Abstract
Neutrophils are immune cells that are well known to be present during many types of lung diseases associated with acute respiratory distress syndrome (ARDS) and may contribute to acute lung injury. Neutrophils are poorly studied with respect to viral infection, and specifically to respiratory viral disease. Influenza A virus (IAV) infection is the cause of a respiratory disease that poses a significant global public health concern. Influenza disease presents as a relatively mild and self-limiting although highly pathogenic forms exist. Neutrophils increase in the respiratory tract during infection with mild seasonal IAV, moderate and severe epidemic IAV infection, and emerging highly pathogenic avian influenza (HPAI). During severe influenza pneumonia and HPAI infection, the number of neutrophils in the lower respiratory tract is correlated with disease severity. Thus, comparative analyses of the relationship between IAV infection and neutrophils provide insights into the relative contribution of host and viral factors that contribute to disease severity. Herein, we review the contribution of neutrophils to IAV disease pathogenesis and to other respiratory virus infections.
Keywords: neutrophil, influenza, acute respiratory distress syndrome, respiratory virus, viral microenvironment
Introduction
Neutrophils are a type of polymorphonuclear granulocyte that differentiate from myeloblasts in the bone marrow to comprise approximately 60% of the circulating blood leukocytes (1). The formation of intracellular granules (azurophilic granules, specific granules, gelatinase granules, and secretory vesicles) and the morphologically characteristic segmentation of nuclei occur during the terminal differentiation process into neutrophils (1). Neutrophils are often considered professional bacteria-responsive immune cells: they express bacteria-specific receptors (e.g., formylated peptide receptors or certain toll-like receptors, “TLRs”) and their granules have anti-bacterial or bacteriostatic properties. Currently, their role in viral infection has received very little scientific attention (2).
Neutrophils are present during many types of lung diseases associated with acute respiratory distress syndrome (ARDS) and may contribute to acute lung injury (3–12). The lung has a global inflammatory response to infection regardless of etiology, and this response includes the infiltration of neutrophils and macrophages in response to chemotactic signaling which originates in the lung (3, 5, 12–20). These phagocytic cells leave circulation and hone to sites within the infected airways where they may deploy potent effector functions to control disease (Figure 1) in response to pathogen associated molecular patterns (PAMPs) and inflammatory cytokines and chemokines (21, 22). In the case of viral infections, the type I interferons (IFN) and IFN-stimulated genes (ISGs) signal an appropriate immune response (23–26). Lethal infections may result from insufficient information or incorrect information about the specific cause(s) of infection, thereby signaling inappropriate (incorrect or excessive) immune responses (27). Neutrophils, as first-responders to many forms of airway infection, may be a keystone species in determining viral disease outcome; however, neutrophils are poorly studied with respect to viral infection and specifically to respiratory viral disease.
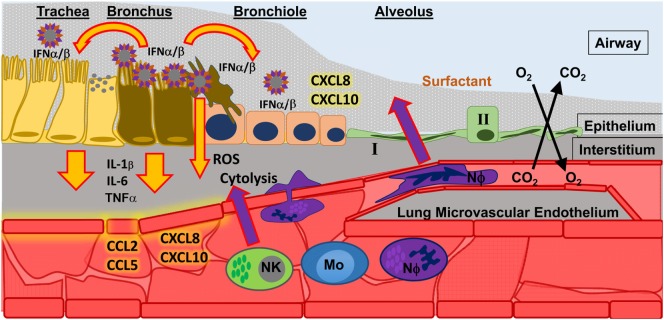
Figure 1.
Influenza A virus (IAV) infection in the upper respiratory tract. Infection of epithelial cells in the bronchus results in the release of type I interferons (IFN α/β) which signal to nearby cells. The result of IFN α/β signaling is the release of pro-inflammatory cytokines (e.g., IL-1β, IL-6, TNFα) that signal to endothelial cells, which help spread inflammatory signals (chemokines, such as CCL2, CCL5, CXCL8, CXCL10) throughout the blood to recruit innate immune cells to the site of infection. Recruited innate immune cells [such as natural killer cells (NK); monocytes (Mo); and neutrophils (NΦ)] must interact with activated endothelium to leave the blood stream and migrate toward the site of infection. There they can perform effector functions to control infection, such as releasing reactive oxygen species (ROS) and directly killing infected cells (cytolysis).
Much research has focused on the role of neutrophils in severe versus mild respiratory disease, as well as their role in bacterial infections. It is, therefore, important to establish a general role of neutrophils in respiratory virus infection to provide a groundwork into more specific questions (e.g., are neutrophils capable of catering to a virus-specific response?). Herein, the evidence for the presence and activities of neutrophils during respiratory viral infection is reviewed. Having established spatio-temporal aspects of the neutrophil response to influenza A virus (IAV), the potential antiviral function(s) of neutrophils during acute virus infection as well as recovery from infection are discussed. The discussion will focus on IAV and neutrophil activities during the course of a “typical” flu infection: activation, migration, and effector functions in situ (Figure 1). IAV are particularly well-suited for the study of neutrophils in viral respiratory disease, since they are well-studied in humans and animal models, and it is well-established that infection with specific viral variants (i.e., genetic point mutations) alter the course of disease from mild to severe (28). More recently, specific IAV viral variants that affect pathogenicity have been linked to alterations of the neutrophil response (29). Thus, a comparison of the neutrophil response between disease phenotypes of a single virus species (Influenza A virus) may elucidate a role for neutrophils in the viral microenvironment. Herein, we review evidence of neutrophil responses during the course of disease in various IAV phenotypes in animal models of infection, as well as comparing these responses to what is known about neutrophil responses during bacterial infection of the airways.
Influenza A Viral Phenotypes
Influenza A virus poses a concern for global public health due to emergence of strains with increased human transmission and/or increased pathology (30–35). In 2009, a novel virus type, influenza A(H1N1)pdm09 IAV, emerged with an increased transmission rate and greater disease, i.e., moderate to severe pathology relative to seasonal human IAV (36–50). Clinical isolates of influenza A(H1N1)pdm09 have relatively little genetic variability yet cause variable clinical outcomes from moderate to severe pathology, including ARDS (39, 41, 47, 48, 51). Therefore, they are well-suited to understand host and viral contributions to IAV pathogenesis. A detailed discussion of the viral replicative cycle is beyond this review, yet excellent reviews are plentiful [e.g., Ref. (52–54)]. IAV is a well-studied model for virus infection in laboratory animals, such as mice and ferrets, and much is known about the contributions of viral and host determinants to severe disease (55–58). Retrospective and experimental infection studies routinely demonstrate common occurrences in the formation of severe IAV [including severe influenza pneumonia (SIP) and ARDS] in humans, ferrets, and mice; these include increased cytokine secretions in the lung, diffuse alveolar damage (“DAD,” bronchointerstitial pneumonia in veterinary pathology), and neutrophilic infiltration (29, 55, 58–66).
In general, IAV infection is an excellent model to investigate the respiratory system’s immune response to viral infection, specifically the pathway leading to severe pneumonia and/or ARDS. Influenza disease is commonly relatively mild and self-limiting, although highly pathogenic forms exist (42, 59, 67–72). The major complication from IAV infection is the formation of SIP which may develop into ARDS (59, 65, 67, 68, 70–73). The reason(s) why infection with IAV may lead to severe viral pneumonia and ARDS is poorly understood, but is thought to involve both host and viral factors. The respective and combined contributions of the host innate immune response and viral factors to the timing and severity of SIP are poorly understood. Neutrophils are present in the respiratory tract during infection with mild seasonal IAV, SIP, and highly pathogenic avian influenza viruses [“HPAI,” which includes avian influenza A (H5N1) virus] (50, 51, 65, 67, 68, 74–76). During SIP and HPAI infection, an increase in the number of neutrophils in the lower respiratory tract (LRT) is correlated with disease severity (50, 51, 65, 67, 68, 76).
Although clinical pathology suggests that a spectrum of disease results from IAV infection, there are at least three disease “phenotypes” caused by infection with IAV, listed by increasing case fatality rate: a mild upper respiratory tract (URT) infection, a SIP which can lead to ARDS, and a LRT infection which can lead to hypercytokinemia. The virological basis for disease phenotype is related to adaptations to mammals—most important are receptor specificity and efficiency of replication—and the major mechanisms have been defined through the use of experimental animal models (30, 32, 33, 57, 77–80). An “ideal” viral infection (i.e., one that is successful for the virus and non-lethal for the host) may be considered a balance between virus replication and an immune response necessary to promote viral shedding, typical of mild seasonal (“epidemic”) IAV. In general, emergent IAV, directly or indirectly from avian enzootic cycles, have increased pathology in humans, the most fatal form of which is a syndrome of complete immune dysregulation (65, 69, 70, 81–84). IAV is genetically highly variable, and mechanisms for increased disease severity are multifactorial, involving host and viral factors.
Uncomplicated Influenza
The majority of yearly, seasonal IAV infections in the world cause a relatively mild, self-limiting URT disease. Influenza disease is characterized by an abrupt onset fever, myalgia, and malaise, with symptoms similar to other URT infections, such as sneezing, coryza, and rhinorrhea (67, 85). Symptoms can last anywhere from 1–5 days and are clinically indistinguishable from other “flu”-like illnesses, including bacterial and viral infections that cause the common cold [e.g., Streptococcus pneumoniae, Haemophilus influenzae, human rhinovirus (HRV) infection, Human respiratory syncytial virus (hRSV) infection, and coronavirus infection] (85–88). Experimental infection of humans with IAV suggests that the virus is mainly restricted to the URT, although sampling the LRT is difficult (67, 68, 87, 89). While fever typically begins 2 days postinfection, virus is shed from the URT in nasal secretions as quickly as 24 h postinfection, allowing efficient transmission prior to symptom onset and continues until 4–5 days postinfection (86, 87, 89) (Figure 2). Rhinorrhea is coincident with neutrophilic rhinitis and shedding of necrotic nasal epithelium (67, 90, 91). Surprisingly, the LRT seems to be involved in uncomplicated IAV infection, although this observation is frequently overlooked or unaddressed in studies (68, 92). In humans, local and systemic concentrations of IL6, CXCL8/IL8, and MCP1/CCL2 correlate with increased disease severity (i.e., symptom severity and increased virus shedding) (87–89, 93) (Figure 2).
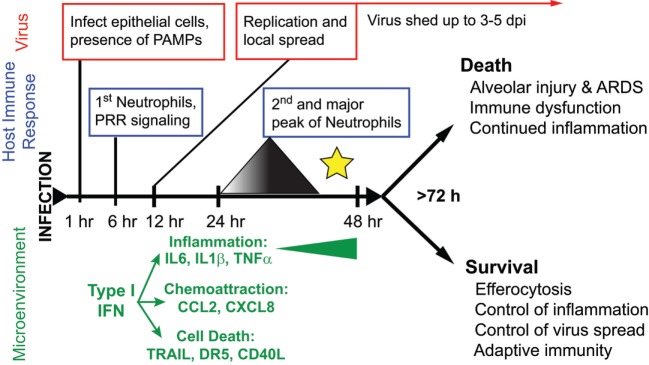
Figure 2.
The course of disease following influenza A virus (IAV) infection. A timeline depicting major events in the viral replication cycle (red), the host immune response (blue), and the effects on the host tissue environment (green) during an IAV infection of the airways. A star marks the critical point for the formation of severe disease versus recovery from infection. This review posits that at this timepoint, coincident with a second wave of increasing neutrophilia and inflammation, the outcome of disease is determined.
Severe Influenza Pneumonia
Influenza A(H1N1)pdm09 virus spread quickly throughout the globe, much like previous pandemic viruses, such as the 1918 H1N1 “Spanish flu” IAV. Humans infected with influenza A(H1N1)pdm09 virus also presented with typical flu-like symptoms (e.g., fever, cough); however, there was an increased number of cases presenting with dyspnea, respiratory distress, and pneumonia (36–38, 40–48, 50, 94, 95). Additionally, retrospective assessments show a proportionately greater number of adolescents and adults with severe disease compared to typical seasonal influenza, and patients with comorbidities, such as obesity and asthma, were at higher risk of severe infection (51, 96–98). In general, the virus causes infection of URT, as well as bronchitis and bronchiolitis, and a high proportion of cases presented with severe disease in the form of viral pneumonia (42, 51, 96). Histopathologic changes in autopsies revealed extensive cytonecrosis, desquamation, and inflammatory infiltration of the bronchus and trachea, mild to severe necrotizing bronchiolitis (42, 50, 51, 99). The primary pathologic finding of SIP was sporadic to DAD with hyaline membrane formation, edema, and occasionally hemorrhage (42, 50, 51, 99). As is typical of influenza infections, some patients experienced bacterial coinfection although this was not in a majority of patients, including those dying from ARDS (42, 48, 50, 51, 93, 99–103). This may distinguish the influenza A(H1N1)pdm09 virus from the 1918 H1N1 IAV, for which bacterial superinfection was determined to cause a majority of the deaths (104, 105), although this may more accurately reflect improved hygiene and standard of care. As discussed below, studies of the reconstructed 1918 H1N1 IAV using animal models suggest that this virus was highly pathogenic irrespective of secondary bacterial pneumonia (62, 106–108).
Across many cohorts of clinical patients, serum concentration of IL6, CCL2, and CXCL8 were significantly elevated in severe cases of influenza A(H1N1)pdm09 viral pneumonia compared to patients with other confirmed illnesses including seasonal IAV, milder forms of influenza A(H1N1)pdm09 virus, bacterial pneumonia, or other viral respiratory infection [hRSV, HRV, human adenovirus (hAdv)] (93, 100–102, 109). These cytokines and chemokines remained elevated over time (up to 6 days following hospital admission) in cases of severe influenza A(H1N1)pdm09 viral pneumonia, whereas they decreased as patients recovered from seasonal and mild influenza A(H1N1)pdm09 virus infection (93, 100). In severe cases of influenza A(H1N1)pdm09 virus infection, decreased type I IFN and ISG production was occasionally noticed compared to adult patients with seasonal IAV infection (93). The influenza A(H1N1)pdm09 viruses have received much scrutiny, and a large dataset of the genetics and pathogenic phenotypes of virus isolates exists in human and animal models. The H1N1 subtype IAV are highly important viruses due to their pandemic potential, as supported by the historical record (62, 68, 110). Some influenza A(H1N1)pdm09 viruses can infect the LRT in humans and in the ferret animal model, which makes them excellent laboratory viruses to investigate the involvement of the LRT in pathogenesis, specifically in the development of severe disease (42, 111–115). Their overall genetic similarity makes for excellent comparison studies between natural clinical isolates, and reverse genetics systems exist to study molecular pathogenicity (41, 95, 113, 116–126).
HPAI and Cytokine Storm
Avian IAV has been recorded sporadically entering the human population over the last 20 years, beginning with an H5N1 subtype virus which emerged in China in 1997 (70, 82, 127, 128). It is likely that this avian influenza A (H5N1) virus and other emergent strains result from contact with infected domestic poultry that are infected with HPAI (82). The disease caused by avian influenza A (H5N1) virus is characterized by DAD, alveolar necrosis, and alveolar hemorrhage [human disease, including pathology, is reviewed in Ref. (83)]. There is evidence of viremia and systemic spread; IAV antigen has been detected in the trachea, bronchi, and alveolar pneumocytes (69, 76), as well as infrequently in the brain and gastric epithelium (59, 76, 83). The innate cellular immune response in the lungs was characterized by an increase in inter-alveolar macrophages/histiocytes (59, 65, 69, 71, 81, 129) and only moderate infiltration of lymphocytes and neutrophils in the few patients that were analyzed postmortem (69, 76). Systemically, patient serum had high concentrations of CXCL10, CCL2, IL6, IL8, and IL10 compared to matched-control patients with seasonal H3 and H1 IAV, and these concentrations were correlated with viral load in throat (59, 65, 69). In lethal cases, the result of infection and immune dysregulation led to multiple organ failure (e.g., kidney tubular inflammation, necrotic lesions in brain, impaired liver function) and abnormal clotting. Reactive histiocytes undergoing hemophagocytosis were frequently found in bone marrow and lungs of patients, which is indicative of diseases involving hypercytokinemia (59, 65, 70, 81, 83, 130).
Other events involving avian IAV transmission to humans are known and are often associated with veterinary or other animal workers; for example, a 2004 case of avian influenza A (H7N7) virus infection in a veterinarian in Europe showed severe fatal pneumonia and DAD (131). It was reported that 1 L of serosanguineous fluid was drained from his chest upon autopsy. In 2013, another avian IAV emerged in Southeast Asia; this time an avian influenza A (H7N9) virus (71, 72, 129, 132). The histopathology was similar to avian influenza A (H5N1) virus: severe pneumonia, DAD, and epithelial necrosis were common features of infection with both viruses (71, 72). Therefore, it seems the typical presentation of human patients infected with either virus includes high levels of CXCL10, CCL2, IL-6, and CXCL8 in the plasma, peripheral blood leukopenia, and lung neutrophilia (65, 69, 81, 129, 130, 132), and this is also similar to experimental infection of laboratory animal models (111, 133). There were slightly more bacterial coinfections in cases of H7N9 compared to H5N1 avian IAV (71, 72, 81). In a direct comparison, serum from patients with infected with avian influenza A (H5N1) virus had higher concentrations of IFNα and IFNγ in the blood and lower levels of IL8, whereas the opposite was true for patients with avian influenza A (H7N9) virus (134). Similarly, CXCL9 and CXCL10 were higher in patients with avian influenza A (H5N1) virus, whereas CCL4 concentrations were higher in patients with avian influenza A (H7N9) virus (134). Infection with either virus resulted in higher blood C-reactive protein (CRP) (129). Variability between patients may account for the apparent discrepancies between specific immune responses. Therefore, experimental infection of laboratory animals removes individual variability and provides a clear picture of general disease progression, with important caveats for their comparison to human disease as discussed in more detail in the next section (58, 135).
Neutrophils in IAV Course of Disease
Neutrophils are increased in the lungs and blood after infection with pathogenic IAV in mice, humans, and ferrets (28, 136, 137). Cell depletion studies have demonstrated that neutrophils are necessary for recovery from severe, but not mild, IAV infection (29, 138, 139). Studies in mice show that neutrophils have effects during both early and late stages of disease (140). As discussed in detail below, initial pathogen sensing through various pathogen recognition receptors (PRRs) stimulates inflammatory signals from resident macrophages to initiate neutrophil chemotaxis to the infected airways (Figure 1). For example, TLR7 recognition of IAV dsRNA- and Myd88-mediated release of TNFα and CCL3 by mononuclear cells is important for neutrophil recruitment to the site of infection (141, 142). Transgenic mice have been used to study the contribution of specific cytokines and chemokines to inflammation following IAV infection, particularly as this relates to “hypercytokinemia,” and are summarized in Tables S1–S4 in Supplementary Material. The signals from the infected lung are propagated systemically by endothelial cells, which recruit and tether neutrophils. The importance of endothelial signaling in the development of severe disease has been shown recently using sphingosine-1 phosphate agonist to prevent severe disease in animal models of both influenza virus and respiratory syncytial virus (143, 144). The complex interactions governing neutrophil extravasation, migration through the interstitium, and crossing the alveolar epithelium are well known in relation to many forms of ALI and the development of ARDS with the exception of conditions surrounding viral infection, although mechanistically they should be quite similar (3–5, 7, 145, 146).
Neutrophil Migration
In the ferret model, the migration of neutrophils to the lungs occurs in two distinct waves: a first wave within hours of challenge, peaking after 24 h then decreasing; and a second wave that increases over time until disease resolution or death (111) (Figure 2). In the ferret model, we have shown that the neutrophils become concentrated at specific foci in the lungs coincident with influenza-positive epithelium and the expression of chemoattractant chemokine genes (22). Neutrophil chemotaxis in humans is thought to be mediated by many factors, such as the chemokine CXCL8, cytokines IL-1 and TNFα, and complement C5a (145, 147, 148). During both mild and severe IAV disease, patients show increased blood CRP and activation of C5a (149), as well as increased secretion of CXCL8, TNFα, and IL-1 in nasal washes, which correlate with disease severity (87, 89, 93, 150–152). In the mouse model of influenza virus infection, chemical reduction of C5a during IAV infection reduced lung neutrophilia (153). Similarly, knockout mice deficient in the inflammasome pathway or mice not expressing cytokines, such as IL-1b and IL-6, have decreased neutrophil activation and migration to the lungs during IAV infection (Table S1 in Supplementary Material) (154–156). Mice do not possess CXCL8, but CXCL1 and CXCL2 have equivalent functions. Neutrophils contribute CXCL2 to the IAV-infected mouse lung to further stimulate neutrophil recruitment (157). More recently, it was shown that removing a CXCL1 repressor (Setdb2) does not increase recruitment of neutrophils to the lungs of mice infected with IAV PR8, rather it reduces the ability to respond to bacterial superinfection (158). In addition, it was shown that another ISG, CXCL10, operates on a unique subset of CXCR3+ neutrophils present during mouse IAV infection in an autocrine manner, increasing chemotaxis, oxidative burst, and enhancing inflammation (157) (Tables S3 and S4 in Supplementary Material). Finally, aryl hydrocarbon receptor is somehow linked to increases in NO and neutrophilia in the lungs of IAV-infected mice independently of known neutrophil chemoattractants or mechanisms of neutrophil extravasation (159–162).
Neutrophil Extracellular PRRs and Phagocytosis
In cell coculture, human neutrophils were seen to interact specifically with IAV-infected cells (163), although the nature of this interaction in the infected lung is unknown. Neutrophils are phagocytic cells, and their methods for sensing extracellular pathogens rely on TLRs (2, 147). Stimulation of neutrophils through cell surface TLRs has been recorded to promote cytokine secretion (CXCL8 and TNFα via NFκB and AP-1), formation of reactive oxygen species (ROS), phagocytosis, granule secretion, neutrophil extracellular trap (NET) formation, and migration (145, 147, 148). Human neutrophils highly express nucleic acid-detecting TLRs, specifically endosomal TLR8 (164, 165), but do not express nor respond to activators of TLR3 or TLR7 (164–166). [Interestingly, TLR3−/− mice have increased neutrophilia and fewer macrophages in the lungs, yet have increased survival after infection with IAV (167, 168).] TLR4 is required for LPS-induced neutrophil migration to the lung (169) and can stimulate immunostimulatory responses via TRIF adaptors (170); however, TLR4-stimulation does not lead to the production of type I IFN in neutrophils (166, 171).
Several innate immune effector proteins with opsonizing functions that are present in airway mucosae are known to interact with both IAV and neutrophils. Surfactant protein D, a lung collectin, is an innate immune defense against a variety of viruses, opsonizing the viruses for phagocytosis by neutrophils, which in turn causes the production of ROS (172–178). Human neutrophil defensins are short basic peptides released from neutrophil granules during inflammation (145, 178, 179). They have been shown to interact with IAV, reducing infectivity, and promote neutrophil phagocytosis and clearance of IAV (178–182). Defensins may also buffer the oxidative burst from neutrophils that follows from phagocytosis of viruses that have been opsonized by surfactant protein D (178–180). One study indicated that neutrophils do not interact with immunoglobulin-bound IAV (183); however, another showed that protective anti-IAV antibody therapy only protected mice in the presence of neutrophils (184). The hypothesis that IAV pathogenicity can be partially explained by infection with viral variants that can evade opsonization by innate immune effectors is attractive and deserves further study (176).
Neutrophil Intracellular PRRs
Neutrophils express sialic acid receptors and may become infected with IAV (74, 185, 186). Neutrophils infected with IAV have increased apoptosis, but infection does not result in the production of virus (186). Infection of human neutrophils with IAV treated at 56°C to denature the viral replicase but not HA suggested that infection alone, but not replication, is sufficient to stimulate the release of CXCL8 and CCL4 in human neutrophils (187). Neutrophils infected with IAV have rapid upregulation (<9 hpi) of type I IFN pathways, including cytoplasmic PRRs, IFNβ, and ISGs (186), which is counter to the long-held dogma that neutrophils were incapable of gene expression. This is may be due to RIG-like receptors (RLRs) sensing of viral dsRNA, as neutrophils transfected with poly(I:C) (a viral RNA mimic) have a similar response (166). Additionally, neutrophils express nod-like receptors but it is unclear how these interact with IAV infection (188, 189). In general, inflammasome and pro-IL1 activation following IAV infection is poorly understood (154, 155, 190, 191). However, studies of IAV infection using caspase-1, IL-1β, or IL-1R transgenic mice show modulation of neutrophil infiltration and pathology and suggest that it is an IAV subtype-dependent effect (154, 191–195) (Tables S1 and S2 in Supplementary Material). Furthermore, it has been demonstrated that the HA of some IAV isolates suppresses neutrophil activation, providing further evidence for IAV subtype-dependent effects on neutrophils (185, 186, 196).
Neutrophil Activation and Degranulation
Activation of TLRs and RLRs trigger degranulation and the expression of surface CD11b (166), which pairs with CD18 to form the “Mac-1” integrin dimer that binds collagen (197). This facilitates migration through tissues, and release of gelatinase or collagenase (MMP-2 and MMP-9) from neutrophils assist in clearing connective tissue from the path. At the site of infection, neutrophils release microbial effectors [reviewed in Ref. (145, 198)]. Neutrophils develop granules sequentially (azurophilic, specific, gelatinase, secretory) and secrete granules in the reverse order (145). Secretory and gelatinase granules are released shortly after endothelial transmigration and contain membrane proteins essential for movement [extracellular matrix (ECM)-binding integrins] and pathogen recognition [immunoglobulin (FcR) and complement receptors] (145). Specific and azurophilic granules contain tissue-destroying enzymes and antimicrobial proteins. For example, neutrophil myeloperoxidase (MPO) may contribute to lung injury during IAV infection (199); however, it may have direct antiviral effects on IAV (200). An investigation found no difference between IAV infection of a wild-type and neutrophil elastase knockout mouse, measuring lung function, chemokine secretion, and neutrophil recruitment (201) (Table S5 in Supplementary Material). The contents of the granules can be secreted to destroy ECM (such as MMP-9) or directed toward phagosomes to destroy engulfed microbes. The production of hypochlorous acid (HOCl) is the main oxidant used in phagosomal killing, and its production is dependent on the generation of ROS by the neutrophils. Interestingly, infection by IAV was related to the inhibition of phagosomal killing of bacteria (196).
Neutrophil degranulation primes neutrophils for ROS generation by mobilizing NADPH oxidase components to the plasma membrane (145) and exocytosis of MPO. IAV infection causes the generation of ROS in neutrophils (202). Oxidative burst is thought to have direct microbial effects; however, the direct effect on IAV has not been published (203). IAV infection benefits from the presence of ROS in the environment (204, 205), yet IAV also suppresses NADPH oxidase activity within infected phagocytes (206, 207). Many have investigated the effects of ROS and NO on lung inflammation during IAV infection and found that reduction of oxidative stress in the form of both ROS and NO alieves IAV-dependent lung injury (206, 208–213) (Table S5 in Supplementary Material). For example, oxidized lipids in the lung environment may trigger TLR4, activating immune cells and contributing to increased lung injury (214–216).
Neutrophil Netosis
Neutrophils undergo a form of programmed cell death called netosis, in which NETs are formed (217). NETs are extracellular strands DNA wrapped in histones and enriched in neutrophil effector proteins (e.g., neutrophil elastase and MPO) (218). NETs have the effect of killing many pathogens, including bacteria (146), fungi (219), protozoans (220), and more recently viruses (221). NETs are becoming the focus of study in autoimmune disease atherosclerosis, since they damage endothelium (222–224). Recently, it was shown that hantavirus stimulates NET production during infection, which leads to the generation of autoantibodies and may provide a mechanism for the hemorrhagic fever caused by Old World hantaviruses (225). NETs are typically found with histones, MPO, and neutrophil elastase, and the effect is to isolate the effects of these molecules directly onto the pathogen surface with a “sticky” NET of nucleic acid. NETs contribute to acute lung injury and alveolar capillary damage during IAV infection (139). Yet, very little is known about the relationship between NETs and viral infection in viral disease pathogenesis (226).
In summary, neutrophils are capable of recognizing viruses via PRRs as either opsonized virions or via endosomal TLR. Although the signaling cascade differs from other phagocytic cells, neutrophils are capable of responding to viral PAMPs with respiratory burst, degranulation of proteases and cytokines, and/or netosis. It is not clear if these responses are effective against influenza virus; in fact, evidence exists that suggest influenza viruses may take advantage of the inflammatory environment. More importantly, it is not clear if there are differences in response to different influenza virus subtypes or strains. As is true for many respiratory etiologies, neutrophil responses must be balanced during influenza virus infection to adequately control of inflammation while promoting pro-immune responses. The timing of neutrophils during disease progression correlates with a key point in divergent disease outcomes (Figure 2), and neutrophils may act both globably and locally at foci of infection (22, 111). Thus, neutrophils are focused in the airways at critical timepoints following infection and therefore balancing their potent inflammatory effector functions may determine disease outcome.
Comparisons of Neutrophils in Viral Respiratory Diseases
As discussed above, there appears to be a correlation between the timing and location of IAV infection and the action of neutrophils, but evidence directly linking these phenomena together remains overall circumstantial. In contrast, during bacterial pneumonia there is direct evidence of the importance of neutrophils in disease: bacterial PAMPs directly upregulate neutrophil activating and chemoattractant chemokines, bacteria have defined anti-neutrophil functions, and some bacteria, e.g., Mycobacterium tuberculosis, rely on neutrophils to establish their granulomatous niche (6, 227, 228). Similarly, we suggest that viruses may interact with respiratory cells to create a viral microenvironmental niche. To further substantiate a link between neutrophils and virus infection, evidence for the role of neutrophils in selected viral respiratory diseases is summarized in Tables 1 and 2. Discussion below focuses on common patterns of neutrophil responses in severe and mild forms of respiratory viral infection.
Table 1.
(−) Sense RNA respiratory viruses that cause increased neutrophil infiltration during infection.
| Virus family | Virus type | Host/model | Primary airway target cell | Pathology | Neutrophil abundance | Reference |
|---|---|---|---|---|---|---|
| Orthomyxoviridae | Influenza A virus | Hu, NHP, Mo, Fe, Sw | Epithelial cells [upper respiratory tract (URT), lower respiratory tract (LRT)] | Mild: necrotic rhinitis and tracheitis; moderate: necrotic bronchiolitis and alveolitis; severe: diffuse alveolar damage and hypercytokinemia | (+++) | (229–231) |
| Paramyxoviridae | Human respiratory syncytial virus | Hu | Epithelial cells (URT, LRT) | Severe: necrotic bronchiolitis and alveolitis, obstructed bronchioles, and giant cell formation | (+++) | (232, 233) |
| Paramyxoviridae | Human metapneumovirus | Hu | Epithelium | Mild: airway inflammation and epithelial degeneration | (+) | (234, 235) |
| Paramyxoviridae | Hendrah/Nipah virus | Hu | Not defined | Severe: interstitial pneumonia, but primarily vasotropic or neurotropic | (++) | (236) |
| Paramyxoviridae | Measles virus | Hu, Mo | Resident myeloid cells of the lung | Severe: bronchiolitis obliterans | (++) | (237) |
| Paramyxoviridae | Human parainfluenza virus | Hu | Ciliated epithelium | Bronchiolitis and alveolitis | Not defined | (238) |
| Bunyaviridae | New World hantavirus | Hu | Lung microvascular endothelium | Hantavirus pulmonary syndrome, endothelial infilammation, and focal antigen-positive sites in lung | (+) | (239) |
Hu, human; NHP, non-human primate; Mo, mouse; Fe; ferret; Sw, swine.
Table 2.
(+) Sense RNA and DNA respiratory viruses that cause increased neutrophil infiltration during infection.
| Virus family | Virus type | Host/model | Primary cell target | Pathology | Neutrophil abundance | Reference |
|---|---|---|---|---|---|---|
| Picornaviridae (+RNA) | Human rhinovirus | Hu | Epithelium | Mild to moderate: neutrophilic rhinitis; severe: acute LRT, bronchiolitis, and alveolitis | (+++) | (231) |
| Adenoviridae (dsDNA) | Human adenovirus (HAdv3, HAdv7) | Hu | Epithelium | Bronchitis and alveolitis | (+) | (240) |
| Coronaviridae (+RNA) | Human coronavirus (NL-63 or OC43) | Hu | Epithelium | Mild | (+/−) | (241) |
| Coronaviridae (+RNA) | Severe acute respiratory syndrome coronavirus | Hu, Fe | Epithelium | Alveolitis, acute respiratory distress syndrome (ARDS), hypercytokinemia | (++) | (10) |
| Coronaviridae (+RNA) | Middle east respiratory syndrome coronavirus | Hu, Fe | Epithelium | Alveolitis, ARDS, hypercytokinemia | (++) | (11) |
Hu, humans; Fe, ferrets.
Severe Viral Respiratory Disease
For both viral and bacterial etiologies, the most severe clinical complications result from infection of the LRT. Infection of the LRT by viruses, such as human parainfluenza viruses, influenza A(H1N1)pdm09, HPAI, New World hantavirus infections (causing hantavirus pulmonary syndrome), Severe acute respiratory syndrome-related coronavirus (SARS-Cov), and Middle East respiratory syndrome coronavirus (MERS-CoV), are all associated with neutrophilic infiltration at sites of infection to various degrees and may develop into ARDS (9–11, 19, 60, 131, 242–245). Clinically defined, ARDS has three phases; and most patents die within the first phase, the acute or “exudative” phase [reviewed in Ref. (12–14)]. This phase is characterized by an increased immune response with high production of pro-inflammatory cytokines and chemokines, increased neutrophil infiltration and accumulation in the alveoli, and disruption of the alveolar epithelial–capillary barrier, which leads to increased vascular permeability and edema (13). The distinctive role for lung neutrophil infiltration in viral infection is summarized as follows: some, but not all, viruses that infect the LRT result in clinically defined ARDS, and lung neutrophil infiltration is associated with viruses that do and do not lead to ARDS (3, 5, 9, 10, 60, 131, 199, 242–244, 246, 247). In ARDS, there are data that directly support the role of neutrophils as both beneficial and detrimental (3, 13, 14). Perhaps, there are general factors (host or virus) that lead to a common antiviral response of neutrophils.
Mild Viral Respiratory Disease
In contrast to emergent highly pathogenic respiratory viruses, notable “mild” human respiratory viruses also involve increased neutrophils at the site of infection (e.g., hRSV). As expected, infection with these viruses is typically associated with the increase of neutrophil chemoattractant chemokines. For example, infection with HRV is a well-studied virus for which there are several studies on the link between neutrophils and disease (229–231, 248) (Table 2). HRV virions enter nasal epithelial cells via endocytosis, yet, unlike influenza, infection does not cause major damage to the nasal epithelium (249–251). Neutrophilic rhinitis, increased vascular permeability, and mucus hypersecretion are the key pathological features of HRV infection (229–231, 249), and infected epithelium seems to be the source of large amounts of neutrophil chemotactic molecules, particularly CXCL8 and kinins (229, 248, 252, 253). Interestingly, in vitro studies have shown that viral recognition of HRV shares features with hRSV, but is somewhat different than with IAV (254–256). It has been established that there are virus-specific and cell-specific differences in sensing RNA viruses via primarily TLR- and/or RLR-pathways (and even in a preference for RIG-I versus MDA5), yet these pathways may have similar general endpoints, such as chemokine and cytokine signaling (255, 257–259).
Finally, a key question is whether virus-induced cytopathy drives neutrophilia or whether it is the result of host response to viral infection. Infections with influenza A(H1N1)pdm09 virus, HPAI, SARS-CoV, and MERS-CoV are thought to cause acute lung injury which results in ARDS; characterized by excessive damage to the alveolar epithelium and involving the infiltration of neutrophils (67, 68) (Tables 1 and 2). However, less pathogenic strains such as HRV infections do not cause significant damage to the respiratory mucosa, yet neutrophils are present (249, 250). Conversely, viruses that cause moderate, focal cytopathy in the lungs, for example seasonal IAV and hRSV, are known to cause neutrophilic infiltration (250, 252, 253). Therefore, neutrophils are not necessarily associated with direct cytopathic effect nor are they exclusively associated with severe disease.
Bacterial Respiratory Disease
Despite many years of searching, there is no single reliable biomarker to indicate a bacterial versus viral infection (although CRP is a good candidate). This is surprising, given the otherwise significant fundamental differences between the biology of these two types of pathogens; differences which are reflected in general immune responses and begin with pathogen detection. Signaling through TLR on the plasma membrane versus endosomal or cytoplasmic PRRs is controlled by complex intracellular adaptor proteins [reviewed in Ref. (260)]. For example, the complexities of signaling allow the characteristically bacteria-specific TLR4 to signal the upregulation of immunostimulatory type I IFN characteristic of a virus infection (170). As evidenced above, many viral infections associated with neutrophil infiltration have RNA genomes. Host cells detect RNA viruses primarily through RLR as well as TLR, whereas bacteria rely on a different group of PRRs to detect extracellular PAMPs (255, 261–263). Interestingly, the lung microenvironment to hRSV has been shown to be different from IAV, specifically in the presence of IL-4 (264, 265). It is thought that this is driven by the presence of alternatively activated macrophages during RSV infection (266). It is not known if this directs differences in neutrophil chemoattraction, yet IL-4 is known to drive a Th2 (“bacterial” or antibody-biased) immune response (267). In sum, surprisingly little is clinically different between the innate immune responses to viral versus bacterial infection; however, perhaps comparative studies that focus on neutrophils can uncover virus-specific responses.
Neutrophils in the Viral Microenvironment
In general, immune activation pathways that involve the activation of NFκB lead to the secretion of neutrophil chemotactic chemokines [reviewed in Ref. (268)]. This is heavily driven by PAMP recognition and activation pathways, and during a viral infection type I IFNs and ISGs are the unique elements in the virus-inflamed lung environment [reviewed in Ref. (24–26, 269, 270)]. This single difference between viral and bacterial infections could have drastic effects on the actions of neutrophils once in the lung. Moreover, without this information (e.g., pathogenic viruses that suppress type I IFN) neutrophils may respond to inflammation in an inefficient way, potentially with pathologic effects (23, 186, 256, 271–277). Neutrophils are known to respond to IAV and type I IFN by upregulation of ISGs (186). In systemic lupus erythematosus, neutrophils may be a large contributor of type I IFN (278, 279). During increased inflammation, left-shifted or immature neutrophils emerge from the bone marrow—a classic sign for sepsis, but also known to be present during some severe viral respiratory diseases (e.g., HPS) (239). It is known that immature neutrophils do neither express IFNα/β receptor nor many other cytokine receptors (273). It is unknown what affect this would have during increased inflammation during pathogenic influenza infection, although their role in other inflammatory conditions suggests it may affect their functions (271, 273, 280, 281).
Resolving Inflammation
All forms of respiratory infection require resolution of the infection and inflammation. IFNs are essential components of initiating sterilizing immunity to virus infection via the adaptive immune system (i.e., resolution of infection), at which point the resolution of inflammation can effectively proceed (25, 26, 282). Although the mechanisms are poorly understood, through their direct antiviral actions and indirect actions on the lung microenvironment (e.g., efferocytosis of apoptotic neutrophils by macrophages), neutrophils have the ability to influence outcomes toward successful resolution as well as toward the formation of ARDS (3, 5, 12, 13, 21, 74, 166, 184, 198, 199, 283–285). Thus, there is evidence that the role of neutrophils in viral infections of the respiratory system is not limited to inflammation, but likely includes recovery from infection and the initiation of adaptive immunity (74, 138, 284). Apart from initiating adaptive immunity, resolution of inflammation may be partially regulated by secretion of IL-1RA and chemokine-destroying factors by recruited macrophages (286, 287). Additionally, it has been proposed that efferocytosis of apoptotic neutrophils is a key step in resolution of inflammation (288–290), and occurs in the lung during bacterial pneumonia (291). It is unclear if this happens during IAV infection or infection with other respiratory viruses.
Prolonging Inflammation
Factors that prolong the life span of neutrophils in the lungs increase the probability that they may contribute to immunopathology. IL-6 and G-CSF are immune mediators present in the lung during infection and are known to prolong survival of neutrophils in mouse lungs following IAV infection (292). Both neutrophils and macrophages are known to phagocytose apoptotic epithelial cells in mouse lungs during IAV infection (283). The cells may be recruited via chemokines or damage receptors. For example, necrotic IAV-infected epithelial cells are a source of CXCL8 (293), which attract neutrophils to dying cells. In addition, neutrophils can detect DAMPs such as S100A9 (294). It has been shown that extracellular S100A9 is abundant during IAV infection in mice (295). Antibody-mediated neutralization of S100A9 decreased lung inflammation in mice and improved disease outcome (295). Apart from potential tissue-destroying effects of neutrophil proteases, the presence of NETs may induce even more inflammation in the lungs (21, 139). Thus, there are limited data supporting directly malevolent actions of neutrophils (Table S5 in Supplementary Material), yet factors that increase their presence and prolong their survival in the lung are correlated with increased disease severity.
Concluding Remarks
There are substantial data that suggest neutrophils are a part of a viral response to infection. Neutrophils are among the first responders to IAV infection in the lung, and they remain in great numbers throughout the development of ARDS. Although neutrophils are an important component of the general response to infection in the respiratory system, as is discussed herein, neutrophils are capable of recognizing viruses (via viral PAMPs), responding to viruses with specific effector functions, and may be instrumental in determining disease outcome. Evidence exists to support the hypothesis that neutrophils respond specifically to the focal nature of viral infection, and they act to influence this microenvironment via their virus-specific effector functions. Factors that influence successful recovery from respiratory viral infection (versus lethal outcome) are complex and both host- and virus-specific. However, a better understanding of the role neutrophils, previously underappreciated with respect to viral infections, will reveal important information about disease outcome. Many questions remain before it is determined the part neutrophils play in mild and severe disease.
Author Contributions
Both authors contributed to the formulation of this scientific research topic. JC wrote the manuscript as part of his Ph.D. dissertation under the mentorship of CJ (296).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors are grateful for the helpful discussions provided by Silvia M. Uriarte, Ph.D. in pursuit of this topic of research.
Funding
Financial support was provided in part by the Commonwealth of Kentucky as a Clinical and Translational Science Pilot Project Program at the University of Louisville to CJ. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplementary Material
The Supplementary Material for this article can be found online at http://journal.frontiersin.org/article/10.3389/fimmu.2017.00550/full#supplementary-material.
References
- 1.Cowland JB, Borregaard N. Granulopoiesis and granules of human neutrophils. Immunol Rev (2016) 273:11–28. 10.1111/imr.12440 [DOI] [PubMed] [Google Scholar]
- 2.Galani IE, Andreakos E. Neutrophils in viral infections: current concepts and caveats. J Leukoc Biol (2015) 98:557–64. 10.1189/jlb.4VMR1114-555R [DOI] [PubMed] [Google Scholar]
- 3.Abraham E. Neutrophils and acute lung injury. Crit Care Med (2003) 31:S195–9. 10.1097/01.CCM.0000057843.47705.E8 [DOI] [PubMed] [Google Scholar]
- 4.Burns AR, Smith CW, Walker DC. Unique structural features that influence neutrophil emigration into the lung. Physiol Rev (2003) 83:309–36. 10.1152/physrev.00023.2002 [DOI] [PubMed] [Google Scholar]
- 5.Grommes J, Soehnlein O. Contribution of neutrophils to acute lung injury. Mol Med (2011) 17:293–307. 10.2119/molmed.2010.00138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lowe DM, Redford PS, Wilkinson RJ, O’Garra A, Martineau AR. Neutrophils in tuberculosis: friend or foe? Trends Immunol (2012) 33:14–25. 10.1016/j.it.2011.10.003 [DOI] [PubMed] [Google Scholar]
- 7.McDonald B, Kubes P. Cellular and molecular choreography of neutrophil recruitment to sites of sterile inflammation. J Mol Med (Berl) (2011) 89:1079–88. 10.1007/s00109-011-0784-9 [DOI] [PubMed] [Google Scholar]
- 8.Smith PK, Wang SZ, Dowling KD, Forsyth KD. Leucocyte populations in respiratory syncytial virus-induced bronchiolitis. J Paediatr Child Health (2001) 37:146–51. 10.1046/j.1440-1754.2001.00618.x [DOI] [PubMed] [Google Scholar]
- 9.Nicholls JM, Poon LL, Lee KC, Ng WF, Lai ST, Leung CY, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet (2003) 361:1773–8. 10.1016/S0140-6736(03)13413-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tse GM, To KF, Chan PK, Lo AW, Ng KC, Wu A, et al. Pulmonary pathological features in coronavirus associated severe acute respiratory syndrome (SARS). J Clin Pathol (2004) 57:260–5. 10.1136/jcp.2003.013276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.van den Brand JM, Smits SL, Haagmans BL. Pathogenesis of Middle East respiratory syndrome coronavirus. J Pathol (2015) 235:175–84. 10.1002/path.4458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Matthay MA, Ware LB, Zimmerman GA. The acute respiratory distress syndrome. J Clin Invest (2012) 122:2731–40. 10.1172/JCI60331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med (2000) 342:1334–49. 10.1056/NEJM200005043421806 [DOI] [PubMed] [Google Scholar]
- 14.Abraham E, Matthay MA, Dinarello CA, Vincent JL, Cohen J, Opal SM, et al. Consensus conference definitions for sepsis, septic shock, acute lung injury, and acute respiratory distress syndrome: time for a reevaluation. Crit Care Med (2000) 28:232–5. 10.1097/00003246-200001000-00039 [DOI] [PubMed] [Google Scholar]
- 15.Aherne W, Bird T, Court SD, Gardner PS, McQuillin J. Pathological changes in virus infections of the lower respiratory tract in children. J Clin Pathol (1970) 23:7–18. 10.1136/jcp.23.1.7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Craig A, Mai J, Cai S, Jeyaseelan S. Neutrophil recruitment to the lungs during bacterial pneumonia. Infect Immun (2009) 77:568–75. 10.1128/IAI.00832-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herold S, Mayer K, Lohmeyer J. Acute lung injury: how macrophages orchestrate resolution of inflammation and tissue repair. Front Immunol (2011) 2:65. 10.3389/fimmu.2011.00065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lien DC, Wagner WW, Jr, Capen RL, Haslett C, Hanson WL, Hofmeister SE, et al. Physiological neutrophil sequestration in the lung: visual evidence for localization in capillaries. J Appl Physiol (1985) (1987) 62:1236–43. [DOI] [PubMed] [Google Scholar]
- 19.Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, et al. Incidence and outcomes of acute lung injury. N Engl J Med (2005) 353:1685–93. 10.1056/NEJMoa050333 [DOI] [PubMed] [Google Scholar]
- 20.Reutershan J, Basit A, Galkina EV, Ley K. Sequential recruitment of neutrophils into lung and bronchoalveolar lavage fluid in LPS-induced acute lung injury. Am J Physiol Lung Cell Mol Physiol (2005) 289:L807–15. 10.1152/ajplung.00477.2004 [DOI] [PubMed] [Google Scholar]
- 21.Jenne CN, Wong CH, Zemp FJ, McDonald B, Rahman MM, Forsyth PA, et al. Neutrophils recruited to sites of infection protect from virus challenge by releasing neutrophil extracellular traps. Cell Host Microbe (2013) 13:169–80. 10.1016/j.chom.2013.01.005 [DOI] [PubMed] [Google Scholar]
- 22.Camp JV, Bagci U, Chu YK, Squier B, Fraig M, Uriarte SM, et al. Lower respiratory tract infection of the ferret by 2009 H1N1 pandemic influenza A virus triggers biphasic, systemic, and local recruitment of neutrophils. J Virol (2015) 89:8733–48. 10.1128/JVI.00817-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Davidson S, Crotta S, McCabe TM, Wack A. Pathogenic potential of interferon alphabeta in acute influenza infection. Nat Commun (2014) 5:3864. 10.1038/ncomms4864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garcia-Sastre A, Biron CA. Type 1 interferons and the virus-host relationship: a lesson in detente. Science (2006) 312:879–82. 10.1126/science.1125676 [DOI] [PubMed] [Google Scholar]
- 25.Le Bon A, Tough DF. Links between innate and adaptive immunity via type I interferon. Curr Opin Immunol (2002) 14:432–6. 10.1016/S0952-7915(02)00354-0 [DOI] [PubMed] [Google Scholar]
- 26.Stetson DB, Medzhitov R. Type I interferons in host defense. Immunity (2006) 25:373–81. 10.1016/j.immuni.2006.08.007 [DOI] [PubMed] [Google Scholar]
- 27.Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev (2012) 76:16–32. 10.1128/MMBR.05015-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Watanabe T, Tisoncik-Go J, Tchitchek N, Watanabe S, Benecke AG, Katze MG, et al. 1918 Influenza virus hemagglutinin (HA) and the viral RNA polymerase complex enhance viral pathogenicity, but only HA induces aberrant host responses in mice. J Virol (2013) 87:5239–54. 10.1128/JVI.02753-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vidy A, Maisonnasse P, Da Costa B, Delmas B, Chevalier C, Le Goffic R. The influenza virus protein PB1-F2 increases viral pathogenesis through neutrophil recruitment and NK cells inhibition. PLoS One (2016) 11:e0165361. 10.1371/journal.pone.0165361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev (1992) 56:152–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Webster RG. Predictions for future human influenza pandemics. J Infect Dis (1997) 176(Suppl 1):S14–9. 10.1086/514168 [DOI] [PubMed] [Google Scholar]
- 32.Webster RG. Influenza virus: transmission between species and relevance to emergence of the next human pandemic. Arch Virol Suppl (1997) 13:105–13. [DOI] [PubMed] [Google Scholar]
- 33.Webster RG, Shortridge KF, Kawaoka Y. Influenza: interspecies transmission and emergence of new pandemics. FEMS Immunol Med Microbiol (1997) 18:275–9. 10.1111/j.1574-695X.1997.tb01056.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Webster RG. Influenza: an emerging disease. Emerg Infect Dis (1998) 4:436–41. 10.3201/eid0403.980325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cox NJ, Subbarao K. Global epidemiology of influenza: past and present. Annu Rev Med (2000) 51:407–21. 10.1146/annurev.med.51.1.407 [DOI] [PubMed] [Google Scholar]
- 36.CDC. Outbreak of swine-origin influenza A (H1N1) virus infection – Mexico, March-April 2009. MMWR Morb Mortal Wkly Rep (2009) 58:467–70. [PubMed] [Google Scholar]
- 37.Cao B, Li XW, Mao Y, Wang J, Lu HZ, Chen YS, et al. Clinical features of the initial cases of 2009 pandemic influenza A (H1N1) virus infection in China. N Engl J Med (2009) 361:2507–17. 10.1056/NEJMoa0906612 [DOI] [PubMed] [Google Scholar]
- 38.Donaldson LJ, Rutter PD, Ellis BM, Greaves FE, Mytton OT, Pebody RG, et al. Mortality from pandemic A/H1N1 2009 influenza in England: public health surveillance study. BMJ (2009) 339:b5213. 10.1136/bmj.b5213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Garten RJ, Davis CT, Russell CA, Shu B, Lindstrom S, Balish A, et al. Antigenic and genetic characteristics of swine-origin 2009 A(H1N1) influenza viruses circulating in humans. Science (2009) 325:197–201. 10.1126/science.1176225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jain S, Kamimoto L, Bramley AM, Schmitz AM, Benoit SR, Louie J, et al. Hospitalized patients with 2009 H1N1 influenza in the United States, April-June 2009. N Engl J Med (2009) 361:1935–44. 10.1056/NEJMoa0906695 [DOI] [PubMed] [Google Scholar]
- 41.Smith GJ, Vijaykrishna D, Bahl J, Lycett SJ, Worobey M, Pybus OG, et al. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature (2009) 459:1122–5. 10.1038/nature08182 [DOI] [PubMed] [Google Scholar]
- 42.Gill JR, Sheng ZM, Ely SF, Guinee DG, Beasley MB, Suh J, et al. Pulmonary pathologic findings of fatal 2009 pandemic influenza A/H1N1 viral infections. Arch Pathol Lab Med (2010) 134:235–43. 10.1043/1543-2165-134.2.235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Libster R, Bugna J, Coviello S, Hijano DR, Dunaiewsky M, Reynoso N, et al. Pediatric hospitalizations associated with 2009 pandemic influenza A (H1N1) in Argentina. N Engl J Med (2010) 362:45–55. 10.1056/NEJMoa0907673 [DOI] [PubMed] [Google Scholar]
- 44.Agarwal PP, Cinti S, Kazerooni EA. Chest radiographic and CT findings in novel swine-origin influenza A (H1N1) virus (S-OIV) infection. AJR Am J Roentgenol (2009) 193:1488–93. 10.2214/AJR.09.3599 [DOI] [PubMed] [Google Scholar]
- 45.Mollura DJ, Asnis DS, Crupi RS, Conetta R, Feigin DS, Bray M, et al. Imaging findings in a fatal case of pandemic swine-origin influenza A (H1N1). AJR Am J Roentgenol (2009) 193:1500–3. 10.2214/AJR.09.3365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Neumann G, Noda T, Kawaoka Y. Emergence and pandemic potential of swine-origin H1N1 influenza virus. Nature (2009) 459:931–9. 10.1038/nature08157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Peiris JS, Poon LL, Guan Y. Emergence of a novel swine-origin influenza A virus (S-OIV) H1N1 virus in humans. J Clin Virol (2009) 45:169–73. 10.1016/j.jcv.2009.06.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Perez-Padilla R, de la Rosa-Zamboni D, Ponce de Leon S, Hernandez M, Quinones-Falconi F, Bautista E, et al. Pneumonia and respiratory failure from swine-origin influenza A (H1N1) in Mexico. N Engl J Med (2009) 361:680–9. 10.1056/NEJMoa0904252 [DOI] [PubMed] [Google Scholar]
- 49.Woo PC, Tung ET, Chan KH, Lau CC, Lau SK, Yuen KY. Cytokine profiles induced by the novel swine-origin influenza A/H1N1 virus: implications for treatment strategies. J Infect Dis (2010) 201:346–53. 10.1086/649785 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Calore EE, Uip DE, Perez NM. Pathology of the swine-origin influenza A (H1N1) flu. Pathol Res Pract (2011) 207:86–90. 10.1016/j.prp.2010.11.003 [DOI] [PubMed] [Google Scholar]
- 51.Shieh WJ, Blau DM, Denison AM, Deleon-Carnes M, Adem P, Bhatnagar J, et al. 2009 pandemic influenza A (H1N1): pathology and pathogenesis of 100 fatal cases in the United States. Am J Pathol (2010) 177:166–75. 10.2353/ajpath.2010.100115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fukuyama S, Kawaoka Y. The pathogenesis of influenza virus infections: the contributions of virus and host factors. Curr Opin Immunol (2011) 23:481–6. 10.1016/j.coi.2011.07.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Medina RA, Garcia-Sastre A. Influenza A viruses: new research developments. Nat Rev Microbiol (2011) 9:590–603. 10.1038/nrmicro2613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tscherne DM, Garcia-Sastre A. Virulence determinants of pandemic influenza viruses. J Clin Invest (2011) 121:6–13. 10.1172/JCI44947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Belser JA, Katz JM, Tumpey TM. The ferret as a model organism to study influenza A virus infection. Dis Model Mech (2011) 4(5):575–9. 10.1242/dmm.007823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Smith H, Sweet C. Lessons for human influenza from pathogenicity studies with ferrets. Rev Infect Dis (1988) 10:56–75. 10.1093/clinids/10.1.56 [DOI] [PubMed] [Google Scholar]
- 57.Barnard DL. Animal models for the study of influenza pathogenesis and therapy. Antiviral Res (2009) 82:A110–22. 10.1016/j.antiviral.2008.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Belser JA, Szretter KJ, Katz JM, Tumpey TM. Use of animal models to understand the pandemic potential of highly pathogenic avian influenza viruses. Adv Virus Res (2009) 73:55–97. 10.1016/S0065-3527(09)73002-7 [DOI] [PubMed] [Google Scholar]
- 59.de Jong MD, Simmons CP, Thanh TT, Hien VM, Smith GJ, Chau TN, et al. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med (2006) 12:1203–7. 10.1038/nm1477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van den Brand JM, Haagmans BL, van Riel D, Osterhaus AD, Kuiken T. The pathology and pathogenesis of experimental severe acute respiratory syndrome and influenza in animal models. J Comp Pathol (2014) 151:83–112. 10.1016/j.jcpa.2014.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van Riel D, Munster VJ, de Wit E, Rimmelzwaan GF, Fouchier RA, Osterhaus AD, et al. Human and avian influenza viruses target different cells in the lower respiratory tract of humans and other mammals. Am J Pathol (2007) 171:1215–23. 10.2353/ajpath.2007.070248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Watanabe T, Kawaoka Y. Pathogenesis of the 1918 pandemic influenza virus. PLoS Pathog (2011) 7:e1001218. 10.1371/journal.ppat.1001218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cameron CM, Cameron MJ, Bermejo-Martin JF, Ran L, Xu L, Turner PV, et al. Gene expression analysis of host innate immune responses during lethal H5N1 infection in ferrets. J Virol (2008) 82:11308–17. 10.1128/JVI.00691-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lu X, Tumpey TM, Morken T, Zaki SR, Cox NJ, Katz JM. A mouse model for the evaluation of pathogenesis and immunity to influenza A (H5N1) viruses isolated from humans. J Virol (1999) 73:5903–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.To KF, Chan PK, Chan KF, Lee WK, Lam WY, Wong KF, et al. Pathology of fatal human infection associated with avian influenza A H5N1 virus. J Med Virol (2001) 63:242–6. [DOI] [PubMed] [Google Scholar]
- 66.Zitzow LA, Rowe T, Morken T, Shieh WJ, Zaki S, Katz JM. Pathogenesis of avian influenza A (H5N1) viruses in ferrets. J Virol (2002) 76:4420–9. 10.1128/JVI.76.9.4420-4429.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kuiken T, Taubenberger JK. Pathology of human influenza revisited. Vaccine (2008) 26(Suppl 4):D59–66. 10.1016/j.vaccine.2008.07.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Taubenberger JK, Morens DM. The pathology of influenza virus infections. Annu Rev Pathol (2008) 3:499–522. 10.1146/annurev.pathmechdis.3.121806.154316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Peiris JS, Yu WC, Leung CW, Cheung CY, Ng WF, Nicholls JM, et al. Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet (2004) 363:617–9. 10.1016/S0140-6736(04)15595-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yuen KY, Chan PK, Peiris M, Tsang DN, Que TL, Shortridge KF, et al. Clinical features and rapid viral diagnosis of human disease associated with avian influenza A H5N1 virus. Lancet (1998) 351:467–71. 10.1016/S0140-6736(98)01182-9 [DOI] [PubMed] [Google Scholar]
- 71.Gao HN, Lu HZ, Cao B, Du B, Shang H, Gan JH, et al. Clinical findings in 111 cases of influenza A (H7N9) virus infection. N Engl J Med (2013) 368:2277–85. 10.1056/NEJMoa1305584 [DOI] [PubMed] [Google Scholar]
- 72.Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med (2013) 368:1888–97. 10.1056/NEJMoa1304459 [DOI] [PubMed] [Google Scholar]
- 73.Rello J, Pop-Vicas A. Clinical review: primary influenza viral pneumonia. Crit Care (2009) 13:235. 10.1186/cc8183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hufford MM, Richardson G, Zhou H, Manicassamy B, Garcia-Sastre A, Enelow RI, et al. Influenza-infected neutrophils within the infected lungs act as antigen presenting cells for anti-viral CD8(+) T cells. PLoS One (2012) 7:e46581. 10.1371/journal.pone.0046581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sprenger H, Meyer RG, Kaufmann A, Bussfeld D, Rischkowsky E, Gemsa D. Selective induction of monocyte and not neutrophil-attracting chemokines after influenza A virus infection. J Exp Med (1996) 184:1191–6. 10.1084/jem.184.3.1191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gu J, Xie Z, Gao Z, Liu J, Korteweg C, Ye J, et al. H5N1 infection of the respiratory tract and beyond: a molecular pathology study. Lancet (2007) 370:1137–45. 10.1016/S0140-6736(07)61515-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chandrasekaran A, Srinivasan A, Raman R, Viswanathan K, Raguram S, Tumpey TM, et al. Glycan topology determines human adaptation of avian H5N1 virus hemagglutinin. Nat Biotechnol (2008) 26:107–13. 10.1038/nbt1375 [DOI] [PubMed] [Google Scholar]
- 78.Imai M, Watanabe T, Hatta M, Das SC, Ozawa M, Shinya K, et al. Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in ferrets. Nature (2012) 486:420–8. 10.1038/nature10831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.de Graaf M, Fouchier RA. Role of receptor binding specificity in influenza A virus transmission and pathogenesis. EMBO J (2014) 33:823–41. 10.1002/embj.201387442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Herfst S, Schrauwen EJ, Linster M, Chutinimitkul S, de Wit E, Munster VJ, et al. Airborne transmission of influenza A/H5N1 virus between ferrets. Science (2012) 336:1534–41. 10.1126/science.1213362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chan PK. Outbreak of avian influenza A(H5N1) virus infection in Hong Kong in 1997. Clin Infect Dis (2002) 34(Suppl 2):S58–64. 10.1086/338820 [DOI] [PubMed] [Google Scholar]
- 82.Claas EC, Osterhaus AD, van Beek R, De Jong JC, Rimmelzwaan GF, Senne DA, et al. Human influenza A H5N1 virus related to a highly pathogenic avian influenza virus. Lancet (1998) 351:472–7. 10.1016/S0140-6736(97)11212-0 [DOI] [PubMed] [Google Scholar]
- 83.Korteweg C, Gu J. Pathology, molecular biology, and pathogenesis of avian influenza A (H5N1) infection in humans. Am J Pathol (2008) 172:1155–70. 10.2353/ajpath.2008.070791 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Nicholls JM, Chan MC, Chan WY, Wong HK, Cheung CY, Kwong DL, et al. Tropism of avian influenza A (H5N1) in the upper and lower respiratory tract. Nat Med (2007) 13:147–9. 10.1038/nm1529 [DOI] [PubMed] [Google Scholar]
- 85.Hilleman MR. Realities and enigmas of human viral influenza: pathogenesis, epidemiology and control. Vaccine (2002) 20:3068–87. 10.1016/S0264-410X(02)00254-2 [DOI] [PubMed] [Google Scholar]
- 86.Carrat F, Vergu E, Ferguson NM, Lemaitre M, Cauchemez S, Leach S, et al. Time lines of infection and disease in human influenza: a review of volunteer challenge studies. Am J Epidemiol (2008) 167:775–85. 10.1093/aje/kwm375 [DOI] [PubMed] [Google Scholar]
- 87.Hayden FG, Fritz R, Lobo MC, Alvord W, Strober W, Straus SE. Local and systemic cytokine responses during experimental human influenza A virus infection. Relation to symptom formation and host defense. J Clin Invest (1998) 101:643–9. 10.1172/JCI1355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Skoner DP, Gentile DA, Patel A, Doyle WJ. Evidence for cytokine mediation of disease expression in adults experimentally infected with influenza A virus. J Infect Dis (1999) 180:10–4. 10.1086/314823 [DOI] [PubMed] [Google Scholar]
- 89.Kaiser L, Fritz RS, Straus SE, Gubareva L, Hayden FG. Symptom pathogenesis during acute influenza: interleukin-6 and other cytokine responses. J Med Virol (2001) 64:262–8. 10.1002/jmv.1045 [DOI] [PubMed] [Google Scholar]
- 90.Francis T, Stuart-Harris CH. Studies on the nasal histology of epidemic influenza virus infection in the ferret: I. The development and repair of the nasal lesion. J Exp Med (1938) 68:789–802. 10.1084/jem.68.6.789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Haff RF, Schriver PW, Stewart RC. Pathogenesis of influenza in ferrets: nasal manifestations of disease. Br J Exp Pathol (1966) 47:435–44. [PMC free article] [PubMed] [Google Scholar]
- 92.Walsh JJ, Dietlein LF, Low FN, Burch GE, Mogabgab WJ. Bronchotracheal response in human influenza. Type A, Asian strain, as studied by light and electron microscopic examination of bronchoscopic biopsies. Arch Intern Med (1961) 108:376–88. 10.1001/archinte.1961.03620090048006 [DOI] [PubMed] [Google Scholar]
- 93.Lee N, Wong CK, Chan PK, Chan MC, Wong RY, Lun SW, et al. Cytokine response patterns in severe pandemic 2009 H1N1 and seasonal influenza among hospitalized adults. PLoS One (2011) 6:e26050. 10.1371/journal.pone.0026050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Glinsky GV. Genomic analysis of pandemic (H1N1) 2009 reveals association of increasing disease severity with emergence of novel hemagglutinin mutations. Cell Cycle (2010) 9:958–70. 10.4161/cc.9.5.10913 [DOI] [PubMed] [Google Scholar]
- 95.Baillie GJ, Galiano M, Agapow PM, Myers R, Chiam R, Gall A, et al. Evolutionary dynamics of local pandemic H1N1/2009 influenza virus lineages revealed by whole-genome analysis. J Virol (2012) 86:11–8. 10.1128/JVI.05347-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Zarychanski R, Stuart TL, Kumar A, Doucette S, Elliott L, Kettner J, et al. Correlates of severe disease in patients with 2009 pandemic influenza (H1N1) virus infection. CMAJ (2010) 182:257–64. 10.1503/cmaj.091884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Louie JK, Acosta M, Winter K, Jean C, Gavali S, Schechter R, et al. Factors associated with death or hospitalization due to pandemic 2009 influenza A(H1N1) infection in California. JAMA (2009) 302:1896–902. 10.1001/jama.2009.1583 [DOI] [PubMed] [Google Scholar]
- 98.Nguyen-Van-Tam JS, Openshaw PJ, Hashim A, Gadd EM, Lim WS, Semple MG, et al. Risk factors for hospitalisation and poor outcome with pandemic A/H1N1 influenza: United Kingdom first wave (May-September 2009). Thorax (2010) 65:645–51. 10.1136/thx.2010.135210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Rosen DG, Lopez AE, Anzalone ML, Wolf DA, Derrick SM, Florez LF, et al. Postmortem findings in eight cases of influenza A/H1N1. Mod Pathol (2010) 23:1449–57. 10.1038/modpathol.2010.148 [DOI] [PubMed] [Google Scholar]
- 100.Hagau N, Slavcovici A, Gonganau DN, Oltean S, Dirzu DS, Brezoszki ES, et al. Clinical aspects and cytokine response in severe H1N1 influenza A virus infection. Crit Care (2010) 14:R203. 10.1186/cc9324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Kim YH, Kim JE, Hyun MC. Cytokine response in pediatric patients with pandemic influenza H1N1 2009 virus infection and pneumonia: comparison with pediatric pneumonia without H1N1 2009 infection. Pediatr Pulmonol (2011) 46:1233–9. 10.1002/ppul.21496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Takano T, Tajiri H, Kashiwagi Y, Kimura S, Kawashima H. Cytokine and chemokine response in children with the 2009 pandemic influenza A (H1N1) virus infection. Eur J Clin Microbiol Infect Dis (2011) 30:117–20. 10.1007/s10096-010-1041-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.To KK, Hung IF, Li IW, Lee KL, Koo CK, Yan WW, et al. Delayed clearance of viral load and marked cytokine activation in severe cases of pandemic H1N1 2009 influenza virus infection. Clin Infect Dis (2010) 50:850–9. 10.1086/650581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Stevens J, Blixt O, Glaser L, Taubenberger JK, Palese P, Paulson JC, et al. Glycan microarray analysis of the hemagglutinins from modern and pandemic influenza viruses reveals different receptor specificities. J Mol Biol (2006) 355:1143–55. 10.1016/j.jmb.2005.11.002 [DOI] [PubMed] [Google Scholar]
- 105.Morens DM, Taubenberger JK, Fauci AS. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J Infect Dis (2008) 198:962–70. 10.1086/591708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Memoli MJ, Tumpey TM, Jagger BW, Dugan VG, Sheng ZM, Qi L, et al. An early ’classical’ swine H1N1 influenza virus shows similar pathogenicity to the 1918 pandemic virus in ferrets and mice. Virology (2009) 393:338–45. 10.1016/j.virol.2009.08.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kobasa D, Jones SM, Shinya K, Kash JC, Copps J, Ebihara H, et al. Aberrant innate immune response in lethal infection of macaques with the 1918 influenza virus. Nature (2007) 445:319–23. 10.1038/nature05495 [DOI] [PubMed] [Google Scholar]
- 108.Tumpey TM, Basler CF, Aguilar PV, Zeng H, Solorzano A, Swayne DE, et al. Characterization of the reconstructed 1918 Spanish influenza pandemic virus. Science (2005) 310:77–80. 10.1126/science.1119392 [DOI] [PubMed] [Google Scholar]
- 109.Wang SM, Liao YT, Hu YS, Ho TS, Shen CF, Wang JR, et al. Immunophenotype expressions and cytokine profiles of influenza A H1N1 virus infection in pediatric patients in 2009. Dis Markers (2014) 2014:195453. 10.1155/2014/195453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Palese P. Influenza: old and new threats. Nat Med (2004) 10:S82–7. 10.1038/nm1141 [DOI] [PubMed] [Google Scholar]
- 111.van den Brand JM, Stittelaar KJ, van Amerongen G, Reperant L, de Waal L, Osterhaus AD, et al. Comparison of temporal and spatial dynamics of seasonal H3N2, pandemic H1N1 and highly pathogenic avian influenza H5N1 virus infections in ferrets. PLoS One (2012) 7:e42343. 10.1371/journal.pone.0042343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.van den Brand JM, Stittelaar KJ, van Amerongen G, Rimmelzwaan GF, Simon J, de Wit E, et al. Severity of pneumonia due to new H1N1 influenza virus in ferrets is intermediate between that due to seasonal H1N1 virus and highly pathogenic avian influenza H5N1 virus. J Infect Dis (2010) 201:993–9. 10.1086/651132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Belser JA, Jayaraman A, Raman R, Pappas C, Zeng H, Cox NJ, et al. Effect of D222G mutation in the hemagglutinin protein on receptor binding, pathogenesis and transmissibility of the 2009 pandemic H1N1 influenza virus. PLoS One (2011) 6:e25091. 10.1371/journal.pone.0025091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Maines TR, Belser JA, Gustin KM, van Hoeven N, Zeng H, Svitek N, et al. Local innate immune responses and influenza virus transmission and virulence in ferrets. J Infect Dis (2012) 205:474–85. 10.1093/infdis/jir768 [DOI] [PubMed] [Google Scholar]
- 115.Maines TR, Jayaraman A, Belser JA, Wadford DA, Pappas C, Zeng H, et al. Transmission and pathogenesis of swine-origin 2009 A(H1N1) influenza viruses in ferrets and mice. Science (2009) 325:484–7. 10.1126/science.1177238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Neumann G, Ozawa M, Kawaoka Y. Reverse genetics of influenza viruses. Methods Mol Biol (2012) 865:193–206. 10.1007/978-1-61779-621-0_12 [DOI] [PubMed] [Google Scholar]
- 117.Abed Y, Pizzorno A, Hamelin ME, Leung A, Joubert P, Couture C, et al. The 2009 pandemic H1N1 D222G hemagglutinin mutation alters receptor specificity and increases virulence in mice but not in ferrets. J Infect Dis (2011) 204:1008–16. 10.1093/infdis/jir483 [DOI] [PubMed] [Google Scholar]
- 118.Camp JV, Chu YK, Chung DH, McAllister RC, Adcock RS, Gerlach RL, et al. Phenotypic differences in virulence and immune response in closely related clinical isolates of influenza A 2009 H1N1 pandemic viruses in mice. PLoS One (2013) 8:e56602. 10.1371/journal.pone.0056602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chutinimitkul S, Herfst S, Steel J, Lowen AC, Ye J, van Riel D, et al. Virulence-associated substitution D222G in the hemagglutinin of 2009 pandemic influenza A(H1N1) virus affects receptor binding. J Virol (2010) 84:11802–13. 10.1128/JVI.01136-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Itoh Y, Shinya K, Kiso M, Watanabe T, Sakoda Y, Hatta M, et al. In vitro and in vivo characterization of new swine-origin H1N1 influenza viruses. Nature (2009) 460:1021–5. 10.1038/nature08260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Jhung MA, Swerdlow D, Olsen SJ, Jernigan D, Biggerstaff M, Kamimoto L, et al. Epidemiology of 2009 pandemic influenza A (H1N1) in the United States. Clin Infect Dis (2011) 52(Suppl 1):S13–26. 10.1093/cid/ciq008 [DOI] [PubMed] [Google Scholar]
- 122.Melidou A, Gioula G, Exindari M, Chatzidimitriou D, Diza E, Malisiovas N. Molecular and phylogenetic analysis of the haemagglutinin gene of pandemic influenza H1N1 2009 viruses associated with severe and fatal infections. Virus Res (2010) 151:192–9. 10.1016/j.virusres.2010.05.005 [DOI] [PubMed] [Google Scholar]
- 123.Meunier I, Embury-Hyatt C, Stebner S, Gray M, Bastien N, Li Y, et al. Virulence differences of closely related pandemic 2009 H1N1 isolates correlate with increased inflammatory responses in ferrets. Virology (2012) 422:125–31. 10.1016/j.virol.2011.10.018 [DOI] [PubMed] [Google Scholar]
- 124.Otte A, Gabriel G. 2009 pandemic H1N1 influenza A virus strains display differential pathogenicity in C57BL/6J but not BALB/c mice. Virulence (2011) 2:563–6. 10.4161/viru.2.6.18148 [DOI] [PubMed] [Google Scholar]
- 125.Safronetz D, Rockx B, Feldmann F, Belisle SE, Palermo RE, Brining D, et al. Pandemic swine-origin H1N1 influenza A virus isolates show heterogeneous virulence in macaques. J Virol (2011) 85:1214–23. 10.1128/JVI.01848-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ye J, Sorrell EM, Cai Y, Shao H, Xu K, Pena L, et al. Variations in the hemagglutinin of the 2009 H1N1 pandemic virus: potential for strains with altered virulence phenotype? PLoS Pathog (2010) 6:e1001145. 10.1371/journal.ppat.1001145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gubareva LV, McCullers JA, Bethell RC, Webster RG. Characterization of influenza A/HongKong/156/97 (H5N1) virus in a mouse model and protective effect of zanamivir on H5N1 infection in mice. J Infect Dis (1998) 178:1592–6. 10.1086/314515 [DOI] [PubMed] [Google Scholar]
- 128.Shortridge KF, Zhou NN, Guan Y, Gao P, Ito T, Kawaoka Y, et al. Characterization of avian H5N1 influenza viruses from poultry in Hong Kong. Virology (1998) 252:331–42. 10.1006/viro.1998.9488 [DOI] [PubMed] [Google Scholar]
- 129.Chen Y, Liang W, Yang S, Wu N, Gao H, Sheng J, et al. Human infections with the emerging avian influenza A H7N9 virus from wet market poultry: clinical analysis and characterisation of viral genome. Lancet (2013) 381:1916–25. 10.1016/S0140-6736(13)60903-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wong SS, Yuen KY. Avian influenza virus infections in humans. Chest (2006) 129:156–68. 10.1378/chest.129.1.156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Fouchier RA, Schneeberger PM, Rozendaal FW, Broekman JM, Kemink SA, Munster V, et al. Avian influenza A virus (H7N7) associated with human conjunctivitis and a fatal case of acute respiratory distress syndrome. Proc Natl Acad Sci U S A (2004) 101:1356–61. 10.1073/pnas.0308352100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chen E, Chen Y, Fu L, Chen Z, Gong Z, Mao H, et al. Human infection with avian influenza A(H7N9) virus re-emerges in China in winter 2013. Euro Surveill (2013) 18:20616. 10.2807/1560-7917.ES2013.18.43.20616 [DOI] [PubMed] [Google Scholar]
- 133.Shinya K, Gao Y, Cilloniz C, Suzuki Y, Fujie M, Deng G, et al. Integrated clinical, pathologic, virologic, and transcriptomic analysis of H5N1 influenza virus-induced viral pneumonia in the rhesus macaque. J Virol (2012) 86:6055–66. 10.1128/JVI.00365-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zhou J, Wang D, Gao R, Zhao B, Song J, Qi X, et al. Biological features of novel avian influenza A (H7N9) virus. Nature (2013) 499:500–3. 10.1038/nature12379 [DOI] [PubMed] [Google Scholar]
- 135.Bouvier NM, Lowen AC. Animal models for influenza virus pathogenesis and transmission. Viruses (2010) 2:1530–63. 10.3390/v20801530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Long JP, Kotur MS, Stark GV, Warren RL, Kasoji M, Craft JL, et al. Accumulation of CD11b(+)Gr-1(+) cells in the lung, blood and bone marrow of mice infected with highly pathogenic H5N1 and H1N1 influenza viruses. Arch Virol (2013) 158:1305–22. 10.1007/s00705-012-1593-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zhu H, Wang D, Kelvin DJ, Li L, Zheng Z, Yoon SW, et al. Infectivity, transmission, and pathology of human-isolated H7N9 influenza virus in ferrets and pigs. Science (2013) 341:183–6. 10.1126/science.1239844 [DOI] [PubMed] [Google Scholar]
- 138.Tate MD, Ioannidis LJ, Croker B, Brown LE, Brooks AG, Reading PC. The role of neutrophils during mild and severe influenza virus infections of mice. PLoS One (2011) 6:e17618. 10.1371/journal.pone.0017618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Narasaraju T, Yang E, Samy RP, Ng HH, Poh WP, Liew AA, et al. Excessive neutrophils and neutrophil extracellular traps contribute to acute lung injury of influenza pneumonitis. Am J Pathol (2011) 179:199–210. 10.1016/j.ajpath.2011.03.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Tate MD, Brooks AG, Reading PC. The role of neutrophils in the upper and lower respiratory tract during influenza virus infection of mice. Respir Res (2008) 9:57. 10.1186/1465-9921-9-57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Bradley LM, Douglass MF, Chatterjee D, Akira S, Baaten BJ. Matrix metalloprotease 9 mediates neutrophil migration into the airways in response to influenza virus-induced toll-like receptor signaling. PLoS Pathog (2012) 8:e1002641. 10.1371/journal.ppat.1002641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Seo SU, Kwon HJ, Song JH, Byun YH, Seong BL, Kawai T, et al. MyD88 signaling is indispensable for primary influenza A virus infection but dispensable for secondary infection. J Virol (2010) 84:12713–22. 10.1128/JVI.01675-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Walsh KB, Teijaro JR, Brock LG, Fremgen DM, Collins PL, Rosen H, et al. Animal model of respiratory syncytial virus: CD8+ T cells cause a cytokine storm that is chemically tractable by sphingosine-1-phosphate 1 receptor agonist therapy. J Virol (2014) 88:6281–93. 10.1128/JVI.00464-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Oldstone MB, Rosen H. Cytokine storm plays a direct role in the morbidity and mortality from influenza virus infection and is chemically treatable with a single sphingosine-1-phosphate agonist molecule. Curr Top Microbiol Immunol (2014) 378:129–47. 10.1007/978-3-319-05879-5_6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Borregaard N, Sorensen OE, Theilgaard-Monch K. Neutrophil granules: a library of innate immunity proteins. Trends Immunol (2007) 28:340–5. 10.1016/j.it.2007.06.002 [DOI] [PubMed] [Google Scholar]
- 146.Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, et al. Neutrophil extracellular traps kill bacteria. Science (2004) 303:1532–5. 10.1126/science.1092385 [DOI] [PubMed] [Google Scholar]
- 147.Prince LR, Whyte MK, Sabroe I, Parker LC. The role of TLRs in neutrophil activation. Curr Opin Pharmacol (2011) 11:397–403. 10.1016/j.coph.2011.06.007 [DOI] [PubMed] [Google Scholar]
- 148.Thomas CJ, Schroder K. Pattern recognition receptor function in neutrophils. Trends Immunol (2013) 34:317–28. 10.1016/j.it.2013.02.008 [DOI] [PubMed] [Google Scholar]
- 149.Bjornson AB, Mellencamp MA, Schiff GM. Complement is activated in the upper respiratory tract during influenza virus infection. Am Rev Respir Dis (1991) 143:1062–6. 10.1164/ajrccm/143.5_Pt_1.1062 [DOI] [PubMed] [Google Scholar]
- 150.Fritz RS, Hayden FG, Calfee DP, Cass LM, Peng AW, Alvord WG, et al. Nasal cytokine and chemokine responses in experimental influenza A virus infection: results of a placebo-controlled trial of intravenous zanamivir treatment. J Infect Dis (1999) 180:586–93. 10.1086/314938 [DOI] [PubMed] [Google Scholar]
- 151.Lee NL. Role of cytokines and chemokines in severe and complicated influenza infections. Hong Kong Med J (2009) 15(Suppl 8):38–41. [PubMed] [Google Scholar]
- 152.Lee N, Chan PK, Wong CK, Wong KT, Choi KW, Joynt GM, et al. Viral clearance and inflammatory response patterns in adults hospitalized for pandemic 2009 influenza A(H1N1) virus pneumonia. Antivir Ther (2011) 16:237–47. 10.3851/IMP1722 [DOI] [PubMed] [Google Scholar]
- 153.Garcia CC, Weston-Davies W, Russo RC, Tavares LP, Rachid MA, Alves-Filho JC, et al. Complement C5 activation during influenza A infection in mice contributes to neutrophil recruitment and lung injury. PLoS One (2013) 8:e64443. 10.1371/journal.pone.0064443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Schmitz N, Kurrer M, Bachmann MF, Kopf M. Interleukin-1 is responsible for acute lung immunopathology but increases survival of respiratory influenza virus infection. J Virol (2005) 79:6441–8. 10.1128/JVI.79.10.6441-6448.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Ichinohe T, Lee HK, Ogura Y, Flavell R, Iwasaki A. Inflammasome recognition of influenza virus is essential for adaptive immune responses. J Exp Med (2009) 206:79–87. 10.1084/jem.20081667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Crowe CR, Chen K, Pociask DA, Alcorn JF, Krivich C, Enelow RI, et al. Critical role of IL-17RA in immunopathology of influenza infection. J Immunol (2009) 183:5301–10. 10.4049/jimmunol.0900995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ichikawa A, Kuba K, Morita M, Chida S, Tezuka H, Hara H, et al. CXCL10-CXCR3 enhances the development of neutrophil-mediated fulminant lung injury of viral and nonviral origin. Am J Respir Crit Care Med (2013) 187:65–77. 10.1164/rccm.201203-0508OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Schliehe C, Flynn EK, Vilagos B, Richson U, Swaminathan S, Bosnjak B, et al. The methyltransferase Setdb2 mediates virus-induced susceptibility to bacterial superinfection. Nat Immunol (2015) 16:67–74. 10.1038/ni.3046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Wheeler JL, Martin KC, Lawrence BP. Novel cellular targets of AhR underlie alterations in neutrophilic inflammation and inducible nitric oxide synthase expression during influenza virus infection. J Immunol (2013) 190:659–68. 10.4049/jimmunol.1201341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Teske S, Bohn AA, Hogaboam JP, Lawrence BP. Aryl hydrocarbon receptor targets pathways extrinsic to bone marrow cells to enhance neutrophil recruitment during influenza virus infection. Toxicol Sci (2008) 102:89–99. 10.1093/toxsci/kfm282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Neff-LaFord H, Teske S, Bushnell TP, Lawrence BP. Aryl hydrocarbon receptor activation during influenza virus infection unveils a novel pathway of IFN-gamma production by phagocytic cells. J Immunol (2007) 179:247–55. 10.4049/jimmunol.179.1.247 [DOI] [PubMed] [Google Scholar]
- 162.Teske S, Bohn AA, Regal JF, Neumiller JJ, Lawrence BP. Activation of the aryl hydrocarbon receptor increases pulmonary neutrophilia and diminishes host resistance to influenza A virus. Am J Physiol Lung Cell Mol Physiol (2005) 289:L111–24. 10.1152/ajplung.00318.2004 [DOI] [PubMed] [Google Scholar]
- 163.Ratcliffe D, Migliorisi G, Cramer E. Translocation of influenza virus by migrating neutrophils. Cell Mol Biol (1992) 38:63–70. [PubMed] [Google Scholar]
- 164.Hayashi F, Means TK, Luster AD. Toll-like receptors stimulate human neutrophil function. Blood (2003) 102:2660–9. 10.1182/blood-2003-04-1078 [DOI] [PubMed] [Google Scholar]
- 165.Hattermann K, Picard S, Borgeat M, Leclerc P, Pouliot M, Borgeat P. The toll-like receptor 7/8-ligand resiquimod (R-848) primes human neutrophils for leukotriene B4, prostaglandin E2 and platelet-activating factor biosynthesis. FASEB J (2007) 21:1575–85. 10.1096/fj.06-7457com [DOI] [PubMed] [Google Scholar]
- 166.Tamassia N, Le Moigne V, Rossato M, Donini M, McCartney S, Calzetti F, et al. Activation of an immunoregulatory and antiviral gene expression program in poly(I:C)-transfected human neutrophils. J Immunol (2008) 181:6563–73. 10.4049/jimmunol.181.9.6563 [DOI] [PubMed] [Google Scholar]
- 167.Le Goffic R, Balloy V, Lagranderie M, Alexopoulou L, Escriou N, Flavell R, et al. Detrimental contribution of the toll-like receptor (TLR)3 to influenza A virus-induced acute pneumonia. PLoS Pathog (2006) 2:e53. 10.1371/journal.ppat.0020053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Leung YH, Nicholls JM, Ho CK, Sia SF, Mok CK, Valkenburg SA, et al. Highly pathogenic avian influenza A H5N1 and pandemic H1N1 virus infections have different phenotypes in toll-like receptor 3 knockout mice. J Gen Virol (2014) 95:1870–9. 10.1099/vir.0.066258-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Andonegui G, Goyert SM, Kubes P. Lipopolysaccharide-induced leukocyte-endothelial cell interactions: a role for CD14 versus toll-like receptor 4 within microvessels. J Immunol (2002) 169:2111–9. 10.4049/jimmunol.169.4.2111 [DOI] [PubMed] [Google Scholar]
- 170.Kolb JP, Casella CR, SenGupta S, Chilton PM, Mitchell TC. Type I interferon signaling contributes to the bias that toll-like receptor 4 exhibits for signaling mediated by the adaptor protein TRIF. Sci Signal (2014) 7:ra108. 10.1126/scisignal.2005442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.van Bruggen R, Drewniak A, Tool AT, Jansen M, van Houdt M, Geissler J, et al. Toll-like receptor responses in IRAK-4-deficient neutrophils. J Innate Immun (2010) 2:280–7. 10.1159/000268288 [DOI] [PubMed] [Google Scholar]
- 172.Hartshorn K, Chang D, Rust K, White M, Heuser J, Crouch E. Interactions of recombinant human pulmonary surfactant protein D and SP-D multimers with influenza A. Am J Physiol (1996) 271:L753–62. [DOI] [PubMed] [Google Scholar]
- 173.Hartshorn KL, Crouch EC, White MR, Eggleton P, Tauber AI, Chang D, et al. Evidence for a protective role of pulmonary surfactant protein D (SP-D) against influenza A viruses. J Clin Invest (1994) 94:311–9. 10.1172/JCI117323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Hartshorn KL, White MR, Voelker DR, Coburn J, Zaner K, Crouch EC. Mechanism of binding of surfactant protein D to influenza A viruses: importance of binding to haemagglutinin to antiviral activity. Biochem J (2000) 351(Pt 2):449–58. 10.1042/bj3510449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.LeVine AM, Whitsett JA, Hartshorn KL, Crouch EC, Korfhagen TR. Surfactant protein D enhances clearance of influenza A virus from the lung in vivo. J Immunol (2001) 167:5868–73. 10.4049/jimmunol.167.10.5868 [DOI] [PubMed] [Google Scholar]
- 176.Qi L, Kash JC, Dugan VG, Jagger BW, Lau YF, Sheng ZM, et al. The ability of pandemic influenza virus hemagglutinins to induce lower respiratory pathology is associated with decreased surfactant protein D binding. Virology (2011) 412:426–34. 10.1016/j.virol.2011.01.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.van Eijk M, Bruinsma L, Hartshorn KL, White MR, Rynkiewicz MJ, Seaton BA, et al. Introduction of N-linked glycans in the lectin domain of surfactant protein D: impact on interactions with influenza A viruses. J Biol Chem (2011) 286:20137–51. 10.1074/jbc.M111.224469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Hartshorn KL, White MR, Tecle T, Holmskov U, Crouch EC. Innate defense against influenza A virus: activity of human neutrophil defensins and interactions of defensins with surfactant protein D. J Immunol (2006) 176:6962–72. 10.4049/jimmunol.176.11.6962 [DOI] [PubMed] [Google Scholar]
- 179.Tecle T, White MR, Gantz D, Crouch EC, Hartshorn KL. Human neutrophil defensins increase neutrophil uptake of influenza A virus and bacteria and modify virus-induced respiratory burst responses. J Immunol (2007) 178:8046–52. 10.4049/jimmunol.178.12.8046 [DOI] [PubMed] [Google Scholar]
- 180.Doss M, White MR, Tecle T, Gantz D, Crouch EC, Jung G, et al. Interactions of alpha-, beta-, and theta-defensins with influenza A virus and surfactant protein D. J Immunol (2009) 182:7878–87. 10.4049/jimmunol.0804049 [DOI] [PubMed] [Google Scholar]
- 181.Salvatore M, Garcia-Sastre A, Ruchala P, Lehrer RI, Chang T, Klotman ME. alpha-Defensin inhibits influenza virus replication by cell-mediated mechanism(s). J Infect Dis (2007) 196:835–43. 10.1086/521027 [DOI] [PubMed] [Google Scholar]
- 182.Daher KA, Selsted ME, Lehrer RI. Direct inactivation of viruses by human granulocyte defensins. J Virol (1986) 60:1068–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Ratcliffe DR, Michl J, Cramer EB. Neutrophils do not bind to or phagocytize human immune complexes formed with influenza virus. Blood (1993) 82:1639–46. [PubMed] [Google Scholar]
- 184.Fujisawa H. Neutrophils play an essential role in cooperation with antibody in both protection against and recovery from pulmonary infection with influenza virus in mice. J Virol (2008) 82:2772–83. 10.1128/JVI.01210-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Hartshorn KL, Liou LS, White MR, Kazhdan MM, Tauber JL, Tauber AI. Neutrophil deactivation by influenza A virus. Role of hemagglutinin binding to specific sialic acid-bearing cellular proteins. J Immunol (1995) 154:3952–60. [PubMed] [Google Scholar]
- 186.Ivan FX, Tan KS, Phoon MC, Engelward BP, Welsch RE, Rajapakse JC, et al. Neutrophils infected with highly virulent influenza H3N2 virus exhibit augmented early cell death and rapid induction of type I interferon signaling pathways. Genomics (2013) 101:101–12. 10.1016/j.ygeno.2012.11.008 [DOI] [PubMed] [Google Scholar]
- 187.Wang JP, Bowen GN, Padden C, Cerny A, Finberg RW, Newburger PE, et al. Toll-like receptor-mediated activation of neutrophils by influenza A virus. Blood (2008) 112:2028–34. 10.1182/blood-2008-01-132860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Allen IC, Moore CB, Schneider M, Lei Y, Davis BK, Scull MA, et al. NLRX1 protein attenuates inflammatory responses to infection by interfering with the RIG-I-MAVS and TRAF6-NF-kappaB signaling pathways. Immunity (2011) 34:854–65. 10.1016/j.immuni.2011.03.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Jaworska J, Coulombe F, Downey J, Tzelepis F, Shalaby K, Tattoli I, et al. NLRX1 prevents mitochondrial induced apoptosis and enhances macrophage antiviral immunity by interacting with influenza virus PB1-F2 protein. Proc Natl Acad Sci U S A (2014) 111:E2110–9. 10.1073/pnas.1322118111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Stasakova J, Ferko B, Kittel C, Sereinig S, Romanova J, Katinger H, et al. Influenza A mutant viruses with altered NS1 protein function provoke caspase-1 activation in primary human macrophages, resulting in fast apoptosis and release of high levels of interleukins 1beta and 18. J Gen Virol (2005) 86:185–95. 10.1099/vir.0.80422-0 [DOI] [PubMed] [Google Scholar]
- 191.Allen IC, Scull MA, Moore CB, Holl EK, McElvania-TeKippe E, Taxman DJ, et al. The NLRP3 inflammasome mediates in vivo innate immunity to influenza A virus through recognition of viral RNA. Immunity (2009) 30:556–65. 10.1016/j.immuni.2009.02.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Belisle SE, Tisoncik JR, Korth MJ, Carter VS, Proll SC, Swayne DE, et al. Genomic profiling of tumor necrosis factor alpha (TNF-alpha) receptor and interleukin-1 receptor knockout mice reveals a link between TNF-alpha signaling and increased severity of 1918 pandemic influenza virus infection. J Virol (2010) 84:12576–88. 10.1128/JVI.01310-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Huang CH, Chen CJ, Yen CT, Yu CP, Huang PN, Kuo RL, et al. Caspase-1 deficient mice are more susceptible to influenza A virus infection with PA variation. J Infect Dis (2013) 208:1898–905. 10.1093/infdis/jit381 [DOI] [PubMed] [Google Scholar]
- 194.Perrone LA, Szretter KJ, Katz JM, Mizgerd JP, Tumpey TM. Mice lacking both TNF and IL-1 receptors exhibit reduced lung inflammation and delay in onset of death following infection with a highly virulent H5N1 virus. J Infect Dis (2010) 202:1161–70. 10.1086/656365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Szretter KJ, Gangappa S, Lu X, Smith C, Shieh WJ, Zaki SR, et al. Role of host cytokine responses in the pathogenesis of avian H5N1 influenza viruses in mice. J Virol (2007) 81:2736–44. 10.1128/JVI.02336-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Abramson JS, Lewis JC, Lyles DS, Heller KA, Mills EL, Bass DA. Inhibition of neutrophil lysosome-phagosome fusion associated with influenza virus infection in vitro. Role in depressed bactericidal activity. J Clin Invest (1982) 69:1393–7. 10.1172/JCI110580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Walzog B, Schuppan D, Heimpel C, Hafezi-Moghadam A, Gaehtgens P, Ley K. The leukocyte integrin Mac-1 (CD11b/CD18) contributes to binding of human granulocytes to collagen. Exp Cell Res (1995) 218:28–38. 10.1006/excr.1995.1127 [DOI] [PubMed] [Google Scholar]
- 198.Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol (2006) 6:173–82. 10.1038/nri1785 [DOI] [PubMed] [Google Scholar]
- 199.Sugamata R, Dobashi H, Nagao T, Yamamoto K, Nakajima N, Sato Y, et al. Contribution of neutrophil-derived myeloperoxidase in the early phase of fulminant acute respiratory distress syndrome induced by influenza virus infection. Microbiol Immunol (2012) 56:171–82. 10.1111/j.1348-0421.2011.00424.x [DOI] [PubMed] [Google Scholar]
- 200.Yamamoto K, Miyoshi-Koshio T, Utsuki Y, Mizuno S, Suzuki K. Virucidal activity and viral protein modification by myeloperoxidase: a candidate for defense factor of human polymorphonuclear leukocytes against influenza virus infection. J Infect Dis (1991) 164:8–14. 10.1093/infdis/164.1.8 [DOI] [PubMed] [Google Scholar]
- 201.Foong RE, Sly PD, Larcombe AN, Zosky GR. No role for neutrophil elastase in influenza-induced cellular recruitment, cytokine production or airway hyperresponsiveness in mice. Respir Physiol Neurobiol (2010) 173:164–70. 10.1016/j.resp.2010.08.003 [DOI] [PubMed] [Google Scholar]
- 202.Abramson JS, Mills EL, Giebink GS, Quie PG. Depression of monocyte and polymorphonuclear leukocyte oxidative metabolism and bactericidal capacity by influenza A virus. Infect Immun (1982) 35:350–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Schwarz KB. Oxidative stress during viral infection: a review. Free Radic Biol Med (1996) 21:641–9. 10.1016/0891-5849(96)00131-1 [DOI] [PubMed] [Google Scholar]
- 204.Knobil K, Choi AM, Weigand GW, Jacoby DB. Role of oxidants in influenza virus-induced gene expression. Am J Physiol (1998) 274:L134–42. [DOI] [PubMed] [Google Scholar]
- 205.Lazrak A, Iles KE, Liu G, Noah DL, Noah JW, Matalon S. Influenza virus M2 protein inhibits epithelial sodium channels by increasing reactive oxygen species. FASEB J (2009) 23:3829–42. 10.1096/fj.09-135590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Snelgrove RJ, Edwards L, Rae AJ, Hussell T. An absence of reactive oxygen species improves the resolution of lung influenza infection. Eur J Immunol (2006) 36:1364–73. 10.1002/eji.200635977 [DOI] [PubMed] [Google Scholar]
- 207.Cooper JA, Jr, Carcelen R, Culbreth R. Effects of influenza A nucleoprotein on polymorphonuclear neutrophil function. J Infect Dis (1996) 173:279–84. 10.1093/infdis/173.2.279 [DOI] [PubMed] [Google Scholar]
- 208.Buffinton GD, Christen S, Peterhans E, Stocker R. Oxidative stress in lungs of mice infected with influenza A virus. Free Radic Res Commun (1992) 16:99–110. 10.3109/10715769209049163 [DOI] [PubMed] [Google Scholar]
- 209.Perrone LA, Belser JA, Wadford DA, Katz JM, Tumpey TM. Inducible nitric oxide contributes to viral pathogenesis following highly pathogenic influenza virus infection in mice. J Infect Dis (2013) 207:1576–84. 10.1093/infdis/jit062 [DOI] [PubMed] [Google Scholar]
- 210.Suliman HB, Ryan LK, Bishop L, Folz RJ. Prevention of influenza-induced lung injury in mice overexpressing extracellular superoxide dismutase. Am J Physiol Lung Cell Mol Physiol (2001) 280:L69–78. [DOI] [PubMed] [Google Scholar]
- 211.Vlahos R, Stambas J, Bozinovski S, Broughton BR, Drummond GR, Selemidis S. Inhibition of Nox2 oxidase activity ameliorates influenza A virus-induced lung inflammation. PLoS Pathog (2011) 7:e1001271. 10.1371/journal.ppat.1001271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Zhang WJ, Wei H, Tien YT, Frei B. Genetic ablation of phagocytic NADPH oxidase in mice limits TNFalpha-induced inflammation in the lungs but not other tissues. Free Radic Biol Med (2011) 50:1517–25. 10.1016/j.freeradbiomed.2011.02.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Akaike T, Noguchi Y, Ijiri S, Setoguchi K, Suga M, Zheng YM, et al. Pathogenesis of influenza virus-induced pneumonia: involvement of both nitric oxide and oxygen radicals. Proc Natl Acad Sci U S A (1996) 93:2448–53. 10.1073/pnas.93.6.2448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G, et al. Identification of oxidative stress and toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell (2008) 133:235–49. 10.1016/j.cell.2008.02.043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Nhu QM, Shirey K, Teijaro JR, Farber DL, Netzel-Arnett S, Antalis TM, et al. Novel signaling interactions between proteinase-activated receptor 2 and toll-like receptors in vitro and in vivo. Mucosal Immunol (2010) 3:29–39. 10.1038/mi.2009.120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Shirey KA, Lai W, Scott AJ, Lipsky M, Mistry P, Pletneva LM, et al. The TLR4 antagonist Eritoran protects mice from lethal influenza infection. Nature (2013) 497:498–502. 10.1038/nature12118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Fuchs TA, Abed U, Goosmann C, Hurwitz R, Schulze I, Wahn V, et al. Novel cell death program leads to neutrophil extracellular traps. J Cell Biol (2007) 176:231–41. 10.1083/jcb.200606027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. J Immunol (2012) 189:2689–95. 10.4049/jimmunol.1201719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Urban CF, Reichard U, Brinkmann V, Zychlinsky A. Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell Microbiol (2006) 8:668–76. 10.1111/j.1462-5822.2005.00659.x [DOI] [PubMed] [Google Scholar]
- 220.Guimaraes-Costa AB, Nascimento MT, Froment GS, Soares RP, Morgado FN, Conceicao-Silva F, et al. Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps. Proc Natl Acad Sci U S A (2009) 106:6748–53. 10.1073/pnas.0900226106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Agraz-Cibrian JM, Giraldo DM, Mary FM, Urcuqui-Inchima S. Understanding the molecular mechanisms of NETs and their role in antiviral innate immunity. Virus Res (2017) 228:124–33. 10.1016/j.virusres.2016.11.033 [DOI] [PubMed] [Google Scholar]
- 222.Kessenbrock K, Krumbholz M, Schonermarck U, Back W, Gross WL, Werb Z, et al. Netting neutrophils in autoimmune small-vessel vasculitis. Nat Med (2009) 15:623–5. 10.1038/nm.1959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Marcos V, Zhou Z, Yildirim AO, Bohla A, Hector A, Vitkov L, et al. CXCR2 mediates NADPH oxidase-independent neutrophil extracellular trap formation in cystic fibrosis airway inflammation. Nat Med (2010) 16:1018–23. 10.1038/nm.2209 [DOI] [PubMed] [Google Scholar]
- 224.Hakkim A, Furnrohr BG, Amann K, Laube B, Abed UA, Brinkmann V, et al. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc Natl Acad Sci U S A (2010) 107:9813–8. 10.1073/pnas.0909927107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Raftery MJ, Lalwani P, Krautkrmer E, Peters T, Scharffetter-Kochanek K, Kruger R, et al. beta2 integrin mediates hantavirus-induced release of neutrophil extracellular traps. J Exp Med (2014) 211:1485–97. 10.1084/jem.20131092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Hemmers S, Teijaro JR, Arandjelovic S, Mowen KA. PAD4-mediated neutrophil extracellular trap formation is not required for immunity against influenza infection. PLoS One (2011) 6:e22043. 10.1371/journal.pone.0022043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Eruslanov EB, Lyadova IV, Kondratieva TK, Majorov KB, Scheglov IV, Orlova MO, et al. Neutrophil responses to Mycobacterium tuberculosis infection in genetically susceptible and resistant mice. Infect Immun (2005) 73:1744–53. 10.1128/IAI.73.3.1744-1753.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Seiler P, Aichele P, Bandermann S, Hauser AE, Lu B, Gerard NP, et al. Early granuloma formation after aerosol Mycobacterium tuberculosis infection is regulated by neutrophils via CXCR3-signaling chemokines. Eur J Immunol (2003) 33:2676–86. 10.1002/eji.200323956 [DOI] [PubMed] [Google Scholar]
- 229.Kennedy JL, Turner RB, Braciale T, Heymann PW, Borish L. Pathogenesis of rhinovirus infection. Curr Opin Virol (2012) 2:287–93. 10.1016/j.coviro.2012.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Levandowski RA, Weaver CW, Jackson GG. Nasal-secretion leukocyte populations determined by flow cytometry during acute rhinovirus infection. J Med Virol (1988) 25:423–32. 10.1002/jmv.1890250406 [DOI] [PubMed] [Google Scholar]
- 231.Turner RB. The role of neutrophils in the pathogenesis of rhinovirus infections. Pediatr Infect Dis J (1990) 9:832–5. 10.1097/00006454-199011000-00011 [DOI] [PubMed] [Google Scholar]
- 232.Domachowske JB, Rosenberg HF. Respiratory syncytial virus infection: immune response, immunopathogenesis, and treatment. Clin Microbiol Rev (1999) 12:298–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Pickles RJ, DeVincenzo JP. Respiratory syncytial virus (RSV) and its propensity for causing bronchiolitis. J Pathol (2015) 235:266–76. 10.1002/path.4462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Schildgen V, van den Hoogen B, Fouchier R, Tripp RA, Alvarez R, Manoha C, et al. Human metapneumovirus: lessons learned over the first decade. Clin Microbiol Rev (2011) 24:734–54. 10.1128/CMR.00015-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Vargas SO, Kozakewich HP, Perez-Atayde AR, McAdam AJ. Pathology of human metapneumovirus infection: insights into the pathogenesis of a newly identified respiratory virus. Pediatr Dev Pathol (2004) 7:478–86; discussion 421. 10.1007/s10024-004-1011-2 [DOI] [PubMed] [Google Scholar]
- 236.Hooper P, Zaki S, Daniels P, Middleton D. Comparative pathology of the diseases caused by Hendra and Nipah viruses. Microbes Infect (2001) 3:315–22. 10.1016/S1286-4579(01)01385-5 [DOI] [PubMed] [Google Scholar]
- 237.Koh YY, Jung da E, Koh JY, Kim JY, Yoo Y, Kim CK. Bronchoalveolar cellularity and interleukin-8 levels in measles bronchiolitis obliterans. Chest (2007) 131:1454–60. 10.1378/chest.06-0188 [DOI] [PubMed] [Google Scholar]
- 238.Downham MA, Gardner PS, McQuillin J, Ferris JA. Role of respiratory viruses in childhood mortality. Br Med J (1975) 1:235–9. 10.1136/bmj.1.5952.235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Zaki SR, Greer PW, Coffield LM, Goldsmith CS, Nolte KB, Foucar K, et al. Hantavirus pulmonary syndrome. Pathogenesis of an emerging infectious disease. Am J Pathol (1995) 146:552–79. [PMC free article] [PubMed] [Google Scholar]
- 240.Wu W, Booth JL, Duggan ES, Patel KB, Coggeshall KM, Metcalf JP. Human lung innate immune cytokine response to adenovirus type 7. J Gen Virol (2010) 91:1155–63. 10.1099/vir.0.017905-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Pyrc K, Berkhout B, van der Hoek L. The novel human coronaviruses NL63 and HKU1. J Virol (2007) 81:3051–7. 10.1128/JVI.01466-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Moolenaar RL, Dalton C, Lipman HB, Umland ET, Gallaher M, Duchin JS, et al. Clinical features that differentiate hantavirus pulmonary syndrome from three other acute respiratory illnesses. Clin Infect Dis (1995) 21:643–9. 10.1093/clinids/21.3.643 [DOI] [PubMed] [Google Scholar]
- 243.Smits SL, van den Brand JM, de Lang A, Leijten LM, van Ijcken WF, van Amerongen G, et al. Distinct severe acute respiratory syndrome coronavirus-induced acute lung injury pathways in two different nonhuman primate species. J Virol (2011) 85:4234–45. 10.1128/JVI.02395-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Schomacker H, Schaap-Nutt A, Collins PL, Schmidt AC. Pathogenesis of acute respiratory illness caused by human parainfluenza viruses. Curr Opin Virol (2012) 2:294–9. 10.1016/j.coviro.2012.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Jiang Y, Xu J, Zhou C, Wu Z, Zhong S, Liu J, et al. Characterization of cytokine/chemokine profiles of severe acute respiratory syndrome. Am J Respir Crit Care Med (2005) 171:850–7. 10.1164/rccm.200407-857OC [DOI] [PubMed] [Google Scholar]
- 246.Bataki EL, Evans GS, Everard ML. Respiratory syncytial virus and neutrophil activation. Clin Exp Immunol (2005) 140:470–7. 10.1111/j.1365-2249.2005.02780.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol (2013) 13:159–75. 10.1038/nri3399 [DOI] [PubMed] [Google Scholar]
- 248.van Kempen M, Bachert C, Van Cauwenberge P. An update on the pathophysiology of rhinovirus upper respiratory tract infections. Rhinology (1999) 37:97–103. [PubMed] [Google Scholar]
- 249.Winther B. Effects on the nasal mucosa of upper respiratory viruses (common cold). Dan Med Bull (1994) 41:193–204. [PubMed] [Google Scholar]
- 250.Winther B, Gwaltney JM, Jr, Mygind N, Hendley JO. Viral-induced rhinitis. Am J Rhinol (1998) 12:17–20. 10.2500/105065898782102954 [DOI] [PubMed] [Google Scholar]
- 251.Racaniello V. Picornaviridae: the viruses and their replication. 5th ed In: Knipe DM, Howley PM, editors. Fields Virology. (Vol. 1), Philadelphia, PA: Lippincott Williams and Wilkins; (2007). p. 795–838. [Google Scholar]
- 252.Gern JE, Busse WW. Association of rhinovirus infections with asthma. Clin Microbiol Rev (1999) 12:9–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Gern JE, Martin MS, Anklam KA, Shen K, Roberg KA, Carlson-Dakes KT, et al. Relationships among specific viral pathogens, virus-induced interleukin-8, and respiratory symptoms in infancy. Pediatr Allergy Immunol (2002) 13:386–93. 10.1034/j.1399-3038.2002.01093.x [DOI] [PubMed] [Google Scholar]
- 254.Murawski MR, Bowen GN, Cerny AM, Anderson LJ, Haynes LM, Tripp RA, et al. Respiratory syncytial virus activates innate immunity through toll-like receptor 2. J Virol (2009) 83:1492–500. 10.1128/JVI.00671-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Loo YM, Fornek J, Crochet N, Bajwa G, Perwitasari O, Martinez-Sobrido L, et al. Distinct RIG-I and MDA5 signaling by RNA viruses in innate immunity. J Virol (2008) 82:335–45. 10.1128/JVI.01080-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Mibayashi M, Martinez-Sobrido L, Loo YM, Cardenas WB, Gale M, Jr, Garcia-Sastre A. Inhibition of retinoic acid-inducible gene I-mediated induction of beta interferon by the NS1 protein of influenza A virus. J Virol (2007) 81:514–24. 10.1128/JVI.01265-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Kato H, Sato S, Yoneyama M, Yamamoto M, Uematsu S, Matsui K, et al. Cell type-specific involvement of RIG-I in antiviral response. Immunity (2005) 23:19–28. 10.1016/j.immuni.2005.04.010 [DOI] [PubMed] [Google Scholar]
- 258.Koyama S, Ishii KJ, Kumar H, Tanimoto T, Coban C, Uematsu S, et al. Differential role of TLR- and RLR-signaling in the immune responses to influenza A virus infection and vaccination. J Immunol (2007) 179:4711–20. 10.4049/jimmunol.179.7.4711 [DOI] [PubMed] [Google Scholar]
- 259.Kelly JT, Busse WW. Host immune responses to rhinovirus: mechanisms in asthma. J Allergy Clin Immunol (2008) 122:671–82; quiz 683–74. 10.1016/j.jaci.2008.08.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Honda K, Taniguchi T. IRFs: master regulators of signalling by toll-like receptors and cytosolic pattern-recognition receptors. Nat Rev Immunol (2006) 6:644–58. 10.1038/nri1900 [DOI] [PubMed] [Google Scholar]
- 261.Dixit E, Kagan JC. Intracellular pathogen detection by RIG-I-like receptors. Adv Immunol (2013) 117:99–125. 10.1016/B978-0-12-410524-9.00004-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Zinman G, Brower-Sinning R, Emeche CH, Ernst J, Huang GT, Mahony S, et al. Large scale comparison of innate responses to viral and bacterial pathogens in mouse and macaque. PLoS One (2011) 6:e22401. 10.1371/journal.pone.0022401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Diebold SS, Kaisho T, Hemmi H, Akira S, Reis e Sousa C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science (2004) 303:1529–31. 10.1126/science.1093616 [DOI] [PubMed] [Google Scholar]
- 264.Byeon JH, Lee JC, Choi IS, Yoo Y, Park SH, Choung JT. Comparison of cytokine responses in nasopharyngeal aspirates from children with viral lower respiratory tract infections. Acta Paediatr (2009) 98:725–30. 10.1111/j.1651-2227.2008.01208.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Sung RY, Hui SH, Wong CK, Lam CW, Yin J. A comparison of cytokine responses in respiratory syncytial virus and influenza A infections in infants. Eur J Pediatr (2001) 160:117–22. 10.1007/s004310000676 [DOI] [PubMed] [Google Scholar]
- 266.Shirey KA, Pletneva LM, Puche AC, Keegan AD, Prince GA, Blanco JC, et al. Control of RSV-induced lung injury by alternatively activated macrophages is IL-4R alpha-, TLR4-, and IFN-beta-dependent. Mucosal Immunol (2010) 3:291–300. 10.1038/mi.2010.6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Paul WE, Seder RA. Lymphocyte responses and cytokines. Cell (1994) 76:241–51. 10.1016/0092-8674(94)90332-8 [DOI] [PubMed] [Google Scholar]
- 268.Bonizzi G, Karin M. The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol (2004) 25:280–8. 10.1016/j.it.2004.03.008 [DOI] [PubMed] [Google Scholar]
- 269.Schindler C, Levy DE, Decker T. JAK-STAT signaling: from interferons to cytokines. J Biol Chem (2007) 282:20059–63. 10.1074/jbc.R700016200 [DOI] [PubMed] [Google Scholar]
- 270.Borden EC, Williams BR. Interferon-stimulated genes and their protein products: what and how? J Interferon Cytokine Res (2011) 31:1–4. 10.1089/jir.2010.0129 [DOI] [PubMed] [Google Scholar]
- 271.Furuya Y, Steiner D, Metzger DW. Does type I interferon limit protective neutrophil responses during pulmonary Francisella tularensis infection? Front Immunol (2014) 5:355. 10.3389/fimmu.2014.00355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Garcia-Sastre A. Induction and evasion of type I interferon responses by influenza viruses. Virus Res (2011) 162:12–8. 10.1016/j.virusres.2011.10.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Martinelli S, Urosevic M, Daryadel A, Oberholzer PA, Baumann C, Fey MF, et al. Induction of genes mediating interferon-dependent extracellular trap formation during neutrophil differentiation. J Biol Chem (2004) 279:44123–32. 10.1074/jbc.M405883200 [DOI] [PubMed] [Google Scholar]
- 274.Meunier I, von Messling V. NS1-mediated delay of type I interferon induction contributes to influenza A virulence in ferrets. J Gen Virol (2011) 92:1635–44. 10.1099/vir.0.032193-0 [DOI] [PubMed] [Google Scholar]
- 275.Seo SU, Kwon HJ, Ko HJ, Byun YH, Seong BL, Uematsu S, et al. Type I interferon signaling regulates Ly6C(hi) monocytes and neutrophils during acute viral pneumonia in mice. PLoS Pathog (2011) 7:e1001304. 10.1371/journal.ppat.1001304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Svitek N, Rudd PA, Obojes K, Pillet S, von Messling V. Severe seasonal influenza in ferrets correlates with reduced interferon and increased IL-6 induction. Virology (2008) 376:53–9. 10.1016/j.virol.2008.02.035 [DOI] [PubMed] [Google Scholar]
- 277.Wang X, Li M, Zheng H, Muster T, Palese P, Beg AA, et al. Influenza A virus NS1 protein prevents activation of NF-kappaB and induction of alpha/beta interferon. J Virol (2000) 74:11566–73. 10.1128/JVI.74.24.11566-11573.2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Garcia-Romo GS, Caielli S, Vega B, Connolly J, Allantaz F, Xu Z, et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci Transl Med (2011) 3:73ra20. 10.1126/scitranslmed.3001201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Decker P. Neutrophils and interferon-alpha-producing cells: who produces interferon in lupus? Arthritis Res Ther (2011) 13:118. 10.1186/ar3345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Stock AT, Smith JM, Carbone FR. Type I IFN suppresses Cxcr2 driven neutrophil recruitment into the sensory ganglia during viral infection. J Exp Med (2014) 211:751–9. 10.1084/jem.20132183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Xin L, Vargas-Inchaustegui DA, Raimer SS, Kelly BC, Hu J, Zhu L, et al. Type I IFN receptor regulates neutrophil functions and innate immunity to Leishmania parasites. J Immunol (2010) 184:7047–56. 10.4049/jimmunol.0903273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Durbin JE, Fernandez-Sesma A, Lee CK, Rao TD, Frey AB, Moran TM, et al. Type I IFN modulates innate and specific antiviral immunity. J Immunol (2000) 164:4220–8. 10.4049/jimmunol.164.8.4220 [DOI] [PubMed] [Google Scholar]
- 283.Hashimoto Y, Moki T, Takizawa T, Shiratsuchi A, Nakanishi Y. Evidence for phagocytosis of influenza virus-infected, apoptotic cells by neutrophils and macrophages in mice. J Immunol (2007) 178:2448–57. 10.4049/jimmunol.178.4.2448 [DOI] [PubMed] [Google Scholar]
- 284.Tate MD, Brooks AG, Reading PC, Mintern JD. Neutrophils sustain effective CD8(+) T-cell responses in the respiratory tract following influenza infection. Immunol Cell Biol (2012) 90:197–205. 10.1038/icb.2011.26 [DOI] [PubMed] [Google Scholar]
- 285.Tate MD, Deng YM, Jones JE, Anderson GP, Brooks AG, Reading PC. Neutrophils ameliorate lung injury and the development of severe disease during influenza infection. J Immunol (2009) 183:7441–50. 10.4049/jimmunol.0902497 [DOI] [PubMed] [Google Scholar]
- 286.Herold S, Tabar TS, Janssen H, Hoegner K, Cabanski M, Lewe-Schlosser P, et al. Exudate macrophages attenuate lung injury by the release of IL-1 receptor antagonist in gram-negative pneumonia. Am J Respir Crit Care Med (2011) 183:1380–90. 10.1164/rccm.201009-1431OC [DOI] [PubMed] [Google Scholar]
- 287.Dean RA, Cox JH, Bellac CL, Doucet A, Starr AE, Overall CM. Macrophage-specific metalloelastase (MMP-12) truncates and inactivates ELR+ CXC chemokines and generates CCL2, -7, -8, and -13 antagonists: potential role of the macrophage in terminating polymorphonuclear leukocyte influx. Blood (2008) 112:3455–64. 10.1182/blood-2007-12-129080 [DOI] [PubMed] [Google Scholar]
- 288.Fadok VA, Bratton DL, Konowal A, Freed PW, Westcott JY, Henson PM. Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J Clin Invest (1998) 101:890–8. 10.1172/JCI1112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.El Kebir D, Filep JG. Targeting neutrophil apoptosis for enhancing the resolution of inflammation. Cells (2013) 2:330–48. 10.3390/cells2020330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Godson C, Mitchell S, Harvey K, Petasis NA, Hogg N, Brady HR. Cutting edge: lipoxins rapidly stimulate nonphlogistic phagocytosis of apoptotic neutrophils by monocyte-derived macrophages. J Immunol (2000) 164:1663–7. 10.4049/jimmunol.164.4.1663 [DOI] [PubMed] [Google Scholar]
- 291.Poe SL, Arora M, Oriss TB, Yarlagadda M, Isse K, Khare A, et al. STAT1-regulated lung MDSC-like cells produce IL-10 and efferocytose apoptotic neutrophils with relevance in resolution of bacterial pneumonia. Mucosal Immunol (2013) 6:189–99. 10.1038/mi.2012.62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Dienz O, Rud JG, Eaton SM, Lanthier PA, Burg E, Drew A, et al. Essential role of IL-6 in protection against H1N1 influenza virus by promoting neutrophil survival in the lung. Mucosal Immunol (2012) 5:258–66. 10.1038/mi.2012.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Arndt U, Wennemuth G, Barth P, Nain M, Al-Abed Y, Meinhardt A, et al. Release of macrophage migration inhibitory factor and CXCL8/interleukin-8 from lung epithelial cells rendered necrotic by influenza A virus infection. J Virol (2002) 76:9298–306. 10.1128/JVI.76.18.9298-9306.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Simard JC, Girard D, Tessier PA. Induction of neutrophil degranulation by S100A9 via a MAPK-dependent mechanism. J Leukoc Biol (2010) 87:905–14. 10.1189/jlb.1009676 [DOI] [PubMed] [Google Scholar]
- 295.Tsai SY, Segovia JA, Chang TH, Morris IR, Berton MT, Tessier PA, et al. DAMP molecule S100A9 acts as a molecular pattern to enhance inflammation during influenza A virus infection: role of DDX21-TRIF-TLR4-MyD88 pathway. PLoS Pathog (2014) 10:e1003848. 10.1371/journal.ppat.1003848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Camp JV. Critical Insights into the Pathogenesis of Clinical Isolates of Pandemic Influenza A(H1N1) 2009 Virus in Mouse and Ferret Models [Doctoral Electronic Theses and Dissertations]. Louisville, KY: University of Louisville; (2015). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.