ABSTRACT
This work analyzes the high-pressure (HP) germination of spores of the food-borne pathogen Clostridium perfringens (with inner membrane [IM] germinant receptors [GRs]) and the opportunistic pathogen Clostridium difficile (with no IM GRs), which has growing implications as an emerging food safety threat. In contrast to those of spores of Bacillus species, mechanisms of HP germination of clostridial spores have not been well studied. HP treatments trigger Bacillus spore germination through spores' IM GRs at ∼150 MPa or through SpoVA channels for release of spores' dipicolinic acid (DPA) at ≥400 MPa, and DPA-less spores have lower wet heat resistance than dormant spores. We found that C. difficile spores exhibited no germination events upon 150-MPa treatment and were not heat sensitized. In contrast, 150-MPa-treated unactivated C. perfringens spores released DPA and became heat sensitive, although most spores did not complete germination by fully rehydrating the spore core, but this treatment of heat-activated spores led to almost complete germination and greater heat sensitization. Spores of both clostridial organisms released DPA during 550-MPa treatment, but C. difficile spores did not complete germination and remained heat resistant. Heat-activated 550-MPa-HP-treated C. perfringens spores germinated almost completely and became heat sensitive. However, unactivated 550-MPa-treated C. perfringens spores did not germinate completely and were less heat sensitive than spores that completed germination. Since C. difficile and C. perfringens spores use different mechanisms for sensing germinants, our results may allow refinement of HP methods for their inactivation in foods and other applications and may guide the development of commercially sterile low-acid foods.
IMPORTANCE Spores of various clostridial organisms cause human disease, sometimes due to food contamination by spores. Because of these spores' resistance to normal decontamination regimens, there is continued interest in ways to kill spores without compromising food quality. High hydrostatic pressure (HP) under appropriate conditions can inactivate bacterial spores. With growing use of HP for food pasteurization, advancement of HP for commercial production of sterile low-acid foods requires understanding of mechanisms of spores' interactions with HP. While much is known about HP germination and inactivation of spores of Bacillus species, how HP germinates and inactivates clostridial spores is less well understood. In this work we have tried to remedy this information deficit by examining germination of spores of Clostridium difficile and Clostridium perfringens by several HP and temperature levels. The results may give insight that could facilitate more efficient methods for spore eradication in food sterilization or pasteurization, biodecontamination, and health care.
INTRODUCTION
A number of Clostridium species are of significant concern in the food and health care industries as they can cause some human disorders, including food poisoning (Clostridium perfringens), botulism (Clostridium botulinum), and severe and potentially fatal diarrhea (Clostridium difficile) (1–3). Notably, the spore form of these organisms is the etiological agent of these diseases, and spores are extremely resistant to many normal decontamination regimens. As a consequence, eradication of such spores from foodstuffs, health care facilities, or medical products requires rather severe treatments such as very high temperatures, a high radiation flux, or strong and in some cases rather toxic chemical agents (2, 4, 5).
One emerging technology that in recent years has attracted significant attention for reducing spore levels in foods or other materials is very high hydrostatic pressure (HP). In particular, HPs of >500 MPa in conjunction with elevated temperatures (T > 90°C, and with compression heating generating T ≈ 121°C) have commercial potential for the preservation of low-acid, shelf-stable, high-value foods (6, 7). Research in recent years on how HP at elevated temperatures results in spore killing, primarily on spores of various Bacillus species, has indicated that HP generally kills spores by triggering spore germination (8–12), and the elevated temperatures can now kill the heat-sensitive germinated spores (6, 7, 10–12).
The mechanisms whereby HP triggers Bacillus spore germination have been well studied (8, 9, 13, 14). HPs of 50 to 300 MPa and T = 37°C activate spores' inner membrane (IM) germinant receptors (GRs), which recognize nutrient germinants, thus triggering spore germination. Treatment of B. subtilis spores at 150 MPa can also result in spores that are activated for germination, although these activated spores can decay to the unactivated dormant spore state depending on the incubation conditions (15). Interestingly, the 150-MPa HP germination of spores of some Bacillus species can be greatly potentiated by a heat activation step prior to HP treatment, most likely by activating at least some IM GRs (16). In contrast, HPs of 400 to 1,000 MPa (550 MPa and 50°C for B. subtilis spores) trigger the opening of a SpoVA protein channel in spores' IM, allowing the release of the spore core's huge depot (∼25% of dry weight) of dipicolinic acid (DPA) in a 1:1 chelate primarily with Ca2+ (14). The DPA-less spores that are generated by very high pressure then germinate spontaneously (17), as one or more enzymes that degrade the spores' peptidoglycan (PG) cortex (cortex-lytic enzymes [CLEs]) become activated upon DPA release, allowing for cortex hydrolysis to complete spore germination. This overall process thus converts a dormant, highly resistant spore that appears bright in phase-contrast microscopy to a metabolically active, fully germinated spore that has lost the dormant spores' resistance properties and appears dark in phase-contrast microscopy.
While how HP triggers Bacillus spore germination is fairly well known, less is known of how clostridial spores are germinated by HP. This is particularly notable, since there are important differences in the mechanisms of germination of spores of Bacillus and at least some Clostridium species (18–21). As noted above, germination of spores of Bacillus species is normally triggered by the binding of nutrient germinants to IM GRs, and this triggers DPA release. DPA release then activates CLEs in spores' outer layers either directly or indirectly, leading to initiation of cortex PG hydrolysis; accordingly, genetically CaDPA-less Bacillus spores rapidly hydrolyze cortex PG spontaneously (17). While spores of C. perfringens have similar IM GRs and many of the components of the SpoVA channel found in Bacillus spores and might be expected to germinate similarly to B. subtilis spores with HP (21), ultimately a protease in spores' outer layers (OL) external to spores' IM must be activated to cleave and activate a zymogen of the only essential CLE, pro-SleC, and active SleC then initiates cortex PG degradation (19, 21). The OL includes spores' PG cortex and germ cell wall, coat, and exosporium. While the signaling pathway leading to DPA release and generation of active SleC in C. perfringens spores is not clear, IM GR activation in these spores may directly trigger DPA release, since (i) when these spores lack all known CLEs, CaDPA release still takes place, although ∼10-fold slower than in wild-type spores, and (ii) mutant C. perfringens spores that lack DPA are stable, do not spontaneously proceed to cortex PG lysis, and thus fail to complete germination (22–24). However, since DPA-less C. perfringens spores still respond to GR-dependent germinants by triggering SleC activation, cortex hydrolysis, and completion of germination (24), activated GRs may also trigger generation of active SleC directly. The situation is even more different in spores of some other clostridial species, most notably C. difficile, that lack IM GRs (21). In C. difficile, the GR appears to be a pseudoprotease in spores' OL that when bound to an appropriate bile salt activates the protease that activates the CLE zymogen pro-SleC (19, 20, 25–27). DPA release in C. difficile spore germination is absolutely dependent on initiation of cortex hydrolysis (28, 29), unlike the situation in Bacillus and C. perfringens spore germination (18, 22–24). As predicted from the latter result, genetically DPA-less C. difficile spores are also stable and do not spontaneously complete spore germination (26).
In the current work, we have examined the germination of spores of C. perfringens with and without a prior heat activation step and of spores of C. difficile following treatment with 150 MPA at 37°C and 550 MPa at 50°C. The effects of HP on these spores were followed by monitoring DPA release, completion of spore germination, and loss of spore viability and heat resistance. The results of this work provide new insight into mechanisms of HP germination of spores of Clostridium species and suggest ways to improve at least C. perfringens spore killing by HP and perhaps that of other Clostridium species as well.
MATERIALS AND METHODS
C. difficile and C. perfringens strains and spore preparation.
C. difficile strain ATCC 43601 (ribotype 031) was used in all experiments, and spores of C. difficile strain ATCC 43593 were also used in a few experiments. Spores of these strains were prepared anaerobically on 70:30 sporulation medium agar (30) and harvested, purified, and stored as described previously (31). Spore purification involved extensive washes with water, separation of dormant spores from debris, growing and sporulating cells and, germinated spores on a Histodenz gradient, and then several water washes (32). Spore purity was assessed by phase-contrast microscopy, and all preparations used were effectively free of growing cells, germinated spores, or cell debris (<2%).
The C. perfringens strain used was MRS101, a derivative of the food poisoning isolate SM101 lacking the gene encoding the enterotoxin CPE (33). Spores of this strain were prepared, purified, and stored as described previously (23, 33). The purified spores of this strain used in this work were effectively free of sporulating cells, germinated spores, and debris (<2%) as observed by phase-contrast microscopy.
HP treatments.
Spores at an optical density at 600 nm (OD600) of ∼3 (∼7 · 108 spores/ml; ∼1.5 ml) were dispensed in flexible pouches that were double sealed, placed in a second pouch that was also double sealed to prevent cross-contamination, and then treated for various HP hold times at either 37°C and 150 MPa or 50°C and 550 MPa as described previously (14, 34). Samples for time zero of HP treatment were subjected to the normal come-up to the target HP, which took ∼40 s (come-up time), but this was released within 1 to 2 s after the target pressure was achieved. C. perfringens spores were treated either directly or after a heat activation step (80°C; 10 min) with subsequent cooling, since previous work showed that C. perfringens spore germination was stimulated tremendously by heat activation (23), while C. difficile spore germination is not (29, 35). After HP treatments, all treated samples were placed on ice and then frozen ∼6 h later and kept frozen until further analyses.
Analysis of germination of HP-treated spores.
HP-treated samples were centrifuged in a microcentrifuge, and the supernatant fraction was analyzed for DPA by its fluorescence with TbCl3. Specifically, various aliquots of the supernatant fluid were mixed with 50 μl of 100 mM K-HEPES buffer (pH 7.4)–200 μM TbCl3, the volume was made to 200 μl, and the Tb-DPA fluorescence was measured in a multiwell fluorometer (Molecular Devices, Sunnyvale, CA), as described previously (36). Total DPA in spore samples was determined by boiling samples for 30 min, centrifuging, and determining the DPA level in the supernatant fluid as described above.
For analysis of individual spores that retained DPA, pelleted samples of HP-treated spores were suspended in ∼20 μl H2O, and ∼250 individual spores were examined simultaneously by differential interference contrast (DIC) microscopy at 25°C to identify spores that exhibited a significant decrease in their refractive index and either had only released their DPA or had both released DPA and also degraded their PG cortex (37). In this analysis, individual spores were captured by laser tweezers, and their Raman spectra were determined. The spectra of 100 individual spores were acquired to determine the percentage of spores in any sample that retained DPA. To determine the DPA concentration in the spores that retained DPA, Raman spectra of individual spores were acquired using a laser power of 20 mW at 780 nm with an integration time of 20 s. The DPA concentration in these spores was determined from the intensity of the major CaDPA Raman peak at 1,017 cm−1 relative to the peak intensity of this same band from a CaDPA solution of known concentration. The Raman spectra of 30 treated spores that did or did not retain CaDPA were averaged as described previously (37–40).
Aliquots of HP-treated spores were also examined in a phase-contrast microscope to measure the percentage of spores that had become phase dark (100 to 200 individual spores were examined) and had thus released DPA, hydrolyzed cortex PG, and undergone core swelling; these spores have undoubtedly completed the germination process. In this microscopy, it was often notable that many HP-treated spores had become slightly dimmer than the starting dormant spores. While the appearance of these dimmer spores generally tracked with the levels of spores that had lost DPA, it was not easy to consistently see the difference between phase-bright and phase-dim spores in our microscope, and thus numbers of phase-dim and phase-bright spores were added together.
Analysis of the viability and heat sensitivity of HP-treated spores.
The viability of C. perfringens spores given various HP treatments was determined as described previously on brain heart infusion (BHI) agar plates (24) with or without a heat treatment at 89°C for 30 min on the HP-treated spores. Aliquots of spores were serially diluted in water, duplicate 10-μl aliquots of various dilutions were applied to BHI plates that were incubated anaerobically at 37°C for 24 h, and colonies were counted.
The viability of C. difficile spores after the HP treatments, and with or without subsequent heat treatment, was determined as previously described (26). Approximately 3 × 107 spores (equivalent to 1.10 OD600 units) were resuspended in 300 μl of water, and the sample was equally divided into three sets. One set was left untreated, while the other two sets were heated at either 70°C or 80°C for 15 min. Aliquots of these spores were then serially diluted in phosphate-buffered saline, and 10-μl aliquots of the resulting dilutions were plated in duplicate or in triplicate onto BHIS agar plates containing 0.1% taurocholate (TA). After ∼22 h, colonies arising from germinated spores were counted.
RESULTS
Germination of C. difficile spores by HP treatment at 150 and 550 MPa.
Previous work has shown that an HP of ∼150 MPa triggers Bacillus spore germination by activating GRs in spores' IM; this germination is accompanied by DPA release as well as cortex hydrolysis, and >90% of these spores exhibit these changes after 7 min of 150-MPa HP treatment at 37°C (8, 13). Since C. difficile spores lack IM GRs, it was not surprising that C. difficile spores released very little DPA during exposure to 150 MPa of HP, even after 20 min; this was the case when DPA release was measured either by bulk determinations or by assessing the percentage of individual spores that had released DPA (Fig. 1 [note scale on vertical axis]). In addition, <4% of the spores treated for periods of up to 20 min with 150 MPa of HP had become dark in the phase-contrast microscope, consistent with minimal completion of spore germination with this HP level (Fig. 1). Not only did the level of C. difficile spores that retained DPA not decrease significantly during HP treatment at 150 MPa, but the average DPA concentration in these individual spores also did not decrease significantly during this treatment (Table 1). This finding was consistent with the minimal levels of DPA release, if any, from spores during this HP treatment (Fig. 1). C. difficile ATCC 43593 spores behaved almost identically to ATCC 43601 spores during 150-MPa treatment (data not shown).
FIG 1.
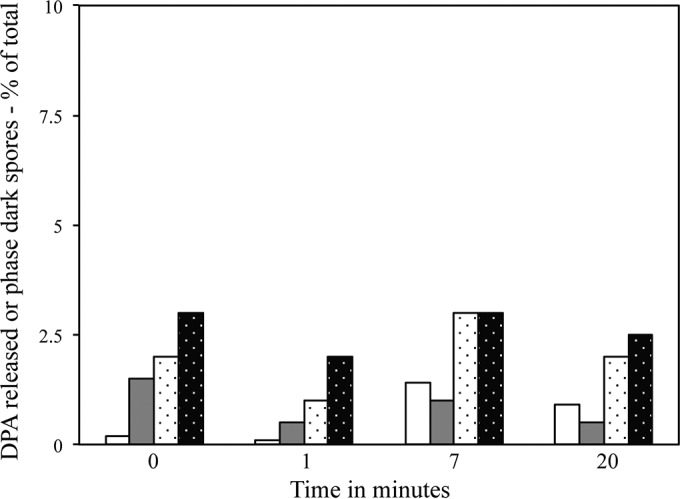
DPA release from and completion of germination of C. difficile spores given 150-MPa HP treatment. C. difficile ATCC 43601 spores were exposed to 150 MPa of HP for various times as described in Materials and Methods. Samples treated for various times were assayed for DPA release in separate experiments by measuring either DPA released in germination supernatant fluid (white bars) or the percentage of individual spores that had lost DPA (white stippled bars). In the two experiments, the percentages of spores that were phase dark and had thus completed germination were also measured (gray bars and black stippled bars). All values were corrected for the small percentage (<2%) of phase-dark spores in the starting spores and the small percentage (<5%) of DPA that was present in the supernatant fluid or absent from untreated spores, all as described in Materials and Methods. In a separate experiment, <5% of individual C. difficile spores had lost DPA or had become phase dark after 15 min of 150 MPa treatment (data not shown).
TABLE 1.
Average DPA concentrations in HP-treated clostridial spores that retained DPA
| Spores | HP hold time (min) | DPA concn (mM) with HP treatment ata: |
|
|---|---|---|---|
| 150 MPa | 550 MPa | ||
| C. difficile | 0 | 663 ± 153 | 663 ± 95 |
| 1 | 697 ± 170 | 669 ± 121 | |
| 3 | ND | 601 ± 76 | |
| 5 | 527 ± 119 | 702 ± 119 | |
| 7 | 544 ± 119 | 629 ± 80 | |
| 15 | ND | 684 ± 117 | |
| 20 | 510 ± 117 | ND | |
| C. perfringensb | 0 | 715 ± 243 | 715 ± 151 |
| 1 | 710 ± 234 | 779 ± 235 | |
| 3 | 651 ± 236 | 652 ± 144 | |
| 5 | 627 ± 214 | 711 ± 220 | |
| 7 | 779 ± 179 | 786 ± 160 | |
| 15 | 781 ± 259 | 623 ± 163 | |
DPA concentrations in C. difficile or C. perfringens spores that were HP treated for various times; retained DPA concentrations were determined as described in Materials and Methods. Values are averages and standard deviations for 30 individual spores that retained DPA. ND, not determined
These spores were not heat activated prior to HP treatment; so few spores retained DPA after HP treatment of heat-activated C. perfringens spores that measurements of DPA concentrations in these spores was too difficult.
In contrast to the effects of 150 MPa of HP on C. difficile spores, 550 MPa of HP triggers Bacillus spore germination in an IM GR-independent manner, almost certainly by activating the SpoVA proteins' DPA release channel in the spores' IM (13, 14, 41). C. difficile does have a spoVA operon expressed late in sporulation that was recently shown to be essential for transporting DPA into developing spores (26, 42, 43). One protein encoded by this operon, SpoVAC, has been shown to function as a mechanosensitive protein in B. subtilis (21, 41). Therefore, it was expected that 550 MPa of HP would stimulate DPA release in C. difficile spores, and this was indeed the case (Fig. 2A and B). Spores of C. difficile rapidly released DPA with 550 MP of HP, as measured either by bulk measurements of DPA released or by the percentage of individual spores that had released DPA (Fig. 2A and B). However, phase-contrast microscopy of spores treated with HP at 550 MPa for various times revealed that after all times of this HP treatment, <2% of the treated spores had become phase dark (Fig. 2A and B), although many appeared dimmer than starting spores in phase-contrast microscopy (see Materials and Methods) and had DIC image intensity lower than that of starting spores but higher than that of fully germinated spores (data not shown). Thus, even though essentially all DPA had been released after 15 or 20 min of 550-MPa HP exposure, the treated spores failed to hydrolyze their cortex and complete the germination process. The finding that cortex hydrolysis was not carried out in 550-MPa-treated C. difficile spores is similar to a previous finding that there is minimal if any cortex hydrolysis in C. difficile spores germinating with dodecylamine (29) (see Discussion). While almost all 550-MPa-treated C. difficile spores did release their DPA, those spores that retained their DPA had levels similar to those in the initial dormant spores (Table 1), indicating that DPA release by this HP is an all-or-none phenomenon, similar to the situation in Bacillus spores (15).
FIG 2.
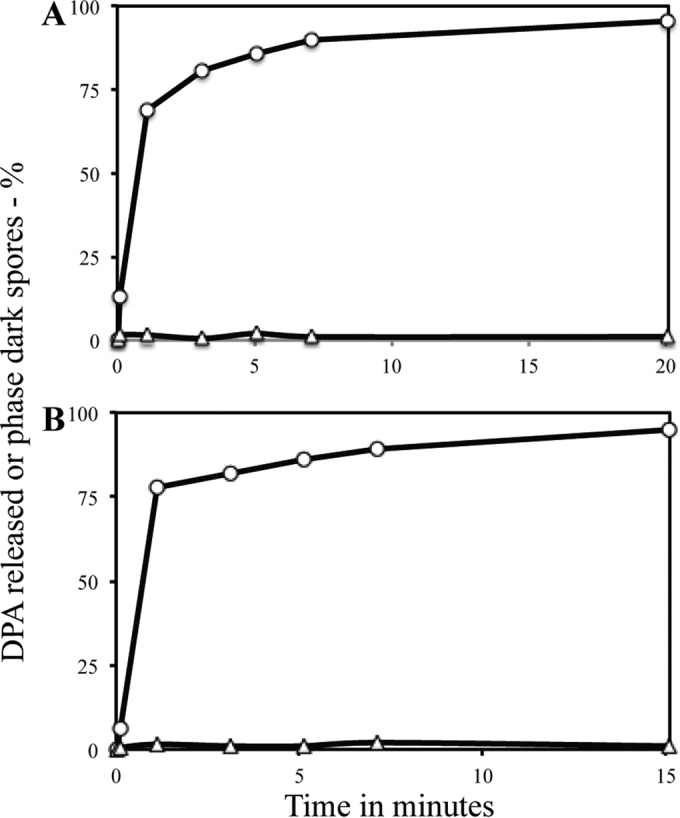
Percentages of individual C. difficile spores that had lost DPA or became phase dark after HP treatment at 550 MPa. C. difficile ATCC 43601 spores were exposed to 550 MPa of HP for various times as described in Materials and Methods. Samples treated for various times were assayed for DPA release (○) by measuring either DPA released in germination supernatant fluid (A) or the percentage of individual spores that had lost DPA or spores that had become phase dark (B) (△). All values were corrected for the small percentage (≤2%) of phase-dark spores in the starting spores and the small percentage (<5%) of DPA that was present in the supernatant fluid or absent from untreated spores, all as described in Materials and Methods. In a separate experiment, ∼90% of individual C. difficile spores had lost DPA after 15 min of 550-MPa treatment, while ≤3% of these individual spores had become phase dark (data not shown).
Germination of C. perfringens spores by HP treatment at 150 or 550 MPa.
In contrast to the minimal level of germination events, if any, in C. difficile spores treated with HP of 150 MPa, ∼75% of C. perfringens spores that had not been heat activated released their DPA after ∼1 min of this HP treatment (Fig. 3A). However, the appearance of phase-dark, fully germinated spores was much slower than DPA release in this experiment; after 15 min, only ∼30% of the spores had become phase dark, while ∼90% of spores had lost their DPA. As found with C. difficile spores treated with 550 MPa, C. perfringens spores that retained their DPA after 150-MPa treatment had DPA levels essentially identical to those in starting spores (Table 1).
FIG 3.
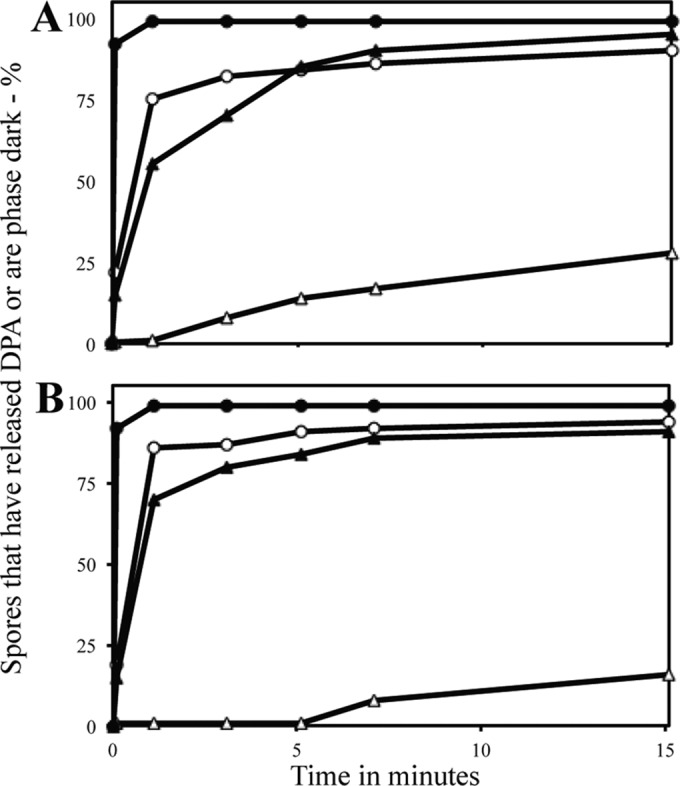
Percentages of individual C. perfringens spores that had released DPA or became phase dark after HP treatment at 150 MPa or 550 MPa with or without prior heat activation. C. perfringens spores that were unactivated (○, △) or heat activated (●, ▲) were HP-treated for various times with either 150 MPa (A) or 550 MPa (B) of HP, and ∼200 individual spores were examined for either DPA by DIC microscopy (○, ●) or dark spores by phase-contrast microscopy (△, ▲), all as described in Materials and Methods. All viabilities were measured in duplicate, and values shown were ±25%.
Previous work showed that germination of C. difficile spores is not stimulated by prior heat activation (29, 35). However, the germination of C. perfringens MRS101 spores with germinants recognized by IM GRs is stimulated >10-fold by brief heat activation at 80°C (23). Consequently, we examined the germination of heat-activated C. perfringens spores during a 150-MPa treatment (Fig. 3A). Strikingly, germination events as measured by either DPA release or appearance of phase-dark spores were much faster and more complete with the heat-activated spores (Fig. 3A). Indeed, most DPA was lost in the time to come to the target HP of 150 MPa. However, again, the appearance of phase-dark, fully germinated spores was significantly slower than the loss of DPA.
As seen with treatment at 150 MPa, treatment of unactivated and heat-activated C. perfringens spores with 550 MPa triggered release of DPA from most spores (Fig. 3B). With unactivated spores, the appearance of phase-dark spores was slower than DPA release, and it was even slower than with 150 MPa of HP. Heat-activated spores treated at 550 MPa of HP also had higher rates of DPA release and appearance of phase-dark spores than unactivated spores, although the appearance of phase-dark spores was still slower than DPA release.
Average Raman spectra of HP-treated spores that did or did not retain CaDPA.
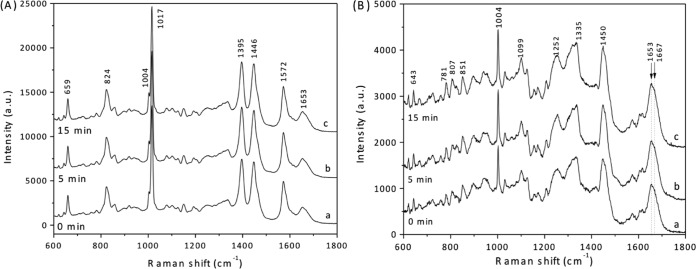
In order to obtain further information about the effects of HP on Clostridium spores, the average Raman spectra of ∼30 individual C. difficile and C. perfringens spores that retained DPA during HP treatment was determined (Fig. 4A and 5A and B; Table 1). The average Raman spectra of HP-treated C. difficile spores that retained DPA were similar for spores treated at 550 MPa either only during the ∼40-s come-up time to the target HP or for many minutes (Fig. 4A); this was also true for C. difficile spores treated at 150 MPa, as expected (data not shown). This finding indicates that the average DPA concentrations in the 550-MPa-treated spores that retained DPA did not decrease appreciably, if they decreased at all, during HP treatment, as noted above and found previously for Bacillus spores (14). This result again indicates that DPA release triggered by HP is an all-or-none phenomenon. Consistent with this conclusion, HP-treated (550 MPa) C. difficile spores that were scored as releasing DPA exhibited no DPA-specific peaks in their Raman spectra (Fig. 4B). Another region of spores' Raman spectra that can give significant information is the spectral region from 1,653 to 1,667 cm−1. In this region of the Raman spectrum, the peak at 1,653 cm−1 is due to the α-helical structure of proteins, while the peak at ∼1,665 cm−1 is due to disordered protein (38−40, 44). While there were some very small changes in this region of HP spores' Raman spectra, none were significant, and no trends in these changes were seen (Fig. 4B).
FIG 4.
Average Raman spectra of C. difficile spores exposed to HP for various times. C. difficile ATCC 43601 spores were exposed to an HP of 550 MPa for various times, and average Raman spectra of ∼30 spores that retained DPA (A) and ∼30 spores that did not retain DPA (B) after HP exposure for 0 min (curves a), 5 min (curves b), or 15 min (curves c) were obtained as described in Materials and Methods. Raman peaks at 824, 1,017, 1,395, and 1,572 cm−1 are due to CaDPA. The region around 1,653 to 1,667 cm−1 in these Raman spectra contains peaks that are from proteins in either the α-helical (1,653 cm−1) or unstructured (1,667 cm−1) conformation, as noted in the text.
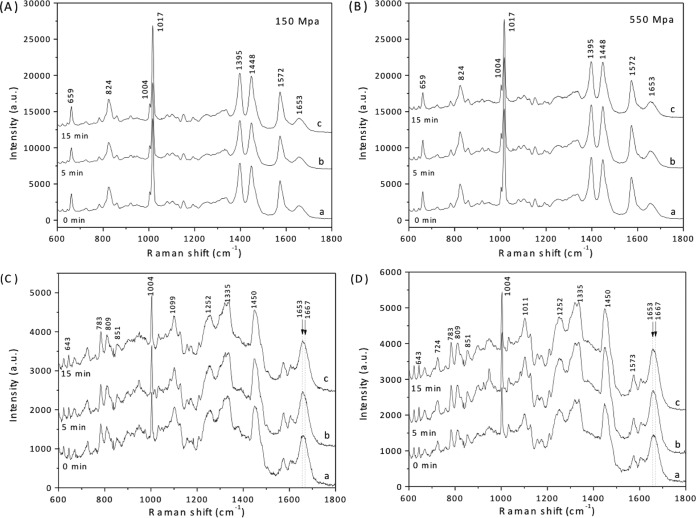
Average Raman spectra of C. perfringens spores that were HP treated at either 150 MPa or 550 MPa and that had retained their DPA also exhibited heights of DPA-specific peaks that were similar in spores from all HP treatment times examined (Fig. 5A and B). This result, combined with the absence of DPA-specific Raman peaks in the spectra of spores that were scored as not retaining DPA (Fig. 5C and D), further indicates that DPA release triggered from C. perfringens spores by HP treatment is an all-or-none phenomenon. In addition, the average Raman spectra of HP-treated C. perfringens spores exhibited little change in the region from 1,653 to 1,667 cm−1 irrespective of these spores' DPA levels (Fig. 5A to D). This finding suggests that these HP treatments led to minimal protein denaturation.
FIG 5.
Average Raman spectra of C. perfringens spores exposed to HP for various times. Unactivated C. perfringens spores were exposed to HP of 150 (A and C) or 550 (B and D) MPa for various times, and average Raman spectra of ∼30 spores that retained DPA (A and B) and ∼30 spores that did not retain DPA (C and D) after HP exposure for 0 min (curves a), 5 min (curves b), or 15 min (curves c) were obtained as described in Materials and Methods. Raman peaks at 824, 1,017, 1,395, and 1,572 cm−1 are due to CaDPA. The region around 1,653 to 1,667 cm−1 in these Raman spectra contains peaks that are from proteins in either the α-helical (1,653 cm−1) or unstructured (1,667 cm−1) conformation, as described in the text.
Effects of HP treatment on C. difficile and C. perfringens spore viability and heat resistance.
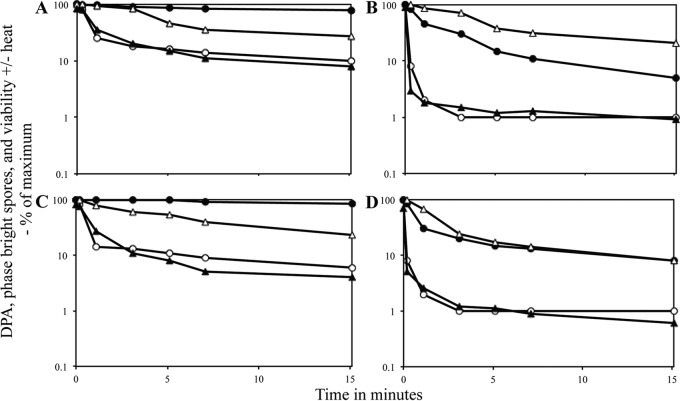
It was clear from the results noted above that 550 MPa of HP could trigger DPA release from C. difficile spores but not the completion of spore germination, while 150 and 550 MPa of HP triggered DPA release and completion of germination of C. perfringens spores. Important questions arising from these HP treatments are whether (i) triggering DPA release alone will be sufficient to lead to significant spore killing by some mechanism and (ii) spores that have released DPA but have not completed germination have become more heat sensitive than dormant spores. To test the effects of HP on spore viability, samples of HP-treated spores were applied to appropriate plates and spore viability was determined. In addition, treated spores were first given a heat treatment (C. difficile for 15 min and 70°C or 80°C; C. perfringens for 30 min at 89°C), and then spore viability was determined. Such heat treatments are relatively innocuous to fully dormant C. difficile ATCC 43601 spores and cause only minimal killing of fully dormant C. perfringens spores (see below). These heat treatments might well rapidly kill DPA-less spores that have an elevated core water content, even if they have not hydrolyzed their PG cortex and completed germination, and should kill fully germinated spores that have an even higher core water content (17, 24, 26).
The results of the experiments noted above with C. difficile spores treated at 150 MPa of HP were that (i) there was no significant decrease in spore viability over a 15-min treatment and (ii) the treated spores exhibited no decrease in viability upon wet heat treatment (Fig. 6A). Notably, mutant spores of C. difficile that lack DPA because of the absence of DPA synthase remain phase bright because cortex PG is not hydrolyzed, but they are killed more rapidly at 80°C than are DPA-replete spores, presumably because the DPA-less spores have a higher core water content than DPA-replete spores (26). Surprisingly, with spores treated at 550 MPa of HP there was (i) no decrease in spore viability during the HP treatment and (ii) no decrease in spore resistance to 80°C or 70°C as this HP treatment continued (Fig. 6B and data not shown).
FIG 6.
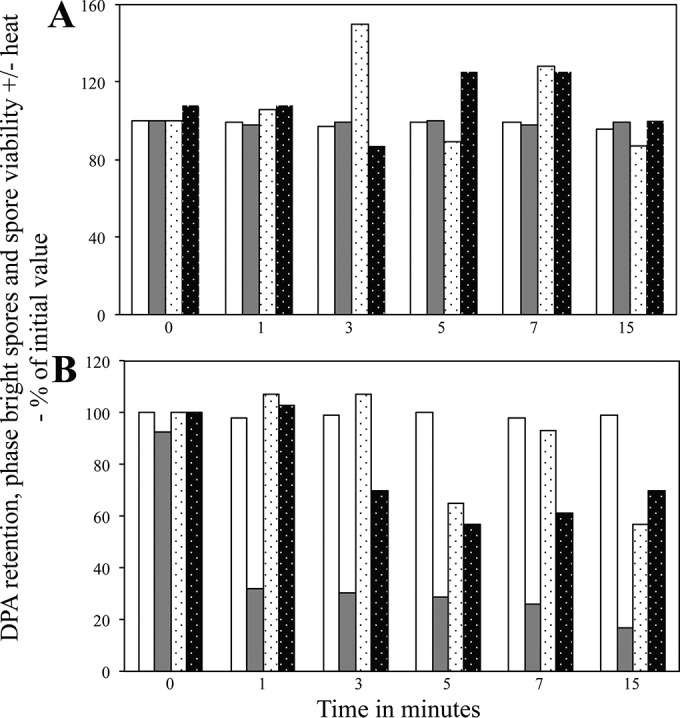
Viability, wet heat sensitivity, and germination events in C. difficile spores exposed to HP. Spores of C. difficile ATCC 43601 were exposed to HPs of either 150 MPa (A) or 550 MPa (B) for various times, and phase-bright spores, spores that retained DPA, spore viability, and spore viability after exposure to 80°C in water for 30 min were measured, all as described in Materials and Methods. The various parameters measured are shown as follows: phase-bright spores, white bars (100 individual spores examined); spores retaining DPA, gray bars (100 individual spores examined); spore viability, white stippled bars (determined in triplicate); and spore viability after heat treatment, black stippled bars (determined in duplicate for panel A and in triplicate for panel B). Values for spores that retained DPA and were phase bright are expressed relative to the average values in untreated spores; values for viability with and without a heat treatment are expressed relative to the viability of spores that were HP treated for 0 min and not then heat treated. In panel B, the values for viable spores at time zero are the averages of values for spores with and without heat treatment. Average standard deviations for viability values without heat treatment were ±38% in panel A and ±43% in panel B, and those for viability values with heat treatment were ±25% in panel A and ±43% in panel B.
In contrast to the results noted above with C. difficile spores given a 150-MPa HP treatment, similar treatment of unactivated C. perfringens spores resulted in (i) an ∼4-fold decrease in spore viability as well as (ii) a marked loss in spore heat resistance (Fig. 7A). Treatment of unactivated C. perfringens spores with 550 MPa of HP gave results very similar to those seen with 150 MPa; there was some loss in spore viability and some loss in wet heat resistance, as this HP treatment was continued, in contrast to results with C. difficile spores treated with 550 MPa of HP (Fig. 6B and compare Fig. 7A and C).
FIG 7.
Viability and wet heat sensitivity of C. perfringens spores exposed to HP. Spores of C. perfringens were exposed to an HP of either 150 MPa (A and B) or 550 MPa (C and D) for various times, either without (A and C) or with (B and D) prior heat activation, and spores that retained DPA, phase-bright spores, spore viability, and spore viability after exposure to 89°C in water for 30 min were measured, all as described in Materials and Methods. Symbols: ○, spores that retained DPA (100 individual spores examined); ●, phase-bright spores (100 individual spores examined); △, spore viability determined in duplicate; and ▲, spore viability after heat treatment determined in duplicate. Values for spores that retained DPA and were phase bright are expressed relative to the average values in untreated spores; values for viability with and without a heat treatment are expressed relative to the viability of spores that were neither HP nor heat treated. Standard deviations for viability values were between ±30 and 50% in panels A and B. An experiment identical to that for this figure gave very similar results (data not shown).
The results of measurements of spore viability and heat sensitivity upon HP treatment of heat-activated C. perfringens spores were similar in some ways to those with unactivated spores, in that spore viability decreased 3- to 4-fold after 15 min of HP at 150 or 550 MPa (Fig. 7B and D). However, spore viability after heat treatment decreased ∼10-fold more than that of unactivated spores when heat-activated spores were treated at either HP (compare Fig. 7A and C with Fig. 7B and D). It was also striking that when either unactivated or heat-activated spores were heat treated following various HP treatment times, the levels of the decreases in spore viability tracked well with the levels of spores that had lost DPA (Fig. 7A to D). In contrast, levels of phase-dark spores that had most likely completed germination triggered by HP treatments were not correlated with loss of either spore viability or DPA (Fig. 7A to D), although certainly all fully germinated spores have lost DPA.
DISCUSSION
In this study, we have shown that HP treatment triggers DPA release in spores of C. difficile and C. perfringens but does not induce complete C. difficile spore germination, in contrast with the case for C. perfringens spores, and that prior heat activation greatly stimulates HP germination of C. perfringens spores. There are three major events in the germination of spores of Bacillus spp. and C. difficile and C. perfringens (18–21, 28, 29, 45): (i) triggering of germination by germinants acting on either IM or OL GRs, (ii) release of DPA via IM SpoVA protein channels, and (iii) hydrolysis of the spores' cortex by CLEs (Fig. 8). Some components involved in these major events are similar in spores of Bacillus species and C. difficile and C. perfringens, in particular SpoVA proteins and IM GRs in C. perfringens, but others, such as the OL GR in C. difficile spores and CLEs in the clostridial spores, are different. One of the most striking differences is the order of the three major germination events with spores of Bacillus spp. and C. difficile (Fig. 8). Germinant sensing by GRs is the first event, but in Bacillus spores the second step is DPA release followed by cortex hydrolysis, while this order is reversed in C. difficile spore germination. In Bacillus spp., DPA release triggers CLE activation and CLE action is not essential for DPA release, although can accelerate it (18, 45–49); in contrast, in C. difficile spore germination, initiation of cortex hydrolysis by SleC precedes and triggers DPA release (28), much as lysozyme treatment of decoated Bacillus species in hypertonic medium triggers DPA release (48). Indeed, in C. difficile spores, the absence of SleC eliminates DPA release with physiological germinants (28, 29). Notably, DPA-less Bacillus spores are extremely unstable and germinate spontaneously (17, 49), while DPA-less C. difficile spores are stable (26).
FIG 8.

Major events and their order in the germination of spores of Bacillus species, C. difficile, and C. perfringens and where HPs feed in to the germination processes. Results supporting the outline of germination events and the signal transduction in this process are from references 17, 18, 19, 20, 22, 23, 24, 25, 26, 27, 28, 29, 33, 40, and 42. Germinants referred to are physiological germinants, such as amino acids, sugars, salts, or bile salts. Arrows show signal transduction pathways, and OL GRs are exemplified by the pseudoprotease in C. difficile spores' OL. While spores of C. perfringens have IM GRs similar to those in Bacillus spores, C. difficile spores have no IM GRs but rather have OL GRs. The question marks between the two pathways in C. perfringens spore germination indicate that the pathway(s) operating has not been definitively established.
DPA-less C. perfringens spores are also stable but are germinated by IM GR-dependent germinants, suggesting that the IM GRs in these spores can somehow trigger cortex hydrolysis (24). However, SleC is not essential for DPA release in C. perfringens spore germination, although SleC's absence slows DPA release from individual spores ∼10-fold in IM GR-dependent germination (22, 23). It has been generally suggested that in C. perfringens spore germination, IM GRs open the SpoVA protein channel for DPA (20). However, all these results suggest that in C. perfringens spores, activated IM GRs may trigger spore germination by activating either SleC or DPA release somewhat independently (Fig. 8).
The major differences in the signal transduction in germination of spores of Bacillus species, C. difficile, and C. perfringens likely explain much of the differential responses to HP treatments observed for C. difficile and C. perfringens spores. Thus, 150 MPa of HP has no effect on C. difficile spores, since IM GRs are absent and presumably the OL pseudoprotease that recognizes germinants (25) is not affected by this HP. Indeed, B. subtilis spores lacking all IM GRs germinate extremely poorly with 150 MPa of HP (8). However, with C. perfringens spores, which contain IM GRs, germination events, including DPA release and at least some cortex hydrolysis, are triggered by 150 MPa of HP. Presumably the IM GRs activated by this HP in turn either (i) activate the SpoVA channel leading to DPA release, with this release somehow leading to generation of active SleC, (ii) directly stimulate the activity of a Csp protease which then proteolytically activates pro-SleC, leading to initiation of cortex hydrolysis and then DPA release, or (iii) perhaps activate both pathways, leading to completion of spore germination. There is also evidence that a second CLE in C. perfringens spores, SleM, can play a small role in cortex hydrolysis in spore germination, although SleM alone is not sufficient for the germination of >99% of C. perfringens spores (22). The more rapid and complete germination of heat-activated C. perfringens spores exposed to 150 MPa of HP is consistent with the 150-MPa germination of spores of at least two Bacillus species being stimulated markedly by heat activation, a treatment that is thought to exert its effects primarily by increasing the responsiveness of IM GRs (16).
With spores of B. subtilis, treatment at 550 MPa leads to rapid completion of germination, even in the absence of all GRs. In contrast, with C. difficile spores, while 550-MPa treatment triggered DPA release, spore germination was not completed, presumably because these spores' signal transduction system leading to cortex hydrolysis is not activated by DPA release. This finding is consistent with the stability of genetically DPA-less C. difficile spores noted above. In contrast, not only did C. perfringens spores treated with 550 MPa release DPA but many of these DPA-less spores, in particular when previously heat activated, completed germination and presumably degraded their cortex. In contrast, genetically DPA-less C. perfringens spores are stable and do not complete spore germination (24). At present we cannot definitively explain (i) why heat activation increases rates of 550-MPa germination so dramatically and (ii) why 550-MPa-treated C. perfringens spores can complete germination. A simple hypothesis to answer both of these questions is that the IM GRs in C. perfringens spores actually become activated by HPs lower than 550 MPa during the come-up time to this pressure (see Materials and Methods). Thus, the 550 MPa of HP treatment actually triggers germination in two ways: (i) by action of 550 MPa directly on the SpoVA channel stimulating DPA release and (ii) by activation of IM GRs during the come-up time to 550 MPa of HP, with this activated state of the IM GRs maintained for some time as shown for B. subtilis spores (15). This hypothesis is consistent with the loss of almost all DPA even before the target pressure of 550 MPa is reached, and with the large increase in 550-MPa germination with heat-activated spores, since heat activation is thought to act on IM GRs, and would explain the completion of germination by 550-MP-treated spores (Fig. 8) (16). Measuring DPA release in C. perfringens gerKC spores, which lack what appears to be the major GR protein involved in C. perfringens spore germination with physiological germinants (50), would be one way to test this hypothesis.
It is also informative here to consider spore germination by dodecylamine, a molecule that germinates spores of Bacillus species and C. difficile and C. perfringens (18, 23, 29, 48). Dodecylamine germination of spores of at least B. subtilis is independent of IM GRs or CLEs, but rather this agent directly activates the SpoVA channel for DPA release, probably by binding to the highly conserved SpoVAC protein (18, 41, 48). This agent results in complete germination of B. subtilis spores, since DPA release itself as well as the absence of DPA in the spore core triggers CLE action. However, while rapid DPA release is triggered by dodecylamine from C. difficile and C. perfringens spores, the DPA release is not followed by cortex hydrolysis (23, 27). Thus, the release of DPA from spores of the two clostridial organisms alone is not sufficient to activate spore cortex hydrolysis. Since 550 MPa of HP is thought to trigger spore germination in a manner similar to that of dodecylamine (41, 48), this finding is consistent with the C. perfringens spore germination seen at 550 MPa, in particular with heat-activated spores, being triggered by heat-activated IM GRs that had been further activated during the come-up time to 550 MPa.
Another notable result is that HP treatment at either 150 or 550 MPa resulted in ∼75% killing of C. perfringens spores in 15 min, whether they were or were not heat activated. However, why there is this killing is not clear. Raman spectra of individual treated spores that had lost DPA showed no evidence of significant protein denaturation, although we cannot rule out inactivation of one or a few essential HP-sensitive proteins. Another possibility is that while the untreated spores are not oxygen sensitive, perhaps spores that have been HP treated become oxygen sensitive even when they only have lost DPA, and this viability loss certainly does not correlate with HP-treated spores having completed germination and becoming phase dark.
The lack of any loss in spore viability upon treatment of C. difficile spores with 150 MPa of HP was not surprising, because these spores exhibited no germination events. However, that the 550-MPa HP-treated spores exhibited no viability loss was surprising, since these spores lost most DPA (Fig. 2). What was more surprising was that the 550-MPa-HP-treated C. difficile spores lost no viability after heat treatment (Fig. 6), in contrast to the decreased heat resistance of genetically DPA-less C. difficile spores (26). This discrepancy could be due to strain-specific differences in heat sensitivity, since the ATCC 43601 strain used here is a different ribotype than the JIR8094 strain used to generate the genetically DPA-less spores. However, another possible explanation is based on the observation that the DPA-less spores generated by disruption of dpaAB, encoding DPA synthetase, have subtle but visible alterations in spore morphology as seen by transmission electron microscopy (26), suggesting that the formation of spores in the absence of DPA leads to structural adaptations in these spores that are unlikely to occur in the HP-treated spores that have been exogenously induced to release their DPA.
Certainly a striking result in this work is that unactivated and heat-activated C. perfringens spores that were HP germinated for ∼15 min at either 150 or 550 MPa of HP had lost much heat resistance after ∼15 min of HP treatment. This led to a total decrease in spore viability of 1 to 2 logs, with the largest decreases seen with heat-activated spores. The most likely reason for loss of heat resistance during these HP treatments is release of DPA resulting in an increase in spore core water content that reduces spore wet heat resistance markedly (51). Indeed, for all HP germination conditions tested, decreases in spore DPA and viability after wet heat treatment were similar, and losses in phase-bright spores and spore viability before a heat treatment were much slower. These observations are consistent with the significantly decreased wet heat resistance of genetically DPA-less spoVA C. perfringens spores (24).
One further conclusion from the analysis given above is that elimination of C. difficile spores by HP processing will not be as effective in reducing levels of these spores as with spores of Bacillus species, since these HP-treated clostridial spores do not become as heat sensitive as they would if germination had gone to completion. In addition, it seems likely that use of a heat treatment step before HP processing to ensure that any C. perfringens spores present are heat activated could lead to more effective HP regimens to increase the killing of spores of this species, as well as spores of other clostridial organisms which have IM GRs, such as C. botulinum.
ACKNOWLEDGMENTS
C.J.D, Y.-Q.L., M.R.S., A.S., and P.S. conceived of the project; P.K.T. and A.S. prepared spores and quantitated them; C.J.D., F.E.F., W.L., F.C.N., B.S., P.S., A.S., and S.W. collected the data; C.J.D., Y.-Q.L., A.S., M.R.S., and P.S. analyzed the data; and P.S., C.J.D., F.E.F., Y.-Q.L., M.R.S., and A.S. wrote the manuscript.
REFERENCES
- 1.Kelly CP, LaMont JT. 2008. Clostridium difficile—more difficult than ever. N Engl J Med 359:1932–1940. doi: 10.1056/NEJMra0707500. [DOI] [PubMed] [Google Scholar]
- 2.Setlow P, Johnson EA. 2012. Spores and their significance, p 45–79. In Doyle MP, Buchanan R (ed), Food microbiology: fundamentals and frontiers, 4th ed ASM Press, Washington, DC. [Google Scholar]
- 3.Hoover DG, Rodriguez-Palacios A. 2013. Transmission of Clostridium difficile in foods. Infect Dis Clin North Am 27:675–685. doi: 10.1016/j.idc.2013.05.004. [DOI] [PubMed] [Google Scholar]
- 4.Setlow P. 2006. Spores of Bacillus subtilis: their resistance to radiation, heat and chemicals. J Appl Microbiol 101:514–525. doi: 10.1111/j.1365-2672.2005.02736.x. [DOI] [PubMed] [Google Scholar]
- 5.Nicholson WL, Munakata N, Horneck G, Melosh HJ, Setlow P. 2000. Resistance of Bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol Mol Biol Rev 64:548–572. doi: 10.1128/MMBR.64.3.548-572.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Knorr D, Froehling A, Jaeger H, Reineke K, Schlueter O, Schoessler K. 2011. Emerging technologies in food processing. Annu Rev Food Sci Technol 2:203–235. doi: 10.1146/annurev.food.102308.124129. [DOI] [PubMed] [Google Scholar]
- 7.Balasubramaniam VM, Martínez-Monteagudo SI, Gupta R. 2015. Principles and application of high pressure-based technologies in the food industry. Annu Rev Food Sci Technol 6:435–462. doi: 10.1146/annurev-food-022814-015539. [DOI] [PubMed] [Google Scholar]
- 8.Paidhungat M, Setlow B, Daniels WB, Hoover D, Papafragkou E, Setlow P. 2002. Mechanisms of induction of germination of Bacillus subtilis spores by high pressure. Appl Environ Microbiol 68:3172–3175. doi: 10.1128/AEM.68.6.3172-3175.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wuytack EY, Boven S, Michiels CW. 1998. Comparative study of pressure-induced germination of Bacillus subtilis spores at low and high pressures. Appl Environ Microbiol 64:3220–3224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reineke K, Mathys A, Heinz V, Knorr D. 2013. Mechanisms of endospore inactivation under high pressure. Trends Microbiol 21:296–304. doi: 10.1016/j.tim.2013.03.001. [DOI] [PubMed] [Google Scholar]
- 11.Lenz CA, Reineke K, Knorr D, Vogel RF. 2015. High pressure thermal inactivation of Clostridium botulinum type E endospores—kinetic modeling and mechanistic insights. Front Microbiol 6:652. doi: 10.3389/fmicb.2015.00652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sarker MR, Akhtar S, Torres JA, Paedes-Sabja D. 2015. High hydrostatic pressure-induced inactivation of bacterial spores. Crit Rev Microbiol 41:18–26. doi: 10.3109/1040841X.2013.788475. [DOI] [PubMed] [Google Scholar]
- 13.Black EP, Koziol-Dube KK, Guan D, Wei J, Setlow B, Cortezzo DE, Hoover DG, Setlow P. 2005. Factors influencing the germination of Bacillus subtilis spores via the activation of nutrient receptors by high pressure. Appl Environ Microbiol 71:5879–5887. doi: 10.1128/AEM.71.10.5879-5887.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Black EP, Wei J, Atluri S, Cortezzo DE, Koziol-Dube K, Hoover DG, Setlow P. 2007. Analysis of factors influencing the rate of germination of spores of Bacillus subtilis by very high pressure. J Appl Microbiol 102:65–76. doi: 10.1111/j.1365-2672.2006.03062.x. [DOI] [PubMed] [Google Scholar]
- 15.Kong L, Doona CJ, Setlow P, Li Y-Q. 2014. Monitoring rates and heterogeneity of high-pressure germination of Bacillus spores using phase-contrast microscopy of individual spores. Appl Environ Microbiol 80:345–353. doi: 10.1128/AEM.03043-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Luu S, Cruz-Mora J, Setlow B, Feeherry FE, Doona CJ, Setlow P. 2015. The effects of heat activation on Bacillus spore germination, with nutrients or under high pressure, with or without various germination proteins. Appl Environ Microbiol 81:2927–2938. doi: 10.1128/AEM.00193-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paidhungat M, Setlow B, Driks A, Setlow P. 2000. Characterization of spores of Bacillus subtilis which lack dipicolinic acid. J Bacteriol 182:5505–5512. doi: 10.1128/JB.182.19.5505-5512.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Setlow P. 2013. When the sleepers wake: the germination of spores of Bacillus species. J Appl Microbiol 115:1251–1268. doi: 10.1111/jam.12343. [DOI] [PubMed] [Google Scholar]
- 19.Paredes-Sabja D, Shen A, Sorg J. 2014. Clostridium difficile spore biology: sporulation, germination and spore structural proteins. Trends Microbiol 22:406–416. doi: 10.1016/j.tim.2014.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Olguin-Araneda V, Banawas S, Sarker MR, Paredes-Sabja D. 2015. Recent advances in germination of Clostridium spores. Res Microbiol 166:236–243. doi: 10.1016/j.resmic.2014.07.017. [DOI] [PubMed] [Google Scholar]
- 21.Paredes-Sabja D, Setlow P, Sarker MR. 2011. Germination of spores of Bacillales and Clostridiales species: mechanisms and proteins involved. Trends Microbiol 19:85–94. doi: 10.1016/j.tim.2010.10.004. [DOI] [PubMed] [Google Scholar]
- 22.Paredes-Sabja D, Setlow P, Sarker MR. 2009. SleC is essential for cortex peptidoglycan hydrolysis during germination of spores of the pathogenic bacterium Clostridium perfringens. J Bacteriol 191:2711–2720. doi: 10.1128/JB.01832-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang G, Zhang P, Paredes-Sabja D, Green C, Setlow P, Sarker MR, Li YQ. 2011. Analysis of the germination of individual Clostridium perfringens spores and its heterogeneity. J Appl Microbiol 111:1212–1223. doi: 10.1111/j.1365-2672.2011.05135.x. [DOI] [PubMed] [Google Scholar]
- 24.Paredes-Sabja D, Setlow B, Setlow P, Sarker MR. 2008. Characterization of Clostridium perfringens spores that lack SpoVA proteins and dipicolinic acid. J Bacteriol 190:4648–4659. doi: 10.1128/JB.00325-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Francis MB, Allen CA, Shrestha R, Sorg JA. 2013. Bile acid recognition by the Clostridium difficile germinant receptor, CspC, is important for establishing infection. PLoS Pathog 9:e1003356. doi: 10.1371/journal.ppat.1003356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Donnelly ML, Fimlaid KA, Shen A. 2016. Characterization of Clostridium difficile spores lacking either SpoVAC or DPA synthase. J Bacteriol 198:1694–1707. doi: 10.1128/JB.00986-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fimlaid KA, Jensen O, Donnelly ML, Francis MB, Sorg JA, Shen A. 2015. Identification of a novel lipoprotein regulator of Clostridium difficile spore germination. PLoS Pathog 11:e1005239. doi: 10.1371/journal.ppat.1005239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Francis MB, Allen CA, Sorg JA. 2015. Spore cortex hydrolysis precedes dipicolinic acid release during Clostridium difficile spore germination. J Bacteriol 197:2276–2283. doi: 10.1128/JB.02575-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang S, Shen A, Setlow P, Li Y-Q. 2015. Characterization of the dynamic germination of individual Clostridium difficile spores using Raman spectroscopy and differential interference contrast microscopy. J Bacteriol 197:2361–2373. doi: 10.1128/JB.00200-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Putnam EE, Nock AM, Lawley TD, Shen A. 2013. SpoIVA and SipL are Clostridium difficile spore morphogenetic proteins. J Bacteriol 195:1214–1225. doi: 10.1128/JB.02181-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Adams CM, Eckenroth BE, Putnam EE, Doublie S, Shen A. 2013. Structural and functional analysis of the CspB protease required for Clostridium spore germination. PLoS Pathog 9:1003165. doi: 10.1371/journal.ppat.1003165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paredes-Sabja D, Sarker MR. 2012. Adherence of Clostridium difficile spores to Caco-2 cells in culture. J Med Microbiol 61:1208–1218. doi: 10.1099/jmm.0.043687-0. [DOI] [PubMed] [Google Scholar]
- 33.Sarker MR, Carman RJ, McClane BA. 1999. Inactivation of the gene (cpe) encoding the Clostridium perfringens enterotoxin eliminates the ability of two cpe-positive C. perfringens type A human gastrointestinal isolates to affect rabbit ileal loops. Mol Microbiol 33:946–958. doi: 10.1046/j.1365-2958.1999.01534.x. [DOI] [PubMed] [Google Scholar]
- 34.Doona CJ, Ghosh S, Feeherry FF, Ramirez-Peralta A, Huang Y, Chen H, Setlow P. 2014. High pressure germination of Bacillus subtilis spores with alterations in levels and types of germination proteins. J Appl Microbiol 117:711–720. doi: 10.1111/jam.12557. [DOI] [PubMed] [Google Scholar]
- 35.Dembek M, Stable RA, Witney AA, Wren BW, Fairweather NF. 2013. Transcriptional analysis of temporal gene expression in germinating Clostridium difficile 630 endospores. PLoS One 15:e64011. doi: 10.1371/journal.pone.0064011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yi X, Setlow P. 2010. Studies of the commitment step in the germination of spores of Bacillus species. J Bacteriol 192:3424–3433. doi: 10.1128/JB.00326-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Huang S-S, Chen D, Pelczar PL, Vepachedu VR, Setlow P, Li Y-Q. 2007. Levels of Ca2+-dipicolinic acid in individual Bacillus spores determined using microfluidic Raman tweezers. J Bacteriol 189:4681–4687. doi: 10.1128/JB.00282-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhang P, Kong L, Setlow P, Li YQ. 2010. Characterization of wet heat inactivation of single spores of Bacillus species by dual-trap Raman spectroscopy and elastic light scattering. Appl Environ Microbiol 76:1796–1805. doi: 10.1128/AEM.02851-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Coleman WH, Chen D, Li Y-Q, Cowan AE, Setlow P. 2007. How moist heat kills spores of Bacillus subtilis. J Bacteriol 189:8458–8466. doi: 10.1128/JB.01242-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Coleman WH, Zhang P, Li Y-Q, Setlow P. 2010. Mechanism of killing of spores of Bacillus cereus and Bacillus megaterium by wet heat. Lett Appl Microbiol 50:507–514. doi: 10.1111/j.1472-765X.2010.02827.x. [DOI] [PubMed] [Google Scholar]
- 41.Velásquez J, Schuurman-Wolters G, Birkner JP, Abee T, Poolman B. 2014. Bacillus subtilis spore protein SpoVAC functions as a mechanosensitive channel. Mol Microbiol 92:813–823. doi: 10.1111/mmi.12591. [DOI] [PubMed] [Google Scholar]
- 42.Fimlaid KA, Bond JP, Schutz KC, Putnam EE, Leung JM, Lawley TD, Shen A. 2013. Global analysis of the sporulation pathway of Clostridium difficile. PLoS Genet 9:1003660. doi: 10.1371/journal.pgen.1003660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saujet L, Pereira FC, Serrano M, Soutourina O, Monot M, Shelyakin PV, Gelfand MS, Dupuy B, Henriques AO, Martin-Verstraete I. 2013. Genome-wide analysis of cell type-specific gene transcription during spore formation in Clostridium difficile. PLoS Genet 9:1003756. doi: 10.1371/journal.pgen.1003756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xie CA, Li Y-Q, Tang W, Newton RJ. 2003. Study of dynamic process of heat denaturation in optically trapped single microorganisms by near-infrared Raman spectroscopy. J Appl Phys 94:6138–6142. doi: 10.1063/1.1617359. [DOI] [Google Scholar]
- 45.Setlow P. 2014. Germination of spores of Bacillus species: what we know and don't know. J Bacteriol 196:1297–1305. doi: 10.1128/JB.01455-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Setlow B, Peng L, Loshon CA, Li YQ, Christie G, Setlow P. 2009. Characterization of the germination of Bacillus megaterium spores lacking enzymes that degrade the spore cortex. J Appl Microbiol 107:318–328. doi: 10.1111/j.1365-2672.2009.04210.x. [DOI] [PubMed] [Google Scholar]
- 47.Peng LD, Chen D, Setlow P, Li Y-Q. 2009. Elastic and inelastic light scattering from single bacterial spores in an optical trap allows monitoring of spore germination dynamics. Anal Chem 81:4035–4042. doi: 10.1021/ac900250x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vepachedu VR, Setlow P. 2007. Role of SpoVA proteins in the release of dipicolinic acid during germination of Bacillus subtilis spores triggered by dodecylamine or lysozyme. J Bacteriol 189:1565–1572. doi: 10.1128/JB.01613-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tovar-Rojo F, Chander M, Setlow B, Setlow P. 2002. The products of the spoVA operon are involved in dipicolinic acid uptake into developing spores of Bacillus subtilis. J Bacteriol 184:584–587. doi: 10.1128/JB.184.2.584-587.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Banawas S, Paredes-Sabja D, Korza G, Li Y, Hao B, Setlow P, Sarker MR. 2013. The Clostridium perfringens germinant receptor protein, GerKC, is located in the spore inner membrane and is crucial for spore germination. J Bacteriol 195:5084–5891. doi: 10.1128/JB.00901-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gerhardt P, Marquis RE. 1989. Spore thermoresistance mechanisms, p 43–63. In Smith I, Slepecky RA, Setlow P (ed), Regulation of prokaryotic development. American Society for Microbiology, Washington, DC. [Google Scholar]