Abstract
Mammalian cells are confronted with changes in extracellular osmolality at various sites, including the aqueous layer above the lung epithelium. Hypertonic shock induces the activation of mitogen-activated protein kinases and the expression of a defined set of genes, including aquaporins. We investigated upstream components of the response to hypertonicity in lung epithelial cells and found that before extracellular signal-regulated kinase activation and aquaporin synthesis, the membrane-bound prohormone neuregulin 1-β is cleaved and binds to human epidermal growth factor receptor 3 (HER3). The signaling is prevented by matrix metalloproteinase inhibition, inhibition of neuregulin 1-β binding to HER3, and inhibition of HER tyrosine kinase activity. Inhibition of HER activation interferes with the hypertonic induction of two different aquaporins in three distinct cell lines of mouse and human origin. We propose that ligand-dependent HER activation constitutes a generalized signaling principle in the mammalian hypertonic stress response relevant to aquaporin expression.
Keywords: cell volume, epithelium, osmotic stress, proteolytic cleavage, extracellular signal-regulated kinase
Detection of changes in the environment and communication of information into the cell is fundamental to cellular function. Numerous extracellular stimuli elicit highly specific cellular responses. Whereas certain environmental factors such as heat (1) or UV light (2, 3) appear to elicit direct intracellular effects, ligand-dependent activation (e.g., by hormones, growth factors, or cytokines) of membrane-bound receptors is a frequent theme.
Cells at all levels of life are exposed to changes in extracellular osmolality that require cellular defense mechanisms to maintain cell homeostasis and volume (4, 5). Hypertonic stress leads to cell shrinkage over seconds to minutes followed by uptake of ions, primarily potassium (K+), and water to restore cell volume. Over the course of several hours, cells exchange ions for organic osmolytes, relatively nontoxic molecules taken up from the extracellular space (e.g., betaine, myo-inositol, taurine) or produced within the cell (e.g., sorbitol).
The mammalian kidney, where osmolality in the renal medulla may reach 1 M, provides an exquisite example of cellular adaptation to hypertonicity (6, 7, 8). In the lung, the osmolality of the aqueous surface layer covering the airway epithelium may range from being hypotonic to ≥400 milliosmolal (mOsm) (9, 10). Changes in the physical properties of the surface layer, including its osmolality, can alter the effectiveness of mucociliary clearance (11), provoke airway constriction (12, 13), or reduce the bactericidal activity of defensins, antibacterial peptides that are secreted into the airways (14). These events are relevant to disease development in the lung (e.g., in the pathogenesis of infections and in some forms of asthma).
In addition to up-regulation of organic osmolyte transporters, hypertonicity induces expression of aquaporin (AQP) water-channel proteins (15, 16, 17). Aquaporins are the primary determinants of osmotic gradient-dependent rapid water movement in or out of cells (18). Hypertonic induction of AQP5, a water-channel protein expressed in lung and several secretory epithelia (16), and AQP1, which is expressed at multiple sites including lung and kidney (16, 17), require activation of mitogen-activated protein kinase (MAPK) signaling cascades, which are known to participate in osmotic signaling in yeast and mammals (19). MAPK activation often follows the activation of G protein-coupled receptors (GPCRs) and/or receptor tyrosine kinases (RTKs), e.g., the epidermal growth factor receptor (EGFR) or human epidermal growth factor receptor (HER). Early signaling events that lead to MAPK activation in response to osmotic stress in mammalian cells are not well defined.
Here, we describe links between osmotic stress, the cleavage of an EGF-family prohormone, neuregulin 1-β (NRG1-β), extracellular signal-regulated kinase (ERK) activation, and the expression of aquaporins. We show that metalloproteinase-dependent cleavage of pro-NRG1-β and subsequent HER2/3 dimer activation is a necessary early step in the hypertonic induction of AQP5 in lung epithelial cells. In addition, we find that HER activation is required for the hypertonic induction of aquaporins in a human lung epithelial cell line as well as in primary human keratinocytes, suggesting that HER-dependent regulation of AQP expression represents a generalized signaling paradigm.
Materials and Methods
Materials. The following materials were used for these studies: electrophoresis reagents from Bio-Rad, enhanced chemiluminescence (ECL+) from Amersham Pharmacia Biotech, a bicinchoninic acid (BCA) protein assay kit and crosslinker 3,3′-dithiobis(sulfosuccinimidyl propionate) (DTSSP) from Pierce, and AG1478 and AG1296 (solubilized in DMSO) from Calbiochem. The following antibodies were used: anti-total and antiphosphorylated ERK from New England Biolabs; anti-EGF receptor isoforms 1–4 (HER1–4), anti-NRG1-precursor -α and -β, anti-phosphatidylinositol 3-kinase (PI3-kinase) subunit p85α, anti-growth factor receptor-bound protein 2 (GRB2), and anti-GRB7 from Santa Cruz Biotechnology; anti-phosphotyrosine from Upstate Biotechnology (Lake Placid, NY); anti-Shc from BD Transduction Laboratories (San Diego); anti-AQP3 from Alpha Diagnostics (San Antonio, TX); neutralizing antibodies to NRG1-α and NRG1-β and recombinant NRG1-α and NRG1-β (EGF domain) from R & D Systems; and horseradish peroxidase-coupled secondary antibodies from Amersham Pharmacia Biotech (mouse and rabbit) and Santa Cruz Biotechnology (goat). Affinity-purified polyclonal rabbit antibodies to the carboxyl terminus of rat AQP5 (20) and human AQP5 (21) have been described. Other reagents, including mouse and goat IgG, were from Sigma. The matrix metalloproteinase (MMP) inhibitor BB-94 was kindly provided by Robert Huber and Klaus Maskos (Max Planck Institute for Biochemistry, Martinsried, Germany).
Cell Culture and Harvest. Cells of the mouse lung epithelial cell line 12 (MLE-12) (a gift from Jeff Whitsett, University of Cincinnati) (22) were grown in RPMI medium 1640 supplemented with 3% (vol/vol) FBS, 2 mM l-glutamine, 100 units·ml-1 penicillin, and 100 μg·ml-1 streptomycin at 37°C in 5% CO2. Calu-3 cells (a gift from William Guggino, The Johns Hopkins University), a human lung epithelial cell line, were grown in DMEM supplemented with 10% (vol/vol) FBS, 4 mM l-glutamine, 100 units·ml-1 penicillin, and 100 units·ml-1 streptomycin (Life Technologies, Rockville, MD) at 37°C in 5% CO2. Human epidermal keratinocytes (Cascade Biologics, Portland, OR) were grown as recommended by the vendor. Cells were grown in uncoated plates (Falcon). MLE-12 and Calu-3 cells were serumstarved for 15 h before each experiment. Keratinocytes were grown in a proprietary, growth-factor-enriched medium (Cascade Biologics) and were not starved before study. In preliminary studies, hypertonic activation of ERK and induction of AQP5 in a dose-responsive fashion were observed after the addition of as little as 50 mOsm sorbitol (well within the range observed for changes in airway surface layer osmolality) and were maximal when medium was supplemented with 200 mOsm sorbitol. The studies reported here were performed in normal medium made hypertonic with 200 mOsm sorbitol. Pharmacological inhibitors were added to isotonic medium at the specified concentration for 15–30 min before hypertonic exposure and remained in the medium for the duration of the experiment.
Preparation of Cell Extracts and Immunoblotting. After incubation, cells were washed with ice-cold PBS, then scraped and pelleted (10,000 × g, 5 min, 4°C). Samples for immunoblots of AQP5, HER family, and NRG were prepared as described, with freeze–thawing and pipetting to produce lysis (16). Immunoblots for analysis of total or phosphorylated ERK were performed as described after processing in a phosphoprotective lysis buffer (16). Total protein concentrations were determined by a bicinchoninic acid assay on supernatant fractions with BSA standards. Depending on the protein to be assayed, 10–100 μg of total protein per lane in 1.5% (wt/vol) SDS was subjected to SDS/PAGE (23). Duplicate gels were stained with Coomassie brilliant blue to confirm equivalent loading. Immunoblots (24) were visualized with enhanced chemiluminescence and autoradiography.
Immunoprecipitation. Our immunoprecipitation protocol has been described (25). Phosphotyrosine immunoblots were blocked with gelatin, and primary and secondary antibodies were diluted in NET buffer [1.5 M NaCl/0.05 M EDTA, pH 8.0/0.5 M Tris, pH 7.5/0.5% (vol/vol) Triton X-100/0.25% gelatin]. HER4 immunoprecipitation samples were lysed in modified phosphoprotective buffer [50 mM β-glycerophosphate, pH 7.2/0.5% (vol/vol) Triton X-100/0.2 mM sodium orthovanadate/4 mM MgCl2/2 mM EGTA/4 μg·ml-1 leupeptin/8 μg·ml-1 aprotinin], and immunoprecipitated samples were washed four times in this buffer and once in wash buffer [20 mM Tris, pH 8.0/1.0% (vol/vol) Triton X-100/5 mM EDTA/5 mM EGTA] (26).
Crosslinking. After stimulation, monolayers were washed twice with ice-cold PBS before the addition of 2 mM DTSSP in PBS/25 mM Hepes for 60 min at room temperature. Reaction was quenched by the addition of 20 mM Tris (pH 7.5) for 15 min on ice. Monolayers were then washed twice with ice-cold PBS and lysed (25) for immunoprecipitation as described (26).
Neutralization Assay. MLE-12 cells grown in 24-well plates were serum-starved ≈15 h. For neutralization of exogenous NRG1, neutralizing antibody (100 μg·ml-1) was preincubated in isotonic, serum-free medium with 10 ng·ml-1 NRG1-α or NRG1-β at 37°C for 60 min with intermittent rocking. The medium was decanted and replaced by 50 μl of preincubated NRG1-antibody mix and incubated at 37°C for 5 min. For neutralization of endogenous NRG1, antibodies (100 μg·ml-1; final volume 50 μl) were preincubated (37°C, 60 min) directly with cells after culture medium removal and before the addition of hypertonic medium (200 mOsm sorbitol) for 5 min. Monolayers were processed for total and phospho-ERK immunoblots as described (16).
Results
Hypertonic Activation of ERK Requires HER Tyrosine Kinase Activity. We described activation of ERK and induction of AQP5 by hypertonicity in MLE-15 cells (16). Like MLE-15 cells, MLE-12 cells are lung epithelial cells that spontaneously express AQP5 on the apical membrane similarly to lung epithelium in vivo, with lower baseline tyrosine phosphorylation than MLE-15 cells (data not shown). MLE-12 cells responded strongly to hypertonic shock (+ 200 mOsm sorbitol) with ERK activation (Fig. 1) but not to jun-N-terminal kinase (JNK) or p38 activation; NaCl and raffinose produced similar results (data not shown).
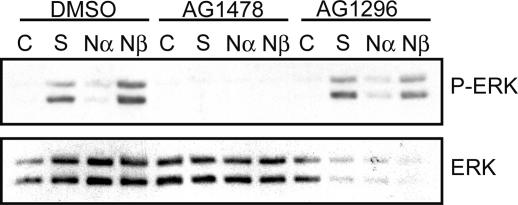
Fig. 1.
EGF receptor inhibition blocks hypertonic activation of ERK in lung epithelial cells. MLE-12 cells were incubated in isotonic (-) or hypertonic (+ 200 mOsm sorbitol) medium for 15 min in the presence of the vehicle control (DMSO) or EGF inhibitor AG1478 (1 μM) or platelet-derived growth factor (PDGF) RTK inhibitor AG1296 (10 μM). Cell lysates were probed for total ERK or phosphorylated (activated) ERK (P-ERK). Inhibition of HER activation blocked hypertonic activation of ERK.
In mammalian cells, ERK activation frequently involves prior RTK phosphorylation. Hypertonic shock of MLE-12 cells induced tyrosine phosphorylation of proteins of the approximate size of HER and PDGF receptor isoforms (data not shown). Incubation of MLE-12 cells with established HER tyrosine kinase inhibitor AG1478 (27) completely blocked ERK activation by hypertonic stress (Fig. 1), indicating that HER receptors may participate in the hypertonic response in MLE-12 cells. PDGF receptor inhibitor AG1296 (28) had no effect.
HER2·HER3 Heterodimers Mediate ERK Activation by Hypertonic Shock. The HER family consists of four members (HER1–4), which function as dimers. HERs are activated by selective transmembrane prohormones that require regulated cleavage to signal (29, 30). HER1 and HER4 can act as homo- or heterodimers. HER2 is devoid of a ligand-binding domain, and HER3 carries an impaired intracellular kinase domain. Consequently, these receptor isoforms signal only as heterodimers. Protein immunoblots of MLE-12 cell lysates revealed strong expression of HER2 and HER3, barely detectable HER1 and HER4, and no evidence of PDGF receptor isoforms (data not shown). The addition of the natural HER1 ligand EGF (up to 100 ng/ml) to MLE-12 cells failed to activate ERK but produced robust ERK activation in BALB/c fibroblasts (data not shown). This finding, coupled with the low abundance of HER1, indicated that HER1 was unlikely to contribute to signaling in MLE-12 cells.
Within the HER family, HER2·HER3 appears to be the preferred heterodimer combination and forms a potent signaling complex (31). Receptor activation is followed by the recruitment of adaptor molecules to the cytoplasmic tails of the receptors and downstream propagation to signaling cascades, e.g., the Ras–ERK–MAP kinase and PI3-kinase pathways (31). NRG1, with differentially spliced isoforms NRG1-α and NRG1-β, is a natural ligand for HER3 (32) present in MLE-12 cells (see below). The addition of exogenous mature NRG1-β to MLE-12 cells activated ERK much more potently than did the -α isoform and was blocked by AG1478 (Fig. 2). After immunoprecipitation of HER2 or HER3 from cell lysates after NRG1-β stimulation, immunoblots revealed that within 1 min, tyrosine phosphorylation of HER2 increased, as did binding of adaptor molecule Shc to HER2 and binding of both Shc and p85α (PI3-kinase subunit) to HER3 (Fig. 3A). The recruitment of adapter molecules for the Ras-ERK-MAP kinase pathway to both receptors correlated with NRG1-β-induced ERK activation (Fig. 3A) and could be tested in response to hypertonic stress. Similar to the response to exogenous NRG1-β, the addition of sorbitol to MLE-12 cells induced tyrosine phosphorylation of HER2, adaptor molecule binding to both HER2 and HER3, and ERK activation (Fig. 3B). Tyrosine phosphorylation of HER2 and HER3 in response to hypertonic shock was abolished by AG1478 (Fig. 4), consistent with its effect on ERK activation (Fig. 1).
Fig. 2.
NRG1-β activates ERK in MLE-12 cells. MLE-12 cells were incubated with isotonic medium (control, C), or hypertonic medium [+ 200 mOsm sorbitol (S)], NRG1-α (Nα; 10 ng/ml), or NRG1-β (Nβ; 10 ng/ml) for 10 min in the presence of vehicle (1% DMSO), the HER tyrosine kinase inhibitor AG1478 (10 μM), or the PDGF RTK inhibitor AG1296 (20 μM). Cell lysates were probed for P-ERK or total ERK. Sorbitol and NRG1-β produced strong ERK activation; both were inhibited by AG1478.
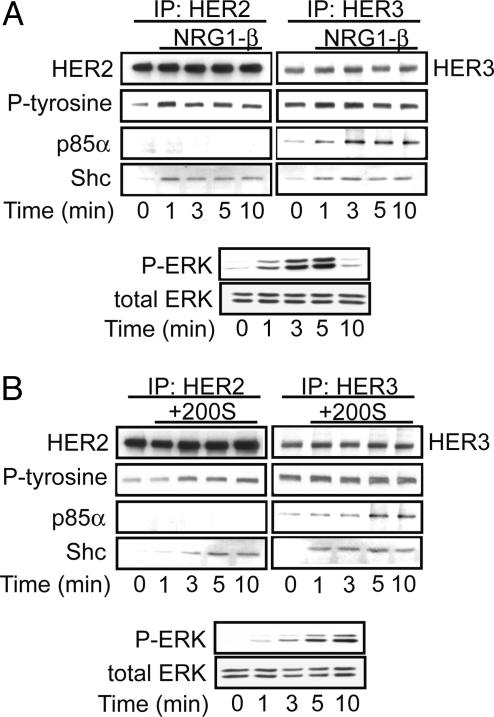
Fig. 3.
HER2 and HER3 are activated in MLE-12 cells. (A) To examine cells for activation of HER2 and HER3, exogenous NRG1-β (10 ng/ml) was added to the medium, and cells were harvested at the designated times for immunoprecipitation (IP) of HER2 or HER3. Immunoprecipitates were probed for total HER2 or HER3, phosphotyrosine (P-tyrosine), and coimmunoprecipitation of adaptor molecules p85α (PI3-kinase subunit) and Shc. An aliquot of the cell lysate was probed for P-ERK or total ERK. Within 1 min, NRG1-β increased tyrosine phosphorylation of HER2, Shc binding to HER2, and p85α and Shc binding to HER3. Addition of NRG1-β led to ERK phosphorylation within 1 min that was maximal at 5 min. (B) To determine whether hypertonic stress activated HER2 or HER3, hypertonic medium was added and cells were harvested at the designated times for analysis as in A. Consistent with the effects of exogenous NRG1-β, hypertonic shock increased tyrosine phosphorylation of HER2, Shc binding to HER2, and p85α and Shc binding to HER3 within 1–3 min. P-ERK was detectable by 1 min and increased to 10 min.
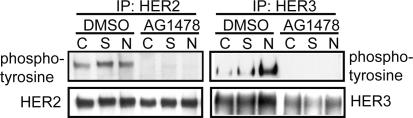
Fig. 4.
The HER RTK inhibitor AG1478 blocks activation of HER2 and HER3. MLE-12 cells were exposed to isotonic control medium (C), hypertonic medium [+ 200 mOsm sorbitol (S)], or NRG1-β (N) in the presence of vehicle control (DMSO) or AG1478 (5 μM). Immunoprecipitates (IP) of HER2 and HER3 were probed for phosphotyrosine and total HER2 or HER3. Tyrosine phosphorylation of both HER2 and HER3 increased slightly after stimulation with sorbitol or NRG1-β and was completely blocked by AG1478.
In contrast to HER2 and HER3, the addition of hypertonic medium produced only subtle changes in tyrosine phosphorylation of HER4 with no recruitment of p85α, Shc, GRB2, or GRB7 (data not shown). Whereas all four HER isoforms are detectable in MLE-12 cells, the HER2·HER3 heterodimer is the most likely to mediate hypertonic activation of ERK.
HER2·HER3 Activation by Hypertonic Shock Requires the Ligand NRG1-β. To distinguish whether hypertonic stress activated HER2·HER3 in a ligand-dependent or -independent manner, we attempted to crosslink a putative ligand to the receptor after hypertonic shock. After hypertonic stimulation of MLE-12 cells for 1 min, endogenous NRG1-β could be coimmunoprecipitated with HER3 in the presence of the reversible crosslinker DTSSP (Fig. 5). At 5 min after osmotic shock, the crosslinked complex was not detectable.
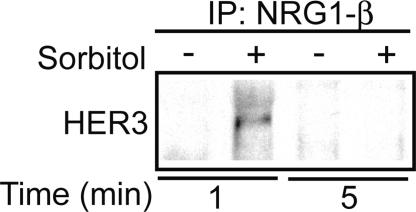
Fig. 5.
NRG1-β binds to HER3 after hypertonic shock. MLE-12 cells were incubated in isotonic (-) or hypertonic (+ 200 mOsm sorbitol) medium for the designated time, then the crosslinker DTSSP was added. Immunoprecipitates (IP) of NRG1-β were subjected to SDS/PAGE under reducing conditions, and immunoblots were probed for HER3. NRG1-β binding to HER3 could be detected at 1 min (up to 2 min in other experiments) but was not detectable at later times.
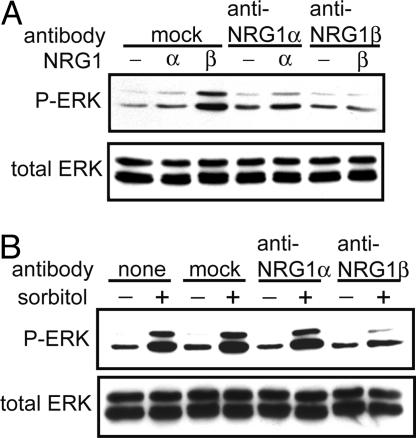
If NRG1-β were a necessary intermediate in osmotic activation of ERK, inhibitory antibodies against the EGF-signaling domain of NRG1-β should block hypertonic ERK activation. Preincubation of exogenous mature NRG1-β with neutralizing antibodies indeed blocked ERK activation by NRG1-β (Fig. 6A). More importantly, preincubation of MLE-12 cells with NRG1-β-neutralizing antibodies blocked ERK activation after hypertonic shock, whereas preincubation with nonspecific IgG or neutralizing antibodies to NRG1-α had no effect (Fig. 6B). These studies indicate that hypertonic activation of ERK requires the ligand NRG1-β.
Fig. 6.
NRG1-β participates in hypertonic activation of ERK. (A) NRG1-α or NRG1-β (10 ng/ml) was incubated with nonspecific IgG (mock) or neutralizing antibodies to NRG1-α or NRG1-β for 60 min, then added to MLE-12 cells for 5 min, at which time cells were harvested and cell lysates were probed for P-ERK or total ERK. NRG1-β-neutralizing antibodies blocked NRG1-β-induced ERK phosphorylation. (B) MLE-12 cells were preincubated for 60 min with no antibody, nonspecific IgG, or neutralizing antibodies to NRG1-α or NRG1-β (final concentration, 100 μg/ml), then hypertonic medium (+) was added for 5 min. Cells were harvested, and lysates were probed for P-ERK. Addition of NRG1-β-neutralizing antibodies blocked ERK activation by hypertonic stress.
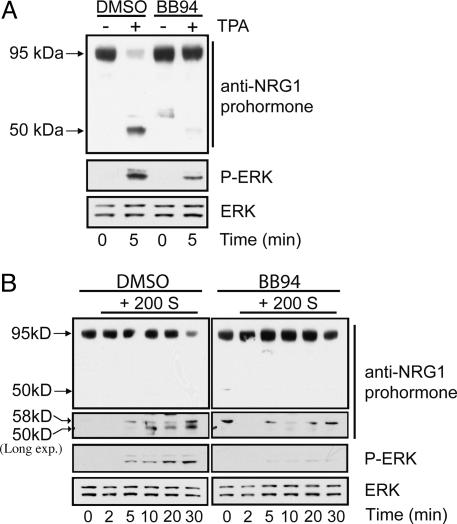
Cleavage of the NRG1 Prohormone in Response to Hypertonic Stress Involves Metalloproteinase Activity. Neuregulin is produced as a membrane-bound proform whose ectodomain acts as a hormone once cleaved above the membrane (32). Although heterogeneous molecular weights have been reported for the precursor and cleaved forms, a membrane-bound 50-kDa stem structure can be detected as a measure of induced cleavage. The phorbol 12-tetradecanoate 13-acetate diester (TPA) induces ectodomain cleavage of HER-family prohormones (33, 34). In MLE-12 cells, TPA produced a decrease in the abundance of the 95-kDa prohormone, an increase in the 50-kDa cleaved product, and ERK activation, all of which were almost completely blocked by preincubation with the MMP inhibitor BB94 (Fig. 7A). In MLE-12 cells exposed to 200 mOsm sorbitol, cleavage of the 95-kDa proform and increased abundance of the 50-kDa cleavage product were detected, however at lower efficiency than with TPA (Fig. 7B). Both hypertonic NRG1 cleavage and ERK activation were blocked by BB94. These findings indicate that MMP-dependent cleavage of NRG1-β is necessary for hypertonic activation of ERK.
Fig. 7.
Hypertonic NRG1 cleavage in MLE-12 cells requires MMP activity. (A) MLE-12 cells were treated with TPA (64 μM) for 5 min after a 30-min pretreatment with vehicle (DMSO) or the MMP inhibitor BB94 (5 μM). The 95-kDa prohormone and a 50-kDa cleavage product were identified in cell lysates after TPA exposure, along with P-ERK. Both NRG1-β cleavage and ERK activation were inhibited by BB94. (B) MLE-12 cells were incubated in hypertonic medium (+ 200 mOsm sorbitol) for the designated times, and cell lysates were probed as in A. In both studies, P-ERK and total ERK were examined (same samples, separate immunoblots). Hypertonic shock increased the 50-kDa cleavage product; by 30 min, a decrease in the prohormone was detected. An unspecific 58-kDa band is evident in both panels. The MMP inhibitor BB94 completely blocked hypertonic cleavage of the prohormone and activation of ERK.
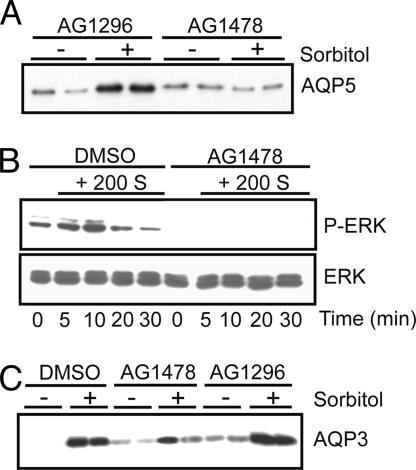
Hypertonic Induction of Aquaporins Is Dependent on HER Activation in Different Cell Lines. To examine the general importance of HER-dependent signaling for AQP induction, we examined the cellular responses to hypertonicity in three distinct cell types. In MLE-12 cells, hypertonic induction of AQP5 protein was markedly reduced in the presence of AG1478 (Fig. 8A) and completely blocked by BB94 (Fig. 8B), confirming the role of MMP-dependent HER2·HER3 activation in AQP5 induction. Of note, the addition of exogenous mature NRG1-β to MLE-12 cells did not induce AQP5 expression (data not shown) despite stimulating HER2·HER3 (Fig. 3) and ERK activation (Fig. 2), indicating that NRG1-β-mediated HER2·HER3 activation is an essential but not sufficient signal for AQP5 induction in MLE-12 cells. Calu-3 cells, a human lung epithelial cell line (35), also exhibited polarized apical expression of AQP5 and ERK activation by hypertonic stress (data not shown). Hypertonic induction of AQP5 in Calu-3 cells was completely blocked by preincubation with AG1478 but not AG1296 (Fig. 9A). Human primary epidermal keratinocytes, in which hypertonic induction of AQP3 has been reported (36), exhibited baseline ERK activation that increased with hypertonic stress and was blocked by AG1478 (Fig. 9B). Hypertonic induction of AQP3 in keratinocytes also was blocked by AG1478 but not by AG1296 (Fig. 9C). Taken together, these observations in three distinct cell types indicate that HER-mediated aquaporin induction represents a general mechanism of signaling in response to hypertonic stress in mammalian cells.
Fig. 8.
Inhibition of HER activation or NRG1-β cleavage blocks hypertonic induction of AQP5 in MLE-12 cells. (A) MLE-12 cells were incubated in isotonic (-) or hypertonic (+) medium in the presence or absence of AG1478 (5 μM). Samples were harvested after 20 h to determine AQP5 protein abundance by protein immunoblot. (B) MLE-12 cells were incubated in the presence or absence of the MMP inhibitor BB94 (5 μM), and harvested at 20 h for assay of AQP5 protein abundance by immunoblot. Hypertonic induction of AQP5 was markedly reduced by both agents.
Fig. 9.
Inhibition of HER activation blocks aquaporin induction. (A) Calu-3 human lung epithelial cells were incubated in isotonic (-) or hypertonic (+) medium for 20 h in the presence or absence of inhibitors of the PDGF receptor (AG1296, 10 μM) or the HER receptor (AG1478, 1 μM), and immunoblots of cell lysates were probed for AQP5. (B) Human epidermal keratinocytes were incubated in hypertonic medium (+ 200 mOsm sorbitol) in the presence of vehicle (DMSO) or AG1478 (1 μM) and harvested at the designated times. Immunoblots were probed for P-ERK or total ERK. (C) Human keratinocytes were incubated in isotonic (-) or hypertonic (+) medium in the presence or absence of DMSO, AG1478 (1 μM), or AG1296 (10 μM) for 20 h, and cell lysates were probed for AQP3 by immunoblot. HER inhibition blocked induction of AQP5 and AQP3, as well as ERK activation.
Discussion
Activation of MAPK-mediated signaling in response to osmotic stress is conserved among eukaryotes and is best defined for the high-osmolarity glycerol (HOG) pathway in Saccharomyces cerevisiae (19, 37). Yeast Hog1, a homologue of mammalian JNK and p38 kinases, can be activated through at least two distinct transmembrane proteins, Sln1 (38, 39), and Sho1 (38, 40). Uhlik et al. (41) recently described a scaffolding protein in mammalian cells that mediates organization and regulation of MAPK signaling during hypertonic stress similarly to yeast. However, homologous transmembrane osmosensing components have not been identified in mammalian cells, and upstream signaling events in osmotic responses remain incompletely defined. In this report, we identify MMP-dependent cleavage of a membrane-bound prohormone, NRG1-β, and ligand-dependent HER activation as necessary steps in hypertonic ERK activation and aquaporin induction.
HERs and their ligands have been implicated as central signaling stations in multiple cellular responses (30). Participation of HERs in the mammalian osmotic stress response was originally suggested by King et al. (42), who showed HER1 tyrosine phosphorylation in response to hypertonicity. In mammalian cells, UV light and osmotic stress lead to RTK activation followed by activation of the JNK signaling cascade (43). HER1 activation by vanadate or UV light is mediated by the inhibition of a negatively regulating phosphatase (2, 44). Response to osmotic stress, however, was suggested to be ligand-independent, resulting from the physical perturbation of the cellular membrane and/or a stress-induced change in RTK conformation (43).
Recent studies have suggested the involvement of a cleavable ligand in the osmotic stress response. The HER1 ligand heparin-binding EGF (HB-EGF) was found in a complex with the osmoinducible molecules CD9 and β-1 integrin in the renal medulla of thirsted rats, suggesting a potential signaling role (45). When heterologously expressed in Chinese hamster ovary cells, the HER ligands pro-TGF-α and pro-NRG1-α2c were cleaved by hypertonic stress and cleavage appeared to involve MMP activity, but no relation to target protein expression was defined (34). Hypertonic induction of cyclooxygenase-2 in the kidney cell line IMCD3 was partially reduced by HER inhibitors and MMP inhibition with N-{dk-[2-(hydroxyaminocarbonyl) methyl]-4-methyl-pentanoyl}-l-3-(2′-naphthyl)-alanyl-l-alanine 2-aminoethyl-amide (TAPI) and doxycycline, but participation of a ligand in the activation mechanism was not demonstrated (46). Very recently, Fischer et al. (47) identified HB-EGF as a mediator of osmotic and oxidative stress-induced activation of MAPKs in tumor cells and suggested an antiapoptotic role for HB-EGF signaling in doxorubicin-induced apoptosis.
NRG1 isoforms and their HER-family receptors are involved in key developmental interactions (32), and deletion of NRG1 leads to embryonic lethal defects in cardiac and neuronal development (48, 49). Studies in cultured cells have implicated neuregulin-mediated signaling in myogenic differentiation (50), glucose transport (51), and fetal lung epithelial development (52). In human tracheal epithelium, NRG1-α participates in the regulation of epithelial integrity: apical NRG1-α gains access to its basolateral receptors during epithelial wounding to stimulate repair (53). Participation of NRG1 in osmotic signaling has not been described, and it is not known whether cell size and shape changes provoked by osmotic stress might similarly facilitate interaction between otherwise segregated ligands and receptors. We detected only transient binding of the native ligand NRG1-β to HER3 after hypertonic shock, consistent with rapid internalization and degradation of the complex after ligand binding (54). Our observation of MMP-mediated cleavage of NRG1 is consistent with protease-dependent NRG1-β cleavage described by others (55). To date, we have not identified the specific protease mediating NRG1 cleavage in response to hypertonic stress.
We propose that HER-mediated aquaporin expression in response to osmotic stress is highly relevant to lung physiology. AQP5 plays an important role in the generation of the airway surface liquid, as evidenced by reduced surface layer secretions in AQP5-null mice (56). Tight regulation of the constitution and volume of the airway surface layer is necessary for the maintenance of normal lung defense. The efficiency of mucociliary transport and the removal of inhaled particulate matter trapped in the airway secretions depends on the water content and fluidity of the surface layer. In addition, the activity of defensins, bactericidal proteins secreted onto the airway surface, depends on the osmolality of the aqueous environment (14). Increased osmolality inhibits the function of defensins and would be predicted to alter local lung defense. In this context, HER-mediated signaling may couple epithelial secretion and water permeability with the release of secreted products (e.g., mucins or defensins) into the airways. The inhibition of HER signaling in a cultured human lung epithelial cell line blocked mucin gene expression as well as airway goblet cell hyperplasia in rats stimulated with tumor necrosis factor α or ovalbumin (57). Similarly, primary cultures of human bronchial epithelium exhibited decreased baseline goblet cell density when treated with the EGF receptor antagonist AG-1478 (58). Finally, water for the humidification of air inspired into the lung comes from the surface layer. Sustained changes in ambient humidity or respiratory pattern can alter surface layer composition (13) and may stimulate HER-mediated changes in aquaporin expression to maintain normal airway water homeostasis.
Our observations in keratinocytes reinforce the potential significance of the described link between HER activation and aquaporin expression. AQP3 is permeated by glycerol in addition to water. In keratinocytes, glycerol entering the cell through AQP3 provides the carbon backbone necessary for the generation of lipids that are released into the epidermis to limit evaporative water loss from the skin. Consistent with that role, AQP3-null mice have increased transdermal water loss, compared with wild-type mice (59). We hypothesize that HER-mediated hypertonic induction of AQP3 in keratinocytes is a fundamental mechanism for limiting water loss through the skin.
In summary, our findings, along with the observations of others, indicate that HER-dependent regulation of gene expression represents a widely used paradigm for cellular osmotic responses.
Acknowledgments
We thank Peter Herrlich and Peter Agre for helpful discussions, and Harvey Lodish, David Hellmann, and Roy Ziegelstein for generous support of A.H. We thank Robert Huber Klaus Maskos for providing BB94. This work was supported by National Heart, Lung, and Blood Institute Grant HL-70217 and a Grant-in-Aid from the American Heart Association (to L.S.K.).
Author contributions: A.H., V.L., and L.S.K. designed research; A.H., V.L., and L.S.K. performed research; A.H., V.L., and L.S.K. analyzed data; and A.H. and L.S.K. wrote the paper.
Abbreviations: AQP, aquaporin; DTSSP, 3,3′-dithiobis(sulfosuccinimidyl propionate); HER, human epidermal growth factor receptor; ERK, extracellular signal-regulated kinase; P-ERK, phosphorylated ERK; MAPK, mitogen-activated protein kinase; MLE, mouse lung epithelial cell line; MMP, matrix metalloproteinase; mOsm, milliosmolal; NRG1, neuregulin 1; PDGF, platelet-derived growth factor; PI3-kinase, phosphatidylinositol 3-kinase; RTK, receptor tyrosine kinase; PA, phorbol 12-tetradecanoate 13-acetate.
References
- 1.Dorion, S. & Landry, J. (2002) Cell Stress Chaperones 7, 200-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Knebel, A., Rahmsdorf, H. J., Ullrich, A. & Herrlich, P. (1996) EMBO J. 15, 5314-5325. [PMC free article] [PubMed] [Google Scholar]
- 3.Karin, M., Takahashi, T., Kapahi, P., Delhase, M., Chen, Y., Makris, C., Rothwarf, D., Baud, V., Natoli, G., Guido, F. & Li, N. F. (2001) Biofactors 15, 87-89. [DOI] [PubMed] [Google Scholar]
- 4.Burg, M. B., Kwon, E. D. & Kultz, D. (1996) FASEB J. 10, 1598-1606. [DOI] [PubMed] [Google Scholar]
- 5.McManus, M. L., Churchwell, K. B. & Strange, K. (1995) N. Engl. J. Med. 333, 1260-1266. [DOI] [PubMed] [Google Scholar]
- 6.Handler, J. S. & Kwon, H. M. (1993) Am. J. Physiol. 265, C1449-C1455. [DOI] [PubMed] [Google Scholar]
- 7.Garcia-Perez, A. & Burg, M. B. (1991) Physiol. Rev. 71, 1081-1115. [DOI] [PubMed] [Google Scholar]
- 8.Woo, S. K. & Kwon, H. M. (2002) Int. Rev. Cytol. 215, 189-202. [DOI] [PubMed] [Google Scholar]
- 9.Man, S. F., Adams, G. K., III, & Proctor, D. F. (1979) J. Appl. Physiol. 46, 205-210. [DOI] [PubMed] [Google Scholar]
- 10.Boucher, R. C., Stutts, M. J., Bromberg, P. A. & Gatzy, J. T. (1981) J. Appl. Physiol. 50, 613-620. [DOI] [PubMed] [Google Scholar]
- 11.Winters, S. L. & Yeates, D. B. (1997) J. Appl. Physiol. 83, 1348-1359. [DOI] [PubMed] [Google Scholar]
- 12.Freed, A. N., Yiin, K. T. & Stream, C. E. (1989) J. Appl. Physiol. 67, 2571-2578. [DOI] [PubMed] [Google Scholar]
- 13.Freed, A. N. & Davis, M. S. (1999) Am. J. Respir. Crit. Care Med. 159, 1101-1107. [DOI] [PubMed] [Google Scholar]
- 14.Smith, J. J., Travis, S. M., Greenberg, E. P. & Welsh, M. J. (1996) Cell 85, 229-236. [DOI] [PubMed] [Google Scholar]
- 15.Jenq, W., Cooper, D. R., Bittle, P. & Ramirez, G. (1999) Biochem. Biophys. Res. Commun. 256, 240-248. [DOI] [PubMed] [Google Scholar]
- 16.Hoffert, J. D., Leitch, V., Agre, P. & King, L. S. (2000) J. Biol. Chem. 275, 9070-9077. [DOI] [PubMed] [Google Scholar]
- 17.Umenishi, F. & Schrier, R. W. (2003) J. Biol. Chem. 278, 15765-15770. [DOI] [PubMed] [Google Scholar]
- 18.King, L. S., Kozono, D. & Agre, P. (2004) Nat. Rev. Mol. Cell Biol. 5, 687-698. [DOI] [PubMed] [Google Scholar]
- 19.de Nadal, E., Alepuz, P. M. & Posas, F. (2002) EMBO Rep. 3, 735-740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.King, L. S., Nielsen, S. & Agre, P. (1997) Am. J. Physiol. 273, C1541-C1548. [DOI] [PubMed] [Google Scholar]
- 21.Steinfeld, S., Cogan, E., King, L. S., Agre, P., Kiss, R. & Delporte, C. (2001) Lab. Invest. 81, 143-148. [DOI] [PubMed] [Google Scholar]
- 22.Wikenheiser, K. A., Vorbroker, D. K., Rice, W. R., Clark, J. C., Bachurski, C. J., Oie, H. K. & Whitsett, J. A. (1993) Proc. Natl. Acad. Sci. USA 90, 11029-11033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Laemmli, U. K. (1970) Nature 227, 680-685. [DOI] [PubMed] [Google Scholar]
- 24.Davis, J. Q. & Bennett, V. (1984) J. Biol. Chem. 259, 1874-1881. [PubMed] [Google Scholar]
- 25.Herrlich, A., Daub, H., Knebel, A., Herrlich, P., Ullrich, A., Schultz, G. & Gudermann, T. (1998) Proc. Natl. Acad. Sci. USA 95, 8985-8990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leitch, V., Agre, P. & King, L. S. (2001) Proc. Natl. Acad. Sci. USA 98, 2894-2898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Egeblad, M., Mortensen, O. H., van Kempen, L. C. & Jaattela, M. (2001) Biochem. Biophys. Res. Commun. 281, 25-31. [DOI] [PubMed] [Google Scholar]
- 28.Kovalenko, M., Gazit, A., Bohmer, A., Rorsman, C., Ronnstrand, L., Heldin, C. H., Waltenberger, J., Bohmer, F. D. & Levitzki, A. (1994) Cancer Res. 54, 6106-6114. [PubMed] [Google Scholar]
- 29.Olayioye, M. A., Neve, R. M., Lane, H. A. & Hynes, N. E. (2000) EMBO J. 19, 3159-3167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Prenzel, N., Fischer, O. M., Streit, S., Hart, S. & Ullrich, A. (2001) Endocr. Relat. Cancer 8, 11-31. [DOI] [PubMed] [Google Scholar]
- 31.Hynes, N. E., Horsch, K., Olayioye, M. A. & Badache, A. (2001) Endocr. Relat. Cancer 8, 151-159. [DOI] [PubMed] [Google Scholar]
- 32.Falls, D. L. (2003) Exp. Cell Res. 284, 14-30. [DOI] [PubMed] [Google Scholar]
- 33.Izumi, Y., Hirata, M., Hasuwa, H., Iwamoto, R., Umata, T., Miyado, K., Tamai, Y., Kurisaki, T., Sehara-Fujisawa, A., Ohno, S. & Mekada, E.S. (1998) EMBO J. 17, 7260-7272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Montero, J. C., Yuste, L., Diaz-Rodriguez, E., Esparis-Ogando, A. & Pandiella, A. (2002) Biochem. J. 363, 211-221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Trout, L., Gatzy, J. T. & Ballard, S. T. (1998) Am. J. Physiol. 275, L1095-L1099. [DOI] [PubMed] [Google Scholar]
- 36.Sugiyama, Y., Ota, Y., Hara, M. & Inoue, S. (2001) Biochim. Biophys. Acta 1522, 82-88. [DOI] [PubMed] [Google Scholar]
- 37.Galcheva-Gargova, Z., Derijard, B., Wu, I. H. & Davis, R. J. (1994) Science 265, 806-808. [DOI] [PubMed] [Google Scholar]
- 38.Posas, F. & Saito, H. (1998) EMBO J. 17, 1385-1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Reiser, V., Raitt, D. C. & Saito, H. (2003) J. Cell Biol. 161, 1035-1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hohmann, S. (2002) Microbiol. Mol. Biol. Rev. 66, 300-372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Uhlik, M. T., Abell, A. N., Johnson, N. L., Sun, W., Cuevas, B. D., Lobel-Rice, K. E., Horne, E. A., Dell'Acqua, M. L. & Johnson, G. L. (2003) Nat. Cell Biol. 5, 1104-1110. [DOI] [PubMed] [Google Scholar]
- 42.King, C. R., Borrello, I., Porter, L., Comoglio, P. & Schlessinger, J. (1989) Oncogene 4, 13-18. [PubMed] [Google Scholar]
- 43.Rosette, C. & Karin, M. (1996) Science 274, 1194-1197. [DOI] [PubMed] [Google Scholar]
- 44.Sachsenmaier, C., Radler-Pohl, A., Zinck, R., Nordheim, A., Herrlich, P. & Rahmsdorf, H. J. (1994) Cell 78, 963-972. [DOI] [PubMed] [Google Scholar]
- 45.Sheikh-Hamad, D., Suki, W. N. & Zhao, W. (1997) Am. J. Physiol. 273, C902-C908. [DOI] [PubMed] [Google Scholar]
- 46.Zhao, H., Tian, W., Tai, C. & Cohen, D. M. (2003) Am. J. Physiol. 285, F281-F288. [DOI] [PubMed] [Google Scholar]
- 47.Fischer, O. M., Hart, S., Gschwind, A., Prenzel, N. & Ullrich, A. (2004) Mol. Cell. Biol. 24, 5172-5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kramer, R., Bucay, N., Kane, D. J., Martin, L. E., Tarpley, J. E. & Theill, L. E. (1996) Proc. Natl. Acad. Sci. USA 93, 4833-4838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Britsch, S., Li, L., Kirchhoff, S., Theuring, F., Brinkmann, V., Birchmeier, C. & Riethmacher, D. (1998) Genes Dev. 12, 1825-1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kim, D., Chi, S., Lee, K. H., Rhee, S., Kwon, Y. K., Chung, C. H., Kwon, H. & Kang, M. S. (1999) J. Biol. Chem. 274, 15395-15400. [DOI] [PubMed] [Google Scholar]
- 51.Suarez, E., Bach, D., Cadefau, J., Palacin, M., Zorzano, A. & Guma, A. (2001) J. Biol. Chem. 276, 18257-18264. [DOI] [PubMed] [Google Scholar]
- 52.Patel, N. V., Acarregui, M. J., Snyder, J. M., Klein, J. M., Sliwkowski, M. X. & Kern, J. A. (2000) Am. J. Respir. Cell Mol. Biol. 22, 432-440. [DOI] [PubMed] [Google Scholar]
- 53.Vermeer, P. D., Einwalter, L. A., Moninger, T. O., Rokhlina, T., Kern, J. A., Zabner, J. & Welsh, M. J. (2003) Nature 422, 322-326. [DOI] [PubMed] [Google Scholar]
- 54.Wiley, H. S. (2003) Exp. Cell Res. 284, 78-88. [DOI] [PubMed] [Google Scholar]
- 55.Shirakabe, K., Wakatsuki, S., Kurisaki, T. & Fujisawa-Sehara, A. (2001) J. Biol. Chem. 276, 9352-9358. [DOI] [PubMed] [Google Scholar]
- 56.Song, Y. & Verkman, A. S. (2001) J. Biol. Chem. 276, 41288-41292. [DOI] [PubMed] [Google Scholar]
- 57.Takeyama, K., Dabbagh, K., Lee, H. M., Agusti, C., Lausier, J. A., Ueki, I. F., Grattan, K. M. & Nadel, J. A. (1999) Proc. Natl. Acad. Sci. USA 96, 3081-3086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Atherton, H. C., Jones, G. & Danahay, H. (2003) Am. J. Physiol. 285, L730-L739. [DOI] [PubMed] [Google Scholar]
- 59.Ma, T., Hara, M., Sougrat, R., Verbavatz, J. M. & Verkman, A. S. (2002) J. Biol. Chem. 277, 17147-17153. [DOI] [PubMed] [Google Scholar]