Abstract
This review highlights the principal effects of steroid hormones at central and peripheral levels in the neuroendocrine axis. The data discussed here points out the principal role of estrogens and testosterone in hormonal programming in relation to sexual orientation, reproductive and metabolic programming, and in the neuroendocrine mechanism involved in development of polycystic ovary syndrome phenotype. Moreover, consistent with the wide range of processes in which steroid hormones take part, we discuss the protective effects of progesterone on neurodegenerative disease and the signaling mechanism involved in the genesis of estrogen-induced pituitary prolactinomas.
Keywords: Hormonal Programming, Estrogens, Androgens, Steroid Hormones, Prolactin
Introduction
Hormones influence many biological processes throughout the lifespan, and have the potential to cause permanent tissue-specific alterations in anatomy and physiology during important developmental periods. Hormones exert transient effects in adult animals that activate or facilitate physiological processes or behaviors while the hormone is present and permanent effects that act during development to program the pattern and extent of adult responses and contribute to sex differences. The role of steroid hormones in organizational and activational processes participating in reproductive anatomy is well described. These processes affect sexual differences of the neuroendocrine system and behavior. Organization refers to the actions of hormones in critical periods in early development obtaining permanent establishment of sexual phenotypes, sexual genitalia and future potential for masculine and feminine mating behavior (1–5). Activation is related to the high levels of hormones in puberty or adulthood, linked with changes in masculine and feminine anatomy, as well as breeding behavior depending the season of the year (1–5).
The developmental and programming effects of steroid hormones on the brain only occur during a sensitive period in early life, commonly referred to as the critical period. This period is in close relation with the theory of organizational effects of steroid hormones, is an empirical concept that differs among species with regard to timing and brain functions involved (6–11). These early effects are permanent and act to hardwire the brain.
Steroid hormones are lipophilic molecules utilized as chemical messengers by organisms, in which they act on a wide range of tissues and biological functions (6,12). A number of pathological states arise because of problems related to steroid hormone action. These include cancer, steroid insensitivity, abnormal fertility, endocrine alterations. Steroid hormones derive from a common cholesterol precursor (Cholestane C27). There are four major types of steroids: progestins, androgens, estrogens, and corticoids. The rate-limiting step in the synthesis of steroid hormones is the transport of free cholesterol (C27) from the cytoplasm into mitochondria, which is controlled by Steroidogenic Acute Regulatory Protein (STAR). The enzymatic step from cholesterol (C27) to pregnenolone (C21) (the common branch point for synthesis of progestins, corticoids, androgens, and therefore estrogens) is the limiting step for steroidogenesis once cholesterol is inside the mitochondria. Pregnenolone is subsequently converted to progesterone by 3β-hydroxysteroid dehydrogenase (3β-HSD). Progesterone is generated in the ovary, the adrenal gland, the placenta during pregnancy and the nervous system, in which it plays an important role as a neurosteroid. This steroid is the principal intermediate for circulating androgens and estrogens. (13–17).
Leydig cells of the testes, thecal cells of the ovary, and cells in the reticularis region of the adrenals are responsible for androgen synthesis and secretion (7,11,18). Luteinizing hormone (LH) enhances the synthesis in the testes of testosterone (T). T can be reduced by 5α-reductase to yield subsequently a more active metabolite: 5α-dihydrotestosterone (DHT). This process takes part mainly in the target tissues. In some target tissues, T and androstenedione can also be transformed into the estrogens, such as 17β-estradiol (E2) and estrone by the cytochrome P450 enzyme aromatase (19).
In females, estrogens and progestins are synthesized and secreted principally by maturing ovarian follicles, corpora lutea and during pregnancy, the placenta. The dominant estrogen secreted is E2. The follicle is composed of the primary oocyte surrounded by granulosa cells and theca cells. The granulosa cells are responsible for producing estrogens from androgen precursors synthesized from the theca cells; this hormone activates estrogen receptors (ERs) in target cells exerting their effects slowly, as a typical steroid receptor. Although most characterized effects are mediated via nuclear receptors and genomic pathways, there are many examples of very rapid, nongenomic effects of steroids (20,21). Classical ERs present in the cell nucleus (i.e. ERα and ERβ) act as ligand-activated transcriptional regulators, whereas ERs present in the cell membrane and cytoplasm regulate various intracellular signaling pathways and can converge with nuclear ERs to exert genotropic effects. Putative mERs include membrane-associated splice variants of ERα (mERα) and ERβ (mERβ) as well as G-protein receptors such as GPR30 and Gq-mER (20,21).
The mechanisms of action for different steroid hormones are relatively similar in the different target tissues. In the physiological situation of low amounts of hormone, classical ER, androgen (AR), and progesterone (PR) receptors are principally localized in the nucleus (22), whereas glucocorticoid receptors (GR) are located in the cytoplasm. Steroid hormones move passively from the circulation and interstitial spaces across cell membranes, and bind to and activate nuclear steroid receptor proteins. Then the hormone-receptor binds to specific short DNA sequences in the promoter region of genes (i.e., hormone response elements) to enhance or repress the transcription of genes.
Steroid hormones are implicated in many biological processes including development, hypothalamic programming, sexual differentiation, reproductive physiology, behavior, osmoregulation, metabolism, regulation of the hypothalamic-pituitary-gonadal axis, and hypothalamic-pituitary-adrenal axis (6,16,23–26).
In this review, the programming effect of steroid hormones during prenatal development will be discussed. In particular, we will analyze the effects of prenatal steroids on epigenetic programming, and their impact in pathologies such as polycystic ovarian disease, and their role in behavioral processes like sexual partner preferences. Furthermore, we will discuss the protective effects of progesterone in the neurodegenerative disease amyotrophic lateral sclerosis (ALS) and consider how the Notch signaling pathway may mediate the formation of estrogen-induced prolactinomas.
I- Steroid Hormones and Programming
Estrogen
Developmental and Programming Effects of Steroids During Rat Ovary Development
Exposure to high levels of steroidal hormones disrupts normal endocrine function and decreases fertility in mammals including humans, especially when the exposure occurs during critical periods of vulnerability during development.
The established view holds that reproductive function is regulated through the integration of information that comes from the hypothalamus, hypophysis, and ovaries, and that gonadotropins modulate folliculogenesis and steroidogenesis in the ovary (27). Numerous studies performed to understand the neural circuits and molecular mechanisms that regulate GnRH release and steroid feedback demonstrate important roles for classical steroid receptors, membrane steroids receptors and neurosteroids in the hypothalamus (28–30).
Parallel to the endocrine control of reproductive function, experimental evidence indicates that there is complementary regulation through the hypothalamus-celiac ganglion-ovary axis (31). The primary neurotransmitter acting in the ovary is noradrenaline (NA), which is released from neuron terminals originating in the celiac ganglia and acting on the thecal layer of ovarian follicles (32).
In mammals, ovarian folliculogenesis starts with the formation of primordial follicles, a process known as nest breakdown, which allows the oocytes to be surrounded by a layer of somatic cells, thus forming the primordial follicles in a process known as follicular assembly (33). In humans, this process occurs during the third trimester of gestation, whereas in rats it occurs between 24 and 72 hours after birth (25,34–36). Once follicular development begins, it continues throughout postnatal life in both species. During this time the oocyte enlarges while the granulosa and theca cells proliferate, increasing the layers of cells surrounding the oocyte. This proliferative phase ends as follicular fluid begins to accumulate and the antral cavity forms (33,34). Each of the different steps of follicular development is controlled by different endocrine and paracrine factors (gonadotropins, growth factors, and steroidal hormones); making this process vulnerable to hormonal changes induced by external factors (37).
The growing incidence of infertility and reproductive disorders in humans and wildlife has alerted many researchers to the influence that products with estrogenic activity that are produced and released into the environment have over reproductive function (38). The molecules that mimic or block hormonal activity are known as endocrine disruptors (EDs). They may be synthetic or natural in origin and can alter homeostasis and the hormonal system, either by environmental exposure or by inappropriate exposure during development (35). Exposure to EDs during the sensitive periods can alter the normal development of the ovary, causing alterations in morphological and follicular development and malfunctions during the adult period (39–43). These alterations in the rat ovary can be inherited by the next generation through changes in the pattern of DNA methylation, because cellular differentiation of the rat ovary begins around the time of birth. The germ cell re-methylation is initiated during the postnatal period and continues throughout the oocyte growth period until the preantral follicle stage (34,43,44). Regardless of the source of hormones or EDs during this period, they would alter the normal development of the offspring due to a reprogramming of the genes.
There are several pathological conditions in which the hormonal environment is altered during development, such as adrenal hyperplasia, obesity, and polycystic ovary syndrome (PCOS) (45,46). PCOS is a complex endocrine disorder characterized by hyperandrogenism, ovulatory/menstrual irregularity, and polycystic ovaries, which affects 5–10% of women of reproductive age (47). Women with PCOS exhibit a significant increase in androgen concentrations during pregnancy (48). An important proportion of the first-degree female relatives of women with PCOS have been shown to be at risk for developing PCOS (49). In fact, in comparison with control girls, PCOS girls exhibit higher levels of anti-Müllerian hormone (AMH), a marker of growing follicles, beginning at the peripubertal stage (50,51). It has been proposed that this inheritance is not the result of a genetic condition, but is due to fetal programming (47–52). Supporting this, experimental treatment of PCOS gestating mothers with the insulin sensitizer drug metformin improved the altered endocrine-metabolic environment of the PCOS mothers and the AMH levels in their daughters, suggesting the follicular alterations described in adult PCOS women may appear early during development (53). This is supported by several studies in animal models that have demonstrated a relationship between programmed polycystic ovary (PCO) morphology during adulthood and prenatal or neonatal exposure to endocrine-disrupting compounds such as estrogens or aromatizable androgens (41,54–56).
The administration of a single dose of estradiol valerate (EV) to neonatal rats (at 12 hours postnatal) induces early vaginal opening, disrupted cyclicity, appearance of a PCO phenotype, absence of corpus luteum, and infertility (41). In addition, this exposure decreases the total number of ovarian follicles mainly due to a reduced number of primordial follicles suggesting that estradiol acts in the first stages of folliculogenesis when primordial follicles are organizing and reprogramming the genes that control ovarian function (42,57). At the molecular level, AMH expression is increased in the ovary of these rats when adults. In contrast to AMH expression, AR expression in granulosa cells decreased at the same stage of development suggesting that the regulatory region of AR and AMH genes could be involved. These results have been confirmed by protein expression data found by immunohistochemistry. In summary, these data suggest that estradiol exposure during the neonatal critical period reprograms AR and AMH expression in the ovary possibly through epigenetic mechanisms that become evident in the adult period, when the full PCO phenotype is acquired (57).
Testosterone
Prenatal Programming of Sexual Partner Preferences: The Ram Model
Mammals are exposed to gonadal, placental and maternal hormones during early development. Hormones must be maintained within an appropriate range over time to program proper development of the reproductive axis and adult behavioral responses (58). Males develop in an environment of elevated T secreted by the fetal testes that acts to masculinize and defeminize brain structures, physiological processes and behaviors. Differences in T concentrations and sensitivity occur naturally between individuals and can result from environmental challenges during pregnancy. The question of whether the occurrence of same sex partner preferences originates from variations in the prenatal hormonal environment has been studied in using a unique ram model (59).
Domestic Rams Display Variations in Sexual Partner Preferences
Domestic sheep are one of the few mammals apart from humans that exhibit exclusive and durable same sex partner preferences. Approximately 8–10% of domestic rams exhibit a sexual preference for other rams i.e., male-oriented rams (60–63). These rams not only mount, but also direct all courtship activities (anal-genital smelling, kicks, nibbles) to other rams, while their sexual interest and activity towards females is extremely low or nonexistent. Several hypotheses have been proposed to explain the development of same-sex preferences in rams. These include effects attributed to same-sex rearing, genes, olfactory responsiveness and brain differences (59). Although none of these mechanisms has been extensively investigated, the most compelling evidence supports the idea that this behavior is partially attributable to brain differences.
Medial Preoptic Area
The medial preoptic area is essential for mating behavior in vertebrates (64). Thus, this structure has been of great interest in searching for anatomic differences that could be causally related to sexual preferences. In rats, there is a cluster of neurons in the medial preoptic area, called the sexually dimorphic nucleus of the preoptic area (SDN-POA), which is 5 to 7 times larger in males than in females (65). The SDN-POA is part of a forebrain circuit that integrates sensory cues with hormonal status to modulate sexual behavior. The larger volume of the male SDN-POA is correlated to the higher concentration of fetal and neonatal T levels in males than in females. Males castrated at birth have much smaller SDN-POAs in adulthood, whereas females treated with T perinatally have larger male-like SDN-POAs as adults. Also, there is evidence that conversion of T to estradiol by the aromatase enzyme is required to masculinize the SDN-POA (66). Much like in rats, sheep have a homologue of the SDN-POA, called the ovine SDN (oSDN) that is twice as large in rams than in ewes (62). The oSDN is a dense cluster of cells comprising the central component of the medial preoptic nucleus that can be identified by Nissl staining and by abundant expression of aromatase mRNA. Moreover, it is larger in female-oriented rams than in male-oriented rams and does not differ between male-oriented rams and ewes. The differences in volume persist even after adults are gonadectomized and treated with T, demonstrating they are most likely the result of the organizational actions of gonadal steroids occurring during fetal development in sheep (67).
Brains Of Straight And Gay Men Differ
The observation that male-oriented rams have a smaller oSDN than female-oriented rams is reminiscent of the anatomical difference observed between the brains of gay and straight men (68). The sexually dimorphic nucleus identified in the hypothalamus of humans is called the 3rd interstitial nucleus of the anterior hypothalamus (INAH3). The INAH3 is twice as large in straight men than in gay men and women. These cross-species results are some of the strongest evidence that sexual partner preferences and sexual orientation are regulated at the level of the hypothalamus-preoptic area. But neither study can address the question of whether the difference is the cause or consequence of the behavior. In fact, for obvious ethical reasons this question can only be experimentally tested using an animal model.
Which Comes First?
If the smaller size of the oSDN causes males to be attracted to other males, the difference in size should be present prior to expression of the behavior. Ideally, the volume of the oSDN should be measured over time as the animals become more sexually experienced in order to determine whether size differences emerge before sexual preferences are expressed. In lieu of this approach, which is not technically feasible, it can be asked whether the oSDN develops before animals have social experiences, i.e. prenatally. Masculinization of sexual behavior in sheep occurs during a critical period that begins shortly after the testes differentiate at gestational day (GD)30 and persists until about GD90 (term pregnancy in sheep is ~150 days)(69). The oSDN is clearly apparent after this critical period in lamb fetuses (GD135) and is twice as large in males as in females (70). The larger oSDN in males correlates with higher levels of T during the critical period. Thus, development of the oSDN occurs independently from sexual experiences and prior to expression of sexual preferences.
Experiments were performed to directly test whether T exposure determines oSDN volume in late gestation fetuses (70). Biweekly maternal treatment with T from GD30 to 90 significantly enlarged the fetal oSDN in females, but had no effect in males. Coincident with this, T-exposed females exhibited masculinized genitalia consisting of a pseudopenis and empty scrotum. These results show that in sheep, like rats, T acting during the critical period for sexual differentiation masculinizes both the brain and external genitalia. Typically, the sex of the brain matches the genitals. But male-oriented rams have male genitals and a female-typical oSDN suggesting that separate critical periods for the genitals and the brain might exist within the broad period of sexual differentiation. To test this possibility, biweekly maternal treatments with T were administered from GD30 to 60 (early T) and GD60 to 90 (late T) (71). Early T masculinized the genitalia of genetic females, but had no effect on the fetal oSDN. Conversely, late T masculinized the fetal oSDN of females and had no effect on the genitalia. Neither maternal treatment significantly affected male fetuses. These results demonstrate that T affects differentiation of the brain and genitals in different timeframes. Individual critical periods exist for other sexually dimorphic traits in sheep, such as urination posture, the LH surge mechanism and the timing of puberty. Distinct temporal requirements for T action or different sensitivities to T metabolites could explain how hormone variations during gestation can produce rams that prefer to mate with other rams but still possess normal masculine genitals and other typical male neuroendocrine traits.
Is Aromatization Of T to E2 Needed For Masculinization Of The SDN?
The fetal oSDN is characterized by an abundant expression of aromatase and ERα suggesting that like in rodents, sheep brain masculinization and defeminization may require conversion of T to estradiol. However, daily treatment of mothers with the aromatase inhibitor androstatrienedione (ATD) had no effects on the sexual preferences and oSDN volumes of adult offspring (72,73). Nor did ATD-exposed rams show LH surge responses or behavioral receptivity in response to estradiol injections. The only difference was that ATD-exposed rams showed lower mounting activity than controls when they reached 18 months of age. Thus, it appears that estrogens are not essential for masculinization and defeminization of sheep brain. These results raise the question of whether masculinization of mate preferences and oSDN volume are controlled entirely through an androgen receptor mechanism or through the combined effects of both androgens and estrogens.
Is Androgen Activity Responsible for Differentiation of Male oSDN?
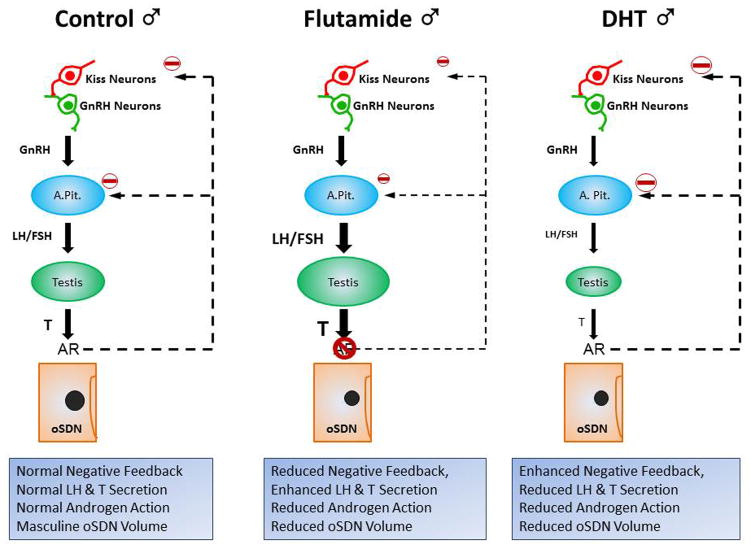
Androgen receptors are also expressed in the developing fetal oSDN (74). Experiments were performed to test the involvement of androgen receptors in the sexual differentiation of sheep brain (75). If androgen receptors mediate masculinization, fetal exposure to the antiandrogen flutamide should reduce the size of the oSDN in males, but not in females. On the other hand, exposure to the androgen agonist DHT should increase the size of the oSDN in females, but not in males. Maternal flutamide treatment from GD 60 to 90 significantly reduced mean oSDN volumes in GD135 male fetuses. Paradoxically, maternal DHT treatment during the same period also significantly reduced the oSDN volume of males. Neither treatment affected females. In a second experiment, fetuses were delivered during the final maternal treatments (GD85) to analyze whether these treatments elicited compensatory hormonal responses from the hypothalamic-pituitary-gonadal axis. Exposure to flutamide blocked negative feedback in males resulting in elevated serum levels of LH and T, but had no effect in females. Thus, flutamide exerts sufficient androgen receptor antagonism to reduce oSDN volume even when T is elevated. Exposure to DHT significantly suppressed LH concentrations in males and females and reduced T concentrations in males. These results demonstrate that DHT exerts negative feedback on the pituitary and consequently inhibits T secretion, which can explain why the oSDN volume was reduced in males and unaffected in females. Apparently the levels of DHT achieved in the fetal circulation were insufficient to support masculinization of the oSDN. These results provide convincing evidence that the prenatal program that masculinizes the oSDN in eugonadal male fetuses acts through the androgen receptor. However, it is also apparent that hypothalamic and pituitary are tonically suppressed in males during the gestational critical period for oSDN masculinization and respond to disruptions in androgen action with an opposing hormone response intended to stabilize the endocrine milieu (Figure 1). It is not yet known whether flutamide exposure exerts sufficient androgen receptor antagonism to alter male-typical sexual partner preference despite the compensatory increase in circulating T.
Figure 1.
Response of male fetal hypothalamic-pituitary-testis axis to vehicle (Control), androgen antagonist flutamide and androgen agonist dihydrotestosterone (DHT). Pregnant ewes received injections of vehicle, flutamide and DHT from gestation day (GD) 60 to 84 and fetuses were delivered on GD85 for evaluation. In control eugonadal male fetuses, LH secretion is tonically suppressed by androgens at this age to regulate normal testosterone (T) secretion driving normal androgen action which, in turn, masculinizes the ovine sexually dimorphic nucleus (oSDN). Exposure to flutamide blocks negative feedback resulting in elevated LH concentrations, enlarged testis & increased testicular T secretion. The increased T competes with the competitive antagonist flutamide for the androgen receptor and leads to partial, instead of full, inhibition of oSDN masculinization. Exogenous DHT exposure enhances negative feedback further suppressing LH secretion leading to smaller testis and reduced levels of testicular T. The result of DHT treatment is reduced exposure of the developing SDN to endogenous T and incomplete masculinization. Both results are consistent with a role for androgen receptor activation in the masculinization of the ovine SDN.
Fetal Reprogramming by T of Reproductive and Metabolic Parameters in Male Sheep
Inappropriate exposure to androgens during gestation can alter the trajectory of lamb development and alter adult physiology. For example, excess of androgen exposure during gestation induces PCOS-like and metabolic traits in the female offspring in mammals, suggesting that the intrauterine environment may play a role in the etiology of PCOS (76–78). Studies from several laboratories demonstrate that pregnant sheep injected with T during part of their gestation give birth to female offspring, which have several reproductive and metabolic features resembling those observed in PCOS women. In the sheep model, T-treated pregnant ewes exhibit high plasma levels of T resembling those of adult males and plasma concentrations of insulin are similar to control pregnant sheep. The hyperandrogenic sheep model differs from human PCOS mothers, which are both hyperandrogenic and hyperinsulinemic. Thus, the sheep model isolates the effect of androgens from other stimuli such as insulin, and provides tissue samples for studies in the offspring.
In addition to the results obtained in the female offspring, recent studies have demonstrated that male sheep born to T-exposed mothers, exhibit an altered reproductive and metabolic phenotype. Biometric analysis of testis of adult males showed that Sertoli cell number was higher in males born to hyperandrogenic mothers (T-males) than in control males (C-males) whereas spermatogonia, spermatocytes and spermatids were lower (79,80). Higher numbers of Sertoli cells in the seminiferous tubules was found to be a common feature in testis from T-males from infancy through puberty (81) but not during fetal life (Recabarren et al., unpublished results). Paradoxically, the number of sperm cells was lower in the ejaculate of postpubertal and adult T-males (82). It is widely accepted that there is a strong correlation between number of Sertoli cells and the sperm production in animals and humans (83,84). Consequently, a functional discrepancy exists in these T-males because there is no correlation between the number of sperm cells and the number of Sertoli cells. The ontogeny of this discrepancy is unknown. It may be initiated during fetal life and completed before or after puberty (81,85). In this regard, the mRNA expression of the Sertoli cell marker antimullerian hormone (AMH) was higher in fetal and in peripubertal T-males testis (86,87).
The hypothalamic-pituitary-gonadal axis is also altered in T-males. This is apparent in the characteristics of the LH pulsatility, and the response of the pituitary gland and testis to a GnRH stimulus. Features of LH pulsatility such as LH pulse amplitude and LH pulse nadir were higher in T-males than in C-males, suggesting a reprogramming of GnRH secretion or a higher pituitary responsiveness to GnRH since the LH pulse frequency was not modified (88). The latter explanation was supported by another study that found pituitary gland responsiveness to a GnRH challenge was higher in T-males of 30 weeks of age than in T-males of 20 weeks of age. However, the release of T stimulated by the endogenous LH in T-males of both ages was similar to that of C-males, suggesting that the responsiveness of the Leydig cell to the endogenous LH release was lower in T-males than in C-males (85). This may be due to modifications in the bioactivity of the LH released by the GnRH challenge because the T response to human chorionic gonadotropin (HCG) in adult T-males was similar to that of C-males (82). This reasoning was supported by the finding that mRNA expression of steroidogenic testicular enzymes in T-males was comparable to that of C-males (81), suggesting that the testicular steroidogenesis was not altered. However, it is also possible that lower T secretion could be related to a low number of LH receptors, because the expression of LH receptors mRNA was lower in T-males than in C-males (81,89). In summary, the exposure to T during fetal development may be followed by: hypothalamic dysregulation of GnRH secretion, disturbances in the processing of LH secretion after a GnRH stimulus and probable modification in the isoforms of LH released. On the other hand, T secretion after LH stimulation may be also affected due to LH receptor availability. As a whole, fertility of these males may be impaired.
The intravenous glucose tolerance test (IVGTT) has been employed to evaluate various indices of insulin sensitivity in T- and C-males including, basal glucose/insulin ratio, the area under the insulin curve or mean insulin secretion, and the Insulin Sensitivity Index-Composite (ISI-C) (90). Studies in human PCOS mothers find that their sons have an altered metabolic profile from infancy to adulthood, and they develop insulin resistance independent of body mass index (82). In the sheep model, the ISI-C and all other indices were similar between T-males and C-males during infancy, the prepubertal and postpubertal periods. It could be inferred from these results that an excess of T during fetal development has no impact on the insulin sensitivity during postnatal development. To further analyze the fetal reprogramming of the insulin-glucose homeostasis in males and the contribution of T, the insulin sensitivity was assessed in orchidectomized postpubertal C-males and T-males before and 48-h after an acute T challenge. Basal levels of insulin and glucagon were not different between groups before and after the T challenge. However, T-males released higher insulin compared to C-males during the first 20-min of the test after the T challenge. Plasma levels of glucose were not different between groups during the IVGTT, suggesting that a T challenge was more effective in the release of insulin in T-males under the glucose stimulation. However, the ISI-C was lower in T-males after the T administration, suggesting a decrease in the insulin sensitivity in peripheral tissues. These findings are in contrast to those found in studies in human males born to PCOS mothers, where males exhibited metabolic disarrangements from infancy to adulthood including the lipid profile and the insulin sensitivity, while there was no alteration in the sperm cell concentrations or the GnRH responsiveness (91,92). However, plasma AMH concentrations were higher in children born to PCOS mothers than those born to control mothers suggesting that the Sertoli cell numbers were increased in the male offspring of PCOS-mothers, indicating a similar reproductive phenotype may exist in hyperandrogenic male offspring of humans and sheep (91). Further research is needed to explore this possibility.
In conclusion, the sheep model has advantages and disadvantages for studying the programming effect of T during fetal life on reproductive and metabolic parameters in offspring. Despite its limitations (93) it could help elucidate the sequelae associated with inappropriate androgen exposure during fetal development and uncover the cause of the neuroendocrine disturbances observed in the affected offspring. It is now apparent that both females and males are susceptible to the reprogramming effects of a hyperandrogenic intrauterine environment.
II-Protective Effects of Steroid Hormones
Progesterone
Protective Effects of Progesterone in the Degenerative Spinal Cord
Under physiological conditions, progesterone is traditionally associated with female reproductive functions and pregnancy. Additionally, this steroid exerts neuroprotective and pro-myelinating effects in the central and peripheral nervous system in acute and chronic diseases such as traumatic brain injury, stroke, ischemia, peripheral neuropathy of traumatic or diabetic origin, Alzheimer’s dementia and amyotrophic lateral sclerosis (ALS)(94–99). At the cellular and molecular level, progesterone modulates neuronal survival and plasticity, increases adult neurogenesis, favors the myelination process, inhibits lipid peroxidation, exerts anti-inflammatory properties and regulates astroglial plasticity (99,100). The central nervous system (CNS) expresses several specific progesterone receptors such as: 1) the classical intracellular progesterone receptor (PR), 2) several isoforms of the membrane PR (mPRα, β and γ), 3) the progesterone receptor membrane component type 1 (abbreviated PGRMC1 and formerly known as 25DX), and 4) sigma 1 receptors (99,101,102). Once progesterone reaches the nervous system, either from systemic circulation or produced locally in the brain, it can be metabolized into 5 alpha dihydroprogesterone (DHP), which is further converted into 3 alpha, 5 alpha tetrahydroprogesterone or allopregnanolone (103). Thus, the metabolism of progesterone inside the nervous system has a profound impact on its mechanism of action: while progesterone and DHP interact with the classical intracellular PR, allopregnanolone is a potent allosteric modulator of GABAa receptors.
The Wobbler mouse is an animal model of ALS, the most common motoneuron disease. Wobblers develop a chronic, progressive motoneuron degeneration with selective involvement of brain stem and cervical motoneurons (104). In contrast to ALS patients who show a sex difference in disease incidence, with higher frequency in men than in women (105), the onset or the progression of the Wobbler disease did not correlate with sex (106). Histologically, ventral horn motoneurons of the cervical spinal cord experience a dramatic cytoplasmic vacuolar degeneration (107), associated with astrocytosis (108–110) and microglial activation (111). Oxidative stress events participate in this mechanism, a finding supported by abnormalities of mitochondrial function in Wobbler mice. In this regard, mitochondria contribute to the production of certain free radicals; namely, superoxide anion and nitric oxide (NO); the latter caused by the elevated activity of a mitochondrial nitric oxide synthase (mtNOS) (112). Excess levels of NO in association with increased generation of superoxide anions produce the formation of peroxynitrite (ONOO-) leading to oxidative damage. This situation leads to mitochondrial swelling and inhibition of the electron transport chain (113). At the ultrastructural level, vacuolated motoneurons from Wobbler mice present cristolysis and disruption of outer and inner mitochondrial membranes (114).
Progesterone administration to Wobblers exerts neuroprotective and anti-inflammatory effects such as: 1) lower number of damaged/vacuolated motoneurons, 2) increased expression of brain derived neurotrophic factor in motoneurons and oligodendrocytes, 3) restoration of cholinergic neurotransmission and of axonal transport and 4) inhibitory effects on astrocytosis (94,114). Recent work suggests that the motoneuron protective effects of progesterone may also depend on the regulation of mitochondrial function.
Progesterone Prevents Mitochondrial Dysfunction In The Degenerative Spinal Cord Of Wobbler Mice
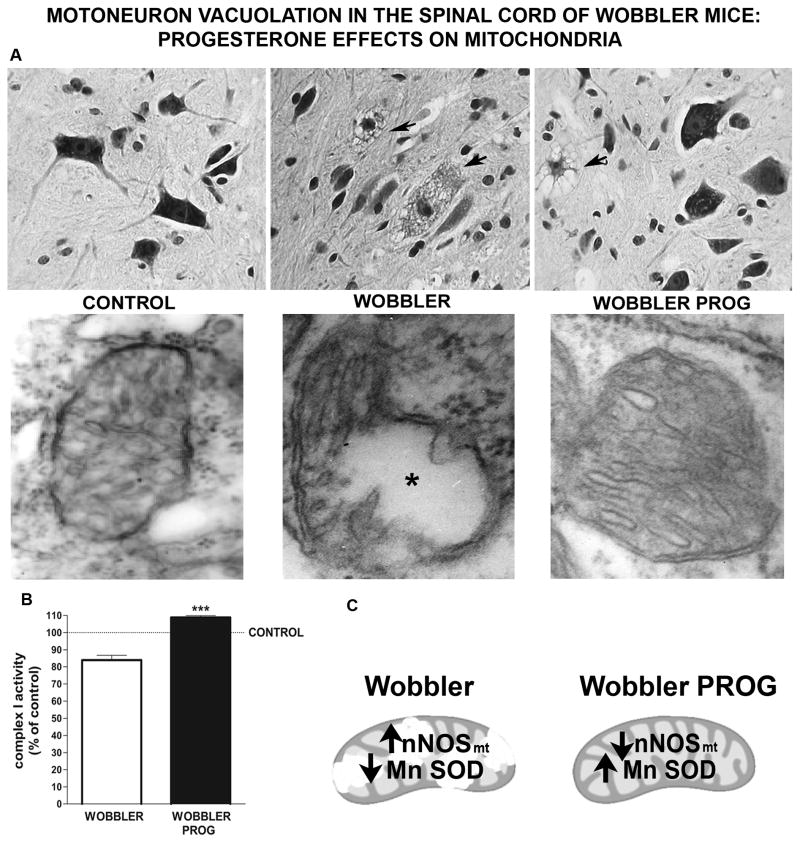
Mitochondria from early stage Wobblers show higher content of nNOS (mtNOS) in the cervical, but not lumbar spinal cord compared to controls. On the contrary, this region presents unchanged levels of cytosolic nNOS in the same groups. These events are associated with increased staining of NADPH-diaphorase/NOS in Wobbler’s motoneurons. The changes in intramitochondrial nNOS in Wobblers have deleterious consequences for the activity of the electron transport chain. In this regard, cervical cord of Wobblers shows compromised activities of complexes I and II–III in contrast to normal activities of cytochrome oxidase. Progesterone treatment decreases the level of mitochondrial nNOS in the cervical region and prevents the fall in the activity of complex I (106). These progesterone effects are associated with a reduction of the percentage of vacuolated/damaged motoneurons in Wobbler’s cervical region, indicating that mitochondrial abnormalities are linked to motoneuron degeneration (Figure 2). Additionally, decreased activity and immunostaining of the intramitochondrial enzyme manganese superoxide dismutase (MnSOD) as well as an accumulation of the amyloid precursor protein (APP) are also present in Wobblers motoneurons. The increase of APP at the level of the soma suggests impaired anterograde transport of APP to the terminal. Likewise, transport impairment may facilitate accumulation of nNOS in the cell body and enhanced access of this enzyme to the mitochondria. Exogenous administration of progesterone prevents these abnormalities (106). Hence, administration of progesterone to clinically afflicted Wobblers (1) blocks the abnormal increase of mitochondrial nNOS and normalizes respiratory complex I; (2) decreases APP accumulation, a signal of axonal degeneration, and (3) enhances superoxide dismutation. Therefore, progesterone neuroprotection reduces mitochondriopathy of the cervical spinal cord of Wobbler mice. The enhancement of MnSOD activity and immunostaining by progesterone in the Wobbler supports the concept that this steroid may act as an antioxidant molecule arresting neurodegeneration.
Figure 2.
A: Digital images of paraffin sections of the ventral horn from cervical spinal cord stained with cresyl violet. Images show motoneurons with normal appearance (upper left), two intensely vacuolated cells (arrows) in an untreated Wobbler mouse (middle graph), and 4 motoneurons with normal appearance and 1 vacuolated motoneuron (arrow) in a progesterone treated Wobbler (upper right) Magnification: 600X. Lower graphs show electron microscopy of mitochondria from a motoneuron of a control mouse (lower left), an untreated Wobbler mouse (middle) and a Wobbler mouse receiving progesterone (PROG) (lower right). Motoneurons from Wobblers show massive vacuolation disrupting the outer, inner mitochondrial membranes and cristae (lower middle graph, asterisk). Motoneurons from PROG treated Wobblers show some mitochondria with a better conservation of the membrane system, including the cristae. Magnification: 50,000X. B: Activity of the mitochondrial respiratory enzyme complex I in the cervical region of the spinal cord of Wobbler mice and Wobbler mice receiving progesterone treatment. Progesterone increased complex I activity in cervical spinal cord from Wobblers (*** p<0.001 vs. Wobbler). Results were expressed as % of complex I activity of control. C: Content of nNOS and MnSOD in mitochondrial fractions from the cervical cord of Wobbler mice and Wobbler receiving PROG. Wobbler vacuolated mitochondria showed high expression of mitochondrial nNOS (nNOSmt) and low activity and expression of manganese superoxide dismutase (MnSOD). Progesterone treatment in Wobblers significantly modified the high nNOSmt and the low MnSOD contents in mitochondria.
Steroid Hormones and Human ALS
ALS is an adult progressive neurodegenerative disorder affecting the upper and lower motor neurons (115). Respiratory failure is the most frequent cause of death in these patients (116). Worse prognostic factors in ALS are: a) bulbar onset, b) advanced age, and c) short time between onset and diagnosis (117). Epidemiological studies show higher frequency of ALS in men than women until menopause when it becomes the same for both sexes. Thus, the suggestion has been made that sex steroid hormones may play a role in the disease susceptibility (105,118). Recently, ALS has been considered a hormonal disorder involving changes of circulating gonadal steroids (118). This neurodegenerative disease also represents a stressful condition, in which changes in the hypothalamic-pituitary-adrenal (HPA) axis have been reported sporadically in ALS patients. Thus, loss of circadian rhythm of cortisol in ALS was first reported by Patacchioli et al (119), whereas Monachelli et al. has recently found serum cortisol levels increases in ALS patients, further suggesting HPA axis dysfunction (120,121).
Circulating levels of progesterone are increased in male ALS patients in comparison to male control subjects. Such levels correlate positively with survival time and factors predicting better prognosis. In this regard, endogenous progesterone levels are elevated in benign forms of the disease such as in patients younger than 55 years of age and presenting spinal onset. In the elderly, peripheral circulation of progesterone may depend on adrenal steroid production (122). Moreover, changes of androgens and estrogens also occur in ALS and their animal models (123). In some men with ALS, free T levels are in the low to normal range, whereas the finding of a low index-to-ring finger length ratio (2D:4D ratio) suggests that greater prenatal T exposure may play a role in motor neuronal vulnerability in adulthood (124). In contrast, women ALS patients show higher circulating levels of T than healthy female subjects. In this group of patients, circulating levels of T do not decline with increasing age as it does in controls. The concentration of sex steroids in ALS patients also bears a differential relationship with respiratory status and disease progression. Thus, respiratory symptoms and a decline in forced vital capacity (FVC %) are associated with a shorter survival in ALS patients. ALS patients with higher T levels and lower progesterone/free T ratio exhibit a greater loss of respiratory function or a more rapid decline of FVC% (122). These results suggest that certain steroids may play a “protective” role while others have a negative influence on parameters vital for the progression and final outcome of the disease. In the transgenic superoxide dismutase-1 (SOD1) and Wobbler mouse models of ALS, it has been shown that ovariectomy leads to a significant acceleration of the disease, whereas estradiol or progesterone treatment significantly delays disease progression (125).
Given that progesterone’s biological effects are essentially mediated by binding to the classical intracellular PR, Gargiulo-Monachelli et al (126) demonstrated the presence of PR immunoreactivity in the cytoplasm of motor neurons and, more prominently, in axonal processes and large arteries. Indeed, the authors reported that PR staining was stronger in nerve roots and large arteries from ALS compared to control spinal cords (126). Immunocytochemistry studies carried out in the rat spinal cord demonstrated that neurons and glial cells localized in the ventral horn are PR positive not only in the cytoplasm but also in the nucleus (127). Evidence of cytoplasmic PR in the human spinal cord may suggest the intriguing possibility that progesterone could be acting through extranuclear mechanisms of hormone action. The presence of extranuclear PR has also been reported in the pre- and post-synaptic structures in the rat hippocampus that, may be linked to the control of neuronal excitability and synaptic plasticity (128). The colocalization of PR with markers of phosphorylated high molecular weight neurofilaments such as SMI-31 suggests that the PR may have an as yet unknown role in these cells. As mentioned above, progesterone displays a neuroprotective role in different pathologies of the nervous system (126). In addition, progesterone and DHP reduce axonal supernumerary sprouts and promote nerve repair by influencing the expression of the peripheral myelin proteins P0 and PMP22. The finding that progesterone and DHP are able to interact with the PR further suggests a role for this classical steroid receptor in nerve repair (103). Until the role of PR in ALS is clarified, it should be mentioned that expression of this receptor in ALS affected spinal cord is not identical to that seen in controls. These findings suggest a role for the PR and progesterone in this disease, probably in relation to regeneration and neuroprotection, as reported for animal models. Consequently, future studies are needed to clearly establish progesterone neuroprotection in ALS, as may be inferred from previous studies in other neurodegenerative and injury models.
III-Estrogen-induced Tumorigenesis
Prolactinomas and Notch-signaling
Pituitary Cell line derived from estrogen treated rats: a model for resistant prolactinomas
Prolactinomas are the most frequent tumors in adults accounting for 60% of all functioning pituitary tumors. They are usually treated with dopaminergic agents and are frequently benign. In addition, 15% may be resistant to classical pharmacological therapy, become invasive and aggressive, and require extirpation. In these cases, the decrease of dopamine D2 receptor (D2R) expression is considered hallmark for loss of dopamine responsiveness and indicate that alternative therapies are needed to treat these tumors.
The estrogen-treated rat is an interesting and well-studied model of pituitary hyperplasia. Increased pituitary weight, hyperprolactinemia, lactotrope hyperplasia, and reduced dopaminergic action at the pituitary level are physiological consequences in chronically estrogenized female rats (129,130). The GH3 cell line, one of the best models developed to study prolactinomas in vitro was generated by treating a rat with high doses of estrogens, in which a prolactinoma developed (131). By extracting this tumor, the GH3 cell line was generated through cellular culture methods. Interestingly, this cell line lacks D2R, which is the principal feature of clinical dopamine agonist resistant prolactinoma. This feature makes the GH3 cell line a good experimental model to study prolactinomas and describe the molecular characteristics in vitro, or even in vivo, in a more physiological context. It is well known that GH3 cells secrete large amounts of prolactin (Prl) and growth hormone (GH), when they are maintained in cell culture or even when used for in vivo experiments (131–137).
In humans, Prl-secreting pituitary adenomas arise most commonly from the lateral wings of the anterior pituitary and fill the sella turcica as they progress leading to compression of the normal anterior and posterior lobes (130,138,139). Tumors range in size from small microadenomas to large invasive tumors with extra-sellar extension. Microadenomas, tumors with less than 1cm in diameter at diagnosis, are observed in high proportions in patients.(140,141).
The physiological symptoms of prolactinomas are galactorrhea and amenorrhea in women, and decreased libido or impotence in men (142). Gonadal dysfunction generally associated with amenorrhea, oligomenorrhea with anovulation, or infertility is present in approximately 90% of women with prolactinomas (143,144). Gonadal dysfunction in these women is due to interference with the hypothalamic-pituitary-gonadal axis by the hyperprolactinemia and, except in patients with large or invasive adenomas, is not due to destruction of the gonadotropin secreting cells. On the other hand, in men, the usual manifestations for clinical consultation are those of hypogonadism. The initial symptom is decreased libido, which may be initially regarded by both the patient and physician as a psychological factor; thus the recognition of prolactinomas in men is frequently delayed and marked hyperprolactinemia occurs. (145–148)
In the last 10 years a subset population of cells of the pituitary gland called the Side Population (SP) has been identified. This small subset of SP cells, have several characteristics reminiscent of stem/progenitor cells and of early-embryonic pituitary cells (149–151).
Pituitary adenomas were found to contain self-renewing sphere-forming cells, considered a property of Cancer Stem Cells (CSC). Whole-genome expression profiling performed in SP cells, which contain CSC, compared to the tumor bulk cells from somatotropinomas and non-functioning adenomas revealed an upregulation of several Notch system (receptors and ligands) components in humans (152). The Notch system has a broad expression both in human (153,154) (Zubeldia Brenner L. unpublished results) and mice pituitary (149–151), as well as in rat pituitary (Zubeldia Brenner L. unpublished results).
The cellular and molecular mechanisms that initiate the formation of prolactinomas are largely unknown. In this regard, the participation of Notch receptors in prolactinoma development has not been studied in detail. The Notch receptors are involved in a wide group of processes during development of eukaryotic cells such as proliferation, migration, differentiation and apoptosis (155–158). Thus, the Notch system seems to function as a general developmental tool that is used to direct cell fate and, consequently, to shape a living organism (155,156).
Mammals possess four different Notch receptors, referred to as Notch-1, Notch-2, Notch-3, and Notch-4. The Notch receptor is a single-pass transmembrane receptor protein. It is a hetero-oligomer composed of a large extracellular portion, which associates in a calcium-dependent, non-covalent interaction with a smaller piece of the Notch protein composed of a short extracellular region, a single transmembrane-pass, and a small intracellular region. The receptor is normally triggered via direct cell-to-cell contact, in which the transmembrane proteins of the cells in direct contact form the ligands that, interacts with the extracellular domain of the Notch receptor on an adjacent cell (159). Upon ligand binding, the receptor suffers two proteolytic events due to the activation mechanism (160,161). Notch receptors are cleaved near the exterior side of the plasma membrane, and then a posterior cleavage in the transmembrane domain mediated by a gamma-secretase enzyme complex liberates the intracellular domain of the receptor (Notch intracellular domain, NICD) to the cytoplasm. NICD acts as a transcription factor, translocating to the nucleus after the cleavage to form a multiprotein complex with the ubiquitously expressed CSL transcription factor. In the absence of NICD, CSL is complexed with co-repressors. When NICD binds CSL, specific co-activators are recruited resulting in a transcriptional activation of the target genes (162). The transcriptional targets of the Notch system include differentiation related factors, cell cycle regulators (p21 and cyclin D1) and regulators of apoptosis. Transcription factors of the Hairy/enhancer of split (Hes) and Hes related (HRT/HRP/Hey) families (163) are also part of the principal targets. These proteins belong to the basic helix-loop-helix family of transcription factors and bind specific sequences in the promoter region of target genes and repress the transcription through recruitment of a set of co-repressors.
Notch receptors are involved in several physiological processes. Furthermore, there are several pathologies in which Notch receptors are seriously dysregulated. In cancer and abnormal neoplasic proliferation, Notch receptors are altered, and may be upregulated or downregulated depending on the tissue and context involved (164–166). Within the four receptors Notch-1 and Notch-3 are the most frequently mentioned in tumor development and cancer (153,154,159).
Tumorigenesis and neural development are part of the processes in which Notch-3 has been described. Furthermore, the Notch-3 receptor and its ligands are implicated in cellular differentiation and in some cases promote pituitary cell growth and tumor formation in non- functioning adenomas (153,154).
Notch-3 is also expressed in the SP cells of the adult anterior pituitary gland. In humans, Notch-3 expression in non-functioning pituitary tumors is markedly higher than in normal pituitary tissue (153,154). Strikingly the Dlk-1 gene deletion, one of the Notch ligands, results in a developmental defect in somatolactotrophs (159).
Notch-1 was also detected in the anterior pituitary of adult mice (151). Recently Zubeldia-Brenner L et al. (unpublished results), detected the additional mammalian Notch receptors (Notch-2, 3, and 4) and other key downstream target genes (Hes-1, Hes-5 as well as Hey-1 and Hey-2) using RT-PCR (Zubeldia-Brenner L et al. unpublished results).
In recent experiments, xenotropic tumors were generated by injecting GH3 cells into Nude/Nude mice to test the role of Notch signaling in tumor growth and progression. Once the tumors developed, mice were treated with DAPT, a drug that blocks the Notch pathway by inhibiting the gamma-secretase. Tumor volumes were significantly smaller in mice treated with DAPT compared with the controls (Zubeldia-Brenner unpublished data). These results suggest that Notch receptors play a critical role in the hormonal function of these pituitary tumors, as well as in the normal physiological context of the pituitary enhancing or downregulating the secretion of Prl and GH.
Summary and Conclusion
This review highlights the important effects of steroid and peptidergic hormones at central and peripheral levels of the neuroendocrine axis and illustrates a wide variety of neural processes in which they participate. The impact of steroids on neuroendocrine processes is very broad, and in this review we included their impact on PCO, brain sexual differentiation, ALS, and the etiology of prolactinomas.
Evidence has been presented to show that inappropriate prenatal exposure to environmental estrogen can alter normal development of the ovary leading to adult ovarian pathologies. This can be transmitted to the next generation through epigenetic mechanisms that reprogram genes. Moreover, the prenatal hormone milieu also plays an important role in brain sexual differentiation revealing that sexual preferences are also under the control of steroid hormones.
Further examples in which steroid hormones are involved are the protective effect of progesterone in the degenerative spinal cord and ALS, and the role of estrogen and Notch signaling in prolactin secreting pituitary tumors.
In conclusion, the developmental studies presented here implicate fetal exposure to hormonal and environmental compounds as an important variable that may developmentally reprogram the neural mechanisms regulating adult reproductive function. They reveal important fetal antecedents of adult gonadal dysfunctions that impact fertility and provide new insights into the prenatal hormone influences on adult sexuality. Moreover, the functional studies of neurodegeneration and tumorigenesis have identified novel cellular and molecular control points regulated by steroid hormones that may constitute future therapeutic targets. The global processes influenced by steroid hormones are tightly regulated and so precise that the presence of hormonal disruptors or imbalances in the neuroendocrine milieu may generate altered phenotypes or predispose individuals to serious pathologies. The insights gained from this research further our understanding of the elaborate and intricate regulation that steroid hormones exert on the central and peripheral nervous systems throughout life.
Acknowledgments
Funding for these studies has been obtained by the following sources: National Science Foundation (NSF) (IOS # 1328157) which supported the 2015 International Workshop in Neuroendocrinology. HE Lara: Fondecyt 1130049 and Conicyt PIA DFG10; CE Roselli: National Institutes of Health Grant number: R01 OD011047; S Recabarren: Fondecyt 1050915, 1090031 and 1140433; Gonzalez Deniselle: MC FONCYT (PICT 2012-0009), the National Research Council of Argentina (CONICET, PIP 112 20120100016), Roemmers and Barón Foundations and the University of Buenos Aires (20020100100062 and 20020100100089); L Zubeldia-Brenner: Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Fundación René Barón.
Footnotes
The authors of the manuscript have no conflicts of interest to declare.
References
- 1.Diamond M. Clinical implications of the organizational and activational effects of hormones. Horm Behav. 2009;55:621–632. doi: 10.1016/j.yhbeh.2009.03.007. [DOI] [PubMed] [Google Scholar]
- 2.Thornton J, Zehr JL, Loose MD. Effects of prenatal androgens on rhesus monkeys: a model system to explore the organizational hypothesis in primates. Horm Behav. 2009;55:633–645. doi: 10.1016/j.yhbeh.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wallen K. The Organizational Hypothesis: Reflections on the 50th anniversary of the publication of Phoenix, Goy, Gerall, and Young (1959) Horm Behav. 2009;55:561–565. doi: 10.1016/j.yhbeh.2009.03.009. [DOI] [PubMed] [Google Scholar]
- 4.Arnold AP. The organizational-activational hypothesis as the foundation for a unified theory of sexual differentiation of all mammalian tissues. Horm Behav. 2009;55:570–578. doi: 10.1016/j.yhbeh.2009.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Guillette LJ, Jr, Crain DA, Rooney AA, Pickford DB. Organization versus activation: the role of endocrine-disrupting contaminants (EDCs) during embryonic development in wildlife. Environ Health Perspect. 1995;103(Suppl 7):157–164. doi: 10.1289/ehp.95103s7157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Albrecht ED, Pepe GJ. Steroid hormone regulation of angiogenesis in the primate endometrium. Front Biosci. 2003;8:d416–d429. doi: 10.2741/1001. [DOI] [PubMed] [Google Scholar]
- 7.Hirschberg AL. Sex hormones, appetite and eating behaviour in women. Maturitas. 2012;71:248–256. doi: 10.1016/j.maturitas.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 8.Jost A. Hormonal factors in the sex differentiation of the mammalian foetus. Philos Trans R Soc Lond B Biol Sci. 1970;259:119–130. doi: 10.1098/rstb.1970.0052. [DOI] [PubMed] [Google Scholar]
- 9.Lenz KM, Nugent BM, McCarthy MM. Sexual differentiation of the rodent brain: dogma and beyond. Front Neurosci. 2012;6:26. doi: 10.3389/fnins.2012.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rahman Q. The neurodevelopment of human sexual orientation. Neurosci Biobehav Rev. 2005;29:1057–1066. doi: 10.1016/j.neubiorev.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 11.Michael Conn P, Melmed Shlomo. Endocrinology Basic and Clinical Principles. Humana Press; 1997. [Google Scholar]
- 12.Chowen JA, Frago LM, Argente J. The regulation of GH secretion by sex steroids. Eur J Endocrinol. 2004;151(Suppl 3):U95–100. doi: 10.1530/eje.0.151u095. [DOI] [PubMed] [Google Scholar]
- 13.Austin J, Short M. Book. Vol. 3. Cambridge: Cambridge University Press; 1987. Hormonal control of reproduction; pp. 244–252. [Google Scholar]
- 14.Cho B, Suh Y, Yoon Y, Lee C, Kim K. Progesterone inhibits the estrogen -induced prolactin gene expression in the rat pituitary. Mol Cell Endocrinology. 1993;93:47–52. doi: 10.1016/0303-7207(93)90138-a. [DOI] [PubMed] [Google Scholar]
- 15.Cidlowski JA, Cidlowski NB. Regulation of glucocorticoid receptors by glucocorticoids in cultured HeLa S3 cells. Endocrinology. 1981;109:1975–1982. doi: 10.1210/endo-109-6-1975. [DOI] [PubMed] [Google Scholar]
- 16.Close FT, Freeman ME. Effects of ovarian steroid hormones on dopamine-controlled prolactin secretory responses in vitro. Neuroendocrinology. 1997;65:430–435. doi: 10.1159/000127206. [DOI] [PubMed] [Google Scholar]
- 17.Goyeneche AA, Deis RP, Gibori G, Telleria CM. Progesterone promotes survival of the rat corpus luteum in the absence of cognate receptors. Biol Reprod. 2003;68:151–158. doi: 10.1095/biolreprod.102.007898. [DOI] [PubMed] [Google Scholar]
- 18.Gardner David G, Shoback Dolores. Greenspan’s Basic and Clinical Endocrinology. McGraw-Hill; 2007. [Google Scholar]
- 19.Michael Conn P, Maurice Goodman H. Section 7 The Endocrine System, Volumen I: Cellular Endocrinology. Vol. 1998 OUP; USA: Jun 4, 1998. Subsequent edition. [Google Scholar]
- 20.Mani SK, Mermelstein PG, Tetel MJ, Anesetti G. Convergence of multiple mechanisms of steroid hormone action. Horm Metab Res. 2012;44:569–576. doi: 10.1055/s-0032-1306343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Micevych PE, Kelly MJ. Membrane estrogen receptor regulation of hypothalamic function. Neuroendocrinology. 2012;96:103–110. doi: 10.1159/000338400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Walters MR. Steroid hormone receptors and the nucleus. Endocr Rev. 1985;6:512–543. doi: 10.1210/edrv-6-4-512. [DOI] [PubMed] [Google Scholar]
- 23.Arnold AP, Gorski RA. Gonadal steroid induction of structural sex differences in the central nervous system. Annu Rev Neurosci. 1984;7:413–442. doi: 10.1146/annurev.ne.07.030184.002213. [DOI] [PubMed] [Google Scholar]
- 24.Collaer ML, Hines M. Human behavioral sex differences: a role for gonadal hormones during early development? Psychol Bull. 1995;118:55–107. doi: 10.1037/0033-2909.118.1.55. [DOI] [PubMed] [Google Scholar]
- 25.Danzo BJ. The effects of environmental hormones on reproduction. Cell Mol Life Sci. 1998;54:1249–1264. doi: 10.1007/s000180050251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ezzat S. The role of hormones, growth factors and their receptors in pituitary tumorigenesis. Brain Pathol. 2001;11:356–370. doi: 10.1111/j.1750-3639.2001.tb00405.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ojeda SR, Urbanski HF. Puberty in the rat. In: Knobil E, Neill J, editors. The Physiology of Reproduction. New York: Raven Press, Ltd; 1994. pp. 363–409. [Google Scholar]
- 28.Kwakowsky A, Cheong RY, Herbison AE, Abraham IM. Non-classical effects of estradiol on cAMP responsive element binding protein phosphorylation in gonadotropin-releasing hormone neurons: mechanisms and role. Front Neuroendocrinol. 2014;35:31–41. doi: 10.1016/j.yfrne.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 29.Herbison AE. Physiology of the adult gonadotropin-releasing hormone neuronal network. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill’s Physiology of Reproduction. Academic Press; 2015. pp. p399–p467. [Google Scholar]
- 30.Micevych PE, Wong AM, Mittelman-Smith MA. Estradiol Membrane-Initiated Signaling and Female Reproduction. Compr Physiol. 2015;5:1211–1222. doi: 10.1002/cphy.c140056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lara HE, Dorfman M, Venegas M, Luza SM, Luna SL, Mayerhofer A, Guimaraes MA, Rosa E, Silva AA, Ramirez VD. Changes in sympathetic nerve activity of the mammalian ovary during a normal estrous cycle and in polycystic ovary syndrome: Studies on norepinephrine release. Microsc Res Tech. 2002;59:495–502. doi: 10.1002/jemt.10229. [DOI] [PubMed] [Google Scholar]
- 32.Lawrence IE, Jr, Burden HW. The origin of the extrinsic adrenergic innervation to the rat ovary. Anat Rec. 1980;196:51–59. doi: 10.1002/ar.1091960106. [DOI] [PubMed] [Google Scholar]
- 33.Pepling ME. Follicular assembly: mechanisms of action. Reproduction. 2012;143:139–149. doi: 10.1530/REP-11-0299. [DOI] [PubMed] [Google Scholar]
- 34.Rajah R, Glaser EM, Hirshfield AN. The changing architecture of the neonatal rat ovary during histogenesis. Dev Dyn. 1992;194:177–192. doi: 10.1002/aja.1001940303. [DOI] [PubMed] [Google Scholar]
- 35.Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, Hauser R, Prins GS, Soto AM, Zoeller RT, Gore AC. Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev. 2009;30:293–342. doi: 10.1210/er.2009-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Patisaul HB, Adewale HB. Long-term effects of environmental endocrine disruptors on reproductive physiology and behavior. Front Behav Neurosci. 2009;3:10. doi: 10.3389/neuro.08.010.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oktem O, Urman B. Understanding follicle growth in vivo. Hum Reprod. 2010;25:2944–2954. doi: 10.1093/humrep/deq275. [DOI] [PubMed] [Google Scholar]
- 38.De Rosa CT, Pohl HR, Williams M, Ademoyero AA, Chou CH, Jones DE. Public health implications of environmental exposures. Environ Health Perspect. 1998;106(Suppl 1):369–378. doi: 10.1289/ehp.98106s1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jefferson W, Newbold R, Padilla-Banks E, Pepling M. Neonatal genistein treatment alters ovarian differentiation in the mouse: inhibition of oocyte nest breakdown and increased oocyte survival. Biol Reprod. 2006;74:161–168. doi: 10.1095/biolreprod.105.045724. [DOI] [PubMed] [Google Scholar]
- 40.Cruz G, Barra R, Gonzalez D, Sotomayor-Zarate R, Lara HE. Temporal window in which exposure to estradiol permanently modifies ovarian function causing polycystic ovary morphology in rats. Fertil Steril. 2012;98:1283–1290. doi: 10.1016/j.fertnstert.2012.07.1060. [DOI] [PubMed] [Google Scholar]
- 41.Sotomayor-Zarate R, Dorfman M, Paredes A, Lara HE. Neonatal exposure to estradiol valerate programs ovarian sympathetic innervation and follicular development in the adult rat. Biol Reprod. 2008;78:673–680. doi: 10.1095/biolreprod.107.063974. [DOI] [PubMed] [Google Scholar]
- 42.Sotomayor-Zarate R, Tiszavari M, Cruz G, Lara HE. Neonatal exposure to single doses of estradiol or testosterone programs ovarian follicular development-modified hypothalamic neurotransmitters and causes polycystic ovary during adulthood in the rat. Fertil Steril. 2011;96:1490–1496. doi: 10.1016/j.fertnstert.2011.09.011. [DOI] [PubMed] [Google Scholar]
- 43.Uzumcu M, Kuhn PE, Marano JE, Armenti AE, Passantino L. Early postnatal methoxychlor exposure inhibits folliculogenesis and stimulates anti-Mullerian hormone production in the rat ovary. J Endocrinol. 2006;191:549–558. doi: 10.1677/joe.1.06592. [DOI] [PubMed] [Google Scholar]
- 44.Hirshfield AN. Development of follicles in the mammalian ovary. Int Rev Cytol. 1991;124:43–101. doi: 10.1016/s0074-7696(08)61524-7. [DOI] [PubMed] [Google Scholar]
- 45.Rittmaster RS, Deshwal N, Lehman L. The role of adrenal hyperandrogenism, insulin resistance, and obesity in the pathogenesis of polycystic ovarian syndrome. J Clin Endocrinol Metab. 1993;76:1295–1300. doi: 10.1210/jcem.76.5.8388405. [DOI] [PubMed] [Google Scholar]
- 46.Maliqueo M, Lara HE, Sanchez F, Echiburu B, Crisosto N, Sir-Petermann T. Placental steroidogenesis in pregnant women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol. 2013;166:151–155. doi: 10.1016/j.ejogrb.2012.10.015. [DOI] [PubMed] [Google Scholar]
- 47.Goodarzi MO, Dumesic DA, Chazenbalk G, Azziz R. Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nat Rev Endocrinol. 2011;7:219–231. doi: 10.1038/nrendo.2010.217. [DOI] [PubMed] [Google Scholar]
- 48.Sir-Petermann T, Maliqueo M, Angel B, Lara HE, Perez-Bravo F, Recabarren SE. Maternal serum androgens in pregnant women with polycystic ovarian syndrome: possible implications in prenatal androgenization. Hum Reprod. 2002;17:2573–2579. doi: 10.1093/humrep/17.10.2573. [DOI] [PubMed] [Google Scholar]
- 49.Kahsar-Miller MD, Nixon C, Boots LR, Go RC, Azziz R. Prevalence of polycystic ovary syndrome (PCOS) in first-degree relatives of patients with PCOS. Fertil Steril. 2001;75:53–58. doi: 10.1016/s0015-0282(00)01662-9. [DOI] [PubMed] [Google Scholar]
- 50.Sir-Petermann T, Codner E, Maliqueo M, Echiburu B, Hitschfeld C, Crisosto N, Perez-Bravo F, Recabarren SE, Cassorla F. Increased anti-Mullerian hormone serum concentrations in prepubertal daughters of women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2006;91:3105–3109. doi: 10.1210/jc.2005-2693. [DOI] [PubMed] [Google Scholar]
- 51.Crisosto N, Codner E, Maliqueo M, Echiburu B, Sanchez F, Cassorla F, Sir-Petermann T. Anti-Mullerian hormone levels in peripubertal daughters of women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2007;92:2739–2743. doi: 10.1210/jc.2007-0267. [DOI] [PubMed] [Google Scholar]
- 52.Padmanabhan V, Veiga-Lopez A. Sheep models of polycystic ovary syndrome phenotype. Mol Cell Endocrinol. 2013;373:8–20. doi: 10.1016/j.mce.2012.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Crisosto N, Echiburu B, Maliqueo M, Perez V, Ladron dG, Preisler J, Sanchez F, Sir-Petermann T. Improvement of hyperandrogenism and hyperinsulinemia during pregnancy in women with polycystic ovary syndrome: possible effect in the ovarian follicular mass of their daughters. Fertil Steril. 2012;97:218–224. doi: 10.1016/j.fertnstert.2011.10.002. [DOI] [PubMed] [Google Scholar]
- 54.Rosa-E-Silva, Guimaraes MA, Padmanabhan V, Lara HE. Prepubertal administration of estradiol valerate disrupts cyclicity and leads to cystic ovarian morphology during adult life in the rat: role of sympathetic innervation. Endocrinology. 2003;144:4289–4297. doi: 10.1210/en.2003-0146. [DOI] [PubMed] [Google Scholar]
- 55.Abbott DH, Padmanabhan V, Dumesic DA. Contributions of androgen and estrogen to fetal programming of ovarian dysfunction. Reprod Biol Endocrinol. 2006;4:17. doi: 10.1186/1477-7827-4-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Crain DA, Janssen SJ, Edwards TM, Heindel J, Ho SM, Hunt P, Iguchi T, Juul A, McLachlan JA, Schwartz J, Skakkebaek N, Soto AM, Swan S, Walker C, Woodruff TK, Woodruff TJ, Giudice LC, Guillette LJ., Jr Female reproductive disorders: the roles of endocrine-disrupting compounds and developmental timing. Fertil Steril. 2008;90:911–940. doi: 10.1016/j.fertnstert.2008.08.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lara HE. Abstract book International workshop in Neuroendocrinology. 2015. Developmental and Programming Effects of Steroids. Induced epigenetic changes after Neonatal Exposure to Rats; p. 20. [Google Scholar]
- 58.Wilson CA, Davies DC. The control of sexual differentiation of the reproductive system and brain. Reproduction. 2007;133:331–359. doi: 10.1530/REP-06-0078. [DOI] [PubMed] [Google Scholar]
- 59.Roselli CE, Stormshak F. Prenatal programming of sexual partner preference: the ram model. J Neuroendocrinol. 2009;21:359–364. doi: 10.1111/j.1365-2826.2009.01828.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Edward O. Price LSK1SJRWJJZ2. The relationship of male-male mounting to the sexual preferences of young rams. 1988 [Google Scholar]
- 61.Perkins A, Fitzgerald JA, Price EO. Luteinizing hormone and testosterone response of sexually active and inactive rams. J Anim Sci. 1992;70:2086–2093. doi: 10.2527/1992.7072086x. [DOI] [PubMed] [Google Scholar]
- 62.Roselli CE, Larkin K, Schrunk JM, Stormshak F. Sexual partner preference, hypothalamic morphology and aromatase in rams. Physiol Behav. 2004;83:233–245. doi: 10.1016/j.physbeh.2004.08.017. [DOI] [PubMed] [Google Scholar]
- 63.Hulet CVBRES. Observations on Sexually Inhibited Rams. J Anim Sci. 1964;23:1095–1097. [Google Scholar]
- 64.Hull EMMRSB. Male sexual behavior. In: Pfaff DW, Arnold AP, Etgen AM, Fahrbach SE, Rubin RT, editors. Hormones, Brain and Behavior. 2002. [Google Scholar]
- 65.Gorski RA, Gordon JH, Shryne JE, Southam AM. Evidence for a morphological sex difference within the medial preoptic area of the rat brain. Brain Res. 1978;148:333–346. doi: 10.1016/0006-8993(78)90723-0. [DOI] [PubMed] [Google Scholar]
- 66.Gorski RA. Sexual differentiation of the brain: a model for drug-induced alterations of the reproductive system. Environ Health Perspect. 1986;70:163–175. doi: 10.1289/ehp.8670163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Roselli CE, Estill CT, Stadelman HL, Stormshak F. The volume of the ovine sexually dimorphic nucleus of the preoptic area is independent of adult testosterone concentrations. Brain Res. 2009;1249:113–117. doi: 10.1016/j.brainres.2008.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Levay S. A difference in hypothalamic structure between heterosexual and homosexual men. Science. 1991;253:1034–1037. doi: 10.1126/science.1887219. [DOI] [PubMed] [Google Scholar]
- 69.Ford JJ, D’Occhio MJ. Differentiation of sexual behavior in cattle, sheep and swine. J Anim Sci. 1989;67:1816–1823. doi: 10.2527/jas1989.6771816x. [DOI] [PubMed] [Google Scholar]
- 70.Roselli CE, Stadelman H, Reeve R, Bishop CV, Stormshak F. The ovine sexually dimorphic nucleus of the medial preoptic area is organized prenatally by testosterone. Endocrinology. 2007;148:4450–4457. doi: 10.1210/en.2007-0454. [DOI] [PubMed] [Google Scholar]
- 71.Roselli CE, Estill CT, Stadelman HL, Meaker M, Stormshak F. Separate critical periods exist for testosterone-induced differentiation of the brain and genitals in sheep. Endocrinology. 2011;152:2409–2415. doi: 10.1210/en.2010-1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Roselli CE, Schrunk JM, Stadelman HL, Resko JA, Stormshak F. The effect of aromatase inhibition on the sexual differentiation of the sheep brain. Endocrine. 2006;29:501–511. doi: 10.1385/ENDO:29:3:501. [DOI] [PubMed] [Google Scholar]
- 73.Roselli CE, Stormshak F. The neurobiology of sexual partner preferences in rams. Horm Behav. 2009;55:611–620. doi: 10.1016/j.yhbeh.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Reddy RC, Estill CT, Meaker M, Stormshak F, Roselli CE. Sex differences in expression of oestrogen receptor alpha but not androgen receptor mRNAs in the foetal lamb brain. J Neuroendocrinol. 2014;26:321–328. doi: 10.1111/jne.12152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Roselli CE, Reddy RC, Estill CT, Scheldrup M, Meaker M, Stormshak F, Montilla HJ. Prenatal influence of an androgen agonist and antagonist on the differentiation of the ovine sexually dimorphic nucleus in male and female lamb fetuses. Endocrinology. 2014;155:5000–5010. doi: 10.1210/en.2013-2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Recabarren SE, Padmanabhan V, Codner E, Lobos A, Duran C, Vidal M, Foster DL, Sir-Petermann T. Postnatal developmental consequences of altered insulin sensitivity in female sheep treated prenatally with testosterone. Am J Physiol Endocrinol Metab. 2005;289:E801–E806. doi: 10.1152/ajpendo.00107.2005. [DOI] [PubMed] [Google Scholar]
- 77.Padmanabhan V, Veiga-Lopez A, Abbott DH, Recabarren SE, Herkimer C. Developmental programming: impact of prenatal testosterone excess and postnatal weight gain on insulin sensitivity index and transfer of traits to offspring of overweight females. Endocrinology. 2010;151:595–605. doi: 10.1210/en.2009-1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Veiga-Lopez A, Moeller J, Patel D, Ye W, Pease A, Kinns J, Padmanabhan V. Developmental programming: impact of prenatal testosterone excess on insulin sensitivity, adiposity, and free fatty acid profile in postpubertal female sheep. Endocrinology. 2013;154:1731–1742. doi: 10.1210/en.2012-2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rojas-Garcia PP, Recabarren MP, Sarabia L, Schon J, Gabler C, Einspanier R, Maliqueo M, Sir- Petermann T, Rey R, Recabarren SE. Prenatal testosterone excess alters Sertoli and germ cell number and testicular FSH receptor expression in rams. Am J Physiol Endocrinol Metab. 2010;299:E998–E1005. doi: 10.1152/ajpendo.00032.2010. [DOI] [PubMed] [Google Scholar]
- 80.Rojas-García P, Recabarren MP, Palma S, Tovar H, Gabler C, Einspanier R, Maliqueo M, Sir-Petermann T, Recabarren SE. Prenatal testosterone excess alters seminal and cellular characteristics in male sheep via its androgenic actions. Abstract 8th Annual Meeting of the Androgen Excess & PCO Society; 2010. [Google Scholar]
- 81.Rojas-Garcia PP, Recabarren MP, Sir-Petermann T, Rey R, Palma S, Carrasco A, Perez-Marin CC, Padmanabhan V, Recabarren SE. Altered testicular development as a consequence of increase number of sertoli cell in male lambs exposed prenatally to excess testosterone. Endocrine. 2013;43:705–713. doi: 10.1007/s12020-012-9818-5. [DOI] [PubMed] [Google Scholar]
- 82.Recabarren SE, Rojas-Garcia PP, Recabarren MP, Alfaro VH, Smith R, Padmanabhan V, Sir-Petermann T. Prenatal testosterone excess reduces sperm count and motility. Endocrinology. 2008;149:6444–6448. doi: 10.1210/en.2008-0785. [DOI] [PubMed] [Google Scholar]
- 83.Johnson L, Zane RS, Petty CS, Neaves WB. Quantification of the human Sertoli cell population: its distribution, relation to germ cell numbers, and age-related decline. Biol Reprod. 1984;31:785–795. doi: 10.1095/biolreprod31.4.785. [DOI] [PubMed] [Google Scholar]
- 84.Berndtson WE, Igboeli G, Pickett BW. Relationship of absolute numbers of Sertoli cells to testicular size and spermatogenesis in young beef bulls. J Anim Sci. 1987;64:241–246. doi: 10.2527/jas1987.641241x. [DOI] [PubMed] [Google Scholar]
- 85.Rojas-García PP, Recabarren MP, Sandoval DCA, Fornes RRR, Sir-Petermann T, Recabarren SE. Prenatal testosterone programming: ontogeny of changes in testis of fetal and prepubertal male sheep. Endocrine Abstract. 2013;32:250. [Google Scholar]
- 86.Rojas-Garcia Pedro P, Recabarren MP, Sir-Petermann T, Rey Rodolfo, Carrasco A, Fornes Romina, Recabarren SE. Prenatal Testosterone Excess Decreases FSH Levels in Umbilical Cord Blood and Modifies the Expression of Key Reproductive Factors in Testis of Fetal Male Lambs. The Endocrine Society’s 94th Annual Meeting; June 23–26, 2012; Houston, TX. 2012. [Google Scholar]
- 87.Rojas-Garcia PP, Sandoval Daniel, Recabarren Monica P, Carrasco Albert, Sir-Petermann T, Recabarren SE. Testosterone Excess Alters the Number of Sertoli and Germ Cells Associated to a Higher Expression of SOX9, AMH and a Lower Expression of Lhr and AR in Prepubertal Male Sheep. Endocrine Society’s 96th Annual Meeting and Expo; Chicago, Illinois. 2014. [Google Scholar]
- 88.Recabarren SE, Recabarren M, Rojas-Garcia PP, Cordero M, Reyes C, Sir-Petermann T. Prenatal exposure to androgen excess increases LH pulse amplitude during postnatal life in male sheep. Horm Metab Res. 2012;44:688–693. doi: 10.1055/s-0032-1316291. [DOI] [PubMed] [Google Scholar]
- 89.Recabarren MP, Rojas-Garcia PP, Einspanier R, Padmanabhan V, Sir-Petermann T, Recabarren SE. Pituitary and testis responsiveness of young male sheep exposed to testosterone excess during fetal development. Reproduction. 2013;145:567–576. doi: 10.1530/REP-13-0006. [DOI] [PubMed] [Google Scholar]
- 90.Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22:1462–1470. doi: 10.2337/diacare.22.9.1462. [DOI] [PubMed] [Google Scholar]
- 91.Recabarren SE, Sir-Petermann T, Rios R, Maliqueo M, Echiburu B, Smith R, Rojas-Garcia P, Recabarren M, Rey RA. Pituitary and testicular function in sons of women with polycystic ovary syndrome from infancy to adulthood. J Clin Endocrinol Metab. 2008;93:3318–3324. doi: 10.1210/jc.2008-0255. [DOI] [PubMed] [Google Scholar]
- 92.Recabarren SE, Smith R, Rios R, Maliqueo M, Echiburu B, Codner E, Cassorla F, Rojas P, Sir-Petermann T. Metabolic profile in sons of women with polycystic ovary syndrome. J Clin Endocrinol Metab. 2008;93:1820–1826. doi: 10.1210/jc.2007-2256. [DOI] [PubMed] [Google Scholar]
- 93.Franks S. Animal models and the developmental origins of polycystic ovary syndrome: increasing evidence for the role of androgens in programming reproductive and metabolic dysfunction. Endocrinology. 2012;153:2536–2538. doi: 10.1210/en.2012-1366. [DOI] [PubMed] [Google Scholar]
- 94.Gonzalez Deniselle MC, Garay L, Gonzalez S, Saravia F, Labombarda F, Guennoun R, Schumacher M, De Nicola AF. Progesterone modulates brain-derived neurotrophic factor and choline acetyltransferase in degenerating Wobbler motoneurons. Exp Neurol. 2007;203:406–414. doi: 10.1016/j.expneurol.2006.08.019. [DOI] [PubMed] [Google Scholar]
- 95.Melcangi RC, Panzica G. Neuroactive steroids: an update of their roles in central and peripheral nervous system. Psychoneuroendocrinol. 2009;34(Suppl 1):S1–S8. doi: 10.1016/j.psyneuen.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 96.Sayeed I, Guo Q, Hoffman SW, Stein DG. Allopregnanolone, a progesterone metabolite, is more effective than progesterone in reducing cortical infarct volume after transient middle cerebral artery occlusion. Ann Emerg Med. 2006;47:381–389. doi: 10.1016/j.annemergmed.2005.12.011. [DOI] [PubMed] [Google Scholar]
- 97.Stein DG, Cekic MM. Progesterone and vitamin d hormone as a biologic treatment of traumatic brain injury in the aged. PM R. 2011;3:S100–S110. doi: 10.1016/j.pmrj.2011.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wang JM, Singh C, Liu L, Irwin RW, Chen S, Chung EJ, Thompson RF, Brinton RD. Allopregnanolone reverses neurogenic and cognitive deficits in mouse model of Alzheimer’s disease. Proc Natl Acad Sci U S A. 2010;107:6498–6503. doi: 10.1073/pnas.1001422107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Schumacher M, Mattern C, Ghoumari A, Oudinet JP, Liere P, Labombarda F, Sitruk-Ware R, De Nicola AF, Guennoun R. Revisiting the roles of progesterone and allopregnanolone in the nervous system: resurgence of the progesterone receptors. Prog Neurobiol. 2014;113:6–39. doi: 10.1016/j.pneurobio.2013.09.004. [DOI] [PubMed] [Google Scholar]
- 100.De Nicola AF, Labombarda F, Gonzalez Deniselle MC, Gonzalez SL, Garay L, Meyer M, Gargiulo G, Guennoun R, Schumacher M. Progesterone neuroprotection in traumatic CNS injury and motoneuron degeneration. Front Neuroendocrinol. 2009;30:173–187. doi: 10.1016/j.yfrne.2009.03.001. [DOI] [PubMed] [Google Scholar]
- 101.Labombarda F, Gonzalez SL, Gonzalez DM, Guennoun R, Schumacher M, De Nicola AF. Cellular basis for progesterone neuroprotection in the injured spinal cord. J Neurotrauma. 2002;19:343–355. doi: 10.1089/089771502753594918. [DOI] [PubMed] [Google Scholar]
- 102.Labombarda F, Meffre D, Delespierre B, Krivokapic-Blondiaux S, Chastre A, Thomas P, Pang Y, Lydon JP, Gonzalez SL, De Nicola AF, Schumacher M, Guennoun R. Membrane progesterone receptors localization in the mouse spinal cord. Neuroscience. 2010;166:94–106. doi: 10.1016/j.neuroscience.2009.12.012. [DOI] [PubMed] [Google Scholar]
- 103.Guennoun R, Labombarda F, Gonzalez Deniselle MC, Liere P, De Nicola AF, Schumacher M. Progesterone and allopregnanolone in the central nervous system: response to injury and implication for neuroprotection. J Steroid Biochem Mol Biol. 2015;146:48–61. doi: 10.1016/j.jsbmb.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 104.Moser JM, Bigini P, Schmitt-John T. The wobbler mouse, an ALS animal model. Mol Genet Genomics. 2013;288:207–229. doi: 10.1007/s00438-013-0741-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Blasco H, Guennoc AM, Veyrat-Durebex C, Gordon PH, Andres CR, Camu W, Corcia P. Amyotrophic lateral sclerosis: a hormonal condition? Amyotroph Lateral Scler. 2012;13:585–588. doi: 10.3109/17482968.2012.706303. [DOI] [PubMed] [Google Scholar]
- 106.Gonzalez Deniselle MC, Carreras MC, Garay L, Gargiulo-Monachelli G, Meyer M, Poderoso JJ, De Nicola AF. Progesterone prevents mitochondrial dysfunction in the spinal cord of wobbler mice. J Neurochem. 2012;122:185–195. doi: 10.1111/j.1471-4159.2012.07753.x. [DOI] [PubMed] [Google Scholar]
- 107.Mitsumoto H, Bradley WG. Murine motor neuron disease (the wobbler mouse): degeneration and regeneration of the lower motor neuron. Brain. 1982;105(Pt 4):811–834. doi: 10.1093/brain/105.4.811. [DOI] [PubMed] [Google Scholar]
- 108.Gonzalez Deniselle MC, Gonzalez SL, De Nicola AF. Cellular basis of steroid neuroprotection in the wobbler mouse, a genetic model of motoneuron disease. Cell Mol Neurobiol. 2001;21:237–254. doi: 10.1023/A:1010943104315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Hantaz-Ambroise D, Jacque C, Ait IA, Parmentier C, Leclerc P, Cambier D, Zadigue G, Rieger F. Specific features of chronic astrocyte gliosis after experimental central nervous system (CNS) xenografting and in Wobbler neurological mutant CNS. Differentiation. 2001;69:100–107. doi: 10.1046/j.1432-0436.2001.690204.x. [DOI] [PubMed] [Google Scholar]
- 110.Laage S, Zobel G, Jockusch H. Astrocyte overgrowth in the brain stem and spinal cord of mice affected by spinal atrophy, wobbler. Dev Neurosci. 1988;10:190–198. doi: 10.1159/000111969. [DOI] [PubMed] [Google Scholar]
- 111.Boillee S, Viala L, Peschanski M, Dreyfus PA. Differential microglial response to progressive neurodegeneration in the murine mutant Wobbler. Glia. 2001;33:277–287. [PubMed] [Google Scholar]
- 112.Finocchietto PV, Franco MC, Holod S, Gonzalez AS, Converso DP, Antico AV, Serra MP, Poderoso JJ, Carreras MC. Mitochondrial nitric oxide synthase: a masterpiece of metabolic adaptation, cell growth, transformation, and death. Exp Biol Med (Maywood ) 2009;234:1020–1028. doi: 10.3181/0902-MR-81. [DOI] [PubMed] [Google Scholar]
- 113.Poderoso JJ. The formation of peroxynitrite in the applied physiology of mitochondrial nitric oxide. Arch Biochem Biophys. 2009;484:214–220. doi: 10.1016/j.abb.2008.12.020. [DOI] [PubMed] [Google Scholar]
- 114.Gonzalez Deniselle MC, Lopez-Costa JJ, Saavedra JP, Pietranera L, Gonzalez SL, Garay L, Guennoun R, Schumacher M, De Nicola AF. Progesterone neuroprotection in the Wobbler mouse, a genetic model of spinal cord motor neuron disease. Neurobiol Dis. 2002;11:457–468. doi: 10.1006/nbdi.2002.0564. [DOI] [PubMed] [Google Scholar]
- 115.Mitchell JD, Borasio GD. Amyotrophic lateral sclerosis. Lancet. 2007;369:2031–2041. doi: 10.1016/S0140-6736(07)60944-1. [DOI] [PubMed] [Google Scholar]
- 116.Baumann F, Henderson RD, Morrison SC, Brown M, Hutchinson N, Douglas JA, Robinson PJ, McCombe PA. Use of respiratory function tests to predict survival in amyotrophic lateral sclerosis. Amyotroph Lateral Scler. 2010;11:194–202. doi: 10.3109/17482960902991773. [DOI] [PubMed] [Google Scholar]
- 117.del Aguila MA, Longstreth WT, Jr, McGuire V, Koepsell TD, van Belle G. Prognosis in amyotrophic lateral sclerosis: a population-based study. Neurology. 2003;60:813–819. doi: 10.1212/01.wnl.0000049472.47709.3b. [DOI] [PubMed] [Google Scholar]
- 118.Gargiulo MG, Meyer M, Rodriguez GE, Garay LI, Sica RE, De Nicola AF, Gonzalez Deniselle MC. Endogenous progesterone is associated to amyotrophic lateral sclerosis prognostic factors. Acta Neurol Scand. 2011;123:60–67. doi: 10.1111/j.1600-0404.2010.01385.x. [DOI] [PubMed] [Google Scholar]
- 119.Patacchioli FR, Monnazzi P, Scontrini A, Tremante E, Caridi I, Brunetti E, Buttarelli FR, Pontieri FE. Adrenal dysregulation in amyotrophic lateral sclerosis. J Endocrinol Invest. 2003;26:RC23–RC25. doi: 10.1007/BF03349149. [DOI] [PubMed] [Google Scholar]
- 120.Monachelli GG, Meyer M, Rodriguez G, Garay L, Sica RE, De Nicola AF, Deniselle MC. Progesterone and cortisol levels in sporadic amyotrophic lateral sclerosis (sALS): correlation with prognostic factors. Horm Mol Biol Clin Investig. 2011;6:167–173. doi: 10.1515/HMBCI.2011.006. [DOI] [PubMed] [Google Scholar]
- 121.Spataro R, Volanti P, Vitale F, Meli F, Colletti T, Di Natale A, La BV. Plasma cortisol level in amyotrophic lateral sclerosis. J Neurol Sci. 2015;358:282–286. doi: 10.1016/j.jns.2015.09.011. [DOI] [PubMed] [Google Scholar]
- 122.Gargiulo-Monachelli GM, Sivori M, Meyer M, Sica RE, De Nicola AF, Gonzalez-Deniselle MC. Circulating gonadal and adrenal steroids in amyotrophic lateral sclerosis: possible markers of susceptibility and outcome. Horm Metab Res. 2014;46:433–439. doi: 10.1055/s-0034-1371891. [DOI] [PubMed] [Google Scholar]
- 123.Militello A, Vitello G, Lunetta C, Toscano A, Maiorana G, Piccoli T, La BV. The serum level of free testosterone is reduced in amyotrophic lateral sclerosis. J Neurol Sci. 2002;195:67–70. doi: 10.1016/s0022-510x(01)00688-8. [DOI] [PubMed] [Google Scholar]
- 124.Vivekananda U, Manjalay ZR, Ganesalingam J, Simms J, Shaw CE, Leigh PN, Turner MR, Al Chalabi A. Low index-to-ring finger length ratio in sporadic ALS supports prenatally defined motor neuronal vulnerability. J Neurol Neurosurg Psychiatry. 2011;82:635–637. doi: 10.1136/jnnp.2010.237412. [DOI] [PubMed] [Google Scholar]
- 125.Groeneveld GJ, Van Muiswinkel FL, Sturkenboom JM, Wokke JH, Bar PR, Van den Berg LH. Ovariectomy and 17beta-estradiol modulate disease progression of a mouse model of ALS. Brain Res. 2004;1021:128–131. doi: 10.1016/j.brainres.2004.06.024. [DOI] [PubMed] [Google Scholar]
- 126.Gargiulo-Monachelli GM, Campos-Melo D, Droppelmann CA, Keller BA, Leystra-Lantz C, De Nicola AF, Gonzalez Deniselle MC, Volkening K, Strong MJ. Expression and cellular localization of the classical progesterone receptor in healthy and amyotrophic lateral sclerosis affected spinal cord. Eur J Neurol. 2014;21:273–280. doi: 10.1111/ene.12291. [DOI] [PubMed] [Google Scholar]
- 127.Labombarda F, Guennoun R, Gonzalez S, Roig P, Lima A, Schumacher M, De Nicola AF. Immunocytochemical evidence for a progesterone receptor in neurons and glial cells of the rat spinal cord. Neurosci Lett. 2000;288:29–32. doi: 10.1016/s0304-3940(00)01191-5. [DOI] [PubMed] [Google Scholar]
- 128.Waters EM, Torres-Reveron A, McEwen BS, Milner TA. Ultrastructural localization of extranuclear progestin receptors in the rat hippocampal formation. J Comp Neurol. 2008;511:34–46. doi: 10.1002/cne.21826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Suarez C, García-Tornadú I, Khalil W, Becu-Villalobos D. Dehydroepiandrosterone treatment attenuates estrogen induced pituitary hyperplasia. J Endocrinol. 2002;174:447–454. doi: 10.1677/joe.0.1740447. [DOI] [PubMed] [Google Scholar]
- 130.Brue T, Pellegrini I, Priou A, Morange I, Jaquet P. Prolactinomas and resistance to dopamine agonists. Horm Res. 1992;38:84–89. doi: 10.1159/000182496. [DOI] [PubMed] [Google Scholar]
- 131.Sonnenschein C, Richardson UI, Tashjian AH., Jr Chromosomal analysis, organ-specific function and appearance of six clonal strains of rat pituitary tumor cells. Exp Cell Res. 1970;61:121–128. doi: 10.1016/0014-4827(70)90264-8. [DOI] [PubMed] [Google Scholar]
- 132.Heaney AP, Fernando M, Melmed S. PPAR-gamma receptor ligands: novel therapy for pituitary adenomas. J Clin Invest. 2003;111:1381–1388. doi: 10.1172/JCI16575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Kim JM, Lee YH, Ku CR, Lee EJ. The cyclic pentapeptide d-Arg3FC131, a CXCR4 antagonist, induces apoptosis of somatotrope tumor and inhibits tumor growth in nude mice. Endocrinology. 2011;152:536–544. doi: 10.1210/en.2010-0642. [DOI] [PubMed] [Google Scholar]
- 134.Schaaf C, Shan B, Buchfelder M, Losa M, Kreutzer J, Rachinger W, Stalla GK, Schilling T, Arzt E, Perone MJ, Renner U. Curcumin acts as anti-tumorigenic and hormone-suppressive agent in murine and human pituitary tumour cells in vitro and in vivo. Endocr Relat Cancer. 2009;16:1339–1350. doi: 10.1677/ERC-09-0129. [DOI] [PubMed] [Google Scholar]
- 135.Kanasaki H, Oride A, Mijiddorj T, Kyo S. Role of thyrotropin-releasing hormone in prolactin-producing cell models. Neuropeptides. 2015;54:73–77. doi: 10.1016/j.npep.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 136.Boockfor FR, Hoeffler JP, Frawley LS. Cultures of GH3 cells are functionally heterogeneous: thyrotropin-releasing hormone, estradiol and cortisol cause reciprocal shifts in the proportions of growth hormone and prolactin secretors. Endocrinology. 1985;117:418–420. doi: 10.1210/endo-117-1-418. [DOI] [PubMed] [Google Scholar]
- 137.Dai C, Zhang B, Liu X, Ma S, Yang Y, Yao Y, Feng M, Bao X, Li G, Wang J, Guo K, Ma W, Xing B, Lian W, Xiao J, Cai F, Zhang H, Wang R. Inhibition of PI3K/AKT/mTOR pathway enhances temozolomide-induced cytotoxicity in pituitary adenoma cell lines in vitro and xenografted pituitary adenoma in female nude mice. Endocrinology. 2013;154:1247–1259. doi: 10.1210/en.2012-1908. [DOI] [PubMed] [Google Scholar]
- 138.Mallea-Gil MS, Cristina C, Perez-Millan MI, Ballarino MC, Rodriguez Villafañe AM, Stalldecker G, Becu-Villalobos D. Invasive giant prolactinoma with loss of therapeutic response to cabergoline: expression of angiogenic markers. Endocr Pathol. 2009;20:35–50. doi: 10.1007/s12022-009-9057-3. [DOI] [PubMed] [Google Scholar]
- 139.Baik JS, Lee MH, Ahn KJ, Choi HS, Jung SL, Kim BS, Jeun SS, Hong YK. Characteristic location and growth patterns of functioning pituitary adenomas: correlation with histological distribution of hormone-secreting cells in the pituitary gland. Clin Imaging. 2015;39:770–774. doi: 10.1016/j.clinimag.2015.05.016. [DOI] [PubMed] [Google Scholar]
- 140.Nobels FR, de Herder WW, van den Brink WM, Kwekkeboom DJ, Hofland LJ, Zuyderwijk J, De Jong FH, Lamberts SW. Long-term treatment with the dopamine agonist quinagolide of patients with clinically non-functioning pituitary adenoma. Eur J Endocrinol. 2000;143:615–621. doi: 10.1530/eje.0.1430615. [DOI] [PubMed] [Google Scholar]
- 141.Liu W, Zhou H, Neidert MC, Schmid C, Bernays RL, Ni M, Zhou D, Jia W, Jia G. Growth hormone secreting pituitary microadenomas and empty sella - An under-recognized association? Clin Neurol Neurosurg. 2014;126:18–23. doi: 10.1016/j.clineuro.2014.08.012. [DOI] [PubMed] [Google Scholar]
- 142.Ciccarelli A, Daly AF, Beckers A. The epidemiology of prolactinomas. Pituitary. 2005;8:3–6. doi: 10.1007/s11102-005-5079-0. [DOI] [PubMed] [Google Scholar]
- 143.Vlotides G, Siegel E, Donangelo I, Gutman S, Ren SG, Melmed S. Rat prolactinoma cell growth regulation by epidermal growth factor receptor ligands. Cancer Res. 2008;68:6377–6386. doi: 10.1158/0008-5472.CAN-08-0508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Paez-Pereda M, Giacomini D, Refojo D, Nagashima AC, Hopfner U, Grubler Y, Chervin A, Goldberg V, Goya R, Hentges ST, Low MJ, Holsboer F, Stalla GK, Arzt E. Involvement of bone morphogenetic protein 4 (BMP-4) in pituitary prolactinoma pathogenesis through a Smad/estrogen receptor crosstalk. Proc Natl Acad Sci U S A. 2003;100:1034–1039. doi: 10.1073/pnas.0237312100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gillam MP, Molitch ME, Lombardi G, Colao A. Advances in the treatment of prolactinomas. Endocr Rev. 2006;27:485–534. doi: 10.1210/er.2005-9998. [DOI] [PubMed] [Google Scholar]
- 146.Molitch ME. Dopamine resistance of prolactinomas. Pituitary. 2003;6:19–27. doi: 10.1023/a:1026225625897. [DOI] [PubMed] [Google Scholar]
- 147.Molitch ME. Medical treatment of prolactinomas. Endocrinol Metab Clin North Am. 1999;28:143–69. vii. doi: 10.1016/s0889-8529(05)70061-x. [DOI] [PubMed] [Google Scholar]
- 148.Molitch ME. Pharmacologic resistance in prolactinoma patients. Pituitary. 2005;8:43–52. doi: 10.1007/s11102-005-5085-2. [DOI] [PubMed] [Google Scholar]
- 149.Chen J, Gremeaux L, Fu Q, Liekens D, Van Laere S, Vankelecom H. Pituitary progenitor cells tracked down by side population dissection. Stem Cells. 2009;27:1182–1195. doi: 10.1002/stem.51. [DOI] [PubMed] [Google Scholar]
- 150.Chen J, Hersmus N, Van DV, Caesens P, Denef C, Vankelecom H. The adult pituitary contains a cell population displaying stem/progenitor cell and early embryonic characteristics. Endocrinology. 2005;146:3985–3998. doi: 10.1210/en.2005-0185. [DOI] [PubMed] [Google Scholar]
- 151.Chen J, Crabbe A, Van DV, Vankelecom H. The notch signaling system is present in the postnatal pituitary: marked expression and regulatory activity in the newly discovered side population. Mol Endocrinol. 2006;20:3293–3307. doi: 10.1210/me.2006-0293. [DOI] [PubMed] [Google Scholar]
- 152.Mertens F, Gremeaux L, Chen J, Fu Q, Willems C, Roose H, Govaere O, Roskams T, Cristina C, Becu-Villalobos D, Jorissen M, Poorten VV, Bex M, van Loon J, Vankelecom H. Pituitary tumors contain a side population with tumor stem cell-associated characteristics. Endocr Relat Cancer. 2015;22:481–504. doi: 10.1530/ERC-14-0546. [DOI] [PubMed] [Google Scholar]
- 153.Lu R, Gao H, Wang H, Cao L, Bai J, Zhang Y. Overexpression of the Notch3 receptor and its ligand Jagged1 in human clinically non-functioning pituitary adenomas. Oncol Lett. 2013;5:845–851. doi: 10.3892/ol.2013.1113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Miao Z, Miao Y, Lin Y, Lu X. Overexpression of the Notch3 receptor in non-functioning pituitary tumours. J Clin Neurosci. 2012;19:107–110. doi: 10.1016/j.jocn.2011.07.029. [DOI] [PubMed] [Google Scholar]
- 155.Fiuza UM, Arias AM. Cell and molecular biology of Notch. J Endocrinol. 2007;194:459–474. doi: 10.1677/JOE-07-0242. [DOI] [PubMed] [Google Scholar]
- 156.Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999;284:770–776. doi: 10.1126/science.284.5415.770. [DOI] [PubMed] [Google Scholar]
- 157.Raetzman LT, Ross SA, Cook S, Dunwoodie SL, Camper SA, Thomas PQ. Developmental regulation of Notch signaling genes in the embryonic pituitary: Prop1 deficiency affects Notch2 expression. Dev Biol. 2004;265:329–340. doi: 10.1016/j.ydbio.2003.09.033. [DOI] [PubMed] [Google Scholar]
- 158.Zanotti S, Canalis E. Notch signaling in skeletal health and disease. Eur J Endocrinol. 2013;168:R95–103. doi: 10.1530/EJE-13-0115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Cheung LY, Rizzoti K, Lovell-Badge R, Le Tissier PR. Pituitary phenotypes of mice lacking the notch signalling ligand delta-like 1 homologue. J Neuroendocrinol. 2013;25:391–401. doi: 10.1111/jne.12010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Hansson EM, Lendahl U, Chapman G. Notch signaling in development and disease. Semin Cancer Biol. 2004;14:320–328. doi: 10.1016/j.semcancer.2004.04.011. [DOI] [PubMed] [Google Scholar]
- 161.Gordon WR, Vardar-Ulu D, Histen G, Sanchez-Irizarry C, Aster JC, Blacklow SC. Structural basis for autoinhibition of Notch. Nat Struct Mol Biol. 2007;14:295–300. doi: 10.1038/nsmb1227. [DOI] [PubMed] [Google Scholar]
- 162.Kopan R, Ilagan MX. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell. 2009;137:216–233. doi: 10.1016/j.cell.2009.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Iso T, Kedes L, Hamamori Y. HES and HERP families: multiple effectors of the Notch signaling pathway. J Cell Physiol. 2003;194:237–255. doi: 10.1002/jcp.10208. [DOI] [PubMed] [Google Scholar]
- 164.Stylianou S, Clarke RB, Brennan K. Aberrant activation of notch signaling in human breast cancer. Cancer Res. 2006;66:1517–1525. doi: 10.1158/0008-5472.CAN-05-3054. [DOI] [PubMed] [Google Scholar]
- 165.Fan X, Mikolaenko I, Elhassan I, Ni X, Wang Y, Ball D, Brat DJ, Perry A, Eberhart CG. Notch1 and notch2 have opposite effects on embryonal brain tumor growth. Cancer Res. 2004;64:7787–7793. doi: 10.1158/0008-5472.CAN-04-1446. [DOI] [PubMed] [Google Scholar]
- 166.Ranganathan P, Weaver KL, Capobianco AJ. Notch signalling in solid tumours: a little bit of everything but not all the time. Nat Rev Cancer. 2011;11:338–351. doi: 10.1038/nrc3035. [DOI] [PubMed] [Google Scholar]