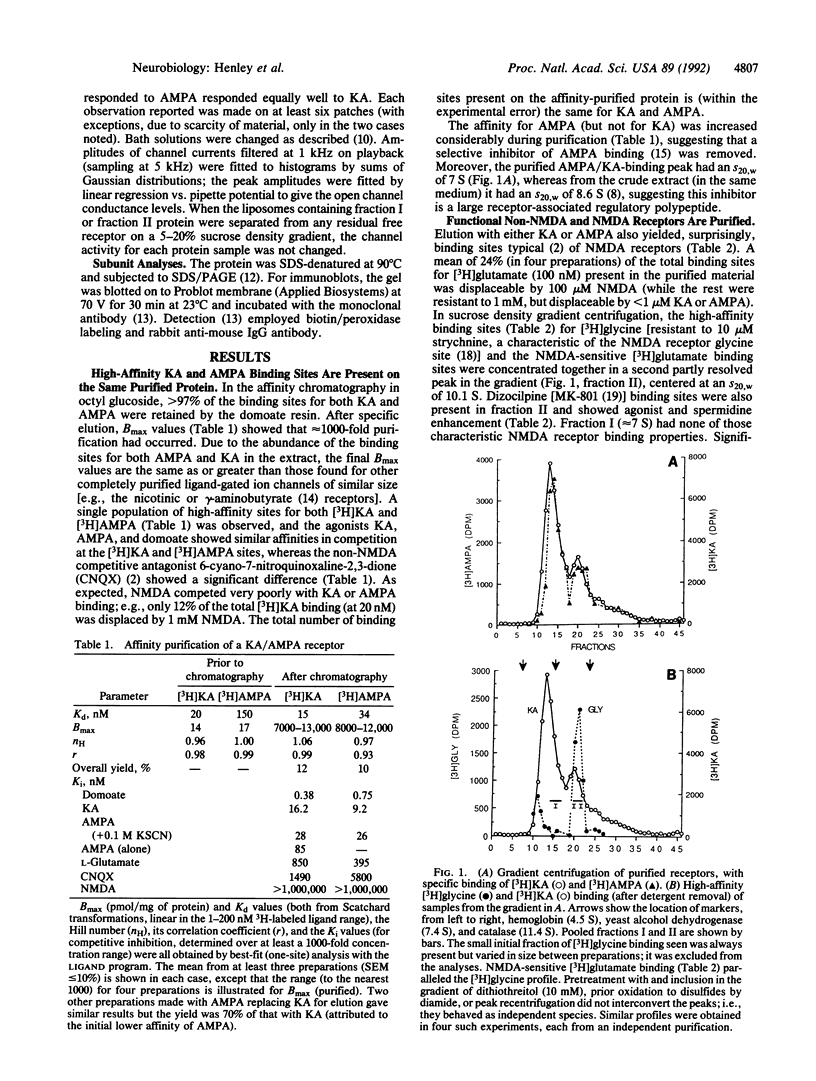
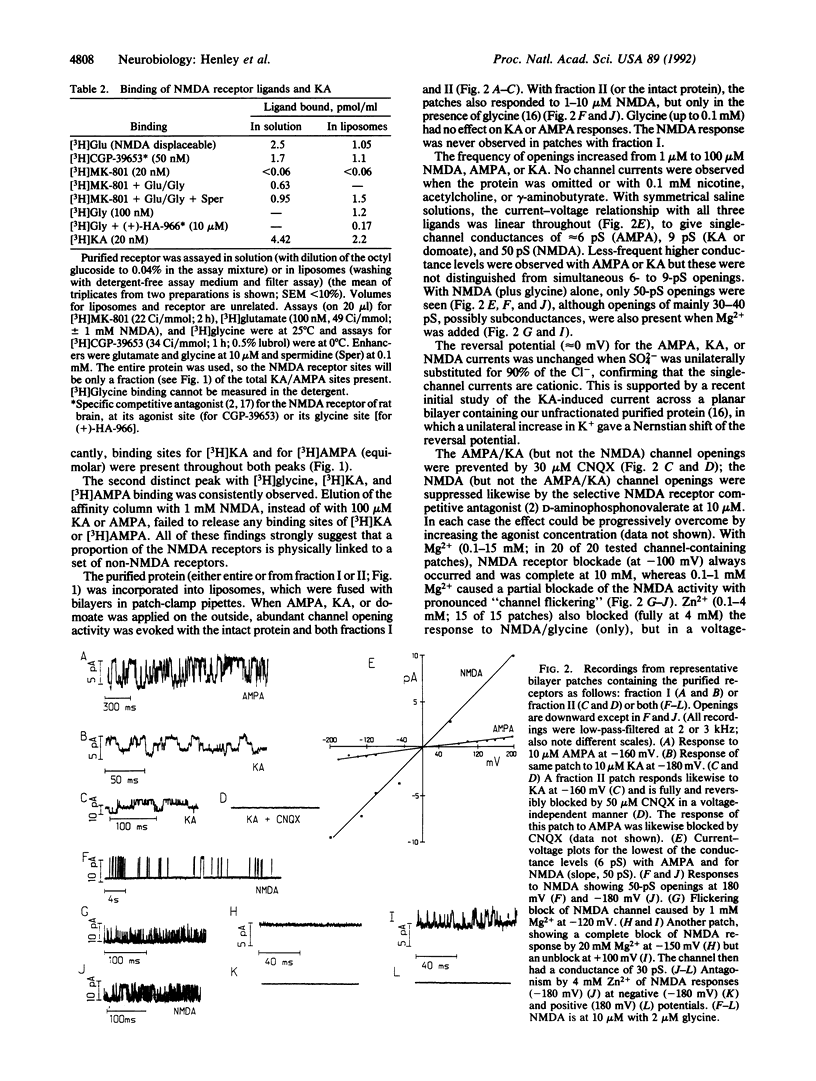
Abstract
We have purified and characterized two vertebrate excitatory amino acid ionotropic receptors from the Xenopus central nervous system. Each is a unitary receptor (i.e., having more than one class of excitatory amino acid agonist specificity within one protein oligomer). The first is a unitary non-N-methyl-D-aspartate (non-NMDA) receptor and the second is a unitary NMDA/non-NMDA receptor. The specific agonist-activated channel activity and pharmacology of each type were recognized by patch-clamping lipid bilayers in which the isolated protein was reconstituted. In the second case, the NMDA and the non-NMDA sites could not be physically separated and exhibited functional interaction. Parallel evidence for this was obtained when poly(A) RNA from Xenopus brain was translated in oocytes: a noncompetitive inhibition of the response to L-kainate is produced by NMDA to a maximum depression of 30% at 1 mM NMDA. Each isolated oligomer contains 42-kDa subunits of the non-NMDA ligand binding type, but the second type has an additional NMDA-receptor-specific 100-kDa subunit. Thus, a subunit-exchange hypothesis can account for the known multiplicity of excitatory amino acid receptor types.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ambrosini A., Barnard E. A., Prestipino G. AMPA and kainate-operated channels reconstituted in artificial bilayers. FEBS Lett. 1991 Apr 9;281(1-2):27–29. doi: 10.1016/0014-5793(91)80350-c. [DOI] [PubMed] [Google Scholar]
- Barnard E. A., Henley J. M. The non-NMDA receptors: types, protein structure and molecular biology. Trends Pharmacol Sci. 1990 Dec;11(12):500–507. doi: 10.1016/0165-6147(90)90051-9. [DOI] [PubMed] [Google Scholar]
- Boulter J., Hollmann M., O'Shea-Greenfield A., Hartley M., Deneris E., Maron C., Heinemann S. Molecular cloning and functional expression of glutamate receptor subunit genes. Science. 1990 Aug 31;249(4972):1033–1037. doi: 10.1126/science.2168579. [DOI] [PubMed] [Google Scholar]
- Brackley P., Goodnow R., Jr, Nakanishi K., Sudan H. L., Usherwood P. N. Spermine and philanthotoxin potentiate excitatory amino acid responses of Xenopus oocytes injected with rat and chick brain RNA. Neurosci Lett. 1990 Jun 22;114(1):51–56. doi: 10.1016/0304-3940(90)90427-b. [DOI] [PubMed] [Google Scholar]
- Coronado R., Latorre R. Phospholipid bilayers made from monolayers on patch-clamp pipettes. Biophys J. 1983 Aug;43(2):231–236. doi: 10.1016/S0006-3495(83)84343-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hampson D. R., Wenthold R. J. A kainic acid receptor from frog brain purified using domoic acid affinity chromatography. J Biol Chem. 1988 Feb 15;263(5):2500–2505. [PubMed] [Google Scholar]
- Henley J. M., Ambrosini A., Krogsgaard-Larsen P., Barnard E. A. Evidence for a single glutamate receptor of the ionotropic kainate/quisqualate type. New Biol. 1989 Nov;1(2):153–158. [PubMed] [Google Scholar]
- Henley J. M., Barnard E. A. Kainate receptors in Xenopus central nervous system: solubilisation with n-octyl-beta-D-glucopyranoside. J Neurochem. 1989 Jan;52(1):31–37. doi: 10.1111/j.1471-4159.1989.tb10894.x. [DOI] [PubMed] [Google Scholar]
- Henley J. M., Bond A., Barnard E. A. Autoradiographic localisations of glutamatergic ligand binding sites in Xenopus brain. Neurosci Lett. 1991 Aug 5;129(1):35–38. doi: 10.1016/0304-3940(91)90714-5. [DOI] [PubMed] [Google Scholar]
- Hollmann M., O'Shea-Greenfield A., Rogers S. W., Heinemann S. Cloning by functional expression of a member of the glutamate receptor family. Nature. 1989 Dec 7;342(6250):643–648. doi: 10.1038/342643a0. [DOI] [PubMed] [Google Scholar]
- Honoré T., Drejer J. Chaotropic ions affect the conformation of quisqualate receptors in rat cortical membranes. J Neurochem. 1988 Aug;51(2):457–461. doi: 10.1111/j.1471-4159.1988.tb01060.x. [DOI] [PubMed] [Google Scholar]
- Honoré T., Drejer J., Nielsen M. Calcium discriminates two [3H]kainate binding sites with different molecular target sizes in rat cortex. Neurosci Lett. 1986 Mar 28;65(1):47–52. doi: 10.1016/0304-3940(86)90118-7. [DOI] [PubMed] [Google Scholar]
- Honoré T., Lauridsen J., Krogsgaard-Larsen P. The binding of [3H]AMPA, a structural analogue of glutamic acid, to rat brain membranes. J Neurochem. 1982 Jan;38(1):173–178. doi: 10.1111/j.1471-4159.1982.tb10868.x. [DOI] [PubMed] [Google Scholar]
- Johnson J. W., Ascher P. Glycine potentiates the NMDA response in cultured mouse brain neurons. Nature. 1987 Feb 5;325(6104):529–531. doi: 10.1038/325529a0. [DOI] [PubMed] [Google Scholar]
- Klein A. U., Niederoest B., Winterhalter K. H., Cuénod M., Streit P. A kainate binding protein in pigeon cerebellum: purification and localization by monoclonal antibody. Neurosci Lett. 1988 Dec 19;95(1-3):359–364. doi: 10.1016/0304-3940(88)90685-4. [DOI] [PubMed] [Google Scholar]
- Kushner L., Lerma J., Zukin R. S., Bennett M. V. Coexpression of N-methyl-D-aspartate and phencyclidine receptors in Xenopus oocytes injected with rat brain mRNA. Proc Natl Acad Sci U S A. 1988 May;85(9):3250–3254. doi: 10.1073/pnas.85.9.3250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mamalaki C., Barnard E. A., Stephenson F. A. Molecular size of the gamma-aminobutyric acidA receptor purified from mammalian cerebral cortex. J Neurochem. 1989 Jan;52(1):124–134. doi: 10.1111/j.1471-4159.1989.tb10906.x. [DOI] [PubMed] [Google Scholar]
- Mayer M. L., Westbrook G. L., Vyklický L., Jr Sites of antagonist action on N-methyl-D-aspartic acid receptors studied using fluctuation analysis and a rapid perfusion technique. J Neurophysiol. 1988 Aug;60(2):645–663. doi: 10.1152/jn.1988.60.2.645. [DOI] [PubMed] [Google Scholar]
- Petrov A. G., Ramsey R. L., Usherwood P. N. Curvature-electric effects in artificial and natural membranes studied using patch-clamp techniques. Eur Biophys J. 1989;17(1):13–17. doi: 10.1007/BF00257141. [DOI] [PubMed] [Google Scholar]
- Sigel E., Barnard E. A. A gamma-aminobutyric acid/benzodiazepine receptor complex from bovine cerebral cortex. Improved purification with preservation of regulatory sites and their interactions. J Biol Chem. 1984 Jun 10;259(11):7219–7223. [PubMed] [Google Scholar]
- Sommer B., Keinänen K., Verdoorn T. A., Wisden W., Burnashev N., Herb A., Köhler M., Takagi T., Sakmann B., Seeburg P. H. Flip and flop: a cell-specific functional switch in glutamate-operated channels of the CNS. Science. 1990 Sep 28;249(4976):1580–1585. doi: 10.1126/science.1699275. [DOI] [PubMed] [Google Scholar]
- Verdoorn T. A., Dingledine R. Excitatory amino acid receptors expressed in Xenopus oocytes: agonist pharmacology. Mol Pharmacol. 1988 Sep;34(3):298–307. [PubMed] [Google Scholar]
- Vlachová V., Vyklický L., Vyklický L., Jr, Vyskocil F. The action of excitatory amino acids on chick spinal cord neurones in culture. J Physiol. 1987 May;386:425–438. doi: 10.1113/jphysiol.1987.sp016542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watkins J. C., Krogsgaard-Larsen P., Honoré T. Structure-activity relationships in the development of excitatory amino acid receptor agonists and competitive antagonists. Trends Pharmacol Sci. 1990 Jan;11(1):25–33. doi: 10.1016/0165-6147(90)90038-a. [DOI] [PubMed] [Google Scholar]
- Wong E. H., Kemp J. A., Priestley T., Knight A. R., Woodruff G. N., Iversen L. L. The anticonvulsant MK-801 is a potent N-methyl-D-aspartate antagonist. Proc Natl Acad Sci U S A. 1986 Sep;83(18):7104–7108. doi: 10.1073/pnas.83.18.7104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young A. B., Fagg G. E. Excitatory amino acid receptors in the brain: membrane binding and receptor autoradiographic approaches. Trends Pharmacol Sci. 1990 Mar;11(3):126–133. doi: 10.1016/0165-6147(90)90199-i. [DOI] [PubMed] [Google Scholar]



