Abstract
Background
Helicobacter pylori is a major human pathogenic bacterium in gastric mucosa. Although the association between gastric cancer and H. pylori has been well-established, the molecular mechanisms underlying H. pylori-induced carcinogenesis are still under investigation. MicroRNAs (miRNAs) are small noncoding RNAs that modulate gene expression at the posttranscriptional level. Recently, studies have revealed that miRNAs are involved in immune response and host cell response to bacteria. Also, microRNA-375 (miR-375) is a key regulator of epithelial properties that are necessary for securing epithelium-immune system cross-talk. It has been recently reported that miR-375 acts as an inhibitor of H. pylori-induced gastric carcinogenesis. There are few reports on miRNA-mediated targeting long noncoding RNAs (lncRNAs).
Objectives
This study aimed to examine the possible effect of miR-375 as an inhibitor of H. pylori-induced carcinogenesis on the expression of lncRNA SOX2 overlapping transcript (SOX2OT) and SOX2, a master regulator of pluripotency of cancer stem cells.
Materials and Methods
In a model cell line, NT-2 was transfected with the constructed expression vector pEGFP-C1 contained miR-375. The RNA isolations and cDNA synthesis were performed after 48 hours of transformation. Expression of miR-375 and SOX2OT and SOX2 were quantified using real-time polymerase chain reaction and compared with control cells transfected with pEGFP-C1-Mock clone. Cell cycle modification was also compared after transfections using the flow cytometry analysis.
Results
Following ectopic expression of miR-375, SOX2OT and SOX2 expression analysis revealed a significant decrease in their expression level (P < 0.05) in NT-2 cells compared to the control. Cell cycle analysis following ectopic expression of miR-375 in the NT-2 cells using propidium iodine staining revealed significant extension in sub-G1 cell cycle.
Conclusions
This is the first report to show down-regulation of SOX2OT and SOX2 following induced expression of miR-375. This finding may suggest expression regulation potential between different classes of ncRNAs, for example between miR-375 and SOX2OT. This data not only extends our understanding of possible ncRNA interactions in cancers but also may open novel investigation lines towards elucidation of molecular mechanisms controlling H. pylori inflammation and carcinogenesis.
Keywords: MicroRNAs, miR-375, RNA, Long Noncoding, Helicobacter pylori
1. Background
Helicobacter pylori is a Gram-negative, microaerophilic, spiral-shaped bacterium that colonizes the gastric mucosa. Helicobacter pylori infection is the most common bacterial infection worldwide and as a major risk factor for gastric carcinoma, interferes with cell proliferation and apoptosis of the gastric epithelium (1). Helicobacter pylori is a critical risk factor during the process of gastric carcinogenesis. Gastric cancer is one of the most common cancers worldwide. However, many aspects of molecular mechanism underlying H. pylori-induced gastric carcinogenesis remain to be elucidated (2, 3).
Emerging evidence shows that miRNAs play an important role in pathogen-host interactions (4, 5). MicroRNAs are a class of small (18 - 22nt) ncRNAs that regulate gene expression at the posttranscriptional level. The miRNAs make contribution to various cellular processes such as cell proliferation, growth, development, cellular stress response, and apoptosis. Alterations in the expression of miRNAs have been widely reported in numerous diseases including almost all types of cancers (6). Act as oncogenes (oncomiRs) or tumor suppressors, miRNAs are playing prominent roles in cancer-related processes such as proliferation, apoptosis, metastasis, and angiogenesis. Due to their high stability and also their cell- and tissue-specific expression patterns, miRNAs have received tremendous attention as potential diagnostic, prognostic, and therapeutic agents over the past decade (7).
To date, there are more than 2000 annotated human mature miRNAs in the official registry (the MicroRNA registry) (8). Also, miRNAs have been domestrated to modulate a wide range of biological processes, including cellular development, differentiation, proliferation, apoptosis, metabolism and immune response and microbial infections (9, 10).
The role of miRNAs in bacterial infections has been initially demonstrated in plants, in which a miRNA induced by Pseudomonas syringae in Arabidopsis thaliana mediates the resistance against the bacterium (11). Recent studies have characterized deregulation of host miRNAs following infection with extracellular (H. pylori) (12), or intracellular (Salmonella enterica) (13), Gram-negative bacteria, as well as in the response to Gram-positive (Listeria monocytogenes) (14) and other pathogenic bacteria (Mycobacterium (15) and Francisella species (16)).
The first evidence for the ability of a bacterial pathogen to induce changes in the miRNAs of host infected cells was obtained from H. pylori. Also, miRNA expression studies in response to various bacterial infections have shown common miRNAs as key regulators in the host innate immune response. An increasing number of studies have described the occurrence of mysregulation of miRNA expression and its involvement in the regulation of gene expression in H. pylori-associated gastric cancer. Some miRNAs (let-7b; miR-103; miR-370; miR-371-5p; miR-372; miR-373; miR-375; miR-449b) are down-regulated, and some miRNAs are up-regulated (miR-21; miR-25; miR-93; miR-146a; miR-155; miR-194; 196; miR-200b, 200c; miR-222; miR-223; miR584; miR-1290) in H. pylori-induced inflammation and gastric carcinogenesis (17).
Also, the miR-375 was previously reported to be involved in gastric cancer. Recently, Isomoto et al. found that some miRNAs including miR-375 had significant correlations with H. pylori-associated gastritis based on endoscopic biopsies. Although the complex role of miR-375 in different cancers has been shown, the role of miR-375 in H. pylori-induced gastritis or even gastric carcinogenesis is poorly understood (18-20).
The SRY (sex determining region Y)-box 2 (SOX2) gene is a master regulator of pluripotency, lies within an intron region of another gene named as SOX2 overlapping transcript (SOX2OT), an lncRNA which is transcribed in the same orientation like SOX2. An ncRNA is a functional RNA that is not translated into a protein. The genes of ncRNAs include highly abundant and functionally important RNAs such as transfer RNAs (tRNAs) and ribosomal RNAs (rRNAs), as well as RNAs such as snoRNAs, miRNAs, siRNAs, snRNAs, piRNAs and the lncRNAs. An increasing number of lncRNAs are being identified, characterized and functionally annotated. lncRNAs are still among the least well-understood of RNAs and the underlying mechanisms by which they function remain largely unexplored. Recent evidences have suggested that lncRNAs are biologically functional. Thus, lncRNA are receiving attention in molecular medicine (21).
LncRNAs represent a large fraction of transcriptome as a new level of gene expression regulation. lncRNAs are found to be involved in epigenetic events, regulation of pluripotency and differentiation processes. Also, lncRNAs function as new oncogenes and tumor-suppressor genes. Moreover, lncRNAs reveal an enhancer-like function in human cells. The human genome contains thousands of lncRNAs that are expressed at low levels and in a tissue specific manner. LncRNAs are, in general, poorly conserved and about one-third of them are primate-specific (21).
A few studies describe the interactions between miRNAs and lncRNAs. The miRNAs act either as inhibitory decoys or as regulatory targets of lncRNA, but such interactions are still poorly explored. While the functions of small ncRNAs, such as miRNAs, in the cell have been more understood, lncRNAs have recently started to emerge as abundant regulators of cell physiology and their functions may be diverse (22). Some researchers believe that conserved miRNA-lncRNA interactions could not be reliably detected with our methodology (23). Here, we focused on the expression level mainly.
A recent study revealed miR-375 down-regulation in H. pylori inflammation and gastric cancerinogenesis. The miR-375 has bioinformatically target site on lncRNA SOX2OT. In our previous work we revealed SOX2OT upregulated with master regulators of pluripotency, SOX2 and OCT4, in Esophageal Squamous Cell Carcinoma (ESCC). The SOX2OT varients differentially expressed and spliced in ESCC. Two novel variants of SOX2OT, SOX2OT-S1 and SOX2OT-S2, were up-regulated in ESCC. The novel variants of SOX2OT revealed distinct expression patterns and down-regulated during the process of neural differentiation of human embryonic stem cells known as NTERA-2 or NT-2 cell line (21, 24). There is no report on possible miR-375-SOX2OT interaction and the effect of miR-375 on SOX2 gene expression so far.
2. Objectives
One putative target site has been found on lncRNA SOX2OT and SOX2 gene based on sequence homology search for miR-375 sequence using bioinformatics analyses tool miRcode. In this work we aimed to study the possible effect of miR-375 as an inhibitor of H. pylori-induced carcinogenesis on the expression of lncRNA SOX2OT which its expression is coregulated with SOX2, a master regulator of pluripotency. This study was the first step of the possible miRNA-lncRNA interactions in H. pylori-induced carcinogenesis, because there is no report on this possible interaction in physiologically or pathologic conditions of any cell.
3. Materials and Methods
3.1. Finding Putative Target Site for miR-375 on lncRNA SOX2OT and SOX2
Target site for miR-375 has been searched using bioinformatic tool named miRcode (www.mircode.org) to find its complementary seed on SOX2 and SOX2OT transcripts. miRcode (designed by institute of biomedicine, university of Gothenburg, Sweden) is a comprehensive search tool for putative miRNA target sites across the complete GENCODE (www.gencodegenes.org) annotated transcriptome, including 10,419 lncRNA genes in the current version (miRcode 11, June 2012) The miRcode pipeline is implemented using Matlab, MySQL, Perl, and PHP searches for complementary matches to established miRNA seed families across GENCODE transcripts (25).
3.2. Cell Culture
Human embryonic carcinoma NT-2 stem cells highly express SOX2OT and SOX2 genes that is an excellent prototype model of gene expression in pluripotent stem cells and differentiations associated to human embryonic development. This cell line was kindly provided by Dr. Peter Andrews (Sheffield university, UK). Cells were cultured in DMEM/F12 medium (invitrogen, carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; invitrogen, Carlsbad, CA, USA) and 100 U/mL penicillin/streptomycin (Sigma, St Louis, MO, USA) and incubated in a humidified incubator in an atmosphere of 5% CO2 at 37ºC.
3.3. The miR-375 Expression Plasmid
The expression plasmid pEGFP-C1 containing miR-375 and the empty plasmid mock vector were purchased from (ParsGenome, Iran). Both plasmids express Neomycin and GFP genes to enable selection and detection of transfected cells. pEGFP-C1-miR-375 vector contained EcoR1 and BamHI sites on their respective 5’ and 3’ ends of miR-375 were used to amplify the 334 bp pre-miR-375 sequence by specific primers (Table 1). Restriction enzymes digestions and PCR analysis pEGFP-C1-miR-375 vector were performed for confirmation of miR-375 precursor (Table 1 and Figure 1).
Table 1. Sequences of Primers for Amplification Using the Real-Time Polymerase Chain Reaction.
| Genes | Sequences |
|---|---|
| miR-375 precursor | |
| Forward | CCGGAATTCCGGAGATGCGTTCAGGTGAGG |
| Reverse | CGCGGATCCGCGCCGTACGGTTGAGATGGC |
| SOX2OT | |
| Forward | GATCACCTATTATAATTTTACC |
| Reverse | GAAACCTGTCAGGCTTTCTTC |
| SOX2 | |
| Forward | AAGAGAACACCAATCCCATCC |
| Reverse | TCCAGATCTATACAAGGTCCATTC |
| GAPDH | |
| Forward | GCCACATCGCTCAGACAC |
| Reverse | GGCAACAATATCCACTTTACCAG |
Figure 1. Confirmation of Insert, miR-375 Precursor, in pEGFP-C1-miR-375 Vector by Restriction Digestion and Polymerase Chain Reaction Analysis.
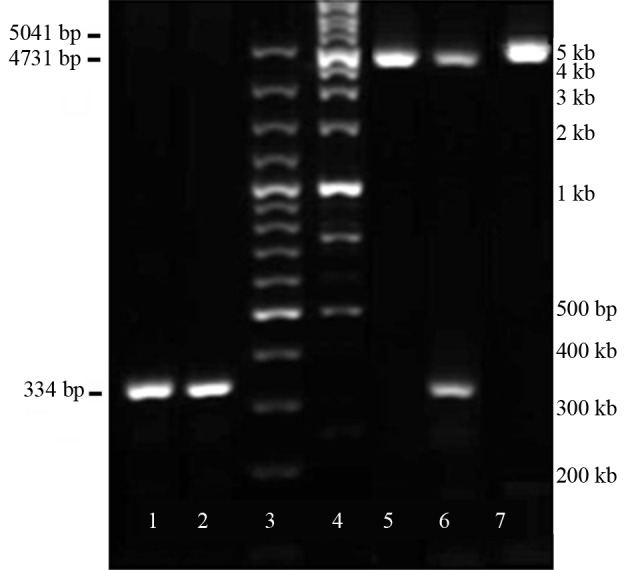
Lane 1 and 2, 334 bp PCR band amplified by specific primers for the insert on the bottom; Lanes 3 and 4 are, 100 bp and 1kbp ladders, respectively; Lane 5, single digestion of pEGFP-C1-Mock digested with EcoRI produced a 5041 bp-fragment; Lane 6, double-digest of pEGFP-C1-miR-375 with EcoRI and BamHI produced 334 bp related to miR-375 precursor insert and 4731 bp vector; Lane 7, single digestion of pEGFP-C1-miR-375 by EcoRI produced a 5041bp fragment.
3.4. Ectopic Expression of miR-375 in NT-2 Cells
The NT-2 cells were seeded at a concentration of 4 × 104 cells per well in 12-well plates and incubated for 24 hours in culture medium. They were transfected with 1.5 µg of pEGFP-C1-miR-375 vector in a test culture and 1.5 µg pEGFP-C1 mock vectors in a negative control group culture, using Lipofectamin 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
3.5. RNA Extraction
After 48 hours of transfections, cells were harvested and total RNAs were extracted from two test and control groups using Trizol reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instruction. The precipitated RNA plate was resuspended in 20 - 30 µL RNase-free dH2O and was treated with DNaseI (Sigma, St Louis, MO, USA) to remove any DNA contaminations. The quality and quantity of total RNA were determined using electrophoresis on agarose gel and NanoDrop2000c Spectrophotometer at 260nm (Thermo Fisher ScientificInc, Wilmington, DE, USA) respectively.
3.6. cDNA Synthesis
The first strand of cDNA for detection of SOX2 and SOX2OT expressions was synthesized by Reverse Transcriptase (Takara, Japan), using both oligo dT and random hexamer primers (Takara, Japan), according to the manufacturer’s instructions. For each sample, a no-reverse transcription (No-RT) control was used in parallel to the DNase-treated RNA, to detect nonspecific amplification of genomic DNA. cDNA synthesis for expression analysis of miR-375 was performed separately by different procedure using ParsGenome’s miRNA kit (ParsGenome, Iran).
3.7. Quantitative Real-Time Polymerase Chain Reaction
Expression of miR-375, SOX2OT and SOX2 were quantified in transfected NT2 cells with pEGFP-C1-miR-375 and pEGFP-C1-Mock vectors 48 hours after transfection. The expression level of miR-375 was determined using the ParsGenome’s miRNA amplification Kit (ParsGenome, Iran).
This was carried out in three main steps including addition of polyA tail, specific cDNA synthesis and RT- PCR amplification, according to the manufacturer’s instructions. All experiments were performed at least in two duplicate using the Bio-Rad thermal cycler (Hercules, California, USA) with the following conditions: 95°C for 10 seconds, 62°C for 10 seconds and 72°C for 30 seconds. Melting curve were determined in 60°C to 95°C. U6 was chosen as an internal control and PCR efficiency for miR-375 and U6 was measured using standard curves generated by serial dilutions of cDNA.
Specific RT-PCR primers also were designed for SOX2OT and SOX2 and GAPDH using the AlleleID 6.0 software (Primer BioSoft, Palo Alto, CA, USA; www.premierbiosoft.com) and Gene runner software (version 3.02; Hastings software Inc.). The sequences of forward and reverse primers used for SOX2, SOX2OT and GAPDH have been listed in Table 1 (length of products: 162, 128, 115 bp, respectively) (Table 1 and Figure 2). Takara SYBR Premix Ex Taq ІІ master mix (2X), supplemented with ROX reference Dye II (Takara, Japan), were used for all RT-PCR reactions. For compensating variations in the amount of input RNA and the efficacy of reverse transcriptase, GAPDH mRNA was also quantified as an internal control, and related expressions of SOX2 and SOX2OT were normalized to its expression value. All RT-PCR reactions were carried out by ABI 7500 RT-PCR PCR systems (applied biosystems, Foster City, CA) using the following cycling conditions: initiation at 94°C for 1 minute, amplification for 40 cycles with denaturation at 94°C for 10 seconds, annealing at 62°C for 10 seconds and extending at 720°C for 40 seconds. The expression of a gene was defined based on the threshold cycle (Ct), and relative expression levels were determined as 2-[Ct of target gene)-(Ct of internal control)].
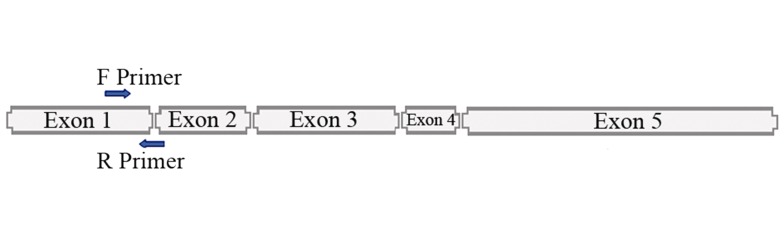
Figure 2. Amplification of 128 bp Fragment of SOX2OT From cDNA by Designing Forward Primer on Exon 1 and Reverse Primer at Junction Between Exon 1 and Exon 2.
3.8. Cell Cycle Analysis by Flow Cytometry
For cell cycle analysis, cells were collected 48 hours after transfection with pEGFP-C1-miR-375 or pEGFP-C1-mock vectoe as control. The cells were washed in PBS, trypsinized with 0.025% trypsin-EDTA to yield single cell suspensions. Then, cells fixed in ice-cold 70% ethanol and stained with 50 μg/mL propidium iodide (PI) solution containing 10 μg/mL RNaseA (Takara, Japan) and 0.1% Triton X-100. Two groups of cells were used for flow cytometric analysis, using Attune acoustic focusing cytometer (applied biosystems, USA). Experiments were repeated at least twice and cell cycle profiles were analyzed using flowing software version 2.5.
3.9. Statistical Analysis
Data are presented as the means ± SD from at least three separate experiments. Significance was analyzed using the student’s t-test. The RT-PCR data were adjusted based on the exact PCR efficiency. The SPSS software, version 17.0 (SPSS, Inc., Chicago, Illinois, USA) was used for performing all the analyses. P < 0.05 were considered as statistically significant difference.
4. Results
4.1. The miR-375 Putative Target Site on lncRNA SOX2OT and SOX2
Bioinformatic and in silico analysis represented a miR-375 putative target site in the third intron of SOX2OT sequence (NR-004053.2) by miRcode, Gene Runner, NCBI databases (Figure 3). The SOX2OT gene contains five exons that intron-less gene of SOX2 embedded within the third intron of SOX2OT.
Figure 3. SOX2OT Gene.
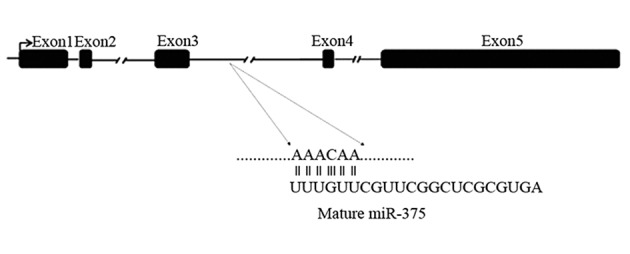
According to our bioinformatic analysis on miR-375 and SOX2OT transcripts, miR-375 has a putative target site on SOX2OT. The SOX2OT gene contains five exons that intron-less gene of SOX2 embedded within the third intron of SOX2OT. Seed sequence of mature form of miR-375 has been represented in the first line.
4.2. Ectopic Expression of miR-375 in NT2 Cells
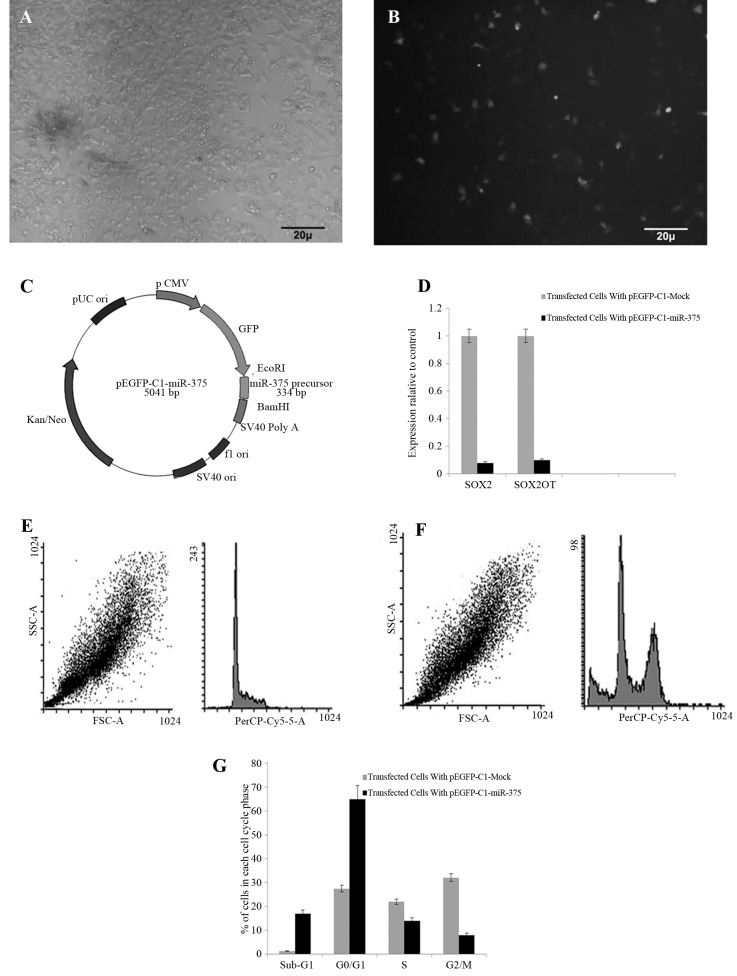
The NT-2 cell line was chosen because of its high SOX2OT basal expression and being a model for pluripotent stem cells to study gene expression and differentiation associated to human embryonic development and stem-cell studies. NT-2 cells transfected with pEGFP-C1-miR-375 (Figure 4A - C) overexpressed miR-375 by about 12-fold in comparison to pEGFP-C1-mock transfected cells 48 hours after transfection with P < 0.05.
Figure 4. NT-2 cell line.
A, Phase contrast micrograph X20 and; B, fluorescence micrograph using green filter show the expression of GFP in transfected NT2 cells 48 hours after transfection with pEGFP-C1-miR-375. that represent cellular clone expression; C, pEGFP-C1 clone contained precursor of miR-375 insert constructed by ParsGenome Co, Tehran, Iran; D, relative expression of SOX2 and SOX2OT in NT-2 cells transfected with pEGFP-C1-miR-375 vector and pEGFP-C1-mock vector as control, Ectopic expression of the miR-375 precursor significantly reduced the expression of SOX2 and SOX2OT; E and F, alteration in cell cycle distribution following overexpression of miR-375 in NT-2 cells was performed by flow cytometry; G, Flow cytometry analysis of NT-2 cells 48 hours after transfection with pEGFP-C1-miR-375 and pEGFP-C1-Mock as control revealed a significant increase in sub-G1 and G0/G1 cell number and significant decrease in S and G2/M cell number after transfection. It suggests sub-G1 extension following ectopic expression of miR-375.
4.3. Over-Expression of miR-375 Down-Regulates SOX2 and SOX2OT
The expression of SOX2 and SOX2OT was measured using the RT-PCR. The relative expression of SOX2OT was decreased by about 9-fold in pEGFP-C1-miR-375 transfected cells in comparison to the pEGFP-C1-mock vector, while 10-fold down-regulation was observed in SOX2 expression, P < 0.05 (Figure 4D).
4.4. Effects of miR-375 Over-Expression on the Cell Cycle Progression in NT-2 Cells
The data obtained from flow cytometry cell cycle analysis following overexpression of miR-375 48 hours after transfection in NT-2 cells compared to cells transfected with pEGFP-C1-mock as control revealed some effects on cell cycle progression. Different alterations in the cell cycle were observed in NT-2 cells transfected with pEGFP-C1-miR-375 compared to control cells. A significant decrease in the percentage of cells in S phase and G2/M phase was observed. Also, a significant arrest in sub-G1 and G0/G1 increase was observed in transfected cells with miR-375 (Figure 4E - G).
5. Discussion
In this work, we aimed to study the possible effect of miR-375 as an inhibitor of H. pylori-induced carcinogenesis on the expression of lncRNA SOX2OT which its expression is co-regulates with SOX2, a master regulator of pluripotency. There is no report on possible interaction of miR-375-SOX2OT in any physiologically or pathologic cells, and thus this study maybe the first step towards to the possible miRNA-lncRNA interactions in H. pylori-induced carcinogenesis.
Helicobacter pylori is considered to be a critical risk factor of gastric cancer. Related mechanisms by which H. pylori could promote cancer are still under investigation. One proposed mechanism has been called perigenetic pathway involves enhancement of the transformed host cell phenotype by means of alteration in gene expression without the need for additional mutations in tumor suppressor genes (26). The miR-375 is one of the miRNAs regulating inflammation and carcinogenetic processes induced by H. pylori acts through perigenetic pathway (19, 27).
Our finding from our unbiased bioinformatic analyses showed that SOX2OT has a predicted seed for miR-375. MiR-375 has a putative site on SOX2OT intron 3 (AGGGAG) and one putative site on SOX2 unique exon sequence. This implicated that miR-375 may interact with SOX2OT and change its expression level. Two common forward and reverse primers were designed on exon 1 and border with exon 2 respectively to amplified 128 bp fragment from all common variations of SOX2OT that have been already identified consist of SOX2OT, SOX2OT-S1 and SOX2OT-S2. The aim of this study was to analyze for the first time the expression regulation of ectopic expression of miR-375 with SOX2OT and SOX2 expressions in NT2 cells using RT-PCR. Transcription pattern of SOX2OT and SOX2 has been proposed to be regulated in the same manner in pluripotent stem cells such as NT2 cells (21). In this study, simultaneously decreased expressions of SOX2OT and SOX2 has been demonstrated along with miR-375 ectopic expression in NT2 cells that is in agreement with the hypothesis proposed by Amaral et al. (24) and Shahryari et al. on the possible role of SOX2OT in regulating SOX2 expression. Silencing of SOX2OT by specific siRNAs in previous study by Shahryari et al. (21), found a negative effect on SOX2 expression as well as a subG1/G1 cell cycle arrest in NT2 cells.
The observation of SOX2OT down-regulation following miR-375 over-expression in this study is in agreement with the cancer stem cell (CSC) theory in gastric carcinogenesis (28-30). However, our observation of Asonuma et al. (31) about inhibition of SOX2 by H. pylori through IL-4 axis and our finding suggest that more studies are necessary for elucidation of the exact mechanism of H. pylori carcinogenesis. According to the CSC theory in cancers, stem cells are the origin of initiation and development of a cancer. In gastric carcinoma, SOX2 is one of the master regulators of stem-cell pluripotency. Thus, H. pylori may affect the pluripotency of infected cells. The SOX2 gene is located in another gene named SOX2OT. We have recently demonstrated that SOX2 and SOX2OT are coregulated in esophageal squamous cell carcinoma (21). Askarian-Amiri et al. (2014) also have recently showed that these two genes are co regulated in breast cancer (32). There is no report on the relationship between gastric carcinoma and SOX2OT. This is the first work shows co regulation of SOX2 and SOX2OT in a stem cell model. Since SOX2 and SOX2OT bioinformatically are targets for miR-375, this finding maybe of importance for the elucidation of H. pylori pathogenesis because this miRNA is down-regulated in H. pylori infected cells and SOX2 is an important regulator of pluripotency.
Our finding shows that ectopic miR-375 in NT2 model stem cells down-regulates SOX2. The Also, the SOX2 gene is a transcription factor critically involved in the pluripotency and stemness of human embryonic stem cells (33). SOX2 has recently been identified as a common target of genomic amplifications and as an oncogene in cancers (34-36). In addition, SOX2 has been implicated in the function of putative cancer stem cells in tumors (37). It has been previously reported that immune system has the capacity to target SOX2 (38-40) and several groups have studied the presence of humoral responses against SOX2 in cancerous cells (40). On the other hand, miR-375 inhibits H. pylori-induced inflammation and carcinogenesis through JAK2/STAT signaling pathway (19). Our finding implicates that previously reported miR-375 down-regulation in H. pylori-induced inflammation and carcinogenesis (19) may result in SOX2 up-regulation through which cause H. pylori carcinogenesis. Further studies are necessary to establish this hypothesis and involved pathways.
This finding may broad our knowledge on ncRNAs and its possible role in cellular phenomena such as infectious diseases. We need further studies to demonstrate that whether the effect of miR-375 on SOX2 and SOX2OT can also exert by H. pylori infected cells or not. However, this findings maybe important to design new strategies for preventing H. pylori-induced-carcinogenesis and are in agreement with the result of Miao et al. (2014) (19).
In conclusion, our data provides for the first time an insight into the potential function of the human SOX2OT in regulating SOX2 expression and its possible regulation by miR-375. It also highlights a co regulation of SOX2OT, with key stem cell pluripotency gene, SOX2. Altogether, our data has suggested a regulatory mechanism for miR-375 that may govern the key stem-cell pluripotency gene, SOX2, mediated by the lncRNA SOX2OT.
Acknowledgments
The authors would like to thank Dr. Peter Andrews from Sheffield university, UK for his kindly providing NTERA-2 cells. The authors would like to express their gratitude to the secretariat of the 19th world congress on advances in oncology and 17th international symposium on molecular medicine for inviting the first author of this paper to give a talk on the importance of microRNA-lncRNA interactions in molecular medicine section of the parallel conferences.
Footnotes
Authors’ Contribution:Here we confirm that all authors participated in the study concept and design and contributed to the sections of the research: laboratory examinations, data interpretation, manuscript writing and revision.
Financial Disclosure:The authors declare that there is no conflict of interests to publish this article.
Funding/Support:This research was funded by national institute of genetic engineering and biotechnology (NIGEB), Tehran, Iran.
References
- 1.Noto JM, Peek RM. Helicobacter pylori: an overview. Methods Mol Biol. 2012;921:7–10. doi: 10.1007/978-1-62703-005-2_2. [DOI] [PubMed] [Google Scholar]
- 2.Zabaleta J. MicroRNA: A Bridge from H. pylori Infection to Gastritis and Gastric Cancer Development. Front Genet. 2012;3:294. doi: 10.3389/fgene.2012.00294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Noto JM, Gaddy JA, Lee JY, Piazuelo MB, Friedman DB, Colvin DC, et al. Iron deficiency accelerates Helicobacter pylori-induced carcinogenesis in rodents and humans. J Clin Invest. 2013;123(1):479–92. doi: 10.1172/JCI64373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhou A, Li S, Wu J, Khan FA, Zhang S. Interplay between microRNAs and host pathogen recognition receptors (PRRs) signaling pathways in response to viral infection. Virus Res. 2014;184:1–6. doi: 10.1016/j.virusres.2014.01.019. [DOI] [PubMed] [Google Scholar]
- 5.Staedel C, Darfeuille F. MicroRNAs and bacterial infection. Cell Microbiol. 2013;15(9):1496–507. doi: 10.1111/cmi.12159. [DOI] [PubMed] [Google Scholar]
- 6.Farazi TA, Spitzer JI, Morozov P, Tuschl T. miRNAs in human cancer. J Pathol. 2011;223(2):102–15. doi: 10.1002/path.2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cho WC. OncomiRs: the discovery and progress of microRNAs in cancers. Mol Cancer. 2007;6:60. doi: 10.1186/1476-4598-6-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Griffiths-Jones S. The microRNA Registry. Nucleic Acids Res. 2004;32(Database issue):D109–11. doi: 10.1093/nar/gkh023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Link A, Kupcinskas J, Wex T, Malfertheiner P. Macro-role of microRNA in gastric cancer. Dig Dis. 2012;30(3):255–67. doi: 10.1159/000336919. [DOI] [PubMed] [Google Scholar]
- 10.O'Connell RM, Rao DS, Baltimore D. microRNA regulation of inflammatory responses. Annu Rev Immunol. 2012;30:295–312. doi: 10.1146/annurev-immunol-020711-075013. [DOI] [PubMed] [Google Scholar]
- 11.Navarro L, Dunoyer P, Jay F, Arnold B, Dharmasiri N, Estelle M, et al. A plant miRNA contributes to antibacterial resistance by repressing auxin signaling. Science. 2006;312(5772):436–9. doi: 10.1126/science.1126088. [DOI] [PubMed] [Google Scholar]
- 12.Kiga K, Mimuro H, Suzuki M, Shinozaki-Ushiku A, Kobayashi T, Sanada T, et al. Epigenetic silencing of miR-210 increases the proliferation of gastric epithelium during chronic Helicobacter pylori infection. Nat Commun. 2014;5:4497. doi: 10.1038/ncomms5497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang T, Yu J, Zhang Y, Li L, Chen Y, Li D, et al. Salmonella enterica serovar enteritidis modulates intestinal epithelial miR-128 levels to decrease macrophage recruitment via macrophage colony-stimulating factor. J Infect Dis. 2014;209(12):2000–11. doi: 10.1093/infdis/jiu006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Archambaud C, Sismeiro O, Toedling J, Soubigou G, Becavin C, Lechat P, et al. The intestinal microbiota interferes with the microRNA response upon oral Listeria infection. MBio. 2013;4(6):e00707–13. doi: 10.1128/mBio.00707-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Arjomandzadegan M, Owlia P, Ranjbar R, Farazi AA, Sofian M, Sadrnia M, et al. Rapid and simple approach for identification of Mycobacterium tuberculosis and M. bovis by detection of regulatory gene whiB7. Acta Microbiol Immunol Hung. 2011;58(1):65–74. doi: 10.1556/AMicr.58.2011.1.7. [DOI] [PubMed] [Google Scholar]
- 16.Cremer TJ, Butchar JP, Tridandapani S. Francisella Subverts Innate Immune Signaling: Focus On PI3K/Akt. Front Microbiol. 2011;5:13. doi: 10.3389/fmicb.2011.00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cadamuro AC, Rossi AF, Maniezzo NM, Silva AE. Helicobacter pylori infection: host immune response, implications on gene expression and microRNAs. World J Gastroenterol. 2014;20(6):1424–37. doi: 10.3748/wjg.v20.i6.1424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Eulalio A, Schulte L, Vogel J. The mammalian microRNA response to bacterial infections. RNA Biol. 2012;9(6):742–50. doi: 10.4161/rna.20018. [DOI] [PubMed] [Google Scholar]
- 19.Miao L, Liu K, Xie M, Xing Y, Xi T. miR-375 inhibits Helicobacter pylori-induced gastric carcinogenesis by blocking JAK2-STAT3 signaling. Cancer Immunol Immunother. 2014;63(7):699–711. doi: 10.1007/s00262-014-1550-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Isomoto H, Matsushima K, Inoue N, Hayashi T, Nakayama T, Kunizaki M, et al. Interweaving microRNAs and proinflammatory cytokines in gastric mucosa with reference to H. pylori infection. J Clin Immunol. 2012;32(2):290–9. doi: 10.1007/s10875-011-9626-3. [DOI] [PubMed] [Google Scholar]
- 21.Shahryari A, Rafiee MR, Fouani Y, Oliae NA, Samaei NM, Shafiee M, et al. Two novel splice variants of SOX2OT, SOX2OT-S1, and SOX2OT-S2 are coupregulated with SOX2 and OCT4 in esophageal squamous cell carcinoma. Stem Cells. 2014;32(1):126–34. doi: 10.1002/stem.1542. [DOI] [PubMed] [Google Scholar]
- 22.Yoon JH, Abdelmohsen K, Gorospe M. Functional interactions among microRNAs and long noncoding RNAs. Semin Cell Dev Biol. 2014;34:9–14. doi: 10.1016/j.semcdb.2014.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alaei-Mahabadi B, Larsson E. Limited evidence for evolutionarily conserved targeting of long non-coding RNAs by microRNAs. Silence. 2013;4(1):4. doi: 10.1186/1758-907X-4-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Amaral PP, Neyt C, Wilkins SJ, Askarian-Amiri ME, Sunkin SM, Perkins AC, et al. Complex architecture and regulated expression of the Sox2ot locus during vertebrate development. RNA. 2009;15(11):2013–27. doi: 10.1261/rna.1705309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jeggari A, Marks DS, Larsson E. miRcode: a map of putative microRNA target sites in the long non-coding transcriptome. Bioinformatics. 2012;28(15):2062–3. doi: 10.1093/bioinformatics/bts344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Suganuma M, Yamaguchi K, Ono Y, Matsumoto H, Hayashi T, Ogawa T, et al. TNF-alpha-inducing protein, a carcinogenic factor secreted from H. pylori, enters gastric cancer cells. Int J Cancer. 2008;123(1):117–22. doi: 10.1002/ijc.23484. [DOI] [PubMed] [Google Scholar]
- 27.Ding L, Xu Y, Zhang W, Deng Y, Si M, Du Y, et al. MiR-375 frequently downregulated in gastric cancer inhibits cell proliferation by targeting JAK2. Cell Res. 2010;20(7):784–93. doi: 10.1038/cr.2010.79. [DOI] [PubMed] [Google Scholar]
- 28.Belair C, Baud J, Chabas S, Sharma CM, Vogel J, Staedel C, et al. Helicobacter pylori interferes with an embryonic stem cell micro RNA cluster to block cell cycle progression. Silence. 2011;2(1):7. doi: 10.1186/1758-907X-2-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kountouras J, Kapetanakis N, Zavos C, Polyzos SA, Romiopoulos I, Tsiaousi E, et al. Helicobacter pylori might contribute to cancer and/or bone marrow-derived stem cell-related gastrointestinal oncogenesis. Oncogene. 2014;34(5):670. doi: 10.1038/onc.2013.602. [DOI] [PubMed] [Google Scholar]
- 30.Bessede E, Staedel C, Acuna Amador LA, Nguyen PH, Chambonnier L, Hatakeyama M, et al. Helicobacter pylori generates cells with cancer stem cell properties via epithelial-mesenchymal transition-like changes. Oncogene. 2014;33(32):4123–31. doi: 10.1038/onc.2013.380. [DOI] [PubMed] [Google Scholar]
- 31.Asonuma S, Imatani A, Asano N, Oikawa T, Konishi H, Iijima K, et al. Helicobacter pylori induces gastric mucosal intestinal metaplasia through the inhibition of interleukin-4-mediated HMG box protein Sox2 expression. Am J Physiol Gastrointest Liver Physiol. 2009;297(2):G312–22. doi: 10.1152/ajpgi.00518.2007. [DOI] [PubMed] [Google Scholar]
- 32.Askarian-Amiri ME, Seyfoddin V, Smart CE, Wang J, Kim JE, Hansji H, et al. Emerging role of long non-coding RNA SOX2OT in SOX2 regulation in breast cancer. PLoS One. 2014;9(7):e23464. doi: 10.1371/journal.pone.0102140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lodato MA, Ng CW, Wamstad JA, Cheng AW, Thai KK, Fraenkel E, et al. SOX2 co-occupies distal enhancer elements with distinct POU factors in ESCs and NPCs to specify cell state. PLoS Genet. 2013;9(2):e23464. doi: 10.1371/journal.pgen.1003288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bass AJ, Watanabe H, Mermel CH, Yu S, Perner S, Verhaak RG, et al. SOX2 is an amplified lineage-survival oncogene in lung and esophageal squamous cell carcinomas. Nat Genet. 2009;41(11):1238–42. doi: 10.1038/ng.465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hussenet T, du Manoir S. SOX2 in squamous cell carcinoma: amplifying a pleiotropic oncogene along carcinogenesis. Cell Cycle. 2010;9(8):1480–6. doi: 10.4161/cc.9.8.11203. [DOI] [PubMed] [Google Scholar]
- 36.McCaughan F, Pole JC, Bankier AT, Konfortov BA, Carroll B, Falzon M, et al. Progressive 3q amplification consistently targets SOX2 in preinvasive squamous lung cancer. Am J Respir Crit Care Med. 2010;182(1):83–91. doi: 10.1164/rccm.201001-0005OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Nakatsugawa M, Takahashi A, Hirohashi Y, Torigoe T, Inoda S, Murase M, et al. SOX2 is overexpressed in stem-like cells of human lung adenocarcinoma and augments the tumorigenicity. Lab Invest. 2011;91(12):1796–804. doi: 10.1038/labinvest.2011.140. [DOI] [PubMed] [Google Scholar]
- 38.Spisek R, Kukreja A, Chen LC, Matthews P, Mazumder A, Vesole D, et al. Frequent and specific immunity to the embryonal stem cell-associated antigen SOX2 in patients with monoclonal gammopathy. J Exp Med. 2007;204(4):831–40. doi: 10.1084/jem.20062387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dhodapkar MV, Dhodapkar KM. Spontaneous and therapy-induced immunity to pluripotency genes in humans: clinical implications, opportunities and challenges. Cancer Immunol Immunother. 2011;60(3):413–8. doi: 10.1007/s00262-010-0944-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gure AO, Stockert E, Scanlan MJ, Keresztes RS, Jager D, Altorki NK, et al. Serological identification of embryonic neural proteins as highly immunogenic tumor antigens in small cell lung cancer. Proc Natl Acad Sci U S A. 2000;97(8):4198–203. doi: 10.1073/pnas.97.8.4198. [DOI] [PMC free article] [PubMed] [Google Scholar]