Abstract
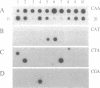
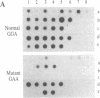
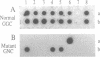
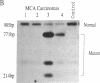
A number of mouse skin tumors initiated by the carcinogens N-methyl-N'-nitro-N-nitrosoguanidine (MNNG), methylnitrosourea (MNU), 3-methylcholanthrene (MCA), and 7,12-dimethylbenz[a]anthracene (DMBA) have been shown to contain activated Ha-ras genes. In each case, the point mutations responsible for activation have been characterized. Results presented demonstrate the carcinogen-specific nature of these ras mutations. For each initiating agent, a distinct spectrum of mutations is observed. Most importantly, the distribution of ras gene mutations is found to differ between benign papillomas and carcinomas, suggesting that molecular events occurring at the time of initiation influence the probability with which papillomas progress to malignancy. This study provides molecular evidence in support of the existence of subsets of papillomas with differing progression frequencies. Thus, the alkylating agents MNNG and MNU induced exclusively G ---- A transitions at codon 12, with this mutation being found predominantly in papillomas. MCA initiation produced both codon 13 G ---- T and codon 61 A ---- T transversions in papillomas; only the G ---- T mutation, however, was found in carcinomas. These findings provide strong evidence that the mutational activation of Ha-ras occurs as a result of the initiation process and that the nature of the initiating event can affect the probability of progression to malignancy.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldaz C. M., Conti C. J., Larcher F., Trono D., Roop D. R., Chesner J., Whitehead T., Slaga T. J. Sequential development of aneuploidy, keratin modifications, and gamma-glutamyltransferase expression in mouse skin papillomas. Cancer Res. 1988 Jun 1;48(11):3253–3257. [PubMed] [Google Scholar]
- Balmain A., Brown K. Oncogene activation in chemical carcinogenesis. Adv Cancer Res. 1988;51:147–182. doi: 10.1016/s0065-230x(08)60222-5. [DOI] [PubMed] [Google Scholar]
- Balmain A., Pragnell I. B. Mouse skin carcinomas induced in vivo by chemical carcinogens have a transforming Harvey-ras oncogene. Nature. 1983 May 5;303(5912):72–74. doi: 10.1038/303072a0. [DOI] [PubMed] [Google Scholar]
- Balmain A., Ramsden M., Bowden G. T., Smith J. Activation of the mouse cellular Harvey-ras gene in chemically induced benign skin papillomas. Nature. 1984 Feb 16;307(5952):658–660. doi: 10.1038/307658a0. [DOI] [PubMed] [Google Scholar]
- Barbacid M. ras genes. Annu Rev Biochem. 1987;56:779–827. doi: 10.1146/annurev.bi.56.070187.004023. [DOI] [PubMed] [Google Scholar]
- Bizub D., Wood A. W., Skalka A. M. Mutagenesis of the Ha-ras oncogene in mouse skin tumors induced by polycyclic aromatic hydrocarbons. Proc Natl Acad Sci U S A. 1986 Aug;83(16):6048–6052. doi: 10.1073/pnas.83.16.6048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bos J. L., Toksoz D., Marshall C. J., Verlaan-de Vries M., Veeneman G. H., van der Eb A. J., van Boom J. H., Janssen J. W., Steenvoorden A. C. Amino-acid substitutions at codon 13 of the N-ras oncogene in human acute myeloid leukaemia. 1985 Jun 27-Jul 3Nature. 315(6022):726–730. doi: 10.1038/315726a0. [DOI] [PubMed] [Google Scholar]
- Burns F. J., Vanderlaan M., Sivak A., Albert R. E. Regression kinetics of mouse skin papillomas. Cancer Res. 1976 Apr;36(4):1422–1427. [PubMed] [Google Scholar]
- Burns P. A., Gordon A. J., Glickman B. W. Influence of neighbouring base sequence on N-methyl-N'-nitro-N-nitrosoguanidine mutagenesis in the lacI gene of Escherichia coli. J Mol Biol. 1987 Apr 5;194(3):385–390. doi: 10.1016/0022-2836(87)90668-1. [DOI] [PubMed] [Google Scholar]
- Chen A. C., Herschman H. R. Tumorigenic methylcholanthrene transformants of C3H/10T1/2 cells have a common nucleotide alteration in the c-Ki-ras gene. Proc Natl Acad Sci U S A. 1989 Mar;86(5):1608–1611. doi: 10.1073/pnas.86.5.1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng S. C., Prakash A. S., Pigott M. A., Hilton B. D., Lee H., Harvey R. G., Dipple A. A metabolite of the carcinogen 7,12-dimethylbenz[a]anthracene that reacts predominantly with adenine residues in DNA. Carcinogenesis. 1988 Sep;9(9):1721–1723. doi: 10.1093/carcin/9.9.1721. [DOI] [PubMed] [Google Scholar]
- Dipple A., Pigott M., Moschel R. C., Costantino N. Evidence that binding of 7,12-dimethylbenz(a)anthracene to DNA in mouse embryo cell cultures results in extensive substitution of both adenine and guanine residues. Cancer Res. 1983 Sep;43(9):4132–4135. [PubMed] [Google Scholar]
- Fasano O., Aldrich T., Tamanoi F., Taparowsky E., Furth M., Wigler M. Analysis of the transforming potential of the human H-ras gene by random mutagenesis. Proc Natl Acad Sci U S A. 1984 Jul;81(13):4008–4012. doi: 10.1073/pnas.81.13.4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Guerrero I., Pellicer A. Mutational activation of oncogenes in animal model systems of carcinogenesis. Mutat Res. 1987 May;185(3):293–308. doi: 10.1016/0165-1110(87)90021-2. [DOI] [PubMed] [Google Scholar]
- Hennings H., Shores R., Mitchell P., Spangler E. F., Yuspa S. H. Induction of papillomas with a high probability of conversion to malignancy. Carcinogenesis. 1985 Nov;6(11):1607–1610. doi: 10.1093/carcin/6.11.1607. [DOI] [PubMed] [Google Scholar]
- Jeffrey A. M. DNA modification by chemical carcinogens. Pharmacol Ther. 1985;28(2):237–272. doi: 10.1016/0163-7258(85)90013-0. [DOI] [PubMed] [Google Scholar]
- Jochemsen A. G., Bernards R., van Kranen H. J., Houweling A., Bos J. L., van der Eb A. J. Different activities of the adenovirus types 5 and 12 E1A regions in transformation with the EJ Ha-ras oncogene. J Virol. 1986 Sep;59(3):684–691. doi: 10.1128/jvi.59.3.684-691.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loveless A. Possible relevance of O-6 alkylation of deoxyguanosine to the mutagenicity and carcinogenicity of nitrosamines and nitrosamides. Nature. 1969 Jul 12;223(5202):206–207. doi: 10.1038/223206a0. [DOI] [PubMed] [Google Scholar]
- Nunn M. F., Seeburg P. H., Moscovici C., Duesberg P. H. Tripartite structure of the avian erythroblastosis virus E26 transforming gene. Nature. 1983 Nov 24;306(5941):391–395. doi: 10.1038/306391a0. [DOI] [PubMed] [Google Scholar]
- Osborne M. R., Brookes P., Lee H. M., Harvey R. G. The reaction of a 3-methylcholanthrene diol epoxide with DNA in relation to the binding of 3-methylcholanthrene to the DNA of mammalian cells. Carcinogenesis. 1986 Aug;7(8):1345–1350. doi: 10.1093/carcin/7.8.1345. [DOI] [PubMed] [Google Scholar]
- Quintanilla M., Brown K., Ramsden M., Balmain A. Carcinogen-specific mutation and amplification of Ha-ras during mouse skin carcinogenesis. Nature. 1986 Jul 3;322(6074):78–80. doi: 10.1038/322078a0. [DOI] [PubMed] [Google Scholar]
- Reddy A. L., Caldwell M., Fialkow P. J. Studies of skin tumorigenesis in PGK mosaic mice: many promoter-independent papillomas and carcinomas do not develop from pre-existing promoter-dependent papillomas. Int J Cancer. 1987 Feb 15;39(2):261–265. doi: 10.1002/ijc.2910390223. [DOI] [PubMed] [Google Scholar]
- Reed K. C., Mann D. A. Rapid transfer of DNA from agarose gels to nylon membranes. Nucleic Acids Res. 1985 Oct 25;13(20):7207–7221. doi: 10.1093/nar/13.20.7207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rigaud G., Grange T., Pictet R. The use of NaOH as transfer solution of DNA onto nylon membrane decreases the hybridization efficiency. Nucleic Acids Res. 1987 Jan 26;15(2):857–857. doi: 10.1093/nar/15.2.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Scribner J. D., Scribner N. K., McKnight B., Mottet N. K. Evidence for a new model of tumor progression from carcinogenesis and tumor promotion studies with 7-bromomethylbenz[a]anthracene. Cancer Res. 1983 May;43(5):2034–2041. [PubMed] [Google Scholar]
- Singer B., Kuśmierek J. T. Chemical mutagenesis. Annu Rev Biochem. 1982;51:655–693. doi: 10.1146/annurev.bi.51.070182.003255. [DOI] [PubMed] [Google Scholar]
- Slaga T. J. Cellular and molecular mechanisms involved in multistage skin carcinogenesis. Carcinog Compr Surv. 1989;11:1–18. [PubMed] [Google Scholar]
- Sukumar S., Notario V., Martin-Zanca D., Barbacid M. Induction of mammary carcinomas in rats by nitroso-methylurea involves malignant activation of H-ras-1 locus by single point mutations. Nature. 1983 Dec 15;306(5944):658–661. doi: 10.1038/306658a0. [DOI] [PubMed] [Google Scholar]
- Toorchen D., Topal M. D. Mechanisms of chemical mutagenesis and carcinogenesis: effects on DNA replication of methylation at the O6-guanine position of dGTP. Carcinogenesis. 1983 Dec;4(12):1591–1597. doi: 10.1093/carcin/4.12.1591. [DOI] [PubMed] [Google Scholar]
- Topal M. D., Eadie J. S., Conrad M. O6-methylguanine mutation and repair is nonuniform. Selection for DNA most interactive with O6-methylguanine. J Biol Chem. 1986 Jul 25;261(21):9879–9885. [PubMed] [Google Scholar]
- Wiseman R. W., Stowers S. J., Miller E. C., Anderson M. W., Miller J. A. Activating mutations of the c-Ha-ras protooncogene in chemically induced hepatomas of the male B6C3 F1 mouse. Proc Natl Acad Sci U S A. 1986 Aug;83(16):5825–5829. doi: 10.1073/pnas.83.16.5825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarbl H., Sukumar S., Arthur A. V., Martin-Zanca D., Barbacid M. Direct mutagenesis of Ha-ras-1 oncogenes by N-nitroso-N-methylurea during initiation of mammary carcinogenesis in rats. 1985 May 30-Jun 5Nature. 315(6018):382–385. doi: 10.1038/315382a0. [DOI] [PubMed] [Google Scholar]