Summary
Ampullary organ electroreceptors excited by weak cathodal electric fields are used for hunting by both cartilaginous and non-teleost bony fishes. Despite similarities of neurophysiology and innervation, their embryonic origins remain controversial: bony fish ampullary organs are derived from lateral line placodes, while a neural crest origin has been proposed for cartilaginous fish electroreceptors. This calls into question the homology of electroreceptors and ampullary organs in the two lineages of jawed vertebrates. Here, we test the hypothesis that lateral line placodes form electroreceptors in cartilaginous fishes by undertaking the first long-term in vivo fate-mapping study in any cartilaginous fish. Using DiI-tracing for up to 70 days in the little skate, Leucoraja erinacea, we show that lateral line placodes form both ampullary electroreceptors and mechanosensory neuromasts. These data confirm the homology of electroreceptors and ampullary organs in cartilaginous and non-teleost bony fishes and indicate that jawed vertebrates primitively possessed a lateral line placode-derived system of electrosensory ampullary organs and mechanosensory neuromasts.
Keywords: lateral line placode, electroreceptor, neuromast hair cell, parvalbumin-3, Eya4, chondrichthyan
Introduction
Electrosensory 'hair cells' excited by weak cathodal electric fields (Bodznick and Montgomery, 2005; Münz et al., 1984; Teeter et al., 1980) and innervated by lateral line nerves that project to a dorsal octavolateral nucleus in the medulla (Bullock et al., 1983) are present in both lineages of jawed vertebrates: all cartilaginous fishes (sharks, skates, rays and holocephalans) and some bony fishes (the lobe-finned salamanders, caecilians, coelacanths and lungfishes, and the ray-finned sturgeons, paddlefishes and bichirs) (Bullock et al., 1983; New, 1997; Northcutt, 1997; Schlosser, 2002a). These electroreceptors are housed in ampullary sense organs, i.e., sensory epithelia at the base of conductive jelly-filled canals that open to the surface via pores (Jørgensen, 2005). Their similar electrophysiology and innervation suggest they are homologous, but conflicting data on their embryonic origins call this homology into question. We have demonstrated experimentally, by direct cell lineage tracing in axolotl (Northcutt et al., 1995) and paddlefish (Modrell et al., 2011a), that ampullary organs in bony fishes are ancestrally derived from lateral line placodes, i.e., patches of thickened neurogenic cranial ectoderm that elongate in characteristic lines over the head and trunk, and which also form neuromasts containing mechanosensory hair cells (Aman and Piotrowski, 2011; Baker et al., 2008; Ghysen and Dambly-Chaudière, 2007; Ma and Raible, 2009; Sarrazin et al., 2010; Schlosser, 2002b; Schlosser, 2006). Analysis of gene and antigen expression in developing shark ampullary organs, however, has led to the suggestion that the electrosensory hair cells of cartilaginous fishes derive from the neural crest (Freitas et al., 2006). We sought to test the hypothesis that lateral line placodes give rise to ampullary organ electroreceptors in cartilaginous fishes by using in vivo fate-mapping techniques in the little skate, Leucoraja erinacea.
Materials and Methods
Embryo collection and DiI-labelling
L. erinacea eggs were obtained from the Marine Biological Laboratory (Woods Hole, MA, USA) and maintained in a flow-through seawater system at ambient temperature to approximately stage 25 (Maxwell et al., 2008). A window was cut in the eggcase, and the embryo and yolk sac removed to a Petri dish. Embryos were anaesthetized in a solution of ethyl 3-aminobenzoate methanesulfonate salt (MS-222, Sigma) in seawater. CellTracker™ CM-DiI (Invitrogen), diluted in 0.3M sucrose from a 5 μg/μl stock diluted in ethanol, was focally injected into the anterodorsal, anteroventral or posterior lateral line placode using a pulled glass capillary needle and a Picospritzer pressure injector. Embryos were allowed to recover in seawater, then replaced in their eggcases and left to develop in a flow-through seawater table for 6-10 weeks. Raja eglanteria embryos for scanning electron microscopy were obtained from Mote Marine Laboratory (Sarasota, FL, USA).
Histology, immunohistochemistry and in situ hybridization
L. erinacea embryos were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) overnight at 4ºC, rinsed three times in PBS and stored at 4ºC in PBS with 0.01% sodium azide. For histological analysis, DiI-labelled embryos were embedded in paraffin wax and sectioned at 10 μm as previously described (O'Neill et al., 2007). Immunohistochemistry with anti-parvalbumin-3 (gift of A. Hudspeth) was performed in wholemount and on sections as previously described (Modrell et al., 2011a). Nuclei were counterstained with DAPI. In situ hybridization for L. erinacea Eya4 (GenBank JQ425114) was performed in wholemount and on sections as previously described (O'Neill et al., 2007) except that slides were not treated with proteinase K before hybridization, and the colour reaction was performed using BM Purple (Roche).
Scanning electron microscopy
After fixation in 2% glutaraldehyde, R. eglanteria embryos were dehydrated into ethanol and processed with dimethoxypropane and CO2 or Freon 113 (1,1,2 trichlorotrifluoroethane) and Freon 13 (chlorotrifluoromethane). Specimens were mounted, sputter-coated with gold palladium alloy (300–400Å thick) and imaged.
Results and Discussion
Parvalbumin-3 is a marker of electrosensory and mechanosensory hair cells in the skate, Leucoraja erinacea
In order to identify ampullary organs more easily in L. erinacea embryos, we examined the expression of parvalbumin-3 (Pv3), a Ca2+-binding protein thought to be the major calcium buffer in mechanosensory hair cells of the inner ear and lateral line (Heller et al., 2002), and which we recently showed also to be expressed in ampullary electroreceptors in the paddlefish, Polyodon spathula (Modrell et al., 2011a), and the axolotl, Ambystoma mexicanum (Modrell and Baker, 2012). Wholemount immunostaining for Pv3 in L. erinacea at stage 33 (Maxwell et al., 2008) revealed expression in the entire cephalic network of lateral line organs (Fig. 1A), i.e., both in the lines of lateral line neuromasts in canals (Fig. 1B), and in the adjacent fields of ampullary organs located deep within the dermis (Fig. 1C). Hence, Pv3 is expressed in electrosensory hair cells in ampullary organs, as well as in mechanosensory hair cells, in both cartilaginous fishes and bony fishes (Modrell and Baker, 2012; Modrell et al., 2011a), suggesting that it acts as a calcium buffer for both types of sensory hair cell in all jawed vertebrates.
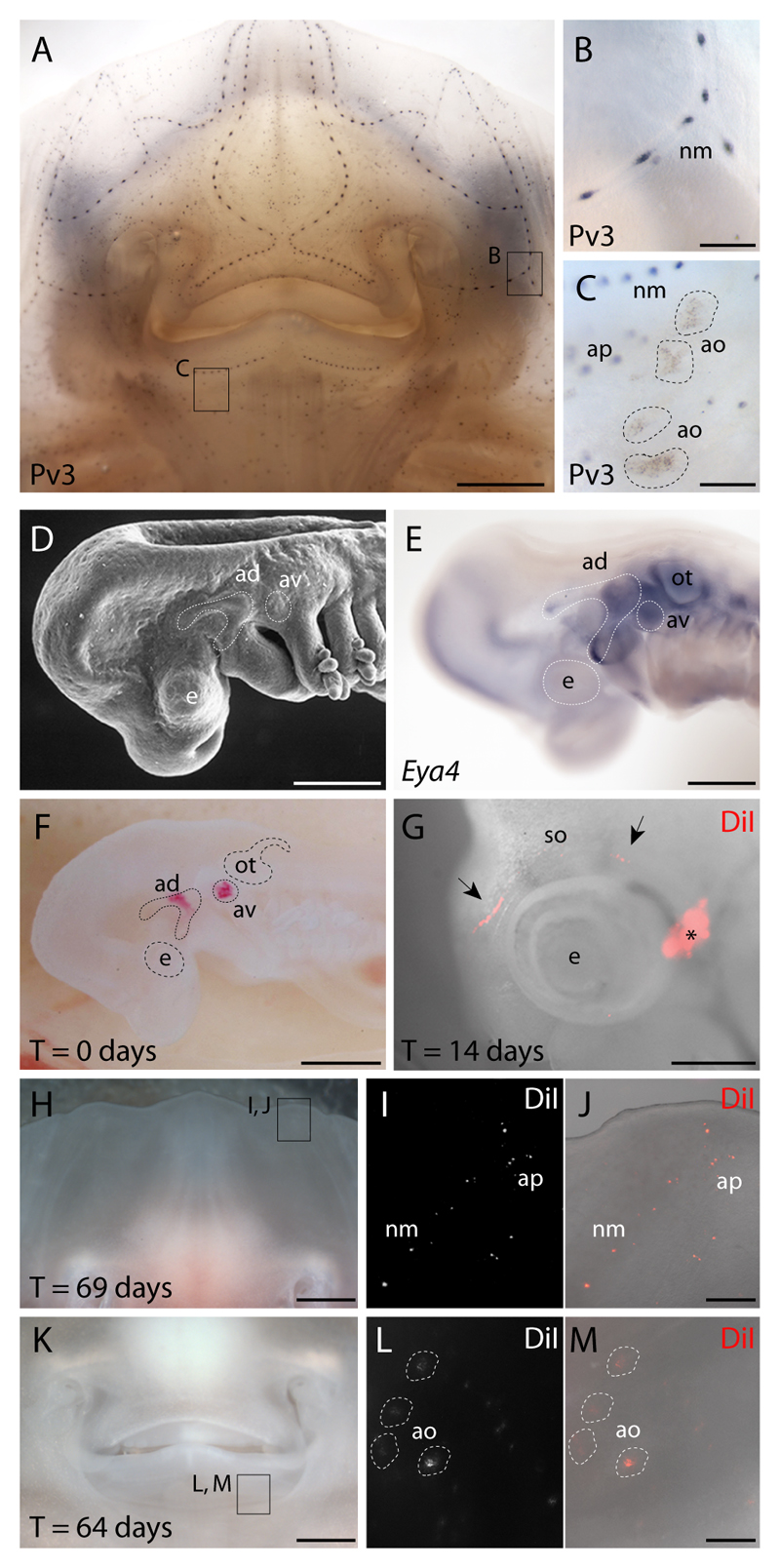
Fig. 1. Lateral line placodal origin of skate ampullary organs and neuromasts.
(A) Wholemount immunostaining for parvalbumin-3 (Pv3) in L. erinacea embryos reveals a cephalic network of (B) mechanosensory neuromasts and (C) electrosensory ampullary organs. In order to test the lateral line placodal origin of neuromasts and ampullary organs, we fate-mapped the anterodorsal or anteroventral lateral line placodes. These placodes were recognizable as (D) ectodermal thickenings caudal to the eye and dorsal to the mandibular and hyoid arches, respectively, which (E) express the transcription co-factor gene Eya4. (F) An embryo in which the anterodorsal and anteroventral lateral line placodes were focally labelled with DiI. (G) In an embryo in which the anterodorsal lateral line placode was labelled, DiI was observed at 14 days post-injection in the supraorbital lateral line primordium (black arrows), far from the original injection site (*). In embryos with DiI-labelled anterodorsal and/or anteroventral lateral line placodes, the distribution of DiI-positive cells at 60-70 days post-injection recapitulated the normal distribution of (H-J) cephalic neuromasts, ampullary pores and (K-M) ampullary organs. ad, anterodorsal lateral line placode; ao, ampullary organ; ap, ampullary tubule pore; av, anteroventral lateral line placode; e, eye; nm, neuromast; ot, otic vesicle; so, supraorbital lateral line primordium. Scale bars: A,H,K: 2.5 mm; B-C,J,M: 0.5 mm; D-E: 0.5 mm; G: 0.8 mm.
Lateral line placodes in L. erinacea give rise to cephalic ampullary organs and neuromasts
We aimed to test the hypothesis that lateral line placodes form both ampullary organs and neuromasts in cartilaginous fishes, by performing in vivo fate-mapping in the little skate. We used the fluorescent lipophilic dye DiI to label focally the anterodorsal lateral line placode alone, or the anterodorsal plus anteroventral lateral line placodes, in L. erinacea embryos at stage 25 (n=18). These placodes were selected because they are the largest cranial lateral line placodes, hence the easiest to target, and they give rise to both ampullary organs and neuromasts in the axolotl (Northcutt et al., 1995) and paddlefish (Modrell et al., 2011a). The anterodorsal and anteroventral lateral line placodes can be recognized in skate embryos both morphologically, as thickenings of ectoderm caudal to the eye and dorsal to the mandibular and hyoid arches, respectively (Fig. 1D), and molecularly, by expression of the gene encoding the transcription co-factor Eya4 (Fig. 1E), an established marker of the developing lateral line system in shark, paddlefish and axolotl embryos (Modrell and Baker, 2012; Modrell et al., 2011a; O'Neill et al., 2007). Figure 1F shows an example of an embryo photographed immediately after receiving a focal injection of DiI in both the anterodorsal and anteroventral lateral line placodes. In different DiI-injected L. erinacea embryos, DiI was incorporated into the elongating primordia of the anterodorsal placode (Northcutt, 2005) and/or anteroventral placodes. For example, in one DiI-labelled embryo in which the anterodorsal lateral line placode was labelled, DiI-positive cells were seen at 14 days post-injection within the migrating supraorbital primordium of the anterodorsal lateral line placode, far from the original injection site (Fig. 1G). At 69 days post-injection (Fig. 1H-J), DiI-positive cells were observed on the ventral surface of the same embryo in patterns that recapitulated the normal distribution of ampullary organs and neuromasts (Fig. 1I,J). We observed similar distributions of DiI in both neuromasts and ampullary organs (Fig. 1K-M) at 60-70 days post-injection in 17/18 injected embryos.
The skate anterodorsal and anteroventral lateral line placodes elongate to form sensory ridges that are closely associated with lines of mechanosensory neuromasts and fields of electrosensory ampullary organs. For example, the anterodorsal lateral line placode splits to form the infraorbital (Fig. 2A) and supraorbital (Fig. 2B) sensory ridges, which are closely associated with the ampullary organs and neuromasts located between the rostrum and the mouth (Northcutt, 2005). Differentiated ampullary organs in cartilaginous fishes are organized into fields of sensory ampullae (Fig. 2C) at the base of long, jelly-filled ampullary tubules that open externally (Jørgensen, 2005). In contrast, neuromasts are organized as lines and are regularly spaced within a continuous epithelial canal (Fig. 2D). Both ampullary organs (Fig. 2E) and neuromasts (Fig. 2F) contain Pv3-positive sensory receptor cells nested among non-sensory support cells. As in paddlefish and axolotl embryos (Modrell and Baker, 2012; Modrell et al., 2011a), the Pv3-positive sensory receptor cells of L. erinacea ampullary organs (Fig. 2G) and neuromasts (Fig. 2H) express Eya4. In order to determine the precise fate of DiI-labelled cells that originated from the lateral line placodes, we immunostained sections of 6 of our experimental embryos for Pv3. In all 6 embryos, DiI was observed in Pv3-positive sensory receptor cells (Fig. 2I), non-sensory support cells (Fig. 2I) and canal cells (Fig. 2J) of ampullary organs, and Pv3-positive sensory hair cells (Fig. 2K), non-sensory support cells (Fig. 2L) and canal cells (Fig. 2K) of neuromasts.
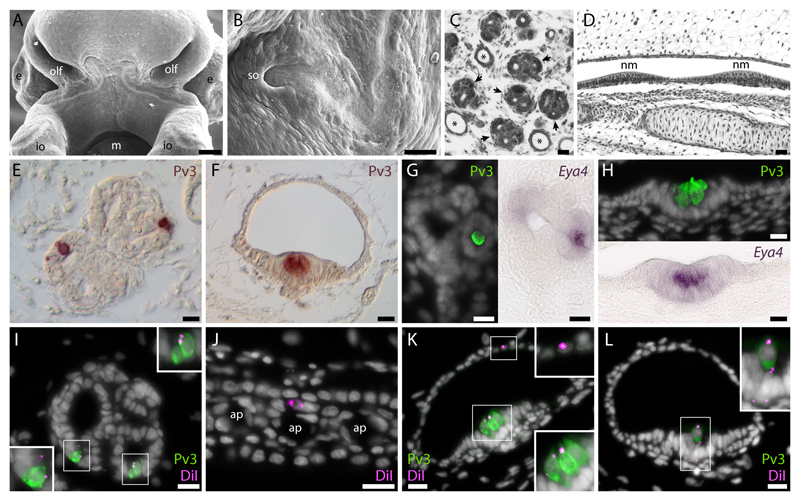
Fig. 2. Lateral line placodal origin of sensory receptor cells, support cells and canal cells in skate ampullary organs and cephalic neuromasts.
(A) A ventral view of the head, illustrating the elongation of the sensory ridge of the anterodorsal lateral line placode that will give rise to the infraorbital neuromasts and closely associated ampullary organs. (B) A lateral view of the head, illustrating the elongation of the sensory ridge of the anterodorsal lateral line placode as it passes rostral and ventral to the eye (where it will run medial to the more laterally situated infraorbital line), giving rise to the supraorbital neuromasts and closely associated ampullary organs. (C) Electrosensory ampullary organs (black arrows) are clustered within the dermis, and each sensory ampulla opens externally via a long jelly-filled ampullary canal (*). (D) Mechanosensory neuromasts are distributed within a continuous network of epithelial canals. Immunohistochemical localization of parvalbumin-3 (Pv3) in (E) ampullary organs and (F) neuromasts reveals small clusters of sensory receptor cells nested among non-sensory support cells. (G) Electrosensory hair cells and (H) mechanosensory hair cells both express Eya4, an established marker of the developing lateral line system. Fate-mapping of the anterodorsal lateral line placode of L. erinacea by DiI injection reveals a placodal origin of (I) the Pv3-positive sensory hair cells, non-sensory support cells and (J) canal cells of electrosensory ampullary organs, and (K) the Pv3-positive sensory hair cells, canal cells and (L) non-sensory support cells of mechanosensory neuromasts. Images in panels I,J,K,L are all from different individuals. ap, ampullary tubule pore; e, eye; io, infraorbital sensory ridge; m, mouth; nm, neuromast; olf, olfactory organ; so, supraorbital sensory ridge. Scale bars: A: 125 µm; B: 50 µm; C-D: 24 µm; E-L: 10 µm.
The posterior lateral line placode in L. erinacea gives rise to trunk neuromasts
Ampullary organs are confined to the head of cartilaginous fishes, but neuromasts are also found on the trunk. Grafting experiments performed a century ago revealed the posterior lateral line (PLL) placode origin of trunk neuromasts in the bony fish clade (Harrison, 1904). The skate PLL placode is easily recognized as a thickening of ectoderm dorsal to the gill arches (Fig. 3A) that elongates caudally along the trunk, forming a distinct ridge in the epidermis (Fig. 3B,C); it also expresses Eya4 (Fig. 3D). To complete our experimental study of the origin of lateral line organs in cartilaginous fishes, we labelled the PLL placode in L. erinacea with DiI (Fig. 3E, n=5): 50-65 days post-injection (Fig. 3F), all labelled PLL placodes had deposited clusters of DiI-positive cells along the trunk (Fig. 3G,H) in a pattern recapitulating the distribution of trunk neuromasts (as illustrated by wholemount Pv3 immunostaining; Fig. 3I). L. erinacea trunk neuromasts resemble head neuromasts, with regularly spaced clusters of Pv3-positive (Fig. 3J) and Eya4-positive (Fig. 3K) hair cells nested among non-sensory support cells in a continuous epithelial canal. In trunk sections of embryos with DiI-labelled PLL placodes, DiI was observed in both Pv3-positive hair cells and support cells (Fig. 3L). Hence, the PLL placode forms trunk neuromasts in cartilaginous fishes, as it does in bony fishes.
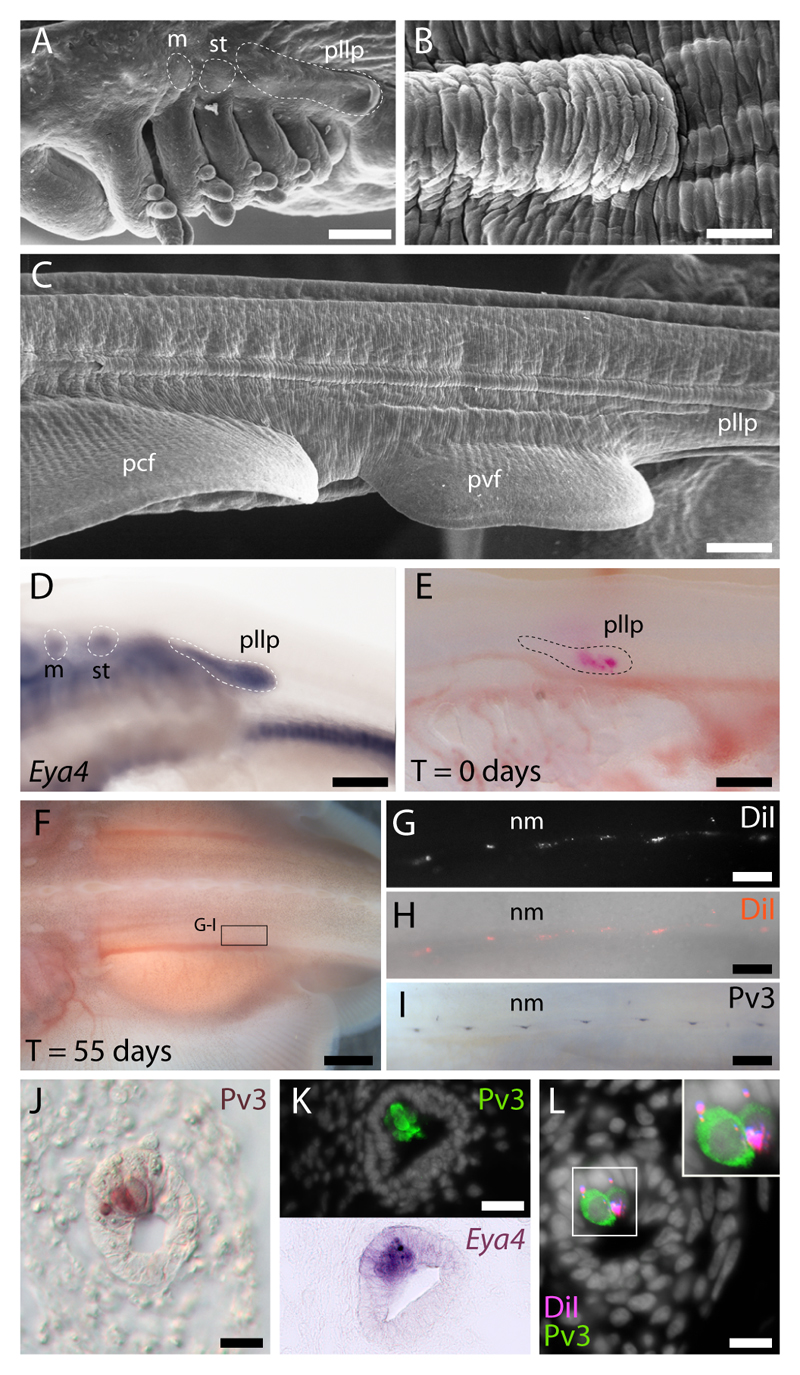
Fig. 3. Lateral line placodal origin of both sensory receptor cells and support cells in skate trunk neuromasts.
(A) The skate posterior lateral line placode is recognizable as an elongated ectodermal thickening dorsal to the pharyngeal arches. (B) This placode elongates caudally along the entire length of the trunk, forming (C) a distinct ridge in the epidermis. (D) The posterior lateral line placode expresses the transcription co-factor gene Eya4. (E) L. erinacea embryos in which the posterior lateral line placode was labelled with DiI show (F-H) DiI-positive cells organized in a pattern that recapitulates the normal distribution of (I) parvalbumin-3 (Pv3)-positive trunk mechanosensory neuromasts. (J) Immunohistochemical localization of Pv3 in trunk neuromasts reveals small clusters of sensory hair cells nested among non-sensory support cells; (K) these sensory hair cells also express Eya4. (L) The presence of DiI in trunk neuromast Pv3-positive sensory hair cells as well as in non-sensory support cells confirms the posterior lateral line placodal origin of these cell types in L. erinacea. m, middle lateral line placode; pcf, pectoral fin; pvf, pelvic fin; st, supratemporal lateral line placode; nm, neuromast; pllp, posterior lateral line placode. Scale bars: A,C-E: 250 µm; B: 100 µm; F: 5 mm; G-I: 500 µm; J-L: 10 µm.
Overall, our in vivo fate-mapping data show that lateral line placodes form both ampullary organs and neuromasts, including the sensory receptor cells within them, in cartilaginous fishes. The proposal that shark electroreceptors are neural crest-derived (Freitas et al., 2006) was based on the expression of Sox8 and HNK1 (neither of which exclusively marks neural crest cells) in developing ampullary organs, and not on lineage-tracing data. Taken together with the previous demonstration of a lateral line placode origin for ampullary organs in both lobe-finned and non-teleost ray-finned bony fishes (Modrell et al., 2011a; Northcutt et al., 1995), our data confirm that ampullary organs in jawed vertebrates are primitively derived from lateral line placodes.
The lateral line placode-derived ampullary organs of jawed vertebrates likely represent the elaboration of an electrosensory system already present in the last common ancestor of vertebrates. Lampreys (jawless fishes) possess epidermal “end bud” electroreceptors (Bodznick and Northcutt, 1981; Jørgensen, 2005). Although not housed in ampullary organs, lamprey electroreceptors share key features with the ampullary electroreceptors of non-teleost jawed fishes: they respond to cathodal stimuli (Bodznick and Preston, 1983) and are innervated by the anterior lateral line nerve, which projects to an electroreceptive dorsal octavolateral nucleus in the medulla (Bodznick and Northcutt, 1981; Bodznick and Preston, 1983; Ronan and Bodznick, 1986). Taken together, these shared characters suggest homology with non-teleost ampullary organ electroreceptors. However, the embryonic origin of lamprey electroreceptors is currently unknown.
Within jawed vertebrates, electroreception has been lost independently at least three times (in the lineages leading to anuran amphibians, amniotes and neopterygian fishes - i.e., gars, bowfins and teleosts) (Bullock et al., 1983; New, 1997; Northcutt, 1997; Schlosser, 2002a) and has also evolved at least twice independently within the teleosts (in siluriforms and gymnotiforms, and in mormyriforms) (Alves-Gomes, 2001; Bullock et al., 1983; New, 1997; Northcutt, 1997) and convergently as a specialization of trigeminal nerve endings in monotremes (Pettigrew, 1999) and dolphins (Czech-Damal et al., 2011). Teleost electroreceptors are innervated by lateral line nerves projecting to a special ‘electrosensory lateral line lobe’ in the hindbrain (Bodznick and Montgomery, 2005), but in contrast to non-teleost electroreceptors (Lu and Fishman, 1995), teleost electroreceptors are excited by anodal stimuli, and the basal membrane is the voltage sensor (Bodznick and Montgomery, 2005). Since neurotransmitter release is triggered in mechanosensory hair cells by opening voltage-gated channels in the basal membrane, electroreceptors may have evolved in some teleosts via genetic modification of the mechanisms underlying neuromast hair cell differentiation (Bodznick and Montgomery, 2005), but this hypothesis remains untested. While the molecular mechanisms underlying mechanosensory hair cell formation have been intensively studied (Driver and Kelley, 2009; Puligilla and Kelley, 2009), few papers have reported gene expression during ampullary organ development (Freitas et al., 2006; Metscher et al., 1997; Modrell and Baker, 2012; Modrell et al., 2011a; Modrell et al., 2011b; O'Neill et al., 2007). Discovering the extent to which the mechanisms of mechanosensory and electrosensory receptor cell formation are conserved – both for ancestral ampullary organ electroreceptors and for independently evolved teleost electroreceptors – may shed further light on the fascinating evolutionary history of this ancient vertebrate sense.
Acknowledgments
We thank Mark Terasaki for assistance with skate embryo husbandry, Kim Cooper for assistance with skate embryo storage and shipping, and the staff of the Woods Hole Marine Biological Laboratory for technical support. This work was funded by a Royal Society Newton International Fellowship (to J.A.G.), Marine Biological Laboratory Spiegel and Colwin Endowed Summer Research Fellowships (to J.A.G.) and the BBSRC (BB/F00818X/1 to C.V.H.B.).
References
- Alves-Gomes JA. The evolution of electroreception and bioelectrogenesis in teleost fish: a phylogenetic perspective. J Fish Biol. 2001;58:1489–1511. [Google Scholar]
- Aman A, Piotrowski T. Cell-cell signaling interactions coordinate multiple cell behaviors that drive morphogenesis of the lateral line. Cell Adh Migr. 2011;5:499–508. doi: 10.4161/cam.5.6.19113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker CVH, O'Neill P, McCole RB. Lateral line, otic and epibranchial placodes: developmental and evolutionary links? J Exp Zool B Mol Dev Evol. 2008;310B:370–383. doi: 10.1002/jez.b.21188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodznick D, Northcutt RG. Electroreception in lampreys: evidence that the earliest vertebrates were electroreceptive. Science. 1981;212:465–467. doi: 10.1126/science.7209544. [DOI] [PubMed] [Google Scholar]
- Bodznick D, Montgomery JC. The physiology of low-frequency electrosensory systems. In: Bullock TH, Hopkins CD, Popper AN, Fay RR, editors. Electroreception. New York: Springer; 2005. pp. 132–153. [Google Scholar]
- Bodznick D, Preston DG. Physiological characterization of electroreceptors in the lampreys Ichthyomyzon unicuspis and Petromyzon marinus. J Comp Physiol A. 1983;152:209–217. [Google Scholar]
- Bullock TH, Bodznick DA, Northcutt RG. The phylogenetic distribution of electroreception: evidence for convergent evolution of a primitive vertebrate sense modality. Brain Res. 1983;287:25–46. doi: 10.1016/0165-0173(83)90003-6. [DOI] [PubMed] [Google Scholar]
- Czech-Damal NU, Liebschner A, Miersch L, Klauer G, Hanke FD, Marshall C, Dehnhardt G, Hanke W. Electroreception in the Guiana dolphin (Sotalia guianensis) Proc R Soc B. 2011;279:663–668. doi: 10.1098/rspb.2011.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driver EC, Kelley MW. Specification of cell fate in the mammalian cochlea. Birth Defects Res C Embryo Today. 2009;87:212–221. doi: 10.1002/bdrc.20154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freitas R, Zhang G, Albert JS, Evans DH, Cohn MJ. Developmental origin of shark electrosensory organs. Evol Dev. 2006;8:74–80. doi: 10.1111/j.1525-142X.2006.05076.x. [DOI] [PubMed] [Google Scholar]
- Ghysen A, Dambly-Chaudière C. The lateral line microcosmos. Genes Dev. 2007;21:2118–2130. doi: 10.1101/gad.1568407. [DOI] [PubMed] [Google Scholar]
- Harrison RG. Experimentelle Untersuchung über die Entwicklung der Sinnesorgane der Seitenlinie bei den Amphibien. Arch Mikrosk Anat EntwMech. 1904;63:35–149. [Google Scholar]
- Heller S, Bell AM, Denis CS, Choe Y, Hudspeth AJ. Parvalbumin 3 is an abundant Ca2+ buffer in hair cells. J Assoc Res Otolaryngol. 2002;3:488–498. doi: 10.1007/s10162-002-2050-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jørgensen JM. Morphology of electroreceptive sensory organs. In: Bullock TH, Hopkins CD, Popper AN, Fay RR, editors. Electroreception. New York: Springer; 2005. pp. 47–67. [Google Scholar]
- Lu J, Fishman HM. Ion channels and transporters in the electroreceptive ampullary epithelium from skates. Biophys J. 1995;69:2467–2475. doi: 10.1016/S0006-3495(95)80117-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma EY, Raible DW. Signaling pathways regulating zebrafish lateral line development. Curr Biol. 2009;19:R381–6. doi: 10.1016/j.cub.2009.03.057. [DOI] [PubMed] [Google Scholar]
- Maxwell EE, Fröbisch NB, Heppleston AC. Variability and conservation in late chondrichthyan development: ontogeny of the winter skate (Leucoraja ocellata) Anat Rec (Hoboken) 2008;291:1079–1087. doi: 10.1002/ar.20719. [DOI] [PubMed] [Google Scholar]
- Metscher BD, Northcutt RG, Gardiner DM, Bryant SV. Homeobox genes in axolotl lateral line placodes and neuromasts. Dev Genes Evol. 1997;207:287–295. doi: 10.1007/s004270050116. [DOI] [PubMed] [Google Scholar]
- Modrell MS, Baker CVH. Evolution of electrosensory ampullary organs: conservation of Eya4 expression during lateral line development in jawed vertebrates. Evol Dev. 2012;14:277–285. doi: 10.1111/j.1525-142X.2012.00544.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modrell MS, Bemis WE, Northcutt RG, Davis MC, Baker CVH. Electrosensory ampullary organs are derived from lateral line placodes in bony fishes. Nat Commun. 2011a;2:496. doi: 10.1038/ncomms1502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modrell MS, Buckley D, Baker CVH. Molecular analysis of neurogenic placode development in a basal ray-finned fish. Genesis. 2011b;49:278–294. doi: 10.1002/dvg.20707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Münz H, Claas B, Fritzsch B. Electroreceptive and mechanoreceptive units in the lateral line of the axolotl Ambystoma mexicanum. J Comp Physiol A. 1984;154:33–44. [Google Scholar]
- New JG. The evolution of vertebrate electrosensory systems. Brain Behav Evol. 1997;50:244–252. doi: 10.1159/000113338. [DOI] [PubMed] [Google Scholar]
- Northcutt RG. Evolution of gnathostome lateral line ontogenies. Brain Behav Evol. 1997;50:25–37. doi: 10.1159/000113319. [DOI] [PubMed] [Google Scholar]
- Northcutt RG. Ontogeny of electroreceptors and their neural circuitry. In: Bullock TH, Hopkins CD, Popper AN, Fay RR, editors. Electroreception. New York: Springer; 2005. pp. 112–131. [Google Scholar]
- Northcutt RG, Brändle K, Fritzsch B. Electroreceptors and mechanosensory lateral line organs arise from single placodes in axolotls. Dev Biol. 1995;168:358–373. doi: 10.1006/dbio.1995.1086. [DOI] [PubMed] [Google Scholar]
- O'Neill P, McCole RB, Baker CVH. A molecular analysis of neurogenic placode and cranial sensory ganglion development in the shark, Scyliorhinus canicula. Dev Biol. 2007;304:156–181. doi: 10.1016/j.ydbio.2006.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettigrew JD. Electroreception in monotremes. J Exp Biol. 1999;202:1447–1454. doi: 10.1242/jeb.202.10.1447. [DOI] [PubMed] [Google Scholar]
- Puligilla C, Kelley MW. Building the world's best hearing aid; regulation of cell fate in the cochlea. Curr Opin Genet Dev. 2009;19:368–373. doi: 10.1016/j.gde.2009.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronan MC, Bodznick D. End buds: non-ampullary electroreceptors in adult lampreys. J Comp Physiol A. 1986;158:9–15. doi: 10.1007/BF00614515. [DOI] [PubMed] [Google Scholar]
- Sarrazin AF, Nuñez VA, Sapède D, Tassin V, Dambly-Chaudière C, Ghysen A. Origin and early development of the posterior lateral line system of zebrafish. J Neurosci. 2010;30:8234–8244. doi: 10.1523/JNEUROSCI.5137-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlosser G. Development and evolution of lateral line placodes in amphibians. II. Evolutionary diversification. Zoology (Jena) 2002a;105:177–193. doi: 10.1078/0944-2006-00062. [DOI] [PubMed] [Google Scholar]
- Schlosser G. Development and evolution of lateral line placodes in amphibians I. Development. Zoology (Jena) 2002b;105:119–146. doi: 10.1078/0944-2006-00058. [DOI] [PubMed] [Google Scholar]
- Schlosser G. Induction and specification of cranial placodes. Dev Biol. 2006;294:303–351. doi: 10.1016/j.ydbio.2006.03.009. [DOI] [PubMed] [Google Scholar]
- Teeter JH, Szamier RB, Bennett MVL. Ampullary electroreceptors in the sturgeon Scaphirhynchus platorynchus (Rafinesque) J Comp Physiol A. 1980;138:213–223. [Google Scholar]