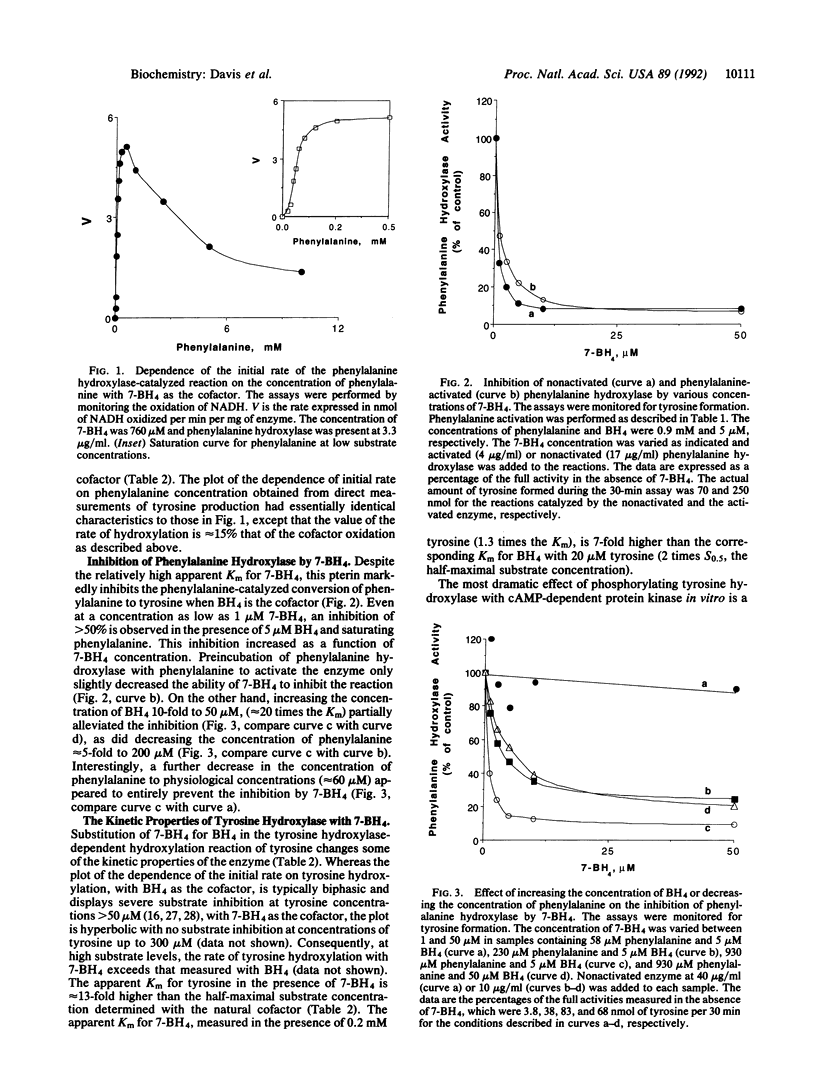
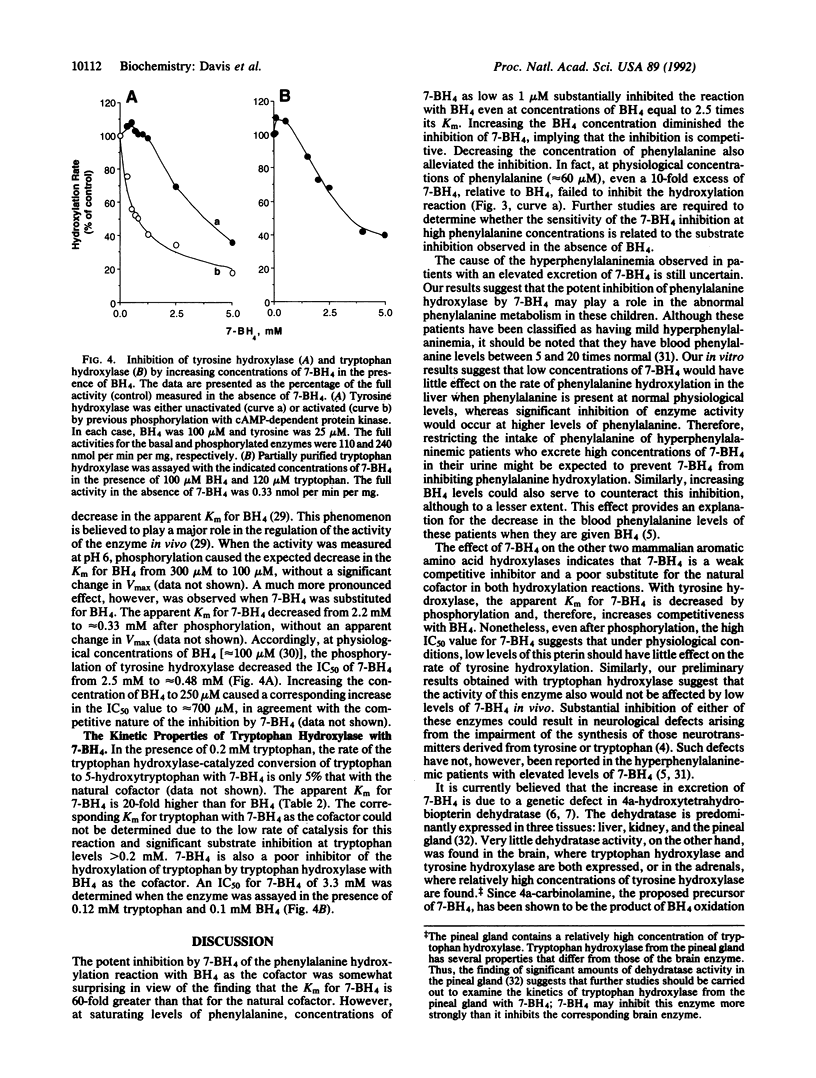
Abstract
The ability of 2-amino-4-hydroxy-7-[dihydroxylpropyl-(L-erythro)-5,6,7,8-tetrahyd ropterin] ("7-tetrahydrobiopterin" or 7-BH4) to substitute for the natural cofactor tetrahydrobiopterin (BH4) has been studied in vitro in the reactions of the three mammalian aromatic amino acid hydroxylases. With rat liver phenylalanine hydroxylase, the apparent Km for 7-BH4 is 160 microM, a value that is approximately 60-fold greater than that for the natural cofactor. In contrast, the hydroxylase reaction is severely inhibited by as little as 1 microM 7-BH4 when assayed in the presence of physiological concentrations of BH4. This inhibition can be overcome either by an increase in the concentration of BH4 or a decrease in the concentration of phenylalanine. With both rat brain tryptophan hydroxylase and rat pheochromocytoma tyrosine hydroxylase, the Km value for 7-BH4 is about one order of magnitude greater than the Km for BH4. Accordingly, 7-BH4 is a poor competitive inhibitor of both tryptophan and tyrosine hydroxylase. Thus, our results suggest that the observed hyperphenylalaninemia in patients who excrete 7-BH4 in their urine may arise directly from the inhibition of phenylalanine hydroxylase by low levels of this pterin. On the other hand, it is less likely that low levels of 7-BH4 would affect the activity of tyrosine or tryptophan hydroxylase in vivo.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blau N., Curtius H. C., Kuster T., Matasovic A., Schoedon G., Dhondt J. L., Guibaud P., Giudici T., Blaskovics M. Primapterinuria: a new variant of atypical phenylketonuria. J Inherit Metab Dis. 1989;12 (Suppl 2):335–338. doi: 10.1007/BF03335415. [DOI] [PubMed] [Google Scholar]
- Craine J. E., Hall E. S., Kaufman S. The isolation and characterization of dihydropteridine reductase from sheep liver. J Biol Chem. 1972 Oct 10;247(19):6082–6091. [PubMed] [Google Scholar]
- Curtius H. C., Adler C., Rebrin I., Heizmann C., Ghisla S. 7-Substituted pterins: formation during phenylalanine hydroxylation in the absence of dehydratase. Biochem Biophys Res Commun. 1990 Nov 15;172(3):1060–1066. doi: 10.1016/0006-291x(90)91554-6. [DOI] [PubMed] [Google Scholar]
- Davis M. D., Kaufman S. 7-Tetrahydrobiopterin is an uncoupled cofactor for rat hepatic phenylalanine hydroxylase. FEBS Lett. 1991 Jul 8;285(1):17–20. doi: 10.1016/0014-5793(91)80714-e. [DOI] [PubMed] [Google Scholar]
- Davis M. D., Kaufman S., Milstien S. Conversion of 6-substituted tetrahydropterins to 7-isomers via phenylalanine hydroxylase-generated intermediates. Proc Natl Acad Sci U S A. 1991 Jan 15;88(2):385–389. doi: 10.1073/pnas.88.2.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis M. D., Kaufman S., Milstien S. Distribution of 4a-hydroxytetrahydropterin dehydratase in rat tissues. Comparison with the aromatic amino acid hydroxylases. FEBS Lett. 1992 May 4;302(1):73–76. doi: 10.1016/0014-5793(92)80288-r. [DOI] [PubMed] [Google Scholar]
- Dix T. A., Kuhn D. M., Benkovic S. J. Mechanism of oxygen activation by tyrosine hydroxylase. Biochemistry. 1987 Jun 16;26(12):3354–3361. doi: 10.1021/bi00386a016. [DOI] [PubMed] [Google Scholar]
- Fisher D. B., Kaufman S. The stimulation of rat liver phenylalanine hydroxylase by lysolecithin and -chymotrypsin. J Biol Chem. 1973 Jun 25;248(12):4345–4353. [PubMed] [Google Scholar]
- Friedman P. A., Kappelman A. H., Kaufman S. Partial purification and characterization of tryptophan hydroxylase from rabbit hindbrain. J Biol Chem. 1972 Jul 10;247(13):4165–4173. [PubMed] [Google Scholar]
- Haavik J., Flatmark T. Isolation and characterization of tetrahydropterin oxidation products generated in the tyrosine 3-monooxygenase (tyrosine hydroxylase) reaction. Eur J Biochem. 1987 Oct 1;168(1):21–26. doi: 10.1111/j.1432-1033.1987.tb13381.x. [DOI] [PubMed] [Google Scholar]
- Huang C. Y., Kaufman S. Studies on the mechanisms of action of phenylalanine hydroxylase and its protein stimulator. I. Enzyme concentration dependence of the specific activity of phenylalanine hydroxylase due to a nonenzymatic step. J Biol Chem. 1973 Jun 25;248(12):4242–4251. [PubMed] [Google Scholar]
- JERVIS G. A. Phenylpyruvic oligophrenia deficiency of phenylalanine-oxidizing system. Proc Soc Exp Biol Med. 1953 Mar;82(3):514–515. [PubMed] [Google Scholar]
- KAUFMAN S. Phenylalanine hydroxylation cofactor in phenylketonuria. Science. 1958 Dec 12;128(3337):1506–1508. doi: 10.1126/science.128.3337.1506. [DOI] [PubMed] [Google Scholar]
- KAUFMAN S. Studies on the mechanism of the enzymatic conversion of phenylalanine to tyrosine. J Biol Chem. 1959 Oct;234:2677–2682. [PubMed] [Google Scholar]
- KAUFMAN S. THE STRUCTURE OF THE PHENYLALANINE-HYDROXYLATION COFACTOR. Proc Natl Acad Sci U S A. 1963 Dec;50:1085–1093. doi: 10.1073/pnas.50.6.1085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufman S. A protein that stimulates rat liver phenylalanine hydroxylase. J Biol Chem. 1970 Sep 25;245(18):4751–4759. [PubMed] [Google Scholar]
- Kaufman S. Phenylalanine 4-monooxygenase from rat liver. Methods Enzymol. 1987;142:3–17. doi: 10.1016/s0076-6879(87)42003-x. [DOI] [PubMed] [Google Scholar]
- Kaufman S. Phenylketonuria and its variants. Adv Hum Genet. 1983;13:217–297. doi: 10.1007/978-1-4615-8342-4_5. [DOI] [PubMed] [Google Scholar]
- Levine R. A., Miller L. P., Lovenberg W. Tetrahydrobiopterin in striatum: localization in dopamine nerve terminals and role in catecholamine synthesis. Science. 1981 Nov 20;214(4523):919–921. doi: 10.1126/science.6117945. [DOI] [PubMed] [Google Scholar]
- Lloyd T., Kaufman S. The stimulation of partially purified bovine caudate tyrosine hydroxylase by phosphatidyl-L-serine. Biochem Biophys Res Commun. 1974 Aug 19;59(4):1262–1270. doi: 10.1016/0006-291x(74)90450-1. [DOI] [PubMed] [Google Scholar]
- NAGATSU T., LEVITT M., UDENFRIEND S. A RAPID AND SIMPLE RADIOASSAY FOR TYROSINE HYDROXYLASE ACTIVITY. Anal Biochem. 1964 Sep;9:122–126. doi: 10.1016/0003-2697(64)90092-2. [DOI] [PubMed] [Google Scholar]
- Ribeiro P., Pigeon D., Kaufman S. The hydroxylation of phenylalanine and tyrosine by tyrosine hydroxylase from cultured pheochromocytoma cells. J Biol Chem. 1991 Aug 25;266(24):16207–16211. [PubMed] [Google Scholar]
- Shiman R., Akino M., Kaufman S. Solubilization and partial purification of tyrosine hydroxylase from bovine adrenal medulla. J Biol Chem. 1971 Mar 10;246(5):1330–1340. [PubMed] [Google Scholar]
- Shiman R., Gray D. W. Substrate activation of phenylalanine hydroxylase. A kinetic characterization. J Biol Chem. 1980 May 25;255(10):4793–4800. [PubMed] [Google Scholar]
- UDENFRIEND S., COOPER J. R. The chemical estimation of tyrosine and tyramine. J Biol Chem. 1952 May;196(1):227–233. [PubMed] [Google Scholar]
- Wang Y. H., Citron B. A., Ribeiro P., Kaufman S. High-level expression of rat PC12 tyrosine hydroxylase cDNA in Escherichia coli: purification and characterization of the cloned enzyme. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8779–8783. doi: 10.1073/pnas.88.19.8779. [DOI] [PMC free article] [PubMed] [Google Scholar]


