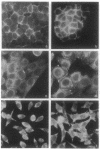
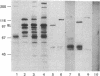
Abstract
The cytoplasmic region of the Ca(2+)-dependent cell-adhesion molecule (CAM) uvomorulin associates with distinct cytoplasmic proteins with molecular masses of 102, 88, and 80 kDa termed alpha, beta, and gamma catenin, respectively. This complex formation links uvomorulin to the actin filament network, which seems to be of primary importance for its cell-adhesion properties. We show here that antibodies against alpha catenin also immunoprecipitate complexes that contain human N-cadherin, mouse P-cadherin, chicken A-CAM (adherens junction-specific CAM; also called N-cadherin) or Xenopus U-cadherin, demonstrating that alpha catenin is complexed with other cadherins. In immunofluorescence tests, alpha catenin is colocalized with cadherins at the plasma membrane. However, in cadherin-negative Ltk- cells, alpha catenin is found uniformly distributed in the cytoplasm, suggesting some additional biological function(s). Expression of uvomorulin in these cells results in a concentration of alpha catenin at membrane areas of cell contacts. We also have cloned and sequenced murine alpha catenin. The deduced amino acid sequence reveals a significant homology to vinculin. Our results suggest the possibility of a new vinculin-related protein family involved in the cytoplasmic anchorage of cell-cell and cell-substrate adhesion molecules.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Angres B., Müller A. H., Kellermann J., Hausen P. Differential expression of two cadherins in Xenopus laevis. Development. 1991 Mar;111(3):829–844. doi: 10.1242/dev.111.3.829. [DOI] [PubMed] [Google Scholar]
- Barstead R. J., Waterston R. H. The basal component of the nematode dense-body is vinculin. J Biol Chem. 1989 Jun 15;264(17):10177–10185. [PubMed] [Google Scholar]
- Belkin A. M., Koteliansky V. E. Interaction of iodinated vinculin, metavinculin and alpha-actinin with cytoskeletal proteins. FEBS Lett. 1987 Aug 17;220(2):291–294. doi: 10.1016/0014-5793(87)80832-3. [DOI] [PubMed] [Google Scholar]
- Bendori R., Salomon D., Geiger B. Identification of two distinct functional domains on vinculin involved in its association with focal contacts. J Cell Biol. 1989 Jun;108(6):2383–2393. doi: 10.1083/jcb.108.6.2383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burridge K., Fath K., Kelly T., Nuckolls G., Turner C. Focal adhesions: transmembrane junctions between the extracellular matrix and the cytoskeleton. Annu Rev Cell Biol. 1988;4:487–525. doi: 10.1146/annurev.cb.04.110188.002415. [DOI] [PubMed] [Google Scholar]
- Coutu M. D., Craig S. W. cDNA-derived sequence of chicken embryo vinculin. Proc Natl Acad Sci U S A. 1988 Nov;85(22):8535–8539. doi: 10.1073/pnas.85.22.8535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eckerskorn C., Mewes W., Goretzki H., Lottspeich F. A new siliconized-glass fiber as support for protein-chemical analysis of electroblotted proteins. Eur J Biochem. 1988 Oct 1;176(3):509–519. doi: 10.1111/j.1432-1033.1988.tb14308.x. [DOI] [PubMed] [Google Scholar]
- Geiger B. A 130K protein from chicken gizzard: its localization at the termini of microfilament bundles in cultured chicken cells. Cell. 1979 Sep;18(1):193–205. doi: 10.1016/0092-8674(79)90368-4. [DOI] [PubMed] [Google Scholar]
- Ginsberg D., DeSimone D., Geiger B. Expression of a novel cadherin (EP-cadherin) in unfertilized eggs and early Xenopus embryos. Development. 1991 Feb;111(2):315–325. doi: 10.1242/dev.111.2.315. [DOI] [PubMed] [Google Scholar]
- Herzberg F., Wildermuth V., Wedlich D. Expression of XBcad, a novel cadherin, during oogenesis and early development of Xenopus. Mech Dev. 1991 Aug;35(1):33–42. doi: 10.1016/0925-4773(91)90039-9. [DOI] [PubMed] [Google Scholar]
- Jones P., Jackson P., Price G. J., Patel B., Ohanion V., Lear A. L., Critchley D. R. Identification of a talin binding site in the cytoskeletal protein vinculin. J Cell Biol. 1989 Dec;109(6 Pt 1):2917–2927. doi: 10.1083/jcb.109.6.2917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemler R., Ozawa M., Ringwald M. Calcium-dependent cell adhesion molecules. Curr Opin Cell Biol. 1989 Oct;1(5):892–897. doi: 10.1016/0955-0674(89)90055-0. [DOI] [PubMed] [Google Scholar]
- Koch P. J., Walsh M. J., Schmelz M., Goldschmidt M. D., Zimbelmann R., Franke W. W. Identification of desmoglein, a constitutive desmosomal glycoprotein, as a member of the cadherin family of cell adhesion molecules. Eur J Cell Biol. 1990 Oct;53(1):1–12. [PubMed] [Google Scholar]
- McNeill H., Ozawa M., Kemler R., Nelson W. J. Novel function of the cell adhesion molecule uvomorulin as an inducer of cell surface polarity. Cell. 1990 Jul 27;62(2):309–316. doi: 10.1016/0092-8674(90)90368-o. [DOI] [PubMed] [Google Scholar]
- Mechanic S., Raynor K., Hill J. E., Cowin P. Desmocollins form a distinct subset of the cadherin family of cell adhesion molecules. Proc Natl Acad Sci U S A. 1991 May 15;88(10):4476–4480. doi: 10.1073/pnas.88.10.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagafuchi A., Takeichi M. Transmembrane control of cadherin-mediated cell adhesion: a 94 kDa protein functionally associated with a specific region of the cytoplasmic domain of E-cadherin. Cell Regul. 1989 Nov;1(1):37–44. doi: 10.1091/mbc.1.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagafuchi A., Takeichi M., Tsukita S. The 102 kd cadherin-associated protein: similarity to vinculin and posttranscriptional regulation of expression. Cell. 1991 May 31;65(5):849–857. doi: 10.1016/0092-8674(91)90392-c. [DOI] [PubMed] [Google Scholar]
- Otto J. J. Detection of vinculin-binding proteins with an 125I-vinculin gel overlay technique. J Cell Biol. 1983 Oct;97(4):1283–1287. doi: 10.1083/jcb.97.4.1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozawa M., Baribault H., Kemler R. The cytoplasmic domain of the cell adhesion molecule uvomorulin associates with three independent proteins structurally related in different species. EMBO J. 1989 Jun;8(6):1711–1717. doi: 10.1002/j.1460-2075.1989.tb03563.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozawa M., Ringwald M., Kemler R. Uvomorulin-catenin complex formation is regulated by a specific domain in the cytoplasmic region of the cell adhesion molecule. Proc Natl Acad Sci U S A. 1990 Jun;87(11):4246–4250. doi: 10.1073/pnas.87.11.4246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price G. J., Jones P., Davison M. D., Patel B., Bendori R., Geiger B., Critchley D. R. Primary sequence and domain structure of chicken vinculin. Biochem J. 1989 Apr 15;259(2):453–461. doi: 10.1042/bj2590453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringwald M., Schuh R., Vestweber D., Eistetter H., Lottspeich F., Engel J., Dölz R., Jähnig F., Epplen J., Mayer S. The structure of cell adhesion molecule uvomorulin. Insights into the molecular mechanism of Ca2+-dependent cell adhesion. EMBO J. 1987 Dec 1;6(12):3647–3653. doi: 10.1002/j.1460-2075.1987.tb02697.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeichi M. The cadherins: cell-cell adhesion molecules controlling animal morphogenesis. Development. 1988 Apr;102(4):639–655. doi: 10.1242/dev.102.4.639. [DOI] [PubMed] [Google Scholar]
- Volk T., Geiger B. A 135-kd membrane protein of intercellular adherens junctions. EMBO J. 1984 Oct;3(10):2249–2260. doi: 10.1002/j.1460-2075.1984.tb02123.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wachsstock D. H., Wilkins J. A., Lin S. Specific interaction of vinculin with alpha-actinin. Biochem Biophys Res Commun. 1987 Jul 31;146(2):554–560. doi: 10.1016/0006-291x(87)90564-x. [DOI] [PubMed] [Google Scholar]
- Weller P. A., Ogryzko E. P., Corben E. B., Zhidkova N. I., Patel B., Price G. J., Spurr N. K., Koteliansky V. E., Critchley D. R. Complete sequence of human vinculin and assignment of the gene to chromosome 10. Proc Natl Acad Sci U S A. 1990 Aug;87(15):5667–5671. doi: 10.1073/pnas.87.15.5667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westmeyer A., Ruhnau K., Wegner A., Jockusch B. M. Antibody mapping of functional domains in vinculin. EMBO J. 1990 Jul;9(7):2071–2078. doi: 10.1002/j.1460-2075.1990.tb07374.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheeler G. N., Parker A. E., Thomas C. L., Ataliotis P., Poynter D., Arnemann J., Rutman A. J., Pidsley S. C., Watt F. M., Rees D. A. Desmosomal glycoprotein DGI, a component of intercellular desmosome junctions, is related to the cadherin family of cell adhesion molecules. Proc Natl Acad Sci U S A. 1991 Jun 1;88(11):4796–4800. doi: 10.1073/pnas.88.11.4796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wheelock M. J., Knudsen K. A. N-cadherin-associated proteins in chicken muscle. Differentiation. 1991 Feb;46(1):35–42. doi: 10.1111/j.1432-0436.1991.tb00863.x. [DOI] [PubMed] [Google Scholar]