Abstract
The role of host genetic variation in pneumonia development and outcome is poorly understood. We studied common polymorphisms in the genes of proinflammatory cytokines (IL6 rs1800795, IL8 rs4073, IL1B rs16944), anti-inflammatory cytokines (IL10 rs1800896, IL4 rs2243250, IL13 rs20541) and toll-like receptors (TLR2 rs5743708 and rs4696480, TLR4 rs4986791, TLR9 rs352139, rs5743836 and rs187084) in patients with community-acquired pneumonia (CAP) (390 cases, 203 controls) and nosocomial pneumonia (355 cases, 216 controls). Experimental data were included in a series of 11 meta-analyses and eight subset analyses related to pneumonia susceptibility and outcome. TLR2 rs5743708 minor genotype appeared to be associated with CAP/Legionnaires’ disease/pneumococcal disease. In CAP patients, the IL6 rs1800795-C allele was associated with severe sepsis/septic shock/severe systemic inflammatory response, while the IL10 rs1800896-A allele protected against the development of these critical conditions. To contribute to deciphering of the above results, we performed an in silico analysis and a qualitative synthesis of literature data addressing basal and stimulated genotype-specific expression level. This data together with database information on transcription factors’ affinity changes caused by SNPs in putative promoter regions, the results of linkage disequilibrium analysis along with SNPs functional annotations supported assumptions about the complexity underlying the revealed associations.
Pneumonia, one of the most common infectious diseases, is associated with high morbidity and mortality worldwide. Pneumonia is classified according to the site of acquisition as community-acquired (CAP) and hospital-acquired (or nosocomial) pneumonia (HAP or NP), which includes healthcare-associated pneumonia (HCAP) and ventilator-associated pneumonia (VAP). HAP tends to be a more serious problem than CAP since HAP patients have an increased risk of infection caused by antibiotic-resistant organisms, besides they are often older and burdened by comorbidities. Pneumonia is considered to be the the most prevalent cause of sepsis associated with the highest mortality rates1.
In infection, host genetic background may be of even more importance than in cancer and cardiovascular diseases which are known to have a high hereditary component2. Most infectious phenotypes result from a complex interplay between multiple genetic factors related to host and pathogen genetics and a variety of non-genetic factors. Since infectious diseases are related to immune-mediated disorders, genetic susceptibility loci for immune traits can be considered as good candidates in genetic association studies of pneumonia and its complications3. The first line of defence of the innate immune system is represented by pattern-recognition receptors that sense bacterial, viral or fungal molecular structures and induce systemic inflammation. The most studied class of pattern-recognition receptors is that of the toll-like receptors (TLRs). Once inflammation has been launched a crosstalk between pro-and anti-inflammatory branches of the immune response should provide a balanced immune reaction crucial for a favorable resolution of infection.
Genetic studies of pneumonia are not numerous while investigations assessing the role of host genetics in sepsis due to pneumonia are even more unfrequent. Furthermore, to date there are no GWAS reported for pneumonia. Meta-analyses of genetic susceptibility to pneumonia provided very limited coverage of common genetic variations being performed for the genes of TNFa4, MBL25, IL66,7, IL107, and FCGR2A6 with no association effect and for the loci ACE8 and TLR49 found to be associated with pneumonia. Several studies showed that genetic associations of sepsis in pneumonia differ from those in other infections10,11,12 and may in particular depend on a specific pathogen13.
Taking into account this scientific background the aim of our study was: (i) to investigate the role of the SNPs in the genes of proinflammatory cytokines (IL6, IL1B, IL8), antiinflammatory cytokines (IL10, IL4, IL13) and toll-like receptors TLR2, TLR4 and TLR9 in CAP and HAP patients, (ii) to combine existent knowledge and our own experimental data on the above genes in a series of meta-analyses of genetic susceptibility to pneumonia development and outcome. The relationship between host genetics and clinical phenotypes is mediated through a large number of factors and interactions, which are essentially a “black box”. To contribute to deciphering of this “black box” in terms of the studied genes several additional investigations were performed. We combined an in silico analysis and literature data analysis to evaluate the genotype-dependent basal and ex-vivo stimulated expression level. Since the majority of common SNPs in the immune genes are located in promoter regions and thus may influence expression level, we provided an in silico analysis of allele-specific binding affinity of TFs. With the use of database information, we also explored linkage disequilibrium structure along with functional annotations of the SNPs within the genes under study.
Results
Genetic association studies of CAP and NP
Data with characteristics of the study populations are given in Supplementary Table S1. In total, the CAP study included 593 subjects, while the NP study comprised 571 patients at high risk of pneumonia development. In both studies, patients and controls were matched by age, sex and ethnicity. Genotyping data are presented in Supplementary Table S2. SNP frequencies were in Hardy–Weinberg equilibrium (HWE) in both control groups with an exception for TLR4 rs4986791 (CAP controls). Several associations revealed in CAP and NP studies became non-significant after correction for multiple comparisons. However one observation is noteworthy. In the recent meta-analysis6 assessing the role of host genetic variations in susceptibility to several respiratory infectious diseases (tuberculosis, influenza, respiratory syncytial virus, SARS-Coronavirus and pneumonia), the IL4 gene allele 2070874-T was the only variant associated with respiratory infections in the pooled group (tuberculosis and respiratory syncytial virus). The IL4 rs2243250-T allele which is in complete linkage disequilibrium with the rs2070874-T allele (r2 = 1.0) was a susceptibility allele for NP in our study (Supplementary Table S2).
Meta-analysis
Studies Characteristics
A total of 346 studies were identified by searching PubMed, Embase and Web of Knowledge resources (Supplementary Figure S1). The characteristics of the included studies are given in Supplementary Table S3. Ten studies were about associations of the selected genes with CAP, six papers were about HAP, five investigations considered pneumococcal disease, two studies were on Legionnaires’ disease and the rest one dealt with an unspecified type of pneumonia. HAP studies comprised the most heterogeneous group of observations. Sixteen studies indicated infectious pathogens (Supplementary Table S4), among them, five researches considered viral pneumonia. Deviations for Hardy-Weinberg equilibrium in controls were reveled mainly in the studies of the IL10 SNPs (two works for each of the three considered polymorphic sites). Eighteen studies were performed in European population, two studies were conducted in Asian population, and three papers analyzed mixed population. All but four investigations were implemented in adult populations. Addressing pneumonia critical complications, the only available type of similar data in different studies was revealed for severe sepsis/septic shock in CAP/pneumococcal disease. In the analysis of this type of poor outcome, we also included our own results and data from the study of systemic inflammatory response syndrome (SIRS) in CAP patients (SIRS IV vs. SIRS I+II)14.
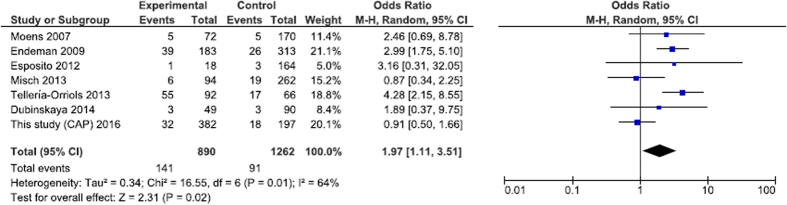
Quantitative data synthesis
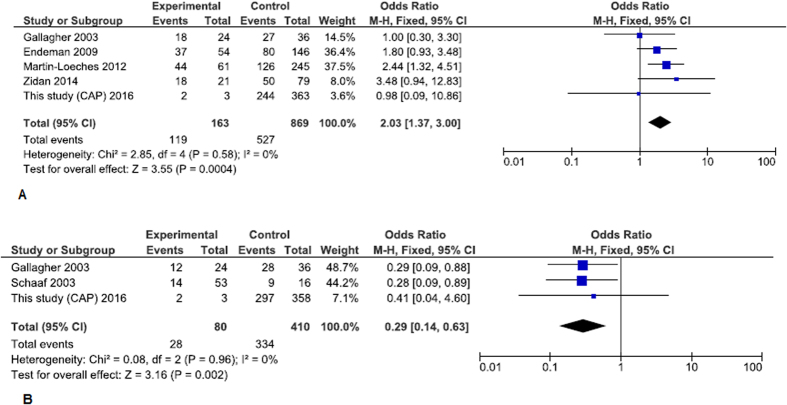
We performed a total of eleven pneumonia susceptibility meta-analyses in four genetic models (Table 1) with no significant results for the whole data sets. A subset analysis after removing studies that were not consistent with HWE equation revealed a borderline significance for the association of the IL10 rs1800896 GA/AA genotype with susceptibility to pneumonia/pneumococcal disease (Table 1). Further meta-analyses were stratified by pneumonia type and ethnicity. In the article of Yuan et al.15 there were no data on ethnicity and a portion of patients with pneumonia in pneumococcal disease. After excluding the study of Yang et al.15 and three HAP studies, TLR2 rs5743708 minor genotype appeared to be associated with CAP/Legionnaires’ disease/pneumococcal disease (Fig. 1). Sensitivity analysis showed that these results were not stable (Supplementary Table S5). An analysis for severe sepsis/septic shock/SIRS in CAP/pneumococcal disease was available only for the SNPs IL6 rs1800795 and IL10 rs1800896 with both series providing significant results (Fig. 2). The so-called “low expression” allele -174C (rs1800795) of pro-inflammatory cytokine IL6 was more frequent in patients with poor outcome (dominant model), while the so-called “low expression” allele -1082A (rs1800896) of anti-inflammatory cytokine IL10 protected against the development of severe critical conditions (dominant model).
Table 1. Results of meta-analyses.
| Gene, SNP | Disease | Studies (n) | Cases/Controls (n) | Genetic model1 | I2 (%) | OR [95% CI] | P |
|---|---|---|---|---|---|---|---|
| Il6 rs1800795 | CAP/HAP/pneumococcal disease | 7 | 2425/1879 | Dom | 6 | 0.92 [0.81–1.04] | 0.20 |
| Subset (CAP) | 5 | 1981/1623 | Dom | 2 | 0.88 [0.77–1.01] | 0.08 | |
| Subset (Pneumococcal CAP/pneumococcal disease) | 4 | 1122/3034 | Add | 94 | 1.44 [0.71–2.95] | 0.32 | |
| IL1B rs16944 | CAP/HAP | 4 | 797/558 | Dom | 35 | 0.88 [0.70–1.11] | 0.30 |
| IL8 rs4073 | CAP/HAP | 4 | 2078/1654 | Add | 88 | 1.29 [0.87–1.90] | 0.21 |
| Severe sepsis/septic shock in CAP | 3 | 222/1444 | Add | 3 | 1.15 [0.82–1.62] | 0.41 | |
| IL10 rs1800896 | CAP/HAP/pneumococcal disease | 6 | 1167/885 | Rec | 72 | 1.41 [0.89–2.22] | 0.14 |
| Subset (CAP) | 4 | 752/625 | Rec | 82 | 1.68 [0.81–3.49] | 0.16 | |
| Subset (controls are in HWE) | 4 | 1033/789 | Rec | 0 | 1.26 [1.00–1.59] | 0.05 | |
| Il10 rs1800871 | CAP/Postoperative pneumonia in patients with esophageal cancer | 3 | 188/194 | Dom | 84 | 1.44 [0.38–5.41] | 0.59 |
| IL10 rs1800872 | CAP/Pneumonia complicated with sepsis/Pneumonia in kidney transplant recipients | 5 | 566/884 | Dom | 80 | 0.63 [0.32–1.24] | 0.18 |
| Subset (controls are in HWE) | 3 | 686/892 | Add | 8 | 0.86 [0.68–1.09] | 0.21 | |
| TLR2 rs5743708 | CAP/Legionnaires’ disease/pneumococcal disease/HAP/pneumonia after allogeneic hematopoietic stem cell transplantation/pneumonia in AML patients after induction chemotherapy | 11 | 1460/2119 | Dom | 71 | 1.60 [0.93–2.75] | 0.09 |
| TLR4 rs4986790 | CAP/Legionnaires’ disease/pneumococcal disease/HAP/pneumonia after allogeneic hematopoietic stem cell transplantation/pneumonia in AML patients after induction chemotherapy | 9 | 1149/1867 | Dom | 63 | 1.18 [0.75–1.86] | 0.48 |
| Subset (CAP/Legionnaires’ disease/pneumococcal diseasea) | 6 | 926/913 | Dom | 52 | 1.59 [0.94–2.69] | 0.09 | |
| TLR4 rs4986791 | CAP/Legionnaires’ disease/pneumococcal disease/HAP/VAP/pneumonia in AML patients after induction chemotherapy | 7 | 1523/1355 | Dom | 73 | 0.90 [0.50–1.62] | 0.73 |
| Subset (CAP/Legionnaires’ disease/pneumococcal disease) | 3 | 885/457 | Dom | 59 | 1.31 [0.54–3.18] | 0.56 | |
| TLR9 rs187084 | CAP/HAP/pneumonia after allogeneic hematopoietic stem cell transplantation | 3 | 798/536 | Dom | 45 | 0.89 [0.70–1.14] | 0.35 |
| TLR9 rs5743836 | CAP/HAP/pneumonia after allogeneic hematopoietic stem cell transplantation | 3 | 809/537 | Dom | 56 | 1.21 [0.81–1.83] | 0.35 |
Genetic models: Add, additive model (major allele versus minor allele); Dom, dominant model (heterozygote-minor homozygote versus major homozygote); Rec, recessive model (minor homozygote versus major homozygote- heterozygote). Significant results are in bold. Abbreviations: AML, acute myeloid leukemia; CAP, community acquired pneumonia; HAP, hospital acquired pneumonia; VAP, ventilator associated pneumonia.
aPneumococcal disease studies with the majority of patients diagnosed with pneumonia are considered.
Figure 1. Forest plot for stratified analysis of the association between the TLR2 (rs5743708) polymorphism and CAP/Legionnaires’ disease/pneumococcal disease (dominant model).
Figure 2.
(A) Forest plot for stratified analysis of the association between the IL6 (rs1800795) polymorphism and severe sepsis/septic shock/severe SIRS in CAP patients (dominant model); (B) Forest plot for stratified analysis of the association between the IL10 (rs1800896) polymorphism and severe sepsis/septic shock/severe SIRS in patients with CAP/pneumococcal disease (dominant model). CAP, community-acquired pneumonia; SIRS, Systemic inflammatory response (score IV vs. score I+II).
Correlation between SNP genotypes and gene expression
Classification of cytokine genotypes as high or low cytokine producers is often based on a relatively small number of initial publications. These papers primarily presented expression data in combination with genotyping data in healthy controls, various patient groups and under different stimulation protocols16. Over time, the results of these investigations were not always replicated; moreover some papers declared an opposite direction of association.
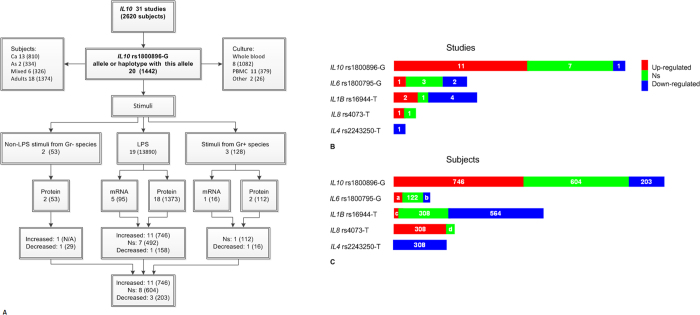
In this part of our investigation, we present an overview of studies comparing induced allele-specific cytokine expression profile. A total of 3612 studies were found on this theme by searching PubMed, Embase and Web of Knowledge resources (Supplementary Figure S2). It was assumed that (i) different stimuli may be more or less specialized in their allele-specific influence on cytokine production; (ii) patients with different diseases may have pre-existing conditions that multi-directionally affect cytokine level. Taking into account these considerations, we limited a spectrum of analyzed studies by the use of pneumonia related pathogen stimuli and considered samples only from healthy volunteers. Additional treatment, if applied, was also indicated to compare results for different stimuli in the same studies. Data from genetic association studies of interleukin genes polymorphisms and ex vivo response to bacterial toxins are collected in Supplementary Table S6. Studies differed by population ethnicity, polymorphic sites, cellular sources, and in vitro incubation protocols. Several studies measured mRNA, but the majority assessed protein secretion. Due to high variability, we could not perform quantitative data synthesis and present here the results of qualitative analysis. Summary for IL10 investigations is given in a block diagram (Fig. 3a). The majority of studies have measured lipopolysaccharides (LPS)-stimulated IL10 production in association with IL10 -1082G/A (rs1800896) genotype or haplotypes including -1082 alleles and testified that IL10 -1082G allele may be a high producing allele. Since LPS is released from Gram-negative bacteria, the results should be extrapolated on Gram-positive species (e.g. Streptococcus pneumonia, Staphylococcus aureus) with caution. Proportions of data with genotype-linked LPS-induced up-regulation, down-regulation or non-significant associations for all studied cytokines are shown in Fig. 3b,c. Proportions are given for the number of studies (Fig. 3b) and for the number of subjects in these studies (Fig. 3c). Data for cytokines other than IL10 could not be interpreted due to a small number of studies/subjects and contradictory associations. Similar analysis was not performed for toll-like receptor genes because genetic variations in these genes were mainly studied in association with cytokine expression.
Figure 3. Summary of literature data on correlation between cytokine genotypes and ex-vivo stimulated expression level.
(A) Block diagram for IL10 investigations; (B) Proportions of studies that have found genotype-linked LPS-induced up-regulation, down-regulation or non-significant associations. (C) Total number of subjects in the above studies.
Further we performed an in silico analysis of correlation between interleukin and toll-like receptor genotypes and basal gene expression levels utilizing SNPexp resource17. We constructed haplotypes with the HaploView software and analyzed association between check SNPs in haplotype blocks and mRNA expression levels in lymphoblastoid cell lines in the Utah HapMap population (CEU). Summary data are provided in Fig. 4. Weak association trends (0.05 < P < 0.10 for at least one SNP in the gene) were seen for IL1B, IL4, IL13 and TLR4 genes (Supplementary Table S7). Significant but also weak associations (P - value in the range 0.02–0.045) were revealed for IL6 and IL10 SNPs (Supplementary Table S7). These weak correlations cannot substantially contribute to differences in genotype-dependent expression of cytokines in acute infection, since cytokine production in these conditions is described in terms of fold-change.
Figure 4. SNPexp data for correlation between SNP-based haplotype markers of the studied genes and mRNA expression level under basal conditions.
Data were obtained from the HapMap phase II release 23 data for EBV-transformed lymphoblastoid cell lines of 60 CEU (Utah HapMap population) parents. Only haplotypes with frequencies more than 3% were included. Differences in gene expression in at least one from a series of available transcripts were considered. SNPs under study are in bold. Small letters for nucleotides correspond to a decreased expression level, while large letters represent an increased expression level. Dark blue color indicates a tendency level of association (P-value in a range 0.05–0.10); red color shows associations with P < 0.05 under additive model. The TLR2 and IL1B genes include two linkage blocks, while other genes consist of one linkage block. Hedrick’s multiallelic D′, which represents the degree of LD between the two blocks when treating each haplotype within a block as an “allele” of that region are shown for the TLR2 and IL1B genes. No haplotype markers were identified in the IL8 gene.
SNPs influence on transcription factors’ binding affinity
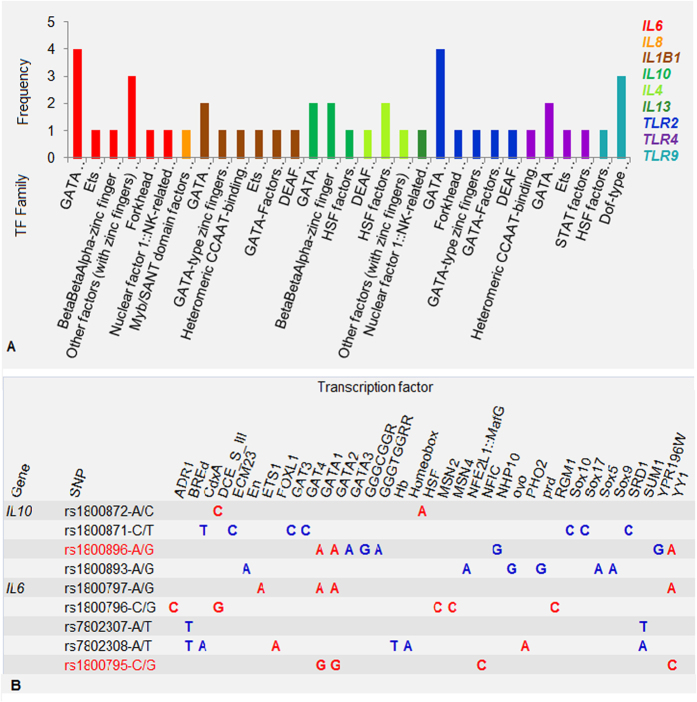
One of the main controlled stages of gene expression is regulation of transcription, which largely depends on recruitment of transcription factors (TFs). Binding affinity of TFs may be affected by SNPs that fall in transcription factor binding sites. In this part of our study, we assessed an influence of different alleles within putative promoter regions of the studied genes on binding affinity of TFs. Twenty seven SNPs including studied SNPs (IL10 rs1800896, IL6 rs1800795, IL1B rs16944, IL8 rs4073, IL4 rs2243250, TLR9 rs5743836 and rs187084) were identified in the putative promoter regions of the studied genes (2 kb upstream of a transcription start site). An in silico analysis of transcription factor DNA-binding specificity for two alleles at each SNP was conducted with RegSNP resource (http://viis.abdn.ac.uk/regsnp/Home.aspx) (Supplementary Table S8). Differences in allele-specific transcription factor DNA-binding ability in the whole set of twenty seven SNPs were predicted for in total 94 TFs. By gene distribution of TFs grouped into families (with the stringent cut-off level 0.20 for the minimum score difference) is shown in Fig. 5a.
Figure 5. RegSNP results for transcription factor DNA-binding specificity for two alleles at each SNP in putative promoter regions of the studied genes.
(A) By gene distribution of TFs grouped into families. The y-axis shows the frequency of SNPs influencing specific transcription factor DNA-binding ability of TF family members. The x-axis presents TF families. (B) An influence of different alleles within putative promoter regions of the IL6 and IL10 genes on binding affinity of TFs. SNPs under study are signed in red. Dark blue color indicates the minimum score difference cut-off in the range of 0.10 to 0.20; red color marks the minimum score difference cut-off >0.20.
In more detail, we present the analysis of the SNPs within putative promoter regions of the IL6 and IL10 genes (Fig. 5b). With the minimum score difference cut-off 0.10, binding affinity of 23 and 22 TFs was predicted to be influenced by four IL10 and five IL6 SNPs respectively. The IL6 SNPs rs1800795 and rs1800797 (r2 = 0.97) affect binding affinity of GATA1, GATA2 and YYI TFs. The linked alleles rs1800795-G and rs1800797-G do influence on binding affinity of GATA1 and GATA2 factors in opposite directions (rs1800795-G has higher binding specificity, rs1800797-G has lower binding specificity), while the influence on transcription factor YYI affinity is unidirectional. We also used RegSNP resource for an analysis of transcription factors NF-1and Sp1 known to have a higher affinity for the rs1800795-C and rs1800896-G alleles respectively18,19. These factors were not covered by the initial analysis due to the stringent cut-off settings (see please Materials and Methods). RegSNP results for these TFs as well as for the above mentioned GATA1 factor (higher affinity for the rs1800795-G allele20) were in line with literature data (Supplementary Table S8, Sheets 1 and 2). Finally, we performed an analysis of the nuclear factor NF-kB, which was shown to be induced by a majority of pneumonia-related pathogens (http://www.bu.edu/nf-kb/gene-resources/target-genes/). The IL10 SNP rs1800795 falls into low-affinity binding site for NF-kB, while the IL6 SNP rs1800896 exerts some influence on NF-kB sequence-specific affinity (allele C higher) (Supplementary Table S8, Sheets 1 and 2).
The analysis of transcription factors’ affinity changes caused by SNPs yielded several noteworthy observations. First, alleles within one SNP could have increased or decreased affinity to different TFs. Second, GATA1-3 factors were highly overrepresented in our series: five of nine genes have SNPs that may influence GATA factors’ binding affinity. Moreover, all SNPs with potential GATA factors binding sites have predicted allele binding score differences >0.20. Third, in the above analysis of the IL6 and IL10 SNPs we showed that SNPs within the same putative promoter region may change the protein-DNA binding affinity of a specific TF /TF family member. This observation was supported by data analysis of the other SNPs (e.g. SOX-related factors and IL10 SNPs; DCE SIII factor, Msn2/Msn4 factors and IL13 SNPs; CCAAT/enhancer-binding proteins (C/EBPdelta and C/EBPbeta), HSF and IL4 SNPs; GATA family members and TLR2 SNPs (Supplementary Table S8).
Linkage disequilibrium and functional analysis of the selected genes
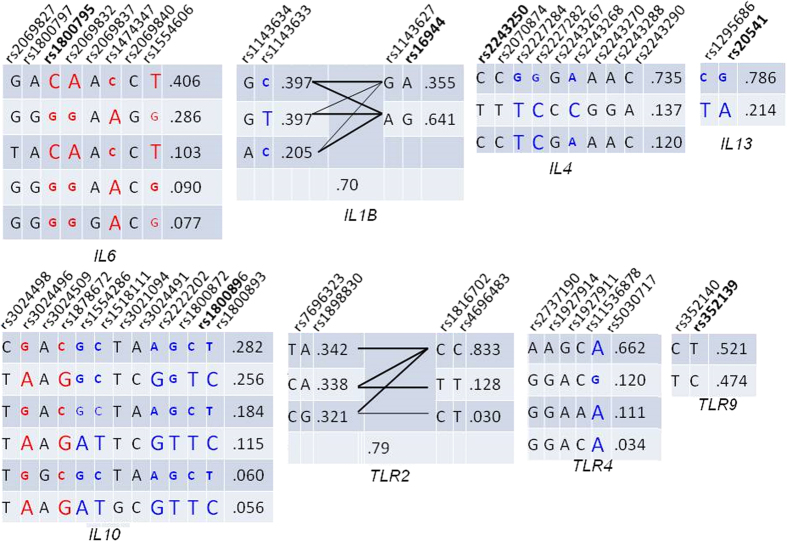
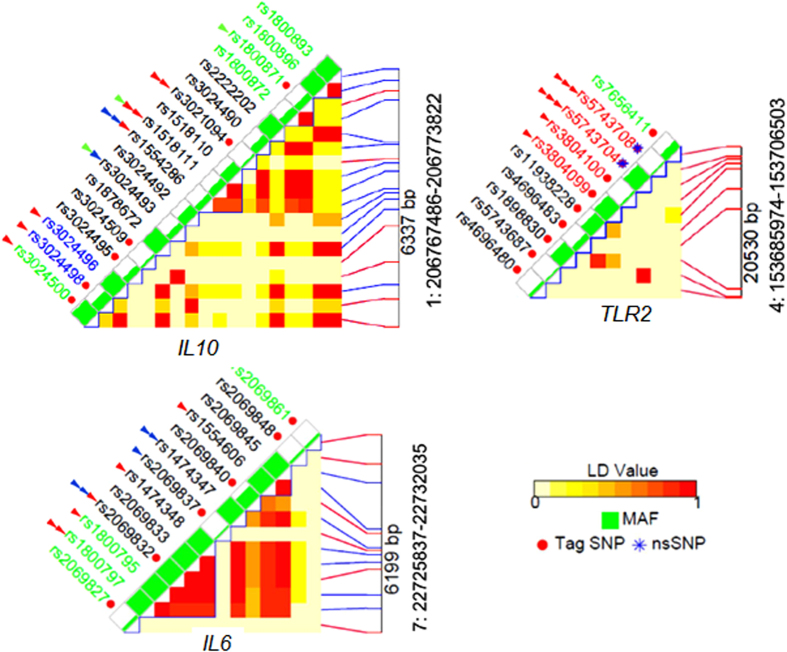
To systematically investigate functional characteristics of the SNPs within the selected genes we constructed LD blocks in European population and collected data on SNPs in these blocks utilizing three web-based resources: RegulomeDB21, SNPinfo22 and the NHGRI GWAS Catalog23 (Supplementary Table S9). IL6, IL10 and TLR2 genes are of the greatest interest in regard with the results of our meta-analysis. Data for these genes are given in Fig. 6, while characteristics of the remaining genes are provided in Supplementary Figure S3. IL6 SNPs are in high LD; SNPs in putative promoter region, including rs1800795 have some regulatory potential, however the highest in silico functionality is predicted for the intron SNP rs2069832 which is adjacent to rs1800795 (distance 788 bp). IL10 SNPs are in weak LD; putative promoter SNPs including rs1800896 have little if any regulatory potential. Three IL10 SNPs were associated with diseases in the NHGRI GWAS Catalog (Supplementary Table S9). Several intron and 3’downstream SNPs that are not linked with the SNP rs1800896 may be more functional than the latter one. An analysis of LD blocks and functional annotations of the IL6 SNPs shows that biological effects of these SNPs may be substantially influenced by the effects of proxy SNPs, while for the IL10 SNPs the influence of neighboring SNPs is not profound. The TLR2 functional variations are SNPs in coding regions, especially non-synonymous SNPs with known deleterious effects due to amino-acid change. Among SNPs in the other genes under study (Supplementary Figure S3), functional SNPs are located predominantly in the IL1B, IL13 and TLR9 gene; two SNPs in the IL13 gene and one SNP in the TLR4 gene have been associated with several disorders and traits in the NHGRI GWAS Catalog (Supplementary Table S9).
Figure 6. Linkage disequilibrium plots of the IL6, IL10 and TLR2 gene regions generated with the LD tag SNP selection tool within SNPinfo resource.
Pair-wise LD values are indicated by different color, which changes from red to white as the D’ value decreases. SNP name is colored with genomic context: black, intron region; red: coding region; blue, UTR (untranslated) region, green, non-genic region. Minor allele frequency for each SNP in European population is denoted by the height of green bar. Functional annotations from the SNP info web server are shown with red arrows: one arrow, regulatory potential is in the range of 0.10–0.20; two arrows, regulatory potential is in the range of 0.20–0.30; three arrows, regulatory potential is higher than 0.30. Functional annotations from the RegulomeDB info web server are represented with dark blue arrows: one arrow, score 2a–f; two arrows, score 1a–f. Scores 3–7 are not provided. SNPs with category 7 score lack evidence of regulatory function, while category 1 variants are assumed to affect binding and expression of a gene target. Within subcategories a–f, variant scored as 1a has the highest confidence on functionality. Functional annotations from the NHGRI GWAS Catalog are marked by green arrows. The number of arrows corresponds to the number of the associations in the Catalog.
Discussion
In this study, we investigated a role of host genetics in pneumonia development and progression. Quality of a genetic association study depends on many factors, with sample size being among the most important among them. In addressing gene candidate studies of pneumonia, the largest sample size across all the studies was used by Martin-Loeches et al.13 followed by our CAP and HAP groups. Nevertheless, we did not obtain results significant after correction for multiple comparisons. This is a rather typical situation, since common polymorphic variants have low penetrance and cannot provide strong association effects, meanwhile, more sites are analyzed the less probability that results will remain significant after testing for multiplicity. In view of this, the revealed association of the IL4 rs2243250-T allele with NP, which is in line with the results of the recent meta-analysis (see please the Results section), seems worthwhile. Although IL4 is usually considered in regard to the immunoglobulin (Ig)-mediated allergic/inflammatory pathway, it is also known to influence humoral response during bacterial infection24 and suppress production of bacterial toxins-induced synthesis of proinflammatory cytokines25. IL4 affects pulmonary surfactant homeostasis26 and may be an important player in pulmonary clearance of bacteria and virus27. Given the results of allele-specific expression study28 together with the results of the meta-analysis of respiratory tract infectious diseases6 and data from our study, the rs2243250-T allele may be a genetic marker or a susceptibility allele in several respiratory infections.
Currently, there are a limited number of pneumonia-related meta-analyses of genetic association studies. In contrary to the majority of other common diseases, which are often subjected to numerous and repetitive meta-analyses, pneumonia meta-analyses considered a narrow range of genes and have never been updated (with the exception for the IL6 association with pneumonia) (https://phgkb.cdc.gov/HuGENavigator/phenoPedia.do). Our study is the first to summarize data from individual papers on the role of TLR2 SNPs in pneumonia/pneumococcal infection. Among TLRs, TLR2 is a receptor with the widest range of recognized pathogen-associated molecular patterns (PAMPs) including PAMPs from Gram-positive bacteria29. There was no association of TLR2 with pneumonia in the whole set of meta-analyzed studies. Given that HAP is an extremely heterogeneous disease, we performed a stratified meta-analysis of CAP/Legionnaires’ disease/pneumococcal disease studies. Minor genotype TLR2 rs5743708 GA/AA was associated with infection in this subset. To access an influence of amino-acid change (753 Arg > Gln) on enzyme structure and activity we utilized the HOPE resource30. The mutant residue differs from the wild-type one by size, charge and hydrophobic properties; multimer contacts peculiar to wild-type protein may be blocked in the mutant protein. To identify numerous PAMPs, TLR2 forms either homodimers or heterodimers with TLR1 or TLR6. The TLR2 rs5743708 G to A transition leads to a functional deficiency in heterodimerization with TLR6 with subsequent diminished activation of intracellular signaling pathways31. This change in TLR2 function correlates with the observed influence of this SNP on susceptibility to infections6,32.
Other noticeable results of our meta-analyses relate to the development of severe complications in pneumonia/pneumococcal infection. Severe sepsis/septic shock were associated with the IL6 rs1800795 GC-CC and IL10 rs1800896 G/G genotypes. The result for IL6 was observed on the relatively large sample therefore it seems more reliable than the summary estimate for IL10 based on the smaller sample size. Data for the IL6 genotype GC-CC were also supported by its association with further worsening of critical illness from severe sepsis to septic shock in pneumonia patients33,34. IL6 is a multi-functional cytokine with both pro- and anti-inflammatory properties and a central role in host defence35. IL10 is the most potent anti-immune and anti-inflammatory cytokine. In infection, it provides a balance between immunosuppression and immunostimulation to maintain a homeostatic state36. Since both cytokines are key players in orchestrating immune response, we assume that alleles that protected against severe complications in pneumonia patients (the IL6 rs1800795-G and IL10 rs1800896-A) may provide more effective regulation in the cytokine network in specific pathophysiological conditions.
An assay for ex vivo cytokine stimulation is often applied to study genetically mediated interindividual variations of cytokine production upon specific stimuli. The results of such an assay provide data for classification of common polymorphic variants as “low” or “high” production ones. Ex vivo stimulated secretion of cytokines is considered to be more standardized assay than in vivo measurement of cytokine level. Ex vivo spontaneous cytokine production was declared not to occur or occur at low levels albeit with some exceptions37. Hence, in genetic association studies, the results of the ex vivo stimulation can be interpreted in terms of more or less inducibility for the carriers of different genotypes. There are many methodical circumstances influencing stimulated expression level, the most important of them, in vivo pre-existent conditions, the use of PBMCs vs. whole blood, the length of the incubation period, single or several concentrations of the stimuli applied and even the origin of this stimuli16,37,38. In general, literature data support an existing classification exclusively for the IL10 “high” and “low” producing genotypes but only in certain cell types and in terms of LPS-induced ex-vivo IL10 secretion. Utilizing the SNPexp resource we showed that basal mRNA expression level was weakly affected by genetic background.
The pattern of cytokine responses to infection pathogens in vivo is thought to be much more complex than in model experiments. Age, sex, socio-economic status and even more so chronic disorders affect cytokine level20,37. Nevertheless, given the results of the meta-analyses it is interesting to consider IL6 and IL10 genotype-dependent expression data in pneumonia or conditions which may precede or follow pneumonia. In CAP studies, there were no significant associations between the IL6 rs1800795 genotype and IL6 production in adult39,40 and pediatric cases41. High IL6 secretion was associated with the -174G allele in patients with severe sepsis34 and after cardiopulmonary bypass (CPB)42. In three other studies in CPB patients, the -174C allele has appeared to be high producing allele43,44,45. IL10 protein level has been not affected by IL10 SNPs in three studies of CAP39,40,46 and one study of NP47. Significant but opposite results have been shown for -1082 G/G genotype associated with lower production of IL10 after CPB48 and with higher secretion of IL10 in burned patients49. Two papers have revealed the significant associations of a “high”- producing haplotype -1082G-819C-592C with higher level of IL10 in adult and pediatric patients after CPB50,51. This brief survey demonstrated that in vivo genetic associations of IL6 and IL10 expression levels were rather contradictory.
Functional common SNPs in cytokine genes are mainly located in putative promoter regions. They may fall inside TFBS and alter transcription factor DNA-binding ability. We explored 2 kbp 5′-upstrem gene region of the studied genes for common SNPs and analyzed their sequence-specific influence on TF binding affinity. Our analysis showed that GATA1-3 factors were highly overrepresented in the set. GATA1-3 factors play a pivotal role in hematopoiesis but also are known to be involved in innate and adaptive immunity. They regulate immune response against different pathogens including those that may cause pneumonia52. Among other common TFs there were HSFs (heat-shock factors). SNPs that influence binding affinity of HSFs were located in anti-inflammatory cytokines IL4 (two SNPs), IL10 and in toll-lire receptor genes TLR4, TLR9. Functional activity of these SNPs may be mediated by HSFs’ dependent activation of the anti-inflammatory limb of the immune response53. We also demonstrated that TF family members have similar allele-specific binding affinity (e.g. GATA factors, SOX-related factors) and linked alleles of SNPs may influence binding affinity of the same TF uni or multi-directionally. Clustering of TFBS for the same TF (homotypic clusters of TFBSs (HCTs)) is known to be enriched in promoters. It has been suggested that HCTs provide functional advantages being favorable for recruitment of TFs54.
It is known that the same TFs can repress or activate transcription depending on cell type and specific promoter sequence. There can be different coexisting modes of TF action: direct activation or repression, indirect activation or repression via interaction with cofactors, induction or suppression by disruption of binding sites and other55. Given complex and optional interactions of TFs with TFBS, our in silico analysis was not either definitive or exhaustive but rather tentative and suggestive with an intension to test the approach for further more detailed investigations supported by experimental data.
Combining LD analysis with functional information data, we showed that a relatively higher overlap with functional annotations was found for the IL6 and IL10 SNPs. We did not reveal predominance of functional SNPs in certain regions of the all studied genes; they were more or less evenly distributed within genes. The TLR2 and TLR4 genes size was several times larger than the cytokine genes size. High LD was evident for the IL6, IL4 and IL8 genes while low LD was detected for the TLR2 and TLR4 genes. This means that the estimated genetic associations reflect non-independent effects of tightly linked SNPs in the IL6 and IL4 genes. The TLR2 SNP rs5743708 is not linked with the other functional TLR2 SNP rs5743704 hence the amino-acid change 753Arg > Gln (rs5743708) may solely contribute to the revealed association of the TLR2 gene with pneumonia.
The study has serious limitations. Own experimental data includes only genotyping of the SNPs in six cytokine genes and three genes of toll-like receptors among CAP and HAP subjects. CAP and HAP samples were modest and the study was powered to detect only relatively large effect sizes (minimum detectable OR~1.6–1.9). Our patients were mainly men and the results may not be generalizable to women. Genes with an important role in the development and output of inflammation such as TNFa, IL1R, LTA, TGFb were not included in the analysis. Highly heterogeneous spectrum of pneumonia-related infections in adult and pediatric populations with very different pathogens with different virulence and different sensitivity to antibiotics were considered. The analysis of SNPs influence on DNA - TF binding affinity was limited by the stringent cut-off settings. However, the study has some biological and methodological implications. We combined own experimental data and the results of the meta-analyses with the analyses of the totality of published data (expression studies) and web-based resources. Our original analysis of transcription factors’ affinity changes caused by SNPs did not and could not provide an unequivocal interpretation in terms of “low” and “high” allele-specific expression phenotypes. Instead in silico obtained data demonstrated complexity in an assessment functionality of SNPs that fall inside TFBS. Consideration of the enrichment for the functional SNPs in LD blocks may help to estimate contribution of the individual SNPs in the observed or presumed associations.
In conclusion, the current study presents some experimental data and the results of the meta-analyses related to the role of host genetics in pneumonia development and progression. Other results were obtained from literature and web-based resources. Since the last data have no experimental support, care must be taken with our tentative and hypothesis generating findings, which require testing in further investigations.
Materials and Methods
CAP and HAP case-control studies
The study protocol was approved by the Ethics Committee of the V. A. Negovsky Research Institute of General Reanimatology (with IRB approval number 2/6/2012), and adhered to the tenets of the Declaration of Helsinki. Informed consent was obtained from all subjects.
From January 2008 to February 2016, CAP and HAP studies were conducted at the hospitals of the V.A. Negovsky Research Institute of General Reanimatology, Moscow, Russia. Three hundred ninety CAP patients were recruited in the study; the corresponding control group consisted of 203 unrelated healthy volunteers without a previous history of pneumonia. Subjects with severe physical trauma due to the accidents and patients with acute diseases requiring extensive surgery were included in the prospective study of NP; among 571 patients, 355 individuals developed NP. The majority of patients and controls in both studies were workers of the Rescue Service, predominantly practically healthy young or middle-aged men. The current study is a continuation of previously published CAP and NP investigations hence inclusion/exclusion criteria, microbiological procedures, genotyping methods and statistical procedures are available elsewhere56,57.
Meta-analysis
A final search was performed on 4th April 2016 of the PubMed, EMBASE, and Web of Science databases. We used the following keyword terms as the criteria for searching: pneumonia, pneumonia outcome, pneumonia severity, sepsis, septic shock, systemic inflammatory response, Streptococcus pneumonia, pneumococcus and gene polymorphism, genotype, alleles, variants and interleukin 6, interleukin-6, IL6, IL-6 or interleukin 10, interleukin-10, IL10, IL-10, or interleukin 8, interleukin-8, IL8, IL-8, or interleukin 1B, interleukin-1B, IL1B, IL-1B, or interleukin 4, interleukin-4, IL4, IL-4, or interleukin 13, interleukin-13, IL13, IL-13, or toll-like receptor 2, TLR2, or toll-like receptor 4, TLR4, or toll-like receptor 9, TLR9. Two investigators (LS and SS) independently extracted data and reached consensus on all the items. We used the following inclusion criteria. The study had to be published in English or in Russian. The article must have evaluated prevalence rates of any type of pneumonia or pneumococcal disease in connection with genotyping data. To be included in the meta-analysis, the studies should provide enough data to calculate odds ratio (OR) with its 95% confidence interval. Additionally we searched for the studies assessing the association of the studied polymorphic variants with severe sepsis/septic shock or systemic inflammatory response (SIRS) in patients with pneumonia. The differences between sepsis and SIRS are that in sepsis individuals meet criteria for SIRS and have a known infection. Patients with SIRS and acute organ dysfunction may be termed patients with severe SIRS (score IV) (https://en.wikipedia.org/wiki/Systemic_inflammatory_response_syndrome#cite_note-rippe-3). In this context, we combined patients with SIRS IV and severe sepsis/septic shock into a single group with the most severe pneumonia-related critical conditions.
Statistical analysis was conducted with the Review Manager 5.3 software58. Pooled effects were calculated under the following genetic models (additive, dominant, recessive and overdominant). The Hardy-Weinberg equilibrium was assessed using Pearson’s χ2 test. The heterogeneity index I2 (I-squared) was used to assess the between-study heterogeneity. Values of I2 in the range of 0–25% were considered indicating negligible heterogeneity with the fixed-effect model applied, 25–50% – modest heterogeneity with both the fixed-effect and random-effect models applied, >50% – significant heterogeneity requiring the random-effect model. We conducted sensitivity analyses to explore the influence of individual studies on the pooled estimate. We did not assess publication bias for revealed associations because, when the number of studies is lower than ten, sensitivity of the tests is low59.
Genotype-specific expression analysis
We collected literature data on genotype-specific ex-vivo induced expression level of the studied genes. We used the following keyword terms as the criteria for searching: expression, secretion, production and gene polymorphism, genotype, alleles, variants and interleukin 6, interleukin-6, IL6, IL-6 or interleukin 10, interleukin-10, IL10, IL-10, or interleukin 8, interleukin-8, IL8, IL-8, or interleukin 1B, interleukin-1B, IL1B, IL-1B, or interleukin 4, interleukin-4, IL4, IL-4, or interleukin 13, interleukin-13, IL13, IL-13.In order not to miss related articles we did not use other terms and assorted papers after reading abstracts.
We performed an in silico analysis of the allele- specific mRNA expression level under basal conditions using SNPexp online tool17. We constructed haplotypes of SNPs in the studied genes in CEU population (Utah residents with ancestry from northern and western Europe) with the HaploView software (version 4.2). All within gene SNPs and SNPs in the 2000-bp upstream 5 flanking and the 500-bp downstream 3 flanking regions were included in the haplotype analysis. Haplotype blocks were defined using the method of confidence intervals60. Phased haplotypes from 210 individuals in the HapMap in correlation with expression data for 47294 transcripts in EBV transformed lymphoblastoid cell lines are available through the SNPexp database. By using web service linear regression testing we correlated mRNA expression level with genotypes in the 60 parents in CEU parent/parent/offspring trios17.
Allele-specific transcription factor DNA-binding affinity analysis
We searched for the SNPs with MAF >0.10 within the putative promoter regions of the studied genes (2 kb upstream of a transcription start site) utilizing the NCBI Gene database. An in silico analysis of transcription factor binding site likelihood scores at positions of SNPs was performed with RegSNP resource (http://viis.abdn.ac.uk/regsnp/Home.aspx). In RegSNP web server, experimentally validated TFBS from the publicTransfac and the Jaspar databases are converted into a positional weight matrix. We used the following analysis options: species, human; cut-off value for minimum transcription factor binding site length, 4; cut-off value for minimum transcription factor likelihood score, 0.85; cut-off value for minimum score difference, 0.1; cut-off value for minimum score ratio, 0.1.
Functional and linkage disequilibrium analysis
We explored LD patterns in the genes under study (including 2 kbp 5′ upstream and 500 bp 3′downstream regions) with the tool LD tag SNP selection within SNPinfo resource22. Since HapMap LD pairwise data do not include all the SNPs considered in our analysis, we used dbSNP genotype data in European population. We calculated pairwise LD values of all SNPs in the studied genes and plotted LD maps with the online program (http://snpinfo.niehs.nih.gov/cgi-bin/snpinfo/snpfunc.cgi)22. Functional analysis was performed with SNPinfo bioinformatic tool SNP Function Prediction (FuncPred; http://snpinfo.niehs.nih.gov/cgi-bin/snpinfo/snpfunc.cgi), RegulomeDB database21 and the NHGRI GWAS Catalog23.
Additional Information
How to cite this article: Smelaya, T. V. et al. Genetic dissection of host immune response in pneumonia development and progression. Sci. Rep. 6, 35021; doi: 10.1038/srep35021 (2016).
Supplementary Material
Acknowledgments
The research was carried out within the state assignment of FASO of Russia (theme “The role of genetic factors in the development and structure of multi-organ failure” No. 0523-2014-0018).
Footnotes
Author Contributions T.V.S. and L.E.S conceived the idea. V.V.M. and A.M.G. supervised the project. T.V.S. and A.N.K. collected and analyzed data. L.E.S. and O.B.B. designed the experiments. O.B.B. and S.V.S. performed the experiments. L.E.S. analyzed the experimental data. S.V.S., N.A.P. and L.E.S. developed and critically assessed the search strategy for the meta-analysis, performed the search, extracted the data and performed the data analysis. S.V.S. and L.E.S. prepared figures. L.E.S. wrote the manuscript. A.N.K. revised the manuscript. All authors reviewed the manuscript.
References
- Mayr F. B., Yende S. & Angus D. C. Epidemiology of severe sepsis. Virulence 2014. 5, 4–11 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angus D. C. et al. The PIRO concept: P is for predisposition. Crit Care 7, 248–251 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar V., Wijmenga C. & Xavier R. J. Genetics of immune-mediated disorders: from genome-wide association to molecular mechanism. Curr Opin Immunol. 31, 51–57 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L., Nie W., Li W., Yuan W. & Huang W. Associations between TNF-α polymorphisms and pneumonia: a meta-analysis. Plos One 2013. 8, e61039 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Laorden M. I. et al. The role of mannose-binding lectin in pneumococcal infection. Eur Respir J. 41, 131–139 (2013). [DOI] [PubMed] [Google Scholar]
- Patarčić I. et al. The role of host genetic factors in respiratory tract infectious diseases: systematic review, meta-analyses and field synopsis. Sci Rep. 5, 16119 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H. et al. Associations of three well-characterized polymorphisms in the IL-6 and IL-10 genes with pneumonia: a meta-analysis. Sci Rep. 5, 8559 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie W. et al. Angiotensin-converting enzyme I/D polymorphism is associated with pneumonia risk: a meta-analysis. J Renin Angiotensin Aldosterone Syst. 15, 585–592 (2014). [DOI] [PubMed] [Google Scholar]
- Cai X., Fu Y. & Chen Q. Association between TLR4 A299G polymorphism and pneumonia risk: a meta-analysis. Med Sci Monit. 21, 625–629 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgitsi M. D. et al. Individualized significance of the -251 A/T single nucleotide polymorphism of interleukin-8 in severe infections. Eur J Clin Microbiol Infect Dis. 35, 563–570 (2016). [DOI] [PubMed] [Google Scholar]
- Wattanathum A., Manocha S., Groshaus H., Russell J. A. & Walley K. R. Interleukin-10 haplotype associated with increased mortality in critically ill patients with sepsis from pneumonia but not in patients with extrapulmonary sepsis. Chest 128, 1690–1698 (2005). [DOI] [PubMed] [Google Scholar]
- Rautanen A. et al. Genome-wide association study of survival from sepsis due to pneumonia: an observational cohort study. Lancet Respir Med. 3, 53–60 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin-Loeches I. et al. Variants at the promoter of the interleukin-6 gene are associated with severity and outcome of pneumococcal community-acquired pneumonia. Intensive Care Med. 38, 256–262 (2012). [DOI] [PubMed] [Google Scholar]
- Gallagher P. M. et al. Association of IL-10 polymorphism with severity of illness in community acquired pneumonia. Thorax. 58, 154–156 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan F. F. et al. Clinical relevance of TLR2, TLR4, CD14 and FcgammaRIIA gene polymorphisms in Streptococcus pneumonia infection. Immunol Cell Biol. 86, 268–270 (2008). [DOI] [PubMed] [Google Scholar]
- Warlé M. C. et al. Are cytokine gene polymorphisms related to in vitro cytokine production profiles? Liver Transpl. 9, 170–181 (2003). [DOI] [PubMed] [Google Scholar]
- Holm K., Melum E., Franke A. & Karlsen T. H. Snpexp - a web tool for calculating and visualizing correlation between hapmap genotypes and gene expression levels. BMC Bioinformatics 11, 600 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaaf B. et al. The interleukin-6 -174 promoter polymorphism is associated with extrapulmonary bacterial dissemination in Streptococcus pneumoniae infection. Cytokine 31, 324–328 (2005). [DOI] [PubMed] [Google Scholar]
- Larsson L. et al. The Sp1 transcription factor binds to the G-allele of the -1087 IL-10 gene polymorphism and enhances transcriptional activation. Genes Immun. 10, 280–284 (2009). [DOI] [PubMed] [Google Scholar]
- Cole S. W. et al. Computational identification of gene-social environment interaction at the human IL6 locus. Proc Natl Acad Sci USA 107, 5681–5686 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyle A. P. et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 22, 1790–1797 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Z. & Taylor J. A. SNPinfo: integrating GWAS and candidate gene information into functional SNP selection for genetic association studies. Nucleic Acids Res. 37, W600–W605 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welter D. et al. The NHGRI GWAS Catalog, a curated resource of SNP-trait associations. Nucleic Acids Res. 42, D1001–D1006 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan A. Q., Shen Y., Wu Z. Q., Wynn T. A. & Snapper C. M. Endogenous pro- and antiinflammatory cytokines differentially regulate an in vivo humoral response to Streptococcus pneumoniae. Infect Immun. 70, 749–761 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumhofer J. M. et al. Gene transfer with IL-4 and IL-13 improves survival in lethal endotoxemia in the mouse and ameliorates peritoneal macrophages immune competence. Eur J Immunol. 28, 610–615 (1998). [DOI] [PubMed] [Google Scholar]
- Ikegami M. et al. IL-4 increases surfactant and regulates metabolism in vivo. Am J Physiol Lung Cell Mol Physiol. 278, L75–L80 (2000). [DOI] [PubMed] [Google Scholar]
- Boyton R. J. & Openshaw P. J. Pulmonary defences to acute respiratory infection. Br Med Bull. 61, 1–12 (2002). [DOI] [PubMed] [Google Scholar]
- Gu W. et al. Clinical relevance of 13 cytokine gene polymorphisms in Chinese major trauma patients. Intensive Care Med. 36, 1261–1265 (2010). [DOI] [PubMed] [Google Scholar]
- Borrello S., Nicolò C., Delogu G., Pandolfi F. & Ria F. TLR2: a crossroads between infections and autoimmunity? Int J Immunopathol Pharmacol. 24, 549–556 (2011). [DOI] [PubMed] [Google Scholar]
- Venselaar H., Te Beek T. A., Kuipers R. K., Hekkelman M. L. & Vriend G. Protein structure analysis of mutations causing inheritable diseases. An e-Science approach with life scientist friendly interfaces. BMC Bioinformatics. 11, 548 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Y., Song C., Snyder G. A., Sundberg E. J. & Medvedev A. E. R753Q polymorphism inhibits Toll-like receptor (TLR) 2 tyrosine phosphorylation, dimerization with TLR6, and recruitment of myeloid differentiation primary response protein 88. J Biol Chem. 287, 38327–38337 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenz E., Mira J. P., Cornish K. L., Arbour N. C. & Schwartz D. A. A novel polymorphism in the toll-like receptor 2 gene and its potential association with staphylococcal infection. Infect Immun. 68, 6398–6401 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng B., Mao Z. R., Pang K., Zhang S. L. & Li L. Association of tumor necrosis factor α -308G/A and interleukin-6 -174G/C gene polymorphism with pneumonia-induced sepsis. J Crit Care. 30, 920–923.(2015) [DOI] [PubMed] [Google Scholar]
- Tischendorf J. J. et al. The interleukin-6 (IL6)-174 G/C promoter genotype is associated with the presence of septic shock and the ex vivo secretion of IL6. Int J Immunogenet. 34, 413–418 (2007). [DOI] [PubMed] [Google Scholar]
- Hunter C. A. & Jones S. A. IL-6 as a keystone cytokine in health and disease. Nat Immunol. 16, 448–457 (2015). [DOI] [PubMed] [Google Scholar]
- Mosser D. M. & Zhang X. Interleukin-10: new perspectives on an old cytokine. Immunol Rev. 226, 205–218 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker D. et al. Spontaneous cytokine production and its effect on induced production. Clin Diagn Lab Immunol. 9, 1049–1056 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathiak G. et al. Lipopolysaccharides from different bacterial sources elicit disparate cytokine responses inwhole blood assays. Int J Mol Med. 11, 41–44 (2003). [PubMed] [Google Scholar]
- Endeman H. et al. Systemic cytokine response in patients with community-acquired pneumonia. Eur Respir J. 37, 1431–1438 (2011). [DOI] [PubMed] [Google Scholar]
- Martinez-Ocaña J. et al. Plasma cytokine levels and cytokine gene polymorphisms in Mexican patients during the influenza pandemicA(H1N1)pdm09. J Clin Virol. 58, 108–113 (2013). [DOI] [PubMed] [Google Scholar]
- Zidan H. E., Elbehedy R. M. & Azab S. F. Il6-174 g/c gene polymorphism and its relation to serum il6 in egyptian children with community-acquired pneumonia. Cytokine. 67, 60–64 (2014). [DOI] [PubMed] [Google Scholar]
- Burzotta F. et al. Relation of the -174 G/C polymorphism of interleukin-6 to interleukin-6 plasma levels and to length of hospitalization after surgical coronary revascularization. Am J Cardiol. 88, 1125–1128 (2001). [DOI] [PubMed] [Google Scholar]
- Bittar M. N. et al. Interleukin 6 G-174C polymorphism influences outcome following coronary revascularization surgery. Heart Surg Forum. 8, E140–E145 (2005). [DOI] [PubMed] [Google Scholar]
- Wypasek E. et al. The increased plasma C-reactive protein and interleukin-6 levels in patients undergoing coronary artery bypass grafting surgery are associated with the interleukin-6-174G > C gene polymorphism. Ann Clin Biochem. 47 (Pt 4), 343–349. (2010). [DOI] [PubMed] [Google Scholar]
- Brull D. J. et al. Interleukin-6 gene −174g>c and −572g>c promoter polymorphisms are strong predictors of plasma interleukin-6levels after coronary artery bypass surgery. Arterioscler Thromb Vasc Biol. 21, 1458–1463 (2001). [DOI] [PubMed] [Google Scholar]
- Romanova E. N. & Govorin A. V. TNF-alpha, IL-10, and eNOS gene polymorphisms in patients with influenza A/H1N1 complicated by pneumonia. Ter Arkh. 85, 58–62 (2013). [PubMed] [Google Scholar]
- Sakamoto K. et al. Relationship between cytokine gene polymorphisms and risk of postoperative pneumonia with esophageal cancer. J Gastrointest Surg. 18, 1247–1253 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galley H. F., Lowe P. R., Carmichael R. L. & Webster N. R. Genotype and interleukin-10 responses after cardiopulmonary bypass. Br. J. Anaesth. 91, 424–426 (2003). [DOI] [PubMed] [Google Scholar]
- Accardo Palumbo A. et al. Analysis of IL-6, IL-10 and IL-17 genetic polymorphisms as risk factors for sepsis development in burned patients. Burns. 38, 208–213 (2012). [DOI] [PubMed] [Google Scholar]
- Schippers E. F., van’t Veer C., van Voorden S., Martina C. A., Huizinga T. W., le Cessie S. & van Dissel J. T. IL-10 and toll-like receptor-4 polymorphisms and the in vivo and ex vivo response to endotoxin. Cytokine. 29(5), 215–228 2005 Mar 7. [DOI] [PubMed] [Google Scholar]
- Allen M. L. et al. Interleukin-10 and its role in clinical immunoparalysis following pediatric cardiac surgery. Crit Care Med. 34, 2658–2665 (2006). [DOI] [PubMed] [Google Scholar]
- Kerry S., TeKippe M., Gaddis N. C. & Aballay A. GATA transcription factor required for immunity to bacterial and fungal pathogens. Plos One. 1, e77 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H. et al. HSF1 is a transcriptional activator of IL-10 gene expression in RAW264.7 macrophages. Inflammation. 35, 1558–1566 (2012). [DOI] [PubMed] [Google Scholar]
- Gotea V. et al. Homotypic clusters of transcription factor binding sites are a key component of human promoters andenhancers. Genome Res. 20, 565–577 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon S., Akopyan G., Garban H. & Bonavida B. Transcription factor YY1: structure, function, and therapeutic implications in cancer biology. Oncogene. 25, 1125–1142 (2006). [DOI] [PubMed] [Google Scholar]
- Salnikova L. E., Smelaya T. V., Golubev A. M., Rubanovich A. V. & Moroz V. V. CYP1A1, GCLC, AGT, AGTR1 gene - gene interactions in community-acquired pneumonia pulmonary complications. Mol Biol Rep. 40, 6163–6176 (2013). [DOI] [PubMed] [Google Scholar]
- Salnikova L. E., Smelaya T. V., Vesnina I. N., Golubev A. M. & Moroz V. V. Genetic susceptibility to nosocomial pneumonia, acute respiratory distress syndrome and poor outcome in patients at risk of critical illness. Inflammation. 37, 295–305 (2014). [DOI] [PubMed] [Google Scholar]
- Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration (2014) Available at: http://tech.cochrane.org/revman/new-releases/whats-new-in-revman-5. (Accessed: 12th April 2016).
- Ioannidis J. P. & Trikalinos T. A. The appropriateness of asymmetry tests for publication bias in meta-analyses: A large survey. CMAJ. 176, 1091–1096 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabriel S. B. et al. The structure of haplotype blocks in the human genome. Science 296, 2225–2229 (2002). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.