Structure-activity relationship studies of AMOR disaccharide in Torenia fournieri reveal the various residues that are critical for AMOR activity.
Abstract
Successful fertilization in flowering plants depends on the precise directional growth control of pollen tube through the female pistil tissue toward the female gametophyte contained in the ovule for delivery of nonmotile sperm cells. Cys-rich peptides LUREs secreted from the synergid cells on either side of the egg cell act as ovular attractants of pollen tubes. Competency control by the pistil is crucial for the response of pollen tubes to these ovular attractants. We recently reported that ovular 4-O-methyl-glucuronosyl arabinogalactan (AMOR) induces competency of the pollen tube to respond to ovular attractant LURE peptides in Torenia fournieri. The beta isomer of the terminal disaccharide 4-O-methyl-glucuronosyl galactose was essential and sufficient for the competency induction. However, critical and noncritical structures in the disaccharide have not been dissected deeply. Herein, we report the synthesis of new AMOR analogs and the structure-activity relationships for AMOR activity in the presence of these synthesized analogs. Removal of 4-O-methyl group or –COOH from the glucuronosyl residue of the disaccharide dramatically reduces AMOR activity. The pyranose backbone of the second sugar of disaccharide is essential for the activity but not hydroxy groups. The role of beta isomer of the disaccharide 4-Me-GlcA-β(1,6)-Gal is very specific for competency control, as there was no difference in effect among the sugar analogs tested for pollen germination. This study represents the first structure-activity relationship study, to our knowledge, of a sugar molecule involved in plant reproduction, which opens a way for modification of the molecule without loss of activity.
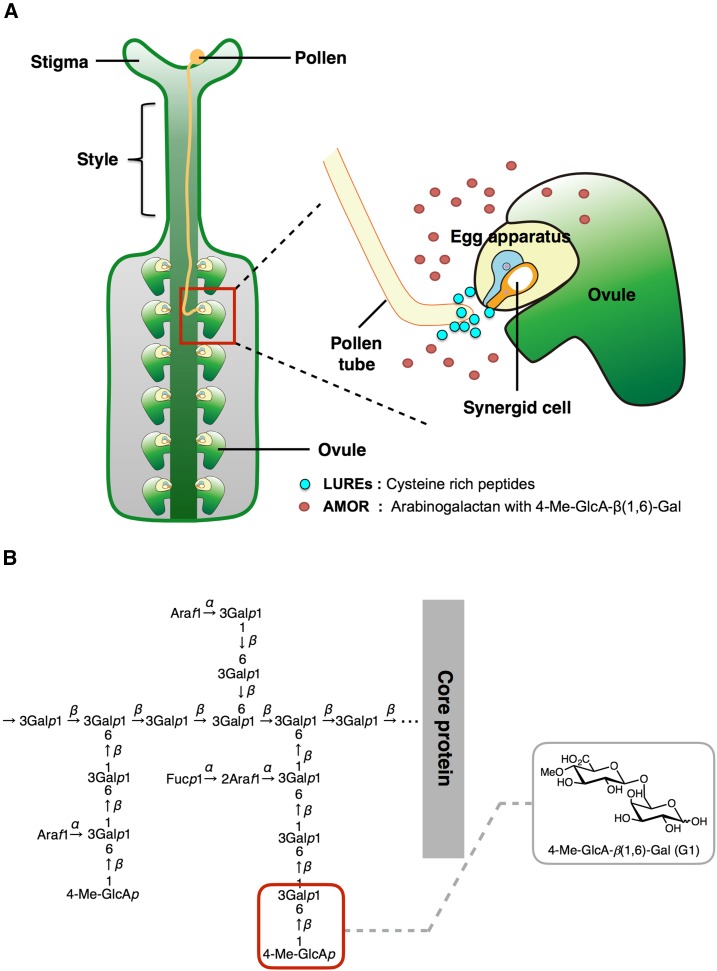
Fertilization in flowering plants is a complex process that involves several steps, starting from the landing of the pollen on the female stigma to the formation of a pollen tube, which is then precisely guided by the pistil tissue for the delivery of nonmotile sperm cells to the female gametes (Fig. 1A). Research spanning a decade has revealed that the pollen tubes communicate extensively with the female sporophytic tissue that provides various guidance cues to enable them to find their way to the ovules (Higashiyama and Takeuchi, 2015). The ovules also secrete Cys-rich peptides named LUREs that attract the pollen tubes to them before fertilization (Okuda et al., 2009). The concept of competency control of pollen tubes was established with the findings that in vitro-germinated pollen tubes of Torenia (Torenia fournieri) are not capable of being guided toward the ovules placed in front of them or to the gelatin beads releasing the LURE attractant peptides (Higashiyama et al., 1998; Okuda et al., 2013). However, the pollen tubes growing through cut, pollinated style of sufficient length responded to the LURE peptides in a semi-in vitro assay, suggesting that unknown factors from the female sporophytic tissues provides competency to the pollen tubes and primes them to respond to the attraction signal (Okuda et al., 2013). In Arabidopsis (Arabidopsis thaliana), an established plant model to study ovular pollen tube guidance (Higashiyama and Takeuchi, 2015), pollen tubes gain higher competency upon contact with the stigma and style (Palanivelu and Preuss, 2006). Though the requirement of female tissues for competency control was established in these different plant species, the female molecule responsible for competency control remained elusive for a decade.
Figure 1.
Ovular factor AMOR induces competency in pollen tubes to respond to ovular attractant. A, Schematic of pollen tube growing through a Torenia pistil toward an ovule. The pollen tube gain competency in presence of the ovular factor AMOR arabinogalactan that enables it to respond to the LURE attractant peptides secreted from the synergid cell. B, The terminal residue (4-O-methyl-glucuronosyl residue, highlighted by a red square) of an arabinogalactan polysaccharide shows AMOR activity.
In our efforts to identify the female molecule responsible for competency control, we discovered an ovular arabinogalactan polysaccharide, named 4-O-methyl-glucuronosyl arabinogalactan (AMOR), which induces competency of the pollen tubes to respond to ovular attractant LURE peptides in Torenia (Mizukami et al., 2016; Fig. 1A). Competency control in Torenia is two-step; pollen tubes have to grow through the style tissue (first step) before they can gain full competency by AMOR in the ovary (second step). The AMOR activity was found to reside in the terminal 4-O-methyl-glucuronosyl residue of the arabinogalactan polysaccharide. A chemically synthesized disaccharide, the beta isomer of 4-O-methyl-glucuronosyl Gal (4-Me-GlcA-β(1,6)-Gal (G1; Fig. 1B) showed AMOR activity and conferred competency to respond to ovular attractant LUREs (Mizukami et al., 2016).
Arabinogalactan polysaccharides are found to be associated with arabinogalactan proteins (AGPs), a complex family of glycoproteins, known as Hyp-rich glycoproteins, that play a major role in various aspects of plant growth, development, and reproduction (Seifert and Roberts, 2007; Ellis et al., 2010; Geshi et al., 2013; Pereira et al., 2014). AGPs are very heterogeneous, with the glycan constituting up to 90% of the mass of the molecule. The glycan part consists of variable side chains of Gal and arabinose sugars that are linked to O6 residue of the β-1,3-linked galactan backbone (Geshi et al., 2013). The galactan backbone is in turn added on to the proteins using O-glycosylation onto Hyp residues in the AGP backbone (Ogawa-Ohnishi and Matsubayashi, 2015). Most of the AGPs are found to have a GPI anchor at the C terminus that allows it to be anchored to the membrane and function as signaling molecules (Borner et al., 2002; Schultz et al., 2004). It has been proposed that AGPs are released to the extracellular matrix by the cleavage of GPI anchor by specific phospholipases (C and D), allowing it to act as a signal by itself or to activate downstream signal transduction pathways by interacting with other proteins (Gaspar et al., 2001; Schultz et al., 2004). There also exists a possibility of carbohydrate moieties being released from the AGPs via cleavage by specific enzymes, which allows these saccharides to function as signaling molecules that activate various signal transduction pathways by binding to specific receptors (Showalter, 2001). A chemical biology approach could enable us to better understand the developmental signaling pathways that are activated by the saccharide moieties of the AGPs.
In this study, we report the structure-activity relationship (SAR) studies of the AMOR disaccharide molecule (G1) that activates pollen tubes for ovular guidance in Torenia. Using a method for assaying response capability of a pollen tube by micromanipulating an ovule of Torenia, we analyzed the activity of various synthetic analogs of G1 that induces competency of pollen tubes and revealed various residues that are critical and noncritical for AMOR activity. We also carried out a pollen tube germination assay with the close analogs of G1 to assess if the AMOR activity was specific for competency control. This work represents the first SAR study, to our knowledge, of a bioactive sugar molecule involved in plant reproduction.
RESULTS
Synthesis of G1 and Its Analogs
The AMOR disaccharide G1 found in the terminal exposed portion of arabinogalactan sugar chain consists of two sugar units, 4-O-methyl GlcA and Gal, linked together by β-1,6 linkage. In the previous work, we designed a methyl etherified (C-4 position) benzyl glucosyluronate trichloroacetimidate as the glycosyl donor and benzyl protected Gal with unsubstituted hydroxy group at C-6 position as the glycosyl acceptor. With these two glycosylation partners (glycosyl donor and acceptor), we successfully obtained the 1,6-glycoside with a good β-selectivity (Mizukami et al., 2016). Both donor and acceptor can be synthesized from readily available Glc and Gal via classical carbohydrate synthesis. To illustrate the structural features of the AMOR disaccharide for its activity, we systematically synthesized a series of analogs of G1 and conducted SAR studies toward uncovering the key structural information (glycosidic bond and functional group on each sugar unit) that is essential for the AMOR activity.
Synthesis of Glycosyl Donors and Acceptors
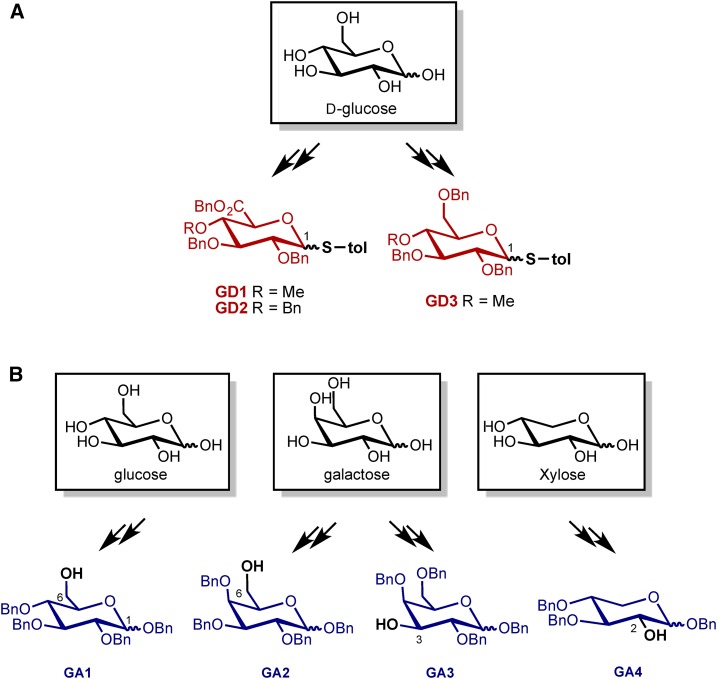
For the programmable synthesis of AMOR disaccharide analogs, we first changed the glycosyl donors from imidate to thioglycosides (Zhang et al., 1999). Thioglycosides are more general and stable glycosyl donors showing broad substrate scope in glycosylation (Codée et al., 2005). Starting from Glc, we synthesized three glycosyl donors, GD1, GD2, and GD3, aiming to disclose the group effect of 4-O-methyl and carboxyl acid (COOH) moieties in glucuronosyl residue (Fig. 2A).
Figure 2.
Synthesis of glycosyl donors from Glc and glycosyl acceptors from monosaccharides. A, The synthesis of glycosyl donors from Glc was demonstrated. Three thioglycoside analogs GD1, GD2, and GD3 as glycosyl donors were obtained by multistep synthesis. B, The glycosyl acceptors GA1, GA2, GA3, and GA4 were synthesized from several commercial available saccharides.
Four glycosyl acceptors (GA1–GA4) were synthesized from three widely commercial available monosaccharides (Fig. 2B). Each synthesized acceptor has one unprotected hydroxy group at a different position that was used for glycosylation with donors. This strategy allowed us to synthesize analogs that would reveal critical information about the second sugar unit (such as linkage effect) essential for AMOR activity.
Glycosylation and Deprotection of Disaccharide Analogs
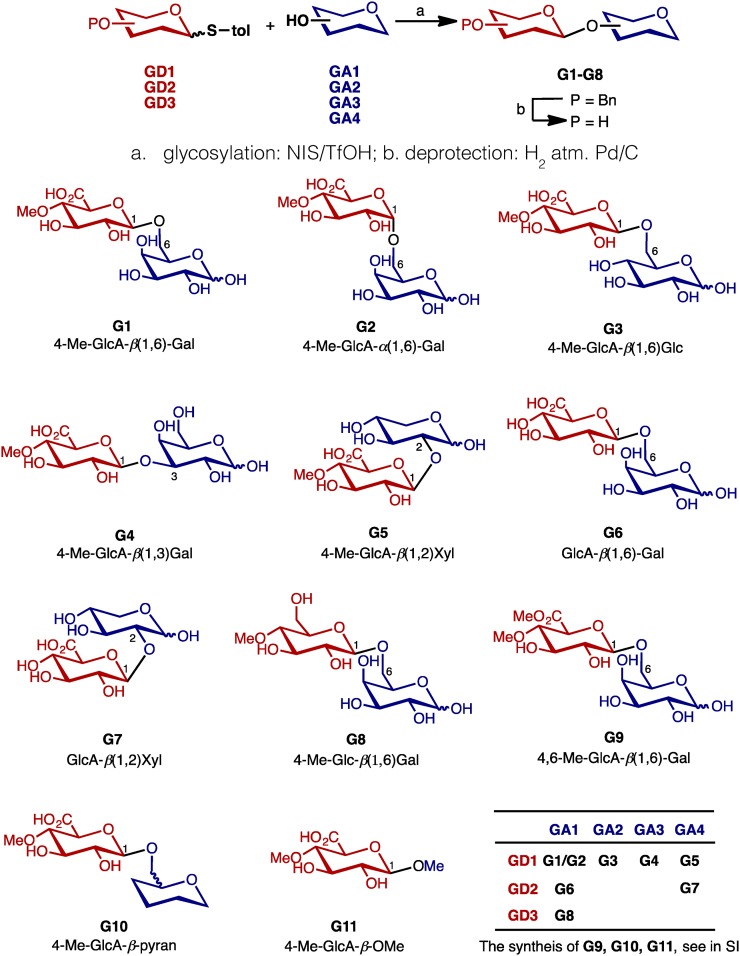
Having a reasonably diverse set of glycosyl donors (GD1–GD3) and acceptors (GA1–GA4) in hand, they were cross-coupled in a combinatorial manner under the influence of NIS/TfOH (Vermeer et al., 2001) to generate protected disaccharides (Fig. 3). The desired beta anomers were isolated from major alpha glycosylation products, and the follow-up deprotection of benzyl groups on hydroxy groups by Pd/C-catalyzed hydrogenation (Oguri et al., 1995) afforded disaccharides G1 to G8. A mild methylation of COOH group in G1 by trimethylsilyldiazomethane (TMSCHN2) gave its ester derivative 4,6-Me-GlcA-β(1,6)-Gal (G9; Presser and Hufner, 2004). To investigate the effect of hydroxy groups on a second sugar moiety (shown in blue), 4H-pyran-2-methanol was reacted with GD1 to synthesize G10. Although the synthesis of a simple methyl glycoside 4-Me-GlcA-β-OMe (G11) was not straightforward (the reaction of GD1 and methanol under NIS/TfOH yielded inseparable mixtures after glycosylation), G11 was synthesized from methyl β-d-glucopyranoside via a modified pathway (see Supplementary Information for details).
Figure 3.
Glycosylation of donor and acceptor for disaccharides analogs. The program glycosylation is shown in the above scheme, using the synthesized glycosyl donor and acceptor mentioned in Figure 2. The structures of all disaccharides obtained are shown below; for G1 to G8, the combinations of their original donor and acceptor were listed in the small table.
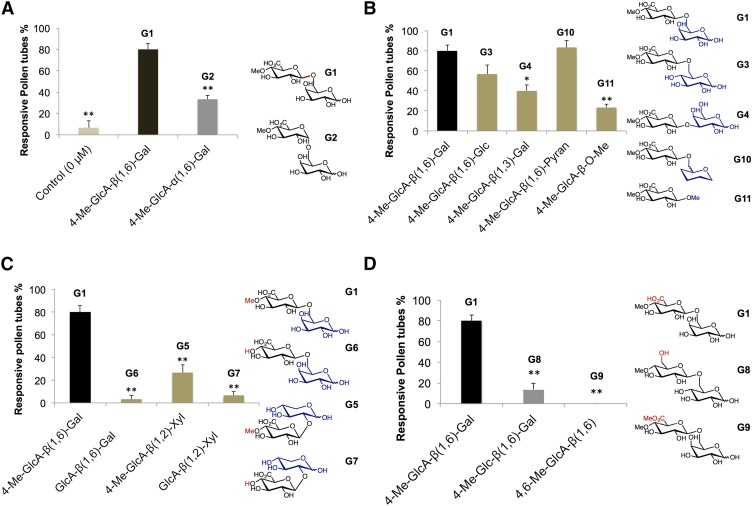
The characteristics of synthesized disaccharide analogs 4-Me-GlcA-α(1,6)-Gal (G2) to G11 (Fig. 3) are as follows. G2, which has an α-glycosidic bond, is an anomer of G1 (stereochemical importance of glycosidic bond). G3 to 4-Me-GlcA-β(1,2)-Xyl (G5) have same 4-O-methyl glucuronosyl residue as G1 but a different second sugar unit (G1: Gal, G3: Glc, G5: Xyl) or different linkage (G1: 1,6-linkage, G4: 1,3-linkage). For disaccharides β-glucuronosyl galactose (G6) and G7, the 4-O-methyl group on the glucuronosyl residue is lacking. Both G8 and G9 have modified COOH functionalities such as hydroxymethyl group (G8) and methyl ester group (G9; the effect of carboxylic acid moiety). The last two specific saccharides (G10 and G11) with simple alkyl aglycones (G10: alkyl = -CH2-4H-pyran, G11: alkyl = CH3) are substrates to illustrate the effect of second sugar moiety. G10 bears a simple pyranose backbone (all hydroxy groups on Gal unit of G1 removed). Methyl glycoside G11 is a monosaccharide that features only a 4-O-methyl glucuronosyl residue.
AMOR Activity of Synthesized Disaccharides
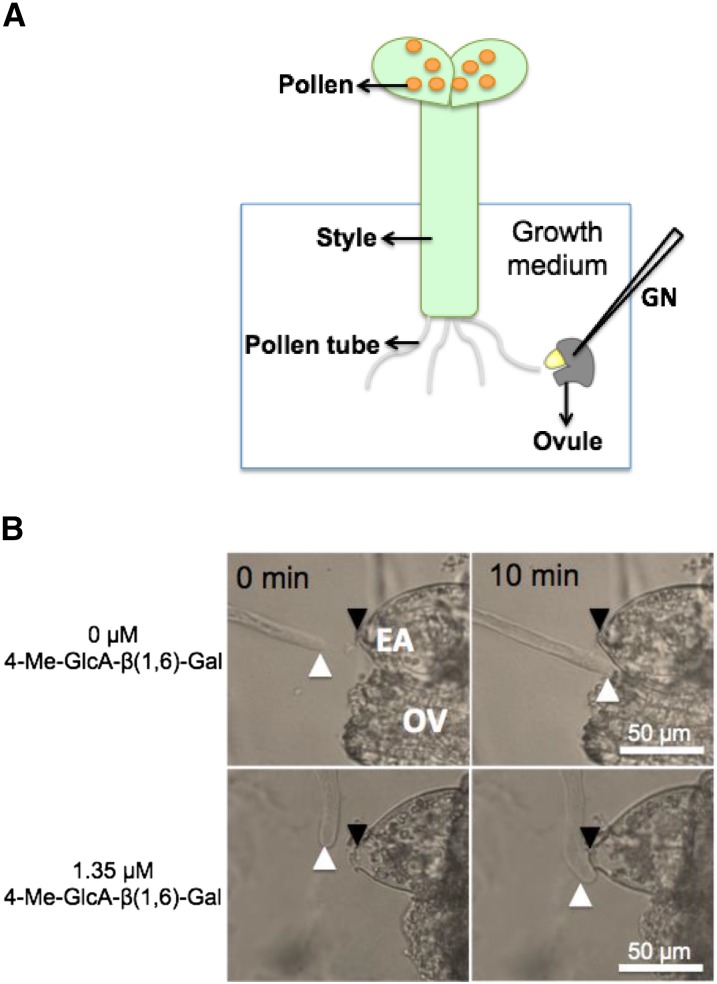
To determine the effect of synthesized disaccharides on the acquisition of competency by the pollen tubes and their response to ovular attraction (AMOR activity), we performed a semi-in vitro Torenia ovule-manipulating assay using a glass needle (Fig. 4A; Mizukami et al., 2016). A hand-pollinated pistil (15 mm) is excised and cultivated in a thin layer of agar growth medium containing the synthesized disaccharide. A single ovule excised from a fresh ovary is positioned in front of the pollen tube with a glass needle. The competent pollen tube is attracted to the micropylar end of the embryo sac when placed within 30 μm in front of the pollen tube (Fig. 4 A and B), while the noncompetent pollen tube fails to target the ovule placed in front of it (Fig. 4B). It was observed that the pollen tube acquired competency when the growth medium contained 1.35 μm of G1 (Fig. 4B).
Figure 4.
AMOR assay to evaluate the responsiveness of a pollen tube. A, Schematic of the semi-in vitro pollen tube guidance assay for quantification of pollen tube competency (responsiveness) to ovular attraction. As shown, pollen tube growing through the cut style is allowed to grow on a growth medium with/without the disaccharide to examine their response capability by placing a freshly prepared ovule near the tip of an elongating pollen tube using a glass needle (GN). B, AMOR assay showing a pollen tube (white arrowhead) growing away from the opening of egg apparatus (black arrowhead, EA) of an ovule (OV) placed in front of it in absence of AMOR disaccharide (top). In the presence of G1 (1.35 µM), the pollen tube is attracted toward the opening of the egg apparatus (bottom).
All of the synthesized disaccharides variants were tested for AMOR activity. The disaccharides have been grouped into different categories based on the type of linkage, presence or absence of functional residues, and their arrangement in sugar molecule to make the interpretations of results easier (Fig. 5).
Figure 5.
AMOR activity of synthesized analogs. A, AMOR activity of G1 versus G2 and the control. B, AMOR activity of G1 versus G3 analog bearing a 1,6-linked Glc, G4 analog bearing a 1,3-linked Gal, G10 analog with a pyran backbone, and G11 analog bearing a methyl glycoside. C, AMOR activity of G1 versus analogs G6 (lacking 4-O-Me) and G5 versus G7 (lacking 4-O-Me). D, AMOR activity of G1 versus hydroxyl analog (G8) and methyl ester analog (G9). Concentration of the synthesized analogs used in the assay is 1.35 μm. Data are means of three independent experiments (n = 30 pollen tubes). Bars represent the sem. All statistical comparisons were made against G1. Significant differences were calculated by Student’s t test, one-way ANOVA with posthoc Tukey HSD test, and Scheffé test (*P < 0.05,**P < 0.01).
G1 having beta linkage showed a significantly greater percentage of pollen tubes attracted to the ovules than the control (growth media without G1) and its alpha anomer G2 (2.4 times) when included in the assay medium (Fig. 5A; Supplemental Table S1) as reported by Mizukami et al. (2016). Replacement of Gal residue (second saccharide) of G1 with a similar sugar residue such as Glc (G3) did not lead to significant difference in the percentage of pollen tubes being attracted to the ovules (Fig. 5B; Supplemental Table S1). Disaccharide G4, with an unusual (1,3)-linkage resulted in a 2-fold drop in percentage of pollen tubes being attracted to the ovules than G1 (Fig. 6B). A similar result was also observed in the case of disaccharide G5 having (1,2)-linkage (Fig. 5C). To our great surprise, addition of the pyranose analog G10 in the assay medium resulted in a greater percentage of pollen tubes being attracted to the ovules, which was comparable with G1 (Fig. 5B; Supplemental Table S1). However, complete removal of sugar-like structure is detrimental, as inclusion of G11 in the assay medium resulted in 3.4-fold drop in the percentage of pollen tubes being attracted to the ovules compared to G1. 4-O-methyl group in the glucuronosyl residue was found to be essential for AMOR activity.
Figure 6.
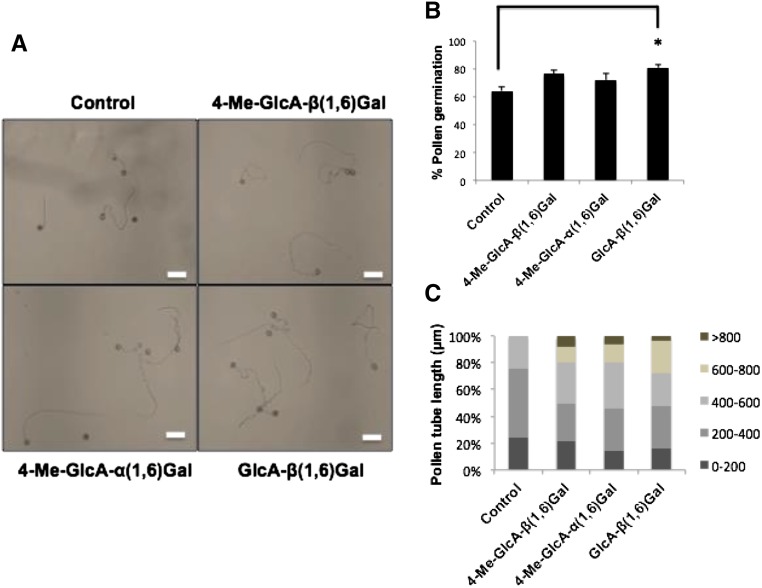
Torenia pollen germination assay with synthesized sugar variants. A, Picture shows the effect of 4-Me-GlcA-β(1,6)-Gal, 4-Me-GlcA-α(1,6)-Gal and GlcA-β(1,6)-Gal (G6) on germination of Torenia pollen (scale bar = 100 μm). B, Percentage pollen germination in the presence of 4-Me-GlcA-β(1,6)-Gal, 4-Me-GlcA-α(1,6)-Gal, and GlcA-β(1,6)-Gal in germination medium compared to control without any of these sugars. Concentration of each of the disaccharide used in the germination media is 1.35 μm. Data are average of six biological replicates. One-way ANOVA with posthoc Tukey HSD test and Bonferroni and Holm tests were used to calculate significant differences and for multiple comparisons (*P < 0.01). C, Percentage of pollen with different pollen tube length (μm) after 2 h of germination in the presence or absence of 4-Me-GlcA-β(1-6)-Gal, 4-Me-GlcA-α(1,6)-Gal, and GlcA-β(1,6)-Gal in the pollen germination medium. Length of 50 pollen tubes was measured for each treatment.
Elimination of methyl group from the C-4 position of G6 led to about 24-fold reduction in the percentage of pollen tubes being attracted to the ovules compared to G1 (Fig. 5C; Supplemental Table S1) as reported by Mizukami et al. (2016). This result was consistent with that of disaccharides having (1,2)-Xyl linkage (G5 and G7). Removal of 4-O-methyl group (G5→G7) resulted in a 4-fold reduction in the percentage of pollen tubes being attracted to the ovules compared to G1 (Fig. 5C; Supplemental Table S1). The COOH group in the C-5 position of G1 was also essential to the functionality of glucuronosyl residue. The addition of hydroxymethyl analog G8 in the assay medium resulted a 6-fold reduction in the percentage of pollen tubes being attracted to the ovules compared to G1, while the methyl ester analog G9 led to complete loss of competency response, as none of the pollen tubes respond to the nearby ovules in its presence compared to G1.
Pollen Germination Activity of Synthesized Disaccharides
Since AGPs are known to promote pollen germination and pollen tube growth, the disaccharide G1 and its closest analogs G2 and G6 were tested for their ability to induce pollen germination and pollen tube growth (Fig. 6). There was no significant difference in the pollen germination rates among these analogs. However, pollen germination was significantly improved (1.2-fold) upon germination of pollen in medium containing G6 compared to the control without synthesized sugars (Fig. 6B). All three synthesized sugars improved the pollen tube growth compared to the control; however, there was no significant difference in pollen tube length between the sugars (Fig. 6, A and C).
DISCUSSION
AGPs play an important role in plant growth and development. The major component of AGPs is the glycan moiety, which often constitutes >90% of the molecule (Knoch et al., 2014). The differences in glycan structure of AGPs could be used as markers in plant development and reproduction (Coimbra et al., 2007; Dilokpimol et al., 2014). The understanding of the functional roles of glycan moiety of AGPs in plants has relied on either the use of β-Yariv reagents that bind to β-1,3-galactan moiety of AGPs (Kitazawa et al., 2013) or monoclonal antibodies that recognize different AGP glycan epitopes (Seifert and Roberts, 2007). Due to the complex and heterogeneous nature of the glyan moieties, it has been difficult to study the structure-function relationship of AGP glycans. In the future, a chemical biology approach together with classical genetics utilizing mutants of enzymes involved in biosynthesis of the glycan moiety of arabinogalactans would enable us to dissect the functional roles of glycan moieties of AGPs.
AGPs are prevalent in the reproductive tissues of plants, and they have been proposed to play prominent roles in plant sexual reproduction (Pereira et al., 2015). Our studies to investigate the process of competency control during pollen tube guidance led to the discovery of an ovular factor AMOR (Arabinogalactan molecule) that induces competency in Torenia pollen tubes to respond to LURE attractants released from the ovules (Mizukami et al., 2016). It was further revealed that AMOR activity resided in the terminal methyl-glucuronosyl residue of the arabinogalactan (Mizukami et al., 2016) polysaccharide and a chemically synthesized disaccharide, the beta epimer of methyl-glucuronosyl Gal (G1), showed full AMOR activity (Mizukami et al., 2016). Though 4-Me-GlcA-Gal is found on the exposed side chains of AGPs, their functional role has remained elusive.
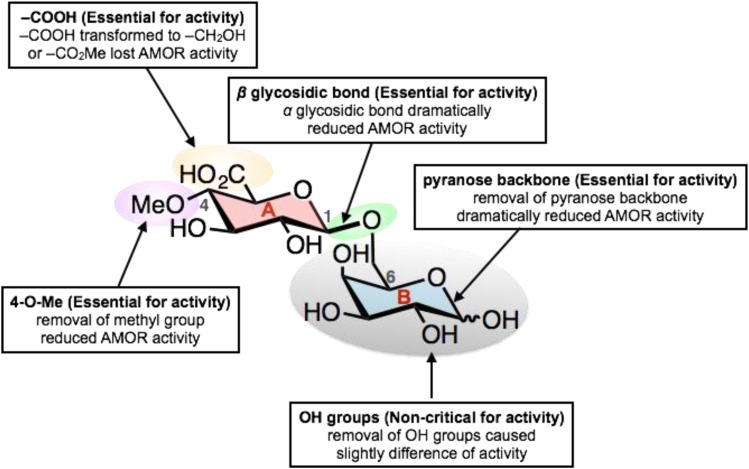
We carried out an SAR study with the goal to decipher the relationship between various residues present on the AMOR disaccharide and their influence on ability of the pollen tubes to respond to ovular guidance cues (competency control). Using a synthetic chemistry approach, we have determined the requirement of disaccharide structure for AMOR activity using G1 and its analogs (Fig. 7). The studies revealed that the presence of 4-O-methyl-glucuronosyl (ring A) is essential for AMOR activity. Both the 4-O-methyl and –COOH found on ring A were irreplaceable for activity. The importance of glucuronosyl residue for AMOR activity is also supported by our previous work, where in hydrolytic degradation of the terminal glucuronosyl residues of AGPs with glucuronidase dramatically decreased the AMOR activity (Mizukami et al., 2016).
Figure 7.
Effect of structural modification of G1 on AMOR activity in Torenia. The SAR results were concluded and showed clearly of all structural information related to AMOR activity.
Stereochemistry of the sugar linkage (β-glycosidic bond) is also an important structural element for the AMOR activity. This is consistent with the fact that β-glycosidic bonds are found naturally between saccharide units of AGPs (Knoch et al., 2014). The pyranose backbone (ring B) of AGPs was also found to be essential for activity, but the surprising discovery was that the simple tetrahydropyran moiety could replace the complex Gal moiety in terms of AMOR activity.
Through this SAR study, we identified the crucial structure of AMOR ligand required to maintain its affinity for the putative receptor. The variation in responses of the pollen tubes to various structural variants of the disaccharide is most likely a result of the differences in affinity of these variants to the putative AMOR receptor. The effect of these structural variants on competency control is presumably dosage dependent, as the presence of higher concentrations of some of the variants in the medium would result in a greater proportion of pollen tubes becoming competent to respond to the guidance cues. The discovery of noncritical residues of the AMOR disaccharide would allow live cell imaging to study AMOR localization, dynamics, and binding to the putative receptor by attaching fluorescent tags to these residues without loss of AMOR activity. The terminal -OH group found on the pyranose backbone of the second sugar would be most suitable for this purpose, as it does not lead to any loss of activity. Use of fluorescent ligands with appropriate imaging properties can also be used to investigate the regulation of putative receptor after activation by the AMOR disaccharide. The use of fluorescent AMOR ligand could reveal the spatial organization of the putative AMOR receptors in the pollen tube and thus aid in their identification.
Arabinogalactan proteins are highly expressed in pollen, and they play an important role in pollen development, germination, and pollen tube growth (Coimbra et al., 2009; Costa et al., 2013; Pereira et al., 2014). Pollen tube growth was severely affected by the Yariv phenyl glycoside, a reagent known to bind specifically to AGPs (Roy et al., 1998; Mollet et al., 2002). Immunolabeling studies using monoclonal antibodies specific to several glycosidic epitopes of AGPs have revealed distinct distribution of AGPs in the in vitro-germinated pollen tubes of Nicotiana tabacum and Arabidopsis, further strengthening their importance in pollen germination and growth (Li et al., 1992; Pereira et al., 2006). The LM2 monoclonal antibody that specifically recognizes the carbohydrate containing epitope of β-linked GlcA produced a ring-like fluorescence around the emerging region of pollen tubes, suggesting a role for GlcA residue in pollen tube growth (Pereira et al., 2006). Thus, we decided to test if Me-GlcA-β(1,6)-Gal and its closest analogs had an effect on pollen germination and pollen tube growth. There was no statistical difference in pollen germination or pollen tube growth in the presence of Me-GlcA-β(1,6)-Gal in the medium compared to control, suggesting that its role was specific to competency control. Interestingly, GlcA-β(1,6)-Gal enhanced pollen germination compared to control, suggesting that the 4-O-methyl group in glucuronosyl residue, which is essential for AMOR, may be detrimental to pollen germination. Stereochemistry of linkage (α vs beta glycosidic bond) is also not important for pollen germination, as both the anomers of Me-GlcA-(1,6)-Gal displayed similar rates of pollen germination and pollen tube growth. It is of interest to know how biosynthesis of arabinogalactan sugar chain is spatiotemporally regulated in the pistil by β-glucuronosyltransferases (for addition of GlcA to the galactan; Geshi et al., 2013; Knoch et al., 2014) and by unidentified methyltransferases (for addition of the 4-O-methyl group to the glucuronosyl galactan).
The identification of the role of 4-Me-GlcA-Gal and the dissection of its critical residues in imparting competency of pollen tubes not only enhance our understanding of the complex fertilization process in flowering plants, but also add to our understanding of plant sugar signaling. The chemical biology approach described in this work provides a great opportunity to gain a molecular-level understanding of these disaccharides and elucidate their functional roles. This SAR study would serve as a guideline for future studies aimed to understand sugar signaling in plant reproduction. This study forms the first step toward understanding the mechanism of AMOR action, including the identification of its putative receptor and the downstream signaling pathways.
MATERIALS AND METHODS
Chemical Synthesis of Disaccharides
See the full experimental details for synthesis of all compounds in the supplemental information. The purity of all synthesized compounds was verified, and pure compounds were used for biological studies.
Plant Materials and Growth Conditions
Torenia fournieri cv ‘blue and white’ were grown on soil at 28°C with a 16-h photoperiod [approximately 150 μmol/(m−2 s−1)]. We used flowers from these plants for the AMOR assay and pollen germination assay.
AMOR Assay
AMOR activity was measured using AMOR assay performed as described previously (Mizukami et al., 2016). A glass-bottom dish (D210402; Matsunami) was covered with modified Nitsch’s medium. The center of the agar plate was cut into an approximately 18- × 22-mm2 square and the agar block was removed. Growth medium (5-fold modified Nitsch’s medium containing 13% polyethylene glycol 4,000 [w/v] and 1% Suc [w/v]) with synthesized saccharides (1.35 µM, 13.5 µM) and 1.5% ultra-low gelling agarose (agarose type IX-A; Sigma) were filled up to 220 µL with 20 mM MES/KOH (pH 6.8) and poured into the square. Before solidification, the cut end of a 15-mm-long hand-pollinated style was embedded at the center of the growth medium. This agar plate was incubated in the dark at 28°C for 14 h. After incubation, water-saturated silicone oil (KF-96-100CS; Shin-Etsu Chemical) was layered to the growth medium and hand-pollinated style. Ovules were dissected from the pistils on the growth medium under observation using a stereomicroscope (SZ61, Olympus). A single ovule was picked up using a glass needle produced with a glass needle puller (PC-10; Narishige) and placed in front of the pollen tubes using manipulator (MMN-1, MMO-202N, MMO-220A, Narishige) under observation using an inverted microscope (IX71, Olympus). Pollen tube response was evaluated based on the attraction of the pollen tube to the micropylar end of the embryo sac. The pollen tube was scored as responsive if, when placed within 30 μm of an ovule, the pollen tube altered its direction of growth and continued growing to make contact with the micropylar end of egg apparatus. The pollen tubes were scored as nonresponsive if they did not alter their direction of growth or did not make contact with the egg apparatus.
Torenia Pollen Germination Assay
Pollen was collected from the anthers of fully open Torenia flowers and suspended in a 2-mL micro centrifuge tube containing liquid growth medium (2-folded modified Nitsch’s medium containing 13% polyethylene glycol 4,000 [w/v] and 2% Suc [w/v], pH 6.5). The suspended pollen-containing medium was mixed well and split equally into different microcentrifuge tubes with synthesized saccharides (1.35 µM) or without it (control). By this method, the pollen density was kept uniform across the treatments to control the population effect. These microcentrifuge tubes were incubated in dark at 28°C for 2 h. After 2 h, the liquid suspension containing germinated pollen was placed on a microslide and observed under bright field with an Axio Imager2 (Zeiss) microscope. Percentage germination was calculated by counting 100 pollen grains from different fields of microslide. Data were collected from six different biological replicates. Pollen tube length was calculated using the ImageJ software from 50 pollen tubes for each treatment selected at random.
Supplemental Data
The following supplemental materials are available.
Supplemental Figure S1. Synthesis of glycosyl donors GD1 and GD2.
Supplemental Figure S2. Synthesis of glycosyl donor GD3.
Supplemental Figure S3. Synthesis of glycosyl acceptor GA1 and GA2.
Supplemental Figure S4. Synthesis of glycosyl acceptor GA3.
Supplemental Figure S5. Synthesis of glycosyl acceptor GA4.
Supplemental Figure S6. Programmable glycosylation of glycosyl donor and acceptor, the yields of glycosylation product G’ and the deprotection product G (the total yield of two anomers and ratio of β:α is shown in parenthesis).
Supplemental Figure S7. Synthesis of G9.
Supplemental Figure S8. Synthesis of 4-Me-GlcA-β-OMe (G11).
Supplemental Table. AMOR activity results for synthetic AMOR and derivatives.
Supplemental Materials and Methods. Supplemental Materials and Methods.
Supplementary Material
Acknowledgments
We thank Dr. Keiko Kuwata (ITbM, Nagoya University) for HRMS measurements. We thank Dr. Toshihisa Kotake (Saitama University, Japan) for critical reading of this manuscript.
Glossary
- AGP
arabinogalactan protein
- AMOR
4-O-methyl-glucuronosyl arabinogalactan
- COOH
carboxyl acid
- G1
4-Me-GlcA-β(1,6)-Gal
- G2
4-Me-GlcA-α(1,6)-Gal
- G5
4-Me-GlcA-β(1,2)-Xyl
- G6
β-glucuronosyl galactose
- G9
4,6-Me-GlcA-β(1,6)-Gal
- G11
4-Me-GlcA-β-OMe
- SAR
structure-activity relationship
Footnotes
S.S. is supported by a postdoctoral fellowship from JSPS. This work was supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (16H06465, 16H06464, 16K21727, and 15F15388 to T.H.), Japan Advanced Plant Science Network, and Japan Science and Technology Agency (ERATO project to T.H.).
Articles can be viewed without a subscription.
References
- Borner GHH, Sherrier DJ, Stevens TJ, Arkin IT, Dupree P (2002) Prediction of glycosylphosphatidylinositol-anchored proteins in Arabidopsis. A genomic analysis. Plant Physiol 129: 486–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Codée JDC, Litjens REJN, van den Bos LJ, Overkleeft HS, van der Marel GA (2005) Thioglycosides in sequential glycosylation strategies. Chem Soc Rev 34: 769–782 [DOI] [PubMed] [Google Scholar]
- Coimbra S, Almeida J, Junqueira V, Costa ML, Pereira LG (2007) Arabinogalactan proteins as molecular markers in Arabidopsis thaliana sexual reproduction. J Exp Bot 58: 4027–4035 [DOI] [PubMed] [Google Scholar]
- Coimbra S, Costa M, Jones B, Mendes MA, Pereira LG (2009) Pollen grain development is compromised in Arabidopsis agp6 agp11 null mutants. J Exp Bot 60: 3133–3142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa M, Nobre MS, Becker JD, Masiero S, Amorim MI, Pereira LG, Coimbra S (2013) Expression-based and co-localization detection of arabinogalactan protein 6 and arabinogalactan protein 11 interactors in Arabidopsis pollen and pollen tubes. BMC Plant Biol 13: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dilokpimol A, Poulsen CP, Vereb G, Kaneko S, Schulz A, Geshi N (2014) Galactosyltransferases from Arabidopsis thaliana in the biosynthesis of type II arabinogalactan: molecular interaction enhances enzyme activity. BMC Plant Biol 14: 90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis M, Egelund J, Schultz CJ, Bacic A (2010) Arabinogalactan-proteins: key regulators at the cell surface? Plant Physiol 153: 403–419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaspar Y, Johnson KL, McKenna JA, Bacic A, Schultz CJ (2001) The complex structures of arabinogalactan-proteins and the journey towards understanding function. Plant Mol Biol 47: 161–176 [PubMed] [Google Scholar]
- Geshi N, Johansen JN, Dilokpimol A, Rolland A, Belcram K, Verger S, Kotake T, Tsumuraya Y, Kaneko S, Tryfona T, et al. (2013) A galactosyltransferase acting on arabinogalactan protein glycans is essential for embryo development in Arabidopsis. Plant J 76: 128–137 [DOI] [PubMed] [Google Scholar]
- Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1998) Guidance in vitro of the pollen tube to the naked embryo sac of Torenia fournieri. Plant Cell 10: 2019–2032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higashiyama T, Takeuchi H (2015) The mechanism and key molecules involved in pollen tube guidance. Annu Rev Plant Biol 66: 393–413 [DOI] [PubMed] [Google Scholar]
- Kitazawa K, Tryfona T, Yoshimi Y, Hayashi Y, Kawauchi S, Antonov L, Tanaka H, Takahashi T, Kaneko S, Dupree P, et al. (2013) β-galactosyl Yariv reagent binds to the β-1,3-galactan of arabinogalactan proteins. Plant Physiol 161: 1117–1126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knoch E, Dilokpimol A, Geshi N (2014) Arabinogalactan proteins: focus on carbohydrate active enzymes. Front Plant Sci 5: 198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li YQ, Bruun L, Pierson ES, Cresti M (1992) Periodic deposition of arabinogalactan epitopes in the cell wall of pollen tubes of Nicotiana tabacum L. Planta 188: 532–538https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=24178385&dopt=Abstract [DOI] [PubMed] [Google Scholar]
- Mizukami AG, Inatsugi R, Jiao J, Kotake T, Kuwata K, Ootani K, Okuda S, Sankaranarayanan S, Sato Y, Maruyama D, et al. (2016) The AMOR arabinogalactan sugar chain induces pollen-tube competency to respond to ovular guidance. Curr Biol 26: 1091–1097 [DOI] [PubMed] [Google Scholar]
- Mollet JC, Kim S, Jauh GY, Lord EM (2002) Arabinogalactan proteins, pollen tube growth, and the reversible effects of Yariv phenylglycoside. Protoplasma 219: 89–98 [DOI] [PubMed] [Google Scholar]
- Ogawa-Ohnishi M, Matsubayashi Y (2015) Identification of three potent hydroxyproline O-galactosyltransferases in Arabidopsis. Plant J 81: 736–746 [DOI] [PubMed] [Google Scholar]
- Oguri H, Hishiyama S, Oishi T, Hirama M (1995) Enantiocontrolled synthesis of the Ab ring moiety of ciguatoxin. Synlett 1252–1254 [Google Scholar]
- Okuda S, Suzuki T, Kanaoka MM, Mori H, Sasaki N, Higashiyama T (2013) Acquisition of LURE-binding activity at the pollen tube tip of Torenia fournieri. Mol Plant 6: 1074–1090 [DOI] [PubMed] [Google Scholar]
- Okuda S, Tsutsui H, Shiina K, Sprunck S, Takeuchi H, Yui R, Kasahara RD, Hamamura Y, Mizukami A, Susaki D, et al. (2009) Defensin-like polypeptide LUREs are pollen tube attractants secreted from synergid cells. Nature 458: 357–361 [DOI] [PubMed] [Google Scholar]
- Palanivelu R, Preuss D (2006) Distinct short-range ovule signals attract or repel Arabidopsis thaliana pollen tubes in vitro. BMC Plant Biol 6: 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira AM, Masiero S, Nobre MS, Costa ML, Solís MT, Testillano PS, Sprunck S, Coimbra S (2014) Differential expression patterns of arabinogalactan proteins in Arabidopsis thaliana reproductive tissues. J Exp Bot 65: 5459–5471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira AM, Pereira LG, Coimbra S (2015) Arabinogalactan proteins: rising attention from plant biologists. Plant Reprod 28: 1–15 [DOI] [PubMed] [Google Scholar]
- Pereira LG, Coimbra S, Oliveira H, Monteiro L, Sottomayor M (2006) Expression of arabinogalactan protein genes in pollen tubes of Arabidopsis thaliana. Planta 223: 374–380 [DOI] [PubMed] [Google Scholar]
- Presser A, Hufner A (2004) Trimethylsilyldiazomethane: a mild and efficient reagent for the methylation of carboxylic acids and alcohols in natural products. Monatsh Chem 135: 1015–1022 [Google Scholar]
- Roy S, Jauh GY, Hepler PK, Lord EM (1998) Effects of Yariv phenylglycoside on cell wall assembly in the lily pollen tube. Planta 204: 450–458 [DOI] [PubMed] [Google Scholar]
- Schultz CJ, Ferguson KL, Lahnstein J, Bacic A (2004) Post-translational modifications of arabinogalactan-peptides of Arabidopsis thaliana. Endoplasmic reticulum and glycosylphosphatidylinositol-anchor signal cleavage sites and hydroxylation of proline. J Biol Chem 279: 45503–45511 [DOI] [PubMed] [Google Scholar]
- Seifert GJ, Roberts K (2007) The biology of arabinogalactan proteins. Annu Rev Plant Biol 58: 137–161 [DOI] [PubMed] [Google Scholar]
- Showalter AM. (2001) Arabinogalactan-proteins: structure, expression and function. Cell Mol Life Sci 58: 1399–1417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vermeer HJ, van Dijk CM, Kamerling JP, Vliegenthart JFG (2001) Fucosylation of linear alcohols: a study of parameters influencing the stereochemistry of glycosylation. Eur J Org Chem 193–203 [Google Scholar]
- Zhang ZY, Ollmann IR, Ye XS, Wischnat R, Baasov T, Wong CH (1999) Programmable one-pot oligosaccharide synthesis. J Am Chem Soc 121: 734–753 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.