Abstract
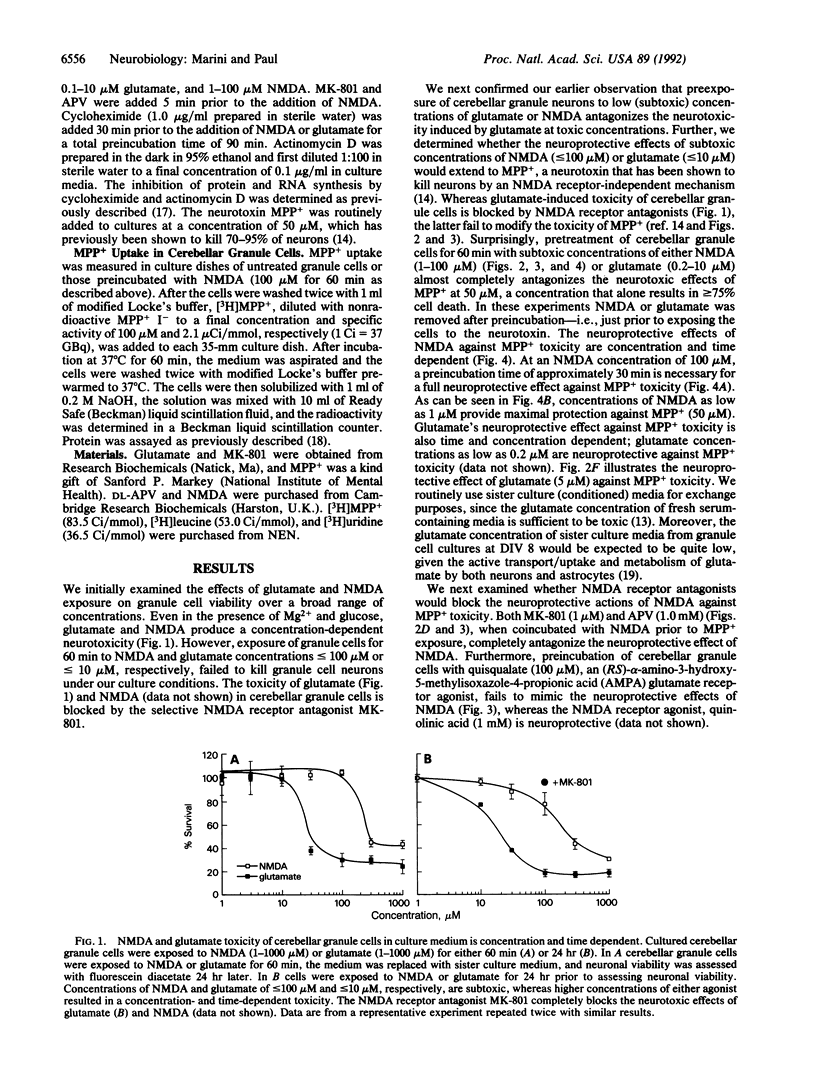
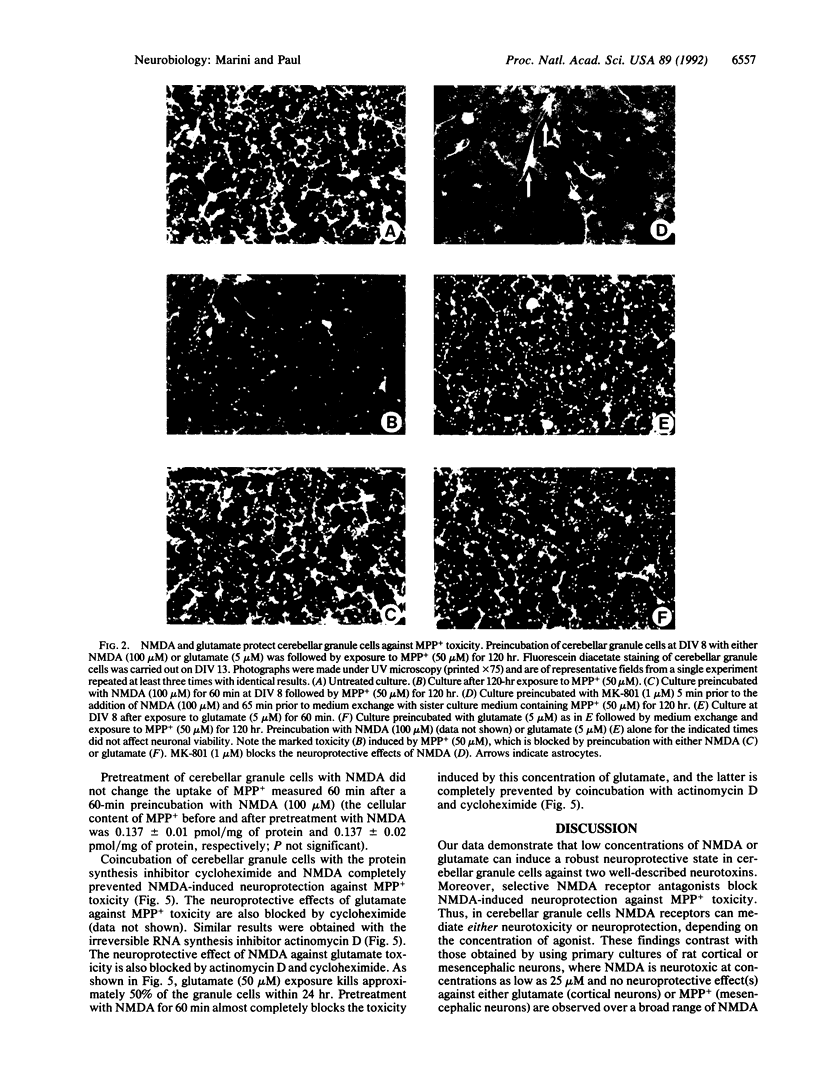
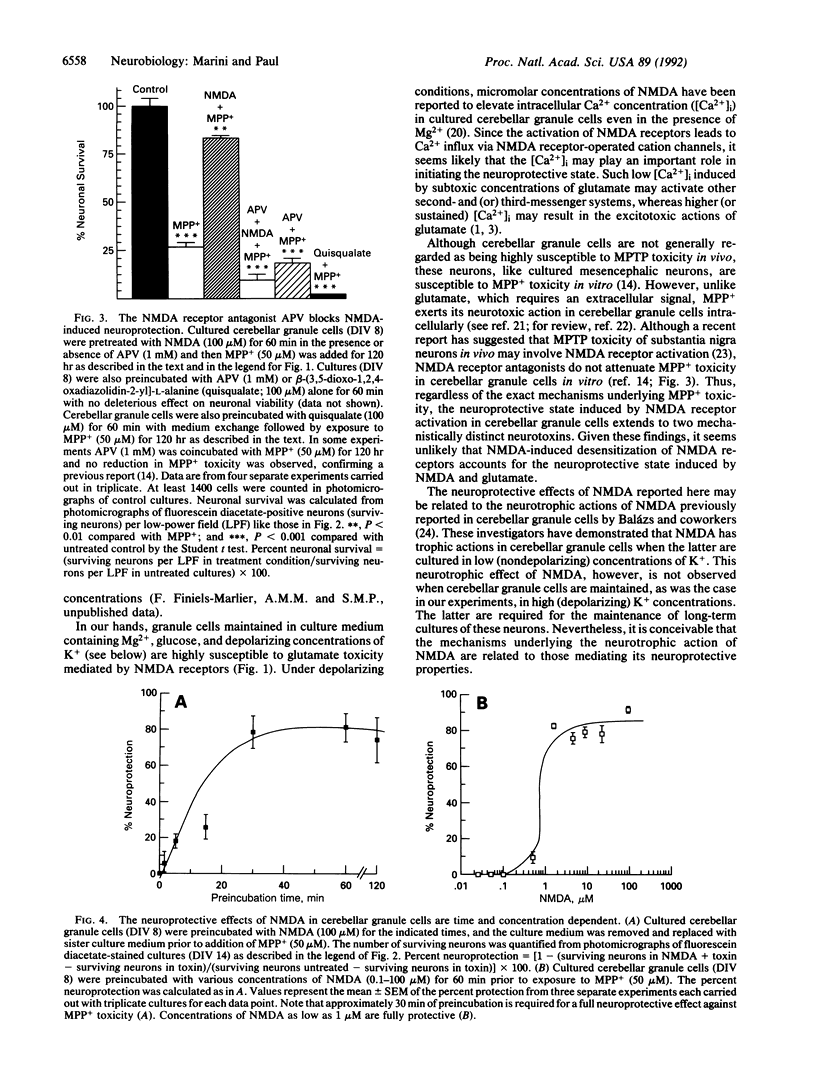
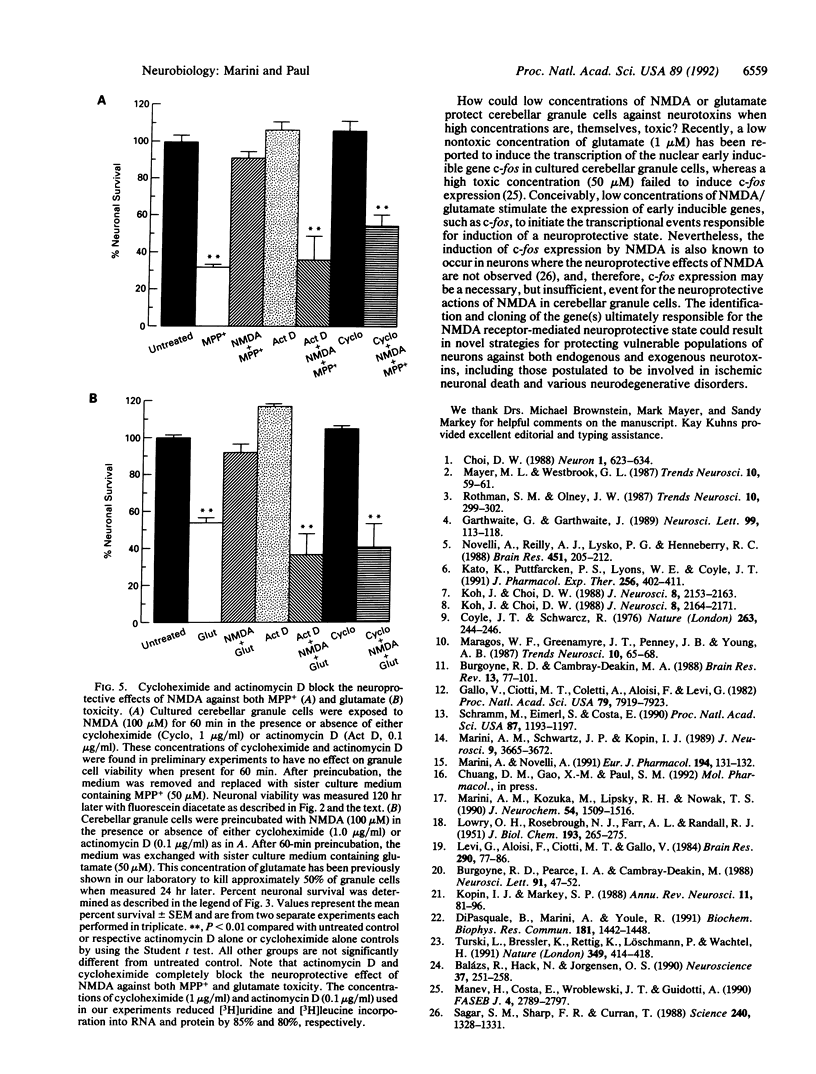
Cerebellar granule cells are susceptible to the excitotoxin glutamate, which acts at N-methyl-D-aspartate (NMDA) receptors, as well as the neurotoxin 1-methyl-4-phenylpyridinium ion (MPP+), the active cytotoxic metabolite of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Paradoxically, preincubation of cultured cerebellar granule cells with low concentrations of NMDA or glutamate markedly antagonizes the neurotoxicity resulting from subsequent exposure to toxic concentrations of either MPP+ or glutamate. The neuroprotective effects of NMDA and glutamate against MPP+ toxicity are observed at agonist concentrations as low as 1 microM, are blocked by specific NMDA receptor antagonists, and require at least 30 min to develop fully. Moreover, NMDA receptor-mediated neuroprotection is prevented by the RNA synthesis inhibitor actinomycin D or the protein synthesis inhibitor cycloheximide. Thus, in cerebellar granule cells activation of NMDA receptors by glutamate can result in either neurotoxicity or neuroprotection, depending on the apparent degree of receptor stimulation. NMDA receptor-mediated neuroprotection requires new RNA and protein synthesis and therefore appears to be mediated by the expression of a neuroprotective protein(s). These data demonstrate the presence of an active NMDA receptor-mediated and transcriptionally directed neuroprotective mechanism in cerebellar granule cells.
Full text
PDF
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Balázs R., Hack N., Jørgensen O. S. Selective stimulation of excitatory amino acid receptor subtypes and the survival of cerebellar granule cells in culture: effect of kainic acid. Neuroscience. 1990;37(1):251–258. doi: 10.1016/0306-4522(90)90211-l. [DOI] [PubMed] [Google Scholar]
- Burgoyne R. D., Pearce I. A., Cambray-Deakin M. N-methyl-D-aspartate raises cytosolic calcium concentration in rat cerebellar granule cells in culture. Neurosci Lett. 1988 Aug 15;91(1):47–52. doi: 10.1016/0304-3940(88)90247-9. [DOI] [PubMed] [Google Scholar]
- Choi D. W. Glutamate neurotoxicity and diseases of the nervous system. Neuron. 1988 Oct;1(8):623–634. doi: 10.1016/0896-6273(88)90162-6. [DOI] [PubMed] [Google Scholar]
- Coyle J. T., Schwarcz R. Lesion of striatal neurones with kainic acid provides a model for Huntington's chorea. Nature. 1976 Sep 16;263(5574):244–246. doi: 10.1038/263244a0. [DOI] [PubMed] [Google Scholar]
- Dipasquale B., Marini A. M., Youle R. J. Apoptosis and DNA degradation induced by 1-methyl-4-phenylpyridinium in neurons. Biochem Biophys Res Commun. 1991 Dec 31;181(3):1442–1448. doi: 10.1016/0006-291x(91)92101-o. [DOI] [PubMed] [Google Scholar]
- Gallo V., Ciotti M. T., Coletti A., Aloisi F., Levi G. Selective release of glutamate from cerebellar granule cells differentiating in culture. Proc Natl Acad Sci U S A. 1982 Dec;79(24):7919–7923. doi: 10.1073/pnas.79.24.7919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garthwaite G., Garthwaite J. Quisqualate neurotoxicity: a delayed, CNQX-sensitive process triggered by a CNQX-insensitive mechanism in young rat hippocampal slices. Neurosci Lett. 1989 Apr 24;99(1-2):113–118. doi: 10.1016/0304-3940(89)90274-7. [DOI] [PubMed] [Google Scholar]
- Kato K., Puttfarcken P. S., Lyons W. E., Coyle J. T. Developmental time course and ionic dependence of kainate-mediated toxicity in rat cerebellar granule cell cultures. J Pharmacol Exp Ther. 1991 Jan;256(1):402–411. [PubMed] [Google Scholar]
- Koh J. Y., Choi D. W. Vulnerability of cultured cortical neurons to damage by excitotoxins: differential susceptibility of neurons containing NADPH-diaphorase. J Neurosci. 1988 Jun;8(6):2153–2163. doi: 10.1523/JNEUROSCI.08-06-02153.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koh J. Y., Choi D. W. Zinc alters excitatory amino acid neurotoxicity on cortical neurons. J Neurosci. 1988 Jun;8(6):2164–2171. doi: 10.1523/JNEUROSCI.08-06-02164.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopin I. J., Markey S. P. MPTP toxicity: implications for research in Parkinson's disease. Annu Rev Neurosci. 1988;11:81–96. doi: 10.1146/annurev.ne.11.030188.000501. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Levi G., Aloisi F., Ciotti M. T., Gallo V. Autoradiographic localization and depolarization-induced release of acidic amino acids in differentiating cerebellar granule cell cultures. Brain Res. 1984 Jan 2;290(1):77–86. doi: 10.1016/0006-8993(84)90737-6. [DOI] [PubMed] [Google Scholar]
- Manev H., Costa E., Wroblewski J. T., Guidotti A. Abusive stimulation of excitatory amino acid receptors: a strategy to limit neurotoxicity. FASEB J. 1990 Jul;4(10):2789–2797. doi: 10.1096/fasebj.4.10.2165013. [DOI] [PubMed] [Google Scholar]
- Marini A. M., Kozuka M., Lipsky R. H., Nowak T. S., Jr 70-kilodalton heat shock protein induction in cerebellar astrocytes and cerebellar granule cells in vitro: comparison with immunocytochemical localization after hyperthermia in vivo. J Neurochem. 1990 May;54(5):1509–1516. doi: 10.1111/j.1471-4159.1990.tb01198.x. [DOI] [PubMed] [Google Scholar]
- Marini A. M., Schwartz J. P., Kopin I. J. The neurotoxicity of 1-methyl-4-phenylpyridinium in cultured cerebellar granule cells. J Neurosci. 1989 Oct;9(10):3665–3672. doi: 10.1523/JNEUROSCI.09-10-03665.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marini A., Novelli A. DL-threo-3-hydroxyaspartate reduces NMDA receptor activation by glutamate in cultured neurons. Eur J Pharmacol. 1991 Feb 26;194(1):131–132. doi: 10.1016/0014-2999(91)90136-e. [DOI] [PubMed] [Google Scholar]
- Novelli A., Reilly J. A., Lysko P. G., Henneberry R. C. Glutamate becomes neurotoxic via the N-methyl-D-aspartate receptor when intracellular energy levels are reduced. Brain Res. 1988 Jun 7;451(1-2):205–212. doi: 10.1016/0006-8993(88)90765-2. [DOI] [PubMed] [Google Scholar]
- Sagar S. M., Sharp F. R., Curran T. Expression of c-fos protein in brain: metabolic mapping at the cellular level. Science. 1988 Jun 3;240(4857):1328–1331. doi: 10.1126/science.3131879. [DOI] [PubMed] [Google Scholar]
- Schramm M., Eimerl S., Costa E. Serum and depolarizing agents cause acute neurotoxicity in cultured cerebellar granule cells: role of the glutamate receptor responsive to N-methyl-D-aspartate. Proc Natl Acad Sci U S A. 1990 Feb;87(3):1193–1197. doi: 10.1073/pnas.87.3.1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turski L., Bressler K., Rettig K. J., Löschmann P. A., Wachtel H. Protection of substantia nigra from MPP+ neurotoxicity by N-methyl-D-aspartate antagonists. Nature. 1991 Jan 31;349(6308):414–418. doi: 10.1038/349414a0. [DOI] [PubMed] [Google Scholar]




