Abstract
Plant trichomes constitute a first line of defence against insect herbivores. The pre- and post-ingestive defensive functions of glandular trichomes are well documented and include direct toxicity, adhesion, antinutrition and defence gene induction. By contrast, the defensive functions of non-glandular trichomes are less well characterized, although these structures are thought to serve as physical barriers that impede herbivore feeding and movement. We experimentally varied the density of stellate non-glandular trichomes in several ways to explore their pre- and post-ingestive effects on herbivores. Larvae of Manduca sexta (Sphingidae) initiated feeding faster and gained more weight on Solanum carolinense (Solanaceae) leaves having lower trichome densities (or experimentally removed trichomes) than on leaves having higher trichome densities. Adding trichomes to artificial diet also deterred feeding and adversely affected caterpillar growth relative to controls. Scanning electron and light microscopy revealed that the ingestion of stellate trichomes by M. sexta caterpillars caused extensive damage to the peritrophic membrane, a gut lining that is essential to digestion and pathogen isolation. These findings suggest that, in addition to acting as a physical barrier to deter feeding, trichomes can inhibit caterpillar growth and development via post-ingestive effects.
Keywords: trichomes, Manduca sexta, Solanum carolinense, herbivory, plant defence, peritrophic matrix
1. Introduction
Trichomes act as a first line of plant defence that can deter herbivory even prior to the initiation of feeding. For example, glandular trichomes may secrete toxic or sticky substances when disturbed by herbivores [1–3] and can also serve as triggers for the activation of defence signalling pathways [4]. By contrast, non-glandular trichomes are thought to enhance plant defence through primarily physical means such as inhibiting herbivore movement and limiting access to leaf or stem tissues [5], while also contributing to non-defence functions such as UV protection and gas exchange [6]. In horsenettle (Solanaceae: Solanum carolinense), non-glandular trichomes were shown to reduce leafminer damage (Tildenia inconspicuella (Murtfeldt) (Lepidoptera: Gelechiidae) by preventing larvae from penetrating the leaf epidermis and forming feeding mines [7]. Other studies suggest that non-glandular trichomes can reduce the feeding efficiency of a range of insect herbivores [8–10] and prolong the duration of developmental periods during which herbivores are vulnerable to predation and environmental stress [11]. However, the specific mechanisms by which non-glandular trichomes deter herbivory are not always clear, as their effects on herbivores can be difficult to disentangle from those of other co-occurring plant defence traits.
In addition to acting as an obstacle to feeding, trichomes may damage herbivores that consume them. Ingestion of trichomes by chewing herbivores is common, and many species have evolved the strategy of ‘mowing’ trichomes to gain access to underlying tissues [9,12]. Previous work has shown that the ingestion of glandular trichomes can harm herbivores [13], for example, via direct toxicity of gland contents [14] or, in one instance, by making caterpillars more attractive to predatory ants [15]. To our knowledge, no previous work has focused explicitly on the post-ingestive effects of non-glandular trichomes on herbivores, although these trichomes often feature sharp, rigid spikes, some reinforced with silica [16], that might be expected to damage herbivore digestive tracts. In particular, ingested trichomes might be expected to damage the peritrophic matrix (PM), a protective sheath that lines the guts of most insects and which serves to prevent mechanical damage to the gut epithelium, inhibit pathogen invasion and assist in digestion and nutrient absorption [17,18]. Disruption of the PM by plant defence proteins (e.g. maize insect resistant 1-cysteine protease toxin [19,20]) or by pathogens has previously been shown to have adverse consequences on insect growth and development [17,20,21].
This study explores pre- and post-ingestive effects of the non-glandular trichomes of horsenettle plants (S. carolinense) on Manduca sexta caterpillars, which are specialist herbivores of Solanaceae. Horsenettle plants exhibit a single type of stellate non-glandular trichome, characterized by a star-like whorl of spines around a primary vertical spike ([22]; electronic supplementary material, figure S1). In previous work, we have found that inbreeding leads to a significant decrease in trichome density in this species (i.e. in selfed versus outcrossed progeny of a given maternal plant) [22]. Furthermore, inbred plants are far less resistant to herbivore feeding [23,24]. However, because inbreeding in S. carolinense has adverse effects on a wide range of physical and chemical plant defences [22–27], previous work has not determined the extent to which the decreased resistance of inbred horsenettle plants to herbivory was caused by the observed effects of inbreeding on trichomes.
The goals of this study were to investigate the specific effects of S. carolinense trichomes on herbivores and, in particular, to test the hypothesis that the ingestion of trichomes by feeding caterpillars might result in damage to the gut PM. To achieve these goals, we manipulated caterpillar exposure to trichomes in three ways, including (i) via plant breeding (i.e. comparing inbred versus outbred progeny as in our previous studies), (ii) by physically removing trichomes (comparing ‘shaved’ versus intact leaves) and (iii) by adding trichomes collected from leaves to artificial diet (comparing diet with and without trichomes). We then assessed differences in caterpillar behaviour and performance across these treatments, as well as damage inflicted on the PM of feeding caterpillars (via scanning electron and light microscopy of excised tissues). Finally, we assessed PM damage in M. sexta caterpillars fed on two additional Solanaceae species that differ in their trichome profiles: Solanum eleaegnifolium, which produces non-glandular stellate trichomes similar to those of S. carolinense, and Nicotiana tabacum, which primarily produces glandular trichomes.
2. Material and methods
(a). Study organisms
(i). Solanum carolinense
Horsenettle (S. carolinense L.) is a perennial weed native to Eastern North America that inhabits early successional habitats [28]. In the field, horsenettle is attacked by a variety of herbivores, many of which also attack closely related crops in the genus Solanum, such as tomato and potato [28]. Previous work has documented a range of anti-herbivore defences in this species, including biochemical defences [27], volatiles [25,26] and structural defences, such as trichomes and spines [22].
This study employed multiple vegetatively produced ramets of 18 different horsenettle genotypes, representing three inbred (selfed) and three outbred progeny from each of three different maternal families (designated B1, A6 and C10). The maternal plants were randomly selected from a population of 16 genotypes originally collected from a natural population near State College, Pennsylvania, USA. Ramets were created from each of these 16 plants by cutting the horizontal root into five or six pieces of similar size (5 cm). These cuttings were grown in 4 l pots in an insect-free greenhouse. Forty flowers produced on one ramet from each of the original 16 field-collected plants were outcrossed, and 40 flowers from a second ramet were self-pollinated (see [24–26,29] for detailed methods). Seeds from these pollinations were germinated and grown in an insect-free greenhouse, and ramets were created from each of the 18 plants used in this study by cutting the horizontal roots into 5 cm segments (as above) after 12 weeks. The cuttings were grown in flat beds in a growth chamber (16 L : 8 D; day/night temperatures 25/22°C; 65% relative humidity) and lightly watered each day. After two weeks, re-sprouted ramets were transplanted to 10 cm tall, 600 ml pots and watered daily. Care was taken to ensure that the leaf area, size, height and growth stage of the plants used in the experiment were similar (plants used in all experiments were about six weeks old and none had yet flowered).
(ii). Solanum elaeagnifolium
Solanum elaeagnifolium Cav. (silverleaf nightshade) is another herbaceous perennial weed native to South and Central America and the southwestern USA [30]. The species is highly invasive in many parts of the world, including the Mediterranean region [31]. The genotypes used in this study were derived from a highly invasive population in Thessaloniki (Greece). Ten fruits (randomly selected) produced on each of 10 genets spaced 10 m apart were collected, and seeds were extracted and pooled together by genet. These seeds were germinated and grown in an insect-free greenhouse and transplanted into 2 l pots. Six weeks after transplanting, seedlings with 8–10 fully developed leaves were used for the biological assays.
(iii). Nicotiana tabacum
Nicotiana tabacum seeds, generously donated by the Zurich Botanischer Garten (Zurich, Switzerland), were germinated and grown under similar conditions as S. elaeagnifolium. Twenty days after germination, seedlings were transplanted into 2 l pots and experiments were conducted three weeks after transplanting.
(iv). Manduca sexta
Manduca sexta commonly feeds on S. carolinense in the field [32], and can successfully complete its life cycle on each of the plant species used in this study [15,33]. Manduca sexta eggs were bought from a commercial vendor (Carolina Biological Supply, NC, USA) and from a colony maintained by Max Plank Institute for Chemical Ecology (Jena, Germany) and were hatched in Petri dishes (90 × 15 mm; Becton Dickinson & Co., Lincoln Park, NJ, USA) on moist Whatman® filter paper in a growth chamber (16 L : 8 D; 25°C; 65% relative humidity). After hatching, neonate larvae were moved to another growth chamber where the experiments were conducted (16 L : 8 D; 25°C L : 22°C D; 65% relative humidity).
(b). Bioassays
(i). No-choice feeding and growth assays with inbred and outbred leaves
To document M. sexta feeding efficiency and growth on inbred and outbred horsenettle plants, 24 newly hatched first-instar larvae were weighed and placed individually onto fully developed young leaves (one larva per plant). Six-week-old plants with six to eight fully developed leaves were used for this experiment. A stopwatch was used to record the time between placement of the larvae and first visual sign of leaf ingestion (indicated by the presence of a small hole on the leaf and a green food bolus inside the caterpillar). Caterpillars were then allowed to feed uninterrupted for 3 days, after which they were removed and weighed to determine relative weight gain according to the following equation:
To quantify differences in trichome numbers on inbred and outbred plants, a circular disc (1 cm diameter) was excised from each leaf where the caterpillars started to feed, and trichome density was quantified using previously reported methods (for details, see [22]).
(ii). No-choice growth assays with ‘shaved’ and control leaves
For this assay, trichomes were completely removed (shaved) from half the leaves of 15 inbred and 15 outbred plants using a twin-blade razor, while the remaining (control) leaves were left unshaved. Complete trichome removal was verified by light microscopy. Leaf discs of 4 cm diameter were then excised from each leaf with a modified bottle cap. The size of the discs ensured that sufficient plant tissue was available throughout the duration of the feeding assay (3 days). The leaf discs were placed in Petri dishes with moist filter paper and presented to pre-weighed first-instar caterpillars (n = 15 per treatment). After 3 days of feeding, caterpillars were removed and weighed again to calculate relative weight gain as described above.
(iii). No-choice feeding and growth assays with artificial diet
To further explore how caterpillar feeding is affected by trichomes, abaxial and adaxial trichomes from 10 fully developed leaves (one each from five inbred and five outbred genets, randomly selected) were shaved as above. Shaved trichomes were carefully added to approximately 100 ml distilled water and mixed with 100 g of caterpillar diet powder (Cookie-dough diet, Educational research company, NE, USA). After solidifying, excess portions of diet (roughly 5 g) were parceled onto a Petri dish with moist filter paper. Control diet was prepared in similar fashion but without trichomes. Newly hatched first-instar larvae (n = 35 per treatment) were weighed and placed directly on the diet pellet (trichomes added or control; no choice test) and allowed to feed for 3 consecutive days. Data on the position of larvae (on the pellet versus away from the pellet) were collected at 1, 6 and 24 h after the start of the experiment. After 3 days, caterpillars were removed and weighed to calculate relative weight gain (as above).
(iv). Dual-choice assays with artificial diet
To test whether caterpillars prefer the trichome-added diet or control diet, we conducted a choice assay with newly hatched first-instar larvae (n = 35). Diet pellets with and without trichomes (as above) were placed 3 cm apart on moist filter paper in a Petri dish. First-instar caterpillars were placed 3 cm away from the centre of the two pellets, and both their initial feeding choice and position after 24 h were recorded.
(v). Scanning electron microscopy of frass and peritrophic matrix from plant- and diet-fed caterpillars
To examine the effects of trichomes on the caterpillar PM, three third-instar caterpillars were randomly selected from a pool of 15 caterpillars reared on inbred and outbred horsenettle leaves (which, as discussed above, exhibit consistent differences in trichome density). Caterpillars grown on the artificial diet were used as controls. To collect PM, caterpillars were immobilized on ice and dissected in a water bath using a dissecting microscope. The gut was removed and gently teased open with fine forceps [20]. For frass samples, three frass pellets per caterpillar were randomly chosen from the same 15 caterpillars used for collecting PM. Both PM and frass pellets were removed and fixed overnight at 4°C in a 25% gluteraldehyde solution. For scanning electron microscopy, tissue was dried through a series of ethanol washes of increasing concentration followed by critical point drying then mounted on aluminium stubs using carbon tape. Mounted stubs were coated with gold–palladium at 2.5 kV and examined by using a Cambridge S360 scanning electron microscope (Huck Institutes of Life Sciences, Microscopy Core Facility, The Pennsylvania State University, USA). While other bioassays employed first-instar caterpillars, third-instar caterpillars were used for SEM (and light) microscopy due to difficulties associated with dissecting PM intact from smaller first instars.
(vi). Light microscopy of PM from plant- and diet-fed caterpillars
To confirm that the observed PM damage was not merely an artefact of the SEM preparation protocol (which involved dehydration rinses and critical point drying), PM from six caterpillars (three feeding on inbred plants and three on outbred plants) were dissected and imaged using an Olympus SZX 10 light microscope. The PM was then viewed and photographed using a Leica M420 microscope with a camera attachment (Leica MC 170 HD) at magnification between ×25 and ×32 (Leica systems, Germany). For comparison, PM and frass from caterpillars grown on two other Solanaceae species: S. eleagnifolium and N. tabacum, were examined using the same microscopy techniques.
(vii). Filming caterpillar–trichome interactions
Neonate M. sexta feeding and movement across trichome-dense horsenettle leaves was filmed using a Canon 60D digital single lens reflex camera with a 100 mm EF L macro lens (electronic supplementary material, video S1).
(c). Statistical analyses
For the horsenettle assays, analysis of variance (ANOVA) was used to assess caterpillar bite time, and weight gain (dependent variables) with maternal family (random), breeding (fixed) and their interaction as predictor variables. Unpaired t-tests were used to analyse trichome density. ANOVAs were also used to analyse caterpillar weight gain on shaved and unshaved horsenettle leaves and on caterpillar diet with and without trichomes. Data were transformed to meet ANOVA assumptions. Caterpillar feeding initiation time and weight gain on shaved and intact leaves data were analysed using unpaired Student's t-tests. Caterpillar choice tests were analysed using χ2. Caterpillar behaviour in no-choice assays with control and trichome-enriched diet was compared using Barnard's exact tests. Data from s.e.m. were analysed visually. Eighteen digitized images from each treatment (six per sample) were examined for the presence of trichomes on PM and frass and reported. Light microscopy data on S. eleagnifolium and N. tabacum were analysed in a similar manner. Barnard's tests were conducted with the software R (v. 3.0); all other analyses were conducted with the software Graph Pad (La Jolla, CA, USA).
3. Results
(a). Caterpillars initiate feeding more rapidly and gain more weight on inbred plants with low trichome densities
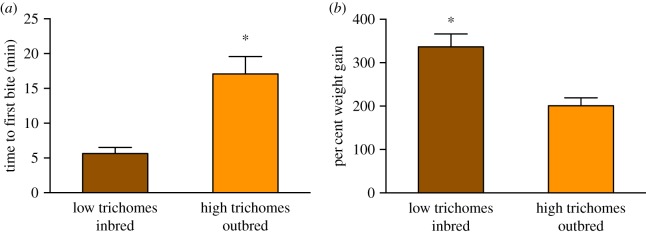
As expected from our previous findings [22], leaves from outbred plants exhibited significantly higher trichome density than leaves from inbred plants (outbred: 48.5 ± 4.7 trichomes per cm2; inbred: 25.1 ± 1.7 trichomes per cm2 (mean ± s.e.); unpaired t-test; p < 0.0001, n = 24 each). Consistent with this difference, first-instar caterpillars took significantly more time to commence feeding on outbred plants than on inbred plants (inbred feeding onset: 5.62 ± 0.88 min, outbred feeding onset: 17.08 ± 2.50 min (mean ± s.e.); figure 1a and electronic supplementary material, table S1a; ANOVA F1,42 = 64.36, p = 0.015). This pattern was consistent across all three maternal families examined (electronic supplementary material, figure S2; and table S1a). Caterpillars also gained more weight over 3 days of feeding on inbred plants than on outbred plants (inbred weight increase was 336.5 ± 29.6%, outbred weight increase was 200.7 ± 18.3% (mean ± s.e.); figure 1b and electronic supplementary material, table S1b; ANOVA F1,42 = 159.2, p = 0.006), consistent with our findings in previous studies [23,33]. There were no family or family × breeding effects on weight gain (electronic supplementary material, figure S3; and table S1b).
Figure 1.
Mean ± s.e. of time until feeding onset (a) and per cent weight gain (b) for first-instar M. sexta caterpillars on inbred (low trichome density) and outbred (high trichome density) horsenettle plants after 3 days of feeding. Asterisk indicates significance (p < 0.05) for post hoc Tukey tests from ANOVA. (Online version in colour.)
(b). Removing trichomes increases caterpillar performance
Over 2 days, caterpillars gained significantly more weight on shaved leaves than on unshaved leaves (shaved: 182.2 ± 23.0%; unshaved: 113.6 ± 28.3%; n = 15 per treatment; figure 2; unpaired t-test; t = 2.583, d.f. = 21, p = 0.01), suggesting that the presence of trichomes reduces caterpillar feeding efficiency. In addition, a two-way ANOVA revealed that both the trichome treatment (shaving; F1,25 = 19.55, p = 0.00) and the breeding effect (F1,25 = 8.56, p = 0.007) were significant (electronic supplementary material, figure S4 and table S2). We were unable to continue the experiment for a third day due to rapid drying of the leaves.
Figure 2.
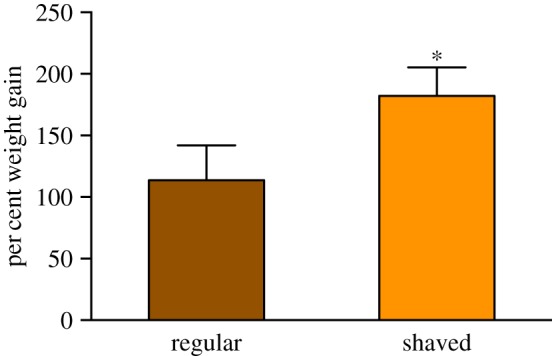
Mean ± s.e. of per cent weight gain of first-instar M. sexta caterpillars after 2 days of feeding on leaves with intact trichomes and ‘shaved’ leaves lacking trichomes. Asterisk indicates significance (p < 0.05) after unpaired t-test. (Online version in colour.)
(c). Adding trichomes to artificial diet alters feeding behaviour and reduces weight gain
In no-choice experiments, caterpillars given an artificial diet with trichomes wandered away from their food more frequently than caterpillars fed the trichome-free control diet—a pattern we observed consistently at 1, 6 and 24 h after the start of the experiment (Barnard's exact tests: 1 h p < 0.0001; 6 h p = 0.026; 24 h p = 0.004; n = 36 per treatment; table 1). Over 3 days of feeding, the caterpillars given the diet with trichomes also gained less weight than caterpillars fed on the control diet (control: 223.0 ± 28.7%; trichome: 72.6 ± 13.3% mean ± s.e.; unpaired t-test: t = 4.84, d.f. = 70, p < 0.0001; figure 3).
Table 1.
Barnard's exact test of 2 × 2 contingency tables comparing caterpillar position distribution (on diet versus away) between regular diet and trichome diet no-choice assays.
| time point (hours) | control diet on/away | trichomes diet on/away | p-value |
|---|---|---|---|
| 1 | 27/8 | 10/26 | 2.858×10−5 |
| 6 | 23/12 | 14/22 | 0.02623 |
| 24 | 26/9 | 14/21 | 0.004027 |
Figure 3.
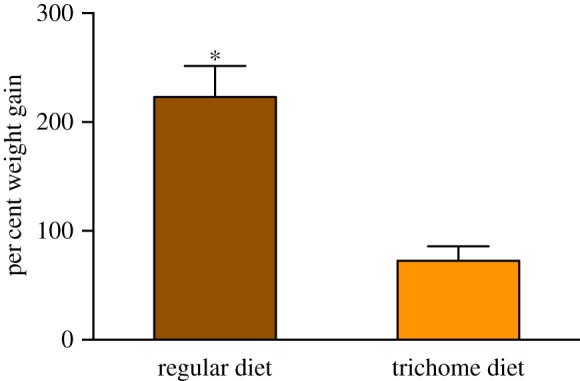
Mean ± s.e. of per cent weight gain of first-instar M. sexta caterpillars on control artificial diet and artificial diet enriched with trichomes. Data collected after 3 days of feeding. Asterisk indicates significance (p < 0.05) after unpaired t-test. (Online version in colour.)
(d). Caterpillars avoid trichomes in preference assays
When first-instar caterpillars were given a choice between artificial diet with and without trichomes, they preferentially fed on the trichome-free control diet (χ2 goodness of fit tests: 1 h, χ2 = 2.83, d.f. = 1, p = 0.09; 24 h, χ2 = 6.17, d.f. = 1, p = 0.01; figure 4).
Figure 4.
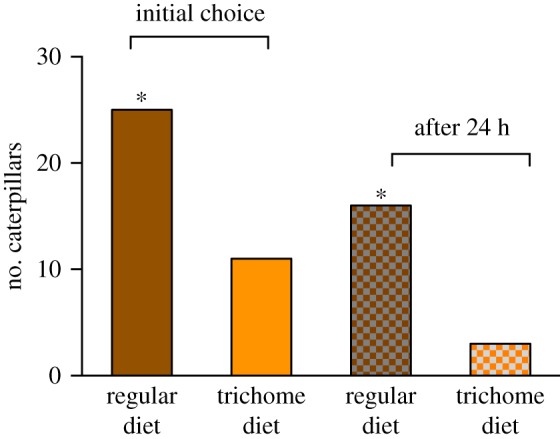
First-instar M. sexta choice between regular artificial diet and diet enriched with trichomes. First choice (initial) and choice after 24 h were recorded. Asterisk indicates significance (p < 0.05) for χ2 analyses. (Online version in colour.)
(e). Ingested trichomes and trichome pieces pass intact through the digestive tract
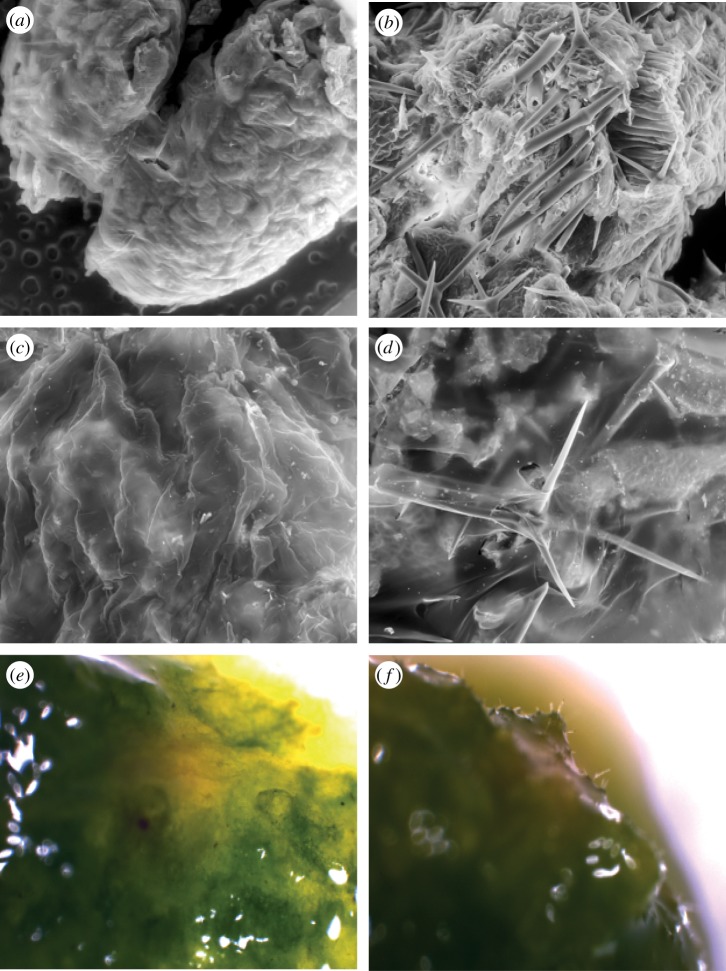
SEM imaging of caterpillar frass revealed that stellate trichomes from inbred and outbred plants survive passage through the caterpillars' guts. Frass from plant-fed caterpillars was riddled with extruding trichome spikes that were not observed on frass from diet-fed caterpillars (figure 5a,b). Cross-sectional views of the pellets also revealed a high concentration of trichomes embedded inside the pellet (SEM and light microscopy; not pictured). Both intact and broken trichomes were evident in the frass.
Figure 5.
Images of frass (a,b) and PM (c–f) from third-instar M. sexta caterpillars fed diets varying in trichome content. (a) Frass from caterpillar fed trichome-free artificial diet. (b) Frass from a caterpillar fed intact S. carolinense leaves, showing embedded trichomes. (c) PM from a caterpillar fed trichome-free artificial diet. (d) PM from a caterpillar fed intact S. carolinense leaves, showing protruding trichome spines. (e) PM from a caterpillar fed intact Nicotiana tabacum leaves (which lack stellate trichomes), showing no damage. (f) PM from a caterpillar fed intact S. eleagnifolium leaves, showing protruding trichomes. Scanning electron microscopy was used for a–d, light microscopy for e and f.
(f). Ingested trichomes damage the peritrophic matrix
SEM imaging of caterpillar PM samples revealed major differences between horsenettle-fed and diet-fed caterpillars. In the latter group, the PM was invariably smooth and unbroken. By contrast, PM from plant-fed larvae was consistently stretched, punctured and torn by trichome spikes in the periphery of the food bolus (figure 5c,d). Light microscopy revealed similar damage. We also examined the PM of caterpillars that ingested leaves from two other Solanaceous species, N. tabacum, which produces mainly glandular trichomes, and S. eleagnifolium, which exhibits stellate trichomes similar to those of S. carolinense. We found no evidence of PM damage in caterpillars that fed on N. tabacum (figure 5e); however, caterpillars that fed on S. eleagnifolium exhibited damage (figure 5f) similar to that observed for those that fed on S. carolinense.
4. Discussion
Our microscopy studies revealed that the PM of caterpillars is severely damaged by ingestion of the non-glandular stellate trichomes of S. carolinese (as well as those of S. eleagnifolium). Furthermore, our behavioural and performance assays revealed consistent adverse effects of trichomes on caterpillar feeding performance and growth. Taken together, these results indicate that trichomes can deter herbivory via both pre- and post-ingestive effects and suggest that the reduced density of trichomes on inbred relative to outbred plants observed in this as well as previous studies [22] likely contributes to the observed decrease in plant resistance to herbivory associated with inbreeding in horsenettle [22,23,33–35].
(a). Overall effects of trichomes on caterpillar performance
We manipulated trichomes on leaves both via plant breeding (taking advantage of the fact that inbred plants consistently exhibit reduced trichome density relative to outbred plants) and via the physical removal of trichomes; in each case, we found evidence that the presence and density of trichomes had adverse effects on caterpillar feeding and performance (figures 1–2). Neither of these techniques allows us to completely isolate the effects of trichomes from other defensive traits, as inbreeding has been shown to have adverse effects on a wide range of defence traits in S. carolinense [22–24], while shaving leaves to remove trichomes inevitably causes minor damage to the epidermis that might be expected to result in the induction of chemical defences, which have previously been shown to be induced by mechanical damage in horsenettle [22,27]. However, the confounding effects of these manipulations should work in opposite directions, as inbreeding is expected to reduce defence competence across the board [23–25,36], while the removal of trichomes might be expected to elicit higher levels of chemical defence.
To disentangle the effects of trichomes from other plant defence traits, we also performed experiments in which trichomes removed from leaves were added to an artificial diet. Here again we observed adverse effects of trichomes on caterpillar feeding performance and weight gain (figure 3), as well as a preference for the trichome-free diet in choice assays (figure 4).
In combination, these results provide compelling evidence that non-glandular trichomes adversely affect herbivore performance. Our findings are consistent with results from other plant–insect systems, which also revealed a feeding preference of caterpillars for leaf tissues without trichomes [37,38] or with reduced trichome density [39]. For example, in Lupinus arboreus Sims (Fabaceae), caterpillars of Platyprepia virginalis (Boisduval) (Lepidoptera: Arctiidae) preferred leaves with lower trichome densities in laboratory assays, and were found more on plants with lower trichome densities in the field [39].
(b). Pre-ingestive effects of stellate trichomes on caterpillars
As with larvae of other lepidopteran species, M. sexta caterpillars begin feeding on leaves shortly after hatching. We filmed neonate movement during this critical stage and observed that trichomes present a formidable barrier to reaching the leaf epidermis (figure 1; electronic supplementary material, video S1). In addition to suspending caterpillars above the leaf surface, trichomes damaged caterpillars and caused them to bleed haemolymph, which might conceivably also increase their conspicuity to natural enemies. Caterpillars responded to trichome jabs with violent head tilting motions, a common defence response in M. sexta that is typically observed in the context of predation [40].
Navigating the hostile landscape created by dense trichomes in order to find a suitable feeding site appears to be a difficult task for early instar caterpillars. We found that caterpillars took on average more than three times as long to initiate feeding on outbred plants than on inbred plants (with less dense trichomes; figure 1a). Meanwhile, caterpillars invariably initiated feeding almost immediately on plants where the trichomes had been experimentally removed. This delay in initiating feeding may increase caterpillar vulnerability to abiotic stressors such as desiccation as well as predation (particularly for poorly camouflaged neonate larvae, such as those of M. sexta, which require plant-derived pigments to blend in to green leaves [11]) and may also increase the risk of being dislodged from the plant.
In addition to acting as a barrier, trichomes may also increase time spent foraging rather than feeding. In no-choice assays with artificial diet, caterpillars spent more time foraging (categorized as ‘away’ from food) and less time feeding when they have to contend with stellate trichomes. Furthermore, in choice assays, caterpillars consistently preferred the trichome-free diet to the trichome-enriched diet (figure 4). Although these results reflect an artificial feeding environment, they suggest that the presence of trichomes might increase the time that caterpillars spend searching for less well-defended feeding sites.
Taken together, these results reveal significant pre-ingestive effects on caterpillar feeding behaviour and performance that likely contribute to the overall adverse effects of trichomes on caterpillar growth that were consistently observed across our bioassays (figures 1b, 2 and 3). These findings are also consistent with previous studies that have reported adverse pre-ingestive effects of trichomes on caterpillars via wounding, obstruction of access to the leaf surface and the elicitation of foraging rather than feeding behaviour [37,41].
(c). Post-ingestive effects of trichomes on caterpillars
In addition to pre-ingestive effects, we found evidence that trichomes can cause adverse effects on caterpillars following ingestion. The presence of intact trichomes and trichome pieces embedded in and protruding through the frass indicates that consumed trichomes are not digested sufficiently to render them harmless as they move through the gut in the bolus (figure 5b). Indeed, visualization of the PM via SEM and light microscopy revealed that trichomes cause severe damage to this critical membrane (figure 5d). Compared to the intact PM (figure 5c), the PM of caterpillars that fed on trichomes had massive tears due to protruding trichomes (figure 5d; electronic supplementary material, figure S5). Similar damage was observed for caterpillars that had fed on another Solanaceous species, S. eleagnifolium, which also produces stellate non-glandular trichomes similar to those of S. carolinense. By contrast, no PM damage was apparent in caterpillars that had fed on the artificial diet or on N. tabacum, which produces mainly glandular trichomes. These findings suggest that the non-glandular stellate trichomes of Solanum spp. function not only as a mechanical barrier to feeding, but also play a mechanical post-ingestive defensive role by rupturing the PM of caterpillars. Moreover, it appears that the glandular trichomes of N. tabacum do not function in post-ingestive mechanical defence.
With the exception of hemipterans, all insects have a PM, and PM composition (chitins, structural and non-structural proteins) and function have been well studied, especially in lepidopteran larvae [19]. The PM facilitates digestion and nutrient absorption, protects the endothelium from mechanical abrasion by the food bolus as it moves through the midgut, plays important roles in detoxification of plant toxins and provides a barrier against pathogens and parasites [19,20].
A previous study reported that fall armyworm larvae compensate for PM damage caused by 1-cysteine protease, a defensive protein found in an insect-resistant line of maize, by upregulating genes involved in repairing and replacing the disrupted PM and by upregulating the expression of several families of digestive enzymes [19]. The costs associated with the increased production of proteins involved in PM repair and digestion were furthermore hypothesized to contribute to the impaired growth of larvae feeding on the resistant line [19]. Increases in the expression of PM repair genes and enzymes have also been found following damage by pathogens that disrupt the PM [20,42]. In addition to these costs, the mechanical damage to the PM caused by the non-glandular stellate trichomes of S. carolinense is likely to be associated with inefficiencies in digestion and nutrient absorption (e.g. [20,43]). Unlike defence proteins that have been shown to increase PM permeability by chemical means [17,44], stellate trichomes open breaches in the PM mechanically (figure 5d and electronic supplementary material, figure S5), and these large openings should further enhance invasion by pathogens and parasites. Immune responses are energetically costly and frequently associated with a reduction in development rate (e.g. [45,46]). Thus, immune induction could also contribute to the overall reduction caterpillar growth that we observed in this study, a possibility we are currently exploring.
The findings presented here indicate that the non-glandular trichomes of Solanum spp. serve as both pre- and post-ingestion defences against M. sexta caterpillars by limiting their performance in short-term growth studies and influencing their food preferences. It is reasonable to speculate that long-term feeding on leaves with stellate trichomes may have additional long-term effects on caterpillar survival, development and reproductive output.
Supplementary Material
Acknowledgements
The authors thank Erica Smyers and Claude Fornallaz for assistance with M. sexta rearing and the cultivation of the plants, Aisling B. Ryan and Sean Hardison for helping with shaving trichomes, Drs Theodora Petanidou and Thomas Tscheulin for helping with the collection of S. elaeagnifolium and John Cantolina at the Penn State Huck Institute of Life Sciences Microscopy Core Facility for helping us with the SEM sample preparation and data collection.
Data accessibility
Data are available from the Dryad Digital Repository: http://dx.doi.org/10.5061/dryad.jd417 [47].
Authors' contributions
R.R.K., J.D.S., M.C.M., C.D.M. and A.G.S. designed the study; R.R.K. conducted most experiments, and J.D.S. assisted with data collection and analyses; R.R.K. and J.D.S. drafted the manuscript, and all authors contributed significantly to revisions.
Competing interests
We declare we have no competing interests.
Funding
This study was supported by The David and Lucile Packard Foundation grant to Consuelo M De Moraes, by National Science Foundation grant DEB1050998, and by grant nos. 2008-35302-04577 and 2009-33120-20093 from the USDA National Institute of Food and Agriculture.
References
- 1.Kennedy GG. 2003. Tomato, pests, parasitoids, and predators: tritrophic interactions involving the genus Lycopersicon. Annu. Rev. Entomol. 48, 51–72. ( 10.1146/annurev.ento.48.091801.112733) [DOI] [PubMed] [Google Scholar]
- 2.Shepherd RW, Wagner GJ. 2007. Phylloplane proteins: emerging defenses at the aerial frontline? Trends Plant Sci. 12, 51–56. ( 10.1016/j.tplants.2006.12.003) [DOI] [PubMed] [Google Scholar]
- 3.Glas J, Schimmel B, Alba J, Escobar-Bravo R, Schuurink R, Kant M. 2012. Plant glandular trichomes as targets for breeding or engineering of resistance to herbivores. Int. J. Mol. Sci. 13, 17 077–17 103. ( 10.3390/ijms131217077) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Peiffer M, Tooker JF, Luthe DS, Felton GW. 2009. Plants on early alert: glandular trichomes as sensors for insect herbivores. New Phytol. 184, 644–656. ( 10.1111/j.1469-8137.2009.03002.x) [DOI] [PubMed] [Google Scholar]
- 5.Szyndler MW, Haynes KF, Potter MF, Corn RM, Loudon C. 2013. Entrapment of bed bugs by leaf trichomes inspires microfabrication of biomimetic surfaces. J. R. Soc. Interface 10, 20130174 ( 10.1098/rsif.2013.0174) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bickford CP. 2016. Ecophysiology of leaf trichomes. Funct. Plant Bio. 43, 807–814. ( 10.1071/FP16095) [DOI] [PubMed] [Google Scholar]
- 7.Gross P, Price PW. 1988. Plant influences on parasitism of two leafminers: a test of enemy-free space. Ecology 69, 1506–1516. ( 10.2307/1941648) [DOI] [Google Scholar]
- 8.Schillinger JA, Gallun RL. 1968. Leaf pubescence of wheat as a deterrent to the cereal leaf beetle, Oulema melanopus. Ann. Entomol. Soc. Am. 61, 900–903. ( 10.1093/aesa/61.4.900) [DOI] [Google Scholar]
- 9.Baur R, Binder S, Benz G. 1991. Nonglandular leaf trichomes as short-term inducible defense of the grey alder, Alnus incana (L.), against the chrysomelid beetle, Agelastica alni L. Oecologia 87, 219–226. ( 10.1007/bf00325259) [DOI] [PubMed] [Google Scholar]
- 10.Medeiros L, Moreira GR. 2002. Moving on hairy surfaces: modifications of Gratiana spadicea larval legs to attach on its host plant Solanum sisymbriifolium. Entomol. Exp. Appl. 102, 295–305. ( 10.1046/j.1570-7458.2002.00950.x) [DOI] [Google Scholar]
- 11.Zalucki MP, Clarke AR, Malcolm SB. 2002. Ecology and behavior of first-instar larval Lepidoptera. Annu. Rev. Entomol. 47, 361–393. ( 10.1146/annurev.ento.47.091201.145220) [DOI] [PubMed] [Google Scholar]
- 12.Hulley PE. 1988. Caterpillar attacks plant mechanical defence by mowing trichomes before feeding. Ecol. Entomol. 13, 239–241. ( 10.1111/j.1365-2311.1988.tb00351.x) [DOI] [Google Scholar]
- 13.Chu C-C, Freeman TP, Buckner JS, Henneberry TJ, Nelson DR, Natwick ET. 2001. Susceptibility of upland cotton cultivars to Bemisia tabaci biotype B (Homoptera: Aleyrodidae) in relation to leaf age and trichome density. Ann. Entomol. Soc. Am. 94, 743–749. ( 10.1603/0013-8746(2001)094%5B0743:soucct%5D2.0.co;2) [DOI] [Google Scholar]
- 14.Hurley KW, Dussourd DE. 2014. Toxic geranium trichomes trigger vein cutting by soybean loopers, Chrysodeixis includens (Lepidoptera: Noctuidae). Arthropod-Plant Int. 9, 33–43. ( 10.1007/s11829-014-9348-6) [DOI] [Google Scholar]
- 15.Weinhold A, Baldwin IT. 2011. Trichome-derived O-acyl sugars are a first meal for caterpillars that tags them for predation. Proc. Natl Acad. Sci. USA 108, 7855–7859. ( 10.1073/pnas.1101306108) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lanning FC, Eleuterius LN. 1985. Silica and ash in tissues of some plants growing in the coastal area of Mississippi, USA. Ann. Bot 56, 157–172. [Google Scholar]
- 17.Terra WR. 2001. The origin and functions of the insect peritrophic membrane and peritrophic gel. Arch. Insect. Biochem. Physiol. 47, 47–61. ( 10.1002/arch.1036) [DOI] [PubMed] [Google Scholar]
- 18.Wang P, Granados RR. 2001. Molecular structure of the peritrophic membrane (PM): identification of potential PM target sites for insect control. Arch. Insect. Biochem. Physiol. 47, 110–118. ( 10.1002/arch.1041) [DOI] [PubMed] [Google Scholar]
- 19.Fescemyer HW, Sandoya GV, Gill TA, Ozkan S, Marden JH, Luthe DS. 2013. Maize toxin degrades peritrophic matrix proteins and stimulates compensatory transcriptome responses in fall armyworm midgut. Insect. Biochem. Mol. Biol. 43, 280–291. ( 10.1016/j.ibmb.2012.12.008) [DOI] [PubMed] [Google Scholar]
- 20.Pechan T, Cohen A, Williams WP, Luthe DS. 2002. Insect feeding mobilizes a unique plant defense protease that disrupts the peritrophic matrix of caterpillars. Proc. Natl Acad. Sci. USA 99, 13 319–13 323. ( 10.1073/pnas.202224899) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoover K, Humphries MA, Gendron AR, Slavicek JM. 2010. Impact of viral enhancin genes on potency of Lymantria dispar multiple nucleopolyhedrovirus in L. dispar following disruption of the peritrophic matrix. J. Invertebr. Pathol. 104, 150–152. ( 10.1016/j.jip.2010.02.008) [DOI] [PubMed] [Google Scholar]
- 22.Kariyat RR, Balogh CM, Moraski RP, De Moraes CM, Mescher MC, Stephenson AG. 2013. Constitutive and herbivore-induced structural defenses are compromised by inbreeding in Solanum carolinense (Solanaceae). Am. J. Bot. 100, 1014–1021. ( 10.3732/ajb.1200612) [DOI] [PubMed] [Google Scholar]
- 23.Delphia CM, De Moraes CM, Stephenson AG, Mescher MC. 2009. Inbreeding in horsenettle influences herbivore resistance. Ecol. Entomol. 34, 513–519. ( 10.1111/j.1365-2311.2009.01097.x) [DOI] [Google Scholar]
- 24.Kariyat RR, Mena-Alí J, Forry B, Mescher MC, De Moraes CM, Stephenson AG. 2012. Inbreeding, herbivory, and the transcriptome of Solanum carolinense. Entomol. Exp. Appl. 144, 134–144. ( 10.1111/j.1570-7458.2012.01269.x) [DOI] [Google Scholar]
- 25.Kariyat RR, Mauck KE, De Moraes CM, Stephenson AG, Mescher MC. 2012. Inbreeding alters volatile signaling phenotypes and influences tri-trophic interactions in horsenettle (Solanum carolinense L.). Ecol. Lett. 15, 301–309. ( 10.1111/j.1461-0248.2011.01738.x) [DOI] [PubMed] [Google Scholar]
- 26.Kariyat RR, Mauck KE, Balogh CM, Stephenson AG, Mescher MC, De Moraes CM. 2013. Inbreeding in horsenettle (Solanum carolinense) alters night-time volatile emissions that guide oviposition by Manduca sexta moths. Proc. R. Soc. B 280, 20130020 ( 10.1098/rspb.2013.0020) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Campbell SA, Thaler JS, Kessler A. 2012. Plant chemistry underlies herbivore-mediated inbreeding depression in nature. Ecol. Lett. 16, 252–260. ( 10.1111/ele.12036) [DOI] [PubMed] [Google Scholar]
- 28.Wise MJ. 2007. The herbivores of Solanum carolinense (Horsenettle) in northern Virginia: natural history and damage assessment. Southeastern Nat. 6, 505–522. ( 10.1656/1528-7092(2007)6%5B505:thosch%5D2.0.co;2) [DOI] [Google Scholar]
- 29.Mena-Ali JI, Stephenson AG. 2007. Segregation analyses of partial self-incompatibility in self and cross progeny of Solanum carolinense reveal a leaky S-allele. Genetics 177, 501–510. ( 10.1534/genetics.107.073775) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Boyd JW, Murray DS, Tyrl RJ. 1984. Silverleaf nightshade, Solarium elaeagnifolium, origin, distribution, and relation to man. Econ. Bot. 38, 210–217. ( 10.1007/bf02858833) [DOI] [Google Scholar]
- 31.Petanidou T, Godfree RC, Song DS, Kantsa A, Dupont YL, Waser NM. 2012. Self-compatibility and plant invasiveness: comparing species in native and invasive ranges. Persp. Plant Ecol. Evol. Syst. 14, 3–12. ( 10.1016/j.ppees.2011.08.003) [DOI] [Google Scholar]
- 32.Delphia CM, Rohr JR, Stephenson AG, De Moraes CM, Mescher MC. 2009. Effects of genetic variation and inbreeding on volatile production in a field population of horsenettle. Int. J. Plant Sci. 170, 12–20. ( 10.1086/593039) [DOI] [Google Scholar]
- 33.Portman SL, Kariyat RR, Johnston MA, Stephenson AG, Marden JH. 2015. Cascading effects of host plant inbreeding on the larval growth, muscle molecular composition, and flight capacity of an adult herbivorous insect. Funct. Ecol. 29, 328–337. ( 10.1111/1365-2435.12358) [DOI] [Google Scholar]
- 34.Portman SL, Kariyat RR, Johnston MA, Stephenson AG, Marden JH. 2015. Inbreeding compromises host plant defense gene expression and improves herbivore survival. Plant Signal. Behav. 10, e998548 ( 10.1080/15592324.2014.998548) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kariyat RR, Portman SL. 2016. Plant–herbivore interactions: thinking beyond larval growth and mortality. Am. J. Bot. 103, 789–791. ( 10.3732/ajb.1600066) [DOI] [PubMed] [Google Scholar]
- 36.Kalske A, Mutikainen P, Muola A, Scheepens JF, Laukkanen L, Salminen J-P, Leimu R. 2013. Simultaneous inbreeding modifies inbreeding depression in a plant–herbivore interaction. Ecol. Lett. 17, 229–238. ( 10.1111/ele.12223) [DOI] [PubMed] [Google Scholar]
- 37.Fordyce JA, Agrawal AA. 2001. The role of plant trichomes and caterpillar group size on growth and defence of the pipevine swallowtail Battus philenor. J. Anim. Ecol. 70, 997–1005. ( 10.1046/j.0021-8790.2001.00568.x) [DOI] [Google Scholar]
- 38.Schmitz OJ. 1994. Resource edibility and trophic exploitation in an old-field food web. Proc. Natl Acad. Sci. USA 91, 5364–5367. ( 10.1073/pnas.91.12.5364) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Eaton KM, Karban R. 2014. Effects of trichomes on the behavior and distribution of Platyprepia virginalis caterpillars. Entomol. Exp. Appl. 151, 144–151. ( 10.1111/eea.12178) [DOI] [Google Scholar]
- 40.Walters E, Illich P, Lewin M. 2001. Defensive responses of larval Manduca sexta and their sensitization by noxious stimuli in the laboratory and field. J. Exp. Biol. 204, 457–469. [DOI] [PubMed] [Google Scholar]
- 41.Werker E. 2000. Trichome diversity and development. Adv. Bot. Res. 31, 1–35. ( 10.1016/s0065-2296(00)31005-9) [DOI] [Google Scholar]
- 42.Buchon N, Broderick NA, Poidevin M, Pradervand S, Lemaitre B. 2009. Drosophila intestinal response to bacterial infection: activation of host defense and stem cell proliferation. Cell Host Microbe 5, 200–211. ( 10.1016/j.chom.2009.01.003) [DOI] [PubMed] [Google Scholar]
- 43.Campbell PM, Cao AT, Hines ER, East PD, Gordon KH. 2008. Proteomic analysis of the peritrophic matrix from the gut of the caterpillar, Helicoverpa armigera. Insect. Biochem. Mol. Biol. 38, 950–958. ( 10.1016/j.ibmb.2008.07.009) [DOI] [PubMed] [Google Scholar]
- 44.Bolognesi R, Ribeiro AF, Terra WR, Ferreira CL. 2001. The peritrophic membrane of Spodoptera frugiperda: secretion of peritrophins and role in immobilization and recycling digestive enzymes. Arch. Insect. Biochem. Physiol. 47, 62–75. ( 10.1002/arch.1037) [DOI] [PubMed] [Google Scholar]
- 45.Lochmiller RL, Deerenberg C. 2000. Trade-offs in evolutionary immunology: just what is the cost of immunity? Oikos 88, 87–98. ( 10.1034/j.1600-0706.2000.880110.x) [DOI] [Google Scholar]
- 46.Rantala MJ, Roff DA. 2005. An analysis of trade-offs in immune function, body size and development time in the Mediterranean field cricket, Gryllus bimaculatus. Funct. Ecol. 19, 323–330. ( 10.1111/j.1365-2435.2005.00979.x) [DOI] [Google Scholar]
- 47.Kariyat RR, Smith JD, Stephenson AG, De Moraes CM, Mescher MC. 2017. Data from: Non-glandular trichomes of Solanum carolinense deter feeding by Manduca sexta caterpillars and cause damage to the gut peritrophic matrix. Dryad Digital Repository. ( 10.5061/dryad.jd417) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Kariyat RR, Smith JD, Stephenson AG, De Moraes CM, Mescher MC. 2017. Data from: Non-glandular trichomes of Solanum carolinense deter feeding by Manduca sexta caterpillars and cause damage to the gut peritrophic matrix. Dryad Digital Repository. ( 10.5061/dryad.jd417) [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Data are available from the Dryad Digital Repository: http://dx.doi.org/10.5061/dryad.jd417 [47].