Abstract
BACKGROUND
Studies have reported that transcranial direct current stimulation (tDCS) can modulate human behaviors, symptoms, and neural activity; however, the neural effects during stimulation are unknown. Most studies compared the effects of tDCS before and after stimulation. The objective of our study was to measure the neurobiological effect of a single tDCS dose during stimulation.
METHODS
We conducted an online and offline protocol combining tDCS and magnetic resonance spectroscopy (MRS) in 17 healthy participants. We applied anodal tDCS over the left dorsolateral prefrontal cortex (DLPFC) and cathodal tDCS over the right DLPFC for 30 minutes, one of the most common montages used with tDCS. We collected MRS measurements in the left DLPFC and left striatum during tDCS and an additional MRS measurement in the left DLPFC immediately after the end of stimulation.
RESULTS
During stimulation, active tDCS, as compared with sham tDCS, elevated prefrontal N-acetylaspartate and striatal glutamate + glutamine but did not induce significant differences in prefrontal or striatal gamma-aminobutyric acid level. Immediately after stimulation, active tDCS, as compared with sham tDCS, did not significantly induce differences in glutamate + glutamine, N-acetylaspartate, or gamma-aminobutyric acid levels in the left DLPFC.
CONCLUSIONS
These observations indicate that tDCS over the DLPFC has fast excitatory effects, acting on prefrontal and striatal transmissions, and these effects are short lived. One may postulate that repeated sessions of tDCS might induce similar longer lasting effects of elevated prefrontal N-acetylaspartate and striatal glutamate + glutamine levels, which may contribute to its behavioral and clinical effects.
Keywords: Dorsolateral prefrontal cortex, Glx, Magnetic resonance spectroscopy, N-acetylaspartate, Striatum, Transcranial direct current stimulation
Studies have reported that transcranial direct current stimulation (tDCS) can noninvasively modulate human behaviors in vivo. Applied over the dorsolateral prefrontal cortex (DLPFC), tDCS can influence mood, emotional perception, and various cognitive processes including decision making (1), problem solving (2), and working memory (3) However, recent meta-analyses questioned some of these effects of tDCS (4,5) Some of these questions raised the issue that most studies have tested the effects of tDCS on behavioral and clinical outcomes. The neurophysiologic mechanisms of action of tDCS remain relatively unknown. The few studies that tested the effects of tDCS on neural outcomes used offline designs, measuring and comparing neural substrates before and after stimulation (6) Although these studies greatly contributed to elucidating the neural effects of tDCS, they were limited to reporting neural differences after tDCS delivery. There is still a need to demonstrate whether tDCS instantaneously changes neural substrates during stimulation. Such a demonstration would provide evidence that tDCS directly modulates the brain.
Characterization of the neural effects of tDCS is also important to further investigate the clinical potentials of tDCS (7,8) The possibility of modulating the brain and consequently inducing behavioral and cognitive changes indicates therapeutic potential for tDCS. Most studies modulating behaviors and cognition in healthy volunteers reported such effects with a single tDCS session, and therapeutic potential has mainly been reported when repetitive sessions of tDCS were applied over the DLPFC. Studies reported reduction of depressive symptoms in patients with major depressive disorders (9,10), positive symptoms in patients with schizophrenia (11), and craving in patients with substance use disorders (12) Again, how the brain is modulated when tDCS induces such clinical benefits is largely unknown. Better characterization of the neural effects of tDCS over the DLPFC would likely contribute to identifying optimal parameters to enhance clinical outcomes.
The goal of this study was to develop an online design using tDCS and magnetic resonance spectroscopy (MRS) to characterize the simultaneous and subsequent neurometabolism differences induced by tDCS using 1H MRS. Our hypotheses were that tDCS would 1) elevate glutamate + glutamine (Glx) levels in the left DLPFC (under the anode electrode) and left ventral striatum, 2) elevate N-acetylaspartate (NAA) levels in the left DLPFC, and 3) decrease gamma-aminobutyric acid (GABA) levels in the left DLPFC. Furthermore, it was hypothesized that these neurometabolic differences in the left DLPFC would be observed during and after delivery of tDCS. To test these hypotheses, we simultaneously delivered anodal and cathodal tDCS over the left and right DLPFC, respectively, and measured total Glx, GABA, and NAA concentrations in the left DLPFC and left striatum with MRS. We selected this electrode montage (anodal tDCS over the left DLPFC and cathodal tDCS over the right DLPFC) because these regions are the most targeted areas to modulate behaviors and cognition and to alleviate neuropsychiatric symptoms. We studied metabolites in the left DLPFC and left striatum because of the importance of corticostriatal fibers as connections within the forebrain and to probe potential subcortical effects of tDCS. We studied Glx and GABA because the effects of tDCS are primarily thought to be ascribable to local differences in cortical excitability, implicating glutamate (13) and GABA transmissions (14) We also measured NAA, a metabolite implicated in neuronal regulatory processes such as protein synthesis and lipid production (15) and an indicator of neuronal viability and metabolism activity (16) Finally, we also focused on these neurotransmitters because they have been shown in numerous articles to be affected in the aforementioned pathologic conditions (17) in which tDCS has shown some clinical potential.
METHODS AND MATERIALS
This study used a randomized, crossover, sham-controlled, blinded at three levels design (participant, MRS experimenter, data analysis conductor) Each participant took part in two experimental sessions: one with active tDCS and one with sham tDCS. The order of the tDCS sessions was randomized with a Latin square (eight participants received active tDCS first and sham tDCS second) Sessions were separated by 7 days to minimize potential carryover effects of tDCS.
Participants
We recruited 17 healthy participants through the electronic mail distribution service of Université Laval. The local institutional review board committee (Institut de Réadaptation en Déficience Physique de Québec) approved the study. We obtained informed written consent from all participants and screened them for neurologic, medical, and psychiatric conditions. Two participants demonstrated moving artifacts during scanning and were omitted from further analysis. The remaining 15 participants (eight men) had an average age of 27 years (range, 21–41 years) and were right-handed as assessed by the Edinburgh Handedness Inventory. Table 1 summarizes participant characteristics.
Table 1.
Participant Characteristics
| Participant No. | Sex | Age, Years | Handednessa |
|---|---|---|---|
| 1 | Male | 31 | 80 |
| 2 | Female | 27 | 50 |
| 3 | Male | 29 | 100 |
| 4 | Male | 22 | 80 |
| 5 | Female | 41 | 100 |
| 6 | Female | 27 | 100 |
| 7 | Male | 23 | 90 |
| 8 | Male | 23 | 100 |
| 9 | Female | 24 | 50 |
| 10 | Female | 23 | 100 |
| 11 | Female | 28 | 60 |
| 12 | Female | 21 | 88 |
| 13 | Male | 28 | 78 |
| 14 | Male | 26 | 100 |
| 15 | Male | 33 | 100 |
Scores on the Edinburgh Handedness Inventory.
tDCS Parameters
We delivered stimulation using a magnetic resonance imaging–compatible DC-STIMULATOR (neuroConn GmbH, Ilmenau, Germany) We placed the anode electrode over the left DLPFC (F3) and the cathode electrode over the right DLPFC (F4) using the electroencephalography 10–20 system. We used 35-mm2 electrodes, and electrode positioning was verified on T1-weighted scan. Active stimulation was delivered for 30 minutes at a current intensity of 1 mA. Sham stimulation was delivered for 30 minutes following standard procedure with a ramp up and a ramp down of 30 seconds with the remaining time with no active current (18) Participants and the MRS experimenter completed a questionnaire on the stimulation conditions for each session to test the integrity of blinding at the end of the study. Of 15 participants, 11 guessed which tDCS session (active or sham) was conducted with a confidence level of 55% determined on a visual analog scale. The MRS experimenter (AH-B) had minimal interaction with the participants and remained fully blinded to the tDCS conditions (active vs. sham; delivered by SF), with a confidence level of 100%, until the interpretation of results.
tDCS and MRS Design
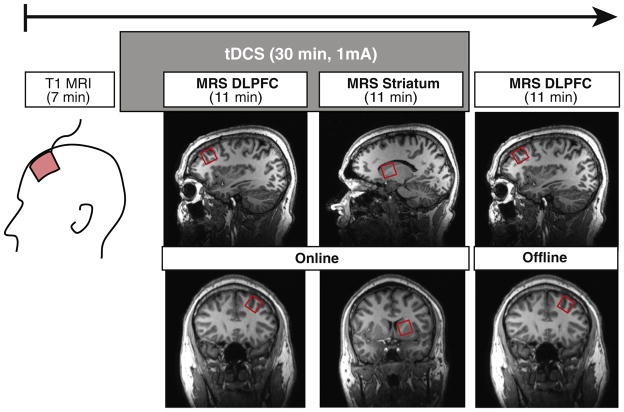
We delivered tDCS during MRS scanning (Figure 1) We started tDCS 5 minutes before acquiring the first spectroscopy scan. To the best of our knowledge, no studies reported online neural effects of tDCS when targeting the DLPFC. However, results of studies reported that tDCS over the primary motor cortex had to be delivered for 5 minutes to induce significant differences in the amplitude of motor evoked potentials as captured by electromyography (19) We are aware that the effects of tDCS over the DLPFC or the primary motor cortex may have a different timeline, but we also made this choice of starting the MRS scan after 5 minutes of stimulation because the most likely side effect of tDCS is an itching sensation during ramp periods (the first and last 30 seconds of tDCS delivery), which might cause head movement and affect data quality. Each scanning period lasted 50 minutes at our facility. Following the acquisition of the 7-minute anatomic magnetic resonance imaging scan and the 30-minute tDCS/MRS session, we had time for only one poststimulation measurement. Because the main goal of our study was to capture the effect of tDCS applied over the DLPFC, we chose to focus on the region under the anode, the left DLPFC.
Figure 1.
Experimental timeline. Following the acquisition of a T1-weighted anatomic image, we delivered active or sham stimulation to the dorsolateral prefrontal cortex (DLPFC) with the anode electrode over the left DLPFC and the cathode electrode over the right DLPFC. We acquired glutamate + glutamine, N-acetylaspartate, and gamma-aminobutyric acid levels in the left DLPFC (ipsilateral to the anode) and in the left striatum beginning 5 minutes after the start of stimulation. We acquired the same metabolites in the left DLPFC immediately after stimulation. MRI, magnetic resonance imaging; MRS, magnetic resonance spectroscopy; tDCS, transcranial direct current stimulation.
MRS Measurements
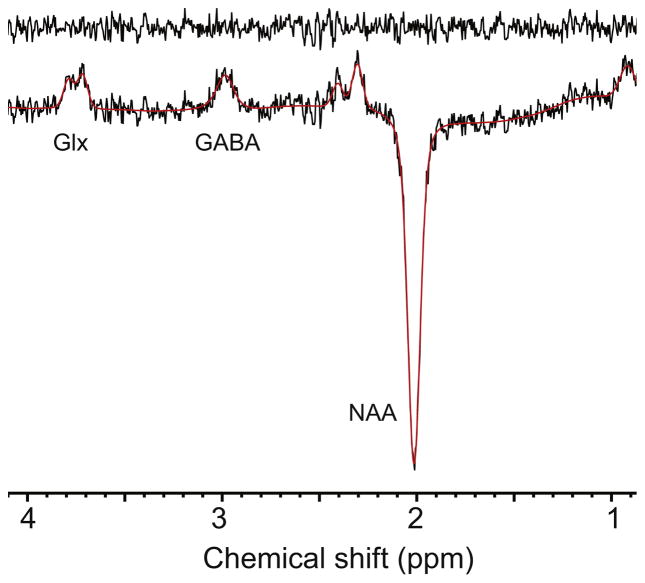
Scanning was performed with a Philips 3-tesla Achieva scanner (Philips Healthcare, Best, The Netherlands) T1-weighted structural magnetic resonance images were acquired with an magnetization prepared rapid acquisition gradient-echo sequence (repetition time = 8.2 ms, echo time = 3.7 ms, field of view = 250 mm, flip angle = 8°, 256 × 256 matrix, 180 slices/volume, slice thickness = 1 mm, no gap) Spectroscopy measurements were acquired during and after stimulation. We obtained each MRS scan lasting 10 minutes 33 seconds in the same order for all participants: 1) left DLPFC during stimulation, 2) left striatum during stimulation, and 3) left DLPFC immediately after the end of stimulation. We obtained spectroscopy measurements using the MEGA PRESS acquisition sequence (20), an efficient and reliable sequence for detecting endogenous GABA (21,22) and other brain metabolites. The MEGA PRESS spectra were acquired from 3 × 3 × 3 cm3 voxels; Figure 2 shows sample MEGA PRESS spectra. We positioned the left DLPFC voxel over Brodmann areas 46 and 9, located in the middle frontal gyrus, with one face parallel to the skull. The voxel might have encompassed more than this gyrus because of the large voxel volume. We positioned the striatum voxel over the head and tail parts of the caudate nucleus, encompassing the internal capsule and ventral striatum on the coronal plane. On the sagittal plane, the voxel was positioned under the lateral ventricle with one face of the voxel parallel to the ventricle (Figure 1) We used the following spectroscopy parameters: repetition time/echo time = 2000/68 ms, spectral bandwidth = 2000 Hz, 2048 samples with 320 averages, and 14-ms Gaussian editing pulses applied to the GABA spins either at 1.9 ppm or at 7.46 ppm in an interleaved manner. This typical acquisition protocol (22) results in a GABA signal that is contaminated by macromolecular signal and often referred to as GABA+.
Figure 2.
Sample MEGA PRESS spectra from the prefrontal voxel illustrated in Figure 1. GABA, gamma-aminobutyric acid; Glx, glutamate + glutamine; NAA, N-acetylaspartate.
We analyzed Glx and NAA measurements with Tarquin 4.3.4 (23) and GABA measurements with Gannet 2.0 (24) Tarquin has comparable reliability in metabolite levels quantification to other software, such as LCModel (22) Metabolite levels were calculated relative to the unsuppressed water signal from the same voxel, and the Tarquin fitted amplitudes were summed to give a total estimate of Glx. No data were excluded from the study because of poor quality, other than the two subjects who demonstrated movement artifacts. We assessed quantification reliability of Tarquin results with Cramer-Rao lower bounds, with a Cramer-Rao lower bound value >20% considered unreliable. The normalized residuals of the Gannet model of GABA data all fell within the range 10%–15%. We compiled and entered data for all participants in IBM SPSS Statistics version 22.0 (IBM Corp., Armonk, New York) to compare mean differences between active and sham conditions for each metabolite (Glx, NAA, and GABA) and all measures (left DLPFC during stimulation, left striatum during stimulation, left DLPFC after the end of stimulation) within each participant. We used a multiple-related samples Wilcoxon test (Wilcoxon related samples t test), which enables one to test the difference between matched pairs when the population cannot be assumed to be distributed normally.
RESULTS
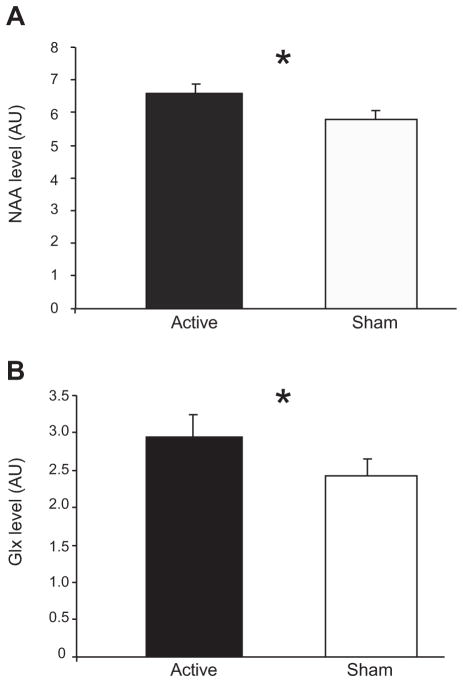
During stimulation, tDCS induced a significant change in NAA level (active vs. sham, Wilcoxon related samples t test; p = .041) (Figure 3A) but no change in Glx (p = .906) or GABA (p = .850) level in the left DLPFC. In the left striatum, tDCS also induced a significant change in Glx level (active vs. sham, Wilcoxon related samples t test; p = .027) (Figure 3B) but no change in NAA (p = .246) or GABA (p = .342) level. No significant difference was induced in NAA (p = .820), Glx (p = .619), or GABA (p = .243) level by tDCS when measured in the left DLPFC immediately after the end of stimulation (Table 2).
Figure 3.
Elevation of prefrontal N-acetylaspartate (NAA) and striatal glutamate + glutamine (Glx) levels by transcranial direct current stimulation (tDCS) applied over the dorsolateral prefrontal cortex. (A) NAA levels (n = 14) in the left dorsolateral prefrontal cortex during active and sham tDCS. (B) Glx levels (n = 15) in the left striatum during active and sham tDCS. Light gray bars represent group averages for active and sham stimulation. Results are in arbitrary units (AU) Error bars represent SEM. *p < .05.
Table 2.
Results for Glx and NAA Levels for Each Voxel of Interest During and After tDCS
| DLPFC During tDCS | Striatum During tDCS | DLPFC After tDCS | |
|---|---|---|---|
| Active tDCS | |||
| Glx | 5.19 (.24) | 6.62 (.38) | 4.45 (.37) |
| NAA | 6.59 (.28) | 5.52 (.48) | 4.49 (.39) |
| Sham tDCS | |||
| Glx | 5.13 (.31) | 5.85 (.33) | 4.38 (.37) |
| NAA | 5.82 (.22) | 4.99 (.46) | 4.35 (.48) |
Values are mean (SEM).
DLPFC, dorsolateral prefrontal cortex; Glx, glutamate + glutamine; NAA, N-acetylaspartate; tDCS, transcranial direct current stimulation.
When comparing online and offline metabolite concentrations within the DLPFC during active and sham tDCS, there was a difference in NAA levels (Wilcoxon related samples t test; active, p = .002; sham, p = .056) but not in GABA (active, p = .344; sham, p = .943) or Glx (active, p = .407; sham, p = .136) levels. More specifically, NAA concentration within the left DLPFC was significantly elevated during tDCS compared with after tDCS in the active condition.
We coregistered the active and sham MRS voxels for the DLPFC and striatum using the header information of native T1 images in Gannet 2.0 (25) Subsequently, we used SPM12 statistical parametric mapping to segment tissues within the voxel into gray matter, white matter, and cerebrospinal fluid. Results from these supplementary analyses suggest there was no bias in voxel placement in relative gray matter proportion between sessions in the left DLPFC (active vs. sham, Wilcoxon related samples t test; p = .959) and striatum (p = .878) Additionally, using the same analysis, there was no change in cerebrospinal fluid between the two sessions DLPFC (p = .163) and striatum (p = .233) voxel, and there was no change in the white matter voxel compositions DLPFC (p = .332) and striatum (p = .502) voxel. These results suggest there was no significant bias in voxel placement between sessions. Furthermore, water tissue content was not different between acquisitions.
We administered a standardized side effect form and a 16-item visual analog scale questionnaire on mood before and after each experimental session. All side effects reported are listed in Table 3. There were no significant differences in the number of reported side effects (paired samples t test, p = .332) or in mood (paired samples t test, p = .438) between active and sham tDCS conditions.
Table 3.
Side Effects Reported by Participants at Each tDCS/MRS Sessiona
| Participant No. | Active tDCS | Sham tDCS |
|---|---|---|
| 1 | Headache (mild) | Trouble concentrating (mild) |
| 4 | Trouble concentrating (mild) | |
| 7 | Neck pain (mild) | Neck pain (mild) |
| 9 | Light tingling (mild) | |
| 11 | Trouble concentrating (mild) | |
| 12 | Headache (mild) | |
| 13 | Trouble concentrating (mild) | Trouble concentrating (mild) |
| 14 | Neck pain (mild) | Neck pain (mild) |
MRS, magnetic resonance spectroscopy; tDCS, transcranial direct current stimulation.
Side effects were rated as absent, mild, moderate, or severe.
DISCUSSION
In this study, we found that a mild (1 mA) dose of anodal tDCS over the left DLPFC coupled with cathodal tDCS over the right DLPFC induced a significant and rapid elevation in prefrontal NAA (within 15 minutes) and striatal Glx (within 30 minutes) This elevation was normalized immediately after the end of stimulation. Overall, these results may suggest that tDCS has an excitatory effect on the prefrontal cortex and that this effect is primarily represented in the ipsilateral striatum, down the corticostriatal pathways, and within the basal ganglia. Although there was no direct elevation of prefrontal glutamate, these results support work that had proposed excitatory effects of anodal stimulation promoting synaptic plasticity, mediated through N-methyl-D-aspartate receptor activity. For example, anodal stimulation can enhance motor cortical excitability, and N-methyl-D-aspartate receptor partial agonist D-cycloserine can prolong the length of this augmented motor cortical plasticity (26).
The present study demonstrated an elevation of NAA levels in the DLPFC during active compared with sham tDCS. This effect was no longer significant immediately after the end of stimulation delivery, suggesting that this effect is normalized immediately after the end of stimulation. The metabolite NAA is critical to cellular regulatory processes, protein synthesis, and lipid production and is a marker of mitochondrial function (27) Decreases in NAA levels, reflecting cellular dysfunction or impaired functioning, have been correlated with neurologic lesions and psychopathology. Reduced NAA levels have been observed in major depressive disorders (28) and schizophrenia (29,30) Elevated NAA levels are known to occur after psychostimulant medication (31,32), antipsychotic medication (33), and repetitive transcranial magnetic stimulation administration (34) Also, NAA is metabolically close to glutamate, with aspartate the intermediate in a two-step conversion, and NAA and glutamate may track in MRS studies, as both metabolites are found in neurons. Previous results demonstrated that tDCS applied to the parietal cortices, with the anode over P4 and cathode over the contralateral arm, elevated NAA and glutamatergic metabolites under the anode after stimulation (35) Our results concur with the results of Clarke et al. (35) with a similar effect over NAA levels in the prefrontal cortex. However, we did not measure a significant elevation of Glx in the prefrontal cortex.
The present study also shows a transient and rapid elevation of Glx levels in the striatal region. This result indicates that excitatory stimulation of the DLPFC may provoke an ipsilaterally distal effect over the circuitry of the basal ganglia. We believe that such elevation of Glx in the ipsilateral striatum may result from increased glutamatergic transmission as a result of tDCS applied to the prefrontal cortex. Stimulation of neuronal metabolism in the prefrontal cortex could lead to glutamate release in downstream structures; this may partly explain some behavioral effects of tDCS. The corticostriatal glutamate pathways play a crucial neuromodulatory role within the striatum (17,36) This is of particular interest, as corticostriatal and mesocorticolimbic circuitries are often found impaired in several neuropsychiatric conditions (37), including substance use disorders (38) and schizophrenia (39,40) Although our results did not show a significant modulation of Glx within the prefrontal cortex, they suggest that tDCS may act through glutamatergic transmission. Glutamate transmission is impaired in depression as seen with lower prefrontal Glx levels (41) and decreased Glu metabolism of individuals with depression (42) Repeated sessions of tDCS applied to the prefrontal cortex can reduce depressive symptoms in patients with major depressive disorder (8,10) These therapeutic effects may be attributable to stimulation of glutamatergic transmission, supporting tDCS as a nonpharmacologic alternative treatment in major depression. A systemic explanation for therapeutic potential of tDCS would be that direct stimulation of cortical glutamatergic neurons entails important modulation on a wide array of cerebral structures, as the prefrontal cortex has crucial glutamatergic outputs to the nucleus accumbens and striatum.
We did not find significant differences in Glx under the anode during or after stimulation, whereas Clarke et al. (35) observed differences in Glx levels under the anode after stimulation. This discrepancy may be explained by methodologic differences between these studies, such as the current intensity (1 mA vs. 2 mA) and electrode montage (unilateral vs. bilateral stimulation paradigm) Specifically, Clarke et al. delivered 2 mA with the anode over P4 and cathode on the contralateral arm; we delivered 1 mA with the anode over F3 and cathode over F4. They found differences by comparing Glx levels before and after tDCS at P4; we found no significant differences in Glx levels by comparing active and sham tDCS at F3 or during and after active tDCS. Our results also differ from the results of Stagg et al. (6) These authors delivered 1 mA for 10 minutes with the anode over the primary motor cortex (M1) and cathode over the supraorbital area and compared Glx levels before and after tDCS. They reported that tDCS with the cathode applied to M1 caused a local decrease in Glx compared with sham, whereas tDCS with the anode over M1 did not change Glx compared with sham tDCS. Again, methodologic choices differ greatly between the work of Stagg et al. (6) and our study, which makes it difficult to compare results.
The present study also shows that the observed neurometabolism differences, elevated levels of prefrontal NAA and striatal Glx, during tDCS delivery were no longer significant immediately after the end of stimulation. This finding may partially explain some discrepancies on the effects of tDCS in the literature, especially when comparing online and offline designs with single tDCS sessions. One might find behavioral and cognitive differences when testing during stimulation, but these differences might disappear immediately after stimulation delivery. Longer lasting effects of tDCS might be observed with repeated tDCS sessions.
Finally, this study also shows that anodal stimulation applied over the left DLPFC coupled with cathodal stimulation over the right DLPFC did not induce significant differences in prefrontal and striatal GABA levels. It has been shown that anodal tDCS–related excitatory effects over M1 are silenced by administration of the N-methyl-D-aspartate antagonist dextromethorphan (13) and reduced by GABA receptors agonist lorazepam (14), suggesting a paramount role for glutamatergic and GABAergic transmission in tDCS effects. Stagg et al. (6) reported differences in GABA levels when comparing before and after 1 mA of stimulation for 10 minutes with the anode over M1 and cathode over the supraorbital area. They reported that after tDCS with the anode over M1, GABA levels decreased compared with sham tDCS. The lack of significant differences in GABA levels in the present study may result from important technical discrepancies in current intensity, tDCS duration, and MRS parameters.
Future studies are needed to characterize the neurophysiologic effects of tDCS and, to a greater extent, the effect of repeated sessions of tDCS on Glx and GABA levels. Given that most striatal medium spiny neurons contain GABA, prefrontal glutamatergic activation of corticostriatal fibers may facilitate GABAergic transmission and GABA release from the striatum to nearby subcortical structures in the basal ganglia. Therefore, it is possible that modulation of GABA transmission may be perceptible only in other structures rather than the striatal region, such as the internal and external globus pallidus. Our methodology and timeline prevented us from taking such measurements. It is also possible that repeated sessions of tDCS over the DLPFC at 2 mA, as mainly delivered in studies targeting clinical populations, may have greater and longer lasting effects on glutamatergic facilitation, and this may modulate prefrontal and striatal GABA release. Also, we used a 3 × 3 × 3 cm3 voxel to obtain three MRS measurements within the 50-minute scan period. However, the large voxel volume is difficult to position in the studied regions, the DLPFC and basal ganglia, which is a limitation of our work. Future studies should investigate the effects of tDCS using smaller voxel volumes to test whether similar neurochemical differences are observed. An additional limitation to the interpretation of our results is that if applying a post-hoc Bonferroni-type correction, the corrected α level of significance would prevent our data from reaching significance.
In conclusion, prefrontal NAA and striatal Glx neurochemical differences were shown during a single session of 1 mA tDCS. To the best of our knowledge, this is the first study combining tDCS and MRS online demonstrating the direct modulation of metabolites with tDCS in real time. An offline design would not be sensitive to the transient differences we observed. Future studies are needed to address the proper mechanistic effect of tDCS in the prefrontal cortex, the region most likely to be targeted in future clinical practice. Findings from this work indicate the importance of further evaluation of the safe dosage and optimal stimulation target of tDCS to delimitate its full potential for both clinical and healthy populations.
Acknowledgments
This work was supported by the National Sciences and Engineering Research Council of Canada Grant No. 402629-2011 (to SF) SF is supported by the Canada Research Chair in Cognitive Neuroplasticity; AH-B by Ph.D. awards from the Fonds de Recherche en Santé du Québec and the Centre Interdisciplinaire de Recherche en Réadaptation et Intégration Sociale; and RAE by National Institutes of Health Grant Nos. R01 EB016089 and P41 EB015909. This study applies tools developed under the National Institutes of Health Grant Nos. R01 EB016089 and P41 EB015909 (to RAE) and the Consortium d’imagerie en neuroscience et santé mentale de Québec via a Platform Support Grant from the Brain Canada Foundation (to SF).
We thank G. Gilbert and A. Harris for their help with magnetic resonance imaging and J. Leblond for his advice on statistical analysis.
Footnotes
DISCLOSURES
The authors report no biomedical financial interests or potential conflicts of interest.
References
- 1.Nihonsugi T, Ihara A, Haruno M. Selective increase of intention-based economic decisions by noninvasive brain stimulation to the dorsolateral prefrontal cortex. J Neurosci. 2015;35:3412–3419. doi: 10.1523/JNEUROSCI.3885-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Metuki N, Sela T, Lavidor M. Enhancing cognitive control components of insight problems solving by anodal tDCS of the left dorsolateral prefrontal cortex. Brain Stimul. 2012;5:110–115. doi: 10.1016/j.brs.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 3.Zaehle T, Sandmann P, Thorne JD, Jäncke L, Herrmann CS. Transcranial direct current stimulation of the prefrontal cortex modulates working memory performance: Combined behavioural and electrophysiological evidence. BMC Neurosci. 2011;12:2. doi: 10.1186/1471-2202-12-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Horvath JC, Forte JD, Carter O. Evidence that transcranial direct current stimulation (tDCS) generates little-to-no reliable neurophysiologic effect beyond MEP amplitude modulation in healthy human subjects: A systematic review. Neuropsychologia. 2015;66:213–236. doi: 10.1016/j.neuropsychologia.2014.11.021. [DOI] [PubMed] [Google Scholar]
- 5.Horvath JC, Forte JD, Carter O. Quantitative review finds no evidence of cognitive effects in healthy populations from single-session transcranial direct current stimulation (tDCS) Brain Stimul. 2015;8:535–550. doi: 10.1016/j.brs.2015.01.400. [DOI] [PubMed] [Google Scholar]
- 6.Stagg CJ, Best JG, Stephenson MC, O’Shea J, Wylezinska M, Kincses ZT, et al. Polarity-sensitive modulation of cortical neurotransmitters by transcranial stimulation. J Neurosci. 2009;29:5202–5206. doi: 10.1523/JNEUROSCI.4432-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.George MS, Aston-Jones G. Noninvasive techniques for probing neurocircuitry and treating illness: Vagus nerve stimulation (VNS), transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS) Neuropsychopharmacology. 2012;35:301–316. doi: 10.1038/npp.2009.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brunoni AR, Nitsche MA, Bolognini N, Bikson M, Wagner T, Merabet L, et al. Clinical research with transcranial direct current stimulation (tDCS): Challenges and future directions. Brain Stimul. 2012;5:175–195. doi: 10.1016/j.brs.2011.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brunoni AR, Valiengo L, Baccaro A, Zañao TA, de Oliveira JF, Goulart A, et al. The sertraline vs electrical current therapy for treating depression clinical study. JAMA Psychiatry. 2013;70:383–391. doi: 10.1001/2013.jamapsychiatry.32. [DOI] [PubMed] [Google Scholar]
- 10.Loo C, Alonzo A, Martin D, Mitchell PB, Galvez V, Sachdev P. Transcranial direct current stimulation for depression: 3-week, randomized, sham-controlled trial. Br J Psychiatry. 2012;200:52–59. doi: 10.1192/bjp.bp.111.097634. [DOI] [PubMed] [Google Scholar]
- 11.Brunelin J, Mondino M, Gassab L, Haesebaert F, Gaha L, Suaud-Chagny MF, et al. Examining transcranial direct-current stimulation (tDCS) as a treatment for hallucinations in schizophrenia. Am J Psychiatry. 2012;169:719–724. doi: 10.1176/appi.ajp.2012.11071091. [DOI] [PubMed] [Google Scholar]
- 12.Jansen JM, Daams JG, Koeter MWJ, Veltman DJ, van den Brink W, Goudriaan AE. Effects of non-invasive neurostimulation on craving: A meta-analysis. Neurosci Biobehav Rev. 2013;37:2472–2480. doi: 10.1016/j.neubiorev.2013.07.009. [DOI] [PubMed] [Google Scholar]
- 13.Liebetanz D, Nitsche MA, Tergau F, Paulus W. Pharmacological approach to the mechanisms of transcranial DC-stimulation-induced after-effects of human motor cortex excitability. Brain. 2002;125:2238–2247. doi: 10.1093/brain/awf238. [DOI] [PubMed] [Google Scholar]
- 14.Nitsche MA, Liebetanz D, Schlitterlau A, Henschkle U, Fricke K, Fromman K, et al. GABAergic modulation of DC stimulation-induced motor cortex excitability shifts in humans. Eur J Neurosci. 2004;19:2720–2726. doi: 10.1111/j.0953-816X.2004.03398.x. [DOI] [PubMed] [Google Scholar]
- 15.Urenjak J, Williams SR, Gadian DG, Noble M. Proton nuclear magnetic resonance spectroscopy unambiguously identifies different neural cell types. J Neurosci. 1993;13:981–989. doi: 10.1523/JNEUROSCI.13-03-00981.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Moffett JR, Ross B, Arun P, Madhavarao CN, Namboodiri AM. N-Acetylaspartate in the CNS: From neurodiagnostics to neurobiology. Prog Neurobiol. 2007;81:89–131. doi: 10.1016/j.pneurobio.2006.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kelley AE. Memory and addiction: Shared neural circuitry and molecular mechanisms. Neuron. 2004;44:161–179. doi: 10.1016/j.neuron.2004.09.016. [DOI] [PubMed] [Google Scholar]
- 18.Gandiga PC, Hummel FC, Cohen L. Transcranial DC stimulation (tDCS): A tool for double-blind sham-controlled clinical studies in brain stimulation. Clin Neurophysiol. 2006;117:845–850. doi: 10.1016/j.clinph.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 19.Nitsche M, Paulus W. Sustained excitability elevations induced by transcranial DC motor cortex stimulation in humans. Neurology. 2001;57:1899–1901. doi: 10.1212/wnl.57.10.1899. [DOI] [PubMed] [Google Scholar]
- 20.Mescher M, Merkle H, Kirsch J, Garwood M. Simultaneous in vivo spectral editing and water suppression. NMR Biomed. 1998;11:266–272. doi: 10.1002/(sici)1099-1492(199810)11:6<266::aid-nbm530>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 21.Puts NA, Edden RA. In vivo magnetic resonance spectroscopy of GABA: A methodological review. Prog Nucl Magn Reson Spectrosc. 2012;60:29–41. doi: 10.1016/j.pnmrs.2011.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mullins PG, McGonigle DJ, O’Gorman RL, Puts NA, Vidyasagar R, Evans CJ, et al. Current practice in the use of MEGA-PRESS spectroscopy for the detection of GABA. Neuroimage. 2014;86:43–52. doi: 10.1016/j.neuroimage.2012.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wilson M, Reynolds G, Kauppinen RA, Arvanatis TN, Peet AC. A constrained least-squares approach to the automated quantitation of in vivo 1H magnetic resonance spectroscopy data. Magn Reson Med. 2011;65:1–12. doi: 10.1002/mrm.22579. [DOI] [PubMed] [Google Scholar]
- 24.Edden RA, Puts NA, Harris AD, Barker PB, Evans CJ. Gannet: A batch-processing tool for the quantitative analysis of gamma-aminobutyric acid-edited MR spectroscopy spectra. J Magn Reson Imaging. 2013;40:1445–1452. doi: 10.1002/jmri.24478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harris AJ, Puts NA, Edden RA. Tissue correction for GABA-edited MRS: Considerations of voxel composition, tissue segmentation and tissue relaxation. J Magn Reson Imaging. 2015;42:1431–1440. doi: 10.1002/jmri.24903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nitsche MA, Jaussi W, Liebetanz D, Lang N, Tergau F, Paulus W. Consolidation of human motor cortical neuroplasticity by D-cycloserine. Neuropsychopharmacology. 2004;29:1573–1578. doi: 10.1038/sj.npp.1300517. [DOI] [PubMed] [Google Scholar]
- 27.Van der Graaf M. In vivo magnetic resonance spectroscopy: Basic methodology and clinical applications. Eur Biophys J. 2010;39:527–540. doi: 10.1007/s00249-009-0517-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gruber S, Frey R, Mlyranick V, Stadlbauer A, Heiden A, Kasper S, et al. Quantification of metabolic differences in the brain of depressive patients and controls obtained by 1H-MRS at 3 Tesla. Invest Radiol. 2003;38:403–410. doi: 10.1097/01.rli.0000073446.43445.20. [DOI] [PubMed] [Google Scholar]
- 29.Mondino M, Brunelin J, Poulet E. N-acetyl-aspartate level is decreased in the prefrontal cortex in subjects at-risk for schizophrenia. Front Psychiatry. 2013;4:1–6. doi: 10.3389/fpsyt.2013.00099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Steen RG, Hamer RM, Lieberman JA. Measurements of brain metabolites by 1H magnetic resonance spectroscopy in patients with schizophrenia: A systematic review and meta-analysis. Neuropsychopharmacology. 2005;30:1949–1962. doi: 10.1038/sj.npp.1300850. [DOI] [PubMed] [Google Scholar]
- 31.Kronenberg G, Ende G, Alm B, Deuschle M, Heuser I, Colla M. Increased NAA and reduced choline levels in the anterior cingulum following chronic methyphenidate. Eur Arch Psychiatry Clin Neurosci. 2008;258:446–450. doi: 10.1007/s00406-008-0810-2. [DOI] [PubMed] [Google Scholar]
- 32.Wiguna T, Guerrero A, Wibisono S, Sastroasmoro S. Effect of 12-week administration of 20-mg long-acting methylphenidate on Glu/Cr, NAA/Cr, Cho/Cr, and mI/Cr rations in the prefrontal cortices on school-age children in Indonesia: a study using 1H resonance spectroscopy (MRS) Clin Neuropharmacol. 2012;35:81–85. doi: 10.1097/WNF.0b013e3182452572. [DOI] [PubMed] [Google Scholar]
- 33.Ertugrul A, Volkan-Salanci B, Basar K, Oguz KK, Demir B, Ergun EL, et al. The effect of clozapine on regional cerebral blood flow and brain metabolite ratios in schizophrenia: Relationship with treatment response. Psychiatry Res. 2009;174:121–129. doi: 10.1016/j.pscychresns.2009.04.007. [DOI] [PubMed] [Google Scholar]
- 34.Fregni F, Potvin K, Dasilva D, Wang X, Lenkinski RE, Freedman SD, Pascual-Leone A. Clinical effects and brain metabolic correlates in non-invasive cortical neuromodulation for visceral pain. Eur J Pain. 2011;15:53–60. doi: 10.1016/j.ejpain.2010.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Clarke VP, Coffman BA, Trumbo MC, Gasparovic C. Trans-cranial direct current stimulation (tDCS) produces localized and specific alterations in neurochemistry: A 1H magnetic resonance spectroscopy study. Neurosci Lett. 2011;500:67–71. doi: 10.1016/j.neulet.2011.05.244. [DOI] [PubMed] [Google Scholar]
- 36.Kalivas PW, Duffy P, Barry J. Regulation of the mesocorticolimbic dopamine system by glutamic acid receptor subtypes. J Pharmacol Exp Ther. 1989;251:378–387. [PubMed] [Google Scholar]
- 37.Shepherd GMG. Corticostriatal connectivity and its role in disease. Nat Rev Neurosci. 2013;14:278–291. doi: 10.1038/nrn3469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Koob G, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2009;35:217–238. doi: 10.1038/npp.2009.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fornito A, Zalesky A, Pantelis C, Bullmore ET. Schizophrenia, neuroimaging and connectomics. Neuroimage. 2012;4:2296–2314. doi: 10.1016/j.neuroimage.2011.12.090. [DOI] [PubMed] [Google Scholar]
- 40.Foerde K, Poldrack RA, Khan BJ, Bookheimer SY, Bilder RM, Guthrie D, et al. Selective corticostriatal dysfunction in schizophrenia: Examination of motor and cognitive skill learning. Neuropsychology. 2008;22:100–109. doi: 10.1037/0894-4105.22.1.100. [DOI] [PubMed] [Google Scholar]
- 41.Hasler G, van der Veen JW, Tumonis T, Meyers N, Shen J, Drevets WC. Reduced prefrontal glutamate/glutamine and gamma-aminobutyric acid levels in major depression determined using proton magnetic resonance spectroscopy. Arch Gen Psychiatry. 2007;64:193–200. doi: 10.1001/archpsyc.64.2.193. [DOI] [PubMed] [Google Scholar]
- 42.Abdallah CG, Jiang L, De Feyter HM, Fasula M, Krystal JH, Rothman DL, et al. Glutamate metabolism in major depressive disorder. Am J Psychiatry. 2014;171:1320–1327. doi: 10.1176/appi.ajp.2014.14010067. [DOI] [PMC free article] [PubMed] [Google Scholar]