Abstract
The inflammatory bowel diseases ulcerative colitis and Crohn’s disease are associated with an increased risk for the development of colorectal cancer. During recent years, several immune signaling pathways have been linked to colitis-associated cancer (CAC), largely owing to the availability of suitable preclinical models. Among these, chronic intestinal inflammation has been shown to support tumor initiation through oxidative stress–induced mutations. A proinflammatory microenvironment that develops, possibly as a result of defective intestinal barrier function and host–microbial interactions, enables tumor promotion. Several molecular pathways such as tumor necrosis factor/nuclear factor-κB or interleukin 6/signal transducer and activator of transcription 3 signaling have been identified as important contributors to CAC development and could be promising therapeutic targets for the prevention and treatment of CAC.
Keywords: Colorectal Cancer, Crohn's Disease, Cytokines, Inflammatory Bowel Disease, Interleukin-6, Tumor Necrosis Factor Alpha, Ulcerative Colitis
Abbreviations used in this paper: AOM-DSS, azoxymethane–dextran sulfate sodium; APC, adenomatous polyposis coli; CAC, colitis-associated cancer; CD, Crohn’s disease; CRC, colorectal cancer; DDR, DNA damage response; gp, glycoprotein; IBD, inflammatory bowel disease; IKK, IκB kinase; IL, interleukin; IL6R, interleukin 6 receptor; LPS, lipopolysaccharide; Myd88, myeloid differentiation primary response gene 88; NF-κB, nuclear factor-κB; NLR, NOD- and leucine-rich repeat–containing protein; NLRP, nucleotide-binding oligomerization domain- and leucine-rich repeat–containing protein family, pyrin domain-containing; NOD, nucleotide-binding oligomerization domain; RONS, reactive oxygen and nitrogen species; STAT3, signal transducer and activator of transcription 3; Th17, T-helper 17; TLR, Toll-like receptor; TNF, tumor necrosis factor; TNFR, tumor necrosis factor receptor; UC, ulcerative colitis
Summary.
This review discusses recent data on immune signaling pathways involved in the pathogenesis of colitis-associated cancer. These include molecular mechanisms activating the innate and adaptive immune system and thereby contributing to cancer initiation and promotion in inflammatory bowel diseases.
Concepts about inflammation-associated cancer development go back to Rudolph Virchow’s observation of increased immune cell infiltration at tumor sites more than 150 years ago.1 Today, inflammatory conditions including infection and immune-mediated disease increase the risk of cancer. Well-known examples in the gastrointestinal tract are the increased risk for gastric cancer and gastric lymphoma in Helicobacter pylori infection and the increased risk for colorectal cancer (CRC) in inflammatory bowel disease (IBD).2
The first reports of colorectal cancer in IBD patients occurred in the early 1900s, when Crohn and Rosenberg3 described a case of colonic adenocarcinoma in a patient with long-term ulcerative colitis (UC). The CRC risk in IBD patients initially was attributed mostly to UC and not to Crohn’s disease (CD) because epidemiologic studies in the 1960s had proposed an up to 10 times greater CRC risk in UC, but not in CD, patients in comparison with the general population.4 Disease extent and duration are regarded as the most important parameters affecting the individual CRC risk in patients with UC. Recent data also have shown an association between the degree of inflammation and the development of colonic neoplasia.5, 6 Additional risk factors include primary sclerosing cholangitis and a family history of CRC.7 Together, the cumulative risk for CRC in UC patients has been reported as 1.6% after 10 years, 8.3% after 20 years, and 18.4% after 30 years of disease duration.8 Because these data are based on studies from academic centers, which frequently have patients with more severe disease, true incidence rates may be lower. For instance, Jess et al9 reported a 2.4-fold increased risk for CRC in UC patients after 15 years of disease in a meta-analysis of population-based cohort studies.
In contrast to UC, the influence of CD on CRC risk has been under debate for many decades. Although several cases of CRC were reported in CD patients beginning in the 1950s, subsequent studies could not detect increased incidence rates in comparison with the general population.10 Recent studies have reported that the risk for CRC in patients with CD patients depends on large-bowel involvement. Similar to UC, the extent and duration of colonic inflammation are the most important risk factors for CRC development in CD patients. In this regard, the cumulative risk for CRC in CD patients has been reported to be 2.9%, 5.6%, and 8.3% after 10, 20, and 30 years of disease, respectively, in a meta-analysis.11 Again, these data are based on studies from academic centers and therefore may overstate the actual incidence rates in patients with CD.
Because of the availability of reasonable preclinical models, our knowledge regarding the molecular mechanisms connecting inflammation and cancer development in colitis-associated cancer (CAC) has increased rapidly in recent years. Chronic inflammation has been linked to tumor initiation, in which normal cells acquire genomic alterations that initiate tumorigenesis, as well as promotion driven by the sustained proliferation of initiated cells.12 This review discusses recent progress in understanding immune signaling pathways involved in these steps during colitis-associated cancer development.
Oxidative Stress–Induced DNA Damage in CAC
For tumor initiation, distinct mutations of oncogenes or tumor-suppressor genes are required to allow subsequent tumor development. These include mutations that result in resistance to apoptosis as well as acquisition of malignant potential. Mutations involved in the initiation of sporadic colorectal carcinoma have been well characterized and accumulate along the individual steps of described adenoma–carcinoma sequence pathways.13, 14 Similarly, a sequence of distinct mutations occurs during the stepwise development of colitis-associated cancer. This can be referred to as the inflammation–dysplasia–carcinoma pathway, which describes the development of low-grade dysplasia in a background of intestinal inflammation, with subsequent progression to high-grade dysplasia, and, finally, invasive carcinoma.15 Notably, not all lesions follow this stepwise evolution. Because the tumorigenic pathway and individual genes affected differ between sporadic and colitis-associated CRC, it is reasonable to propose that the mechanisms inducing these mutations also differ. Mutations in sporadic CRC have been attributed to several kinds of genomic and epigenetic instability including chromosomal instability, CpG island methylator phenotype, global hypomethylation, and mutations in mismatch repair genes that lead to microsatellite instability.16 Although these genomic and epigenetic alterations also occur in CAC,17 growing evidence supports a central role for inflammation-dependent oxidative stress in the induction of mutations that lead to CAC.
Oxidative stress occurs as an imbalance of the generation and elimination of reactive oxygen and nitrogen species (RONS).18 Increased oxidative stress is one of the key features of chronic inflammation because cells of the innate immune system release various kinds of RONS including superoxide, hydrogen peroxide, singlet oxygen, hydroxyl radicals, and nitric oxide into the tissue microenvironment on activation. These RONS interact with the DNA of resident cells and induce various forms of DNA damage including single- and double-strand breaks, abasic sites, and nucleotide modification, all of which contribute to tumor initiation when they affect oncogenes or tumor-suppressor genes.19
In human IBD, studies have shown that increased RONS correlates with disease activity, as well as reduced antioxidant levels. For instance, 8-oxo-7,8-dihydro-2,-deoxyguanosine, an oxidative stress–dependent base modification, is common in inflamed and dysplastic tissue, but not in healthy mucosa.20 Similarly, concentrations of nitric oxide are increased and correlate with oxidative damage in tissue samples of active and even inactive IBD.21
To repair reactive oxygen species-induced mutations, the DNA damage response (DDR) is activated, which comprises various mechanisms including direct repair, nucleotide excision repair, and others (see the article by Curtin22 for a review). Furthermore, the DDR can regulate cellular proliferation through activation of premature cellular senescence, an irreversible arrest of cell-cycle progression that protects against the amplification of defective DNA and proliferation of mutant clones.23 Senescence occurs in various precancerous lesions, and evasion as a result of mutations in senescence-associated genes has been regarded as a requirement for malignant transformation.
In IBD, Sohn et al24 found an increase of DDR (Histon gamma H2A.X, phospho-checkpoint kinase 2) and senescence (Heterochromatin protein 1 gamma) markers in inflamed tissue samples from IBD patients. In UC samples, increased DDR and senescence correlated with infiltration of macrophages as a possible source of RONS. Supporting the activation of senescence as a protective mechanism against malignant transformation in CAC, high levels of senescence markers have been reported in low-grade dysplasia in comparison with nondysplastic inflamed tissues and also high-grade dysplasia of UC patients, proposing an evasion of senescence during the progression from low- to high-grade dysplasia.25, 26
Evidence for the pathogenic role of oxidative stress and protection afforded by the DDR is provided by preclinical studies using CAC models. Meira et al27 showed that mice deficient in alkyladenine DNA glycosylase, an enzyme involved in base excision repair, developed more DNA base lesions and higher numbers of tumors in the widely used azoxymethane–dextran sodium sulfate (AOM-DSS) model of CAC. In the same model, mice deficient in the transcription factor nuclear factor-erythroid 2–related factor 2, which regulates genes involved in antioxidant signaling pathways, also had increased numbers of aberrant crypt foci.28 Furthermore, mice lacking the glutathione peroxidase Glutathione peroxidase 3, which is regulated by nuclear factor-erythroid 2–related factor 2 and acts as a redox enzyme, develop more tumors with higher grades of dysplasia in the AOM-DSS model.29 GPX3-deficient mice even developed polyps after DSS treatment without AOM, indicating that increased oxidative stress without an effective DDR is sufficient for tumor initiation. After tumor initiation, the proinflammatory microenvironment also contributes to tumor promotion.
Proinflammatory Signaling Pathways: The Role of Nuclear Factor-κB in Tumor Initiation and Promotion
Inflammation occurring as a response to infection or tissue damage removes dead cells and promotes restoration of tissue integrity via stem cell and myofibroblast activation, cell proliferation, angiogenesis, and other processes. Because of the overlap between mechanisms involved in wound healing and tumorigenesis, tumors have been described as “wounds that do not heal.”30 In fact, chronic inflammation can result in excessive tissue regeneration and thereby enhance the promotion and progression of initiated tumor cells.31 During recent years, major proinflammatory pathways have been implicated in inflammation-associated tumor development. Among these, the group of nuclear factor-κB (NF-κB) transcription factors takes center stage. NF-κB transcription factors comprise dimers of the subunits RelA (p65), c-Rel, RelB, p50/NF-κB1, and p52/NF-κB2.32 In a resting state, NF-κB dimers are bound to specific inhibitors (Iκ) within the cytoplasm. Two signaling pathways leading to NF-κB activation have been reported. In the classic pathway, proinflammatory stimuli including tumor necrosis factor α (TNFα), interleukin 1 (IL1), lipopolysaccharide (LPS), and CD40 ligand lead to activation of the IκB kinase (IKK) subunits IKK-α, IKK-β, and IKK-γ (NF-kappa-B essential modulator), which together target IκB proteins for proteosomal degradation. Subsequently, unbound NF-κB dimers (primarily RelA/p50) translocate to the nucleus and initiate transcription of target genes. The alternative pathway largely is dependent on activation by lymphotoxin α/β and CD40L, but not TNFα, IL1, or LPS. Alternative pathway activation leads to the conversion of p100 to p52 via IKK-α–dependent phosphorylation and subsequent proteosomal cleavage. The resulting p52/RelB heterodimers translocate to the nucleus and lead to target gene transcription (for review see the article by Karin et al33). Both pathways of NF-κB activation lead to increased expression of genes involved in the regulation of cell-cycle progression, apoptosis, and other cancer-relevant signaling pathways.
In CAC, the initial evidence for the functional relevance of NF-κB signaling was provided in a preclinical study by Greten et al,34 which showed that deletion of IKK-β in intestinal epithelial cells resulted in decreased numbers of tumors, increased apoptosis, and defective Bcl-Xl signaling. In contrast, deletion of IKK-β in myeloid cells led to diminished tumor size along with reduced expression of proinflammatory cytokines including IL1β, TNFα, and IL6 in the AOM-DSS model. These data suggest that tumor cell–specific NF-κB activation is required for inflammation-associated tumor initiation, and NF-κB activation in myeloid cells contributes to tumor promotion and progression. In line with these data, mice with constitutive activation of IKK-β in intestinal epithelial cells and germline deletion of the tumor-suppressor gene adenomatous polyposis coli (APC) show enhanced tumor development with marked DNA damage and DDR activation.35 Because differences in tumor load were largely dependent on tumor numbers, and tumors even developed spontaneously in mice with constitutively active IKK-β (even without APC loss), these data again highlight the central roles of epithelial NF-κB activation and oxidative stress in tumor initiation.
Interestingly, a study by Cooks et al36 found increased NF-κB signaling in mice with a gain-of-function mutation of TP53. The p53 protein generally acts as a mediator of cellular senescence by triggering cell-cycle arrest. Mutations affecting TP53 occur at late stages of sporadic CRC, usually resulting in loss of p53 function, bypass of senescence, and infiltrative and metastatic tumor growth.37, 38 In contrast to sporadic CRC, TP53 mutations occur at early steps of CAC, before infiltrative or metastatic tumor growth. Therefore, the functional role of TP53 mutations at early steps of CAC has been controversial. The data discussed earlier suggest that gain-of-function mutations in TP53 at early steps of CAC development enhance NF-κB signaling in tumor cells.36 Despite these advances, very few studies have analyzed NF-κB signaling in human CAC.39, 40
TNFα as a Link Between Chronic Intestinal Inflammation and CAC
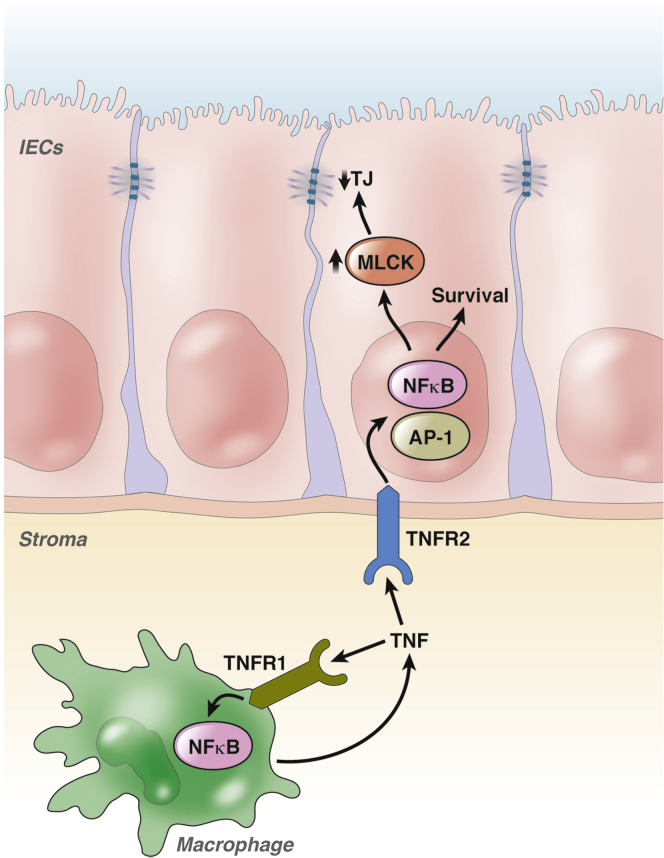
Because of its central role in maintaining chronic inflammation in IBD, TNFα signaling has been proposed as a tumor-promoting mechanism in CAC. The contribution of TNFα to IBD is highlighted by the efficacy of anti-TNFα therapeutics that now routinely are used to treat IBD patients.41 TNFα, which is expressed as pro-TNF on the plasma membrane of myeloid and T cells, is released after cleavage by converting enzymes such as ADAM-17. Soluble TNFα then binds to 1 of 2 receptors: TNF-Rp55 (TNFR1) or TNF-Rp75 (TNFR2).41 Although activation of TNFR1 results in apoptosis via caspase-3 activation, activation of TNFR2 promotes cell survival via activation of NF-κB and other signaling pathways.42 Although TNF initially was discovered and named as a factor that induces tumor necrosis, it now has been implicated in the pathogenesis of inflammation-associated cancer, likely as a consequence of proinflammatory signaling. Functional evidence for this concept has been provided by preclinical studies using the AOM-DSS CAC model. TNFR2 expression is up-regulated on intestinal epithelial cells in adoptive transfer and DSS colitis,43, 44 and increased NF-κB signaling after TNFR2 activation in the AOM-DSS model.43 Treatment of mice with an anti-TNF antibody reduced tumor number and size. TNFR2 activation in intestinal epithelial cells also leads to myosin light chain kinase up-regulation with subsequent release of pro-tumorigenic cytokines and breakdown of tight junctions.44, 45, 46 Similar to TNFR2, tumor growth was reduced in TNFR1-deficient mice as well as in wild-type mice treated with the anti-TNF inhibitor entanercept in the AOM-DSS model.47 The effectiveness of entanercept is somewhat surprising because this anti-TNF biologic is not effective in human IBD. Nevertheless, reduced TNF signaling is accompanied by decreased colonic infiltration by neutrophils and macrophages. Bone marrow chimera mice also were used to show that the effects of TNFR1 on tumor growth were caused by expression on bone marrow–derived rather than stromal and parenchymal cell types. Thus, TNFR1 activation in infiltrating immune cells seems to be important for tumor-promoting effects of chronic inflammation, whereas TNFR2 activation seems to be important for NF-κB–dependent tumor cell survival, tight junction barrier loss, and tumor-promoting cytokine release (Figure 1).
Figure 1.
TNFα-dependent activation of NF-κB in CAC. TNF, a major proinflammatory cytokine involved in the pathogenesis of IBD, contributes to CAC development through the activation of NF-κB in myeloid and intestinal epithelial cells. Although TNFR1 activation in myeloid cells leads to the release of additional proinflammatory cytokines that contribute to tumor growth, TNFR2 activation in epithelial cells promotes cell survival as well as the activation of myosin light chain kinase (MLCK), which supports epithelial barrier loss through direct effects on tight junctions (TJ). After TNFR2 activation, the MLCK promoter can be regulated via NF-κB or activator protein 1. IEC, intestinal epithelial cell.
Importantly, these data on TNF signaling in CAC were derived from preclinical studies, whereas data from human CAC are rare. Moreover, despite the widespread use of anti-TNF therapeutics in IBD treatment, there are currently no data available that clearly show a preventive effect on CAC development.
IL6-Dependent Signal Transducer and Activator of Transcription 3 Activation in IECs
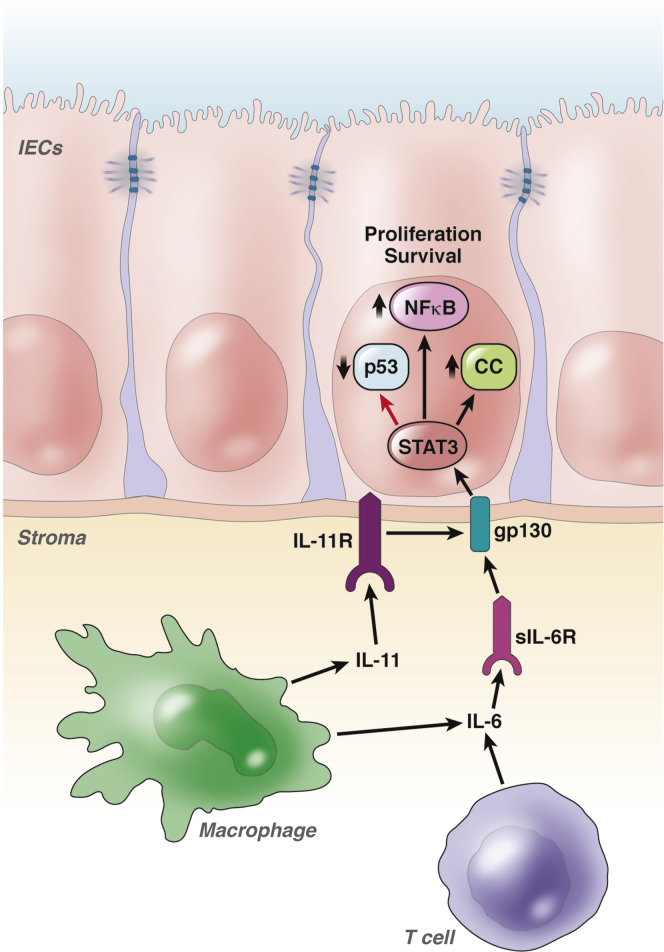
Another cytokine implicated in the pathogenesis of CAC is IL6. On activation of proinflammatory signaling pathways involving NF-κB or nuclear factor of activated T-cells, cytoplasmic 2, IL6 is released by monocytes, macrophages, fibroblasts, endothelial cells, lymphocytes, and cancer cells. This IL6 binds to membrane-bound IL6-receptor (IL6R) on target cells, and the complex then binds to a homodimer of glycoprotein 130 (gp130), which mediates further downstream signaling.48 In addition, IL6 can act on cells that do not express IL6R. In this trans-signaling, IL6 binds to a soluble form of IL6R and the complex binds to membrane-bound gp130 on target cells to activate downstream signaling. Initially, a role for IL6 in cancer development was suggested by in vitro studies of growth-promoting effects on colon cancer cell lines.49, 50 Because these cells do not express membrane-bound IL6R, it was argued that IL6 signaling might act through trans-signaling.50 Evidence for this concept was provided by Becker et al,51 who showed that not only IL6R antibodies, but also sgp130Fc, a designer protein that specifically blocks trans-signaling, limited tumor development in the AOM-DSS model. Similar data recently were provided by Matsumoto et al52 and Grivennikov et al,53 who also found increased activation of signal transducer and activator of transcription 3 (STAT3) in tumor cells, suggesting that this transcription factor contributes to increased tumor cell proliferation after IL6R and Janus kinase activation. STAT3 induces target genes that are important for tumor cell survival, proliferation, angiogenesis, metastasis, cell adhesion, and inflammation.54 The functional importance of STAT3 was emphasized by data from Bollrath et al,55 who showed reduced tumor growth in mice with a conditional deletion of STAT3 in intestinal epithelial cells. Conversely, increased STAT3 activity in gp130Y757F mice promoted tumor growth in the AOM-DSS model.55 In that model, STAT3 activation resulted in hyperproliferation of intestinal epithelial cells caused by direct effects on regulators of G1 and G2/M transition. Recent data from Brighenti et al56 also suggested that p53 down-regulation was induced by IL6 via increased proteosomal degradation. Rokavec et al57 recently studied mice deficient in miR-34a and found that they developed increased numbers of tumors with increased STAT3 phosphorylation and invasive tumor growth partners in the AOM-DSS model. This was caused by a feedback loop, in which miR-34a inhibits IL6R expression and STAT3 inhibits miR-34 expression. STAT3 activation also is necessary for persistent NF-κB activation in tumor cells.58 This process is mediated by STAT3-dependent nuclear retention of RelA, which further amplifies sustained transcription effects of STAT3 and NF-κB, which are required for malignant transformation.
A growing number of cytokines beyond IL6 now are being recognized as activators of STAT3. For instance, a recent study proposed a dominant role for IL11 in STAT3 activation, relative to IL6 in the AOM-DSS model, because IL11-receptor a1–deficient mice developed fewer tumors than IL6-deficient mice.59 Furthermore, autocrine vascular endothelial growth factor signaling in intestinal epithelial cells can promote STAT3 activation in AOM-DSS–treated mice.60 Although the relative importance of the individual mechanisms leading to STAT3 activation remains to be defined, these preclinical data suggest that STAT3 may be a promising therapeutic target for prevention of CAC (Figure 2).
Figure 2.
IL6- and IL11-dependent STAT3 activation promotes tumor proliferation and survival. IL6 and IL11 released from immune cells such as macrophages and effector T cells bind to their specific receptors and induce STAT3 activation via gp130. Phosphorylated STAT3 promotes survival and proliferation of intestinal epithelial cells via direct effects on cell-cycle regulators (CC) and down-regulation of p53. Furthermore, activated STAT3 promotes sustained NF-κB activity. IL11R, IL11 receptor; sIL6R, soluble IL6 receptor. IEC, intestinal epithelial cell.
Tumor-Promoting Effects of T-Helper 17 Cells
In addition to IL6 and TNF, proinflammatory cytokines released by T-helper 17 (Th17) cells, which are important for the adaptive immune response in IBD, have been implicated in the pathogenesis of CAC. Th17 cells are a heterogenous population of effector T cells characterized by up-regulation of the transcription factor RAR-related orphan receptor γT and release of proinflammatory cytokines including IL17A, IL17F, IL21, and IL22.61 A role of Th17 cells in tumor development initially was suggested by Langowski et al,62 who showed that deficiency of IL23, a member of the IL12 family of cytokines that contributes to the differentiation of Th17 cells, led to increased tumor resistance and decreased tissue inflammation in a cutaneous tumor model. Consistent with this, anti-IL17A antibody treatment of mice exposed to 1,2-dimethylhydralazine and DSS also reduced tumor development.63 Furthermore, tumor development in the AOM-DSS model was reduced in IL17A-deficient mice.64 Because this reduced tumor development was accompanied by reduced intestinal inflammation with reduced levels of IL6, TNF, and other cytokines, Th17 cells seem to mediate tumor development by providing a proinflammatory microenvironment rather than by direct effects on tumor cells. Similar data are available for the Th17 family cytokine IL21. Stolfi et al65 analyzed IL21-deficient mice in the AOM-DSS model and found reduced tumor number and reduced expression of proinflammatory cytokines, including IL6 relative to wild-type mice. In contrast to the indirect effects of IL17A and IL21, IL22 may signal directly to intestinal epithelial cells. For example, IL22 leads to STAT3 activation in intestinal epithelial cells during DSS-induced colitis and is required for epithelial regeneration.66 Conversely, mice deficient in the IL22 binding protein, which acts as an IL22 scavenger, show increased tumor growth in the AOM-DSS model owing to increased epithelial IL22-dependent STAT3 activation.63 IL22, IL22-receptor a1, and IL23 expression all have been shown to be increased in human UC and colon cancer tissues.67, 68
Overall, these data indicate that Th17 cells provide a proinflammatory microenvironment that activates tumor-promoting pathways. This concept is supported further by data showing that mice with conditional deletion of APC in colonial epithelial cells develop tumors in the distal colon, but that crossing these mice with IL-23-deficient, IL-23-receptor-deficient, or IL17A receptor–deficient mice reduced both tumor growth and expression of proinflammatory cytokines, including IL6, IL17A, and IL22.69 Of note, IL23 signaling was attributed mainly to the activation of myeloid cells through the commensal microflora. Thus, the authors of this article proposed a sequence in which genetic defects lead to increased infiltration of tumor tissue with microbial products and subsequent development of an IL23/IL17-dependent inflammatory microenvironment that leads to tumor promotion and progression. Although these data were generated in a model of sporadic CRC, studies of the role of microbiota and innate immune pathways in CAC suggest that the conclusions are relevant to CAC.
Innate Immunity and the Microbiome in CAC
Among the studies discussed earlier, Grivennikov et al69 attributed intestinal barrier loss to genetic alterations occurring during sporadic CRC development. This may, however, be relevant to CAC because increased intestinal permeability has been linked to IBD pathogenesis.70, 71, 72, 73 Although the mechanisms leading to intestinal barrier loss and the relationship between this, host–microbial interactions, and chronic innate and adaptive immune responses are not completely understood, barrier dysfunction also may be important to the development of CAC. For example, AOM administration to IL10-deficient mice, which spontaneously develop colitis, leads to tumor development under normal conditions, but neither colitis nor tumorigenesis occur in IL10-deficient mice housed under germ-free conditions.74 Although some bacteria such as Helicobacter hepaticus or Escherichia coli NC101 have been associated with CAC development, a clear association of specific microbiota with human CAC has not been identified.75, 76
Although the factors leading to barrier defects in IBD still are unknown, the resulting influx of microbial products can activate innate immunity, several pathways of which have been implicated in CAC development. Among these, the activation of Toll-like receptors (TLRs) recently has gained attention because these receptors enable the detection of pathogenic molecules and activate downstream pathways to initiate a host immune response. Recent studies particularly have implicated TLR4 signaling in the pathogenesis of CAC. TLR4 is expressed by intestinal epithelial cells and various cells of the immune system, and can be activated by LPS.77 Fukata et al78 showed that TLR4 is overexpressed in CAC tissue of UC patients. Furthermore, TLR4-deficient mice were protected against CAC development in the AOM-DSS model and had reduced myeloid cell infiltration and cyclooxygenase-2 expression.78 These effects primarily were owing to TLR4 signaling on colonic epithelial cells, rather than myeloid cells, as shown in bone marrow chimera experiments.79 In contrast, mice deficient in TLR2 showed increased tumor development in the AOM-DSS model.80 These effects have been attributed to a dysregulated immune response after exposure to AOM-DSS rather than direct effects on epithelial cells because TLR2-deficient mice had increased IL6 and IL17A expression.
Because signaling by TLR2 and TLR4 both are mediated by the myeloid differentiation primary response gene 88 (Myd88), there is currently no explanation for the differences in tumor growth between TLR2- and TLR4-deficient mice. The situation is complicated further by the fact that Myd88 either promotes or reduces tumor growth, depending on the CAC models used. For example, Myd88-deficient mice were more susceptible to AOM-DSS–induced tumors, similar to the phenotype TLR2-deficient, but not TLR4-deficient, mice.81 In contrast, Myd88 function is required for inflammation-associated tumor development in IL10–deficient mice exposed to AOM and in wild-type mice exposed to AOM and oxazolone.74, 82 The mechanisms for these differences in individual models and by the different TLRs cannot be explained at a molecular level. The conflicting data could be attributed to differences in microbiota of mice with innate immune defects.83
Similar to TLRs, nucleotide-binding oligomerization domain (NOD)- and leucine-rich repeat–containing proteins (NLRs) have been implicated in the pathogenesis of IBD and CAC. For example, mutations of the CARD15 gene encoding NOD2 have been associated with Crohn’s disease.73, 84 NOD2 deficiency leads to dysbiosis with increased intestinal inflammation that promotes tumor development after AOM injection.85 CAC risk was transmitted by transferring fecal microbiota from NOD2-deficient mice to wild-type mice, suggesting that NOD2 effects were caused primarily by effects on microbial communities.
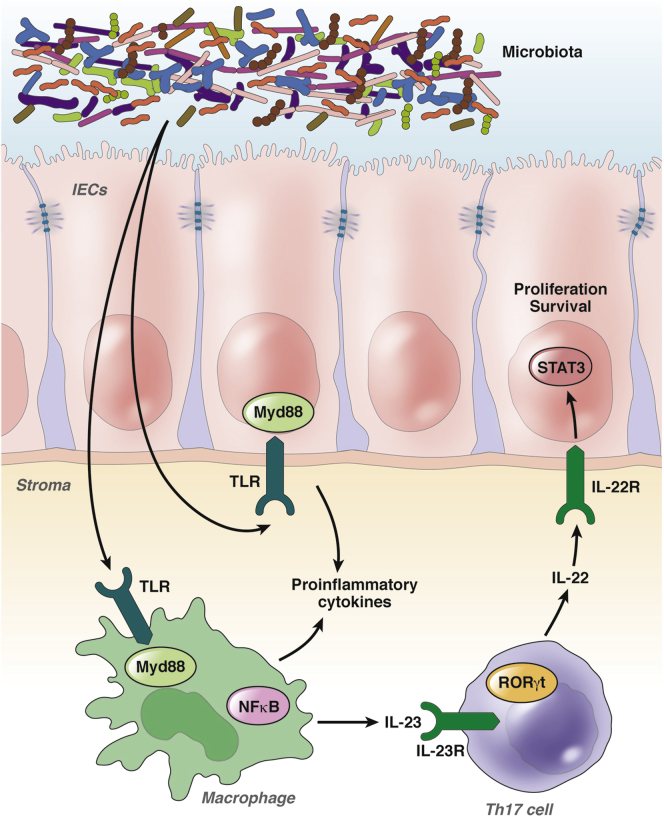
Another downstream mechanism after NLR activation is activation of the inflammasome, a multiprotein complex containing NLR family, pyrin domain-containing (NLRP) proteins NLRC4 and NAIP.70 This complex leads to the activation of caspase-1, which cleaves pro-IL1β and pro-IL18 to facilitate release of these proinflammatory cytokines. Interestingly, mice lacking the NLRP3 component of the inflammasome show more tumor development in the AOM-DSS model, suggesting a protective role for inflammasome activation in CAC.86 Data were similar for NLRP6 and NLRP12.87, 88, 89, 90 Together, the results suggest that the host response to intestinal microbiota can lead to both tumor-promoting or tumor-inhibiting effects (Figure 3). We hope that the growing body of literature regarding these mechanisms will allow future growth in understanding individual contributions of these and other mechanisms to human CAC.
Figure 3.
Microbiota and innate immune mechanisms regulate a proinflammatory microenvironment promoting tumor growth. Possibly as the result of a defective intestinal barrier, microbial products activate innate immune cells or intestinal epithelial cells (IECs), perhaps via TLRs, which then create a proinflammatory microenvironment with subsequent activation of Th17 cells characterized by the transcription factor RAR-related orphan receptor γT (RORγt). Proinflammatory cytokines released by activated innate immune cells, intestinal epithelial cells, or Th17 cells contribute to tumor cell proliferation and survival.
Conclusions
During recent decades, our knowledge of the molecular mechanisms involved in the development of colitis-associated cancer has improved dramatically. In addition to the tumor-initiating effects of oxidative stress, several regulatory pathways, including TNF/NF-κB and IL6/STAT3 signaling, have been identified that are important mediators of tumor promotion and progression. Parallel efforts to determine the pathogenesis of IBD and roles of host–microbial interactions have led to the conclusion that many of these also are modulators of intestinal tumor development (Table 1 shows a summary of the anticipated mechanisms). Despite these advances, not all data acquired in preclinical models can be translated to human disease. For instance, despite the involvement of TNF signaling in CAC models, current clinical data do not show a distinct reduction of CAC risk in IBD patients treated with anti-TNF therapeutics. However, this information might become available when there are more long-term data. Furthermore, it will be important to see how new therapeutic strategies targeting different molecular pathways, such as IL6 or integrins, influence the development of CAC.
Table 1.
Immune Signaling Pathways Involved in the Development of CAC
| Pathway | Molecule | Function | Preclinical data | Evidence in human disease | References |
|---|---|---|---|---|---|
| OS | 8-OH-dG | Marker for OS | Increased 8-OH-dG in inflamed and dysplastic tissue samples of UC patients | 20 | |
| NO | Marker for OS | Increased NO concentrations in active and inactive IBD | 21 | ||
| DDR | γH2A.X | Marker for DDR activation | Increased in IBD tissue | 24 | |
| Aag | Enzyme involved in base excision repair | Aag deficiency: increased number of DNA base lesions and tumors in the AOM-DSS model | 27 | ||
| Nrf2 | Transcription factor involved in the regulation of redox mechanisms | Nrf2 deficiency: Increased number of tumors in the AOM-DSS model | 28 | ||
| Glutathione peroxidase GPX3 | Redox enzyme | GPX3 deficiency: increased tumors in the AOM-DSS model, even tumor development after DSS treatment without AOM | 29 | ||
| TNFα/NF-κB signaling | NF-κB | Proinflammatory transcription factor | Deletion of IKK-β in myeloid cells: reduced tumor size and deletion of IKK-β in intestinal epithelial cells decreased tumor number in AOM-DSS model. Overexpression of constitutively active IKK-β in APC-deficient mice increased tumor development, DNA damage, and DDR |
Increased activity of NF-κB in IBD tissue, no evidence in human CAC | 34, 35, 39, 40 |
| TNF2 | Proinflammatory cytokine involved in NF-κB activation | Anti-TNFα treatment protective in AOM-DSS model, TNFR1 signaling in myeloid cells promotes proinflammatory microenvironment, TNFR2 signaling in intestinal epithelial cells promotes tumor cell survival | Increased TNFα signaling and anti-TNF therapy in IBD patients. No distinct proof for functional role of TNFα in human CAC | 45, 47, 50 | |
| IL6/IL11/STAT3 signaling | IL6 | Proinflammatory cytokine | Mice with deficient IL6 signaling or treatment with anti-IL6 antibodies reduces tumor growth in AOM-DSS model | IL6 promotes growth of human colorectal cancer cell lines | 51, 52, 53, 54, 91 |
| IL11 | Proinflammatory cytokine, member of IL6 family | IL11-receptor a1 deficiency: protection against tumor development in the AOM-DSS model | 60 | ||
| STAT3 | Transcription factor mediating effects of IL6-receptor activation | Conditional deletion of STAT3 in intestinal epithelial cells protects against, whereas constitutive activation of STAT3 promotes tumor development in the AOM-DSS model | 56 | ||
| miR-34 | miR-34 induced by the tumor-suppressor gene p53 | miR-34 deficiency: increased IL6/STAT3 signaling and tumor growth in the AOM-DSS model | 58 | ||
| Th17 cells | IL17A | Th17 effector cytokines | Anti-IL17A antibody–treated and IL17A-deficient mice: protection in AOM-DSS model | 64, 65 | |
| IL-21 | IL21-deficient mice: reduced tumor growth in AOM-DSS model | 66 | |||
| IL-22 | IL22 induces intestinal epithelial cell proliferation via STAT3 activation. IL22 binding protein deficiency promotes tumor growth in the AOM-DSS model | Increased IL22 and IL22-receptor expression in UC and CRC tissue | 67, 68, 92 | ||
| TLR signaling | TLR4 | TLR activated by LPS | TLR4 deficiency: reduced tumor growth in the AOM-DSS model | Overexpressed in human CAC tissue | 79, 80 |
| TLR2 | TLR activated by bacterial cell wall components | TLR2 deficiency: increased tumor development in AOM + DSS model | 81 | ||
| Myd88 | Downstream mediator of TLR activation | Role of Myd88, depending on specific model. Protective effect in AOM-DSS model; tumor-promoting effect in IL10-deficient mice treated with AOM or wild-type mice treated with AOM and oxazolone | 75, 82, 93 | ||
| Inflammasome/NLR family | NLRP3 | Inflammasome components | NLRP3 deficiency: more tumors in the AOM-DSS model | 88 | |
| NLRP6 | NLRP6 deficiency: increased intestinal inflammation and tumor development in AOM-DSS model | 60, 87, 90 | |||
| NLRP12 | NLRP12 deficiency: increased NF-κB signaling and tumor development | 89 |
Aag, alkyladenine DNA glycosylase; 8-OH-dG, 8-oxo-7,8-dihydro-2,-deoxyguanosine; NO, nitric oxide; Nrf2, nuclear factor-erythroid 2–related factor 2; OS, oxidative stress.
Footnotes
Conflicts of interest The authors disclose no conflicts.
Funding Supported by the Clinical Research Group Chronisch entzündliche Darmerkrankungen - Erlangen of the German Research Council (KFO257) (M.J.W. and M.F.N.).
References
- 1.Balkwill F., Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–545. doi: 10.1016/S0140-6736(00)04046-0. [DOI] [PubMed] [Google Scholar]
- 2.Terzic J., Grivennikov S., Karin E. Inflammation and colon cancer. Gastroenterology. 2010;138:2101–2114 e5. doi: 10.1053/j.gastro.2010.01.058. [DOI] [PubMed] [Google Scholar]
- 3.Crohn B., Rosenberg H. The sigmoidoscopic picture of chronic ulcerative colitis (non-specific) Am J Med Sci. 1925;170:220–228. [Google Scholar]
- 4.Morson B.C. Cancer in ulcerative colitis. Gut. 1966;7:425–426. doi: 10.1136/gut.7.5.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rubin D.T., Huo D., Kinnucan J.A. Inflammation is an independent risk factor for colonic neoplasia in patients with ulcerative colitis: a case-control study. Clin Gastroenterol Hepatol. 2013;11:1601–1608. doi: 10.1016/j.cgh.2013.06.023. e1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nieminen U., Jussila A., Nordling S. Inflammation and disease duration have a cumulative effect on the risk of dysplasia and carcinoma in IBD: a case-control observational study based on registry data. Int J Cancer. 2014;134:189–196. doi: 10.1002/ijc.28346. [DOI] [PubMed] [Google Scholar]
- 7.Lakatos P.L., Lakatos L. Risk for colorectal cancer in ulcerative colitis: changes, causes and management strategies. World J Gastroenterol. 2008;14:3937–3947. doi: 10.3748/wjg.14.3937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Eaden J.A., Abrams K.R., Mayberry J.F. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526–535. doi: 10.1136/gut.48.4.526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jess T., Rungoe C., Peyrin-Biroulet L. Risk of colorectal cancer in patients with ulcerative colitis: a meta-analysis of population-based cohort studies. Clin Gastroenterol Hepatol. 2012;10:639–645. doi: 10.1016/j.cgh.2012.01.010. [DOI] [PubMed] [Google Scholar]
- 10.Jones J.H. Colonic cancer and Crohn's disease. Gut. 1969;10:651–654. doi: 10.1136/gut.10.8.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Canavan C., Abrams K.R., Mayberry J. Meta-analysis: colorectal and small bowel cancer risk in patients with Crohn's disease. Aliment Pharmacol Ther. 2006;23:1097–1104. doi: 10.1111/j.1365-2036.2006.02854.x. [DOI] [PubMed] [Google Scholar]
- 12.Philip M., Rowley D.A., Schreiber H. Inflammation as a tumor promoter in cancer induction. Semin Cancer Biol. 2004;14:433–439. doi: 10.1016/j.semcancer.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 13.Fearon E.R., Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61:759–767. doi: 10.1016/0092-8674(90)90186-i. [DOI] [PubMed] [Google Scholar]
- 14.Vogelstein B., Fearon E.R., Hamilton S.R. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319:525–532. doi: 10.1056/NEJM198809013190901. [DOI] [PubMed] [Google Scholar]
- 15.Zisman T.L., Rubin D.T. Colorectal cancer and dysplasia in inflammatory bowel disease. World J Gastroenterol. 2008;14:2662–2669. doi: 10.3748/wjg.14.2662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Waldner M.J., Neurath M.F. Potential avenues for immunotherapy of colitis-associated neoplasia. Immunotherapy. 2012;4:397–405. doi: 10.2217/imt.12.15. [DOI] [PubMed] [Google Scholar]
- 17.Ullman T.A., Itzkowitz S.H. Intestinal inflammation and cancer. Gastroenterology. 2011;140:1807–1816. doi: 10.1053/j.gastro.2011.01.057. [DOI] [PubMed] [Google Scholar]
- 18.Gorrini C., Harris I.S., Mak T.W. Modulation of oxidative stress as an anticancer strategy. Nat Rev Drug Discov. 2013;12:931–947. doi: 10.1038/nrd4002. [DOI] [PubMed] [Google Scholar]
- 19.Jackson A.L., Loeb L.A. The contribution of endogenous sources of DNA damage to the multiple mutations in cancer. Mutat Res. 2001;477:7–21. doi: 10.1016/s0027-5107(01)00091-4. [DOI] [PubMed] [Google Scholar]
- 20.D'Inca R., Cardin R., Benazzato L. Oxidative DNA damage in the mucosa of ulcerative colitis increases with disease duration and dysplasia. Inflamm Bowel Dis. 2004;10:23–27. doi: 10.1097/00054725-200401000-00003. [DOI] [PubMed] [Google Scholar]
- 21.Keshavarzian A., Banan A., Farhadi A. Increases in free radicals and cytoskeletal protein oxidation and nitration in the colon of patients with inflammatory bowel disease. Gut. 2003;52:720–728. doi: 10.1136/gut.52.5.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Curtin N.J. DNA repair dysregulation from cancer driver to therapeutic target. Nat Rev Cancer. 2012;12:801–817. doi: 10.1038/nrc3399. [DOI] [PubMed] [Google Scholar]
- 23.Nardella C., Clohessy J.G., Alimonti A. Pro-senescence therapy for cancer treatment. Nat Rev Cancer. 2011;11:503–511. doi: 10.1038/nrc3057. [DOI] [PubMed] [Google Scholar]
- 24.Sohn J.J., Schetter A.J., Yfantis H.G. Macrophages, nitric oxide and microRNAs are associated with DNA damage response pathway and senescence in inflammatory bowel disease. PLoS One. 2012;7:e44156. doi: 10.1371/journal.pone.0044156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Risques R.A., Lai L.A., Brentnall T.A. Ulcerative colitis is a disease of accelerated colon aging: evidence from telomere attrition and DNA damage. Gastroenterology. 2008;135:410–418. doi: 10.1053/j.gastro.2008.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Risques R.A., Lai L.A., Himmetoglu C. Ulcerative colitis-associated colorectal cancer arises in a field of short telomeres, senescence, and inflammation. Cancer Res. 2011;71:1669–1679. doi: 10.1158/0008-5472.CAN-10-1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Meira L.B., Bugni J.M., Green S.L. DNA damage induced by chronic inflammation contributes to colon carcinogenesis in mice. J Clin Invest. 2008;118:2516–2525. doi: 10.1172/JCI35073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Osburn W.O., Karim B., Dolan P.M. Increased colonic inflammatory injury and formation of aberrant crypt foci in Nrf2-deficient mice upon dextran sulfate treatment. Int J Cancer. 2007;121:1883–1891. doi: 10.1002/ijc.22943. [DOI] [PubMed] [Google Scholar]
- 29.Barrett C.W., Ning W., Chen X. Tumor suppressor function of the plasma glutathione peroxidase gpx3 in colitis-associated carcinoma. Cancer Res. 2013;73:1245–1255. doi: 10.1158/0008-5472.CAN-12-3150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dvorak H.F. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med. 1986;315:1650–1659. doi: 10.1056/NEJM198612253152606. [DOI] [PubMed] [Google Scholar]
- 31.Kuraishy A., Karin M., Grivennikov S.I. Tumor promotion via injury- and death-induced inflammation. Immunity. 2011;35:467–477. doi: 10.1016/j.immuni.2011.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Karin M. Nuclear factor-kappaB in cancer development and progression. Nature. 2006;441:431–436. doi: 10.1038/nature04870. [DOI] [PubMed] [Google Scholar]
- 33.Karin M., Yamamoto Y., Wang Q.M. The IKK NF-kappa B system: a treasure trove for drug development. Nat Rev Drug Discov. 2004;3:17–26. doi: 10.1038/nrd1279. [DOI] [PubMed] [Google Scholar]
- 34.Greten F.R., Eckmann L., Greten T.F. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. 2004;118:285–296. doi: 10.1016/j.cell.2004.07.013. [DOI] [PubMed] [Google Scholar]
- 35.Shaked H., Hofseth L.J., Chumanevich A. Chronic epithelial NF-kappaB activation accelerates APC loss and intestinal tumor initiation through iNOS up-regulation. Proc Natl Acad Sci U S A. 2012;109:14007–14012. doi: 10.1073/pnas.1211509109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cooks T., Pateras I.S., Tarcic O. Mutant p53 prolongs NF-kappaB activation and promotes chronic inflammation and inflammation-associated colorectal cancer. Cancer Cell. 2013;23:634–646. doi: 10.1016/j.ccr.2013.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Collado M., Serrano M. Senescence in tumours: evidence from mice and humans. Nat Rev Cancer. 2010;10:51–57. doi: 10.1038/nrc2772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schwitalla S., Ziegler P.K., Horst D. Loss of p53 in enterocytes generates an inflammatory microenvironment enabling invasion and lymph node metastasis of carcinogen-induced colorectal tumors. Cancer Cell. 2013;23:93–106. doi: 10.1016/j.ccr.2012.11.014. [DOI] [PubMed] [Google Scholar]
- 39.Andresen L., Jorgensen V.L., Perner A. Activation of nuclear factor kappaB in colonic mucosa from patients with collagenous and ulcerative colitis. Gut. 2005;54:503–509. doi: 10.1136/gut.2003.034165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kojima M., Morisaki T., Sasaki N. Increased nuclear factor-kB activation in human colorectal carcinoma and its correlation with tumor progression. Anticancer Res. 2004;24:675–681. [PubMed] [Google Scholar]
- 41.Bradley J.R. TNF-mediated inflammatory disease. J Pathol. 2008;214:149–160. doi: 10.1002/path.2287. [DOI] [PubMed] [Google Scholar]
- 42.Faustman D., Davis M. TNF receptor 2 pathway: drug target for autoimmune diseases. Nat Rev Drug Discov. 2010;9:482–493. doi: 10.1038/nrd3030. [DOI] [PubMed] [Google Scholar]
- 43.Onizawa M., Nagaishi T., Kanai T. Signaling pathway via TNF-alpha/NF-kappaB in intestinal epithelial cells may be directly involved in colitis-associated carcinogenesis. Am J Physiol Gastrointest Liver Physiol. 2009;296:G850–G859. doi: 10.1152/ajpgi.00071.2008. [DOI] [PubMed] [Google Scholar]
- 44.Su L., Nalle S.C., Shen L. TNFR2 activates MLCK-dependent tight junction dysregulation to cause apoptosis-mediated barrier loss and experimental colitis. Gastroenterology. 2013;145:407–415. doi: 10.1053/j.gastro.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Suzuki M., Nagaishi T., Yamazaki M. Myosin light chain kinase expression induced via tumor necrosis factor receptor 2 signaling in the epithelial cells regulates the development of colitis-associated carcinogenesis. PLoS One. 2014;9:e88369. doi: 10.1371/journal.pone.0088369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang F., Schwarz B.T., Graham W.V. IFN-gamma-induced TNFR2 expression is required for TNF-dependent intestinal epithelial barrier dysfunction. Gastroenterology. 2006;131:1153–1163. doi: 10.1053/j.gastro.2006.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Popivanova B.K., Kitamura K., Wu Y. Blocking TNF-alpha in mice reduces colorectal carcinogenesis associated with chronic colitis. J Clin Invest. 2008;118:560–570. doi: 10.1172/JCI32453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rose-John S. IL-6 trans-signaling via the soluble IL-6 receptor: importance for the pro-inflammatory activities of IL-6. Int J Biol Sci. 2012;8:1237–1247. doi: 10.7150/ijbs.4989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Schneider M.R., Hoeflich A., Fischer J.R. Interleukin-6 stimulates clonogenic growth of primary and metastatic human colon carcinoma cells. Cancer Lett. 2000;151:31–38. doi: 10.1016/s0304-3835(99)00401-2. [DOI] [PubMed] [Google Scholar]
- 50.Lahm H., Petral-Malec D., Yilmaz-Ceyhan A. Growth stimulation of a human colorectal carcinoma cell line by interleukin-1 and -6 and antagonistic effects of transforming growth factor beta 1. Eur J Cancer. 1992;28A:1894–1899. doi: 10.1016/0959-8049(92)90031-v. [DOI] [PubMed] [Google Scholar]
- 51.Becker C., Fantini M.C., Schramm C. TGF-beta suppresses tumor progression in colon cancer by inhibition of IL-6 trans-signaling. Immunity. 2004;21:491–501. doi: 10.1016/j.immuni.2004.07.020. [DOI] [PubMed] [Google Scholar]
- 52.Matsumoto S., Hara T., Mitsuyama K. Essential roles of IL-6 trans-signaling in colonic epithelial cells, induced by the IL-6/soluble-IL-6 receptor derived from lamina propria macrophages, on the development of colitis-associated premalignant cancer in a murine model. J Immunol. 2010;184:1543–1551. doi: 10.4049/jimmunol.0801217. [DOI] [PubMed] [Google Scholar]
- 53.Grivennikov S., Karin E., Terzic J. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell. 2009;15:103–113. doi: 10.1016/j.ccr.2009.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Waldner M.J., Neurath M.F. Master regulator of intestinal disease: IL-6 in chronic inflammation and cancer development. Semin Immunol. 2014;26:75–79. doi: 10.1016/j.smim.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 55.Bollrath J., Phesse T.J., von Burstin V.A. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell. 2009;15:91–102. doi: 10.1016/j.ccr.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 56.Brighenti E., Calabrese C., Liguori G. Interleukin 6 downregulates p53 expression and activity by stimulating ribosome biogenesis: a new pathway connecting inflammation to cancer. Oncogene. 2014;33:4396–4406. doi: 10.1038/onc.2014.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rokavec M., Oner M.G., Li H. IL-6R/STAT3/miR-34a feedback loop promotes EMT-mediated colorectal cancer invasion and metastasis. J Clin Invest. 2014;124:1853–1867. doi: 10.1172/JCI73531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lee H., Herrmann A., Deng J.H. Persistently activated Stat3 maintains constitutive NF-kappaB activity in tumors. Cancer Cell. 2009;15:283–293. doi: 10.1016/j.ccr.2009.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Putoczki T.L., Thiem S., Loving A. Interleukin-11 is the dominant IL-6 family cytokine during gastrointestinal tumorigenesis and can be targeted therapeutically. Cancer Cell. 2013;24:257–271. doi: 10.1016/j.ccr.2013.06.017. [DOI] [PubMed] [Google Scholar]
- 60.Waldner M.J., Wirtz S., Jefremow A. VEGF receptor signaling links inflammation and tumorigenesis in colitis-associated cancer. J Exp Med. 2010;207:2855–2868. doi: 10.1084/jem.20100438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ahern P.P., Izcue A., Maloy K.J. The interleukin-23 axis in intestinal inflammation. Immunol Rev. 2008;226:147–159. doi: 10.1111/j.1600-065X.2008.00705.x. [DOI] [PubMed] [Google Scholar]
- 62.Langowski J.L., Zhang X., Wu L. IL-23 promotes tumour incidence and growth. Nature. 2006;442:461–465. doi: 10.1038/nature04808. [DOI] [PubMed] [Google Scholar]
- 63.Qi H., Yang H., Xu G. Therapeutic efficacy of IL-17A antibody injection in preventing the development of colitis associated carcinogenesis in mice. Immunobiology. 2015;220:54–59. doi: 10.1016/j.imbio.2014.09.002. [DOI] [PubMed] [Google Scholar]
- 64.Hyun Y.S., Han D.S., Lee A.R. Role of IL-17A in the development of colitis-associated cancer. Carcinogenesis. 2012;33:931–936. doi: 10.1093/carcin/bgs106. [DOI] [PubMed] [Google Scholar]
- 65.Stolfi C., Rizzo A., Franze E. Involvement of interleukin-21 in the regulation of colitis-associated colon cancer. J Exp Med. 2011;208:2279–2290. doi: 10.1084/jem.20111106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pickert G., Neufert C., Leppkes M. STAT3 links IL-22 signaling in intestinal epithelial cells to mucosal wound healing. J Exp Med. 2009;206:1465–1472. doi: 10.1084/jem.20082683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huber S., Gagliani N., Zenewicz L.A. IL-22BP is regulated by the inflammasome and modulates tumorigenesis in the intestine. Nature. 2012;491:259–263. doi: 10.1038/nature11535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jiang R., Wang H., Deng L. IL-22 is related to development of human colon cancer by activation of STAT3. BMC Cancer. 2013;13:59. doi: 10.1186/1471-2407-13-59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Grivennikov S.I., Wang K., Mucida D. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17-mediated tumour growth. Nature. 2012;491:254–258. doi: 10.1038/nature11465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Saleh M., Trinchieri G. Innate immune mechanisms of colitis and colitis-associated colorectal cancer. Nat Rev Immunol. 2011;11:9–20. doi: 10.1038/nri2891. [DOI] [PubMed] [Google Scholar]
- 71.Marchiando A.M., Graham W.V., Turner J.R. Epithelial barriers in homeostasis and disease. Annu Rev Pathol. 2010;5:119–144. doi: 10.1146/annurev.pathol.4.110807.092135. [DOI] [PubMed] [Google Scholar]
- 72.Su L., Shen L., Clayburgh D.R. Targeted epithelial tight junction dysfunction causes immune activation and contributes to development of experimental colitis. Gastroenterology. 2009;136:551–563. doi: 10.1053/j.gastro.2008.10.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Buhner S., Buning C., Genschel J. Genetic basis for increased intestinal permeability in families with Crohn's disease: role of CARD15 3020insC mutation? Gut. 2006;55:342–347. doi: 10.1136/gut.2005.065557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Uronis J.M., Muhlbauer M., Herfarth H.H. Modulation of the intestinal microbiota alters colitis-associated colorectal cancer susceptibility. PLoS One. 2009;4:e6026. doi: 10.1371/journal.pone.0006026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Arthur J.C., Perez-Chanona E., Muhlbauer M. Intestinal inflammation targets cancer-inducing activity of the microbiota. Science. 2012;338:120–123. doi: 10.1126/science.1224820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nagamine C.M., Rogers A.B., Fox J.G. Helicobacter hepaticus promotes azoxymethane-initiated colon tumorigenesis in BALB/c-IL10-deficient mice. Int J Cancer. 2008;122:832–838. doi: 10.1002/ijc.23175. [DOI] [PubMed] [Google Scholar]
- 77.Kamdar K., Nguyen V., DePaolo R.W. Toll-like receptor signaling and regulation of intestinal immunity. Virulence. 2013;4:207–212. doi: 10.4161/viru.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Fukata M., Chen A., Vamadevan A.S. Toll-like receptor-4 promotes the development of colitis-associated colorectal tumors. Gastroenterology. 2007;133:1869–1881. doi: 10.1053/j.gastro.2007.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Fukata M., Hernandez Y., Conduah D. Innate immune signaling by Toll-like receptor-4 (TLR4) shapes the inflammatory microenvironment in colitis-associated tumors. Inflamm Bowel Dis. 2009;15:997–1006. doi: 10.1002/ibd.20880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lowe E.L., Crother T.R., Rabizadeh S. Toll-like receptor 2 signaling protects mice from tumor development in a mouse model of colitis-induced cancer. PLoS One. 2010;5:e13027. doi: 10.1371/journal.pone.0013027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Salcedo R., Worschech A., Cardone M. MyD88-mediated signaling prevents development of adenocarcinomas of the colon: role of interleukin 18. J Exp Med. 2010;207:1625–1636. doi: 10.1084/jem.20100199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Schiechl G., Bauer B., Fuss I. Tumor development in murine ulcerative colitis depends on MyD88 signaling of colonic F4/80+CD11b(high)Gr1(low) macrophages. J Clin Invest. 2011;121:1692–1708. doi: 10.1172/JCI42540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Elinav E., Henao-Mejia J., Flavell R.A. Integrative inflammasome activity in the regulation of intestinal mucosal immune responses. Mucosal Immunol. 2013;6:4–13. doi: 10.1038/mi.2012.115. [DOI] [PubMed] [Google Scholar]
- 84.Lees C.W., Barrett J.C., Parkes M. New IBD genetics: common pathways with other diseases. Gut. 2011;60:1739–1753. doi: 10.1136/gut.2009.199679. [DOI] [PubMed] [Google Scholar]
- 85.Couturier-Maillard A., Secher T., Rehman A. NOD2-mediated dysbiosis predisposes mice to transmissible colitis and colorectal cancer. J Clin Invest. 2013;123:700–711. doi: 10.1172/JCI62236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Allen I.C., TeKippe E.M., Woodford R.M. The NLRP3 inflammasome functions as a negative regulator of tumorigenesis during colitis-associated cancer. J Exp Med. 2010;207:1045–1056. doi: 10.1084/jem.20100050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Allen I.C., Wilson J.E., Schneider M. NLRP12 suppresses colon inflammation and tumorigenesis through the negative regulation of noncanonical NF-kappaB signaling. Immunity. 2012;36:742–754. doi: 10.1016/j.immuni.2012.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hu B., Elinav E., Huber S. Microbiota-induced activation of epithelial IL-6 signaling links inflammasome-driven inflammation with transmissible cancer. Proc Natl Acad Sci U S A. 2013;110:9862–9867. doi: 10.1073/pnas.1307575110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen G.Y., Liu M., Wang F. A functional role for Nlrp6 in intestinal inflammation and tumorigenesis. J Immunol. 2011;186:7187–7194. doi: 10.4049/jimmunol.1100412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Normand S., Delanoye-Crespin A., Bressenot A. Nod-like receptor pyrin domain-containing protein 6 (NLRP6) controls epithelial self-renewal and colorectal carcinogenesis upon injury. Proc Natl Acad Sci U S A. 2011;108:9601–9606. doi: 10.1073/pnas.1100981108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gerlach K., Daniel C., Lehr H.A. Transcription factor NFATc2 controls the emergence of colon cancer associated with IL-6-dependent colitis. Cancer Res. 2012;72:4340–4350. doi: 10.1158/0008-5472.CAN-11-4155. [DOI] [PubMed] [Google Scholar]
- 92.Grivennikov S.I., Wang K., Mucida D. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17-mediated tumour growth. Nature. 2012;491:254–258. doi: 10.1038/nature11465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Elinav E., Nowarski R., Thaiss C.A. Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat Rev Cancer. 2013;13:759–771. doi: 10.1038/nrc3611. [DOI] [PubMed] [Google Scholar]