Abstract
Environmental factors play an important role in the development of rheumatoid arthritis (RA). Among these factors, smoking is generally considered to be an established risk factor for RA. Data regarding the impact of diet on risk of RA development is limited. This study assessed the impact of dietary patterns on RA susceptibility in Chinese populations. This was a large scale, case-control study composed of 968 patients with RA and 1037 matched healthy controls. Subjects were recruited from 18 teaching hospitals. Socio-demographic characteristics and dietary intakes 5 years prior to the onset of RA were reported by a self-administered questionnaire. Differences in quantity of consumption between cases and controls were analyzed by Student’s t test. Multiple logistic regression analysis was applied to identify independent dietary risk factor(s) responsible for RA susceptibility. Compared to healthy individuals, RA patients had decreased consumption of mushrooms (P = 0.000), beans (P = 0.006), citrus (P = 0.000), poultry (P = 0.000), fish (P = 0.000), edible viscera (P = 0.018), and dairy products (P = 0.005). Multivariate analyses revealed that several dietary items may have protective effects on RA development, such as mushrooms (aOR = 0.669; 95%CI = 0.518–0.864, P = 0.002), citrus fruits (aOR = 0.990; 95%CI = 0.981–0.999, P = 0.04), and dairy products (aOR = 0.921; 95%CI 0.867–0.977, P = 0.006). Several dietary factors had independent effects on RA susceptibility. Dietary interventions may reduce the risk of RA.
Keywords: Chinese population, Dietary factors, Disease susceptibility, Rheumatoid arthritis
Introduction
Rheumatoid arthritis (RA) is a chronic systemic inflammatory disease that leads to progressive joint erosion and destruction and affects 0.5–1 % of the population. RA is considered to be a complex disease that is triggered by both genetic and environmental factors, and the pathogenesis of RA has not yet been fully elucidated. Recent advances in high-throughput genome scanning have enabled the identification of many genetic risk factors that contribute to RA susceptibility. However, it has been estimated that the genetic factors account for only 60 % of the risk for RA susceptibility [1], while environmental and other non-genetic factors account for the remaining 40 %.
Among environmental factors, while only smoking is currently considered to be an established risk factor for RA [2, 3], other factors, such as diet, may contribute to RA susceptibility. Diet is an environmental factor that affects inflammation, antigen presentation, antioxidant defense mechanisms, allergies, and gut microbiota, and the exact impact of diet on RA risk remains uncertain. Several case-control studies have suggested that omega-3 fatty acids, which are present in fish, soybean, safflower, sunflower, and corn oils, may confer protection against RA [4, 5]. Alcohol may reduce the risk of RA in women [6]. While reactive oxygen species (ROS) are produced in rheumatoid joints [7], the roles of dietary factors such as antioxidants and vitamins in the pathogenesis of RA remain unclear. Vitamin C is both an intracellular and an extracellular scavenger of ROS, and in models of rat adjuvant arthritis supplementation with vitamin C contributed to improved arthritis scores, evidenced by a reduction in paw volume [8, 9]. However, there is little clinical evidence on the effects of antioxidants, such as vitamins E and C, on individuals with RA [10, 11]. A 2-year study on the effects of the Mediterranean diet on RA patients revealed that the consumption of cereals, vegetables, legumes, fruits, and olive oil decreased the risk of new onset of inflammatory polyarthritis [12].
While there is evidence on the effects of diet on RA pathogenesis in Western populations, there is little information to date concerning the impact of diet on the development of RA in Chinese populations. Here, we assess the impact of dietary factors on the development of RA in Chinese populations.
Patients and methods
Study population
A total of 968 RA patients and 1037 ethnic and geographically matched healthy controls were recruited from 18 hospitals (Fig. 1). All RA patients fulfilled the American College of Rheumatology (ACR) criteria for RA [13]. Healthy controls were randomly selected from medical workers without any history of chronic diseases. A self-administered food frequency questionnaire (FFQ) was designed to measure socio-demographic characteristics and dietary intakes. Patients were asked to complete a detailed weekly retrospective FFQ (i.e., over the last 5 years prior to RA onset). Healthy controls were required to complete a similar weekly retrospective FFQ, to gather information on their dietary intakes for the previous 5 years. FFQs were coded and analyzed. The patients and healthy controls with incomplete data from FFQs were excluded from the study.
Fig. 1.
Patient flow chart. RA rheumatoid arthritis, HC healthy controls. A total of 968 RA patients and 1037 healthy controls were recruited from 18 hospitals
The study was conducted in China between May 2012 and September 2013. A survey was designed following the guidelines established by the Chinese Rheumatology Association (CRA). The study was approved by the Medical Ethics Committee of Peking University People’s Hospital. Informed written consent forms were obtained from all study participants.
Demographic data
Demographics and personal characteristics were gathered, including gender, age, marital status, education, occupation, personal income, height, and weight.
Dietary assessment
Dietary assessment on the FFQ includes the frequency and the amount of dietary intake. The frequency of red meat, poultry, fish, edible viscera, vegetables, potatoes, mushrooms, beans, nuts, milk or yogurt, eggs, citrus, and other fruit except for citrus was measured with a response range from 1 to 8 (1 = less than once per month, 2 = 1–3 times per month, 3 = 1–2 times per week, 4 = 3–4 times per week, 5 = 5–6 times per week, 6 = 1 time per day, 7 = 2 times per day, and 8 = 3 or more times per day). The intake amount per serving of rice, flour meal, red meat, poultry, fish, edible viscera, vegetables, potatoes, mushrooms, beans, and nuts was measured with a response range from 1 to 4 (1 = 50 g or less, 2 = 50–100 g, 3 = 100–150 g, 4 = 150–200 g or more). The amount of eggs, citrus, and other fruit except for citrus was measured with a response range from 1 to 5 (1 = 1/2 or less, 2 = 1/2–2/3, 3 = 2/3–1, 4 = 1–2, 5 = 2–3 or more). The intake amount per serving of milk or yogurt was measured with a response range from 1 to 5 (1 = 50 ml or less, 2 = 50–75 ml, 3 = 75–100 ml, 4 = 100–200 ml, 5 = 200–300 ml or more). The total amount of dietary intake per month of a food item is equal to the intake amount per serving of that food item × the frequency of dietary intake.
Statistical analyses
Differences in quantity of consumption between cases and controls were assessed using t test analyses. Multiple logistic regression analysis was applied to identify independent dietary risk factor(s) responsible for RA susceptibility. Odds ratios (ORs) with 95 % confidence intervals (CIs) were calculated to estimate the relative risk. P values less than 0.05 were considered significant.
Results
Demographic profile of RA patients
The study subjects were mainly recruited from two ethnic groups: Han (n = 913) and Hui (n = 18). Additionally, there were 37 subjects coming from other ethnic groups, including Man (n = 27), Mongol (n = 7), Yi (n = 1), and Zhuang (n = 1). The demographic characteristics of patients and controls are shown in Table 1.
Table 1.
Characteristics of RA patients and healthy controls
| RA (n = 968) | HC (n = 1037) | |
|---|---|---|
| Gender (%) | ||
| Male | 202 (20.9) | 220 (21.2) |
| Female | 766 (79.1) | 817 (78.8) |
| Age (years), (mean ± SD) | 52.1 ± 13.4 | 49.7 ± 15.2 |
| Age group (years), (%) | ||
| ≤20 | 22 (1.3) | 27 (2.6) |
| 21–30 | 50 (5.2) | 76 (7.3) |
| 31–40 | 83 (8.7) | 88 (8.5) |
| 41–50 | 183 (19.1) | 237 (22.9) |
| 51–60 | 324 (33.8) | 370 (35.7) |
| 61–70 | 212 (22.1) | 160 (15.4) |
| ≥71 | 94 (9.8) | 79 (7.6) |
| Ethnicity (%) | ||
| Han | 913 (94.3) | 967 (93.2) |
| Hui | 18 (1.9) | 20 (1.9) |
| Others | 37 (3.8) | 50 (4.8) |
RA rheumatoid arthritis, HC healthy controls
Dietary intakes in RA patients and healthy controls
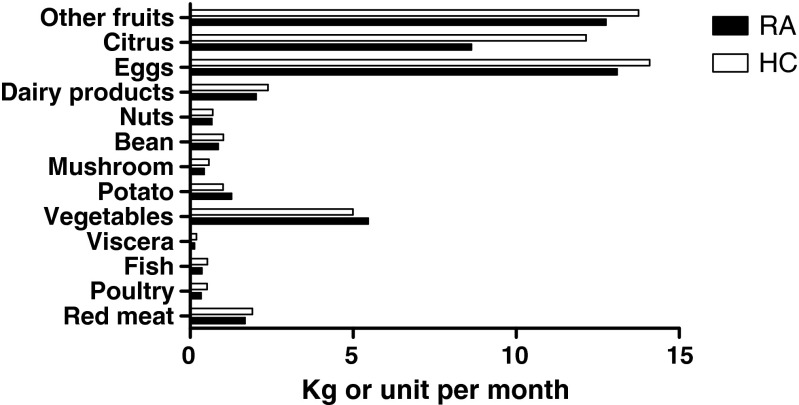
The results of the dietary intakes of both groups are shown in Fig. 2 and Table 2. Compared with healthy controls, RA patients consumed lower amounts of chicken (P = 0.0001), fish (P = 0.0001), mushrooms (P = 0.0001), beans (P = 0.006), dairy products (P = 0.005), citrus fruits (P = 0.000), and organ meats (P = 0.018). There were no significant differences in consumption of red meat between RA patients and healthy controls.
Fig. 2.
Dietary intakes of RA patients and healthy controls (HC). Compared with healthy controls, RA patients had higher amount of consumption in potatoes and lower amount of consumption in chicken, fish, mushrooms, beans, dairy products, citrus fruits, and organ meats. There were no significant differences in consumption of red meat between RA patients and healthy controls
Table 2.
Differences in monthly dietary consumption between RA patients and healthy controls
| RA (n = 968)a | HC (n = 1037)a | P | |
|---|---|---|---|
| Red meat (kg/month), mean ± SD | 1.688 ± 1.621 | 1.911 ± 1.620 | 0.363 |
| Poultry (kg/month), mean ± SD | 0.339 ± 0.498 | 0.521 ± 0.890 | 0.000 |
| Fish (kg/month), mean ± SD | 0.361 ± 0.546 | 0.526 ± 1.029 | 0.000 |
| Edible viscera (kg/month), mean ± SD | 0.139 ± 0.224 | 0.195 ± 0.635 | 0.018 |
| Potatoes (kg/month), mean ± SD | 1.275 ± 1.884 | 1.015 ± 1.095 | 0.001 |
| Vegetables (kg/month), mean ± SD | 5.465 ± 4.368 | 4.997 ± 3.912 | 0.020 |
| Mushrooms (kg/month), mean ± SD | 0.427 ± 0.636 | 0.577 ± 0.989 | 0.000 |
| Beans (kg/month), mean ± SD | 0.866 ± 0.993 | 1.019 ± 1.301 | 0.006 |
| Citrus fruits (unit/month), mean ± SD | 8.630 ± 17.163 | 12.143 ± 20.726 | 0.000 |
| Other fruits (unit/month), mean ± SD | 12.753 ± 15.160 | 13.753 ± 20.824 | 0.268 |
| Nuts (kg/month), mean ± SD | 0.672 ± 1.420 | 0.696 ± 1.260 | 0.715 |
| Dairy products (milk or yogurt) (L/month), mean ± SD | 2.029 ± 2.697 | 2.388 ± 2.578 | 0.005 |
| Eggs (unit/month) mean ± SD | 13.093 ± 13.686 | 14.099 ± 14.566 | 0.143 |
RA rheumatoid arthritis, HC healthy controls
aEntries may not add up to 968 or 1037 because of the responses obtained
Comparison of dietary intake between Han and Hui populations in both RA and HC groups
We compared the dietary consumption between the Han and Hui populations in the RA and HC cohorts, respectively. As shown in Table 3, except for dairy products, there were no significant differences in dietary consumption between Han and Hui groups.
Table 3.
Differences in monthly dietary consumption between Han patients and Hui patients
| HC | RA | |||||
|---|---|---|---|---|---|---|
| Han (n = 967)a | Hui (n = 20)a | P | Han (n = 913)a | Hui (n = 18)a | P | |
| Red meat (kg/month), mean ± SD | 1.838 ± 1.781 | 2.733 ± 2.309 | 0.136 | 1.652 ± 1.924 | 1.144 ± 1.095 | 0.431 |
| Poultry (kg/month), mean ± SD | 0.495 ± 0.626 | 1.565 ± 4.265 | 0.317 | 0.345 ± 0.516 | 0.306 ± 0.246 | 0.765 |
| Fish (kg/month), mean ± SD | 0.514 ± 0.862 | 0.484 ± 0.345 | 0.893 | 0.373 ± 0.563 | 0.327 ± 0.256 | 0.750 |
| Edible viscera (kg/month), mean ± SD | 0.172 ± 0.414 | 0.181 ± 0.206 | 0.940 | 0.135 ± 0.210 | 0.181 ± 0.383 | 0.394 |
| Potatoes (kg/month), mean ± SD | 1.023 ± 1.104 | 0.632 ± 0.501 | 0.146 | 1.269 ± 1.823 | 0.683 ± 0.662 | 0.174 |
| Vegetables (kg/month), mean ± SD | 5.025 ± 3.901 | 5.033 ± 3.458 | 0.993 | 5.538 ± 4.361 | 5.182 ± 4.345 | 0.739 |
| Mushrooms (kg/month), mean ± SD | 0.586 ± 1.018 | 0.553 ± 0.389 | 0.902 | 0.430 ± 0.651 | 0.462 ± 0.479 | 0.844 |
| Beans (kg/month), mean ± SD | 1.026 ± 1.319 | 0.829 ± 0.731 | 0.541 | 0.883 ± 1.012 | 0.681 ± 0.725 | 0.429 |
| Citrus fruits (unit/month), mean ± SD | 11.767 ± 19.992 | 20.359 ± 29.988 | 0.256 | 8.709 ± 17.639 | 6.350 ± 5.475 | 0.582 |
| Other fruits (unit/month), mean ± SD | 13.413 ± 19.391 | 14.421 ± 11.191 | 0.831 | 12.778 ± 15.438 | 10.188 ± 9.941 | 0.504 |
| Nuts (kg/month), mean ± SD | 0.691 ± 1.271 | 0.935 ± 1.199 | 0.433 | 0.654 ± 1.378 | 1.278 ± 3.143 | 0.412 |
| Dairy products (milk or yogurt) (L/month), mean ± SD | 2.366 ± 2.581 | 2.385 ± 3.198 | 0.976 | 1.984 ± 2.550 | 3.491 ± 2.976 | 0.020* |
| Eggs (unit/month), mean ± SD | 14.115 ± 14.276 | 19.429 ± 28.746 | 0.472 | 13.352 ± 13.956 | 10.006 ± 13.4103 | 0.374 |
RA rheumatoid arthritis, HC healthy controls
aEntries may not add up to 967 or 20 or 913 or 18 because of the responses obtained
*p < 0.05
Comparison of dietary intakes between Han and non-Han in both RA and HC groups
We compared the dietary consumption between the Han and non-Han populations in both RA and HC groups. Han people with RA intake more fish and less dairy products than non-Han people with RA. There were no other significant differences in dietary consumption between the Han and non-Han groups (Table 4).
Table 4.
Differences in monthly dietary consumption between Han patients and Non-Han patients
| HC | RA | |||||
|---|---|---|---|---|---|---|
| Han (n = 967)a | Non-Han (n = 70)a | P | Han (n = 913)a | Non-Han (n = 55)a | P | |
| Red meat (kg/month) mean ± SD | 1.838 ± 1.781 | 3.400 ± 4.517 | 0.139 | 1.652 ± 1.924 | 1.456 ± 1.522 | 0.608 |
| Poultry (kg/month) mean ± SD | 0.495 ± 0.626 | 0.878 ± 2.843 | 0.406 | 0.345 ± 0.516 | 0.279 ± 0 .215 | 0.420 |
| Fish (kg/month) mean ± SD | 0.514 ± 0.862 | 0.849 ± 2.921 | 0.490 | 0.373 ± 0.563 | 0.251 ± 0.247 | 0.009* |
| Edible viscera (kg/month) mean ± SD | 0.172 ± 0.414 | 0.614 ± 2.330 | 0.285 | 0.135 ± 0.210 | 0.231 ± 0.432 | 0.197 |
| Potatoes (kg/month) mean ± SD | 1.023 ± 1.104 | 0.826 ± 0 .699 | 0.254 | 1.269 ± 1.823 | 1.341 ± 2.763 | 0.809 |
| Vegetables (kg/month) mean ± SD | 5.025 ± 3.901 | 4.906 ± 3.978 | 0.839 | 5.538 ± 4.361 | 4.664 ± 4.134 | 0.222 |
| Mushrooms (kg/month) mean ± SD | 0.586 ± 1.018 | 0.473 ± 0 .405 | 0.491 | 0.430 ± 0.651 | 0.422 ± 0.459 | 0.936 |
| Beans (kg/month) mean ± SD | 1.026 ± 1.319 | 0.884 ± 1.077 | 0.519 | 0.883 ± 1.012 | 0.722 ± 0.718 | 0.328 |
| Citrus fruits (unit/month) mean ± SD | 11.767 ± 19.992 | 17.317 ± 23.719 | 0.079 | 8.709 ± 17.639 | 9.277 ± 11.397 | 0.841 |
| Other fruits (unit/month) mean ± SD | 13.413 ± 19.391 | 16.093 ± 13.838 | 0.400 | 12.778 ± 15.438 | 12.492 ± 10.974 | 0.909 |
| Nuts (kg/month) mean ± SD | 0.691 ± 1.271 | 0.682 ± 0.945 | 0.964 | 0.654 ± 1.378 | 0.955 ± 2.140 | 0.180 |
| Dairy products (milk or yogurt) (L/month) mean ± SD | 2.366 ± 2.581 | 2.495 ± 2.540 | 0.747 | 1.984 ± 2.550 | 3.557 ± 4.677 | 0.000* |
| Eggs (unit/month) mean ± SD | 14.115 ± 14.276 | 13.752 ± 19.794 | 0.878 | 13.352 ± 13.956 | 11.043 ± 10.580 | 0.309 |
RA rheumatoid arthritis, HC, healthy controls
aEntries may not add up to 967 or 70 or 913 or 55 because of the responses obtained
*p < 0.05
Dietary intake as independent risk factor for RA development
The multivariate analyses revealed that females and individuals older than 50 years of age were more susceptible to RA (Table 5). Consumption of several dietary items were independent factors for higher risk of RA development, including potatoes (OR = 1.160; 95 % CI = 1.035–1.300, P = 0.011) and fruits except for citrus fruits (OR = 1.013; 95 % CI = 1.003–1.023, P = 0.013). In contrast, some dietary items were found to have protective effects on RA, including mushrooms (OR = 0.669; 95 % CI = 0.518–0.864, P = 0.002), dairy products (OR = 0.921; 95 % CI = 0.867–0.977, P = 0.006), and citrus fruits (OR = 0.990; 95 % CI = 0.981–0.999, P = 0.040). Consumption of red meats, fruits, and vegetables was not found to have any effect on RA risk. The analyses revealed that higher intakes of carbohydrates and lower intakes of dairy products, mushrooms, and citrus fruits may associate with RA development. There was a trend for high intakes of fish (OR = 0.864; 95%CI = 0.647–1.156, P = 0.325) and beans (OR = 0.938; 95%CI = 0.811–1.086, P = 0.391) to have protective effects against RA, though these did not reach statistical significance. Additionally, no significant associations were observed between the consumption of red meat and RA risk.
Table 5.
Multiple logistic regression of factors associated with RA (n = 968)
| Β | OR (95 % CI) | P | |
|---|---|---|---|
| Gender (male, reference group) | 0.875 | 2.398 (1.722–3.340) | 0.000 |
| Age group (≤20, reference group) | |||
| 21–30 | −0.476 | 0.621 (0.228–1.689) | 0.351 |
| 31–40 | 0.154 | 1.167 (0.425–3.205) | 0.765 |
| 41–50 | 0.506 | 1.658 (0.627–4.390) | 0.308 |
| 51–60 | 1.227 | 3.411 (1.305–8.914) | 0.012 |
| 61–70 | 1.712 | 5.543 (2.033–15.112) | 0.001 |
| ≥71 | 1.336 | 3.803 (1.305–11.082) | 0.014 |
| Ethnicity (Han, reference group) | |||
| Hui | 0.719 | 2.051 (0.342–12.289) | 0.431 |
| Others | 0.250 | 1.284 (0.426–3.872) | 0.657 |
| Red meat (kg/month) | 0.046 | 1.047 (0.945–1.160) | 0.382 |
| Poultry (kg/month) | −0.119 | 0.887 (0.672–1.171) | 0.399 |
| Fish (kg/month) | −0.146 | 0.864 (0.647–1.156) | 0.325 |
| Edible viscera (kg/month) | 0.138 | 1.148 (0.716–1.842) | 0.566 |
| Potato (kg/month) | 0.148 | 1.160 (1.035–1.300) | 0.011* |
| Vegetables (kg/month) | 0.029 | 1.030 (0.991–1.070) | 0.135 |
| Mushroom (kg/month) | −0.402 | 0.669 (0.518–0.864) | 0.002* |
| Bean (kg/month) | −0.064 | 0.938 (0.811–1.086) | 0.391 |
| Citrus (unit/month), | −0.010 | 0.990 (0.981–0.999) | 0.040* |
| Other fruits except for citrus (unit/month) | 0.013 | 1.013 (1.003–1.023) | 0.013* |
| Dairy products (L/month) | −0.083 | 0.921 (0.867–0.977) | 0.006* |
| Eggs (unit/month) | −0.004 | 0.996 (0.984–1.008) | 0.502 |
| BMI | 0.011 | 1.011 (0.975–1.048) | 0.559 |
RA rheumatoid arthritis
*p < 0.05
Discussion
The etiology of RA remains an area of considerable interest. RA is triggered by genetic factors; however, environmental factors may also play a role in the pathogenesis of RA. Dietary and lifestyle factors (e.g., smoking) [2, 3] contribute to RA. Smoking is so far the most well-established environmental risk factor for development of RA [2, 3]. Additionally, infectious agents such as viruses, bacteria, and fungi have long been suspected risk factors for RA, but there is no conclusive evidence to support the hypothesis [14]. Influences of other environmental factors on RA, such as hormones, have also been suggested. Hormones are believed to influence RA based on the fact that females are more prone to develop RA than are men, with a peak onset at 50–60 years of age [1, 15].
Salminen et al. [15] reported that 33–75 % patients believe that food plays an important role in their symptom severity and approximately 50 % will have tried dietary manipulation in an attempt to improve their symptoms. Diets rich in fiber, omega-3 fatty acids, and antioxidants, and low in red meat have been reported associated with decreased RA risks [16–18]. Comparing with the survey by others [16–18], Chinese diets are characterized by higher levels of vegetables and lower intakes of red meat consumption and the average meat consumption is much lower than Western diets. In this study, there were no significant differences in red meat intakes between RA patients and healthy controls, as reported in other studies [19, 20]. Whether the association between red meat consumption and inflammatiory arthritis remains unclear, meat consumption might affect the gut microbiota or contribute to high energy intakes, which provide some explanation for the association.
The results of this study revealed that high intakes of carbohydrates (e.g., potatoes) might be associated with increased RA risks. High carbohydrate and lower fiber intakes lead to excess energy intake. With excess energy intake and reduced energy expenditure, body weight and adiposity increase. Although there was no signifcant difference in body mass index (BMI) between RA patients and HCs in present work, other studies have reported that BMI is positively correlated with chronic inflammatory disorders [21]. It is recognized that adipocytes release the proinflammatory cytokines TNF, IL-1β and IL-6; thus, adipose tissue is metabolically active and contribute to sysytemic inflammatory responses [3, 21].
Studies have reported that ROS are involved in the pathogenesis of RA. Antioxidant-rich diets have beneficial effects on several diseases. However, in this study, high intakes of fruits or vegetables had no significant effects. Interestingly, citrus fruits have a protective role in the pathogenesis of RA. Citrus fruits are rich in hesperidin. Kawaguchi [22] reported that the consumption of citrus flavanone and naringin suppressed the inflammatory responses in collagen-induced arthritis in mice, possibly by decreasing tumor necrosis factor-α (TNF-α) levels. In an 8-week, placebo-controlled, randomized, double-blind clinical trial, Oben et al. reported that citrus extracts improved knee joint pain and flexibility and reduced C-reactive protein levels [23]. In addition to hesperidin and naringin, citrus fruits are rich in vitamin C, which has protective roles according to some studies and no clinical benefits according to other studies [11].
Mushroom consumption was significantly lower in RA patients than in healthy controls. Mushrooms have food and pharmaceutical applications due to the presence of β-glucans, polysaccharopeptides, and polysaccharide-protein conjugates, which have immunomodulatory and antitumor activities. These compounds have demonstrated mitogenicity and activation of immune effector cells, such as lymphocytes, macrophages, and natural killer cells, resulting in the production of cytokines, including interleukins, TNF-α, and interferon gamma [24]. Yu et al. reported that mushrooms regulate immunity in vitro [25]. A number of mushroom components have been shown to modulate immunity and therefore might play a role in preventing RA.
In this study, there were no significant benefits from fish consumption; however, fish contains omega-3 fatty acids, which have protective roles. More than 20 randomized controlled trials have reported that omega-3 fatty acids have modest benefits on RA treatment. Additionally, symptoms got worse in RA patients who discontinued the fish oil supplements [16, 26, 27].
Dairy products, such as milk and yogurt, had protective roles against RA in this study. Yogurt contains probiotics, which maintain a healthy gut microbiota. A metagenomic approach using Illumina sequencing of pooled milk samples revealed that the genera and types of bacteria in milk may affect bacterial establishment and stability in this food matrix [28]. Supplementation with Lactobacillus casei improves the inflammatory status of patients with RA [29]. A 3-month, double-blind, placebo-controlled study reported considerable improvements in RA patients following supplementation with Lactobacillus rhamnosus and Lactobacillus reuteri [30]. Additionally, the administration of probiotics in different animal models improved inflammatory bowel disease, atopic dermatitis, and RA, probably as a result of enrichment of CD4+Foxp3+ Tregs in inflamed body areas [31]. Therefore, probiotics are recommended to patients with RA.
In conclusion, dietary factors contribute to the development of RA. Dietary modification might reduce RA risk and prevent disease progression. There is a need for large-scale prospective, placebo-controlled studies to assess the effects of multiple dietary compounds on RA.
Authors’ contributions
All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final manuscript. Dr. He and Dr. Li had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Study conception and design: J. He, Y. Wang, J. Guo, and Z. Li.
Acquisition of data: Y. Jin, M. Feng, X. Zhang, X. Li, L. Su, A. Wang, L. Wu, X. Yu, L. Sun, X. Liu, Y. Wang, B. Jia, J. Li, J. Tao, F. Zhang, P. Yu, L. Cui, J. Yang, Z. Li, J. Xie, P. Wei, W. Sun, L. Gong, Y. Cheng, C. Huang, X. Wang, Y. Wang, H. Guo, H. Jin, X. Liu, G. Wang, Y. Wang, L. He, Y. Zhao, X. Li, and Y. Zhang.
Analysis and interpretation of data: J. He, Y. Wang, J. Guo, and Z. Li.
Compliance with ethical standards
Disclosures
None.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81373117 and No. 31270914) and the Beijing Municipal Science & Technology Commission (No. Z131107002213060).
Footnotes
Jing He, Yu Wang and Min Feng have equally contributed to this work.
Contributor Information
Jian-Ping Guo, Email: jianping.guo@bjmu.edu.cn.
Zhan-Guo Li, Email: li99@bjmu.edu.cn.
References
- 1.Liao KP, Alfredsson L, Karlson EW. Environmental influences on risk for rheumatoid arthritis. Curr Opin Rheumatol. 2009;21:279–283. doi: 10.1097/BOR.0b013e32832a2e16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Di Giuseppe D, Discacciati A, Orsini N, et al. Cigarette smoking and risk of rheumatoid arthritis: a dose-response meta-analysis. Arthritis Res Ther. 2014;16:R61. doi: 10.1186/ar4498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Hair MJ, Landewe RB, van de Sande MG, et al. Smoking and overweight determine the likelihood of developing rheumatoid arthritis. Ann Rheum Dis. 2013;72:1654–1658. doi: 10.1136/annrheumdis-2012-202254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Galli C, Calder PC. Effects of fat and fatty acid intake on inflammatory and immune responses: a critical review. Ann Nutr Metab. 2009;55:123–139. doi: 10.1159/000228999. [DOI] [PubMed] [Google Scholar]
- 5.Karlson EW, Mandl LA, Aweh GN, et al. Coffee consumption and risk of rheumatoid arthritis. Arthritis Rheum. 2003;48:3055–3060. doi: 10.1002/art.11306. [DOI] [PubMed] [Google Scholar]
- 6.Lu B, Solomon DH, Costenbader KH, et al. Alcohol consumption and risk of incident rheumatoid arthritis in women: a prospective study. Arthritis Rheumatol. 2014;66:1998–2005. doi: 10.1002/art.38634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hu F, Mu R, Zhu J, et al. Hypoxia and hypoxia-inducible factor-1alpha provoke toll-like receptor signalling-induced inflammation in rheumatoid arthritis. Ann Rheum Dis. 2014;73:928–936. doi: 10.1136/annrheumdis-2012-202444. [DOI] [PubMed] [Google Scholar]
- 8.Sakai A, Hirano T, Okazaki R, et al. Large-dose ascorbic acid administration suppresses the development of arthritis in adjuvant-infected rats. Arch Orthop Trauma Surg. 1999;119:121–126. doi: 10.1007/s004020050374. [DOI] [PubMed] [Google Scholar]
- 9.Davis RH, Rosenthal KY, Cesario LR, et al. Vitamin C influence on localized adjuvant arthritis. J Am Podiatr Med Assoc. 1990;80:414–418. doi: 10.7547/87507315-80-8-414. [DOI] [PubMed] [Google Scholar]
- 10.Remans PH, Sont JK, Wagenaar LW, et al. Nutrient supplementation with polyunsaturated fatty acids and micronutrients in rheumatoid arthritis: clinical and biochemical effects. Eur J Clin Nutr. 2004;58:839–845. doi: 10.1038/sj.ejcn.1601883. [DOI] [PubMed] [Google Scholar]
- 11.Mangge H, Hermann J, Schauenstein K. Diet and rheumatoid arthritis—a review. Scand J Rheumatol. 1999;28:201–209. doi: 10.1080/03009749950155553. [DOI] [PubMed] [Google Scholar]
- 12.Skoldstam L, Hagfors L, Johansson G. An experimental study of a Mediterranean diet intervention for patients with rheumatoid arthritis. Ann Rheum Dis. 2003;62:208–214. doi: 10.1136/ard.62.3.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aletaha D, Neogi T, Silman AJ, et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European league against rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569–2581. doi: 10.1002/art.27584. [DOI] [PubMed] [Google Scholar]
- 14.Ebringer A, Wilson C. HLA molecules, bacteria and autoimmunity. J Med Microbiol. 2000;49:305–311. doi: 10.1099/0022-1317-49-4-305. [DOI] [PubMed] [Google Scholar]
- 15.Salminen E, Heikkila S, Poussa T, et al. Female patients tend to alter their diet following the diagnosis of rheumatoid arthritis and breast cancer. Prev Med. 2002;34:529–535. doi: 10.1006/pmed.2002.1015. [DOI] [PubMed] [Google Scholar]
- 16.Kremer JM, Bigauoette J, Michalek AV, et al. Effects of manipulation of dietary fatty acids on clinical manifestations of rheumatoid arthritis. Lancet. 1985;1:184–187. doi: 10.1016/S0140-6736(85)92024-0. [DOI] [PubMed] [Google Scholar]
- 17.Adam O, Beringer C, Kless T, et al. Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis. Rheumatol Int. 2003;23:27–36. doi: 10.1007/s00296-002-0234-7. [DOI] [PubMed] [Google Scholar]
- 18.Geusens P, Wouters C, Nijs J, et al. Long-term effect of omega-3 fatty acid supplementation in active rheumatoid arthritis. A 12-month, double-blind, controlled study. Arthritis Rheum. 1994;37:824–829. doi: 10.1002/art.1780370608. [DOI] [PubMed] [Google Scholar]
- 19.Pedersen M, Stripp C, Klarlund M, et al. Diet and risk of rheumatoid arthritis in a prospective cohort. J Rheumatol. 2005;32:1249–1252. [PubMed] [Google Scholar]
- 20.Benito-Garcia E, Feskanich D, Hu FB, et al. Protein, iron, and meat consumption and risk for rheumatoid arthritis: a prospective cohort study. Arthritis Res Ther. 2007;9:R16. doi: 10.1186/ar2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Varma V, Yao-Borengasser A, Rasouli N, et al. Human visfatin expression: relationship to insulin sensitivity, intramyocel-lular lipids, and inflammation. J Clin Endocrinol Metab. 2007;92:666–672. doi: 10.1210/jc.2006-1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kawaguchi K, Maruyama H, Hasunuma R, et al. Suppression of inflammatory responses after onset of collagen-induced arthritis in mice by oral administration of the citrus flavanone naringin. Immunopharmacol Immunotoxicol. 2011;33:723–729. doi: 10.3109/08923973.2011.564186. [DOI] [PubMed] [Google Scholar]
- 23.Oben J, Enonchong E, Kothari S, et al. Phellodendron and citrus extracts benefit joint health in osteoarthritis patients: a pilot, double-blind, placebo-controlled study. Nutr J. 2009;8:38. doi: 10.1186/1475-2891-8-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lull C, Wichers HJ, Savelkoul HF. Antiinflammatory and immunomodulating properties of fungal metabolites. Mediat Inflamm. 2005;2005:63–80. doi: 10.1155/MI.2005.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yu S, Weaver V, Martin K, et al. The effects of whole mushrooms during inflammation. BMC Immunol. 2009;10:12. doi: 10.1186/1471-2172-10-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fortin PR, Lew RA, Liang MH, et al. Validation of a meta-analysis: the effects of fish oil in rheumatoid arthritis. J Clin Epidemiol. 1995;48:1379–1390. doi: 10.1016/0895-4356(95)00028-3. [DOI] [PubMed] [Google Scholar]
- 27.Ariza-Ariza R, Mestanza-Peralta M, Cardiel MH. Omega-3 fatty acids in rheumatoid arthritis: an overview. Semin Arthritis Rheum. 1998;27:366–370. doi: 10.1016/S0049-0172(98)80016-4. [DOI] [PubMed] [Google Scholar]
- 28.Ward TL, Hosid S, Ioshikhes I, et al. Human milk metagenome: a functional capacity analysis. BMC Microbiol. 2013;13:116. doi: 10.1186/1471-2180-13-116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vaghef-Mehrabany E, Alipour B, Homayouni-Rad A, et al. Probiotic supplementation improves inflammatory status in patients with rheumatoid arthritis. Nutrition. 2014;30:430–435. doi: 10.1016/j.nut.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 30.Pineda Mde L, Thompson SF, Summers K, et al. A randomized, double-blinded, placebo-controlled pilot study of probiotics in active rheumatoid arthritis. Med Sci Monit. 2011;17:CR347–CR354. doi: 10.12659/MSM.881808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kwon HK, Lee CG, So JS, et al. Generation of regulatory dendritic cells and CD4 + Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci U S A. 2010;107:2159–2164. doi: 10.1073/pnas.0904055107. [DOI] [PMC free article] [PubMed] [Google Scholar]