Abstract
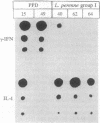
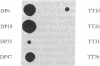
We have established a large panel of T-cell clones (TCCs) specific for Dermatophagoides pteronyssinus and Lolium perenne group I grass pollen allergens (total, 61) and for tetanus toxoid and protein purified derivative bacterial antigens (total, 38) from the peripheral blood of two atopic individuals and then analyzed their ability to produce interleukin 4 (IL-4), IL-5, and interferon gamma (IFN-gamma). Upon stimulation with phorbol 12-myristate 13-acetate plus anti-CD3 antibody, the great majority of TCCs specific for bacterial components was able to produce both IL-4 and IFN-gamma, whereas most D. pteronyssinus- and L. perenne group I-specific TCCs produced IL-4, but no, or limited, IFN-gamma. Moreover, the mean amounts of IL-4 and IFN-gamma released by allergen-specific TCCs were significantly higher and lower, respectively, than the mean amounts produced by TCCs specific for bacterial components. Under the same experimental conditions, virtually all allergen-specific TCCs, but only one-third of tested TCCs specific for bacterial components, expressed IL-5 RNA and secreted IL-5 in their supernatants. Eighteen TCCs (nine specific for allergens and nine specific for bacterial components) were also assessed for their ability to induce IgE synthesis by autologous B cells in response to stimulation with the specific antigen. Under these experimental conditions, all allergen-specific TCCs, but only one-third of TCCs specific for bacterial components that produced IL-4 but no, or little, IFN-gamma induced the synthesis of detectable amounts of IgE. The demonstration that most allergen-specific helper T cells in atopic individuals are able to produce high amounts of IL-4 (and IL-5), but no IFN-gamma, may explain why allergens induce production of IgE antibodies and increase eosinophils.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ansari A. A., Kihara T. K., Marsh D. G. Immunochemical studies of Lolium perenne (rye grass) pollen allergens, Lol p I, II, and III. J Immunol. 1987 Dec 15;139(12):4034–4041. [PubMed] [Google Scholar]
- Ben-Sasson S. Z., Le Gros G., Conrad D. H., Finkelman F. D., Paul W. E. IL-4 production by T cells from naive donors. IL-2 is required for IL-4 production. J Immunol. 1990 Aug 15;145(4):1127–1136. [PubMed] [Google Scholar]
- Cherwinski H. M., Schumacher J. H., Brown K. D., Mosmann T. R. Two types of mouse helper T cell clone. III. Further differences in lymphokine synthesis between Th1 and Th2 clones revealed by RNA hybridization, functionally monospecific bioassays, and monoclonal antibodies. J Exp Med. 1987 Nov 1;166(5):1229–1244. doi: 10.1084/jem.166.5.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coffman R. L., Carty J. A T cell activity that enhances polyclonal IgE production and its inhibition by interferon-gamma. J Immunol. 1986 Feb 1;136(3):949–954. [PubMed] [Google Scholar]
- Del Prete G., Maggi E., Parronchi P., Chrétien I., Tiri A., Macchia D., Ricci M., Banchereau J., De Vries J., Romagnani S. IL-4 is an essential factor for the IgE synthesis induced in vitro by human T cell clones and their supernatants. J Immunol. 1988 Jun 15;140(12):4193–4198. [PubMed] [Google Scholar]
- Fattah D., Quint D. J., Proudfoot A., O'Malley R., Zanders E. D., Champion B. R. In vitro and in vivo studies with purified recombinant human interleukin 5. Cytokine. 1990 Mar;2(2):112–121. doi: 10.1016/1043-4666(90)90005-e. [DOI] [PubMed] [Google Scholar]
- Finkelman F. D., Katona I. M., Urban J. F., Jr, Snapper C. M., Ohara J., Paul W. E. Suppression of in vivo polyclonal IgE responses by monoclonal antibody to the lymphokine B-cell stimulatory factor 1. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9675–9678. doi: 10.1073/pnas.83.24.9675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanzavecchia A., Santini P., Maggi E., Del Prete G. F., Falagiani P., Romagnani S., Ferrarini M. In vitro selective expansion of allergen specific T cells from atopic patients. Clin Exp Immunol. 1983 Apr;52(1):21–28. [PMC free article] [PubMed] [Google Scholar]
- Maggi E., Biswas P., Del Prete G., Parronchi P., Macchia D., Simonelli C., Emmi L., De Carli M., Tiri A., Ricci M. Accumulation of Th-2-like helper T cells in the conjunctiva of patients with vernal conjunctivitis. J Immunol. 1991 Feb 15;146(4):1169–1174. [PubMed] [Google Scholar]
- Maggi E., Del Prete G. F., Parronchi P., Tiri A., Macchia D., Biswas P., Simonelli C., Ricci M., Romagnani S. Role for T cells, IL-2 and IL-6 in the IL-4-dependent in vitro human IgE synthesis. Immunology. 1989 Nov;68(3):300–306. [PMC free article] [PubMed] [Google Scholar]
- Maggi E., Del Prete G., Macchia D., Parronchi P., Tiri A., Chrétien I., Ricci M., Romagnani S. Profiles of lymphokine activities and helper function for IgE in human T cell clones. Eur J Immunol. 1988 Jul;18(7):1045–1050. doi: 10.1002/eji.1830180712. [DOI] [PubMed] [Google Scholar]
- Marsh D. G., Meyers D. A., Freidhoff L. R., Ehrlich-Kautzky E., Roebber M., Norman P. S., Hsu S. H., Bias W. B. HLA-Dw2: a genetic marker for human immune response to short ragweed pollen allergen Ra5. II. Response after ragweed immunotherapy. J Exp Med. 1982 May 1;155(5):1452–1463. doi: 10.1084/jem.155.5.1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosmann T. R., Cherwinski H., Bond M. W., Giedlin M. A., Coffman R. L. Two types of murine helper T cell clone. I. Definition according to profiles of lymphokine activities and secreted proteins. J Immunol. 1986 Apr 1;136(7):2348–2357. [PubMed] [Google Scholar]
- Noma T., Nakakubo H., Sugita M., Kumagai S., Maeda M., Shimizu A., Honjo T. Expression of different combinations of interleukins by human T cell leukemic cell lines that are clonally related. J Exp Med. 1989 May 1;169(5):1853–1858. doi: 10.1084/jem.169.5.1853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norman P. S. An overview of immunotherapy: implications for the future. J Allergy Clin Immunol. 1980 Feb;65(2):87–96. doi: 10.1016/0091-6749(80)90191-8. [DOI] [PubMed] [Google Scholar]
- Parronchi P., Tiri A., Macchia D., De Carli M., Biswas P., Simonelli C., Maggi E., Del Prete G., Ricci M., Romagnani S. Noncognate contact-dependent B cell activation can promote IL-4-dependent in vitro human IgE synthesis. J Immunol. 1990 Mar 15;144(6):2102–2108. [PubMed] [Google Scholar]
- Pène J., Rousset F., Brière F., Chrétien I., Bonnefoy J. Y., Spits H., Yokota T., Arai N., Arai K., Banchereau J. IgE production by normal human lymphocytes is induced by interleukin 4 and suppressed by interferons gamma and alpha and prostaglandin E2. Proc Natl Acad Sci U S A. 1988 Sep;85(18):6880–6884. doi: 10.1073/pnas.85.18.6880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roebber M., Klapper D. G., Goodfriend L., Bias W. B., Hsu S. H., Marsh D. G. Immunochemical and genetic studies of Amb.t. V (Ra5G), an Ra5 homologue from giant ragweed pollen. J Immunol. 1985 May;134(5):3062–3069. [PubMed] [Google Scholar]
- Romagnani S., Del Prete G., Maggi E., Parronchi P., Tiri A., Macchia D., Giudizi M. G., Almerigogna F., Ricci M. Role of interleukins in induction and regulation of human IgE synthesis. Clin Immunol Immunopathol. 1989 Jan;50(1 Pt 2):S13–S23. doi: 10.1016/0090-1229(89)90110-4. [DOI] [PubMed] [Google Scholar]
- Suemura M., Shiho O., Deguchi H., Yamamura Y., Böttcher I., Kishimoto T. Characterization and isolation of IgE class-specific suppressor factor (IgE-TsF) I. The presence of the binding site(s) for IgE and of the H-2 gene products in IgE-TsF. J Immunol. 1981 Aug;127(2):465–471. [PubMed] [Google Scholar]
- Swain S. L., McKenzie D. T., Weinberg A. D., Hancock W. Characterization of T helper 1 and 2 cell subsets in normal mice. Helper T cells responsible for IL-4 and IL-5 production are present as precursors that require priming before they develop into lymphokine-secreting cells. J Immunol. 1988 Nov 15;141(10):3445–3455. [PubMed] [Google Scholar]
- Uede T., Ishizaka K. Formation of IgE-binding factors by rat T lymphocytes. VI. Cellular mechanisms for the formation of IgE-potentiating factor and IgE-suppressive factor by antigenic stimulation of antigen-primed spleen cells. J Immunol. 1982 Oct;129(4):1391–1397. [PubMed] [Google Scholar]
- Vercelli D., Jabara H. H., Arai K., Geha R. S. Induction of human IgE synthesis requires interleukin 4 and T/B cell interactions involving the T cell receptor/CD3 complex and MHC class II antigens. J Exp Med. 1989 Apr 1;169(4):1295–1307. doi: 10.1084/jem.169.4.1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vercelli D., Jabara H. H., Arai K., Yokota T., Geha R. S. Endogenous interleukin 6 plays an obligatory role in interleukin 4-dependent human IgE synthesis. Eur J Immunol. 1989 Aug;19(8):1419–1424. doi: 10.1002/eji.1830190811. [DOI] [PubMed] [Google Scholar]
- White B. A., Bancroft F. C. Cytoplasmic dot hybridization. Simple analysis of relative mRNA levels in multiple small cell or tissue samples. J Biol Chem. 1982 Aug 10;257(15):8569–8572. [PubMed] [Google Scholar]
- Wierenga E. A., Snoek M., de Groot C., Chrétien I., Bos J. D., Jansen H. M., Kapsenberg M. L. Evidence for compartmentalization of functional subsets of CD2+ T lymphocytes in atopic patients. J Immunol. 1990 Jun 15;144(12):4651–4656. [PubMed] [Google Scholar]