Abstract
The sexually dimorphic expression of H19/IGF2 is evolutionarily conserved. To investigate whether the expression of H19/IGF2 in the female porcine eye is sex-dependent, gene expression and methylation status were evaluated using quantitative real-time PCR (qPCR) and bisulfite sequencing PCR (BSP). We hypothesized that H19/IGF2 might exhibit a different DNA methylation status in the female eye. In order to evaluate our hypothesis, parthenogenetic (PA) cells were used for analysis by qPCR and BSP. Our results showed that H19 and IGF2 were over-expressed in the female eye compared with the male eye (3-fold and 2-fold, respectively). We observed a normal monoallelic methylation pattern for H19 differentially methylated regions (DMRs). Compared with H19 DMRs, IGF2 DMRs showed a different methylation pattern in the eye. Taken together, these results suggest that elevated expression of H19/IGF2 is caused by a specific chromatin structure that is regulated by the DNA methylation status of IGF2 DMRs in the female eye.
Keywords: H19/IGF2, gene expression, DNA methylation, parthenogenetic, pig
Introduction
The imprinted gene IGF2 has been reported to be paternally expressed and it is a growth factor in mammals (Barlow et al., 1991). The H19 gene, which is maternally expressed, encodes a non-coding RNA (Bartolomei et al., 1991; Brunkow and Tilghman, 1991; Gabory et al., 2006). Numerous studies have demonstrated that the parent-specific expression of H19/IGF2 depends on differentially methylated regions (DMRs) (Park et al., 2011; Thorvaldsen et al., 2006). Studies have also shown that DMRs play an important role in the regulation of gene expression in the H19/IGF2 cluster within promoters and enhancers (Braunschweig et al., 2011).
It has been reported that the H19 DMRs in mice contain three CTCF-binding motifs (Han et al., 2008). Through CTCF binding to the DMRs, IGF2 is inactivated and maternal H19 is expressed. However, when that binding is prevented, paternal IGF2 is expressed. This is known as parent-specific chromatin loops (Wei et al., 2005), and, as part of this mechanism, IGF2 DMRs are considered an epigenetic switch that regulates H19 and IGF2 expressions.
H19/IGF2 are reciprocally imprinted in most tissues, although they are expressed at different levels, a fact which contributes to sex-bias in the female mouse eye, but not in other tissues (Hudson et al., 2010; Park et al., 2009; Reinius and Kanduri, 2013). In this study, we investigated the expression of H19 and IGF2 in the eye of female pigs to detect sex-specific imprinting effects. We hypothesized that the DNA methylation patterns of the H19 DMRs and IGF2 DMRs interact in the eye, and that this might regulate H19 and IGF2 expression through a specific chromatin structure. To test this hypothesis, we analyzed the expression pattern and methylation status of H19/IGF2 in parthenogenetic (PA) cells by qPCR and bisulfite sequencing PCR (BSP).
Materials and Methods
Ethics statement
Pig experiments were carried out in accordance with the guidelines on animal care and use of animals in research, which were approved by the Animal Care and Use Committee of Jilin University, Changchun, China.
Sample collection
The tissues of porcine eye were obtained from three female and three male newborn piglets. They were, rinsed in Dulbecco's phosphate-buffered saline (PBS, Sigma, St. Louis, MO, USA), immediately frozen in liquid nitrogen and stored in −80 °C until use.
The protocol for cell harvesting was previously described in detail (Han et al., 2013; Wang et al., 2014b). Briefly, matured eggs were electrically and parthenogenetically activated (PA) by two direct current pulses of 1.2 kV/cm for 30 μs using a BTX Electro Cell Manipulator 2001 (BTX, San Diego, CA) and then cultured in cytochalasin B to suppress the extrusion of the second polar body. PA embryos were then transferred into the oviduct of a surrogate sow on the next day. PA fetuses were collected from each uterine horn at day 28.
Gene expression analysis
Total RNA was isolated from porcine eye cells using the TRNzol reagent (TIANGEN, Beijing, China) according to the instructions of the manufacturer. The RNA was treated with DNase I (Fermentas) and reverse transcribed to cDNA using the BioRT cDNA first strand synthesis kit (Bioer Technology, Hangzhou, China). Quantitative real-time PCR was performed to determine the expression of H19 and IGF2. The primer sequences are listed in Table 1. Quantitative gene expression analysis was carried out according to the manufacturer's instructions using the BIO-RAD iQ5 Multicolor Real-Time PCR Detection System with the BioEasy SYBR Green I Real Time PCR Kit (Bioer Technology, Hangzhou, China). The thermal cycling conditions were 95 °C for 3 min, followed by 40 cycles of denaturation at 95 °C for 10 s, annealing at 55 °C for 15 s, and extension at 72 °C for 30 s. The 2-ΔΔCT formula was used to determine relative gene expression, which was normalized to the quantity of GAPDH mRNA. All experiments were repeated three times for each gene. Data are expressed as means ± S.E.M.
Table 1. Primers for qRT-PCR analysis.
| Genes | Annealing (°C) | Primer sequences (5′ → 3′) | Size (bp) | Reference/accession |
|---|---|---|---|---|
| H19 | 55 | F: CTCAAACGACAAGAGATGGT | 122 | (Park et al., 2011) |
| R: AGTGTAGTGGCTCCAGAATG | ||||
| IGF2 | 55 | F: AAGAGTGCTCTTCCGTAG | 156 | (Park et al., 2011) |
| R: TGTCATAGCGGAAGAACTTG | ||||
| GAPDH | 55 | F: ATTCCACGGCACAGTCAAGG | 120 | NM_001206359.1 |
| R: ACATACTCAGCACCAGCATCG |
Methylation pattern of H19 DMR3, IGF2 DMR1/2
The procedure for BSP was previously described by Clark et al. (1994). Briefly, genomic DNA of porcine eye cells was isolated using the TIANamp Genomic DNA Kit (TIANGEN, Beijing, China) and treated with the CpGenome Turbo Bisulfite Modification Kit (Millipore) according to the manufacturer's instructions. Nested PCR using Taq Plus PCR Master Mix (TIANGEN, Beijing, China) was performed for the amplification of the H19 DMR3 and IGF2 DMR1/2. The primer sequences are listed in Table 2. PCR products were purified and subjected to BSP (10 positive clones) and Combined Bisulfite Restriction Analysis (COBRA), which have been described in a previous study (Watanabe et al., 2010; Huntriss et al., 2013).
Table 2. Primers for bisulfite sequencing PCR analysis.
| Sequence | Annealing (°C) | Primer sequences (5′ → 3′) | Size (bp) | Reference/ |
|---|---|---|---|---|
| accession | ||||
| H19 DMR1 | 55 °C | Outer F:AGGAGATTAGGTTTAGGGGAAT | 260 | (Park et al., 2011) |
| R: CTACCACTCCCCTCATACCTAA | ||||
| Inner F: AGTGTTTGGGGATTTTTTTTTT | ||||
| R: CACCCCATCCCCTAAATAACCCTC | ||||
| H19 DMR2 | 55°C | Outer F:TATGTTTAGGGGTGATAAAAGT | 216 | (Park et al., 2011) |
| R: CCCCACTTCTACAATTCAAC | ||||
| Inner F: AGGTGTTATTTTGTTTGTTGGT | ||||
| R:ATAAAATAACCTAAAAAAACTCAA | ||||
| H19 DMR3 | 55 °C | Outer F: GGTTTTAGGGGGATATTTTTT | 208 | (Park et al., 2011) |
| R:TTAAAAAAACATTACTTCCATATAC | ||||
| Inner F: GATTTTTAGGTTTGTTATTATTT | ||||
| R: CAAATATTCAATAAAAAAACCC | ||||
| IGF2 DMR1 | 55 °C | Outer F:GGAAGTTTTGTTTAGTTGGTTTTT | 389 | (Braunschweig et al., 2011) |
| R:AAATCTAAAAACAAAAACAAAAAAC | ||||
| Inner F:GTTAGGTTTAGTGTTTAGTATTGGTTT | ||||
| R: TCCAAAACCAAACCTCTCCTAC | ||||
| IGF2 DMR2 | 55 °C | Outer F:GTTAGGGGGGGGTTTGGTTTTTTAG | 268 | (Braunschweig et al., 2011) |
| R: CTCCCCTTAATCCTATAAAACTTCC | ||||
| Inner F: GGTAGTATTTGAAGTTTAAGAG | ||||
| R: CTATAAAACTTCCAAACAAACC |
Statistical analysis
The obtained qRT-PCR and BSP data were analyzed by t-tests using SPSS 16.0 software (SPSS Inc., Chicago, IL, USA). A value of p < 0.05 was considered statistically significant. The methylation status was analyzed using the online BiQ Analyzer software (http://biq-analyzer.bioinf.mpi-inf.mpg.de/tools/MethylationDiagrams/index.php).
Results
Imprinted gene expression analysis of H19 and IGF2
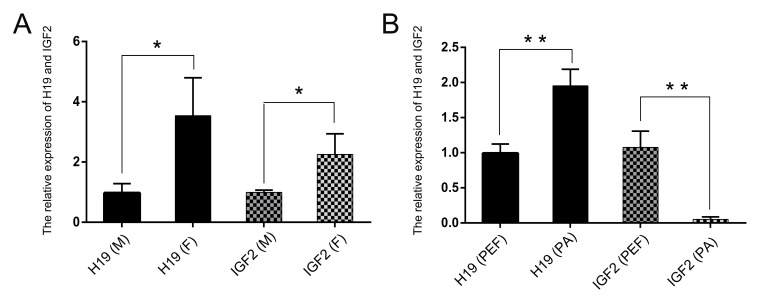
The qPCR results showed that the expression of IGF2 was up-regulated 2-fold in female eyes compared with male eyes. Furthermore, H19 expression was also up-regulated 3-fold (Figure 1A).
Figure 1. Relative expression levels of H19/IGF2. qRT-PCR analysis of H19/IGF2 in (A) male and female eye, (B) porcine embryonic fibroblast (PEF) and parthenogenetic (PA) cells. F: female; M: male. Data are reported as means ± SEM (n = 3). * p < 0.05, ** p < 0.01.
In order to investigate if the elevated expression was regulated by a specific chromatin structure in H19/IGF2 clusters in female eyes, PA cells were analyzed using qPCR. As expected, the expression of H19 in PA cells was up-regulated, while IGF2 expression was significantly reduced compared with porcine embryonic fibroblast (PEF) cells (Figure 1B).
Methylation analysis of H19 DMR3 and IGF2 DMR1/2
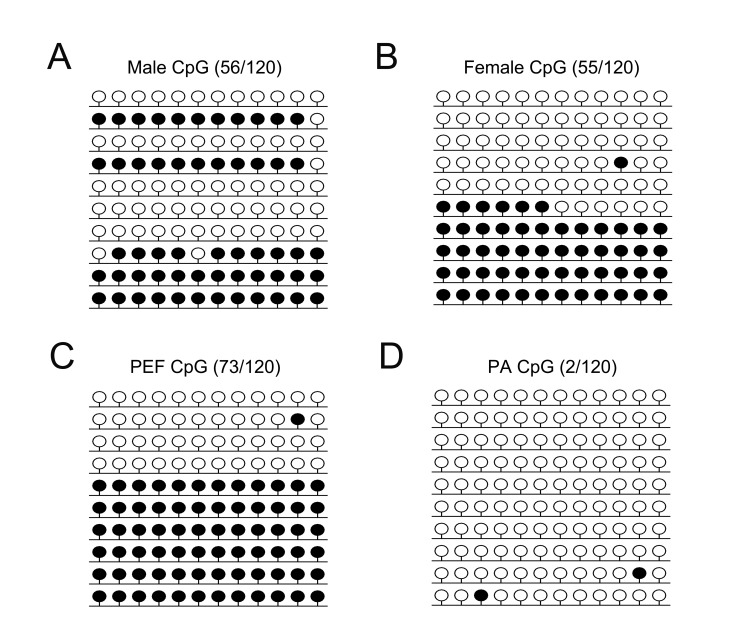
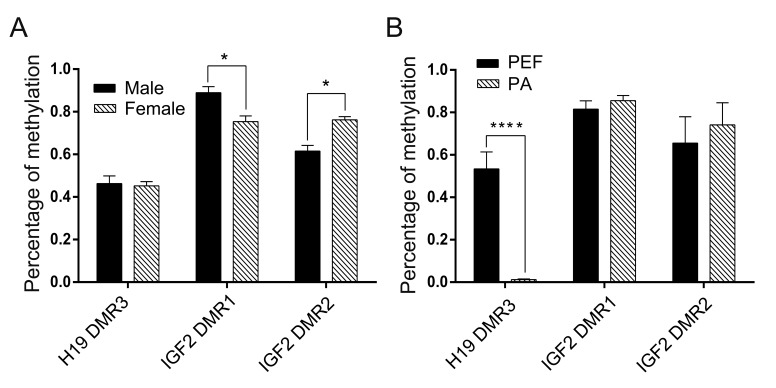
The methylation level of H19 DMR3 was lower in the female eye compared to the male eye (45.2 ± 0.8% and 51.3 ± 4.4%, respectively) (Figure 2A,B). The PCR products were subjected to COBRA analysis, which confirmed our results (Figure S1A (241.5KB, pdf) ). Statistical analyses confirmed the significant difference (Figure 4A). These results suggested that the H19 DMRs were hemi-methylated in both female and male eyes.
Figure 2. Methylation pattern of H19 DMR3. CpG methylation profiles of H19 DMR3 in male eye (A), female eye (B), porcine embryonic fibroblast (PEF) cells (C) and parthenogenetic (PA) cells (D). The black and white circles indicate methylated and unmethylated CpGs, respectively.
Figure 4. Percentage of methylated CpG sites within H19 DMD and IGF2 DMRs between male and female eyes (A), and between porcine embryonic fibroblast (PEF) and parthenogenetic (PA) cells (B). * p < 0.05, *** p < 0.005.
Compared with H19 DMRs, IGF2 DMR1/2 was found to be the hypermethylated in porcine eyes. The results of BSP showed that 75.4 ± 1.1% of IGF2 DMR1 were hypomethylated in the female eye compared with 89 ± 1.1% in the male eye (Figure 3A,B), while 76.3 ± 0.6% of IGF2 DMR2 were hypermethylated in the female eye compared with 64.1 ± 1% in the male eye (Figure 3C,D). The PCR products were subjected to COBRA analysis, which confirmed the results (Figure S1B (241.5KB, pdf) ). Statistical analyses revealed that there was a significant difference in IGF2 DMR1 and DMR2 between female and male eyes (Figure 4A). Taken together, these results suggested that the IGF2 DMRs methylation status play a crucial role in the regulation of imprinted gene expression in the female eye.
Figure 3. Methylation pattern of IGF2 DMRs. CpG methylation profiles of IGF2 DMR1 and DMR2 in (A-D) male eye and female eye, (E, G) porcine embryonic fibroblast (PEF) cells (F,H) parthenogenetic (PA) cells. The black and white circles indicate methylated and unmethylated CpGs, respectively.
Methylation status analysis of PA cells
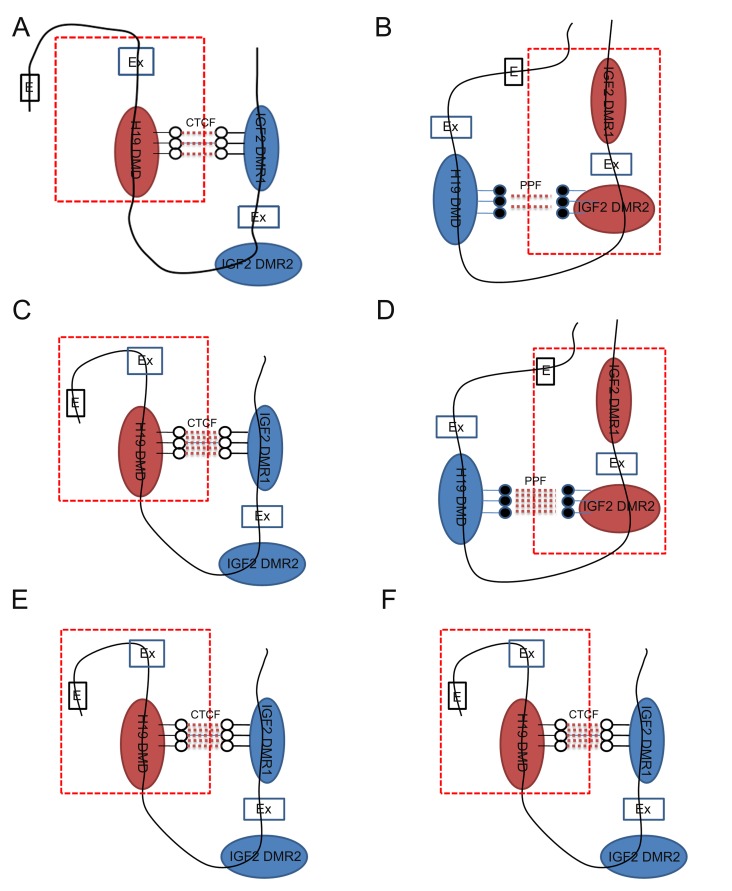
The expression pattern and methylation status of H19/IGF2 led us to hypothesize that a specific chromatin structure was located in the porcine female eye (Figure 5). To further investigate whether the expression of H19 and IGF2 were associated with their respective DMRs patterns, H19 DMRs and IGF2 DMRs were analyzed in PA cells. The BSP data showed that H19 DMRs in PA cells were significantly demethylated (p < 0.05) compared with those in PEF cells (Figure 2C,D), while IGF2 DMR1/2 were found to be hypermethylated in both PA and PEF cells (Figure 3E-H). The PCR products were subjected to COBRA analysis, which confirmed our BSP results (Figure S1C,D (241.5KB, pdf) ). Statistical analyses revealed that there was no difference in the methylation status of IGF2 DMR1 and DMR2 between PA and PEF cells (Figure 4B). Taken together, our results demonstrate that the hypomethylated patterns of DMRs regulated the H19 over-expression in maternal chromatin. These results indicate that the interaction of DNA methylation could change chromatin structure to regulate gene expression in H19/IGF2 clusters.
Figure 5. Schematic representations of H19/IGF2. DNA methylation interactions of H19 DMD and IGF2 DMRs on the maternal (A) and paternal allele (B) in the male eye. The specific chromatin structure is described on the maternal (C) and paternal allele (D) in the female eye. As expected, the specific chromatin structures are established as in parthenogenetic (PA) cells (E and F). Ex: exon; E: enhancer; Red dotted line: active domain; CTCF: CCCTC-binding factor - binding sites; PPF: putative protein factors.
Discussion
The chromatin-loop model was first developed to account for H19/IGF2 imprinting expression by IGF2 DMRs (Murrell et al., 2004). Usually, the IGF2 DMR1 moves to an active domain, while IGF2 DMR2 moves to an inactive domain on the maternal allele. When H19 DMRs and IGF2 DMR1 are both unmethylated, H19 promoters in close proximity become enhancers. This interaction between the H19 DMRs and IGF2 DMR1 results in H19 expression. In contrast, when the IGF2 DMR2 and DMR1 both move to an active domain on the paternal allele, H19 DMRs interacts with IGF2 DMR2. Because the H19 DMRs are methylated, IGF2 promoters come close to enhancers, resulting in IGF2 expression. In this model, IGF2 DMRs play an important role in the regulation of H19 and IGF2 expression. In the present study, the results of our BSP analysis suggested that there was a specific chromatin structure that might be regulated by DNA methylation in the porcine female eye. Compared with the male eye (Figure 5A,B), hypomethylated IGF2 DMR1 closely interacts with H19 DMRs, and thus forces H19 over-expression on the maternal chromosome (Figure 5C). Compared with the male eye, hypermethylated DMR2 of IGF2 closely interacts with H19 DMRs on the paternal chromosome in the female eye. This forces IGF2 over-expression (Figure 5D).
Due to the fact that H19 DMRs were found to be hemi-methylated in both male and female eyes, we explored whether the hypomethylation status of DMRs closely interacted with each other on the maternal chromosome. In this study, we used PA cells to determine if the elevated expression of H19/IGF2 in the porcine female eye was regulated by the DNA methylation interaction. Indeed, evolutionarily conserved sexually dimorphic expression of H19/IGF2 suggests a functional sex difference. PA cells contain exclusively maternal chromatin, thus, H19/IGF2 expression from paternal chromatin was not a confounding factor in the analyses (Zhu et al., 2003).
In our previous study, BSP results suggested that there was no difference in IGF2 DMR1 and DMR2 between PA and normal fetuses (Wang et al., 2014a). These data are in accordance with the present results from PA cells. In addition, the H19 DMRs were hypomethylated in PA cells, confirming previous reports of aberrant methylation profiles in oocytes, PA embryos and fetuses (Park et al., 2009; Wang et al., 2014a). Our results indicate that H19 DMRs closely interact with IGF2 DMR1 on the maternal chromosome in PA samples (Figure 5E,F). Due to the hypomethylated DMRs, H19 expression was higher in PEF cells, while IGF2 was not expressed. The results from the PA cells indirectly show that IGF2 was over-expressed by the hypermethylation status of IGF2 DMR2 on the paternal chromosome in the female eye.
In summary, the results of the present study demonstrate the elevated expression of H19/IGF2 in female porcine eye, which was associated with the methylation patterns of IGF2 DMRs. Our detailed analysis of PA and PEF cells suggest that a specific chromatin structure was formed due to the methylation of DMRs, which regulated the expression of H19/IGF2.
Acknowledgments
The authors express their gratitude to Xue Chen and Tingting Yu at the Embryo Engineering Center for technical assistance. This work was financially supported by the National Natural Science Foundation of China (Grant No. 31601003). The Project was also funded through a grant from the Key Laboratory of Regenerative Biology, Guangzhou Institutes of Biomedicine and Health, Chinese Academy of Sciences.
Supplementary Material
The following online material is available for this article:
Footnotes
Associate editor: Bertram Brenig
References
- Barlow DP, Stoger R, Herrmann BG, Saito K, Schweifer N. The mouse insulin-like growth factor type-2 receptor is imprinted and closely linked to the Tme locus. Nature. 1991;349:84–87. doi: 10.1038/349084a0. [DOI] [PubMed] [Google Scholar]
- Bartolomei MS, Zemel S, Tilghman SM. Parental imprinting of the mouse H19 gene. Nature. 1991;351:153–155. doi: 10.1038/351153a0. [DOI] [PubMed] [Google Scholar]
- Braunschweig MH, Owczarek-Lipska M, Stahlberger-Saitbekova N. Relationship of porcine IGF2 imprinting status to DNA methylation at the H19 DMD and the IGF2 DMRs 1 and 2. BMC Genet. 2011;12:47–47. doi: 10.1186/1471-2156-12-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunkow ME, Tilghman SM. Ectopic expression of the H19 gene in mice causes prenatal lethality. Genes Dev. 1991;5:1092–1101. doi: 10.1101/gad.5.6.1092. [DOI] [PubMed] [Google Scholar]
- Clark SJ, Harrison J, Paul CL, Frommer M. High sensitivity mapping of methylated cytosines. Nucleic Acids Res. 1994;22:2990–2997. doi: 10.1093/nar/22.15.2990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabory A, Ripoche MA, Yoshimizu T, Dandolo L. The H19 gene: Regulation and function of a non-coding RNA. Cytogenet Genome Res. 2006;113:188–193. doi: 10.1159/000090831. [DOI] [PubMed] [Google Scholar]
- Han DW, Im YB, Do JT, Gupta MK, Uhm SJ, Kim JH, Scholer HR, Lee HT. Methylation status of putative differentially methylated regions of porcine IGF2 and H19. Mol Reprod Dev. 2008;75:777–784. doi: 10.1002/mrd.20802. [DOI] [PubMed] [Google Scholar]
- Han X, Ouyang H, Chen X, Huang Y, Song Y, Zhang M, Pang D, Lai L, Li Z. Aberrant expression of Igf2/H19 in porcine parthenogenetic fetuses and placentas. Anim Reprod Sci. 2013;139:101–108. doi: 10.1016/j.anireprosci.2013.04.008. [DOI] [PubMed] [Google Scholar]
- Hudson QJ, Kulinski TM, Huetter SP, Barlow DP. Genomic imprinting mechanisms in embryonic and extraembryonic mouse tissues. Heredity. 2010;105:45–56. doi: 10.1038/hdy.2010.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huntriss JD, Hemmings KE, Hinkins M, Rutherford AJ, Sturmey RG, Elder K, Picton HM. Variable imprinting of the MEST gene in human preimplantation embryos. Eur J Hum Genet. 2013;21:40–47. doi: 10.1038/ejhg.2012.102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murrell A, Heeson S, Reik W. Interaction between differentially methylated regions partitions the imprinted genes Igf2 and H19 into parent-specific chromatin loops. Nat Genet. 2004;36:889–893. doi: 10.1038/ng1402. [DOI] [PubMed] [Google Scholar]
- Park CH, Kim HS, Lee SG, Lee CK. Methylation status of differentially methylated regions at Igf2/H19 locus in porcine gametes and preimplantation embryos. Genomics. 2009;93:179–186. doi: 10.1016/j.ygeno.2008.10.002. [DOI] [PubMed] [Google Scholar]
- Park CH, Uh KJ, Mulligan BP, Jeung EB, Hyun SH, Shin T, Ka H, Lee CK. Analysis of imprinted gene expression in normal fertilized and uniparental preimplantation porcine embryos. PLoS ONE. 2011;6:e22216. doi: 10.1371/journal.pone.0022216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinius B, Kanduri C. Elevated expression of H19 and Igf2 in the female mouse eye. PLoS ONE. 2013;8:e56611. doi: 10.1371/journal.pone.0056611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorvaldsen JL, Fedoriw AM, Nguyen S, Bartolomei MS. Developmental profile of H19 differentially methylated domain (DMD) deletion alleles reveals multiple roles of the DMD in regulating allelic expression and DNA methylation at the imprinted H19/Igf2 locus. Mol Cell Biol. 2006;26:1245–1258. doi: 10.1128/MCB.26.4.1245-1258.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Song Y, Huang Y, Duan F, Lv Q, Ouyang H, Lai L, Li Z. Genomic imprinting analysis of Igf2/H19 in porcine cloned fetuses using parthenogenetic somatic cells as nuclear donors. Biotechnol Lett. 2014a;36:1945–1952. doi: 10.1007/s10529-014-1572-8. [DOI] [PubMed] [Google Scholar]
- Wang DX, Song YN, Huang YY, Duan FF, Lv QY, Ouyang HS, Lai LX, Li ZJ. Genomic imprinting analysis of Igf2/H19 in porcine cloned fetuses using parthenogenetic somatic cells as nuclear donors. Biotechnol Lett. 2014b;36:1945–1952. doi: 10.1007/s10529-014-1572-8. [DOI] [PubMed] [Google Scholar]
- Watanabe K, Emoto N, Sunohara M, Kawakami M, Kage H, Nagase T, Ohishi N, Takai D. Treatment of PCR products with exonuclease I and heat-labile alkaline phosphatase improves the visibility of combined bisulfite restriction analysis. Biochem Biophys Res Commun. 2010;399:422–424. doi: 10.1016/j.bbrc.2010.07.093. [DOI] [PubMed] [Google Scholar]
- Wei GH, Liu DP, Liang CC. Chromatin domain boundaries: Insulators and beyond. Cell Res. 2005;15:292–300. doi: 10.1038/sj.cr.7290298. [DOI] [PubMed] [Google Scholar]
- Zhu J, King T, Dobrinsky J, Harkness L, Ferrier T, Bosma W, Schreier LL, Guthrie HD, DeSousa P, Wilmut I. In vitro and in vivo developmental competence of ovulated and in vitro matured porcine oocytes activated by electrical activation. Cloning Stem Cells. 2003;5:355–365. doi: 10.1089/153623003772032853. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.