Abstract
Studies investigating the association of food and nutrient consumption with the risk of urothelial cell carcinoma (UCC) have produced mixed results. We used three common dietary scores, the Mediterranean Diet Score (MDS), the Alternate Healthy Eating Index 2010 (AHEI-2010), and the Dietary Inflammatory Index (DII) to assess the evidence of an association between diet and the risk of UCC. Over a median follow-up time of 21.3 years, 379 incident UCC cases were diagnosed. Dietary scores were calculated using data from a 121-item food frequency questionnaire administered at baseline. We used Cox models to compute hazard ratios (HR) for the association between dietary scores (per one standard deviation) and UCC risk. In order to reflect overall adherence to a healthy diet, a metascore was constructed by summing the quintiles of each of the three scores. None of the dietary scores was associated with the risk of UCC overall. A healthier diet was found to be inversely associated with the risk of invasive (MDS: HR=0.86, 95% CI: 0.74–1.00, metascore: HR=0.84, 95% CI: 0.71–0.98), but not superficial disease (heterogeneity between subtypes p=0.04 and p=0.03, respectively). Results were consistent but weaker for DII and AHEI-2010. We found some evidence of effect modification by smoking, in particular for the metascore (Current: HR=0.77, 95% CI: 0.58–1.01, Former: HR=0.77, 95% CI: 0.64–0.92, Never: HR=1.01, 95% CI: 0.81–1.26, p for heterogeneity=0.05). A healthy diet may be protective against the risk of invasive, but not superficial, UCC. Promoting healthy dietary habits may help lower the risk of invasive UCC, especially for current and former smokers.
Keywords: dietary score, urothelial cell, bladder cancer, Mediterranean Diet Score, Dietary Inflammatory Index, Alternate Healthy Eating Index
INTRODUCTION
Urothelial cell carcinoma (UCC) includes tumours of the transitional epithelium of the renal pelvis, the ureter, proximal urethra and, predominantly, the urinary bladder. UCC is the fourth most commonly diagnosed cancer in males in developed countries1. More than half of cases are superficial but recurrence frequently occurs, which constitutes a substantial healthcare burden2. Bladder cancer patients experience low survival rates, especially women, for whom the 5-year relative survival in 2009–2013 in Australia was 45%, compared with 56% for men3. Well-established risk factors for UCC include smoking, sex (3 to 4 times more frequent in males), and occupational and environmental exposures to pollutants4. Genetic variants associated with bladder cancer risk have been discovered4 and some appear to interact with environmental factors, for example GSTM1, GSTT1 and NAT2 polymorphisms with polycyclic aromatic hydrocarbons and aromatic amines5.
Braver’s urogenous contact theory6 provides a biologically plausible rationale proposing that the urothelium is exposed to dietary factors in the urine, which may have an influence on urothelial cell carcinogenesis. However, after suggesting a probable chemopreventive effect of fruit and vegetables in their first report (1997), the World Cancer Research Fund/American Cancer Research Fund concluded in their most recent report that “the evidence was too limited to conclude that any aspect of food, nutrition and physical activity directly modifies the risk of bladder cancer”7.
Studies that have evaluated the association between diet and the risk of UCC have mostly focused on specific vitamins or nutrients, and produced mixed and inconclusive results. Hypotheses tested thus far include the protective effects of certain vitamins8, 9, intake of vegetables and fruit10, 11 or their specific compounds such as flavonoids12, and specific food types such as milk13 and eggs14. Other hypothesised risk factors include the intake or total fluids15, or of coffee and tea16. The hypothesis that cancer is caused by single nutrients is difficult to test, in particular because the intakes of specific nutrients are correlated with each other and can be combined in numerous ways. Hence, dietary scores reflecting entire dietary patterns may provide greater insight into the effect of a wide range of dietary exposures, and offer an attractive alternative to study complex associations between diet and diseases such as UCC17.
In this study, we sought to examine whether specific patterns of diet, as defined by dietary scores, were associated with the risk of UCC. We focused on three commonly used dietary scores: the Mediterranean Diet Score (MDS), the Alternate Healthy Eating Index 2010 (AHEI-2010), and the recently developed Dietary Inflammatory Index (DII). These dietary scores are based on different theoretical approaches to defining a healthy diet and each has been found to be associated with various health outcomes18–20.
MATERIAL AND METHODS
Study participants
The Melbourne Collaborative Cohort Study (MCCS) is a prospective cohort study comprising 17,045 men and 24,469 women, aged 27 to 76 years (99% aged 40–69 years) at the time of recruitment (1990–1994). Study participants were recruited from the metropolitan area of Melbourne, Australia. People of Southern European descent (Italy and Greece) were oversampled to extend the range of lifestyle exposures, including dietary habits. More details about the cohort can be found elsewhere21. The protocol for this study was approved by the Cancer Council Victoria’s Human Research Ethics Committee.
Dietary score definitions
At baseline, participants were administered a validated 121-item food frequency questionnaire (FFQ) developed for the MCCS22. A summary of the components of the dietary scores is presented in the Supplementary Table. The three dietary scores we examined are based on different theoretical approaches: the MDS represents the dietary pattern consumed by populations bordering the Mediterranean Sea, for whom a high life expectancy and particularly low rates of cardiovascular disease, certain cancers, and other diet-related chronic diseases were observed; the AHEI-2010 is the up-to-date version of the Alternate Healthy Eating Index, which was developed to summarise an individual’s overall intake of foods and nutrients found to be associated with the risk of chronic disease in the medical literature; the DII, based on the evidence that chronic inflammation appears to be associated with the risk of several chronic diseases, aims to summarise the overall inflammatory potential of an individual’s diet, using a comprehensive literature search of inflammation-associated foods and nutrients.
Mediterranean Diet Score
The MDS was calculated using the 9-item score developed by Trichopoulou23. For vegetables, fruits, cereals, legumes, and fish, participants were allocated 1 point if their intake was above the sex-specific median. For dairy and red meat, participants were allocated 1 point if their intake was below the median. For alcohol, men with an intake between 10 and 50g per day and women with an intake between 5 and 25g per day were allocated 1 point. We replaced the ratio of dietary monounsaturated (MUFA) to saturated (SFA) fats used by Trichopoulou with the intake of olive oil, as olive oil was found to correlate more closely than MUFA with the MDS24. The MDS potentially ranges from 0 to 9, a higher value representing a more Mediterranean diet.
Alternate Healthy Eating Index-2010
The AHEI-2010 is based on 11 components25, each given a score between 0 (less healthy diet) and 10 (healthier diet), with intermediate values scored proportionally to their intake. Intakes of vegetables, fruit, whole grains, nuts and legumes, long chain omega-3 fats, and polyunsaturated fatty acids contribute to the score positively; intakes of sugar sweetened drinks and fruit juice, red and processed meat, trans fat, and sodium contribute negatively, and alcohol intake is considered to be part of healthier diet if consumed in moderation. The AHEI-2010 score potentially ranges from 0 to 110, a higher value representing healthier eating habits.
Dietary Inflammatory Index
To calculate the DII for the participants in this study, the dietary data were first linked to a worldwide database that provided robust estimates of a mean and standard deviation for each food parameter included in the DII. These parameters then became the multipliers to express an individual’s exposure relative to the “standard global mean” as a z-score. To minimize the effect of right skewing, this value was then converted to a centred percentile score. The centred percentile score for each food parameter for each participant was then multiplied by the corresponding food parameter effect score in order to obtain a food parameter-specific DII score. All parameter-specific DII scores were then summed to create the overall DII score. In the MCCS, DII scores were calculated using 29 foods and nutrients, out of the 45 possible items26, for which the intake data were available: energy, carbohydrate, protein, fat, alcohol, fibre, cholesterol, saturated fatty acid, mono-unsaturated fatty acid, poly-unsaturated fatty acid, omega 3 fat, omega 6 fat, trans fat, niacin, thiamin, riboflavin, vitamin B12, vitamin B6, iron, magnesium, zinc, vitamin A, vitamin C, vitamin E, folic acid, beta carotene, garlic, onion, and tea. DII scores ranging from −8.9 to 8.0 were estimated from simulated diets based on international intake data26. A higher value represents a more pro-inflammatory diet.
Overall adherence to a healthy diet
Overall adherence to a healthy diet was summarised by creating a metascore. The metascore was constructed from the 3 dietary scores by summing the quintiles of each score (with the DII scale inverted so a higher score reflected a healthier diet). The metascore thus potentially ranged from 3 to 15, a higher value representing overall good adherence to a healthy diet.
Exclusion criteria
To avoid misclassification of dietary habits, we excluded from the analysis individuals with heart disease, angina, and diabetes as they may have changed their diet in response to their diagnoses (N=6,121). Individuals in the 1st and 99th percentile of the energy intake distribution were also excluded as they may represent aberrant reporting of food habits (N=951), i.e. males with energy intakes (in kilocalories) of <995 or >5,514 kcal, and females with energy intakes of <777 or >4,568 kcal.
Case ascertainment
All incident UCC cases diagnosed between baseline (1990–1994) and 31 December 2012 were identified from the Victorian Cancer Registry and the Australian Cancer Database, which contain virtually complete data on cancers diagnosed in MCCS participants. We used the following ICD-O-3 morphology codes: 8120, 8122, 8130 and 8131. Disease subtypes were defined according to behaviour. Invasive UCC included any tumour that had penetrated or invaded the basement membrane. Superficial UCC included papillary transitional cell neoplasm of low malignant potential or carcinoma in situ (CIS) that was completely confined within the epithelium. For individuals who were diagnosed with both superficial and invasive UCC, only the first diagnosis was considered. Individuals who received both diagnoses on the same day were considered to be diagnosed with invasive disease (N=2 cases). Tumours of uncertain diagnosis (not histologically confirmed) were censored at the time of diagnosis, as were those with borderline behaviour, and those with a vaginal histology code (ICD-4: C529).
Statistical analysis
Correlations between dietary scores were assessed using Spearman correlations and represented graphically using box plots. Dietary scores were used as continuous variables or divided into quintiles. Because the MDS and the AHEI-2010 are pseudo-continuous, we could only split them into groups of similar size, named quintiles hereafter. We used Cox regression models to estimate hazard ratios (HRs) for the association between dietary scores and the risk of UCC. Age was used as the timescale to adjust for age effects27. For all dietary scores, the proportional hazards assumption was assessed by visual inspection of Schoenfeld residuals28. Other potential confounders included in the models were: sex; country of birth (Australia, New Zealand, UK, Southern Europe, Northern Europe); smoking (never, former: quit ≤15 years prior, former: quit >15 years prior, current ≤20 cigarettes per day, current >20 cigarettes per day); alcohol drinking (defined by sex-specific Australian NHMRC recommendation thresholds: never, 1–39 g/d (males) and 1–19 g/d (females), 40–59 g/d (males) and 20–39 g/d (females), 60+ g/d (males) and 40+ g/d (females)), body mass index (BMI) (<25 kg/m2, 25–30 kg/m2, >30 kg/m2); physical activity (continuous score developed by MacInnis et al.29); education (primary school only, some high/technical school, completed high/technical school, degree or diploma); socioeconomic status (defined by quintiles of relative socioeconomic disadvantage of area of residence30) and reported intake of nonsteroidal anti-inflammatory drugs (NSAIDS) (none, aspirin only, other NSAID only, both aspirin and other NSAID). All HRs for dietary scores are given per one standard deviation increase of the variable.
All models were fitted separately for overall UCC and for invasive and superficial disease. Person-years of follow-up were calculated from baseline until the date of diagnosis, censoring at death or end of follow-up (31 December 2012). Censoring was also done at diagnosis of other UCC subtype (invasive or superficial) in the competing risk model31. Potential interactions of dietary scores with smoking, alcohol and sex were examined by adding interaction terms and applying a likelihood ratio test. Tests for heterogeneity in the HRs between invasive and superficial tumours were performed using the data duplication method32.
RESULTS
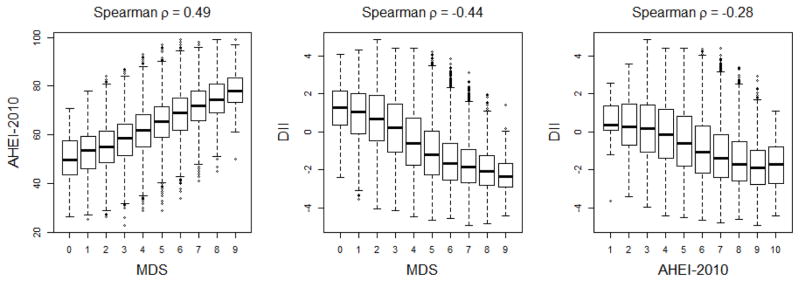
During follow-up, 379 individuals were diagnosed with UCC, including 165 invasive and 214 superficial cases. The median follow-up time was 21.3 years, interquartile range (IQR): 6.1–22.4 (in cases only: 13.6 years, IQR: 8.2–17.5), and the median age at cancer diagnosis was 73.8 years, IQR: 66.6–78.0. UCC cases were more likely to be male, to smoke, be migrants from Southern Europe, drink alcohol and be less highly educated; other covariates had little association with UCC risk (Table 1). These associations did not appear to differ substantially by UCC subtype (data not shown). A moderate positive rank correlation was found between the MDS and the AHEI-2010 (Spearman ρ=0.49), and moderate negative rank correlations were found for the DII with each of the MDS and the AHEI-2010 (ρ=−0.44, and ρ=−0.28, respectively) (Figure 1). For all dietary scores, no evidence of departure from the proportional hazard assumption was observed for UCC risk overall or by subtype.
Table 1.
Characteristics of the study participants, Melbourne Collaborative Cohort Study (MCCS), 1990–2012
| Cases (N=379) | Non-cases (37,063) | Pa | ||
|---|---|---|---|---|
| Age at baseline | Median (IQR) | 61.5 [55.5–65.8] | 54.4 [46.9–61.9] | <0.001 |
|
| ||||
| Sex | Male (%) | 290 (76%) | 13,420 (39%) | <0.001 |
|
| ||||
| Country of birth | Australia, NZ, UK | 218 (58%) | 23,390 (69%) | 0.001 |
| Northern Europe | 31 (8%) | 2,231 (7%) | ||
| Southern Europe | 130 (34%) | 8442 (25%) | ||
|
| ||||
| Education | Primary school | 109 (29%) | 6351 (19%) | 0.01 |
| Some high/technical school | 125 (33%) | 12908 (38%) | ||
| Completed high/technical school | 85 (23%) | 6991 (20%) | ||
| Degree/diploma | 60 (16%) | 7813 (23%) | ||
|
| ||||
| SEIFA | Q1 | 57 (15%) | 4766 (14%) | 0.84 |
| Q2 | 89 (24%) | 6928 (20%) | ||
| Q3 | 75 (20%) | 6266 (18%) | ||
| Q4 | 68 (18%) | 7109 (21%) | ||
| Q5 | 90 (23%) | 8994 (26%) | ||
|
| ||||
| Smoking | Never | 116 (31%) | 20156 (59%) | <0.001 |
| Former, quit ≥15 years | 98 (26%) | 4707 (14%) | ||
| Former, quit <15 years | 94 (24%) | 5416 (16%) | ||
| Current <20 cigs/day | 29 (8%) | 1784 (5%) | ||
| Current ≥20 cigs/day | 42 (11%) | 2000 (6%) | ||
|
| ||||
| Alcohol consumption | 0 | 91 (24%) | 10797 (32%) | <0.001 |
| 1–39 g/d (M), 1–19 g/d (F) | 219 (58%) | 18767 (55%) | ||
| 40–59 g/d (M), 20–39 g/d (F) | 41 (11%) | 3015 (9%) | ||
| 60+ g/d (M), 40+ g/d (F) | 28 (7%) | 1480 (4%) | ||
|
| ||||
| NSAIDs | None | 285 (81%) | 26803 (79%) | 0.85 |
| Aspirin only | 47 (11%) | 4084 (12%) | ||
| Other NSAID only | 40 (7%) | 2592 (8%) | ||
| Both aspirin and other NSAID | 7 (2%) | 584 (2%) | ||
|
| ||||
| Physical activity score | Median (IQR) | 4.0 [0.0–5.5] | 4.0 [1.5–5.5] | 0.19 |
|
| ||||
| BMI | Median (IQR) | 26.9 [24.6–29.8] | 26.2 [23.7–29.1] | 0.10 |
|
| ||||
| AHEI-2010 | Median (IQR) | 62.5[54.5–70.5] | 64.5 [57.0–72.0] | 0.001 |
|
| ||||
| MDS | Median (IQR) | 5 [4–6] | 5 [4–6] | 0.91 |
|
| ||||
| DII | Median (IQR) | −0.84 [−2.05–0.61] | −0.98[−2.14–0.40] | 0.04 |
|
| ||||
| Tumour subtype | Invasive | 165 (44%) | ||
| Superficial | 214 (56%) | |||
| Age at diagnosis | Median (IQR) | 73.8 [66.6–78.0] | ||
IQR = interquartile range, SEIFA: Socio-Economic Indexes for Areas, NSAIDs: Nonsteroidal anti-inflammatory drugs, BMI: body-mass index (in kg/m2), AHEI-2010: Alternate Healthy Eating Index 2010, MDS: Mediterranean Diet Score, DII: Dietary inflammatory Index
p-values from likelihood ratio tests comparing age-adjusted Cox models with and without the variable (except for the variable age, for which a Wilcoxon signed-rank test was used)
Figure 1. Correlation between Mediterranean Diet Score (MDS), Alternate Health Eating Index-2010 (AHEI-2010), and Dietary Inflammatory Index (DII), Melbourne Collaborative Cohort Study (MCCS), 1990–2012.
Boxplots and Spearman correlations between dietary scores. The AHEI-2010 was divided into deciles for graphical representation.
We found no evidence of association between dietary scores and the risk of UCC overall: MDS: HR (per 1 SD)=0.97, 95% CI: 0.88–1.08; AHEI-2010: HR=1.03, 95% CI: 0.92–1.15; DII: HR=1.06, 95% CI: 0.96–1.18; metascore: HR=0.96, 95% CI: 0.86–1.06 (not shown), and no association of dietary scores with superficial UCC for the MDS: HR=1.06, 95% CI:0.92–1.21, the DII: HR=1.05, 95% CI: 0.92–1.20, or the metascore: HR=1.05, 95% CI: 0.91–1.20 (Table 2). The risk of superficial UCC was, on the contrary, increased for individuals with better adherence to the AHEI-2010 (HR=1.17, 95% CI: 1.01–1.34).
Table 2.
HR estimates and 95% CI for the association between dietary scores and the risk of UCC, by subtype, Melbourne Collaborative Cohort Study (MCCS), 1990–2012
| All cases | Superficial | Invasive | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||||
| N cases | HRa | 95% CI | p | N cases | HRa | 95% CI | p | N cases | HRa | 95% CI | p | phetb | ||
| MDS | Q1 (unhealthy) | 83 | 1 (ref) | - | - | 42 | 1 (ref) | - | - | 41 | 1 (ref) | - | - | |
| Q2 | 81 | 1.04 | 0.76 1.41 | 0.82 | 44 | 1.08 | 0.73–1.71 | 0.61 | 37 | 0.95 | 0.61–1.49 | 0.83 | ||
| Q3 | 82 | 0.87 | 0.64 1.19 | 0.39 | 47 | 0.99 | 0.65–1.51 | 0.98 | 35 | 0.75 | 0.47–1.18 | 0.21 | ||
| Q4 | 73 | 0.92 | 0.66 1.27 | 0.61 | 44 | 1.10 | 0.72–1.70 | 0.64 | 29 | 0.73 | 0.45–1.17 | 0.20 | ||
| Q5 (healthy) | 60 | 0.89 | 0.62 1.26 | 0.50 | 37 | 1.10 | 0.69–1.73 | 0.69 | 23 | 0.67 | 0.46–1.14 | 0.14 | ||
|
| ||||||||||||||
| Continuous, per 1 SD | 0.97 | 0.87 1.07 | 0.52 | 1.06 | 0.92–1.21 | 0.44 | 0.86 | 0.74–1.00 | 0.06 | 0.04 | ||||
|
| ||||||||||||||
| DII | Q1 (anti-inflammatory) | 74 | 1 (ref) | - | - | 41 | 1 (ref) | - | - | 33 | 1 (ref) | - | - | |
| Q2 | 71 | 1.00 | 0.72 1.39 | 0.98 | 39 | 0.97 | 0.52–1.21 | 0.91 | 32 | 1.04 | 0.64–1.69 | 0.87 | ||
| Q3 | 69 | 0.96 | 0.69 1.34 | 0.83 | 46 | 1.15 | 0.75–1.76 | 0.52 | 23 | 0.73 | 0.42–1.24 | 0.24 | ||
| Q4 | 72 | 0.98 | 0.71 1.37 | 0.92 | 42 | 1.01 | 0.65–1.56 | 0.98 | 30 | 0.94 | 0.57–1.55 | 0.80 | ||
| Q5 (pro-inflammatory) | 93 | 1.24 | 0.90 1.70 | 0.19 | 46 | 1.10 | 0.72–1.70 | 0.65 | 47 | 1.46 | 0.91–2.31 | 0.11 | ||
|
| ||||||||||||||
| Continuous, per 1 SD | 1.07 | 0.97 1.19 | 0.18 | 1.05 | 0.92–1.20 | 0.47 | 1.10 | 0.95–1.28 | 0.20 | 0.61 | ||||
|
| ||||||||||||||
| AHEI-2010 | Q1 (unhealthy) | 104 | 1 (ref) | - | - | 54 | 1 (ref) | - | - | 50 | 1 (ref) | - | - | |
| Q2 | 82 | 0.89 | 0.66 1.19 | 0.43 | 38 | 0.80 | 0.49–1.14 | 0.29 | 44 | 0.98 | 0.65–1.48 | 0.94 | ||
| Q3 | 66 | 0.93 | 0.68 1.29 | 0.68 | 40 | 1.10 | 0.72–1.67 | 0.65 | 26 | 0.76 | 0.47–1.22 | 0.26 | ||
| Q4 | 61 | 0.82 | 0.61 1.18 | 0.34 | 38 | 1.03 | 0.67–1.58 | 0.89 | 23 | 0.66 | 0.40–1.09 | 0.10 | ||
| Q5 (healthy) | 66 | 1.02 | 0.73 1.43 | 0.89 | 44 | 1.33 | 0.87–2.02 | 0.19 | 22 | 0.70 | 0.42–1.18 | 0.18 | ||
|
| ||||||||||||||
| Continuous, per 1 SD | 1.04 | 0.93 1.16 | 0.54 | 1.17 | 1.01–1.34 | 0.03 | 0.88 | 0.75–1.04 | 0.13 | 0.006 | ||||
|
| ||||||||||||||
| Metascore | Q1 (unhealthy) | 115 | 1 (ref) | - | - | 62 | 1 (ref) | - | - | 57 | 1 (ref) | - | - | |
| Q2 | 68 | 0.82 | 0.61 1.11 | 0.20 | 35 | 0.82 | 0.54–1.24 | 0.34 | 33 | 0.83 | 0.54–1.27 | 0.38 | ||
| Q3 | 81 | 0.96 | 0.72 1.28 | 0.79 | 45 | 1.03 | 0.70–1.52 | 0.88 | 36 | 0.88 | 0.58–1.35 | 0.56 | ||
| Q4 | 55 | 0.73 | 0.53 1.02 | 0.06 | 37 | 0.96 | 0.63–1.45 | 0.83 | 18 | 0.49 | 0.29–0.84 | 0.01 | ||
| Q5 (healthy) | 56 | 0.98 | 0.70 1.36 | 0.89 | 35 | 1.18 | 0.77–1.81 | 0.44 | 21 | 0.75 | 0.45–1.26 | 0.27 | ||
|
| ||||||||||||||
| Continuous, per 1 SD | 0.95 | 0.85 1.06 | 0.35 | 1.05 | 0.91–1.20 | 0.52 | 0.84 | 0.71–0.98 | 0.03 | 0.03 | ||||
MDS: Mediterranean diet score, DII: Dietary inflammatory index, AHEI-2010: Alternate Healthy Eating Index 2010
Models were adjusted for sex, country of birth, smoking, alcohol consumption, body mass index physical activity, education, and socioeconomic status
phet: p-value for heterogeneity of the association between dietary score and risk of UCC by subtype (i.e. invasive vs. superficial)
Regarding invasive UCC, inverse associations were found with MDS (HR=0.86, 95% CI: 0.74–1.00) and the metascore (HR=0.84, 95% CI: 0.71–0.98). The direction of the HR estimates were consistent, in terms of healthier eating being associated with protection, for a more pro-inflammatory diet (HR=1.10, 95% CI: 0.95–1.28) and the AHEI-2010 (HR=0.88, 95% CI: 0.75–1.04), (Table 2). For the MDS, the AHEI-2010, and the metascore, the association with the risk of UCC was different by tumour subtype: p for heterogeneity=0.04 for the MDS, p=0.006 for the AHEI-2010, and p=0.03 for the metascore (Table 2).
The association of dietary scores with risk of invasive UCC appeared to be stronger for current and former smokers. There was evidence of effect modification by smoking status for the metascore (current smokers: HR=0.77, 95% CI: 0.58–1.01; former smokers: HR=0.77, 95% CI: 0.64–0.92; never smokers: HR=1.01, 95% CI: 0.81–1.26, p interaction=0.05). For each dietary score taken individually, the association also appeared stronger for current and former smokers, although the evidence was weaker, p interaction=0.12, p=0.33, p=0.11 for the MDS, the DII, and the AHEI-2010, respectively. We found no evidence that the association between dietary scores and the risk of invasive UCC was modified by alcohol consumption or by sex, although a more pro-inflammatory diet appeared to be associated with an increased risk of invasive UCC in participants reporting low alcohol consumption (HR=1.26, 95% CI: 1.01–1.57) (Table 3). This was also the case for the metascore for which the association appeared to be highest in low and moderate drinkers (HR=0.77, 95% CI: 0.61–0.98 and HR=0.83, 95% CI: 0.70–1.00, respectively).
Table 3.
HR estimates (per 1 SD increase) and 95% CI of the risk of invasive UCC for dietary scores by smoking, alcohol, and sex, Melbourne Collaborative Cohort Study (MCCS), 1990–2012
| Smoking | Alcohol intake | Sex | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| ||||||||||||
| HR1 | 95% CI | Pint | HR | 95% CI | Pint | HR | 95% CI | Pint | ||||
| MDS | Never | 1.01 | 0.81–1.26 | 0.12 | Low | 0.82 | 0.64–1.05 | 0.86 | Male | 0.85 | 0.72–1.00 | 0.80 |
| Current | 0.82 | 0.63–1.05 | Medium | 0.88 | 0.74–1.05 | Female | 0.88 | 0.69–1.11 | ||||
| Former | 0.80 | 0.67–0.95 | High | 0.85 | 0.65–1.10 | |||||||
|
| ||||||||||||
| DII | Never | 0.98 | 0.79–1.22 | 0.33 | Low | 1.26 | 1.01–1.57 | 0.21 | Male | 1.09 | 0.93–1.28 | 0.70 |
| Current | 1.17 | 0.92–1.49 | Medium | 1.08 | 0.91–1.29 | Female | 1.14 | 0.91–1.45 | ||||
| Former | 1.16 | 0.96–1.38 | High | 0.98 | 0.75–1.26 | |||||||
|
| ||||||||||||
| AHEI-2010 | Never | 1.04 | 0.83–1.31 | 0.11 | Low | 0.85 | 0.67–1.08 | 0.88 | Male | 0.88 | 0.75–1.04 | 0.98 |
| Current | 0.81 | 0.62–1.05 | Medium | 0.88 | 0.73–1.06 | Female | 0.88 | 0.68–1.13 | ||||
| Former | 0.83 | 0.69–0.99 | High | 0.93 | 0.71–1.21 | |||||||
|
| ||||||||||||
| Metascore | Never | 1.01 | 0.81–1.26 | 0.05 | Low | 0.77 | 0.61–0.98 | 0.52 | Male | 0.83 | 0.71–0.98 | 0.96 |
| Current | 0.77 | 0.58–1.01 | Medium | 0.83 | 0.70–1.00 | Female | 0.84 | 0.66–1.07 | ||||
| Former | 0.77 | 0.64–0.92 | High | 0.94 | 0.71–1.24 | |||||||
Pint: p-value for interaction between dietary scores and relevant variables on the risk of invasive UCC.
DISCUSSION
Main findings
We observed inverse associations between dietary scores reflecting healthy eating and the risk of invasive UCC. Although no dietary score appeared to be a substantially better predictor of the risk of invasive UCC, the strongest association was obtained from computing a summary dietary score aiming to represent an overall healthy diet. The association we observed between overall adherence to a healthy diet and protection from invasive disease was stronger for current and former smokers than for never smokers.
Limitations
We could not adjust our results for environmental and occupational exposures to chemicals (e.g. polycyclic aromatic hydrocarbons and aromatic amines), which may introduce a bias if exposed workers in some industries had unhealthier dietary habits than other cohort members. Overall in Australia, workplace exposure to carcinogens is estimated to be responsible for 14% of bladder cancer in men and 0.7% in women33. As exposed individuals probably constituted a small proportion of our cohort, any resulting confounding effect from differential eating habits is likely to be small. Additionally, adjusting for socioeconomic status and educational level (although imperfect proxies for such exposures) did not meaningfully change the observed associations.
We were also unable to control for exposure to arsenic in drinking water, which has been shown to strongly increase the risk of UCC7, but the prevalence of such exposures in Melbourne, Australia is low according to the Australian Drinking Water Guidelines34. Fluid intake, in particular of water, may be associated with health awareness and thus with a healthier diet, but an accurate measure of this parameter could not be obtained from the MCCS FFQ. In any case, the association of fluid intake with the risk of UCC has been inconsistent, with the largest studies and a meta-analysis finding little or no association16.
Another potential limitation is that food frequency questionnaires measure dietary intakes with considerable error18. We evaluated diet only once, at baseline, and cannot exclude the possibility that people changed their diets over follow-up, which would lead to misclassification of long-term diet. Although using dietary scores does not overcome the weaknesses inherent in FFQs, they may be better able to distinguish between individuals than absolute amounts of specific foods or nutrients. Further, it is unlikely that an individual reports dietary intake erroneously for all items of a score. In this regard, dietary scores may be a more objective measure obtained from FFQs.
There also are limitations inherent to the construction of dietary scores. Regarding the DII, we could include only 29 items out of the 45 included in the original score. The missing food items were anthocyanin, eugenol, flavan-3-ols, flavones, flavonols, flavonones, isoflavones, caffeine, ginger, saffron, turmeric, vitamin D, pepper, thyme and oregano, rosemary and selenium. Several of these items are not consumed frequently in the Australian population. Others food items are more frequently consumed and therefore might have influenced the DII computed in our study; this may be especially true for flavonoids which are frequently consumed and have recently been associated with the risk of UCC12. Although we could not test and confirm this result in our cohort, other studies have observed relatively strong correlations between inflammatory biomarkers and the DII when it was computed with fewer than 30 food parameters35, 36. Because the items of the MDS are scored relative to the median intake, the values taken by the MDS depend on the overall levels of consumption of each item in the study sample, which can bias the score towards certain food items. In our study, more than 25% of participants were Italians and Greeks which may have contributed to relatively high observed consumption (for an Australian cohort) of the MDS items. Although our results may not be strictly comparable to those of other studies because of wide variations in food consumption usually found by country and cohort, it has been shown that the magnitude of the association of the MDS with other chronic diseases is fairly consistent across studies19, and only slightly sensitive to each of its constitutive items37.
An association between high AHEI-2010 and increased risk of superficial UCC was an unexpected finding from our analyses. We considered the possibility that individuals in this cohort who adhered more closely to the AHEI may have been more health-conscious and sought more prompt and regular medical checks leading to increased detection of early disease. The AHEI may better represent what people understood to be ‘healthy’ when the study was conducted, especially in relation to polyunsaturated fats. The DII on the other hand is based on less well known nutrients and food items so people would be unlikely to knowingly select for more anti-inflammatory scores. Other considerations include possible limitations of using the AHEI-2010 as a dietary assessment tool. For example, maximum scores are given by the AHEI-2010 for consumption of four or more serves of fruit per day which increases the sugar content of the diet, and is not consistent with the Australian dietary guidelines (ADG) which recommend only 2 servings of fruit per day38. While the AHEI-2010 does not distinguish between the different types of vegetables consumed, the WCRF and the ADG recommend limiting consumption of starchy vegetables. The AHEI-2010 also gives a higher score for a consumption of alcohol greater than that recommended in the ADG, particularly for women e.g. up to 1.5 standard drinks versus up to 1 standard drink per day, respectively. The ADG also recommends at least 2 alcohol-free days per week, which is not specified in the AHEI-2010. These limitations pertaining to the contribution of alcohol to dietary scores are also true for the MDS and the DII and so are unlikely to explain this unexpected result for the AHEI-2010 only.
Strengths
Diet was determined at baseline and the risk of UCC assessed prospectively, thus minimising the possibility of recall and selection biases. The prospective cohort study design is superior to the case-control design and provides more conservative estimates of the association between diet and cancer17. Our study also had lengthy and virtually complete follow-up.
Comparison with other studies
Only a handful of studies have examined the association between dietary scores and the risk of UCC. The EPIC cohort reported that the risk of bladder cancer was non-significantly decreased in participants with a higher MDS score39. Partly consistent with the finding from our study, an inverse association between MDS and bladder cancer was found in current, but not former or never smokers in the EPIC study (HR=0.66, 95% CI: 0.47–0.93 for high vs. low MDS for UCC overall, results for invasive disease not shown), although the evidence for interaction was relatively weak. A recent meta-analysis of the association between the MDS and cancer found only this study assessing the risk of bladder cancer40. Results from cohort studies will accumulate in the future, thus strengthening the evidence regarding the association between dietary scores and the risk of UCC, and potential effect modification by subtype. A Belgian case-control study that examined some items of the MDS concluded that there were possible associations between olive oil intake and decreased risk of bladder cancer, and between cheese consumption and increased risk41. To our knowledge, the recently created DII and the AHEI-2010 have never been assessed regarding their association with the risk of UCC, only with the risk of cancer overall. In the Nurse’s Health Study and the Health Professional’s Follow-up Study, the AHEI-2010 showed a relatively weak association with overall cancer risk25. In the NIH-AARP cohort the AHEI-2010, alternate Mediterranean Diet, the Healthy Eating Index-2005 and the DASH diet score, were all inversely associated with all-cause, cardiovascular and cancer mortality18. The DII has also been found to be associated with increased risk of all-cause, CVD, and cancer mortality in the NHANES III Follow-up Study based on a single 24-hour dietary recall42. In these studies, cancers of different sites were not analysed separately and UCC would have constituted only a small proportion of these.
Previous studies of specific foods or nutrients have found no strong associations with risk of UCC, as concluded in previous reviews4, 7. Three studies using the large EPIC cohort did not find any association for intake of fruit and vegetables10, diversity of fruit and vegetables11, or red meat and associated nitrosamines or haeme iron43. An alternative approach used by some authors has been to evaluate a score based on adherence to the WCRF/AICR recommendations for reducing cancer risk. These recommendations are specific to cancer and extend beyond diet to include weight management and physical activity44. In contrast to the dietary indices we have assessed, the WCRF/AICR recommendations also emphasises minimizing alcohol intake7. This is however unlikely to have strongly influenced the results of our study, as a recent meta-analysis found no evidence for an association between alcohol and UCC risk45.
In this study, we chose to focus on three widely used dietary scores17. Many other dietary scores have been proposed including the Dietary Diversity Score, the Dietary Approaches to Stop Hypertension Score, and the Recommended Food Score46–48. An alternative approach would be to evaluate specific national guidelines, for example, relevant to our setting, the Australian Dietary Guidelines38. Because the three dietary scores we studied were only moderately correlated with each other, we were able to pool them to create a summary measure of healthy eating. This metascore appeared to be more strongly associated with invasive UCC risk in our study. It should be noted that none of the aforementioned dietary scores, or those used in our study, were specifically designed for evaluating the risk of cancer. This may result in underestimation of the potential association between dietary patterns and risk of UCC.
Possible mechanisms
The MDS has been shown to be associated with reduced oxidative stress49, and reduced inflammation50, and the DII correlates with a range of inflammatory markers26, 35. The healthy eating index, although less correlated with the DII in our study, has also been associated with reduced chronic inflammation51. A recent systematic review concluded that inflammatory biomarkers are associated with poor UCC prognosis52 suggesting that a pro-inflammatory diet could accelerate the progression of tumours, and might explain why we observed trends of association with the risk of invasive disease only. Although we cannot rule out the possibility that this finding was due to an increased probability of type I error when conducting subgroup analyses, it also may be due to the heterogeneity of UCC, whose various subtypes are well recognized to be subject to divergent mechanistic pathways and clinical characteristics53.
A diet consisting of a large intake of plant and limited intake of animal products, along with consumption of healthy fats, as indicated by the MDS and other healthy dietary patterns, not only provides antioxidants, key nutrients for cell differentiation and carcinogen metabolism, but may also optimise the pH of the urine54. An acidic urinary pH potentially promotes carcinogenic activity in the bladder compared with a more alkaline urinary pH55. In addition, consumption of olive oil, which although predominantly a monounsaturated fat resistant to lipid peroxidation also contains tocopherols, carotenes and essential fatty acids, may help to maintain the integrity of urothelial cell membranes and protect against more invasive disease56. To our knowledge, studies evaluating associations between inflammatory biomarkers and the risk of UCC are scarce, particularly in terms of dietary exposures. A recent, albeit relatively small study concluded that anti-inflammatory cytokines measured in urine could be a useful biomarker of bladder cancer risk57.
Finally, there is some plausible biological basis for a stronger effect of the MDS in smokers. First, cigarette smoking lowers urinary pH levels58 and, among workers exposed to similar industrial carcinogens present in aromatic dyes, those with an acidic urine (pH <6.0) had 10-fold higher levels of exfoliated urothelial cell DNA adduct levels59. Higher levels of these carcinogens have been detected in smokers compared with non-smokers60. Second, the Mediterranean diet may help reduce the inflammatory and DNA damaging effects of smoking, which could in turn decrease the risk of cancer, as observed in other studies61. The fact that in our study, a similar risk reduction was observed in former smokers is perhaps more surprising but may be due to the long-term effect of these exposures, or latencies in the development and diagnosis of cancer.
Implications
Our findings of associations between different measures of healthy eating and reduced risk of invasive UCC are consistent with the current Australian Dietary Guidelines (ADG), which are overall similar to the AHEI-2010 and borrow items from the three scores we evaluated. The ADG promote consuming a variety of foods from the five food groups: vegetables, fruit, grains (mostly wholegrain), lean protein rich foods (including plant foods such as legumes, nuts and seeds, and low fat dairy foods). They suggest minimising intake of: foods rich in saturated fat (replacing them with poly- or monounsaturated fat); processed foods rich in sugar and salt, and alcoholic beverages. Our study adds to the evidence that a healthy diet might help reduce the risk of invasive urothelial cell carcinoma, for which survival is low. This may be particularly true for smokers.
CONCLUSION
We found evidence that healthy diets, as defined by commonly used dietary scores, were associated with reduced risk of invasive UCC. These associations seemed to be stronger in current and former smokers. Promoting healthy dietary habits might help lower the incidence of invasive UCC, especially in current and former smokers.
Supplementary Material
Novelty and impact.
Most studies on the association between diet and the risk of urothelial cell carcinoma (UCC) have focused on individual foods and nutrients and produced inconclusive evidence. In this paper, we used three common dietary scores to assess the evidence of an association between healthy eating and UCC risk. Our findings indicate that a healthy diet may be protective against invasive UCC. The associations we observed were particularly strong in current and past smokers.
Acknowledgments
FUNDING SOURCES
Drs. Shivappa and Hébert were supported by grant number R44DK103377 from the United States National Institute for Diabetes and Digestive and Kidney Diseases.
This study was made possible by the contribution of many people, including the original investigators and the diligent team who recruited the participants and who continue working on follow-up. We would also like to express our gratitude to the many thousands of Melbourne residents who took part in the study.
Cases and their vital status were ascertained through the Victorian Cancer Registry (VCR) and the Australian Institute of Health and Welfare (AIHW), including the National Death Index and the Australian Cancer Database.
Abbreviations
- UCC
urothelial cell carcinoma
- MDS
Mediterranean Diet Score
- AHEI
Alternate Healthy Eating Index
- DII
Dietary Inflammatory Index
- MCCS
Melbourne Collaborative Cohort Study
- WCRF
World Cancer Research Fund
- ADG
Australian Dietary Guidelines
Footnotes
CONFLICTS OF INTEREST AND FINANCIAL DISCLOSURES
PAD, AMH, MB, JKB, NS, JRH, JLH, RLM, DRE, GGG declare no potential conflicts of interests.
Disclosure: Dr. James R. Hébert owns controlling interest in Connecting Health Innovations LLC (CHI), a company planning to license the right to his invention of the dietary inflammatory index (DII) from the University of South Carolina in order to develop computer and smart phone applications for patient counselling and dietary intervention in clinical settings. Dr. Nitin Shivappa is an employee of CHI.
References
- 1.Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 2.Sievert KD, Amend B, Nagele U, Schilling D, Bedke J, Horstmann M, Hennenlotter J, Kruck S, Stenzl A. Economic aspects of bladder cancer: what are the benefits and costs? World J Urol. 2009;27:295–300. doi: 10.1007/s00345-009-0395-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Thursfield VFH. Cancer in Victoria: Statistics & Trends. 2014. Melbourne: Cancer Council Victoria; 2015. [Google Scholar]
- 4.Burger M, Catto JW, Dalbagni G, Grossman HB, Herr H, Karakiewicz P, Kassouf W, Kiemeney LA, La Vecchia C, Shariat S, Lotan Y. Epidemiology and risk factors of urothelial bladder cancer. Eur Urol. 2013;63:234–41. doi: 10.1016/j.eururo.2012.07.033. [DOI] [PubMed] [Google Scholar]
- 5.Hung RJ, Boffetta P, Brennan P, Malaveille C, Hautefeuille A, Donato F, Gelatti U, Spaliviero M, Placidi D, Carta A. GST, NAT, SULT1A1, CYP1B1 genetic polymorphisms, interactions with environmental exposures and bladder cancer risk in a high-risk population. Int J Cancer. 2004;110:598–604. doi: 10.1002/ijc.20157. [DOI] [PubMed] [Google Scholar]
- 6.Braver DJ, Modan M, Chetrit A, Lusky A, Braf Z. Drinking, micturition habits, and urine concentration as potential risk factors in urinary bladder cancer. J Natl Cancer Inst. 1987;78:437–40. [PubMed] [Google Scholar]
- 7.WCRF/AICR. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Research WCRFAIfC. 2007 [Google Scholar]
- 8.Brinkman M, Buntinx F, Muls E, Zeegers MP. Use of selenium in chemoprevention of bladder cancer. Lancet Oncol. 2006;7:766–74. doi: 10.1016/S1470-2045(06)70862-2. [DOI] [PubMed] [Google Scholar]
- 9.Chen F, Li Q, Yu Y, Yang W, Shi F, Qu Y. Association of vitamin C, vitamin D, vitamin E and risk of bladder cancer: a dose-response meta-analysis. Sci Rep. 2015;5:9599. doi: 10.1038/srep09599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Buchner FL, Bueno-de-Mesquita HB, Ros MM, Kampman E, Egevad L, Overvad K, Raaschou-Nielsen O, Tjonneland A, Roswall N, Clavel-Chapelon F, Boutron-Ruault MC, Touillaud M, et al. Consumption of vegetables and fruit and the risk of bladder cancer in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2009;125:2643–51. doi: 10.1002/ijc.24582. [DOI] [PubMed] [Google Scholar]
- 11.Buchner FL, Bueno-de-Mesquita HB, Ros MM, Kampman E, Egevad L, Overvad K, Tjonneland A, Roswall N, Clavel-Chapelon F, Boutron-Ruault MC, Touillaud M, Kaaks R, et al. Variety in vegetable and fruit consumption and risk of bladder cancer in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2011;128:2971–9. doi: 10.1002/ijc.25636. [DOI] [PubMed] [Google Scholar]
- 12.Zamora-Ros R, Sacerdote C, Ricceri F, Weiderpass E, Roswall N, Buckland G, St-Jules DE, Overvad K, Kyro C, Fagherazzi G, Kvaskoff M, Severi G, et al. Flavonoid and lignan intake in relation to bladder cancer risk in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br J Cancer. 2014;111:1870–80. doi: 10.1038/bjc.2014.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Larsson SC, Andersson SO, Johansson JE, Wolk A. Cultured milk, yogurt, and dairy intake in relation to bladder cancer risk in a prospective study of Swedish women and men. Am J Clin Nutr. 2008;88:1083–7. doi: 10.1093/ajcn/88.4.1083. [DOI] [PubMed] [Google Scholar]
- 14.Li F, Zhou Y, Hu RT, Hou LN, Du YJ, Zhang XJ, Olkkonen VM, Tan WL. Egg consumption and risk of bladder cancer: a meta-analysis. Nutr Cancer. 2013;65:538–46. doi: 10.1080/01635581.2013.770041. [DOI] [PubMed] [Google Scholar]
- 15.Zhou J, Smith S, Giovannucci E, Michaud DS. Reexamination of total fluid intake and bladder cancer in the Health Professionals Follow-up Study Cohort. Am J Epidemiol. 2012;175:696–705. doi: 10.1093/aje/kwr359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zeegers MP, Kellen E, Buntinx F, van den Brandt PA. The association between smoking, beverage consumption, diet and bladder cancer: a systematic literature review. World J Urol. 2004;21:392–401. doi: 10.1007/s00345-003-0382-8. [DOI] [PubMed] [Google Scholar]
- 17.Steck SE, Guinter M, Zheng J, Thomson CA. Index-Based Dietary Patterns and Colorectal Cancer Risk: A Systematic Review. Adv Nutr. 2015;6:763–73. doi: 10.3945/an.115.009746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reedy J, Krebs-Smith SM, Miller PE, Liese AD, Kahle LL, Park Y, Subar AF. Higher diet quality is associated with decreased risk of all-cause, cardiovascular disease, and cancer mortality among older adults. J Nutr. 2014;144:881–9. doi: 10.3945/jn.113.189407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sofi F, Macchi C, Abbate R, Gensini GF, Casini A. Mediterranean diet and health status: an updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014;17:2769–82. doi: 10.1017/S1368980013003169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shivappa N, Sandin S, Lof M, Hebert JR, Adami HO, Weiderpass E. Prospective study of dietary inflammatory index and risk of breast cancer in Swedish women. Br J Cancer. 2015;113:1099–103. doi: 10.1038/bjc.2015.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Giles GG, English DR. The Melbourne Collaborative Cohort Study. IARC Sci Publ. 2002;156:69–70. [PubMed] [Google Scholar]
- 22.Ireland P, Jolley D, Giles G, O’Dea K, Powles J, Rutishauser I, Wahlqvist ML, Williams J. Development of the Melbourne FFQ: a food frequency questionnaire for use in an Australian prospective study involving an ethnically diverse cohort. Asia Pac J Clin Nutr. 1994;3:19–31. [PubMed] [Google Scholar]
- 23.Trichopoulou A, Costacou T, Bamia C, Trichopoulos D. Adherence to a Mediterranean diet and survival in a Greek population. N Engl J Med. 2003;348:2599–608. doi: 10.1056/NEJMoa025039. [DOI] [PubMed] [Google Scholar]
- 24.Martinez-Gonzalez MA, de la Fuente-Arrillaga C, Nunez-Cordoba JM, Basterra-Gortari FJ, Beunza JJ, Vazquez Z, Benito S, Tortosa A, Bes-Rastrollo M. Adherence to Mediterranean diet and risk of developing diabetes: prospective cohort study. BMJ. 2008;336:1348–51. doi: 10.1136/bmj.39561.501007.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chiuve SE, Fung TT, Rimm EB, Hu FB, McCullough ML, Wang M, Stampfer MJ, Willett WC. Alternative dietary indices both strongly predict risk of chronic disease. J Nutr. 2012;142:1009–18. doi: 10.3945/jn.111.157222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shivappa N, Steck SE, Hurley TG, Hussey JR, Hebert JR. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 17:1689–96. doi: 10.1017/S1368980013002115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thiebaut AC, Benichou J. Choice of time-scale in Cox’s model analysis of epidemiologic cohort data: a simulation study. Stat Med. 2004;23:3803–20. doi: 10.1002/sim.2098. [DOI] [PubMed] [Google Scholar]
- 28.Grambsch PM, Therneau TM. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81:515–26. [Google Scholar]
- 29.MacInnis RJ, English DR, Hopper JL, Haydon AM, Gertig DM, Giles GG. Body size and composition and colon cancer risk in men. Cancer Epidemiol Biomarkers Prev. 2004;13:553–9. [PubMed] [Google Scholar]
- 30.Pink B. Socio-Economic Indexes for Areas (SEIFA) Australian Bureau of Statistics; 2013. [Google Scholar]
- 31.Andersen PK, Geskus RB, de Witte T, Putter H. Competing risks in epidemiology: possibilities and pitfalls. Int J Epidemiol. 2012;41:861–70. doi: 10.1093/ije/dyr213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lunn M, McNeil D. Applying Cox regression to competing risks. Biometrics. 1995;51:524–32. [PubMed] [Google Scholar]
- 33.Fritschi L, Driscoll T. Cancer due to occupation in Australia. Aust N Z J Public Health. 2006;30:213–9. doi: 10.1111/j.1467-842x.2006.tb00860.x. [DOI] [PubMed] [Google Scholar]
- 34.NHMRC, NHMRC. Australian Drinking Water Guidelines 6. National Health and Medical Research Council, National Resource Management Ministerial Council; 2011. [Google Scholar]
- 35.Shivappa N, Hebert JR, Rietzschel ER, De Buyzere ML, Langlois M, Debruyne E, Marcos A, Huybrechts I. Associations between dietary inflammatory index and inflammatory markers in the Asklepios Study. Br J Nutr. 2015;113:665–71. doi: 10.1017/S000711451400395X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shivappa N, Steck SE, Hurley TG, Hussey JR, Ma Y, Ockene IS, Tabung F, Hebert JR. A population-based dietary inflammatory index predicts levels of C-reactive protein in the Seasonal Variation of Blood Cholesterol Study (SEASONS) Publ Health Nutr. 2014;17:1825–33. doi: 10.1017/S1368980013002565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tognon G, Rothenberg E, Eiben G, Sundh V, Winkvist A, Lissner L. Does the Mediterranean diet predict longevity in the elderly? A Swedish perspective Age. 2011;33:439–50. doi: 10.1007/s11357-010-9193-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.NHMRC, NHMRC. Australian Dietary Guidelines. National Health and Medical Research Council, National Resource Management Ministerial Council; 2013. [Google Scholar]
- 39.Buckland G, Ros MM, Roswall N, Bueno-de-Mesquita HB, Travier N, Tjonneland A, Kiemeney LA, Sacerdote C, Tumino R, Ljungberg B, Gram IT, Weiderpass E, et al. Adherence to the Mediterranean diet and risk of bladder cancer in the EPIC cohort study. Int J Cancer. 2014;134:2504–11. doi: 10.1002/ijc.28573. [DOI] [PubMed] [Google Scholar]
- 40.Schwingshackl L, Hoffmann G. Adherence to Mediterranean diet and risk of cancer: an updated systematic review and meta-analysis of observational studies. Cancer Med. 2015 doi: 10.1002/cam4.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Brinkman MT, Buntinx F, Kellen E, Van Dongen MC, Dagnelie PC, Muls E, Zeegers MP. Consumption of animal products, olive oil and dietary fat and results from the Belgian case-control study on bladder cancer risk. Eur J Cancer. 2011;47:436–42. doi: 10.1016/j.ejca.2010.09.027. [DOI] [PubMed] [Google Scholar]
- 42.Shivappa N, Steck SE, Hussey JR, Ma Y, Hebert JR. Inflammatory potential of diet and all-cause, cardiovascular, and cancer mortality in National Health and Nutrition Examination Survey III Study. Eur J Nutr. 2015 doi: 10.1007/s00394-015-1112-x. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jakszyn P, Gonzalez CA, Lujan-Barroso L, Ros MM, Bueno-de-Mesquita HB, Roswall N, Tjonneland AM, Buchner FL, Egevad L, Overvad K, Raaschou-Nielsen O, Clavel-Chapelon F, et al. Red meat, dietary nitrosamines, and heme iron and risk of bladder cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) Cancer Epidemiol Biomarkers Prev. 2011;20:555–9. doi: 10.1158/1055-9965.EPI-10-0971. [DOI] [PubMed] [Google Scholar]
- 44.Romaguera D, Vergnaud AC, Peeters PH, van Gils CH, Chan DS, Ferrari P, Romieu I, Jenab M, Slimani N, Clavel-Chapelon F, Fagherazzi G, Perquier F, et al. Is concordance with World Cancer Research Fund/American Institute for Cancer Research guidelines for cancer prevention related to subsequent risk of cancer? Results from the EPIC study. Am J Clin Nutr. 2012;96:150–63. doi: 10.3945/ajcn.111.031674. [DOI] [PubMed] [Google Scholar]
- 45.Bagnardi V, Rota M, Botteri E, Tramacere I, Islami F, Fedirko V, Scotti L, Jenab M, Turati F, Pasquali E, Pelucchi C, Galeone C, et al. Alcohol consumption and site-specific cancer risk: a comprehensive dose-response meta-analysis. Br J Cancer. 2015;112:580–93. doi: 10.1038/bjc.2014.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kennedy GL, Pedro MR, Seghieri C, Nantel G, Brouwer I. Dietary diversity score is a useful indicator of micronutrient intake in non-breast-feeding Filipino children. J Nutr. 2007;137:472–7. doi: 10.1093/jn/137.2.472. [DOI] [PubMed] [Google Scholar]
- 47.Kant AK, Schatzkin A, Graubard BI, Schairer C. A prospective study of diet quality and mortality in women. JAMA. 2000;283:2109–15. doi: 10.1001/jama.283.16.2109. [DOI] [PubMed] [Google Scholar]
- 48.Karanja NM, Obarzanek E, Lin PH, McCullough ML, Phillips KM, Swain JF, Champagne CM, Hoben KP. Descriptive characteristics of the dietary patterns used in the Dietary Approaches to Stop Hypertension Trial. DASH Collaborative Research Group. J Am Diet Assoc. 1999;99:S19–27. doi: 10.1016/s0002-8223(99)00412-5. [DOI] [PubMed] [Google Scholar]
- 49.Dai J, Jones DP, Goldberg J, Ziegler TR, Bostick RM, Wilson PW, Manatunga AK, Shallenberger L, Jones L, Vaccarino V. Association between adherence to the Mediterranean diet and oxidative stress. Am J Clin Nutr. 2008;88:1364–70. doi: 10.3945/ajcn.2008.26528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schwingshackl L, Hoffmann G. Mediterranean dietary pattern, inflammation and endothelial function: a systematic review and meta-analysis of intervention trials. Nutr Metab Cardiovasc Dis. 2014;24:929–39. doi: 10.1016/j.numecd.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 51.Akbaraly TN, Shipley MJ, Ferrie JE, Virtanen M, Lowe G, Hamer M, Kivimaki M. Long-term adherence to healthy dietary guidelines and chronic inflammation in the prospective Whitehall II study. Am J Med. 2015;128:152–60. e4. doi: 10.1016/j.amjmed.2014.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Masson-Lecomte A, Rava M, Real FX, Hartmann A, Allory Y, Malats N. Inflammatory biomarkers and bladder cancer prognosis: a systematic review. Eur Urol. 2014;66:1078–91. doi: 10.1016/j.eururo.2014.07.033. [DOI] [PubMed] [Google Scholar]
- 53.Wu XR. Urothelial tumorigenesis: a tale of divergent pathways. Nat Rev Cancer. 2005;5:713–25. doi: 10.1038/nrc1697. [DOI] [PubMed] [Google Scholar]
- 54.Welch AA, Mulligan A, Bingham SA, Khaw KT. Urine pH is an indicator of dietary acid-base load, fruit and vegetables and meat intakes: results from the European Prospective Investigation into Cancer and Nutrition (EPIC)-Norfolk population study. Br J Nutr. 2008;99:1335–43. doi: 10.1017/S0007114507862350. [DOI] [PubMed] [Google Scholar]
- 55.Cohen SM, Masui T, Garland EM, Arnold LL. Effects of diet on urinary bladder carcinogenesis and cancer prevention. J Nutr. 1997;127:826S–9S. doi: 10.1093/jn/127.5.826S. [DOI] [PubMed] [Google Scholar]
- 56.Cicerale S, Conlan XA, Sinclair AJ, Keast RS. Chemistry and health of olive oil phenolics. Crit Rev Food Sci Nutr. 2009;49:218–36. doi: 10.1080/10408390701856223. [DOI] [PubMed] [Google Scholar]
- 57.Margel D, Pevsner-Fischer M, Baniel J, Yossepowitch O, Cohen IR. Stress proteins and cytokines are urinary biomarkers for diagnosis and staging of bladder cancer. Eur Urol. 2011;59:113–9. doi: 10.1016/j.eururo.2010.10.008. [DOI] [PubMed] [Google Scholar]
- 58.Pathak KV, Chiu TL, Amin EA, Turesky RJ. Methemoglobin Formation and Characterization of Hemoglobin Adducts of Carcinogenic Aromatic Amines and Heterocyclic Aromatic Amines. Chem Res Toxicol. 2016;29:255–69. doi: 10.1021/acs.chemrestox.5b00418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rothman N, Talaska G, Hayes RB, Bhatnagar VK, Bell DA, Lakshmi VM, Kashyap SK, Dosemeci M, Kashyap R, Hsu FF, Jaeger M, Hirvonen A, et al. Acidic urine pH is associated with elevated levels of free urinary benzidine and N-acetylbenzidine and urothelial cell DNA adducts in exposed workers. Cancer Epidemiol Biomarkers Prev. 1997;6:1039–42. [PubMed] [Google Scholar]
- 60.Yu J, Wang S, Zhao G, Wang B, Ding L, Zhang X, Xie J, Xie F. Determination of urinary aromatic amines in smokers and nonsmokers using a MIPs-SPE coupled with LC-MS/MS method. J Chromatogr B Analyt Technol Biomed Life Sci. 2014;958:130–5. doi: 10.1016/j.jchromb.2014.03.023. [DOI] [PubMed] [Google Scholar]
- 61.Vardavas CI, Flouris AD, Tsatsakis A, Kafatos AG, Saris WH. Does adherence to the Mediterranean diet have a protective effect against active and passive smoking? Public Health. 2011;125:121–8. doi: 10.1016/j.puhe.2010.11.012. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.