Abstract
The contribution of paternal obesity to pregnancy outcomes has been little described. Our aims were to determine whether the effect of an antenatal maternal dietary and lifestyle intervention among women who are overweight or obese on newborn adiposity, was modified by paternal obesity. We conducted a secondary analysis of a multicenter randomised trial. Pregnant women with BMI ≥25 kg/m2 received either Lifestyle Advice or Standard Care. Paternal anthropometric measures included height, weight, BMI; waist, hip, calf and mid-upper arm circumferences; biceps and calf skinfold thickness measurements (SFTM); and percentage body fat. Newborn anthropometric outcomes included length; weight; head, arm, abdominal, and chest circumferences; biceps, triceps, subscapular, suprailiac, thigh, and lateral abdominal wall SFTM; and percentage body fat. The effect of an antenatal maternal dietary and lifestyle intervention among women who were overweight or obese on neonatal anthropometric measures, was significantly modified by paternal BMI ≥35.0 kg/m2, with a significantly smaller infant triceps, suprailiac, and thigh SFTM, and percent fat mass, compared with that observed in offspring of lean fathers. Further research is required to determine whether our observed associations are causal, and whether paternal weight loss prior to conception is a potential strategy to reduce the intergenerational effects of obesity.
Introduction
Globally 2.1 billion adults are overweight or obese1. In many developed countries, overweight and obesity has increased over the past three decades1, with over 70% of adult males and 60% of females having a body mass index (BMI) above 25.0 kg/m2 1. Obesity contributes significantly to overall burden of disease2, being associated with increased risks of cardiovascular disease, type-2 diabetes and many malignancies3, contributing indirectly to more than 3.4 million deaths annually4.
Overweight and obesity during pregnancy represents a significant health burden, with almost 50% of women having a BMI above 25 kg/m2 on entering pregnancy5. Adverse effects of high maternal BMI on maternal pregnancy outcomes are well documented, the risks increasing as BMI increases6, 7. Furthermore, maternal obesity is associated with an increased risk of high infant birth weight, nursery admission, preterm birth, congenital anomalies, and both jaundice and hypoglycaemia7, with both high maternal BMI and gestational weight gain significant predictors of offspring obesity8. In an attempt to improve maternal and infant outcomes, interest has focused on interventions in the antenatal period to limit gestational weight gain, with systematic reviews and meta-analyses of randomized trials identifying only a modest effect on maternal weight change9, 10.
Paternal contributions to fetal growth, infant birth weight and body composition are less well described. Paternal height has been consistently correlated with birth weight and measures of skeletal growth, primarily birth length11–15. While some report positive associations between paternal weight and infant birth weight16, 17, this is not universal14. Furthermore, while some have identified associations between paternal BMI and infant birth weight16, 18, 19, others have not11, 13, 20. These studies have limitations. Most have not evaluated the contribution of paternal BMI to newborn anthropometric measures of adiposity, have involved relatively lean individuals, and have not adequately controlled for maternal factors contributing to fetal growth, or have relied upon maternal reporting of paternal weight and height.
The impact of paternal obesity on the efficacy of pregnancy interventions to limit gestational weight gain, particularly among overweight or obese women, has not been examined. We have reported the primary findings of the LIMIT randomised trial evaluating provision of antenatal dietary and lifestyle advice to women who were overweight or obese, indicating an 18% relative risk reduction in infant birth weight above 4 kg21, following improvements in maternal diet and physical activity22. The planned secondary study reported here, evaluated whether the effect of a randomised antenatal maternal dietary and lifestyle intervention among women who are overweight or obese on newborn adiposity measures, was modified by paternal BMI.
Results
We randomized 2,212 women, (1,108 Lifestyle Advice; 1,104 Standard Care). There were 2,142 live-born infants (1,075 Lifestyle Advice; 1,067 Standard Care), with detailed anthropometric measurements for 913 fathers and infants (442 Lifestyle Advice; 471 Standard Care) (Fig. 1). Baseline characteristics of mothers of these infants were similar between treatment groups (Table 1), and similar to the full randomised groups21. Paternal (Table 2) and neonatal23 anthropometric measurements were also similar between treatment groups. There was a statistically significant correlation between maternal and paternal BMI (Pearson correlation coefficient 0.22, p < 0.001); a higher proportion of fathers were in the obese category for the highest maternal BMI groups compared to the lowest; however more than 50% of fathers had BMI <30 in every maternal BMI category.
Figure 1.
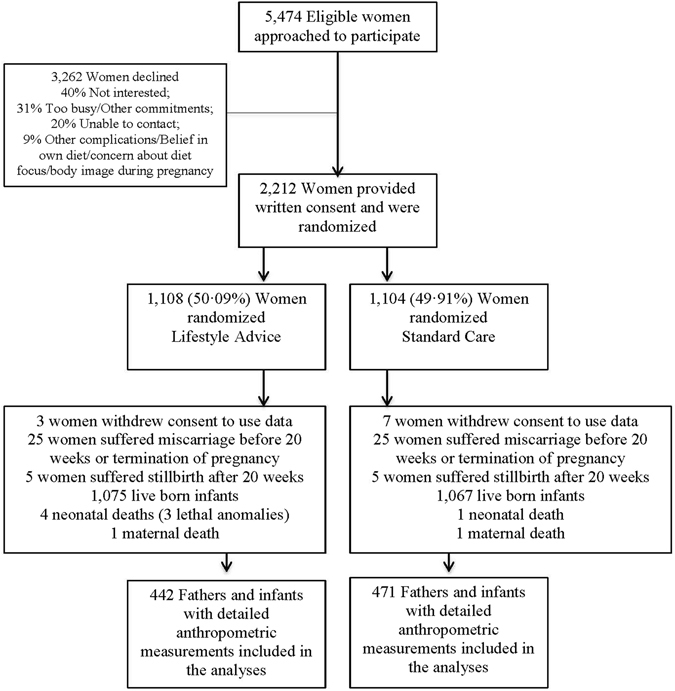
Flow of participants through the trial.
Table 1.
Maternal baseline characteristics by randomised group.
| Characteristic | Lifestyle Advice (N = 442) | Standard Care (N = 471) |
|---|---|---|
| Maternal Age (Years)* | 29.5 (5.7) | 29.6 (5.4) |
| Gestational Age at Entry (Weeks)+ | 14.0 (11.9–16.6) | 14.1 (11.7–16.7) |
| Body Mass Index (kg/m2)+ | 31.1 (27.9–36.2) | 31.3 (27.6–36.3) |
| Body Mass Index Category# | ||
| BMI 25.0–29.9 | 187 (42.3) | 190 (40.3) |
| BMI 30.0–34.9 | 112 (25.3) | 135 (28.7) |
| BMI 35.0–39.9 | 90 (20.4) | 79 (16.8) |
| BMI >=40.0 | 53 (12.0) | 67 (14.2) |
| Public Patient# | 429 (97.1) | 461 (97.9) |
| Caucasian# | 412 (93.2) | 431 (91.5) |
| Smoker# | 41 (9.3) | 48 (10.2) |
| Nulliparous# | 210 (47.5) | 231 (49.0) |
| Index of Socio-economic Disadvantage^ | ||
| Quintile 1 (Most Disadvantaged) | 123 (27.8) | 130 (27.6) |
| Quintile 2 | 115 (26.0) | 135 (28.7) |
| Quintile 3 | 69 (15.6) | 67 (14.2) |
| Quintile 4 | 64 (14.5) | 74 (15.7) |
| Quintile 5 (Least Disadvantaged) | 71 (16.1) | 65 (13.8) |
*Mean and standard deviation. +Median and interquartile range. #Number and %. ^Socioeconomic index as measured by SEIFA.
Table 2.
Paternal anthropometric characteristics by maternal randomised treatment group.
| Characteristic | Lifestyle Advice (N = 442) | Standard Care (N = 471) |
|---|---|---|
| Weight (kg)* | 91.9 (18.4) | 91.9 (18.7) |
| Height (cm)* | 179.0 (6.8) | 179.1 (7.5) |
| Body Mass Index (kg/m2)* | 28.7 (5.6) | 28.6 (5.4) |
| Body Mass Index Category# | ||
| Missing | 8 (1.8) | 10 (2.1) |
| BMI <25 | 110 (24.9) | 110 (23.4) |
| BMI 25-<30 | 183 (41.4) | 193 (41.0) |
| BMI 30-<35 | 89 (20.1) | 110 (23.4) |
| BMI >=35 | 52 (11.8) | 48 (10.2) |
| Age (years)*,^ | 32.58 (6.34) | 32.54 (6.35) |
| Calculated Fat Mass (kg)+ | 17.2 (11.3–24.8) | 17.7 (11.8–24.8) |
| Calculated Percent Body Fat+ | 19.4 (14.3–25.0) | 19.3 (14.4–25.2) |
*Mean and standard deviation. +Median and interquartile range. #Number and %. ^Based on a subset of 197 fathers (101 Lifestyle Advice, 96 Standard Care) for whom information on date of birth was available.
Modification of the antenatal dietary and lifestyle intervention on neonatal anthropometry by paternal BMI
Increasing paternal BMI significantly modified the effect of the antenatal intervention on infant triceps (p = 0.03) and suprailiac (p = 0.02) skinfold thickness measurement (SFTM), with some evidence to suggest paternal BMI was an effect modifier for thigh SFTM (p = 0.06) (Table 3). Specifically, in infants born to a father with BMI ≥35.0 kg/m2 the antenatal intervention was associated with smaller average triceps SFTM by 0.89 mm while no significant effect of the intervention was seen for infants born to fathers with BMI <25.0 kg/m2, or 30.0–34.9 kg/m2. Paternal BMI ≥35.0 kg/m2 modified the effect of the intervention for average infant suprailiac SFTM, which was smaller by 1.87 mm in the intervention group, in contrast to infants of fathers with BMI <25.0 kg/m2, 25.0–29.9 kg/m2 and 30.0–34.9 kg/m2 respectively.
Table 3.
Treatment effect modification of infant morphology, according to paternal body mass index category.
| Infant Outcome | Paternal BMI Category | Lifestyle Advice (N = 442) | Standard Care (N = 471) | Adjusted Treatment Effect (95% CI) | Adjusted P-value | Adjusted Interaction P-value |
|---|---|---|---|---|---|---|
| Birth Weight (g) | BMI <25 | 3470.45 (473.04) | 3492.44 (547.94) | −26.19 (−167.95, 115.57) | 0.712 | 0.93 |
| BMI 25–<30 | 3555.73 (466.05) | 3520.90 (547.15) | 22.06 (−86.62, 130.74) | 0.69 | ||
| BMI 30–<35 | 3515.25 (546.53) | 3512.65 (708.76) | −20.99 (−169.61, 127.63) | 0.78 | ||
| BMI >=35 | 3538.54 (491.83) | 3560.77 (509.84) | −34.80 (−244.12, 174.52) | 0.75 | ||
| Birth Length (cm) | BMI <25 | 49.94 (2.14) | 50.00 (2.37) | −0.08 (−0.71, 0.55) | 0.8 | 0.64 |
| BMI 25–<30 | 50.18 (1.90) | 50.09 (2.31) | 0.06 (−0.42, 0.54) | 0.79 | ||
| BMI 30–<35 | 50.01 (2.31) | 49.59 (3.24) | 0.38 (−0.28, 1.03) | 0.26 | ||
| BMI >=35 | 49.64 (2.30) | 49.84 (2.13) | −0.31 (−1.24, 0.62) | 0.51 | ||
| Head Circumference (cm) | BMI <25 | 34.94 (1.29) | 34.98 (1.70) | −0.10 (−0.52, 0.33) | 0.66 | 0.41 |
| BMI 25–<30 | 34.91 (1.41) | 34.88 (1.64) | 0.03 (−0.29, 0.36) | 0.84 | ||
| BMI 30–<35 | 34.92 (1.42) | 34.63 (2.01) | 0.24 (−0.21, 0.68) | 0.29 | ||
| BMI >=35 | 34.66 (1.78) | 35.06 (1.71) | −0.40 (−1.04, 0.23) | 0.21 | ||
| Chest Circumference (cm) | BMI <25 | 34.23 (1.78) | 34.45 (1.91) | −0.25 (−0.93, 0.44) | 0.48 | 0.4 |
| BMI 25–<30 | 34.39 (1.79) | 34.09 (2.02) | 0.35 (−0.18, 0.89) | 0.2 | ||
| BMI 30–<35 | 34.27 (2.12) | 34.45 (2.12) | −0.32 (−1.04, 0.40) | 0.39 | ||
| BMI >=35 | 33.92 (1.87) | 33.86 (2.35) | −0.15 (−1.11, 0.82) | 0.76 | ||
| Mid Upper Arm Circumference (cm) | BMI <25 | 11.29 (0.89) | 11.19 (1.18) | 0.02 (−0.34, 0.38) | 0.91 | 0.82 |
| BMI 25–<30 | 11.30 (1.01) | 11.18 (1.11) | 0.10 (−0.18, 0.38) | 0.49 | ||
| BMI 30–<35 | 11.22 (0.90) | 11.12 (0.98) | 0.03 (−0.35, 0.41) | 0.88 | ||
| BMI >=35 | 11.23 (1.05) | 10.90 (1.08) | 0.30 (−0.21, 0.81) | 0.25 | ||
| Abdominal Circumference (cm) | BMI <25 | 32.95 (2.10) | 32.82 (2.35) | −0.02 (−0.79, 0.75) | 0.95 | 0.18 |
| BMI 25–<30 | 32.99 (2.13) | 32.59 (2.34) | 0.40 (−0.20, 1.00) | 0.2 | ||
| BMI 30–<35 | 32.72 (2.23) | 33.15 (2.24) | −0.67 (−1.48, 0.13) | 0.1 | ||
| BMI >=35 | 32.69 (1.96) | 32.26 (2.52) | 0.40 (−0.68, 1.49) | 0.47 | ||
| Biceps SFTM (mm) | BMI <25 | 4.23 (0.94) | 4.13 (1.36) | 0.07 (−0.34, 0.48) | 0.74 | 0.81 |
| BMI 25–<30 | 4.60 (1.33) | 4.41 (1.15) | 0.16 (−0.16, 0.49) | 0.32 | ||
| BMI 30–<35 | 4.38 (1.26) | 4.19 (1.02) | 0.19 (−0.24, 0.63) | 0.39 | ||
| BMI >=35 | 4.20 (1.07) | 4.35 (1.03) | −0.13 (−0.72, 0.45) | 0.65 | ||
| Triceps SFTM (mm) | BMI <25 | 5.33 (1.27) | 5.37 (1.57) | −0.13 (−0.61, 0.36) | 0.61 | 0.03 |
| BMI 25–<30 | 5.61 (1.30) | 5.32 (1.44) | 0.27 (−0.11, 0.65) | 0.16 | ||
| BMI 30–<35 | 5.25 (1.48) | 5.33 (1.29) | −0.14 (−0.64, 0.37) | 0.59 | ||
| BMI >=35 | 5.03 (1.12) | 5.94 (1.50) | −0.89 (−1.57, −0.22) | 0.01 | ||
| Suprailiac SFTM (mm) | BMI <25 | 5.57 (1.76) | 5.55 (1.67) | −0.09 (−0.75, 0.57) | 0.79 | 0.002 |
| BMI 25–<30 | 5.94 (1.67) | 5.73 (1.87) | 0.21 (−0.30, 0.72) | 0.42 | ||
| BMI 30–<35 | 5.72 (2.20) | 5.77 (2.11) | −0.08 (−0.77, 0.62) | 0.83 | ||
| BMI >=35 | 5.12 (1.52) | 7.03 (2.20) | −1.87 (−2.81, −0.93) | 0.0001 | ||
| Subscapular SFTM (mm) | BMI <25 | 5.25 (1.17) | 5.05 (1.13) | 0.14 (–0.32, 0.60) | 0.55 | 0.64 |
| BMI 25–<30 | 5.13 (1.42) | 5.08 (1.27) | 0.06 (−0.30, 0.42) | 0.75 | ||
| BMI 30–<35 | 5.02 (1.49) | 5.10 (1.16) | −0.14 (−0.62, 0.34) | 0.56 | ||
| BMI >=35 | 5.12 (1.19) | 5.44 (1.64) | −0.31 (−0.96, 0.33) | 0.34 | ||
| Lateral Abdominal SFTM (mm) | BMI <25 | 3.72 (1.05) | 3.65 (1.03) | 0.06 (−0.32, 0.43) | 0.77 | 0.55 |
| BMI 25–<30 | 4.02 (1.21) | 3.83 (1.04) | 0.19 (−0.11, 0.49) | 0.21 | ||
| BMI 30–<35 | 3.84 (1.07) | 3.66 (1.01) | 0.13 (−0.27, 0.52) | 0.53 | ||
| BMI >=35 | 3.63 (0.92) | 3.93 (0.88) | −0.26 (−0.79, 0.28) | 0.35 | ||
| Thigh SFTM (mm) | BMI <25 | 6.68 (1.79) | 6.54 (1.81) | 0.06 (−0.60, 0.71) | 0.86 | 0.06 |
| BMI 25–<30 | 7.40 (1.92) | 6.99 (1.89) | 0.45 (−0.06, 0.95) | 0.08 | ||
| BMI 30–<35 | 6.97 (2.21) | 6.92 (1.72) | −0.06 (−0.74, 0.62) | 0.86 | ||
| BMI >=35 | 6.61 (1.65) | 7.67 (2.14) | −1.01 (−1.92, −0.09) | 0.03 | ||
| Calculated Fat Mass (g) | BMI <25 | 519.15 (161.25) | 514.15 (169.81) | −0.37 (−67.79, 67.05) | 0.99 | 0.12 |
| BMI 25–<30 | 546.39 (169.81) | 508.34 (196.11) | 37.26 (−14.61, 89.13) | 0.16 | ||
| BMI 30–<35 | 520.08 (239.72) | 528.87 (197.73) | −20.21 (−89.36, 48.94) | 0.57 | ||
| BMI >=35 | 478.74 (137.34) | 555.62 (200.03) | −89.49 (−182.90, 3.93) | 0.06 | ||
| Calculated Fat Free Mass (g)* | BMI <25 | 3058.33 (282.29) | 3062.06 (357.21) | −7.80 (−131.49, 115.90) | 0.9 | 0.88 |
| BMI 25–<30 | 3069.61 (305.55) | 3009.87 (360.21) | 52.80 (−42.36, 147.97) | 0.28 | ||
| BMI 30–<35 | 3066.72 (380.31) | 3027.67 (389.34) | 5.19 (−121.68, 132.05) | 0.94 | ||
| BMI >=35 | 2986.76 (375.95) | 2910.80 (380.12) | 28.40 (−142.99, 199.78) | 0.75 | ||
| Calculated Percent Body Fat | BMI <25 | 14.31 (3.39) | 14.09 (3.29) | 0.12 (−1.13, 1.38) | 0.85 | 0.02 |
| BMI 25–<30 | 14.85 (3.28) | 14.03 (3.61) | 0.84 (−0.12, 1.81) | 0.09 | ||
| BMI 30–<35 | 13.93 (4.28) | 14.50 (3.27) | −0.70 (−1.99, 0.59) | 0.29 | ||
| BMI >=35 | 13.63 (2.78) | 15.63 (3.77) | −2.15 (−3.89, −0.40) | 0.02 |
Values are mean (SD) and treatment effects are differences in means.
Where the interaction p-value is not significant, treatment group comparisons within subgroups should be interpreted with caution.
Results have been adjusted for centre, maternal parity, maternal BMI, maternal age, socioeconomic status and maternal smoking status.
*Calculated using equation from Deierlein et al.42. Note that ‘ethnicity’ (Hispanic vs non-Hispanic) is included in this equation but is not applicable to the LIMIT cohort (all participants are ‘non-Hispanic’).
Paternal BMI ≥35.0 kg/m2 also positively modified the effect of the antenatal intervention on infant thigh SFTM, being associated with smaller average thigh SFTM of 1.01 mm, when compared with the small, non-significant treatment effects seen among infants of fathers with BMI <25.0 kg/m2, 25.0–29.9 kg/m2, and 30.0–34.9 kg/m2 respectively. This finding should be interpreted with caution however, as the treatment group by paternal BMI category interaction test did not reach statistical significance (p = 0.06).
Paternal BMI ≥35 kg/m2 also positively modified infant percentage body fat in response to the intervention (p = 0.02; Table 3). Treatment effects were again seen for infants of fathers in the highest BMI category only, where the intervention was associated with significantly smaller percent body fat by 2.15%.
Increasing paternal BMI did not significantly modify the effect of the antenatal intervention on mean infant birth weight, birth length, or body circumferences (Table 3).
In the Standard Care group only, paternal BMI category was associated with significant differences in mean suprailiac SFTM (p = 0.005) and thigh SFTM (p = 0.04), and there was some evidence of a difference in mean percent body fat (p = 0.06), but there were no other statistically significant differences between subgroups (Table 4).
Table 4.
Associations between infant morphology and paternal body mass index category from participants in the Standard Care Group.
| Infant Characteristic | Paternal BMI <25 (N = 110) | Paternal BMI 25-<30 (N = 193) | Paternal BMI 30-<35 (N = 110) | Paternal BMI >=35 (n = 48) | P value |
|---|---|---|---|---|---|
| Weight (g) | 3492.44 (547.94) | 3520.90 (547.15) | 3512.65 (708.76) | 3560.77 (509.84) | 0.37 |
| Length (cm) | 50.00 (2.37) | 50.09 (2.31) | 49.59 (3.24) | 49.84 (2.13) | 0.84 |
| Head Circumference (cm) | 34.98 (1.70) | 34.88 (1.64) | 34.63 (2.01) | 35.06 (1.71) | 0.18 |
| Chest Circumference (cm) | 34.45 (1.91) | 34.09 (2.02) | 34.45 (2.12) | 33.86 (2.35) | 0.12 |
| Mid Upper Arm Circumference (cm) | 11.19 (1.18) | 11.18 (1.11) | 11.12 (0.98) | 10.90 (1.08) | 0.83 |
| Abdominal Circumference (cm) | 32.82 (2.35) | 32.59 (2.34) | 33.15 (2.24) | 32.26 (2.52) | 0.13 |
| Biceps SFTM (mm) | 4.13 (1.36) | 4.41 (1.15) | 4.19 (1.02) | 4.35 (1.03) | 0.45 |
| Triceps SFTM (mm) | 5.37 (1.57) | 5.32 (1.44) | 5.33 (1.29) | 5.94 (1.50) | 0.07 |
| Suprailiac SFTM (mm) | 5.55 (1.67) | 5.73 (1.87) | 5.77 (2.11) | 7.03 (2.20) | 0.005 |
| Subscapular SFTM (mm) | 5.05 (1.13) | 5.08 (1.27) | 5.10 (1.16) | 5.44 (1.64) | 0.15 |
| Lateral Abdominal SFTM (mm) | 3.65 (1.03) | 3.83 (1.04) | 3.66 (1.01) | 3.93 (0.88) | 0.51 |
| Thigh SFTM (mm) | 6.54 (1.81) | 6.99 (1.89) | 6.92 (1.72) | 7.67 (2.14) | 0.04 |
| Fat Mass (g) | 514.15 (169.81) | 508.34 (196.11) | 528.87 (197.73) | 555.62 (200.03) | 0.22 |
| Fat Free Mass (g) | 3062.06 (357.21) | 3009.87 (360.21) | 3027.67 (389.34) | 2910.80 (380.12) | 0.24 |
| Percent Body Fat | 14.09 (3.29) | 14.03 (3.61) | 14.50 (3.27) | 15.63 (3.77) | 0.06 |
Values are mean (SD).
*P-value from global test for effect of paternal BMI category. Results have been adjusted for maternal weight, height, BMI category, gestational weight gain, gestational diabetes, smoking status, socio-economic status, gestational age at birth and infant sex.
Discussion
Our findings identify an association between high paternal BMI and some measures of newborn adiposity. Specifically, the effect of the antenatal intervention in terms of smaller anthropometric measures of adiposity was significantly enhanced in infants whose fathers had a BMI ≥35.0 kg/m2, as demonstrated by smaller average infant triceps, suprailiac, and thigh SFTM, and infant percent fat mass in the intervention group.
Strengths of our study include its prospective, randomised nature and rigorous methodology, utilising standardised and well-accepted research standard anthropometric measures, adhering to defined protocols23, 24. In analysing the effect of paternal BMI on infant adiposity, we have controlled for maternal BMI, gestational weight gain, and other pregnancy factors influencing fetal growth and therefore birth weight. Importantly, our study allows us to address the potential contribution of paternal obesity to a range of infant anthropometric measures that include adiposity, which has not been possible to date, as previously reported cohorts have utilised relatively lean populations, with few participants at the upper extremes of the BMI spectrum11–16, 25.
Limitations include lack of a gold standard measure of paternal and infant adiposity, however the use of dual x-ray absorptiometry or air displacement plethysmography was prohibitive in the context of this large randomised trial. We have previously reported good reproducibility with infant skin fold measurements23. Timing of partner measurements would ideally have been at the woman’s first pregnancy visit, however women were not enrolled until several weeks after this visit, and paternal measures were taken on average at about 90 days after randomisation. Average young adult male weight change in one year is 0.6 kg26, and over the five-month time frame in which paternal measurements were obtained were considered unlikely to change. We also conducted further analyses (not reported) to check for any effect of the intervention on paternal measures; these found no differences between the Lifestyle Advice and Standard Care groups.
The effects of paternal height on newborn length and measures of skeletal growth11, 25 and weight11–16, have been reasonably well described and appear to be independent of both maternal height and weight. There are well reported associations between paternal height and both newborn length or measures of skeletal growth11, 25, and weight11–16, across varying populations world-wide, and over different time periods. Catalano and colleagues27 reported a weak positive association between paternal height and estimated infant fat free mass, the outcome more strongly associated with infant sex, gestational age at birth, maternal gestational weight gain, and maternal BMI prior to conception.
The contribution of paternal weight and BMI to infant anthropometric measures has been less clearly established to date. While the majority of studies have failed to identify associations between either paternal weight or BMI and infant birth weight11–15, Mikulandra and colleagues16 report on almost 1,600 newborns from Croatia, identifying a significant positive association between increasing paternal weight and BMI, and increasing infant birth weight. These findings are consistent with those of Chen and colleagues in an Asian population18, and Klebanoff and colleagues, who utilized Danish population linked data from women birthing between 1974 and 198919. In contrast, other studies conducted in predominantly Caucasian populations11, 13, 20 have not identified an association between increasing paternal BMI and infant birth weight. Most of these studies11, 13, 16, 18–20 have focused on infant birth weight, rather than investigating more direct measures of infant adiposity as we have.
The contribution of paternal obesity to human pregnancy outcomes has been previously poorly documented. The association between paternal BMI and infant adiposity described here may reflect paternal genetic influences, or paternal influences acting through the gamete environment prior to conception, or both. Our novel findings indicate that paternal obesity also significantly modifies the effect of an antenatal dietary and lifestyle intervention among pregnant women who are overweight or obese, on neonatal anthropometric measures of adiposity. One potential hypothesis to explain this observation may be that with increasing paternal BMI there is greater “familial” awareness of obesity, with women whose partners are of higher BMI being more supported to initiate changes to dietary and physical activity patterns. We investigated this possibility in a post-hoc analysis, but found no supporting evidence, with no substantial variation in maternal dietary and physical activity patterns across the paternal BMI spectrum.
There is increasing experimental evidence to support a male contribution to the development of offspring obesity, independent of genetics and a shared postnatal environment. Observational human literature identifies well recognized associations between paternal obesity, and an altered hormonal environment, impaired spermatogenesis, with reduced sperm counts28, 29 and impaired DNA integrity and mitochondrial function, secondary to increased reactive oxygen species28. Furthermore, these obesity related effects on spermatogenesis appear reversible through weight loss, either from dietary modification28, or following bariatric surgery29.
Animal based studies have identified similar reversible changes in spermatogenesis following diet-induced obesity in rodents30, 31, with suggestions that this impacts blastocyst function in early conception32. Furthermore, rodents fed a high-fat diet to induce obesity, both in the presence33 and absence34 of impaired glucose homeostasis, father offspring, particularly females, who are heavier, have reduced glucose tolerance, increased insulin resistance, and increased adiposity33, 34. Many of these adiposity effects persist in subsequent generations35.
While there is evidence to support a role for a male contribution to the development of offspring obesity, the precise mechanism whereby this “transmission” occurs remains uncertain36. Hypotheses include aberrant repair of spermatozoa DNA damage37, epigenetic changes reflecting hypo-methylation of histone bound DNA regions33, 38, and changes to non-coding small RNAs, such as micro RNAs39.
Of particular interest are the mechanisms underlying the effect of paternal BMI on the efficacy of the intervention in substantially ameliorating the increased infant adiposity associated with a high paternal BMI. The intervention, which improved maternal diet quality and physical activity22, presumably targets pathways in the fetus that mediate paternal genetic or preconception environmental influences on infant adiposity. These appear distinct from those harnessed by maternal obesity as by contrast, the intervention did not improve infant adiposity associated with a high maternal BMI23. Identifying the paternal pathways contributing to excess fetal adipose accumulation may reveal new targets and potential interventions to limit this.
Our findings highlight the need for ongoing research within human male populations into the impact of obesity on pregnancy and offspring health. Further research is required to determine whether our observed associations are causal, and, if so, whether paternal weight loss prior to conception would affect infant adiposity, as a potential strategy to reduce the adverse intergenerational effects of obesity.
Subjects and Methods
Study Design and Participants
The methodology40 and clinical findings have been reported21, 22, 41, and the trial registered on the Australian and New Zealand Clinical Trials Registry (ACTRN12607000161426). The LIMIT Study has been conducted in accordance with ethical and legal guidelines at the time the study was undertaken. Protocols used in this study were approved by the Women’s and Children’s Local Health Network Human Research and Ethics Committee at the Women’s and Children’s Hospital, the Central Northern Adelaide Health Service Ethics of Human Research Committee (Lyell McEwin Hospital) and the Flinders Clinical Research Ethics Committee (Flinders Medical Centre). Study methods complied with the Australian National Health and Medical Research Council’s National Statement on Ethical Conduct in Human Research (2007) - Updated May 2015 (https://www.nhmrc.gov.au/guidelines-publications/e72). Women with a singleton pregnancy between 10+0–20+0 weeks gestation, with BMI ≥25 kg/m2 were eligible.
Intervention
At the first antenatal appointment, women had their height and weight measured, and BMI calculated. Eligible women were presented with written information prior to providing consent. The central randomisation service utilised a computer-generated schedule with balanced variable blocks, and stratification for parity (0 versus 1/more), BMI at booking (25–29.9 kg/m2 versus ≥30 kg/m2), and collaborating centre. Women were randomised to either ‘Lifestyle Advice’ or ‘Standard Care’.
Lifestyle Advice Group
Women participated in a comprehensive dietary and lifestyle intervention over pregnancy, delivered by a research dietician and research assistants21, 22.
Standard Care Group
Women continued their care according to local hospital guidelines, which did not include dietary, exercise, or gestational weight gain advice.
Outcome Variables
Maternal BMI and gestational weight gain
At antenatal booking, women were categorised as overweight (BMI 25.0–29.9 kg/m2) or obese (BMI ≥30 kg/m2). Gestational weight gain was the difference in weight between 36 weeks gestation (or nearest to birth) and trial entry.
Paternal BMI and Anthropometry
Following informed consent, trained research assistants obtained detailed anthropometric measurements from the infant’s father during his partner’s pregnancy. Weight and height were measured to the nearest 0.1 kg and 0.1 cm respectively, in light clothing without shoes. BMI was categorised as normal (BMI <25 kg/m2), overweight (BMI 25.0–29.9 kg/m2), obese (BMI 30.0–34.9 kg/m2), or morbidly obese (BMI ≥35.0 kg/m2).
Waist, hip, calf, and mid-upper arm circumferences were obtained, and Harpenden skinfold callipers used to measure biceps and calf skinfold thicknesses. The following formula, derived from a demographically matched sample within an extensive database of complete anthropometric measurements, was used as a surrogate estimate of percentage body fat, total fat mass, and total non-fat mass (personal communication, Professor Timothy Olds, 17/10/12).
where BF = Body fat (%), Mass = Body Weight (kg), Ht = Height (cm), BicepsSF = Biceps skin fold thickness (mm), ArmG = Arm girth (cm), CalfSF = Calf skinfold thickness (mm), and WaistG = Waist Girth (cm).
Newborn Anthropometry
Prior to discharge from hospital, a trained research assistant obtained newborn anthropometric measures on healthy newborn infants on the postnatal ward, including birth weight, length, body circumference (head, arm, chest, and abdomen) and skinfold thickness measurements (SFTM) (biceps, triceps, subscapular, supra-iliac, thigh and lateral abdominal)23.
Infant gender, age in days and the sum of triceps, subscapular and thigh SFTM were used to calculate fat mass42. Percent body fat was calculated (fat mass/body weight at birth) × 100.
Statistical Analyses
To evaluate whether the effect of a randomised antenatal maternal dietary and lifestyle intervention among women who are overweight or obese on newborn adiposity measures, was modified by paternal BMI, we conducted intention to treat analyses, according to the woman’s allocated treatment group at randomisation. Live born infants were included if their mother consented to anthropometric measurements being taken, and one or more paternal anthropometric measurements were available. Linear regression models were used to test whether the effect of treatment was modified by paternal BMI category and other measures of paternal adiposity, by including an intervention-by-paternal BMI category term in the model. Post-hoc tests were performed to estimate the effect of treatment group within each paternal BMI category and results are presented as differences in means with 95% CI. Adjustment was made for centre, parity, maternal BMI category, maternal age, socio-economic status and maternal smoking status.
To evaluate whether there was an association between paternal BMI and infant adiposity, analyses were conducted using data from the Standard Care group only. Linear regression models were used to test whether there was a difference in infant outcome by paternal BMI category (categories being used rather than continuous BMI to allow for nonlinearity of association and to aid interpretability). Separate estimates of difference in means were derived for each paternal BMI category. Analyses were adjusted for maternal BMI, gestational weight gain, gestational diabetes, socioeconomic status, gestational age at delivery, and maternal smoking.
Statistical significance was assessed at the 2-sided p < 0.05 level. As these are secondary analyses (with the findings considered exploratory), no adjustment for multiple comparisons has been made43, 44. All analyses were performed using SAS v9.3 (Cary, NC, USA).
Acknowledgements
The LIMIT Trial was funded by a four-year project grant from the National Health and Medical Research Council (NHMRC), Australia (ID 519240). J.M. Dodd is supported through a NHMRC Practitioner Fellowship (ID 627005). R.M. Grivell is supported through a NHMRC Early Career Fellowship (ID 1073514). L.N. Yelland is supported through a NHMRC Early Career Fellowship (ID 1052388). Infrastructure support was provided by The University of Adelaide, and the Women’s and Children’s Hospital, Flinders Medical Centre, and Lyell McEwin Hospital, Adelaide. We are indebted to the women, their infants and their partners, who participated in the LIMIT randomised trial.
Author Contributions
J.D., A.M., J.R., and J.O. conceived the study; J.D., A.M., J.R., J.O., L.D., A.D., R.G. conducted the study; J.D., J.L., L.Y. conducted the statistical analyses; J.D., L.D. drafted the manuscript; all authors contributed intellectually to the manuscript and its revisions.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ng M, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384:766–781. doi: 10.1016/S0140-6736(14)60460-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ezzati M, et al. Selected major risk factors and global and regional burden of disease. Lancet. 2002;360:1347–1360. doi: 10.1016/S0140-6736(02)11403-6. [DOI] [PubMed] [Google Scholar]
- 3.World Health Organisation. Health in 2015: from MDGs, Millennium Development Goals to SDGs, Sustainable Development Goals. (2015).
- 4.Lim SS, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2224–2260. doi: 10.1016/S0140-6736(12)61766-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Scheil, W., Scott, J., Catcheside, B., Sage, L. & Kennare, R. (ed. Pregnancy Outcome Unit SA Health) (Government of South Australia, Adelaide, 2014).
- 6.Callaway LK, Prins JB, Chang AM, McIntyre HD. The prevalence and impact of overweight and obesity in an Australian obstetric population. MJA. 2006;184:56–59. doi: 10.5694/j.1326-5377.2006.tb00115.x. [DOI] [PubMed] [Google Scholar]
- 7.Dodd JM, Grivell RM, Nguyen A-M, Chan A, Robinson JS. Maternal and perinatal health outcomes by body mass index category. ANZJOG. 2011;51:136–140. doi: 10.1111/j.1479-828X.2010.01272.x. [DOI] [PubMed] [Google Scholar]
- 8.Baird J, et al. Being big or growing fast: systematic review of size and growth in infancy and later obesity. BMJ. 2005;331:929–0. doi: 10.1136/bmj.38586.411273.E0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dodd JM, Grivell RM, Crowther CA, Robinson JS. Antenatal interventions for overweight or obese pregnant women: a systematic review of randomised trials. British Journal of Obstetrics and Gynaecology. 2010;117:1316–1326. doi: 10.1111/j.1471-0528.2010.02540.x. [DOI] [PubMed] [Google Scholar]
- 10.Thangaratinam S, et al. Effects of interventions in pregnancy on maternal weight and obstetric outcomes: meta-analysis of randomised evidence. BMJ May. 2012;16(344):e2088–e2088. doi: 10.1136/bmj.e2088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Knight B, et al. Evidence of genetic regulation of fetal longitudinal growth. Early Hum Dev. 2005;81:823–831. doi: 10.1016/j.earlhumdev.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 12.Morrison J, Williams GM, Najman JM, Andersen MJ. The influence of paternal height and weight on birth-weight. Aust N Z J Obstet Gynaecol. 1991;31:114–116. doi: 10.1111/j.1479-828X.1991.tb01795.x. [DOI] [PubMed] [Google Scholar]
- 13.Nahum GG, Stanislaw H. Relationship of paternal factors to birth weight. J Reprod Med. 2003;48:963–968. [PubMed] [Google Scholar]
- 14.To WW, Cheung W, Kwok JS. Paternal height and weight as determinants of birth weight in a Chinese population. Am J Perinatol. 1998;15:545–548. doi: 10.1055/s-2007-994058. [DOI] [PubMed] [Google Scholar]
- 15.Wilcox MA, Newton CS, Johnson IR. Paternal influences on birthweight. Acta Obstet Gynecol Scand. 1995;74:15–18. doi: 10.3109/00016349509009936. [DOI] [PubMed] [Google Scholar]
- 16.Mikulandra F, Tadin I, Grgurić J, Zakanj Z, Perisa M. Influence of father’s weight and height on weight of male and female newborns. Coll Antropol. 2001;25:59–63. [PubMed] [Google Scholar]
- 17.Miletić T, et al. Effect of parental anthropometric parameters on neonatal birth weight and birth length. Coll Antropol. 2007;31:993–997. [PubMed] [Google Scholar]
- 18.Chen YP, et al. Paternal body mass index (BMI) is associated with offspring intrauterine growth in a gender dependent manner. PLoS One. 2012;7:e36329. doi: 10.1371/journal.pone.0036329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klebanoff MA, Mednick BR, Schulsinger C, Secher NJ, Shiono PH. Father’s effect on infant birth weight. Am J Obstet Gynecol. 1998;178:1022–1026. doi: 10.1016/S0002-9378(98)70542-3. [DOI] [PubMed] [Google Scholar]
- 20.Botton J, et al. Parental body size and early weight and height growth velocities in their offspring. Early Hum Dev. 2010;86:445–450. doi: 10.1016/j.earlhumdev.2010.06.001. [DOI] [PubMed] [Google Scholar]
- 21.Dodd JM, et al. Antenatal lifestyle advice for women who are overweight or obese: the LIMIT randomised trial. BMJ. 2014;348:g1285–g1285. doi: 10.1136/bmj.g1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dodd JM, et al. Effects of antenatal lifestyle advice for women who are overweight or obese on maternal diet and physical activity: the LIMIT randomised trial. BMC Med. 2014;12:161. doi: 10.1186/s12916-014-0161-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dodd JM, et al. The effect of antenatal lifestyle advice for women who are overweight or obese on neonatal body composition: the LIMIT randomised trial. BJOG. 2016;123:244–253. doi: 10.1111/1471-0528.13796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dodd JM, et al. Effects of an antenatal dietary intervention on maternal anthropometric measures in pregnant women with. obesity. Obesity (Silver Spring) 2015;23:1555–1562. doi: 10.1002/oby.21145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Veena, S. R., Krishnaveni, G. V., Wills, A. K., Hill, J. C. & Fall, C. H. A principal components approach to parent-to-newborn body composition associations in South India. BMC Pediatrics Feb 24; 9:16, doi:10.1186/1471-2431-9-16 (2009). [DOI] [PMC free article] [PubMed]
- 26.Kaikkonen JE, et al. Factors associated with six-year weight change in young and middle-aged adults in the Young Finns Study. Scand J Clin Lab Invest. 2015;75:133–144. doi: 10.3109/00365513.2014.992945. [DOI] [PubMed] [Google Scholar]
- 27.Catalano PM, Drago NM, Amini SB. Factors affecting fetal growth and body composition. Am J Obstet Gynecol. 1995;172:1459–1463. doi: 10.1016/0002-9378(95)90478-6. [DOI] [PubMed] [Google Scholar]
- 28.Håkonsen LB, et al. Does weight loss improve semen quality and reproductive hormones? Results from a cohort of severely obese men. Reprod Health. 2011;8:24. doi: 10.1186/1742-4755-8-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Reis LO, et al. Bariatric surgery does not interfere with sperm quality–a preliminary long-term study. Reprod Sci. 2012;19:1057–1062. doi: 10.1177/1933719112440747. [DOI] [PubMed] [Google Scholar]
- 30.Palmer NO, Bakos HW, Fullston T, Lane M. Impact of obesity on male fertility, sperm function and molecular composition. Spermatogenesis. 2012;2:253–263. doi: 10.4161/spmg.21362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Palmer NO, Bakos HW, Owens JA, Setchell BP, Lane M. Diet and exercise in an obese mouse fed a high-fat diet improve metabolic health and reverse perturbed sperm function. Am J Physiol Endocrinol Metab. 2012;302:E768–768. doi: 10.1152/ajpendo.00401.2011. [DOI] [PubMed] [Google Scholar]
- 32.McPherson NO, Bakos HW, Owens JA, Setchell BP, Lane M. Improving metabolic health in obese male mice via diet and exercise restores embryo development and fetal growth. PLoS One. 2013;19:e71459. doi: 10.1371/journal.pone.0071459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ng SF, et al. Chronic high fat diet in fathers programs beta cell dysfunction in female rat offspring. Nature. 2010;467:963–966. doi: 10.1038/nature09491. [DOI] [PubMed] [Google Scholar]
- 34.Fullston T, et al. Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content. FASEB Journal. 2013;27:4226–4243. doi: 10.1096/fj.12-224048. [DOI] [PubMed] [Google Scholar]
- 35.Ng SF, et al. Paternal high-fat diet consumption induces common changes in the transcriptomes of retroperitoneal adipose and pancreatic islet tissues in female rat offspring. FASEB Journal. 2014;28:1830–1841. doi: 10.1096/fj.13-244046. [DOI] [PubMed] [Google Scholar]
- 36.McPherson NO, Fullston T, Aitken RJ, Lane M. Paternal obesity, interventions, and mechanistic pathways to impaired health in offspring. Ann Nutr Metab. 2014;64:231–238. doi: 10.1159/000365026. [DOI] [PubMed] [Google Scholar]
- 37.Aitken RJ, Koopman P, Lewis SE. Seeds of concern. Nature. 2004;432:48–52. doi: 10.1038/432048a. [DOI] [PubMed] [Google Scholar]
- 38.Tunc O, Tremellen K. Oxidative DNA damage impairs global sperm DNA methylation in infertile men. J Assist Reprod Genet. 2009;26:537–544. doi: 10.1007/s10815-009-9346-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gapp K, et al. Implication of sperm RNAs in transgenerational inheritance of the effects of early trauma in mice. Nat Neurosci. 2014;17:667–669. doi: 10.1038/nn.3695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dodd, J. M. et al. Limiting weight gain in overweight and obese women during pregnancy to improve health outcomes: the LIMIT randomised controlled trial. BMC Pregnancy and Childbirth Oct 26; 11:79 (2011). [DOI] [PMC free article] [PubMed]
- 41.Dodd JM, et al. The effect of antenatal lifestyle advice for women who are overweight or obese on neonatal health: the LIMIT randomised trial. BMC Med. 2014;12:163. doi: 10.1186/s12916-014-0163-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Deierlein A, Thornton J, Hull H, Gallagher D. An anthropomettric model to estimate neonatal fat mass using air displacement pelthysmography. Nutrition & Metabolism. 2012;9:21. doi: 10.1186/1743-7075-9-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bender R, Lange S. Adjusting for multiple testing – when and how? Journal of Clinical Epidemiology. 2001;54:343–9. doi: 10.1016/S0895-4356(00)00314-0. [DOI] [PubMed] [Google Scholar]
- 44.Schulz K, Grimes DA. Multiplicity in randomised trials I: endpoints and treatments. Lancet. 2005;365:1591–1595. doi: 10.1016/S0140-6736(05)66461-6. [DOI] [PubMed] [Google Scholar]


