Abstract
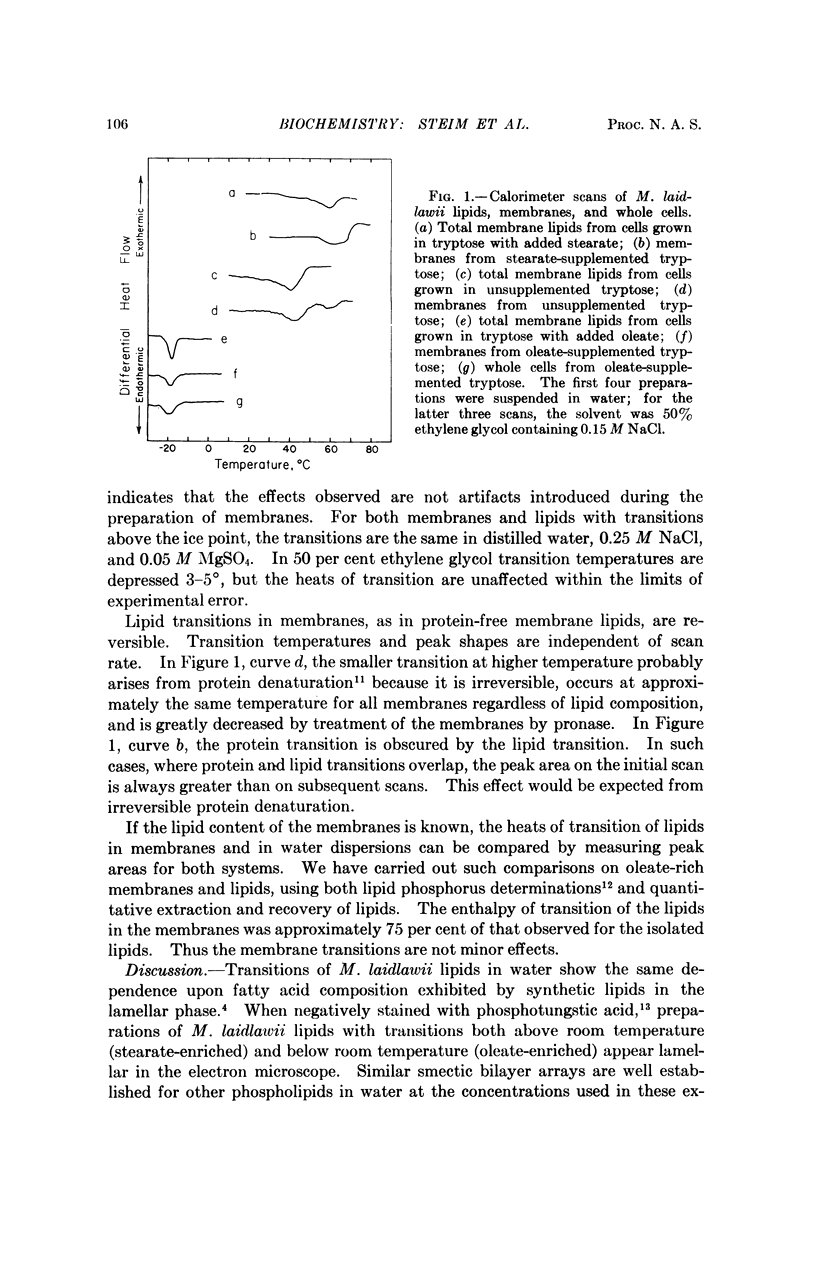
Both membranes of Mycoplasma laidlawii and water dispersions of protein-free membrane lipids exhibit thermal phase transitions that can be detected by differential scanning calorimetry. The transition temperatures are lowered by increased unsaturation in the fatty acid residues, but in each case they are the same for membranes and lipids. The transitions resemble those observed for synthetic lipids in the lamellar phase in water, which arise from melting of the hydrocarbon chains within the phospholipid bilayers. Such melts are cooperative phenomena and would be greatly perturbed by apolar binding to protein. Thus the identity of membrane and lipid transition temperatures suggests that in the membranes, as in water, the lipids are in the bilayer conformation in which the hydrocarbon chains associate with each other rather than with proteins. Observations of morphological changes indicate that osmotic imbalance occurs when the membrane transition temperature exceeds the growth temperature, and that for transport processes to function properly the hydrocarbon chains must be in a liquid-like state.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BANGHAM A. D., HORNE R. W. NEGATIVE STAINING OF PHOSPHOLIPIDS AND THEIR STRUCTURAL MODIFICATION BY SURFACE-ACTIVE AGENTS AS OBSERVED IN THE ELECTRON MICROSCOPE. J Mol Biol. 1964 May;8:660–668. doi: 10.1016/s0022-2836(64)80115-7. [DOI] [PubMed] [Google Scholar]
- Engelman D. M., Terry T. M., Morowitz H. J. Characterization of the plasma membrane of Mycoplasma laidlawii. I. Sodium dodecyl sulfate solubilization. Biochim Biophys Acta. 1967 Jul 3;135(3):381–390. doi: 10.1016/0005-2736(67)90028-4. [DOI] [PubMed] [Google Scholar]
- Korn E. D. Structure of biological membranes. Science. 1966 Sep 23;153(3743):1491–1498. doi: 10.1126/science.153.3743.1491. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Ladbrooke B. D., Williams R. M., Chapman D. Studies on lecithin-cholesterol-water interactions by differential scanning calorimetry and X-ray diffraction. Biochim Biophys Acta. 1968 Apr 29;150(3):333–340. doi: 10.1016/0005-2736(68)90132-6. [DOI] [PubMed] [Google Scholar]
- Lenard J., Singer S. J. Protein conformation in cell membrane preparations as studied by optical rotatory dispersion and circular dichroism. Proc Natl Acad Sci U S A. 1966 Dec;56(6):1828–1835. doi: 10.1073/pnas.56.6.1828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Razin S., Cosenza B. J., Tourtellotte M. E. Variations in Mycoplasma morphology induced by long-chain fatty acids. J Gen Microbiol. 1966 Jan;42(1):139–145. doi: 10.1099/00221287-42-1-139. [DOI] [PubMed] [Google Scholar]
- Razin S., Tourtellotte M. E., McElhaney R. N., Pollack J. D. Influence of lipid components of Mycoplasma laidlawii membranes on osmotic fragility of cells. J Bacteriol. 1966 Feb;91(2):609–616. doi: 10.1128/jb.91.2.609-616.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steim J. M. Differential thermal analysis of protein denaturation in solution. Arch Biochem Biophys. 1965 Dec;112(3):599–604. doi: 10.1016/0003-9861(65)90101-3. [DOI] [PubMed] [Google Scholar]
- Terry T. M., Engelman D. M., Morowitz H. J. Characterization of the plasma membrane of Mycoplasma laidlawii. II. Modes of aggregation of solubilized membrane components. Biochim Biophys Acta. 1967 Jul 3;135(3):391–405. doi: 10.1016/0005-2736(67)90029-6. [DOI] [PubMed] [Google Scholar]


