Significance
How does the “mind” (brain) influence the “body” (internal organs)? We identified key areas in the primate cerebral cortex that are linked through multisynaptic connections to the adrenal medulla. The most substantial influence originates from a broad network of motor areas that are involved in all aspects of skeletomotor control from response selection to motor preparation and movement execution. A smaller influence originates from a network in medial prefrontal cortex that is involved in the regulation of cognition and emotion. Thus, cortical areas involved in the control of movement, cognition, and affect are potential sources of central commands to influence sympathetic arousal. These results provide an anatomical basis for psychosomatic illness where mental states can alter organ function.
Keywords: cerebral cortex, sympathetic, psychosomatic, rabies virus
Abstract
Modern medicine has generally viewed the concept of “psychosomatic” disease with suspicion. This view arose partly because no neural networks were known for the mind, conceptually associated with the cerebral cortex, to influence autonomic and endocrine systems that control internal organs. Here, we used transneuronal transport of rabies virus to identify the areas of the primate cerebral cortex that communicate through multisynaptic connections with a major sympathetic effector, the adrenal medulla. We demonstrate that two broad networks in the cerebral cortex have access to the adrenal medulla. The larger network includes all of the cortical motor areas in the frontal lobe and portions of somatosensory cortex. A major component of this network originates from the supplementary motor area and the cingulate motor areas on the medial wall of the hemisphere. These cortical areas are involved in all aspects of skeletomotor control from response selection to motor preparation and movement execution. The second, smaller network originates in regions of medial prefrontal cortex, including a major contribution from pregenual and subgenual regions of anterior cingulate cortex. These cortical areas are involved in higher-order aspects of cognition and affect. These results indicate that specific multisynaptic circuits exist to link movement, cognition, and affect to the function of the adrenal medulla. This circuitry may mediate the effects of internal states like chronic stress and depression on organ function and, thus, provide a concrete neural substrate for some psychosomatic illness.
Everyone has experienced an “adrenaline rush,” an acute response to stress. Cannon (1) showed a century ago that this psychological experience and its physiological correlates (e.g., increased heart rate, sweating, pupillary dilation) involve secretion from the adrenal medulla triggered by sympathetic neurons in the thoracic spinal cord. He noted that these responses are anticipatory, preparing the body for “fight or flight.”
Sympathetic activation is equally essential for precise, organ-specific responses during exercise, exposure to heat or cold, and hypoglycemia, and activation can occur during cognitive deliberations and stressful social situations (2, 3). For example, we all have our own “hot buttons”—issues or events that can trigger an immediate intense reaction. Commonly, the event can be a critical remark from a supervisor, parent, or spouse. The most effective behavior in these circumstances is neither fight nor flight, but rather a more nuanced response that is adjusted to the context of the situation. Clearly, the response to stress is subject to extensive “top-down” or cognitive control (2, 4–6). Here, we used transneuronal transport of rabies virus (RV) to identify the areas of the cerebral cortex that are responsible for the top-down control of the adrenal medulla in a nonhuman primate.
RV is transported exclusively in the retrograde direction and moves transneuronally in a time-dependent manner (7). By varying the survival time, it is possible to trace synaptically linked circuits of up to six neurons in length. In the past, we have used the transneuronal transport of RV to define the location of cortical neurons that control specific muscles (8, 9) (Fig. 1, Left). Here, we injected RV (N2c strain) into the adrenal medulla and set the survival time in different animals to allow transport through chains of 2–4 synaptically linked neurons (second-order animal: n = 1; third-order animal: n = 1; fourth-order animals: n = 4) (Fig. 1, Right, and Fig. S1). Infected neurons were distributed throughout the brainstem and diencephalon in second- to fourth-order animals in patterns comparable to those described for rodents when similar studies were performed by using pseudorabies virus (10). This report will focus on the distribution of infected neurons in the cerebral cortex.
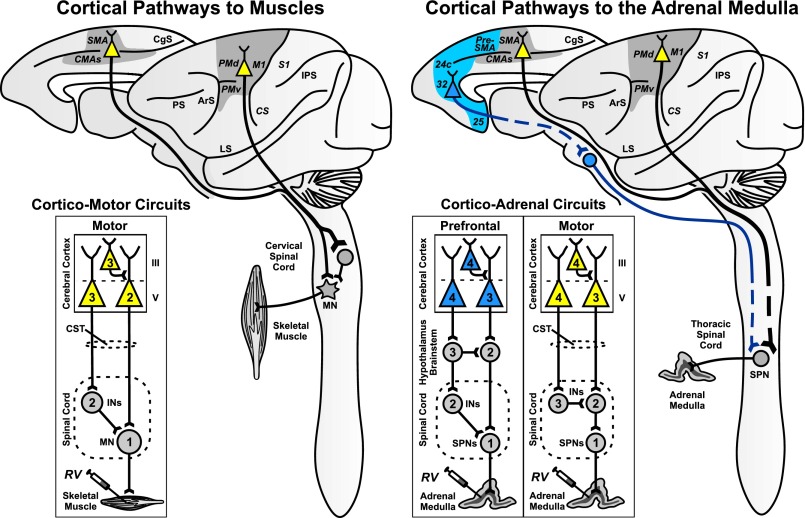
Fig. 1.
Mind–body circuits. (Left) Cortical pathways to muscles (8, 9). Cortical areas on the lateral surface and the medial wall of the hemisphere are the source of neurons that project to the spinal cord and influence motoneurons that innervate muscles. Inset (cortico-motor circuits) is a schematic diagram of the circuits that connect neurons in motor areas of the cerebral cortex to single muscles. When RV is injected into a muscle, it is transported transneuronally in the retrograde direction through these circuits. Depending on the survival time, the virus will infect first-order (1), second-order (2), or third-order (3) neurons. (Right) Cortical pathways to the adrenal medulla. Cortical areas on the lateral surface and the medial wall of the hemisphere are the source of neurons that influence the adrenal medulla. Inset (cortico-adrenal circuits) is a schematic diagram of the multisynaptic circuits that connect neurons in the motor and prefrontal areas of the cerebral cortex to the adrenal medulla. When RV is injected into the adrenal medulla, it is transported transneuronally in the retrograde direction through these circuits. Depending on the survival time, the virus will infect first-order (1), second-order (2), third-order (3), or fourth-order (4) neurons. Gray shading, cortical motor areas; blue shading, medial prefrontal areas. ArS, arcuate sulcus; CgS, cingulate sulcus; CMAs, cingulate motor areas; CS, central sulcus; CST, corticospinal tract; INs, interneurons; IPS, intraparietal sulcus; LS, lateral sulcus; M1, primary motor cortex; MN, motoneurons; PMd and PMv, dorsal and ventral premotor areas; PS, principal sulcus; S1, primary somatosensory cortex; SMA, supplementary motor area; SPNs, sympathetic preganglionic neurons.
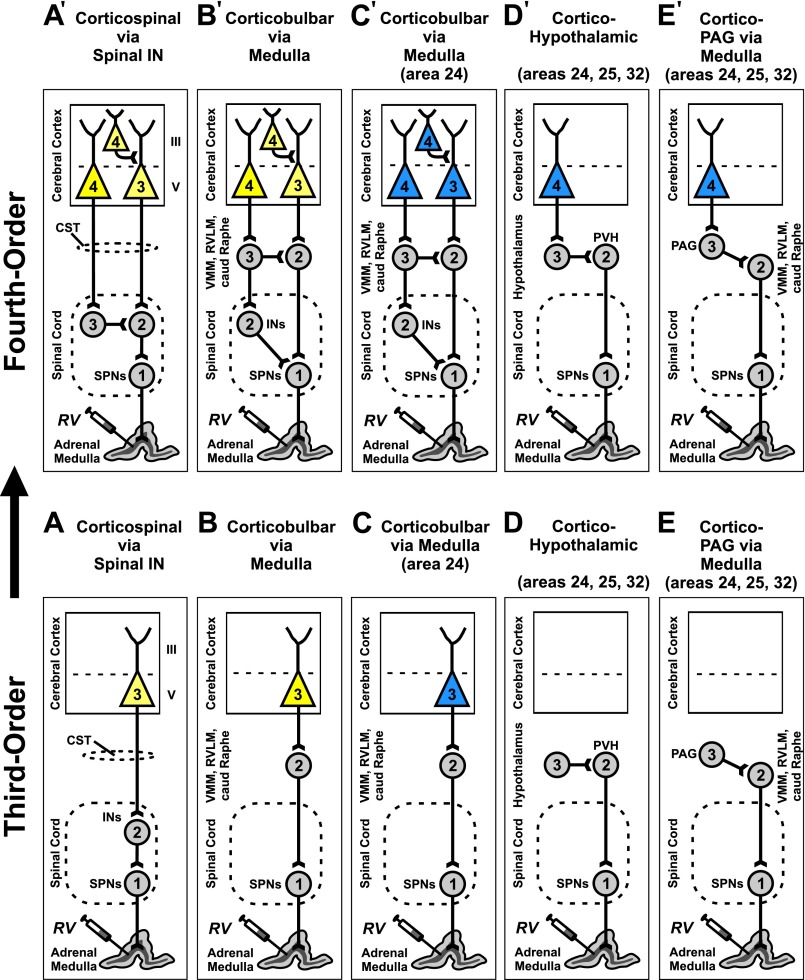
Fig. S1.
Routes for viral transport from the adrenal medulla to the cerebral cortex. Circuit diagrams illustrate likely pathways for retrograde, transneuronal transport of RV from the adrenal medulla to the cerebral cortex. (Lower) All potential pathways begin with retrograde transport of RV to the first-order sympathetic preganglionic neurons (SPNs) in the spinal cord. Second-order neurons are then labeled by retrograde, transneuronal transport of RV from the SPNs. These neurons include spinal interneurons (INs) and neurons in the medullary reticular formation with axons descending into the spinal cord [i.e., ventromedial medulla (VMM), rostral ventrolateral medulla (RVLM), caudal raphe complex (caud Raphe)]. Third-order neurons in layer V of the cerebral cortex can be labeled transneuronally from second-order neurons in the spinal cord or second-order neurons in the medullary reticular formation (A–C). Three interconnected neurons represents the absolute minimum circuit for transport to the cerebral cortex. In one animal, we found that only a small number of cortical neurons (256) were labeled in this manner. These neurons were found in layer V of the motor areas in the frontal lobe (A and B) (yellow) and in area 24 of the medial prefrontal network (C) (blue). (Upper) Extending the survival time to enable transport through four interconnected neurons labeled many more neurons and additional areas of the cerebral cortex (n = 4, mean = 5,232) (A′–E′). See Materials and Methods for the basis of determining the “order” of transport. CST, corticospinal tract; PAG, periaqueductal gray; PVN, paraventricular nucleus of the hypothalamus.
We first observed substantial numbers of infected neurons (mean = 5,232) in the cerebral cortex in fourth-order animals. Most of these infected neurons were located in layer V, the main source of descending outputs from the cerebral cortex (Fig. S2). Retrograde transneuronal transport of RV from an injection site in the adrenal medulla infected neurons largely (87%) in two nonoverlapping sets of cortical areas: a “motor” network (11–13) and a “medial prefrontal” network (14) (Fig. 2). Small numbers of infected neurons also were located in areas within the lateral sulcus (∼7%) and orbitofrontal cortex (∼3%).
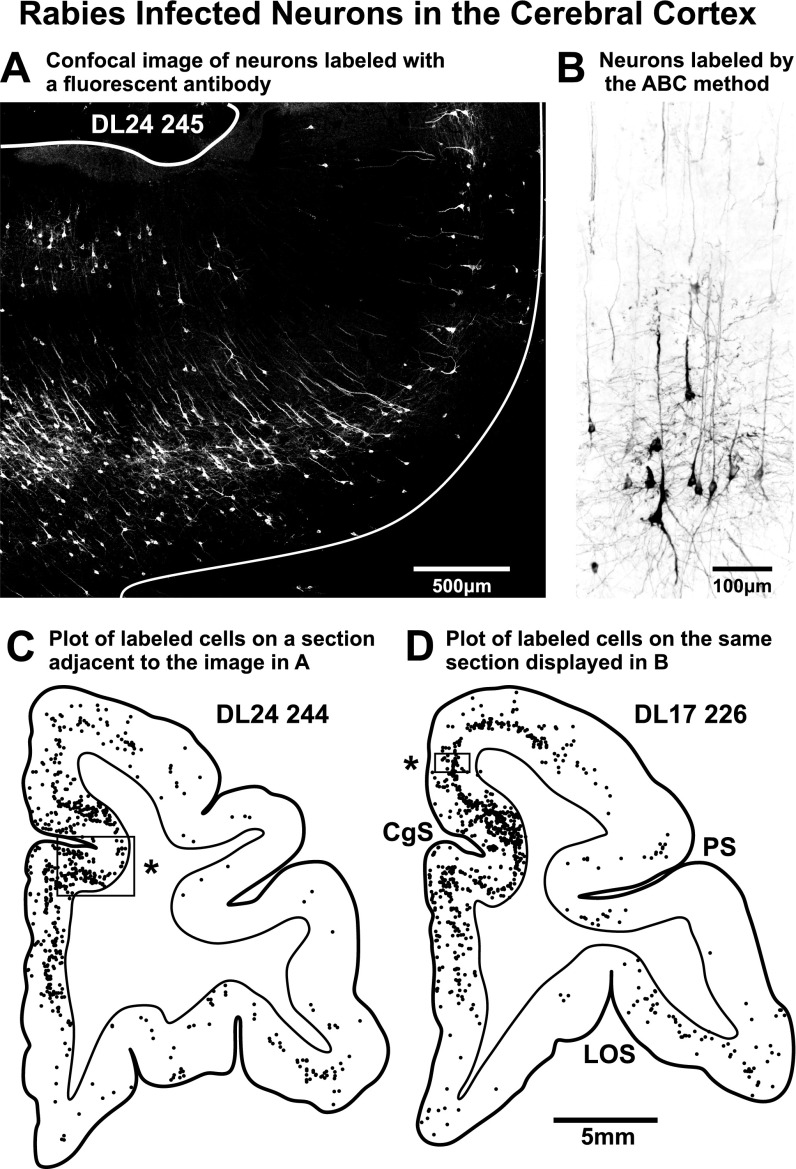
Fig. S2.
Labeled neurons in the cerebral cortex. (A) Confocal image of the ventral bank of the cingulate sulcus showing neurons infected with RV. The section was processed with a fluorescent secondary antibody (Alexa 488, diluted 1:300, Jackson ImmunoResearch) to reveal Golgi-like filling of infected neurons. This image was stitched together from multiple frames with Nikon’s confocal software and postprocessed by adjusting brightness, contrast, and intensity (Corel Photopaint). Then, the reduce noise function (auto) of Corel Draw was applied. (B) Rabies infected neurons in the cerebral cortex. This material was processed by using the ABC method (Vectastain; Materials and Methods). (C) Chart of labeled neurons found on a section of frontal cortex. One dot = one infected cell. The region photographed in A is indicated by the box and asterisk in C, but the photograph was taken of an adjacent section. (D) Chart of labeled neurons found on a section of frontal cortex. The region photographed in B is indicated by the box and asterisk in D. CgS, cingulate sulcus; LOS, lateral orbital sulcus; PS, principal sulcus.
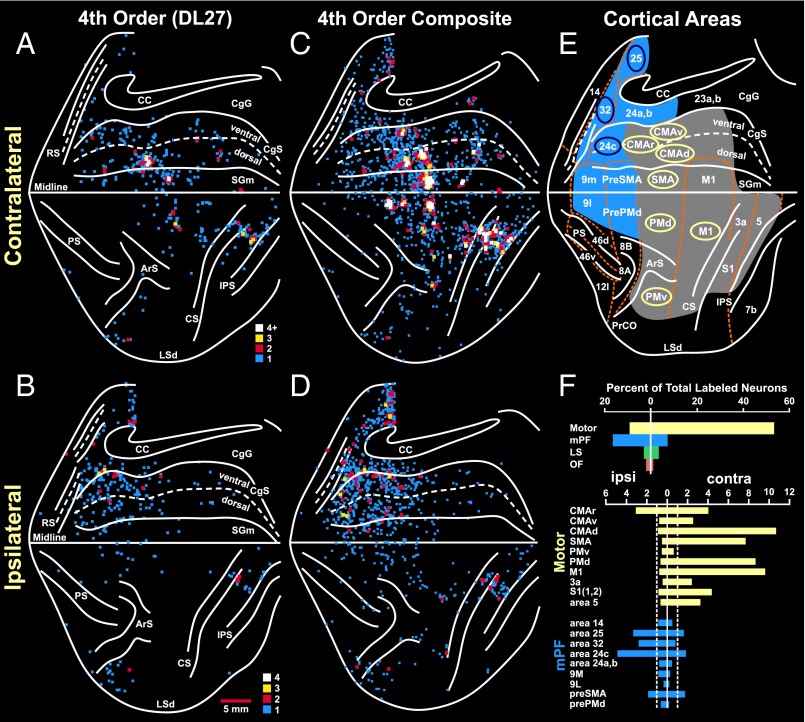
Fig. 2.
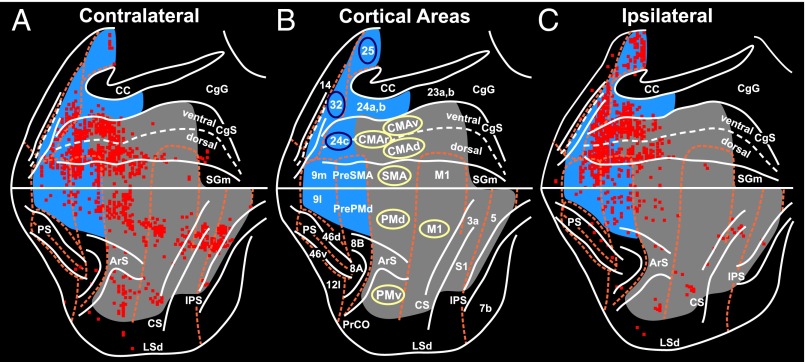
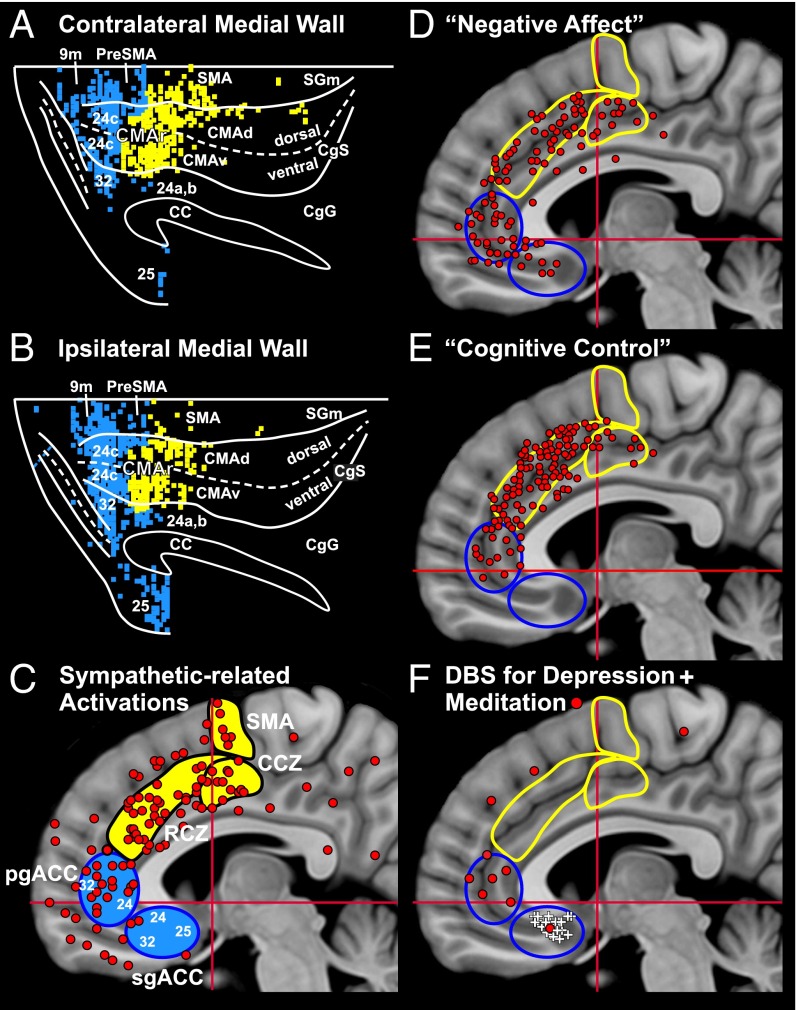
Cortical origin of multisynaptic inputs to the adrenal medulla. (A and B) Flat map of the distribution of neurons in the frontal and parietal lobes after RV was injected into the adrenal medulla (case DL27). (A) Contralateral hemisphere. (B) Ipsilateral hemisphere. The medial wall of the hemisphere is reflected upwards (11). These results were obtained by using a survival time that allowed retrograde transneuronal transport of rabies to label fourth-order neurons. The colored squares indicate the number of labeled neurons found in successive 200-μm bins found on each 50-μm section (color key). (C and D) Composite maps of the distribution of fourth-order neurons in the frontal and parietal lobes of four animals injected with rabies into the adrenal medulla. (C) Contralateral hemisphere. (D) Ipsilateral hemisphere. (E) Cortical areas under analysis. Motor and somatosensory areas are shaded gray. Medial prefrontal areas are shaded blue. Cortical motor areas in the frontal lobe are indicated by the yellow ellipses; specific areas of cingulate cortex are indicated by blue ellipses. (F) Histograms of the distribution of labeled neurons in cortical areas contralateral (right) and ipsilateral (left) to the injection site. Motor areas, yellow bars; medial prefrontal areas, blue bars; lateral sulcus areas (LS), green bar; orbitofrontal areas (OF), red bar. Top histogram, summary distribution; bottom histogram, distribution in specific areas. Dashed lines indicate 1% level of the total number of infected cortical neurons. CC, corpus callosum; CgG, cingulate gyrus; CMAd, CMAr, and CMAv, dorsal, rostral, and ventral cingulate motor areas; LSd, dorsal lip of the lateral sulcus; mPF, medial prefrontal cortex; RS, rostral sulcus; SGm, medial superior frontal gyrus. Other abbreviations defined in the legend of Fig. 1.
The motor network was the major source of descending influence over the adrenal medulla (63% of all labeled neurons) (Fig. 2F). This network included all seven of the cortical motor areas in the frontal lobe and specific regions of somatosensory cortex (areas 3a, 1, 2) and posterior parietal cortex (area 5) (Figs. 2 and 3). All of the motor areas projected primarily to the contralateral adrenal medulla (contralateral to ipsilateral ratio ∼85:15) except for the rostral cingulate motor area (CMAr), which projected bilaterally. The strongest projections originated from the “trunk” representation of the primary motor cortex (M1), from the dorsal premotor area (PMd) on the lateral surface of the hemisphere, and from the four motor areas on the medial wall of the hemisphere [the supplementary motor area (SMA), and the rostral, dorsal, and ventral cingulate motor areas (CMAr, CMAd, and CMAv)]. Each of the cortical motor areas has a human equivalent. On the medial wall, the CMAr and CMAv of the monkey are considered to be the rostral cingulate zone (RCZ) in humans and the CMAd is the caudal cingulate zone (CCZ) in humans (15, 16) (Fig. 4 A–C).
Fig. 3.
Densest cortical origin of multisynaptic inputs to the adrenal medulla. The survival time in this animal allowed retrograde transneuronal transport of rabies to label sixth-order neurons. The red squares indicate 200-μm bins on single sections that contained the largest number of labeled neurons (top 15%). (A) Contralateral hemisphere. (C) Ipsilateral hemisphere. (B) Areas of the cerebral cortex in the diagram. Conventions and abbreviations as in Fig. 2.
Fig. 4.
Monkey anatomy and human neuroimaging: comparison of results. The densest origin of projections to the adrenal medulla are color-coded: motor areas, yellow bins; medial prefrontal areas, blue bins (from Fig. 3). (A) Contralateral medial wall of the hemisphere. (B) Ipsilateral medial wall. Dorsal is at the top and anterior is to the left for all of the diagrams in this figure. (C–F) Sites of activation on the medial wall of the hemisphere found in human studies. Each diagram shows the location of the SMA, CCZ, RCZ, pgACC, and sgACC. Motor areas are filled or outlined in yellow; medial prefrontal areas are filled or outlined in blue. (C) Sympathetic-related activations, red circles (36 studies, see SI Materials and Methods for the studies included). (D) Negative affect, red circles (data from ref. 31). (E) Cognitive control, red circles (data from ref. 31). (F) Sites of deep brain stimulation (DBS) for treatment-resistant depression, white pluses (data from ref. 33); sites of activation associated with meditation, red circles (see SI Materials and Methods for studies included). CA-CP, red horizontal lines; Vca; red vertical lines.
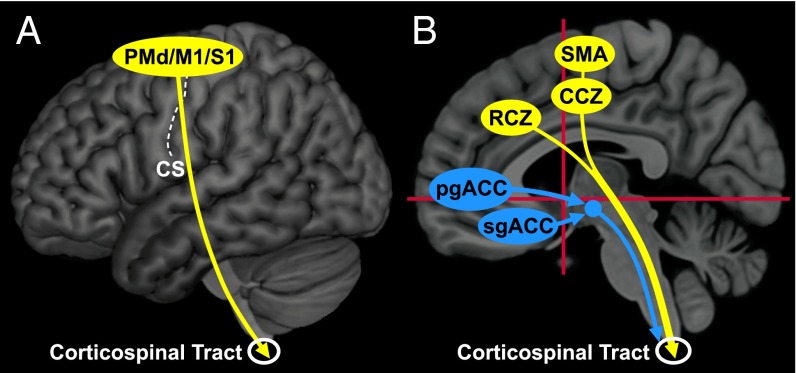
The cortical areas that form the motor network are richly interconnected (12, 13). Furthermore, all of these cortical motor areas project directly to the spinal cord (11, 13) and to regions of the reticular formation (17). Thus, descending cortico-spinal and corticobulbo-spinal pathways may mediate some or all of the influence of the motor network on the adrenal medulla (Fig. 5 and Fig. S1). This conclusion is supported by classic studies that demonstrated that surface stimulation in restricted regions of M1 and the PMd evoked changes in blood pressure (18). These changes survived lesions of the fifth nerve and hypothalamus, but were abolished by lesions of the pyramidal tract (18, 19).
Fig. 5.
Origin and pathways for top-down influence over the adrenal medulla by the cerebral cortex. Motor areas are filled yellow, and medial prefrontal areas are filled blue. (A) Major cortical areas for top-down control on the lateral surface of the hemisphere. All of these areas send projections to the spinal cord. (B) Major cortical areas for top-down control on the medial wall of the hemisphere. The motor areas send projections to the spinal cord, whereas the output of medial prefrontal areas is likely to be mediated by various subcortical centers. CA-CP, red horizontal lines; Vca; red vertical lines.
The medial prefrontal network represented nearly 25% of the descending cortical output to the adrenal medulla (Fig. 2F). This network originated from multiple, distinct regions of medial prefrontal cortex on the medial wall of the hemisphere and on the lateral surface of the hemisphere (Figs. 2 and 3). Ipsilateral projections from the medial prefrontal network to the adrenal medulla were approximately twice as numerous as the contralateral projections. The core of this network originated from two regions of anterior cingulate cortex (ACC)—a “pregenual” region including portions of areas 32 and 24 and a “subgenual” region consisting mainly of area 25 (Figs. 2 and 3). Comparable pregenual and subgenual regions of the ACC have been identified in humans (Fig. 4 A–C) (20–22).
The pregenual and subgenual regions of the anterior cingulate cortex are densely interconnected (14). These cortical areas in nonhuman primates do not project directly to the spinal cord. Instead, neurons in these regions were probably infected through their reported projections to the medullary reticular formation, hypothalamus, and periaqueductal gray (14, 17, 23), which, in turn, project to brainstem sympathetic nuclei and to the spinal cord (Fig. 1 and Fig. S1) (24–26).
To identify cortical areas that may be less directly connected to the adrenal medulla, we extended the survival time to allow transport through chains of 5–6 synaptically linked neurons (fifth-order animals: n = 2; and sixth-order animals: n = 2). Prolonging the survival time resulted in a dramatic increase (20- to 100-fold) in the numbers of labeled neurons in the cerebral cortex (fifth order, mean = ∼104,000; sixth order, mean = ∼480,000). Large numbers of labeled neurons were located not only in layer V, but also in supra- and infragranular layers of cortex. However, the cortical areas with dense labeling (i.e., local peak density, top 15% of bins) in sixth-order animals remained the same as those that were densely labeled in fourth-order animals (Figs. 2 and 3). As with the shorter survival times, smaller concentrations of infected neurons also were located in cortical areas in the lateral sulcus (<11%) and in regions of orbitofrontal cortex (<6%). No other cortical regions contained more than 2% of the labeled neurons. Thus, independent of the survival time, the motor and prefrontal networks are the major sources of descending influence over the adrenal medulla.
Discussion
Our results have a number of important functional implications. First, they emphasize the importance of the cortical motor areas in the top-down influence over the adrenal medulla. Normal limb and body movements are accompanied by coordinated changes in sympathetic output that are proportional to the metabolic demands of the task. An important component of this sympathetic activity is thought to be due to a “central command” that can precede any change in muscle activity (4–6). This command is a “predictive” response because it prepares the body to meet the anticipated metabolic needs of activities like exercise, emotional states, and fight or flight. Our results suggest that the central commands to prepare for and generate visceromotor output originate from the same cortical areas that are involved in the preparation and generation of skeletomotor output. The colocalization of visceromotor and skeletomotor function may provide a mechanism to facilitate the coordination of these two motor systems (Fig. 5).
Second, we found that the top-down influence over the adrenal medulla by M1 originates largely from its trunk representation. This region of M1 projects to thoracic segments of the spinal cord (27). These spinal segments also contain the sympathetic preganglionic neurons that form the final common pathway to the adrenal medulla (10). These observations suggest that there is a link between the descending control of “core muscles” and the regulation of sympathetic output. This link could provide a neural substrate for the control of stress through “core” exercises, such as yoga and pilates (28).
Third, our results indicate that a major component of the cortical influence over the adrenal medulla arises from multiple motor and nonmotor areas on the medial wall of the hemisphere (Figs. 2, 3, and 5B). This finding differs from the view that autonomic outflow from the cingulate cortex originates from a single region, the subgenual ACC (22). Our perspective is supported by functional imaging data from human studies that show clusters of activations associated with sympathetic arousal in multiple areas of the cingulate cortex including the RCZ, CCZ, and pregenual ACC (Fig. 4C).
The human counterparts of the CMAr, CMAv, CMAd, and pregenual and subgenual ACC (Fig. 4 A and B) all display activation in tasks that are associated with negative affect (Fig. 4D). The RCZ of humans and the CMAr of monkeys are consistently activated in tasks requiring “cognitive control” such as response selection, conflict, and error awareness (Fig. 4E) (22, 29–32). In essence, our results provide the neural circuitry that links negative affect (e.g., sadness) and cognitive control processes (e.g., awareness of errors) to stress responses. The same substrate may mediate comparable stress responses when a sad situation or an error is recalled. Furthermore, abnormal activation of this circuitry may be fundamental to conditions like posttraumatic stress disorder.
The cortical areas we have identified as regulating the adrenal medulla overlap substantially with those included in “depression networks” (20, 21). One node in this network, subgenual ACC, displays histological changes in subjects with bipolar familial depression (21), and deep brain stimulation near subgenual ACC mitigates some symptoms of depression (20, 33) (Fig. 4F). Another node in the network, the pregenual ACC, is a site of activation during mindful meditation (Fig. 4F), a behavioral technique for reducing anxiety. Similarly, the pregenual ACC in monkeys appears to be uniquely linked with reward-related systems in the basal ganglia (34) and to be involved in regulating anxiety and emotional valence while deciding on a course of action (22, 35–37). Taken together, these findings raise the possibility that the areas of the cerebral cortex that influence the adrenal medulla (Fig. 5) also are key cortical nodes of a “stress and depression connectome.” Perhaps some of these nodes represent additional targets for therapeutic intervention in affective disorders.
In summary, the control of the adrenal medulla is embedded in cortical areas that are involved in the control of movement, cognition and affect (Fig. 5). This means that mental operations like the preparation to move, the appreciation of conflict and feelings of sadness may be linked to the regulation of adrenal function. The cerebral cortex is a site of neural plasticity and information storage. As a consequence, aberrant plasticity could be a cause of or contribute to psychosomatic illnesses, just as the engagement of cortical plasticity through moving, thinking and feeling may help to cure or ameliorate them.
Materials and Methods
This report is based on observations from Cebus monkeys (1.4–3.7 kg; 3 male, 6 female), which received injections of RV (CVS-N2c; 5.0 × 108 pfu/mL) into the adrenal medulla. RV was especially useful for these experiments because current evidence indicates that RV is transported transneuronally in all types of systems and across all types of synapses (38–40). In addition, there is no evidence that the N2c strain is transported more efficiently in some pathways than others.
All procedures were in accordance with the Association for Assessment and Accreditation of Laboratory Animal Care and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The experimental protocol was approved by both the Institutional Animal Care and Use Committee and the Biosafety Committee at the University of Pittsburgh. Biosafety practices conformed to Biosafety Level 2 regulations outlined in Biosafety in Microbiological and Biomedical Laboratories (41). Procedural details for handling virus and virus-infected animals have been published (38).
Adrenal Medulla Injections.
Surgeries were performed under general anesthesia [15 mg/kg Ketamine IM for sedation and 1–3% (vol/vol) isoflurane in 1–3 liters/min O2 for general anesthesia] and aseptic conditions. Analgesics (buprenorphine 0.01–0.03 mg/kg s.c.) were given perioperatively. The left adrenal medulla was accessed via a paralumbar incision. We placed multiple RV injections (total of 80–100 μL) throughout the central regions of the adrenal gland by using a Hamilton microsyringe with a 30-gauge needle. The injection syringe was held in place for 30 s. After removal of the injection needle, the site was blotted with a sterile cotton swab to prevent any leakage through the injection tract. Following the injections, the wound was sutured, and the animal was returned to a cage designed for housing virus-infected animals.
Survival Period.
We varied the survival times from 97 to 136 h. The mean survival times were 120.2 h (fourth-order labeling, n = 4), 126.8 h (fifth-order labeling, n = 2), and 131.5 h (sixth-order labeling, n = 2). At the end of the survival period, each animal was sedated with ketamine (25 mg/kg), deeply anesthetized with sodium pentobarbital (40 mg/kg IP), and perfused transcardially with three solutions: (i) 0.1 M phosphate buffer, (ii) formalin (3.7 gm formaldehyde gas/100 ml of 0.1 M phosphate buffer), and (iii) 10% (vol/vol) glycerol. Two to three liters of each solution was delivered over a period of 7–10 min per solution. After perfusion, the brains and spinal cords were removed and stored at 4 °C in 10% (vol/vol) phosphate-buffered formalin with 20% (vol/vol) glycerol.
Histological Procedures.
We cut serial frozen sections (50 μm) in the coronal plane of a brain block including the cerebral cortex from its rostral pole to the parietal-occipital sulcus. The cerebellum and brainstem were cut separately. We also cut serial frozen sections (50 μm) of a spinal cord block containing the fourth thoracic segment (T4) to the first lumbar segment (L1) in the transverse plane. We stained every 10th section of the brain and every 20th of the spinal cord with cresyl violet to enable analysis of cytoarchitecture. To identify virus-infected neurons, we performed immunohistochemical reactions on free-floating sections according to the avidin-biotin peroxidase method (Vectastain; Vector Laboratories). The reactions used a mouse monoclonal antibody (M957, diluted 1:300; supplied by A. Wandeler, Animal Disease Research Institute, Nepean, ON, Canada) that is specific for the P antigen expressed by RV (42). All reacted sections were mounted on gelatin-coated glass slides, air-dried, and coverslipped with Cytoseal.
Analytic Procedures.
Sections through the entire brain (every fourth or eighth) and spinal cord (every 10th) were examined under the microscope for reaction product by using brightfield and/or polarized light illumination. Another series of sections (every 10th) was stained and examined for cytoarchitecture. We plotted section outlines, labeled neurons, gray-white matter boundaries, cytoarchitectonic borders, and other anatomic features by using a computer-based charting system (MD2 or MD3; Accustage). Then, we used these plots along with software written in the laboratory to create unfolded cortical maps that displayed the distribution of labeled neurons, cortical sulci, and cytoarchitectonic borders on a 2D surface. The procedures used to unfold the cortex and generate maps of the lateral surface and medial wall of the hemisphere have been described (11). The criteria we used to define cytoarchitectonic borders in the cebus monkey have been presented in prior studies (refs. 12 and 43; see also ref. 44). To create the composite maps (Fig. 2 C and D), we overlaid the unfolded maps from four of the fourth-order animals. Then, the unfolded maps of the lateral surface were aligned on the central and arcuate sulci and the medial edge of the hemisphere. The maps of the medial wall were aligned on the anterior posterior extent of the corpus callosum and the medial edge of the hemisphere. The maps of individual animals required only small shifts (<1 mm) in the anterior-posterior and medio-lateral directions to achieve a best fit.
Determining Order of Transport.
Survival time, in and of itself, is not sufficient to define the number of synapses mediating transneuronal transport (i.e., order of transport) in any experiment. Instead, we use the distribution of infected neurons within well-established pathways as internal controls (Fig. 1, Right). Specifically, after injections of rabies into the adrenal medulla, we determined the order of transport based on the following patterns of labeling: third-order transport, infected neurons are first seen in layer V of the cerebral cortex; fourth-order transport, infected neurons are first seen in cortical layers outside of layer V and in the deep cerebellar nuclei; fifth-order transport, infected neurons are first seen in cerebellar cortex (i.e., Purkinje cells); and sixth-order transport, infected neurons are first seen in the inferior olive.
SI Materials and Methods
Criteria for Inclusion of Sympathetic-Related Activations (Fig. 4C and refs. 45–80).
The initial selection of studies used the Neurosynth.org database with the search term “autonomic.” Ninety-one studies were retrieved. Additional studies were found by using the references cited in these papers or gleaned from reviews of imaging studies. All activation coordinates were plotted in Montreal Neurological Institute (MNI) space on a brain template image at x = 5 from the ICBM 2009b Nonlinear Asymmetric 0.5 × 0.5 × 0.5 mm template (www.bic.mni.mcgill.ca/ServicesAtlases/ICBM152NLin2009). Because coordinates derived from PET studies were usually in Talairach space, these coordinates were converted to MNI coordinates with Ginger ALE 2.3.5 (www.brainmap.org/ale). As a further check on the accuracy of the plots, the location of each site plotted on our MNI brain template was required to match the location shown in the cited paper.
Studies were included on the basis of the following criteria: (i) Only increases in regional cerebral blood flow (rCBF) that were on the medial wall of the hemisphere were plotted. Both fMRI and PET studies were included. (ii) The activation was reported to be related to a parameter that reflects sympathetic arousal. These parameters included skin conductance responses, mean arterial pressure, low frequency heart rate interval, pupil dilation, increased heart rate, suppression of baroreflex activity, sympathetic nerve activity, and increases in epinephrine or cortisol. (iii) Redundant coordinates in a single paper were filtered by requiring them to be at least 1 cm apart or if there were symmetrical coordinates in each hemisphere, only the coordinate on the right side was plotted to avoid biasing the results. (iv) The measurements of sympathetic arousal were performed simultaneously with the imaging in the scanner.
Criteria for Inclusion of Cognitive Control and Negative Affect Activations (Fig. 4 D and E).
D and E in Fig. 4 were adapted from Shackman et al. (31). All activations were derived from a metaanalysis by using the BrainMap database.
“Negative Affect” in Fig. 3D is represented by 117 activations from 59 publications (ref. 31 and this section). Negative affect included negative emotions (38%), anxiety/fear (28%), sadness (17%), disgust, anger, and guilt. These negative emotions were evoked by mixed induction/provocation (34%), visual stimuli (film/images) (24%), aversive Pavlovian conditioning (11%), aversive instrumental, anticipate monetary penalty, threat of shock, aversive visual, anticipate tastants, and CO2 challenge.
Cognitive Control in Fig. 3E is represented by 168 activations from 90 publications (ref. 31 and this section). Tasks that evoked cognitive processes were designed to manage conflict during a decision that required suppressing reflexive actions to achieve a goal. These tasks included Stroop (30%), go/no go (16%), antisaccade/antipointing (12%), stimulus-response reversal, flanker, card sort, miscellaneous conflict, stop, drawing, picture naming, and motion prediction. These tasks were thought to be produced by cognitive processes that were categorized as response selection (53%), response inhibition (13%), error detection/correction (12%), negative feedback/set shifting (11%), task switching, interference, and phonological/semantic interference. Studies using monetary punishment were excluded from cognitive control activations.
Criteria for Inclusion of Meditation-Related Activations (Fig. 4F).
Recent publications were found using the terms “meditation, stress, fMRI” in PubMed (n = 26). Coordinates were plotted in MNI space as described above. Only sites where rCBF increased during meditation versus controls were plotted (81–84).
Acknowledgments
We thank M. Schnell (Thomas Jefferson University) for supplying the N2c strain of rabies virus; A. Wandeler (Animal Disease Research Institute) for supplying the antibody to the rabies virus; M. Page and M. Semcheski for the development of computer programs; D. Sipula, T. Cox, M. O’Malley, and M. Watach for their technical assistance; and P. Sterling for comments on the manuscript. This work was supported in part by NIH Grants R01 NS24328 (to P.L.S.), P40 OD010996 (to P.L.S.), P30 NS076405 (to P.L.S.), T32 DK063922 (to D.J.L.), and K08 DK101756-01 (to D.J.L.). This project was also funded, in part, under a grant with the Pennsylvania Department of Health.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1605044113/-/DCSupplemental.
References
- 1.Cannon WB. Bodily Changes in Pain, Hunger, Fear and Rage. 2nd Ed Appleton; New York: 1929. [Google Scholar]
- 2.Critchley HD. Neural mechanisms of autonomic, affective, and cognitive integration. J Comp Neurol. 2005;493(1):154–166. doi: 10.1002/cne.20749. [DOI] [PubMed] [Google Scholar]
- 3.Goldstein DS, Kopin IJ. Adrenomedullary, adrenocortical, and sympathoneural responses to stressors: A meta-analysis. Endocr Regul. 2008;42(4):111–119. [PMC free article] [PubMed] [Google Scholar]
- 4.Cechetto DF, Saper CB. Role of the cerebral cortex in autonomic function. In: Lowey AD, Spyer KM, editors. Central Regulation of Autonomic Function. Oxford Univ Press; Oxford: 1990. pp. 208–223. [Google Scholar]
- 5.Williamson JW, Fadel PJ, Mitchell JH. New insights into central cardiovascular control during exercise in humans: A central command update. Exp Physiol. 2006;91(1):51–58. doi: 10.1113/expphysiol.2005.032037. [DOI] [PubMed] [Google Scholar]
- 6.Shoemaker JK, Wong SW, Cechetto DF. Cortical circuitry associated with reflex cardiovascular control in humans: Does the cortical autonomic network “speak” or “listen” during cardiovascular arousal. Anat Rec (Hoboken) 2012;295(9):1375–1384. doi: 10.1002/ar.22528. [DOI] [PubMed] [Google Scholar]
- 7.Dum RP, Strick PL. Transneuronal tracing with neurotropic viruses reveals network macroarchitecture. Curr Opin Neurobiol. 2013;23(2):245–249. doi: 10.1016/j.conb.2012.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rathelot JA, Strick PL. Muscle representation in the macaque motor cortex: An anatomical perspective. Proc Natl Acad Sci USA. 2006;103(21):8257–8262. doi: 10.1073/pnas.0602933103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rathelot JA, Strick PL. Subdivisions of primary motor cortex based on cortico-motoneuronal cells. Proc Natl Acad Sci USA. 2009;106(3):918–923. doi: 10.1073/pnas.0808362106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Strack AM, Sawyer WB, Platt KB, Loewy AD. CNS cell groups regulating the sympathetic outflow to adrenal gland as revealed by transneuronal cell body labeling with pseudorabies virus. Brain Res. 1989;491(2):274–296. doi: 10.1016/0006-8993(89)90063-2. [DOI] [PubMed] [Google Scholar]
- 11.Dum RP, Strick PL. The origin of corticospinal projections from the premotor areas in the frontal lobe. J Neurosci. 1991;11(3):667–689. doi: 10.1523/JNEUROSCI.11-03-00667.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dum RP, Strick PL. Frontal lobe inputs to the digit representations of the motor areas on the lateral surface of the hemisphere. J Neurosci. 2005;25(6):1375–1386. doi: 10.1523/JNEUROSCI.3902-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dum RP, Strick PL. In: Motor Cortex in Voluntary Movements. Riehle A, Vaadia E, editors. CRC; Boca Raton, FL: 2005. pp. 3–47. [Google Scholar]
- 14.Price JL. Definition of the orbital cortex in relation to specific connections with limbic and visceral structures and other cortical regions. Ann N Y Acad Sci. 2007;1121:54–71. doi: 10.1196/annals.1401.008. [DOI] [PubMed] [Google Scholar]
- 15.Picard N, Strick PL. Motor areas of the medial wall: A review of their location and functional activation. Cereb Cortex. 1996;6(3):342–353. doi: 10.1093/cercor/6.3.342. [DOI] [PubMed] [Google Scholar]
- 16.Amiez C, Petrides M. Neuroimaging evidence of the anatomo-functional organization of the human cingulate motor areas. Cereb Cortex. 2014;24(3):563–578. doi: 10.1093/cercor/bhs329. [DOI] [PubMed] [Google Scholar]
- 17.Keizer K, Kuypers HG. Distribution of corticospinal neurons with collaterals to the lower brain stem reticular formation in monkey (Macaca fascicularis) Exp Brain Res. 1989;74(2):311–318. doi: 10.1007/BF00248864. [DOI] [PubMed] [Google Scholar]
- 18.Wall PD, Pribram KH. Trigeminal neurotomy and blood pressure responses from stimulation of lateral cerebral cortex of Macaca mulatta. J Neurophysiol. 1950;13(6):409–412. doi: 10.1152/jn.1950.13.6.409. [DOI] [PubMed] [Google Scholar]
- 19.Wall PD, Davis GD. Three cerebral cortical systems affecting autonomic function. J Neurophysiol. 1951;14(6):507–517. doi: 10.1152/jn.1951.14.6.507. [DOI] [PubMed] [Google Scholar]
- 20.Mayberg HS. Targeted electrode-based modulation of neural circuits for depression. J Clin Invest. 2009;119(4):717–725. doi: 10.1172/JCI38454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Price JL, Drevets WC. Neural circuits underlying the pathophysiology of mood disorders. Trends Cogn Sci. 2012;16(1):61–71. doi: 10.1016/j.tics.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 22.Vogt BA. Submodalities of emotion in the context of cingulate subregions. Cortex. 2014;59:197–202. doi: 10.1016/j.cortex.2014.04.002. [DOI] [PubMed] [Google Scholar]
- 23.Ongür D, An X, Price JL. Prefrontal cortical projections to the hypothalamus in macaque monkeys. J Comp Neurol. 1998;401(4):480–505. [PubMed] [Google Scholar]
- 24.Veazey RB, Amaral DG, Cowan WM. The morphology and connections of the posterior hypothalamus in the cynomolgus monkey (Macaca fascicularis). II. Efferent connections. J Comp Neurol. 1982;207(2):135–156. doi: 10.1002/cne.902070204. [DOI] [PubMed] [Google Scholar]
- 25.Holstege G. Subcortical limbic system projections to caudal brainstem and spinal Cord. In: Paxinos G, editor. The Human Nervous System. Academic; San Diego: 1990. pp. 261–286. [Google Scholar]
- 26.Barbas H, Saha S, Rempel-Clower N, Ghashghaei T. Serial pathways from primate prefrontal cortex to autonomic areas may influence emotional expression. BMC Neurosci. 2003;4:25. doi: 10.1186/1471-2202-4-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hatanaka N, Nambu A, Yamashita A, Takada M, Tokuno H. Somatotopic arrangement and corticocortical inputs of the hindlimb region of the primary motor cortex in the macaque monkey. Neurosci Res. 2001;40(1):9–22. doi: 10.1016/s0168-0102(01)00210-3. [DOI] [PubMed] [Google Scholar]
- 28.Innes KE, Bourguignon C, Taylor AG. Risk indices associated with the insulin resistance syndrome, cardiovascular disease, and possible protection with yoga: A systematic review. J Am Board Fam Pract. 2005;18(6):491–519. doi: 10.3122/jabfm.18.6.491. [DOI] [PubMed] [Google Scholar]
- 29.Bush G, Luu P, Posner MI. Cognitive and emotional influences in anterior cingulate cortex. Trends Cogn Sci. 2000;4(6):215–222. doi: 10.1016/s1364-6613(00)01483-2. [DOI] [PubMed] [Google Scholar]
- 30.Shima K, Tanji J. Role for cingulate motor area cells in voluntary movement selection based on reward. Science. 1998;282(5392):1335–1338. doi: 10.1126/science.282.5392.1335. [DOI] [PubMed] [Google Scholar]
- 31.Shackman AJ, et al. The integration of negative affect, pain and cognitive control in the cingulate cortex. Nat Rev Neurosci. 2011;12(3):154–167. doi: 10.1038/nrn2994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen C, et al. Anterior cingulate cortex cells identify process-specific errors of attentional control prior to transient prefrontal-cingulate inhibition. Cereb Cortex. 2015;25(8):2213–2228. doi: 10.1093/cercor/bhu028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hamani C, et al. The subcallosal cingulate gyrus in the context of major depression. Biol Psychiatry. 2011;69(4):301–308. doi: 10.1016/j.biopsych.2010.09.034. [DOI] [PubMed] [Google Scholar]
- 34.Eblen F, Graybiel AM. Highly restricted origin of prefrontal cortical inputs to striosomes in the macaque monkey. J Neurosci. 1995;15(9):5999–6013. doi: 10.1523/JNEUROSCI.15-09-05999.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Matsumoto K, Suzuki W, Tanaka K. Neuronal correlates of goal-based motor selection in the prefrontal cortex. Science. 2003;301(5630):229–232. doi: 10.1126/science.1084204. [DOI] [PubMed] [Google Scholar]
- 36.Kennerley SW, Walton ME, Behrens TE, Buckley MJ, Rushworth MF. Optimal decision making and the anterior cingulate cortex. Nat Neurosci. 2006;9(7):940–947. doi: 10.1038/nn1724. [DOI] [PubMed] [Google Scholar]
- 37.Amemori K, Graybiel AM. Localized microstimulation of primate pregenual cingulate cortex induces negative decision-making. Nat Neurosci. 2012;15(5):776–785. doi: 10.1038/nn.3088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kelly RM, Strick PL. Rabies as a transneuronal tracer of circuits in the central nervous system. J Neurosci Methods. 2000;103(1):63–71. doi: 10.1016/s0165-0270(00)00296-x. [DOI] [PubMed] [Google Scholar]
- 39.Kelly RM, Strick PL. Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. J Neurosci. 2003;23(23):8432–8444. doi: 10.1523/JNEUROSCI.23-23-08432.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ugolini G. Advances in viral transneuronal tracing. J Neurosci Methods. 2010;194(1):2–20. doi: 10.1016/j.jneumeth.2009.12.001. [DOI] [PubMed] [Google Scholar]
- 41. Centers for Disease Control and Prevention; National Institutes of Health (2009) Biosafety in Microbiological and Biological Laboratories, Department of Health and Human Services Publication No. 21-1112 (Centers for Disease Control and Prevention, Atlanta), 5th Ed.
- 42.Nadin-Davis SA, et al. A panel of monoclonal antibodies targeting the rabies virus phosphoprotein identifies a highly variable epitope of value for sensitive strain discrimination. J Clin Microbiol. 2000;38(4):1397–1403. doi: 10.1128/jcm.38.4.1397-1403.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Middleton FA, Strick PL. Cerebellar projections to the prefrontal cortex of the primate. J Neurosci. 2001;21(2):700–712. doi: 10.1523/JNEUROSCI.21-02-00700.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Carmichael ST, Price JL. Architectonic subdivision of the orbital and medial prefrontal cortex in the macaque monkey. J Comp Neurol. 1994;346(3):366–402. doi: 10.1002/cne.903460305. [DOI] [PubMed] [Google Scholar]
- 45.Beissner F, Deichmann R, Henke C, Bär KJ. Acupuncture--deep pain with an autonomic dimension? Neuroimage. 2012;60(1):653–660. doi: 10.1016/j.neuroimage.2011.12.045. [DOI] [PubMed] [Google Scholar]
- 46.Critchley HD, Elliott R, Mathias CJ, Dolan RJ. Neural activity relating to generation and representation of galvanic skin conductance responses: A functional magnetic resonance imaging study. J Neurosci. 2000;20(8):3033–3040. doi: 10.1523/JNEUROSCI.20-08-03033.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Critchley HD, Corfield DR, Chandler MP, Mathias CJ, Dolan RJ. Cerebral correlates of autonomic cardiovascular arousal: A functional neuroimaging investigation in humans. J Physiol. 2000;523(Pt 1):259–270. doi: 10.1111/j.1469-7793.2000.t01-1-00259.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Critchley HD, Melmed RN, Featherstone E, Mathias CJ, Dolan RJ. Volitional control of autonomic arousal: A functional magnetic resonance study. Neuroimage. 2002;16(4):909–919. doi: 10.1006/nimg.2002.1147. [DOI] [PubMed] [Google Scholar]
- 49.Critchley HD, Mathias CJ, Dolan RJ. Fear conditioning in humans: The influence of awareness and autonomic arousal on functional neuroanatomy. Neuron. 2002;33(4):653–663. doi: 10.1016/s0896-6273(02)00588-3. [DOI] [PubMed] [Google Scholar]
- 50.Critchley HD, et al. Human cingulate cortex and autonomic control: Converging neuroimaging and clinical evidence. Brain. 2003;126(Pt 10):2139–2152. doi: 10.1093/brain/awg216. [DOI] [PubMed] [Google Scholar]
- 51.Critchley HD, Tang J, Glaser D, Butterworth B, Dolan RJ. Anterior cingulate activity during error and autonomic response. Neuroimage. 2005;27(4):885–895. doi: 10.1016/j.neuroimage.2005.05.047. [DOI] [PubMed] [Google Scholar]
- 52.Dubé AA, et al. Brain activity associated with the electrodermal reactivity to acute heat pain. Neuroimage. 2009;45(1):169–180. doi: 10.1016/j.neuroimage.2008.10.024. [DOI] [PubMed] [Google Scholar]
- 53.Fan J, et al. Spontaneous brain activity relates to autonomic arousal. J Neurosci. 2012;32(33):11176–11186. doi: 10.1523/JNEUROSCI.1172-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Farrow TF, et al. Neural correlates of the behavioral-autonomic interaction response to potentially threatening stimuli. Front Hum Neurosci. 2013;6:349. doi: 10.3389/fnhum.2012.00349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Fechir M, et al. Functional imaging of sympathetic activation during mental stress. Neuroimage. 2010;50(2):847–854. doi: 10.1016/j.neuroimage.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 56.Gianaros PJ, Van Der Veen FM, Jennings JR. Regional cerebral blood flow correlates with heart period and high-frequency heart period variability during working-memory tasks: Implications for the cortical and subcortical regulation of cardiac autonomic activity. Psychophysiology. 2004;41(4):521–530. doi: 10.1111/1469-8986.2004.00179.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gianaros PJ, et al. Anterior cingulate activity correlates with blood pressure during stress. Psychophysiology. 2005;42(6):627–635. doi: 10.1111/j.1469-8986.2005.00366.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gianaros PJ, Onyewuenyi IC, Sheu LK, Christie IC, Critchley HD. Brain systems for baroreflex suppression during stress in humans. Hum Brain Mapp. 2012;33(7):1700–1716. doi: 10.1002/hbm.21315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Goswami R, Frances MF, Steinback CD, Shoemaker JK. Forebrain organization representing baroreceptor gating of somatosensory afferents within the cortical autonomic network. J Neurophysiol. 2012;108(2):453–466. doi: 10.1152/jn.00764.2011. [DOI] [PubMed] [Google Scholar]
- 60.Guillot A, et al. Functional neuroanatomical networks associated with expertise in motor imagery. Neuroimage. 2008;41(4):1471–1483. doi: 10.1016/j.neuroimage.2008.03.042. [DOI] [PubMed] [Google Scholar]
- 61.Harrison NA, Gray MA, Gianaros PJ, Critchley HD. The embodiment of emotional feelings in the brain. J Neurosci. 2010;30(38):12878–12884. doi: 10.1523/JNEUROSCI.1725-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kimmerly DS, Wong SW, Salzer D, Menon R, Shoemaker JK. Forebrain regions associated with postexercise differences in autonomic and cardiovascular function during baroreceptor unloading. Am J Physiol Heart Circ Physiol. 2007;293(1):H299–H306. doi: 10.1152/ajpheart.00044.2007. [DOI] [PubMed] [Google Scholar]
- 63.Knight DC, Nguyen HT, Bandettini PA. The role of the human amygdala in the production of conditioned fear responses. Neuroimage. 2005;26(4):1193–1200. doi: 10.1016/j.neuroimage.2005.03.020. [DOI] [PubMed] [Google Scholar]
- 64.LaBar KS, Gatenby JC, Gore JC, LeDoux JE, Phelps EA. Human amygdala activation during conditioned fear acquisition and extinction: A mixed-trial fMRI study. Neuron. 1998;20(5):937–945. doi: 10.1016/s0896-6273(00)80475-4. [DOI] [PubMed] [Google Scholar]
- 65.Lemche E, et al. Human attachment security is mediated by the amygdala: Evidence from combined fMRI and psychophysiological measures. Hum Brain Mapp. 2006;27(8):623–635. doi: 10.1002/hbm.20206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Maihöfner C, Seifert F, Decol R. Activation of central sympathetic networks during innocuous and noxious somatosensory stimulation. Neuroimage. 2011;55(1):216–224. doi: 10.1016/j.neuroimage.2010.11.061. [DOI] [PubMed] [Google Scholar]
- 67.Nagai Y, Critchley HD, Featherstone E, Trimble MR, Dolan RJ. Activity in ventromedial prefrontal cortex covaries with sympathetic skin conductance level: A physiological account of a “default mode” of brain function. Neuroimage. 2004;22(1):243–251. doi: 10.1016/j.neuroimage.2004.01.019. [DOI] [PubMed] [Google Scholar]
- 68.Nagai Y, et al. Brain activity relating to the contingent negative variation: An fMRI investigation. Neuroimage. 2004;21(4):1232–1241. doi: 10.1016/j.neuroimage.2003.10.036. [DOI] [PubMed] [Google Scholar]
- 69.Nowak M, et al. Command-related distribution of regional cerebral blood flow during attempted handgrip. J Appl Physiol (1985) 1999;86(3):819–824. doi: 10.1152/jappl.1999.86.3.819. [DOI] [PubMed] [Google Scholar]
- 70.Ohira H, et al. Association of neural and physiological responses during voluntary emotion suppression. Neuroimage. 2006;29(3):721–733. doi: 10.1016/j.neuroimage.2005.08.047. [DOI] [PubMed] [Google Scholar]
- 71.Ohira H, et al. Imaging brain and immune association accompanying cognitive appraisal of an acute stressor. Neuroimage. 2008;39(1):500–514. doi: 10.1016/j.neuroimage.2007.08.017. [DOI] [PubMed] [Google Scholar]
- 72.Piché M, Arsenault M, Rainville P. Dissection of perceptual, motor and autonomic components of brain activity evoked by noxious stimulation. Pain. 2010;149(3):453–462. doi: 10.1016/j.pain.2010.01.005. [DOI] [PubMed] [Google Scholar]
- 73.Qin S, Hermans EJ, van Marle HJ, Fernández G. Understanding low reliability of memories for neutral information encoded under stress: Alterations in memory-related activation in the hippocampus and midbrain. J Neurosci. 2012;32(12):4032–4041. doi: 10.1523/JNEUROSCI.3101-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Reinhardt I, et al. Neural correlates of aversive conditioning: Development of a functional imaging paradigm for the investigation of anxiety disorders. Eur Arch Psychiatry Clin Neurosci. 2010;260(6):443–453. doi: 10.1007/s00406-010-0099-9. [DOI] [PubMed] [Google Scholar]
- 75.Rubio A, et al. Uncertainty in anticipation of uncomfortable rectal distension is modulated by the autonomic nervous system--a fMRI study in healthy volunteers. Neuroimage. 2015;107:10–22. doi: 10.1016/j.neuroimage.2014.11.043. [DOI] [PubMed] [Google Scholar]
- 76.Seifert F, et al. Brain activity during sympathetic response in anticipation and experience of pain. Hum Brain Mapp. 2013;34(8):1768–1782. doi: 10.1002/hbm.22035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Teves D, Videen TO, Cryer PE, Powers WJ. Activation of human medial prefrontal cortex during autonomic responses to hypoglycemia. Proc Natl Acad Sci USA. 2004;101(16):6217–6221. doi: 10.1073/pnas.0307048101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wager TD, et al. Brain mediators of cardiovascular responses to social threat: Part I: Reciprocal dorsal and ventral sub-regions of the medial prefrontal cortex and heart-rate reactivity. Neuroimage. 2009;47(3):821–835. doi: 10.1016/j.neuroimage.2009.05.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wager TD, et al. Brain mediators of cardiovascular responses to social threat, part II: Prefrontal-subcortical pathways and relationship with anxiety. Neuroimage. 2009;47(3):836–851. doi: 10.1016/j.neuroimage.2009.05.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Williams LM, et al. BOLD, sweat and fears: fMRI and skin conductance distinguish facial fear signals. Neuroreport. 2005;16(1):49–52. doi: 10.1097/00001756-200501190-00012. [DOI] [PubMed] [Google Scholar]
- 81.Hölzel BK, et al. Differential engagement of anterior cingulate and adjacent medial frontal cortex in adept meditators and non-meditators. Neurosci Lett. 2007;421(1):16–21. doi: 10.1016/j.neulet.2007.04.074. [DOI] [PubMed] [Google Scholar]
- 82.Kilpatrick LA, et al. Impact of Mindfulness-Based Stress Reduction training on intrinsic brain connectivity. Neuroimage. 2011;56(1):290–298. doi: 10.1016/j.neuroimage.2011.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lazar SW, et al. Functional brain mapping of the relaxation response and meditation. Neuroreport. 2000;11(7):1581–1585. [PubMed] [Google Scholar]
- 84.Wang DJ, et al. Cerebral blood flow changes associated with different meditation practices and perceived depth of meditation. Psychiatry Res. 2011;191(1):60–67. doi: 10.1016/j.pscychresns.2010.09.011. [DOI] [PubMed] [Google Scholar]