Abstract
The adult form of attention-deficit/hyperactivity disorder has a prevalence of up to 5% and is the most severe long-term outcome of this common disorder. Family studies in clinical samples as well as twin studies suggest a familial liability and consequently different genes were investigated in association studies. Pharmacotherapy with methylphenidate (MPH) seems to be the first-line treatment of choice in adults with attention-deficit hyperactive disorder (ADHD) and some studies were conducted on the genes influencing the response to this drug. Finally some peripheral biomarkers were identified in ADHD adult patients. We believe this work is the first systematic review and meta-analysis of candidate gene association studies, pharmacogenetic and biochemical (metabolomics) studies performed in adults with ADHD to identify potential genetic, predictive and peripheral markers linked specifically to ADHD in adults. After screening 5129 records, we selected 87 studies of which 61 were available for candidate gene association studies, 5 for pharmacogenetics and 21 for biochemical studies. Of these, 15 genetic, 2 pharmacogenetic and 6 biochemical studies were included in the meta-analyses. We obtained an association between adult ADHD and the gene BAIAP2 (brain-specific angiogenesis inhibitor 1-associated protein 2), even after Bonferroni correction, with any heterogeneity in effect size and no publication bias. If we did not apply the Bonferroni correction, a trend was found for the carriers allele 9R of dopamine transporter SLC6A3 40 bp variable tandem repeat polymorphism (VNTR) and for 6/6 homozygotes of SLC6A3 30 bp VNTR. Negative results were obtained for the 9-6 haplotype, the dopamine receptor DRD4 48 bp VNTR, and the enzyme COMT SNP rs4680. Concerning pharmacogenetic studies, no association was found for the SLC6A3 40 bp and response to MPH with only two studies selected. For the metabolomics studies, no differences between ADHD adults and controls were found for salivary cortisol, whereas lower serum docosahexaenoic acid (DHA) levels were found in ADHD adults. This last association was significant even after Bonferroni correction and in absence of heterogeneity. Other polyunsaturated fatty acids (PUFAs) such as AA (arachidonic acid), EPA (eicosapentaenoic acid) and DyLA (dihomogammalinolenic acid) levels were not different between patients and controls. No publication biases were observed for these markers. Genes linked to dopaminergic, serotoninergic and noradrenergic signaling, metabolism (DBH, TPH1, TPH2, DDC, MAOA, MAOB, BCHE and TH), neurodevelopment (BDNF and others), the SNARE system and other forty genes/proteins related to different pathways were not meta-analyzed due to insufficient data. In conclusion, we found that there were not enough genetic, pharmacogenetic and biochemical studies of ADHD in adults and that more investigations are needed. Moreover we confirmed a significant role of BAIAP2 and DHA in the etiology of ADHD exclusively in adults. Future research should be focused on the replication of these findings and to assess their specificity for ADHD.
Introduction
Attention-deficit hyperactivity disorder (ADHD), which is commonly known to occur during childhood, is characterized by excessive inattention and/or hyperactivity and impulsivity as well as executive dysfunction, lack of emotional self-control and motivation. However a significant number of affected children (65%) continue manifesting symptoms into adulthood.1, 2
The prevalence of ADHD was reported as 5.3% in childhood,3, 4, 5 whereas it is estimated as 2.5–4.9% in adulthood.6, 7 Clinical research showed that the predominant features of ADHD in adults differ from ADHD in children, with adults showing less obvious symptoms of hyperactivity or impulsivity and more inattentive symptoms.8 A recent study reported follow-back analyses of ADHD cases diagnosed in adulthood, alongside follow-forward analyses of ADHD cases diagnosed in childhood in a representative birth cohort from Dunedin.9 The findings suggested that adults with ADHD may not have a childhood-onset disorder. There was little overlap between participants who had been diagnosed with ADHD in childhood and those diagnosed in adulthood. This finding is intriguing but difficult to interpret because it is not consistent with a very large literature documenting the persistence of ADHD into adulthood. Pharmacological treatment is considered effective and safe both for children and adults, but there is considerable inter-individual variability among patients regarding response to medication, required doses, and adverse events. Pharmacotherapy with methylphenidate (MPH) seems to be the first-line treatment of choice both in children10, 11 and in adults12, 13, 14, 15 with ADHD.
ADHD is a complex and heterogeneous disorder and its etiology is not yet completely understood. Despite evidences that environmental factors have an important role in its etiology, classical genetics studies support a strong genetic contribution for ADHD with the heritability estimates averaging 75% in children.16 Genetic liability comprises a polygenic liability with both common and rare variants involved. Individuals with large, rare copy-number variations (CNVs) require less loading of multiple common genetic risk variants for developing ADHD.17
In adults, initial heritability estimates of ADHD were substantially lower (~30–50%),18, 19, 20 even though other evidence had suggested a stronger genetic component to the etiology of adult ADHD.21, 22, 23, 24 A more recent review suggests that the low heritability of ADHD in adults is unlikely to reflect a true developmental change. Instead, the drop in heritability is better explained by rater effects related to a switch from using one rater for both twins in a pair (parent/teacher) in childhood, to relying on self-ratings (where each twin rates themselves) of ADHD symptoms in adulthood. When rater effects are addressed using cross-informant approaches, the heritability of ADHD in adults appears to be comparable to the heritability of ADHD in childhood.25 Many candidate gene association studies (http://adhd.psych.ac.cn/index.do), several meta-analyses including the most recent,26 genome-wide association (single nucleotide polymorphism, SNP-GWA) and CNV studies, summarized in some recent reviews,27, 28, 29 have been reported for children with ADHD. These studies have implicated dopaminergic, serotoninergic and glutamatergic signaling along with synaptic vesicle, neurite outgrowth and cell adhesion pathways. A recent review of the literature on ADHD pharmacogenetics in childhood supported a significant effect of neurodevelopmental and noradrenergic systems in the MPH response, whereas no or contrasting results were obtained for dopaminergic and serotonininergic signaling, synaptosomal-associated protein 25 (SNAP25), metabolic enzymes.30 Finally, the results of a series of meta-analyses reviewed in Scassellati et al.31 on biochemical studies performed in childhood, showed a significant association with peripheral biomarkers linked to monoaminergic pathways and to the hypothalamic–pituitary–adrenal (HPA) axis.
In contrast to a large genetics literature about childhood ADHD, fewer studies have addressed the adult form of the disorder. A summary of genetic studies of adult ADHD was reported in Franke et al.32 in a comprehensive review. The literature about adult ADHD now comprises six meta-analyses of candidate genes in International multi-center persistent ADHD collaboration (IMpACT) samples,33, 34, 35, 36, 37, 38 two SNP-GWAS,39, 40 two CNV-GWAS41, 42 studies and three reviews of pharmacogenetics studies.30, 43, 44 No meta-analyses of biochemical studies of adult ADHD have been reported.
This work has the aim to review candidate gene association studies, pharmacogenetic and biochemical (metabolomics) studies performed in adults with ADHD and to conduct meta-analyses where possible in order to identify potential genetic, predictive and peripheral markers linked specifically to ADHD in adults.
Materials and methods
Search strategy and selection
To identify eligible studies for the review and meta-analyses, we searched these electronic databases: PubMed, EMBASE (Ovid), PsycINFO (Ovid), Biosis (Web of Science) from inception 1971 until August 2015. We searched for all available original studies regarding candidate gene associations, pharmacogenetics and biochemical markers in adults with ADHD. The search was limited to articles published in English (399 studies were reported in other languages, most in German). The following keywords were employed: adults, attention-deficit/hyperactivity disorder or ADHD combined with the following terms: gene*, genetics, polymorphism, association, pharmacogen*, response to medication, stimulant response, MPH, amphetamine, atomoxetine, peripheral levels, biomarkers, plasma, serum, saliva, urine, cerebrospinal fluid and red blood cells. The initial search produced 7746 on Pubmed, 4708 on EMBASE (Ovid), 3425 on PsycINFO (Ovid) and 9796 on Biosis (Web of Science).
We selected articles that met the following inclusion criteria: (1) ADHD diagnosis according to the Diagnostic and Statistical Manual of Mental Disorders—version IV (DSM-IV) or the International Classification of Diseases—10th Revision (ICD-10); (2) having a case–control or family-based study design for candidate gene association studies; (3) having a drug-with/without placebo design for pharmacogenetic studies; (4) having a drug-free/naive case–control study design for biochemical studies. We excluded Dresler et al.45 and Hirvikoski et al.46 in the analyses of SLC6A3 40 bp variable tandem repeat polymorphism (VNTR) genotyping and salivary cortisol levels respectively, because they did not use a structured interview tool with high reliability and less reliable methods for the assessment of childhood ADHD were applied.
Supplementary Figure 1 presents a PRISMA flow diagram depicting our selection procedure for review and meta-analysis.
Meta-analyses were performed for all markers for which usable data were reported in at least three published studies.
Data extraction for meta-analyses and statistical analyses
The literature search was performed by two individuals independently (CS and CB). When data were not available, authors were contacted. For candidate gene association studies, we extracted the following data from the original publications: biological systems, genes analyzed, location and polymorphisms, first author, populations studied, sample size, results and eligibility for meta-analysis (Table 1). For pharmacogenetic studies on MPH drug, we extracted: genes analyzed, location and polymorphisms, first author, populations, sample size, dosages, duration times, results and eligibility for meta-analysis (Table 2). For biochemical studies, we extracted: biological systems, biomarkers analyzed, biological fluids, first author, populations, sample size, results and eligibility for meta-analysis (Table 3). In Supplementary Tables 1, 2, 3, we showed the candidate gene association, pharmacogenetic and biochemical studies for which the meta-analyses were not possible to conduct due to insufficient data.
Table 1. Candidate gene association studies available for meta-analysis.
| Systems | Genes | Location | Polymorphisms | Authors | Populations |
Sample size |
Results | Eligibility | Reasons | |
|---|---|---|---|---|---|---|---|---|---|---|
| ADHD | CTRL | |||||||||
| Dopaminergic | ||||||||||
| SLC6A3 | Intron, intron, intron, intron, exon, intron, intron and 3′UTR | rs460700, rs37020, rs13161905, rs27048, rs6347, rs11133767, rs40184, rs2975292 | Ribases et al.48 | Spain and German | 211 | 211 | No | Excluded | Other polymorphisms | |
| Promoter | rs2652511 | de Azeredo et al.49 | Brazil | 501 | 569 | Yes | Excluded | Another polymorphism | ||
| Intron 8 | 30 bp VNTR | Bruggerman et al.50 | German | 122 | 174 | No | Included | |||
| Silva et al.51 | Brazil | 94 | 481 | Yes | Included | |||||
| Franke et al.34 | IMpACT | 1438 | 1767 | Yes | Included | |||||
| Hoogman et al.52 | Dutch (IMpACT) | 88 | 77 | Yes | Included | |||||
| Spencer et al.53 | Boston | 34 | 36 | No | Included | |||||
| de Azeredo et al.49 | Brazil | 497 | 596 | No | Included | |||||
| 3′UTR | 40 bp VNTR | Bruggerman et al.50 | German | 122 | 174 | No | Included | |||
| Johansson et al.54 | Norwegian | 358 | 340 | No | Included | |||||
| Dresler et al.45 | German | 161 | 109 | No | Excluded | No diagnosis available | ||||
| Franke et al.34 | IMpACT | 1440 | 1769 | Yes | Included | |||||
| Aparacida da Silva et al.55 | Brazil | 102 | 479 | No | Included | |||||
| Brown et al.56 | Boston | 53 | 38 | Yes | Excluded | No data available | ||||
| Hoogman et al.52 | Dutch (IMpACT) | 87 | 77 | Yes | Included | |||||
| Spencer et al.53 | Boston | 34 | 36 | No | Included | |||||
| de Azeredo et al.49 | Brazil | 476 | 587 | No | Included | |||||
| Hasler et al.57 | European | 77 | 474 | No | Included | |||||
| DRD4 | Exon 3 | 48 bp VNTR | Muglia et al.58 | European 94% | 66 | 66 | Yes | Included | ||
| Johansson et al.54 | Norwegian | 358 | 340 | No | Included | |||||
| Sanchez-Mora et al.33 | IMpACT | 1410 | 1751 | No | Included | |||||
| Tovo-rodrigues et al.59 | Brazil | 308 | 230 | No | Excluded | Only haplotypes | ||||
| Carpentier et al.60 | Netherlands (IMpACT) | 176 | 500 | No | Included | |||||
| Spencer et al.53 | Boston | 34 | 36 | No | Included | |||||
| Hasler et al.57 | European | 77 | 474 | Yes | Included | |||||
| Promoter | rs4646984 (120 bp ins/del) | Sanchez-Mora et al.33 | IMpACT | 1447 | 2062 | No | Excluded | No enough studies | ||
| Promoter and exon | rs4646984 (120 bp ins/del), rs4646983 | Ghosh et al.61 | Indian | 170 | 180 | Yes | Excluded | No enough studies, cADHD | ||
| 5′UTR and 3′UTR | rs3758653, rs936465 | Ribases et al.48 | Spain and German | 211 | 211 | No | Excluded | Other polymorphisms | ||
| Metabolic enzymes | ||||||||||
| COMT | Promoter, exon, exon and exon | rs6269, rs4633, rs4818, rs4680 | Halleland et al.62 | Norwegian | 435 | 383 | No: rs4680 | Included | ||
| 5′UTR, intron, intron, intron, intron, exon, intron, intron, intron and intron | rs2020917, rs933271, rs1544325, rs740603, rs740601, rs4680, rs4646316, rs174696, rs165774, rs9332377 | Ribases et al.48 | Spain and German | 211 | 211 | No | Excluded | No frequencies reported | ||
| Exon | rs4680 | Carpentier et al.60 | Netherlands (IMpACT) | 176 | 500 | No | Included | |||
| Exon | rs4680 | Biehl et al.63 | German | 35 | 35 | No | Included | |||
| Exon | rs4818 | Pazvantoğlu et al.64 | Turkish | 120 | 109 | No | Excluded | Parents of children with ADHD | ||
| Intron and 3′UTR | rs740603, rs165599 | Ghosh et al.61 | Indian | 170 | 180 | No | Excluded | Other polymorphisms, cADHD | ||
| Neurodevelopmental network | ||||||||||
| BAIAP2 | All in introns | 8079626, rs8079781, rs7210438, rs4969385 | Ribases et al.65 | Spain | 270 | 531 | Yes | Included | ||
| Ribases et al.65 | German | 639 | 612 | Yes | Included | |||||
| Ribases et al.65 | Norwegian | 417 | 469 | Yes | Included | |||||
Abbreviations: ADHD, attention-deficit hyperactive disorder; BAIAP2, brain-specific angiogenesis inhibitor 1-associated protein 2; COMT, catechol-O-methyl-transferase; DRD4, dopamine D4 receptor; IMpACT, International multi-center persistent ADHD collaboration; SLC6A3, dopamine transporter; UTR, untranslated region; VNTR, variable tandem repeat polymorphism.
The results indicating no mean absence of association, yes presence of association.
Table 2. Pharmacogenetic studies on MPH drug available for meta-analysis.
| Genes | Location | Polymorphisms | Authors | Populations |
Sample size |
Dosages | Duration times | Results | Eligibility | Reasons | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| R | NR /PL | ||||||||||
| SLC6A3 | 3′UTR | 40 bp VNTR | Kooij et al.66 | Netherlands | 16 | 26 | 0.5 mg kg−1 per day; 1 mg kg−1 per day | 3 Weeks | No | Included | |
| Contini et al.67 | Brazil | 136 | 35 | 0.3 mg kg−1 per day | 4 Weeks | No | Included | ||||
| Mick et al.68 | Boston | 66 | 40 | 0.5 mg kg−1 per day; 1 mg kg−1 per day | 6 Weeks | No | Excluded | Placebo arm | |||
| 5′UTR | rs2652511 | Contini et al.67 | Brazil | 136 | 35 | 0.3 mg kg−1 per day | 4 Weeks | No | Excluded | Another polymorphism | |
| Intron 8 | 30 bp VNTR | Contini et al.67 | Brazil | 136 | 35 | 0.3 mg kg−1 per day | 4 Weeks | No | Excluded | No enough studies | |
Abbreviations: NR, no responders; PL, placebo (Mick et al.68); R, responders; SLC6A3, dopamine transporter; UTR, untranslated region; VNTR, variable tandem repeat polymorphism.
The results indicating no mean absence of association.
Table 3. Biochemical studies (metabolomics) available for meta-analysis.
| Systems | Biomarkers | Biological fluids | Authors | Populations |
Sample size |
Results | Eligibility | Reasons | |
|---|---|---|---|---|---|---|---|---|---|
| ADHD (N) | CTRL (N) | ||||||||
| Circadian rhythms | Cortisol | Saliva | Hirvikoski et al.46 | Swedish | 28 | 28 | No | Excluded | No diagnosis available |
| Lackschewitz et al.69 | German | 18 | 18 | No | Included | ||||
| Baird et al.70 | UK | 13 | 19 | Alterations | Included | ||||
| Raz et al.71 | Israel | 24 | 25 | No | Included | ||||
| PUFAs | AA | RBC | Young et al.72 | Canada | 36 | 35 | No | Excluded | No enough studies |
| Serum | Young et al.72 | Canada | 36 | 35 | No | Included | |||
| Laasonen et al.73 | Finland | 26 | 36 | No | Included | ||||
| Irmish et al.74 | Germany | 15 | 15 | Alterations | Included | ||||
| DHA | RBC | Young et al.72 | Canada | 36 | 35 | Alterations | Excluded | No enough studies | |
| Serum | Young et al.72 | Canada | 36 | 35 | Alterations | Included | |||
| Laasonen et al.73 | Finland | 26 | 36 | No | Included | ||||
| Irmish et al.74 | Germany | 15 | 15 | Alterations | Included | ||||
| EPA | RBC | Young et al.72 | Canada | 36 | 35 | No | Excluded | No enough studies | |
| Serum | Young et al.72 | Canada | 36 | 35 | No | Included | |||
| Laasonen et al.73 | Finland | 26 | 36 | No | Included | ||||
| Irmish et al.74 | Germany | 15 | 15 | Alterations | Included | ||||
| DyLA | RBC | Young et al.72 | Canada | 36 | 35 | No | Excluded | No enough studies | |
| Serum | Young et al.72 | Canada | 36 | 35 | No | Included | |||
| Laasonen et al.73 | Finland | 26 | 36 | No | Included | ||||
| Irmish et al.74 | Germany | 15 | 15 | Alterations | Included | ||||
Abbreviations: AA, arachidonic acid; ADHD, attention-deficit hyperactivity disorder; DHA, docosahexaenoic acid; DyLA, dihomogammalinolenic acid; EPA, eicosapentaenoic acid; N, sample size; PUFAs, polyunsaturated fatty acids; RBC, red blood cells.
No indicates absence of alterations on levels of a biomarker.
Review Manager was used to analyze the data (RevMan Version 5.1.6; Copenhagen, The Nordic Cochrane Centre, The Cochrane Collaboration, 2008). We used the fixed-effects model to generate a pooled effect size and 95% confidence interval (CI) from individual study effect sizes (the odd ratios for genetic/pharmacogenetic studies or the standardized mean difference for biochemical studies) using the Mantel–Haenszel and inverse variance methods, respectively. The significance of the pooled effect sizes was determined by the z-test. Between-study heterogeneity was assessed using a χ2 test of goodness of fit test and the I2 statistic. We used a P-value of 0.05 to assert statistical significance. Where the results showed a significant effect in the presence of significant between-study heterogeneity, a random effects model was used, with effect sizes pooled using the DerSimonian and Laird method.
Publication bias was estimated by the method of Egger et al.47 which uses a linear regression approach to measure funnel plot asymmetry on the natural logarithm scale of the effect size. The significance of the intercept (a) was determined by the t-test.47 The rank correlation method and regression method tests were conducted by MIX version 1.7. (http://www.mix-for-meta-analysis.info).
Because we conducted 6, 1 and 2 meta-analyses to assess the significance of genetic, pharmacogenetic and biochemical markers, respectively, our Bonferroni corrected significance levels were 0.008, 0.05 and 0.025, respectively. This provides a stringent approach for preventing false-positive findings, with the cost of reducing statistical power. In contrast, for our analyses of publication biases and heterogeneity, we use an uncorrected alpha level to ensure that any of these potential problems with the findings could be detected.
Results
During the screening of 5129 records, we selected 87 studies meeting our eligibility criteria: 61 were available for candidate gene association studies, 5 for pharmacogenetics and 21 for biochemical studies (Supplementary Figure 1). Of these, 15 candidate gene association, 2 pharmacogenetic and 6 biochemical studies were included in the meta-analyses.
Because most of the meta-analyses did not show significant heterogeneity in effect size across studies, meta-regressions were not run.
Here below we described candidate gene association studies (Table 1),33, 34, 45, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65 pharmacogenetic (Table 2)66, 67, 68 and biochemical (metabolomic) (Table 3)46, 69, 70, 71, 72, 73, 74 studies for which it was possible to conduct meta-analysis. Moreover we described the studies for which the meta-analyses were not possible.
Candidate gene association studies available for meta-analysis
From the selection of 61 studies, 15 association studies were included in the meta-analyses (Table 1). Ten studies were excluded in which no enough data were reported on a polymorphism; 43 on a gene; and 5 lacking statistical data for patients and controls (Supplementary Table 1).
Neurotransmitter systems
Dopamine transporter gene (SLC6A3)
The main function had by the dopamine transporter (DAT1) is to regulate dopamine (DA) availability in the synapse. It removes DA from the synaptic cleft into the pre-synaptic neuron or releases DA into the extracellular space. Its involvement in the etiology of ADHD is supported by the fact that DAT1 is the main target for the stimulant drugs that treat ADHD.
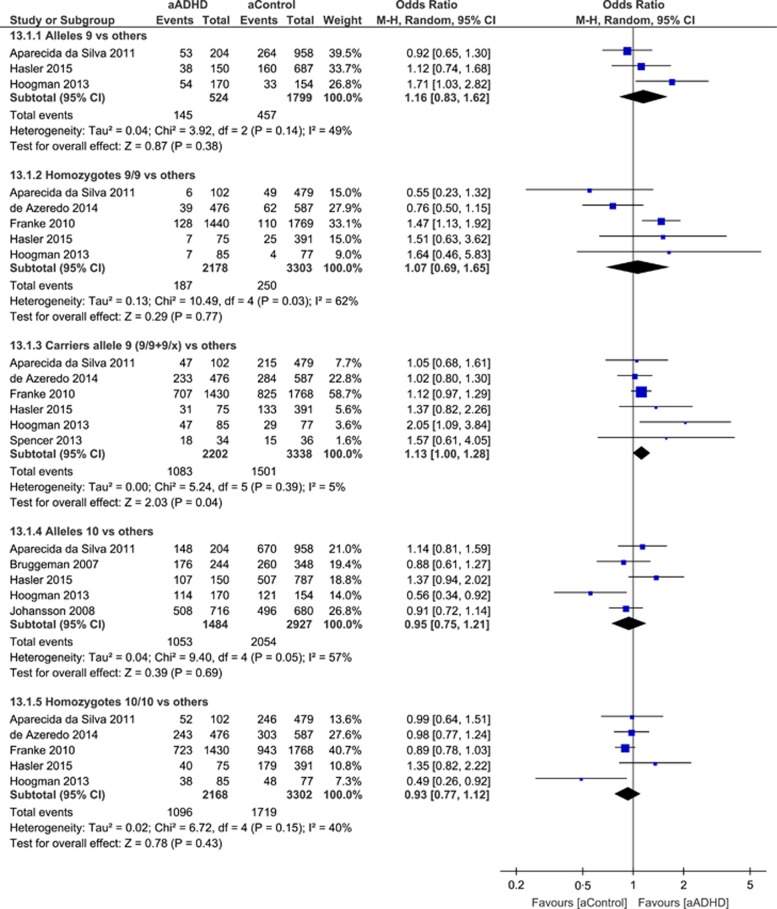
The most studied SLC6A3 variant is a VNTR of 40 base pairs located at the 3′-untranslated region (3′-UTR) of the gene. The ten repeat (10R) and nine repeat (9R) alleles are the most common. A recent meta-analysis,75 assessing the association of this variant with DAT1 activity, showed that the 9R allele regulates dopamine activity in the striatal brain regions independent of the presence of neuropsychiatric illness. In childhood, the 10R allele has been associated to ADHD susceptibility.26 In adults, nine studies34, 49, 50, 52, 53, 54, 55, 57 were available for this polymorphism. Brown et al.56 was excluded because they porfermed the analyses in a selected sample of patients and controls carrying only 9/9, 9/10 and 10/10 genotype. Because the authors showed different calculations for this VNTR, we reported the results according to: (a) allele 9 versus the others52, 55, 57 (Figures 1, Z=0.87, P=0.38, absence of heterogeneity in effect size across studies P=0.14, I2=49%); (b) 9/9 homozygotes versus the other genotypes34, 49, 52, 55, 57 (Figure 1, Z=0.29, P=0.77, slight heterogeneity in effect size across studies P=0.03, I2=62%); (c) carriers allele 9 (9/9+9/x) versus others,34, 49, 52, 53, 55, 57 (Figure 1, Z=2.03, P=0.04, no heterogeneity in effect size P=0.39, I2=5%). This trend of association was lost after Bonferroni correction; (d) allele 10 versus others50, 52, 54, 55, 57 (Figure 1, Z=0.39, P=0.69, slight heterogeneity in effect size P=0.05, I2=57%); (e) 10/10 homozygotes versus other genotypes,34, 49, 52, 55, 57 (Figure 1 Z=0.78, P=0.43, no heterogeneity in effect size P=0.15, I2=40%).
Figure 1.
Forest plot for odds ratio from meta-analysis of the 40 bp variable tandem repeat polymorphism (VNTR) in the dopamine transporter SLC6A3 gene. ADHD, attention-deficit hyperactive disorder; CI, confidence interval; Chi2, χ2 test of goodness of fit; Tau2, estimate of the between-study variance in a random-effects meta-analysis.
Another VNTR in intron 8 of the DAT gene was investigated by several studies. This VNTR is a 30- bp repeat sequence with two common alleles of five (5R) and six (6R) repeats. The 6R allele has been associated with ADHD susceptibility in childhood.26 In adults, six studies34, 49, 50, 51, 52, 53 were available for this polymorphism. As above, the authors reported different calculations, therefore we showed the results according to: (a) allele 5 versus others34, 51, 52 (Supplementary Figure 2, Z=1.72, P=0.09, no heterogeneity in effect size P=0.06, I2=64%); (b) 5/5 homozygotes versus others34, 49, 51, 52 (Supplementary Figure 2, Z=0.48, P=0.63, no heterogeneity in effect size P=0.63, I2=0%); (c) allele 6 versus others51, 52 (Supplementary Figure 2, Z=2.46, P=0.01, no heterogeneity in effect size P=0.64, I2=0%). This trend of association is due to only two studies; (d) 6/6 homozygotes versus others34, 49, 51, 52 (Supplementary Figure 2, Z=2.28, P=0.02, no heterogeneity in effect size P=0.13, I2=47%). This association was lost after Bonferroni correction; (e) carriers allele 6 versus others34, 49, 51, 52, 53 (Supplementary Figure 2, Z=0.58, P=0.56, no heterogeneity in effect size P=0.92, I2=0%).
Concerning the haplotype formed by the two polymorphisms, the results indicated no association between carriers of 9-6 haplotype and ADHD susceptibility34, 49, 52 (Supplementary Figure 3, d=1.45, 95% CI: 0.90–2.34, Z=1.55, P=0.12) with significant heterogeneity in effect size among the studies (P=0.002, I2=84%).
Finally according to Egger's test, the results indicated for the all meta-analyses related to the two polymorphisms 30 and 40 bp, no publication bias (see Supplementary Table 4 for all allele/genotype/haplotype combinations).
Further studies investigated other polymorphisms in this gene (Table 1), however no meta-analyses were possible due to the small number of studies.
Dopamine D4 receptor gene
The dopamine D4 receptor (DRD4) is a G-protein-coupled receptor belonging to the DA D2-like receptor family. It has been considered a candidate for the etiology of ADHD because of its high expression in brain regions implicated in attention and inhibition such as anterior cingulate cortex. Moreover, DRD4 was first associated with a personality trait common in ADHD (novelty seeking).
A highly polymorphic functional VNTR in the third exon of DRD4 has been frequently studied in association studies. It comprises 11 copies of a 48- bp repeat sequence, where the 4, 7 and 2 repeat alleles are the most prevalent. The 7R allele has been associated with ADHD in many studies of youth.26, 28, 29, 76, 77 The current meta-analysis included six studies.33, 53, 54, 57, 58, 60 Tovo-rodrigues et al.59 was excluded because insufficient data were reported. Also for this polymorphism different calculations were performed. Thus we represented the results according to: (a) allele 7 versus others33, 54, 57, 58 (Supplementary Figure 4, Z=0.79, P=0.43, in absence of heterogeneity in effect size across the studies P=0.06, I2=60%); (b) 7/7 homozygotes versus others33, 57 (Supplementary Figure 4, Z=1.24, P=0.21, in presence of heterogeneity in effect size across the studies P=0.006, I2=87%). These results considered only two studies; (c) carriers allele 7 versus others33, 53, 57, 60 (Supplementary Figure 4, Z=0.76, P=0.44, no heterogeneity in effect size P=0.57, I2=0%). No publication bias was observed (see Supplementary Table 4 for all allele/genotype combinations).
Concerning the rs4646984 polymorphism (120-bp tandem insertion/deletion up stream of exon 1), not enough studies were available for meta-analysis (Table 1).
Metabolic enzymes
Catechol-O-methyltransferase
Catechol-O-methyltransferase (COMT) is an enzyme responsible for the degradation of DA and NE. It is highly expressed in frontal lobe where it regulates synaptic DA levels. Most of the association studies between COMT and ADHD susceptibility examined a well-known Val158Met functional polymorphism in exon 4. Homozygotes for the valine allele show greater COMT activity than homozygotes for the methionine allele. Several meta-analyses26, 78, 79 indicated no association between ADHD and the Val158Met in childhood. For the current meta-analysis in adults, three studies were available for COMT.60, 62, 63 Here we showed the results in relation to: (a) allele Val versus Met (Supplementary Figure 5, Z=1.16, P=0.25, no heterogeneity in effect sizes across the studies was found P=0.31, I2=15%); (b) homozygotes Val/Val versus carriers allele Met (Supplementary Figure 5, Z=0.22 P=0.82, no heterogeneity in effect sizes across the studies was found P=0.66, I2=0%); (c) carriers allele Val versus homozygotes Met/Met (Supplementary Figure 5, Z=0.37, P=0.71, no heterogeneity in effect sizes across the studies was found P=0.80, I2=0%). No publication bias was observed (see Supplementary Table 4 for all allele/genotype combinations). Further studies investigated other polymorphisms (Table 1).
Neurodevelopmental network
BAI1-associated protein 2
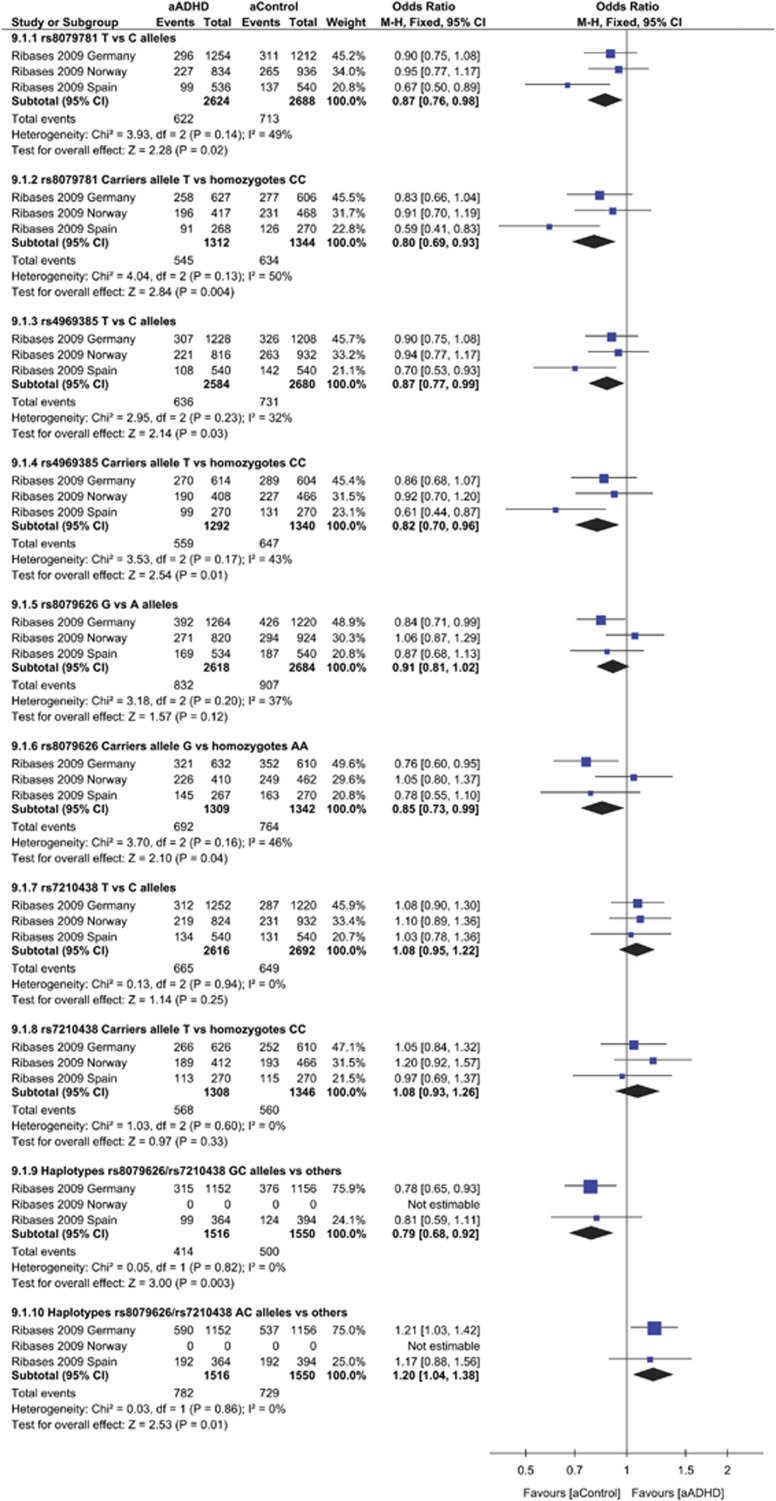
There is evidence suggesting that abnormal left–right brain asymmetries in ADHD patients are involved in a variety of ADHD-related cognitive processes, including sustained attention, working memory, response inhibition and planning. Although mechanisms underlying cerebral lateralization are unknown, left–right cortical asymmetry has been associated with transcriptional asymmetry at embryonic stages and several genes differentially expressed between hemispheres have been identified. One of these is brain-specific angiogenesis inhibitor 1-associated protein 2 (BAIAP2). rs8079781, rs4969385, rs8079626 and 7210438 were genotyped in samples from Spain, German and Norway.65 The results of our meta-analyses indicated significant differences for rs8079781 (Figure 2 allele T versus C, Z=2.28, P=0.02; carriers allele T versus homozygotes CC Z=2.84, P=0.004) with no significant heterogeneity in effect sizes across studies (P=0.14, I2=49% P=0.13, I2=50%, respectively). This last difference survived to Bonferroni correction. Similarly a significant association was observed for rs4969385 with allele T versus C (Z=2.14, P=0.03) and carriers of allele T versus homozygotes CC (Z=2.54, P=0.01), with no significant heterogeneity in effect size across the studies (P=0.23, I2=32% P=0.17, I2=43%, respectively; Figure 2). After applying the Bonferroni correction, this difference was lost. For the other two polymorphisms no associations were observed, except for a slight trend for the rs8079626 carriers allele G versus homozygotes AA (Z=2.10, P=0.04). Interesting results came from the meta-analysis of rs8079626-rs7210438 haplotypes. The GC haplotype versus others showed a protective effect (d=0.79, 95% CI: 0.68–0.92, Z=3.00, P=0.003), which remained significant after Bonferroni correction. The AC haplotype was the risk haplotype (d=1.20, 95% CI: 1.04–1.38, Z=2.53 P=0.01). In all cases, there was no heterogeneity in effect sizes across studies (P=0.82, I2=0% P=0.86, I2=0%, respectively). No pubblication bias were observed (see Supplementary Table 4 for all allele/genotype combinations).
Figure 2.
Forest plot for odds ratio from meta-analysis of the polymorphisms rs8079781, rs4969385, rs8079626 and 7210438 in the BAI1-associated protein 2 (BAIAP2) gene. ADHD, attention-deficit hyperactive disorder; Chi2, χ2 test of goodness of fit. CI, confidence interval.
Candidate gene association studies not available for meta-analysis
There are several genes for which there are not enough studies to perform meta-analysis. These are showed in Supplementary Table 1.35, 48, 53, 54, 57, 60, 64, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113 In particular, among the most known, DRD5, whose highly polymorphic dinucleotide repeat (CA)n has been the most studied in adults, three studies54, 60, 80 were available, but Squassina et al.80 was excluded because no frequencies were reported. Ribases et al.48 investigated other polymorphisms. Similar conclusions are reached for other dopamine receptor genes (DRD3, DRD2 and DRD1).
Serotonin transporter gene (SLC6A4, 5-HTT) is a further well-known gene. The 5HTTLPR, a 44- bp insertion/deletion yielding long and short alleles, was investigated in adults by two studies,35, 53 whereas Ribases et al.83 studied other polymorphisms. Similar conclusions were reported for other serotonin receptor genes among which serotonin 1B receptor gene (HTR1B) whose G861C polymorphism, rs6296, was investigated by only two studies,60, 83 and serotonin 2A receptor gene (HTR2A; 5HT2A).
Studies on norepinephrine transporter gene (NET1, SLC6A2) along with alpha-2A-adrenergic receptor gene (ADRA2A), were not enough to conduct meta-analyses in adults.
Among the metabolic enzymes, there are dopamine beta hydroxylase (DBH), tryptophan hydroxylases (TPH1, TPH2), of which a meta-analysis on IMPaCT samples was available for both genes,36 Dopamine decarboxylase (DDC), Monoamine oxidases MAOA and MAOB, acetylcholine metabolizing butyrylcholinesterase (BCHE) and tyrosine hydroxylase (TH).
Concerning the neurodevelopmental network, brain-derived neurotrophic factor (BDNF), ciliary neurotrophic factor receptor (CNTFR), neurotrophin NTF3, nerve growth factor (NGF) were too infrequent to perform meta-analyses. Similarly also for LPHN3, CDH13, GTP-binding RAS-like 2 (DIRAS2) genes no enough studies were available.
As regard the genes for synaptic vesicle proteins, SNAP25, showed in adults, a significant association of the MnlI polymorphism with ADHD susceptibility,97 even though this result was not confirmed by Olgiati et al.98 Other SNARE genes such as for instance Synaptobrevin-2 (VAMP-2), Syntaxin 1A (STX1A), Synapsin III (SYNIII) were investigated in some studies,98, 99, 100, 101 however, because different polymorphisms were genotyped, no meta-analyses can be performed.
Finally the neuronal isoform (NOS-I, encoded by NOS1), the main source of nitric oxide in the central nervous system, was investigated by two studies that reported an association with NOS1 gene.102, 103
Other genes were studied in single studies: for instance circadian locomotor output cycles kaput (CLOCK),105 protein kinase G (PRKG1),106 mineralocorticoid receptor (NR3C2),107 CKLF-like MARVEL transmembrane domain containing 8 (CMTM8)/diacylglycerol kinase eta (DGKH)/neuronal PAS domain protein 3 (NPAS3)/solute carrier family 39 (zinc transporter), member 3 (SLC39A3)/deafness, autosomal recessive 31 (DFNB31)/epidermal growth factor receptor (EGFR),108 neural cell adhesion molecule 1 (NCAM1)/tetratricopeptide repeat domain 12 (TTC12)/ankyrin repeat and kinase domain containing 1 (ANKK1),82 Ca(2+)-binding extracellular heparan/chondroitin sulfate proteoglycan (SPOCK3),109 disrupted in schizophrenia 1 (DISC1),110 Kv channel-interacting protein 4 (KCNIP4),111 phosphatase 2, regulatory subunit B, gamma (PPP2R2C),112 forkhead box P2 (FOXP2),113 αN-catenin protein (CTNNA2),96 u-opioid receptor (OPRM1), and others.60 Thus we did not perform meta-analyses.
Pharmacogenetic studies available for meta-analysis
Five studies were conducted on the pharmacogenetics of MPH in adults, of these only two studies were available to perform meta-analysis (Supplementary Figure 1). The gene implicated is SLC6A3 and its 40 bp VNTR and was investigated with MPH response in three studies66, 67, 68 (Table 2). However Mick et al.68 was not estimable due to a placebo study design, whereas the other studies66, 67 showed the results in relation to responders versus no responders. Notwithstanding we performed the meta-analysis. We report different combinations as showed by the authors: (a) allele 10 versus the others (Supplementary Figure 6, Z=1.20, P=0.23); (b) homozygotes 10/10 versus others (Supplementary Figure 6, Z=1.59, P=0.11), in absence of heterogeneity in effect sizes across the studies (Supplementary Figure 6, Z=0.53, I2=0% Z=0.23, I2=30%, respectively). The Egger test was not applied due to the presence of only two studies.
Pharmacogenetic studies not available for meta-analysis
The studies are reported in Supplementary Table 2.66, 114, 115 Kooij et al.66 investigated polymorphisms in DRD4 (120-bp insertion/deletion, 48-bp VNTR) and SLC6A2 (4-bp insertion/deletion in the promoter region) in 42 adults treated with IR-MPH. The primary study outcome was clinical response, defined a priori as a decrease of at least two points on the Investigator-based Clinical Global Impression-Severity scale for ADHD (CGI-S), as well as a 30% or greater symptom reduction as measured with the self-reported DSM-IV ADHD-rating scale (ADHD-RS). The two secondary measures of response were the same scales, taken separately. No associations were found with outcomes of treatment response.
Negative associations were also found for ADRA2A (rs1800544, rs1800545 and rs553668)115 and other 11 polymorphisms in 7 genes (HTR1B, SLC6A4, TPH2, DBH, DRD4, COMT and SNAP25).114 These studies were naturalistic and investigated almost 200 patients, treated with IR-MPH. The outcome measures of MPH response were the Swanson, Nolan and Pelham Rating Scale version IV (SNAP-IV) and the CGI-S scale. The final measurements were taken after the 30th day of treatment and the a priori definition of clinical response was a 30% or greater symptom reduction in SNAP-IV and a CGI-S score of two points or less.
Biochemical studies (metabolomics) available for meta-analysis
Twenty-one studies were identified for peripheral biochemical/metabolomic measures. Of these, 6 studies provided data for meta-analyses (Supplementary Figure 1) and were reported in Table 3.46, 69, 70, 71, 72, 73, 74
HPA axis (salivary cortisol)
Individuals with ADHD suffer from increased vulnerability to environmental and mental stressors and may be at increased risk for chronic stress. The HPA axis is a critical physiological system that mediates responses to stress. In the face of environmental stressors, the HPA axis is activated, resulting in the secretion of cortisol from the adrenal cortex to the blood stream. The secretion of cortisol, which represents a biological indicator of arousal, is an adaptive response that alerts the individual to environmental changes and promotes the recovery of homeostasis. Studies have found the measurement of cortisol as an indicator of adrenocortical activity to be of high predictive value for psychological stress.
Some studies were conducted on adults with ADHD measuring salivary cortisol at baseline as compared with control subjects.69, 70, 71 Corominas et al.116 was excluded because it did not include a comparison group of healthy individuals. Also Hirvikoski et al.46 was excluded. The results of our meta-analysis indicated no significant differences between patients and controls (Supplementary Figure 7, Z=1.52, P=0.13). Significant heterogeneity in effect sizes across studies was found (P<0.0003, I2=88%). No publication bias was observed (P=0.09; Supplementary Table 5).
Mono- and polyunsaturated fatty acid
The connection between ADHD and lipids has not been sufficiently investigated so far in adults. It has been hypothesized that ADHD symptomatology in adults could be associated with a deficiency or imbalance of polyunsaturated fatty acids (PUFAs), due to insufficient dietary intake of essential FAs, and/or impaired absorption of FAs, and/or an increased metabolism of FAs.74
Three studies investigated PUFA levels in adults with ADHD.72, 73, 74 Young et al.117 was excluded because it reported the effect of the FA treatment on the biochemical assays of the biomarker (Supplementary Table 3). The results of our meta-analyses indicated no significant differences between patients and controls for: serum arachidonic acid (AA) concentrations (Supplementary Figure 8, Z=0.99, P=0.32 with heterogeneity in effect size across the studies P=0.03, I2=72%), serum eicosapentaenoic acid (Supplementary Figure 8, Z=0.83, P=0.41; no heterogeneity in effect size across the studies P=0.11, I2=54%) and serum dihomogammalinolenic acid (Supplementary Figure 8, Z=0.70, P=0.49; heterogeneity in effect size across the studies P=0.005, I2=81%). A significant difference between patients and controls was observed for serum docosahexaenoic acid (DHA; Supplementary Figure 8, Z=4.14, P<0.0001), significant after Bonferroni correction. For DHA, no heterogeneity in effect size across the studies was observed (P=0.54, I2=0%) as well as no publication bias (P=0.19, Supplementary Table 5).
Biochemical studies (metabolomics) not available for meta-analysis
There are studies on biochemical levels of some proteins that are not enough to conduct meta-analysis (Supplementary Table 3).70, 73, 74, 104, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126, 127, 128, 129 Some studies regarded the circadian rhythms analyzing the melatonin levels,70, 118 others the oxidative stress pathway. In particular in a pioneering study,120 plasma homocysteine levels were lower and serum folic acid levels higher in adult ADHD patients when compared with controls, whereas no difference was found in serum vitamin B12, total antioxidant status, total oxidant status and the oxidative stress index. Contrarily another study reported that patients' total antioxidant status, total oxidant status and oxidative stress index were significantly higher than controls.123 Bulut et al.121 demonstrated alterations in malondialdehyde level in patients as compared with controls. In a successive study the same authors reported higher malondialdehyde and lower paraoxonase and arylesterase levels in ADHD adult patients when compared with healthy subjects.119 Finally a study demonstrated that the mean nitric oxide metabolite levels in patients were significantly higher than those of controls and superoxide dismutase activity of patients was significantly lower.122 In contrast, in another study the ADHD adult group did not show significant differences regarding NOx—levels compared with controls.104
Other relevant peripheral biomarkers are represented by the serum BDNF,126 adiponectin,124 albumin125 as we as the cerebrospinal fluid metabolites homovanillic acid and 5-hydroxyindoleacetic acid.128
Discussion
This work meta-analyzed available candidate gene association, pharmacogenetic and biochemical (metabolomics) studies of ADHD in adults. The main result obtained from the systematic review is that there are not enough studies to perform meta-analyses. Indeed among a total of 87 studies, 23 were effectively suitable for meta-analyses. The genes and biochemical measures showed by the remaining were too infrequently reported for meta-analysis. We obtained a positive association between adult ADHD and rs8079781 in BAIAP2 and with the rs8079626-rs7210438 GC haplotype, even after Bonferroni correction. A trend towards association was observed for rs4969385 in this gene. We found no heterogeneity in effect size and no publication bias for the BAIAP2 analyses. If we did not apply the Bonferroni correction, a trend was found for the carriers allele 9R of SLC6A3 40 bp VNTR and for 6/6 homozygotes of SLC6A3 30 bp, whereas negative results were obtained for the 9-6 haplotype, the DRD4 48 bp VNTR, and the COMT SNP, rs4680. Concerning pharmacogenetic studies, a negative association was found for the SLC6A3 40 bp VNTR for response to MPH, with only two studies available. For the metabolomics studies, no differences between ADHD adults and controls were found for salivary cortisol, whereas lower serum DHA levels were observed in ADHD adults. This latter association was significant even after Bonferroni correction and heterogeneity was not significant. Other PUFAs such as AA, eicosapentaenoic acid and dihomogammalinolenic acid serum levels were not different between patients and controls. No publication biases were observed for these markers. Genes linked to dopaminergic, serotoninergic and noradrenergic signaling, metabolism (DBH, TPH1, TPH2, DDC, MAOA, MAOB, BCHE and TH), neurodevelopment (BDNF and others), the SNARE system and other 40 genes/proteins related to different pathways were not meta-analyzed due to insufficient data.
SLC6A3 is a well-known gene having an important role in the pathophysiology of different psychiatric illnesses including ADHD. Dopamine is an important neuromodulator that is released prominently in frontal and striatal areas known to have an important role in cognitive function. Moreover the DAT has a key role in regulating striatal dopamine levels and the amount of DAT expression in striatum has in previous meta-analyses been shown to be associated with the 40 bp VNTR polymorphism in the SLC6A3 gene.75 Finally, the DAT is a key pharmacological target of two ADHD medications, amphetamine and MPH.130 Despite this robust rationale for being involved in ADHD, the meta-analyses on ADHD susceptibility as well as on MPH response showed no significant associations and lacking of consistent conclusions. If we do not apply the Bonferroni correction, the carriers of 9R allele seems to be associated to ADHD in adults, as well as the 6/6 homozygotes of 30 bp VNTR. Further studies are thus mandatory for confirming the hypothesized differential effect of the allele 9R associated to adults and of the allele 10R to children32, 34, 131 as well as the susceptibility of allele 6R observed both in adults (present data) and in children.26
Negative results were obtained also for one of the most studied potential susceptibility genes for the disorder, DRD4 and its well-known 48 bp VNTR, a potential functional polymorphism located in the third exon encoding the putative third cytoplasmic loop of the receptor that modulates the receptor's signal transduction proprieties by altering intracellular cyclic AMP levels.
Even the results for the COMT Val66Met variant showed no association to ADHD in adults, as has been observed for ADHD in children.78, 79 COMT is the major catecholamine-degrading enzyme involved in the degradation of catecholamines in synapses in the cerebral cortex, preferentially affecting prefrontal cortical dopamine metabolism. The Val66Met variant affects COMT enzyme activity, leading to significant alterations in dopamine levels in the post-synaptic neuron.
Thus all together these results suggest that it is needed to conduct further studies on SLC6A3, DRD4 and COMT genes given their relevant importance in ADHD. For instance it could be needed to consider other polymorphisms and/or haplotypes inside of these gene and a deep sequencing of implicated genomic regions may allow identification of the functional variants directly involved in the genetic background of ADHD. In that regard it is interesting to note that an increased burden of rare variants has been observed in the 7R allele of DRD4 in children with ADHD132 and in adults.59
Interesting data were obtained for BAIAP2. There are consistent data that support the existence of functional asymmetry in the brain.133, 134 In addition to environmental effects, growing evidence supports that genes have an essential role in the development of the human brain.135, 136 Although little is known about genetic factors underlying brain lateralization, several genes are differentially expressed in the two hemispheres, some of which could be involved in the development of right–left asymmetries. A relationship between genes differentially expressed in brain hemispheres and the vulnerability to ADHD was suggested, because patients with ADHD show deviations from the typical pattern of cerebral asymmetry that may account for a large number of ADHD-related symptoms.136, 137 Our meta-analyses conducted on three different population samples confirmed the role of BAIAP2 in the ADHD susceptibility in adults. BAIAP2 is expressed at higher levels in the left human cerebral cortex and participates in neuronal proliferation, survival and maturation. It encodes the insulin receptor tyrosine kinase substrate protein of 53 kDa (IRSp53), a member of a group of downstream signaling molecules that participate in the signal transduction pathways of insulin and insulin-like growth factor 1. Moreover BAIAP2 expression in rat cerebral cortices is enhanced by treatment with MPH. As demonstrated by Ribases et al.65 this association was found for ADHD in adults but not in children, suggesting a distinct genetic load between persistent and remitting ADHD22, 24, 83 and a potential genetic marker for persistent ADHD.
With regards to the biochemical studies (metabolomics), our meta-analysis on basal salivary cortisol levels showed no difference between patients and controls. The presence of heterogeneity due to the paper from Baird et al.70 suggests the needing to replicate this finding. Contrarily our meta-analysis on PUFAs supported a significant role of DHA in adult ADHD. Irmisch et al.74 showed that the DHA was associated with hyperactivity in adults with ADHD, and it seems to be essential for pre- and postnatal brain development.138 The role of n-3PUFAs had in ADHD may be explained by their impact on the immune system through the formation of immunosuppressive prostaglandins, a system involved in ADHD.139 Another hypothesis140 is based on the evidence that PUFA deficiency in rodents results in behavioral changes such as increased motor activity and decreased learning abilities on the grounds of dysregulated monoamine neurotransmission. A differential impact of cortisol and DHA on ADHD in childhood and in adulthood can be suggested. In fact a recent meta-analysis31 on peripheral biomarkers in ADHD childhood showed a significant role of salivary cortisol but not of serum DHA. Here in adults we demonstrated the opposite effect, supporting the idea that the concentrations of cortisol and FAs can vary in relation to age. However the course of salivary cortisol and serum FA composition during the lifespan needs to be further investigated.
It is intriguing that we found much more heterogeneity of results for the child studies26 compared with the adult studies. This may be due to the fact that child samples comprise those who will persist with ADHD in adulthood and those who will not. Also, because biological and psychological development change markedly in childhood, differences in developmental stage could account for the greater heterogeneity in childhood. Another possibility is that there is greater phenotypic heterogeneity in the child samples. We could not, however, address these issues with the data available to us. Nevertheless, future studies should use these heterogeneity findings to better design studies and to more thoroughly report dimensions of phenotypic heterogeneity in their sample.
All data suffer from one important limitation, which is their specificity for ADHD. In fact SLC6A3, DRD4, COMT have each been associated to other psychiatric pathologies such as major depressive disorder, schizophrenia, anxiety disorders, bipolar disorder and obsessive-compulsive disorder.141 The issue of comorbidity is vital because it is pervasive among adults with ADHD.142 Despite this fact, many studies of Axis-I disorders in adults do not address comorbidity, possibly because, for many years, ADHD had been perceived as a childhood disorder. For example, most of the adult studies used the SCID as diagnostic interview. As ADHD is not included in the SCID, it would have been overlooked. Thus, the lack of specificity for genetic associations could be due to the fact that studies of these other disorders did not assess ADHD. Non-specificity also suggests the possibility that common biological mechanisms linked to dopaminergic and noradrenergic systems are shared among different psychiatric disorders, as was recently supported.141 Future studies should include ADHD in the assessment of comorbidities and also address the concomitant treatment of ADHD and comorbid Axis-I disorders, which is new and an important issue.
This work presents some limitations. (1) For some meta-analyses, the sample sizes were small and with only two studies. (2) Differential proportion of remitting and persisting ADHD within the children samples may also explain discordant results among studies. (3) With regard to the pharmacogenetic studies, one study included a placebo arm.68
In conclusion, we found that there were not enough genetic, pharmacogenetic and biochemical studies of ADHD in adults and that more investigations are needed. Moreover we confirmed significant role of BAIAP2 and DHA in the etiology of ADHD exclusively in adults. Future research should be focused on the replication of these findings and to assess their specificity for ADHD.
Acknowledgments
This research was supported by grants from the Italian Ministry of Health (Ricerca Corrente). Dr Faraone is supported by the K.G. Jebsen Centre for Research on Neuropsychiatric Disorders, University of Bergen, Bergen, Norway, the European Community's Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 602805 and NIMH grant R01MH094469.
Footnotes
Supplementary Information accompanies the paper on the Molecular Psychiatry website (http://www.nature.com/mp)
In the past year, Dr Faraone received income, potential income, travel expenses and/or research support from Arbor, Pfizer, Ironshore, Shire, Akili Interactive Labs, CogCubed, Alcobra, VAYA Pharma, Neurovance, Impax, NeuroLifeSciences. With his institution, he has US patent US20130217707 A1 for the use of sodium-hydrogen exchange inhibitors in the treatment of ADHD. The remaining authors declare no conflict of interest.
Supplementary Material
References
- Wilens TE, Faraone SV, Biederman J. Attention-deficit/hyperactivity disorder in adults. JAMA 2004; 292: 619–623. [DOI] [PubMed] [Google Scholar]
- Faraone S, Biederman J, Mick E. The age dependent decline of attention-deficit/hyperactivity disorder: a meta-analysis of follow-up studies. Psychol Med 2006; 36: 159–165. [DOI] [PubMed] [Google Scholar]
- Polanczyk GV, Willcutt EG, Salum GA, Kieling C, Rohde LA. ADHD prevalence estimates across three decades: an updated systematic review and meta-regression analysis. Int J Epidemiol 2014; 43: 434–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willcutt EG. The prevalence of DSM-IV attention-deficit/hyperactivity disorder: a meta-analytic review. Neurotherapeutics 2012; 9: 490–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polanczyk G, de Lima MS, Horta BL, Biederman J, Rohde LA. The worldwide prevalence of ADHD: a systematic review and metaregression analysis. Am J Psychiatry 2007; 164: 942–948. [DOI] [PubMed] [Google Scholar]
- Ramos-Quiroga JA, Nasillo V, Fernández-Aranda F, Casas M. Addressing the lack of studies in attention-deficit/hyperactivity disorder in adults. Expert Rev Neurother 2014; 14: 553–567. [DOI] [PubMed] [Google Scholar]
- Simon V, Czobor P, Balint S, Meszaros A, Bitter I. Prevalence and correlates of adult attention-deficit hyperactivity disorder: meta-analysis. Br J Psychiatry 2009; 194: 204–211. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Swanson JM. Clinical practice: Adult attention deficit-hyperactivity disorder. N Engl J Med 2013; 369: 1935–1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moffitt TE, Houts R, Asherson P, Belsky DW, Corcoran DL, Hammerle M et al. Is adult ADHD a childhood-onset neurodevelopmental disorder? Evidence from a four-decade longitudinal cohort study. Am J Psychiatry 2015; 172: 967–977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katzman MA, Sternat T. A review of OROS methylphenidate (Concerta(®)) in the treatment of attention-deficit/hyperactivity disorder. CNS Drugs 2014; 28: 1005–1033. [DOI] [PubMed] [Google Scholar]
- Sugrue D, Bogner R, Ehret MJ. Methylphenidate and dexmethylphenidate formulations for children with attention-deficit/hyperactivity disorder. Am J Health Syst Pharm 2014; 71: 1163–1170. [DOI] [PubMed] [Google Scholar]
- Simon N, Rolland B, Karila L. Methylphenidate in adults with attention deficit hyperactivity disorder and substance use disorders. Curr Pharm Des 2015; 21: 3359–3366. [DOI] [PubMed] [Google Scholar]
- Fredriksen M, Halmøy A, Faraone SV, Haavik J. Long-term efficacy and safety of treatment with stimulants and atomoxetine in adult ADHD: a review of controlled and naturalistic studies. Eur Neuropsychopharmacol 2013; 23: 508–527. [DOI] [PubMed] [Google Scholar]
- Surman CB, Hammerness PG, Pion K, Faraone SV. Do stimulants improve functioning in adults with ADHD? A review of the literature. Eur Neuropsychopharmacol 2013; 23: 528–533. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Asherson P, Banaschewski T, Biederman J, Buitelaar JK, Ramos-Quiroga JA et al. Attention deficit hyperactivity disorder. Nat Rev Dis Primer 2015; 1: doi:10.1038/nrdp.2015.20. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Mick E. Molecular genetics of attention deficit hyperactivity disorder. Psychiatr Clin North Am 2010; 33: 159–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin J, O'Donovan MC, Thapar A, Langley K, Williams N. The relative contribution of common and rare genetic variants to ADHD. Transl Psychiatry 2015; 5: e506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kan KJ, Dolan CV, Nivard MG, Middeldorp CM, van Beijsterveldt CE, Willemsen G et al. Genetic and environmental stability in attention problems across the lifespan: evidence from the Netherlands twin register. J Am Acad Child Adolesc Psychiatry 2013; 52: 12–25. [DOI] [PubMed] [Google Scholar]
- Larsson H, Asherson P, Chang Z, Ljung T, Friedrichs B, Larsson JO et al. Genetic and environmental influences on adult attention deficit hyperactivity disorder symptoms: a large Swedish population-based study of twins. Psychol Med 2013; 43: 197–207. [DOI] [PubMed] [Google Scholar]
- Boomsma DI, Saviouk V, Hottenga JJ, Distel MA, de Moor MH, Vink JM et al. Genetic epidemiology of attention deficit hyperactivity disorder (ADHD index) in adults. PLoS One 2010; 5: e10621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biederman J, Faraone SV, Mick E, Spencer T, Wilens T, Kiely K et al. High risk for attention deficit hyperactivity disorder among children of parents with childhood onset of the disorder: a pilot study. Am J Psychiatry 1995; 152: 431–435. [DOI] [PubMed] [Google Scholar]
- Biederman J, Faraone S, Milberger S, Curtis S, Chen L, Marrs A et al. Predictors of persistence and remission of ADHD into adolescence: results from a four-year prospective follow-up study. J Am Acad Child Adolesc Psychiatry 1996; 35: 343–351. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Biederman J, Feighner JA, Monuteaux MC. Assessing symptoms of attention deficit hyperactivity disorder in children and adults: which is more valid? J Consult Clin Psychol 2000; 68: 830–842. [PubMed] [Google Scholar]
- Faraone SV, Biederman J, Monuteaux MC. Toward guidelines for pedigree selection in genetic studies of attention deficit hyperactivity disorder. Genet Epidemiol 2000; 18: 1–16. [DOI] [PubMed] [Google Scholar]
- Brikell I, Kuja-Halkola R, Larsson H. Heritability of attention-deficit hyperactivity disorder in adults. Am J Med Genet B Neuropsychiatr Genet 2015; 30; doi: 10.1002/ajmg.b.32335. [DOI] [PubMed] [Google Scholar]
- Gizer IR, Ficks C, Waldman ID. Candidate gene studies of ADHD: a meta-analytic review. Hum Genet 2009; 126: 51–90. [DOI] [PubMed] [Google Scholar]
- Hawi Z, Cummins TD, Tong J, Johnson B, Lau R, Samarrai W et al. The molecular genetic architecture of attention deficit hyperactivity disorder. Mol Psychiatry 2015; 20: 289–297. [DOI] [PubMed] [Google Scholar]
- Gao Q, Liu L, Qian Q, Wang Y. Advances in molecular genetic studies of attention deficit hyperactivity disorder in China. Shanghai Arch Psychiatry 2014; 26: 194–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Z, Chang SH, Zhang LY, Gao L, Wang J. Molecular genetic studies of ADHD and its candidate genes: a review. Psychiatry Res 2014; 219: 10–24. [DOI] [PubMed] [Google Scholar]
- Bruxel EM, Akutagava-Martins GC, Salatino-Oliveira A, Contini V, Kieling C, Hutz MH et al. ADHD pharmacogenetics across the life cycle: new findings and perspectives. Am J Med Genet B Neuropsychiatr Genet 2014; 165B: 263–282. [DOI] [PubMed] [Google Scholar]
- Scassellati C, Bonvicini C, Faraone SV, Gennarelli M. Biomarkers and attention-deficit/hyperactivity disorder: a systematic review and meta-analyses. J Am Acad Child Adolesc Psychiatry 2012; 51: e20. [DOI] [PubMed] [Google Scholar]
- Franke B, Faraone SV, Asherson P, Buitelaar J, Bau CH, Ramos-Quiroga JA et al. The genetics of attention deficit/hyperactivity disorder in adults, a review. Mol Psychiatry 2012; 17: 960–987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Mora C, Ribasés M, Casas M, Bayés M, Bosch R, Fernàndez-Castillo N et al. Exploring DRD4 and its interaction with SLC6A3 as possible risk factors for adult ADHD: a meta-analysis in four European populations. Am J Med Genet B Neuropsychiatr Genet 2011; 156B: 600–612. [DOI] [PubMed] [Google Scholar]
- Franke B, Vasquez AA, Johansson S, Hoogman M, Romanos J, Boreatti-Hümmer A et al. Multicenter analysis of the SLC6A3/DAT1 VNTR haplotype in persistent ADHD suggests differential involvement of the gene in childhood and persistent ADHD. Neuropsychopharmacology 2010; 35: 656–664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landaas ET, Johansson S, Jacobsen KK, Ribasés M, Bosch R, Sánchez-Mora C et al. An international multicenter association study of the serotonin transporter gene in persistent ADHD. Genes Brain Behav 2010; 9: 449–458. [DOI] [PubMed] [Google Scholar]
- Johansson S, Halmøy A, Mavroconstanti T, Jacobsen KK, Landaas ET, Reif A et al. Common variants in the TPH1 and TPH2 regions are not associated with persistent ADHD in a combined sample of 1,636 adult cases and 1,923 controls from four European populations. Am J Med Genet B Neuropsychiatr Genet 2010; 153B: 1008–1015. [DOI] [PubMed] [Google Scholar]
- Sánchez-Mora C, Ribasés M, Ramos-Quiroga JA, Casas M, Bosch R, Boreatti-Hümmer A et al. Meta-analysis of brain-derived neurotrophic factor p.Val66Met in adult ADHD in four European populations. Am J Med Genet B Neuropsychiatr Genet 2010; 153B: 512–523. [DOI] [PubMed] [Google Scholar]
- Reif A, Nguyen TT, Weissflog L, Jacob CP, Romanos M, Renner TJ et al. DIRAS2 is associated with adult ADHD, related traits, and co-morbid disorders. Neuropsychopharmacology 2011; 36: 2318–2327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Mora C, Ramos-Quiroga JA, Bosch R, Corrales M, Garcia-Martínez I, Nogueira M et al. Case-control genome-wide association study of persistent attention-deficit hyperactivity disorder identifies FBXO33 as a novel susceptibility gene for the disorder. Neuropsychopharmacology 2015; 40: 915–926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lesch KP, Timmesfeld N, Renner TJ, Halperin R, Röser C, Nguyen TT et al. Molecular genetics of adult ADHD: converging evidence from genome-wide association and extended pedigree linkage studies. J Neural Transm (Vienna) 2008; 115: 1573–1585. [DOI] [PubMed] [Google Scholar]
- Akutagava-Martins GC, Salatino-Oliveira A, Genro JP, Contini V, Polanczyk G, Zeni C et al. Glutamatergic copy number variants and their role in attention-deficit/hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet 2014; 165B: 502–509. [DOI] [PubMed] [Google Scholar]
- Ramos-Quiroga JA, Sánchez-Mora C, Casas M, Garcia-Martínez I, Bosch R, Nogueira M et al. Genome-wide copy number variation analysis in adult attention-deficit and hyperactivity disorder. J Psychiatr Res 2014; 49: 60–67. [DOI] [PubMed] [Google Scholar]
- Rovaris DL, Mota NR, da Silva BS, Girardi P, Victor MM, Grevet EH et al. Should we keep on? Looking into pharmacogenomics of ADHD in adulthood from a different perspective. Pharmacogenomics 2014; 15: 1365–1381. [DOI] [PubMed] [Google Scholar]
- Contini V, Rovaris DL, Victor MM, Grevet EH, Rohde LA, Bau CH. Pharmacogenetics of response to methylphenidate in adult patients with attention-deficit/hyperactivity disorder (ADHD): a systematic review. Eur Neuropsychopharmacol 2013; 23: 555–560. [DOI] [PubMed] [Google Scholar]
- Dresler T, Ehlis AC, Heinzel S, Renner TJ, Reif A, Baehne CG et al. Dopamine transporter (SLC6A3) genotype impacts neurophysiological correlates of cognitive response control in an adult sample of patients with ADHD. Neuropsychopharmacology 2010; 35: 2193–2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirvikoski T, Lindholm T, Nordenstrom A, Nordstrom AL, Lajic S. High self-perceived stress and many stressors, but normal diurnal cortisol rhythm, in adults with ADHD (attention-deficit/hyperactivity disorder). Horm Behav 2009; 55: 418–424. [DOI] [PubMed] [Google Scholar]
- Egger M, Smith GD, Phillips AN. Meta-analysis: principles and procedures. BMJ 1997; 315: 1533–1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ribasés M, Ramos-Quiroga JA, Hervás A, Sánchez-Mora C, Bosch R, Bielsa A et al. Candidate system analysis in ADHD: evaluation of nine genes involved in dopaminergic neurotransmission identifies association with DRD1. World J Biol Psychiatry 2012; 13: 281–292. [DOI] [PubMed] [Google Scholar]
- de Azeredo LA, Rovaris DL, Mota NR, Polina ER, Marques FZ, Contini V et al. Further evidence for the association between a polymorphism in the promoter region of SLC6A3/DAT1 and ADHD: findings from a sample of adults. Eur Arch Psychiatry Clin Neurosci 2014; 264: 401–408. [DOI] [PubMed] [Google Scholar]
- Brüggemann D, Sobanski E, Alm B, Schubert T, Schmalzried H, Philipsen A et al. No association between a common haplotype of the 6 and 10-repeat alleles in intron 8 and the 3'UTR of the DAT1 gene and adult attention deficit hyperactivity disorder. Psychiatr Genet 2007; 17: 121. [DOI] [PubMed] [Google Scholar]
- Silva MA, Cordeiro Q, Louzã M, Vallada H. Association between a SLC6A3 intron 8 VNTR functional polymorphism and ADHD in a Brazilian sample of adult patients. Rev Bras Psiquiatr 2009; 31: 390–395. [DOI] [PubMed] [Google Scholar]
- Hoogman M, Onnink M, Cools R, Aarts E, Kan C, Arias Vasquez A et al. The dopamine transporter haplotype and reward-related striatal responses in adult ADHD. Eur Neuropsychopharmacol 2013; 23: 469–478. [DOI] [PubMed] [Google Scholar]
- Spencer TJ, Biederman J, Faraone SV, Madras BK, Bonab AA, Dougherty DD et al. Functional genomics of attention-deficit/hyperactivity disorder (ADHD) risk alleles on dopamine transporter binding in ADHD and healthy control subjects. Biol Psychiatry 2013; 74: 84–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson S, Halleland H, Halmøy A, Jacobsen KK, Landaas ET, Dramsdahl M et al. Genetic analyses of dopamine related genes in adult ADHD patients suggest an association with the DRD5-microsatellite repeat, but not with DRD4 or SLC6A3 VNTRs. Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 1470–1475. [DOI] [PubMed] [Google Scholar]
- Aparecida da Silva M, Cordeiro Q, Louzã M, Vallada H. Lack of association between a 3'UTR VNTR polymorphism of dopamine transporter gene (SLC6A3) and ADHD in a Brazilian sample of adult patients. J Atten Disord 2011; 15: 305–309. [DOI] [PubMed] [Google Scholar]
- Brown AB, Biederman J, Valera E, Makris N, Doyle A, Whitfield-Gabrieli S et al. Relationship of DAT1 and adult ADHD to task-positive and task-negative working memory networks. Psychiatry Res 2011; 193: 7–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasler R, Salzmann A, Bolzan T, Zimmermann J, Baud P, Giannakopoulos P et al. DAT1 and DRD4 genes involved in key dimensions of adult ADHD. Neurol Sci 2015; 36: 861–869. [DOI] [PubMed] [Google Scholar]
- Muglia P, Jain U, Macciardi F, Kennedy JL. Adult attention deficit hyperactivity disorder and the dopamine D4 receptor gene. Am J Med Genet 2000; 96: 273–277. [DOI] [PubMed] [Google Scholar]
- Tovo-Rodrigues L, Rohde LA, Roman T, Schmitz M, Polanczyk G, Zeni C et al. Is there a role for rare variants in DRD4 gene in the susceptibility for ADHD? Searching for an effect of allelic heterogeneity. Mol Psychiatry 2012; 17: 520–526. [DOI] [PubMed] [Google Scholar]
- Carpentier PJ, Arias Vasquez A, Hoogman M, Onnink M, Kan CC, Kooij JJ et al. Shared and unique genetic contributions to attention deficit/hyperactivity disorder and substance use disorders: a pilot study of six candidate genes. Eur Neuropsychopharmacol 2013; 23: 448–457. [DOI] [PubMed] [Google Scholar]
- Ghosh P, Sarkar K, Bhaduri N, Ray A, Sarkar K, Sinha S et al. Catecholaminergic gene variants: contribution in ADHD and associated comorbid attributes in the eastern Indian probands. Biomed Res Int 2013; 2013: 918410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halleland H, Lundervold AJ, Halmøy A, Haavik J, Johansson S. Association between catechol O-methyltransferase (COMT) haplotypes and severity of hyperactivity symptoms in adults. Am J Med Genet B Neuropsychiatr Genet 2009; 150B: 403–410. [DOI] [PubMed] [Google Scholar]
- Biehl SC, Gschwendtner KM, Guhn A, Müller LD, Reichert S, Heupel J et al. Does adult ADHD interact with COMT val (158) met genotype to influence working memory performance? Atten Defic Hyperact Disord 2015; 7: 19–25. [DOI] [PubMed] [Google Scholar]
- Pazvantoğlu O, Güneş S, Karabekiroğlu K, Yeğin Z, Erenkuş Z, Akbaş S et al. The relationship between the presence of ADHD and certain candidate gene polymorphisms in a Turkish sample. Gene 2013; 528: 320–327. [DOI] [PubMed] [Google Scholar]
- Ribasés M, Bosch R, Hervás A, Ramos-Quiroga JA, Sánchez-Mora C, Bielsa A et al. Case-control study of six genes asymmetrically expressed in the two cerebral hemispheres: association of BAIAP2 with attention-deficit/hyperactivity disorder. Biol Psychiatry 2009; 66: 926–934. [DOI] [PubMed] [Google Scholar]
- Kooij JS, Boonstra AM, Vermeulen SH, Heister AG, Burger H, Buitelaar JK et al. Response to methylphenidate in adults with ADHD is associated with a polymorphism in SLC6A3 (DAT1). Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 201–208. [DOI] [PubMed] [Google Scholar]
- Contini V, Victor MM, Marques FZ, Bertuzzi GP, Salgado CA, Silva KL et al. Response to methylphenidate is not influenced by DAT1 polymorphisms in a sample of Brazilian adult patients with ADHD. J Neural Transm 2010; 117: 269–276. [DOI] [PubMed] [Google Scholar]
- Mick E, Biederman J, Spencer T, Faraone SV, Sklar P. Absence of association with DAT1 polymorphism and response to methylphenidate in a sample of adults with ADHD. Am J Med Genet B Neuropsychiatr Genet 2006; 141B: 890–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lackschewitz H, Huther G, Kroner-Herwig B. Physiological and psychological stress responses in adults with attention-deficit/hyperactivity disorder (ADHD). Psychoneuroendocrinology 2008; 33: 612–624. [DOI] [PubMed] [Google Scholar]
- Baird AL, Coogan AN, Siddiqui A, Donev RM, Thome J. Adult attention-deficit hyperactivity disorder is associated with alterations in circadian rhythms at the behavioural, endocrine and molecular levels. Mol Psychiatry 2012; 17: 988–995. [DOI] [PubMed] [Google Scholar]
- Raz S, Leykin D. Psychological and cortisol reactivity to experimentally induced stress in adults with ADHD. Psychoneuroendocrinology 2015; 60: 7–17. [DOI] [PubMed] [Google Scholar]
- Young GS, Maharaj NJ, Conquer JA. Blood phospholipid fatty acid analysis of adults with and without attention deficit/hyperactivity disorder. Lipids 2004; 39: 117–123. [DOI] [PubMed] [Google Scholar]
- Laasonen M, Hokkanen L, Leppamaki S, Tani P, Erkkila AT. Project DyAdd: fatty acids in adult dyslexia, ADHD, and their comorbid combination. Prostaglandins Leukot Essent Fatty Acids 2009; 81: 89–96. [DOI] [PubMed] [Google Scholar]
- Irmisch G, Richter J, Thome J, Sheldrick AJ, Wandschneider R. Altered serum mono- and polyunsaturated fatty acid levels in adults with ADHD. ADHD Atten Def Hyp Disord 2013; 5: 303–311. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Spencer TJ, Madras BK, Zhang-James Y, Biederman J. Functional effects of dopamine transporter gene genotypes on in vivo dopamine transporter functioning: a meta-analysis. Mol Psychiatry 2014; 19: 880–889. [DOI] [PubMed] [Google Scholar]
- Wu J, Xiao HF, Sun HJ, Zou L, Zhu LQ. Role of dopamine receptors in ADHD: a systematic meta-analysis. Mol Neurobiol 2012; 45: 605–620. [DOI] [PubMed] [Google Scholar]
- Smith TF. Meta-analysis of the heterogeneity in association of DRD4 7-repeat allele and AD/HD: stronger association with AD/HD combined type. Am J Med Genet B Neuropsychiatr Genet 2010; 153B: 1189–1199. [DOI] [PubMed] [Google Scholar]
- Lee YH, Song GG. BDNF 196 G/A and COMT Val158Met polymorphisms and susceptibility to ADHD: a meta-analysis. J Atten Disord 2015; doi: 10.1177/1087054715570389. [DOI] [PubMed]
- Sun HJ, Yuan FF, Shen XM, Xiong GL, Wu J. Role of COMT in ADHD: a systematic meta-analysis. Mol Neurobiol 2014; 49: 251–261. [DOI] [PubMed] [Google Scholar]
- Squassina A, Lanktree M, De Luca V, Jain U, Krinsky M, Kennedy JL et al. Investigation of the dopamine D5 receptor gene (DRD5) in adult attention deficit hyperactivity disorder. Neurosci Lett 2008; 432: 50–53. [DOI] [PubMed] [Google Scholar]
- Muglia P, Jain U, Kennedy JL. A transmission disequilibrium test of the Ser9/Gly dopamine D3 receptor gene polymorphism in adult attention-deficit hyperactivity disorder. Behav Brain Res 2002; 130: 91–95. [DOI] [PubMed] [Google Scholar]
- Mota NR, Rovaris DL, Kappel DB, Picon FA, Vitola ES, Salgado CA et al. NCAM1-TTC12-ANKK1-DRD2 gene cluster and the clinical and genetic heterogeneity of adults with ADHD. Am J Med Genet B Neuropsychiatr Genet 2015; doi: 10.1002/ajmg.b.32317. [DOI] [PubMed]
- Ribasés M, Ramos-Quiroga JA, Hervás A, Bosch R, Bielsa A, Gastaminza X et al. Exploration of 19 serotoninergic candidate genes in adults and children with attention-deficit/hyperactivity disorder identifies association for 5HT2A, DDC and MAOB. Mol Psychiatry 2009; 14: 71–85. [DOI] [PubMed] [Google Scholar]
- De Luca V, Muglia P, Jain U, Kennedy JL. No evidence of linkage or association between the norepinephrine transporter (NET) gene MnlI polymorphism and adult ADHD. Am J Med Genet B Neuropsychiatr Genet 2004; 124B: 38–40. [DOI] [PubMed] [Google Scholar]
- de Cerqueira CC, Polina ER, Contini V, Marques FZ, Grevet EH, Salgado CA et al. ADRA2A polymorphisms and ADHD in adults: possible mediating effect of personality. Psychiatry Res 2011; 186: 345–350. [DOI] [PubMed] [Google Scholar]
- De Luca V, Muglia P, Vincent JB, Lanktree M, Jain U, Kennedy JL. Adrenergic alpha 2C receptor genomic organization: association study in adult ADHD. Am J Med Genet B Neuropsychiatr Genet 2004; 127B: 65–67. [DOI] [PubMed] [Google Scholar]
- Inkster B, Muglia P, Jain U, Kennedy JL. Linkage disequilibrium analysis of the dopamine beta-hydroxylase gene in persistent attention deficit hyperactivity disorder. Psychiatr Genet 2004; 14: 117–120. [DOI] [PubMed] [Google Scholar]
- Hess C, Reif A, Strobel A, Boreatti-Hümmer A, Heine M, Lesch KP et al. A functional dopamine-beta-hydroxylase gene promoter polymorphism is associated with impulsive personality styles, but not with affective disorders. J Neural Transm 2009; 116: 121–130. [DOI] [PubMed] [Google Scholar]
- Baehne CG, Ehlis AC, Plichta MM, Conzelmann A, Pauli P, Jacob C et al. Tph2 gene variants modulate response control processes in adult ADHD patients and healthy individuals. Mol Psychiatry 2009; 14: 1032–1039. [DOI] [PubMed] [Google Scholar]
- Jacob CP, Weber H, Retz W, Kittel-Schneider S, Heupel J, Renner T et al. Acetylcholine-metabolizing butyrylcholinesterase (BCHE) copy number and single nucleotide polymorphisms and their role in attention-deficit/hyperactivity syndrome. J Psychiatr Res 2013; 47: 1902–1908. [DOI] [PubMed] [Google Scholar]
- Lanktree M, Squassina A, Krinsky M, Strauss J, Jain U, Macciardi F et al. Association study of brain-derived neurotrophic factor (BDNF) and LIN-7 homolog (LIN-7) genes with adult attention-deficit/hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 945–951. [DOI] [PubMed] [Google Scholar]
- Ribasés M, Hervás A, Ramos-Quiroga JA, Bosch R, Bielsa A, Gastaminza X et al. Association study of 10 genes encoding neurotrophic factors and their receptors in adult and child attention-deficit/hyperactivity disorder. Biol Psychiatry 2008; 63: 935–945. [DOI] [PubMed] [Google Scholar]
- Arcos-Burgos M, Jain M, Acosta MT, Shively S, Stanescu H, Wallis D et al. A common variant of the latrophilin 3 gene, LPHN3, confers susceptibility to ADHD and predicts effectiveness of stimulant medication. Mol Psychiatry 2010; 15: 1053–1066. [DOI] [PubMed] [Google Scholar]
- Ribasés M, Ramos-Quiroga JA, Sánchez-Mora C, Bosch R, Richarte V, Palomar G et al. Contribution of LPHN3 to the genetic susceptibility to ADHD in adulthood: a replication study. Genes Brain Behav 2011; 10: 149–157. [DOI] [PubMed] [Google Scholar]
- Mavroconstanti T, Johansson S, Winge I, Knappskog PM, Haavik J. Functional properties of rare missense variants of human CDH13 found in adult attention deficit/hyperactivity disorder (ADHD) patients. PLoS One 2013; 8: e71445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salatino-Oliveira A, Genro JP, Polanczyk G, Zeni C, Schmitz M, Kieling C et al. Cadherin-13 gene is associated with hyperactive/impulsive symptoms in attention/deficit hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet 2015; 168B: 162–169. [DOI] [PubMed] [Google Scholar]
- Herken H, Erdal ME, Kenar AN, Unal GA, Cakaloz B, Ay ME et al. Association of SNAP-25 gene Ddel and Mnll polymorphisms with adult attention deficit hyperactivity disorder. Psychiatry Investig 2014; 11: 476–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olgiati P, Mandelli L, Alberti S, Lia L, Serretti A, Tiwari AK et al. Role of synaptosome-related (SNARE) genes in adults with attention deficit hyperactivity disorder. Psychiatry Res 2014; 215: 799–800. [DOI] [PubMed] [Google Scholar]
- Sánchez-Mora C, Cormand B, Ramos-Quiroga JA, Hervás A, Bosch R, Palomar G et al. Evaluation of common variants in 16 genes involved in the regulation of neurotransmitter release in ADHD. Eur Neuropsychopharmacol 2013; 23: 426–435. [DOI] [PubMed] [Google Scholar]
- Kenar AN, Oİ Ay, Herken H, Erdal ME. Association of VAMP-2 and Syntaxin 1 A genes with adult attention deficit hyperactivity disorder. Psychiatry Investig 2014; 11: 76–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenar AN, Edgünlü T, Herken H, Erdal ME. Association of synapsin III gene with adult attention deficit hyperactivity disorder. DNA Cell Biol 2013; 32: 430–434. [DOI] [PubMed] [Google Scholar]
- Reif A, Jacob CP, Rujescu D, Herterich S, Lang S, Gutknecht L et al. Influence of functional variant of neuronal nitric oxide synthase on impulsive behaviors in humans. Arch Gen Psychiatry 2009; 66: 41–50. [DOI] [PubMed] [Google Scholar]
- Hoogman M, Aarts E, Zwiers M, Slaats-Willemse D, Naber M, Onnink M et al. Nitric oxide synthase genotype modulation of impulsivity and ventral striatal activity in adult ADHD patients and healthy comparison subjects. Am J Psychiatry 2011; 168: 1099–1106. [DOI] [PubMed] [Google Scholar]
- Kittel-Schneider S, Reuß M, Meyer A, Weber H, Gessner A, Leistner C et al. Multi-level biomarker analysis of nitric oxide synthase isoforms in bipolar disorder and adult ADHD. J Psychopharmacol 2015; 29: 31–38. [DOI] [PubMed] [Google Scholar]
- Kissling C, Retz W, Wiemann S, Coogan AN, Clement RM, Hünnerkopf R et al. A polymorphism at the 3'-untranslated region of the CLOCK gene is associated with adult attention-deficit hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet 2008; 147: 333–338. [DOI] [PubMed] [Google Scholar]
- De Luca V, Muglia P, Jain U, Basile VS, Sokolowski MB, Kennedy JL. A drosophila model for attention deficit hyperactivity disorder (ADHD): no evidence of association with PRKG1 gene. Neuromol Med 2002; 2: 281–287. [DOI] [PubMed] [Google Scholar]
- Kortmann GL, Contini V, Bertuzzi GP, Mota NR, Rovaris DL, Paixão-Côrtes VR et al. The role of a mineralocorticoid receptor gene functional polymorphism in the symptom dimensions of persistent ADHD. Eur Arch Psychiatry Clin Neurosci 2013; 263: 181–188. [DOI] [PubMed] [Google Scholar]
- Weber H, Kittel-Schneider S, Gessner A, Domschke K, Neuner M, Jacob CP et al. Cross-disorder analysis of bipolar risk genes: further evidence of DGKH as a risk gene for bipolar disorder, but also unipolar depression and adult ADHD. Neuropsychopharmacology 2011; 36: 2076–2085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber H, Scholz CJ, Jacob CP, Heupel J, Kittel-Schneider S, Erhardt A et al. SPOCK3, a risk gene for adult ADHD and personality disorders. Eur Arch Psychiatry Clin Neurosci 2014; 64: 409–421. [DOI] [PubMed] [Google Scholar]
- Jacobsen KK, Halmøy A, Sánchez-Mora C, Ramos-Quiroga JA, Cormand B, Haavik J et al. DISC1 in adult ADHD patients: an association study in two European samples. Am J Med Genet B Neuropsychiatr Genet 2013; 162B: 227–234. [DOI] [PubMed] [Google Scholar]
- Weißflog L, Scholz CJ, Jacob CP, Nguyen TT, Zamzow K, Groß-Lesch S et al. KCNIP4 as a candidate gene for personality disorders and adult ADHD. Eur Neuropsychopharmacol 2013; 23: 436–447. [DOI] [PubMed] [Google Scholar]
- Jacob C, Nguyen TT, Weißflog L, Herrmann M, Liedel S, Zamzow K et al. PPP2R2C as a candidate gene of a temperament and character trait-based endophenotype of ADHD. Atten Defic Hyperact Disord 2012; 4: 145–152. [DOI] [PubMed] [Google Scholar]
- Rebases M, Sánchez-Mora C, Ramos-Quiroga JA, Bosch R, Gómez N, Nogueira M et al. An association study of sequence variants in the forkhead box P2 (FOXP2) gene and adulthood attention-deficit/hyperactivity disorder in two European samples. Psychiatr Genet 2012; 22: 155–160. [DOI] [PubMed] [Google Scholar]
- Contini V, Victor MM, Bertuzzi GP, Salgado CA, Picon FA, Grevet EH et al. No significant association between genetic variants in seven candidate genes and response to methylphenidate treatment in adult patients with ADHD. J Clin Psychopharmacol 2012; 32: 820–823. [DOI] [PubMed] [Google Scholar]
- Contini V, Victor MM, Cerqueira CC, Polina ER, Grevet EH, Salgado CA et al. Adrenergic alpha 2 A receptor gene is not associated with methylphenidate response in adults with ADHD. Eur Arch Psychiatry Clin Neurosci 2011; 261: 205–211. [DOI] [PubMed] [Google Scholar]
- Corominas M, Ribes V, Palomar G, Ramos-Quiroga JA, Ferrer R, Catalan R et al. Cortisol awakening response (CAR)in adults with attention deficit hyperactivity disorder (ADHD). Eur Psychiatry 2013; 28: 1.21920709 [Google Scholar]
- Young GS, Conquer JA, Thomas R. Effect of randomized supplementation with high dose olive, flax or fish oil on serum phospholipid fatty acid levels in adults with attention deficit hyperactivity disorder. Reprod Nutr Dev 2005; 45: 549–558. [DOI] [PubMed] [Google Scholar]
- Van Veen MM, Kooij JJ, Boonstra AM, Gordijn MC, Van Someren EJ. Delayed circadian rhythm in adults with attention-deficit/hyperactivity disorder and chronic sleep-onset insomnia. Biol Psychiatry 2010; 67: 1091–1096. [DOI] [PubMed] [Google Scholar]
- Bulut M, Selek S, Bez Y, Kaya MC, Gunes M, Karababa F et al. Lipid peroxidation markers in adult attention deficit hyperactivity disorder: new findings for oxidative stress. Psychiatry Res 2013; 209: 638–642. [DOI] [PubMed] [Google Scholar]
- Karababa IF, Savas SN, Selek S, Cicek E, Cicek EI, Asoglu M et al. Homocysteine Levels and Oxidative Stress Parameters in Patients With Adult ADHD. J Atten Disord 2014; doi: 10.1177/1087054714538657. [DOI] [PubMed]
- Bulut M, Selek S, Gergerlioglu HS, Savas HA, Yilmaz HR, Yuce M et al. Malondialdehyde levels in adult attention-deficit hyperactivity disorder. J Psychiatry Neurosci 2007; 32: 435–438. [PMC free article] [PubMed] [Google Scholar]
- Selek S, Savas H, Gergerlioglu S, Bulut M, Yilmaz HR. Oxidative imbalance in adult attention deficit/hyperactivity disorder. Biol Psychol 2008; 79: 256–259. [DOI] [PubMed] [Google Scholar]
- Selek S, Bulut M, Ocak AR, Kalenderoglu A, Savas H. Evaluation of total oxidative status in adult attention deficit hyperactivity disorder and its diagnostic implications. J Psychiatr Res 2012; 46: 451–455. [DOI] [PubMed] [Google Scholar]
- Mavroconstanti T, Halmøy A, Haavik J. Decreased serum levels of adiponectin in adult attention deficit hyperactivity disorder. Psychiatry Res 2014; 216: 123–130. [DOI] [PubMed] [Google Scholar]
- Grabemann M, Mette C, Zimmermann M, Wiltfang J, Kis B. Serum albumin correlates with affective prosody in adult males with attention-deficit hyperactivity disorder. Psychiatry Res 2014; 217: 198–201. [DOI] [PubMed] [Google Scholar]
- Corominas-Roso M, Ramos-Quiroga JA, Ribases M, Sanchez-Mora C, Palomar G, Valero S et al. Decreased serum levels of brain-derived neurotrophic factor in adults with attention-deficit hyperactivity disorder. Int J Neuropsychopharmacol 2013; 30: 1–9. [DOI] [PubMed] [Google Scholar]
- Ramos-Quiroga JA, Corominas-Roso M, Palomar G, Gomez-Barros N, Ribases M, Sanchez-Mora C et al. Changes in the serum levels of brain-derived neurotrophic factor in adults with attention deficit hyperactivity disorder after treatment with atomoxetine. Psychopharmacology (Berl) 2014; 231: 1389–1395. [DOI] [PubMed] [Google Scholar]
- Reimherr FW, Wender PH, Ebert MH, Wood DR. Cerebrospinal fluid homovanillic acid and 5-hydroxy-indoleacetic acid in adults with attention deficit disorder, residual type. Psychiatry Res 1984; 11: 71–78. [DOI] [PubMed] [Google Scholar]
- Ernst M, Liebenauer LL, Tebeka D, Jons PH, Eisenhofer G, Murphy DL et al. Selegiline in ADHD adults: plasma monoamines and monoamine metabolites. Neuropsychopharmacology 1997; 16: 276–284. [DOI] [PubMed] [Google Scholar]
- Coghill DR, Banaschewski T, Lecendreux M, Soutullo C, Zuddas A, Adeyi B et al. Post hoc analyses of the impact of previous medication on the efficacy of lisdexamfetamine dimesylate in the treatment of attention-deficit/hyperactivity disorder in a randomized, controlled trial. Neuropsychiatr Dis Treat 2014; 10: 2039–2047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franke B, Hoogman M, Arias Vasquez A, Heister JG, Savelkoul PJ, Naber M et al. Association of the dopamine transporter (SLC6A3/DAT1) gene 9-6 haplotype with adult ADHD. Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 1576–1579. [DOI] [PubMed] [Google Scholar]
- Grady DL, Chi HC, Ding YC, Smith M, Wang E, Schuck S et al. High prevalence of rare dopamine receptor D4 alleles in children diagnosed with attention-deficit hyperactivity disorder. Mol Psychiatry 2003; 8: 536–545. [DOI] [PubMed] [Google Scholar]
- Sun T, Patoine C, Abu-Khalil A, Visvader J, Sum E, Cherry TJ et al. Early asymmetry of gene transcription in embryonic human left and right cerebral cortex. Science 2005; 308: 1794–1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geschwind DH, Miller BL, DeCarli C, Carmelli D. Heritability of lobar brain volumes in twins supports genetic models of cerebral laterality and handedness. Proc Natl Acad Sci USA 2002; 99: 3176–3181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson PM, Cannon TD, Narr KL, van Erp T, Putanen VP, Huttunen M et al. Genetic influences on brain structure. Nat Neurosci 2001; 4: 1253–1258. [DOI] [PubMed] [Google Scholar]
- Smalley SL, Loo SK, Yang MH, Cantor RM. Toward localizing genes underlying cerebral asymmetry and mental health. Am J Med Genet B Neuropsychiatr Genet 2005; 135: 79–84. [DOI] [PubMed] [Google Scholar]
- Oades RD. Frontal, temporal and lateralized brain function in children with attention-deficit hyperactivity disorder: a psychophysiological and neuropsychological viewpoint on development. Behav Brain Res 1998; 94: 83–95. [DOI] [PubMed] [Google Scholar]
- Rogers CE, Anderson PJ, Thompson DK, Kidokoro H, Wallendorf M, Treyvaud K et al. Regional cerebral development at term relates to school-age social-emotional development in very preterm children. J Am Acad Child Adolesc Psychiatry 2012; 51: 181–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verlaet AA, Noriega DB, Hermans N, Savelkoul HF. Nutrition, immunological mechanisms and dietary immunomodulation in ADHD. Eur Child Adolesc Psychiatry 2014; 23: 519–529. [DOI] [PubMed] [Google Scholar]
- Transler C, Mitchell S, Eilander A. Could polyunsaturated fatty acids deficiency explain some dysfunctions found in ADHD? Hypotheses from animal research. J Atten Disord 2013; 17: 20–28. [DOI] [PubMed] [Google Scholar]
- Gatt JM, Burton KL, Williams LM, Schofield PR. Specific and common genes implicated across major mental disorders: a review of meta-analysis studies. J Psychiatr Res 2015; 60: 1–13. [DOI] [PubMed] [Google Scholar]
- Kessler RC, Adler L, Barkley R, Biederman J, Conners CK, Demler O et al. The prevalence and correlates of adult ADHD in the United States: results from the National Comorbidity Survey Replication. Am J Psychiatry 2006; 163: 716–723. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.