Abstract
Methylenetetrahydrofolate reductase (MTHFR) c.677C>T and c.1298A>C variants were known to be associated with prostate cancer (PCa) risk with conflicting results, because of MTHFR and nutrient status interaction in the prostate development. In this large-scale, hospital-based, case-control study of 1817 PCa cases and 2026 cancer-free controls, we aimed to clarify the association between these two MTHFR variants and PCa risk in Shanghai and to explore the underlying molecular mechanisms. We found that both the heterozygous CT (adjusted OR = 0.78, 95% CI: 0.67–0.92) and the homozygous TT genotypes (adjusted OR = 0.68, 95% CI: 0.55–0.83) of c.677C>T were associated with a significantly decreased risk of PCa compared with homozygous wild-type CC genotype, respectively, using multivariate logistic regression. Furthermore, we confirmed that MTHFR c.677T allele was related to an increased serum homocysteine level in the Han Chinese population in Shanghai. In the cultured PCa cell lines, we observed that MTHFR c.677T could elevate the cellular homocysteine level and cause DNA damage, thus increasing cell apoptosis and finally inhibiting cell proliferation. In conclusion, MTHFR c.677T was a protective factor of PCa risk in ethnic Han Chinese males by inducing DNA damage and cell apoptosis.
Prostate cancer (PCa) is the second most frequently diagnosed cancer and the fifth leading cause of cancer death worldwide among men, and remains the first frequently diagnosed cancer in more developed countries1. Although PCa incidence is much lower than in Western countries, it is still in the top 10 most common cancers in Chinese males2. The increased incidence rate of PCa in China was due to prolonged life expectancy, dietary changes, more PSA testing and Westernized lifestyle3. The PCa age-standardized incidence (12.96 per 100,000) and age-standardized mortality (4.97 per 100,000) in Shanghai were the highest among all provinces of China4. This phenomenon aroused many researchers’ interest. Higher economic development level, better medical level and being more receptive to western culture, lifestyle and diet may partly explain the difference5, but specific connections remained unknown.
PCa is a complex disease; and many etiological factors, including genetic profile, nutrition, environmental exposures, etc., are thought to play important roles in cancer development6. It is widely acknowledged that genetic polymorphisms may act as a predictors of some characteristics of PCa, including incidence rate7,8,9, pathology10, progression11, etc.
Folates are cofactors and cosubstrates for biological methylation and nucleic acid synthesis and also act as regulatory molecules. The folate-mediated, one-carbon metabolic pathway is fundamental to DNA synthesis, repair and methylation12; and plays an important role in many types of diseases, such as cancers, birth defects and cardiovascular diseases13. Over the past decades, a series of clinical studies showed that the concentration of homocysteine, folate and vitamin B12—the key metabolites in the folate pathway—were related to the occurrence of multiple types of cancers, including PCa14, breast cancer15, and gastric cancer16. Numerous studies reported that increasing plasma homocysteine level was associated with a reduced PCa risk17,18. Thus, extensive analyses of genes involved in the folate metabolism pathway have been conducted. The methylenetetrahydrofolate reductase (MTHFR) gene encodes an enzyme that catalyzes the irreversible conversion of 5′,10′-methylenetetrahydrofolate to 5-methyltetrahydro folate, which serves as a methyl donor in the remethylation of homocysteine to methionine. Until now, the MTHFR gene has been identified to possess 98 different variants with benign, pathogenic or unknown significance which are listed on ClinVar database (http://www.ncbi.nlm.nih.gov/clinvar/). Among these variants, rs1801133 (c. 677C>T) and rs1801131 (c.1298A>C) are two most extensively reported polymorphisms with a global minor allele frequency (GMAP) 0.245 and 0.249, respectively19,20. These two variants have shown to be associated with the risk of PCa, although somewhat conflicting results have been reported21,22,23,24.
Wu et al. reported a significant reduction of PCa risk in yellow people who carried c.677T variant in Taiwan25, and many groups achieved similar results in other ethnic regions26,27. However, a recent meta-analysis found no significant association between the those variants and PCa risk21, and subgroup analysis indicated that the association between variants and PCa risk may vary among ethnic groups. In this meta-analysis, only two studies enrolled focused on the ethnic Han Chinese population. Wu et al. performed study in Taiwan while Cai et al. performed study in the Liaoning Province, both far different regions from Shanghai. There was a limited sample size in the previous study regarding MTFHR variants in Han Chinese PCa susceptibility; and more importantly, this association has not been surveyed in Shanghai city, which had the highest PCa incidence in China. The current study aimed to investigate the association between these two common variants of MTHFR and PCa risk in a large-scale, hospital-based, case-control cohort in Shanghai city and to explore the underlying molecular mechanisms.
Results
Characteristics of the study population
The demographic characteristics of the 1817 PCa patients and 2026 normal controls were presented in Table 1. Since we performed a frequency match between PCa patients and cancer-null controls, the age of diagnosis between the two groups were well matched with a mean age of 66.7 in the PCa patients group and a mean age of 66.9 in the control group. Besides, there were no significant differences in the distribution between the two groups in the following indexes: BMI, hypertension, diabetes mellitus, and cardiovascular diseases. Apparently, serum PSA level was significantly higher in the PCa patients group (28.5 ng/ml) due to the nature of PCa and the filtration we performed before enrollment. Among all PCa patients, 605 patients (33.3%) had a Gleason score ≥ 8, 526 patients (32.2%) had extracapsular extension, 154 patients (8.5%) had lymph node involvement and 352 patients (19.4%) had positive surgical margins.
Table 1. Distribution of demographic and clinicopathological characteristics of 1817 PCa patients and 2026 controls included in the study.
| Variables | Cases (n = 1817) | Controls (n = 2026) | P |
|---|---|---|---|
| Age (yr), mean ± SD | 66.7 ± 7.2 | 66.9 ± 6.8 | 0.437 |
| BMI (kg/m2), n (%) | 0.877 | ||
| <25 | 1308 (72.0) | 1463 (72.2) | |
| ≥25 | 509 (28.0) | 563 (27.8) | |
| Hypertension, n (%) | 0.424 | ||
| No | 1054 (58.0) | 1201 (59.3) | |
| Yes | 763 (42.0) | 825 (40.7) | |
| Cardiovascular disease, n (%) | 0.631 | ||
| No | 1660 (91.4) | 1842 (90.9) | |
| Yes | 157 (8.6) | 184 (9.1) | |
| Diabetes mellitus, n (%) | 0.926 | ||
| No | 1636 (90.0) | 1826 (90.1) | |
| Yes | 181 (10.0) | 200 (9.9) | |
| PSA (ng/mL), mean ± SD | 28.5 ± 1.3 | 1.2 ± 0.3 | <0.001 |
| Gleason score, n (%) | |||
| ≤6 | 289 (15.9) | ||
| 7 | 923 (50.8) | ||
| ≥8 | 605 (33.3) | ||
| Pathological tumor stage, n (%) | |||
| T2 | 1231 (67.7) | ||
| T3a | 160 (8.8) | ||
| T3b | 426 (23.4) | ||
| Lymph node involvement, n (%) | 154 (8.5) | ||
| Positive surgical margins, n (%) | 352 (19.4) |
PCa, prostate cancer.
Association between two MTHFR gene single nucleotide polymorphisms (SNPs) and PCa risk
The genotype frequencies and their associations with the risk of PCa were summarized in Table 2. All the genotype frequencies of the two selected MTHFR gene SNPs among controls met the criteria of Hardy-Weinberg Equilibrium (all P > 0.05). We found a significantly different genotype frequency distribution of rs1801133 (C677T) between the PCa group and the cancer-free group (P < 0.001), while no different genotype frequency distribution of rs1801131 (A1298C) was observed between cases and controls (P = 0.690). Both the heterozygous CT genotype (crude OR = 0.79, 95% CI: 0.68–0.91) and the homozygous TT genotype (crude OR = 0.65, 95% CI: 0.54–0.78) of rs1801133 were associated with a significantly decreased risk of PCa, compared with homozygous wild-type CC genotype, respectively. In addition, the relationship between the two MTHFR genotypes and the risk of PCa was evaluated using dominant models, recessive models and additive models, respectively, with all three models achieving statistical significance; which indicated that people who carried T allele of rs1801133 had a lower risk of developing PCa. Besides, no clear association between genotype frequency of rs1801131 and PCa risk was observed. Furthermore, we used multivariate logistic regression analyses to reanalyze the above-mentioned genotypes after adjusting for age at diagnosis, BMI, hypertension, diabetes mellitus, and cardiovascular disease. We found that all of the genotypes maintained the same relationship with PCa risk as declared above in a univariate logistic regression model. Finally, we found a 32% decreased risk of PCa for people who carried homozygous TT genotype of rs1801133 (adjusted OR = 0.68, P < 0.001) compared with those who carried homozygous wild-type CC genotype.
Table 2. Association between genetic polymorphisms in folate metabolism genes and PCa risk in Han Chinese men.
| Gene | SNP | Type | Genotype | Cases (n = 1817) | Controls (n = 2026) | PHWE | Crude OR (95% CI) | P | Adjusted OR (95% CI)c | Pa |
|---|---|---|---|---|---|---|---|---|---|---|
| MTHFR | rs1801133 | Nonsynonymous (exon 4) | CC | 654 (36.0) | 599 (29.6) | 0.580 | 1.00 | 1 × 10−5 | 1.00 | 3 × 10−4 |
| CT | 876 (48.2) | 1022 (50.4) | 0.79 (0.68–0.91) | 0.78 (0.67–0.92) | ||||||
| TT | 287 (15.8) | 405 (20.0) | 0.65 (0.54–0.78) | 0.68 (0.55–0.83) | ||||||
| Dominant model | 0.75 (0.65–0.85) | 2 × 10−5 | 0.75 (0.65–0.87) | 2 × 10−4 | ||||||
| Recessive model | 0.75 (0.64–0.89) | 7 × 10−4 | 0.78 (0.65–0.94) | 0.008 | ||||||
| Additive model | 0.80 (0.73–0.88) | 6 × 10−5 | 0.82 (0.74–0.90) | 1 × 10−4 | ||||||
| rs1801131 | Nonsynonymous (exon 7) | AA | 1192 (65.6) | 1355 (66.9) | 0.220 | 1.00 | 0.690 | 1.00 | 0.680 | |
| AC | 569 (31.3) | 609 (30.1) | 1.06 (0.92–1.22) | 1.07 (0.92–1.24) | ||||||
| CC | 56 (3.1) | 62 (3.1) | 1.03 (0.71–1.49) | 1.06 (0.72–1.58) | ||||||
| Dominant model | 1.06 (0.93–1.21) | 0.400 | 1.07 (0.92–1.24) | 0.380 | ||||||
| Recessive model | 1.01 (0.70–1.45) | 0.970 | 1.04 (0.70–1.55) | 0.830 | ||||||
| Additive model | 1.05 (0.93–1.17) | 0.460 | 1.06 (0.93–1.20) | 0.410 | ||||||
PCa, prostate cancer; OR, odds ratio; 95%CI, 95% confidence interval.
HWEP value for the Hardy–Weinberg equilibrium test in controls subjects.
aAdjusted for age, BMI, hypertension, diabetes mellitus and cardiovascular disease in multivariant logistic regression models.
Stratification analysis
We performed a stratified analysis to evaluate the association between SNPs mentioned above and PCa risk in different subgroups using a recessive model. Analytic results were presented in Table 3. In multivariate logistic regression analyses, we found that the homozygous TT genotype of rs1801133 was significantly associated with a decreased risk of PCa risk in subgroups of patients with a Gleason score ≥8, patients with extracapsular extension, patients with seminal vesicle invasion, and patients with or without lymph node involvement. The results were all further supported by a homogeneity test using the χ2-based Q test (all P < 0.05). Besides, a decreased risk of PCa was observed among subgroups age >68 years, a BMI of <25 kg/m2, with hypertension, without diabetes mellitus and without cardiovascular disease for patients carrying MTHFR rs1801133 TT genotype; and an increased risk of PCa was observed among subgroups with cardiovascular disease with lymph node involvement and those carrying the MTHFR rs1801131 CC genotype, further homogeneity tests didn’t support the differences in PCa risk found between these strata.
Table 3. Stratified analysis for associations between genetic polymorphisms in folate metabolism genes and PCa risk by recessive genetic model in Han Chinese men.
| Variables | rs1801133 (cases/controls) |
Adjusted ORa (95% CI) | P | Phom | rs1801131 (cases/controls) |
Adjusted ORa (95% CI) | P | Phom | ||
|---|---|---|---|---|---|---|---|---|---|---|
| CC/CT | TT | AA/AC | CC | |||||||
| Age (yr), median | ||||||||||
| ≤68 | 850/1053 | 167/252 | 0.86 (0.67–1.11) | 0.250 | 0.194 | 985/1260 | 32/45 | 0.93 (0.53–1.61) | 0.780 | 0.390 |
| >68 | 680/568 | 120/153 | 0.67 (0.48–0.92) | 0.014 | 776/704 | 24/17 | 1.19 (0.57–2.52) | 0.640 | ||
| BMI (kg/m2) | ||||||||||
| <25 | 1112/1178 | 196/285 | 0.73 (0.60–0.89) | 0.002 | 0.596 | 1263/1424 | 45/39 | 1.30 (0.84–2.02) | 0.230 | 0.034 |
| ≥25 | 418/443 | 91/120 | 0.82 (0.60–1.11) | 0.190 | 498/540 | 11/23 | 0.53 (0.25–1.09) | 0.076 | ||
| Hypertension | ||||||||||
| No | 887/975 | 167/226 | 0.87 (0.68–1.11) | 0.250 | 0.276 | 1023/1166 | 31/35 | 0.94 (0.54–1.64) | 0.830 | 0.982 |
| Yes | 643/646 | 120/179 | 0.66 (0.49–0.90) | 0.007 | 738/798 | 25/27 | 1.34 (0.73–2.45) | 0.340 | ||
| Diabetes mellitus | ||||||||||
| No | 1382/1467 | 254/359 | 0.77 (0.63–0.94) | 0.008 | 0.982 | 1584/1771 | 52/55 | 1.07 (0.71–1.63) | 0.740 | 0.427 |
| Yes | 148/154 | 33/46 | 0.78 (0.43–1.42) | 0.410 | 177/193 | 4/7 | 0.55 (0.13–2.40) | 0.420 | ||
| Cardiovascular disease | ||||||||||
| No | 1399/1483 | 261/359 | 0.80 (0.66–0.98) | 0.027 | 0.671 | 1611/1783 | 49/59 | 0.92 (0.61–1.40) | 0.700 | 0.123 |
| Yes | 131/150 | 26/34 | 0.73 (0.35–1.52) | 0.390 | 150/181 | 7/3 | 6.49 (1.08–39.01) | 0.028 | ||
| Gleason score | ||||||||||
| ≤7 | 997/1621 | 215/405 | 0.94 (0.76–1.16) | 0.560 | 0.005 | 1177/1964 | 35/62 | 1.05 (0.66–1.68) | 0.820 | 0.570 |
| ≥8 | 533/1621 | 72/405 | 0.53 (0.39–0.71) | 3 × 10−5 | 584/1964 | 21/62 | 1.03 (0.57–1.84) | 0.930 | ||
| Extracapsular extension | ||||||||||
| No | 1018/1621 | 213/405 | 0.94 (0.77–1.16) | 0.590 | 0.025 | 1194/1964 | 37/62 | 1.09 (0.68–1.74) | 0.720 | 0.818 |
| Yes | 512/1621 | 74/405 | 0.51 (0.38–0.70) | 8 × 10−5 | 567/1964 | 19/62 | 1.10 (0.61–2.00) | 0.750 | ||
| Seminal vesicle invasion | ||||||||||
| No | 1154/1621 | 237/405 | 0.92 (0.76–1.12) | 0.420 | 0.018 | 1351/1964 | 40/62 | 1.03 (0.65–1.61) | 0.910 | 0.433 |
| Yes | 376/1621 | 50/405 | 0.43 (0.29–0.62) | 2 × 10−5 | 410/1964 | 16/62 | 1.21 (0.63–2.32) | 0.560 | ||
| Positive surgical margin | ||||||||||
| No | 1234/1621 | 231/405 | 0.82 (0.67–1.00) | 0.045 | 0.953 | 1419/1964 | 46/62 | 1.02 (0.67–1.56) | 0.920 | 0.796 |
| Yes | 296/1621 | 56/405 | 0.69 (0.47–1.00) | 0.046 | 342/1964 | 10/62 | 1.27 (0.57–2.82) | 0.570 | ||
| Lymph node involvement | ||||||||||
| No | 1382/1621 | 281/405 | 0.87 (0.72–1.04) | 0.130 | <0.001 | 1616/1964 | 47/62 | 0.92 (0.61–1.40) | 0.700 | 0.067 |
| Yes | 148/1621 | 6/405 | 0.13 (0.05–0.32) | 1 × 10−6 | 145/1964 | 9/62 | 3.38 (1.41–8.13) | 0.011 | ||
PCa, prostate cancer; OR, odds ratio; 95%CI, 95% confidence interval.
aAdjusted for age, BMI, hypertension, diabetes mellitus, and cardiovascular disease in multivariant logistic regression models.
homP value for homogeneity test using the χ2-based Q-test.
Variants of MTHFR alter plasma and cellular homocysteine level
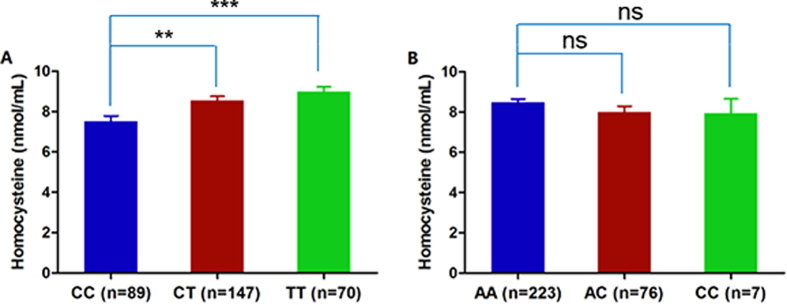
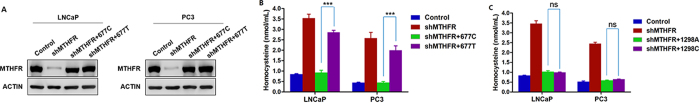
MTHFR is an essential enzyme in one-carbon metabolism, producing methyl donor for the conversion of homocysteine to methionine. Thus, variants of MTHFR may alter enzymatic function and change the serum homocysteine level. To test this hypothesis, we randomly selected 306 participants from a control group and detected the serum homocysteine levels. Among the 306 participants, 89 (29.1%) carried the wild-type CC genotype of rs1801133, 147 (48.0%) carried the heterozygous CT genotype and 70 (22.9%) carried the homozygous TT genotype. We found that people with the heterozygous CT genotype or homozygous TT genotype had a significantly higher serum homocysteine level compared with wild-type CC carriers (Fig. 1A). However, both heterozygous AC genotype and homozygous CC genotype of rs1801131 had no significant effects on plasma homocysteine concentration (Fig. 1B). Furthermore, we set up MTHFR-knockdown LNCaP and PC3 cell lines which were validated by western blotting (Fig. 2A) and tested cellular homocysteine level. It showed that both in LNCaP and PC3 cell lines, homocysteine level in MTHFR-knockdown cells was significantly higher than in control cells (Fig. 2B,C). Then, we overexpressed c.677C MTHFR, c.677T MTHFR, c.1298A MTHFR and c.1298C MTHFR, respectively, in MTHFR-knockdown LNCaP and PC3 cell lines. We found that that the cellular homocysteine level of c.677T MTHFR-overexpressed cells was significantly higher than that of wild-type MTHFR-overexpressed cells (Fig. 2B). However, we did not observe a significant difference in cellular homocysteine levels between c.1298C MTHFR-overexpressed cells and wild-type MTHFR-overexpressed cells (Fig. 2C).
Figure 1. MTHFR c.677T was related to homocysteine level in the Han Chinese population.
(A) Both heterozygous CT genotype and homozygous TT genotype of rs1801133 are significantly related with a higher serum homocysteine level. (B) Neither heterozygous AC genotype nor homozygous CC genotype of rs1801131 is related with a serum homocysteine level. **Indicates P < 0.01, ***indicates P < 0.001.
Figure 2. MTHFR c.677T variant leads to a notably increased cellular homocysteine level in PCa cell lines.
(A) Knockdown efficiency of shMTHFR and protein expression of MTHFR after restoration of wild-type or mutant MTHFR gene was measured by western blot. The full-length blots were displayed in Supplementary Figure 1. (B) c.677T variant of MTHFR gene leads to an increased cellular homocysteine level in both LNCaP and PC3 cell lines. (C) c.1298C variant of MTHFR gene has no effect on the cellular homocysteine level in LNCaP and PC3 cell lines. ***Indicates P < 0.001, ns indicates no significance.
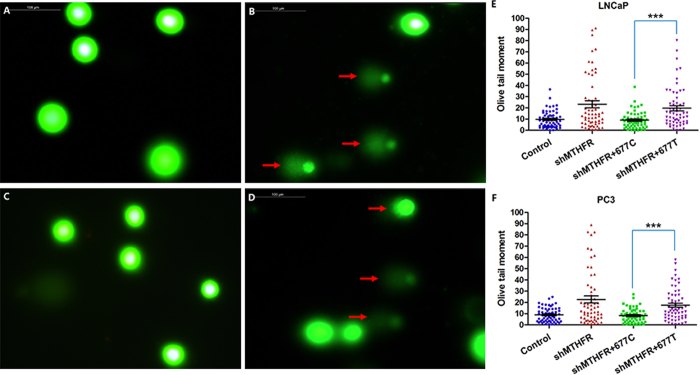
MTHFR c.677T causes DNA damage
We performed Comet Assay to assess DNA damage. In normal cells, the fluorescence is confined mostly to the nucleus because undamaged DNA cannot migrate. In cells with DNA damage, DNA is denatured by the alkali solution used for single-strand break detection or the neutral solution used for double-strand break detection. The negatively charged DNA fragments are then released from the nucleus and migrate toward the anode. The results of control cells and MTHFR-knockdown cells were presented in Fig. 3A,B. The results of wild-type MTHFR-overexpression and c.677T MTHFR-overexpression in MTHFR-knockdown cell lines were presented in Fig. 3C,D. It showed that MTHFR knockdown could lead to DNA damage. Overexpression of wild-type MTHFR in these cells could repair DNA damage, but overexpression of c.677T MTHFR could not. Quantitative Comet Assay results were presented in Fig. 3E,F, which showed that c.677T variant of MTHFR could lead to DNA damage (P < 0.001). Overexpression of c.1298A and c.1298C MTHFR in MTHFR-knockdown cell lines both could repair DNA damage (data not shown).
Figure 3. MTHFR c.677T variant causes DNA damage.
(A) Comet Assay results in control cells. (B) Comet Assay results in MTHFR-knockdown cells, indicating that down-regulation of MTHFR leads to DNA damage. (C) Comet Assay results in MTHFR-knockdown + wild-type overexpressed cells, indicating that overexpression of 677C-MTHFR can reduce DNA damage. (D) Comet Assay results in MTHFR-knockdown + 677T-MTHFR overexpressed cells, indicating that overexpression of 677T-MTHFR cannot reduce DNA damage. (E) Quantitative Comet Assay results in LNCaP cells, indicating that c.677T variant might cause DNA damage compared with wild-type MTHFR. (F) Same results are achieved in PC3 cell lines. Red arrow points out the tails, which indicate DNA damage.
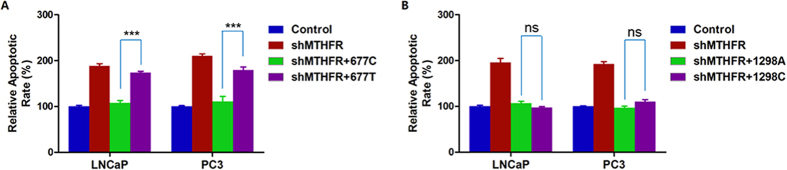
MTHFR c.677T variant increases cell apoptosis
We tested cell apoptosis in four cell groups (control, MTHFR-knockdown, shMTHFR + wild-type MTHFR and shMTHFR + c.677T/c.1298C MTHFR) in LNCaP and PC3 cell lines, respectively. We found that the c.677T variant of MTHFR significantly increased cell apoptosis compared with wild-type genotype in both cell lines (Fig. 4A). However, we did not observe a significant difference between the c.1298C variant and wild-type MTHFR overexpressed cell lines (Fig. 4B).
Figure 4. MTHFR c.677T variant increases cell apoptosis.
(A) c.677T variant of MTHFR increases cell apoptosis remarkably in both LNCaP and PC3 cell lines. (B) c.1298C variant of MTHFR has no effect on cell apoptosis. ***Indicates P < 0.001, ns indicates no significance.
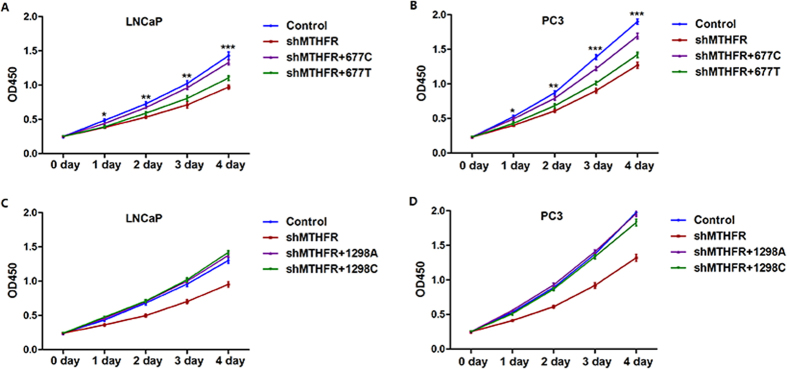
MTHFR c.677T variant inhibits cell proliferation
CCK-8 assays were performed to evaluate cell proliferation in the above-mentioned four cell groups in LNCaP and PC3 cell lines, respectively. We found that the c.677T variant of MTHFR could inhibit cell proliferation in both cell lines, compared with wild-type MTHFR (Fig. 5A,B). In addition, we did not find any effect on cell proliferation in c.1298C variant overexpressed cell lines, compared with wild-type MTHFR overexpressed cell lines (Fig. 5C,D).
Figure 5. MTHFR c.677T variant inhibits cell proliferation.
(A) c.677T variant of MTHFR inhibits cell proliferation significantly compared with wild-type MTHFR in LNCaP cells. (B) c.677T variant of MTHFR inhibits cell proliferation significantly compared with wild-type MTHFR in PC3 cells. (C) c.1298C variant of MTHFR has no significant effect on cell proliferation compared with wild-type MTHFR in LNCaP cells. (D) c.1298C variant of MTHFR has no significant effect on cell proliferation compared with wild-type MTHFR in PC3 cells. *Indicates P < 0.05, **indicates P < 0.01, ***indicates P < 0.001, all comparisons were performed between groups of shMTHFR + wild-type MTHFR and shMTHFR + c.677T/c.1298C MTHFR.
Discussion
We performed a large-scale, hospital-based, case-controlled study in Han Chinese men native to Shanghai to determine whether MTHFR c.677C>T and c.1298A>C were associated with PCa risk. We found that both heterozygous CT genotype and homozygous TT genotype carriers of rs1801133 had a significantly lower risk of developing PCa compared with those carrying wild-type CC genotype, which is in agreement with previous studies performed in Taiwanese patients25 and the Liaoning Province28. In addition, after stratification analysis using a recessive model, we found that homozygous TT genotype of rs1801133 was clearly associated with a decreased risk of PCa in patients with Gleason score ≥8, patients with extracapsular extension, patients with seminal vesicle invasion and patients with or without lymph node involvement. Further functional analyses revealed that MTHFR c.677T contributed to elevating homocysteine level, increasing DNA damage and cell apoptosis, and inhibiting proliferation of PCa cells.
Variants of MTHFR, which is a critical enzyme for intracellular folate homeostasis and metabolism, were reported to be associated with elevated circulating homocysteine levels and many diseases including PCa. Most of the studies focusing on the relationship between PCa and SNPs of MTHFR paid close attention to MTHFR polymorphism and PCa risk and have conflicting results. A recent meta-analysis which enrolled 22 studies revealed that the MTHFR C677T polymorphism did not contribute to the risk of PCa in the overall population of Caucasians, but indicated that it may play a role in PCa development in Asian males23. Another recently published meta-analysis which included over 20,000 participants declared that the C677T CT polymorphism of MTHFR may be a risk factor of PCa in East Asians, and the association between A1298C variant and PCa risk may vary in different ethnic populations21. Clearly, these results indicated that the function of these two common MTHFR polymorphisms may vary in different ethnic populations, which suggested that further, large-scale research in different regions and races was needed. Furthermore, among all of the studies enrolled by these meta-analysis publications, only the studies performed by Wu25 and Cai28 focused on Asian males of Mongoloid race. Wu et al. performed studies in Taiwan while Cai et al. performed studies in the Liaoning province, regions in different latitudes. It’s reported that activity and variant distribution of MTHFR might vary among regions in different latitudes and UV-exposure levels29,30. On the other hand, ethnic Han Chinese people account for over 92% of the population in China, with more than 1 billion people. Hence, genetic background varied immensely among people in different regions. Furthermore, sample sizes were relatively small in Wu’s and Cai’s research. In the present large-scale study from Shanghai, we confirmed that T allele of rs1801133 may be a protective factor of PCa and rs1801131 may have no significant association with PCa risk.
It has long been recognized that deficiency of folate may increase the incidence of cancer and many other diseases. Deficiency of folate contributes to a higher homocysteine level, thus leading to increased cytosolic calcium and reactive oxygen species (ROS)31. Also, DNA damage and chromosome aberrations were induced via ROS-mediated signaling pathways32. Moreover, the methylation of cytosine in DNA will be reduced when there is a folate deficiency, and will eventually cause an aberrant expression of pro-oncogene and a potential malignant transformation in carcinogenesis33. MTHFR is directly involved in the metabolism of folate and is considered to have direct consequences to cancer incidence if the enzymatic function is partly lost. It’s reported that the homozygous status of these two common variants of the MTHFR gene (rs1801133 and rs1801131) will cause about a 70% and 40% reduction of enzyme function compared with wild-type genotype, respectively20. Therefore, it was ironic to find that T allele of C677T polymorphism of the MTHFR gene was a protective factor of PCa risk. However, some research groups found that the 677TT genotype of the MTHFR gene might allow the expression of tumor suppressor genes and reduce the risk of cancer development while it partly caused enzymatic dysfunction34. Furthermore, MTHFR inhibition was proven to arrest the growth of cancer cells in in vitro studies because of limited methionine supply35. Cell apoptosis could also be induced by an increased ROS level and could play a favorable role in cancer cells36. These findings may partly support our results, and we still need more basic, functional research to determine the exact function of MTHFR variants in PCa carcinogenesis, development, and progression.
We acknowledged that there were some limitations in our study design and performance. Firstly, there may be some biases from samples collection in this hospital-based, case-controlled study. Although these biases could be minimized by taking into consideration confounding factors and age-matching between cases and controls. Secondly, in the stratified analysis, the case number in subgroups was relatively small and it would decrease the statistical power to examine the genetic variants with regard to PCa risk. Thirdly, folate intake, which is an important factor affecting the function of MTHFR polymorphism on PCa risk, was another condition not available from participants. Finally, potential mechanisms of the long-distance regulatory effect of the two studied MTHFR polymorphisms on the expression of MTHFR was not explored in-depth, which warrants further investigation in our future study. Notwithstanding these limitations, our research underscores that ethnic Han Chinese males carrying MTHFR c.667T allele had a significantly lower risk for developing PCa compared with those with wild-type MTHFR genotype of rs1801133. Further functional analyses revealed that the MTHFR c.677T contributes to elevating homocysteine level, increasing DNA damage and cell apoptosis, and inhibiting proliferation of PCa cells, thereby decreasing PCa risk.
In conclusion, our findings indicated that MTHFR c.677T variant acts as a protective factor of PCa risk by elevating homocysteine level, increasing DNA damage and cell apoptosis, and inhibiting proliferation of PCa cells. Further research focusing on gene-gene or gene-environment interactions should be conducted to explore more legible mechanisms of the role MTHFR plays in PCa risk.
Materials and Methods
Study population
This hospital-based, case-controlled study recruited 1817 newly diagnosed PCa patients, and 2026 matched cancer-null controls from genetically unrelated ethnic Han Chinese men treated at Fudan University Shanghai Cancer Center from January 2008 to June 2015. Two well-trained pathologists, specializing in genitourinary cancer, assessed the histological type of the PCa patients independently. All of the cases were defined as primary prostate adenocarcinoma according to the WHO criteria for PCa. We excluded patients with malignancies other than primary PCa, patients with a family history of PCa, and patients who received chemotherapy or radiotherapy before enrollment. Tumor stage was assessed and classified according to the TNM classification system provided by the American Joint Committee on Cancer (AJCC) in 201037. The Gleason score system was employed to determine histological grading of the PCa patients. We collected patients’ clinical and pathological information at the time they were enrolled, including general clinical features (age at diagnosis, height, weight, circulating PSA level), characteristics at surgery (tumor grade, tumor stage, surgical margin status and lymph node involvement) and comorbidities (hypertension, cardiovascular disease and diabetes mellitus).
Frequency-match was performed in the 2026 cancer-null Chinese men by age at diagnosis and geographic area according to the PCa patients’ characteristics. Since serum PSA screening and digital rectal examination were not routinely performed in China, we advised people with urinary tract symptoms to have a serum PSA measurement and a digital rectal examination. Participants with a serum PSA level >4 ng/ml, with or without an abnormal digital rectal examination, were also excluded from the control database.
All of the study designs and testing procedures were performed according to the ethical standards of the Helsinki Declaration II and approved by the Scientific and Ethical Committee of Fudan University Shanghai Cancer Center. Written informed consents were obtained from all participants before any study-specific investigation was performed.
SNP identification and genotyping
We extracted genomic DNA from peripheral leukocytes using the Qiagen Blood DNA Mini Kit (Qiagen Inc., Valencia, CA, USA) following the standard protocols. Our selected SNPs of MTHFR gene, rs1801133 (NM_005957.4:c.665C>T; also known as C677T) and rs1801131 (NM_005957.4:c.1286A>C; also known as A1298T), were all located in exon region and were genotyped by SNaPshot analysis (ABI). Among all of the testing results, we used direct dye terminator sequencing of PCR to validate 5% of them according to the manufacturer’s instructions of ABI Prism BigDye system (ABI, Foster City, CA, USA). The samples for sequencing and genotyping were run on an ABI 3730 automated sequencer and analyzed by SeqMan and Peakscan, respectively.
Cell lines and cell culture
Human LNCaP and PC3 cell lines (The Cell Bank of Chinese Academy of Sciences, Shanghai) were cultured in RPMI 1640 media (Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum (Invitrogen). The cells were maintained in 5% CO2 at 37 °C and the media was replaced every other day.
Plasmid construction, mutation genesis, and transfection
Full-length MTHFR cDNA was amplified by PCR, and CDS was cloned into the pcDNA3.1 vector (Invitrogen, Carlsbad, CA, USA). The corresponding C677T and A1298C mutated plasmid was generated by site-directed mutagenesis with the MutanBEST Kit (Takara, Berkeley, CA) to ensure a uniform backbone sequence. We verified all recombinant clones by DNA sequence. The expression plasmids were transfected into cells using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions.
Western blot
Standard procedures were followed for western blot analysis. Antibodies used for western blotting included anti-MTHFR (Cat NO. ab203786, abcam) and anti-β-ACTIN (Cat NO. A00702, Gensctipt).
RNAi silencing of MTHFR
Lentiviral vector (pLKO.1) expressing shRNA clones were generated by the Stealth RNAiTM siRNAs platform. The shRNA against 3′ UTR of human MTHFR gene was generated using the following target sequence: 5′-CAGTGGCAGTGAGAGCTCCAAAGAT-3′.
1 × 107 cells were seeded in a 10 cm dish with 7 ml viral medium containing 8 μg/ml Polybrene for lentiviral infection. Cells were selected for infection by the addition of 2 μg/ml puromycin after 24 hours. Uninfected cells demonstrated 100% cell death after cultured with puromycin within 3 days.
Measurement of serum and cellular homocysteine level
We randomly selected 306 controls to test the circulating homocysteine level. EDTA-plasma samples were collected, centrifuged immediately, and stored at −80 °C for analyzing. Plasma and cellular homocysteine levels were detected using a tHcy Detection kit (Kuake Biotechnology, Zhejiang, China) by AU5800 biochemistry analyzer (Beckman Coulter, California, USA) according to the manufacturer’s protocol.
Comet Assay for DNA damage
The Comet Assay Kit (Trevigen, Gaithersburg, MD) was used to detect single- and double-stranded DNA breaks in cultured cells. Slides were viewed (excitation 425–500 nm) with a Leica DMI 4000B epifluorescence microscope. These slides were used for each condition. In normal cells, the fluorescence is confined mostly to the nucleus because undamaged DNA cannot migrate. In cells with DNA damage, DNA is denatured by the alkali solution used for single-strand break detection, or the neutral solution used for double-strand break detection; and the negatively charged DNA fragments are released from the nucleus and migrate toward the anode.
Cell apoptosis detection
Cells were harvested and stained with Annexin V-FITC and propidium iodide (Annexin V-FITC apoptosis detection kit, B.D. Biosciences Pharmingen, San Jose, CA, USA) for 15 minutes at room temperature and protected from light. Finally, the cell suspension was filtered by a nylon sieve and analyzed by an Accuri C6 flow cytometer (BD Biosciences).
Cell proliferation assay
Cell proliferation after transfection was measured using the Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan). In brief, LNCaP and PC3 cells were seeded in 96-well plates and allowed to adhere. A 10 μl CCK-8 solution was added to each well and incubated in a humidified CO2 incubator at 37 °C for 2 hours. Then, samples taken from each well were measured at 450 nm, and the percentage of surviving cells in each treatment group was plotted relative to the untreated one.
Statistical analysis
The body mass index (BMI, kg/m2) was calculated as the weight in kilograms divided by the height in square meters. We used the WHO cut point as an index for defining overweight status38 (BMI ≥ 25 kg/m2) in the Asian population. Continuous variables were presented as mean ± standard deviation (SD) and categorical variables were reported in the form of a number (proportion).
The goodness-of fit χ2 test was performed to calculate Hardy-Weinberg equilibrium (HWE) for evaluating genotype distribution in control subjects, and P value < 0.05 was considered deviated from equilibrium. We used univariate and multivariate unconditional logistic regression models to calculate crude and adjusted odds ratios (ORs) and 95% confidence intervals (CIs), respectively, to assess associations between the genotypes and PCa risk. In a multivariate model, covariants included age at diagnosis, BMI, hypertension, diabetes mellitus, and cardiovascular disease. Furthermore, we performed a stratified analysis to explore the relationship between the genotypes and risk of PCa among subgroups of age at diagnosis (≤68 vs. >68), BMI (<25 vs. ≥25), hypertension, diabetes mellitus, cardiovascular disease, Gleason score, extracapsular extension, seminal vesicle invasion, positive surgical margin and lymph node involvement. We used Chi-square-based Q test to evaluate the homogeneity of associations between subgroups. In all statistical analyses, a two-sided P < 0.05 was viewed as significant. We performed all statistical analyses using SPSS software version 16.0 (SPSS Inc., Chicago, IL, USA).
Additional Information
How to cite this article: Wu, J.-L. et al. MTHFR c.677C>T Inhibits Cell Proliferation and Decreases Prostate Cancer Susceptibility in the Han Chinese Population in Shanghai. Sci. Rep. 6, 36290; doi: 10.1038/srep36290 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
This work was supported by grants from the 863 Program (number 2015AA020913), the 973 Program (Number 2015CB943300), the National Science Foundation of China (Numbers 81471454 and 31521003), Commission for Science and Technology of Shanghai Municipality (Numbers 14ZR1402000 and 15XD1500500) to Zhao J.Y., grant from the National Science Foundation of China (Number 81472377) to Ye D.W. and grant from Shanghai rising star project (Number 16QA1401100) to Zhu Y.
Footnotes
Author Contributions J.-L.W. and S.-X.Z. acquired, analyzed and interpreted the data, and drafted the manuscript. R.Z., X.Z., K.C. and C.-Y.G. analyzed and interpreted the data. H.-L.G. reviewed Haematoxylin and eosin sections to confirm tumor histology. B.D. and Y.Z. prepared all figures. H.-L.Z. and G.-H.S. edited all tables. Y.-Y.Q., J.-Y.Z. and D.-W.Y. designed and supervised the study, and provided funding support. All authors reviewed and approved the manuscript.
References
- Torre L. A. et al. Global cancer statistics, 2012. CA: a cancer journal for clinicians 65, 87–108, doi: 10.3322/caac.21262 (2015). [DOI] [PubMed] [Google Scholar]
- Chen W. et al. Cancer statistics in China, 2015. CA: a cancer journal for clinicians 66, 115–132, doi: 10.3322/caac.21338 (2016). [DOI] [PubMed] [Google Scholar]
- Qu Y. et al. Constitutively active AR-V7 plays an essential role in the development and progression of castration-resistant prostate cancer. Scientific reports 5, 7654, doi: 10.1038/srep07654 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qi D. et al. Trends of prostate cancer incidence and mortality in Shanghai, China from 1973 to 2009. The Prostate 75, 1662–1668, doi: 10.1002/pros.23046 (2015). [DOI] [PubMed] [Google Scholar]
- Hu Y. et al. Longitudinal trends in prostate cancer incidence, mortality, and survival of patients from two Shanghai city districts: a retrospective population-based cohort study, 2000–2009. BMC public health 14, 356, doi: 10.1186/1471-2458-14-356 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J. Y., Huang Y. & Sellers T. A. Single nucleotide polymorphisms in DNA repair genes and prostate cancer risk. Methods in molecular biology (Clifton, N.J.) 471, 361–385, doi: 10.1007/978-1-59745-416-2_18 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao D. L. et al. Association of glutathione S-transferase T1 and M1 polymorphisms with prostate cancer susceptibility in populations of Asian descent: a meta-analysis. Oncotarget 6, 35843–35850, doi: 10.18632/oncotarget.5346 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C. et al. Significant association between the Axin2 rs2240308 single nucleotide polymorphism and the incidence of prostate cancer. Oncology letters 8, 789–794, doi: 10.3892/ol.2014.2177 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao C. X. et al. 8q24 rs4242382 polymorphism is a risk factor for prostate cancer among multi-ethnic populations: evidence from clinical detection in China and a meta-analysis. Asian Pacific journal of cancer prevention: APJCP 15, 8311–8317 (2014). [DOI] [PubMed] [Google Scholar]
- Lopez-Cortes A. et al. Genetic polymorphisms in MTHFR (C677T, A1298C), MTR (A2756G) and MTRR (A66G) genes associated with pathological characteristics of prostate cancer in the Ecuadorian population. The American journal of the medical sciences 346, 447–454, doi: 10.1097/MAJ.0b013e3182882578 (2013). [DOI] [PubMed] [Google Scholar]
- Levesque E. et al. Steroidogenic germline polymorphism predictors of prostate cancer progression in the estradiol pathway. Clinical cancer research: an official journal of the American Association for Cancer Research 20, 2971–2983, doi: 10.1158/1078-0432.ccr-13-2567 (2014). [DOI] [PubMed] [Google Scholar]
- Choi S. W. & Mason J. B. Folate and carcinogenesis: an integrated scheme. The Journal of nutrition 130, 129–132 (2000). [DOI] [PubMed] [Google Scholar]
- Rycyna K. J., Bacich D. J. & O’Keefe D. S. Opposing roles of folate in prostate cancer. Urology 82, 1197–1203, doi: 10.1016/j.urology.2013.07.012 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tio M., Andrici J., Cox M. R. & Eslick G. D. Folate intake and the risk of prostate cancer: a systematic review and meta-analysis. Prostate cancer and prostatic diseases 17, 213–219, doi: 10.1038/pcan.2014.16 (2014). [DOI] [PubMed] [Google Scholar]
- Chen P. et al. Higher dietary folate intake reduces the breast cancer risk: a systematic review and meta-analysis. British journal of cancer 110, 2327–2338, doi: 10.1038/bjc.2014.155 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tio M., Andrici J., Cox M. R. & Eslick G. D. Folate intake and the risk of upper gastrointestinal cancers: a systematic review and meta-analysis. Journal of gastroenterology and hepatology 29, 250–258, doi: 10.1111/jgh.12446 (2014). [DOI] [PubMed] [Google Scholar]
- Hultdin J., Van Guelpen B., Bergh A., Hallmans G. & Stattin P. Plasma folate, vitamin B12, and homocysteine and prostate cancer risk: a prospective study. International journal of cancer 113, 819–824, doi: 10.1002/ijc.20646 (2005). [DOI] [PubMed] [Google Scholar]
- de Vogel S. et al. Serum folate and vitamin B12 concentrations in relation to prostate cancer risk–a Norwegian population-based nested case-control study of 3000 cases and 3000 controls within the JANUS cohort. International journal of epidemiology 42, 201–210, doi: 10.1093/ije/dys199 (2013). [DOI] [PubMed] [Google Scholar]
- Izmirli M. A literature review of MTHFR (C677T and A1298C polymorphisms) and cancer risk. Molecular biology reports 40, 625–637, doi: 10.1007/s11033-012-2101-2 (2013). [DOI] [PubMed] [Google Scholar]
- Moll S. & Varga E. A. Homocysteine and MTHFR Mutations. Circulation 132, e6–e9, doi: 10.1161/circulationaha.114.013311 (2015). [DOI] [PubMed] [Google Scholar]
- Chen P. L. et al. Association between MTHFR gene polymorphisms (C677T, A1298C) and genetic susceptibility to prostate cancer: a meta-analysis. Genetics and molecular research: GMR 14, 19191–19202, doi: 10.4238/2015.December.29.29 (2015). [DOI] [PubMed] [Google Scholar]
- Guo S. et al. The protective effect of methylenetetrahydrofolate reductase C677T polymorphism against prostate cancer risk: Evidence from 23 case-control studies. Gene 565, 90–95, doi: 10.1016/j.gene.2015.03.067 (2015). [DOI] [PubMed] [Google Scholar]
- Abedinzadeh M., Zare-Shehneh M., Neamatzadeh H., Abedinzadeh M. & Karami H. Association between MTHFR C677T Polymorphism and Risk of Prostate Cancer: Evidence from 22 Studies with 10,832 Cases and 11,993 Controls. Asian Pacific journal of cancer prevention: APJCP 16, 4525–4530 (2015). [DOI] [PubMed] [Google Scholar]
- Li D. et al. No association of the MTHFR gene A1298C polymorphism with the risk of prostate cancer: A meta-analysis. Experimental and therapeutic medicine 3, 493–498, doi: 10.3892/etm.2012.445 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H. C. et al. Significant association of methylenetetrahydrofolate reductase single nucleotide polymorphisms with prostate cancer susceptibility in taiwan. Anticancer research 30, 3573–3577 (2010). [PubMed] [Google Scholar]
- Kucukhuseyin O. et al. Effects of the MTHFR C677T polymorphism on prostate specific antigen and prostate cancer. Asian Pacific journal of cancer prevention: APJCP 12, 2275–2278 (2011). [PubMed] [Google Scholar]
- Zhang W. B., Zhang J. H., Pan Z. Q., Yang Q. S. & Liu B. The MTHFR C677T polymorphism and prostate cancer risk: new findings from a meta-analysis of 7306 cases and 8062 controls. Asian Pacific journal of cancer prevention: APJCP 13, 2597–2604 (2012). [DOI] [PubMed] [Google Scholar]
- Cai D. et al. Association of polymorphisms in folate metabolic genes and prostate cancer risk: a case-control study in a Chinese population. Journal of genetics 89, 263–267 (2010). [DOI] [PubMed] [Google Scholar]
- Botto L. D. & Yang Q. 5,10-Methylenetetrahydrofolate reductase gene variants and congenital anomalies: a HuGE review. American journal of epidemiology 151, 862–877 (2000). [DOI] [PubMed] [Google Scholar]
- Jablonski N. G. & Chaplin G. Colloquium paper: human skin pigmentation as an adaptation to UV radiation. Proceedings of the National Academy of Sciences of the United States of America 107 Suppl 2, 8962–8968, doi: 10.1073/pnas.0914628107 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho P. I. et al. Folate deprivation induces neurodegeneration: roles of oxidative stress and increased homocysteine. Neurobiology of disease 14, 32–42 (2003). [DOI] [PubMed] [Google Scholar]
- Schieber M. & Chandel N. S. ROS function in redox signaling and oxidative stress. Current biology: CB 24, R453–R462, doi: 10.1016/j.cub.2014.03.034 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi S. W. & Mason J. B. Folate status: effects on pathways of colorectal carcinogenesis. The Journal of nutrition 132, 2413s–2418s (2002). [DOI] [PubMed] [Google Scholar]
- Bai J. L. et al. MTHFR C677T polymorphism contributes to prostate cancer risk among Caucasians: A meta-analysis of 3511 cases and 2762 controls. European journal of cancer (Oxford, England: 1990) 45, 1443–1449, doi: 10.1016/j.ejca.2009.01.020 (2009). [DOI] [PubMed] [Google Scholar]
- Pavillard V. et al. Analysis of cell-cycle kinetics and sulfur amino acid metabolism in methionine-dependent tumor cell lines; the effect of homocysteine supplementation. Biochemical pharmacology 67, 1587–1599, doi: 10.1016/j.bcp.2004.01.006 (2004). [DOI] [PubMed] [Google Scholar]
- Sabharwal S. S. & Schumacker P. T. Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles’ heel? Nature reviews. Cancer 14, 709–721, doi: 10.1038/nrc3803 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng L., Montironi R., Bostwick D. G., Lopez-Beltran A. & Berney D. M. Staging of prostate cancer. Histopathology 60, 87–117, doi: 10.1111/j.1365-2559.2011.04025.x (2012). [DOI] [PubMed] [Google Scholar]
- Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet (London, England) 363, 157–163, doi: 10.1016/s0140-6736(03)15268-3 (2004). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.