Abstract
Xanthomonas oryzae pv. oryzicola (Xoc) and X. oryzae pv. oryzae (Xoo) cause bacterial leaf streak (BLS) and bacterial leaf blight (BLB) in rice, respectively. Unlike Xoo, endogenous avirulence-resistance (avr-R) gene interactions have not been identified in the Xoc-rice pathosystem; however, both pathogens possess transcription activator-like effectors (TALEs) that are known to modulate R or S genes in rice. The transfer of individual tal genes from Xoc RS105 (hypervirulent) into Xoc YNB0-17 (hypovirulent) led to the identification of tal7, which suppressed avrXa7-Xa7 mediated defense in rice containing an Xa7 R gene. Mobility shift and microscale thermophoresis assays showed that Tal7 bound two EBE sites in the promoters of two rice genes, Os09g29100 and Os12g42970, which encode predicted Cyclin-D4-1 and GATA zinc finger family protein, respectively. Assays using designer TALEs and a TALE-free strain of Xoo revealed that Os09g29100 was the biologically relevant target of Tal7. Tal7 activates the expression of rice gene Os09g29100 that suppresses avrXa7-Xa7 mediated defense in Rice. TALEN editing of the Tal7-binding site in the Os09g29100 gene promoter further enhanced resistance to the pathogen Xoc RS105. The suppression of effector-trigger immunity (ETI) is a phenomenon that may contribute to the scarcity of BLS resistant cultivars.
Introduction
Xanthomonas oryzae pv. oryzicola (Xoc) causes bacterial leaf streak (BLS) in rice1, 2, a disease that is endemic in rice-growing regions worldwide. In China, BLS is more devastating than bacterial leaf blight (BLB) of rice caused by X. oryzae pv. oryzae (Xoo). BLS routinely reduces yield by 10–20% and causes yield losses up to 40%3, 4. Theoretically, the most effective way to control BLS is to grow resistant rice varieties; however, with the exception of Rxo1 from maize5, only Xo1 was reported early in 2016 as a source of resistance to African Xoc strain6. Very little novel host resistance (R) genes for BLS have been reported.
Xoc and Xoo show over 87% similarity when compared by DNA-DNA hybridization2. Despite their relatedness, the two pathovars show distinctly different infection strategies; Xoc colonizes the apoplast of mesophyll parenchyma cells, whereas Xoo invades the xylem systemically7. The genome sequences of Xoc and Xoo strains revealed the conservation of many pathogenicity and virulence genes between these two pathogens, such as two Xoc strains BLS256 (CP003057) and RS105 (CP011961) and three Xoo strains KACC10331 (AE013598), MAFF311018 (AP008229), and PXO99A (CP000967). Both pathovars contain similar genes for exopolysaccharide (EPS) synthesis, the regulation of pathogenicity factors (rpf), two-component signal transduction systems, the type III secretion system (T3SS), AvrBs3/PthA family proteins that function as transcription activator-like effectors (TALEs), and other T3S effectors (T3SEs) that are translocated into host cells8. Genomic studies have proven helpful in understanding the tissue- and host-specificity differences between these two pathovars9.
The T3SEs in X. oryzae have been classified into two groups: TALEs and Non-TAL effectors (NTALEs, also nominated as Xops (Xanthomonas outer protein))10. Many NTALEs share similar features including co-regulated expression with hrp genes11, conserved N-terminal aminoacid motifs12, 13, and a conserved plant-inducible promoter that is bound by HrpX14, 15. Novel NTALEs presumably contribute to host specificity and inhibit pathogen-triggered immunity16.
The first reported TALE was AvrBs3, which was discovered in X. campestris pv. vesicatoria and triggered HR in pepper cultivars containing an R gene Bs3 17, 18. AvrBs3 was shown to function as a transcription factor and bound to the promoter region of Bs3 19, 20. All TALEs in X. oryzae are members of the avrBs3/pthA family17, 21–26, which share several features including a central repeat region containing nearly identical repeats of 34–35 amino acids. The number of repeats varies in and across strains and contributes to host specificity through repeat variable diresidues (RVDs) that recognize unique DNA codes in plants20, 27–31. AvrXa7, AvrXa10, and AvrXa27 are typical TALEs that elicit BLB resistance in hosts that contain the cognate R genes, Xa7, Xa10 and Xa27, respectively22, 24, 32–35. However, interacting TALEs and cognate R genes have not been identified in the Xoc-rice pathosystem. Intriguingly, when avrXa7 and avrXa10 of Xoo strain PXO86 were transferred into Xoc BLS303 and BLS256, a compatible, water-soaked response was observed in rice cultivars containing the R genes Xa7 and Xa10, respectively36, suggesting that Xoc might encode unidentified effectors that interfere with effector-triggered immunity (ETI) which might help explain the lack of effective R genes for resistance to BLS36.
In this study, we isolated Xoc YNB0-17, a hypovirulent strain containing nine putative tal genes, which is considerably smaller than the 24 tal genes identified in Xoc RS105. The heterologous expression of avrXa7 in Xoc YNB0-17 but not in Xoc RS105, triggered ETI in rice plants containing Xa7. This led us to investigate whether RS105 contains unidentified effectors that enable the pathogen to suppress avr-R mediated immunity and cause disease in rice. The transfer of individual tal genes from Xoc RS105 into YNB0-17 led to the identification of tal7, which suppressed avrXa7-Xa7 mediated defense. Furthermore, gel shift assays and microscale thermophoresis (MST) indicated that Tal7 bound to Os09g29100 and Os12g42970 promoters in rice. These two genes encode predicted Cyclin-D4-1 and GATA zinc finger family protein, respectively. Functional assays confirmed that activation of cyclin-D4-1 could suppress AvrXa7-Xa7 mediated defense in rice.
Results
Heterologous expression of Xoo tal genes in Xoc YNB0-17 triggers ETI in rice
Multiple strains of Xoc were isolated and evaluated for virulence on rice cv. Nipponbarre. Xoc strain YNB0-17, which was originally isolated from Yuelianggu rice (data not shown), was hypovirulent strain on Nipponbare as compared to the highly virulent Xoc strain RS10537. Although endogenous avirulence-R gene interactions have not been identified in the Xoc-rice pathosystem, Xoc is known to contain TALEs that could potentially modulate R or S genes in rice38, 39. To investigate whether Xoo tal genes could trigger R-gene resistance in these strains, plasmids pHZWavrXa7 and pHZWavrXa27 (Supplemental Table S1), which contained avrXa7 and avrXa27, respectively, were transferred into YNB0-17 and RS105, respectively. YNB0-17(pHZWavrXa7) and YNB0-17(pHZWavrXa27) triggered an HR when infiltrated into leaves of rice cultivars IRBB7 and 87-15, which carry resistance genes Xa7 and Xa27, respectively (Fig. 1a). When the YNB0-17 derivatives were inoculated to rice cultivar IR24, typical water-soaked symptoms resulted, which is the predicted response since IR24 lacks the corresponding R genes (Fig. 1a). Xoc RS105(pHZWavrXa27) elicited an HR in rice cv. 87-15; however, inoculation with RS105(pHZWavrXa7) resulted in a susceptible (water-soaked) reaction in rice IRBB7 (Fig. 1a). Disease lesion lengths and growth curves of Xoc strains containing avrXa7 are shown in Fig. 1b and c. BLS lesion lengths and growth of YNB0-17(pHZWavrXa7) in IRBB7 rice was significantly reduced in comparison to YNB0-17 containing the empty vector pHM1 (Fig. 1b and c). Remarkably, BLS lesion size and bacterial growth of RS105(pHZWavrXa7) was not significantly different from RS105(pHM1) (Fig. 1b and c). Collectively, the results shown in Fig. 1 indicated that the expression of avrXa7 and avrXa27 in YNB0-17 triggered an HR and decreased bacterial growth (avrXa7) and BLS lesion length. However, the expression of avrXa7 in RS105 did not result in the HR and did not decrease bacterial growth in planta. These data suggest that like BLS256 and BLS303 strains36, RS105 has inhibitor(s) that may suppress avrXa7-Xa7 ETI, and the inhibitor(s) is (are) either absent or nonfunctional in YNB0-17.
Figure 1.
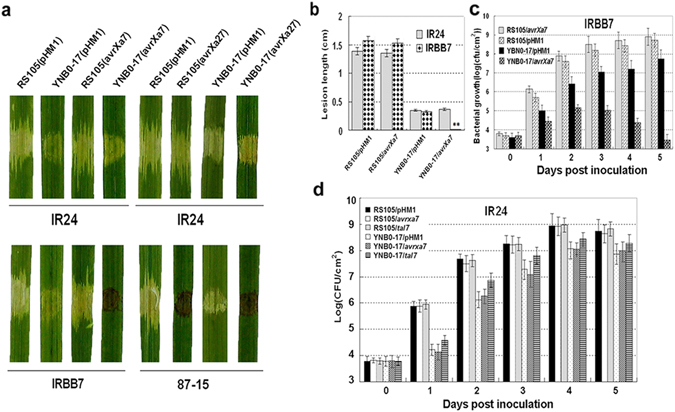
Heterologous expression of avrXa7 and avrXa27 in X. oryzae pv. oryzicola (Xoc) strains YNB0-17 and RS105 and inoculation to rice cv. IR24 (susceptible) and IRBB7 (contains R gene Xa7) and 87-15 (contains R gene Xa27). (a) YNB0-17 and RS105 containing avrXa7 and avrXa27 were infiltrated into seedlings of IRBB7 (Xa7) and 87-15 (Xa27) with needleless syringes. The susceptible rice cultivar IR24 and strains containing the empty vector pHM1 were used as controls. Leaves were scored for water-soaked symptoms or HRs within the infiltrated area 2 dpi and were designated as susceptible or resistant (showing an HR). Leaves were photographed 3 dpi. (b) Strains YNB0-17 and RS105 carrying avrXa7 were inoculated to adult rice plants of IRBB7 (Xa7) and susceptible line IR24. Lesion lengths were recorded 14 dpi. The vertical columns and intersecting bars represent the mean lesion length and SD from five replicate plants. The asterisks in each horizontal data column indicate significant differences at P = 0.01 using the Student’s t test. (c) Bacterial growth in rice IRBB7 inoculated with Xoc YNB0-17 and RS105 containing avrXa7 or the empty vector control (pHM1). (d) Bacterial growth of Xoc YNB0-17 and RS105 containing avrXa7, tal7 or pHM1 in the BLS susceptible rice line IR24. Leaf discs (0.8 cm diameter) were excised from the inoculated areas, homogenized in sterile water, diluted and plated on to NA. Data points represent the mean ± SD from three replicates. All experiments were repeated three times, and similar results were obtained.
X. oryzae pv. oryzicola Tal7 suppresses AvrXa7-Xa7 mediated immunity
Comparison of the Southern blot profiles of Xoc RS105 and YNB0-17 indicated that the main difference between the strains was the larger number of tal genes in RS105 (Supplemental Fig. 1). To identify whether RS105 contains a tal gene that suppresses AvrXa7-Xa7 defense, we isolated individual tal genes from RS105 based on Southern blot analysis (Supplemental Fig. 1); the isolated fragments containing putative tal genes were then cloned into pUFR034 containing avrXa7 (Supplemental Fig. 2). Remarkably, when tal7 was co-expressed with avrXa7 in strain YNB0-17, infiltrated tissue showed water-soaked symptoms indicative of a susceptible interaction (Fig. 2b). These results suggest that tal7 suppresses avrXa7-Xa7 resistance. The nuclear localization signals (NLS) and acidic transcriptional activation domains (AD) that are located at the C-terminus of TALEs and enable these proteins to function as transcription activators in plant nuclei22, 25. Thus, we speculated that a truncation at the C-terminus of Tal7 would impair its ability to suppress avrXa7-Xa7 immunity in rice. To confirm this, we generated truncated tal7∆NA (Fig. 2a), which lacks the NLS and AD domains (p707∆NA, Supplemental Table S1). The HR was not suppressed when rice IRBB7 was infiltrated with Xoc YNB0-17 containing tal7∆NA (Fig. 2b), indicating that suppression of avrXa7-Xa7 defense by Tal7 requires the NLS and AD domains. Immunoblotting experiments indicated that AvrXa7 was present in the supernatant (SN) and total extracts (TE) of YNB0-17 containing avrXa7, avrXa7 + tal7, and avrXa7 + tal7∆NA (Fig. 2c). Furthermore, the SN and TEs of YNB0-17 containing avrXa7 + tal7, and avrXa7 + tal7∆NA also contained Tal7 (Fig. 2d). The absence of AvrXa7 and Tal7 in the supernatant of the T3SS mutant, R∆hrcV (Fig. 2c and d), indicates that both effectors are delivered into plant cells via T3SS.
Figure 2.
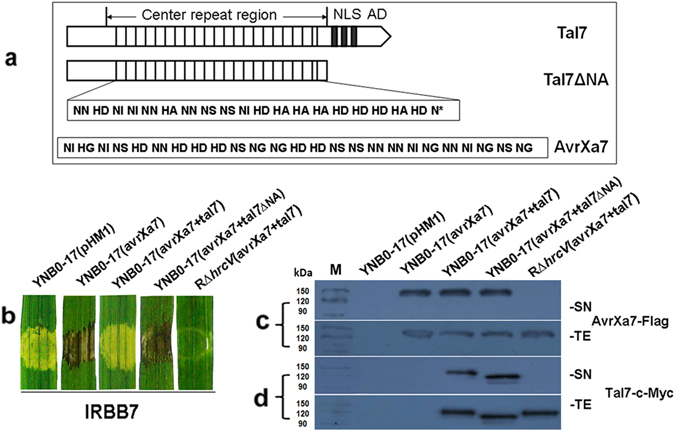
Tal7 suppresses avrXa7-Xa7 mediated defense. (a) Diagram showing the RVDs of Tal7 and AvrXa7 and schematic construction of Tal7∆NA. (b) Tal7 suppressesthe avrXa7-Xa7 interaction in rice. Strain YNB0-17 harboring pHM1, avrXa7, avrXa7 + tal7, and avrXa7 + tal7∆NA were infiltrated into rice seedlings of IRBB7(Xa7) with needleless syringes. The T3SS-deficient strain of RS105, R∆hrcV (Supplemental Table S1), containing avrXa7 + tal7, was used as a control. Leaves were scored for water-soaked symptoms or the HR at 3 dpi and were designated as susceptible or resistant, respectively. Leaves were photographed 3 dpi. (c) Secretion of AvrXa7-Flag and (d) Tal7-c-Myc by strain YNB0-17 harboring the constructs mentioned in panel b. Bacterial cells containing p707 or p707ΔNA (Supplemental Table S1) were induced in XOM3 medium as described in Methods. Bacterial supernatants (SN) and total extracts (TE) were analyzed by SDS-PAGE, transferred to polyvinylidene difluoride membranes, and used for immunoblotting with anti-FLAG (c) or anti-C-Myc (d) as the primary antibody. The experiments were repeated three times, and similar results were obtained each time.
Tal7 targets rice genes Os09g29100 and Os12g42970
Cernadas et al.38 demonstrated that up-regulation of Os01g31220, Os02g14770, Os07g47790, Os09g29100 and Os12g42970 genes depends on Tal7 of BLS256 (in that paper tal7 was nominated as tal6). Two of these genes, Os09g29100 and Os12g42970, which encode predicted Cyclin-D4-1 and GATA zinc finger family protein, respectively, are among the most highly-induced genes when YNB0-17(tal7) were infiltrated into IR24 rice (Fig. 3a). Os09g29100 expression was 8-fold (12 hpi) and 5-fold (24 hpi) higher than the control (YNB0-17) after Xoc YNB0-17(tal7) was infiltrated to IR24 rice; whereas Os12g42970 expression was 6.5- and 4.5-fold higher at 12 hpi and 24 hpi, respectively (Fig. 3a). These data suggest that Os09g29100 and Os12g42970 are possibly the Tal7 targets and others are not.
Figure 3.
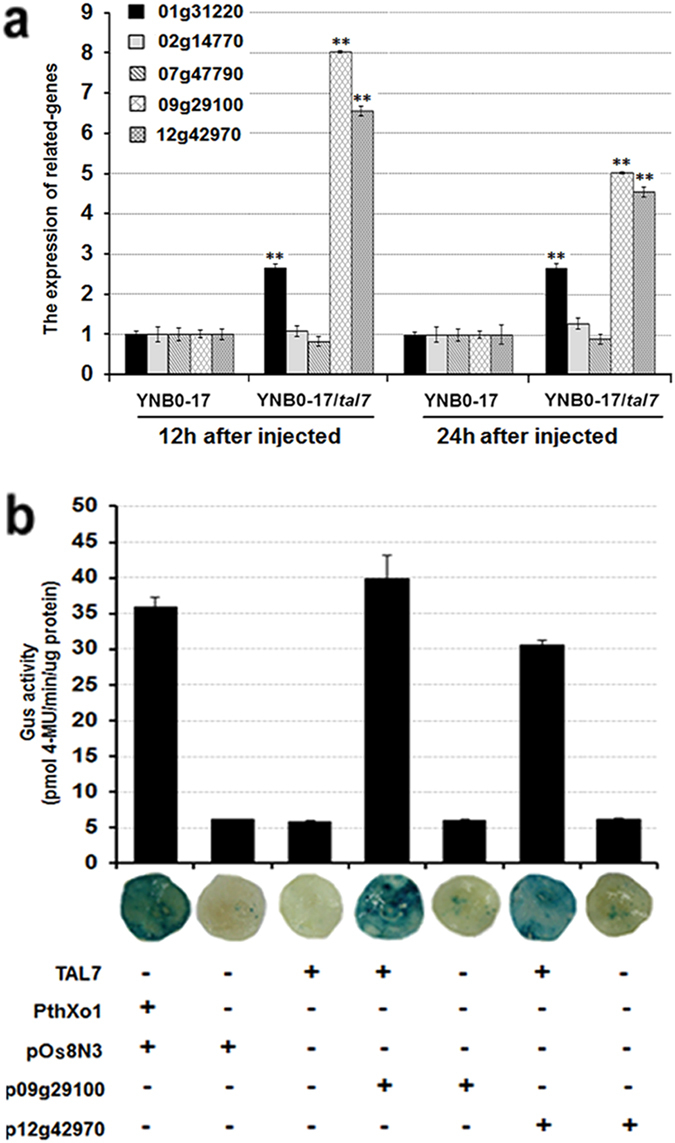
Xoc Tal7 targets rice genes Os09g29100 and Os12g42970. (a) The expression of candidate targets of Tal7. Xoc strains YNB0-17, and YNB0-17(tal7) were infiltrated into IR24 rice, and the expression of five rice genes (Os01g31220, Os02g14770, Os07g47790, Os09g29100, and Os12g42970) was measured 12 and 24 hpi by real time qRT-PCR. The expression levels of Actin and 18S rRNA used as internal standards. The asterisks in each horizontal data column indicate significant differences at P = 0.01 using the Student’s t test. Data are the mean ± SD of triplicate measurements from a representative experiment; and similar results were obtained in two other independent experiments. (b) Transcriptional activation of rice genes Os09g29100 and Os12g42970 by TALEs of Xoc Tal7. The TALE PthXo1 from Xoo and its target rice gene, Os8N3 (pOs8N3), were used as a positive control. Reporter fusions containing the rice promoters fused to GUS were codelivered via A. tumefaciens into N. benthamiana with (+) and without (−) constructs encoding TAL7 and PthXo1. pOs8N3, p09G29100 and p12g42970 represent the Os8N3, 09g29100 and 12g42970 promoters fused to GUS (see Methods). For quantitative assays, two leaf discs (0.9 cm diameter) were sampled 2 dpi, and GUS activity was determined using 4-methyl-umbelliferyl-β-D-glucuronide (MUG). 4-MU is 4-methyl-umbelliferone. Error bars indicate standard deviations (n = 3 samples from different plants). For qualitative assays, GUS activity in excised leaf discs was determined 3 dpi with X-Gluc (5-bromo-4-chloro-3-indolyl-b-D-glucuronide). A blue color indicatesa positive reaction. All experiments were performed twice with similar results.
To validate that Tal7 activates the expression of selected rice genes, 500-bp fragments containing the promoter regions of Os09g29100 and Os12g42970 were fused to the promoterless gusA gene and cloned in pCAMBIA1381 (Supplemental Table S1, p09g29100 and p12g42970). Agrobacterium strains containing GUS reporter constructs and tal genes were co-transformed into N. benthamiana leaves (see Methods). The Os8N3 promoter fused to gusA (Supplemental Table S1, pOs8N3) and a tal gene pthXo1 were included as controls (Fig. 3b). Tal7 triggered Os09g29100- and Os12g42970-mediated GUS activity. Quantitative assays indicated that GUS activation was higher for the Os09g29100 promoter (Fig. 3b), potentially due to weaker effector binding or/and reduced activation of the GATA zinc finger family protein gene promoter.
Tal7 binds EBEs of rice genes Os09g29100 and Os12g42970
As described above, Tal7 induced expression of Os09g29100 and Os12g42970. Thus, it was tempting to speculate that Tal7 targets the promoter regions of the two genes in rice. We then examined the Os09g29100 and Os12g42970 promoters to validate the predicted Tal7 binding site (EBEtal7). By examining the RVDs of Tal7 in the context of the TALE recognition code, which governs the interaction between TALE RVDs and their target nucleotides20, 27, we could visually corroborate the predicted EBEs for Tal7 in the Os09g29100 and Os12g42970 promoter regions using the TALE-NT program (http://tale-nt.cac.cornell.edu/, Supplemental Fig. 3a,b). The two promoter regions each contained a similar sequence, and the predicted binding sequence was identified with most of the identical nucleotides in the 5′ region (Supplemental Fig. 3a,b).
Potential binding of Tal7 to the candidate EBEtal7 sequences was investigated by EMSA using His-Tal7 fusion proteins and biotin-labeled Os09g29100 and Os12g42970 promoter fragments (Supplemental Fig. 3a,b). These assays showed that Tal7 bound to fragments 09g29100A and 12g42970A, but not 09g29100B and 12g42970B (Fig. 4a). The specificity of binding was further confirmed by competition assays with labeled and unlabeled (0, 5X, 20X, 100X) 09g29100A and 12g42970A fragments (Fig. 4b). These results indicate that Tal7 bind Os09g29100 and Os12g42970 promoters at the EBEtal7 location (fragments 09g29100A and 12g42970A, Fig. 4a).
Figure 4.
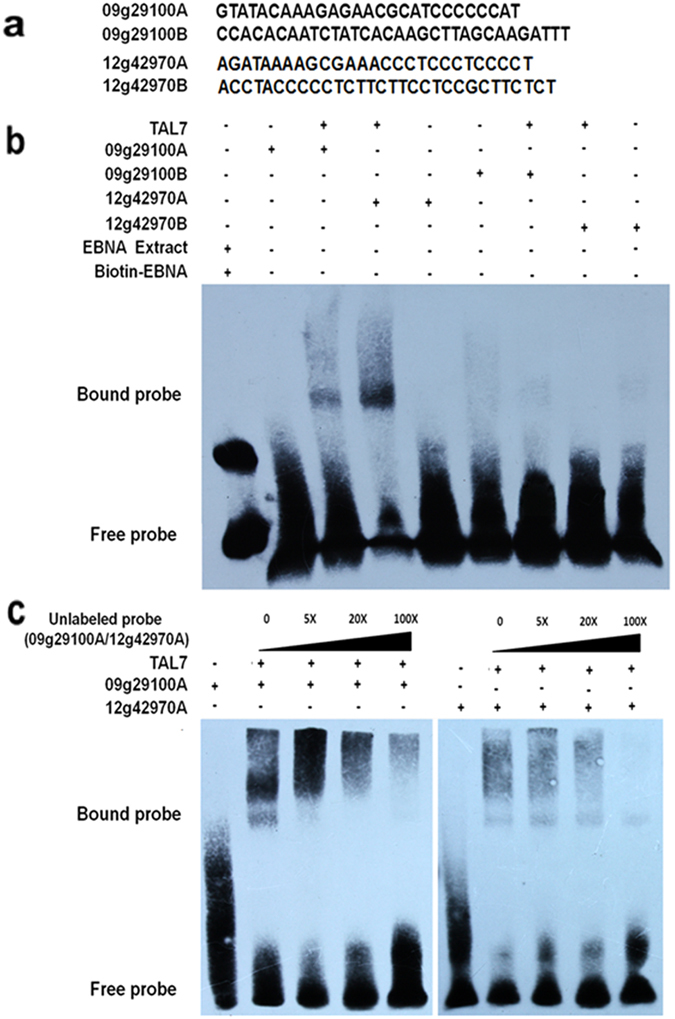
Xoc Tal7 binds the EBEs of the Os09g29100 and Os12g42970 promoter regions. Electromobility shift assays (EMSA) were performed using 20 fM biotin-labeled DNA fragments derived from the two promoter regions as probes. Unlabeled probes were used as competitor DNA. The presence of DNA or protein is indicated by (+), and absence by (−). (a) The DNA sequences of the four probes for EMSA, 09g29100A, 09g29100B, 12g42970A and 12g42970B, are shown at the top of the panel. (b) EMSA results for the probes from Os09g29100 and Os12g42970. EBNA extract protein and biotin-EBNA coming from EMSA Kit (Thermo, USA) used as positive contral. (c) Competition of the biotinylated probes with unlabeled incubated with His-Tal7. Unlabeled probe 09g29100A and 12g42970A were incubated at increasingly higher concentrations; e.g. 5, 20, and 100 X times more than the 20 fM of biotinylated 09g29100A and 12g42970A, respectively. The experiments were repeated two or more times with similar results.
MST (Supplemental Methods S3) supported the EMSA data and indicated that Tal7 interacted with the Os09g29100 and Os12g42970 promoter regions (Supplemental Fig. 4). The Kd values for the Tal7-09g29100 and Tal7-12g42970 interactions were 2.26 and 2.926 µM (Supplemental Fig. 4). AvrXa7 showed a binding affinity for Os11N3, which was used as a positive control (Supplemental Fig. 4) and Tal7 binding affinity for Os11N3 was used as a negative control (Supplemental Fig. 4).
Activation of Os09g29100 results in the suppression of AvrXa7-Xa7 ETI
Xoc strain RS105, containing tal7 (genomic copy) and avrXa7 (in trans), failed to elicit an HR in IRBB7 rice containing the R gene Xa7 (Fig. 1a). This led us to speculate that Tal7 was responsible for suppressing AvrXa7-Xa7 ETI. The EMSA and MST experiments both indicated that Tal7 bind the EBEs in the promoters of Os09g29100 and Os12g42970 (Fig. 4 and Supplemental Fig. 4); consequently, experiments were designed to determine if the Tal7-EBEtal7 interaction might induce target genes expression and then suppress AvrXa7-Xa7 ETI.
To evaluate which rice gene is the biologically relevant, we engineered two designer TAL effectors (dTALEs), dtal2-8 and dtal3-3 (Supplemental Table S1), to specifically bind Os09g29100 and Os12g42970 promoter regions outside EBEtal7 loci (Supplemental Fig. 3a,b). To investigate whether the two dTALEs can also suppress AvrXa7-Xa7 ETI in IRBB7 rice, the constructs for co-expression of dtal2-8 or dtal3-3 with avrXa7 were transferred into YNB0-17 and infiltrated rice tissue showed only dtal2-8 suppressed Xa7 defense (data not shown). To exclude possible interference of other TALEs in AvrXa7-Xa7 defense, an Xoo tal-free strain PH40 of PXO99A were transferred with the plasmids pHZWavrXa7, pHZWtal7, pHZWdtal2-8 and pHZWdtal3-3 containing avrXa7, tal7, dtal2-8 and dtal3-3, respectively and then were inoculated in rice with needleless syringes. First, Os09g29100 and Os12g42970 gene expression levels in IR24 rice were evaluated after PH, PH(avrXa7), PH(tal7), PH(dtal2-8) and PH(dtal3-3) were infiltrated at 0, 12, and 24 h. Real-time qPCR assays indicated that dtal2-8 and dtal3-3 specifically and significantly activated the expression of Os09g29100 and Os12g42970 genes, respectively, beginning 12 hpi (Fig. 5a). Functional assays indicated that only dtal3-3 suppressed AvrXa7-Xa7 ETI in a manner analogous to tal7 (Fig. 5b). Suppression of AvrXa7-Xa7 ETI was only observed when dtal3-3 and avrXa7 were co-expressed (e.g. PH/dtal3-3 + PH/avrXa7; Fig. 5b). Collectively, these data indicate that only Os09g29100, which encodes Cyclin-D4-1, is the relevant Tal7 target, and the activation of cyclin-D4-1 may suppress AvrXa7-Xa7 defense in IRBB7 rice.
Figure 5.
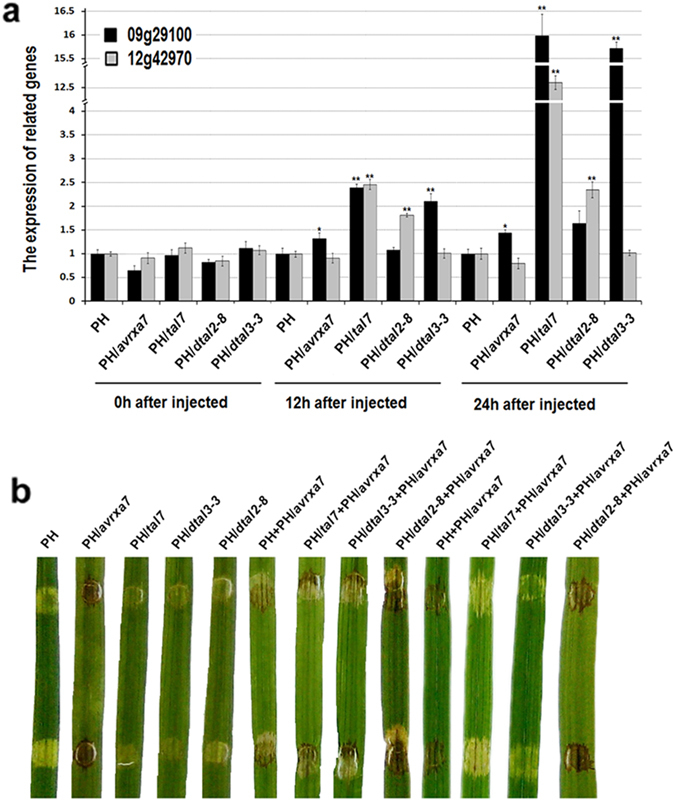
Os09g29100 is a major susceptibility gene in rice that can mask or suppress AvrXa7-Xa7 ETI. Strain PH, a tal-free strain of Xoo (Supplemental Table S1) was used in these experiments. (a) Expression of rice genes Os09g29100 and Os12g42970 in planta by real-time qRT-PCR. Xoo strain PH, PH/avrXa7, PH/tal7, PH/dtal3-3 and PH/dtal2-8 were infiltrated into IR24 rice leaves, and expression of Os09g29100 and Os12g42970 was evaluated 0, 12 and 24 hpi. Data are the mean ± SD of triplicate measurements from a representative experiment, and similar results were obtained in two other independent experiments. The asterisks in each column indicate significant differences at P = 0.01 by t test. (b) Role of Tal7 and two dTALEs in suppressing avrXa7-Xa7 ETI. Xoo strain PH and PH harboring avrXa7, tal7, dtal3-3 or dtal2-8 were infiltrated into rice seedlings of IRBB7 (Xa7) with needleless syringes. Images show inoculation with a single strain (five panels on left), one strain followed 3 h later with another strain (four middle panels), and two co-infiltrated strains (four panels at right). Leaves were scored for water-soaked symptoms or the HR within the infiltrated area 3 dpi and were designated as susceptible or resistant (showing an HR). Leaves were photographed 3 dpi.
TALEN-mediated disruption of EBEtal7 in cyclin-D4-1
A pair of TALENs was used to induce mutations in EBEtal7 of rice gene Os09g29100. The TALEN pair was designed to recognize sequences on both sides of EBE tal7 (Supplemental Fig. 5). After failure in obtaining transgenic plants from IRBB7, forty transgenic plants (T0) from the donor rice Nipponbare were obtained after transformation with pCAMBIA1301-EBEtal7 and grown to maturity. Seeds (T1, n = 145) were cultivated and leaves were harvested for analysis of potential mutations in EBEtal7. Digestion with TE7I to detect TALEN-mediated mutations41 and sequence analysis confirmed the existence of six TALEN-edited lines, which were designated A97-1, A97-2, A97-3, A97-4, and A97-5, and A97-6 (Fig. 6a). There were no differences in resistance phenotypes of the six T1 transgenic lines (data not shown). Consequently, line A97-4 was used for further study since more seed were obtained from this line.
Figure 6.
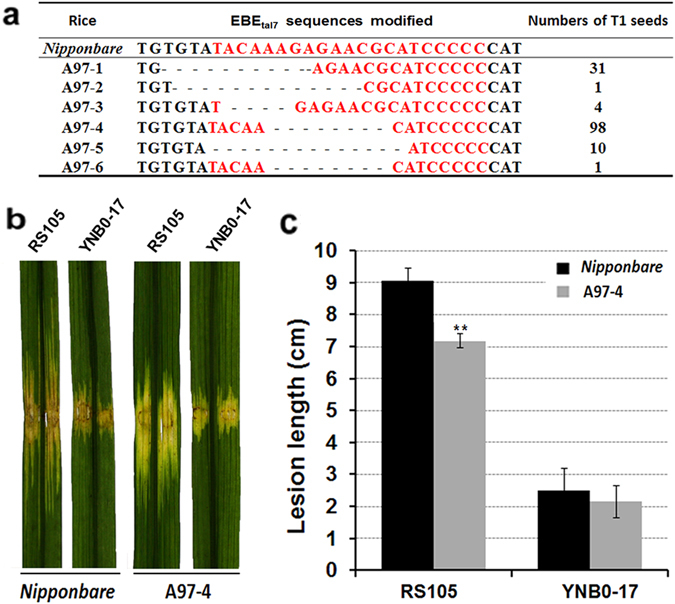
TALEN-modified editing of EBEtal7 in rice gene Os09g29100. (a) Colored letters indicate the nucleotide preference of Tal7 according to the TALE code. Letter height represents the preferences relative to other nucleotides for the RVD (generated with TALGetter: http://galaxy.informatik.uni-halle.de/root?tool_id=TALgetter). The RVDs of Tal7 DNA-binding domain and nucleotide sequence of the EBEtal7 region in Os09g29100 are shown. The table shows the EBEtal7 sequences in Nipponbare and six TALEN-edited lines (A97-1, A97-2, A97-3, A97-4, A97-5, and A97-6). The EBEtal7 sequence is shown in red font; the dashed lines (−) indicate deletions obtained from TALEN editing. The number of T1 seeds obtained from each line is indicated. (b) Inoculation of Xoc RS105 (hypervirulent) and YNB0-17 (hypovirulent) to rice cv. Nipponbare and transgenic line A97-4 containing the TALEN-modified EBEtal7 in Os09g29100. Strains YNB0-17 and RS105 (OD600 = 0.5) were inoculated to adult rice plants with needleless syringes and photographed 14 dpi. (c) Disease lesion lengths on cv. Nipponbare and rice transgenic line A97-4 at 14 dpi. Column height represents mean lesion length and vertical bars show the ± SD from five replicate plants. All experiments were repeated three times, and similar results were obtained.
Xoc RS105 and YNB0-17 were inoculated to rice cv. Nipponbare and transgenic line A97-4 containing the TALEN-modified EBEtal7 in Os09g29100. The hypervirulent RS105 showed reduced symptom severity on transgenic line A97-4 as compared to Nipponbare, whereas the hypovirulent YNB0-17 did not (Fig. 6b and c), strongly suggesting that Os09g29100 is the target of Tal7 contributing to the BLS development.
Discussion
Xoc and Xoo show a high level of DNA/DNA similarity, but have very different infection strategies3, 42. Although both Xoc and Xoo contain TALEs, effector/R gene interactions have not been described in Xoc-rice pathosystem. In the present study, we focused on the hypervirulent Xoc strain RS105 and hypovirulent YNB0-17. The latter strain was isolated from rice cv. Yuelianggu in the remote mountainous regions of the Yunnan province and could be a progenitor of more highly-evolved Xoc strains. The Xoo avrXa7 and avrXa27 were transferred into Xoc YNB0-17 and RS105; as expected, YNB0-17 containing avrXa7 and avrXa27 elicited the HR in rice containing R genes Xa7 and Xa27, respectively (Fig. 1a). Xoc RS105 expressing avrXa27 also elicited an HR in Xa27 rice; however, a compatible, virulent phenotype was observed when RS105/avrXa7 was inoculated to Xa7 rice (Fig. 1a). These results are reminiscent of those reported previously where the expression of avrXa7 and avrXa10 in Xoc BLS256 and BLS303 failed to elicit Xa7 mediated defense36.
We speculated that unidentified TALEs or NTALEs exist in Xoc RS105 that enabled the strain to suppress ETI and cause disease in rice. Since Xoc YNB0-17 lacks avrRxo1 and xopO, which are present in RS105 (Supplemental Fig. 6a,b), we investigated whether these two effectors suppress ETI in YNB0-17 when co-expressed with avrXa7. The co-expression of avrRxo1 or xopO in YNB0-17/avrXa7 did not inhibit avrXa7-Xa7 mediated ETI (Supplemental Fig. 6c,d), which prompted us to undertake a more global search for unidentified TALE(s) in RS105. Individual tal genes were cloned from Xoc RS105 based on Southern blot experiments (Supplemental Fig. 1) and transferred into YNB0-17 individually. This approach led to the discovery of Tal7, which suppressed AvrXa7-Xa7 mediated immunity in YNB0-17 expressing avrXa7 (Fig. 2b). This leads us to postulate that more virulent Xoc strains like RS105 have acquired a larger, more diverse repertoire of tal genes so they can bypass, circumvent, or suppress ETI.
Cernadas et al.38 reported five rice genes, Os01g31220, Os02 g14770, Os07g47790, Os09g29100 and Os12 g42970, as predicted targets of Tal7. In our study, the expression of Os09g29100 and Os12g42970 were significantly up-regulated than other three when IR24 rice was infiltrated with YNB0-17/tal7 (Fig. 3a), suggesting that the two genes are Tal7 target genes in rice.
We then used the TALE-NT program43 to identify potential EBEs recognized by Tal7 (EBEtal7) in the promoters of Os09g29100 and Os12g42970. Four EBEtal7 sequences were found in the promoters of these two genes (Supplemental Fig. 3a,b) and were designated 09g29100a, 09g29100b, 12g42970a, and 12g42970b (Fig. 4a). Interestingly, Cernadas et al.38 also predicted two candidate EBEs for Tal7 in the promoter of Os12g42970 but not Os09g29100. In the currently study, EMSA indicated that Tal7 bound 09g29100a and12g42970a, but not 09g29100b or 12g42970b (Fig. 4a).
Although the results indicated that Tal7 bound the promoter regions of both 09g29100A and 12g42970A, it wasn’t clear which interaction was significant in the suppression of AvrXa7-Xa7 ETI. This was investigated by engineering dtal3-3 and dtal2-8 for specific activation of Os09g29100 and Os12g42970, respectively (Supplemental Fig. 3). The two dTALEs were introduced into Xanthomonas strains YNB0-17 and PH, and evaluated for suppression of AvrXa7-Xa7 ETI in IRBB7 rice. Functional assays indicated that only dtal3-3 suppressed AvrXa7-Xa7 ETI in a manner analogous to tal7 (Fig. 5b). More importantly, these results indicate that Os09g29100 is the biologically relevant target of Tal7 with respect to suppression of ETI; this is evident because dtal3-3 (but not dtal2-8) targets Os09g29100 promoter. This study further illustrates the discriminatory ability of dTALEs to target specific genes and assess biological relevance, which has been validated in other Xanthomonas-plant interactions38, 44, 45.
Numerous effectors are produced by microbial pathogens and many are known to promote disease by suppressing plant defense46. In the present study, we demonstrated that Tal7 from Xoc suppressed AvrXa7-Xa7 mediated defense. Tal7 exhibits the modular structure typical of TALEs (Fig. 2a) and contains highly conserved C-terminal domains with NLS and AD features; both the NLS and AD domains of TALEs are known to be required for activation of plant gene expression47. Correspondingly, a truncated form of Tal7 lacking the NLS and AD domains (Tal7ΔNA) failed to suppress avrXa7-Xa7 mediated immunity in Xa7 rice; instead, an HR (defense) was observed in Xa7 rice (Fig. 2b), indicating that transcriptional activation of the target rice gene Os09g29100 is required for suppression of the defense response. In this regard, Os09g29100, which encodes a Cyclin-D4-1 protein, may promote BLS development in rice, since the TALEN-modified rice in Os09g29100 promoter enhanced resistance to the hypervirulent RS105 containing tal7 (Fig. 6). This is consistent with the previous observation that the mutation in tal7 of BLS256 strain decreased Xoc virulence in rice Nipponbare where the Os09g29100 gene is present38. We assume that the presence of Tal7 in Xoc strains leads to rice parenchyma cells to produce more nutrients to fit Xoc proliferation, since Cyclin-D4-1 is a member of the cyclin protein family that is involved in regulating cell cycle progression and putatively bind to and activate protein kinases named cyclin-dependent kinases (CDKs)48. The activation of CDKs in rice may alter signal induction in HR induction in rice mediated by AvrXa7-Xa7. Nevertheless, this needs first the cloning of Xa7 from IRBB7 and the generation of mutants either in Cyclin-D4-1 or/and Xa7 in IRBB7 rice.
Several TALEs produced by the related pathogen Xoo are known to target rice susceptibility genes in the SWEET family variety of plant genes. SWEET susceptibility genes (e.g. Os8N3 and OS11N3) are known to export sugars from the plant cell, a process that supports bacterial proliferation30, 44, 45, 49. It is likely that the S genes in rice that are targeted by Xoc TALEs differ from those produced by Xoo 50. Recently, Cernadas et al.38 identified Tal2g, which is an Xoc TALE that targets an S gene OsSULTR3;6, a predicted sulfate transporter. Interestingly, Tal2g did not suppress avrX7-Xa7 defense in our investigation (data not shown).
Our results indicate that Tal7 activated Os09g29100 expression, leading to suppress avrXa7-Xa7 defence in rice (Figs 3a, 2b and 5). Multiple TALEs have been shown to bind to the promoters of S genes in rice; for example, overlapping EBEs have been identified in the Os11N3 and Os8N3 promoter regions30, 49–51. Our data indicate that genetic modification of the EBEtal7 binding site via TALEN editing could be deployed to reduce Tal7 binding, which could potentially reduce disease severity (Fig. 6b and c). This approach shows great promise in engineering rice cultivars with reduced susceptibility to X. oryzae 52–54.
Over 40 distinct BLB resistance genes have been identified from cultivated rice varieties and wild relatives, and many R genes have been used in breeding programs for disease control10, 55, 56. Interestingly, R genes resistant to Xoo infection are ineffective to Xoc, and several factors may explain this phenomenon. (i) Xoc lacks the cognate tal genes that interact with BLB R genes; for example, Xoc strains RS105 and YNB0-17 do not encode avrXa7 and avrXa27. (ii) Xoc has major TALEs activating S gene expression that differs from Xoo. (iii) Xoc TALEs may function to suppress BLB resistance, such as Tal7 in the present study. Continued efforts to decipher tal-gene functions will increase our understanding of BLS and BLB in rice and will enhance our ability to control these diseases via R genes.
Materials and Methods
Bacterial strains, plasmids and growth conditions
The bacterial strains and plasmids used in this study are listed in Supplemental Table S1. Escherichia coli strains were cultivated in Luria-Bertani medium57 at 37 °C. All X. oryzae strains were grown in nutrient agar or NB broth (NA without agar) at 28 °C58. Antibiotics were used at the following final concentrations as required: ampicillin, 100 μg ml−1; rifampicin, 75 μg ml−1; kanamycin, 25 μg ml−1; spectinomycin, 50 μg ml−1.
DNA manipulation
DNA isolations, restriction enzyme digestions, electroporation, PCR and Southern blots were performed according to standard procedures59. The primers used for PCR are listed in Supplemental Table S1. Fragments of tal genes were verified by sequence analysis and analyzed with Vector NTI software (http://www.invitrogen.com).
To express avrXa7 in X. oryzae strains, primers were utilized to amplify a 280-bp conserved promoter (tal-pF and tal-pR) (Supplemental Table S2) and a 210-bp terminal region (tal-tF and tal-tR) (Supplemental Table S2) from Xoo PXO99A. These regions are identical in tal loci in both Xoo and Xoc (data not shown). The amplified products were digested with Kpn I and BamH I and fused; the resulting fragment was amplified again using tal-pF and tal-tR and cloned into pMD18-T (Takara, Dalian, China). The BamH I fragment containing avrXa7, which was obtained from pHZWavrXa7 (Supplemental Table S1), was inserted into pMD18-T containing the fused fragments. The Kpn I fragment from the above constructs was transferred into vector pUFR034, generating pavrXa7 (Supplemental Table S1).
To isolate tal genes from RS105, genomic DNA was digested with BamH I and subjected to electrophoresis in 1.2% agarose gels (Supplemental Methods S1). The region containing tal genes was gel-purified and ligated into BamH I-digested pBluescriptII SK(−) for in situ hybridization, digestion and sequencing. tal genes of interest were then transferred into pavrXa7 to replace avrXa7 at BamH I sites. To identify a tal from RS105 strain that may suppress avrXa7-Xa7 defense, a vector was constructed using cluster 2 of tal genes in strain PXO99A 60 as the basic frame (Supplemental Fig. 1). Four primers pairs (tal-KHKF/tal-KHKR, tal-KHHF/tal-KHHR, tal-HKHF/tal-HKHR and tal-HKKF/tal-HKKR; Supplemental Table S2) were synthesized to amplify the sequences from pthXo1 and tal2a (Supplemental Fig. 2). Genomic DNA of PXO99A was used as template, and a Hind III site was created by a single nucleotide mutation (G → C) (Supplemental Fig. 2). The BamH I fragment of avrXa7 was inserted into pthXo1 and ligated into pMD18-T, thus assembling the KH1 and KH2 fragments. Later, single BamH I fragments of tal genes isolated from RS105 were individually introduced into the tal2a locus flanked by HK1 and HK2 (Supplemental Fig. 2). The new frame harboring avrXa7 and a single tal candidate from RS105 strain was generated by fusion at the Hind III site (Supplemental Fig. 2). The fusions were then transferred into pUFR034 at Kpn I sites, and the constructs were transformed into YNB0-17 for further investigation.
Plant assays
Nipponbare (Oryzae sativa sp. japonica) and IR24 are BLB- and BLS-susceptible lines. IRBB7 (Xa7) and 87-15 (Xa27) are rice lines carrying different resistance genes for BLB32, 50. These lines were used for assaying the response to avrXa7 and avrXa27 to monitor the virulence and avirulence of X. oryzae strains containing different tal genes. Plant experiments were conducted in the greenhouse at Shanghai Jiao Tong University. Rice plants were grown under 14 h light (30 °C)/10 h dark (25 °C) conditions. Bacterial cultures were grown overnight, washed twice, and resuspended to an OD600 = 0.6 in sterile distilled water for inoculation to rice. Four-week old rice seedlings were inoculated by infiltrating secondary leaves with a needleless syringe58. Water-soaked symptoms or an HR were recorded two days post inoculation (dpi). Virulence assays on adult rice plants (two months old) were performed by leaf-needling with bacterial suspensions at OD600 = 0.6, and lesion lengths were measured 14 dpi. Ten leaves were used for each strain in each experiment, and independent experiments were conducted at least three times.
Measurement of bacterial growth in rice
Bacterial suspensions (OD600 = 0.6) were infiltrated into secondary leaves of four-week old rice seedlings using needleless syringes. Three 0.8 cm diameter leaf discs were harvested with a cork borer from each area after infiltration. After sterilization in 20% sodium hypochlorite, 75% ethanol and sterile water, the discs were ground with a sterile mortar and pestle in 1 ml of distilled water, diluted, and plated to determine the cfu cm−2. Serial dilutions were spotted in triplicate on NA with appropriate antibiotics. Plates were incubated at 28 °C for 3–4 days until single colonies could be counted. The cfu cm−2 was estimated, and the standard deviation (SD) was calculated using colony counts from the three replicate spots of three samples taken at each time point. Experiments were repeated at least three times.
Immunoblotting assays
Immunoblotting with Flag and c-Myc labeled antibodies was used to detect the secretion of AvrXa7-Flag and Tal7-c-Myc by Xoc YNB0-17. AvrXa7 was cloned in frame with C-terminal 3X FLAG-tag epitopes, and Tal7 and Tal7∆NA were cloned with C-terminal 3X c-Myc-tag epitopes. YNB0-17 bacterial cells containing the C-terminally tagged tal genes in constructs p707 and p707ΔNA (Supplemental Table S1) were cultured in NB to the logarithmic phase. The harvested bacteria were washed twice, and the OD600 was adjusted to 2.0 with sterile distilled water. The bacterial suspension (1 ml) was added to 40 ml filter-sterilized XOM3 medium61 and incubated at 28 °C for 6 h. The medium was centrifuged to separate total extracts (TE) and supernatant fractions (SN), and secreted proteins in the supernatant were precipitated with 12.5% trichloroacetic acid62. Proteins were separated on 8% SDS-PAGE gels and transferred to polyvinylidene difluoride membranes for immunoblotting using anti-FLAG or anti-C-Myc (Transgene, Beijing, China) as the primary antibody. Primary antibodies were detected using goat anti-rabbit IgG (H + L) (Transgene) and visualized with the EasySee Western Kit (Transgene).
Real-time quantitative RT-PCR
Real-time quantitative PCR was conducted to evaluate gene expression in different culture conditions using the Applied Biosystems 7500 Real-time PCR System and SYBR Premix-Ex Taq (TaKaRa). The SV Total RNA Isolation System (Promega, USA) was used to isolate RNA from rice leaves inoculated with X. oryzae. cDNA fragments were synthesized using the Revert Aid First Strand cDNA Synthesis Kit (TaKaRa). The resulting first-strand cDNA was diluted to a final volume of 20 μl, and SYBR green-labeled PCR fragments were amplified using primers designed to amplify related genes (Supplemental Table S2). PCR conditions were as follows: denaturation at 95 °C for 30 s and 41 cycles of 95 °C for 5 s and 60 °C for 34 s. The expression levels of Actin and 18 S rRNA used as internal standards. The comparative threshold method was used to calculate the relative mRNA levels. qRT-PCR experiments were performed in two independent trials with three replicatesin each test.
GUS assays of TAL effector activity
To assay glucuronidase (GUS) reporter activity, Agrobacterium tumefaciens strains delivering TALEs and GUS reporter constructs were mixed 1:1 (OD600 = 0.8), and inoculated into 5–7 week old Nicotiana benthamiana leaves. For qualitative GUS assays, leaf discs were sampled 3 dpi, incubated in X-Gluc (5-bromo-4-chloro-3-indolyl-β-D-glucuronide)30, destained in ethanol, and dried on acetate foil sheets. For quantitative assays, two leaf discs (0.9 cm diameter) were sampled 2 dpi, and GUS activity was determined using 4-methyl-umbelliferyl-β-D-glucuronide (MUG). Proteins were quantified using Bradford assays. Data correspond to triplicate samples from different plants, and experiments were performed twice.
Electrophoretic mobility shift assays (EMSA)
Promoter fragments 09g29100A (27 bp) and 09g29100B (32 bp) were amplified from rice cv. Nipponbare genomic DNA using EMSA primers EMSA9g-F2/EMSA9g-R2 and EMSA9g-F1/EMSA9g-R1, respectively (Supplemental Table S2). Promoter fragments 12g42970A (27 bp) and 12g42970B (30 bp) were bond using EMSA12g-F2/EMSA12g-R2 and EMSA12g-F1/EMSA12g-R1, respectively, in the binding buffer. The bond dsDNA sequences were labeled with the Biotin 3′ End DNA Labeling Kit (Thermo, USA). EMSA was performed using protocols supplied with the Light Shift Chemoluminescent EMSA Kit (Thermo).
Recombinant His-Tal7 was purified from E. coli BL21(DE3) containing pETtal7 (Supplemental Table S1). Bacteria were cultured in LB medium, induced, and harvested as described previously9. Cells were sonicated, centrifuged and fusion proteins were purified using Ni-NTA HisBind Resin (Novagen). 1 μl purified fusion proteins (20 μM His-Tal7) was mixed with 18 μl of binding buffer and 1 μl 20 fM of biotin-labeled DNA. The mixtures were incubated at room temperature for 30 min. Samples were then loaded on 5% polyacrylamide gels in 0.5 X TBE buffer, pH 8.359. Gels were transferred to Hybond N+ membranes, and signals were detected by chemoluminescence according to the manufacturer’s instructions.
Designer TAL effectors
TAL Effector-Nucleotide Targeter 2.043 was used to design TAL effectors (dTALEs) that targeted 18-bp promoter regions in rice genes Os09g29100 and Os12g42970 (Supplemental Fig. 3). Four basic RVDs (NI, NG, NN, and HD) were used to recognize their respective target nucleotides (A, T, G and C) (Supplemental Fig. 3) described previously63. These RVDs were used to assemble the TAL effector central repeats based on the DNA sequence of the respective Os09g29100 and Os12g42970 promoter sites (dtal3-3 and dtal2-8, Supplemental Fig. 3). The central repeat regions of the dTALEs were synthesized by ViewSolid Biotechnology (Beijing, China) and cloned in pUAVPD64, resulting in pdTAL3-3 and pdTAL2-8 (Supplemental Table S1). These two plasmids were digested with Sph I to release the dTALEs and used to replace avrXa7 in pZWavrXa7 at the Sph I site, resulting in pZWdtal3-3 and pZWdtal2-8 (Supplemental Table S1). The T3S signal, NLS and ADdomains of AvrXa7 were preserved in the two dTALE constructs (Supplemental Fig. 3c). Plasmids pZWdtal3-3 and pZWdtal2-8 were then linked to vector pHM1 at the Hind III site, thus generating pHZWdtal3-3 and pHZWdtal2-8 (Supplemental Table S1); these constructs were transferred into X. oryzae and used in biological assays.
Design and assembly of TALENs targeting the EBEtal7 in Os09g29100
We used TALENs to edit the EBE binding site in the promoter region of Os09g29100. The TALEN targeter program (https://tale-nt.cac.cornell.edu) was used to select two Os09g29100 sequences as follows: left arm, 5′CCTACCCTCCACGCGGCT, and right arm, 5′GAGCAATGGGGGGAT (Supplemental Fig. 5b,c). The targeted TALENs were constructed by Sidansai Biotechnology (Shanghai, China) using the FastTALETM TALEN Assembly Kit and vectors pL20 and pR16, which contain FokI DNA cleavage domains, Nos termini, and nuclear localization signals65. The TALEN backbone vectors pL20 and pR16 contain ubiquitin and 35S promoters, respectively (Supplemental Fig. 5b,c). The two TALEN expression cassettes were excised from pL20 and pR16 and ligated into the Hind III and Sac I sites of plasmid pCAMBIA1301, resulting in pCAMBIA1301-EBEtal7 (Supplemental Fig. 5). The construct pCAMBIA1301-EBEtal7 was introduced into rice cv. Nipponbare by Agrobacterium-mediated transformation66, serviced by Wuhan Biorun Bio-Tech Co. Ltd. (Wuhan, China).
Detection of mutations in the Os09g29100 promoter
To detect and analyze mutations derived from TALEN editing, genomic DNA was extracted from T0 and T1 transgenic rice leaves and used for amplification of the Os09g29100 promoter region with primers A97-F/R (Supplemental Table S2). The PCR products were subjected to T7 Endonuclease I (T7EI) assays to detect mismatched nucleotides as follows: PCR products (a mixture from mutant and wild-type) were heated at 95 °C for 5 min, then cooled from 95 to 75 °C at 0.05 °C s−1, 75 to 16 °C at 0.1 °C s−1, and then maintained at 16 °C for 2 min. The denatured and reannealed PCR products were digested with T7 endonuclease I (New England BioLabs) for 30 min at 37 °C; they were then subjected to 2% agarose gel electrophoresis. Mutant alleles were confirmed by subcloning and sequencing. Nipponbare and selected TALEN-edited Homozygous lines were inoculated with Xoc RS105 and YNB0-17 and virulence was assessed by phenotype and lesion length.
Electronic supplementary material
Acknowledgements
We thank Dr. Bing Yang, Iowa State University, for providing pHZWavrXa7 and pHZWavrXa27, Dr. Chaozu He, Chinese Academy of Sciences, for providing a rice cultivar 87-15 that contains Xa27, and Dr. Carol L. Bender, Oklahoma State University, for critically reading the manuscript. This study is sported by the National Key Research and Development Program of China (2016YFD0100601) and the National Natural Science Foundation of China (U1302265, 31471742).
Author Contributions
G.C.: Conceiving and designing the experiments, writing the paper. Z.C.:Conceiving and designing the experiments. L.C.: Performing the experiments, writing the paper. Y.C.: Conceiving and analysis the data. Z.X.: Conceiving and analysis the data. W.M.: Contributing materials/analysis tools.M.Z.: Contributing materials/analysis tools.L.Z.: Conceiving and designing the experiments.
Competing Interests
The authors declare that they have no competing interests.
Footnotes
Electronic supplementary material
Supplementary information accompanies this paper at doi:10.1038/s41598-017-04800-8
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Zaiquan Cheng, Email: czquan-99@163.com.
Gongyou Chen, Email: gyouchen@sjtu.edu.cn.
References
- 1.Fang CT, et al. A comparison of the rice bacterial leaf blight organism with the bacterial leaf streak organism of rice and Leersia hexandra Swartz. Acta Microbiol Sinica. 1957;3:99–124. [Google Scholar]
- 2.Swings J, et al. Reclassification of the causal agents of bacterial blight Xanthomonas campestris pathovar oryzae and bacterial leaf streak Xanthomonas campestris pathovar oryzicola of rice as pathovars of Xanthomonas oryzae new species ex ishiyama 1922. sp. nov, nom. rev. Int J Syst Bacteriol. 1990;40:309–311. doi: 10.1099/00207713-40-3-309. [DOI] [Google Scholar]
- 3.Niño-Liu DO, Ronald PC, Bogdanove AJ. Xanthomonas oryzae pathovars: model pathogens of a model crop. Mol Plant Pathol. 2006;7:303–324. doi: 10.1111/j.1364-3703.2006.00344.x. [DOI] [PubMed] [Google Scholar]
- 4.He WA, et al. Research progress on rice resistance to bacterial leaf streak. J. Plant Genet Resour. 2010;11:116–119. [Google Scholar]
- 5.Zhao BY, et al. A maize resistance gene functions against bacterial streak disease in rice. Proc. Nati. Acad. Sci. USA. 2005;102:15383–15388. doi: 10.1073/pnas.0503023102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Triplett LR, et al. A resistance locus in the American heirloom rice variety Carolina Gold Select is triggered by TAL effectors with diverse predicted targets and is effective against African strains of Xanthomonas oryzae pv. oryzicola. The plant J. 2016;87:472–583. doi: 10.1111/tpj.13212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ou, S. H. Rice Disease. Kew, Surrey: Commonwealth Agricultural Bureau (1985).
- 8.Lu H, et al. Acquisition and evolution of plant pathogenesis-associated gene clusters and candidate determinants of tissue-specificity in Xanthomonas. PLoS One. 2008;3:e3828. doi: 10.1371/journal.pone.0003828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zou HS, et al. EcpA, an extracellular protease, is a specific virulence factor required by Xanthomonas oryzae pv. oryzicola but not by X. oryzae pv. oryzae in rice. Microbiol. 2012;58:2372–2383. doi: 10.1099/mic.0.059964-0. [DOI] [PubMed] [Google Scholar]
- 10.White FF, Potnis N, Jones JB, Koebnik R. The type III effectors of Xanthomonas. Mol Plant Pathol. 2009;10:749–766. doi: 10.1111/j.1364-3703.2009.00590.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tang X, Xiao Y, Zhou JM. Regulation of the type III secretion system in phytopathogenic bacterial. Mol. Plant Microbe Interact. 2006;19:1159–1166. doi: 10.1094/MPMI-19-1159. [DOI] [PubMed] [Google Scholar]
- 12.Song, C. F. & Yang, B. Mutagenesis of 18 type III effectors reveals virulence function of XopZPXO99 In Xanthomonas oryzae pv. oryzae. Mol. Plant Microbe Interact. 23, 893–902 (2010). [DOI] [PubMed]
- 13.Dean P. Functional domains and motifs of bacterial type III effector proteins and their roles in infection. FEMS Microbiol Rev. 2011;35:1100–1125. doi: 10.1111/j.1574-6976.2011.00271.x. [DOI] [PubMed] [Google Scholar]
- 14.Furutani A, et al. Identification of novel HrpXo regulons preceded by two cis-acting elements, a plant-inducible promoter box and a -10 box-like sequence, from the genome database of Xanthomonas oryzae pv. oryzae. FEMS Microbiol Lett. 2006;259:133–141. doi: 10.1111/j.1574-6968.2006.00265.x. [DOI] [PubMed] [Google Scholar]
- 15.Koebnik R, et al. Specific binding of the Xanthomonas campestris pv. vesicatoria AraC-type transcriptional activator HrpX to plant-inducible promoter boxes. J. Bacteriol. 2006;188:7652–7660. doi: 10.1128/JB.00795-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim JG, et al. Xanthomonas T3S effector XopN suppresses PAMP-triggered immunity and interacts with a tomato atypical receptor-like kinase and TFT1. Plant Cell. 2009;21:1305–1323. doi: 10.1105/tpc.108.063123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bonas U, Stall RE, Staskawicz B. Genetic and structural characterization of the avirulence gene avrBs3 from Xanthomonas campestris pv. vesicatoria. Mol Gen Genet. 1989;218:127–136. doi: 10.1007/BF00330575. [DOI] [PubMed] [Google Scholar]
- 18.Ackerveken GV, Marois E, Bonas U. Recognition of the-bacterial avirulence protein AvrBs3 occurs inside the host plant cell. Cell. 1996;87:1307–1316. doi: 10.1016/S0092-8674(00)81825-5. [DOI] [PubMed] [Google Scholar]
- 19.Römer P, et al. Plant pathogen recognition mediated by promoter activation of pepper. Science. 2007;318:645–648. doi: 10.1126/science.1144958. [DOI] [PubMed] [Google Scholar]
- 20.Boch J, et al. Breaking the code of DNA binding specificity of TAL-Type III effectors. Science. 2009;326:1509–1512. doi: 10.1126/science.1178811. [DOI] [PubMed] [Google Scholar]
- 21.Hopkins CM, et al. Identification of a family of avirulence genes from Xanthomonas oryzae pv. oryzae. Mol. Plant Microbe Interact. 1992;5:451–459. doi: 10.1094/MPMI-5-451. [DOI] [PubMed] [Google Scholar]
- 22.Zhu W, et al. AvrXa10 contains an acidic transcriptional activation domain in the functionally conserved C terminus. Mol. Plant Microbe Interact. 1998;11:824–832. doi: 10.1094/MPMI.1998.11.8.824. [DOI] [PubMed] [Google Scholar]
- 23.Bai J, et al. Xanthomonas oryzae pv. oryzaea, virulence genes contribute differently and specifically to pathogen aggressiveness. Mol. Plant Microbe Interact. 2000;13:1322–1329. doi: 10.1094/MPMI.2000.13.12.1322. [DOI] [PubMed] [Google Scholar]
- 24.Yang B, Zhu W, Johnson LB, White FF. The virulence factor AvrXa7 of Xanthomonas oryzae pv. oryzae is a type III secretion pathway-dependent nuclear-localized double-stranded DNA-binding protein. Proc. Nati. Acad. Sci. USA. 2000;97:9807–9812. doi: 10.1073/pnas.170286897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang B, Sugio A, White FF. Avoidance of host recognition by alterations in the repetitive and C-terminal regions of AvrXa7, a type III effector of Xanthomonas oryzae pv. oryzae. Mol. Plant Microbe Interact. 2005;18:142–149. doi: 10.1094/MPMI-18-0142. [DOI] [PubMed] [Google Scholar]
- 26.Yang B, White FF. Diverse members of the AvrBs3/PthA family of type III effectors are major virulence determinants in bacterial blight disease of rice. Mol. Plant Microbe Interact. 2004;17:1192–1200. doi: 10.1094/MPMI.2004.17.11.1192. [DOI] [PubMed] [Google Scholar]
- 27.Moscou MJ, Bogdanove AJ. A simple cipher governs DNA recognition by TAL effectors. Science. 2009;326:1501. doi: 10.1126/science.1178817. [DOI] [PubMed] [Google Scholar]
- 28.Römer P, Recht S, Lahaye T. A single plant resistance gene promoter engineered to recognize multiple TAL effectors from disparate pathogens. Proc. Nati. Acad. Sci. USA. 2009;106:20526–20531. doi: 10.1073/pnas.0908812106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Römer P, et al. Recognition of AvrBs3-like proteins is mediated by specific binding to promoters of matching pepper Bs3 alleles. Plant Physiol. 2009;150:1697–1712. doi: 10.1104/pp.109.139931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Antony G, et al. Rice xa13 recessive resistance to bacterial blight is defeated by induction of the disease susceptibility gene Os11N3. Plant Cell. 2010;22:3864–3876. doi: 10.1105/tpc.110.078964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mak AN, et al. The crystal structure of TAL effector PthXo1 bound to its DNA target. Science. 2012;335:716–19. doi: 10.1126/science.1216211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gu K, et al. R gene expression induced by a type III effector triggers disease in rice. Nature. 2005;435:1122–1125. doi: 10.1038/nature03630. [DOI] [PubMed] [Google Scholar]
- 33.Tian DS, Yin ZC. Constitutive heterologous expression of avrXa27 in rice containing the R gene Xa27 confers enhanced resistance to compatible Xanthomonas oryzae strains. Mol Plant Pathol. 2009;10:29–39. doi: 10.1111/j.1364-3703.2008.00509.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hummel AW, Doyle EL, Bogdanove AJ. Addition of transcription activator-like effector binding sites to a pathogen strain-specific rice bacterial blight resistance gene makes it effective against additional strains and against bacterial leaf streak. New Phytol. 2012;195:883–893. doi: 10.1111/j.1469-8137.2012.04216.x. [DOI] [PubMed] [Google Scholar]
- 35.Tian DS, et al. The rice TAL effector-dependent resistance protein Xa10 triggers cell death and calcium depletion in the endoplasmic reticulum. Plant Cell. 2014;26:497–515. doi: 10.1105/tpc.113.119255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Makino S, Sugio A, White FF, Bogdanove AJ. Inhibition of resistance gene-mediated defense in rice by Xanthomonas oryzae pv. oryzicola. Mol. Plant Microbe Interact. 2006;19:240–249. doi: 10.1094/MPMI-19-0240. [DOI] [PubMed] [Google Scholar]
- 37.Zou LF, et al. Elucidation of the hrp clusters of Xanthomonas oryzae pv. oryzicola that control the hypersensitive response in nonhost tobacco and pathogenicity in susceptible host rice. App Environ Microb. 2006;72:6212–6224. doi: 10.1128/AEM.00511-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cernadas RA, et al. Code-assisted discovery of TAL effector targets in bacterial leaf streak of rice reveals contrast with bacterial blight and a novel susceptibility gene. PLoS Pathog. 2014;10:e1003972. doi: 10.1371/journal.ppat.1003972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hummel, A. W. et al. A transcription activator-like effector from Xanthomonas oryzae pv. oryzicola elicits dose-dependent resistance in rice. Mol Plant Pathol. in press, doi:10.1111/mpp.12377 (2016). [DOI] [PMC free article] [PubMed]
- 40.Ji, Z. Y. et al. Interfering TAL effectors of Xanthomonas oryzae neutralize R-gene-mediated plant disease resistance. Nat. Commun. in press, doi:10.1038/ncomms13435 (2016). [DOI] [PMC free article] [PubMed]
- 41.Li T, Liu B, Chen CY, Yang B. TALEN utilization in rice genome modifications. Methods. 2014;69:9–16. doi: 10.1016/j.ymeth.2014.03.019. [DOI] [PubMed] [Google Scholar]
- 42.White FF, Yang B. Host and pathogen factors controlling the rice/Xanthomonas oryzae interaction. Plant Physiol. 2009;150:1677–1686. doi: 10.1104/pp.109.139360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Doyle, E. L. et al. TAL Effector-Nucleotide Targeter (TALE-NT) 2.0: tools for TAL effector design and target prediction. Nucleic Acids Res. 40, doi:10.1093/nar/gks608 (2012). [DOI] [PMC free article] [PubMed]
- 44.Streubel J, et al. Five phylogenetically close rice SWEET genes confer TAL effector-mediated susceptibility to Xanthomonas oryzae pv. oryzae. New Phytol. 2013;200:808–819. doi: 10.1111/nph.12411. [DOI] [PubMed] [Google Scholar]
- 45.Boch J, Bonas U, Lahaye T. TAL effectors–pathogen strategies and plant resistance engineering. New Phytol. 2014;204:823–832. doi: 10.1111/nph.13015. [DOI] [PubMed] [Google Scholar]
- 46.Dou D, Zhou JM. Phytopathogen effectors subverting host immunity: different foes, similar battleground. Cell Host Microbe. 2012;12:484–495. doi: 10.1016/j.chom.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 47.Richter A, Streubel J, Boch J. TAL effector DNA-binding principles and specificity. Methods in Mol Biol. 2016;1338:9–25. doi: 10.1007/978-1-4939-2932-0_2. [DOI] [PubMed] [Google Scholar]
- 48.Tamirisa S, Vudem DR, Khareedu VR. A Cyclin Dependent Kinase Regulatory Subunit (CKS) Gene of Pigeonpea Imparts Abiotic Stress Tolerance and Regulates Plant Growth and Development in Arabidopsis. Front Plant Sci. 2017;8:165. doi: 10.3389/fpls.2017.00165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yang B, Sugio A, White FF. Os8N3 is a host disease-susceptibility gene for bacterial blight of rice. Proc. Nati. Acad. Sci. USA. 2006;103:10503–10508. doi: 10.1073/pnas.0604088103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Verdier V, et al. Transcription activator-like (TAL) effectors targeting OsSWEET genes enhance virulence on diverse rice (Oryza sativa) varieties when expressed individually in a TAL effector-deficient strain of Xanthomonas oryzae. New Phytol. 2012;196:1197–1207. doi: 10.1111/j.1469-8137.2012.04367.x. [DOI] [PubMed] [Google Scholar]
- 51.Hutin M, et al. A knowledge-based molecular screen uncovers a broad spectrum OsSWEET14 resistance allele to bacterial blight from rice. The Plant J. 2015;84:694–703. doi: 10.1111/tpj.13042. [DOI] [PubMed] [Google Scholar]
- 52.Li T, et al. Modularly assembled designer TAL effector nucleases for targeted gene knockoutand gene replacement in eukaryotes. Nucleic Acids Res. 2011;39:6315–6325. doi: 10.1093/nar/gkr188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li T, et al. High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat. Biotechnol. 2012;30:390–392. doi: 10.1038/nbt.2199. [DOI] [PubMed] [Google Scholar]
- 54.Blanvillain-Baufumé, S. et al. Targeted promoter editing for rice resistance to Xanthomonas oryzae pv. oryzae reveals differential activities for SWEET14-inducing TAL effectors. Plant Biotech J. in press, doi:10.1111/pbi.12613 (2016). [DOI] [PMC free article] [PubMed]
- 55.Verdier V, Vera, Cruz. C, Leach JE. Controlling rice bacterial blight in Africa: needs and prospects. J. Biotechnol. 2012;159:320–328. doi: 10.1016/j.jbiotec.2011.09.020. [DOI] [PubMed] [Google Scholar]
- 56.Zhang H, Wang S. Rice versus Xanthomonas oryzae pv. oryzae: a unique pathosystem. Curr Opin Plant Biol. 2013;16:188–195. doi: 10.1016/j.pbi.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 57.Miller, J. H. Experiments in molecular genetics. Cold Spring Harb, NY: Cold Spring Harb. Laboratory (1972).
- 58.Li YR, et al. A novel regulatory role of HrpD6 in regulating hrp-hrc-hpa genes in Xanthomonas oryzae pv. oryzicola. Mol. Plant Microbe Interact. 2011;24:1086–1101. doi: 10.1094/MPMI-09-10-0205. [DOI] [PubMed] [Google Scholar]
- 59.Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular Cloning: A Laboratory Manual. Cold Spring Harb: Cold Spring Harb. Laboratory Press (1989).
- 60.Salzberg SL, et al. Genome sequence and rapid evolution of the rice pathogen Xanthomonas oryzae pv. oryzae PXO99A. BMC Genomics. 2008;9:204. doi: 10.1186/1471-2164-9-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Xiao YL, et al. Establishment of the hrp-inducing systems for the expression of the hrp genes of Xanthomonas oryzae pv. oryzicola. Acta Microbiol Sinica. 2007;47:396–401.60. [PubMed] [Google Scholar]
- 62.Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 63.Li T, Huang S, Zhou J, Yang B. Designer TAL effectors induce disease susceptibility and resistance to Xanthomonas oryzae pv. oryzae in rice. Mol Plant. 2013;6:781–789. doi: 10.1093/mp/sst034. [DOI] [PubMed] [Google Scholar]
- 64.Wu XM, Li YR, Zou LF, Chen GY. Gene-for-gene relationships between rice and diverse avrBs3/pthA avirulence genes in Xanthomonas oryzae pv. oryzae. Plant Pathol. 2007;56:26–34. [Google Scholar]
- 65.Zhao, J. et al. A one-step system for convenient and flexible assembly of transcription activator-like effector nucleases (TALENs). Mol Cells. in press, doi:10.14348/molcells.2016.0140 (2016). [DOI] [PMC free article] [PubMed]
- 66.Hiei Y, Komari T. Agrobacterium-mediated transformation of rice using immature embryos or calli induced from mature seed. Nat. Proto. 2008;3:824–834. doi: 10.1038/nprot.2008.46. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


