Abstract
The incidence and prevalence of inflammatory bowel disease (IBD) are increasing. Although the etiology of IBD is unknown, it is thought that genetically susceptible individuals display an inappropriate inflammatory response to commensal microbes, resulting in intestinal tissue damage. Key proteins involved in regulating the immune response, and thus in inflammation, are the small triphosphate-binding protein Rac and its regulatory network. Recent data suggest these proteins to be involved in (dys)regulation of the characteristic inflammatory processes in IBD. Moreover, Rac-gene variants have been identified as susceptibility risk factors for IBD, and Rac1 GTPase signaling has been shown to be strongly suppressed in non-inflamed mucosa compared with inflamed colonic mucosa in IBD. In addition, first-line immunosuppressive treatment for IBD includes thiopurine therapy, and its immunosuppressive effect is primarily ascribed to Rac1 suppression. In this review, we focus on Rac modification and its potential role in the development of IBD, Rac as the molecular therapeutic target in current thiopurine therapy, and the modulation of the Rac signal transduction pathway as a promising novel therapeutic strategy.
Key Points
| Genetic variants of Rac1 and NCF2 are identified as susceptibility single nucleotide polymorphisms for IBD. |
| The immunosuppressive mechanism of thiopurine treatment relies on the modification of Rac1. |
| Rac1 may serve as a potential biomarker to predict (pharmacological) effectiveness of thiopurine therapy. |
Introduction
Inflammatory bowel disease (IBD), with its two main entities Crohn’s disease (CD) and ulcerative colitis (UC), is characterized by chronic inflammation of the gastrointestinal tract and causes a significant disease burden, and the incidence is rising globally [1]. Although the etiology of IBD is yet unknown, it is thought that IBD results from an inappropriate inflammatory reaction to commensal microbes in genetically susceptible individuals [2]. In genetic association studies, several gene variants and genetic risk loci, crucial for intestinal homeostasis and affecting the immune response, which contribute to the development of (or protection against) IBD have been identified [3]. This inappropriate inflammatory response to commensal microbes results in intestinal tissue damage. The small triphosphate-binding protein Rac and a connected network of regulatory proteins are key proteins involved in the regulation of the immune response, and thus in inflammation [4]. Therefore, these proteins may be involved in (dys)regulation of the specific inflammatory processes in IBD. Moreover, Rac-gene variants have been identified as susceptibility risk factors for IBD [5, 6]. This may indicate that Rac has a pathogenetic or, at least, disease-modifying role. In general, therapeutic strategies for IBD are based on targeting inflammatory responses, most often by suppression, consequently modifying the disease course rather than curing it. First-line immunosuppressive maintenance treatment, as recommended in current Dutch and international guidelines for IBD, consists of the conventional thiopurine derivates azathioprine and mercaptopurine [7–9]. Interestingly, the immunosuppressive molecular mechanism of thiopurine treatment is based on the modification of Rac1. Based on these findings, new therapeutic opportunities are now being explored [10].
In this review, we focus on Rac(-modification) and its potential role in IBD; that is, Rac being a molecular therapeutic target in current thiopurine therapy and modulation of the Rac signal transduction pathway as a promising novel therapeutic strategy.
Regulation of Rac
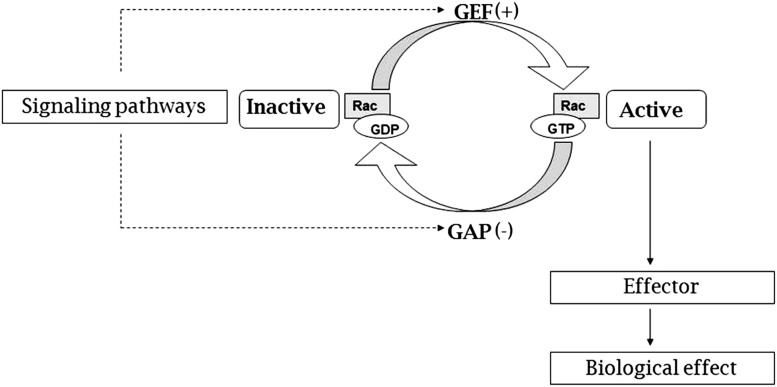
Rac proteins form a subfamily of the Rho family, comprising small guanosine triphosphate-binding proteins that consist of Rac 1, 2, 3 and RhoG [11]. Like other Rho GTPases, Rac acts as a molecular switch between guanosine diphosphate (GDP)-bound inactive and guanosine triphosphate (GTP)-bound active states. As Rac is of critical importance in many inflammation-regulating cellular processes, its activity needs to be very tightly regulated. Therefore, inactivation of Rac, by means of hydrolysis of GTP to GDP, is promoted by GTPase activating proteins (GAPs). Activation of Rac, the result of exchanging GDP for GTP, is stimulated by guanine nucleotide exchange factors (GEFs) (Fig. 1) [4, 11–14]. However, it is more complicated than this; for example, various subtypes of GEFs and GAPs modify the level of activation of Rac [15]. Additionally, various Rho GTPases, including RhoA, may exert antagonistic activity towards the Rac proteins. To date, 20 members of GAP proteins have been identified to inactivate Rac in humans [16, 17]. In total, 20 GEFs are shown to activate Rac, of which two, Vav and Tiam1, are the most extensively studied [15, 18, 19]. The structure of Rac is complex and consists of several functional features including five G motifs, a core effector domain, and a membrane targeting sequence. All three parts may interact with different selectivity with GEFs, GAPs or effector proteins. Pre-clinical studies have shown feasibility of pharmacological modification of these interactions [20]. Although no Rac-targeted drug molecules have yet been clinically approved, this may provide interesting pharmaceutical opportunities to design specific drugs in order to modify the (in)activation of Rac [21–24].
Fig. 1.
Simplified scheme of the ‘role’ of Rac and the molecular switch of active Rac and inactive Rac, regulated by guanine nucleotide exchange factors (GEF) and GTPase-activating proteins (GAP). Moreover, Rac GEF reaction may proceed by competitive displacement of bound GDP by GTP through a transient intermediate of GEF (GTP-Rac-GDP) [65]. GDP guanosine diphosphate, GTP guanosine triphosphate
Rac and Inflammatory Bowel Disease (IBD)
Established Genetic Associations of Rac With IBD
A potential role for Rac in the pathogenesis of IBD has been put forward by the discovery of genetic variants of Rac1 and NCF2 (which is involved in reducing binding to Rac2) as susceptibility single nucleotide polymorphisms (SNPs) for IBD [5, 6]. Rac1 (located on chromosome 7) is associated with colonic IBD, whereas Rac2 (located on chromosome 22) is associated with occurrence of CD in general. Moreover, loss of Rac1 expression (in Rac1 knockout mice) protects against developing experimental (dextran sulfate sodium [DSS]-induced) colitis [5]. The Rac1 risk allele results in an increased Rac1 expression in peripheral blood cells, leading to an inflammatory response in the colonic tissue [5]. Rac2 knockout mice develop more severe disease when subjected to a C. rodentium-induced model of infectious colitis compared with wild-type mice, suggesting that impaired Rac2 function may promote the development of IBD [25].
Rac, Inflammation, and IBD
Rac is a key protein in many inflammatory pathways including apoptosis, intestinal barrier function by cytoskeletal regulation, reactive oxygen species (ROS) generation, and leukocyte trafficking. Therefore, modulation of the Rac pathway may be of pathogenetic importance [4, 26–28]. Rac1 GTPase signaling has been shown to be strongly suppressed in non-inflamed colonic mucosa when compared with the inflamed colonic mucosa in IBD patients. Moreover, a recent study established that activation of Rac1 is increased in experimentally induced mucosal wounds.
Intestinal epithelial apoptosis is mediated in part by Rac1, via the JNK signaling pathway, and as such co-regulates proapoptotic pathways in intestinal epithelial cells [29]. In IBD, the role of T cell apoptosis is crucial and has been extensively described earlier by Peppelenbosch and van Deventer [30]. Inducing T cell apoptosis is believed to be a central goal of most drug treatment modalities in IBD, and accordingly, a common final denominator of many therapeutic strategies, such as anti-tumor necrosis factor alpha (TNFα), JAK-STAT inhibition, corticosteroids, methotrexate, and thiopurine derivatives.
The intestinal barrier is crucial in protecting human organisms against luminal pathogens. Increased paracellular permeability of the intestinal barrier results in systemic contamination, potentially leading to systemic inflammation. Cytoskeletal regulation also plays a role in intestinal epithelial barrier function. Rac is a key protein involved in the regulation of the cytoskeletal construction, and thus plays a role in intestinal epithelial barrier function [31]. This pathway may contribute to the development of IBD. Activation of Rac1 leads to actin polymerization and formation of lamellipodia, whereas activated Rac1 is additionally involved in actin turnover [32]. Cytoskeletal rearrangement is of critical importance in the immune modulatory capacities of Rac1, as it involves mobility and plasticity of (intestinal) leukocytes [33]. It has been suggested that part of the therapeutic JAK-STAT inhibition is mediated by this mechanism [34, 35]. ROS generation via nicotinamide adenine dinucleotide phosphate (NADPH) oxidase enzymes is well known to be the classical effector following neutrophil activation in systemic and intestinal inflammation [26]. Rac2 belongs to the NADPH complex, which can produce ROS, underlying the mechanism of killing microbes by phagocytes. In the absence of Rac2, macrophages and/or neutrophils display suppressed ROS production, defective chemotaxis, impaired phagocytosis, and decreased microbial killing [6, 36]. One of the subunits of the NADPH complex is subunit Gp91phox, which contributes to the development of experimental IBD [37]. Other SNPs in NADPH oxidase complex genes, including NCF4, are also associated with CD [38].
Another aspect of the chronic inflammatory process which is characteristic for IBD is leukocyte trafficking into the gut mucosa, regulated by an integrin-mediated adhesive interaction. Rac has been identified to regulate the α4-paxillin interaction required for effective (α4β1) dependent leukocyte trafficking. Paxillin binding to α4 blocks lamellipodia formation by inhibiting Rac activation [39].
It seems evident that modification of Rac by genetic variation or drug interaction influences apoptosis, the intestinal barrier, ROS generation, and leukocyte trafficking, and thus may play a role in the pathogenesis of IBD.
Modulation of the Rac Pathways by Thiopurine Therapy
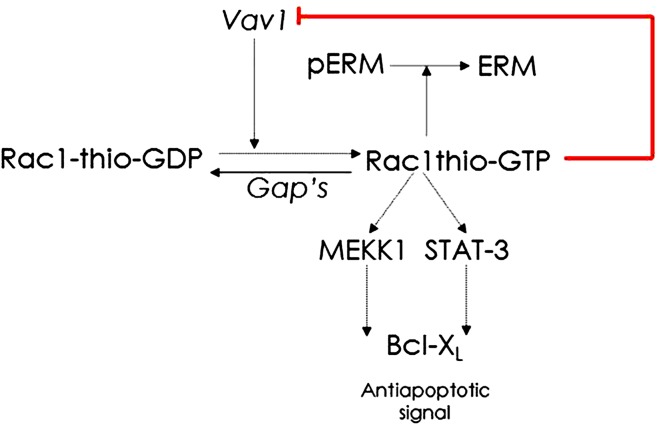
Conventional thiopurines, azathioprine and mercaptopurine, comprise first-line immunosuppressive maintenance treatment in IBD [7, 9]. In previous reports, it has been suggested that thiopurine therapy is effective in inducing, but mainly in maintaining remission in IBD patients [40–43]. Although these thiopurine derivates have been prescribed for years, the immunosuppressive mechanism has only recently been clarified, at least partly, and appears to be based on inducing apoptosis in activated (CD28 co-stimulated) gut inflammation-associated T cells [44]. An antimetabolite effect of thiopurine derivatives is only expected when administered in relatively high dosages (like in oncological treatment) [45]. Interference with in vitro activation of Rac1 is identified to be the molecular target of azathioprine and mercaptopurine therapy [33, 44]. Azathioprine and mercaptopurine first need to be biologically activated in order to generate the pharmacologically active end-metabolites 6-thioguanine nucleotides (6-TGN), via several enzymatic steps [46]. The major component (80 %) of the 6-TGN pool consists of 6-thioguanine triphosphate (6-TGTP) [47]. Instead of GTP, this thiopurine metabolite 6-TGTP binds to Rac 1. Consecutively, 6-TGTP-bound Rac1 induces a mitochondrial pathway of T cell apoptosis via inhibition of Rac1 activation in T cells by blocking the GEF Vav1 (Fig. 2) [33, 44]. In this way, inhibition of the activation of Rac1 results in less active Rac1 and, secondarily, in increased T cell apoptosis. In addition, recently it has been proposed that the related small GTPases RhoA and Cdc42 are pharmacological targets of 6-TGTP. RhoA has diverse functions related with regulation of vasculature. Consequently, the authors suggested that interference with RhoA may result in vascular complications such as non cirrhotic portal hypertension [48]. This may therefore explain why in some patients thiopurine therapy has previously been associated with vascular liver irregularities, and nodular regenerative hyperplasia in particular.
Fig. 2.
Model for thiopurine-mediated immunosuppression. The guanine nucleotide exchange factor (GEF) Vav1 activates Rac1 by the exchange of GDP for GTP. The active thiopurine metabolite thio-GTP binds to Rac1 instead of GTP. This thio-GTP-bound Rac 1 (Rac1thio-GTP) induces T-cell apoptosis (by a decrease of Bcl-XL [anti-apoptotic signal]) via inhibition of Rac 1 activation in T cells. Active Rac1 also ensures de-phosphorylation of Ezrin–Radixin–Moesin (pERM) to ERM. GDP guanosine diphosphate, GTP guanosine triphosphate
Moreover, an association has been identified in adult IBD patients between successful thiopurine therapy and an SNP in the RAC1 gene (rs 34932801). This association has not been observed in a cohort of children with IBD, maybe due to the limited number of observations [49]. Patients with wild-type genotype (G/G) RAC1 c-289 demonstrate a better clinical response to thiopurine therapy compared with patients with variant alleles [50]. The functional consequences of this wild-type genotype have been assessed in an in vitro experiment, in which less promoter activity (less relatively luciferase activity) is observed for the G/G than for the G>C genotype [51]. This corroborates the presumed pivotal role for Rac1 in the mechanism of action of thiopurine therapy. Recently, an in vivo study showed that IBD patients treated with thiopurine therapy have a lower median expression of Rac1 compared with IBD patients without maintenance immunosuppressive treatment. Particularly, patients with active disease who clinically responded to mercaptopurine therapy showed significantly decreased concentrations of Rac1-GTP and Rac1 expression [52].
Leukocyte adhesion and cell migration, processes known to modify the immune response, are also disrupted by inhibition of Rac1 activation in several ways. Via an as-yet unknown mechanism, active Rac1 ensures the dephosphorylation of Ezrin–Radixin–Moesin (pERM to ERM). The ERM protein complex is involved in cytoskeletal dynamics of cells and consequently in the rigidity of the cell membrane [33, 53]. The suppression of Rac1 activation by azathioprine or mercaptopurine leads to less pERM being dephosphorylated, thereby resulting in T-cell-APC conjugation suppression [33]. Additionally, azathioprine inhibits the adhesion and transmigration of leukocytes through the endothelial barrier by selectively decreasing TNFα-induced vascular cell adhesion molecule (VCAM-1) protein levels [54].
Additionally, in one in vivo study, it has been shown that mercaptopurine and 6-TGTP may also reduce pro-inflammatory signaling pathways in macrophages, by reducing expression of the chemokines interleukin (IL)-8 and CCL2, and thus, may induce an antiproliferative and anti-inflammatory effect on gut epithelial cells, by inhibition of Rac1 [54].
Moreover, TNFα has been shown to stimulate activation of Rac1 [54]. Anti-TNFα therapy, a well known and highly effective treatment for IBD, may suppress the activation of Rac1–GTP similar to thiopurine therapy which may contribute to the immunosuppressive effect of anti-TNFα therapy [55]. The SONIC (Study of Biologic and Immunomodulator Naive Patients in Crohn’s Disease) and SUCCESS (Efficacy & Safety of Infliximab Monotherapy vs Combination Therapy vs AZA Monotherapy in Ulcerative Colitis) trials have shown that, in IBD patients, combination therapy consisting of anti-TNFα and thiopurine therapy is more effective than monotherapy with either anti-TNFα or azathioprine [56, 57].
Therapeutic Drug Monitoring of Thiopurine Therapy
Measuring the concentrations of thiopurine metabolites (6-TGN and 6-methylmercaptopurines-ribonucleotides [6-MMPR]) in red blood cells (RBCs) is currently advocated to monitor and optimize thiopurine therapy in IBD patients [46]. 6-TGN concentrations above 230 pmol/8 × 108 RBCs have been associated with therapeutic effectiveness, and concentrations above 450 pmol/8 × 108 RBCs with myelosuppression [58]. Additionally, 6-MMPR concentrations above 5700 pmol/8 × 108 RBCs have been associated with toxicity, mainly hepatotoxicity, and therapeutic ineffectiveness [59, 60]. The possibility of routine assessment of these thiopurine metabolites is still debated as it seems of limited clinical value to predict therapeutic efficacy [61–63]. One of the problems is that these metabolites are pharmacokinetic parameters, which ‘solely’ describe the level of metabolites of the drugs in RBCs, while the mechanism of action of the drug cannot be assessed. A pharmacodynamic parameter can measure the ‘real effect’ of therapy and appears to be more apt in predicting therapeutic success. Hence, a pharmacodynamic parameter associated with the effect of thiopurine therapy is warranted. As Rac1 activation is the molecular target for thiopurine therapy, activated Rac1 (GTP-bound Rac1), Rac1/ERM, and pERM/ERM levels may serve as potential (pharmacodynamic) biomarkers to predict the effectiveness of thiopurine therapy [33, 44]. Another problem of thiopurine pharmacokinetics (6-TGN and 6-MMPR) is that the metabolites of interest are usually measured in erythrocytes, while the immunosuppressive effect of thiopurine therapy is exerted in leukocytes. Measuring the therapeutic pharmacodynamic effect in leukocytes, such as with Rac1-GTP and Rac1/ERM determinations, may thus be a better matrix for monitoring this therapy. These parameters may be helpful in identifying patients who do not experience the intended pharmacodynamic effect of thiopurines, whether due to too low concentrations of the drug (e.g., skewed metabolism, lack of compliance) or due to ‘truly pharmacological’ refractoriness. These ‘truly pharmacological’ refractory patients are defined as being unresponsive to thiopurine therapy, with therapeutic levels of 6-TGN, and unchanged concentrations of Rac1-GTP and Rac1 expression. Additionally, there is a group of patients, the so-called ‘truly pharmacodynamic’ refractory patients, in whom concentrations of Rac1-GTP and Rac1 expression decrease (i.e., the intended pharmacodynamic effect) but no clinical improvement is established. The proposed pharmacodynamic parameters may, therefore, be (early) biomarkers for thiopurine (in)efficacy.
Novel Therapeutic Strategies in IBD and Modulation of Rac1 Pathways
Analogs of the alleged pharmacologically active thiopurine metabolite 6-TGTP have been developed to improve therapeutic effectiveness and decrease toxicity. These analogs have shown, in vitro, low toxicity as well as high immunosuppressive efficacy [10, 64]. These promising findings indicate that a more targeted modulation of the Rac pathway has potential as a novel therapeutic target for IBD patients.
Tiede and colleagues assessed that thiopurine therapy not only suppressed activation of Rac1 but also of Rac2 [44]. Another previous study showed that Rac2 knockout mice develop more severe disease when subjected to a C. rodentium-induced model of infectious colitis [25]. This suggests that Rac1 and Rac2 may actually have opposite effects. Suppression of Rac2 activation could be a negative effect of thiopurine therapy and thus a specific Rac1 blocker may be more effective in IBD treatment.
Conclusion
This review aims to provide insight into the mechanism of action of thiopurine therapy by modulation of Rac. Moreover, the role of Rac in IBD has been put forward by the discovery of Rac gene variants as a susceptibility risk factor for IBD and that Rac1 GTPase signaling is strongly suppressed in non-inflamed compared with inflamed colonic mucosa in IBD. Recent findings provide opportunities for improving drug monitoring of thiopurine therapy by analyzing the modulation of Rac1 as a pharmacodynamic marker for effective therapy in IBD patients.
Compliance with Ethical Standards
Conflict of interest
ML. Seinen: not applicable; G.P. van Nieuw Amerongen: not applicable; N.K.H. de Boer: not applicable; A.A. van Bodegraven: not applicable.
Funding
Open access was funded by the VU University Medical Center, Amsterdam, as part of the COMPACT agreement between Springer and the Association of Dutch Universities and Academy Institutes.
References
- 1.Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142:46–54. doi: 10.1053/j.gastro.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 2.Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–2078. doi: 10.1056/NEJMra0804647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Khor B, Gardet A, Xavier RJ. Genetics and pathogenesis of inflammatory bowel disease. Nature. 2011;474:307–317. doi: 10.1038/nature10209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bokoch GM. Regulation of innate immunity by Rho GTPases. Trends Cell Biol. 2005;15:163–171. doi: 10.1016/j.tcb.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 5.Muise AM, Walters T, Xu W, et al. Single nucleotide polymorphisms that increase expression of the guanosine triphosphatase RAC1 are associated with ulcerative colitis. Gastroenterology. 2011;141:633–641. doi: 10.1053/j.gastro.2011.04.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muise AM, Xu W, Guo CH, et al. NADPH oxidase complex and IBD candidate gene studies: identification of a rare variant in NCF2 that results in reduced binding to RAC2. Gut. 2012;61:1028–1035. doi: 10.1136/gutjnl-2011-300078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dignass A, Van AG, Lindsay JO, et al. The second European evidence-based consensus on the diagnosis and management of Crohn’s disease: current management. J Crohns Colitis. 2010;4:28–62. doi: 10.1016/j.crohns.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 8.Terdiman JP, Gruss CB, Heidelbaugh JJ, et al. American Gastroenterological Association Institute guideline on the use of thiopurines, methotrexate, and anti-TNF-alpha biologic drugs for the induction and maintenance of remission in inflammatory Crohn’s disease. Gastroenterology. 2013;145:1459–1463. doi: 10.1053/j.gastro.2013.10.047. [DOI] [PubMed] [Google Scholar]
- 9.van Bodegraven AA, van Everdingen JJ, Dijkstra G, et al. Guideline ‘Diagnosis and treatment of inflammatory bowel disease in adults’. I. Diagnosis and treatment. Ned Tijdschr Geneeskd. 2010;154:A1899. [PubMed] [Google Scholar]
- 10.Atreya R, Hofmann U, Fritz G, et al. Superior immunosuppressive efficacy and decreased toxicity of noven thiopurine analogues. Gut. 2011;60:A398. [Google Scholar]
- 11.Tybulewicz VL, Henderson RB. Rho family GTPases and their regulators in lymphocytes. Nat Rev Immunol. 2009;9:630–644. doi: 10.1038/nri2606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Beckers CM, van Hinsbergh VW, van Nieuw Amerongen GP. Driving Rho GTPase activity in endothelial cells regulates barrier integrity. Thromb Haemost. 2010;103:40–55. doi: 10.1160/TH09-06-0403. [DOI] [PubMed] [Google Scholar]
- 13.Citi S, Spadaro D, Schneider Y, et al. Regulation of small GTPases at epithelial cell–cell junctions. Mol Membr Biol. 2011;28:427–444. doi: 10.3109/09687688.2011.603101. [DOI] [PubMed] [Google Scholar]
- 14.DerMardirossian C, Bokoch GM. GDIs: central regulatory molecules in Rho GTPase activation. Trends Cell Biol. 2005;15:356–363. doi: 10.1016/j.tcb.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 15.Rossman KL, Der CJ, Sondek J. GEF means go: turning on RHO GTPases with guanine nucleotide-exchange factors. Nat Rev Mol Cell Biol. 2005;6:167–180. doi: 10.1038/nrm1587. [DOI] [PubMed] [Google Scholar]
- 16.Tcherkezian J, Lamarche-Vane N. Current knowledge of the large RhoGAP family of proteins. Biol Cell. 2007;99:67–86. doi: 10.1042/BC20060086. [DOI] [PubMed] [Google Scholar]
- 17.Yang C, Kazanietz MG. Chimaerins: GAPs that bridge diacylglycerol signalling and the small G-protein Rac. Biochem J. 2007;403:1–12. doi: 10.1042/BJ20061750. [DOI] [PubMed] [Google Scholar]
- 18.Cook DR, Rossman KL, Der CJ. Rho guanine nucleotide exchange factors: regulators of Rho GTPase activity in development and disease. Oncogene. 2014;33:4021–4035. doi: 10.1038/onc.2013.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schmidt A, Hall A. Guanine nucleotide exchange factors for Rho GTPases: turning on the switch. Genes Dev. 2002;16:1587–1609. doi: 10.1101/gad.1003302. [DOI] [PubMed] [Google Scholar]
- 20.Marei H, Malliri A. Rac1 in human diseases: the therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases. 2016;1–25. doi:10.1080/21541248.2016.1211398. [DOI] [PMC free article] [PubMed]
- 21.Bustelo XR, Sauzeau V, Berenjeno IM. GTP-binding proteins of the Rho/Rac family: regulation, effectors and functions in vivo. Bioessays. 2007;29:356–370. doi: 10.1002/bies.20558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Smithers CC, Overduin M. Structural mechanisms and drug discovery prospects of Rho GTPases. Cells. 2016;5(2). doi:10.3390/cells5020026. [DOI] [PMC free article] [PubMed]
- 23.Vetter IR, Wittinghofer A. The guanine nucleotide-binding switch in three dimensions. Science. 2001;294:1299–1304. doi: 10.1126/science.1062023. [DOI] [PubMed] [Google Scholar]
- 24.Wertheimer E, Gutierrez-Uzquiza A, Rosemblit C, et al. Rac signaling in breast cancer: a tale of GEFs and GAPs. Cell Signal. 2012;24:353–362. doi: 10.1016/j.cellsig.2011.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fattouh R, Guo CH, Lam GY, et al. Rac2-deficiency leads to exacerbated and protracted colitis in response to Citrobacter rodentium infection. PLoS One. 2013;8:e61629. doi: 10.1371/journal.pone.0061629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bokoch GM, Zhao T. Regulation of the phagocyte NADPH oxidase by Rac GTPase. Antioxid Redox Signal. 2006;8:1533–1548. doi: 10.1089/ars.2006.8.1533. [DOI] [PubMed] [Google Scholar]
- 27.Etienne-Manneville S, Hall A. Rho GTPases in cell biology. Nature. 2002;420:629–635. doi: 10.1038/nature01148. [DOI] [PubMed] [Google Scholar]
- 28.Huang Y, Xiao S, Jiang Q. Role of Rho kinase signal pathway in inflammatory bowel disease. Int J Clin Exp Med. 2015;8:3089–3097. [PMC free article] [PubMed] [Google Scholar]
- 29.Jin S, Ray RM, Johnson LR. Rac1 mediates intestinal epithelial cell apoptosis via JNK. Am J Physiol Gastrointest Liver Physiol. 2006;291:G1137–G1147. doi: 10.1152/ajpgi.00031.2006. [DOI] [PubMed] [Google Scholar]
- 30.Peppelenbosch MP, van Deventer SJ. T cell apoptosis and inflammatory bowel disease. Gut. 2004;53:1556–1558. doi: 10.1136/gut.2004.040824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schlegel N, Meir M, Spindler V, et al. Differential role of Rho GTPases in intestinal epithelial barrier regulation in vitro. J Cell Physiol. 2011;226:1196–1203. doi: 10.1002/jcp.22446. [DOI] [PubMed] [Google Scholar]
- 32.Murali A, Rajalingam K. Small Rho GTPases in the control of cell shape and mobility. Cell Mol Life Sci. 2014;71:1703–1721. doi: 10.1007/s00018-013-1519-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Poppe D, Tiede I, Fritz G, et al. Azathioprine suppresses ezrin-radixin-moesin-dependent T cell-APC conjugation through inhibition of Vav guanosine exchange activity on Rac proteins. J Immunol. 2006;176:640–651. doi: 10.4049/jimmunol.176.1.640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sandborn WJ, Ghosh S, Panes J, et al. Tofacitinib, an oral Janus kinase inhibitor, in active ulcerative colitis. N Engl J Med. 2012;367:616–624. doi: 10.1056/NEJMoa1112168. [DOI] [PubMed] [Google Scholar]
- 35.Sandborn WJ, Ghosh S, Panes J, et al. A phase 2 study of tofacitinib, an oral Janus kinase inhibitor, in patients with Crohn’s disease. Clin Gastroenterol Hepatol. 2014;12:1485–1493. doi: 10.1016/j.cgh.2014.01.029. [DOI] [PubMed] [Google Scholar]
- 36.Bosco EE, Kumar S, Marchioni F, et al. Rational design of small molecule inhibitors targeting the Rac GTPase-p67(phox) signaling axis in inflammation. Chem Biol. 2012;19:228–242. doi: 10.1016/j.chembiol.2011.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bao S, Carr ED, Xu YH, et al. Gp91(phox) contributes to the development of experimental inflammatory bowel disease. Immunol Cell Biol. 2011;89:853–860. doi: 10.1038/icb.2011.4. [DOI] [PubMed] [Google Scholar]
- 38.Roberts RL, Hollis-Moffatt JE, Gearry RB, et al. Confirmation of association of IRGM and NCF4 with ileal Crohn’s disease in a population-based cohort. Genes Immun. 2008;9:561–565. doi: 10.1038/gene.2008.49. [DOI] [PubMed] [Google Scholar]
- 39.Rose DM. The role of the alpha4 integrin-paxillin interaction in regulating leukocyte trafficking. Exp Mol Med. 2006;38:191–195. doi: 10.1038/emm.2006.23. [DOI] [PubMed] [Google Scholar]
- 40.Gisbert JP, Nino P, Cara C, et al. Comparative effectiveness of azathioprine in Crohn’s disease and ulcerative colitis: prospective, long-term, follow-up study of 394 patients. Aliment Pharmacol Ther. 2008;28:228–238. doi: 10.1111/j.1365-2036.2008.03732.x. [DOI] [PubMed] [Google Scholar]
- 41.Pearson DC, May GR, Fick G, et al. Azathioprine for maintaining remission of Crohn’s disease. Cochrane Database Syst Rev. 2000;(2):CD000067. doi:10.1002/14651858.CD000067. [DOI] [PubMed]
- 42.Sandborn W, Sutherland L, Pearson D, et al. Azathioprine or 6-mercaptopurine for inducing remission of Crohn’s disease. Cochrane Database Syst Rev. 2000;(2):CD000545. doi:10.1002/14651858.CD000545. [DOI] [PubMed]
- 43.Timmer A, McDonald JW, Tsoulis DJ, et al. Azathioprine and 6-mercaptopurine for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012;9:CD000478. doi: 10.1002/14651858.CD000478.pub3. [DOI] [PubMed] [Google Scholar]
- 44.Tiede I, Fritz G, Strand S, et al. CD28-dependent Rac1 activation is the molecular target of azathioprine in primary human CD4+ T lymphocytes. J Clin Invest. 2003;111:1133–1145. doi: 10.1172/JCI16432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Quemeneur L, Gerland LM, Flacher M, et al. Differential control of cell cycle, proliferation, and survival of primary T lymphocytes by purine and pyrimidine nucleotides. J Immunol. 2003;170:4986–4995. doi: 10.4049/jimmunol.170.10.4986. [DOI] [PubMed] [Google Scholar]
- 46.van Asseldonk DP, de Boer NK, Peters GJ, et al. On therapeutic drug monitoring of thiopurines in inflammatory bowel disease; pharmacology, pharmacogenomics, drug intolerance and clinical relevance. Curr Drug Metab. 2009;10:981–997. doi: 10.2174/138920009790711887. [DOI] [PubMed] [Google Scholar]
- 47.Neurath MF, Kiesslich R, Teichgraber U, et al. 6-thioguanosine diphosphate and triphosphate levels in red blood cells and response to azathioprine therapy in Crohn’s disease. Clin Gastroenterol Hepatol. 2005;3:1007–1014. doi: 10.1016/S1542-3565(05)00697-X. [DOI] [PubMed] [Google Scholar]
- 48.Shin JY, Wey M, Umutesi HG, et al. Thiopurine prodrugs mediate immunosuppressive effects by interfering with Rac1 function. J. Biol. Chem. 2016;291:13699–13714. doi: 10.1074/jbc.M115.694422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lev-Tzion R, Renbaum P, Beeri R, et al. Rac1 polymorphisms and thiopurine efficacy in children with inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 2015;61:404–407. doi: 10.1097/MPG.0000000000000820. [DOI] [PubMed] [Google Scholar]
- 50.Koifman E, Karban A, Mazor Y, et al. Thiopurine effectiveness in patients with Crohn’s disease: a study of genetic and clinical predictive factors. Inflamm Bowel Dis. 2013;19:1639–1644. doi: 10.1097/MIB.0b013e31828828d3. [DOI] [PubMed] [Google Scholar]
- 51.Bourgine J, Garat A, Allorge D, et al. Evidence for a functional genetic polymorphism of the Rho-GTPase Rac1. Implication in azathioprine response? Pharmacogenet Genom. 2011;21:313–324. doi: 10.1097/FPC.0b013e3283449200. [DOI] [PubMed] [Google Scholar]
- 52.Seinen ML, van Nieuw Amerongen GP, de Boer NK, et al. Rac1 as a potential pharmacodynamic biomarker for thiopurine therapy in inflammatory bowel disease. Ther Drug Monit. 2016. [DOI] [PubMed]
- 53.Cernuda-Morollon E, Millan J, Shipman M, et al. Rac activation by the T-cell receptor inhibits T cell migration. PLoS One. 2010;5:e12393. doi: 10.1371/journal.pone.0012393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Marinkovic G, Hamers AA, de Vries CJ, et al. 6-Mercaptopurine reduces macrophage activation and gut epithelium proliferation through inhibition of GTPase Rac1. Inflamm Bowel Dis. 2014;20:1487–1495. doi: 10.1097/MIB.0000000000000122. [DOI] [PubMed] [Google Scholar]
- 55.Parikh K, Zhou L, Somasundaram R, et al. Suppression of p21Rac signaling and increased innate immunity mediate remission in Crohn’s disease. Sci Transl Med. 2014;6:233ra53. doi: 10.1126/scitranslmed.3006763. [DOI] [PubMed] [Google Scholar]
- 56.Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N Engl J Med. 2010;362:1383–1395. doi: 10.1056/NEJMoa0904492. [DOI] [PubMed] [Google Scholar]
- 57.Panaccione R, Ghosh S, Middleton S, et al. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology. 2014;146:392–400. doi: 10.1053/j.gastro.2013.10.052. [DOI] [PubMed] [Google Scholar]
- 58.Dubinsky MC, Lamothe S, Yang HY, et al. Pharmacogenomics and metabolite measurement for 6-mercaptopurine therapy in inflammatory bowel disease. Gastroenterology. 2000;118:705–713. doi: 10.1016/S0016-5085(00)70140-5. [DOI] [PubMed] [Google Scholar]
- 59.Dubinsky MC, Yang H, Hassard PV, et al. 6-MP metabolite profiles provide a biochemical explanation for 6-MP resistance in patients with inflammatory bowel disease. Gastroenterology. 2002;122:904–915. doi: 10.1053/gast.2002.32420. [DOI] [PubMed] [Google Scholar]
- 60.Kreijne JE, Seinen ML, Wilhelm AJ, et al. Routinely established skewed thiopurine metabolism leads to a strikingly high rate of early therapeutic failure in patients with inflammatory bowel disease. Ther. Drug. Monit. 2015;37:797–804. doi: 10.1097/FTD.0000000000000213. [DOI] [PubMed] [Google Scholar]
- 61.Gilissen LP, Wong DR, Engels LG, et al. Therapeutic drug monitoring of thiopurine metabolites in adult thiopurine tolerant IBD patients on maintenance therapy. J Crohns Colitis. 2012;6:698–707. doi: 10.1016/j.crohns.2011.12.003. [DOI] [PubMed] [Google Scholar]
- 62.Gonzalez-Lama Y, Bermejo F, Lopez-Sanroman A, et al. Thiopurine methyl-transferase activity and azathioprine metabolite concentrations do not predict clinical outcome in thiopurine-treated inflammatory bowel disease patients. Aliment Pharmacol Ther. 2011;34:544–554. doi: 10.1111/j.1365-2036.2011.04756.x. [DOI] [PubMed] [Google Scholar]
- 63.Haines ML, Ajlouni Y, Irving PM, et al. Clinical usefulness of therapeutic drug monitoring of thiopurines in patients with inadequately controlled inflammatory bowel disease. Inflamm Bowel Dis. 2011;17:1301–1307. doi: 10.1002/ibd.21458. [DOI] [PubMed] [Google Scholar]
- 64.Atreya I, Diall A, Dvorsky R, et al. Designer thiopurine-analogues for optimized immunosuppression in inflammatory bowel diseases. J Crohns Colitis. 2016. doi:10.1093/ecco-jcc/jjw091. [DOI] [PubMed]
- 65.Zhang B, Yang L, Zheng Y. Novel intermediate of Rac GTPase activation by guanine nucleotide exchange factor. Biochem Biophys Res Commun. 2005;331:413–421. doi: 10.1016/j.bbrc.2005.03.179. [DOI] [PubMed] [Google Scholar]