Abstract
Background
Cancers are heterogeneous comprising of distinct tumor subtypes. Therefore, presenting the burden of cancer in the population and trends over time by these tumor subtypes is important to identify patterns and differences in the occurrence of these subtypes, especially to generalize findings to the US general population.
Methods
Using SEER cancer registry data, we present incidence rates according to subtypes for diagnosis years (1992–2013) among men and women for five major cancer sites: breast (female only), esophagus, kidney and renal pelvis, lung and bronchus, and thyroid. We also describe estimates of 5-year relative survival according to subtypes and diagnosis year (1992–2008). We used Joinpoint models to identify years when incidence rate trends changed slope. Finally, recent 5-year age-adjusted incidence rates (2009–2013) are presented for each subtype by race and age.
Results
Hormone receptor positive and HER-2 negative was the most common subtype (about 74%) of breast cancers. Adenocarcinoma made up about 69% of esophagus cases among men. Adenocarcinoma also is the most common lung subtype (43% in men and 52% in women). Ninety percent of thyroid subtypes were papillary. Distinct incidence and survival patterns emerged by these subtypes over time among men and women.
Conclusions
Histologic or molecular subtype revealed different incidence and/or survival trends that are masked when cancer is considered as a single disease based on anatomic site.
Impact
Presenting incidence and survival trends by subtype, whenever possible, is critical to provide more detailed and meaningful data to patients, providers and the public.
Keywords: Incidence, survival, SEER, subtype, histology
Introduction
The conventional method of reporting population-based cancer statistics, solely by anatomic site, does not leverage advances in characterization of neoplasms based on their detailed biological characteristics.(1) Cancer subtypes are increasingly defined by detailed anatomic site(2, 3), histology(4, 5), or molecular characteristics. (6) Important patterns of cancer occurrence emerge when cancers are examined based on these biological characteristics. Thus, reporting cancer statistics by these clinically important subtypes from population-based registries may identify important trends within the US population or among population subgroups that would otherwise not be evident.
Patterns of disparity can emerge when characterizing cancers based on underlying biology, such as the elevated rate of triple negative breast cancer among African American women, which is a more aggressive subtype than the predominant HR+/HER−breast cancer subtype.(6, 7) Cancer subtypes may also often have distinct risk factors associated with particular histologies. Understanding trends over time and risk may be useful in targeting interventions or prevention strategies to specific subgroups.(8–10) Further, providing data by subtypes such as biomarker presence or genetic test result is essential for determining the impact of major improvements in cancer therapy such as targeted therapies at the population level outside of clinical trials.(11)
The Surveillance Epidemiology and End Results (SEER) Program has traditionally presented cancer statistics by organ site (12), however presenting cancer statistics by tumor subtypes is an important contribution that reflects advances in knowledge about the heterogeneity of cancer and to understand the differential burden of cancer in populations. In this report, SEER cancer incidence and survival data are presented for selected cancer subsites. The objective of this population-based report is to illustrate unique patterns of incidence, time trends, and survival for breast, esophageal, thyroid, lung, kidney and renal pelvis cancer subtypes that represent one change in how SEER data will be presented in the future.
Materials and Methods
Population-based cancer incidence data have been collected by the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) Program since 1973. Incidence and survival data included in this report are from the SEER 13 registries which cover about 13% of the United States population. Cancers from five organ sites diagnosed from 1992 to 2013 were selected to illustrate the potential value in examining tumor subtypes and include: Female breast, esophagus, kidney and renal pelvis, lung and bronchus, and thyroid. Since joint expression of hormone receptor and HER-2 status to classify breast cancer subtypes was not collected until 2010, only female breast cancer cases diagnosed from 2010 to 2013 were included.
The SEER site recode variable based on the World Health Organization International Classification of Disease for Oncology, 3rd edition (ICD-O-3) was used to define the primary site. All cases were included in reporting by the primary site as is done in standard reports.(1, 13) Subtypes for each cancer site that were defined by histologic type and restricted to cases with microscopic confirmation of histology (Supplementary Table 1). Two exceptions were that clinically relevant subtypes for breast cancer were defined by hormone receptor and HER2 status and kidney and renal pelvis were defined by anatomy; so these were not restricted to cases with histologic confirmation. Although kidney and renal pelvis tumors were defined by anatomy, each subsite had a predominant histologic type. The vast majority of renal pelvis tumors were transitional cell carcinomas while kidney NOS tumors were almost all adenocarcinomas and renal cell carcinomas.
Five-year cancer incidence rates (2009–2013) and four-year rates (2010–2013) for breast cancer are presented for each subtype by race and age. Race groups include White, Black, Asian and Pacific Islander (API), and American Indian/Alaska Native (AI/AN). Differences between race and age groups were compared using the relative rate ratio and its 95% confidence interval.(14) All incidence rates were age-adjusted to the 2000 US standard population. The population estimates used as the denominators to calculate incidence rates were a modification of the intercensal and Vintage 2014.(15)
Incidence rates were estimated from 1992 to 2013. In addition, trends and changes over time were estimated using a Joinpoint model.(16) This is a technique that fits a series of joined straight lines on a logarithm scale to the age-adjusted rates over time, a maximum of 4 joinpoints were considered for fitting trends. Breast cancer trends were not estimated since data were only available from 2010. Incidence rates used to calculate trends were also adjusted for reporting delay which may occur because of a lag in reporting to the cancer registry or data corrections.(17) Delay adjustment factors were not available by subtype therefore these rates are adjusted by the overall reporting delay for that primary site. In this report, trends that are reported as increasing or decreasing refer to statistically significant increasing or decreasing trends estimated from the Joinpoint model. Non-statistically significant trends are referred to as stable.
Finally, we present estimates of 5-year relative survival according to cancer subtypes and diagnosis year among men and women. Relative survival was calculated as the ratio of observed all-cause survival to expected survival using the actuarial method in SEER*Stat (18, 19) It represents survival associated with a cancer diagnosis and it is the standard method for reporting cancer-specific survival from registry data as it does not rely on causes of death which may be missing or misclassified (refs). Expected survival rates were calculated using life tables based on individual year 1970–2011, individual age 0–99 years, sex and race (White, Black, Other (American Indian/Alaska Native, Asian/Pacific Islander and were matched on age, sex, year of diagnosis, and race (white, black, and other) to the cancer cohort.(20) Survival analyses included cases diagnosed in 1992–2008 and follow-up until December 31, 2012. Cases diagnosed in 2009 and after are not included because we do not have complete 5 years of follow-up for them. For the same reasons, we were unable to examine 5-year survival data for breast cancer cases diagnosed after 2010.
Results
Female Breast Cancer
Hormone receptor positive and HER-2 negative (HR+/HER2−) breast cancer was the most common subtype comprising 74% of all cases (Supplementary Figure 1). Incidence rates for breast cancer subtype varied by race. For example, white women had the highest incidence rate for this subtype followed by black, API and AI/AN women (Table 1). In contrast, triple-negative breast cancer which made up the second largest component at 11% of cases had the highest rates among black women. The HR+/HER2+ and HR−/HER2+ subtypes had relatively small difference in incidence in white compared to black women.
Table 1.
5-year incidence rates (2009–2013) for men and women by race
| Race | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| White* | Black | AI/AN | API | ||||||||||||
| Total N | N | Rate | N | Rate | RR | 95% CI | N | Rate | RR | 95% CI | N | Rate | RR | 95% CI | |
| Men | |||||||||||||||
| Esophagus | |||||||||||||||
| Adenocarcinoma | 4,639 | 4,372 | 5.7 | 121 | 1.4 | 0.2 | (0.2, 0.3) | 28 | 3.4 | 0.6 | (0.4, 0.9) | 118 | 1.0 | 0.2 | (0.1, 0.2) |
| Squamous Cell | 1,796 | 1,108 | 1.5 | 388 | 4.5 | 3.1 | (2.7, 3.5) | 15 | 2.0 | 1.4 | (0.7, 2.3) | 285 | 2.3 | 1.6 | (1.4, 1.8) |
| Other | 304 | 257 | 0.3 | 24 | 0.3 | 0.9 | (0.6, 1.4) | ~ | 21 | 0.2 | 0.5 | (0.3, 0.8) | |||
| Kidney & Renal Pelvis | |||||||||||||||
| Kidney, NOS | 19,406 | 4,816 | 6.6 | 901 | 10.5 | 1.6 | (1.5, 1.7) | 56 | 8.6 | 1.3 | (1.0, 1.7) | 708 | 6.1 | 0.9 | (0.9, 1.0) |
| Renal Pelvis | 1,082 | 937 | 1.3 | 44 | 0.6 | 0.5 | (0.3, 0.6) | ~ | 96 | 0.8 | 0.6 | (0.5, 0.8) | |||
| Lung & Bronchus | |||||||||||||||
| Squamous | 13,022 | 10,277 | 14.1 | 1547 | 20.6 | 1.5 | (1.4, 1.5) | 90 | 15.3 | 1.1 | (0.9, 1.4) | 1,108 | 9.7 | 0.7 | (0.6, 0.7) |
| Small cell | 6,173 | 5,095 | 6.7 | 549 | 6.8 | 1.0 | (0.9, 1.1) | 47 | 7.1 | 1.1 | (0.8, 1.4) | 482 | 4.1 | 0.6 | (0.6, 0.7) |
| Adenocarcinoma | 22,187 | 16,660 | 22.5 | 2644 | 31.4 | 1.4 | (1.3, 1.5) | 99 | 14.9 | 0.7 | (0.5, 0.8) | 2,784 | 23.2 | 1.0 | (1.0, 1.1) |
| Large cell | 926 | 704 | 0.9 | 143 | 1.7 | 1.8 | (1.5, 2.2) | ~ | 78 | 0.6 | 0.7 | (0.5, 0.9) | |||
| Malignant neoplasm & Carcinoma unspecified |
6,481 | 4,816 | 6.6 | 901 | 10.5 | 1.6 | (1.5, 1.7) | 56 | 8.6 | 1.3 | (1.0, 1.7) | 708 | 6.1 | 0.9 | (0.9, 1.0) |
| Thyroid | |||||||||||||||
| Papillary | 6,137 | 5,100 | 6.5 | 262 | 2.7 | 0.4 | (0.4, 0.5) | 38 | 4.1 | 0.6 | (0.4, 0.9) | 737 | 5.4 | 0.8 | (0.8, 0.9) |
| Follicular | 558 | 433 | 0.6 | 52 | 0.6 | 1.0 | (0.7, 1.4) | ~ | 69 | 0.5 | 1.0 | (0.7, 1.3) | |||
| Medullary | 176 | 148 | 0.2 | 13 | 0.1 | 0.7 | (0.4, 1.3) | ~ | 14 | 0.1 | 0.6 | (0.3, 1.0) | |||
| Anaplastic | 98 | 74 | 0.1 | 8 | 0.1 | 0.9 | (0.4, 2.0) | ~ | 16 | 0.1 | 1.5 | (0.8, 2.6) | |||
| Women | |||||||||||||||
| Breast | |||||||||||||||
| HR+/HER2+ | 10,968 | 8,159 | 9.6 | 1257 | 10.1 | 1.1 | (1.0, 1.1) | 101 | 9.7 | 1.0 | (0.8, 1.2) | 1451 | 9.0 | 0.9 | (0.9, 1.0) |
| HR−/HER2+ | 4,832 | 3,386 | 3.9 | 602 | 4.9 | 1.2 | (1.1, 1.4) | 37 | 3.7 | 0.9 | (0.7, 1.3) | 807 | 4.9 | 1.2 | (1.1, 1.3) |
| HR+/HER2− | 80,317 | 63608 | 72.4 | 7075 | 59.4 | 0.8 | (0.8, 0.8) | 491 | 51.8 | 0.7 | (0.7, 0.8) | 9143 | 56.8 | 0.8 | (0.8, 0.8) |
| Triple negative | 12,143 | 8,555 | 10.0 | 2330 | 18.9 | 1.9 | (1.8, 2.0) | 67 | 7.1 | 0.7 | (0.5, 0.9) | 1191 | 7.4 | 0.7 | (0.7, 0.8) |
| Esophagus | |||||||||||||||
| Adenocarcinoma | 784 | 717 | 0.8 | 37 | 0.3 | 0.4 | (0.3, 0.6) | ~ | 22 | 0.1 | 0.2 | (0.1, 0.3) | |||
| Squamous Cell | 1,057 | 726 | 0.8 | 212 | 1.8 | 2.2 | (1.9, 2.6) | ~ | 114 | 0.7 | 0.9 | (0.7, 1.1) | |||
| Other | 110 | 89 | 0.1 | 15 | 0.1 | 1.5 | (0.8, 2.6) | ~ | ~ | ||||||
| Kidney & Renal Pelvis | |||||||||||||||
| Kidney, NOS | 10,807 | 8,450 | 9.6 | 1,376 | 11.6 | 1.2 | (1.1, 1.3) | 155 | 16.0 | 1.7 | (1.4, 2.0) | 826 | 5.2 | 0.5 | (0.5, 0.6) |
| Renal Pelvis | 815 | 663 | 0.7 | 58 | 0.6 | 0.8 | (0.6, 1.0) | ~ | 91 | 0.6 | 0.8 | (0.6, 1.0) | |||
| Lung | |||||||||||||||
| Squamous | 7,905 | 6,495 | 7.4 | 924 | 8.5 | 1.2 | (1.1, 1.2) | 65 | 8.0 | 1.1 | (0.8, 1.4) | 421 | 2.7 | 0.4 | (0.3, 0.4) |
| Small cell | 6,068 | 5,185 | 5.8 | 566 | 5.0 | 0.9 | (0.8, 0.9) | 47 | 5.1 | 0.9 | (0.6, 1.2) | 270 | 1.7 | 0.3 | (0.3, 0.3) |
| Adenocarcinoma | 24,720 | 18,911 | 21.1 | 2,758 | 23.7 | 1.1 | (1.1, 1.2) | 86 | 9.9 | 0.5 | (0.4, 0.6) | 2,965 | 18.9 | 0.9 | (0.9, 0.9) |
| Large cell | 747 | 585 | 0.7 | 106 | 0.9 | 1.4 | (1.1, 1.7) | ~ | 51 | 0.3 | 0.5 | (0.4, 0.7) | |||
| Malignant neoplasm & Carcinoma unspecified |
5,110 | 3,971 | 4.4 | 697 | 6.0 | 1.4 | (1.3, 1.5) | 31 | 4.0 | 0.9 | (0.6, 1.3) | 411 | 2.7 | 0.6 | (0.5, 0.7) |
| Thyroid | |||||||||||||||
| Papillary | 20,401 | 15,973 | 20.2 | 1,378 | 11.0 | 0.5 | (0.5, 0.6) | 152 | 15.2 | 0.8 | (0.6, 0.9) | 2,898 | 18.2 | 0.9 | (0.9, 0.9) |
| Follicular | 1,381 | 1,025 | 1.3 | 179 | 1.5 | 1.2 | (1.0, 1.4) | 14 | 1.5 | 1.2 | (0.6, 2.1) | 163 | 1.0 | 0.8 | (0.7, 1.0) |
| Medullary | 290 | 237 | 0.3 | 33 | 0.3 | 0.9 | (0.6, 1.3) | ~ | 19 | 0.1 | 0.4 | (0.3, 0.7) | |||
| Anaplastic | 167 | 133 | 0.1 | 11 | 0.1 | 0.7 | (0.3, 1.3) | ~ | 23 | 0.2 | 1.1 | (0.6, 1.7) | |||
Reference group
Counts of 10 or less were suppressed
In addition, incidence peaked among women age 65 to 74 years among all subtypes. The HER2 over-expressing tumors (i.e., HR−/HER2+) were the least common subtypes with fewer observed variations by race or age groups compared to both the HR+/HER2− and triple-negative subtypes.
Esophageal Cancer
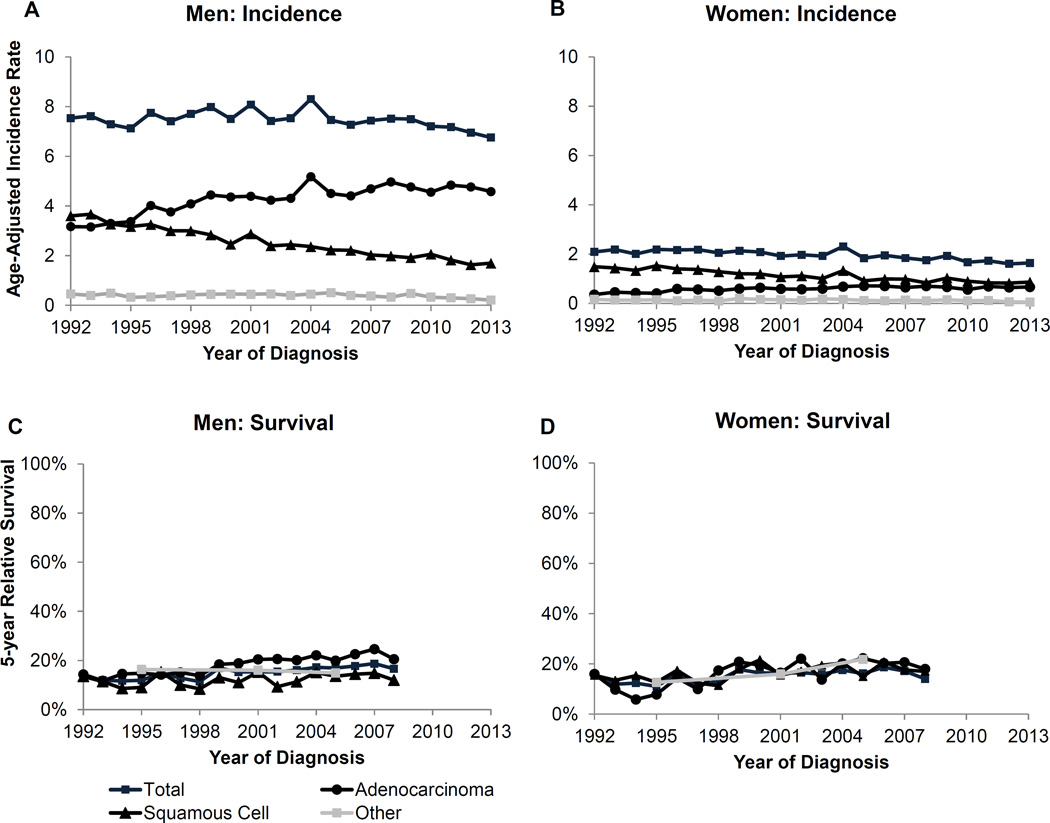
The overall trend for esophageal cancer shows a decline in incidence for both men and women (Figure 1). The 5-year survival is relatively stable over time, however there are differences between the subtypes for both incidence and survival. Specifically, the incidence trends by subtype, in particular for men, revealed an increasing incidence for adenocarcinoma contrasted with a decline for squamous cell and other histologic subtypes (Supplementary Table 2). Incidence for squamous cell carcinoma is declining for women but incidence among the other subtypes remains stable. Among men, who have much higher rates of esophageal cancer compared to women, adenocarcinoma makes up approximately 69% of all esophageal cancers while squamous cell carcinoma makes up the second largest component at 27% (Supplementary Figure 1).
Figure 1.
Esophageal cancer age-adjusted incidence rates and 5-year relative survival over time by subtype for men and women. Survival estimates for Other are based on 5 year groups (1992–1998, 1999–2003, 2004–2008).
The incidence rates by race show that, while white men have the highest rates of adenocarcinoma (5.7 per 100,000), black men have the highest rate of squamous cell carcinoma (4.5 per 100,000; Table 1). Incidence rates for squamous cell carcinoma are also higher among AI/AN and API compared to white men (2.0 and 2.3 vs. 0.3, respectively). Incidence for adenocarcinoma is similar for white and black women but black women had higher rates of squamous cell carcinoma than white and API women (Table 1).
There are also differences in incidence by age for both men and women. Incidence increased dramatically for men by age for both adeno- and squamous cell carcinoma (Table 2). However, men had the highest incidence of adenocarcinoma for all age groups compared to women. There is an increased risk with advancing age in men for both adeno- and squamous cell carcinoma compared with the youngest age group. For women the highest incidence was squamous cell carcinoma and similar to men, the incidence increased after age 55 (Table 2).
Table 2.
5-year incidence rates (2009–2013) for men and women by age
| <55* | 55–64 | 65–74 | 75+ | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | Rate | N | Rate | RR | 95% CI | N | Rate | RR | 95% CI | N | Rate | RR | 95% CI | ||
| Men | |||||||||||||||
| Esophagus | |||||||||||||||
| Adenocarcinoma | 644 | 0.8 | 1,348 | 11.4 | 14.9 | (13.5, 16.4) | 1,381 | 21.6 | 28.2 | (25.7, 31.0) | 1,288 | 27.8 | 36.3 | (33.0, 40.0) | |
| Squamous Cell | 218 | 0.3 | 525 | 4.4 | 17.7 | (15.1, 20.9) | 545 | 8.5 | 34.1 | (29.0, 40.1) | 515 | 11.1 | 44.5 | (37.9, 52.4) | |
| Other | 32 | 0.0 | 85 | 0.7 | 19.0 | (12.5, 29.6) | 95 | 1.5 | 39.2 | (25.9, 60.7) | 96 | 2.1 | 54.3 | (35.9, 83.9) | |
| Kidney & Renal Pelvis | |||||||||||||||
| Kidney, NOS | 5144 | 6.3 | 5503 | 46.6 | 7.4 | (7.1, 7.7) | 5148 | 80.0 | 12.8 | (12.3, 13.3) | 3838 | 83.2 | 13.3 | (12.7, 13.8) | |
| Renal Pelvis | 80 | 0.1 | 214 | 1.8 | 19.3 | (14.8, 25.2) | 325 | 5.1 | 54.3 | (42.3, 70.4) | 472 | 10.1 | 107.6 | (84.6, 138.3) | |
| Lung | |||||||||||||||
| Squamous | 819 | 0.9 | 2,722 | 23.0 | 24.4 | (22.5, 26.4) | 4,532 | 72.1 | 76.5 | (70.9, 82.5) | 5,006 | 108.4 | 114.9 | (106.6, 123.9) | |
| Small cell | 579 | 0.7 | 1,577 | 13.3 | 19.9 | (18.1, 22.0) | 2,236 | 35.0 | 52.4 | (47.7, 57.5) | 1,801 | 39.1 | 58.5 | (53.2, 64.3) | |
| Adenocarcinoma | 2,205 | 2.6 | 5,267 | 44.5 | 17.3 | (16.4, 18.1) | 7,247 | 114.3 | 44.3 | (42.2, 46.5) | 7,571 | 163.9 | 63.5 | (60.5, 66.6) | |
| Large cell | 104 | 0.1 | 238 | 2.0 | 16.6 | (13.1, 21.1) | 310 | 4.8 | 39.9 | (31.8, 50.4) | 277 | 6.0 | 49.7 | (39.5, 62.9) | |
| Malignant neoplasm & Carcinoma unspecified |
608 | 0.7 | 1,518 | 12.8 | 18.1 | (16.4, 19.9) | 2,009 | 31.6 | 44.5 | (40.6, 48.8) | 2,381 | 51.2 | 72.0 | (65.8, 78.9) | |
| Thyroid | |||||||||||||||
| Papillary | 3,352 | 4.2 | 1,415 | 12.0 | 2.9 | (2.7, 3.1) | 986 | 15.3 | 3.7 | (3.4, 3.9) | 479 | 10.4 | 2.5 | (2.3, 2.8) | |
| Follicular | 246 | 0.3 | 126 | 1.1 | 3.5 | (2.8, 4.4) | 112 | 1.8 | 5.8 | (4.6, 7.3) | 81 | 1.8 | 5.9 | (4.5, 7.6) | |
| Medullary | 97 | 0.1 | 34 | 0.3 | 2.4 | (1.6, 3.6) | 28 | 0.4 | 3.6 | (2.3, 5.6) | 21 | 0.5 | 3.8 | (2.3, 6.2) | |
| Anaplastic | 15 | 0.0 | 20 | 0.2 | 10.0 | (4.8, 21.1) | 33 | 0.5 | 29.8 | (15.6, 59.2) | 31 | 0.7 | 39.5 | (20.5, 78.7) | |
| Women | |||||||||||||||
| Breast | |||||||||||||||
| HR+/HER2+ | 4790 | 5.9 | 2,902 | 22.9 | 3.9 | (3.7, 4.1) | 1,911 | 25.4 | 4.3 | (4.1, 4.6) | 1,459 | 21.0 | 3.6 | (3.4, 3.8) | |
| HR−/HER2+ | 1985 | 2.4 | 1,399 | 11.1 | 4.6 | (4.3, 4.9) | 828 | 11.0 | 4.6 | (4.2, 5.0) | 653 | 9.5 | 3.9 | (3.6, 4.3) | |
| HR+/HER2− | 24,066 | 29.1 | 20,796 | 163.5 | 5.6 | (5.5, 5.7) | 19,592 | 262.6 | 9.0 | (8.9, 9.2) | 16,501 | 240.3 | 8.3 | (8.1, 8.4) | |
| Triple negative | 4840 | 5.9 | 3201 | 25.2 | 4.3 | (4.1, 4.4) | 2274 | 30.4 | 5.1 | (4.9, 5.4) | 1923 | 27.6 | 4.7 | (4.4, 4.9) | |
| Esophagus | |||||||||||||||
| Adenocarcinoma | 102 | 0.1 | 181 | 1.4 | 12.0 | (9.3, 15.5) | 187 | 2.6 | 21.6 | (16.8, 27.8) | 318 | 4.3 | 36.3 | (28.8, 45.9) | |
| Squamous Cell | 116 | 0.1 | 239 | 1.9 | 14.4 | (11.5, 18.2) | 321 | 4.4 | 33.7 | (27.1, 42.1) | 390 | 5.5 | 42.2 | (34.1, 52.5) | |
| Other | 10 | 0.0 | 15 | 0.1 | 10.3 | (4.3, 25.7) | 24 | 0.3 | 30.1 | (13.7, 70.6) | 62 | 0.9 | 76.5 | (38.4, 168.0) | |
| Kidney & Renal Pelvis | |||||||||||||||
| Kidney, NOS | 2853 | 3.5 | 2682 | 21.1 | 6.1 | (5.8, 6.4) | 2681 | 36.2 | 10.5 | (9.9, 11.1) | 2697 | 38.9 | 11.3 | (10.7, 11.9) | |
| Renal Pelvis | 48 | 0.1 | 97 | 0.8 | 13.6 | (9.5, 19.7) | 218 | 3.0 | 53.3 | (38.7, 74.6) | 459 | 6.4 | 113.9 | (84.1, 157.2) | |
| Lung | |||||||||||||||
| Squamous | 428 | 0.5 | 1,283 | 10.0 | 20.8 | (18.6, 23.2) | 2,781 | 38.0 | 78.7 | (71.0, 87.3) | 3,435 | 51.3 | 106.1 | (95.8, 117.7) | |
| Small cell | 626 | 0.7 | 1,472 | 11.5 | 16.3 | (14.9, 18.0) | 2,107 | 28.5 | 40.4 | (36.9, 44.3) | 1,884 | 28.6 | 40.4 | (36.9, 44.4) | |
| Adenocarcinoma | 2,851 | 3.3 | 5,440 | 42.7 | 13.0 | (12.4, 13.6) | 7,787 | 106.0 | 32.3 | (30.9, 33.7) | 8,759 | 128.9 | 39.3 | (37.6, 41.0) | |
| Large cell | 88 | 0.1 | 190 | 1.5 | 14.8 | (11.4, 19.3) | 227 | 3.1 | 30.2 | (23.4, 39.1) | 244 | 3.7 | 36.4 | (28.3, 47.1) | |
| Malignant neoplasm & Carcinoma unspecified |
568 | 0.6 | 1,030 | 8.1 | 12.4 | (11.2, 13.8) | 1,499 | 20.5 | 31.6 | (28.7, 34.9) | 2,039 | 29.5 | 45.4 | (41.3, 50.0) | |
| Thyroid | |||||||||||||||
| Papillary | 13,586 | 16.9 | 3,910 | 30.9 | 1.8 | (1.8, 1.9) | 2,244 | 29.6 | 1.8 | (1.7, 1.8) | 1,007 | 15.5 | 0.9 | (0.9, 1.0) | |
| Follicular | 778 | 1.0 | 260 | 2.1 | 2.1 | (1.9, 2.5) | 226 | 3.1 | 3.2 | (2.7, 3.7) | 147 | 2.3 | 2.4 | (2.0, 2.8) | |
| Medullary | 132 | 0.2 | 72 | 0.6 | 3.5 | (2.6, 4.7) | 57 | 0.8 | 4.8 | (3.4, 6.6) | 31 | 0.5 | 2.9 | (1.8, 4.3) | |
| Anaplastic | 9 | 0.0 | 28 | 0.2 | 21.3 | (9.7, 51.6) | 45 | 0.6 | 61.7 | (29.4, 144.3) | 85 | 1.1 | 110.7 | (54.8, 252.0) | |
Reference group
Five-year relative survival for esophageal cancer overall is modestly increasing over time for both men and women (Figure 1). Survival is higher among those with adenocarcinoma compared to squamous cell among men (20,5% vs. 16.6% in 2008). However, there is not any difference in survival among the subtypes for women.
Kidney & Renal Pelvis Cancer
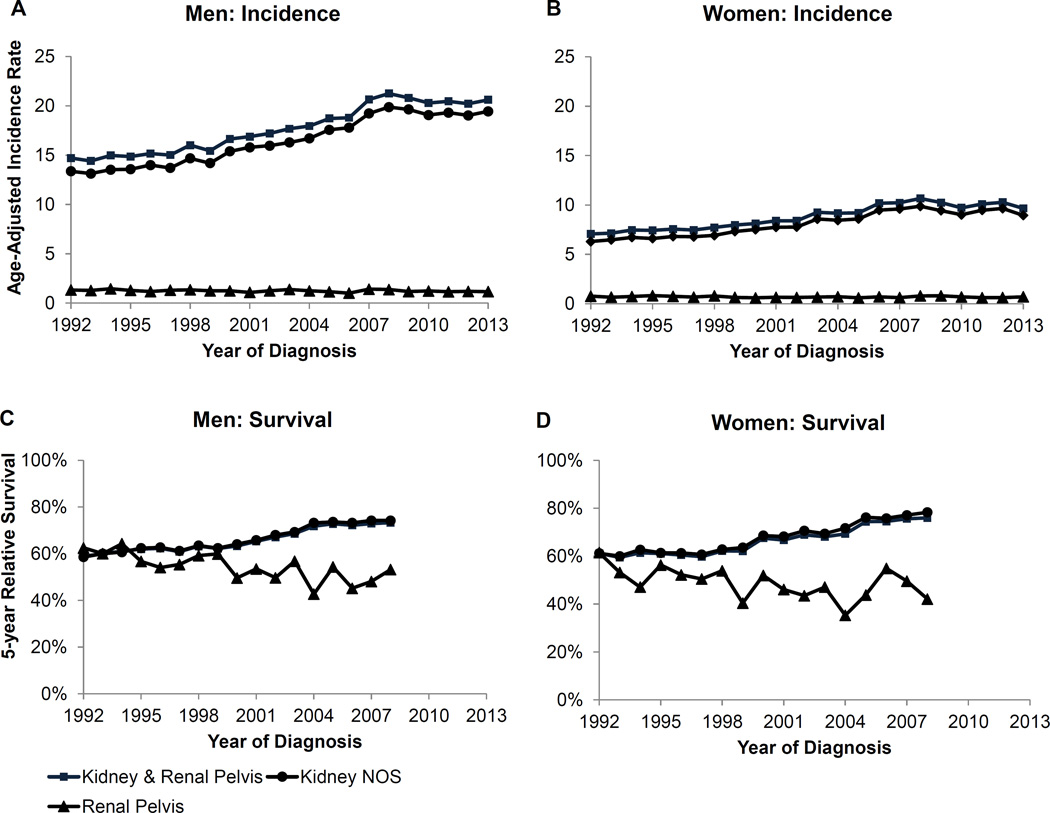
The overall incidence trend for kidney and renal pelvis cancer was increasing from 1992 to 2008 but is now stable among both men and women. The overall trend is driven by the incidence in kidney cancer since these comprise over 90% of cases. Indeed, the incidence trend for kidney cancer followed the same pattern. Cancer of the renal pelvis has a much smaller incidence rate and has been stable since 1992. Although the pattern is similar among men and women, the incidence is lower among women.
The incidence rates of kidney cancer are highest among black and AI/AN men (10.5 and 8.6 per 100,000, respectively; Table 1). Black and AI/AN women also have higher rates compared to white women. In contrast, incidence rates for cancer of the renal pelvis are highest among whites for both men and women. The incidence rates for cancer of both kidney and renal pelvis increase by age among men and women (Table 2). However, the increase by age is far greater for cancer of the renal pelvis compared to kidney.
Five-year relative survival for kidney cancer is increasing over time among both men and women (Figure 2). Specifically, 5-year survival was 58.6% in 1992 and increased to 74,2% in men and increased from 61.2% to 78.3% in women. Survival is lower for cancer of the renal pelvis but variable due to small case counts.
Figure 2.
Kidney and renal pelvis cancer age-adjusted incidence rates and 5-year relative survival for men and women
Lung & Bronchus Cancer
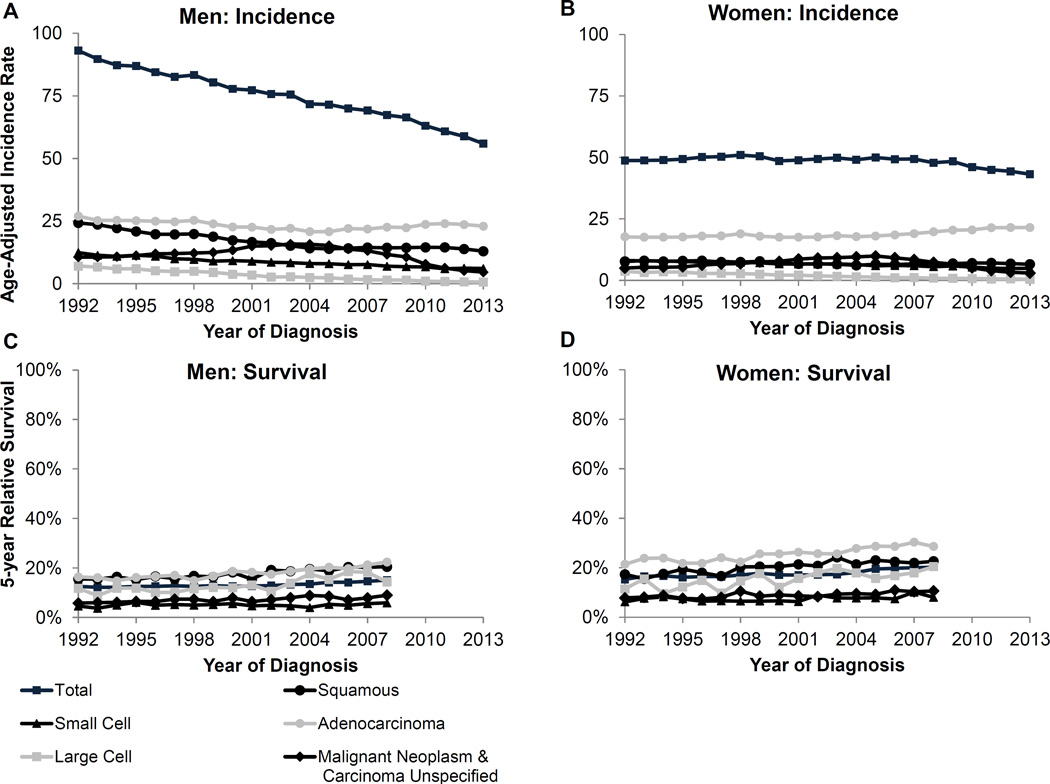
The overall trend for lung cancer is declining among both men and women (Figure 3). This decline is also seen among men and women with small cell, large cell, and malignant neoplasm and carcinoma unspecified subtypes (Supplementary Table 2). However, adenocarcinoma, which is the most common subtype making up 45% of the cases among men and 55% of the cases among women, is increasing among both sexes (Supplementary Figure 1). Squamous cell carcinoma, the second most common histologic subtype comprising 17% of cases among men and 12% among women, is decreasing among men but stable among women.
Figure 3.
Lung and bronchus cancer age-adjusted incidence rates and 5-year relative survival for men and women
The incidence rates by race show that black men have a higher incidence for all subtypes except small cell compared to white men (Table 1). This difference in particularly pronounced for squamous cell, malignant neoplasm and unspecified carcinomas, and large cell carcinoma subtypes. AI/AN men had a higher rate of malignant neoplasms and unspecified carcinoma and a lower rate of adenocarcinoma compared to white men. API men had overall lower rates for all lung subtypes compared to white men with the exception of adenocarcinoma. Compared to white women, black women have higher rates for large cell carcinoma, malignant neoplasms and unspecified carcinoma, and squamous cell carcinoma (Table 1). Incidence rates for adenocarcinoma are nearly equivalent for black and white women. Small cell carcinoma was higher among white women compared with black women. AI/AN and API women had lower rates for all subtypes compared to white women with the only exception of a higher rate of squamous cell carcinoma among AI/AN compared to white women. Among both men and women, incidence rates of lung subtypes by age show each of the subtype incidence rates increases with age (Table 2). Lung cancer incidence for all subtypes is highest among men and women age 75 and older.
Five-year relative survival by lung cancer histologic subtype indicates an increase in survival for each subtype among both men and women (Figure 3), although survival among women is generally higher than among men. Among men, those with the adeno- and squamous cell carcinoma had the highest 5-year relative survival (22.4% and 20.5% in 2008, respectively). A similar pattern was seen for among women with a 5-year relative survival 28.6% in 2008 for adenocarcinoma and 22.6% for squamous cell carcinoma.
Thyroid Cancer
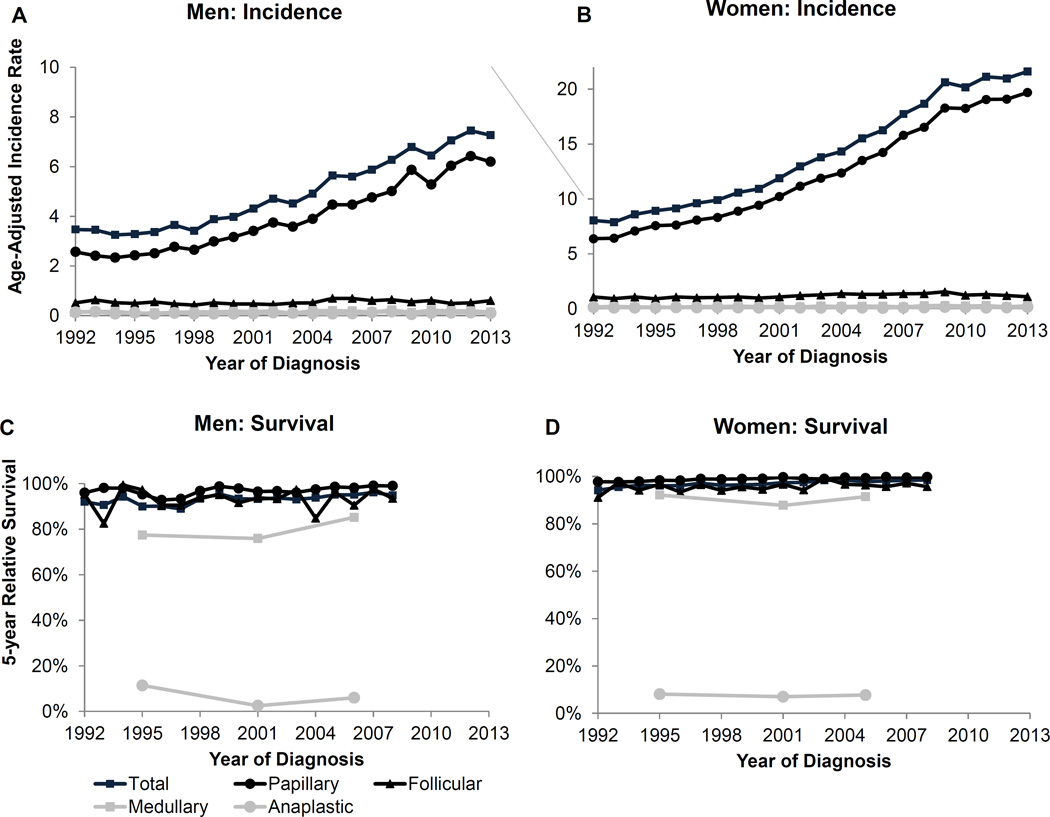
The overall incidence trend for thyroid cancer is increasing among men and women (Figure 4). However, the overall trend is driven by the papillary subtype since it accounts for about 90% of the cases among both men and women (Table 1). Indeed, incidence trends for the papillary subtype are increasing among men and women (Supplementary Table 2). The less common subtypes, in descending order of incidence, are follicular, medullary and anaplastic. The incidence rates for these subtypes are low, however, medullary thyroid cancer has been increasing among men and women. The trend for anaplastic thyroid cancer is stable among men and women.
Figure 4.
Thyroid cancer age-adjusted incidence rates and 5-year relative survival over time by subtype for men and women. Survival estimates for Medullary and Anaplastic are based on 5 year groups (1992–1998, 1999–2003, 2004–2008).
Among men the highest incidence rates for the papillary subtype occurred among whites and APIs with lower rates among blacks and AI/ANs (Table 1). Incidence rates of papillary subtype rose with age, peaked among men 65 to 74 years then dropped among men 75+ years of age (Table 2). Incidence rates for other thyroid cancer subtypes were lower and typically increased with age.
Among women incidence rates for papillary subtype were higher compared to those of men, with higher rates among whites and APIs than among blacks and AI/ANs (Table 1). Rates across racial and ethnic groups were similar for non-papillary subtypes. Incidence rates of papillary subtype peaked among women at 55 to 64 years (Table 2). Incidence rates for follicular and medullary subtypes peaked at 65 to 74 years and those for anaplastic peaked at 75+ years.
Among both men and women, overall five-year relative survival is driven by the papillary subtype for which the survival is very high (Figure 4). Specifically, 99% in 2008 for men and 99.8% for women. However, survival from the other subtypes is poorer including a very poor survival from the anaplastic subtype (<10% five-year survival).
Discussion
For the five cancer sites presented in this report, the analysis by histologic or molecular subtype reveal important differences in incidence trends that are not apparent when only the anatomic site is considered. For example, triple negative breast cancer occurs at a higher rate among black women than white women. Esophageal squamous cell carcinoma incidence rates decreased in men over time as adenocarcinomas emerged to become the predominant esophageal cancer subtype among men in the mid-1990s. Similarly, the rising incidence of papillary thyroid cancer explains almost the entire increase in thyroid cancer incidence.
Differences in survival also emerge when the subtypes are examined. Specifically, papillary thyroid cancer subtypes have favorable survival compared to less common subtypes of these anatomic sites. Patients with small cell carcinoma of the lung had worse survival than those diagnosed with squamous-, adeno-, and other specified carcinomas of the lung. These results clearly illustrate the importance of providing data according to more clinically relevant categories for assessing risk and outcomes, as well as for investigating health disparities. These results can also inform areas of need for targeted drug development and highlight areas where orphan drugs may be required to improve survival for specific cancer subtypes.
Changes in risk factors over time may affect the incidence trends differentially by subtype. For example, tobacco smoking is most strongly related to squamous cell subtype of esophageal and lung cancer (21–23). The declining prevalence of smoking over time in the US general population (24) may have contributed to the decreasing incidence in the squamous cell subtype of esophageal and lung cancer over time. However, changes in tobacco blends and inhalation depth may be resulting in increasing peripheral adenocarcinomas of the lung (25–27). Thus the reporting by subtype is necessary to connect risk factor behavior in the population with clinical awareness and surveillance.
Cancers associated with excess weight such as adenocarcinoma of the esophagus, kidney cancer and postmenopausal breast cancer (28, 29) showed increasing trends among both men and women. Some of the observed patterns may reflect an increase in the rates of overweight and obesity starting from the 1970s.(30) The prevalence of overweight and obesity has slowed since 2000 (31) and may be leveling off in more recent years (32). While the trends for these cancers are influenced by other risk factors, the high prevalence of excess weight likely contributed to these increases (33).
Finally, cancer screening may differentially affect subtype detection. For example, HR- breast cancers are twice as likely to be missed by mammographic screening compared with HR+ breast cancers (6, 34). There are other risk factors such as reproductive history, lactation, weight, physical activity, and postmenopausal hormone use that could explain the apparent differences in breast cancer incidence rates (35) Papillary thyroid cancers are also differentially detected by screening compared to the other more fatal subtypes (36, 37). Also, some of the increasing rates in kidney cancer may be due to an increase in incidental diagnoses resulting from diagnostic imaging for other health conditions unrelated to symptoms of kidney cancer (38, 39). Risk factors are only one component contributing to changes in incidence rates over time. Other factors include improvements in screening methods and changes in screening behaviors. Other factors such as changes in disease classification or data collection procedures, and delays in cancer reporting can also affect observed trends over time.
Our results illustrate the direction that population-based cancer surveillance must take in order to support contemporary cancer research and to provide the most informative information to both clinicians and patients. According to the classical paradigm of cancer surveillance, there are approximately 60 different organs where cancer could develop. More recently, it has been estimated that there are several hundred cancer subtypes. Our understanding of cancer diversity is almost certain to expand with discoveries based on proteomic, genomic and methylomic characterization. The SEER cancer registry program recognizes the need to integrate these advances to support cancer research initiatives. This will enable estimation of the burden of specific cancer subtypes, disparities in the population, and the potential benefits of targeted therapies.
Strengths of the present study include the ability to examine long term incidence and survival trends for cancer subtypes, including by age, race and sex. However, there are also challenges as we move to presenting data in more meaningful categories. These challenges will require both a clear understanding of the data collected including how it changes over time and how the use and development of new methods for surveillance affect the data. For example, cases can be assigned to different histology codes over time based on new biological knowledge and these changes can complicate the interpretation of trends. There have been changes in histology coding for lung cancer that affect trends by moving cases from one histologic subtype to another. (23) Previous work has presented approaches to obtain consistent trends over time through grouping of codes (23) or through imputation.(40, 41) These methods aim to prevent artificial changes in the trends over time created by coding changes. Researchers analyzing subtypes need to be familiar with the changes in histology classification over the time to ensure the proper interpretation of trends. Since the classification of histologic codes in an analysis is at the discretion of the authors, it is of upmost importance that when publishing results by histologic subtype that the definitions are clearly reported so that the results can be compared across studies.
Other potential challenges are that certain cases that have not been microscopically confirmed and cannot be classified into a histologic subtype and cases that are microscopically confirmed but are a nonspecific histology and therefore excluded from this analysis. The proportion of cases not microscopically confirmed is small for esophageal and thyroid cancer (<4%) and moderate for kidney and lung cancer (10% each), however these proportions are stable over time and so should not impact the trends. Similarly, the proportion of cases excluded from the analysis is small (<5%) and also stable over time.
In addition, even when microscopically confirmed the information to define the subtype may be missing or unknown for some organ sites resulting in cases being excluded from the subtype analysis, particularly in the early years of reporting. In this analysis the proportion of cases microscopically confirmed and excluded from the analysis is small (<5%) and also stable over time. One approach that has been used to address this issue is imputation of missing values.(6, 7) When interpreting trends by subtype the impact of cases unable to be categorized should be considered by either including the trend for unknown subtype or applying statistical methods such as imputation to assign cases to specific subtypes.
Despite the challenges described above, presenting trends by more clinically relevant subcategories is useful to provide more detailed and meaningful information to patients, providers and the public in general. This report highlights that for five cancer sites analysis by histologic or molecular subtype reveals different incidence and/or survival trends that are masked when only the generic organ site trends are considered. As precision medicine and targeted therapies are developed it will be increasingly necessary to report population-based cancer rates and trends by clinically meaningful subgroups so that cancer incidence and outcomes remain relevant to patients and researchers.
Supplementary Material
Acknowledgments
The authors would like to thank SEER registries at the following locations: Atlanta, Connecticut, Detroit, Hawaii, Iowa, New Mexico, San Francisco-Oakland, Seattle-Puget Sound, Utah, Los Angeles, San Jose-Monterey, Rural Georgia, and the Alaska. The authors also thank Martin Krapcho and Ken Bishop of Information Management Systems, Inc. (IMS) for their help in creating databases and assistance with programming.
Funding: This study was supported by the Surveillance Research Program in the Division of Cancer Control and Population Sciences at the National Cancer Institute.
Footnotes
Conflicts of interest: None
References
- 1.Ryerson AB, Eheman CR, Altekruse SF, Ward JW, Jemal A, Sherman RL, et al. Annual Report to the Nation on the Status of Cancer, 1975–2012, featuring the increasing incidence of liver cancer. Cancer. 2016 doi: 10.1002/cncr.29936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Altekruse SF, Dickie L, Wu XC, Hsieh MC, Wu M, Lee R, et al. Clinical and prognostic factors for renal parenchymal, pelvis, and ureter cancers in SEER registries: collaborative stage data collection system, version 2. Cancer. 2014;120(Suppl 23):3826–3835. doi: 10.1002/cncr.29051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sirri E, Castro FA, Kieschke J, Jansen L, Emrich K, Gondos A, et al. Recent Trends in Survival of Patients With Pancreatic Cancer in Germany and the United States. Pancreas. 2016 doi: 10.1097/MPA.0000000000000588. [DOI] [PubMed] [Google Scholar]
- 4.Devesa SS, Blot Wj Fau - Fraumeni JF, Jr, Fraumeni JF., Jr Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. [PubMed] [Google Scholar]
- 5.Aschebrook-Kilfoy B, Ward Mh Fau - Sabra MM, Sabra Mm Fau - Devesa SS, Devesa SS. Thyroid cancer incidence patterns in the United States by histologic type, 1992–2006. doi: 10.1089/thy.2010.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Howlader N, Altekruse SF, Li CI, Chen VW, Clarke CA, Ries LA, et al. US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J Natl Cancer Inst. 2014;106 doi: 10.1093/jnci/dju055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kohler BA, Sherman RL, Howlader N, Jemal A, Ryerson AB, Henry KA, et al. Annual Report to the Nation on the Status of Cancer, 1975–2011, Featuring Incidence of Breast Cancer Subtypes by Race/Ethnicity, Poverty, and State. J Natl Cancer Inst. 2015;107:djv048. doi: 10.1093/jnci/djv048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lacey JV, Jr, Swanson CA, Brinton LA, Altekruse SF, Barnes WA, Gravitt PE, et al. Obesity as a potential risk factor for adenocarcinomas and squamous cell carcinomas of the uterine cervix. Cancer. 2003;98:814–821. doi: 10.1002/cncr.11567. [DOI] [PubMed] [Google Scholar]
- 9.Altekruse SF, Lacey JV, Jr, Brinton LA, Gravitt PE, Silverberg SG, Barnes WA, Jr, et al. Comparison of human papillomavirus genotypes, sexual, and reproductive risk factors of cervical adenocarcinoma and squamous cell carcinoma: Northeastern United States. Am J Obstet Gynecol. 2003;188:657–663. doi: 10.1067/mob.2003.132. [DOI] [PubMed] [Google Scholar]
- 10.Lacey JV, Jr, Frisch M, Brinton LA, Abbas FM, Barnes WA, Gravitt PE, et al. Associations between smoking and adenocarcinomas and squamous cell carcinomas of the uterine cervix (United States) Cancer Causes Control. 2001;12:153–161. doi: 10.1023/a:1008918310055. [DOI] [PubMed] [Google Scholar]
- 11.Collins FS, Varmus H. A New Initiative on Precision Medicine. New England Journal of Medicine. 2015;372:793–795. doi: 10.1056/NEJMp1500523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Howlader N, Noone AM, Krapcho M, Miller D, Bishop K, Altekruse SF, et al. Bethesda, MD: 2016. Apr, SEER Cancer Statistics Review, 1975–2013, National Cancer Institute. http://seer.cancer.gov/csr/1975_2013/, based on November 2015 SEER data submission, posted to the SEER web site. [Google Scholar]
- 13.Howlader N, Noone AM, Krapcho M, Garshell J, Miller D, Altekruse SF, et al. Bethesda, MD: 2015. Apr, SEER Cancer Statistics Review, 1975–2012, National Cancer Institute. http://seer.cancer.gov/csr/1975_2012/, based on November 2014 SEER data submission, posted to the SEER web site. [Google Scholar]
- 14.Fay MP. Approximate confidence intervals for rate ratios from directly standardized rates with sparse data. Communications in Statistics - Theory and Methods. 1999;28:2141–2160. [Google Scholar]
- 15.Surveillance, Epidemiology, and End Results (SEER) Program; National Cancer Institute. Population estimates used in NCI's SEER*Stat software. [Last accessed: September 20, 2016]; Available from: http://seer.cancer.gov/popdata/methods.html.
- 16.Kim HJ, Fay MP, Feuer EJ, Midthune DN. Permutation tests for joinpoint regression with applications to cancer rates. Stat Med. 2000;19:335–351. doi: 10.1002/(sici)1097-0258(20000215)19:3<335::aid-sim336>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 17.Clegg LX, Feuer EJ, Midthune DN, Fay MP, Hankey BF. Impact of reporting delay and reporting error on cancer incidence rates and trends. J Natl Cancer Inst. 2002;94:1537–1545. doi: 10.1093/jnci/94.20.1537. [DOI] [PubMed] [Google Scholar]
- 18.Ederer F, Axtell LM, Cutler SJ. The relative survival rate: a statistical methodology. Natl Cancer Inst Monogr. 1961;6:101–121. [PubMed] [Google Scholar]
- 19.Surveillance Research Program; National Cancer Institute. SEER*Stat Program Version 8.3.2. 2016 Apr; Available from: http://seer.cancer.gov/seerstat/
- 20.Surveillance Research Program; National Cancer Institute. Expected Survival Life Tables. [Accessed September 20, 2016]; Available from: http://seer.cancer.gov/expsurvival/
- 21.Cook MB, Chow WH, Devesa SS. Oesophageal cancer incidence in the United States by race, sex, and histologic type, 1977–2005. Br J Cancer. 2009;101:855–859. doi: 10.1038/sj.bjc.6605246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Freedman ND, Abnet CC, Leitzmann MF, Mouw T, Subar AF, Hollenbeck AR, et al. A prospective study of tobacco, alcohol, and the risk of esophageal and gastric cancer subtypes. Am J Epidemiol. 2007;165:1424–1433. doi: 10.1093/aje/kwm051. [DOI] [PubMed] [Google Scholar]
- 23.Lewis DR, Check DP, Caporaso NE, Travis WD, Devesa SS. US lung cancer trends by histologic type. Cancer. 2014;120:2883–2892. doi: 10.1002/cncr.28749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Prevention CfDCa. Cancer and Tobacco Use. [2016 November 16, 2016];Vital Signs. 2016 Nov 10; [Google Scholar]
- 25.Thun MJ, Carter BD, Feskanich D, Freedman ND, Prentice R, Lopez AD, et al. 50-year trends in smoking-related mortality in the United States. N Engl J Med. 2013;368:351–364. doi: 10.1056/NEJMsa1211127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thun MJ, Henley SJ, Calle EE. Tobacco use and cancer: an epidemiologic perspective for geneticists. Oncogene. 2002;21:7307–7325. doi: 10.1038/sj.onc.1205807. [DOI] [PubMed] [Google Scholar]
- 27.Thun MJ, Lally CA, Flannery JT, Calle EE, Flanders WD, Heath CW., Jr Cigarette smoking and changes in the histopathology of lung cancer. J Natl Cancer Inst. 1997;89:1580–1586. doi: 10.1093/jnci/89.21.1580. [DOI] [PubMed] [Google Scholar]
- 28.Eheman C, Henley SJ, Ballard-Barbash R, Jacobs EJ, Schymura MJ, Noone AM, et al. Annual Report to the Nation on the status of cancer, 1975–2008, featuring cancers associated with excess weight and lack of sufficient physical activity. Cancer. 2012;118:2338–2366. doi: 10.1002/cncr.27514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Research WCRFAIfC. Washington, DC: American Institute for Cancer Research; 2007. Food, Nutrition, Physical Activity, and the Prevention of Cancer: a Global Perspective. [Google Scholar]
- 30.Flegal KM, Carroll MD, Kuczmarski RJ, Johnson CL. Overweight and obesity in the United States: prevalence and trends, 1960–1994. Int J Obes Relat Metab Disord. 1998;22:39–47. doi: 10.1038/sj.ijo.0800541. [DOI] [PubMed] [Google Scholar]
- 31.Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA. 2012;307:491–497. doi: 10.1001/jama.2012.39. [DOI] [PubMed] [Google Scholar]
- 32.Flegal KM, Carroll MD, Ogden CL, Johnson CL. PRevalence and trends in obesity among us adults, 1999–2000. JAMA. 2002;288:1723–1727. doi: 10.1001/jama.288.14.1723. [DOI] [PubMed] [Google Scholar]
- 33.Wang Y, Beydoun MA. The obesity epidemic in the United States--gender, age, socioeconomic, racial/ethnic, and geographic characteristics: a systematic review and meta-regression analysis. Epidemiol Rev. 2007;29:6–28. doi: 10.1093/epirev/mxm007. [DOI] [PubMed] [Google Scholar]
- 34.Porter PL, El-Bastawissi AY, Mandelson MT, Lin MG, Khalid N, Watney EA, et al. Breast tumor characteristics as predictors of mammographic detection: comparison of interval- and screen-detected cancers. J Natl Cancer Inst. 1999;91:2020–2028. doi: 10.1093/jnci/91.23.2020. [DOI] [PubMed] [Google Scholar]
- 35.Toriola AT, Colditz GA. Trends in breast cancer incidence and mortality in the United States: implications for prevention. Breast Cancer Res Treat. 2013;138:665–673. doi: 10.1007/s10549-013-2500-7. [DOI] [PubMed] [Google Scholar]
- 36.Brito JP, Morris JC, Montori VM. Thyroid cancer: zealous imaging has increased detection and treatment of low risk tumours. BMJ. 2013;347:f4706. doi: 10.1136/bmj.f4706. [DOI] [PubMed] [Google Scholar]
- 37.Davies L, Welch HG. Current thyroid cancer trends in the United States. JAMA Otolaryngol Head Neck Surg. 2014;140:317–322. doi: 10.1001/jamaoto.2014.1. [DOI] [PubMed] [Google Scholar]
- 38.Jayson M, Sanders H. Increased incidence of serendipitously discovered renal cell carcinoma. Urology. 1998;51:203–205. doi: 10.1016/s0090-4295(97)00506-2. [DOI] [PubMed] [Google Scholar]
- 39.Miller DC, Ruterbusch J, Colt JS, Davis FG, Linehan WM, Chow WH, et al. Contemporary clinical epidemiology of renal cell carcinoma: insight from a population based case-control study. J Urol. 2010;184:2254–2258. doi: 10.1016/j.juro.2010.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yu M, Feuer EJ, Cronin KA, Caporaso NE. Use of multiple imputation to correct for bias in lung cancer incidence trends by histologic subtype. Cancer Epidemiol Biomarkers Prev. 2014;23:1546–1558. doi: 10.1158/1055-9965.EPI-14-0130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Howlader N, Noone AM, Yu M, Cronin KA. Use of imputed population-based cancer registry data as a method of accounting for missing information: application to estrogen receptor status for breast cancer. Am J Epidemiol. 2012;176:347–356. doi: 10.1093/aje/kwr512. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.