 is required in vivo for selective axon guidance responses to secreted semaphorins. Neuron 25, 29-41. 10.1016/S0896-6273(00)80869-7 [DOI] [PubMed] [Google Scholar]
- Giraudon P., Vincent P., Vuaillat C., Verlaeten O., Cartier L., Marie-Cardine A., Mutin M., Bensussan A., Belin M.-F. and Boumsell L. (2004). Semaphorin CD100 from activated T lymphocytes induces process extension collapse in oligodendrocytes and death of immature neural cells. J. Immunol. 172, 1246-1255. 10.4049/jimmunol.172.2.1246 [DOI] [PubMed] [Google Scholar]
- Goedert M., Otten U. and Thoenen H. (1978). Biochemical effects of antibodies against nerve growth factor on developing and differentiated sympathetic ganglia. Brain Res. 148, 264-268. 10.1016/0006-8993(78)90401-8 [DOI] [PubMed] [Google Scholar]
- Gu C., Rodriguez E. R., Reimert D. V., Shu T., Fritzsch B., Richards L. J., Kolodkin A. L. and Ginty D. D. (2003). Neuropilin-1 conveys semaphorin and VEGF signaling during neural and cardiovascular development. Dev. Cell 5, 45-57. 10.1016/S1534-5807(03)00169-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haupt C., Kloos K., Faus-Kessler T. and Huber A. B. (2010). Semaphorin 3A-Neuropilin-1 signaling regulates peripheral axon fasciculation and pathfinding but not developmental cell death patterns. Eur. J. Neurosci. 31, 1164-1172. 10.1111/j.1460-9568.2010.07154.x [DOI] [PubMed] [Google Scholar]
- He Z. and Tessier-Lavigne M. (1997). Neuropilin is a receptor for the axonal chemorepellent Semaphorin III. Cell 90, 739-751. 10.1016/S0092-8674(00)80534-6 [DOI] [PubMed] [Google Scholar]
- Jacobs W. B., Kaplan D. R. and Miller F. D. (2006). The p53 family in nervous system development and disease. J. Neurochem. 97, 1571-1584. 10.1111/j.1471-4159.2006.03980.x [DOI] [PubMed] [Google Scholar]
- Jiang S. X., Sheldrick M., Desbois A., Slinn J. and Hou S. T. (2007). Neuropilin-1 is a direct target of the transcription factor E2F1 during cerebral ischemia-induced neuronal death in vivo. Mol. Cell. Biol. 27, 1696-1705. 10.1128/MCB.01760-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang S. X., Whitehead S., Aylsworth A., Slinn J., Zurakowski B., Chan K., Li J. and Hou S. T. (2010). Neuropilin 1 directly interacts with Fer kinase to mediate semaphorin 3A-induced death of cortical neurons. J. Biol. Chem. 285, 9908-9918. 10.1074/jbc.M109.080689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joset A., Dodd D. A., Halegoua S. and Schwab M. E. (2010). Pincher-generated Nogo-A endosomes mediate growth cone collapse and retrograde signaling. J. Cell Biol. 188, 271-285. 10.1083/jcb.200906089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurney W. M., Gallo G., Letourneau P. C. and McLoon S. C. (2002). Rac1-mediated endocytosis during ephrin-A2- and semaphorin 3A-induced growth cone collapse. J. Neurosci. 22, 6019-6028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kantari C. and Walczak H. (2011). Caspase-8 and Bid: caught in the act between death receptors and mitochondria. Biochim. Biophys. Acta 1813, 558-563. 10.1016/j.bbamcr.2011.01.026 [DOI] [PubMed] [Google Scholar]
- Karin M. and Gallagher E. (2005). From JNK to pay dirt: jun kinases, their biochemistry, physiology and clinical importance. IUBMB Life 57, 283-295. 10.1080/15216540500097111 [DOI] [PubMed] [Google Scholar]
- Kirkland R. A. and Franklin J. L. (2003). Bax, reactive oxygen, and cytochrome c release in neuronal apoptosis. Antioxid. Redox Signal. 5, 589-596. 10.1089/152308603770310257 [DOI] [PubMed] [Google Scholar]
- Kitsukawa T., Shimizu M., Sanbo M., Hirata T., Taniguchi M., Bekku Y., Yagi T. and Fujisawa H. (1997). Neuropilin-semaphorin III/D-mediated chemorepulsive signals play a crucial role in peripheral nerve projection in mice. Neuron 19, 995-1005. 10.1016/S0896-6273(00)80392-X [DOI] [PubMed] [Google Scholar]
- Kolodkin A. L. and Tessier-Lavigne M. (2011). Mechanisms and molecules of neuronal wiring: a primer. Cold Spring Harb. Perspect. Biol. 3, a001727 10.1101/cshperspect.a001727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolodkin A. L., Levengood D. V., Rowe E. G., Tai Y.-T., Ginger R. J. and Ginty D. D. (1997). Neuropilin is a semaphorin III receptor. Cell 90, 753-762. 10.1016/S0092-8674(00)80535-8 [DOI] [PubMed] [Google Scholar]
- Lee R., Kermani P., Teng K. K. and Hempstead B. L. (2001). Regulation of cell survival by secreted proneurotrophins. Science 294, 1945-1948. 10.1126/science.1065057 [DOI] [PubMed] [Google Scholar]
- Levi-Montalcini R. (1987). The nerve growth factor 35 years later. Science 237, 1154-1162. 10.1126/science.3306916 [DOI] [PubMed] [Google Scholar]
- Luo Y., Raible D. and Raper J. A. (1993). Collapsin: a protein in brain that induces the collapse and paralysis of neuronal growth cones. Cell 75, 217-227. 10.1016/0092-8674(93)80064-L [DOI] [PubMed] [Google Scholar]
- Messersmith E. K., Leonardo E. D., Shatz C. J., Tessier-Lavigne M., Goodman C. S. and Kolodkin A. L. (1995). Semaphorin III can function as a selective chemorepellent to pattern sensory projections in the spinal cord. Neuron 14, 949-959. 10.1016/0896-6273(95)90333-X [DOI] [PubMed] [Google Scholar]
- Nakamura F., Kalb R. G. and Strittmatter S. M. (2000). Molecular basis of semaphorin-mediated axon guidance. J. Neurobiol. 44, 219-229. [DOI] [PubMed] [Google Scholar]
- Newquist G., Drennan J. M., Lamanuzzi M., Walker K., Clemens J. C. and Kidd T. (2013). Blocking apoptotic signaling rescues axon guidance in Netrin mutants. Cell Rep. 3, 595-606. 10.1016/j.celrep.2013.02.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oppenheim R. W. (1991). Cell death during development of the nervous system. Annu. Rev. Neurosci. 14, 453-501. 10.1146/annurev.ne.14.030191.002321 [DOI] [PubMed] [Google Scholar]
- Pasterkamp R. J. and Giger R. J. (2009). Semaphorin function in neural plasticity and disease. Curr. Opin. Neurobiol. 19, 263-274. 10.1016/j.conb.2009.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirvan A., Ziv I., Fleminger G., Shina R., He Z., Brudo I., Melamed E. and Barzilai A. (1999). Semaphorins as mediators of neuronal apoptosis. J. Neurochem. 73, 961-971. 10.1046/j.1471-4159.1999.0730961.x [DOI] [PubMed] [Google Scholar]
- Shirvan A., Kimron M., Holdengreber V., Ziv I., Ben-Shaul Y., Melamed S., Melamed E., Barzilai A. and Solomon A. S. (2002). Anti-semaphorin 3A antibodies rescue retinal ganglion cells from cell death following optic nerve axotomy. J. Biol. Chem. 277, 49799-49807. 10.1074/jbc.M204793200 [DOI] [PubMed] [Google Scholar]
- Takahashi T., Fournier A., Nakamura F., Wang L.-H., Murakami Y., Kalb R. G., Fujisawa H. and Strittmatter S. M. (1999). Plexin-neuropilin-1 complexes form functional semaphorin-3A receptors. Cell 99, 59-69. 10.1016/S0092-8674(00)80062-8 [DOI] [PubMed] [Google Scholar]
- Tamagnone L., Artigiani S., Chen H., He Z., Ming G.L., Song H.-J., Chedotal A., Winberg M. L., Goodman C. S., Poo M.-M. et al. (1999). Plexins are a large family of receptors for transmembrane, secreted, and GPI-anchored semaphorins in vertebrates. Cell 99, 71-80. 10.1016/S0092-8674(00)80063-X [DOI] [PubMed] [Google Scholar]
- Teng K. K., Felice S., Kim T. and Hempstead B. L. (2010). Understanding proneurotrophin actions: recent advances and challenges. Dev. Neurobiol. 70, 350-359. 10.1002/dneu.20768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terman J. R., Mao T., Pasterkamp R. J., Yu H.-H. and Kolodkin A. L. (2002). MICALs, a family of conserved flavoprotein oxidoreductases, function in plexin-mediated axonal repulsion. Cell 109, 887-900. 10.1016/S0092-8674(02)00794-8 [DOI] [PubMed] [Google Scholar]
- Tsui-Pierchala B. A. and Ginty D. D. (1999). Characterization of an NGF-P-TrkA retrograde-signaling complex and age-dependent regulation of TrkA phosphorylation in sympathetic neurons. J. Neurosci. 19, 8207-8218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varela-Echavarria A., Tucker A., Puschel A. W. and Guthrie S. (1997). Motor axon subpopulations respond differentially to the chemorepellents netrin-1 and semaphorin D. Neuron 18, 193-207. 10.1016/S0896-6273(00)80261-5 [DOI] [PubMed] [Google Scholar]
- White F. A. and Behar O. (2000). The development and subsequent elimination of aberrant peripheral axon projections in Semaphorin3A null mutant mice. Dev. Biol. 225, 79-86. 10.1006/dbio.2000.9822 [DOI] [PubMed] [Google Scholar]
- Wright K. M., Smith M. I., Farrag L. and Deshmukh M. (2007). Chromatin modification of Apaf-1 restricts the apoptotic pathway in mature neurons. J. Cell Biol. 179, 825-832. 10.1083/jcb.200708086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaron A. and Zheng B. (2007). Navigating their way to the clinic: emerging roles for axon guidance molecules in neurological disorders and injury. Dev. Neurobiol. 67, 1216-1231. 10.1002/dneu.20512 [DOI] [PubMed] [Google Scholar]
- Yaron A., Huang P.-H., Cheng H.-J. and Tessier-Lavigne M. (2005). Differential requirement for Plexin-A3 and -A4 in mediating responses of sensory and sympathetic neurons to distinct class 3 semaphorins. Neuron 45, 513-523. 10.1016/j.neuron.2005.01.013 [DOI] [PubMed] [Google Scholar]
- Yue Y., Su J., Cerretti D. P., Fox G. M., Jing S. and Zhou R. (1999). Selective inhibition of spinal cord neurite outgrowth and cell survival by the Eph family ligand ephrin-A5. J. Neurosci. 19, 10026-10035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y., Adolfs Y., Pijnappel W. W. M. P., Fuller S. J., Van der Schors R. C., Li K. W., Sugden P. H., Smit A. B., Hergovich A. and Pasterkamp R. J. (2011). MICAL-1 is a negative regulator of MST-NDR kinase signaling and apoptosis. Mol. Cell. Biol. 31, 3603-3615. 10.1128/MCB.01389-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmer M., Palmer A., Köhler J. and Klein R. (2003). EphB-ephrinB bi-directional endocytosis terminates adhesion allowing contact mediated repulsion. Nat. Cell Biol. 5, 869-878. 10.1038/ncb1045 [DOI] [PubMed] [Google Scholar]