Abstract
Klebsiella pneumoniae is considered an urgent health concern due to the emergence of multi-drug-resistant strains for which vaccination offers a potential remedy. Vaccines based on surface polysaccharides are highly promising but need to address the high diversity of surface-exposed polysaccharides, synthesized as O-antigens (lipopolysaccharide, LPS) and K-antigens (capsule polysaccharide, CPS), present in K. pneumoniae. We present a comprehensive and clinically relevant study of the diversity of O- and K-antigen biosynthesis gene clusters across a global collection of over 500 K. pneumoniae whole-genome sequences and the seroepidemiology of human isolates from different infection types. Our study defines the genetic diversity of O- and K-antigen biosynthesis cluster sequences across this collection, identifying sequences for known serotypes as well as identifying novel LPS and CPS gene clusters found in circulating contemporary isolates. Serotypes O1, O2 and O3 were most prevalent in our sample set, accounting for approximately 80 % of all infections. In contrast, K serotypes showed an order of magnitude higher diversity and differ among infection types. In addition we investigated a potential association of O or K serotypes with phylogenetic lineage, infection type and the presence of known virulence genes. K1 and K2 serotypes, which are associated with hypervirulent K. pneumoniae, were associated with a higher abundance of virulence genes and more diverse O serotypes compared to other common K serotypes.
Keywords: Klebsiella pneumoniae, seroepidemiology, surface polysaccharide, K antigen and O antigen, vaccine target
Data summary
Supplementary dataset S1 lists the ENA accession numbers for the 573 publicly available K. pneumoniae whole-genome Illumina read sets analysed in this study (http://www.ebi.ac.uk/ena).
Representative rfb and cps locus sequences have been deposited in GenBank, see Table S2 for the list of GenBank accession numbers (http://www.ncbi.nlm.nih.gov/genbank/).
Impact statement
Vaccines offer a potential remedy against the increasing threat of pan-drug-resistant Klebsiella pneumoniae strains. However the high diversity of surface antigens poses a challenge for vaccine design. This work is the first, to our knowledge, to catalogue naturally occurring polysaccharide antigen biosynthesis gene clusters in a globally representative collection of K. pneumoniae isolates, enabling us to identify novel serotypes and perform an epidemiological analysis. We show that only three O-antigen serotypes account for the majority of infections, offering a promising target for vaccine design.
Introduction
Klebsiella pneumoniae is a leading cause of hospital- and community-acquired infections (including urinary tract infections, pneumonia, bacteraemia and soft tissue infections), primarily afflicting the young and immunocompromised, despite being part of the normal human intestinal microbiota and able to colonize the skin and nasopharynx of healthy individuals (Podschun & Ullmann 1998; Broberg et al. 2014). K. pneumoniae are naturally resistant to antibiotics such as amino-penicillins and carboxy-penicillins. Increasingly, treatment options are diminishing, leaving third-generation cephalosporins and carbapenems as the remaining alternative. However, the emergence of isolates carrying genes encoding extended spectrum beta lactamases (ESBL) and carbapenemases has raised alarm because it removes these last line treatment options and effectively negates the use of a whole class of antibiotics, with few alternatives. Therefore multiple agencies including the WHO, US Centers for Disease Control and Prevention and the UK Department of Health singled out K. pneumoniae as a global health concern (Boucher et al. 2009; Ahmad et al. 2012).
Historically Klebsiella isolates have been classified into serotypes and tracked using typing antisera. Serotyping is based on the recognition of distinct variations of surface-exposed polysaccharides, namely O-antigens and K-antigens, by specific antibodies, resulting in different O and K serotypes. O-antigens are the outermost part of the lipopolysaccharide (LPS), whereas K-antigens belong to the bacterial capsule polysaccharide (CPS). The number of serotypes has been estimated to be eight for O-antigens and 77 for K-antigens (Orskov, Fife-Asbury 1977; Trautmann et al. 1997; Edwards, Fife 1952; Edmunds 1954).
Multivalent protein-conjugate polysaccharide vaccines have been demonstrated to be highly successful and effective against bacterial pathogens, such as Streptococcus pneumoniae (Center, 2007). In K. pneumoniae, polyvalent vaccines based on the K-antigen have been developed and reached Phase I trials in humans (Edelman et al. 1994; Campbell et al. 1996). However, the high diversity of K-antigens and the confusing seroepidemiology render a vaccine with a broad coverage complex to develop and thus very costly. Compared with other Enterobacteriaceae, such as Escherichia coli [161 defined O serotypes (Iguchi et al. 2015a)] and Shigella flexneri [at least 47 O serotypes (Talukder et al. 2003)], Klebsiella has a surprisingly low number of reported O serotypes which promises a more viable alternative for vaccine development compared with K-antigen-based vaccines (Ahmad et al. 2012). Whole-genome sequence data allows us to verify and assess the diversity of these clusters and also estimate their frequency in contemporary isolates to assess the contribution of isolates bearing these markers.
The O-antigen biosynthesis enzymes are encoded on the rfb locus. To date, seven O-antigen clusters have been defined for K. pneumoniae, associated to serotypes O1, O2, O3, O4, O5, O8 and O12 (Table S1) and the rfb O-antigen biosynthetic pathway is well described (Raetz & Whitfield, 2002; Kalynych et al. 2014; Whitfield & Trent 2014). In essence it is an ABC-transporter-dependent pathway functionally composed of three types of enzymes: those responsible for (i) biosynthesis of nucleotide-activated sugars, (ii) polysaccharide repeat-unit synthesis and (iii) assembly of the repeat units and transport across the membrane (flippases).
Both the O1 and O2 antigen polysaccharide chains are based on a repeat-unit designated as d-galactan I. The difference being that the O1 antigen is capped by a distal d-galactan II unit, whereas O2 is not (Vinogradov et al. 2002; Kol et al. 1992). d-galactan II is the only known O-antigen polysaccharide in Klebsiella for which biosynthesis is enabled by genes (wbbY and wbbZ) unlinked to the rfb cluster (Hsieh et al. 2014). The O1/O2 biosynthetic cluster occurs in two variants (Fig. 1a). Variant 1 consists of the transporter genes (wzm and wzt), three glycosyltransferases responsible for repeat-unit synthesis and an UDP-galactopyranose mutase, responsible for the synthesis of the polysaccharide subunit d-galactan I (Clarke & Whitfield 1992). Variant 2 is extended and carries three additional putative glycosyltransferases encoded by gmlABC. These additional genes are thought to explain the existence of a recently defined O2 subtype whereby the gml gene products modify d-galactan I, to give the chemically and antigenically distinct d-galactan III explaining the different subtype (Kelly et al. 1995; Szijártó et al. 2015). The serology of O1 isolates is proposed to be unchanged by GmlABC, because in this instance d-galactan II (encoded by WbbY and WbbZ) is the dominant antigenic epitope (Szijártó et al. 2015).
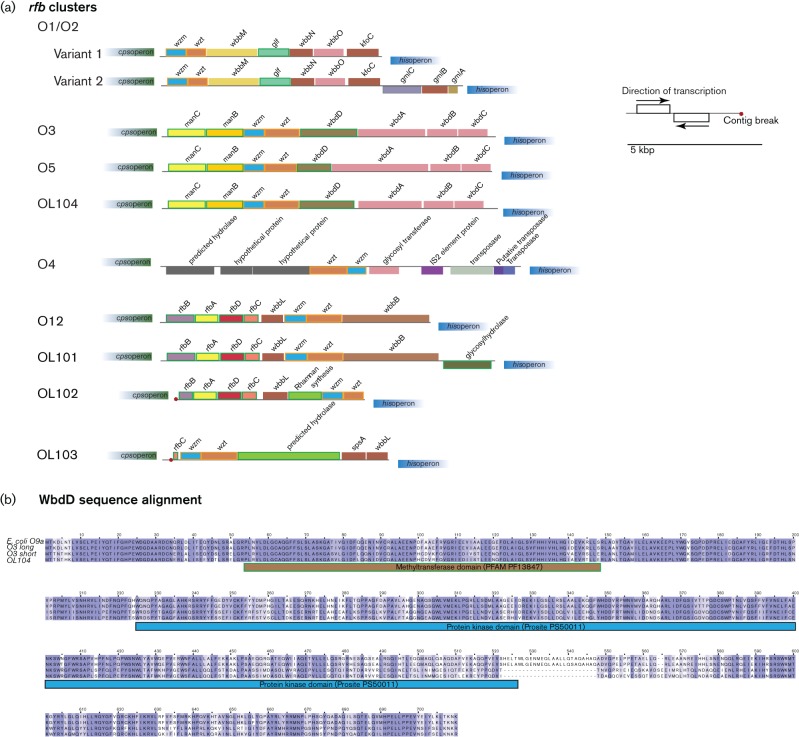
Fig. 1.
(a) rfb gene clusters (see Fig. S1 , available in the online Supplementary Material for full list), genes are colored according to function (see Fig. S1) and above or below the line according to the coding strand and the direction of transcription (black arrows). (b) WbdD sequence alignment. Comparison of E. coli 09a (Uniprot accession number Q47592) and one representative each for the two types of K. pneumoniae O3 WbdD (O3 long (isolate 5151_3#1) and O3 short (9878_1#12)) and OL104 wbdD (5193_7#2). The alignment was colored according to the BLOSUM62 score. Domain detection was performed using InterProScan5 (Jones et al. 2014) based on the E. coli WbdD sequence.
O-antigen polysaccharide structure and rfb sequence of K. pneumoniae serotypes O3 and O5 are identical to those of E. coli serotypes O9a and O8, respectively (Vinogradov et al. 2002; Iguchi et al. 2015b). Biosynthesis enzymes of O3 and O5 differ in the sequence of their mannosyltransferase (WbdA) and methyltransferase (WbdD). WbdD, in complex with WbdA, regulates the mannose chain length by capping the growing chain with a phosphate and methyl group in O3 and a methyl group only in O5 (Hagelueken et al. 2012, 2015).
For completeness the O8 polymer is identical to the O1 antigen, with the exception of the partial O-acetylation of the d-galactan I repeat unit in O8 (Kelly et al. 1993) and serotype O9 may be considered as a subtype of serogroup O2 (Hansen et al. 1999; Trautmann et al. 1997; Kelly & Whitfield 1996). Serotype O9 Wzm and Wzt sequences share >95 % identity to the O8 Wzm and Wzt orthologues (Fang et al. 2015)
Whilst most members of the Enterobacteriaceae have one conserved LPS core, two types of outer cores have been reported for K. pneumoniae; they differ in a set of three genes within the waa operon (Regué et al. 2005), which is independently located in the genome.
The seroepidemiology of K. pneumoniae has previously shown that for human-host-associated isolates the most prevalent O serotypes are O1, O2 and O3, with O1 being dominant in human disease (Trautmann et al. 1997; Hansen et al. 1999; Yu et al. 2007; Fang et al. 2015). The observed prevalence of O4, O5 and O12 differs among the studies, whereas the significance of serotypes O8 and O11 remain equivocal (Trautmann et al. 1997). For O11 this is due to the lack of available rfb sequences and publicly available O11 isolates, so they could not be considered in this study. In addition, little is known about the association between O serotype and disease presentation, the only exception being the observation that serotype O3 isolates are more commonly isolated from blood samples compared with urine samples (Trautmann et al. 1997; Hansen et al. 1999).
The K-antigen biosynthesis enzymes are encoded on the cps (capsule polysaccharide synthesis) locus. To date the cps gene clusters of the 77 serologically defined K-types and nine additional distinct cps operons have been identified and published (Pan et al. 2015; Chung The et al. 2015; Wyres et al. 2015). The biosynthesis pathway is a Wzy-dependent polymerization pathway, identical to E. coli Group 1 capsule synthesis (Whitfield 2006; Larue et al. 2009; Shu et al. 2009; Bushell et al. 2013).
There is some confusion in the literature regarding the prevalence of K serotypes and their association with disease outcome (Cryz et al. 1986; Fung et al. 2000, 2002; Yu et al. 2007). This may be explained by the higher diversity of the K serotypes and possible cross-reactivity, making them much harder to define. In some studies K1 and K2 showed the highest prevalence and were associated with poorer disease outcome (Fung et al. 2000, 2002; Yu et al. 2007). In addition, these K serotypes have been associated with the emergence of hypervirulent K. pneumoniae causing community-acquired invasive infections such as pyogenic liver abscess, which has become of particular concern in parts of Asia (Shon et al. 2013). Hypervirulent K1 strains generally belong to the ST23 lineage, while K2 is found in more diverse backgrounds (Struve et al. 2015; Bialek-Davenet et al. 2014). However, in many studies outside of Asia, including an extensive seroepidemiological study of European and North American cases, K1 isolates are rare (Cryz et al. 1986).
Until now our knowledge of the nature and diversity of O- and K- operons and, by proxy, the antigens they encode is based on sequences of a limited number of reference isolates. Recently, the whole-genome sequences of more than 500 isolates collected from environmental samples, plants, mammals and non-human primates, as well as those from asymptomatic human carriage, from cases of invasive disease and from both the clinical and community setting were sequenced and published (Holt et al. 2015; Ellington, 2016; Chung The et al. 2015; Wand et al. 2015) (Table 1). These studies highlighted that the ability to cause invasive infections is not determined by lineage but is associated with the presence of virulence factors such as siderophore systems and the rmpA mucosity factor, although these virulence determinants are overrepresented in lineages associated with the hypervirulence phenotype (Holt et al. 2015; Bialek-Davenet et al. 2014; Struve et al. 2015).
Table 1. Genome data included in this study and references.
| Dataset | Note | Reference |
|---|---|---|
| Global |
289 isolates; Human and environmental isolates, from six countries (Australia, Indonesia, Laos, Singapore, Vietnam, USA), sampled to maximize diversity and exclude members of a clonal outbreak, metadata includes invasiveness, acquisition type and sample site. Invasiveness status of human isolates consists of three types: carriage (isolates not considered to be the cause of an infection), non-invasive (pneumonia, urinary tract infection, wound infection; with no recorded bacteraemia) and invasive (isolated from normally sterile sites: blood, CSF, intra-ocular, pleural, pericardial, joint fluids, deep-seated tissue abscesses). Acquisition type is either community acquisition (isolated within 48 hours of admission to hospital) or hospital acquisition (isolated after 48 hours after admission) |
(Holt et al. 2015) |
| UK Hospital | 162 isolates; Collection from Cambridge University Hospitals NHS Foundation Trust in the UK over a period of seven years, invasive isolates isolated from normally sterile sites. Biased selection for antimicrobial resistance to three or more of six antimicrobial classes (penicillins, amoxacillin-clavulanate, aminoglycosides, fluoroquinolones, trimethoprim and third-generation cephalosporins), metadata includes sample site | (Ellington 2016) |
| Nepal Hospital | 88 isolates; Nepalese hospital outbreak from May to December 2012, consisting mainly of two clonal lineages; randomly selected blood cultures. | (Chung The et al. 2015) |
| Preantibiotic | 34 isolates; Collection of isolates isolated before the widespread use of antibiotics (pre 1949). No additional metadata available. | (Wand et al. 2015) |
Using this comprehensive whole genome dataset we set out to describe the genetic diversity of O-antigen and K-antigen and LPS core biosynthesis gene clusters and classify them by molecular serotyping. Using these data we then set out to determine if there was an association between O-, K- or LPS core types and disease outcome, phylogenetic lineage or the presence of other known virulence-related genes.
Methods
Bacterial isolates.
Publicly available genome data derived from four different K. pneumoniae collections were analyzed in this study, totaling 573 sequenced isolates (Table 1). The global dataset consists of a globally representative collection from six different countries including isolates from different hosts and different infection types (Holt et al. 2015). The UK hospital dataset consists of a collection from the Cambridge University Hospitals NHS Foundation Trust and contains invasive isolates biased towards those with resistance to third-generation cephalosporins collected over a period of seven years (Ellington 2016). The Nepal hospital dataset contains human isolates from a single Nepalese hospital outbreak in 2012 (Chung The et al. 2015). The preantibiotic dataset contains strains isolated before the widespread use of antibiotics (pre 1949) (Wand et al. 2015).
Genome data, generated in the above studies by paired end Illumina sequencing, were sourced from the European Nucleotide Archive (accession numbers are listed in Supplementary dataset S1, available in the online supplementary material). Reads were de novo assembled using Velvet (Zerbino & Birney 2008) and Velvet Optimiser, and the resulting assemblies annotated using Prokka (Seemann 2014), as described previously (Holt et al. 2015).
In silico serotyping.
O serotyping was performed on the basis of the polysaccharide ABC transporters (flippases; Wzm and Wzt). The specificity of transporters to polysaccharide types has been noted previously (Cuthbertson et al. 2007). The protein sequences of Wzm were located and extracted from the assembled and annotated contigs using tblastn (Camacho et al. 2009) and grouped based on sequence clustering using CD-HIT (Fu et al. 2012) with an identity threshold of 95 %. This threshold was empirically derived and shown to be able to differentiate between the previously described rfb clusters (Table S1. The resulting groups were assigned the O serotype of known Wzm sequences (Table S1), which were included in the clustering step. In cases where Wzm was missing, Wzt homology was used for assignment. Any rfb loci which differed from previously described loci in their sequence and gene content were assigned to putative novel O serotypes. Novel rfb loci sequences were given the designation OL[n], etc, (OL=LPS locus, to differentiate those defined on the basis of rfb locus sequences from those defined serologically; n is a numeric identifier beginning from n=100) (Fig. S1, Table S2). The differentiation of O1 from O2 serotypes was based on the presence of WbbY and WbbZ in O1 (NCBI accession number KJ451390, strain NTUH-K2044 (Hsieh et al. 2014)). LPS core types were assigned as described above, using WaaL clustering with an identity threshold of 80 %. The waa gene cluster was extracted by locating WaaC and WabG using tblastn. Several operons contained a contig break or stretches of low sequencing coverage; when no waaL could be identified or did not fit into any of the two main groups, these were marked as unassigned.
Due to the much higher diversity of the K antigen, grouping was performed based on the full-length cps sequence, which was extracted by locating wzi and wzc using tblastn (Camacho et al. 2009) and extending the presumed locus until a gene on the opposite strand appeared. The extracted cps locus DNA sequences were grouped using UCLUST (Edgar 2010) based on a 95 % identity. The groups were assigned to a serotype if they matched to any of the 86 previously described cps locus sequences (Pan et al. 2015; Chung The et al. 2015; Wyres et al. 2015).
Additionally, a database of wzi (Brisse et al. 2013) and wzc (Pan et al. 2013) sequences with known serotypes was used to verify the classifications. Any cps clusters which differed from previously described loci in their sequence and gene content were assigned to putative novel K serotypes. Putative full-length novel clusters were confirmed by generation of alternative assemblies using SPAdes 3.6.1 (Bankevich et al. 2012) and inspection of the resulting assembly graph using Bandage (Wick et al. 2015). Sequences were confirmed as full length if they spanned from the 5′ galF to the 3′ ugd gene. Full-length clusters were given the designation KL[n], etc: KL=capsule locus, to differentiate those defined on the basis of cps locus sequences from those defined serologically; n is a numeric identifier beginning from n=101 (although the first two loci (KL101 and KL102) have already been named KN1 and KN2 in the literature, and thus we will continue to use these designations here). Representative nucleotide sequences of full length cps and rfb clusters were annotated using Prokka (Seemann 2014) followed by manual curation, and deposited in GenBank (Fig. S1, Table S2). Novel wzi alleles were added to the K. pneumoniae whole-genome sequence typing database BIGSdb at the Institut Pasteur (http://bigsdb.web.pasteur.fr/) (Bialek-Davenet et al. 2014).
Seroepidemiology of human K. pneumoniae infections.
To analyze the prevalence of different O and K serotypes in human disease and their putative association with disease outcome, the 216 human-associated isolates of the global collection were used to investigate their seroepidemiology.
Three different infection properties were examined for association between O and K serotypes and LPS core variant: (i) infection site: we focused on isolates sampled from human blood (representing bacteraemia), sputum (pneumonia) and urine (urinal tract infections, UTI), isolates from other sites were excluded from this category, (ii) acquisition type: hospital-acquired isolates had been sampled in patients from 48 h after admission to hospital, community-acquired isolates had been sampled within 48 h of admission to hospital, isolates where no acquisition type was recorded were excluded from this category, (iii) infection status: carriage isolates are intestinal samples without any infection, non-invasive isolates are infections without bacteraemia (such as wound infection, pneumonia, UTI) and invasive isolates are from infections of normally sterile sites (such as blood).
Gene presence.
A database of putative virulence genes was compiled (Table S3) and the presence of these genes was characterized as described previously (Chung The et al. 2015).
Multi locus sequence typing (MLST).
MLST sequence types for all isolates were determined directly from sequence reads using SRST2 (Inouye et al. 2014) to type against the seven-locus MLST scheme (Diancourt et al. 2005). Sequence types (ST) are listed in Supplementary dataset S1.
Comparative gene analysis and phylogenetic trees.
Comparative gene analysis and core genome definition was performed using Roary (Page et al. 2015), using a blastp percentage identity of 95 % and a core definition of 99 %.
Phylogenies were inferred from either a SNP alignment generated by mapping reads to the K. pneumoniae MGH78578 reference sequence (Fig. S2) or from a concatenated alignment of core genes extracted from Roary output (Page et al. 2015) (Fig. 2). Single-gene alignments (Fig. 1b and Fig. S3) were performed using Clustal Omega (Sievers et al. 2011). Phylogenies were inferred from these alignments by running RAxML using a gamma distribution to model site-specific rate variation and 100 bootstrap replicates (Stamatakis 2006).
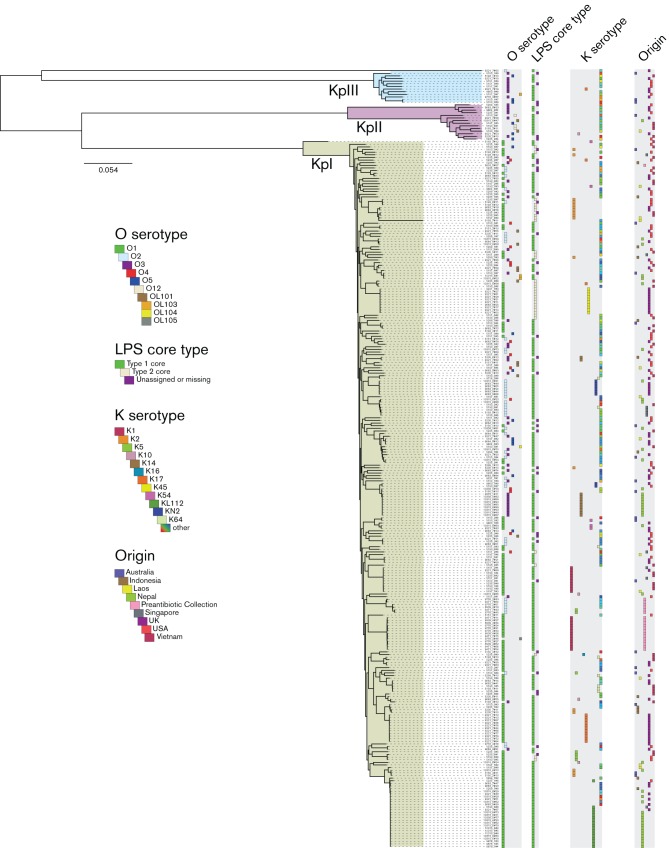
Fig. 2.
Phylogeny of a randomly selected subset of isolates. Lineages are labeled according to the scheme of (Holt et al. 2015), subdividing the K. pneumoniae species into subspecies KpI (K. pneumoniae), KpII (K. quasipneumoniae) and KpIII (K. variicola). O and K serotypes and LPS core types, as well as the geographical origin, are denoted by colored squares as indicated.
Results
Genetic structures of the rfb gene cluster
The rfb gene cluster was located from the genome sequence data and classified on the basis of Wzm and Wzt protein sequence. The rfb cluster was universally located between the cps and his gene clusters in all of our samples (Fig. S1). It was evident from these data that there were rfb operons that did not match any of the known rfb gene clusters (i.e. they did not belong to O1, O2, O3, O4, O5, O8 or O12), either as a result of different gene content or differing nucleotide sequence. These clusters were defined as novel geno-/serotypes. Out of the 573 isolates analyzed in this study, 533 isolates could be assigned to six known O serotypes, 36 isolates to five novel serotypes, and four isolates remain unassigned due to contig breaks or missing sequencing coverage (Supplementary dataset S1).
The clusters themselves range in size from seven to ten genes (8–10 kb). Both O1/O2 variants, O3, O4, O5 and O12, and four novel rfb clusters (OL101, OL102, OL103 and OL104) were observed in our dataset (Fig. 1a). The novel cluster, OL104, is identical in gene repertoire to O3 and O5, both synthesizing a mannose polymer repeated subunit. OL104 possesses the same WbdA as O3, that contains two mannosyltransferase domains and is distinct from the longer O5 WbdA, that contains three mannosyltransferase domains, which have been shown to have functional consequences in the mannosyl linkages they are able to synthesize (Greenfield et al. 2012). Within the isolates designated as serotype O3, two types of WbdD, the chain-length regulator, were observed. One (present in 34 of a total of 86 O3 isolates screened; 40 %) is identical to the E. coli O9a WbdD (Uniprot accession number Q47592), whereas the other type (present in the remaining 52 isolates; 60 %) has a deletion of 25 aa at the C-terminal region of its protein kinase domain. The WbdD of the isolates designated as serotype OL104 shows the same deletion (Fig. 1b). We speculate that OL104 and the two O3 WbdD variants might actually represent serologically distinct subtypes of the O3 serogroup.
The K. pneumoniae O12 antigen is composed of an N-acetylglucosamine and rhamnose polymer (Vinogradov et al. 2002). Based on their gene content and order, O12, OL101 and OL102 are also closely related rfb clusters differing by the presence and absence of single genes: Compared with the O12 cluster, OL101 includes an additional glycosylhydrolase located on the opposite strand, while OL102 lacks the terminal glycosyltransferase gene but possesses an additional rhamnan synthesis gene (Fig. 1a).
It is of note that the direction of transcription in all of the rfb operons run antiparallel to those genes belonging to the adjoining his operon, with exception noted above, the O4 cluster and the extended variant of the O1/O2 O-antigen cluster (O1/O2 Variant 2) (Fig. 1a). The O4 rfb cluster is the only rfb cluster in K. pneumoniae containing a transposase. The O4 antigen is based on a galactose and ribofuranose polymer (Vinogradov et al. 2002).
When assessing the genetic diversity of the waa LPS core biosynthesis operon we detected both of the known core types, but found no conclusive evidence for novel core types.
Genetic structures of the cps gene cluster
The cps gene cluster showed a strikingly higher level of diversity both in sequence and gene content, compared to the rfb clusters (Fig. S1). Clusters ranged in size from 20.5 to 21.6 kb.
Of the 573 isolates analyzed, 387 (68 %) were assigned to 52 known K serotypes by comparison to previously described cps clusters representing the serologically typed reference strains (Pan et al. 2015) (Supplementary dataset S1). Three isolates (0.5 %) where only a partial sequence was available were assigned to a serotype with lower confidence due to only a partial match to the reference sequence. A total of 165 isolates (29 %) for which the cps sequence was >5 % different to those of any of the known cps clusters were putatively assigned to 68 novel serotypes. Eighteen isolates (3 %) remained unassigned because no wzi and wzc sequences could be identified in the genomes. Among the putative novel serotypes, 21 distinct full-length cps sequences were identified (Fig. S1). Five of these sequences matched or were transposase variants of those previously described in (Wyres et al. 2015) and (Chung The et al. 2015), including one which was a transposase-negative variant of the cps cluster from K. pneumoniae HS11286 (genome accession NC_016845.1).
Consistent with results from previous studies, the first gene in all cps clusters was galF (Ebrecht et al. 2015) followed by a putative glucose phosphatase (cpsACP), the JUMPstart sequence (‘just upstream of many polysaccharide starts’) followed by the translocation and surface assembly genes (wzi, wza, wzb and wzc) (Rahn et al., 1999). Although the genes responsible for transport into the periplasm and polymerization (wzx and wzy), UndPP-linkage (wbaP or wcaJ) and a 6-phosphogluconate dehydrogenase [gnd, a housekeeping gene not required for CPS synthesis (Chen et al. 2010)] were universally present, they were not found at fixed locations in these clusters. The cps clusters are usually terminated by UDP-glucose 6-dehydrogenase gene (ugd). The gene encoding glucose dehydratase (rffG) separates the cps and rfb clusters.
The distribution of the rfb and cps gene clusters across K. pneumoniae phylogeny
To understand the distribution of the rfb, cps and waa gene clusters across the K. pneumoniae species we reconstructed a whole-genome-based phylogeny for all isolates included in this study and show their distribution (Fig. 2). This highlights that, with some exceptions, rfb clusters are not restricted to any particular clade. In our dataset the O1 and O2 rfb loci are only found in the KpI sublineage (K. pneumoniae sensu stricto (Holt et al. 2015)), for the other rfb clusters there is extensive evidence of horizontal gene transfer of the different rfb clusters between closely and more distantly related lineages. This is further confirmed by the non-concordance of the phylogeny of the rfb gene clusters and the whole-genome phylogeny [O1 and O2: Fig. S2(a); O3, O5 and Novel 4: Fig. S2(b)].
The two waa LPS core types are equally not restricted to particular clades in the phylogeny, although we note a biased distribution with respect to the O serotypes. Out of the 573 isolates in the full dataset, 70 isolates (12 %) encoded core type 2; these core 2 strains are almost exclusively (65 isolates, 93 %) associated with serotype O1. The remaining isolates are core 1 (446, 78 %) or unassigned (57, 10 %). One previous study (Regué et al. 2005) investigated the distribution of LPS core types in 100 K. pneumoniae strains based on PCR and dot blots and found a ratio of LPS core 1 to LPS core 2 isolates similar to our data (79 : 19), and also noted the association of LPS core 2 with serotype O1 (10 out of 34 O1 strains); however we did not observe an association with K serotype K2 as has been previously reported. Our analysis shows an elevated number of strains carrying both the LPS core type 2 and K2 or K45, however this is likely to be strongly influenced by two clonal expansions within the sample collection we analysed (Fig. 2). Furthermore, we could only observe LPS core type 2 in subspecies KpI (Fig. 2).
Due to the high diversity, K serotype switching is more difficult to analyze. As an example, K1 isolates belong mainly to two lineages, one of which (ST23) is associated with the hypervirulent phenotype, whereas the K2 serotype is much more distributed across the phylogenetic tree, including lineages associated with the hypervirulent phenotype (e.g. ST25) and other non-hypervirulent lineages (e.g. ST14, Fig. 2). This is consistent with previous reports which showed that many K. pneumoniae sequence types include multiple CPS types (Wyres et al. 2015; Holt et al. 2015; Brisse et al. 2009).
O and K Serotype epidemiology
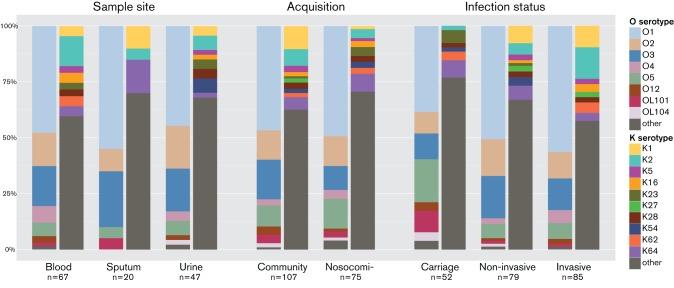
Looking across the full dataset of the 573 isolates the majority were genotyped as O1 (296 isolates, 52 %), followed by O2 (91, 16 %), O3 (86, 15 %), O5 (33, 6 %), OL101 (26, 5 %), O4 (18, 3 %) and O12 (9, 2 %). Each of the OL102 to OL104 serotypes occur in less than 1 % of isolates. The O1/O2 extended rfb cluster variant 2 was observed in 112 (38 %) of O1 genotyped isolates and 52 (57 %) O2 isolates (Supplementary dataset S1).
The seven most common O serotypes described above were associated with 54, 39, 40, 22, 14, 7 and 9 distinct K serotypes respectively, suggesting there is a relationship between the number of representatives of each O serotype in our dataset and the number of distinct K serotypes with which each is associated. This relationship is best explored using only the global isolate set, which is not biased towards any particular K. pneumoniae lineage or isolate source. Within this dataset O1 is the most common O serotype (n=133) and was associated with 43 distinct K serotypes. O3 is the next most common O serotype (n=45), followed by O2 (n=39) and O5 (n=24), these were associated with 29, 30 and 20 distinct K serotypes, respectively. Among the same isolate set the most common K serotype was K2 (n=20), associated with two distinct O serotypes (O1 and O2). K1 (n=15) and K64 (n=14) are the next most common serotypes and are associated with two and four O serotypes respectively. All other K serotypes are represented by eight or fewer isolates in our collection and were each associated with just one or two distinct O serotypes. These data indicate that re-assortment of K and O types occurs frequently in K. pneumoniae.
To investigate the seroepidemiology in human disease, we analyzed the association of O and K serotypes with the three different recorded infection properties (infection site, acquisition type and infection status), using the human-associated isolates of the global collection (Table 2, Fig. 3). Fisher’s exact test was performed to examine whether there was a significant association between serotypes or LPS core types and infection sites, acquisition type or infection status. Significant correlations were found between K2 and invasive infections (odds ratio 4.1, p=0.008), and between OL101 O type and asymptomatic carriage (OR 8.5, p=0.009) (Table 2). Of special interest is also K1, which was virtually restricted to community-acquired isolates (OR 8.4, p=0.016) and completely missing in carriage-associated isolates (p=0.024). No correlation was found for any association of either O or K serotype to infection sites and no correlation of LPS core type with any of the traits as above was observed.
Table 2. Distribution of serotypes and LPS core types in human K. pneumoniae isolates in the global collection (n=216).
The eight most common O-types and ten most common K-types are shown. In the sample site category, samples from other than blood, urine or sputum were excluded. In the acquisition mode category, samples where no acquisition mode was reported were excluded.
| Sample site | Acquisition | Infectiousness | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| O serotype |
Blood n=67 |
Urine n=47 |
Sputum n=20 |
Community n=107 |
Nosocomial n=75 |
Carriage n=52 |
Infection n=79 |
Invasive n=85 |
||||||||
| O1 | 32 | 47.8 % | 21 | 44.7 % | 11 | 55.0 % | 50 | 46.7 % | 37 | 49.3 % | 20 | 38.5 % | 40 | 50.6 % | 48 | 56.5 % |
| O2 | 10 | 14.9 % | 9 | 19.1 % | 2 | 10.0 % | 14 | 13.1 % | 10 | 13.3 % | 5 | 9.6 % | 13 | 16.5 % | 10 | 11.8 % |
| O3 | 12 | 17.9 % | 9 | 19.1 % | 5 | 25.0 % | 19 | 17.8 % | 8 | 10.7 % | 6 | 11.5 % | 15 | 19.0 % | 12 | 14.1 % |
| O4 | 5 | 7.5 % | 2 | 4.3 % | 0 | 0.0 % | 3 | 2.8 % | 3 | 4.0 % | 0 | 0.0 % | 2 | 2.5 % | 5 | 5.9 % |
| O5 | 4 | 6.0 % | 3 | 6.4 % | 1 | 5.0 % | 10 | 9.3 % | 10 | 13.3 % | 10 | 19.2 % | 5 | 6.3 % | 6 | 7.1 % |
| O12 | 2 | 3.0 % | 1 | 2.1 % | 0 | 0.0 % | 4 | 3.7 % | 1 | 1.3 % | 2 | 3.8 % | 1 | 1.3 % | 2 | 2.4 % |
| OL101 | 1 | 1.5 % | 0 | 0.0 % | 1 | 5.0 % | 4 | 3.7 % | 2 | 2.7 % | 5† | 9.6 % | 1 | 1.3 % | 1 | 1.2 % |
| OL104 | 0 | 0.0 % | 1 | 2.1 % | 0 | 0.0 % | 2 | 1.9 % | 1 | 1.3 % | 2 | 3.8 % | 1 | 1.3 % | 0 | 0.0 % |
| Others∗ | 1 | 1.5 % | 1 | 2.1 % | 0 | 0.0 % | 1 | 0.9 % | 3 | 4.0 % | 2 | 3.8 % | 1 | 1.3 % | 1 | 1.2 % |
| K serotype | ||||||||||||||||
| K1 | 3 | 4.5 % | 2 | 4.3 % | 2 | 10.0 % | 11 | 10.3 % | 1 | 1.3 % | 0 | 0.0 % | 6 | 7.6 % | 8 | 9.4 % |
| K2 | 9 | 13.4 % | 3 | 6.4 % | 1 | 5.0 % | 8 | 7.5 % | 3 | 4.0 % | 1 | 1.9 % | 4 | 5.1 % | 12† | 14.1 % |
| K5 | 2 | 3.0 % | 1 | 2.1 % | 0 | 0.0 % | 3 | 2.8 % | 1 | 1.3 % | 0 | 0.0 % | 2 | 2.5 % | 2 | 2.4 % |
| K16 | 3 | 4.5 % | 1 | 2.1 % | 0 | 0.0 % | 2 | 1.9 % | 2 | 2.7 % | 0 | 0.0 % | 1 | 1.3 % | 3 | 3.5 % |
| K23 | 0 | 0.0 % | 0 | 0.0 % | 0 | 0.0 % | 1 | 0.9 % | 3 | 4.0 % | 3 | 5.8 % | 1 | 1.3 % | 0 | 0.0 % |
| K27 | 2 | 3.0 % | 2 | 4.3 % | 0 | 0.0 % | 2 | 1.9 % | 0 | 0.0 % | 0 | 0.0 % | 2 | 2.5 % | 2 | 2.4 % |
| K28 | 2 | 3.0 % | 2 | 4.3 % | 0 | 0.0 % | 3 | 2.8 % | 2 | 2.7 % | 1 | 1.9 % | 2 | 2.5 % | 2 | 2.4 % |
| K54 | 0 | 0.0 % | 3 | 6.4 % | 0 | 0.0 % | 2 | 1.9 % | 2 | 2.7 % | 1 | 1.9 % | 3 | 3.8 % | 0 | 0.0 % |
| K62 | 3 | 4.5 % | 0 | 0.0 % | 0 | 0.0 % | 2 | 1.9 % | 2 | 2.7 % | 2 | 3.8 % | 0 | 0.0 % | 4 | 4.7 % |
| K64 | 3 | 4.5 % | 1 | 2.1 % | 3 | 15.0 % | 6 | 5.6 % | 6 | 8.0 % | 4 | 7.7 % | 5 | 6.3 % | 3 | 3.5 % |
| Others∗ | 40 | 59.7 % | 32 | 68.1 % | 14 | 70.0 % | 67 | 62.6 % | 53 | 70.7 % | 40 | 76.9 % | 53 | 67.1 % | 49 | 57.6 % |
| LPS core type | ||||||||||||||||
| Type 1 | 43 | 64.2 % | 35 | 74.5 % | 12 | 60.0 % | 68 | 63.6 % | 55 | 73.3 % | 37 | 71.2 % | 55 | 69.6 % | 55 | 64.7 % |
| Type 2 | 7 | 10.4 % | 3 | 6.4 % | 2 | 10.0 % | 9 | 8.4 % | 10 | 13.3 % | 7 | 13.5 % | 7 | 8.9 % | 11 | 12.9 % |
| Others∗ | 17 | 25.4 % | 9 | 19.1 % | 6 | 30.0 % | 30 | 28.0 % | 10 | 13.3 % | 8 | 15.4 % | 17 | 21.5 % | 19 | 22.4 % |
∗ Unidentified or not listed † Significant correlation (see text)
Fig. 3.
Distribution of serotypes in human K. pneumoniae isolates in the global collection. The eight most common O-types (left bars) and ten most common K-types (right bars) are shown. Other serotypes are shown in dark grey.
To assess any correlation of serotypes to geographical origins, the dataset was grouped based on acquisition type and infection site in order to remove any spurious relationship. The groups were analyzed as described above; no statistically significant correlation between serotypes or LPS core types and geographical origin could be found. No significant correlation of the presence of O1/O2 rfb cluster extended variant 2 to infection site, infection type or sample site was found.
Association between serotype and virulence gene content
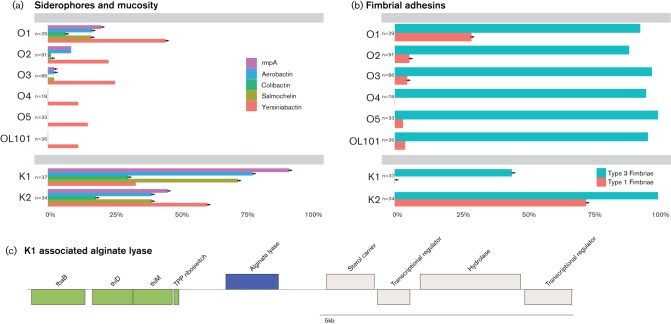
Multiple virulence factors have been defined for Klebsiella (Lawlor et al. 2007; Broberg et al. 2014) including three siderophores (yersiniabactin, salmochelin and aerobactin), the cytotoxin colibactin and rmpA/rmpA2, a regulator of CPS overexpression (Cheng et al. 2010). Fimbrial adhesins are also known to play a major role in biofilm formation and are thus also classified as virulence factors. We determined if the distribution of these known virulence functions was correlated with O- or K-serotype using the existing global data set. The most noteable association was the significant enrichment of an array of siderophore and colibactin clusters and the mucosity regulator rmpA with K serotypes K1 and K2 (Fig. 4a). Since K1 and K2 isolates were almost exclusively also O1 serotypes, the same effect was observed in O1. These results are perhaps unsurprising given that these virulence genes are overrepresented in the hypervirulent K. pneumoniae lineages, which are also associated with serotypes K1 and K2 (Holt et al. 2015; Bialek-Davenet et al. 2014).
Fig. 4.
(a,b) Virulence genes in K. pneumoniae serotypes. Frequency of isolates containing (a) siderophore and mucosity and (b) fimbrial adhesin genes for selected serotypes. →, Significantly overrepresented in this serotype vs all other serotypes (Fisher’s exact test, P-value <0.01). ←, Significantly underrepresented. (c) Genomic context of the K1 associated alginate lyase (blue: alginate lyase, green: thiamine pyrophosphate biosynthesis cluster, grey: other).
Two types of fimbriae are known to be present in K. pneumoniae: type 1 and type 3 fimbriae. Whilst type 3 fimbriae are known to bind to different human cell types, ranging from tracheal epithelia to renal tubular cells, as well as being important for binding to plants and abiotic surfaces (Khater et al. 2015), type 1 fimbriae appear more specific, so far they have been shown to be important for adhesion to uroepithelial cells and also for binding to plants (Klemm, Schembri 2000). Both type 1 and type 3 fimbriae are also associated with biofilm formation in K. pneumoniae (type 3 fimbriae more strongly than type 1 (Schroll et al. 2010; Stahlhut et al. 2012; Klemm, Schembri 2000). Our data showed that serotype O1 isolates had a significantly higher likelihood of possessing type 1 fimbriae compared with any other O serotype (odds ratio 9.7, p=1.1E-16×E-16E-1610–16, Fisher’s exact test) (Fig. 4b). Differences in type 1 and type 3 fimbriae occurrence are very pronounced between K1 and K2: whereas K1 lacks type 1 fimbriae genes (none present, p=0.0019), K2 has a significantly higher occurrence compared with any other K serotype (OR 15.8, p=3.82×10–13); type 3 fimbriae genes are rarer in K1 (OR 0.03, p=6.43×10–17) compared with any other K serotype. The majority of K1 isolates possessing neither type 1 nor type 3 fimbriae (20 isolates without type 1 or type 3 fimbriae out of a total of 35 K1 isolates), stem from a single sequence type (ST82) present in the preantibiotic collection.
To further investigate the fact that K1 occurs almost exclusively in community-acquired isolates and is absent in nosocomial infections, a comparative genomic analysis was performed to search for genes that are overrepresented in K1 compared with other K-serotypes. Strikingly, an alginate lyase isozyme was found to be present in all K1 isolates and virtually absent in all other K serotypes (p=4.82×10–57, Fisher’s exact test). This K1-associated alginate lyase is located just upstream of the thiamine pyrophosphate (TPP) biosynthesis cluster (Fig. 4c). The only other commonly occurring alginate lyase gene in the dataset was present within the K14 cps cluster and has a sequence identity to the K1 associated alginate lyase below 45 %. Alginate lyases enable the use of alginate as carbon and energy source (Wong et al. 2000).
Discussion
The rfb and cps gene clusters give rise to the dominant serotypical properties of K. pneumoniae and are therefore priority vaccine candidates for the treatment of this increasingly multidrug-resistant pathogen. We have catalogued the naturally occurring diversity of these gene clusters within a large collection of isolates taken from different geographic and clinical settings, hosts and disease manifestations. In doing so we identified six known and five novel O-antigen clusters and 45 known and 18 novel complete K-antigen biosynthesis gene clusters. Of particular interest is a previously undescribed rfb cluster (OL101), occurring in 5 % of isolates in our dataset, originating in Europe, Asia and North America and found in human, bovine and environmental samples. We also identified a further 46 putative cps clusters (for which only partial sequences could be obtained) which appear novel. Although this may be an overestimate as some of these may represent transposase variants or divergent forms of other cps types or result from assembly errors; additional work will be required to investigate and validate these further.
This genetic catalogue of rfb and cps clusters enabled in silico serotyping of whole genome sequences. We focused on the seroepidemiology by analyzing the human-associated isolates from the global dataset (Holt et al. 2015), where clinical parameters such as infection site, type and acquisition mode have been recorded. We showed that O serotypes O1, O2 and O3 accounted for 80 % or more of all samples included in this study and that the relative prevalence of these O serotypes was approximately the same for all infection sites, infection types and acquisition modes. This contrasted with the K serotypes, for which an order of magnitude higher diversity was found and no single K serotype dominated this collection.
Notable in the K serotype analysis was the distribution of serotype K1 sequences: most K1 isolates in our study belong to one of two lineages, ST23 and ST82 and showed a strong association with community-acquired infections, consistent with previous reports and the known association of ST23 with the hypervirulent phenotype (Tsay et al. 2002; Yu et al. 2007). K1 isolates were found in six out of seven countries covered by our genome collection. However, despite the fact that K1 is regarded as one of the two most common serotypes it was only found once in 162 isolates collected over a period of seven years in a UK hospital (this study) and is absent in another study based on 703 isolates in 13 hospitals located in Western Europe and Northern America (Cryz et al. 1986). This contrasts with reports from Taiwan, China and South Africa where K1 appears to have been dominant for a considerable period of time (Fung et al. 2000,2002; Yu et al. 2007; Peng et al. 1991; Luo 1990) and has been associated with comparatively higher prevalence of hypervirulent infections in these countries, mostly linked to the ST23 lineage (Bialek-Davenet et al. 2014).
When looking for genes closely associated with K1 we identified the presence of a gene predicted to encode an alginate lyase isozyme. Outside of K1 the only other alginate lyase detected is distantly related and part of the K14 cps cluster. Hence, this gene is almost exclusively found in isolates of the K1 serotype (including those of both major K1 lineages). Pseudomonas aeruginosa is known to produce alginate during biofilm biogenesis in chronic lung infections of cystic fibrosis patients. Alginate lyase enables cell detachment from the biofilm (Boyd & Chakrabarty 1995; Ramsey & Wozniak 2005). However it is not known whether K. pneumoniae produces alginate biofilms. Moreover, since it is unusual to find alginate-lyase-producers, like the K1 K. pneumoniae, that do not use alginate as primary carbon source, it has been proposed that alginate lyase production could be related to coinfection of P. aeruginosa and K. pneumoniae in cystic fibrosis patients (Wong et al. 2000). Given the above it is suggested that the association with cystic fibrosis is important, but probably it is opportunistic, and its role in K. pneumoniae is much broader.
We found no association of LPS core type with the isolates' infection sites, infection types and acquisition modes. This is in contrast to a study showing that LPS core type 2 contributes to the level of virulence in K. pneumoniae, although its mechanism is so far unknown (Regué et al. 2005).
Virulence genes (siderophores, colibactin, rmpA and fimbriae) are expected to be overrepresented in isolates from infections compared with those from carriage or environmental sources and are particularly overrepresented in lineages associated with hypervirulent invasive disease (Holt et al. 2015), thus the significantly high presence in K. pneumoniae serotype O1 isolates was anticipated. However, their significantly lower abundance in isolates of serotype O2 or O3, which are both also prevalent in human infection, was more surprising.
The striking difference in the distribution of the two major fimbriae types found in K. pneumoniae is also of note. Type 3 fimbriae are essential for biofilm formation (Schroll et al. 2010), while type 1 fimbriae are important for adhesion to uroepithelial cells and are thus considered virulence factors for urinary tract infection (Stahlhut et al. 2012). The differential distribution of fimbriae in K. pneumoniae probably allows isolates to adhere to different receptors and perhaps exploit or specialize in different niches, the presence of type 1 fimbriae in K2 isolates and the notable absence of type 1 fimbriae in K1 isolates is intriguing but will require more targeted sampling to unravel its true biological significance.
It is suspected that certain CPS types are able to influence the accessibility of the LPS O-antigen, possibly masking it; the reports are however inconclusive: Hsieh et al. (2012) described the masking of the O1 antigen by K1 capsule but not by K2, whereas Szijártó et al. (2015) showed that the O1 antigen is accessible by antibodies irrespective of the capsule type. The potential masking of O-antigen by the capsule warrants further research and is the crucial next step towards a polysaccharide-based vaccine against K. pneumoniae infection.
Our data indicate that O and K serotypes are frequently re-assorted within the K. pneumoniae population. There are far fewer distinct O serotypes than K serotypes but among the O serotypes, those that are most common are generally associated with a higher number of distinct K serotypes. While there are lineage-specific clustering effects (Fig. 2), these data support the hypothesis that the rfb and/or cps clusters are shuffled within the K. pneumoniae population via horizontal gene transfer. However given the wide diversity of K serotypes, much larger strain collections will need to be examined in order to detect a statistically significant divergence from random re-assortment of CPS and LPS loci.
K. pneumoniae is considered to be a significant threat to human health with the rates of infection increasing globally and appearing to be driven by increasing levels of antimicrobial resistance to front line antimicrobials. The short-term solution has been to turn to old drugs such as colistin that are associated with significant nephrotoxicity. However, the development of resistance to colistin is rapid and explained by both intrinsic mechanisms, such as point mutations (Cheng et al. 2015), as well as by the acquisition of genes by lateral gene transfer (Liu et al. 2015). With this in mind we found that O-serotype prevalence and distribution were stable with regards to different infection types and sites. In addition although O serotype switching has been a common occurrence in the evolution of K. pneumoniae K serotypes show a much bigger variance (Wyres et al. 2015). Consequently O antigens, defined in detail in this study, offer a promising target for vaccine design that warrant further research.
Acknowledgements
We thank Dr Chris Whitfield (University of Guelph, Canada) for the critical review of the manuscript and the helpful comments, Dr Joerg Schneider (LimmaTech Biologics AG, Switzerland) for corrections, and Dr Chi-Tai Fang (National Taiwan University, Taiwan) for providing the O4 and O9 rfb sequences.
Supplementary Data
Supplementary File 1
Supplementary Data
Supplementary File 2
Supplementary Data
Supplementary File 3
Supplementary Data
Supplementary File 4
Supplementary Data
Supplementary File 5
Supplementary Data
Supplementary File 6
Supplementary Data
Supplementary File 7
Abbreviations:
- CPS
capsule polysaccharide
- ESBL
extended spectrum beta lactamases
- MLST
multilocus sequence typing
- TPP
thiamine pyrophosphate
- UndPP
undecaprenyl-pyrophosphate
- LPS
Lipopolysaccharide
References
- Ahmad T. A., El-Sayed L. H., Haroun M., Hussein A. A., El Ashry E. S. H.(2012). Development of immunization trials against Klebsiella pneumoniae. Vaccine 302411–2420. 10.1016/j.vaccine.2011.11.027 [DOI] [PubMed] [Google Scholar]
- Bankevich A., Nurk S., Antipov D., Gurevich A. A., Dvorkin M., Kulikov A. S., Lesin V. M., Nikolenko S. I., Pham S., et al. (2012). SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19455–477. 10.1089/cmb.2012.0021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bialek-Davenet S., Criscuolo A., Ailloud F., Passet V., Jones L., Delannoy-Vieillard A. S., Garin B., Le Hello S., Arlet G., et al. (2014). Genomic definition of hypervirulent and multidrug-resistant Klebsiella pneumoniae clonal groups. Emerg Infect Dis 201812–1820. 10.3201/eid2011.140206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boucher H. W., Talbot G. H., Bradley J. S., Edwards J. E., Gilbert D., Rice L. B., Scheld M., Spellberg B., Bartlett J.(2009). Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Infect Dis 481–12. 10.1086/595011 [DOI] [PubMed] [Google Scholar]
- Boyd A., Chakrabarty A. M.(1995). Pseudomonas aeruginosa biofilms: role of the alginate exopolysaccharide. J Ind Microbiol 15162–168. 10.1007/BF01569821 [DOI] [PubMed] [Google Scholar]
- Brisse S., Fevre C., Passet V., Issenhuth-Jeanjean S., Tournebize R., Diancourt L., Grimont P.(2009). Virulent clones of Klebsiella pneumoniae: identification and evolutionary scenario based on genomic and phenotypic characterization. PLoS One 4e4982. 10.1371/journal.pone.0004982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brisse S., Passet V., Haugaard A. B., Babosan A., Kassis-Chikhani N., Struve C., Decré D.(2013). wzi Gene sequencing, a rapid method for determination of capsular type for Klebsiella strains. J Clin Microbiol 514073–4078. 10.1128/JCM.01924-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broberg C. A., Palacios M., Miller V. L.(2014). Klebsiella: a long way to go towards understanding this enigmatic jet-setter. F1000Prime Rep 6 64. 10.12703/P6-64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bushell S. R., Mainprize I. L., Wear M. A., Lou H., Whitfield C., Naismith J. H.(2013). Wzi is an outer membrane lectin that underpins group 1 capsule assembly in Escherichia coli. Structure 21844–853. 10.1016/j.str.2013.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camacho C., Coulouris G., Avagyan V., Ma N., Papadopoulos J., Bealer K., Madden T. L.(2009). BLAST+: architecture and applications. BMC Bioinformatics 10421. 10.1186/1471-2105-10-421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell W. N., Hendrix E., Cryz S., Cross A. S.(1996). Immunogenicity of a 24-valent Klebsiella capsular polysaccharide vaccine and an eight-valent Pseudomonas O-polysaccharide conjugate vaccine administered to victims of acute trauma. Clin Infect Dis 23179–181. 10.1093/clinids/23.1.179 [DOI] [PubMed] [Google Scholar]
- Center K. J.(2007). Prevenar vaccination: review of the global data, 2006. Vaccine 253085–3089. 10.1016/j.vaccine.2007.01.021 [DOI] [PubMed] [Google Scholar]
- Chen Y. Y., Ko T. P., Chen W. H., Lo L. P., Lin C. H., Wang A. H.(2010). Conformational changes associated with cofactor/substrate binding of 6-phosphogluconate dehydrogenase from Escherichia coli and Klebsiella pneumoniae: Implications for enzyme mechanism. J Struct Biol 16925–35. 10.1016/j.jsb.2009.08.006 [DOI] [PubMed] [Google Scholar]
- Cheng H. Y., Chen Y. S., Wu C. Y., Chang H. Y., Lai Y. C., Peng H. L.(2010). RmpA regulation of capsular polysaccharide biosynthesis in Klebsiella pneumoniae CG43. J Bacteriol 1923144–3158. 10.1128/JB.00031-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y. H., Lin T. L., Pan Y. J., Wang Y. P., Lin Y. T., Wang J. T.(2015). Colistin resistance mechanisms in Klebsiella pneumoniae strains from Taiwan. Antimicrob Agents Chemother 592909–2913. 10.1128/AAC.04763-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung The H., Karkey A., Pham Thanh D., Boinett C. J., Cain A. K., Ellington M., Baker K. S., Dongol S., Thompson C., et al. (2015). A high-resolution genomic analysis of multidrug-resistant hospital outbreaks of Klebsiella pneumoniae. EMBO Mol Med 7227–239. 10.15252/emmm.201404767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke B. R., Whitfield C.(1992). Molecular cloning of the rfb region of Klebsiella pneumoniae serotype O1:K20: the rfb gene cluster is responsible for synthesis of the d-galactan I O polysaccharide. J Bacteriol 1744614–4621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cryz S. J., Mortimer P. M., Mansfield V., Germanier R.(1986). Seroepidemiology of Klebsiella bacteremic isolates and implications for vaccine development. J Clin Microbiol 23687–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuthbertson L., Kimber M. S., Whitfield C.(2007). Substrate binding by a bacterial ABC transporter involved in polysaccharide export. Proc Natl Acad Sci U S A 10419529–19534. 10.1073/pnas.0705709104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diancourt L., Passet V., Verhoef J., Grimont P. A., Brisse S.(2005). Multilocus sequence typing of Klebsiella pneumoniae nosocomial isolates. J Clin Microbiol 434178–4182. 10.1128/JCM.43.8.4178-4182.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebrecht A. C., Orlof A. M., Sasoni N., Figueroa C. M., Iglesias A. A., Ballicora M. A.(2015). On the ancestral UDP-glucose pyrophosphorylase activity of GalF from Escherichia coli. Front Microbiol 61253. 10.3389/fmicb.2015.01253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelman R., Taylor D. N., Wasserman S. S., McClain J. B., Cross A. S., Sadoff J. C., Que J. U., Cryz S. J.(1994). Phase 1 trial of a 24-valent Klebsiella capsular polysaccharide vaccine and an eight-valent Pseudomonas O-polysaccharide conjugate vaccine administered simultaneously. Vaccine 121288–1294. 10.1016/S0264-410X(94)80054-4 [DOI] [PubMed] [Google Scholar]
- Edgar R. C.(2010). Search and clustering orders of magnitude faster than BLAST. Bioinformatics 262460–2461. 10.1093/bioinformatics/btq461 [DOI] [PubMed] [Google Scholar]
- Edmunds P. N.(1954). Further Klebsiella capsule types. J Infect Dis 9465–71. 10.1093/infdis/94.1.65 [DOI] [PubMed] [Google Scholar]
- Edwards P. R., Fife M. A.(1952). Capsule types of Klebsiella. J Infect Dis 9192–104. 10.1093/infdis/91.1.92 [DOI] [PubMed] [Google Scholar]
- Ellington M. J.(2016). Klebsiella pneumoniae collection from Cambridge University Hospitals NHS Foundation Trust. In Manuscript in Preparation. [Google Scholar]
- Fang C. T., Shih Y. J., Cheong C. M., Yi W. C.(2015). Rapid and accurate determination of lipopolysaccharide O-antigen types in Klebsiella pneumoniae with a novel PCR-based O-genotyping method. J Clin Microbiol 54666–675. 10.1128/JCM.02494-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu L., Niu B., Zhu Z., Wu S., Li W.(2012). CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics 283150–3152. 10.1093/bioinformatics/bts565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fung C. P., Hu B. S., Chang F. Y., Lee S. C., Kuo B. I., Ho M., Siu L. K., Liu C. Y.(2000). A 5-year study of the seroepidemiology of Klebsiella pneumoniae: high prevalence of capsular serotype K1 in Taiwan and implication for vaccine efficacy. J Infect Dis 1812075–2079. 10.1086/315488 [DOI] [PubMed] [Google Scholar]
- Fung C. P., Chang F. Y., Lee S. C., Hu B. S., Kuo B. I., Liu C. Y., Ho M., Siu L. K.(2002). A global emerging disease of Klebsiella pneumoniae liver abscess: is serotype K1 an important factor for complicated endophthalmitis? Gut 50420–424. 10.1136/gut.50.3.420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenfield L. K., Richards M. R., Vinogradov E., Wakarchuk W. W., Lowary T. L., Whitfield C.(2012). Domain organization of the polymerizing mannosyltransferases involved in synthesis of the Escherichia coli O8 and O9a lipopolysaccharide O-antigens. J Biol Chem 28738135–38149. 10.1074/jbc.M112.412577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagelueken G., Huang H., Clarke B. R., Lebl T., Whitfield C., Naismith J. H.(2012). Structure of WbdD: a bifunctional kinase and methyltransferase that regulates the chain length of the O antigen in Escherichia coli O9a. Mol Microbiol 86730–742. 10.1111/mmi.12014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagelueken G., Clarke B. R., Huang H., Tuukkanen A., Danciu I., Svergun D. I., Hussain R., Liu H., Whitfield C., et al. (2015). A coiled-coil domain acts as a molecular ruler to regulate O-antigen chain length in lipopolysaccharide. Nat Struct Mol Biol 2250–56. 10.1038/nsmb.2935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen D. S., Mestre F., Alberti S., Hernández-Allés S., Alvarez D., Doménech-Sánchez A., Gil J., Merino S., Tomás J. M., et al. (1999). Klebsiella pneumoniae lipopolysaccharide O typing: revision of prototype strains and O-group distribution among clinical isolates from different sources and countries. J Clin Microbiol 3756–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holt K. E., Wertheim H., Zadoks R. N., Baker S., Whitehouse C. A., Dance D., Jenney A., Connor T. R., Hsu L. Y., et al. (2015). Genomic analysis of diversity, population structure, virulence, and antimicrobial resistance in Klebsiella pneumoniae, an urgent threat to public health. Proc Natl Acad Sci U S A 112E3574–3581. 10.1073/pnas.1501049112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh P. F., Lin T. L., Yang F. L., Wu M. C., Pan Y. J., Wu S. H., Wang J. T.(2012). Lipopolysaccharide O1 antigen contributes to the virulence in Klebsiella pneumoniae causing pyogenic liver abscess. PLoS One 7e33155. 10.1371/journal.pone.0033155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh P. F., Wu M. C., Yang F. L., Chen C. T., Lou T. C., Chen Y. Y., Wu S. H., Sheu J. C., Wang J. T.(2014). d-Galactan II is an immunodominant antigen in O1 lipopolysaccharide and affects virulence in Klebsiella pneumoniae: implication in vaccine design. Front Microbiol 5608. 10.3389/fmicb.2014.00608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iguchi A., Iyoda S., Kikuchi T., Ogura Y., Katsura K., Ohnishi M., Hayashi T., Thomson N. R.(2015a). A complete view of the genetic diversity of the Escherichia coli O-antigen biosynthesis gene cluster. DNA Res 22101–107. 10.1093/dnares/dsu043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iguchi A., Iyoda S., Seto K., Morita-Ishihara T., Scheutz F., Ohnishi M.(2015b). Escherichia coli O-genotyping PCR: a comprehensive and practical platform for molecular O serogrouping. J Clin Microbiol 532427–2432. 10.1128/JCM.00321-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inouye M., Dashnow H., Raven L. A., Schultz M. B., Pope B. J., Tomita T., Zobel J., Holt K. E.(2014). SRST2: Rapid genomic surveillance for public health and hospital microbiology labs. Genome Med 690. 10.1186/s13073-014-0090-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones P., Binns D., Chang H. Y., Fraser M., Li W., McAnulla C., McWilliam H., Maslen J., Mitchell A., et al. (2014). InterProScan 5: genome-scale protein function classification. Bioinformatics 301236–1240. 10.1093/bioinformatics/btu031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalynych S., Morona R., Cygler M.(2014). Progress in understanding the assembly process of bacterial O-antigen. FEMS Microbiol Rev 381048–1065. 10.1111/1574-6976.12070 [DOI] [PubMed] [Google Scholar]
- Kelly R. F., Severn W. B., Richards J. C., Perry M. B., MacLean L. L., Tomás J. M., Merino S., Whitfield C.(1993). Structural variation in the O-specific polysaccharides of Klebsiella pneumoniae serotype O1 and O8 lipopolysaccharide: evidence for clonal diversity in rfb genes. Mol Microbiol 10615–625. 10.1111/j.1365-2958.1993.tb00933.x [DOI] [PubMed] [Google Scholar]
- Kelly R. F., Perry M. B., MacLean L. L., Whitfield C.(1995). Structures of the O-antigens of Klebsiella serotypes O2 (2a,2e), O2 (2a,2e,2h), and O2 (2a,2f,2g), members of a family related d-galactan O-antigens in Klebsiella spp. Journal of Endotoxin Research, 131–140. [Google Scholar]
- Kelly R. F., Whitfield C.(1996). Clonally diverse rfb gene clusters are involved in expression of a family of related d-galactan O antigens in Klebsiella species. J Bacteriol 1785205–5214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khater F., Balestrino D., Charbonnel N., Dufayard J. F., Brisse S., Forestier C.(2015). In silico analysis of usher encoding genes in Klebsiella pneumoniae and characterization of their role in adhesion and colonization. PLoS One 10e0116215. 10.1371/journal.pone.0116215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klemm P., Schembri M. A.(2000). Bacterial adhesins: function and structure. Int J Med Microbiol 29027–35. 10.1016/S1438-4221(00)80102-2 [DOI] [PubMed] [Google Scholar]
- Kol O., Wieruszeski J. M., Strecker G., Fournet B., Zalisz R., Smets P.(1992). Structure of the O-specific polysaccharide chain of Klebsiella pneumoniae O1K2 (NCTC 5055) lipopolysaccharide. A complementary elucidation. Carbohydr Res 236339–344. 10.1016/0008-6215(92)85028-X [DOI] [PubMed] [Google Scholar]
- Larue K., Kimber M. S., Ford R., Whitfield C.(2009). Biochemical and structural analysis of bacterial O-antigen chain length regulator proteins reveals a conserved quaternary structure. J Biol Chem 2847395–7403. 10.1074/jbc.M809068200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawlor M. S., O'connor C., Miller V. L.(2007). Yersiniabactin is a virulence factor for Klebsiella pneumoniae during pulmonary infection. Infect Immun 751463–1472. 10.1128/IAI.00372-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. Y., Wang Y., Walsh T. R., Yi L. X., Zhang R., Spencer J., Doi Y., Tian G., Dong B., et al. (2015). Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16161–168. 10.1016/S1473-3099(15)00424-7 [DOI] [PubMed] [Google Scholar]
- Luo W. T.(1990). Preliminary study on serotyping of Klebsiella pneumoniae and its clinical significance. Zhonghua Jie He He Hu Xi Za Zhi 137325–378. [PubMed] [Google Scholar]
- Orskov I., Fife-Asbury M. A.(1977). New Klebsiella capsular antigen, K82, and the deletion of five of those previously assigned. Int J Syst Evol Microbiol 27386–387. 10.1099/00207713-27-4-386 [DOI] [Google Scholar]
- Page A. J., Cummins C. A., Hunt M., Wong V. K., Reuter S., Holden M. T., Fookes M., Falush D., Keane J. A., et al. (2015). Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics 313691–3693. 10.1093/bioinformatics/btv421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y. J., Lin T. L., Chen Y. H., Hsu C. R., Hsieh P. F., Wu M. C., Wang J. T.(2013). Capsular types of Klebsiella pneumoniae revisited by wzc sequencing. PLoS One 8e80670. 10.1371/journal.pone.0080670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan Y. J., Lin T. L., Chen C. T., Chen Y. Y., Hsieh P. F., Hsu C. R., Wu M. C., Wang J. T.(2015). Genetic analysis of capsular polysaccharide synthesis gene clusters in 79 capsular types of Klebsiella spp. Sci Rep 515573. 10.1038/srep15573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng H. L., Wang P. Y., Wu J. L., Chiu C. T., Chang H. Y.(1991). Molecular epidemiology of Klebsiella pneumoniae. Zhonghua Min Guo Wei Sheng Wu Ji Mian Yi Xue Za Zhi 24264–271. [PubMed] [Google Scholar]
- Podschun R., Ullmann U.(1998). Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11589–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raetz C. R., Whitfield C.(2002). Lipopolysaccharide endotoxins. Annu Rev Biochem 71635–700. 10.1146/annurev.biochem.71.110601.135414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahn A., Drummelsmith J., Whitfield C.(1999). Conserved organization in the cps gene clusters for expression of Escherichia coli group 1 K antigens: relationship to the colanic acid biosynthesis locus and the cps genes from Klebsiella pneumoniae. J Bacteriol 1812307–2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramsey D. M., Wozniak D. J.(2005). Understanding the control of Pseudomonas aeruginosa alginate synthesis and the prospects for management of chronic infections in cystic fibrosis. Mol Microbiol 56309–322. 10.1111/j.1365-2958.2005.04552.x [DOI] [PubMed] [Google Scholar]
- Regué M., Izquierdo L., Fresno S., Piqué N., Corsaro M. M., Naldi T., De Castro C., Waidelich D., Merino S., et al. (2005). A second outer-core region in Klebsiella pneumoniae lipopolysaccharide. J Bacteriol 1874198–4206. 10.1128/JB.187.12.4198-4206.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samuel G., Reeves P.(2003). Biosynthesis of O-antigens: genes and pathways involved in nucleotide sugar precursor synthesis and O-antigen assembly. Carbohydrate Research 3382503–2519. 10.1016/j.carres.2003.07.009 [DOI] [PubMed] [Google Scholar]
- Schroll C., Barken K. B., Krogfelt K. A., Struve C.(2010). Role of type 1 and type 3 fimbriae in Klebsiella pneumoniae biofilm formation. BMC Microbiol 10179. 10.1186/1471-2180-10-179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seemann T.(2014). Prokka: rapid prokaryotic genome annotation. Bioinformatics 302068–2069. 10.1093/bioinformatics/btu153 [DOI] [PubMed] [Google Scholar]
- Shon A. S., Bajwa R. P., Russo T. A.(2013). Hypervirulent (hypermucoviscous) Klebsiella pneumoniae. Virulence 4107–118. 10.4161/viru.22718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shu H. Y., Fung C. P., Liu Y. M., Wu K. M., Chen Y. T., Li L. H., Liu T. T., Kirby R., Tsai S. F.(2009). Genetic diversity of capsular polysaccharide biosynthesis in Klebsiella pneumoniae clinical isolates. Microbiology 1554170–4183. 10.1099/mic.0.029017-0 [DOI] [PubMed] [Google Scholar]
- Sievers F., Wilm A., Dineen D., Gibson T. J., Karplus K., Li W., Lopez R., McWilliam H., Remmert M., et al. (2011). Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol 539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahlhut S. G., Struve C., Krogfelt K. A., Reisner A.(2012). Biofilm formation of Klebsiella pneumoniae on urethral catheters requires either type 1 or type 3 fimbriae. FEMS Immunol Med Microbiol 65350–359. 10.1111/j.1574-695X.2012.00965.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamatakis A.(2006). RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 222688–2690. 10.1093/bioinformatics/btl446 [DOI] [PubMed] [Google Scholar]
- Struve C., Roe C. C., Stegger M., Stahlhut S. G., Hansen D. S., Engelthaler D. M., Andersen P. S., Driebe E. M., Keim P., et al. (2015). Mapping the evolution of hypervirulent Klebsiella pneumoniae. MBio 6e00630-15. 10.1128/mBio.00630-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szijártó V., Guachalla L. M., Hartl K., Varga C., Banerjee P., Stojkovic K., Kaszowska M., Nagy E., Lukasiewicz J., et al. (2015). Both clades of the epidemic KPC-producing Klebsiella pneumoniae clone ST258 share a modified galactan O-antigen type. Int J Med Microbiol 30689–98. 10.1016/j.ijmm.2015.12.002 [DOI] [PubMed] [Google Scholar]
- Talukder K. A., Islam Z., Islam M. A., Dutta D. K., Safa A., Ansaruzzaman M., Faruque A. S., Shahed S. N., Nair G. B., et al. (2003). Phenotypic and genotypic characterization of provisional serotype Shigella flexneri 1c and clonal relationships with 1a and 1b strains isolated in Bangladesh. J Clin Microbiol 41110–117. 10.1128/JCM.41.1.110-117.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trautmann M., Ruhnke M., Rukavina T., Held T. K., Cross A. S., Marre R., Whitfield C.(1997). O-antigen seroepidemiology of Klebsiella clinical isolates and implications for immunoprophylaxis of Klebsiella infections. Clin Diagn Lab Immunol 4550–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsay R. W., Siu L. K., Fung C. P., Chang F. Y.(2002). Characteristics of bacteremia between community-acquired and nosocomial Klebsiella pneumoniae infection: risk factor for mortality and the impact of capsular serotypes as a herald for community-acquired infection. Arch Intern Med 1621021–1027. 10.1001/archinte.162.9.1021 [DOI] [PubMed] [Google Scholar]
- Vinogradov E., Frirdich E., MacLean L. L., Perry M. B., Petersen B. O., Duus Jens Ø., Whitfield C.(2002). Structures of lipopolysaccharides from Klebsiella pneumoniae. Eluicidation of the structure of the linkage region between core and polysaccharide O chain and identification of the residues at the non-reducing termini of the O chains. J Biol Chem 27725070–25081. 10.1074/jbc.M202683200 [DOI] [PubMed] [Google Scholar]
- Wand M. E., Baker K. S., Benthall G., McGregor H., McCowen J. W., Deheer-Graham A., Sutton J. M.(2015). Characterization of pre-antibiotic era Klebsiella pneumoniae isolates with respect to antibiotic/disinfectant susceptibility and virulence in Galleria mellonella. Antimicrob Agents Chemother 593966–3972. 10.1128/AAC.05009-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitfield C.(2006). Biosynthesis and assembly of capsular polysaccharides in Escherichia coli. Annu Rev Biochem 7539–68. 10.1146/annurev.biochem.75.103004.142545 [DOI] [PubMed] [Google Scholar]
- Whitfield C., Trent M. S.(2014). Biosynthesis and export of bacterial lipopolysaccharides. Annu Rev Biochem 8399–128. 10.1146/annurev-biochem-060713-035600 [DOI] [PubMed] [Google Scholar]
- Wick R. R., Schultz M. B., Zobel J., Holt K. E.(2015). Bandage: interactive visualization of de novo genome assemblies. Bioinformatics 313350–3352. 10.1093/bioinformatics/btv383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong T. Y., Preston L. A., Schiller N. L.(2000). Alginate lyase: review of major sources and enzyme characteristics, structure-function analysis, biological roles, and applications. Annu Rev Microbiol 54289–340. 10.1146/annurev.micro.54.1.289 [DOI] [PubMed] [Google Scholar]
- Wyres K. L., Gorrie C., Edwards D. J., Wertheim H. F., Hsu L. Y., Van Kinh N., Zadoks R., Baker S., Holt K. E.(2015). Extensive capsule locus variation and large-scale genomic recombination within the Klebsiella pneumoniae clonal group 258. Genome Biol Evol 71267–1279. 10.1093/gbe/evv062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu V. L., Hansen D. S., Ko W. C., Sagnimeni A., Klugman K. P., von Gottberg A., Goossens H., Wagener M. M., Benedi V. J., International Klebseilla Study Group (2007). Virulence characteristics of Klebsiella and clinical manifestations of K. pneumoniae bloodstream infections. Emerg Infect Dis 13986–993. 10.3201/eid1307.070187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zerbino D. R., Birney E.(2008). Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18821–829. 10.1101/gr.074492.107 [DOI] [PMC free article] [PubMed] [Google Scholar]
Data Bibliography
- 1.Holt Kathryn, E., Wertheim, Heiman., Zadoks Ruth, N., Baker, Stephen., Whitehouse Chris, A., Dance, David., Jenney, A., Connor, T.R., Hsu, L.Y., & other authors Genomic analysis of diversity, population structure, virulence, and antimicrobial resistance in Klebsiella pneumoniae, an urgent threat to public health. Proceedings of the National Academy of Sciences of the United States of America (2015), 112 (27):E3574-81. [DOI] [PMC free article] [PubMed]
- 2.Chung The, H., Karkey, A., Pham Thanh, D., Boinett, C.J., Cain, A.K., Ellington, M., Baker, K.S., Dongol, S., Thompson, C., & other authors A high-resolution genomic analysis of multidrug-resistant hospital outbreaks of Klebsiella pneumoniae. EMBO molecular medicine (2015), 7:227-239. [DOI] [PMC free article] [PubMed]
- Ellington, M.J., Klebsiella pneumoniae collection from Cambridge University Hospitals NHS Foundation Trust. Manuscript in preparation (2016).
- 4.Wand, M.E., Baker, K.S., Benthall, G., McGregor, H., McCowen, J.W., James, W.I., Deheer-Graham, A., Sutton, J.M., Characterization of pre-antibiotic era Klebsiella pneumoniae isolates with respect to antibiotic/disinfectant susceptibility and virulence in Galleria mellonella. Antimicrobial agents and chemotherapy (2015), 59:3966-3972. [DOI] [PMC free article] [PubMed]
- Follador, R., Heinz, E., Wyres, K.L., Ellington, M.J., Kowarik, M., Holt, K.E., Thomson, N.R., GenBank accession numbers LT174532–LT174607 (2016). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary File 1
Supplementary File 2
Supplementary File 3
Supplementary File 4
Supplementary File 5
Supplementary File 6
Supplementary File 7