Abstract
In nature plants are often simultaneously challenged by different biotic and abiotic stresses. Although the mechanisms underlying plant responses against single stress have been studied considerably, plant tolerance mechanisms under combined stress is not understood. Also, the mechanism used to combat independently and sequentially occurring many number of biotic and abiotic stresses has also not systematically studied. From this context, in this study, we attempted to explore the shared response of sunflower plants to many independent stresses by using meta-analysis of publically available transcriptome data and transcript profiling by quantitative PCR. Further, we have also analyzed the possible role of the genes so identified in contributing to combined stress tolerance. Meta-analysis of transcriptomic data from many abiotic and biotic stresses indicated the common representation of oxidative stress responsive genes. Further, menadione-mediated oxidative stress in sunflower seedlings showed similar pattern of changes in the oxidative stress related genes. Based on this a large scale screening of 55 sunflower genotypes was performed under menadione stress and those contrasting in oxidative stress tolerance were identified. Further to confirm the role of genes identified in individual and combined stress tolerance the contrasting genotypes were individually and simultaneously challenged with few abiotic and biotic stresses. The tolerant hybrid showed reduced levels of stress damage both under combined stress and few independent stresses. Transcript profiling of the genes identified from meta-analysis in the tolerant hybrid also indicated that the selected genes were up-regulated under individual and combined stresses. Our results indicate that menadione-based screening can identify genotypes not only tolerant to multiple number of individual biotic and abiotic stresses, but also the combined stresses.
Introduction
Sunflower (Helianthus annuus) is one of the most important oilseed crops worldwide. Sunflower growing regions are characterized by constant occurrence of not only multiple individual biotic and abiotic stresses, but also simultaneous drought, pathogen infection and temperature stresses resulting in substantial loss of crop productivity [1–4]. Adding to this, recent climate changes lead to unpredictable rainfall pattern, temperature and pathogen spread [5–8]. This lead to increased interaction of pathogens with different abiotic stresses in the plant interphase [9–10]. Research in the past had largely focused on understanding plant responses to individual stresses with a limited emphasis on combined stresses [11–15]. Importantly, the available combined stress related literature indicates both shared and unique physiological and molecular responses of plants between combined and individual stresses [9, 16–18]. Therefore, uncovering the shared mechanisms using information from large number of individual stress based studies will be useful for understanding the role of commonly regulated genes under combined and individual stresses.
Transcript profiling data from drought, salt, abscisic acid (ABA), several fungal pathogens, reactive oxygen species (ROS) and cold stress treated sunflower plants are available [19–24]. Such transcriptome profiling from individual stresses such as drought and low temperature has been used to unravel the pathways associated with multiple individual stresses [25, 26]. In spite of large scale transcriptome data available from different individual stresses, a comprehensive effort to identify commonly regulated genes has not yet been made. These shared responses might reveal complex signaling networks and pathways [27, 28] facilitating understanding of both individual and combined stress tolerance mechanisms. From this direction meta-analysis of available data is useful. Recently, meta-analysis of microarray data from rice (Oryza sativa) and Arabidopsis thaliana exposed to drought and bacterial stress identified several commonly regulated stress-responsive genes [29, 30]. In a similar study in rice and A. thaliana plants exposed to drought and bacterial pathogen, ~3100 and 900 differentially expressed genes were identified respectively. About 38.5% and 28.7% differential genes were common to drought and bacterial stresses in rice and A. thaliana, respectively [31]. A large number of commonly regulated genes belonged to ROS mediated signaling and free radical scavenging pathways.
ROS is implicated in complex regulatory networks governing both biotic and abiotic stress responses [32, 33, 34] and also known to play role under combined stresses [35]. The ROS triggered downstream signaling events are also part of shared hormonal responses and metabolic pathways [36, 37]. These signalling networks interact as a part of ‘cross-talk’ and play role in plant adaptation to multiple individual stresses [38–41] and combined stress [10, 11, 17]. Apart from role in signaling pathway, high levels of ROS cause cellular damage due to oxidative stress. The antioxidant defense mechanism is one of the key pathways associated with number of individual and combined stresses [26, 42]. For example, under combined drought and heat stress tolerance, antioxidant enzyme cytosolic ascorbate peroxidase (APX1) plays critical role in H2O2 scavenging [43]. Besides several mutants defective in ROS scavenging enzymes showed increased susceptibility to both biotic and abiotic factors [34, 44, 45]. Catalase-deficient barley showed leaf bleaching [46] and tobacco CAT1 antisense lines showed necrotic lesions linked to the activation of certain pathogen responses [47,48]. In the recent past, many studies have used exogenous ROS generating chemical compounds and ROS scavenging systems as a potential tool to identify plants tolerant to multiple stresses [13, 49]. Genetic variability for oxidative stress tolerance in crop plants has also been explored to identify multiple stress tolerant crops [11, 44, 50, 51]. In our previous work we developed an empherical screening-based approach for identification of individual abiotic stress tolerant crops [50, 52, 53, 54, 55]. The selection criteria for tolerant seedlings during screening involved not only survival under stress after acclimation treatment, but also their high growth rate during recovery.
In the present study the major emphasis was to identify sunflower genotypes contrasting for oxidative stress tolerance using menadione, an oxidative stress inducer and understand the mechanisms involved in multiple individual and combined stress responses (S1 Fig). Initially, meta-analysis was performed on sunflower transcriptome datasets selected from six publically available biotic and abiotic stress experiments to identify commonly regulated genes with most up- and down-regulation. The analysis led to the identification of 526 up-regulated and 4440 down-regulated stress responsive genes which are shared across the stresses. Further RT-qPCR analysis of these genes confirmed their expression pattern observed in microarray. Subsequently expression pattern of the identified genes was studied using the sunflower genotypes having contrasting stress tolerance under multiple individual and combinations of stresses namely, drought, cold, methyl viologen, NaCl and pathogen. Results from this study revealed the plant responses to multiple individual and combined stresses and identified candidate genes for further studies on development of broad-spectrum stress-tolerant sunflower in the future.
Materials and Methods
Plant material and growth conditions
The sunflower genotypes were obtained from different centers of All India Coordinated Research Project (AICRP) for sunflower at University of Agricultural Sciences (UAS), GKVK Bangalore, India. The genetic backgrounds and agronomic characteristics of these 55 lines are described in S1. The sunflower seeds of var. Morden (an open pollinated heterogeneous population) were procured from National Seeds Project, UAS, GKVK, Bangalore, India. Two-day-old seedlings were grown on moist filter paper in Petriplates and incubated at 30°C in seed germination chamber. For the seedling level stress treatment, plants were grown in pots with 2 kg of soil under greenhouse conditions with 10/14 h day/night cycle, 27°C temperature and 80% relative humidity.
Data collection and meta-analysis
The transcriptomic data of individual biotic and abiotic stresses on sunflower was collected from array express database (https://www.ebi.ac.uk/arrayexpress/) (S2 Table). This data (http://www.ebi.ac.uk/arrayexpress/experiments/browse.html?keywords=&organism=Helianthus+annuus&array) was manually curated using Microsoft Excel and control and treatment files were separated. The curated data were used as input files for meta-analysis. Integrative Meta-analysis of Expression data (INMEX) tool [56] was used for meta-analysis of multiple gene-expression datasets for identifying commonly up- and down-regulated genes. Stouffer’s model was used to integrate the data with treatments and controls, thereby commonly expressed (shared) genes among the different stress conditions were identified [57]. This method is used in meta-analysis of data across studies using p-value, sample size and estimated direction of effect for each study. This method can easily execute meta-analyses even when different analytical approaches were used in each individual study [58]. For the data upload, input data (file format.txt or.zip) was arranged in Excel file with gene expression values and corresponding probe ID or gene name in rows and samples or experiments in columns. Each column or treatment was named as per specific treatments. Further the up- and down-regulated gene IDs were converted from Affimetrix to Uniprot IDs. The different dataset were merged together into a mega-dataset (S2 Fig). The functional annotation of the identified genes was derived using Blast2GO tool [59]. Blast2GO identifies the function of a given sequence primarily based on the gene ontology (GO) term. It optimizes the function of a given sequence when compared to homologous sequences considering the similarity and the extent of homology in the selected database (https://www.blast2go.com/).
Individual stress imposition
Oxidative stress in seedlings by menadione
Menadione (2-methyl-1, 4 napthoquinone) sodium bisulfite (Cat No. M2518-100G, Sigma Aldrich), a free radical inducer [50, 60] was used in this study to impose oxidative stress in seedlings 48 h after germination. Menadione is a quinone compound and upon auto oxidation and reduction process, it generates superoxide radicals in the cell. It is suitable for imposing oxidative stress in non-photosynthesizing tissues. Seedlings of 2.5–3 cm length were incubated at a particular concentration of menadione (0.25, 0.5, 1, 2, 3, 4, 5 mM) for 2 h at 30°C under constant shaking. After the treatment, seedlings were washed thoroughly using distilled water. A subset of the control seedlings were transferred to Petriplates on moist filter paper and allowed to recover for 3 d at 30°C. After the recovery period, survival and recovery growth were measured. Another subset of seedlings were allowed to recover for 5 h at 30°C and used to estimate cell death [50, 61, 62]. In all the experiments three replicates were taken and each replicate had 25 seedlings. The seedlings maintained at 30°C throughout the experimental period, were used as absolute controls. Reduction in growth of seedlings was calculated using the following formula.
Ten-day-old sunflower seedlings were treated with 1 mM menadione and after 2 h the tissue from leaf, root and whole seedling were frozen. RNA from these tissues was extracted and cDNA was synthesized using the protocol described under RT-qPCR. Expression of oxidative stress responsive genes namely superoxide dismutase (SOD, accession number AY172569), ascorbate peroxidase (APX, accession number AGU36670), catalase (CAT, accession number L28740), and heat shock protein (HSP17, accession number U96641) were studied using RT-qPCR.
Oxidative stress in leaves by methyl viologen
Methyl viologen (Paraquat dichloride; M2254, Sigma Aldrich), a ROS generating herbicide in chloroplast was used to impose oxidative stress under 1400 μmol m-2s-1 in sunflower leaves. This compound interferes with photosynthetic electron transport chain to produce ROS in photosynthesizing tissues under high light. Oxidative stress was imposed by spraying 5 μM methyl viologen on 7-day-old plants and the tissue was collected after 12 h for different assays (S5 Fig).
Drought stress
Pots were filled with potting mixture of known weight and were irrigated until all the soil macro and micro pores were filled and excess water was drained overnight. Based on water holding capacity for this soil mixture total weight of pot with soil mix for 100% field capacity (FC) was arrived. Drought stress was imposed by gravimetric approach [63]. 10-day-old plants in pots were used for the experiment. Stress was imposed by withholding irrigation and the plants meant for drought stress were maintained at 30–40% FC for one week. At the end of stress period, stress responses were studied in the leaves.
NaCl stress
NaCl 200 mM was dissolved in water and irrigated to the pots having 7-day-old plants. After 5 days of treatment the leaf tissue was frozen for gene expression and biochemical studies (S5 Fig).
Cold stress
Plants (10-day-old) were subjected to cold stress by incubating at 4°C for 2 h and tissue was frozen for further studies (S5 Fig).
Downy mildew pathogen infection
Field grown plants (45-day-old) were naturally allowed to infect with Plasmopara halstedii. The uniformly infected symptomatic leaves were used to rub on the 7-day-old plants grown in pots. At the University of Agricultural Sciences downy mildew infected ‘sick plots’ are maintained for varietal trails [64]. The experiment was carried out during spring (humidity 60–70%, 28°C day and 16°C night temperature). In spring P. halstedii infects sunflower seedlings through germination of overwintered sexual oospores. For the systemic plant colonization by disseminating structures on various plant organs intercellular hyphae play critical role under humid conditions [65]. This pathogen causes seedling damping off, dwarfing of the plant, bleaching of leaves, and visible white sporulation on the lower side of leaves [65]. Disease index was scored after 5 days and tissue was collected for gene expression (S5 Fig). The pathogen infection incidence was assessed by scoring visible white spores and bleaching symptoms. Scoring was done as follows: 0 = no symptoms on the leaves; 1 = <1%; 2 = 1–10%; 3 = 10–25%; 4 = 25–50%; 5 = 50–75%; 6 = > 75% of total leaf area affected. Disease index (DI) was calculated using the following formula [66]:
Six in the formula indicates maximum disease grade.
Combined stress imposition
Ten-day-old plants were used for combined stress imposition. Two types of combined stresses were imposed in this study. One is combination of drought and pathogen, in which plants were initially exposed to drought stress by withholding the water for 3 days. The pathogen was inoculated on the first day of water withholding. The tissue was collected after 3 days of combined stress treatment. Second type of combined stress involved subjecting plants to combination of drought, NaCl, cold, oxidative and pathogen stress as per following procedure. Initially control grown plants were irrigated with 200 mM NaCl and then water was with-held for 3 days. During same period plants were simultaneously exposed to cold stress for 2 h and sprayed with 5 μm methyl viologen and inoculated with pathogen. All these process were carried out within 3 days period and tissue was frozen for further analysis. Minimum of three replicates were maintained for each treatments. The overview of the combined stress experiment is presented in S5 Fig.
Estimation of H2O2
The levels of H202 play critical role in signaling and act as substrate for reactive oxygen species (ROS) [67] and we quantified the stress induced H202 using xylenol orange assay [68]. The xylenol orange reagent was prepared in 50 ml of distilled water containing 1 mL of 50 mM ferrous ammonium sulphate in 2.5 M H2SO4 and 62.5 μL of 125 μM xylenol orange (Sigma chemicals, cat No. 52097-5G, Bangalore) and 0.9019 g sorbitol. The tissue sample was extracted in phosphate buffer (pH 7.5). From this 25 μL supernatant was taken and mixed with 275 μL of xylenol orange reagent. The reaction mix was incubated for 30 min at room temperature and absorbance was measured at 560 nm against xylenol orange reagent only as blank [68]. Standards were prepared by dilution of reagent grade 30% H2O2.
Estimation of Melandialdehyde (MDA) content
Melandialdehyde (MDA) is the end product of lipid peroxidation. MDA levels are indicators of extent of stress impact on plant cell membrane. Leaf tissue (1.0 g) was homogenized in 5 mL of 5% (w/v) trichloroacetic acid and the homogenate was centrifuged at 12,000 g for 15 min at room temperature. The supernatant was mixed with an equal volume of thiobarbituric acid [0.5% in 20% (w/v) trichloroacetic acid], and the mixture was boiled for 25 min at 100°C, followed by centrifugation for 5 min at 7,500 g to get clear solution. Absorbance of the supernatant was measured at 532 nm. MDA content in leaf tissue was calculated using standard graph developed using MDA (Sigma chemicals cat No.63287-1G-F, Bangalore) [69].
Real-time quantitative RT-PCR (RT-qPCR)
Total RNA was extracted according to the protocol described by Datta et al. [70]. First strand cDNA was synthesized by oligo (18 mer dT) primers using Molony murine leukaemia virus reverse transcriptase (MMLV-RT; MBI Fermentas, Hanover, MD, USA) according to manufacturer’s instructions. The cDNA pool was used as a template to perform RT-qPCR analysis. PCR reactions were performed in optical 96-well plates (Applied Biosystems) with an ABI PRISM® 7900 HT sequence detection system, using SYBR® Green to monitor the synthesis of double-stranded DNA. A standard thermal profile with the following conditions was used, 50°C for 2 min, 95°C for 10 min, 40 cycles of 95°C for 15 s, and 60°C for 1 min. Amplicon dissociation curves were recorded after cycle 40 by heating from 60 to 95°C with a ramp speed of 1.9°C min−1. The relative expression levels of the selected genes under a given stress condition was calculated using comparative threshold method by comparing reference control gene [71]. Actin (FJ487620.1) and Ubiquitin (X14333.1) were used as internal controls to normalize RT-qPCR. Details of all primers used in this study are given in S3 Table.
Statistical analysis
The data obtained was analysed using two-way analysis of variance (ANOVA) as per the procedure given by Fischer [72]. Data points with ‘*’ indicate significant differences at P≤0.05.
Results
Identification of commonly regulated genes under abiotic and biotic stresses using meta-analysis of transcriptome data
The sunflower cDNA arrays used in this study were derived from transcriptomic studies available from the public databases. The data from plants exposed to drought, heat, NaCl, oxidative stress, cold stress and an oomycete pathogen, Plasmopara halstedii (causal agent of downy mildew in sunflower) infection were collected to identify stress responsive genes shared among these stresses (S2 Table).
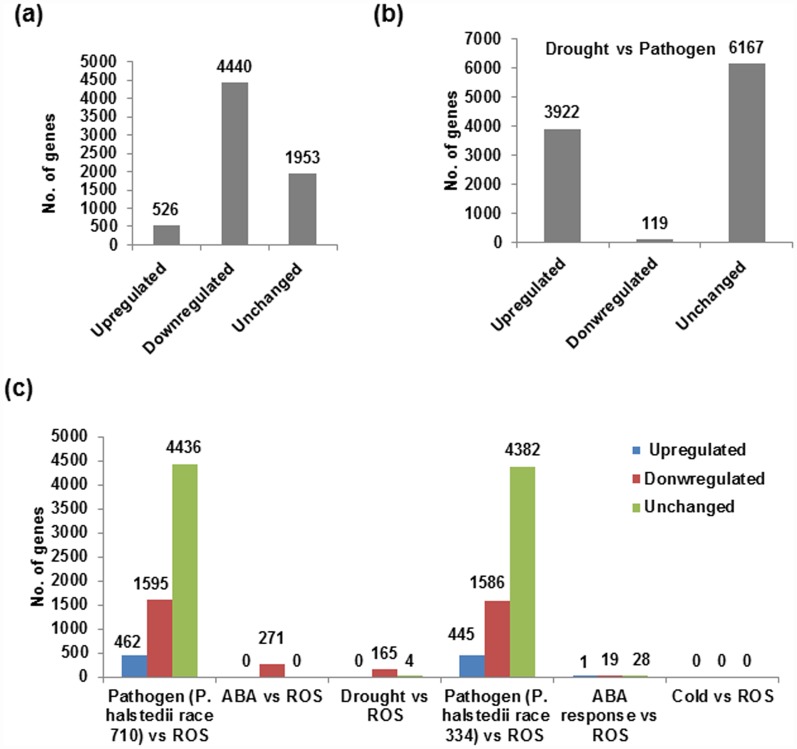
To identify the commonly up or down-regulated genes across the six stresses, meta-analysis was performed. The overall experimental approach followed is detailed in S2 Fig. The analysis showed 526 up-regulated, 4440 down-regulated genes and 1953 genes with similar expression like control (Fig 1). The number of genes upregulated in drought and pathogen was higher than all other stresses. Analysis of differentially expressed genes specifically under drought and pathogen stress showed 3922 up-regulated and 119 down-regulated genes. This data indicated that several genes are shared under multiple individual stresses (Fig 1b). The analysis showed no genes shared between cold and oxidative stress (Fig 1c). On the contrary maximum number of shared genes were found between pathogen stresses (two races of downy mildew pathogen) and oxidative stress. Particularly, 1595 and 1586 genes were down-regulated and 462 and 445 genes were up-regulated in race 710 and race 334, respectively. Further, ABA-ROS and drought-ROS comparison also revealed several commonly regulated genes.
Fig 1. Meta-analysis of sunflower transcriptome data from 6 different experimental datasets.
The raw data were integrated in meta-analysis tool INMEX and differentially expressed genes were identified. The number of differentially expressed genes under all stresses (a), between drought and pathogen stresses (b). Based on individual stress comparisons, commonly up-regulated, down regulated and unchanged genes were identified (c).
The up-regulated genes shared across the stress were classified into different classes based on their molecular function using Blast2GO tool. Large number of genes were found to be involved in protein binding (9.8%), ATP binding function (7.3%), oxidoreductases (4.5%), DNA and RNA binding (4.2 & 4%) in addition to hydrolases, ligases, zinc ion binding, kinase activity, transcription factors and membrane transporters (S6 Fig). The remaining genes had unknown function. Further 29 genes commonly up- or down-regulated in many stresses were directly or indirectly involved in regulation of ROS and oxidative stress tolerance were shortlisted for further analysis (S4 Table). The upregulated genes were subjected to Agrigo tool to map the genes to identify associated pathways and based on biological function, those genes were found to be involved in oxidation reduction process (S7 Fig). Similarly in the large number of down regulated genes were involved in developmental processes, hormone responses, defense responses, transcription, translational events and protein modifications (S8 Fig). The genes involved in plant development, including anatomical structure (23 genes), flower (8 genes), pollen (6 genes) and seed (8 genes) development responsive genes were downregulated. The downregulated genes with response to stimulus include multicellular organismal process (34 genes), responses to stress (34 genes), responses to chemical stimulus (28 genes), abiotic stimulus (19 genes) and defense responsive (14 genes). The genes that were involved in many biosynthetic process (57 genes), macromolecular biological process (82 genes), catabolic process (16 genes), macromolecule modification (25 genes), protein modification (24 genes) and post translational modification (20 genes) were downregulated. Based on these results and literature information [73] we hypothesised that oxidative stress tolerance mechanisms are linked to tolerance of plants to multiple number of individual stresses and also combined stresses.
Menadione induces oxidative stress and broad-spectrum stress effects
Menadione, a compound that produces superoxide radicals, has been used to induce oxidative stress in plants [50, 74, 75]. Seedlings (two day old) of var. Morden were treated with different concentrations of menadione and response was recorded after recovery. Mild concentrations of menadione (0.25 to 2 mM for 2 h) reduced the shoot and root growth compared to that of water treated controls. Root and shoot growth was reduced at concentration higher than 3 mM menadione treatment for 2 h (S9 Fig). Seedling survival and growth after recovery period were reduced as the concentration of menadione and duration of incubation increased. However at high concentrations of menadione (LD50 = 2 mM-3 h or 3 mM-0.5 h), the seedlings abruptly collapsed due to the cell death as quantified by Evans blue staining (S9 Fig; S5 Table).
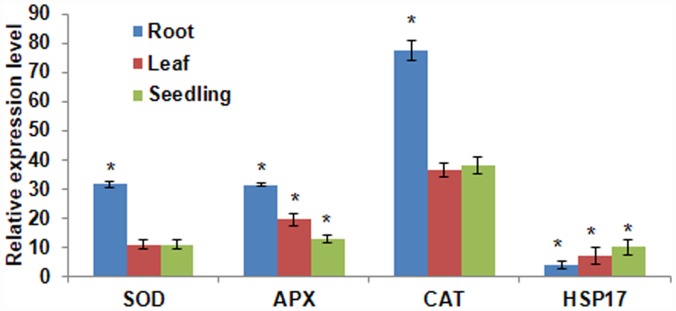
Further the expression of few known stress responsive genes was tested in the menadione stressed seedlings (var. Morden). The expression levels for SOD, APX, CAT and HSP genes were up-regulated compared to non-stress seedlings in roots and leaf. The transcript expression of these genes in seedlings was similar to the expression in roots (Fig 2). This indicated that menadione-induced oxidative stress enhances the expression of genes involved in ROS scavenging and stress adaptation.
Fig 2. Transcript expression profile of oxidative stress responsive genes in menadione treated sunflower seedlings.
Expression pattern of HaSOD, HaAPX, HaCAT, and HaHSP17 were studied using RT-qPCR. Sunflower seedlings were exposed to 1 mM menadione for 2 h. RNA was extracted after the stress period from root, leaf and whole seedling and cDNA was synthesized. ‘*’asterisks indicate a significant difference from the control (two way ANOVA and Duncan’s multiple range test at P<0.05). Error bars indicate standard error of mean of three biological replicates.
Menadione stress screening identifies genotypes contrasting in stress tolerance
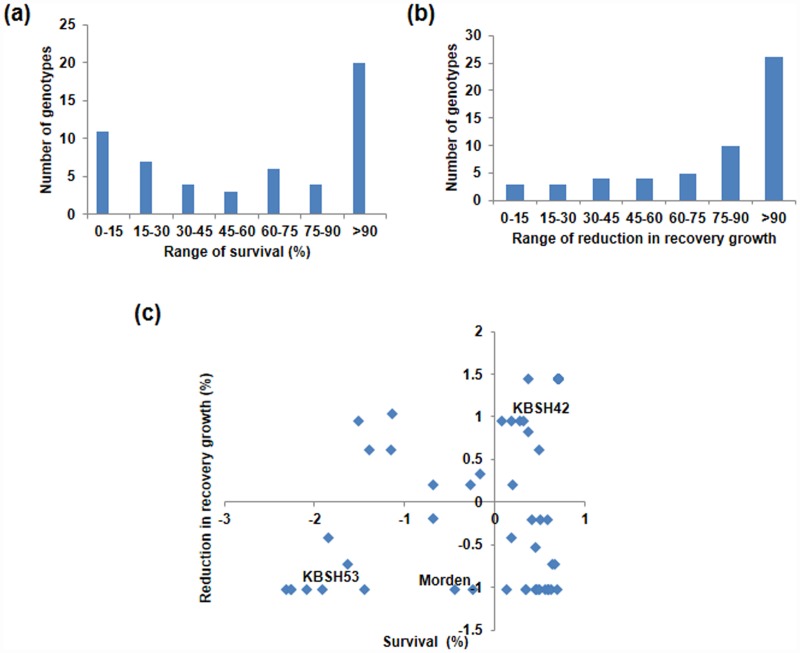
To identify the contrasting genotypes, seedlings of 55 sunflower genotypes were subjected to menadione stress and survival and recovery growth were recorded (Fig 3a & 3b). During the national trials, these genotypes were grown in various geographical locations in India including Akola, Bangalore, Coimbatore, Dholi, Hisar, Ludhiana, Nandyal, Nimpith and Raichur. Owing to the characteristic abiotic stress occurrence in these locations, they were exposed to various stresses during their growth season. Six genotypes were found to be extremely sensitive to oxidative stress based on survival and recovery data (S6 Table). The Z-distribution analysis for both survival and recovery growth was used to identify contrasting genotypes. Based on this, two contrasting genotypes namely KBSH53 and KBSH42 were identified as tolerant and susceptible genotypes, respectively (Fig 3c). These two genotypes were also contrast for resistance to powdery mildew (S1 Table). These two genotypes and another variety Morden identified through temperature induced stress response is moderately tolerant and high yielding, were used to study the effect of multiple individual and combined stresses.
Fig 3. Genetic variability of sunflower genotypes under menadione-induced oxidative stress.
Frequency distribution of genotypes based on survival (%) (a) and, reduction in recovery growth (%) (b) and seedlings were classified using Z- distribution analysis for the 55 genotypes (c). The first and fourth quadrant indicates susceptible and resistant genotypes respectively. The seedlings were exposed to acclimation stress of 1 mM menadione at 30°C and subsequently exposed to a higher concentration of menadione.
Oxidative stress tolerant genotypes exhibits tolerance to multiple individual and combined stresses
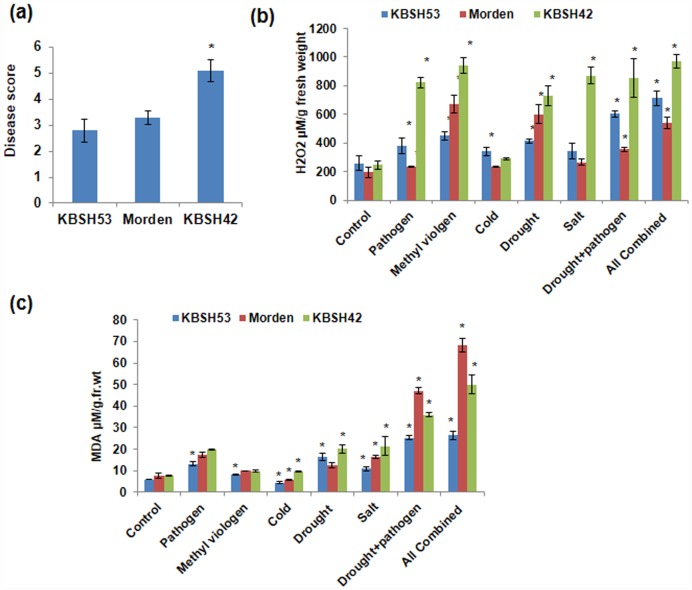
To test tolerant genotypes identified through menadione-based screening for their response to multiple individual and combined abiotic and biotic stresses, these genotypes were subjected to different stresses as shown in the S5 Fig. KBSH53 showed less disease index as compared to KBSH42 (Fig 4a). In these genotypes accumulation of ROS under drought plus pathogen combined stress and all (pathogen, NaCl, drought, cold and methyl viologen) combined stresses is significantly higher than the independent stresses as shown by NBT staining (S10 Fig). KBSH53 showed less NBT staining under pathogen and other treatments and KBSH42 showed higher accumulation of superoxide radicle and higher accumulation of H2O2 as compared to KBSH53 in all stresses (Fig 4b & S10 Fig). Similarly, MDA levels showing lipid peroxidation was high in KBSH42 and low in resistant genotype KBSH53 (Fig 4c). Consistently, susceptible genotype KBSH42 showed higher lipid peroxidation. This confirms that the genotypes identified through menadione screening showed response similar to that exhibited under oxidative stress under multiple individual and combined drought and pathogen stress. Taken together, large scale screening of sunflower genotypes using menadione identified contrasting genotypes KBSH42 and KBSH53 for individual and combined stress tolerance (Fig 4).
Fig 4. Individual and combined stress response of the three genotypes varying in oxidative stress tolerance.
Disease score on plants exposed to P. halstedii (a) 7-day-old sunflower plants were exposed to pathogen spores for 5 days and. Score was assigned from 1–10 based on low to high infection. H2O2 levels were assessed from leaves of different biotic and abiotic stressed plants (b). The leaf tissue was ground in PBS buffer and aliquots were used for estimation of H2O2 levels by using a modified ferrous oxidation-xylenol orange (FOX) assay. MDA levels in stressed plants (c) was quantified by TBARS assay to study the extent of damage on lipids. ‘*’ indicate a significant difference from the control (Student’s t test, P<0.05). Error bars indicate standard error of mean. Data were pooled from two independent experiments representing three biological replicates.
Gene expression analysis under individual and combined stress explains the molecular basis for susceptibility and resistance of genotypes
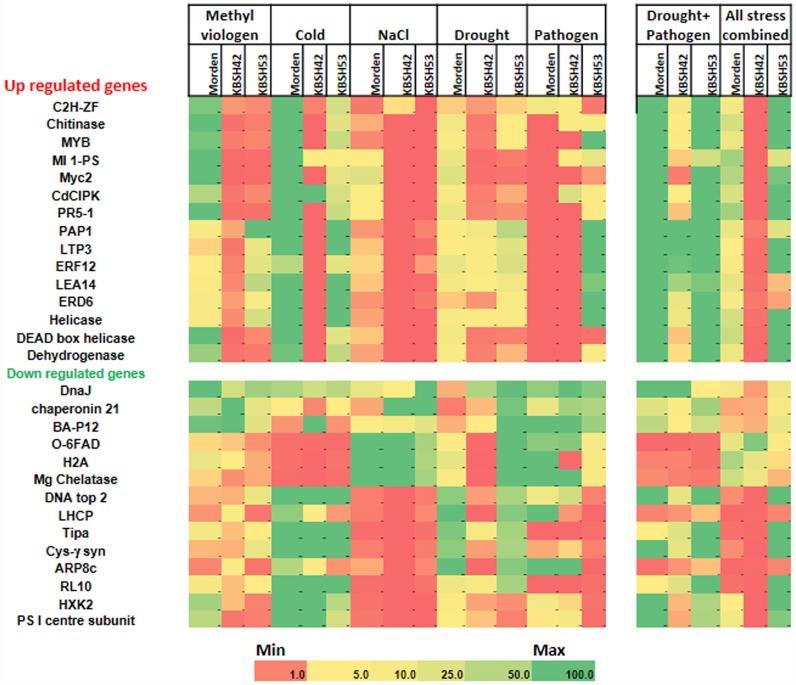
To study the gene expression pattern in contrasting genotypes under individual and combined stresses, a total of 15 up- and 14 down-regulated genes that were selected from meta-analysis were used for RT-qPCR (S4 Table). The transcript analysis showed that many genes identified by meta-analysis were up-regulated in the tolerant genotype KBSH53 in all combined stresses and drought plus pathogen stress. The transcript levels were higher for genes encoding DNA topisomerase, aquoglyceroporin, cystathionine γ-synthase, envelope glycoprotein RL10, hexokinase, and photosystem I reaction center proteins under both type (drought plus pathogen, and pathogen, NaCl, drought, cold and methyl viologen all together) of combined stresses. However, the majority of the transcripts upregulated either one fold or less in individual stresses. Many genes upregulated in meta-analysis data also upregulated in the drought plus pathogen combined stress (Fig 5).
Fig 5. Transcript profiling of sunflower genotypes varying in stress tolerance under individual and combined stresses.
Morden, KBSH42 and KBSH53 plants were subjected to individual stresses namely, methyl viologen-induced oxidative stress, cold, salt, drought and pathogen. Another batch of plants were subjected to two type of combined stresses, namely drought and pathogen and all stresses combined. The stress protocol is described in S5 Fig. From these stressed plants total RNA was isolated and cDNA was prepared and used for RT-qPCR. Three replicates were maintained. The expression was normalized to HaActin and fold change was calculated against the control samples. Two way ANOVA and Duncan’s multiple range test at P<0.05 was carried out using three biological replicates.
In the susceptible genotype KBSH42 several genes showed reduced transcript levels under individual and combined drought plus pathogen stresses. Only Microsomal oleic acid desaturase (O-6FAD) gene showed up-regulation in all combined stress. The DnaJ gene found to be down-regulated in meta-analysis results showed up regulation under combined drought plus pathogen stresses (Fig 5). Similarly, the genes encoding acid phosphatase 1, lipid transfer protein isoform 3, ethylene responsive transcription factor 3 and late embryogenesis abundant 4 were up-regulated in combined drought plus pathogen stress which showed similar trend of transcript levels as predicted by meta-analysis. In nutshell, the transcript profiling of selected genes both under individual and combined stresses showed that the transcriptome response in KBSH53 is different from KBSH42 (Fig 5).
Discussion
Meta-analysis is a useful tool to identify shared genes among multiple individual and combined stresses
Understanding the shared mechanisms contributing to two or more individually or simultaneously occurring stresses is important to improve crop productivity under foreseeable complex stress situations. But, adaptation of plants to such individual and combined stress is imparted through a complex, yet to be fully understood mechanisms. To dissect molecular mechanism behind multiple individual stress and combined stress tolerance in sunflower, meta-analysis approach was employed [29] using publically available transcriptome datasets from individual stress studies. The analysis revealed 526 genes up-regulated and 4440 genes down-regulated among P. halstedii infection, ROS treatment, drought, ABA treatment and cold stresses. Most of the commonly up- or down- regulated genes identified from meta-analysis showed similar expression pattern under all combined stresses. Between ROS and cold stress response no commonly regulated genes were found. A simple explanation is either the genes responsive to cold and ROS are independent or the levels of stress imposed was not sufficient to trigger the shared responsive genes. Role of several of these genes under multiple individual and combined stresses are largely unknown (S4 Table). The genes encoding C2H2 zinc finger, MYB, MYC/bHLH and ethylene responsive factor (ERF) belong to specific family of transcription factors. These transcription factors are known to regulate several downstream functional genes in response to different environmental stresses [9, 11]. Another interesting class of genes found were those encoding H2A, DNA topoisomerase 2, DNaJ and DEAD box helicases. Apart from these, genes involved in histone relaxation, DNA repair and RNA secondary structure removal under stress were also found [76, 77, 78, 79]. Further the downstream genes encoding chitinase, pathogen resistance 5 (PR5), autophagy related protein and myo-inositol-1-phosphate synthase that are involved in plant defense against pathogens were identified along with several chlorophyll and light harvesting complex protein encoding genes. A similar group of genes identified through microarray profiling of sunflower leaves exposed to cold and NaCl stress showed dynamic changes in transcript levels of transcription factors, genes related to translation, protein degradation/folding and ROS production or scavenging mechanisms [80]. Similarly comparative gene expression analysis under highlight (HL), high temperature (HT) and combined HL and HT stresses in sunflower leaves and seeds revealed differential expression of 89, 113 and 186 genes, respectively [81]. Meta-analysis of 6 experimental datasets under different stresses revealed several genes belonging to ATP-, DNA-, RNA-, protein- binding, hydrolase, ligase, oxidoreductase, serine threonine kinase, transcription factor, zinc ion binding and transporter activity. The data suggests that meta-analysis approach can be potentially employed to identify shared stress responsive genes, which can reveal the mechanism of combined and multiple stress tolerance.
Owing to the complexity involved in handling all combined stresses, we further focused on pathogen and drought combination for detailed systematic confirmation of the meta-analysis results and to dissect the shared mechanism between individual and combined stresses. Interestingly the meta-analysis showed that a large number of commonly regulated genes belong to the ROS-responsive or oxidative stress scavenging system (S7 Fig). ROS scavenging proteins are shown to act as early sensors to prevent potential oxidative stress damage [82]. The response of 187 nuclear encoded ROS responsive genes and 1880 transcription factors showed rapid and coordinated expression under H2O2 [83]. This prompted us to further examine the role of ROS pathway related genes using RT-qPCR under combined stress. Under combined drought and pathogen stress, the genes identified as up-regulated by meta-analysis consistently showed higher transcript levels in var. Morden. Interestingly most of the genes identified from the analysis were also induced in sunflower seedlings treated with menadione-induced oxidative stress (Fig 5). Since these genes were also separately confirmed for their up-regulation under combined drought and pathogen stress, we speculated the strong overlap in some gene expression between the methyl viologen or menadione-induced oxidative stress and the combined stress. This overlap can be attributed as shared response of plants among the combined and oxidative stresses. Meta-analysis of drought, bacterial stress response in rice and A. thaliana revealed 38.5% (1214) and 28.7% (272) differentially expressed genes (DEGs) respectively and a majority of these showed conserved expression status in both stresses (30). These studies suggests that several genes act as part of shared response between combined and individual stresses.
Tolerant genotypes identified through menadione-based screen showed multiple individual and combined stress tolerance
We hypothesized that menadione-based screening of genetically diverse sunflower genotypes could identify tolerant and susceptible groups not only for oxidative stress tolerance, but also for tolerance to combined stresses (S5 Fig). Specifically, menadione-based screen has been earlier demonstrated as one of the methods suitable for screening and identification of contrasting stress tolerant genotypes in sunflower [50]. Moreover, owing to highly cross- pollinated nature of sunflower, the selected 55 genotypes are expected to have genetic variability for multiple individual and combined stress tolerance. The pool of genotypes used in this study represent genetic background with tolerance to various abiotic stresses, namely drought, temperature extremes and salinity apart from superior agronomical characteristics (S1 Table). Our screening process identified KBSH53 and KBSH42 as tolerant and susceptible genotypes, respectively. Interestingly, the susceptible genotype also showed susceptibility to combined drought and pathogen stress (Fig 4). Consistently, the resistant genotype showed improved performance under individual drought and pathogen stress and also combined stress. Taken together, these evidences support our hypothesis and show that the approach used in this study can identify not only genes responsible for multiple individual stress tolerance, but also for the combined stress. The usefulness of data from individual stress studies to identify genes for combined stress tolerance is possible because of crosstalk between many signalling pathways during multiple stresses [32, 33, 84, 85].
Meta-analysis identified genes in multiple individual and combined stress tolerance
Meta-analysis of sunflower transcriptome datasets revealed 526 up and 4440 down-regulated genes in all combined stresses. RT-qPCR results for selected 29 genes in the tolerant genotype KBSH53 revealed candidate genes for combined stress tolerance. Amongst these genes, 17 were induced under all combined stress (pathogen, NaCl, drought, cold and methyl viologen stress) in tolerant genotype KBSH53, but susceptible genotype did not show transcript changes over control. In general, under any of the independent stresses the identified genes did not show significant fold change in both up- or down-regulated gene category. Overall the data suggest that under combined or multiple stresses, the meta-analysis can identify candidate shared stress responsive genes.
Cross-talk and role for identified genes
Several genes showed up-regulation in the tolerant genotype KBSH53 including increased transcript levels of transcription factors C2H-ZF, MYB, MYC2, ERF12 and ERD6. Overexpression of some of these transcription factors resulted in multiple stress tolerance [86]. Since the literature information on validation is scarce, many other genes identified in the meta-analysis could not be verified for their functional relevance. However, we subsequently review few other evidences that support correlation between meta-analysis identified genes and their validation in literature. The induction of transcription factors under combined stress has been reported in a previous study, wherein it was observed that combined heat and drought stress lead to upregulation of WRKYs and ERFs [87]. One of our previous study also showed the overexpression of AtWRKY28 in A. thaliana enhances drought and NaCl stress tolerance [88, 89]. The tolerant genotype also showed up-regulation of transcriptional regulators such as DNA topoisomerase 6, DEAD box helicases, ribosomal protein L10 (RPL10), ROS detoxification enzyme encoding genes like dehydrogenases, genes involved in protein stability such as chaperonins, late embryogenesis abundant 14 (LEA14), myo inositol phosphate synthase, calcium induced protein kinase (CIPK), lipid transport proteins and histidine kinases. It is evident that under combined stress, receptor like kinases, protein kinases (MAPK and CIPK), small GTP- binding proteins and membrane intrinsic proteins (MIP) are up-regulated [90]. Further, PR and chitinases also showed up-regulation in tolerant genotype and these genes are independently known to impart resistance to different pathogens [84]. These genes up-regulated under combined stress have potential to improve stress tolerance through complex network mode of mechanisms. Taken together, our data demonstrated that the meta-analysis can efficiently identify the potential candidate genes for combined stress tolerance.
In conclusion, salient features of this study include, one, menadione-based screening can be used as means to generate oxidative stress and explore genetic variability in agronomically superior genotypes for oxidative stress tolerance. Second, meta-analysis can be potentially employed to identify candidate genes for multiple and combined stress tolerance. Third, identified genes are the potential candidates for genetic engineering of plants to combat multiple environmental stresses.
Supporting Information
(PPTX)
(PPTX)
(PPT)
(PPTX)
(PPTX)
(PPTX)
(PDF)
(PDF)
(PPTX)
(PPTX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
Authors thank Drs. Y. G. Shadakshari and K. Karuna, AICRP (sunflower) at GKVK for providing seeds and help with pathogen assay. We also thank Dr. Prachi Pandey for critical reading of the manuscript.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
Combined stress tolerance projects at MS-K lab are supported by National Institute of Plant Genome Research (NIPGR) core funding and DBT-Ramalingaswami re-entry fellowship grant (BT/RLF/re-entry/23/2012). MUK lab is supported by Indian Council of Agricultural Research niche area of excellence program (F. No. 10-(6)/2005 EPD & F.No.10 (15) 2012 EPD). VSR is currently Fulbright-Nehru postdoctoral fellow under United States-India Educational Foundation (USIEF). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Wastemack C, Hause B. Jasmonates and octadecanoids—signals in plant stress responses and development. Progress in Nucleic Acid Research and Molecular Biology 2002;72: 165–220. [DOI] [PubMed] [Google Scholar]
- 2.Albuquerque MC, de Figueiredo, De Carvalho NM. Effect of type of environmental stress on the emergence of sunflower (Helianthus annuus L.), Soybean (Glycine max (L.) Merril) and maize (Zea mays L.) seeds with different levels of vigor. Seed Science and Technology 2003; 31: 465–469. [Google Scholar]
- 3.Rowe HC, Kliebenstein DJ. Complex genetics control natural variation in Arabidopsis thaliana resistance to Botrytis cinerea. Genetics 2008; 180: 2237–2250. 10.1534/genetics.108.091439 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ali D, Naderidarbaghshahi M, Rezaei A, Majdnasiri B. Effect of water deficiency stress on yield and yield component of sunflower cultivars in Isfahan. International Journal of Farming and Allied Sciences 2013; 2: 1319–1324. [Google Scholar]
- 5.Porter JR, Semenov MA. Crop responses to climatic variation. Philosophical Transactions of the Royal Society B: Biological Sciences. 2005; 360: 2021–2035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bale JS, Masters GJ, Hodkinson ID, Awmack C, Bezemer TM, Brown VK, et al. Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Global Change Biology 2002; 8: 1–16. [Google Scholar]
- 7.Madgwick JW, West JS, White RP, Semenov MA, Townsend JA, Turner JA, et al. Impacts of climate change on wheat anthesis and Fusarium ear blight in the UK. European Journal of Plant Pathology 2011; 130: 117–131. [Google Scholar]
- 8.Nicol JM, Turner SJ, Coyne DL, den Nijs L, Hockland S, Tahna Maafi Z. Current nematode threats to world agriculture In: Jones J, Gheysen G, Fenoll C, eds. Genomics and molecular genetics of plant–nematode interactions. London: Springer; 2011. 21–44. [Google Scholar]
- 9.Ramegowda V, Senthil-Kumar M. The interactive effects of simultaneous biotic and abiotic stresses on plants: Mechanistic understanding from drought and pathogen combination. Journal of Plant Physiology 2015; 176: 47–54. 10.1016/j.jplph.2014.11.008 [DOI] [PubMed] [Google Scholar]
- 10.Suzuki N, Rivero RM, Shulaev V, Blumwald E, Mittler R. Abiotic and biotic stress combinations. New Phytologist 2014; 203: 32–43. 10.1111/nph.12797 [DOI] [PubMed] [Google Scholar]
- 11.Atkinson NJ, Urwin PE. The interaction of plant biotic and abiotic stresses: from genes to the field. Journal of Experimental Botany 2012; 63: 3523–3543. 10.1093/jxb/ers100 [DOI] [PubMed] [Google Scholar]
- 12.Ahuja I, de Vos RC, Bones AM, Hall RD. Plant molecular stress responses face climate change. Trends in Plant Science 2010; 15: 664–674. 10.1016/j.tplants.2010.08.002 [DOI] [PubMed] [Google Scholar]
- 13.Mittler R, Blumwald E. Genetic engineering for modern agriculture: challenges and perspectives. Annual Review of Plant Biology 2010; 61: 443–462. 10.1146/annurev-arplant-042809-112116 [DOI] [PubMed] [Google Scholar]
- 14.Mittler R, Finka A, Goloubinoff P. How do plants feel the heat? Trends in Biochemical Sciences 2012; 37: 118–125. 10.1016/j.tibs.2011.11.007 [DOI] [PubMed] [Google Scholar]
- 15.Li J, Lin X, Chen A, Peterson T, Ma K, Bertzky M, et al. Global priority conservation areas in the face of 21st century climate change. PLoS ONE 2013; 8: e54839 10.1371/journal.pone.0054839 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Atkinson NJ, Lilley CJ, Urwin PE. Identification of genes involved in the response of Arabidopsis to simultaneous biotic and abiotic stresses. Plant Physiology 2013; 162: 2028–2041. 10.1104/pp.113.222372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Prasch CM, Sonnewald U. Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiology 2013; 162: 1849–1866. 10.1104/pp.113.221044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rasmussen S, Barah P, Suarez-Rodriguez MC, Bressendorff S, Friis P, Costantino P, et al. Transcriptome responses to combinations of stresses in Arabidopsis. Plant Physiology 2013; 161: 1783–1794. 10.1104/pp.112.210773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lai Z, Livingstone K, Zou Y, Church SA, Knapp SJ, Andrews J, et al. Identification and mapping of SNPs from ESTs in sunflower. Theoretical and Applied Genetics 2005; 111: 1532–1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu HT, Li B, Shang ZL, Li XZ, Mu RL, Sun DY, et al. Calmodulin is involved in heat shock signal transduction in wheat. Plant Physiology 2003; 132: 1186–1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jocić S, Cvejić S, Hladni N, Miladinović D, Miklič V. Development of sunflower genotypes resistant to downy mildew. Helia 2010; 33: 173–180. [Google Scholar]
- 22.Oracz K, El-Maarouf-Bouteau H, Kranner I, Bogatek R, Corbineau F, Bailly C. The mechanisms involved in seed dormancy alleviation by hydrogen cyanide unravel the role of reactive oxygen species as key factors of cellular signaling during germination. Plant Physiology 2009; 150: 494–505. 10.1104/pp.109.138107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alignan M, Hewezi T, Petitprez M, Dechamp-Guillaume G, Gentzbittel L. A cDNA microarray approach to decipher sunflower (Helianthus annuus) responses to the necrotrophic fungus Phoma macdonaldii. New Phytologist 2006; 170: 523–536. [DOI] [PubMed] [Google Scholar]
- 24.Rengel D, Arribat S, Maury P, Martin-Magniette M-L, Hourlier T, Laporte M, et al. A gene-phenotype network based on genetic variability for drought responses reveals key physiological processes in controlled and natural environments. PLoS ONE 2012; 7: e45249 10.1371/journal.pone.0045249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Roche J, Hewezi T, Bouniols A, Gentzbittel L. Transcriptional profiles of primary metabolism and signal transduction-related genes in response to water stress in field-grown sunflower genotypes using a thematic cDNA microarray. Planta 2007; 226: 601–617. [DOI] [PubMed] [Google Scholar]
- 26.Hewezi T, Leger M, El Kayal W, Gentzbittel L. Transcriptional profiling of sunflower plants growing under low temperatures reveals an extensive down-regulation of gene expression associated with chilling sensitivity. Journal of Experimental Botany 2006; 57: 3109–3122. [DOI] [PubMed] [Google Scholar]
- 27.Ma S, Gong Q, Bohnert HJ. An Arabidopsis gene network based on the graphical Gaussian model. Genome Research 2007; 17: 1614–1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Narsai R, Wang C, Chen J, Wu J, Shou H, Whelan J. Antagonistic, overlapping and distinct responses to biotic stress in rice (Oryza sativa) and interactions with abiotic stress. BMC Genomics 2013; 14: 93 10.1186/1471-2164-14-93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shaik R, Ramakrishna W. Genes and co-expression modules common to drought and bacterial stress responses in Arabidopsis and rice. PLoS ONE 2013; 8: e77261 10.1371/journal.pone.0077261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shaik R, Ramakrishna W. Machine learning approaches distinguish multiple stress conditions using stress-responsive genes and identify candidate genes for broad resistance in rice. Plant Physiology 2014; 164: 481–495. 10.1104/pp.113.225862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen W, Provart NJ, Glazebrook J, Katagiri F, Chang HS, Eulgem T, et al. Expression profile matrix of Arabidopsis transcription factor genes suggests their putative functions in response to environmental stresses. Plant Cell 2002; 14: 559–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kreps JA, Wu Y, Chang HS, Zhu T, Wang X, Harper JF. Transcriptome changes for Arabidopsis in response to salt, osmotic, and cold stress. Plant Physiology 2002; 130: 2129–2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kimura M, Yamamotoy Y, Seki M, Sakurai T, Sato M, Abe T, et al. Identification of Arabidopsis genes regulated by high light-stress using cDNA microarray. Photochemistry and Photobiology 2003; 77: 226–233. [DOI] [PubMed] [Google Scholar]
- 34.Sharma R, De Vleesschauwer D, Sharma MK, Ronald PC. Recent advances in dissecting stress-regulatory crosstalk in rice. Molecular Plant. 2013; 6: 250–260. 10.1093/mp/sss147 [DOI] [PubMed] [Google Scholar]
- 35.Ramegowda V, Senthil-Kumar M, Udayakumar M, Mysore KS. A high-throughput virus-induced gene silencing protocol identifies genes involved in multi-stress tolerance. BMC Plant Biology 2013; 13: 193 10.1186/1471-2229-13-193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Singh DP, Moore CA, Gilliland A, Carr JP. Activation of multiple antiviral defence mechanisms by salicylic acid. Molecular Plant Pathology 2004; 5: 57–63. 10.1111/j.1364-3703.2004.00203.x [DOI] [PubMed] [Google Scholar]
- 37.Fujita M, Fujita Y, Noutoshi Y, Takahashi F, Narusaka Y, Yamaguchi-Shinozaki K, et al. Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Current Opinion in Plant Biology 2006; 9: 436–442. [DOI] [PubMed] [Google Scholar]
- 38.Cutler SR, Rodriguez PL, Finkelstein RR, Abrams SR. Abscisic acid: emergence of a core signaling network. Annual Review of Plant Biology 2010; 61: 651–679. 10.1146/annurev-arplant-042809-112122 [DOI] [PubMed] [Google Scholar]
- 39.Hey SJ, Byrne E, Halford NG. The interface between metabolic and stress signaling. Annals of Botany 2010; 105: 197–203. 10.1093/aob/mcp285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pastori GM, Foyer CH. Common components, networks, and pathways of cross-tolerance to stress. The central role of ‘‘redox” and abscisic acid-mediated controls. Plant Physiology 2002; 129: 460–468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wilkinson S, Davies WJ. Drought, ozone, ABA and ethylene: new insights from cell to plant to community. Plant, Cell & Environment 2010; 33: 510–525. [DOI] [PubMed] [Google Scholar]
- 42.Estavillo GM, Crisp PA, Pornsiriwong W, Wirtz M, Collinge D, Carrie C, et al. Evidence for a SAL1-PAP chloroplast retrograde pathway that functions in drought and high light signaling in Arabidopsis. Plant Cell 2011; 23: 3992–4012. 10.1105/tpc.111.091033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Koussevitzky S, Suzuki N, Huntington S, Armijo L, Sha W, et al. Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. The Journal of Biological Chemistry. 2008; 283: 34197–34203. 10.1074/jbc.M806337200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gill SS, Tuteja N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiology and Biochemistry 2010; 48: 909–930. 10.1016/j.plaphy.2010.08.016 [DOI] [PubMed] [Google Scholar]
- 45.Landoni M, De Francesco A, Bellatti S, Delledonne M, Ferrarini A, Venturini L, et al. A mutation in the FZL gene of Arabidopsis causing alteration in chloroplast morphology results in a lesion mimic phenotype. Journal of Experimental Botany 2013; 64: 4313–4328. 10.1093/jxb/ert237 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kendall AC, Keys AJ, Turner JC, Lea PJ, Miflin BJ. The isolation and characterisation of a catalase-deficient mutant of barley (Hordeum vulgare L.). Planta 1983; 159: 505–511. 10.1007/BF00409139 [DOI] [PubMed] [Google Scholar]
- 47.Chamnongpol S, Willekens H, Langebartels C, Van Montagu M, Inzé D, Van Camp W. Transgenic tobacco with a reduced catalase activity develops necrotic lesions and induces pathogenesis-related expression under high light. Plant Journal 1996; 10: 491–503. [Google Scholar]
- 48.Takahashi H, Chen Z, Du H, Liu Y, Klessig DF. Development of necrosis and activation of disease resistance in transgenic tobacco plants with severely reduced catalase levels. Plant Journal 1997; 11: 993–1005. [DOI] [PubMed] [Google Scholar]
- 49.Garg N, Manchanda G. ROS generation in plants: boon or bane? Plant Bio systems 2009; 143: 8e96. [Google Scholar]
- 50.Senthil-Kumar M, Srikanthbabu V, Raju BM, Ganeshkumar, Shivaprakash, Udayakumar M. Screening of inbred lines to develop a thermotolerant sunflower hybrid using the temperature induction response (TIR) technique: a novel approach by exploiting residual variability. Journal of Experimental Botany 2003; 54: 2569–2578. [DOI] [PubMed] [Google Scholar]
- 51.Senthil-Kumar M, Kumar G, Srikanthbabu V, Udayakumar M. Assessment of variability in acquired thermotolerance: Potential option to study genotypic response and the relevance of stress genes. Journal of Plant Physiology 2007; 164: 111–125. [DOI] [PubMed] [Google Scholar]
- 52.Jayaprakash TL, Ramamohan G, Krishna Prasad BT, Kumar G, Prasad TG, Mathew MK, et al. Genotypic variability in differential expression of Lea2 and Lea3 genes and proteins in response to salinity stress in finger millet (Eleusine coracona Gaertn) and rice (Oryza sativa L.) seedlings. Annals of Botany 1998; 82: 513–522. [Google Scholar]
- 53.Kumar G, Krishnaprasad BT, Savitha M, Gopalakrishna R, Mukhopadhyay K, Ramamohan G, et al. Enhanced expression of heat shock proteins in thermotolerant lines of sunflower and their progenies selected on the basis of temperature induction response (TIR). Theoretical and Applied Genetics 1999; 99: 359–367. [Google Scholar]
- 54.Srikanthbabu V, Kumar G, Krishnaprasad BT, Gopalakrishna R, Savitha M, Udayakumar M. Identification of pea genotypes with enhanced thermo tolerance using temperature induction response (TIR) technique. Journal of Plant Physiology 2002; 159: 535–545. [Google Scholar]
- 55.Uma S, Prasad TG, Udayakumar M. Genetic variability in recovery growth and synthesis of stress proteins in response to polyethylene glycol and salt stress in finger millet. Annals of Botany 1995;76: 43–49. [Google Scholar]
- 56.Xia J, Fjell CD, Mayer ML, Pena OM, Wishart DS, Hancock RE. INMEX—a web-based tool for integrative meta-analysis of expression data. Nucleic Acids Research 2013; 41: W63–W70. 10.1093/nar/gkt338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chang LC, Lin HM, Sibille E, Tseng GC. Meta-analysis methods for combining multiple expression profiles: comparisons, statistical characterization and an application guideline. BMC Bioinformatics 2013; 14: 368 10.1186/1471-2105-14-368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kim SC, Lee SJ, Lee WJ, Yum YN, Kim JH, Sohn S, et al. Stouffer’s Test in a large scale simultaneous hypothesis testing. PLoS ONE. 2013; 8: e63290 10.1371/journal.pone.0063290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gotz S, Garcia-Gomez JM, Terol J, Williams TD, Nagaraj SH, Nueda MJ, et al. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Research 2008; 36: 3420–3435 10.1093/nar/gkn176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Reichheld JP, Vernou XT, Lardon F and Montagu MV. Specific checkpoints regulate plant cell cycle progression in response to oxidative stress. Plant Journal 1999; 17: 647–656. [Google Scholar]
- 61.Cecchini NM, Monteoliva MI, Alvarez ME. Proline dehydrogenase contributes to pathogen defense in Arabidopsis. Plant Physiology 2011; 155: 1947–1959. 10.1104/pp.110.167163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Turner TG, Novacky A. The quantitative relation between plant and bacterial cells involved in the hypersensitive reaction. Phytopathology Journal 1974; 64: 885–890. [Google Scholar]
- 63.Hema R, Vemanna RS, Sreeramulu S, Reddy CP, Senthil-Kumar M, Udayakumar M. Stable expression of mtlD gene imparts multiple stress tolerance in finger millet. PLoS ONE 2014; 9: e99110 10.1371/journal.pone.0099110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kumar BRP, Mahesh J, Kulkarni BN, Rao V, Chandrika K, Gowda BVR, et al. Biochemical and histological changes associated with downy mildew (Plasmopara halstedii (Farl.) Berl.and deToni) infection in sunflower (Helianthus annuus L.). HELIA, 2000; 23; 1–18. [Google Scholar]
- 65.Gascuel Q, Martinez Y, Boniface M-C, Vear F, Pichon M, Godiard L. The sunflower downy mildew pathogen Plasmopara halstedii. Molecular Plant Pathology 2014; 16: 109–122. 10.1111/mpp.12164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lyngdoh IE, Channakrishnaiah KM, Shashidhar HE, Gopalakrishna T, Shivanna MB, Prasad DT. PCR analysis of the radiation-induced sunflower mutants for identification of molecular markers associated with Alternaria helianthi resistance. Journal of Plant Biology 2004; 31: 21–27. [Google Scholar]
- 67.Jajic I, Sarna T, Strzalka K. Senescence, stress, and reactive oxygen species. Plants 2015; 4: 393–411. 10.3390/plants4030393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gay C, Collins J, Gebicki JM. Hydroperoxide assay with the ferric-xylenol orange complex. Analytical Biochemistry. 1999; 273: 149–155. [DOI] [PubMed] [Google Scholar]
- 69.Loreto F. Velikova V. Isoprene produced by leaves protects the photosynthetic apparatus against ozone damage, quenches ozone products, and reduces lipid peroxidation of cellular membranes. Plant Physiology 2001; 127: 1781–1787. [PMC free article] [PubMed] [Google Scholar]
- 70.Datta K, Schmidt A, Marcus A. Characterization of two soybean repetitive proline-rich proteins and a cognate cDNA from geminated axes. Plant Cell 1989; 1: 945–952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schmittgen TD, Lee EJ, Jiang J, Sarkar A, Yang L, Elton TS, et al. Real-time PCR quantification of precursor and mature microRNA. Methods 2008; 44: 31–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fisher RA (1960). The design of experiments, 7th Edn Edinburgh: Oliver and Boyd. [Google Scholar]
- 73.Kumar D, Datta R, Hazra S, Sultana A, Mukhopadhyay R, Chattopadhyay R. Transcriptomic profiling of Arabidopsis thaliana mutant pad2.1 in response to combined cold and osmotic stress. PLoS ONE 2015; 10: e0122690 10.1371/journal.pone.0122690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Borges AVS, Djenidi G, Lacroix J, Théate B, Delille, Frankignoulle M. Atmospheric CO2 flux from mangrove surrounding waters. Geophysical Research Letters 2003; 30: 1558. [Google Scholar]
- 75.Prasad TK, Anderson MD, Stewart CR. Acclimation, hydrogen peroxide and abscisic acid protect mitochondria against irreversible chilling injury in maize seedlings. Plant Physiology 1994; 105: 619–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Breuer C, Morohashi K, Kawamura A, Takahashi N, Ishida T, Umeda M, et al. Transcriptional repression of the APC/C activator CCS52A1 promotes active termination of cell growth. EMBO Journal 2012; 31: 4488–4501. 10.1038/emboj.2012.294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yi H, Mysore KS, Gelvin SB. Expression of the Arabidopsis histone H2A-1 gene correlates with susceptibility to Agrobacterium transformation. Plant Journal 2002; 32: 285–298. [DOI] [PubMed] [Google Scholar]
- 78.Linder P, Owttrim GW. Plant RNA helicases: linking aberrant and silencing RNA. Trends in Plant Science. 2009; 4: 344–352. [DOI] [PubMed] [Google Scholar]
- 79.Fernandez P, Rienzo JA, Fernandez L, Hopp HE, Paniego N, Heinz RA. Transcriptome identification of candidate genes involved in sunflower responses to chilling and salt stresses based on cDNA microarray analysis. BMC Plant Biology 2007; 8:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hewezi T,LÉGER M and Gentzbittel L. A comprehensive analysis of the combined effects of high light and high temperature stresses on gene expression in sunflower. Annals of Botany 2008; 102: 127–140. 10.1093/aob/mcn071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Fernández-Ocaña A, Chaki M, Luque F, Gómez-Rodríguez MV, et al. , Functional analysis of superoxide dismutases (SODs) in sunflower under biotic and abiotic stress conditions. Identification of two new genes of mitochondrial Mn-SOD. Journal of Plant Physiology 2011; 168: 1303–1308. 10.1016/j.jplph.2011.01.020 [DOI] [PubMed] [Google Scholar]
- 82.Balazadeh S, Jaspert N, Arif M, Mueller-Roeber B, Maurino VG. Expression of ROS-responsive genes and transcription factors after metabolic formation of H (2) O (2) in chloroplasts. Frontiers in Plant Science 2012; 1; 234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yasudaa M, Ishikawac A, Jikumarud Y, Sekie M, Umezawa T, Asami T, et al. Antagonistic interaction between systemic acquired resistance and the abscisic acid–mediated abiotic stress response in Arabidopsis. Plant Cell 2008; 20: 1678–1692. 10.1105/tpc.107.054296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mentzen W, Wurtele ES. Regulon organization of Arabidopsis. BMC Plant Biology. 2008; 8: 99 10.1186/1471-2229-8-99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Carrera J, Rodrigo G, Jaramillo A, Elena SF. Reverse-engineering Arabidopsis thaliana transcriptional network under changing environmental conditions. Genome Biology 2009; 10: R96 10.1186/gb-2009-10-9-r96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Lindemose S, O’Shea C, Jensen MK, and Skriver K. Structure, function and networks of transcription factors involved in abiotic stress responses. International Journal of Molecular Sciences 2013:14: 5842–5878. 10.3390/ijms14035842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rizhsky L, Liang H, Mittler R. The combined effect of drought stress and heat shock on gene expression in tobacco. Plant Physiology 2002; 130: 1143–1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ramegowda V, Senthil-Kumar M, Nataraja KN, Reddy MK, Mysore KS, Udayakumar M. Expression of a finger millet transcription factor, EcNAC1, in tobacco confers abiotic stress-tolerance. PLoS ONE 2012; 7: e40397 10.1371/journal.pone.0040397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Babitha KC, Ramu SV, Pruthvi V, Mahesh P, Nataraja KN, Udayakumar M. Co-expression of AtbHLH17 and AtWRKY28 confers resistance to abiotic stress in Arabidopsis. Transgenic Research 2013; 22: 327–341. 10.1007/s11248-012-9645-8 [DOI] [PubMed] [Google Scholar]
- 90.Rizhsky L, Liang H, Shuman J, Shulaev V, Davletova S, Mittler R. When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiology 2004: 134: 1683–1696. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PPTX)
(PPTX)
(PPT)
(PPTX)
(PPTX)
(PPTX)
(PDF)
(PDF)
(PPTX)
(PPTX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.