Abstract
Aging has been implicated in the development of pulmonary fibrosis, which has seen a sharp increase in incidence in those older than 50 years. Recent studies demonstrate a role for the nucleotide-binding domain and leucine rich repeat containing family, pyrin domain containing 3 (NLRP3) inflammasome and its regulated cytokines in experimental lung fibrosis. In this study, we tested the hypothesis that age-related NLRP3 inflammasome activation is an important predisposing factor in the development of pulmonary fibrosis. Briefly, young and aged wild-type and NLRP3−/− mice were subjected to bleomycin-induced lung injury. Pulmonary fibrosis was determined by histology and hydroxyproline accumulation. Bone marrow and alveolar macrophages were isolated from these mice. NLRP3 inflammasome activation was assessed by co-immunoprecipitation experiments. IL-1β and IL-18 production was measured by ELISA. The current study demonstrated that aged wild-type mice developed more lung fibrosis and exhibited increased morbidity and mortality after bleomycin-induced lung injury, when compared with young mice. Bleomycin-exposed aged NLRP3−/− mice had reduced fibrosis compared with their wild-type age-matched counterparts. Bone marrow–derived and alveolar macrophages from aged mice displayed higher levels of NLRP3 inflammasome activation and caspase-1–dependent IL-1β and IL-18 production, which was associated with altered mitochondrial function and increased production of reactive oxygen species. Our study demonstrated that age-dependent increases in alveolar macrophage mitochondrial reactive oxygen species production and NLRP3 inflammasome activation contribute to the development of experimental fibrosis.
Keywords: aging, bleomycin, mitochondrial oxidative stress, NLRP3 inflammasome, pulmonary fibrosis
Clinical Relevance
The study demonstrates that aged wild-type mice developed more lung fibrosis and exhibited increased morbidity and mortality after bleomycin-induced lung injury. Age-dependent increases in alveolar macrophage mitochondrial reactive oxygen species production are mediated by NLRP3 inflammasome activation and contribute to the development of experimental fibrosis.
Aging is associated with diminished mitochondrial energy metabolism and increased mitochondrial oxidative stress, mitochondrial reactive oxygen species (mtROS) production, and mitochondrial DNA mutations (1, 2). Low levels of inflammation caused by enhanced mitochondrial dysfunction and oxidative stress can potentiate inflammatory responses that have been associated with age-related degenerative diseases (3). Fibrotic interstitial lung diseases, including idiopathic pulmonary fibrosis (IPF), are more prevalent in aging populations, with a sharp increase in incidence in those older than 50 years (4). IPF is a severe, rapidly progressing, fatal lung disease. Although recent research has identified the pathways underlying the development of IPF, little is known about how oxidative stress, mitochondrial dysfunction, and the process of aging contribute to disease pathogenesis (5, 6).
Recent studies suggest a role for nucleotide-binding domain and leucine rich repeat containing family, pyrin domain containing 3 (NLRP3) inflammasome activation in lung inflammation and fibrosis (7–11). The NLRP3 inflammasome is a key signaling platform that is activated by a variety of signals, such as reactive oxygen species (ROS), to produce IL-1β and IL-18 (12). Bleomycin, one of the most widely used reagents for inducing experimental lung fibrosis, generates intracellular ROS, which can injure lung epithelial cells and activate the NLRP3 inflammasome in macrophages, leading to IL-1β secretion (7, 13). IL-1β promotes the production of transforming growth factor (TGF)-β, which stimulates the proliferation and transdifferentiation of epithelial cells and resident fibroblasts into collagen-producing myofibroblasts (14). Furthermore, IL-1β can induce the secretion of neutrophil chemokines causing neutrophil influx, promote alveolar epithelial cell death, and enhance the production of platelet-derived growth factor, all of which promote fibrosis (14–16).
The NLRP3 inflammasome can be activated in immune cells, including alveolar macrophages, to cause lung injury (10). Alveolar macrophages, the most abundant cells in bronchoalveolar lavage (BAL) fluid, can release cytokines, growth factors, extracellular matrix proteins and metalloproteases that contribute to alveolar injury, aberrant lung repair, and pulmonary fibrosis (17, 18). Several studies have suggested that activated alveolar macrophages and their mediators may play a role in IPF pathogenesis (19–21).
Given the increased incidence of IPF in older populations and the contribution of mitochondrial oxidative stress and IL-1β secretion to pulmonary injury and repair, we investigated the impact of aging on mtROS production and NLRP3 inflammasome–mediated production of IL-1β and IL-18 as potential modulators of bleomycin-induced pulmonary fibrosis (22, 23). We demonstrate that aged mice, compared with their younger counterparts, have greater experimental lung injury and fibrosis that is associated with increased NLRP3 inflammasome activation and mitochondrial dysfunction.
Materials and Methods
Mice
Young (2–4 mo of age) and aged (17–19 mo of age) male C57BL/6 mice were purchased from the NIA rodent facility (Charles River Laboratories, Frederick, MD). NLRP3−/− mice have been described previously (24) and were bred to the age of 13 months. On receipt, the mice were handled under identical husbandry conditions and fed certified commercial feed. Body weights were measured daily, and the mice were killed humanely if they lost more than 15% of their starting body weight. The Institutional Animal Care and Use Committee at the Lovelace Respiratory Research Institute and Weill Cornell Medical College approved the use of animals in this study. No animals were used in the study if they had evidence of skin lesions, weight loss, or lymphadenopathy.
Primary Bone Marrow Isolation and Cell Culture
Bone marrow cells were prepared from the femurs of mice as described previously (25, 26). The cells were cultured in media alone or in media containing LPS (100 ng/ml) for 4 hours before treatment with media alone or media containing bleomycin 0.1 U (1.6 mg/U; Sigma-Aldrich, St. Louis, MO) for 24 hours. In additional experiments, at the time of bleomycin stimulation, the cells were treated with z-VADFMK (10 µM, 1 h) (InvivoGen, San Diego, CA) or glybenclamide (25 µg/ml, 24 h). NLRP3 small interfering RNA (siRNA) (Flexitube GeneSolution siRNA; QIAGEN, Valencia, CA) (15 nmol/sequence) was complexed with GenMute siRNA transfection reagent (SignaGen Laboratories, Ijamsville, MD) for 15 minutes before being added to macrophage cultures. NLRP3 siRNA-transfected cells were cultured for 24 hours before LPS and/or bleomycin stimulation. NLRP3 silencing was confirmed by Western blot analysis and real-time polymerase chain reaction (RT-PCR) (see Figure E4 in the online supplement). LPS-primed macrophages were treated with rotenone (5 µM) 1 hour before treatment with media alone or media containing bleomycin. The cells were treated with mitoTEMPO (100 µM) (Enzo Life Sciences, Plymouth Meeting, PA) for 1 hour before stimulation with media alone or LPS (for 4 h). The cells were subsequently treated with media alone or media containing bleomycin.
Alveolar Macrophage Isolation
Alveolar macrophages were collected using previously published methods (26). Briefly, the lungs were gently infused with 2 ml of 37°C calcium and magnesium-free phosphate-buffered saline (PBS) containing 0.5 mM EDTA. Liquid was gently withdrawn and reinfused back into the lung two additional times. Cells from ∼10 young or aged mice were collected after centrifugation, plated at a cell density of 0.5 × 106 cells/well, and treated with LPS and/or bleomycin, as detailed above.
RNA Isolation and RT-PCR
RNA samples were extracted using the RNeasy Kit (QIAGEN), and primer-specific gene expression was assessed using QuantiTect Primer Assays (Qiagen, Valencia, CA) according to the manufacturer’s instructions. Gene expression was collected using the ABI 7300 RT-PCR machine (Invitrogen) and analyzed using the delta-CT method as described previously (27–29).
ELISA
Culture supernatants or serum were analyzed for IL-1β or IL-18 production using murine ELISA kits purchased from eBioscience (San Diego, CA).
Caspase-1 Assay
Caspase-1 activity was assessed using the Caspase-1 Assay (Abcam, Cambridge, MA). Briefly, 100 µg of each sample was incubated at 37°C for 1 hour before spectrophotometric analysis at 405 nM.
Western Immunoblot Analysis
Proteins were isolated from cultured cells and lung tissue samples and analyzed by Western blot using rabbit antimouse caspase-8, caspase-9, p-SMAD3, SMAD3, and β-actin (Cell Signal Technology, Danvers, MA) or antimouse NLRP3 (Abcam) and ASC (Santa Cruz Biotechnologies, Dallas, TX) as described (27). Briefly, 10–30 µg of each protein sample was loaded onto a 4–12% BIS-TRIS Bolt gel (Invitrogen) and run at 165 V for 35 minutes. Protein was transferred to the nitrocellulose membrane using the iBlot Western blotting system (Invitrogen).
Flow Cytometry
mtROS levels were measured by staining cells with MitoSOX (Invitrogen) at 5 µM for 40 minutes at 37°C. Cells were collected from digested lung samples (collagenase D [1 mg/ml] and DNase I [0.1 mg/ml] [Roche, Indianapolis, IN]) before staining with CD45.2-PE, CD11b-PERCP Cy5.5, CD11c-Alexa Fluor 647, CD64-PE-Cy7, F4/80-APC, and/or GR1-PE (eBioscience) in staining solution (1% fetal bovine serum–supplemented 1X PBS). Alveolar macrophage populations were analyzed using previously published methods (30). Briefly, CD45+ cell populations were selected initially and CD11c+CD11b−CD64+ cells were quantified as alveolar macrophages. Nondetermined macrophage populations were identified as F4/80+CD11b+GR1− cells. All flow cytometry was analyzed using FlowJo software (Tree Star, Inc., Ashland, OR).
Co-Immunoprecipitation
Co-immunoprecipitation of NLRP3 or ASC was performed using the Pierce Co-Immunoprecipitation kit (Thermo Scientific, Rockford, IL) according to the manufacturer’s instructions. Briefly, 500 µg of in vitro or in vivo murine samples were incubated overnight with 1 µg of either anti-NLRP3 or anti-ASC antibodies. Samples were collected in 50 µl of elution buffer before SDS-PAGE analysis.
Bleomycin Administration
Mice were anesthetized with isoflurane (5% for induction and 2% for maintenance) before receiving 0.1 milligram of bleomycin sulfate per mouse (Iktas Trade Co., Ltd., Hong Kong, China) (50-µl volume in PBS) via oral aspiration, as described previously (31). Aged mice were administered the same amount of bleomycin as their younger counterparts, because although their body mass increases with age, their lung volume, once mature, remains stable (32). This approach ensured that all mice would receive the same amount of bleomycin per lung volume and that changes in fibrosis in older mice would not be simply a result of the administration of a weight-corrected increased dose of bleomycin.
Histology
Lungs were isolated on Day 28 after instillation and fixed with 10% formalin under constant pressure. Lung tissue was stained with HandE and trichrome before blinded analysis by a pathologist. CD31 (Dianova, Hamburg, Germany) and TUNEL (Sigma Aldrich) staining on histology slides was performed according to the manufacturer’s instructions (Promega, Madison, WI) and as published previously (33). Images were obtained using the Nanozoomer Digital Pathology System (Hamamutsu, Boston, MA).
Hydroxyproline Assay
Briefly, right lobe tissue collected on Day 28 was dehydrated overnight and digested with 6N HCl before spectrophotometric quantification of hydroxyproline levels. Protein was quantified using the Bio-Rad protein assay (Bio-Rad, Hercules, CA) and was used to normalize hydroxyproline levels.
Statistical Analysis
Survival analysis between groups was calculated using the Mantel Cox test. Comparison of groups was performed using a two-tailed t test or the one-way or two-way analysis of variance, when appropriate. Samples obtained were normally, or approximately normally, distributed. All samples were independent and were the same size for analysis. Variances of the populations were equal. All data were analyzed using GraphPad prism software (San Diego, CA). Statistical significance was considered to be P value <0.05.
Results
Increased Morbidity and Mortality in Bleomycin-Treated Aged Animals
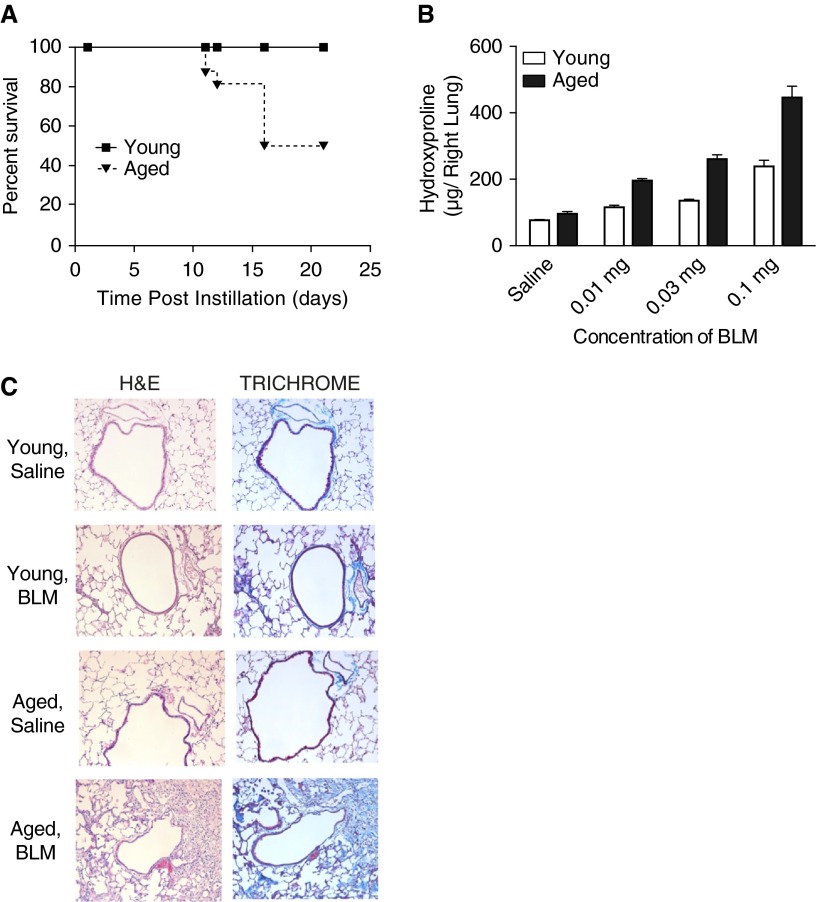
Previous studies have described an association between mouse age and severity of lung disease after intratracheal instillation of bleomycin (32). We administered bleomycin at several doses (0.01–0.1 mg/mouse) via oral aspiration to young (2–4 mo of age) and aged (17–19 mo of age) male C57BL/6 mice. Bleomycin-treated aged mice experienced increased mortality (Figure 1A) and weight loss (Figure E1A) compared with young mice. In addition to higher total leukocyte numbers, there was a dose-dependent increased recruitment of macrophages, neutrophils, and lymphocytes in BAL samples isolated from aged lung after bleomycin treatment (Figures E1B–E1E).
Figure 1.
Increased mortality and fibrosis in aged mice after in vivo bleomycin administration. Young (2–4 mo of age) and aged (17–19 mo of age) C57BL/6 mice were instilled with bleomycin (0.1 mg/ mouse) via oral aspiration. (A) Survival of young (n = 10) and aged (n = 10) C57BL/6 mice after bleomycin instillation (Mantel Cox test, P = 0.0015). (B) Right lobe tissue from mice exposed to a range of bleomycin doses was collected on Day 28, dehydrated overnight, and digested with 6N HCl before quantification of hydroxyproline levels. Results are presented as mean ± SEM. (C) Lungs were isolated on Day 28 after instillation, fixed, and stained with H&E or trichrome before blinded analysis by a pathologist. 20× images were obtained using the Nanozoomer Digital Pathology System. Results are representative of three independent experiments. BLM, bleomycin; H&E, hematoxylin and eosin.
Increased Pulmonary Fibrosis in Aged Mice After Bleomycin Instillation
Increased fibrotic lesions occur in aged lung after bleomycin instillation (32). Consistent with this, we observed a significant increase in lung collagen deposition, as determined by increased hydroxyproline levels and trichrome staining, in aged lung after bleomycin instillation, relative to young lung (Figures 1B and 1C). Furthermore, focal bronchoalveolar hyperplasia and relatively prominent, scattered lymphoid nodules were observed in damaged areas of lungs derived from aged mice (Figure 1C).
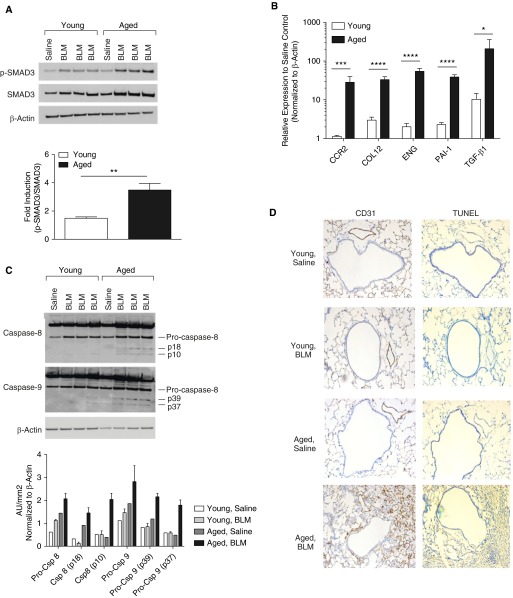
We next examined the expression of key factors that modulate fibroblast proliferation and cellular recruitment to the site of injury. SMAD3 phosphorylation in young and aged lung samples was assayed after bleomycin instillation. Although expression of SMAD3 was detected in both age groups, aged lung exhibited enhanced SMAD3 protein expression and SMAD3 phosphorylation (Figure 2A). Increased TGF-β1 signaling was reflected in the increase in expression of TGF-β1 transcriptional targets (Figure 2B) such as CCR2, collagen type-1, endoglin, PAI-1, and TGF-β mRNA post bleomycin instillation. Next, we performed TUNEL staining on young and aged lung samples to examine apoptosis. Although apoptosis was detected in both young and aged lung, enhanced TUNEL staining that was associated with increased caspase-8 and -9 cleavage was evident in aged lung (Figures 2C and 2D).
Figure 2.
Enhanced transforming growth factor (TGF)-β–mediated signaling and apoptosis in aged lung in response to bleomycin. Young (2–4 mo of age) (n = 10) and aged (17–19 mo of age) (n = 10) C57BL/6 mice were instilled with bleomycin (0.1 mg/mouse) via oral aspiration. (A) Phospho-SMAD3 (p-SMAD3) and SMAD3 levels present in young and aged lung isolated on Day 16 after bleomycin instillation were determined by immunoblotting. The ratio p-SMAD3/total SMAD3 expression was determined by densitometry and is expressed as fold induction in bleomycin-treated mice compared with uninjured control mice. β-actin served as the standard. P = 0.0019 (t test). (B) Relative expression of key TGF-β signaling genes in young and aged lung on Day 16 after bleomycin instillation. Student’s t test for single genes: *P < 0.05; ***P < 0.001; for all genes: two-way analysis of variance, P < 0.0001. (C) Caspase-8 and caspase-9 expression and presence of activated cleavage products in young and aged lung 28 days after bleomycin instillation were assessed by immunoblotting. β-actin served as the standard. (D) Apoptosis was assessed by TUNEL staining. Lung tissue was costained with endothelial marker CD31. Images and immunoblots are representative of at least three experiments. Data are expressed as the mean ± SD. ****P < 0.0001, **P = 0.0019. AU, arbitrary unit; CCR2, C-C motif chemokine receptor 2; COL12, collagen type-1; Csp, caspase; ENG, endoglin; PAI, plasminogen activator inhibitor; SMAD, mothers against decapentaplegic homolog.
Increased NLRP3 Inflammasome Activation in Aged Lung in Response to Bleomycin
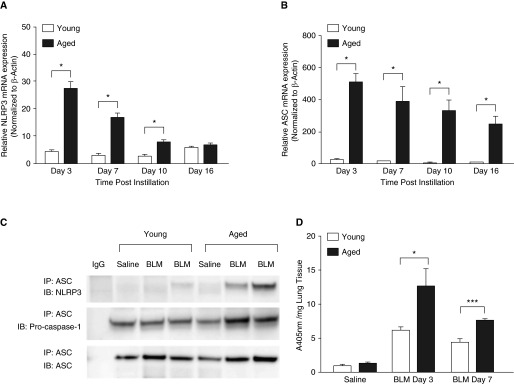
IL-1β and IL-18 play a direct role in promoting the production of TGF-β (34–38). We therefore evaluated the expression and activation of the NLRP3 inflammasome, a key signaling complex for the processing and activation of IL-1β and IL-18, in young and aged lung tissue harvested after bleomycin administration (32, 37). NLRP3 and ASC mRNA expression were significantly increased in aged lung (Figures 3A and 3B). Co-immunoprecipitation of ASC and NLRP3 from young and aged lung homogenates revealed increased NLRP3 inflammasome assembly in aged lung on Day 3 after bleomycin instillation (Figure 3C and Figure E2). Relative to young lung, caspase-1 activity was increased in aged lung homogenates on Days 3 and 7 (Figure 3D).
Figure 3.
Increased nucleotide-binding domain and leucine rich repeat containing family, pyrin domain containing 3 (NLRP3) inflammasome activation in aged lung after treatment with bleomycin. Lung tissue was isolated from saline control mice and bleomycin-instilled young (2–4 mo of age) and aged (17–19 mo of age) mice at select time points after bleomycin instillation. RNA was isolated and (A) NLRP3 and (B) ASC mRNA expression was assessed by real-time polymerase chain reaction. (C) Lung homogenate samples were isolated on Day 3 after bleomycin and co-immunoprecipitation was performed against anti-ASC. Eluted proteins were separated by electrophoresis and immunoblotted with antimouse NLRP3, ASC, and pro–caspase-1. (D) Caspase-1 activity was assessed in young and aged lung homogenates isolated on Days 3 and 7 after bleomycin instillation. Similar results were obtained from two or more independent experiments with an n ≥ 5 per experiment. Data are expressed as mean ± SEM. *P < 0.05, ***P < 0.001, Student’s t test. NLRP3: Day 3, P = 0.014 (t test); Day 7, P = 0.0134 (t test); Day 10, P = 0.0463 (t test). ASC: Day 3, P = 0.0101 (t test); Day 7, P = 0.0496 (t test); Day 10, P = 0.05 (t test); Day 16, P = 0.0402 (t test). Caspase-1: Day 3, P = 0.036 (t test); Day 7, P < 0.001 (t test). ASC, apoptosis-associated speck-like protein containing a CARD domain; IB, immunoblot; IP, immunoprecipitation.
Increased Production of Mature IL-1β and IL-18 in Aged Lung in Response to Bleomycin
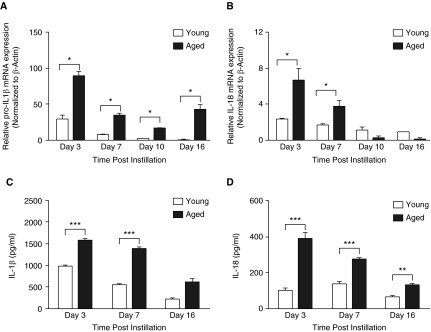
We next evaluated the expression of pro- and mature forms of IL-1β and IL-18 in young and aged lung tissue after bleomycin administration. Both pro–IL-1β and pro–IL-18 mRNA expression were detected in the lung on Day 3 after bleomycin exposure, with heightened expression in aged lung (Figures 4A and 4B). Significantly higher levels of mature IL-1β and IL-18 were detected in lung homogenates from aged, relative to young, mice (Figures 4C and 4D).
Figure 4.
Increased production of mature IL-1β and IL-18 in aged lung after treatment with bleomycin. Lung tissue was isolated from saline control mice and bleomycin-instilled young (2–4 mo of age) and aged (17–19 mo of age) mice at select time points after instillation. RNA was isolated and mRNA expression of (A) pro–IL-1β and (B) pro–IL-18 was assessed by real-time polymerase chain reaction. Homogenates were collected, and production of (C) IL-1β and (D) IL-18 in young and aged lung was assessed by ELISA. Similar results were obtained from two or more independent experiments with an n ≥ 5 per experiment. Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, Student’s t test. pro–IL-1β: Day 3, P = 0.0195 (t test); Day 7, P = 0.0076 (t test); Day 10, P = 0.0044 (t test); Day 16, P = 0.0154 (t test). pro–IL-18: Day 3, P = 0.0016 (t test); Day 7, P = 0.01722 (t test). IL-1β: Day 3, P < 0.0001 (t test); Day 7, P < 0.0001 (t test). IL-18: Day 3, P < 0.0001 (t test); Day 7, P < 0.0001 (t test); Day 16, P = 0.0027 (t test).
NLRP3−/− Aged Mice Are Protected from Bleomycin-Induced Injury and Fibrosis
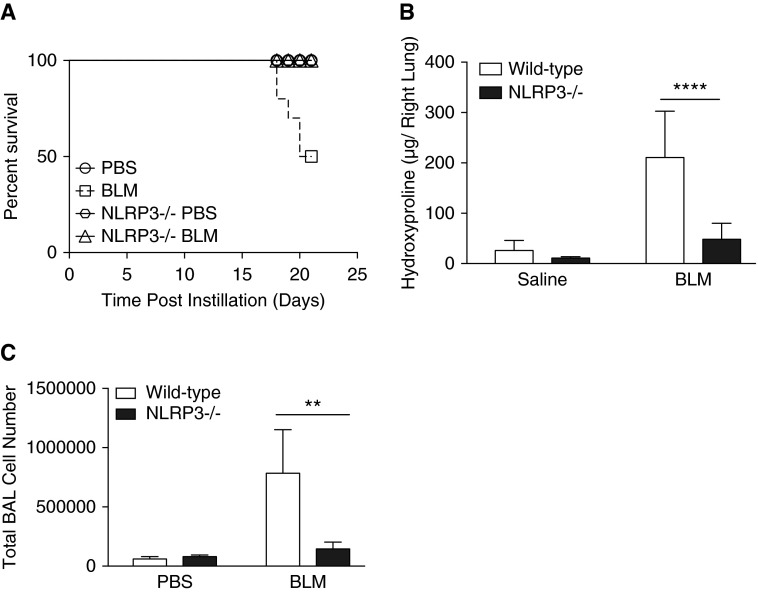
Given the increased fibrosis and activation of the NLRP3 inflammasome in the lungs of bleomycin-treated aged mice compared with young mice, we hypothesized that NLRP3-deficient mice would be protected from bleomycin-induced lung injury. Accordingly, we exposed 13-month-old male NLRP3−/− mice and age-matched wild-type C57BL/6 mice to bleomycin. Compared with wild-type mice, NLRP3−/− mice experienced no mortality (Figure 5A) and significantly less weight loss (Figure E3A) after bleomycin instillation. In addition, when compared with wild-type mice, middle-aged NLRP3−/− mice exhibited significantly less lung fibrosis, lower BAL cell counts (Figures 5B and 5C), and decreased protein after bleomycin instillation (Figure E3B).
Figure 5.
NLRP3−/− aged mice are protected from bleomycin-induced mortality and fibrosis. Wild-type and NLRP3−/− mice (13 mo old) were instilled with bleomycin (0.1 mg/mouse) via oral aspiration. (A) Survival of wild-type (n = 8) and NLRP3−/− (n = 10) mice after bleomycin instillation (Mantel Cox test, P = 0.0049). (B) Right lobe tissue from mice was collected on Day 21 after bleomycin instillation, dehydrated overnight, and digested with 6N HCl before quantification of hydroxyproline levels. (C) Bronchoalveolar lavage (BAL) was performed on mice at Day 21, and cell counts were performed. Data are presented as mean ± SEM. Student’s t test, **P = 0.0022 and ****P < 0.0001.
Increased IL-1β and IL-18 Production by Aged Macrophages in Response to Bleomycin Is Dependent on the NLRP3 Inflammasome
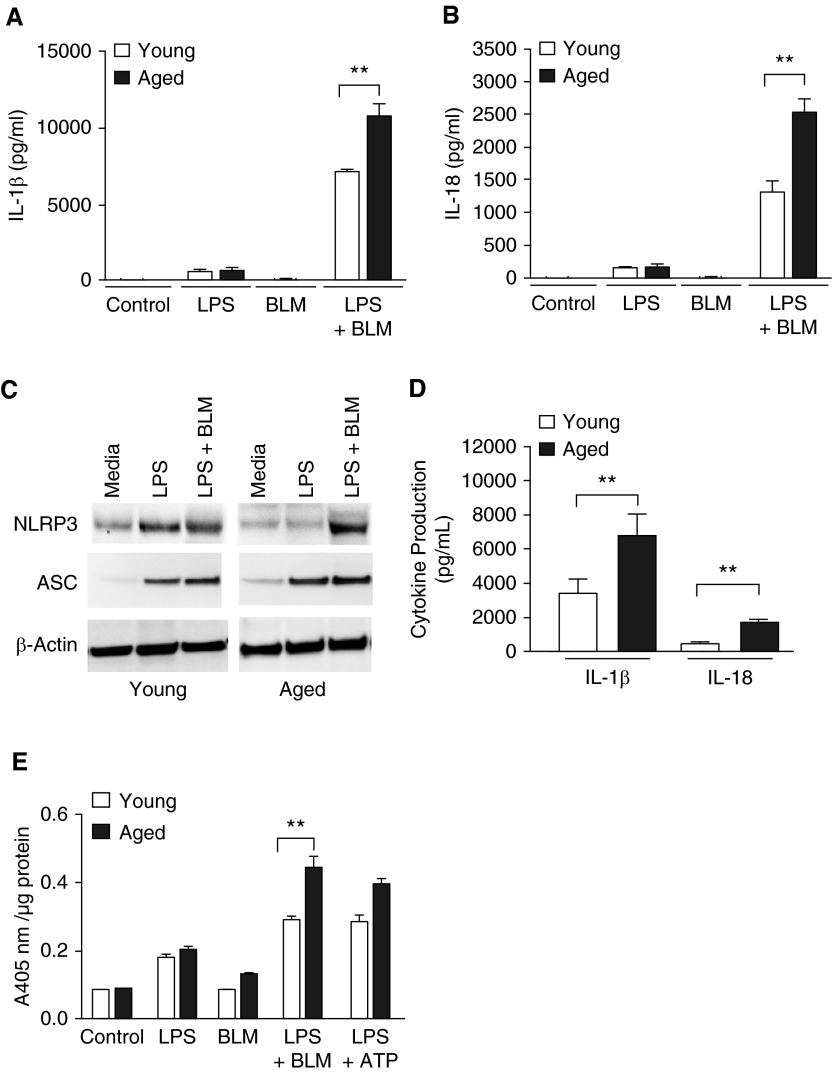
The NLRP3 inflammasome activates caspase-1 in response to many stimuli, including metabolic dysregulation, mitochondrial dysfunction, and ROS, resulting in the processing and secretion of IL-1β and IL-18 (24, 39–43). We examined the impact of aging on NLRP3 inflammasome activity in macrophages in response to bleomycin. Alveolar macrophages from young and aged wild-type mice were pretreated with LPS for 4 hours to prime the NLRP3 inflammasome, as described previously (13), before overnight stimulation with bleomycin. Compared with young macrophages, LPS-primed, bleomycin-stimulated aged macrophages displayed increased expression of IL-1β and IL-18 (Figures 6A and 6B). These results were corroborated in bone marrow–derived macrophages (BMDMs) from the same animals (BMDMs are considerably more abundant and accessible than are alveolar macrophages). NLRP3, ASC, IL-1β and IL-18, and protein expression (Figures 6C and 6D) were all elevated significantly in stimulated aged BMDMs compared with young macrophages. These aged BMDMs also had higher levels of caspase-1 expression and caspase-1 activity (Figure 6E), as well as significantly increased NLRP3, ASC, pro–IL-1β, and pro–IL-18 mRNA expression, relative to young macrophages (Figures E4A and E4B).
Figure 6.
NLRP3 inflammasome–mediated expression and activity in aged alveolar and bone marrow–derived macrophages is increased in response to bleomycin. Young (2–4 mo of age) and aged (17–19 mo of age) alveolar and bone marrow–derived macrophages (BMDMs) were cultured in media alone or in media containing LPS (100 ng/ml) for 4 hours before treatment with media or media containing bleomycin (0.1 U) for 24 hours. BMDMs were obtained from bone marrow cells cultured with murine M-CSF (10 ng/ml) for 7 days in 37°C, 5% CO2. In alveolar macrophages, production of mature (A) IL-1β (P = 0.0039, t test) and (B) IL-18 (P = 0.0052, t test) was assessed by ELISA. In BMDMs treated with LPS and bleomycin, (C) expression of NLRP3 and ASC was examined by Western blotting, (D) production of IL-1β (P = 0.0068, t test) and IL-18 (P = 0.0086, t test) was measured by ELISA, and (E) caspase-1 activity was assessed by the caspase-1 assay (P = 0.0068, t test). Similar results were obtained from three or more independent experiments with an n ≥ 3 per experiment and are expressed as mean ± SEM. **P < 0.05. M-CSF, macrophage-colony stimulating factor.
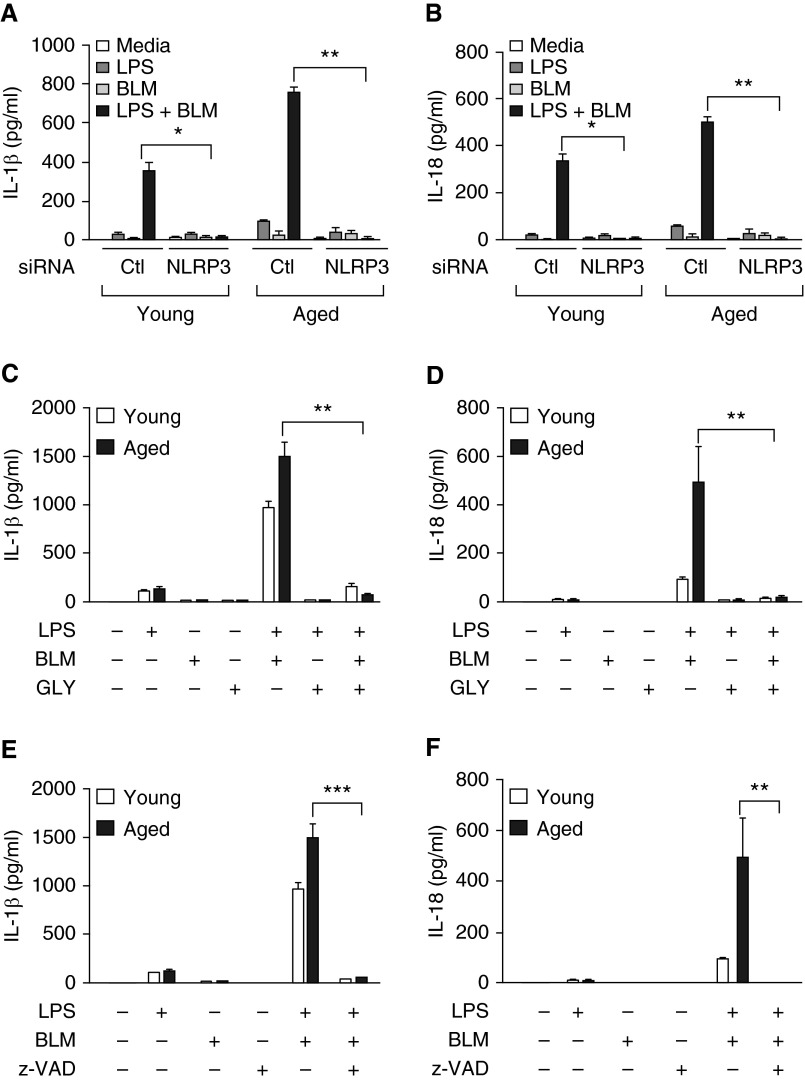
Using NLRP3-specific siRNA, we confirmed that IL-1β and IL-18 production by young and aged BMDMs in response to bleomycin was NLRP3 specific (Figures 7A and 7B and Figures E5A and E5B). Similarly, chemical inhibition of NLRP3 activity by glybenclamide (Figures 7C and 7D) or treatment with pan caspase-1 inhibitor V-ZAD-FMK (Figures 7E and 7F) resulted in decreased production of IL-1β and IL-18 by both young and aged LPS-primed, bleomycin-stimulated BMDMs.
Figure 7.
Production of IL-1β and IL-18 by bleomycin-treated macrophages is NLRP3 and caspase-1 dependent. Young (2–4 mo of age) and aged (17–19 mo of age) bone marrow cells were cultured with murine M-CSF (10 ng/ml) for 7 days in 37°C, 5% CO2. Cells were treated with (A and B) missense or NLRP3-specific small interfering (siRNA) for 24 hours before LPS (100 ng/ml, 4 h) stimulation or with (C and D) glybenclamide (25 µg/ml, 24 h) at the time of LPS stimulation. Cells were subsequently cultured with media or media containing bleomycin (0.1 U) for 24 hours. IL-1β and IL-18 production was assessed by ELISA. (A) IL-1β after siRNA: LPS + BLM treatment in young, *P = 0.022 (t test); LPS + BLM treatment in aged, **P = 0.0019 (t test). (B) IL-18 after siRNA: LPS + BLM treatment in young, *P = 0.022 (t test); LPS + BLM treatment in aged, **P = 0.0106 (t test). (C) IL-1β after glybenclamide: LPS + BLM + GLY treatment in aged, **P = 0.0055 (t test). (D) IL-18 after glybenclamide: LPS + BLM + GLY treatment in aged, **P = 0.0019 (t test). In additional experiments, at the time of bleomycin stimulation, cells were treated with z-VAD (10 µM, 1 h) before IL-1β and IL-18 measurement by ELISA. (E) IL-1β: LPS + BLM + z-VAD: ***P < 0.0001 (t test)] and (F) IL-18: LPS + BLM + z-VAD, **P = 0.0055 (t test). Similar results were obtained from three or more independent experiments with an n ≥ 3 per experiment and are expressed as mean ± SEM. Ctl, control; GLY, glybenclamide; z-VAD, Z-VADFMK.
Alterations in Mitochondrial Function in Aged Macrophages Contribute to Increased NLRP3 Inflammasome Expression in Response to Bleomycin
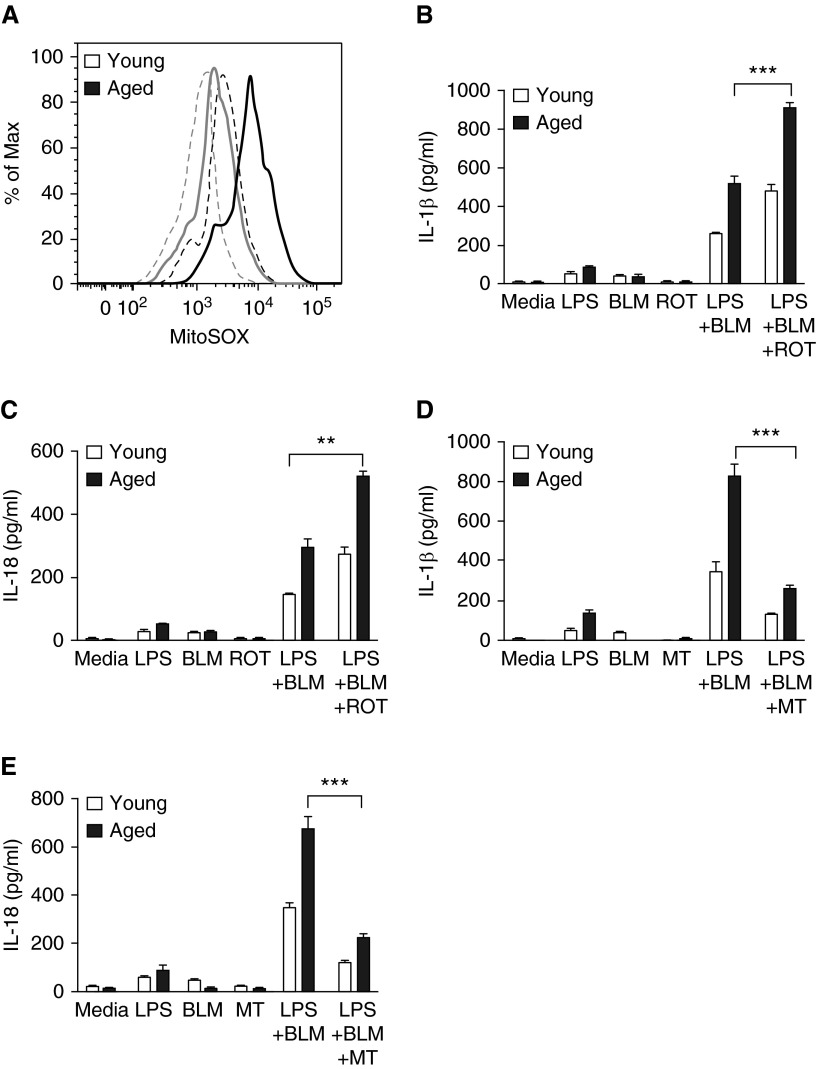
Increased mtROS production is required for the formation of the NLRP3 inflammasome (42, 43). Because aging is associated with increased levels of mtROS (44), we examined the impact of age-induced alterations in mitochondrial integrity on NLRP3 activation after bleomycin exposure. We evaluated whether increased mtROS production in aged BMDMs may contribute to augmented NLRP3 inflammasome activation in response to bleomycin. Elevated levels of mtROS occurred in both control and bleomycin-treated aged macrophages relative to young macrophages, as determined by MitoSOX fluorescence (Figure 8A). At the time of bleomycin stimulation, we treated young and aged macrophages with rotenone, a mitochondrial complex I inhibitor that induces mtROS production. Rotenone treatment increased mtROS in both young and aged macrophages and also enhanced IL-1β (Figures 8B and 8C). Conversely, treatment with mitoTEMPO, a mitochondria-targeted antioxidant, resulted in a significant decrease in IL-1β and IL-18 production in both young and aged macrophages after bleomycin treatment (Figures 8D and 8E).
Figure 8.
Alterations in mitochondrial function in aged macrophages after bleomycin treatment potentially contributes to increased NLRP3 inflammasome function. Young (2–4 mo of age) and aged (17–19 mo of age) bone marrow cells were cultured with M-CSF (10 ng/ml) for 7 days in 37°C, 5% CO2. On Day 7, LPS-primed cells were cultured for 24 hours with bleomycin (0.1 U). (A) Reactive oxygen species levels within mitochondria were measured by staining cells with MitoSOX at 5 µM for 40 min at 37°C. Levels of MitoSOX fluorescence were determined by flow cytometry. Cells treated with bleomycin and untreated cells are represented by solid and dashed lines, respectively. (B–C) Young and aged LPS-primed macrophages were treated with rotenone (5 µM) 1 hour before treatment with media alone or bleomycin. Cell culture supernatants were isolated, and production of IL-1β (B, LPS + BLM + ROT, aged: P = 0.0001 [t test]) and IL-18 (C, LPS + BLM + ROT, aged: P = 0.0013 [t test]) was assessed by ELISA. (D–E) Young and aged macrophages were treated with mitoTEMPO (100 µM) 1 hour before stimulation with media alone or LPS. Cells were subsequently treated with media alone or media containing bleomycin and production of IL-1β (D, LPS + BLM + MT, aged: P = 0.0001 [t test] and IL-18 (E, LPS + BLM + MT, aged: P = 0.0001 [t test]) was assessed at 24 hours by ELISA. Similar results were obtained from three or more independent experiments with an n ≥ 4 per experiment and are expressed as mean ± SEM. **P = 0.0013 and ***P < 0.05. MT, mitoTEMPO; ROT, rotenone.
Discussion
The increased burden of IPF in the aged population (>65 yr old) is well known. However, the molecular mechanisms by which aging influences susceptibility to pulmonary fibrosis remain unclear. Aging is associated with increased production of mtROS, oxidative damage to macromolecules, mitochondrial dysfunction, and alterations in cellular signaling cascades (22). Bleomycin administration, which promotes intracellular ROS production, results in an injury phase characterized by mitochondrial leakage and activation of mitochondria-dependent apoptotic pathways (45, 46). These disturbances may be sensed by the NLRP3 inflammasome, leading to enhanced processing and secretion of IL-1β and IL-18.
We demonstrated that IL-1β and IL-18 production by LPS-primed macrophages in response to bleomycin challenge is dependent on NLRP3 inflammasome activation. In agreement with previous studies, our data show that mtROS are crucial for NLRP3 inflammasome-mediated activation of IL-1β and IL-18 (43). Indeed, rotenone treatment, which elevates mtROS production, resulted in increased IL-1β and IL-18 production in bleomycin-treated young macrophages to levels similar to those of aged macrophages not treated with rotenone. Conversely, scavenging of mtROS using mitoTEMPO decreased IL-1β and IL-18 production by both young and aged bleomycin-treated macrophages. It is possible that treatment with mitoTEMPO could produce off-target effects, leading to cytotoxicity. It is important to note, however, that significantly higher concentrations of these cytokines were still detected in aged macrophages. Overall, these findings suggest that enhanced caspase-1 activity and IL-1β and IL-18 expression in aged macrophages treated with bleomycin may reflect age-associated impairments in mitochondrial function with an associated increase in the production of mtROS. The precise relationship between aging, inflammasome activation, and mitochondrial dysfunction requires further investigation.
Heightened activation of the NLRP3 inflammasome can result in increased acute lung injury (37, 47). Here, we demonstrate that the lungs of aged mice treated with bleomycin have increased fibrosis and NLRP3 inflammasome activation, and that mice deficient in the NLRP3 gene are protected from bleomycin-induced lung injury and fibrosis. Using a murine model of influenza infection, we found previously that age-related decrements in NLRP3 inflammasome activation can be detrimental by weakening antiviral responses, which can be rescued by treatment with nigericin, an activator of the NLRP3 inflammasome (27). Of note, nigericin and bleomycin are ROS-inducing agents that enhance NLRP3 inflammasome activation in aged dendritic cells and macrophages. Paradoxically, increased ROS production and NLRP3 inflammasome activation improve survival and resolution of influenza infection, whereas augmented ROS production in response to bleomycin can promote fibrosis and reduce survival in aged mice. It is plausible that age-related increases in ROS promote host adaptation, which provides protection from robust inflammatory responses. In response to influenza, viral inhibition of innate signaling may be affected disproportionately in aged cells. Changes in K+ efflux and increased ROS generation after nigericin treatment reduces this inhibition and improves inflammasome activation in aged hosts to levels similar to those of young, similarly treated counterparts. A significantly higher increase in ROS above basal levels, such as that which occurs in response to bleomycin, may alter regulatory signaling cascades present in aged lung, thereby resulting in increased NLRP3 inflammasome–mediated injury. Because the generation of ROS appears to be a common cellular response critical for NLRP3 inflammasome activation and because it is associated with responsiveness to a variety of stimuli in aged macrophages, future work is needed to investigate the source of ROS and the mechanism by which the NLRP3 inflammasome senses ROS generation.
Here, we show that NLRP3-dependent production of mature IL-1β and IL-18 is increased in aged macrophages. Expression of pro–IL-1β and pro–IL-18 was also markedly higher in aged lung homogenates and macrophage cultures, which may be attributed to enhanced nuclear factor-κB gene transcription resulting from increased cellular damage and oxidative stress caused by bleomycin. In addition, increased proapoptotic gene expression and levels of apoptosis after in vitro and in vivo administration of bleomycin may result in the release of ATP and the subsequent up-regulation of P2X7 receptor expression. Increased expression of P2X7 receptors in aged macrophages and aged lung (data not shown) would further amplify NLRP3 inflammasome signaling and recruitment of inflammatory mediators. It is important to note that although bleomycin may serve as the initial signaling component for NLRP3 inflammasome activation, dysregulated negative and positive feedback signaling in aged macrophages perpetuates inflammatory signaling, which further enhances apoptosis and subsequent tissue damage.
We have shown that during the fibrotic phase of bleomycin-induced lung injury and repair, there is increased expression of profibrotic genes and enhanced collagen deposition in the aged lung. Augmented expression of IL-1β and IL-18 and subsequent expression of TGF-β may contribute to the failure of the aging lung to resolve the residual inflammation incurred in response to recurrent microinjuries, thereby favoring the development of fibrotic lesions. Enhanced production of TGF-β, IL-1β, and IL-18, and subsequent increases in PAI-1 expression in aged lung, may further inhibit extracellular matrix degradation, increase epithelial cell death, and contribute to the persistent infiltration of inflammatory cells such as macrophages.
We have demonstrated that cellular infiltration is increased in aged lung in response to bleomycin. The observed increase in neutrophil recruitment in aged lung may be attributed to heightened NLRP3 inflammasome–dependent production of IL-1β and IL-18. IL-1β plays an important role in the neutrophil chemotactic response (48), whereas IL-18 interacts with IL-12 to induce IFN-γ, contributing to a wide variety of inflammatory processes (49). Increased neutrophil recruitment in aged hosts can contribute to exacerbated tissue injury and inflammatory responses (28). Aged lung epithelia have also been found to have an elevated expression of CCR2 associated with increased neutrophil and macrophage recruitment in the setting of bleomycin-induced lung injury and fibrosis. The CCR-CCL2 chemotactic system is one of the major signaling pathways that induce inflammation and apoptosis, and the contribution of each of these cell types to bleomycin-induced fibrosis and IPF, and the impact of aging on cellular functions, require further investigation.
Conclusions
In conclusion, our studies suggest that age-associated abnormalities, such as increased oxidative stress and NLRP3 inflammasome activation, potentially contribute to fibrotic lung injury. Therapeutics designed to target the inflammasome pathway and its downstream mediators may serve as novel strategies for the treatment of IPF.
Supplementary Material
Acknowledgments
Acknowledgments
The authors thank Steve Belinsky, Ph.D., for critical review of the manuscript. They also thank Maria Jiao for performing the immunohistochemistry and TUNEL staining (Tri-Institutional Laboratory of Comparative Pathology, Weill Cornell Medical College).
Footnotes
This work was supported by National Institutes of Health grants K01AG034999 and R21AG044755 (H.W.S.-D.), National Heart, Lung, Blood Institute grants U01 HL105371–01 and P01HL114501 (I.O.R.), and the Lovelace Respiratory Research Institute/Brigham and Women's Hospital Consortium for Lung Research.
Author Contributions: Conception and design: H.W.S.-D. and I.O.R.; analysis and interpretation: all authors; drafting/revision of the manuscript: all authors.
This article has an online supplement, which is accessible from this issue’s table of contents at www.atsjournals.org
Originally Published in Press as DOI: 10.1165/rcmb.2015-0222OC on March 2, 2016
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Kujoth GC, Hiona A, Pugh TD, Someya S, Panzer K, Wohlgemuth SE, Hofer T, Seo AY, Sullivan R, Jobling WA, et al. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science. 2005;309:481–484. doi: 10.1126/science.1112125. [DOI] [PubMed] [Google Scholar]
- 2.Vermulst M, Wanagat J, Kujoth GC, Bielas JH, Rabinovitch PS, Prolla TA, Loeb LA. DNA deletions and clonal mutations drive premature aging in mitochondrial mutator mice. Nat Genet. 2008;40:392–394. doi: 10.1038/ng.95. [DOI] [PubMed] [Google Scholar]
- 3.Howcroft TK, Campisi J, Louis GB, Smith MT, Wise B, Wyss-Coray T, Augustine AD, McElhaney JE, Kohanski R, Sierra F. The role of inflammation in age-related disease. Aging (Albany, NY) 2013;5:84–93. doi: 10.18632/aging.100531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Navaratnam V, Fleming KM, West J, Smith CJ, Jenkins RG, Fogarty A, Hubbard RB. The rising incidence of idiopathic pulmonary fibrosis in the U.K. Thorax. 2011;66:462–467. doi: 10.1136/thx.2010.148031. [DOI] [PubMed] [Google Scholar]
- 5.Bueno M, Lai YC, Romero Y, Brands J, St Croix CM, Kamga C, Corey C, Herazo-Maya JD, Sembrat J, Lee JS, et al. PINK1 deficiency impairs mitochondrial homeostasis and promotes lung fibrosis. J Clin Invest. 2015;125:521–538. doi: 10.1172/JCI74942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Selman M, Pardo A. Revealing the pathogenic and aging-related mechanisms of the enigmatic idiopathic pulmonary fibrosis. An integral model. Am J Respir Crit Care Med. 2014;189:1161–1172. doi: 10.1164/rccm.201312-2221PP. [DOI] [PubMed] [Google Scholar]
- 7.Gasse P, Mary C, Guenon I, Noulin N, Charron S, Schnyder-Candrian S, Schnyder B, Akira S, Quesniaux VF, Lagente V, et al. IL-1R1/MyD88 signaling and the inflammasome are essential in pulmonary inflammation and fibrosis in mice. J Clin Invest. 2007;117:3786–3799. doi: 10.1172/JCI32285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gasse P, Riteau N, Charron S, Girre S, Fick L, Pétrilli V, Tschopp J, Lagente V, Quesniaux VF, Ryffel B, et al. Uric acid is a danger signal activating NALP3 inflammasome in lung injury inflammation and fibrosis. Am J Respir Crit Care Med. 2009;179:903–913. doi: 10.1164/rccm.200808-1274OC. [DOI] [PubMed] [Google Scholar]
- 9.Cassel SL, Eisenbarth SC, Iyer SS, Sadler JJ, Colegio OR, Tephly LA, Carter AB, Rothman PB, Flavell RA, Sutterwala FS. The Nalp3 inflammasome is essential for the development of silicosis. Proc Natl Acad Sci USA. 2008;105:9035–9040. doi: 10.1073/pnas.0803933105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu J, Yan Z, Schwartz DE, Yu J, Malik AB, Hu G. Activation of NLRP3 inflammasome in alveolar macrophages contributes to mechanical stretch-induced lung inflammation and injury. J Immunol. 2013;190:3590–3599. doi: 10.4049/jimmunol.1200860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lasithiotaki I, Giannarakis I, Tsitoura E, Samara KD, Margaritopoulos GA, Choulaki C, Vasarmidi E, Tzanakis N, Voloudaki A, Sidiropoulos P, et al. NLRP3 inflammasome expression in idiopathic pulmonary fibrosis and rheumatoid lung. Eur Respir J. 2016;47:910–918. doi: 10.1183/13993003.00564-2015. [DOI] [PubMed] [Google Scholar]
- 12.Latz E, Xiao TS, Stutz A. Activation and regulation of the inflammasomes. Nat Rev Immunol. 2013;13:397–411. doi: 10.1038/nri3452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xu JF, Washko GR, Nakahira K, Hatabu H, Patel AS, Fernandez IE, Nishino M, Okajima Y, Yamashiro T, Ross JC, et al. COPDGene Investigators. Statins and pulmonary fibrosis: the potential role of NLRP3 inflammasome activation. Am J Respir Crit Care Med. 2012;185:547–556. doi: 10.1164/rccm.201108-1574OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu RM. Oxidative stress, plasminogen activator inhibitor 1, and lung fibrosis. Antioxid Redox Signal. 2008;10:303–319. doi: 10.1089/ars.2007.1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Araki T, Katsura H, Sawabe M, Kida K. A clinical study of idiopathic pulmonary fibrosis based on autopsy studies in elderly patients. Intern Med. 2003;42:483–489. doi: 10.2169/internalmedicine.42.483. [DOI] [PubMed] [Google Scholar]
- 16.Trujillo G, O’Connor EC, Kunkel SL, Hogaboam CM. A novel mechanism for CCR4 in the regulation of macrophage activation in bleomycin-induced pulmonary fibrosis. Am J Pathol. 2008;172:1209–1221. doi: 10.2353/ajpath.2008.070832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wilson MS, Madala SK, Ramalingam TR, Gochuico BR, Rosas IO, Cheever AW, Wynn TA. Bleomycin and IL-1β-mediated pulmonary fibrosis is IL-17A dependent. J Exp Med. 2010;207:535–552. doi: 10.1084/jem.20092121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bitterman PB, Adelberg S, Crystal RG. Mechanisms of pulmonary fibrosis. Spontaneous release of the alveolar macrophage-derived growth factor in the interstitial lung disorders. J Clin Invest. 1983;72:1801–1813. doi: 10.1172/JCI111140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Baran CP, Opalek JM, McMaken S, Newland CA, O’Brien JM, Jr, Hunter MG, Bringardner BD, Monick MM, Brigstock DR, Stromberg PC, et al. Important roles for macrophage colony-stimulating factor, CC chemokine ligand 2, and mononuclear phagocytes in the pathogenesis of pulmonary fibrosis. Am J Respir Crit Care Med. 2007;176:78–89. doi: 10.1164/rccm.200609-1279OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gibbons MA, MacKinnon AC, Ramachandran P, Dhaliwal K, Duffin R, Phythian-Adams AT, van Rooijen N, Haslett C, Howie SE, Simpson AJ, et al. Ly6Chi monocytes direct alternatively activated profibrotic macrophage regulation of lung fibrosis. Am J Respir Crit Care Med. 2011;184:569–581. doi: 10.1164/rccm.201010-1719OC. [DOI] [PubMed] [Google Scholar]
- 21.Prasse A, Pechkovsky DV, Toews GB, Jungraithmayr W, Kollert F, Goldmann T, Vollmer E, Müller-Quernheim J, Zissel G. A vicious circle of alveolar macrophages and fibroblasts perpetuates pulmonary fibrosis via CCL18. Am J Respir Crit Care Med. 2006;173:781–792. doi: 10.1164/rccm.200509-1518OC. [DOI] [PubMed] [Google Scholar]
- 22.Cui H, Kong Y, Zhang H. Oxidative stress, mitochondrial dysfunction, and aging. J Signal Transduct. 2012;2012:646354. doi: 10.1155/2012/646354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Okuma T, Terasaki Y, Kaikita K, Kobayashi H, Kuziel WA, Kawasuji M, Takeya M. C-C chemokine receptor 2 (CCR2) deficiency improves bleomycin-induced pulmonary fibrosis by attenuation of both macrophage infiltration and production of macrophage-derived matrix metalloproteinases. J Pathol. 2004;204:594–604. doi: 10.1002/path.1667. [DOI] [PubMed] [Google Scholar]
- 24.Sutterwala FS, Ogura Y, Szczepanik M, Lara-Tejero M, Lichtenberger GS, Grant EP, Bertin J, Coyle AJ, Galán JE, Askenase PW, et al. Critical role for NALP3/CIAS1/Cryopyrin in innate and adaptive immunity through its regulation of caspase-1. Immunity. 2006;24:317–327. doi: 10.1016/j.immuni.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 25.Inaba K, Inaba M, Romani N, Aya H, Deguchi M, Ikehara S, Muramatsu S, Steinman RM. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med. 1992;176:1693–1702. doi: 10.1084/jem.176.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang X, Goncalves R, Mosser DM. The isolation and characterization of murine macrophages. Curr Protoc Immunol. 2008;Chapter 14:Unit 14.1. doi: 10.1002/0471142735.im1401s83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stout-Delgado HW, Vaughan SE, Shirali AC, Jaramillo RJ, Harrod KS. Impaired NLRP3 inflammasome function in elderly mice during influenza infection is rescued by treatment with nigericin. J Immunol. 2012;188:2815–2824. doi: 10.4049/jimmunol.1103051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stout-Delgado HW, Du W, Shirali AC, Booth CJ, Goldstein DR. Aging promotes neutrophil-induced mortality by augmenting IL-17 production during viral infection. Cell Host Microbe. 2009;6:446–456. doi: 10.1016/j.chom.2009.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stout-Delgado HW, Yang X, Walker WE, Tesar BM, Goldstein DR. Aging impairs IFN regulatory factor 7 up-regulation in plasmacytoid dendritic cells during TLR9 activation. J Immunol. 2008;181:6747–6756. doi: 10.4049/jimmunol.181.10.6747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Misharin AV, Morales-Nebreda L, Mutlu GM, Budinger GR, Perlman H. Flow cytometric analysis of macrophages and dendritic cell subsets in the mouse lung. Am J Respir Cell Mol Biol. 2013;49:503–510. doi: 10.1165/rcmb.2013-0086MA. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Egger C, Cannet C, Gérard C, Jarman E, Jarai G, Feige A, Suply T, Micard A, Dunbar A, Tigani B, et al. Administration of bleomycin via the oropharyngeal aspiration route leads to sustained lung fibrosis in mice and rats as quantified by UTE-MRI and histology. PLoS One. 2013;8:e63432. doi: 10.1371/journal.pone.0063432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Redente EF, Jacobsen KM, Solomon JJ, Lara AR, Faubel S, Keith RC, Henson PM, Downey GP, Riches DW. Age and sex dimorphisms contribute to the severity of bleomycin-induced lung injury and fibrosis. Am J Physiol Lung Cell Mol Physiol. 2011;301:L510–L518. doi: 10.1152/ajplung.00122.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gavrieli Y, Sherman Y, Ben-Sasson SA. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J Cell Biol. 1992;119:493–501. doi: 10.1083/jcb.119.3.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rameshwar P, Chang VT, Gascón P. Implication of CD44 in adhesion-mediated overproduction of TGF-β and IL-1 in monocytes from patients with bone marrow fibrosis. Br J Haematol. 1996;93:22–29. doi: 10.1046/j.1365-2141.1996.4631004.x. [DOI] [PubMed] [Google Scholar]
- 35.Kolb M, Margetts PJ, Anthony DC, Pitossi F, Gauldie J. Transient expression of IL-1β induces acute lung injury and chronic repair leading to pulmonary fibrosis. J Clin Invest. 2001;107:1529–1536. doi: 10.1172/JCI12568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gauldie J, Kolb M, Sime PJ. A new direction in the pathogenesis of idiopathic pulmonary fibrosis? Respir Res. 2002;3:1. doi: 10.1186/rr158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hoshino T, Okamoto M, Sakazaki Y, Kato S, Young HA, Aizawa H. Role of proinflammatory cytokines IL-18 and IL-1β in bleomycin-induced lung injury in humans and mice. Am J Respir Cell Mol Biol. 2009;41:661–670. doi: 10.1165/rcmb.2008-0182OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Segawa S, Goto D, Yoshiga Y, Sugihara M, Hayashi T, Chino Y, Matsumoto I, Ito S, Sumida T. Inhibition of transforming growth factor-β signalling attenuates interleukin (IL)-18 plus IL-2-induced interstitial lung disease in mice. Clin Exp Immunol. 2010;160:394–402. doi: 10.1111/j.1365-2249.2010.04094.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kanneganti TD, Ozören N, Body-Malapel M, Amer A, Park JH, Franchi L, Whitfield J, Barchet W, Colonna M, Vandenabeele P, et al. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature. 2006;440:233–236. doi: 10.1038/nature04517. [DOI] [PubMed] [Google Scholar]
- 40.Sutterwala FS, Ogura Y, Zamboni DS, Roy CR, Flavell RA. NALP3: a key player in caspase-1 activation. J Endotoxin Res. 2006;12:251–256. doi: 10.1177/09680519060120040701. [DOI] [PubMed] [Google Scholar]
- 41.Aimanianda V, Haensler J, Lacroix-Desmazes S, Kaveri SV, Bayry J. Novel cellular and molecular mechanisms of induction of immune responses by aluminum adjuvants. Trends Pharmacol Sci. 2009;30:287–295. doi: 10.1016/j.tips.2009.03.005. [DOI] [PubMed] [Google Scholar]
- 42.Elinav E, Strowig T, Henao-Mejia J, Flavell RA. Regulation of the antimicrobial response by NLR proteins. Immunity. 2011;34:665–679. doi: 10.1016/j.immuni.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 43.Nakahira K, Haspel JA, Rathinam VA, Lee SJ, Dolinay T, Lam HC, Englert JA, Rabinovitch M, Cernadas M, Kim HP, et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol. 2011;12:222–230. doi: 10.1038/ni.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lee HC, Wei YH. Mitochondria and aging. Adv Exp Med Biol. 2012;942:311–327. doi: 10.1007/978-94-007-2869-1_14. [DOI] [PubMed] [Google Scholar]
- 45.Wallach-Dayan SB, Izbicki G, Cohen PY, Gerstl-Golan R, Fine A, Breuer R. Bleomycin initiates apoptosis of lung epithelial cells by ROS but not by Fas/FasL pathway. Am J Physiol Lung Cell Mol Physiol. 2006;290:L790–L796. doi: 10.1152/ajplung.00300.2004. [DOI] [PubMed] [Google Scholar]
- 46.Brar SS, Meyer JN, Bortner CD, Van Houten B, Martin WJ., II Mitochondrial DNA-depleted A549 cells are resistant to bleomycin. Am J Physiol Lung Cell Mol Physiol. 2012;303:L413–L424. doi: 10.1152/ajplung.00343.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mitroulis I, Skendros P, Ritis K. Targeting IL-1β in disease; the expanding role of NLRP3 inflammasome. Eur J Intern Med. 2010;21:157–163. doi: 10.1016/j.ejim.2010.03.005. [DOI] [PubMed] [Google Scholar]
- 48.Richter AG, Perkins GD, Chavda A, Sapey E, Harper L, Thickett DR. Neutrophil chemotaxis in granulomatosis with polyangiitis (Wegener's) and idiopathic pulmonary fibrosis. Eur Respir J. 2011;38:1081–1088. doi: 10.1183/09031936.00161910. [DOI] [PubMed] [Google Scholar]
- 49.Okamura H, Tsutsi H, Komatsu T, Yutsudo M, Hakura A, Tanimoto T, Torigoe K, Okura T, Nukada Y, Hattori K, et al. Cloning of a new cytokine that induces IFN-γ production by T cells. Nature. 1995;378:88–91. doi: 10.1038/378088a0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.