Graphical abstract

Keywords: Plants, Microorganisms, Uric acid, Hyperuricemia, Uricase, Uricase encoding genes
Abstract
Uric acid increased accumulation and/or reduced excretion in human bodies is closely related to pathogenesis of gout and hyperuricemia. It is highly affected by the high intake of food rich in purine. Uric acid is present in both higher plants and microorganisms with species dependent concentration. Urate-degrading enzymes are found both in plants and microorganisms but the mechanisms by which plant degrade uric acid was found to be different among them. Higher plants produce various metabolites which could inhibit xanthine oxidase and xanthine oxidoreductase, so prohibit the oxidation of hypoxanthine to xanthine then to uric acid in the purine metabolism. However, microorganisms produce group of degrading enzymes uricase, allantoinase, allantoicase and urease, which catalyze the degradation of uric acid to the ammonia. In humans, researchers found that several mutations caused a pseudogenization (silencing) of the uricase gene in ancestral apes which exist as an insoluble crystalloid in peroxisomes. This is in contrast to microorganisms in which uricases are soluble and exist either in cytoplasm or peroxisomes. Moreover, many recombinant uricases with higher activity than the wild type uricases could be induced successfully in many microorganisms. The present review deals with the occurrence of uric acid in plants and other organisms specially microorganisms in addition to the mechanisms by which plant extracts, metabolites and enzymes could reduce uric acid in blood. The genetic and genes encoding for uric acid in plants and microorganisms are also presented.
Introduction
Uric acid is one of the most important nitrogen compounds in animal and plant bodies. It consists of 2,6,8 trihydroxypurine existing as a keto-enol tautomerism that under physiological conditions can easily be converted to the corresponding urate [1]. It derived from purine, two of which, adenine and guanine, are present in DNA and RNA. In Human, both uric acid and urate are accumulated in the form of calculi in the joints and/or connective tissues causing arthritis and rheumatic pain. They may also deposit in kidneys and/or ureters causing kidney disease or failure [2].
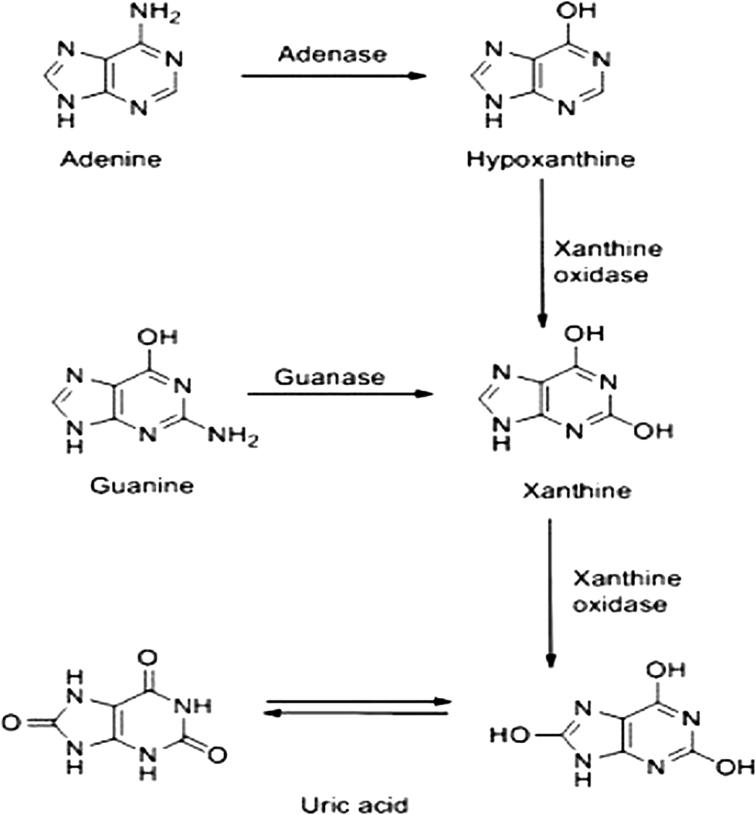
Uric acid is either produced when the body breaks purine occurred naturally [3] (Fig. 1) or supplied from certain foods. Consequently, some animal and plant foods with high purine contents should be avoided from diet especially in persons suffer from gout, as the overproduction of uric acid can induce hyperuricemia which is linked to gout [4].
Fig. 1.
Production of uric acid from purines.
Adapted from Xiang et al. [3].
The normal level of uric acid in the blood is between 3–7 mg/100 mL, which is required to human and animal bodies as antioxidant and prevents damage of blood vessels lining so protect them. Low purine diets including plants, often required to treat gout. The average daily meal for adult in United States contains about 600–1000 mg of purines. Recent research has shown that plant purines (fruits and vegetables) have risk of uric acid accumulation but lower than that of meat and fish [5].
Production of uric acid by fungi and bacteria
Early, Jarmai [6] and Hutyra and Marek [7] reported that gout in birds had been caused by smut fungus Ustilago maydis, a common causal agent of moldy corn. Oosporin, a mycotoxin secreted by U. maydis induce gout in chickens and turkeys [8], [9]. Furthermore, Constantini [10] reported that gout and hyperuricemia have been induced in animals by the fungal species U. maydis, Chaetomium trialterale, Saccharomyces cerevisiae, and Candida utilis. It is also induced by mycotoxins, aflatoxin, ochratoxin, Oosporin, and oxalic acid. Other fungal metabolites such as cyclosporine, ergotamine, and penicillin have been found to induce gout [10].
Gout is documented to be etiologically linked to beer, a Saccharomyces fermented beverage. Researchers found that beers contain significant quantities of ochratoxin and large amount of uric acid produced by the yeast Saccharomyces sp. [10] and accumulated in its vacuoles [11]. They also indicated that drinkers of beer and wine and people who often consume yeast foods such as bread and cheese are more susceptible to develop gout [10] (Table 1). Ochratoxin, a series of nephrotoxins produced by several species of the genera Aspergillius and Penicillium was found in beer and causes gout as early detected by many authors [10], [12], [13], [14]. A synergistic interaction may occur between the alcohol from beer or yeast-fermented wine and ochratoxin. In fact, a study performed with 61 gouty men revealed that nearly all of them were beer drinkers [10].
Table 1.
Uric acid content of various beers.
| Brand of beer | Uric acid (mg/dL) |
|---|---|
| Miller beer | 7.34 |
| Olympia beer | 7.05 |
| Budweiser beer | 8.09 |
| Taiwan beer | 9.35 |
Adapted from Constantini [10].
Furthermore, long term feeding of rats with yeast autolysate has associated with rise in uric acid and anti-DNA antibodies. The elevated anti-DNA level was correlated with severe arthritis [15].
When single-cell protein, as in yeast, is used as a source of edible protein it increases uric acid in body when the individual lacks uricase [16]. Ergotamine, a fungal metabolite produced by Claviceps purpurea, and penicillin, an antibiotic produced by Penicillium notatum, has been shown to induce acute gout in human [17]. Aflatoxin, a common mycotoxin produced by Aspergillus flavus was also found to induce gout. When female Macaque monkey is fed with aflatoxin B1 contaminated food, numerous urate crystals surrounded by inflammatory cells were detected [18] and the kidneys lesions were similar to those found in human patients suffering from hyperuricemia and gout [19].
Oxalic acid, a metabolite produced by many fungal species, induced also, gout in human and chicken. It is one of the degradation products of uric acid. This explains why both oxalate and urate crystals are usually present in kidney stone of gouty patients [20].
Cyclosporine, a fungal metabolite produced by Tolypociladium inflatum and widely used as immunosuppressant, was found to be an inducer of gout in human. Many Organ Transplant Centers recorded that 24% of cyclosporine treated patients suffered from gout compared to patients treated with the immunosuppressant azathioprine where none of the patients suffered from gout [21], [22], [23].
Mushrooms and truffles contain moderate amounts of purine but are still included as a part of healthy diet because of additional benefits they provide. Moreover, Nogaim et al. [24] noticed an increase in uric acid level in blood serum of rats fed with mushroom powder after 15 days of daily diet due to much protein and phosphorus in mushroom. Continuous eating of this fungus can cause decrease in kidney function, leading to more serious high uric acid illness.
Enzymatic degradation of uric acid by microorganisms
The enzyme responsible for purine metabolism is uricase (urate oxidase, oxidoreductase, EC 1.7.3.3). It activates the oxidation of uric acid to soluble allantoin. Most vertebrates possess uricase, except humans and higher apes, which became not functional by point mutation during evolution resulting in the formation of a redundant protein [25]. Uricase is localized inside microorganisms, especially Bacillus pasteurii [26], Proteus mirabilis [27], and Escherichia coli [28], while other microorganisms could produce them extracellularly by changing certain components of the culture media as in Streptomyces albosriseolus [29], Microbacterium [30], Bacillus thermocatenulatus [31], Candida tropicalis [32], and Pseudomonas aeruginosa [33].
Microorganisms induced gout and hyperuricemia
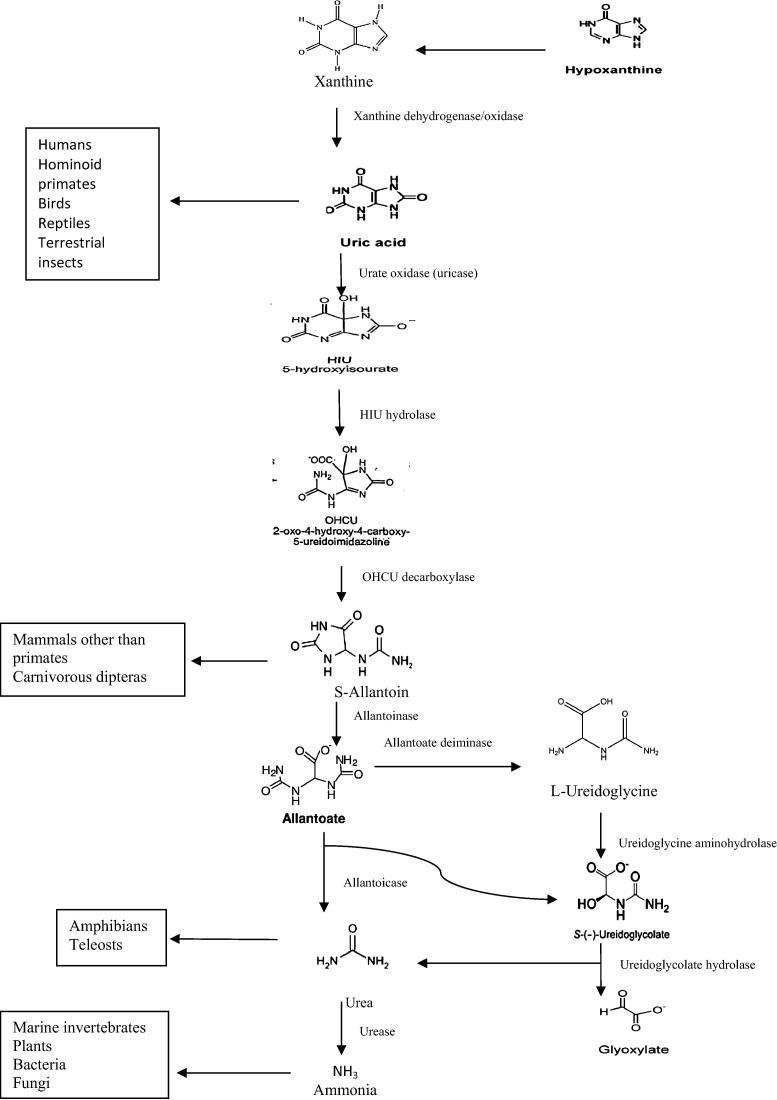
Catabolism of purine to uric acid is conserved among microorganisms; however, the end product of uric acid breakdown varies among species, depending on the kind of active catabolic enzymes. The formed uric acid can either be excreted or degraded in the peroxisomes by active catabolic enzymes [34], Fig. 2. Plants are capable to perform complete purine degradation. The end products, glycoxylate and ammonia, are recycled to synthesized organic molecules, which can be used in growth. Catabolic intermediates, urides, allentoin and allantoate, are likely to be involved in protecting plants against abiotic stress [35]. The first common intermediate of all purine bases is xanthine. It is oxidized to urate in the cytosol by xanthine dehydrogenase, whereas urate is imported into the perixosome and oxidized by uricase to 5-hydroxyisourate, which in turn converted via 2-oxy-4-hydroxy-4-carboxy-5-ureidoimidaoline to S-allantoin by the functional allantoin synthase [35], [36], [37], [38], [39], [40]. In microorganisms, different end products of uric acid degradation are due to evolution of urate oxidase (uricase, allantoinase, and allantoicase). Moreover, most microorganisms possess all the required nitrogen catabolic enzymes to completely break down uric acid to ammonia [41], [42], [43]. In certain fungi and bacteria, allantoate is hydrolyzed by an allantolate amidinohydrolase (allantoicase) generating urea and s-ureidoglycolate [44], [45], [46], while in plants, it generate s-ureidoglycolate, ammonia and carbon dioxide from allantoate as final products [44], [47], [48]. In contrast to plant and microbes, animals degrade purine to intermediate purine compounds such as urates and allentoin, which are then excreted [34], Fig. 2.
Fig. 2.
Pathway of uric acid degradation to ammonia.
Adapted from Lee et al. [34].
El-Nagger and Emara [49] isolated from soil a number of uricolytic fungi belongs to Fusarium, Spondilocladium, Stemphylium, Geotrichum, Mucor, Alternaria, Helminthosporium, Chaetomium, Penicillium, Curvularia and Aspergillus.
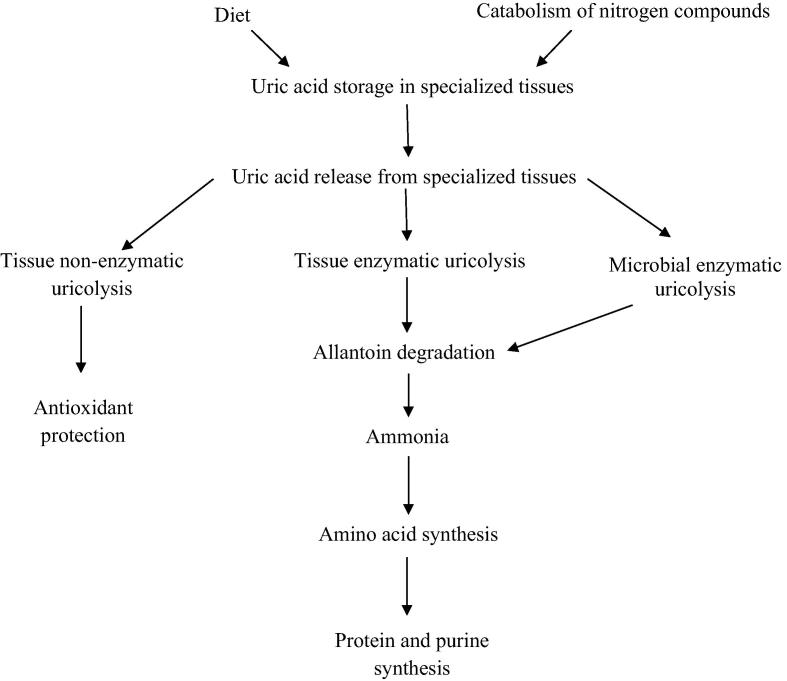
Bacteria (Pseudomonas, Enterobacter, Citrobacter and Lactococcus) isolated from gut of apple snail Pomacea canaliculata possess high uricolytic activity. It symbiotically recycles the combined nitrogen and phosphorus in the snail [50]. Uric acid subjected to either non-enzymatic uricolysis to form antioxidant or enzymatic uricolysis to form allantoin and ammonia in the snail could afford amino acid, protein and purine [50], [51], [52], [53], [54], Fig. 3.
Fig. 3.
Catabolic pathway of uric acid in tissues.
Adapted from Koch et al. [50]; Vega et al. [51], and Giraud-Billoud et al. [52], [53], [54].
Streptomyces exofolitus isolated from soil by Magda et al. [55] were found to be high producer of uricase. They reported that this pure uricase can be used to diagnose and evaluate uric acid in urine and blood. Also, Streptomyces albosriseolus isolated by Ammar et al. [29] potentially produces uricase in media containing uric acid as carbon and nitrogen source.
The “Microbial Index of Gout” was declared as a novel, sensitive and non-invasive way for diagnosing gout via fecal microbiota. They proposed that the intestinal microbiota in gout patients is highly distinguished from that of healthy ones as Bactriiodes caccae and B. xylanisolvens were enriched while Faecalibacterium parusnitzit and Bifidobacterium pseadocatenulate were depressed [56].
Ogawa [57] designed a new prophylaxis for treating hyperurecemia using probiotic effect of microorganisms as bacteria. The term probiotic refers to the living microorganisms that survive through the gastrointestinal tract and have beneficial effect on the host’s health. He used pretreated rats with uricase inhibitor “Potassium oxonate” as a model for hyperuricemia. The serum uric acid level of the group treated with probiotics showed significant repression in rat serum specifically in the presence of Lactobacillus fermentum ONRIC b0185 and b0195 and L. pentosus ONRIC b0223. These bacterial strains could convert nucleosides to purine base because they have nucleosidases activities. Nucleosidases in turn convert guanine and adenosine to hypoxathine then xanthine.
Production of uric acid by plants
Hyperuricemia is highly affected by the high dietary intake of food rich in purine, such as meats, bean seeds, mushrooms and some types of sea foods [58]. Additionally, there is growing interest in fruits, vegetables and herbs high in phytochemical compounds that have been implicated as alternative or additive drugs to gout.
Purines are naturally occurred in all plant foods. It was found that purine at 10–15 mg/100 g food is present in all plant foods. However, some plant foods can contain 100–500 mg uric acid/100 g food [59]. However, some others contain above this range. Plants which have high amounts of purines include spinach, peas, lenticels, cauliflowers and beans. Any food containing yeast extract should be avoided [60]. Several plants contain moderate concentrations of purine ranging from 50–100 mg/100 g of food, as avocado, bananas and asparagus [61], (Table 2), in which one should not consume them on weekly basis in portions larger than one small cup (in fresh state) or half cup (if in cooked state). Some foods, on the other hand, are helped in decreasing uric acid level such as pineapple, lemons, fibrous foods, olive oil, parsley, red cabbage, corn and rice [60].
Table 2.
Occurrence of uric acid in plant foods.
| Plant foods | Total uric acid mg/100 g food (average) | Plant foods | Total uric acid mg/100 g food (average) |
|---|---|---|---|
| Highest in uric acid (400 mg/100 g and higher) | |||
| Mushroom, flat, edible Boletus, dried | 488 | Yeast, Baker’s | 680 |
| Moderately High in uric acid (100–400 mg/100 g) | |||
| Bean, seed, white, dry | 128 | Bean, Soya, seed, dry | 190 |
| Black gram (mungo bean), seed, dry | 222 | Grape, dried, raisin, sultana | 107 |
| Lentil, seed, dry | 127 | Linseed | 105 |
| Peas, dry, chick (garbanzo), seed | 109 | Poppy seed, seed, dry | 170 |
| Sunflower seed, dry | 143 | ||
| Lower in uric acid (100 mg/100 g and lower) | |||
| Almond, sweet | 37 | Apple | 14 |
| Apricot | 73 | Artichoke | 78 |
| Asparagus | 23 | Aubergine | 21 |
| Avocado | 19 | Bamboo shoots | 29 |
| Banana | 57 | Barley without husk, whole grain | 96 |
| Bean sprouts, Soya | 80 | Bread, wheat (flour) or White bread | 14 |
| Broccoli | 81 | Brussel sprouts | 69 |
| Cabbage, red | 32 | Cabbage, savoy | 37 |
| Cabbage, white | 22 | Carrot | 17 |
| Cauliflower | 51 | Celeriac | 30 |
| Cherry, Morello | 17 | Cherry, sweet | 7.1 |
| Chicory | 12 | Chinese leaves | 21 |
| Chives | 67 | Cocoa powder, oil partially removed | 71 |
| Corn, sweet | 52 | Cress | 28 |
| Cucumber | 7.3 | Currant, red | 17 |
| Date, dried | 35 | Elderberry, black | 33 |
| Endive | 17 | Fennel leaves | 14 |
| Fig. (dried) | 64 | Gooseberry | 16 |
| Grape | 27 | Grass, Viper’s (black salsify) | 71 |
| Kale | 48 | Kiwi fruit (Chinise gooseberry, strawberry peach) | 19 |
| Kohlrabi | 25 | Leek | 74 |
| Lettuce | 13 | Melon, Cantelope | 33 |
| Millet, shucked corn | 62 | Morel | 30 |
| Nuts, Brail | 23 | Nuts, hazelnut (cobnut) | 37 |
| Nuts, peanut | 79 | Oats, without husk, whole grain | 94 |
| Lower in uric acid (100 mg/100 g and lower) | |||
| Olive, green, marinated | 29 | Onion | 13 |
| Orange | 19 | Parsley, leaf | 57 |
| Pea, pod and seed, green | 84 | Pea, seed, dry | 95 |
| Peach | 21 | Peppers, green | 12 |
| Pineapple | 55 | Plum | 19 |
| Plum, dried | 24 | Potato | 64 |
| Pumpkin | 18 | Quince | 44 |
| Radish | 30 | Raspberry | 18 |
| Rhubarb | 12 | Rye, Whole grain | 51 |
| Sesame (gingelly) seed, oriental, dry | 62 | Spinach | 57 |
| Squash, summer | 24 | Strawberry | 21 |
| Tomato | 11 | Wheat, whole grain | 51 |
Adapted from Halevi [61].
Vegetables containing higher levels of magnesium and lower level of calcium reduce the amounts of uric acid in the blood and decrease the chance of developing gout. These vegetables include corn, potatoes and avocados. Celery seeds are popular alternative to drugs in reducing uric acid in blood. Furthermore, fruits and vegetables contain vitamin C may help in the reduction of uric acid level in blood. Cherries especially black cherry juices being used in great quantities to help relief the symptoms of gout and reduce uric acid level [62].
Inhibition of uric acid synthesis by some plant metabolites
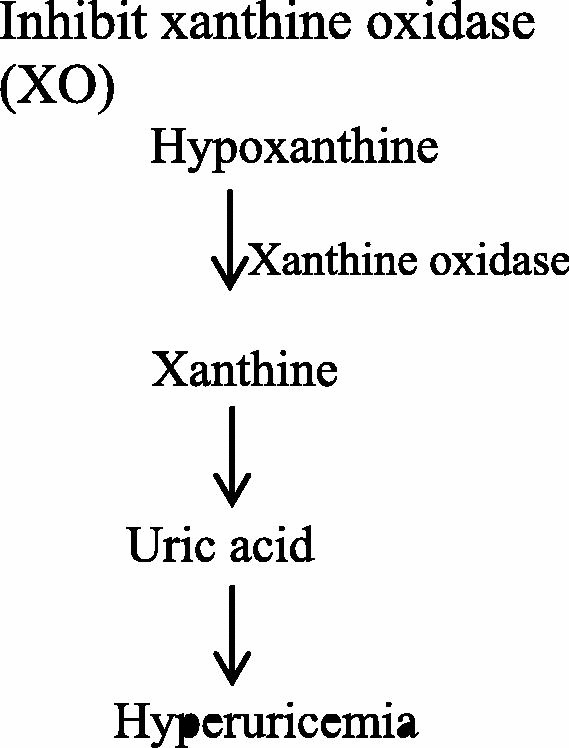
Xanthine oxidoreductase (XOR) has two forms; xanthine oxidase (XO) and xanthine dehydrogenase (XDH), both of them catalyze the oxidation of hypoxanthine to xanthines, then to uric acid in the purine metabolism [4]. Overactivity of both enzymes cause the accumulation of uric acid in the body and form a pathogenethesis condition called gout [63]. Additionally, xanthine oxidase (XO) serves as a valuable biological source of oxygen free radicals that participate in various damages of living tissues leading to many pathological states [58], [64].
Some herbal plant extracts possess antioxidant activity to abolish the oxidative and inflammatory response produced by xanthine oxidase. Xanthine oxidase [XO EC.1.2.3.2] is a key enzyme that plays a role in hyperuricemia catalyzing the oxidation of hypoxanthine to xanthine then to uric acid. The enzyme is situated at the end of the catabolic sequence of purine metabolism [65]. Therefore, several researches are focused on exploring potent XO inhibitors from wide variety of traditional herbal plants [66], [67].
Allopurinol is the efficient clinically used XO inhibitor in the treatment of gout [68]. However, this drug causes numerous side effects such as nephropathy and allergic responses [69]. Thus the search for natural XO inhibitors from plants with higher therapeutic activity and fewer side effects are needed to treat gout and other diseases associated with XO activity. Some medicinal plants represent a potential source of XO inhibitors [67], [70]. Plant flavonoids, anthocyanins and phenolics are known to have antioxidant and anti-inflammatory properties that reduce uric acid in blood [71], [72], [73].
The presence of uricases in plant was established in glyoxysomes of different seed tissues (endosperm, perisperm, scutella and cotyledons) from various plants [74] as well as in peroxisomes from maize root tips [75], soybean nodules [76], in roots but not in leaves of corn and tobacco [74], in pea and soybean leaf extracts [77] and from leaves of chickpea, broad bean and wheat [78].
Many herbal plant species were explored to be antigout and reduce uric acid in blood such as Lagerstroemia speciosa [4], Apium graveolens, Ficus carica, Curcuma domestica, Cinnamomum zeylanicum and Rosmarinus officinalis [79], Erythrina strica [80], Rhuscoriaria [81], Juniperus phoenicea [82], Momordica charantia, Apium gravelens, Petroselium crispum, Linum usitatissmun, Cucurbita pepo, Zingiber officinale, Curcurma longa, Cinnamomum sp., Rosmarinus sp. [56], [83], Origanum majorana [84], Prunus cerasus [85], Phyllanthus niruri [86], Glycine max and Arabidopsis thaliana [87], Vinca sp. [10], [88] and Colchicum sp. [10], [89], [90], [91]. The mechanisms by which these plants reduce uric acid in blood were summarized in Table 3.
Table 3.
The mechanisms by which some plant active metabolites reduce uric acid in blood.
| Plant species | Family | Used part | Active metabolite | Mechanism of action | References |
|---|---|---|---|---|---|
| Lagerstroemia speciosa (L.) Pers. | Lythraceae | Leaves | Valoneic acid dilactone (VAD) Ellagic acid (EA) |
 |
[4] |
| Apium graveolens (Celery) | Umbelliferae | Fresh leaves and seeds | Oleic and Linoleic acid in Celery All rich in phenolics Unsaturated fatty acids, long chain fatty acids, phytosterols and Malondialdehyde |
Antigout, antimicrobial, Anti-inflammatory and antioxidant effects | [79] |
| Ficus carica (Fig) | Moraceae | Dry Fig. fruits | |||
| Curcuma domestica L. (Turmeric) | Zingiberaceae | Rhizomes | |||
| Cinnamomum zeylanicum (Cinnamon) | Lauraceae | Bark | |||
| Rosmarinus officinalis (Rosemary) | Labiatae | Leaves | |||
| Erythrina strica roxb | Papilionaceae | Hydromethanolic extract of leaves | Flavonoids, saponins, tannins, phenolics and triterpenoids | Inhibit xanthine oxidase (XO) and xanthine dehydrogenase (XDH) activities | [80] |
| Rhuscoriaria (sumac or sumak) | Anacardiaceae | Hydroalcoholic extract of fruits | Phenolic (as gallic acid), methyl gallate and protocatechuic acid | – Inhibit xanthine oxidase (XO) activity – Decrease Hyperuricemia |
[81] |
| Juniperus phoenicea | Cupressaceae | Decoction of fresh leaves in water | Phenols | Reduce uric acid level and have antioxidant activity | [82] |
| Momordica charantia (Bitter) | Cucurbitaceae | Methanol-water extract of pulp | Phenols and Flavonoids | Inhibit xanthine oxidase | [58], [83] |
| Apium gravelens (Celery) | Umbelliferae | Dried powdered leaves | |||
| Petroselium crispum | Umbelliferae | Parsly leaves | |||
| Linum usitatissmum (Flax) | Linaceae | Seed | |||
| Cucurbita pepo (Pumpkin) | Cucurbitaceae | Seed | |||
| Zingiber officinale (Ginger) | Zingiberaceae | Rhizome | |||
| Curcurma longa (Turmeric) | Zingiberaceae | Whole plant | |||
| Cinnamomum sp. (Cinnamon) | Lauraceae | Leaves | |||
| Rosmarinus sp. (Rosemary) | Labiatae | Leaves | |||
| Origanum majorana Linn. | Labiatae | Ethanolic and aqueous extracts of root and stem | Phenols, flavonoids, tannins triterpenoids, saponins, polyphenols, coumarins, ellagic acid, valoneic acid dilactone | – Inhibit xanthine oxidase – Anti-gout activity |
[84] |
| Prunus cerasus L. (tart cherry) | Rosaceae | Cherry juice | Anthocyanins | – Antioxidant – Anti-inflammatory |
[85] |
| Phyllanthus niruri Linn. | Euphorbiaceae | Methanolic extract of plant | Lignans | – Uricosoric action – Xanthine oxidase inhibition |
[86] |
| Glycine max | Leguminosae | Plant extract | Allantionase Allantoate amidohydrolase Ureidoglycine aminohydrolase Ureidoglycolate amidohydrolase |
– Release nitrogen from purine nucleotides into amino acids | [87] |
| Arabidopsis thaliana | Brassicaceae | ||||
| Vinca sp. | Apocynaceae | Plant extract | Vinblastine alkaloid | – Antifungal – High potential antigout – antimicrotubule |
[10], [88] |
| Colchicum sp. | Colchicaceae | Plant extract | Colchicine alkaloid | – Antipredator and antifungal (plant protector) – Antitubulin activity – Efficient antigout: combination of colchicine and antiurate drug |
[10], [89], [90], [91] |
Genetics and uricase encoding genes
Schult et al. [92] discovered 14 functional genes encoding enzymes or proteins of the purine catabolic pathway. Five genes (pucA, pucB, pucC, pucD, and pucE) must be expressed for the function of xanthine dehydrogenase, while only 2 genes (pucL and pucM) were encoded for uricase, and pucJ and pucK genes encoded the uric acid transport system. The pucH and pucI genes encoded allantoinase and allantoin permease, respectively. On the other hand, allantoate amidohydrolase is encoded by pucF gene. The pucR-mutant Bacillus subtilis expressed low activity of all tested genes, indicating that PucR is the main regulator of puc genes expression. All 14 genes except pucI are located at 284–285° in the gene cluster on the chromosome and are implicated in six transcription units. Allantoic acid, allantoin, and uric acid were effector compounds that regulate PucR for the expression of puc genes.
Uric acid utilization activates the production of the virulence factors (capsule and urease) in the pathogen Cryptococcus neoformans (the cause of fatal meningitis in the immune-compromised patients), that potentially regulate the immune response in the host during infection. The identified catabolic genes of uric acid in C. neoformans were URO1 (urate oxidase), URO2 (HIU hydrolase), URO3 (OHCU decarboxylase), DAL1 (allantoinase), DAL2,3,3 (allantoicase-ureidoglycolate hydrolase fusion protein), and URE1 (urease) [34].
In Humans, multiple independent evolutionary events cause the pseudogenization (silencing) of the uricase gene in ancestral apes [93]. Uricase exists as insoluble crystalloid that involves the core of peroxisomes in terrestrial vertebrates [94]. Uricases of most microbial and aquatic vertebrate species are soluble and remain in either the cytoplasm (bacteria) or peroxisome (yeast) [93].
Nonsense mutations caused a pseudogenization of the uricase gene in humans. Despite being non-functional, cDNA sequencing ensured that uricase mRNA is present in human liver cells and that these transcripts have two premature stop codons [95], [96], [97].
When functional uricase gene was deleted from mice, the animals died shortly after birth, while the xanthine oxidase inhibitor allopurinol prevented the deaths. The inability of mice to undergo the sudden buildup of uric acid has indicated that ancient apes underwent successive mutations to slowly decrease uricase before pseudogenization [98]. However, other hypothesis to prevent the sudden formation of uric acid in ancient primates may be the gradual attenuation of the uricase activity before pseudogenization events [99].
In most plants, break down of purine bases gives rise to CO2 and ammonia [100]. However, in root nodules of legumes, nodule bacteria incorporated the newly fixed nitrogen into purine nucleotides, then converted to allantoin and allantoic acid, which play a crucial role in the storage and translocation of nitrogen to other tissues [101], [102].
Bacteria and fungi have the capacity to utilize numerous compounds, including purines, as nitrogen and carbon sources. In Pseudomonas aeruginosa, the encoding genes for the initial deamination step of adenine and guanine, used as nitrogen sources, are located on different loci on the chromosome, while the genes encoding the enzymes degrading hypoxanthine to ureidoglycolic acid are linked to each other [103]. Recently, it was reported that E. coli bears gene that encode for guanine deaminase [104] and many encoding genes involved in the purine catabolic pathway [105]. It was found that the expression of these genes was not sufficient to support growth using purines as the sole nitrogen source; however, when aspartate was added as the nitrogen source, purines could stimulate growth [105]. E. coli can utilize allantoin but not hypoxanthine as a nitrogen source under anaerobic conditions. The genes encoding enzymes for both allantoin and glyoxylic acid metabolisms are linked and their expressions are controlled by the allR gene product, when allantoin and glyoxylic acid are used as the effector molecules [106].
Fluri and Kinghorn [107] suggested that a single gene (all2) is involved in uricase induction and activity in Schizosaccharomyces pombe. Five mutants were isolated at the a112 gene on the basis of their inefficacy to utilize hypoxanthine as a sole source of nitrogen. The mutants were found to be unable to utilize the purines adenine, hypoxanthine, xanthine, uric acid, allantoin and allantoic acid, although they could utilize urea and ammonia. The mutants appeared to be unable to produce the enzymes included in purine catabolism.
Mutant uricase enzymes derived from the uricase gene of colonies from Bacillus subtilis by staggered extension process (StEP) mutagenesis yielding two identical active mutant genes. The mutant uricase activity in Bacillus subtilis exhibits high uricase activity [108]. Many efforts have been made to make uric acid sensors using uricase (urate oxidase, EC 1.7.3.3) as a biocatalyst [109], [110], [111], [112], [113].
Under nitrogen-limiting conditions, genes of the hypoxanthine catabolic pathway in Aspergillus nidulans are induced by a globally acting protein and a pathway-specific regulatory protein [114]. Uric acid degradation required the expression of nine unlinked genes implicated in the metabolism of purine compounds [115], [116], [117].
In bacteria, fungi, insects, animals, and plants, oxidized purines, xanthine, hypoxanthine, uric acid, pyrimidine uracil, or ascorbate were transported by nucleobase ascorbate transporters (NATs) [118], [119]. The only functionally characterized plant NAT-maize leaf permease 1 [118] was the high compatibility transporter of xanthine and uric acid that competitively binds but does not transport ascorbate [119].
Arabidopsis possesses purine permease (PUP) and ureide permease (UPS) gene families that are conserved only among plant species. The UPS family transport uracil, allantoin, while the purine permease transports xanthine and hypoxanthine [120], [121]. In French bean, one UPS was found to transport allantoin [122].
Uridine monophosphate synthase and thymidine kinase are the regulatory enzymes for purine uptake. Studies using radiolabelled purins, pirimidines and [14C] fluoroorotic acid revealed that the FOA recessive genes for “1-1/for 1-1” on chromosome 5 were unable to uptake uracil or uracil-like bases in Arabidopsis thaliana mutant [123].
To date, six loci along chromosome 5 of Arabidopsis genome were identified to encode nucleobase transporters: At5g03555 (from PRT family); At5g25420, At5g49990, and At5g62890 (from NAT family); At5g50300 (an AzgA-like transporter); and At5g41160 (from PUP family) [123], [124]. The recently characterized AzgA adenine–guanine–hypoxanthine transporter of Aspergillus nidulans was found to have amino acid similarity to Arabidopsis loci At5g50300 and At3g10960 encode proteins [125]. The amino acid sequence of the FUR4 uracil transporter of Saccharomyces cerevisiae (from PRT family) showed significant similarity to that of Arabidopsis locus At5g03555 encoded protein [123].
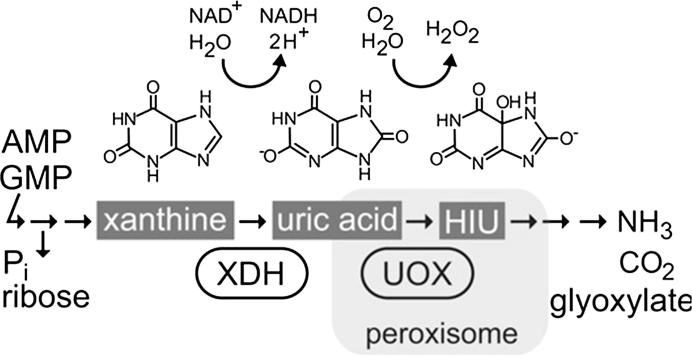
Hauck et al. [126] isolated a urate oxidase (UOX) mutant of Arabidopsis thaliana that accumulate uric acids in the tissues mainly in the embryo due to the suppression in a xanthine dehydrogenase (XDH). The UOX-mutant exhibits a severe inhibition of cotyledon development and nutrient mobilization from the lipid reserves in the cotyledons. The local defect of peroxisomes (glyoxysomes) in the cotyledon of the mature embryo causes the deposit of fatty acids in the dry seeds. Peroxisomes possess part of the purine nucleobase catabolic pathway and play a central role in the breakdown of fatty acids (ᵦ-oxidation) [127]. Without ᵦ-oxidation, seedling establishment cannot proceed and uric acid will accumulated in the embryo due to its weak mobility in lipids [126], Fig. 4.
Fig. 4.
Purine nucleotide catabolism based on reactions catalyzed by xanthine dehydrogenase (XDH) and urate oxidase (UOX). HIU, 5-hydroxyisourate; Pi, phosphate.
Adapted from Hauck et al. [126].
Uric acid is transported into the peroxisomes and oxidized by urate oxidase [UOX] to hydroxyisourate, which is converted to S-allantoin by two further enzymatic reactions [128]. Humans possess a non-functional UOX; therefore, the final product of human purine ring breakdown is uric acid, which is excreted in the urine. In plants, S-allantoin breakdown results in the complete catabolism of the purine ring system in the endoplasmic reticulum, releasing CO2, glyoxylate and ammonia [129], [130], [131].
Hongoh et al. [132] cloned the gene encoding uricase of the yeast-like symbiont of the brown plant-hopper, Nilaparvata lugens, which shows 62% sequence identity with that of Aspergillus flavus. The symbiont uricase possessed all the known consensus motifs, except the C-terminal PTS-1, Ser-basic-Leu. The symbiont's uricase gene expressed in Escherichia coli was as active as those of plants and animals, but less active than those from other fungi.
Yang and Han [133] isolated two functionally allantionase genes, AtALN (Arabidopsis allantoinase) and RpALN (Robinia pseudoacacia allantoinase). The absence of nitrogen in the medium increased the expression of these genes. The cloned AtALN and RpALN encoding allantionase confirmed that allantoin catabolism pathway exists in both Arabidopsis and Robinia spp. Multiple sequence alignment showed that those allantoinase genes share homology with those isolated from E. coli, bullfrog and yeasts.
Recombinant Hansenula polymorpha MU200 was obtained by expressing uricase from Candida utilis. The highest production of recombinant uricase reached 52.3 U/mL (about 2.1 g/L of protein) extracellularly and 60.3 U/mL (about 2.4 g/L of protein) intracellularly in fed-batch fermentation after 58 h of incubation, which are much higher than those expressed in other expression systems [134].
Rasburicase is a recombinant urate oxidase produced from Saccharomyces cerevisiae harboring Aspergillus flavus uricase gene. It acts as an alternative to allopurinol for reducing uric acid levels, so it has been used for the handling of anticancer-therapy-induced hyperuricemia [135].
The cloned uricase gene (UOXu) of Candida utilis contains 909 base pairs and encodes a protein with 303 amino acid residues and a mass of 34,1463 Da [136]. Cloned urate oxidase gene of C. utilis was recombined in the plasmid of the probiotic Lactobacillus bulgaria to produce urate oxidase that breaks down uric acid. The recombinant plasmid PMG36e-U containing urate oxidase gene of 34 KDa molecular weight has an activity up to 0.33 μ/mL [137].
Saeed et al. [138] expressed an uricolytic activity from Escherichia coli harboring uricase gene from Pseudomonas aeruginosa. The sequence of the cloned gene shows 44% similarity to the uricase gene of Cellulomonas flavigena and 35% to that of the yeast Beauveria bassiana.
Meraj et al. [139] induced mutated Bacillus subtilis with the ability for hyperproduction of urate oxidase using ethyl methane sulfonate at 180 min dose rate. The advantages to adopt mutagenesis technique for the productions of many microbial enzymes, are their simplicity and low cost. However, the cloning technique is very expensive and requires high technical facilities.
Conclusions and future perspectives
Uric acid is a catabolic insoluble product of purine metabolism. Humans are unable to further degrade uric acid. In normal cases, uric acid is excreted with urine, but in gouty cases, longstanding elevation of monosodium urate crystal deposit in joints, kidneys and tissues, as a consequence of hyperuricemia. Until now, the future for gouty patients largely depends on whether the best ways of management for gout are widely spread, since we already have excellent standards for diagnosis and very effective chemical and herbal treatments for most patients. Unfortunately, these treatments were hampered by the less knowledge of our genetics, foods nature as well as our bad lifestyle and eating habits which reflect their repercussions on our general health.
This review article focuses on the different types of foods present in our diet in relation to uric acid levels as some dietary plant foods may be low, moderate or even high in uric acid contents. It also point out on how the different life forms (human, animals, plants and microbes) can genetically handle uric acid metabolism and catabolism. Attentions were made on the various mechanisms by which plant secondary metabolites and microbes (bacteria, fungi and actinomycetes) enzymes’ degrade uric acid to soluble ammonia.
Future perspectives must be made in the way of increasing the awareness of populations to these open areas of research basing on the statement ‘prevention is better than cure’. Major advances should also focus on the manufacture of recombinant probiotic microorganisms carrying uricase genes to use it in the treatment of gout in addition to the present chemical and herbal treatments.
Conflict of interest
The authors have declared no conflict of interest.
Compliance with Ethics Requirements
This article does not contain any studies with human or animal subjects.
Biographies

Rehab M. Hafez is a Lecturer of Plant Cytogenetics from Botany and Microbiology Department, Faculty of Science, Cairo University, Egypt. Her research interests lie in the area of Cytogenetics in relation with tissue culture, transformation, biotechnology and nanotechnology. She published one paper and 6 abstract in conferences. She supervises on 3 M.Sc. and one Ph.D. theses. She participates in different committees in her Department as well as The Egyptian society of Botany. She attended numerous training courses; educational, in quality assurance and accreditation and in ISO 9001/20015. She attended 8 scientific training courses, one symposium and 5 international conferences.

Tahany M. Abdel-Rahman is a Professor of Microbiology (since1992) in Botany and Microbiology Department, Faculty of Science, Cairo University. She completed her B.Sc. (1969), M.Sc. Microbiology (1974) and Ph.D. Microbiology (1980). She published over 70 papers in Microbiology and she supervised on 30 M.Sc. and 16 PhD students. She assumed several positions in her Faculty; Deputy Director of Microanalytical Center, Vice Dean for Postgraduate Studies, Vice Chancellor for Scientific committee for promotion of Associate Professor, Deputy Director for project of medicinal plant sustainability and for project of Extraction of medicinally active compound from wild plants.

Rasha M. Naguib is the head of Microbiology Section in Microanalytical Center, Faculty of Science, Cairo University (since 2004). She took her B.Sc. in Botany/Chemistry (2004), M.Sc. in Microbiology (2008) and Ph.D. in Microbiology (2013). She worked as Chemist for 5 months in 2004 at Blood Bank, Cairo University hospitals (El-Kasr El- Eini). She attends 5 scientific workshops, one conference as well as 6 medical and scientific training courses. She performs seasonal teaching and training programs through the Micro Analytical Center (since 2004). She also supervises the graduation project for 4th year students of Biotechnology/Bio-molecular Chemistry Program (2015–2016).
Footnotes
Peer review under responsibility of Cairo University.
References
- 1.Maples K.R., Ronald P.M. Free radical metabolite of uric acid. J Biolog Chem. 1988;263(4):1709–1712. [PubMed] [Google Scholar]
- 2.Cheng X., Changgui L. Review. The principles of gout therapy. Gout Hyperuricemia. 2015;2(1):15–23. [Google Scholar]
- 3.Xiang L.-W., Li J., Lin J.-M., Li H.-F. Determination of gouty arthritis' biomarkers in human urine using reversed-phase high-performance liquid chromatography. J Pharm Anal. 2014;4(2):153–158. doi: 10.1016/j.jpha.2013.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Unno T., Akio S., Takami K. Xanthine oxidase inhibitors from the leaves of Lagerstroemia speciosa (L.) Pers. J Ethnopharmacol. 2004;93(2–3):391–395. doi: 10.1016/j.jep.2004.04.012. [DOI] [PubMed] [Google Scholar]
- 5.Koulouris S. Gout and purines. In: Experiments on battling gout and living a healthier life; 2016 [2016 Oct 15]. Available from: <https://goutandyou.com/purines/>.
- 6.Jarmai Durch schimmeligen Mais verursachte Gicht bei Gausen. Deutsche Tierarzil Wchnschr. 1925;33:580–582. [Google Scholar]
- 7.Hutyra F, Marek J. Special pathology and therapeutics of the diseases of domestic animals. Alexander Eger, Chikago, vol. III; 1926.
- 8.Pegram R.A., Wyatt R.D. Avian gout caused by oosporein, a mycotoxin produced by Chaetomium trilaterale. Poult Sci. 1981;60:2429. doi: 10.3382/ps.0602429. [DOI] [PubMed] [Google Scholar]
- 9.Pegram R.A., Wyatt R.D., Smith T.L. Oosporein-toxicosis in the turkey poult. Avian Dis. 1982;26(1):47–59. [PubMed] [Google Scholar]
- 10.Costantini A.V. The fungal etiology of gout and hyperuricemia: the antifungal mode of action of colchicine. Biomed Rev. 1992;1:47–52. [Google Scholar]
- 11.Svihla G., Dainko J.L., Schlenk F. Ultraviolet microscopy of purine compounds in the yeast vacuole. J Bacht. 1963;85:399–409. doi: 10.1128/jb.85.2.399-409.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Krogh P., Hald B., Gjertsen P., Myken F. Fate of ochratoxin A and citrinin during malting and brewing experiments. Appl Microbiol. 1974;28:31–34. doi: 10.1128/am.28.1.31-34.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chu F.S., Chang C.C., Ashoor S.H., Prentice N. Stability of aflatoxin Bl and ochratoxin A in brewing. Appl Microbiol. 1975;29:313–316. doi: 10.1128/am.29.3.313-316.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nip W.K., Chang F.C., Chu F.S., Prentice N. Fate of ochratoxin A in brewing. Appl Microbiol. 1975;30:1048–1049. doi: 10.1128/am.30.6.1048-1049.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nikolenko lul, Siniachenko O.V., Diadyk Al. Antideoxyribonucleic acid antibodies in podagra. Revmatologiia (Mosk) 1989;2:30–35. [PubMed] [Google Scholar]
- 16.Edozien C., Udo U., Young V.R., Schrmshaw N.S. Effects of high levels of yeast feeding on uric acid metabolism of young men. Nature. 1970;228(5267):180. doi: 10.1038/228180a0. [DOI] [PubMed] [Google Scholar]
- 17.Talbott JH. Gout, 2nd ed. Grune and Stratton, New York; 1964, p. 145.
- 18.Bourgeosis C.H., Shank R.C., Grossman R.A., Johnson D.O., Wooding W.L., Chandavimol P. Acute aflatoxin Bl toxicity in the macaque and its similarity to Reye 's syndrome. Lab Invest. 1971;24:206–216. [PubMed] [Google Scholar]
- 19.Sommers S.C., Churg J. Kidney pathology in hyperuricemia and gout. In: Yü T., Burger L., editors. The Kidney in Gout and Hyperuricemia. Futura Publishing Company; Mount Kisco, New York: 1982. p. 292. [Google Scholar]
- 20.Kossa J. Kunstliche Erzeugung der Gicht durch Gifte. Arch Internal Pharmacodyn. 1899;5:97–109. [Google Scholar]
- 21.West C., Carpenter B.J., Hakala T.R. The incidence of gout in renal transplant recipients. Am J Kidney Dis. 1987;10(5):369–372. doi: 10.1016/s0272-6386(87)80103-8. [DOI] [PubMed] [Google Scholar]
- 22.Gores P.F., Fryd D.S., Sutherland D.E., Najarian J.S., Simmons R.L. Hyperuricemia after renal transplantation. Am J Surg. 1988;156(5):397–400. doi: 10.1016/s0002-9610(88)80196-x. [DOI] [PubMed] [Google Scholar]
- 23.Lin H.Y., Rocher L.L., McQuillan M.A., Schmaltz S., Palella T.D., Fox I.H. Cyclosporine-induced hyperuricemia and gout. N Engl J Med. 1989;321(5):287–292. doi: 10.1056/NEJM198908033210504. [DOI] [PubMed] [Google Scholar]
- 24.Nogaim Q.A., Amra H.A.S., Nada S.A. The medical effects of edible mushroom extract on aflatoxin B1. J Biol Sci. 2011;11:481–486. [Google Scholar]
- 25.Wu X.W., Lee C.C., Muzny D.M., Caskey C.T. Urate oxidase: primary structure and evolutionary implications. Proc Natl Acad Sci (USA) 1989;86:9412–9416. doi: 10.1073/pnas.86.23.9412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cheristians S., Kaltwasser H. Nickel-content of urease from Bacillus pasteurii. Arch Microbiol. 1986;145:51–55. doi: 10.1007/BF00413026. [DOI] [PubMed] [Google Scholar]
- 27.Rando D., Steglitz U., Mörsdorf G., Kaltwasser H. Nickel availability and urease expression in Proteus mirabilis. Arch Microbiol. 1990;154:428–432. doi: 10.1007/BF00245222. [DOI] [PubMed] [Google Scholar]
- 28.Nakagawa S., Ishino S., Teshiba S. Construction of catalase deficient Escherichia coli strains of the production of uricase. Biosci Biotech Bioch. 1996;60:415–420. doi: 10.1271/bbb.60.415. [DOI] [PubMed] [Google Scholar]
- 29.Ammar M.S., Elwan S.H., El-Shahed A.S. A uricolytic Streptomyces albogriseolus from an Egyptian soil. Taxonomy and uricase production and properties. J. Microbiol. 1987;22:261–279. [Google Scholar]
- 30.Zhou X.L., Ma X.H., Sun G.Q., Li X., Guo K.P. Isolation of a thermostable uricase producing bacterium and study on its enzyme production conditions. Process Biochem. 2005;40:3749–3753. [Google Scholar]
- 31.Lofty W.A. Production of a thermostable uricase by a novel Bacillus thermocatenulatus strain. Bioresour Technol. 2008;99:699–702. doi: 10.1016/j.biortech.2007.01.048. [DOI] [PubMed] [Google Scholar]
- 32.Tanaka A., Yamamura M., Kawamoto S., Fukui S. Production of uricase by Candida tropicalis using n-alkane as substrate. Appl Environ Microb. 1977;34:342–346. doi: 10.1128/aem.34.4.342-346.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abd El Fattah Y.R., Saeed H.M., Gohar Y.M., El-Baz M.A. Improved production of Pseudomonas aeruginosa uricase by optimization of process parameters through statistical experimental designs. Process Biochem. 2005;40:1707–1714. [Google Scholar]
- 34.Lee I.R., Yang L., Sebetso G., Allen R., Doan Thi H.N., Ross B. Characterization of the complete uric acid degradation pathway in the Fungal Pathogen Cryptococcus neoformans. PLoS ONE. 2013;8(5):1–13. doi: 10.1371/journal.pone.0064292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gabison L., Chiadmi M., El Hajji M., Castro B., Colloc’h N., Prange T. Near-atomic resolution structures of urate oxidase complexed with its substrate and analogues: the protonation state of the ligand. Acta Crystallogr D. 2010;66:714–724. doi: 10.1107/S090744491001142X. [DOI] [PubMed] [Google Scholar]
- 36.Hesberg C., Hänsch R., Bittner F. Tandem orientation of duplicated xanthine dehydrogenase genes from Arabidopsis thaliana: differential gene expression and enzyme activities. J Biol Chem. 2004;279:13547–13554. doi: 10.1074/jbc.M312929200. [DOI] [PubMed] [Google Scholar]
- 37.Kahn K., Tipton P.A. Spectroscopic characterization of intermediates in the urate oxidase reaction. Biochem. 1998;37:11651–11659. doi: 10.1021/bi980446g. [DOI] [PubMed] [Google Scholar]
- 38.Triplett E.W., Blevins D.G., Randall D.D. Purification and properties of soybean nodule xanthine dehydrogenase. Arch Biochem Biophys. 1982;219:39–46. doi: 10.1016/0003-9861(82)90131-x. [DOI] [PubMed] [Google Scholar]
- 39.Vogels G.D., Van der Drift C. Degradation of purines and pyrimidines by microorganisms. Bacteriol Rev. 1976;40:403–468. doi: 10.1128/br.40.2.403-468.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yesbergenova Z., Yang G., Oron E., Soffer D., Fluhr R., Sagi M. The plant Mo-hydroxylases aldehyde oxidase and xanthine dehydrogenase have distinct reactive oxygen species signatures and are induced by drought and abscisic acid. Plant J. 2005;42:862–876. doi: 10.1111/j.1365-313X.2005.02422.x. [DOI] [PubMed] [Google Scholar]
- 41.Marzluf G.A. Genetic regulation of nitrogen metabolism in the fungi. Microbiol Mol Biol Rev. 1997;61:17–32. doi: 10.1128/mmbr.61.1.17-32.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Magasanik B. Global regulation of gene expression. Proc Natl Acad Sci (USA) 2000;97:14044–14045. doi: 10.1073/pnas.97.26.14044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wong K.H., Hynes M.J., Davis M.A. Recent advances in nitrogen regulation: a comparison between Saccharomyces cerevisiae and filamentous fungi. Eukaryot Cell. 2008;7:917–925. doi: 10.1128/EC.00076-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gravenmade E.J., Vogel G.D., Van der Drift C. Hydrolysis, racemization and absolute configuration of ureidoglycolate, a substrate of allantoicase. Biochim Biophys Acta. 1970;198:569–582. doi: 10.1016/0005-2744(70)90134-8. [DOI] [PubMed] [Google Scholar]
- 45.Todd C.D., Polacco J.C. AtAAH encodes a protein with allantoate amidohydrolase activity from Arabidopsis thaliana. Planta. 2006;223:1108–1113. doi: 10.1007/s00425-006-0236-x. [DOI] [PubMed] [Google Scholar]
- 46.Van der Drift C., de Windt F.E., Vogels G.D. Allantoate hydrolysis by allantoate amidohydrolase. Arch Biochem Biophys. 1970;136:273–279. doi: 10.1016/0003-9861(70)90351-6. [DOI] [PubMed] [Google Scholar]
- 47.Werner A.K., Sparkes I.A., Romeis T., Witte C.-P. Identification, biochemical characterization, and subcellular localization of allantoate amidohydrolases from Arabidopsis and soybean. Plant Physiol. 2008;146:418–430. doi: 10.1104/pp.107.110809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Winkler R.G., Polacco J.C., Blevins D.G., Randall D.D. Enzymic degradation of allantoate in developing soybeans. Plant Physiol. 1985;79:787–793. doi: 10.1104/pp.79.3.787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.El-Naggar M.R., Emara H.A. On the occurrence and identity of uricolytic microorganisms in Asiri soils Proc Saudi. Biol Soc. 1980;4:171–178. [Google Scholar]
- 50.Koch E., Lozada M., Dionisi H., Castro-Vazquez A. Uric acid-degrading bacteria in the gut of the apple snail Pomacea canaliculata and their possible symbiotic significance. Symbiosis. 2014;63:149–155. [Google Scholar]
- 51.Vega I.A., Giraud-Billoud M., Koch E., Gamarra-Luques C., Castro-Vega I.A., Giraud-Billoud M. Uric acid accumulation within intracellular crystalloid corpuscles of the midgut gland in Pomacea canaliculata (Caenogastropoda, Ampullariidae) Veliger. 2007;48:276–283. [Google Scholar]
- 52.Giraud-Billoud M., Koch E., Vega I.A., Gamarra-Luques C., Castro-Vazquez A. Urate cells and tissues in the South American apple-snail Pomacea canaliculata. J Molluscan Stud. 2008;74:259–266. [Google Scholar]
- 53.Giraud-Billoud M., Abud M.A., Cueto J.A., Vega I.A., Castro-Vazquez A. Uric acid deposits and estivation in the invasive apple-snail. Pomacea canaliculata. Comp Biochem Physiol. 2011;158(Part A):506–512. doi: 10.1016/j.cbpa.2010.12.012. [DOI] [PubMed] [Google Scholar]
- 54.Giraud-Billoud M., Vega I.A., Rinaldi Tosi M.E., Abud M.A., Calderón M.L., Castro-Vazquez A. Antioxidant and molecular chaperone defenses during estivation and arousal in the South American apple-snail Pomacea canaliculata. J Exp Biol. 2013;216:614–622. doi: 10.1242/jeb.075655. [DOI] [PubMed] [Google Scholar]
- 55.Magda A., Sanaa T., Saleh A., Reda A. Production and characterization of uricase from Streptomyces exfoliates UR10 isolated from farm wastes. Turk J Biol. 2013;37:520–529. [Google Scholar]
- 56.Zhuang G., Jiachao Z., Zhanli W., Kay Ying A., Shi H., Qiangchuan H. Intestinal microbiota distinguish gout patients from healthy humans. Sci Rep. 2016:1–10. doi: 10.1038/srep20602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ogawa J. Division of Applied Life Sciences, Graduate School of Agriculture, Kyoto University; 2006. Analysis of Microbial Purine Metabolism and Its Application for Hyperuricemia Prevention; pp. 32–34. [Google Scholar]
- 58.Alsultanee I.R., Ewadh M.J., Mohammed M.F. Novel natural anti gout medication extract from Momdica charantia. J Nat Sci Res. 2014;4(17):16–23. [Google Scholar]
- 59.The George Mateljan Foundation [Internet]; 2016 [updated 2017 April; cited 2016 Oct]. Available from: <http://www.whfoods.com/genpage.php?tname=george&dbid=51>.
- 60.Kerns M. List of foods and vegetables that raise uric acid; 2010 [Last Updated 2010 Nov 30]. Available from: <http://www.livestrong.com/article/321742-list-of-foods-vegetables-that-raise-uric-acid/>.
- 61.Halevi S. Various food types and their purine content; 2016. In: AcuMedico, Chinese medicine articles [update 2016 June]. Available from: <http://www.acumedico.com/purine.htm>.
- 62.Cooper E. List of uric acid foods; 2009 [Last Updated 2009 Oct 02]. Available from: <http://www.livestrong.com/article/26296-list-uric-acid-foods/>.
- 63.Liote F. Hyperuricemia and gout. Curr Rheumatol Rep. 2003;5:227–234. doi: 10.1007/s11926-003-0072-y. [DOI] [PubMed] [Google Scholar]
- 64.Matata B.M., Elahi M.M. Sources of reactive oxidants species in biology and disease. Oxid Stress. 2007:23–38. [Google Scholar]
- 65.Harris M.D., Siegel L.B., Alloway J.A. Gout and hyperuricemia. Am Fam Physician. 1999;59(4):925–934. [PubMed] [Google Scholar]
- 66.Kong L.D., Cai Y., Huang W.W., Cheng C.H.K., Tan R.X. Inhibition of xanthine oxidase by some Chinese medicinal plants used to treat gout. J Ethnopharmacol. 2000;73:199–207. doi: 10.1016/s0378-8741(00)00305-6. [DOI] [PubMed] [Google Scholar]
- 67.Sweeney A.P., Wyllie S.G., Shalliker R.A., Markham J.L. Xanthine oxidase inhibitory activity of selected Australian native plants. J Ethnopharmacol. 2001;75:273–277. doi: 10.1016/s0378-8741(01)00176-3. [DOI] [PubMed] [Google Scholar]
- 68.Bustanji Y., Hudaid M., Tawaha K., Mohammad M.K., Almasri I., Hamad S. In vitro xanthine oxidase inhibition by selected Jordanian medicinal plants. Jord J Pharm Sci. 2011;4(1):49–56. [Google Scholar]
- 69.Fagugli R.M., Gentile G., Ferrara G., Brugnano R. Acute renal and hepatic failure associated with allopurinol treatment. Clin Nephrol. 2008;70:523–526. doi: 10.5414/cnp70523. [DOI] [PubMed] [Google Scholar]
- 70.Burke A., Smyth E., FitzGerald G.A. Analgesic – antipyretic agents; pharmacotherapy of gout. In: Brunton L.L., Lazo J.S., Parker K.L., editors. The pharmacological basis of therapeutics. 11th ed. McGraw–Hill Medical Publishing Division; New York: 2006. pp. 706–710. [Google Scholar]
- 71.Blando F., Gerardi C., Nicoletti I. Sour cherry (Prunus cerasus L.) anthocyanins as ingredients for functional foods. J Biomed Biotechnol. 2004:253–258. doi: 10.1155/S1110724304404136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hollands W., Brett G.M., Dainty J.R., Teucher B., Kroon P.A. Urinary excretion of strawberry anthocyanins is dose dependent for physiological oral doses of fresh fruit. Mol Nutr Food Res. 2008;52:1097–1105. doi: 10.1002/mnfr.200700372. [DOI] [PubMed] [Google Scholar]
- 73.Wang H., Nair M.G., Strasburg G.M., Chang Y.C., Booren A.M., Gray J.I. Antioxidant and antiinflammatory activities of anthocyanins and their aglycon, cyanidin, from tart cherries. J Nat Prod. 1999;62:294–296. doi: 10.1021/np980501m. [DOI] [PubMed] [Google Scholar]
- 74.Theimer R.R., Beevers H. Uricase and allantoinase in glyoxysomes. Plant Physiol. 1971;47:246–251. doi: 10.1104/pp.47.2.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Parish R.W. Urate oxidase in peroxisomes from maize root tips. Planta. 1972;104:247–251. doi: 10.1007/BF00387079. [DOI] [PubMed] [Google Scholar]
- 76.Hanks J.F., Tolbert N.E., Schubert K.R. Localization of enzymes of ureide biosynthesis in peroxisomes and microsomes of nodules. Plant Physiol. 1981;68:65–69. doi: 10.1104/pp.68.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Christensen T.M.I.E., Jochimsen B.U. Enzymes of ureide synthesis in pea and soybean. Plant Physiol. 1983;72:56–59. doi: 10.1104/pp.72.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Montalbini P., Redondo J., Caballero J.L., Cárdenas J., Pineda M. Uricase from leaves: its purification and characterization from three different higher plants. Planta. 1997;202:277–283. [Google Scholar]
- 79.Mohamed D.A., Al-Okbi S.Y. Evaluation of anti-gout activity of some plant food extracts. Pol J Food Nutr Sci. 2008;58(3):389–395. [Google Scholar]
- 80.Raju R., Sigimol J., Soniya S., Santhosh M.M., Umamheshwari M. Effect of the fractions of Erythrina stricta leaf extract on serum urate levels and Xo/Xdh activities in oxonate-induced hyperuricaemic mice. J Appl Pharmac Sci. 2012;02(02):89–94. [Google Scholar]
- 81.Mahdabadi M.N., Zahra K., Nadia T.B., Farzaneh L., Asma J., Seyed H.M. RhusCoriaria effect on serum uric acid level and in vivo xanthine oxidase activity in oxonate-induced hyperuricemic mice. J Pharm Biomed Sci. 2013;3(12):1–6. [Google Scholar]
- 82.Gdoura N., Murat J.C., Abdelmouleh A., Elfeki A. Effects of Juniperus phoenicea extract on uricemia and activity of antioxidant enzymes in liver, erythrocyte and testis of hyperuricemic (oxonate-treated) rats. Afr J Pharm Pharmacol. 2013;7(8):416–425. [Google Scholar]
- 83.Al-Azzawie H.F., Abd S.A. Effects of crude flavonoids from ginger (Zingiber officinale), on serum uric acid levels, biomarkers of oxidative stress and xanthine oxidase activity in oxonate-induced hyperuricemic rats. Inter J Adv Res. 2015;3(10):1033–1039. [Google Scholar]
- 84.Vasudeva N., Prerna S., Sneha Das, Surendra K.S. Antigout and antioxidant activity of stem and root of Origanum majorana Linn. Am J Drug Dis Devel. 2014;4(2):102–112. [Google Scholar]
- 85.Bell P.G., David C.G., Gareth W.D., Trevor W.G., Michael J.S., Glyn H. Montmorency tart cherry (Prunus cerasus L.) concentrate lowers uric acid, independent of plasma cyanidin-3-O-glucosiderutinoside. J Funct Foods. 2014;11:82–90. [Google Scholar]
- 86.Murugaiyah V., Chan K.-L. Mechanisms of antihyperuricemic effect of Phyllanthus niruri and its lignin constituents. J Ethnopharmacol. 2009;124:233–239. doi: 10.1016/j.jep.2009.04.026. [DOI] [PubMed] [Google Scholar]
- 87.Werner A.K., Nieves M.-E., Monika Z., Imogen A.S., Feng-Qiu C., Claus-Peter W. The ureide-degrading reactions of purine ring catabolism employ three amidohydrolases and one aminohydrolase in Arabidopsis, soybean, and rice. Plant Physiol. 2013;163:672–681. doi: 10.1104/pp.113.224261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Krakoff I.H. Discussion of conference on gout and purine metabolism. Arthritis Rheum. 1965;8:760. [Google Scholar]
- 89.Ahern M.J., Reid C., Gordon T.P., McCredie M., Brooks P.M., Jones M. Does colchicine work? The results of the first controlled study in acute gout. Austr New Zealand J Med. 1987;17(3):301–304. doi: 10.1111/j.1445-5994.1987.tb01232.x. [DOI] [PubMed] [Google Scholar]
- 90.Famaey J.P. Colchicine in therapy. State of the art and new perspectives for an old drug. Clin Exp Rheumatol. 1988;6(3):305–317. [PubMed] [Google Scholar]
- 91.Dalbeth N., Lauterio T.J., Wolfe H.R. Mechanism of action of colchicine in the treatment of gout. Clin Ther. 2014;36(10):1465–1479. doi: 10.1016/j.clinthera.2014.07.017. [DOI] [PubMed] [Google Scholar]
- 92.Schultz A.C., Nygaard P., Saxild H.H. Functional analysis of 14 genes that constitute the purine catabolic pathway in Bacillus subtilis and evidence for a novel regulon controlled by the PucR transcription activator. J Bacteriol. 2001:3293–3302. doi: 10.1128/JB.183.11.3293-3302.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kratzer J.T., Lanaspac M.A., Murphyd M.N., Cicerchic C., Gravese C.L., Tiptonf P.A. Evolutionary history and metabolic insights of ancient mammalian uricases. PNAS. 2014;11(10):3763–3768. doi: 10.1073/pnas.1320393111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hruban Z., Swift H. Uricase: Localization in hepatic microbodies. Science. 1964;146(3649):1316–1318. doi: 10.1126/science.146.3649.1316. [DOI] [PubMed] [Google Scholar]
- 95.Wu X.W., Lee C.C., Muzny D.M., Caskey C.T. Urate oxidase: primary structure and evolutionary implications. Proc Natl Acad Sci USA. 1989;86(23):9412–9416. doi: 10.1073/pnas.86.23.9412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wu X.W., Muzny D.M., Lee C.C., Caskey C.T. Two independent mutational events in the loss of urate oxidase during hominoid evolution. J Mol Evol. 1992;34(1):78–84. doi: 10.1007/BF00163854. [DOI] [PubMed] [Google Scholar]
- 97.Yeldandi A.V., Vijay Y., Sujata K., Narasimha M.C.V., Xuedong W., Alvares K. Molecular evolution of the urate oxidase-encoding gene in hominoid primates: nonsense mutations. Gene. 1991;109(2):281–284. doi: 10.1016/0378-1119(91)90622-i. [DOI] [PubMed] [Google Scholar]
- 98.Oda M., Satta Y., Takenaka O., Takahata N. Loss of urate oxidase activity in hominoids and its evolutionary implications. Mol Biol Evol. 2002;19(5):640–653. doi: 10.1093/oxfordjournals.molbev.a004123. [DOI] [PubMed] [Google Scholar]
- 99.Yokota S., Kamijo K., Oda T. Degradation of overexpressed wild-type and mutant uricase proteins in cultured cells. J Histochem Cytochem. 1999;47(9):1133–1140. doi: 10.1177/002215549904700905. [DOI] [PubMed] [Google Scholar]
- 100.Ashihara H., Crozier A. Biosynthesis and metabolism of caffeine and related purine alkaloids in plants. Adv Bot Res. 2000;30:117–205. [Google Scholar]
- 101.Mendel R.R., Schwarz G. Molybdoenzymes and molybdenum cofactor in plants. Crit Rev Plant Sci. 1999;18:33–69. [Google Scholar]
- 102.Schubert KR, Boland MJ. The ureides, p. 197–282. In Stumph PK, Cohn EE, editors. The biochemistry of plants. Academic Press Inc, New York, N.Y.; 1990.
- 103.Matsumoto H., Ohta S., Kobayashi R., Terawaki Y. Chromosomal location of genes participating in the degradation of purines in Pseudomonas aeruginosa. Mol Gen Genet. 1978;167:165–176. doi: 10.1007/BF00266910. [DOI] [PubMed] [Google Scholar]
- 104.Maynes J.T., Yuan R.G., Snyder F.F. Identification, expression and characterization of Escherichia coli guanine deaminase. J Bacteriol. 2000;182:4658–4660. doi: 10.1128/jb.182.16.4658-4660.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Xi H., Schneider B.L., Reitzer L. Purine catabolism in Escherichia coli and function of xanthine dehydrogenase in purine salvage. J Bacteriol. 2000;182:5332–5341. doi: 10.1128/jb.182.19.5332-5341.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cusa E., Obradors N., Baldoma L., Badia J., Aguilar J. Genetic analysis of a chromosomal region containing genes required for assimilation of allantoin nitrogen and linked glyoxylate metabolism in Escherichia coli. J Bacteriol. 1999;181:7479–7484. doi: 10.1128/jb.181.24.7479-7484.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Fluri R., Kinghorn J.R. The all2 gene is required for the induction of the purine deamination pathway in Schizosaccharomyces pombe. J Gen Microbiol. 1985;131:527–532. doi: 10.1099/00221287-131-3-527. [DOI] [PubMed] [Google Scholar]
- 108.Huang S.-H., Wu T.-K. Modified colorimetric assay for uricase activity and a screen for mutant Bacillus subtilis uricase genes following StEP mutagenesis. Eur J Biochem. 2004;271:517–523. doi: 10.1046/j.1432-1033.2003.03951.x. [DOI] [PubMed] [Google Scholar]
- 109.Yutaka A., Hiroshi I., Hiroomi N., Tsugutoshi A., Mitsutatk Y. Effects of serum bilirubin on determination of uric acid by the uricase-peroxidase coupled reaction. Clin Chem. 1992;38:1350–1352. [PubMed] [Google Scholar]
- 110.Bhargava A.K., Lal H., Pundir C.S. Discrete analysis of serum uric acid with immobilized uricase and peroxidase. J Biochem Biophys Methods. 1999;39:125–136. doi: 10.1016/s0165-022x(99)00007-x. [DOI] [PubMed] [Google Scholar]
- 111.Nanjo M., Guilbault G.G. Enzyme electrode sensing oxygen for uric acid in serum and urine. Anal Chem. 1974;46:1769–1772. doi: 10.1021/ac60348a058. [DOI] [PubMed] [Google Scholar]
- 112.Uchiyama S., Shimizu H., Hasebe Y. Chemical amplification of uric acid sensor responses by dithiothreitol. Anal Chem. 1991;66(1):873–1876. [Google Scholar]
- 113.Miland E., Ordieres A.J.M., Blanco P.T., Smyth C.O. Poly (o-aminophenol)-modified bienzyme carbon paste electrode for the detection of uric acid. Talanta. 1996;43:785–796. doi: 10.1016/0039-9140(95)01825-5. [DOI] [PubMed] [Google Scholar]
- 114.Marzluf G.A. Genetic regulation of nitrogen metabolism in the fungi. Microbiol Mol Biol Rev. 1997;61:17–32. doi: 10.1128/mmbr.61.1.17-32.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Suarez T., Oestreicher N., Kelly J., Ong G., Sankarsingh T., Scazzocchio C. The uaY positive control gene of Aspergillus nidulans: fine structure, isolation of constitutive mutants and reversion patterns. Mol Gen Genet. 1991;230:359–368. doi: 10.1007/BF00280292. [DOI] [PubMed] [Google Scholar]
- 116.Suarez T., Oestreicher N., Penalva M.A., Scazzocchio C. Molecular cloning of the uaY regulatory gene of Aspergillus nidulans reveals a favored region for DNA insertions. Mol Gen Genet. 1991;230:369–375. doi: 10.1007/BF00280293. [DOI] [PubMed] [Google Scholar]
- 117.Suarez T., de Queiroz M.V., Oestreicher N., Scazzocchio C. The sequence and binding specificity of UaY, the specific regulator of the purine utilization pathway in Aspergillus nidulans, suggest an evolutionary relationship with the PPR1 protein of Saccharomyces cerevisiae. EMBO J. 1995;14:1453–1467. doi: 10.1002/j.1460-2075.1995.tb07132.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Schultes N.P., Brutnell T.P., Allen A., Dellaporta S.L., Nelson T., Chen J. Leaf permease1 gene of maize is required for chloroplast development. Pl Cell. 1996;8:463–475. doi: 10.1105/tpc.8.3.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Argyrou E., Sophianopoulou V., Schultes N., Diallinas G. Functional characterization of a maize purine transporter by expression in Aspergillus nidulans. Pl Cell. 2001;13:953–964. doi: 10.1105/tpc.13.4.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Desimone M., Catoni E., Ludewig U., Hilpert M., Schneider A., Kunze R. A novel superfamily of transporters for allantoin and other oxoderivatives of nitrogen heterocyclic compounds in Arabidopsis. Pl Cell. 2002;14:847–856. doi: 10.1105/tpc.010458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Schmidt A., Su Y.-H., Kunze R., Warner S., Hewitt M., Slocum R.D. UPS1 and UPS2 from Arabidopsis mediate high affinity transport of uracil and 5-fluorouracil. J Biol Chem. 2004;279:44817–44824. doi: 10.1074/jbc.M405433200. [DOI] [PubMed] [Google Scholar]
- 122.Pélissier H.C., Frerich A., Desimone M., Schumacher K., Tegeder M. PvUPS1, an allantoin transporter in nodulated roots of French bean. Plant Physiol. 2004;134:664–675. doi: 10.1104/pp.103.033365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Mourad G.S., Snook B.M., Prabhakar J.T., Mansfield T.A., Schultes N.P. A fluoroorotic acid-resistant mutant of Arabidopsis defective in the uptake of uracil. J Exp Bot. 2006;57(14):3563–3573. doi: 10.1093/jxb/erl107. [DOI] [PubMed] [Google Scholar]
- 124.Diallinas G., Gorfinkiel L., Arst H.N., Jr., Ceccheto G., Scazzocchio C. Genetic and molecular characterization of a gene encoding a wide specificity purine permease of Aspergillus nidulans reveals a novel family of transporters conserved in prokaryotes and eukaryotes. J Biol Chem. 1995;270:8610–8622. doi: 10.1074/jbc.270.15.8610. [DOI] [PubMed] [Google Scholar]
- 125.Cecchetto G., Amillis S., Diallinas G., Scazzocchio C., Drevet C. The AzgA purine transporter of Aspergillus nidulans: characterisation of a protein belonging to a new phylogenetic cluster. J Biol Chem. 2004;279:3132–3141. doi: 10.1074/jbc.M308826200. [DOI] [PubMed] [Google Scholar]
- 126.Hauck O.K., Scharnberg J., Escobar N.M., Wanner G., Giavalisco P., Witte C.-P. Uric Acid Accumulation in an Arabidopsis urate oxidase mutant impairs seedling establishment by blocking peroxisome maintenance. Pl Cell. 2014;26:3090–3100. doi: 10.1105/tpc.114.124008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Theodoulou F.L., Eastmond P.J. Seed storage oil catabolism: a story of give and take. Curr Opin Pl Biol. 2012;15:322–328. doi: 10.1016/j.pbi.2012.03.017. [DOI] [PubMed] [Google Scholar]
- 128.Lamberto I., Percudani R., Gatti R., Folli C., Petrucco S. Conserved alternative splicing of Arabidopsis transthyretin like determines protein localization and S-allantoin synthesis in peroxisomes. Pl Cell. 2010;22:1564–1574. doi: 10.1105/tpc.109.070102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Serventi F., Ramazzina I., Lamberto I., Puggioni V., Gatti R., Percudani R. Chemical basis of nitrogen recovery through the ureide pathway: formation and hydrolysis of S-ureidoglycine in plants and bacteria. ACS Chem Biol. 2010;5:203–214. doi: 10.1021/cb900248n. [DOI] [PubMed] [Google Scholar]
- 130.Werner A.K., Romeis T., Witte C.P. Ureide catabolism in Arabidopsis thaliana and Escherichia coli. Nat Chem Biol. 2010;6:19–21. doi: 10.1038/nchembio.265. [DOI] [PubMed] [Google Scholar]
- 131.Werner A.K., Medina-Escobar N., Zulawski M., Sparkes I.A., Cao F.Q., Witte C.P. The ureide-degrading reactions of purine ring catabolism employ three amidohydrolases and one aminohydrolase in Arabidopsis, soybean, and rice. Plant Physiol. 2013;163:672–681. doi: 10.1104/pp.113.224261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Hongoh Y., Tetsuhiko S., Hajime I. Cloning, sequence analysis and expression in Escherichia coli of the gene encoding a uricase from the yeast-like symbiont of the brown planthopper, Nilaparvata lugens Insect. Biochem Mol Biol. 2000;30:173–182. doi: 10.1016/s0965-1748(99)00116-2. [DOI] [PubMed] [Google Scholar]
- 133.Yang J., Kyung-Hwan H. Functional characterization of allantoinase genes from Arabidopsis and a Nonureide-Type Legume Black Locust. Plant Physiol. 2004;134:1039–1049. doi: 10.1104/pp.103.034637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Chen Z., Zhaoyue W., Xiuping H., Xuena G., Weiwei L., Borun Z. Uricase production by a recombinant Hansenula polymorpha strain harboring Candida utilis uricase gene. Appl Microbiol Biotechnol. 2008;79:545–554. doi: 10.1007/s00253-008-1472-8. [DOI] [PubMed] [Google Scholar]
- 135.Oldfield V., Perry C.M. Rasburicase: a review of its use in the management of anticancer therapy-induced hyperuricaemia. Drugs. 2006;66:529–545. doi: 10.2165/00003495-200666040-00008. [DOI] [PubMed] [Google Scholar]
- 136.Koyama Y., Ichikawa T., Nakano E. Cloning, sequence analysis, and expression in Escherichia coli of the gene encoding the Candida utilis urate oxidase (uricase) J Biochem (Tokyo) 1996;120:969–973. doi: 10.1093/oxfordjournals.jbchem.a021514. [DOI] [PubMed] [Google Scholar]
- 137.Cheng X., Yang B., Liu D., He L.J., Chen G., Chen Y. Genetic engineering of bacteria that can produce urate oxidase. Intern Med. 2012;2:114–117. [Google Scholar]
- 138.Saeed H.M., Abdel-Fattah Y.R., Berekaa M.M., Gohar Y.M., Elbaz M.A. Identification, cloning and expression of Pseudomonas aeruginosa Ps-x Putative Urate Oxidase Gene in Escherichia coli. Polish J Microbiol. 2004;53(4):227–236. [PubMed] [Google Scholar]
- 139.Meraj M., Khalil-ur–Rahman, Amer J., Muhammad A., Rajoka M.I., Sadia J. Bacillus subtilis improvement through UV and chemical mutagenesis for indigenously hyperproduced urate oxidase. Pak J Life Soc Sci. 2012;10(2):123–129. [Google Scholar]