Abstract
Background
Despite evidence for the effects of metals on neurodevelopment, the long-term effects on mental health remain unclear due to methodological limitations. Our objective was to determine the feasibility of studying metal exposure during critical neurodevelopmental periods and to explore the association between early-life metal exposure and adult schizophrenia.
Methods
We analyzed childhood-shed teeth from nine individuals with schizophrenia and five healthy controls. We investigated the association between exposure to lead (Pb2+), manganese (Mn2+), cadmium (Cd2+), copper (Cu2+), magnesium (Mg2+), and zinc (Zn2+), and schizophrenia, psychotic experiences, and intelligence quotient (IQ). We reconstructed the dose and timing of early-life metal exposures using laser ablation inductively coupled plasma mass spectrometry.
Results
We found higher early-life Pb2+ exposure among patients with schizophrenia than controls. The differences in log Mn2+ and log Cu2+ changed relatively linearly over time to postnatal negative values. There was a positive correlation between early-life Pb2+ levels and psychotic experiences in adulthood. Moreover, we found a negative correlation between Pb2+ levels and adult IQ.
Conclusions
In our proof-of-concept study, using tooth-matrix biomarker that provides direct measurement of exposure in the fetus and newborn, we provide support for the role of metal exposure during critical neurodevelopmental periods in psychosis.
Keywords: Environmental exposure, Lead, Neurodevelopmental disorders, Psychosis, Tooth, Trace elements
1. Introduction
According to the neurodevelopmental model of schizophrenia, psychotic symptoms emerge as a result of interactions between brain abnormalities established in early development and brain maturational events that occur much later [1]. Metals have well-known effects on neurodevelopment in children, some acting as essential nutritive elements and others as neurotoxicants [2–8]. For example, zinc (Zn2+) is essential in development of the nervous system, and is involved in neuronal proliferation and migration as well as modulation of synaptic activities and intracellular signaling pathways [9]. Lead (Pb2+) interferes with intraneuronal gene transcription, affects hippocampal neurogenesis, and causes glial dysfunction in developing brain [10]. Furthermore, many essential elements, such as copper (Cu2+), manganese (Mn2+), and Zn2+, might exert toxic effects on brain at higher doses [11–14]. The link between exposure to different metals and adverse early neurodevelopmental outcomes is well known [2,4,6–8,15–22]. However, the effect of metals on later developmental outcomes, such as schizophrenia, is still debated.
Beyond the general effect of metals on neurodevelopment, there are multiple intriguing links between several metals and schizophrenia (Table 1). Lead (Pb2+) and manganese (Mn2+) have been shown to cause alterations in neurotransmitters in the same manner that is often observed in patients with schizophrenia [23–25]. For example, strong evidence links developmental exposure to Pb2+ with disrupted N-methyl-d-aspartate (NMDA) receptors function [25], which can lead to NMDA receptor hypofunction with subsequent dysregulation of brain-derived neurotrophic factor (BDNF) signaling, synaptic function, and long-term potentiation [23,25]. Furthermore, NMDA receptors, partly regulated by the tryptophan–kynurenine pathway, are essential during in utero brain development and influence synaptic formation and plasticity, cell proliferation, and cell migration during prenatal period [26–28].
Table 1.
Clinical and neurobiological similarities of schizophrenia and altered balance of metals.
| Schizophrenia | Pb2+ (excess) | Cd2+ (excess) | Mn2+ (excess) | Cu2+ (excess) | Mg2+ (deficiency) | Zn2+ (deficiency) |
|---|---|---|---|---|---|---|
| Core neuropsychological dysfunctions (IQ, working memory, attention, processing speed) | ** | ** | * | ** | * | * |
| References | [15,16] | [6–8] | [8,56] | [29,57,58] | [59–63] | [18,19] |
| Core neurotransmitter disturbances | ||||||
| • Dopamine | + | + | + | + | + | + |
| • Serotonin | + | + | + | + | + | + |
| • Glutamate | + | + | + | + | + | + |
| References | [64–66] | [67–69] | [14,24] | [70] | [71–73] | [74–76] |
| Clinical evidence of association with psychosis | + | + | + | + | + | + |
| References | [35,36] | [30,77] | [44,45] | [12,30] | [78,79] | [30] |
Evidence of association;
moderate association;
strong association.
Of note, individuals exposed to heavy metals or deficient in nutrient metals frequently experience neuropsychological deficits also observed in schizophrenia [5–7,18,19,29]. Wilson's disease, caused by a hereditary excess in Cu2+, frequently manifests with psychosis [11]. Multiple reports have found different concentration of various metals, such as Cu2+, Mn2+, Zn2+, and cadmium (Cd2+) between patients with schizophrenia and healthy controls [30–34]. Intriguingly, higher delta-aminolevulinic acid levels (marker of Pb2+ exposure) have been detected in mothers whose children later develop schizophrenia [35,36].
The debate on the potential role of metals in schizophrenia is further complicated by methodological limitations of available studies. Importantly, metal exposure is mostly measured after disease development and is seldom determined during critical developmental periods. Studies that did measure metal exposure during critical developmental periods usually used indirect measures, such as maternal blood samples [35,36]. Moreover, most studies relied on samples that do not reflect exact timing of exposure during the high-risk developmental period, and fall short of tracking change in exposure over time. To our knowledge, no prior studies have robustly studied the link between fetal or early childhood metal exposure and risk of psychotic illness in adulthood. Our primary objective was to determine whether the timing and dose of prenatal and early childhood metal exposure influences later development of schizophrenia and psychotic experiences.
2. Methods
In a proof-of-concept study, we analyzed shed deciduous teeth from nine individuals from the Genetic Risk and OUtcome of Psychosis (GROUP) [37] study with a DSM-IV diagnosis of schizophrenia and five healthy controls. GROUP is a prospective cohort of 1120 patients with psychotic disorders and 1648 controls and aims to investigate the genetic and environmental risk factors of psychosis in the Netherlands. A total sample size of 14 was estimated to be sufficient to detect a large effect size in the difference in mean log concentrations between cases and controls.
We investigated the association between exposure to metals, including manganese (Mn2+), lead (Pb2+), cadmium (Cd2+), copper (Cu2+), magnesium (Mg2+), and zinc (Zn2+), and intelligence quotient (IQ), syndromal schizophrenia as well psychotic experiences (as assessed by the Community Assessment of Psychic Experiences (CAPE) scale) [38]. Deciduous teeth from each subject were evaluated for pre- and postnatal metal exposure. We reconstructed the dose and timing of fetal and childhood metal exposures using a novel biomarker method named laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS), which has been described and validated in detail elsewhere [39–45]. All teeth collected in the study were shed naturally and kept by the parents at home, as is often the case in some cultures. Teeth were stored dry in sealed containers of various types. The method for tooth analysis in this study excludes the external layers to avoid any contamination. Importantly, all analytical methods used here have been extensively validated and applied to samples stored over decades and archeological samples that are thousands of years old [44,46]. Briefly, the method combines sophisticated histological and laser-based chemical analyses to precisely sample dentin layers corresponding to specific life stages, generating integrated, longitudinal, 1- to 2-week metal exposure estimates in pregnancy and during early childhood [39]. The time-varying difference between early-life (−4 to 6 months) metal concentrations, as measured in the tooth biomarker, and case/control designation was evaluated using a distributed lag model (DLM).
3. Results
Characteristics of the study participants and of the GROUP cohort are presented in Table 2. Mean [standard deviation, (SD)] age for the patients was 25.2 (1.9) years, and mean (SD) age for the control group was 28.0 ± 8.4 years. Mean (SD) duration of disorder was 3.8 (2.5) years.
Table 2.
Demographic characteristics of all participants in GROUP study and those included in the pilot study.
| Variable | All participants in GROUP study |
Participants in the pilot study |
||
|---|---|---|---|---|
| Psychosis (n = 1120) | Controls (n = 1648) | Cases (n = 9) | Controls (n = 5) | |
| Male (%) | 76 | 45.7 | 100.0 | 20.0 |
| Age, mean (SD) | 27.7 (8.1) | 28.7 (9.3) | 25.2 (1.9) | 28.0 (8.4) |
| IQ, mean (SD) | 94.9 (16.1) | 105.4 (15.6) | 100.2 (11.0) | 114.0 (19.2) |
| Illness Duration, years, mean (SD) | 4.4 (4.1) | – | 3.8 (2.5) | – |
| DSM-IV diagnosis (%) | ||||
| • Schizophrenia | 68.8 | – | 44.4 | – |
| • Schizoaffective | 10.7 | – | 44.4 | – |
| • Psychosis not otherwise specified | 10.4 | – | 11.1 | – |
| • Other | 10.1 | – | – | – |
| PANSS score, mean (SD) | ||||
| Positive | 13.9 (6.6) | – | 11.1 (5.6) | – |
| Negative | 15.0 (6.7) | – | 16.8 (4.7) | – |
| Psychotic episodes, mean (SD) | 1.7 (1.1) | – | 1.7 (0.7) | – |
IQ, intelligence quotient; PANSS, positive and negative syndrome scale; SD, standard deviation.
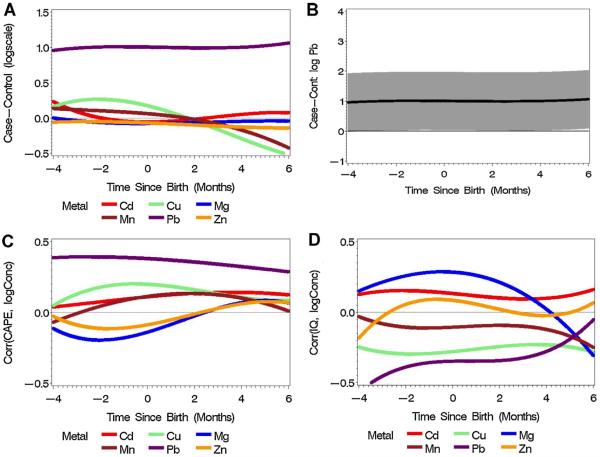
After stringent statistical correction, the longitudinal differences in log Pb2+ were generally estimated above zero, showing statistically significant higher early-life intake of Pb2+ among patients with schizophrenia compared with controls (Fig. 1A and B). The differences in log Mn2+ and log Cu2+ changed relatively linearly over time to postnatal negative values, indicating lower postnatal exposure to Cu2+ and Mn2+ in patients with schizophrenia than controls (Fig. 1A). The largest between group perinatal difference in Cu2+ and Mn2+ was observed six months postnatally (log difference case–control for Cu2+, −0.52, case/control ratio of 59% corresponding to 41% lower Cu2+ concentrations in cases compared to controls; log difference case–control for Mn2+, −0.42, case/control ratio of 66% corresponding to a 34% lower Mn2+ concentrations in cases compared to controls). There was a positive correlation between pre- and postnatal Pb2+ levels and CAPE score in adulthood (Fig. 1C). There was a negative correlation between Pb2+ levels and adult IQ (Fig. 1D), which was strongest during the second trimester of pregnancy (r = −0.39) and decreased gradually in the third trimester and postnatal period. There was a positive correlation between Mg2+ levels and IQ that peaked around birth (r = 0.28) and decreased postnatally (Fig. 1D).
Fig. 1.
Metal concentration in early life and adult psychosis and related phenotypes. (A) The difference of log concentrations between schizophrenia cases (N = 9) and controls (N = 5) for six metals during three developmental periods. (B) The differences of log concentrations between schizophrenia cases (N = 9) and controls (N = 5) for Pb2+ with Bonferroni-adjusted 95% confidence intervals. (C) The association between CAPE and perinatal log concentrations of six metals (N = 14). (D) The association between IQ and perinatal log concentrations of six metals (N = 14).
4. Discussion
Our proof-of-concept study is the first to use a tooth-matrix biomarker as a way to investigate the association between early-life metal exposure and long-term psychiatric outcomes. It provides initial support for the role of metal exposure during critical neurodevelopmental periods in early life and adult psychosis.
We believe that several characteristics make our study important. First, the study introduces a new method to reconstruct environmental exposures longitudinally from the second trimester and through the first year of life and it has the potential to transform research into environmental causes of mental disorders. The method is objective and therefore eliminates the possibility of bias in measuring environmental exposures. Second, correlations between metal exposures and severity of psychotic experiences ranged from 0.1 to 0.4, which is remarkable given the sample size, and that this is a unique longitudinal correlation across a 25-year period. Finally, unlike previous studies that used maternal samples as a way to measure fetal exposure, our method allows for direct measurement of exposure in utero, starting from second trimester of pregnancy. Larger sample sizes would definitely be beneficial as they would allow to increase power (which will lead to detection of more subtle statistical differences), and could help understand mechanisms better because they will allow for more sophisticated statistical analyses taking into account multiple metals.
We should also emphasize that the method is a highly sophisticated imaging technique that in contrast to all previous methods measures value of each metal for each individual hundreds of times across several months. Therefore, the tooth for each individual is represented by 120–180 distinct yet temporally connected values. Furthermore, although the sample size may seem small in clinical research, in molecular research, it is considered a moderate sample size. For example, PET imaging studies in human use comparable sample sizes [47–50]. Importantly, despite the small sample size, we were able to show a statistically significant effect for lead after a Bonferroni correction for multiple comparisons.
A clear pattern emerged from our results linking exposure to Pb2+ during early development with schizophrenia and psychotic experiences. Importantly, the association was both strong and statistically significant (i.e. the 95% CI for the ratios rarely included 1 and the correlation coefficient representing a `small'/`medium' effect size with 95% CI for the correlation coefficient rarely included zero). This confirms the findings of previous studies where more indirect measures of lead exposure were used to study the association between lead exposure during perinatal period and risk of schizophrenia. In two nested case–control studies, Opler et al. [35,36] showed that higher delta-aminolevulinic acid (D-ALA) levels corresponding to ≥15 μg/dL blood lead level in the mother is associated with a twice increase in risk of schizophrenia in the offspring compared with a blood lead level of <15 μg/dL.
We also found an interesting association between higher lead levels and low IQ. While previous studies provided substantial evidence for the association between childhood lead exposure and low IQ [4,51–54], our study takes this evidence one step further. Some previous studies suggested that the effect of lead exposure on mental development was different depending on the trimester of pregnancy. This was based on maternal blood samples [52,53]; but our study provides the first set of direct measurement of fetal in utero exposure. We found that lead levels during prenatal period had a significant negative correlation with IQ (r ~ −0.5); but after reaching a plateau in the third trimester and first two months of life, the magnitude of correlation gradually decreased toward zero. This finding suggests that the prenatal period is the critical time where exposure to lead can have a substantial impact on intellectual development and that the lead impact tends to decrease as the brain gradually becomes more mature. It should be noted however that our sample was small, and therefore further replication of our finding is required.
We also found evidence of potential differential time-related relationship and critical windows of susceptibility for several other metals, such as Mg2+, Cu2+, and Mn2+, and adult outcomes. Although small sample size precludes any definitive interpretation of our findings, it is clear that determining the exact timing of exposure is critical, because exposure during different developmental periods confer different risks for adult outcomes.
Nevertheless, our findings should be interpreted in light of some limitations. First, our study was underpowered by design and therefore 95% CIs for estimates were very wide. Nevertheless and despite wide 95% CIs we could find a statistically significant effect of lead exposure on IQ and risk for psychosis. Furthermore, small sample size precluded statistical adjustment for important confounders, such as socioeconomic status and the interaction between metals. Third, although we tried to choose the most relevant metals for our study, there are several other metals that might be plausibly linked to abnormal neurodevelopment and consequently higher risk of psychosis. For example, iron regulates three key processes in brain development, namely energy metabolism, dopaminergic transmission, and myelination [55]. Importantly, studies suggest that iron deficiency is more prevalent in patients with schizophrenia than healthy controls [33]. Fourth, an inherent limitation of our method is that it cannot assess metal exposure during first trimester, which is considered another critical period in brain development. However, our novel biomarker provides the first set of direct in utero evidence and therefore is extremely valuable in assessing environmental exposures during pregnancy.
In summary, this study provides support for the role of metal exposure during critical early-life neurodevelopmental periods in psychosis. Our new biomarker represents a potential major advance in environmental science research in adult psychiatric disorders, since we can now objectively determine the temporal pattern of early-life environmental exposures. Further studies in a larger sample with a wider array of environmental exposures are required to fully elucidate the association between early-life environment and schizophrenia and related phenotypes in the population.
Acknowledgments
Dr. Velthorst was supported by (VENI) grant 916-15-005 from the Netherlands Organization for Scientific Research.
Footnotes
Financial disclosures None declared.
Disclosure of interest The authors declare that they have no competing interest.
References
- [1].Weinberger DR. Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry. 1987;44:660–9. doi: 10.1001/archpsyc.1987.01800190080012. [DOI] [PubMed] [Google Scholar]
- [2].Wasserman GA, Liu X, Parvez F, Ahsan H, Factor-Litvak P, Kline J, et al. Water arsenic exposure and intellectual function in 6-year-old children in Araihazar, Bangladesh. Environ Health Perspect. 2007;115:285–9. doi: 10.1289/ehp.9501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Jamieson JA, Taylor CG, Weiler HA. Marginal zinc deficiency exacerbates bone lead accumulation and high dietary zinc attenuates lead accumulation at the expense of bone density in growing rats. Toxicol Sci. 2006;92:286–94. doi: 10.1093/toxsci/kfj201. [DOI] [PubMed] [Google Scholar]
- [4].Needleman HL, Gunnoe C, Leviton A, Reed R, Peresie H, Maher C, et al. Deficits in psychologic and classroom performance of children with elevated dentine lead levels. N Engl J Med. 1979;300:689–95. doi: 10.1056/NEJM197903293001301. [DOI] [PubMed] [Google Scholar]
- [5].Bellinger D, Hu H, Titlebaum L, Needleman HL. Attentional correlates of dentin and bone lead levels in adolescents. Arch Environ Health. 1994;49:98–105. doi: 10.1080/00039896.1994.9937461. [DOI] [PubMed] [Google Scholar]
- [6].Wright RO, Amarasiriwardena C, Woolf AD, Jim R, Bellinger DC. Neuropsychological correlates of hair arsenic, manganese, and cadmium levels in school-age children residing near a hazardous waste site. Neurotoxicology. 2006;27:210–6. doi: 10.1016/j.neuro.2005.10.001. [DOI] [PubMed] [Google Scholar]
- [7].Rodriguez-Barranco M, Lacasana M, Gil F, Lorca A, Alguacil J, Rohlman DS, et al. Cadmium exposure and neuropsychological development in school children in southwestern Spain. Environ Res. 2014;134:66–73. doi: 10.1016/j.envres.2014.06.026. [DOI] [PubMed] [Google Scholar]
- [8].Rodriguez-Barranco M, Lacasana M, Aguilar-Garduno C, Alguacil J, Gil F, Gonzalez-Alzaga B, et al. Association of arsenic, cadmium and manganese exposure with neurodevelopment and behavioural disorders in children: a systematic review and meta-analysis. Sci Total Environ. 2013;454–455:562–77. doi: 10.1016/j.scitotenv.2013.03.047. [DOI] [PubMed] [Google Scholar]
- [9].Adamo AM, Oteiza PI. Zinc deficiency and neurodevelopment: the case of neurons. Biofactors. 2010;36:117–24. doi: 10.1002/biof.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Bellinger DC. Very low lead exposures and children's neurodevelopment. Curr Opin Pediatr. 2008;20:172–7. doi: 10.1097/MOP.0b013e3282f4f97b. [DOI] [PubMed] [Google Scholar]
- [11].Akil M, Brewer GJ. Psychiatric and behavioral abnormalities in Wilson's disease. Adv Neurol. 1995;65:171–8. [PubMed] [Google Scholar]
- [12].McDonald LV, Lake CR. Psychosis in an adolescent patient with Wilson's disease: effects of chelation therapy. Psychosom Med. 1995;57:202–4. doi: 10.1097/00006842-199503000-00013. [DOI] [PubMed] [Google Scholar]
- [13].Koh JY, Choi DW. Zinc toxicity on cultured cortical neurons: involvement of N-methyl-d-aspartate receptors. Neuroscience. 1994;60:1049–57. doi: 10.1016/0306-4522(94)90282-8. [DOI] [PubMed] [Google Scholar]
- [14].Guilarte TR. Manganese neurotoxicity: new perspectives from behavioral, neuroimaging, and neuropathological studies in humans and non-human primates. Front Aging Neurosci. 2013;5:23. doi: 10.3389/fnagi.2013.00023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Grandjean P, Landrigan PJ. Neurobehavioural effects of developmental toxicity. Lancet Neurol. 2014;13:330–8. doi: 10.1016/S1474-4422(13)70278-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Grandjean P, Landrigan PJ. Developmental neurotoxicity of industrial chemicals. Lancet. 2006;368:2167–78. doi: 10.1016/S0140-6736(06)69665-7. [DOI] [PubMed] [Google Scholar]
- [17].Wasserman GA, Liu X, Parvez F, Ahsan H, Factor-Litvak P, van Geen A, et al. Water arsenic exposure and children's intellectual function in Araihazar, Bangladesh. Environ Health Perspect. 2004;112:1329–33. doi: 10.1289/ehp.6964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Sandstead HH, Penland JG, Alcock NW, Dayal HH, Chen XC, Li JS, et al. Effects of repletion with zinc and other micronutrients on neuropsychologic performance and growth of Chinese children. Am J Clin Nutr. 1998;68:470S–5S. doi: 10.1093/ajcn/68.2.470S. [DOI] [PubMed] [Google Scholar]
- [19].Umamaheswari K, Bhaskaran M, Krishnamurthy G, Vasudevan H, Vasudevan K. Effect of iron and zinc deficiency on short term memory in children. Indian Pediatr. 2011;48:289–93. doi: 10.1007/s13312-011-0060-7. [DOI] [PubMed] [Google Scholar]
- [20].Cecil KM, Brubaker CJ, Adler CM, Dietrich KN, Altaye M, Egelhoff JC, et al. Decreased brain volume in adults with childhood lead exposure. PLoS Med. 2008;5:e112. doi: 10.1371/journal.pmed.0050112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Wasserman GA, Liu X, Parvez F, Factor-Litvak P, Ahsan H, Levy D, et al. Arsenic and manganese exposure and children's intellectual function. Neurotoxicology. 2011;32:450–7. doi: 10.1016/j.neuro.2011.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Brubaker CJ, Schmithorst VJ, Haynes EN, Dietrich KN, Egelhoff JC, Lindquist DM, et al. Altered myelination and axonal integrity in adults with childhood lead exposure: a diffusion tensor imaging study. Neurotoxicology. 2009;30:867–75. doi: 10.1016/j.neuro.2009.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Neal AP, Guilarte TR. Molecular neurobiology of lead (Pb(2+)): effects on synaptic function. Mol Neurobiol. 2010;42:151–60. doi: 10.1007/s12035-010-8146-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Guilarte TR, Chen MK. Manganese inhibits NMDA receptor channel function: implications to psychiatric and cognitive effects. Neurotoxicology. 2007;28:1147–52. doi: 10.1016/j.neuro.2007.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Guilarte TR, Opler M, Pletnikov M. Is lead exposure in early life an environmental risk factor for Schizophrenia? Neurobiological connections and testable hypotheses. Neurotoxicology. 2012;33:560–74. doi: 10.1016/j.neuro.2011.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Forrest CM, Khalil OS, Pisar M, Darlington LG, Stone TW. Prenatal inhibition of the tryptophan–kynurenine pathway alters synaptic plasticity and protein expression in the rat hippocampus. Brain Res. 2013;1504:1–15. doi: 10.1016/j.brainres.2013.01.031. [DOI] [PubMed] [Google Scholar]
- [27].Forrest CM, Khalil OS, Pisar M, McNair K, Kornisiuk E, Snitcofsky M, et al. Changes in synaptic transmission and protein expression in the brains of adult offspring after prenatal inhibition of the kynurenine pathway. Neuroscience. 2013;254:241–59. doi: 10.1016/j.neuroscience.2013.09.034. [DOI] [PubMed] [Google Scholar]
- [28].Forrest CM, McNair K, Pisar M, Khalil OS, Darlington LG, Stone TW. Altered hippocampal plasticity by prenatal kynurenine administration, kynurenine-3-monoxygenase (KMO) deletion or galantamine. Neuroscience. 2015;310:91–105. doi: 10.1016/j.neuroscience.2015.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Salustri C, Barbati G, Ghidoni R, Quintiliani L, Ciappina S, Binetti G, et al. Is cognitive function linked to serum free copper levels? A cohort study in a normal population. Clin Neurophysiol: Off J Int Fed Clin Neurophysiol. 2010;121:502–7. doi: 10.1016/j.clinph.2009.11.090. [DOI] [PubMed] [Google Scholar]
- [30].Rahman A, Azad MA, Hossain I, Qusar MM, Bari W, Begum F, et al. Zinc, manganese, calcium, copper, and cadmium level in scalp hair samples of schizophrenic patients. Biol Trace Elem Res. 2009;127:102–8. doi: 10.1007/s12011-008-8230-8. [DOI] [PubMed] [Google Scholar]
- [31].Wolf TL, Kotun J, Meador-Woodruff JH. Plasma copper, iron, ceruloplasmin and ferroxidase activity in schizophrenia. Schizophr Res. 2006;86:167–71. doi: 10.1016/j.schres.2006.05.027. [DOI] [PubMed] [Google Scholar]
- [32].Liu T, Lu QB, Yan L, Guo J, Feng F, Qiu J, et al. Comparative study on serum levels of 10 trace elements in schizophrenia. PLoS ONE. 2015;10:e0133622. doi: 10.1371/journal.pone.0133622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Yanik M, Kocyigit A, Tutkun H, Vural H, Herken H. Plasma manganese, selenium, zinc, copper, and iron concentrations in patients with schizophrenia. Biol Trace Elem Res. 2004;98:109–17. doi: 10.1385/BTER:98:2:109. [DOI] [PubMed] [Google Scholar]
- [34].Pfeiffer CC, Iliev V. A study of zinc deficiency and copper excess in the schizophrenias. Int Rev Neurobiol. 1972;1:141–65. [Google Scholar]
- [35].Opler MG, Brown AS, Graziano J, Desai M, Zheng W, Schaefer C, et al. Prenatal lead exposure, delta-aminolevulinic acid, and schizophrenia. Environ Health Perspect. 2004;112:548–52. doi: 10.1289/ehp.6777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Opler MG, Buka SL, Groeger J, McKeague I, Wei C, Factor-Litvak P, et al. Prenatal exposure to lead, delta-aminolevulinic acid, and schizophrenia: further evidence. Environ Health Perspect. 2008;116:1586–90. doi: 10.1289/ehp.10464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Korver N, Quee PJ, Boos HB, Simons CJ, de Haan L, investigators G Genetic Risk and Outcome of Psychosis (GROUP), a multi-site longitudinal cohort study focused on gene-environment interaction: objectives, sample characteristics, recruitment and assessment methods. Int J Methods Psychiatr Res. 2012;21:205–21. doi: 10.1002/mpr.1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Stefanis NC, Hanssen M, Smirnis NK, Avramopoulos DA, Evdokimidis IK, Stefanis CN, et al. Evidence that three dimensions of psychosis have a distribution in the general population. Psychol Med. 2002;32:347–58. doi: 10.1017/s0033291701005141. [DOI] [PubMed] [Google Scholar]
- [39].Hare D, Austin C, Doble P, Arora M. Elemental bio-imaging of trace elements in teeth using laser ablation-inductively coupled plasma-mass spectrometry. J Dent. 2011;39:397–403. doi: 10.1016/j.jdent.2011.03.004. [DOI] [PubMed] [Google Scholar]
- [40].Arora M, Austin C. Teeth as a biomarker of past chemical exposure. Curr Opin Pediatr. 2013;25:261–7. doi: 10.1097/MOP.0b013e32835e9084. [DOI] [PubMed] [Google Scholar]
- [41].Coull BCHB, Wright RO, Arora M. Statistical methods for analyzing critical windows of metal exposures using the tooth biomarker. Int Soc Expo Sci (ISES) 2014 [Google Scholar]
- [42].Arora M, Hare D, Austin C, Smith DR, Doble P. Spatial distribution of manganese in enamel and coronal dentine of human primary teeth. Sci Total Environ. 2011 doi: 10.1016/j.scitotenv.2010.12.018. [DOI] [PubMed] [Google Scholar]
- [43].Arora M, Kennedy BJ, Elhlou S, Pearson NJ, Walker DM, Bayl P, et al. Spatial distribution of lead in human primary teeth as a biomarker of pre- and neonatal lead exposure. Sci Total Environ. 2006;371:55–62. doi: 10.1016/j.scitotenv.2006.07.035. [DOI] [PubMed] [Google Scholar]
- [44].Arora M, Austin C, Sarrafpour B, Hernandez-Avila M, Hu H, Wright RO, et al. Determining prenatal, early childhood and cumulative long-term lead exposure using micro-spatial deciduous dentine levels. PLoS ONE. 2014;9:e97805. doi: 10.1371/journal.pone.0097805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Arora M, Bradman A, Austin C, Vedar M, Holland N, Eskenazi B, et al. Determining fetal manganese exposure from mantle dentine of deciduous teeth. Environ Sci Technol. 2012;46:5118–25. doi: 10.1021/es203569f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Austin C, Smith TM, Bradman A, Hinde K, Joannes-Boyau R, Bishop D, et al. Barium distributions in teeth reveal early-life dietary transitions in primates. Nature. 2013;498:216–9. doi: 10.1038/nature12169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Rodda J, Okello A, Edison P, Dannhauser T, Brooks DJ, Walker Z. (11)C-PIB PET in subjective cognitive impairment. Eur Psychiatry. 2010;25:123–5. doi: 10.1016/j.eurpsy.2009.07.011. [DOI] [PubMed] [Google Scholar]
- [48].Ko JH, Koshimori Y, Mizrahi R, Rusjan P, Wilson AA, Lang AE, et al. Voxel-based imaging of translocator protein 18 kDa (TSPO) in high-resolution PET. J Cereb Blood Flow Metab: Off J Int Soc Cereb Blood Flow Metab. 2013;33:348–50. doi: 10.1038/jcbfm.2012.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Bauer M, Romermann K, Karch R, Wulkersdorfer B, Stanek J, Philippe C, et al. A pilot PET study to assess the functional interplay between ABCB1 and ABCG2 at the human blood-brain barrier. Clin Pharmacol Ther. 2016 doi: 10.1002/cpt.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Park E, Hwang YM, Chu MK, Jung KY. Increased brainstem serotonergic transporter availability in adult migraineurs: an [(18)F]FP-CIT PET imaging pilot study. Nucl Med Mol Imaging. 2016;50:70–5. doi: 10.1007/s13139-015-0373-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Lanphear BP, Hornung R, Khoury J, Yolton K, Baghurst P, Bellinger DC, et al. Low-level environmental lead exposure and children's intellectual function: an international pooled analysis. Environ Health Perspect. 2005;113:894–9. doi: 10.1289/ehp.7688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Hu H, Tellez-Rojo MM, Bellinger D, Smith D, Ettinger AS, Lamadrid-Figueroa H, et al. Fetal lead exposure at each stage of pregnancy as a predictor of infant mental development. Environ Health Perspect. 2006;114:1730–5. doi: 10.1289/ehp.9067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Liu J, Gao D, Chen Y, Jing J, Hu Q, Chen Y. Lead exposure at each stage of pregnancy and neurobehavioral development of neonates. Neurotoxicology. 2014;44:1–7. doi: 10.1016/j.neuro.2014.03.003. [DOI] [PubMed] [Google Scholar]
- [54].Brown AS, Schaefer CA, Quesenberry CP, Jr, Liu L, Babulas VP, Susser ES. Maternal exposure to toxoplasmosis and risk of schizophrenia in adult offspring. Am J Psychiatry. 2005;162:767–73. doi: 10.1176/appi.ajp.162.4.767. [DOI] [PubMed] [Google Scholar]
- [55].Georgieff MK. The role of iron in neurodevelopment: fetal iron deficiency and the developing hippocampus. Biochem Soc Trans. 2008;36:1267–71. doi: 10.1042/BST0361267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Meyer-Baron M, Knapp G, Schaper M, van Thriel C. Performance alterations associated with occupational exposure to manganese – a meta-analysis. Neurotoxicology. 2009;30:487–96. doi: 10.1016/j.neuro.2009.05.001. [DOI] [PubMed] [Google Scholar]
- [57].Wenisch E, De Tassigny A, Trocello JM, Beretti J, Girardot-Tinant N, Woimant F. Cognitive profile in Wilson's disease: a case series of 31 patients. Rev Neurol – France. 2013;169:944–9. doi: 10.1016/j.neurol.2013.06.002. [DOI] [PubMed] [Google Scholar]
- [58].Medalia A, Isaacsglaberman K, Scheinberg IH. Neuropsychological impairment in Wilsons-disease. Arch Neurol – Chicago. 1988;45:502–4. doi: 10.1001/archneur.1988.00520290030009. [DOI] [PubMed] [Google Scholar]
- [59].Schendel DE, Berg CJ, Yeargin-Allsopp M, Boyle CA, Decoufle P. Prenatal magnesium sulfate exposure and the risk for cerebral palsy or mental retardation among very low-birth-weight children aged 3 to 5 years. JAMA. 1996;276:1805–10. [PubMed] [Google Scholar]
- [60].Shrestha KP, Carrera AE. Hair trace elements and mental retardation among children. Arch Environ Health. 1988;43:396–8. doi: 10.1080/00039896.1988.9935857. [DOI] [PubMed] [Google Scholar]
- [61].Slutsky I, Abumaria N, Wu LJ, Huang C, Zhang L, Li B, et al. Enhancement of learning and memory by elevating brain magnesium. Neuron. 2010;65:165–77. doi: 10.1016/j.neuron.2009.12.026. [DOI] [PubMed] [Google Scholar]
- [62].Ghia N, Spong CY, Starbuck VN, Scialli AR, Ghidini A. Magnesium sulfate therapy affects attention and working memory in patients undergoing pre-term labor. Am J Obstet Gynecol. 2000;183:940–4. doi: 10.1067/mob.2000.109045. [DOI] [PubMed] [Google Scholar]
- [63].Starobrat-Hermelin B, Kozielec T. The effects of magnesium physiological supplementation on hyperactivity in children with attention deficit hyperactivity disorder (ADHD). Positive response to magnesium oral loading test. Magnes Res. 1997;10:149–56. [PubMed] [Google Scholar]
- [64].Alkondon M, Costa AC, Radhakrishnan V, Aronstam RS, Albuquerque EX. Selective blockade of NMDA-activated channel currents may be implicated in learning deficits caused by lead. FEBS Lett. 1990;261:124–30. doi: 10.1016/0014-5793(90)80652-y. [DOI] [PubMed] [Google Scholar]
- [65].Nation JR, Frye GD, Von Stultz J, Bratton GR. Effects of combined lead and cadmium exposure: changes in schedule-controlled responding and in dopamine, serotonin, and their metabolites. Behav Neurosci. 1989;103:1108–14. doi: 10.1037//0735-7044.103.5.1108. [DOI] [PubMed] [Google Scholar]
- [66].Zuch CL, O'Mara DJ, Cory-Slechta DA. Low-level lead exposure selectively enhances dopamine overflow in nucleus accumbens: an in vivo electrochemistry time course assessment. Toxicol Appl Pharmacol. 1998;150:174–85. doi: 10.1006/taap.1998.8396. [DOI] [PubMed] [Google Scholar]
- [67].Andersson H, Petersson-Grawe K, Lindqvist E, Luthman J, Oskarsson A, Olson L. Low-level cadmium exposure of lactating rats causes alterations in brain serotonin levels in the offspring. Neurotoxicol Teratol. 1997;19:105–15. doi: 10.1016/s0892-0362(96)00218-8. [DOI] [PubMed] [Google Scholar]
- [68].Gutierrez-Reyes EY, Albores A, Rios C. Increase of striatal dopamine release by cadmium in nursing rats and its prevention by dexamethasone-induced metallothionein. Toxicology. 1998;131:145–54. doi: 10.1016/s0300-483x(98)00126-7. [DOI] [PubMed] [Google Scholar]
- [69].Minami A, Takeda A, Nishibaba D, Takefuta S, Oku N. Cadmium toxicity in synaptic neurotransmission in the brain. Brain Res. 2001;894:336–9. doi: 10.1016/s0006-8993(01)02022-4. [DOI] [PubMed] [Google Scholar]
- [70].Zhang L, Bai R, Liu Y, Meng L, Li B, Wang L, et al. The dose-dependent toxicological effects and potential perturbation on the neurotransmitter secretion in brain following intranasal instillation of copper nanoparticles. Nanotoxicology. 2012;6:562–75. doi: 10.3109/17435390.2011.590906. [DOI] [PubMed] [Google Scholar]
- [71].Okada M, Mizuno K, Okuyama M, Kaneko S. Magnesium ion augmentation of inhibitory effects of adenosine on dopamine release in the rat striatum. Psychiatry Clin Neurosci. 1996;50:147–56. doi: 10.1111/j.1440-1819.1996.tb01680.x. [DOI] [PubMed] [Google Scholar]
- [72].Buck DR, Mahoney AW, Hendricks DG. Effects of cerebral intraventricular magnesium injections and a low magnesium diet on nonspecific excitability level, audiogenic seizure susceptibility and serotonin. Pharmacol Biochem Behav. 1979;10:487–91. doi: 10.1016/0091-3057(79)90222-3. [DOI] [PubMed] [Google Scholar]
- [73].Danysz W, Parsons CG, Mobius HJ, Stoffler A, Quack G. Neuroprotective and symptomatological action of memantine relevant for Alzheimer's disease – a unified glutamatergic hypothesis on the mechanism of action. Neurotox Res. 2000;2:85–97. doi: 10.1007/BF03033787. [DOI] [PubMed] [Google Scholar]
- [74].Garcia-Colunga J, Reyes-Haro D, Godoy-Garcia IU, Miledi R. Zinc modulation of serotonin uptake in the adult rat corpus callosum. J Neurosci Res. 2005;80:145–9. doi: 10.1002/jnr.20421. [DOI] [PubMed] [Google Scholar]
- [75].Pifl C, Wolf A, Rebernik P, Reither H, Berger ML. Zinc regulates the dopamine transporter in a membrane potential and chloride dependent manner. Neuropharmacology. 2009;56:531–40. doi: 10.1016/j.neuropharm.2008.10.009. [DOI] [PubMed] [Google Scholar]
- [76].Hollmann M, Boulter J, Maron C, Beasley L, Sullivan J, Pecht G, et al. Zinc potentiates agonist-induced currents at certain splice variants of the NMDA receptor. Neuron. 1993;10:943–54. doi: 10.1016/0896-6273(93)90209-a. [DOI] [PubMed] [Google Scholar]
- [77].Arinola G, Idonije B, Akinlade K, Ihenyen O. Essential trace metals and heavy metals in newly diagnosed schizophrenic patients and those on antipsychotic medication. J Res Med Sci: Off J Isfahan Univ Med Sci. 2010;15:245–9. [PMC free article] [PubMed] [Google Scholar]
- [78].Matzen TA, Martin RL. Magnesium deficiency psychosis induced by cancer chemotherapy. Biol Psychiatry. 1985;20:788–91. doi: 10.1016/0006-3223(85)90158-1. [DOI] [PubMed] [Google Scholar]
- [79].Nechifor M, Vaideanu C, Palamaru I, Borza C, Mindreci I. The influence of some antipsychotics on erythrocyte magnesium and plasma magnesium, calcium, copper and zinc in patients with paranoid schizophrenia. J Am Coll Nutr. 2004;23:549s–51s. doi: 10.1080/07315724.2004.10719401. [DOI] [PubMed] [Google Scholar]