Abstract
Our previous study showed that the in vivo positive effects of 17α,20β-dihydroxy-4-pregnen-3-one (DHP), the major progestin in zebrafish, on early spermatogenesis was much stronger than the ex vivo ones, which may suggest an effect of DHP on the expression of gonadotropins. In our present study, we first observed that fshb and lhb mRNA levels in the pituitary of male adult zebrafish were greatly inhibited by 3 wk exposure to 10 nM estradiol (E2). However, an additional 24 hr 100 nM DHP exposure not only reversed the E2-induced inhibition, but also significantly increased the expression of fshb and lhb mRNA. These stimulatory effects were also observed in male adult fish without E2 pretreatment, and a time course experiment showed that it took 24 hr for fshb and 12 hr for lhb to respond significantly. Because these stimulatory activities were partially antagonized by a nuclear progesterone receptor (Pgr) antagonist mifepristone, we generated a Pgr-knock out (pgr−/−) model using the TALEN technique. With and without DHP in vivo treatment, fshb and lhb mRNA levels of pgr−/− were significantly lower than those of pgr+/+. Furthermore, ex vivo treatment of pituitary fragments of pgr−/− with DHP stimulated lhb, but not fshb mRNA expression. Results from double-colored fluorescent in situ hybridization showed that pgr mRNA was expressed only in fshb-expressing cells. Taken together, our results indicated that DHP participated in the regulation of neuroendocrine control of reproduction in male zebrafish, and exerted a Pgr-mediated direct stimulatory effect on fshb mRNA at pituitary level.
Keywords: 17α,20β-dihydroxy-4-pregnen-3-one; Gonadotropin; Nuclear progesterone receptor; Zebrafish; Pituitary
Introduction
Gonadal steroid hormones play key roles in regulating gametogenesis, but they also exert both positive and negative feedback effects at the hypothalamic and pituitary levels. In tetrapod vertebrates, progesterone (P4) is the dominant ovarian progestin, which regulates reproductive behavior (Blaustein 1986) and exerts feedback effects on both pituitary gonadotropin secretion (Turgeon & Waring 1990, Waring & Turgeon 1992, Turgeon & Waring 2000) and hypothalamic gonadotropin-releasing hormone (GnRH) release (O’byrne et al. 1991, Skinner et al. 1998). 17α,20β-dihydroxy-4-pregnen-3-one (DHP) and 17α,20β,21-trihydroxy-pregn-4-en-3-one (20β-S) are the most potent and biologically relevant progestins in teleosts (Scott et al. 2010). In male fish, progestins induce spermiation (Ueda et al. 1985), increase seminal fluid production (Baynes & Scott 1985), and stimulate spermatozoa motility (Miura et al. 1992, Tubbs & Thomas 2008). Studies in Japanese eel (Anguilla japonica) demonstrated that DHP induces the entry of male germ cells into meiosis (Miura et al. 2006). Our recent studies in zebrafish showed that DHP treatment increased proliferation and differentiation of early spermatogonial generation (Chen et al. 2013). Interestingly, the in vivo effect of DHP on spermatogonia was more prominent than that ex vivo. One likely mechanism is that the effects of DHP in vivo may involve both direct action at the testicular level and indirect modulation of pituitary gonadotropin release. Alternatively, in vitro incubation could also lead to dysregulation of many paracrine and autocrine signaling around testes that are important for DHP signaling.
In mammalian models, some studies reported no effect of progesterone on luteinizing hormone (LH) (Kerrigan et al. 1993, Park et al. 1996), while others using immortalized gonadotrope-derived LβT2 cells (L beta T2 gonadotrope cell line, Turgeon et al. 1996) demonstrated that progesterone suppressed LH β subunit gene expression, in contrast to its stimulatory effect on the expression of follicle-stimulating hormone (FSH) β subunit (Thackray et al. 2006, Thackray et al. 2009). In teleosts, much progress has been made in understanding the regulation of the GnRH-Fsh/Lh system by androgens and 17β-estradiol (Zohar et al. 2010), but the potential roles of progestins in the neuroendocrine regulation of reproduction have received little attention. In female tilapia, DHP has been implicated in the regulation of Fsh and Lh release (Levavi-Sivan et al. 2006), but these DHP-mediated effects are considered to play a minor role in comparison to the dominant actions of androgen and 17β-estradiol (Van der Kraak 2009). Lack of information hinders an overall understanding of steroid hormones in regulating reproductive processes in teleosts.
The effects of progestins are mainly mediated through an intracellular nuclear progestin receptor (Pgr) that belongs to the nuclear receptor family (Conneely et al. 1986). In addition, multiple membrane progestin receptors (mPRs), which have no structural similarity to Pgr, also can mediate the non-classical action of progestin (Zhu et al. 2003, Hanna & Zhu 2011, Tan & Thomas 2015). In zebrafish, the Pgr is expressed robustly in the preoptic region of the hypothalamus (Hanna et al. 2010), and membrane progestin receptors (mPRs) in scattered cells in the pituitary (Hanna & Zhu 2009), suggesting potential roles of progestins in the brain-pituitary complex and the involvement of multiple progestin receptors and signaling pathways.
Recently, we have clearly shown that Pgr knockout (pgr−/−) causes completely anovulation and infertility in female zebrafish, but male knockout fish is still fertile (Zhu et al. 2015). In current study, we focused on the effects and molecular mechanisms of progestin (DHP) signaling in male zebrafish. We demonstrate that DHP can enhance expression of gonadotropins (fshb and lhb transcripts) in the pituitaries of male adult zebrafish, independently of estradiol (E2) inhibition. We also provide clear evidence from studies of Pgr antagonist, knockout and in situ to indicate that effects of progestin (DHP), especially on fshb are mediated directly at least in part by Pgr that is expressed in Fsh expressing cells in the pituitary of male zebarfish.
Materials and Methods
Zebrafish husbandry
The experimental fish were Tübingen strain, which were housed in the zebrafish (Danio rerio) facility (ESSEN, China) and maintained in recirculating freshwater (pH 7.2–7.6) at 28ºC with a 14L:10D photoperiod (lights on at 08:00). The fish were fed three times per day with commercial tropical fish food (Otohime B2, Reed Mariculture, CA, USA), using standard conditions for this species (Westerfield 2000). All the fish used were mature adult males (age 4~6 months, body length 2.4~2.8cm, body weight 275~440mg). Experimental protocols were approved by the Institutional Animal Care and Use Committee of Xiamen University.
Generating and characterizing pgr−/− zebrafish using TALENs
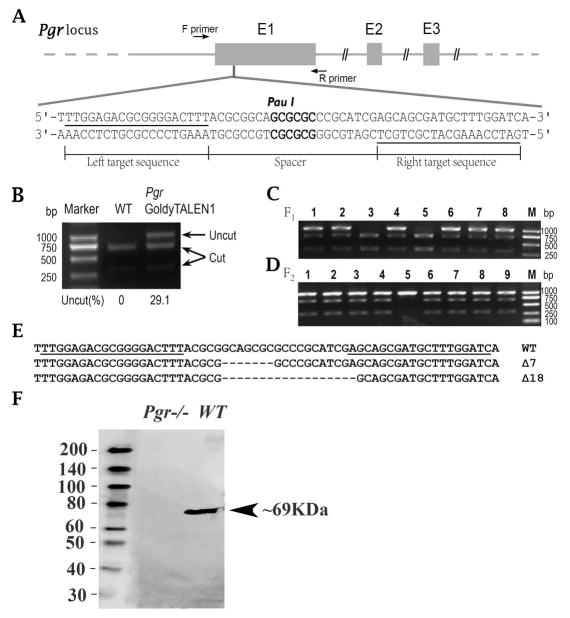
Instead of using an unit assembly protocol and a modified FokI (Huang et al. 2011, Zhu et al. 2015), we used a Golden Gate TALEN assembly protocol and wildtype FokI (Cermak et al. 2011) to generate TALEN expression vectors targeting one the locus same as previous target (Zhu et al. 2015) located in the 1st exon (Fig. 1A) of the zebrafish pgr gene (Ensembl No. ENSDARG00000035966). Thereafter, assembled pgr GoldyTALEN expression vectors were linearized using Sac I (Thermo Scientific, USA), and transcribed into mRNAs using the Mmessage mMACHINE T3 Transcription Kit (Life Technologies, USA).
Figure 1.
Approximately 1 nL containing ~75 pg of each TALEN mRNA was injected into the yolk of each embryo at the one-cell stage. For examining the mutation rate of the injected embryos, a pool of genomic DNA was extracted from 30 injected embryos or the same number of wild type (WT) embryos. With genomic DNA as template, a DNA fragment containing the TALEN target site was amplified using PCR (Table 1; Fig. 1A), followed by restriction endonuclease (Pau I; Thermo Scientific, USA) digestion, and checked with agarose gel electrophoresis. Area densities of cut and uncut bands on the gel were measured using the Gel-Pro Analyzer Program (Media Cybernetics) (Fig. 1B), mutation efficiency being calculated as (%) = uncut/(cut + uncut) × 100%.
Table 1.
Primers used in present study
| Primer name | Primer sequence (5′-3′) | Expected size (bp) | GenBank accession number | Purpose |
|---|---|---|---|---|
| pgr-TALEN Forward | AGGAATACGTCGCACACTTT | 965 | Ensembl No. ENSDARG00000035966 | Positive gene Knockout fish screening |
| pgr-TALEN Reverse | CGCCATTTTAATTCGACCTC | |||
| pgr-Forward | GGGTCTCGCTGCGTAATTTT | 841 | Ensembl No. ENSDARG00000035966 | RT-PCR |
| pgr-Reverse | GCCTGGTAGCACTTTCGAAG | |||
| ef1a-Forward | GGCTGACTGTGCTGTGCTGATTG | 409 | BC064291 | Real-time PCR |
| ef1a-Reverse | CTTGTCGGTGGGACGGCTAGG | |||
| fshb-Forward | CAGATGAGGATGCGTGTGC | 281 | AY424303 | Real-time PCR |
| fshb-Reverse | ACCCCTGCAGGACAGCC | |||
| lhb-Forward | GCAGAGACACTTACAACAGCC | 145 | AY424304/AY424305 | Real-time PCR |
| lhb-Reverse | AAAACCAAGCTCTGAGCAGCC |
To obtain homozygous mutant zebrafish, adult (>90 dpf) F0 founder fish were outcrossed with WT fish. From each cross, a pool of genomic DNA was extracted from 30 randomly selected F1 embryos, and the status of the TALEN target site was analyzed via PCR amplification, and restriction enzyme digestion as described above. Based on the mutation efficiency, the remaining F1 embryos from F0 founder fish with high rates of germline transmission were raised to adulthood and were genotyped individually using tail fin-clip screening assay in order to obtain heterozygous F1. Based on the DNA sequence of the TALEN target sites, heterozygous F1 fish with the same frameshift mutation were intercrossed to produce F2 offspring (Fig. 1C). Homozygous Pgr-knockout (pgr−/−), heterozygotes (pgr+/-) and homozygous wild-type (pgr+/+) in F2 fish were identified via PCR amplification followed by restriction enzyme digestion analysis as described above (Fig. 1D).
In order to examine the pgr mRNA sequence in pgr−/− male fish, cDNA was synthesized using a Revert Aid First Strand cDNA Synthesis Kit (Thermo Scientific, USA) from the total RNA extracted from pooled pituitary samples using RNAzol reagent (MRC, Cincinnati, OH, USA). The specific PCR product was obtained using the Pgr-primers (Table 1), followed by cloning and Sanger sequencing.
In order to examine if Pgr protein was absent in pgr−/− male fish, testicular samples of WT and pgr−/− fish were collected for Western blot assay as described previously (Hanna et al. 2010). Total protein samples were extracted by immediately placing freshly excised testis into 1×SDS buffer, denatured by boiling for 5 min, and then cooled on ice. Equal amounts (60 μg) of protein samples were loaded and separated using a 12% SDS-PAGE gel and were transferred onto a PVDF membrane. The membrane was blocked in TBST containing 0.1% (v:v) tween-20 and 1% (w:v) bovine serum albumin for 1 hr at room temperature (RT), before the membrane was incubated with Pgr antibody (Hanna et al. 2010) for 24 hr at 4ºC. After five washes with TBST, the membrane was incubated for 1 hr at RT with the horseradish peroxidase (HRP)-conjugated secondary antibody (1:1000, v:v). A Bio-Rad ECL kit was used to detect signals on the PVDF membrane. Protein size was determined by comparing blotted protein size to a biotinylated protein ladder (Cell Signaling Technology, #7727s, USA) following the manufacturer’ s directions.
Expression of fshb and lhb mRNA during the diurnal cycle
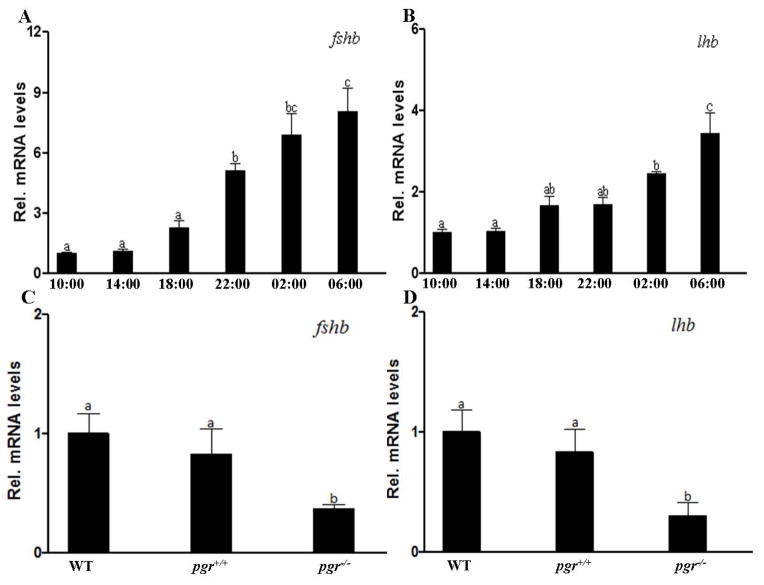
To examine the expression of fshb and lhb mRNA levels in the pituitary during the diurnal cycle, mature WT male fish (>90 dpf) from the same batch were randomly chosen. Pituitary samples were collected every four hours starting at 10:00 until 06:00. Since maximum levels were found at 06:00, expression levels of WT and pgr−/− fish were compared at 06:00. Three pituitaries were pooled as one sample for these mRNA quantification studies.
In vivo exposure to sex steroids
Using a zebrafish model of estrogen-induced androgen insufficiency (De Waal et al. 2009), adult (>90 dpf) males were kept for 3 wk in water containing 10 nM estradiol (E2) (Sigma–Aldrich, China). Fish were then exposed again to 10 nM E2 either with or without 100 nM DHP (Sigma-Aldrich, China) for another 24 hr. From the same batch of male fish, animals without E2 pretreatment were exposed to 100 nM DHP for 24 hr. Male fish were euthanized in ice water; pituitaries were collected (three pituitaries were pooled as one sample), frozen quickly in dry ice and stored at −80ºC for RNA extraction.
For other in vivo exposure experiments, adult (>90 dpf) male fish were randomly divided into the control and DHP treatment groups. To determine an appropriate exposure concentration, fish were exposed to 10 or 100 nM DHP for 24 hr (starting at 13:00), while the control group was exposed to the vehicle (ethyl alcohol at a concentration of 0.0001%, v:v). For time course experiments, treatment groups were exposed to 100 nM DHP at 13:00, and pituitary samples were collected as described above at 16:00, 19:00, 01:00 and 13:00 to determine appropriate length of exposure time.
To study if the effects of DHP on fshb and lhb transcript levels were Pgr-dependent, male fish were exposed to DHP (100 nM) and Pgr specific antagonist (RU486, 0.1 or 1 μM) for 24 hr. An additional control group was exposed only to 1 μM RU486 for 24 hr. Moreover and, in addition, pgr−/− male fish were exposed to the vehicle (as control, 0.0001% ethyl alcohol, v:v) or DHP (100 nM) for a period of 24 hr. We also quantified the basal fshb and lhb mRNA levels in pgr+/+ fish.
Pituitary ex vivo exposure to sex steroids
Pituitaries of mature male fish were removed, washed three times with Hank’s balanced salt solution (without magnesium and calcium) in a 24-well plate (NEST, USA), and then incubated individually in basal culture medium consisting of 15mg/mL Leibovitz’s L-15 medium (Invitrogen, USA), supplemented with 10 mM Hepes (Merck, Germany), 0.5% w/v BSA (MULT Sciences, China), 200 U/mL penicillin and 200 mg/L streptomycin (Invitrogen, USA); pH was adjusted to 7.4 with NaOH. Pituitary samples were incubated for 24 hr in basal culture medium containing either 100 nM DHP, or 0.0001% (v:v) ethyl alcohol as control. Incubation was carried out in a humidified air atmosphere at 28ºC for 24 hr, and three pituitaries were pooled to obtain one sample for gene expression analysis.
Gene expression analysis
Total RNA was extracted from pituitary samples using RNAzol reagent (MRC, Cincinnati, OH, USA). Three pituitaries were pooled as one sample to obtain sufficient RNA for cDNA synthesis. The same amount of total RNA (1.0 μg) was used for the synthesis of the first strand cDNAs using a Revert Aid First Strand cDNA Synthesis Kit (Thermo Scientific, USA) following the manufacturer’s instructions.
The relative expression levels of fshb, lhb and of the house-keeping-gene ef1a were determined using real-time quantitative PCR (qPCR) with gene-specific primers (Table 1), which had been examined for their specificity and amplification efficiency on serial dilutions of respective target gene plasmid DNA (2×102–2×107 copies/2 μL). qPCR was performed in a 20 μL reaction mixture on the 7500 FAST real-time PCR detection system (Applied Biosystems, USA) using default settings. Copies of ef1α, which showed no significant difference among different stages, were used as internal control. The relative mRNA levels of the target genes were determined using the comparative Ct method (Schmittgen & Livak 2008).
Double-colored fluorescent in situ hybridization
The colocalization of pgr with either fshb or lhb mRNA in zebrafish pituitary was investigated using double-colored fluorescent in situ hybridization (FISH), as described previously (Chen & Ge 2012), except that cryosections were used. In brief, pituitaries of adult male zebrafish were dissected and fixed in 4% w/v paraformaldehyde in PBS at 4 ºC overnight, followed by immersion in 25% w/v sucrose in PBS at 4 ºC until sinking, and then embedded in optimal cutting compound (Tissue-Tek™, Sakura, USA) by freezing in liquid nitrogen. Embedded pituitaries were processed for serial frozen sectioning at 10μm thickness, rehydrated and digested with proteinase K (5 μg/mL; Roche Applied Science, Germany) at 37ºC for 5 min, followed by hybridization with fluorescein labeled and DIG-labeled RNA probes at 55ºC overnight. The probes for the detection of fshb, lhb and pgr mRNA were generated as described previously (Chen & Ge 2012, Chen et al. 2010). After hybridization, the cryosections were washed with 2×saline-sodium citrate (SSC; 0.06 M NaCl and 0.006 M sodium citrate) for 30 min at RT, 30% v/v formamide deionized in 2×SSC for 15 min at 65ºC, 0.2×SSC for 15 min at 65ºC, and 0.2×SSC for 15 min at RT. After washing, a TSA Plus Cyanine3/Fluorescein (TSA-Cy3) System (PerkinElmer, USA) was used to detect the hybridization signal. The first signal was detected using HRP-conjugated anti-fluorescein antibody (Roche Applied Science, Germany) with TSA Fluorescein following the manufacturer’s instructions. In order to detect the second signal, cryosections were incubated in 1% H2O2 for 60 min to deactivate the HRP from the first staining. HRP-conjugated anti-DIG antibody (Roche Applied Science, Germany) were added to the sections, followed by detection with the TSA-Cy3 system. After mounting with the medium for fluorescence reagent (Vector, USA), the slides were observed and the images recorded using a Zeiss LSM 780 NLO two photon laser scanning system (Zeiss, Germany).
Statistical analysis
All data are presented as means ± standard error of the mean. Depending on the experimental setup, data were analyzed using either Student’s t-test or one-way ANOVA followed by Tukey’s post hoc test to assess statistical differences between two or more groups. The analyses were performed using the GraphPad Prism 4 software package (GraphPad Software, San Diego, CA).
Results
Generating the pgr−/− zebrafish model and knockout phenotype
Mutant lines targeting two different loci with three different frame shift mutations caused by small deletions and/or insertions of nucleotides in the 1st exon of pgr genomic sequences have been generated and characterized using a unit assembly protocol and modified FokI (for details, see Zhu et al. 2015). In current study, we targeted same locus as one of the previous targets (Fig. 1A) using a different TALEN assembly protocol and wildtype FokI (Cermak et al. 2011), and successfully generated a new knockout line with a different frame shift (7-bp deletion) and a premature stop codon (Fig. 1B–E). Pgr protein was undetectable in the testes of pgr−/− fish (Fig. 1F). We found exact same anovulation and infertility in newly generated knockout female zebrafish, which validated our previous finding (Zhu et al. 2015). Similarly, we found that Pgr knockout had no obvious effect in male fertility. So this new Pgr line was chosen for the following experiments.
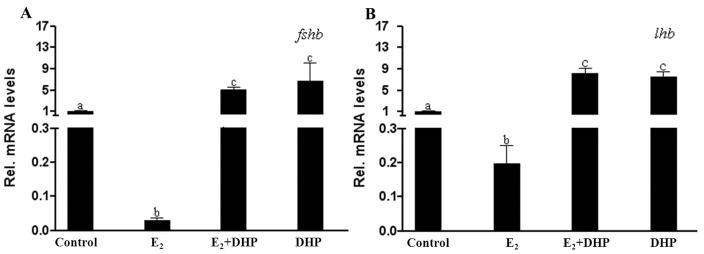
E2 inhibited but DHP stimulated the expression of fshb and lhb, independently from E2 inhibition
Both fshb and lhb transcripts in the pituitaries of male zebrafish decreased significantly to 2 (fshb) or 20% (lhb) of the control after 3 wk of E2 (10 nM) exposure (Fig. 2). In contrast, expression of the fshb and lhb transcripts in the pituitaries of male zebrafish increased significantly compared to the control when male fish were exposed to DHP both with or without E2 (Fig. 2).
Figure 2.
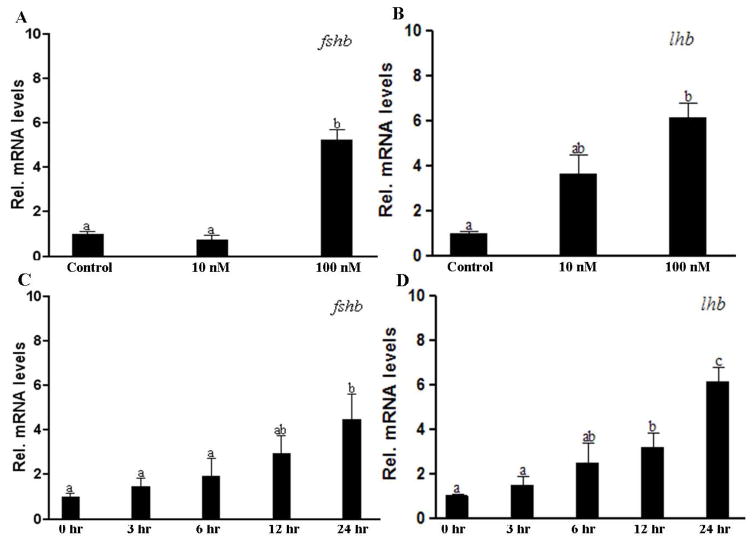
Dose and time dependent stimulatory effects of progestin on the expression of fshb and lhb
Because pituitary fshb and lhb transcript levels increased to similar levels in response to DHP irrespective of an E2 pretreatment, the subsequent DHP exposure experiments were carried out without E2 pretreatment.
Both fshb and lhb transcripts in the pituitaries of male fish increased significantly (5-fold for fshb, 7-fold for lhb) when fish were exposed to 100 nM DHP water for 24 hr; while 10 nM DHP exposure had no significant effects (Fig. 3A, B). The time course experiment indicated that significant increases of fshb and lhb transcript levels in response to DHP (100 nM) required at least 24hr and 12 hr exposure, respectively (Fig. 3C, D).
Figure 3.
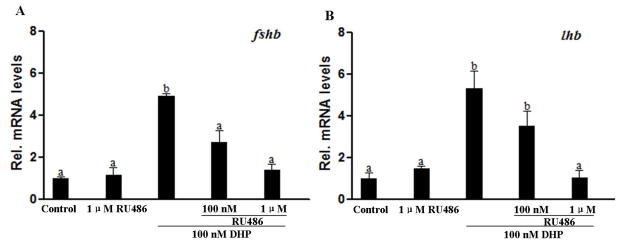
RU486 inhibited DHP induced expression of fshb and lhb in the pituitary
Effects of a Pgr specific antagonist (RU486) were examined in order to determine if DHP-induced fshb and lhb expression was mediated through Pgr. RU486 alone, up to a concentration of 1 μM, did not alter fshb and lhb expression levels (Fig. 4), but RU486 significantly inhibited the stimulatory effects of DHP on the expression of fshb and lhb, in a dose-dependent manner (Fig. 4).
Figure 4.
Daily expression levels of fshb and lhb transcripts in the pituitary of pgr−/− male fish
Expression of fshb and lhb mRNA showed distinct diurnal changes in WT male fish. Both transcripts of fshb and lhb were low in the late morning (10:00) after spawning and in the afternoon (14:00). The levels increased in the evening (18:00) and reached peak levels in early morning (06:00), two hours before the lights came on and the start of spawning activity (Fig. 5A, B). The highest transcript level of fshb was ~7.5-fold higher than the lowest one, while that of lhb was ~4.0-fold higher than the lowest one. Furthermore, we observed that there was no difference in fshb or lhb transcript levels between control WT male (i.e. not treated with TALENs) and pgr+/+ male pituitaries collected at 06:00 (Fig. 5C, D). This excluded potential TALEN induced off-target effects on the site that might affect the reproductive system. In addition, the levels of fshb and lhb mRNA of pgr−/− male fish were significantly lower than those of WT and pgr+/+ collected at 06:00 (Fig. 5C, D).
Figure 5.
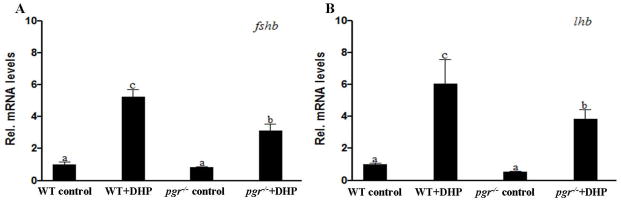
Effects of DHP on the expression of fshb and lhb in pgr−/− male zebrafish
Surprisingly, we found that DHP still significantly increased the mRNA levels of fshb and lhb in pgr−/− fish, but the magnitudes of the increases were significantly lower than those observed in WT fish (Fig. 6A, B).
Figure 6.
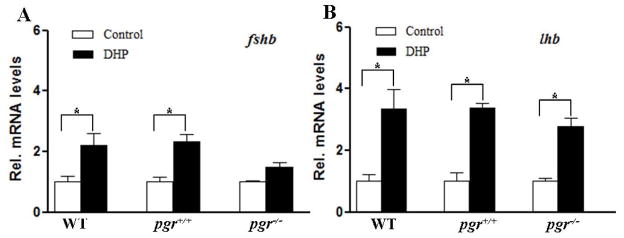
To examine whether DHP acts directly at the pituitary level to increase fshb and lhb mRNA levels, an ex vivo pituitary culture system was used. Both fshb and lhb transcripts increased significantly (~2-fold for fshb, ~2.5-fold for lhb) in the cultured pituitary tissue fragments of both WT and pgr+/+ males exposed to 100 nM DHP for 24 hr (Fig. 7). Furthermore, compared to the pgr−/− control, there was a significant increase (~2-fold) of lhb transcript levels in pituitary fragments of pgr−/− fish following exposure to 100 nM DHP for 24 hr (Fig. 7B), while fshb transcript levels no longer responded to DHP in pituitaries of pgr−/− males (Fig. 7A).
Figure 7.
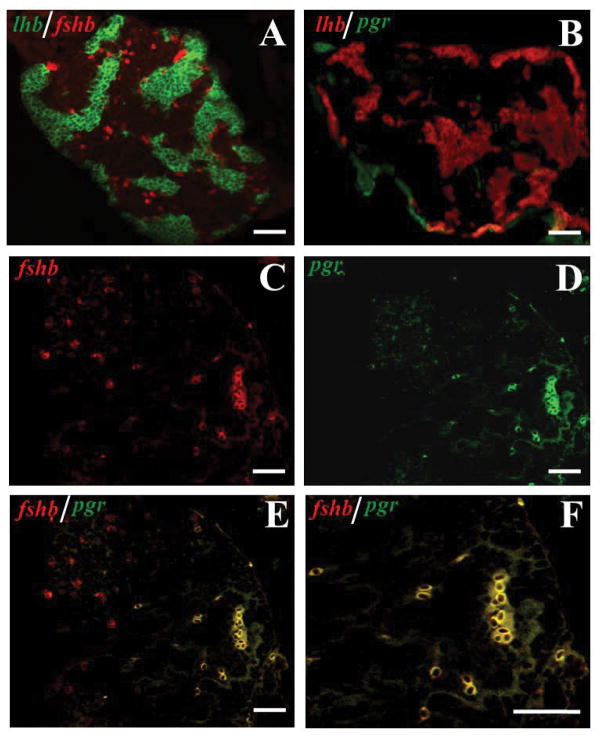
Pgr mRNA were expressed in fshb-expressing cells but not in lhb-expressing cells
Because the above results suggested that at least some part of DHP effects on gonadotropin hormone (GTH) expression were likely mediated by Pgr, we further investigated if pgr mRNA was expressed in gonadotropin cells for a direct action of DHP. Using double-colored FISH, which could detect the expression of two genes in the same section as reported previously (Chen & Ge 2012), we observed that the fshb-expressing cells scattered individually in the pituitary, whereas the lhb-expressing cells normally formed aggregates (Fig. 8A), which is consistent with a recent report (Golan et al. 2016). Because the expression level of pgr mRNA was much lower than those of fshb and lhb mRNA in the pituitary, the FISH signals of pgr mRNA were observed only in a few cells scattered individually (Fig. 8B–F). The FISH signal of pgr transcript was mostly observed in fshb-expressing cells (Fig. 8C–F), but none in lhb-expressing cells (Fig. 8B).
Figure 8.
Discussion
Gonadal steroids exert negative or positive feedbacks on gonadotropin synthesis and secretion in various vertebrates. In the present study, we investigated the potential roles of DHP in the regulation of gonadotropin transcription in adult male zebrafish using morphological, physiological and molecular approaches. The results showed that DHP was able to stimulate the transcription levels of both fshb and lhb. Importantly, using pgr−/− fish we demonstrated that the stimulatory effect of DHP on fshb mRNA was mediated by the Pgr which was expressed in fshb-expressing pituitary cells.
We first applied the long-term E2 treatment model to examine the effects of DHP on gonadotropin subunit expression in vivo. Our results showed that DHP significantly increased gonadotropin subunit mRNA expression in the presence and absence of E2. This was different from that in mammalian models, in which many studies have shown that progesterone can suppress LH mRNA levels in the presence of estrogen (Abbot et al. 1988, Simard et al. 1988, Corbani et al. 1990). Therefore, it seemed that there was no interaction between DHP and E2 in regulating GTH subunit expression in male zebrafish.
Extensive studies in goldfish have demonstrated that waterborne DHP is a pheromone which can increases LH, steroid and seminal fluid production (Stacey & Sorensen 1986, Dulka et al. 1987, Kobayashi et al. 2002). Because of the exposure via water in the present study, it is possible that DHP exerted its stimulatory effects as a pheromone. While the zebrafish ovary can produce steroid glucuronides, including 17,20β-P-glucuronide, which are attractive to males, 17,20β-P-glucuronide has never been tested as a sex pheromone in male zebrafish. However, 17,20β-P-sulphate is the only steroid that males appear to be able to smell (Belanger et al. 2010). Sex pheromones usually induce a rapid endocrine response at low concentrations (pM and low nM). For example, exposure of goldfish to 0.5 nM DHP increases serum LH levels after only 15 min (Dulka et al. 1987). However, in the present study, the significant response of fshb and lhb to DHP exposure required 24 hr and 12 hr, respectively, reaching maximum levels after 24 hr exposure, and DHP induced the up-regulation of fshb mRNA expression only at a high concentration (100 nM). Moreover, using the primary pituitary ex vivo culture system, we observed a stimulatory effect of DHP on GTH expression. Taken together, it was most likely that DHP-induced GTH up-regulation, especially fshb mRNA expression, through an endocrine pathway.
Several studies in fish report that plasma LH levels are very low during the early spermatogenesis stage and become detectable when germ cells entered meiosis; however, the increase is not prominent until the spawning season (Schulz et al. 2010). Similarly, the highest levels of circulating progestins are observed during the entire spermiation process, especially during the spawning season (Schulz et al. 2010). One physiological mechanism to explain this coincidence is that LH induced the production of DHP (Schulz et al. 2010). Our previous study in zebrafish also demonstrates that recombinant zebrafish Lh induces DHP production ex vivo (Chen et al. 2010). Data from the present study showed that DHP had a positive feedback on lhb mRNA expression, which was in agreement with previous studies in female tilapia (Levavi-Sivan et al. 2006). These results might indicate that LH is the main factor regulating the production of the maturation-inducing hormone DHP.
An increase of fshb mRNA levels during spermiation is recorded in several teleost species, which may be due to environment factors, i.e. temperature and photoperiod (Schulz et al. 2010). In the present study, we reported that DHP increased fshb mRNA levels in male zebrafish. It is worth noting that this stimulatory effect required a high concentration of DHP (100 nM), which is supposed to happen during spawning. Therefore, we examined the diurnal changes of fshb expression levels, and the results indicated that both fshb and lhb transcripts reached peak levels at 06:00, 2 hours before spawning. Interestingly, a previous study in female zebrafish also reports an obvious elevation of fshb expression at 01:00 and 04:00 before final oocyte maturation at 07:00 (So et al. 2005). Taken together, these results suggested that FSH may be involved in final gamete maturation, which is different from the current view that FSH in fish is involved in early gametogenesis, i.e. promoting the early stage of spermatogenesis in the testis, and stimulating follicle growth or vitellogenesis in the ovary. As a continuous daily breeder with an asynchronous spermatogenic cyst, it is most likely that zebrafish initiate the early stages of spermatogenesis at some time point every day. Results from our present study supported the suggestion from our previous study that DHP-induced early spermatogenesis is mainly mediated by triggering the release of gonadotropins (Chen et al. 2013). Recent studies in zebrafish indicate that FSH promotion of proliferation and differentiation of spermatogonia does not require androgen, but can also be mediated by suppressing an inhibitor of spermatogenesis (Skaar et al. 2011), or by inducing a stimulator of spermatogenesis (Igf3) (Nóbrega et al. 2015). Moreover, the testes in fshr−/− males showed significant retardation in growth with delayed spermatogenesis and gonad formation, though male fertility did not seem to be affected (Zhang et al. 2015a). In contrast to significant effects and infertility due to LH knockout in female fish, male fertility was also apparently not affected in zebrafish (Zhang et al. 2015b, Chu et al. 2014). However, our present study clearly indicates expressions of fshb and lhb mRNA are regulated by feedback signaling of progestin in zebrafish. Taken together, FSH, LH and progestin are working in concert to appropriately regulate gonadal growth, puberty onset, daily recruitment and maturation of germ cells in male zebrafish. Clearly, additional studies are required to understand effects, regulation, and conserved functions of LH, FSH, progestin, and their receptors during reproduction and evolution.
Three classes of progestin receptors, i.e. Pgr, mPR and progesterone receptor membrane component (Pgrmc), are reported in vertebrates (Zhu et al. 2008, Thomas 2008). Previous reports and the present results showed that both Pgr and mPR are expressed in the zebrafish hypothalamus and pituitary (Chen et al. 2010, Hanna et al. 2010, Hanna & Zhu 2009), so that DHP may induce GTH expression via Pgr or/and mPRs. The results from present study showed that the Pgr antagonist RU486 partially blocked the stimulatory effects of DHP. Furthermore, using a pituitary ex vivo culture system, we did not observe any stimulatory effect of DHP on fshb expression in the pgr−/− model. Moreover, results from double-colored FISH showed that most of the FISH signals revealed by the pgr probe were observed in fshb-expressing cells, but not in lhb-expressing cells. Our results clearly indicated that DHP exerted a direct stimulatory effect on fshb mRNA expression which was mediated by Pgr at the pituitary level. In mammals, it has been demonstrated that the full suppressive effect of progesterone on Lhb gene expression requires the unique amino-terminal region of the Pgr, but this suppression does not require direct binding of Pgr to the Lhb promoter although it is recruited to the endogenous promoter in live cells (Thackray et al. 2009). In contrast, progesterone directly activates Fshb through binding of Pgr in the proximal FSH promoter (Thackray et al. 2006). The molecular mechanism of progestins on fshb gene expression is conserved in vertebrates during evolution. Further study is necessary to show whether other progestin receptors are involved in mediating the stimulatory effect of DHP on lhb gene expression in fish.
Interestingly, DHP still caused up-regulation of GTH expression in vivo in pgr−/− male fish, suggesting that nonnuclear receptor mediated pathways in other brain regions, most likely in the hypothalamus, might be involved in the stimulatory effects of DHP on GTH. However, the relevant information is less documented in teleosts. In mammals, the molecular mechanisms of progesterone on GnRH were also unambiguous. Classic Pgr (Skinner et al. 1998), PAQR (Sleiter et al. 2009) and PGRMC1 (Bashour & Wray 2012) are suggested as the key players in mediating progesterone action on GnRH. Further research is needed to address the involvement of other progestin receptors in mediating this effect.
In teleost fish, estrogenic compounds can inhibit androgen synthesis and prevent spermatogenesis (Van der Ven et al. 2003, Van den Belt et al. 2004, Pawlowski et al. 2004, Van der Ven et al. 2007), but the mechanisms remain unclear. A study in zebrafish suggests that in vivo exposure to E2 causes a state of androgen insufficiency, involving feedback mechanisms on the hypothalamus–pituitary system (De Waal et al. 2009). In the present study, we observed in vivo a clear inhibitory effect of E2 on fshb and lhb mRNA levels, similar to what is described in the closely related goldfish (Kobayashi et al. 2000, Huggard-Nelson et al. 2002). However, direct estrogenic stimulation of pituitary cells elevates fshb and lhb transcript levels in zebrafish (Lin & Ge 2009). Therefore, we concluded that in zebrafish, long-term exposure to E2 inhibited stimulatory or strengthened inhibitory signaling towards the pituitary gonadotropin cells, and these effects overruled the direct, stimulatory effects of E2 on gonadotropin subunit expression.
In summary, the present study indicated that DHP played an important role in the regulation of gonadotropin production in the male zebrafish pituitary. It is likely that the effect of DHP on gonadotropin gene expression may have resulted from integration of multiple effectors acting on the hypothalamus, as well as the pituitary. In addition, Pgr was the main but not the only receptor type for mediating this effect. However, at the pituitary level, DHP exerted a Pgr mediated direct stimulatory effect on fshb mRNA. Despite this, pgr−/− male fish are fertile (Zhu et al. 2015), which is similar to a mice model (Schneider et al. 2005), and the biological significance of this stimulatory effect of DHP on gonadotropin gene expression remains to be clarified.
Acknowledgments
Funding
This study was supported by the National Natural Science Foundation of China (No. 31201977 & No. 41276129), the funding for Doctor Station of the Ministry of Education of China (No. 20120121110029), Xiamen Southern Oceanographic Center (No. 14GZY019NF19), and NIH 1R15GM100461-01A1.
We would like to express our great appreciation to Professor John Hodgkiss for assistance with the English.
Footnotes
Declaration of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author contributions
CLW was involved in entire study. DTL performed TALEN synthesizing, mutation screen. WTC performed double-colored fluorescent in situ hybridization. WG, YZ and WSH supervised the project and prepared manuscript drafting. SXC conceived and supervised the project, analyzed results and wrote the paper.
References
- Abbot SD, Docherty K, Clayton RN. Regulation of LH subunit mRNA levels by gonadal hormones in female rats. Journal of Molecular Endocrinology. 1988;1:49–60. doi: 10.1677/jme.0.0010049. [DOI] [PubMed] [Google Scholar]
- Bashour NM, Wray S. Progesterone directly and rapidly inhibits GnRH neuronal activity via progesterone receptor membrane component 1. Endocrinology. 2012;153:4457–4469. doi: 10.1210/en.2012-1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baynes SM, Scott AP. Seasonal variations in parameters of milt production and in plasma concentration of sex steroids of male rainbow trout (Salmo gairdneri) General and Comparative Endocrinology. 1985;57:150–160. doi: 10.1016/0016-6480(85)90211-4. [DOI] [PubMed] [Google Scholar]
- Belanger RM, Pachkowski MD, Stacey NE. Methyltestosterone-induced changes in electro-olfactogram responses and courtship behaviors of cyprinids. Chemical Senses. 2010;35:65–74. doi: 10.1093/chemse/bjp085. [DOI] [PubMed] [Google Scholar]
- Blaustein JD. Steroid Receptors and Hormone Action in the Brain. Annals of the New York Academy of Sciences. 1986;474:400–414. doi: 10.1111/j.1749-6632.1986.tb28030.x. [DOI] [PubMed] [Google Scholar]
- Cermak T, Doyle EL, Christian M, Wang L, Zhang Y, Schmidt C, Baller JA, Somia NV, Bogdanove AJ, Voytas DF. Efficient design and assembly of custom TALEN and other TAL effector-based constructs for DNA targeting. Nucleic Acids Research. 2011;185:1–9. doi: 10.1093/nar/gkr218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen SX, Bogerd J, García-López Á, Jonge H, de Waal PP, Hong WS, Schulz RW. Molecular cloning and functional characterization of a zebrafish nuclear progesterone receptor. Biology of Reproduction. 2010;82:171–181. doi: 10.1095/biolreprod.109.077644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen SX, Bogerd J, Schoonen NE, Martijn J, de Waal PP, Schulz RW. A progestin (17α, 20β-dihydroxy-4-pregnen-3-one) stimulates early stages of spermatogenesis in zebrafish. General and Comparative Endocrinology. 2013;185:1–9. doi: 10.1016/j.ygcen.2013.01.005. [DOI] [PubMed] [Google Scholar]
- Chen W, Ge W. Ontogenic expression profiles of gonadotropins (fshb and lhb) and growth hormone (gh) during sexual differentiation and puberty onset in female zebrafish. Biology of Reproduction. 2012;86:1–11. doi: 10.1095/biolreprod.111.094730. [DOI] [PubMed] [Google Scholar]
- Chu L, Li J, Liu Y, Hu W, Cheng CH. Targeted gene disruption in zebrafish reveals noncanonical functions of LH signaling in reproduction. Molecular Endocrinology. 2014;28:9292–9296. doi: 10.1210/me.2014-1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conneely OM, Sullivan WP, Toft DO, Birnbaumer M, Cook RG, Maxwell BL, Zarucki-Schulz T, Greene GL, Schrader WT, O’Malley BW. Molecular cloning of the chicken progesterone receptor. Science. 1986;233:767–770. doi: 10.1126/science.2426779. [DOI] [PubMed] [Google Scholar]
- Corbani M, Counis R, Wolinska-Witort E, d’Angelo-Bernard G, Moumni M, Jutisz M. Synergistic effects of progesterone and oestradiol on rat LH subunit mRNA. Journal of Molecular Endocrinology. 1990;4:119–125. doi: 10.1677/jme.0.0040119. [DOI] [PubMed] [Google Scholar]
- De Waal PP, Leal MC, García-López Á, Liarte S, de Jonge H, Hinfray N, Brion F, Schulz RW, Bogerd J. Oestrogen-induced androgen insufficiency results in a reduction of proliferation and differentiation of spermatogonia in the zebrafish testis. Journal of Endocrinology. 2009;202:287–297. doi: 10.1677/JOE-09-0050. [DOI] [PubMed] [Google Scholar]
- Dulka JG, Stacey NE, Sorensen PW, Vanderkraak GJ. A steroid sex pheromone synchronizes male–female spawning readiness in goldfish. Nature. 1987;325:251–253. [Google Scholar]
- Golan M, Martin AO, Mollard P, Levavi-Sivan B. Anatomical and functional gonadotrope networks in the teleost pituitary. Scientific Reports. 2016;6:23777. doi: 10.1038/srep23777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanna RN, Daly SCJ, Pang Y, Anglade I, Kah O, Thomas P, Zhu Y. Characterization and expression of the nuclear progestin receptor in zebrafish gonads and brain. Biology of Reproduction. 2010;82:112–122. doi: 10.1095/biolreprod.109.078527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanna RN, Zhu Y. Expression of membrane progestin receptors in zebrafish (Danio rerio) oocytes, testis and pituitary. General and Comparative Endocrinology. 2009;161:153–157. doi: 10.1016/j.ygcen.2008.10.006. [DOI] [PubMed] [Google Scholar]
- Hanna RN, Zhu Y. Controls of meiotic signaling by membrane or nuclear progestin receptor in zebrafish follicle-enclosed oocytes. Molecular and Cellular Endocrinology. 2011;337:80–88. doi: 10.1016/j.mce.2011.02.004. [DOI] [PubMed] [Google Scholar]
- Huang P, Xiao A, Zhou M, Zhu Z, Lin S, Zhang BN. Heritable gene targeting in zebrafish using customized TALENs. Nature Biotechnology. 2011;29:699–700. doi: 10.1038/nbt.1939. [DOI] [PubMed] [Google Scholar]
- Huggard-Nelson DL, Nathwani PS, Kermouni A, Habibi HR. Molecular characterization of LH-β and FSH-β subunits and their regulation by estrogen in the goldfish pituitary. Molecular and Cellular Endocrinology. 2002;188:171–193. doi: 10.1016/s0303-7207(01)00716-x. [DOI] [PubMed] [Google Scholar]
- Kerrigan JR, Dalkin AC, Haisenleder DJ, Yasin M, Marshall JC. Failure of gonadotropin-releasing hormone (GnRH) pulses to increase luteinizing hormone beta messenger ribonucleic acid in GnRH-deficient female rats. Endocrinology. 1993;133:2071–2079. doi: 10.1210/endo.133.5.8404655. [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Sohn YC, Yoshiur Y, Aida K. Effects of sex steroids on the mRNA levels of gonadotropin subunits in juvenile and ovariectomized goldfish Carassius auratus. Fisheries Science. 2000;66:223–231. [Google Scholar]
- Kobayashi M, Sorensen PW, Stacey NE. Hormonal and pheromonal control of spawning behavior in the goldfish. Fish Physiology Biochemistry. 2002;26:71–84. [Google Scholar]
- Levavi-Sivan B, Biran J, Fireman E. Sex steroids are involved in the regulation of gonadotropin-releasing hormone and dopamine D2 receptors in female tilapia pituitary. Biology of Reproduction. 2006;75:642–650. doi: 10.1095/biolreprod.106.051540. [DOI] [PubMed] [Google Scholar]
- Lin SW, Ge W. Differential regulation of gonadotropins (FSH and LH) and growth hormone (GH) by neuroendocrine, endocrine, and paracrine factors in the zebrafish—an in vitro approach. General and Comparative Endocrinology. 2009;160:183–193. doi: 10.1016/j.ygcen.2008.11.020. [DOI] [PubMed] [Google Scholar]
- Miura T, Higuchi M, Ozaki Y, Ohta T, Miura C. Progestin is an essential factor for the initiation of the meiosis in spermatogenetic cells of the eel. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:7333–7338. doi: 10.1073/pnas.0508419103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miura T, Yamauchi K, Takahashi H, Nagahama Y. The role of hormones in the acquisition of sperm motility in salmonid fish. Journal of Experimental Zoology. 1992;261:359–363. doi: 10.1002/jez.1402610316. [DOI] [PubMed] [Google Scholar]
- Nóbrega RH, Morais RD, Crespo D, de Waal PP, de Franca LR, Schulz RW, Bogerd J. Fsh Stimulates Spermatogonial Proliferation and Differentiation in Zebrafish via Igf3. Endocrinology. 2015;156:3804–17. doi: 10.1210/en.2015-1157. [DOI] [PubMed] [Google Scholar]
- O’byrne KT, Thalabard JC, Grosser PM, Wilson RC, Williams CL, Chen MD, Ladendorf D, Hotchkiss J, Knobil E. Radiotelemetric Monitoring of Hypothalamic Gonadotropin-Releasing Hormone Pulse Generator Activity Throughout the Menstrual Cycle of the Rhesus Monkey. Endocrinology. 1991;129:1207–1214. doi: 10.1210/endo-129-3-1207. [DOI] [PubMed] [Google Scholar]
- Park D, Kim C, Kim K, Ryu K. Progesterone together with estradiol promotes luteinizing hormone β-subunit mRNA stability in rat pituitary cells cultured in vitro. European Journal of Endocrinology. 1996;134:236–242. doi: 10.1530/eje.0.1340236. [DOI] [PubMed] [Google Scholar]
- Pawlowski S, Van Aerle R, Tyler CR, Braunbeck T. Effects of 17α-ethinylestradiol in a fathead minnow (Pimephales promelas) gonadal recrudescence assay. Ecotoxicology and Environmental Safety. 2004;57:330–345. doi: 10.1016/j.ecoenv.2003.07.019. [DOI] [PubMed] [Google Scholar]
- Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nature Protocol. 2008;3:1101–1108. doi: 10.1038/nprot.2008.73. [DOI] [PubMed] [Google Scholar]
- Schneider JS, Burgess C, Sleiter NC, DonCarlos LL, Lydon JD, O’Malley B. Enhanced sexual behaviors and androgen receptor immunoreactivity in the male progesterone receptor knockout mouse. Endocrinology. 2005;146:4340–4348. doi: 10.1210/en.2005-0490. [DOI] [PubMed] [Google Scholar]
- Schulz RW, de França LR, Lareyre JJ, Gac FL, Garcia HC, Nobrega RH, Miura T. Spermatogenesis in fish. General and Comparative Endocrinology. 2010;165:390–411. doi: 10.1016/j.ygcen.2009.02.013. [DOI] [PubMed] [Google Scholar]
- Scott AP, Sumpter JP, Stacey N. The role of the maturation-inducing steroid, 17, 20β-dihydroxypregn-4-en-3-one, in male fishes: a review. Journal of Fish Biology. 2010;76:183–224. doi: 10.1111/j.1095-8649.2009.02483.x. [DOI] [PubMed] [Google Scholar]
- Simard J, Labrie C, Hubert JF, Labrie F. Modulation by sex steroids and [D-TRP6,Des-Gly-NH210] luteinizing hormone (LH)-releasing hormone ethylamide of α-subunit and LH β messenger ribonucleic acid levels in the rat anterior pituitary gland. Molecular Endocrinology. 1988;2:775–784. doi: 10.1210/mend-2-9-775. [DOI] [PubMed] [Google Scholar]
- Skaar KS, Nobrega RH, Magaraki A, Olsen LC, Schulz RW, Male R. Proteolytically activated, recombinant anti-Müllerian hormone inhibits androgen secretion, proliferation, and differentiation of spermatogonia in adult zebrafish testis organ cultures. Endocrinology. 2011;152:3527–3540. doi: 10.1210/en.2010-1469. [DOI] [PubMed] [Google Scholar]
- Skinner DC, Evans NP, Delaleu B, Goodman RL, Bouchard P, Caraty A. The negative feedback actions of progesterone on gonadotropin-releasing hormone secretion are transduced by the classical progesterone receptor. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:10978–10983. doi: 10.1073/pnas.95.18.10978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sleiter N, Pang Y, Park C, Horton TH, Dong J, Thomas P, Levine JE. Progesterone receptor A (PRA) and PRB-independent effects of progesterone on gonadotropin-releasing hormone release. Endocrinology. 2009;150:3833–3844. doi: 10.1210/en.2008-0774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- So WK, Kwok HF, Ge W. Zebrafish gonadotropins and their receptors: II. Cloning and characterization of zebrafish follicle-stimulating hormone and luteinizing hormone subunits—their spatial-temporal expression patterns and receptor specificity. Biology of Reproduction. 2005;72:1382–1396. doi: 10.1095/biolreprod.104.038216. [DOI] [PubMed] [Google Scholar]
- Stacey NE, Sorensen PW. 17α, 20β-dihydroxy-4-pregnen-3-one: a steroidal primer pheromone increasing milt volume in the goldfish, Carassius auratus. Canadian Journal of Zoology. 1986;64:2412–2417. [Google Scholar]
- Tan W, Thomas P. Involvement of epidermal growth factor receptors and mitogen-activated protein kinase in progestin-induction of sperm hypermotility in Atlantic croaker through membrane progestinreceptor-alpha. Molecular and Cellular Endocrinology. 2015;414:194–201. doi: 10.1016/j.mce.2015.06.023. [DOI] [PubMed] [Google Scholar]
- Thackray VG, Hunnicutt JL, Memon AK, Ghochani Y, Mellon PL. Progesterone Inhibits basal and gonadotropin-releasing hormone induction of luteinizing hormone β-subunit gene expression. Endocrinology. 2009;150:2395–2403. doi: 10.1210/en.2008-1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thackray VG, McGillivray SM, Mellon PL. Androgens, progestins, and glucocorticoids induce follicle-stimulating hormone β-subunit gene expression at the level of the gonadotrope. Molecular Endocrinology. 2006;20:2062–2079. doi: 10.1210/me.2005-0316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas P. Characteristics of membrane progestin receptor alpha (mPRα) and progesterone membrane receptor component 1 (PGMRC1) and their roles in mediating rapid progestin actions. Frontiers in Neuroendocrinology. 2008;29:292–312. doi: 10.1016/j.yfrne.2008.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tubbs C, Thomas P. Functional characteristics of membrane progestin receptor alpha (mPRα) subtypes: a review with new data showing mPRα expression in seatrout sperm and its association with sperm motility. Steroids. 2008;73:935–941. doi: 10.1016/j.steroids.2007.12.022. [DOI] [PubMed] [Google Scholar]
- Turgeon JL, Kimura Y, Waring DW, Mellon PL. Steroid and pulsatile gonadotropin-releasing hormone (GnRH) regulation of luteinizing hormone and GnRH receptor in a novel gonadotrope cell line. Molecular Endocrinology. 1996;10:439–450. doi: 10.1210/mend.10.4.8721988. [DOI] [PubMed] [Google Scholar]
- Turgeon JL, Waring DW. Rapid Augmentation by Progesterone of Agonist-Stimulated Luteinizing Hormone Secretion by Cultured Pituitary Cells. Endocrinology. 1990;127:773–780. doi: 10.1210/endo-127-2-773. [DOI] [PubMed] [Google Scholar]
- Turgeon JL, Waring DW. Progesterone Regulation of the Progesterone Receptor in Rat Gonadotropes 1. Endocrinology. 2000;141:3422–3429. doi: 10.1210/endo.141.9.7688. [DOI] [PubMed] [Google Scholar]
- Ueda H, Kambegawa A, Nagahama Y. Involvement of gonadotrophin and steroid hormones in spermiation in the amago salmon, Oncorhynchus rhodurus, and goldfish, Carassius auratus. General and Comparative Endocrinology. 1985;59:24–30. doi: 10.1016/0016-6480(85)90415-0. [DOI] [PubMed] [Google Scholar]
- Van den Belt K, Berckmans P, Vangenechten C, Verheyen R, Witters H. Comparative study on the in vitro/in vivo estrogenic potencies of 17β-estradiol, estrone, 17α-ethynylestradiol and nonylphenol. Aquatic Toxicology. 2004;66:183–195. doi: 10.1016/j.aquatox.2003.09.004. [DOI] [PubMed] [Google Scholar]
- Van der Kraak G. The GnRH system and the neuroendocrine regulation of reproduction. Fish Physiology. 2009;28:115–149. [Google Scholar]
- Van der Ven L, Van den Brandhof EJ, Vos JH, Wester PW. Effects of the estrogen agonist 17β-estradiol and antagonist tamoxifen in a partial life-cycle assay with zebrafish (Danio rerio) Environmental Toxicology Chemistry. 2007;26:92–99. doi: 10.1897/06-092r1.1. [DOI] [PubMed] [Google Scholar]
- Van der Ven L, Wester PW, Vos JG. Histopathology as a tool for the evaluation of endocrine disruption in zebrafish (Danio rerio) Environmental Toxicology Chemistry. 2003;22:908–913. [PubMed] [Google Scholar]
- Waring DW, Turgeon JL. A pathway for luteinizing hormone releasing-hormone self-potentiation: cross-talk with the progesterone receptor. Endocrinology. 1992;130:3275–3282. doi: 10.1210/endo.130.6.1317780. [DOI] [PubMed] [Google Scholar]
- Westerfield M. The zebrafish book: a guide for the laboratory use of zebrafish (Danio rerio) University of Oregon Press; 2000. [Google Scholar]
- Zhang Z, Lau SW, Zhang L, Ge W. Disruption of Zebrafish Follicle-Stimulating Hormone Receptor (fshr) But Not Luteinizing Hormone Receptor (lhcgr) Gene by TALEN Leads to Failed Follicle Activation in Females Followed by Sexual Reversal to Males. Endocrinology. 2015a;156:3747–62. doi: 10.1210/en.2015-1039. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Zhu B, Ge W. Genetic analysis of zebrafish gonadotropin (FSH and LH) functions by TALEN-mediated gene disruption. Molecular Endocrinology. 2015b;29:76–98. doi: 10.1210/me.2014-1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Hanna RN, Schaaf MJM, Spaink HP, Thomas P. Candidates for membrane progestin receptors—past approaches and future challenges. Comparative Biochemistry & Physiology Part C Toxicology & Pharmacology. 2008;148:381–389. doi: 10.1016/j.cbpc.2008.05.019. [DOI] [PubMed] [Google Scholar]
- Zhu Y, Liu D, Shaner ZC, Chen S, Hong W, Stellwag EJ. Nuclear progestin receptor (Pgr) knockouts in zebrafish demonstrate role for Pgr in ovulation but not in rapid non-genomic steroid mediated meiosis resumption. Frontiers in Endocrinology. 2015;6:37. doi: 10.3389/fendo.2015.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Rice CD, Pang Y, Pace M, Thomas P. Cloning, expression, and characterization of a membrane progestin receptor and evidence it is an intermediary in meiotic maturation of fish oocytes. Proceedings of the National Academy of Sciences of the United States of America. 2003;100:2231–2236. doi: 10.1073/pnas.0336132100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zohar Y, Muñoz-Cueto JA, Elizur A, Kah O. Neuroendocrinology of reproduction in teleost fish. General and Comparative Endocrinology. 2010;165:438–455. doi: 10.1016/j.ygcen.2009.04.017. [DOI] [PubMed] [Google Scholar]