Abstract
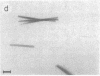
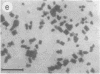
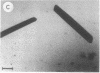
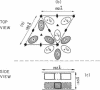
Photosystem I (PSI) reaction centers (RCs) of the thermophilic cyanobacterium Mastigocladus laminosus were purified and characterized. The PSI RC was obtained in two forms, monomeric and trimeric. The two forms contained the same number of pigments per P700 and displayed similar photochemical activities. The two forms had nearly identical polypeptide subunit compositions; the only observed difference was an additional subunit of about 12 kDa observed in the trimeric form. The purified preparations of both the monomeric and the trimeric forms were used for crystallization and preliminary crystallographic analysis. The trimeric PSI RC preparations produced several three-dimensional crystal forms, one of which, the "hexagonal needle" form (THN), had a hexagonal unit cell with dimensions of 300 x 300 x 160 A, containing four PSI RC trimers. The monomeric preparations also produced single crystals of several forms under various crystallization conditions. One of these crystal forms, the "hexagonal plate" (MHP), diffracted to a resolution of about 5.5 A. It had a hexagonal unit cell with dimensions of 192 x 192 x 163 A, containing six PSI RC monomers. Comparison of the PSI RCs in the crystals with those in the precrystallization preparations demonstrated that neither the monomeric nor the trimeric form of PSI RC was altered by the crystallization process. Both forms retained their original polypeptide subunit composition and their pigment content.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen J. P., Feher G. Crystallization of reaction center from Rhodopseudomonas sphaeroides: preliminary characterization. Proc Natl Acad Sci U S A. 1984 Aug;81(15):4795–4799. doi: 10.1073/pnas.81.15.4795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen J. P., Feher G., Yeates T. O., Komiya H., Rees D. C. Structure of the reaction center from Rhodobacter sphaeroides R-26: the cofactors. Proc Natl Acad Sci U S A. 1987 Aug;84(16):5730–5734. doi: 10.1073/pnas.84.16.5730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen J. P., Feher G., Yeates T. O., Komiya H., Rees D. C. Structure of the reaction center from Rhodobacter sphaeroides R-26: the protein subunits. Proc Natl Acad Sci U S A. 1987 Sep;84(17):6162–6166. doi: 10.1073/pnas.84.17.6162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen J. P., Feher G., Yeates T. O., Rees D. C., Deisenhofer J., Michel H., Huber R. Structural homology of reaction centers from Rhodopseudomonas sphaeroides and Rhodopseudomonas viridis as determined by x-ray diffraction. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8589–8593. doi: 10.1073/pnas.83.22.8589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang C. H., Tiede D., Tang J., Smith U., Norris J., Schiffer M. Structure of Rhodopseudomonas sphaeroides R-26 reaction center. FEBS Lett. 1986 Sep 1;205(1):82–86. doi: 10.1016/0014-5793(86)80870-5. [DOI] [PubMed] [Google Scholar]
- Chitnis P. R., Reilly P. A., Miedel M. C., Nelson N. Structure and targeted mutagenesis of the gene encoding 8-kDa subunit of photosystem I from the cyanobacterium Synechocystis sp. PCC 6803. J Biol Chem. 1989 Nov 5;264(31):18374–18380. [PubMed] [Google Scholar]
- Deisenhofer J., Epp O., Miki K., Huber R., Michel H. X-ray structure analysis of a membrane protein complex. Electron density map at 3 A resolution and a model of the chromophores of the photosynthetic reaction center from Rhodopseudomonas viridis. J Mol Biol. 1984 Dec 5;180(2):385–398. doi: 10.1016/s0022-2836(84)80011-x. [DOI] [PubMed] [Google Scholar]
- Ford R. C., Holzenburg A. Investigation of the structure of trimeric and monomeric photosystem I reaction centre complexes. EMBO J. 1988 Aug;7(8):2287–2293. doi: 10.1002/j.1460-2075.1988.tb03071.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ford R. C., Picot D., Garavito R. M. Crystallization of the photosystem I reaction centre. EMBO J. 1987 Jun;6(6):1581–1586. doi: 10.1002/j.1460-2075.1987.tb02403.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garavito R. M., Jenkins J., Jansonius J. N., Karlsson R., Rosenbusch J. P. X-ray diffraction analysis of matrix porin, an integral membrane protein from Escherichia coli outer membranes. J Mol Biol. 1983 Feb 25;164(2):313–327. doi: 10.1016/0022-2836(83)90079-7. [DOI] [PubMed] [Google Scholar]
- Garavito R. M., Rosenbusch J. P. Isolation and crystallization of bacterial porin. Methods Enzymol. 1986;125:309–328. doi: 10.1016/s0076-6879(86)25027-2. [DOI] [PubMed] [Google Scholar]
- Garavito R. M., Rosenbusch J. P. Three-dimensional crystals of an integral membrane protein: an initial x-ray analysis. J Cell Biol. 1980 Jul;86(1):327–329. doi: 10.1083/jcb.86.1.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golbeck J. H. Structure, function and organization of the Photosystem I reaction center complex. Biochim Biophys Acta. 1987;895(3):167–204. doi: 10.1016/s0304-4173(87)80002-2. [DOI] [PubMed] [Google Scholar]
- Kühlbrandt W. Three-dimensional crystallization of membrane proteins. Q Rev Biophys. 1988 Nov;21(4):429–477. doi: 10.1017/s0033583500004625. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Matthews B. W. Solvent content of protein crystals. J Mol Biol. 1968 Apr 28;33(2):491–497. doi: 10.1016/0022-2836(68)90205-2. [DOI] [PubMed] [Google Scholar]
- Michel H., Oesterhelt D. Three-dimensional crystals of membrane proteins: bacteriorhodopsin. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1283–1285. doi: 10.1073/pnas.77.3.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel H. Three-dimensional crystals of a membrane protein complex. The photosynthetic reaction centre from Rhodopseudomonas viridis. J Mol Biol. 1982 Jul 5;158(3):567–572. doi: 10.1016/0022-2836(82)90216-9. [DOI] [PubMed] [Google Scholar]
- Nechushtai R., Muster P., Binder A., Liveanu V., Nelson N. Photosystem I reaction center from the thermophilic cyanobacterium Mastigocladus laminosus. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1179–1183. doi: 10.1073/pnas.80.5.1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reilly P., Hulmes J. D., Pan Y. C., Nelson N. Molecular cloning and sequencing of the psaD gene encoding subunit II of photosystem I from the cyanobacterium, Synechocystis sp. PCC 6803. J Biol Chem. 1988 Nov 25;263(33):17658–17662. [PubMed] [Google Scholar]
- Rögner M., Nixon P. J., Diner B. A. Purification and characterization of photosystem I and photosystem II core complexes from wild-type and phycocyanin-deficient strains of the cyanobacterium Synechocystis PCC 6803. J Biol Chem. 1990 Apr 15;265(11):6189–6196. [PubMed] [Google Scholar]
- Yeates T. O., Komiya H., Rees D. C., Allen J. P., Feher G. Structure of the reaction center from Rhodobacter sphaeroides R-26: membrane-protein interactions. Proc Natl Acad Sci U S A. 1987 Sep;84(18):6438–6442. doi: 10.1073/pnas.84.18.6438. [DOI] [PMC free article] [PubMed] [Google Scholar]