Abstract
Mutual regulation and balance between the endocrine and immune systems facilitate an organism’s stress response, and are impaired following chronic stress or prolonged immune activation. Concurrent alterations in stress physiology and immunity are increasingly recognized as contributing factors to several stress-linked neuropsychiatric disorders including depression, anxiety, and post-traumatic stress disorder. Accumulating evidence suggests that impaired balance and crosstalk between the glucocorticoid receptor (GR) and nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) – effectors of the endocrine and immune axes, respectively – may play a key role in mediating the harmful effects of chronic stress on mood and behavior. Here, we first review the molecular mechanisms of GR and NFκB interactions in health, then describe potential shifts in the GR-NFκB dynamics in chronic stress conditions within the context of brain circuitry relevant to neuropsychiatric diseases. Furthermore, we discuss developmental influences and sex differences in the regulation of these two transcription factors.
Keywords: glucocorticoid receptor, nuclear factor kappa-light-chain-enhancer of activated B cells, stress, depression, anxiety, PTSD, FΚBP5, inflammation, glucocorticoid resistance
Graphical abstract
1. Introduction
Balance between the endocrine and immune systems facilitates function of multiple organ systems, and disruption of this balance can precipitate a host of pathologies including neuropsychiatric diseases. Furthermore, due to the highly choreographed interactions among these systems, disruptions in one system quickly spread to the other system. These multifaceted interactions are illustrated by the coordination and cross-talk between the glucocorticoid receptor (GR) within the endocrine system and nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) within the immune system. These proteins are transcription factors with a delicate balance of interactions such that alterations in their functions and crosstalk lead to a host of physiological and behavioral changes. The balance between these transcription factors is tested when an organism is exposed to excessive stressor burdens, whether those stressors are environmental or psychological.
Transcription factors such as GR and NFκB are prime candidates to mediate the impact of wide-scale endocrine and inflammatory dysregulation because of their rapid activation, ability to regulate hundreds of genes at once, and many levels of regulation that allow fine-tuning of their induction and activity. Because of their opposing effects on inflammatory processes, the substantial mutual regulation and crosstalk between GR and NFκB help shape the effects of stress on the immune system, which in turn, regulates the stress response. During chronic stress, a dysfunctional or less sensitive GR leads to disinhibition of NFκB-mediated inflammation, and the resulting endocrine-immune imbalance is hypothesized to promote the harmful behavioral and physiological consequences of stress. Beyond the immediate context of inflammation, GR and NFκB also each independently regulate neurogenesis, synaptic plasticity, and neurotransmission, processes that are thought to contribute to the etiology of neuropsychiatric disorders. This review will focus on GR and NFκB as a means by which to highlight interactions among the endocrine and immune systems and detail the repercussions associated with dysfunction of these transcription factors. In order to accomplish this goal, the following areas are addressed:
-
2
Function and Regulation of GR and NFκB
-
3
Interactions Between GR and NFκB
-
4
Hallmarks of GR-NFκB dysfunction
-
5
Traversing the Blood Brain Barrier: from peripheral resistance to central mayhem
-
6
Influence of Age and Sex on GR-NFκB Interactions
2. Function and Regulation of GR and NFκB
2.1.1 Structure, Function, and Regulation of GR
The glucocorticoid receptor (GR) is a 777-amino acid nuclear receptor expressed throughout the body (Hollenberg, Weinberger et al. 1985) including the brain (McEwen 1973). The GR is encoded by the Nr3c1 gene and is responsive to its ligand glucocorticoids (Bell and Munck 1972). As a nuclear receptor, the GR contains a DNA binding domain and a ligand binding domain, both of which are required for nuclear receptor activity (Giguere, Hollenberg et al. 1986). GR resides in the cytosol interacting with a chaperone complex that maintains its activity (Levinson, Baxter et al. 1972, Bresnick, Dalman et al. 1989). Following binding of its glucocorticoid ligand, GR translocates to the nucleus (Davies, Ning et al. 2002) where it interacts with specific DNA sequences called glucocorticoid response elements (GREs). As a transcription factor, GR mediates transcription of numerous target genes involved in a range of actions within an organism including effects on metabolism, reproduction, immunity, and regulation of the hypothalamic-pituitary-adrenal (HPA) axis (Baxter, Rousseau et al. 1972, Rousseau, Higgins et al. 1975, Strahle, Klock et al. 1987, Panagiotakopoulos and Neigh 2014). Glucocorticoids also lead to the eventual resolution of prolonged HPA activity, and suppress their own secretion via binding to GR expressed in regulatory brain regions including the hippocampus, prefrontal cortex, and amygdala. Given the pleiotropic and pervasive effects of GR activation, efficient and effective regulation of receptor activity is essential to organism function and adaptation.
2.1.2 Mechanisms of GR Regulation
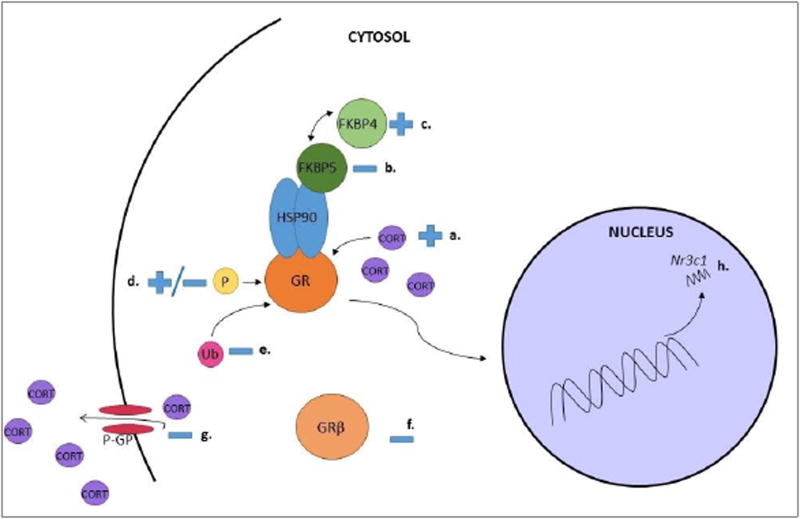
Cellular responsivity to glucocorticoids and activity of the GR can be regulated at multiple levels. The predominant regulatory mechanisms of GR regulation are discussed in the following sections, and an overview of mechanisms of GR regulation is detailed in Figure 1A.
Figure 1.
A. Molecular Regulation of GR Activity of the GR is regulated at multiple levels. Glucocorticoids (CORT), the ligand for the GR, promote nuclear translocation activity of the GR (a.). Co-chaperones of the GR such as FKBP5 (b.) and FKBP4 (c.) impair or promote GR nuclear translocation respectively. The GR is phosphorylated (d.) at multiple sites which may increase or decrease GR activity, and furthermore, the phosphorylation status of GR may alter the transcriptional profile, providing fine-tuning of the genes GR up or down-regulates. Ubiquitination (e.) promotes degradation of the GR, reducing GR activity. Expression of GRβ (f.) may decrease activity of the GR, and increased efflux of the ligand CORT from the cell due to increased p-glycoprotein activity (g.) may also decrease activity of GR. Altered transcription of Nr3c1 (h.) may also impact GR expression and activity.
B. Molecular Regulation of NFκB. The canonical and non-canonical NFκB pathways reside in the cytoplasm, tethered by the inhibitor protein IκBα/p100/p105. Following TAK1-mediated activation by TNF, IL-1, and Toll-like receptor transduction pathways, the canonical IKK complex phosphorylates IκBα, thus releasing the p65-p50 heterodimer to translocate to the nucleus. In the non-canonical pathway, following activation by lymphotoxin-β receptor or CD40 ligand signaling, IKKα phosphorylates p100/p105, thus triggering their cleavage into p52/p50. The newly freed p52/p50-RelB complex then translocates into the nucleus. The NFκB complex may be subject to further post-translational modifications including phosphorylation and acetylation prior to binding to the κREs.
2.1.2a Co-Chaperone Regulation of the GR
As a nuclear receptor, GR resides in the cytosol in a complex with a number of chaperones and co-chaperone proteins. The chaperones and co-chaperones impact function of the receptor, and by modulating the receptor’s activity have the ability to impact glucocorticoid signaling. Following translation of the GR protein, GR interacts with the chaperones heat shock protein 90, 70 (Hsp90, Hsp70), and p60 which are required for efficient folding of GR into a conformation with high-affinity for ligand binding, and interaction with the chaperone p23 further stabilizes the GR-Hsp90 complex (Whitesell and Cook 1996, Dittmar and Pratt 1997). GR resides in the cytosol bound to two molecules of Hsp90, and continued interaction of GR with the chaperone Hsp90 is required to maintain GR’s high-affinity conformation (Bresnick, Dalman et al. 1989). In addition to the two molecules of Hsp90, the GR cytosolic complex also contains an immunophilin. The immunophilins FK506 binding protein 51 (FKBP5) and FK506 binding protein 52 (FKBP4) have been widely studied for their regulatory activity of GR and established links to the expression of mood disorders (Binder 2009). FKBP5 and FKPB4 bind to Hsp90 through a tetratricopeptide repeat (TPR) domain. This domain, as well as amino acids in the immunophilin’s C-terminal domain, impact the immunophilin’s binding affinity to Hsp90 (Silverstein, Galigniana et al. 1999, Cheung-Flynn, Roberts et al. 2003). The GR cytosolic complex consists of two molecules of Hsp90, one molecule of GR, and one immunophilin (FKBP5 or FKBP4). These two proteins, FKBP5 and FKBP4, compete with each other for binding with Hsp90 (Wochnik, Ruegg et al. 2005) and dictate the feasibility of translocation.
Interaction of the GR complex with the immunophilins FKBP5 and FKBP4 have differential action on glucocorticoid signaling. While interaction with FKBP4 facilitates movement of GR to the nucleus, allowing GR to elicit its transcriptional activity, interaction of GR-Hsp90 with FKBP5 inhibits GR-mediated transcriptional activity and efficient translocation of GR to the nucleus. This occurs in part due to reduced interaction of FKBP5 with dynein compared to that of FKBP4 (Wochnik, Ruegg et al. 2005) (Tatro, Everall et al. 2009). When GR interacts with its corticosteroid ligand, the GR-Hsp90 complex dissociates from the co-chaperone FKBP5, and FKBP5 is exchanged for FKBP4. FKBP4 then associates with dynein, a motor protein that assists in nuclear translocation of the GR complex to the nucleus (Davies, Ning et al. 2002). The peptidyl prolyl isomerase (PPIase) domain of FKBP4 is required for association with dynein and movement of the GR complex to the nucleus (Silverstein, Galigniana et al. 1999, Galigniana, Radanyi et al. 2001, Galigniana, Harrell et al. 2002). Dynein then interacts with microtubules to assist in the nuclear transport of GR (Harrell, Murphy et al. 2004). The balance between FKBP5 and FKBP4 in part dictates the extent to which GR can translocate to the nucleus in order to act as a transcription factor.
2.1.2b GR Isoforms
Alternate splicing of GR results in multiple isoforms being expressed, the most commonly studied of which are GRα and GRβ. GRα is the traditional GR isoform that mediates transcriptional activity. Alternatively, GRβ is primarily located in the nucleus and inhibits GRα transcriptional activity (Oakley, Sar et al. 1996). Multiple sources have cited increased expression of the β isoform as a mechanism of glucocorticoid resistance in inflammatory disease (Leung, Hamid et al. 1997, Honda, Orii et al. 2000, Sousa, Lane et al. 2000, Kozaci, Chernajovsky et al. 2007) due to its ability to inhibit GRα-mediated transcriptional activity. However, the inhibitory activity of GRβ on GRα is not universally accepted (Hecht, Carlstedt-Duke et al. 1997), and some cite the relative scarcity of GRβ compared to GRα in multiple cell types to argue against the physiological relevance of GRβ mediating glucocorticoid resistance (Pujols, Mullol et al. 2002). More recent studies have found that GRβ, like GRα, is also able to translocate from the cytosol to the nucleus, a process that is dependent on Hsp90 and dynein for both isoforms (Zhang, Clark et al. 2006). Furthermore, interaction with immunophilin co-chaperones can also alter GRα and GRβ activity such that FKBP5 and FKBP4 have been found to interact with both GRα and GRβ (Zhang, Clark et al. 2008), and overexpression of FKPB5 increases nuclear translocation of GRβ which reduces transcriptional activity of GRα in the nucleus.
2.1.2c Post-translational Modifications
GR is post-translationally modified on multiple serine residues (Dalman, Sanchez et al. 1988), and can be phosphorylated following glucocorticoid stimuli at these serine residues (Orti, Mendel et al. 1989, Wang, Frederick et al. 2002). Altered patterns of GR phosphorylation can change transcriptional activity of GR. S211 phosphorylation is associated with increased transcriptional activity of GR and a conformational change that may facilitate interaction with cofactors at particular target genes (Wang, Frederick et al. 2002, Chen, Dang et al. 2008). However, this is not a universal effect, and the effect of S211 phosphorylation on gene transcription differs among specific GR target genes (Chen, Dang et al. 2008). Differential phosphorylation patterns of GR result in recruitment of GR to different target genes (Blind and Garabedian 2008) and altered mRNA transcript profiles (Galliher-Beckley, Williams et al. 2011), providing a mechanism of altering the transcriptional pattern of GR rather than just increased or decreased global activity.
Kinases and phosphatases have been key research tools in elucidating the role of phosphorylation in altering GR-mediated transcriptional patterns. P38 MAPK phosphorylates S211 of GR, and mutation of this site decreases GR’s transcriptional activity (Miller, Webb et al. 2005). Conversely, c-Jun N-terminal kinase (JNK) inhibits GR-activity and phosphorylates GR at S246 (Rogatsky, Logan et al. 1998, Szatmary, Garabedian et al. 2004) and S226 (Itoh, Adachi et al. 2002). JNK phosphorylation also increases nuclear export of GR (Itoh, Adachi et al. 2002), and JNK inhibition in HT22 cells dose-dependently increases GR’s transcriptional activity (Wang, Wu et al. 2005). This inhibitory activity of JNK-mediated phosphorylation is one mechanism that may contribute to glucocorticoid resistance in a nuanced gene-specific manner.
2.1.2d Receptor Expression and Degradation
Regulation of GR protein expression is an additional mechanism of modulating signaling activity of GR. Glucocorticoid treatment downregulates GR expression in cell models (Cidlowski and Cidlowski 1981, Silva, Powell-Oliver et al. 1994), and chronic corticosterone treatment decreases GR protein in specific brain regions (Hu, Zhang et al. 2016). However, inhibition of proteasomes reverses the effect of glucocorticoid treatment on GR expression, establishing the role of proteasomal degradation in glucocorticoid-mediated regulation of GR protein concentrations. Following treatment with glucocorticoids, the GR can be ubiquitinated, signaling its designation for proteasomal degradation. Changes in GR protein concentrations following glucocorticoid treatment have been found to be functional; reporter gene analysis shows that proteasome inhibition increases dexamethasone-mediated GR transcriptional activity (Wallace and Cidlowski 2001). Degradation of GR has been proposed to be a mechanism of cell-type specific glucocorticoid resistance (Mata-Greenwood, Stewart et al. 2013). Furthermore, interaction with the co-chaperone Bag-1 also promotes GR’s degradation (Demand, Alberti et al. 2001). In dexamethasone-resistant HUVECs, higher levels of Hsp70 recruitment to GR following dexamethasone treatment occurs and increased interaction of GR with Bag-1, and overexpression of Bag1 decreases protein levels of GR (Mata-Greenwood, Stewart et al. 2013).
2.1.2e Glucocorticoid Availability
The previous mechanisms of GR regulation involve direct modulation of GR in terms of availability, function, and structure. Although not as well-delineated at this point, availability of glucocorticoid ligand is also able to modulate GR signaling in the cell. P-glycoprotein expels glucocorticoids from the cell, and reduced activity of this pump may increase intracellular concentration of glucocorticoids, thus increasing activation and subsequent signaling of GR (Pariante, Kim et al. 2003). However, the impact of these cell efflux pumps is disputed. Other studies question the physiological relevance of p-glycoprotein in altering HPA axis responses in rodents (Mason, Pariante et al. 2008). More recently, strain differences or different isoforms of p-glycoprotein have been hypothesized to impact the somewhat conflicting data on the role of pglycoprotein in modulating GR activity (Mason, Pariante et al. 2012).
2.1.2f GR action during transcription
Binding of GR to the GRE binding site results in dimerization of the DNA binding domain of GR (Luisi, Xu et al. 1991) and increases in gene expression of its target genes (Chandler, Maler et al. 1983). GR is also able to bind to negative GRE (nGRE) DNA binding sequences on which the GR reduces transcriptional activity through interaction with transrepression complexes (Surjit, Ganti et al. 2011). GR binds to nGRE as two separate GR monomers on opposite sides of the DNA strand in an orientation that prevents GR dimerization, a conformation in contrast to GRs at positive GREs where GR binds on the same side of the DNA strand (Hudson, Youn et al. 2013).
2.1.2g Non-genomic actions of GR
Though discussion of GR signaling often concerns transcriptional regulation through the classical GR nuclear receptor, GR also can exert non-genomic effects. Rapid effects have been observed following glucocorticoid stimulation, and these effects occur too quickly to be explained by genomic GR actions through altered transcription. Much research in non-genomic actions of GR has centered around fast feedback inhibition of the HPA axis (Iwasaki, Aoki et al. 1997, Deng, Riquelme et al. 2015), and non-genomic effects of glucocorticoids have been studied in multiple cell types, including cells that mediate immune and inflammatory responses. Membrane-bound GR has been found to be responsible for much of the non-genomic actions of glucocorticoids (Perez, Cormack et al. 2013). Membrane-bound GRs have been found in monocytes, B-cells, T-cells, and neurons (Bartholome, Spies et al. 2004, Lowenberg, Verhaar et al. 2006, Tryc, Spies et al. 2006, Shaqura, Li et al. 2016).
2.2.1 Structure, Function, and Regulation of NFκB
The family of NFκB proteins serve as transcription factors that regulate immune and inflammatory responses, cell survival, development, synaptic plasticity, and neurogenesis (Kaltschmidt and Kaltschmidt 2009). NFκB is present in all cell types (Hoffmann and Baltimore 2006) and due to its prominent role in inflammatory signaling, is extensively studied in many diseases including cancer, rheumatoid arthritis, asthma, cardiovascular and metabolic disease. NFκB can be activated by numerous inflammatory, physiological, and cellular stress signals (for a complete list, refer to www.nf-kb.com). When activated, it regulates the transcription of hundreds of target genes relevant to a diverse array of physiological functions. Multiple receptor-specific intracellular transduction pathways such as toll-like receptors (TLRs), TNF-α receptor, IL-1 receptor, T- and B-cell receptor signaling can converge upon the NFκB pathway. Furthermore, adjacent inflammatory signaling cascades including the p38/MAPK, JNK pathways can interact with and mutually regulate the NFκB pathway (Oeckinghaus, Hayden et al. 2011).
There are five polypeptides in the NFκB family that contribute to the diversity of this transcription factor (p65, cRel, Rel, as well as the precursor proteins p100 and p105 that get proteasomally cleaved into the subunits p52 and p50). All five subunits contain a conserved Rel-homology domain, which allows dimerization between subunits and enables DNA-binding. These subunits form 15 possible dimers that display either transcriptional activator, repressor, or no DNA-binding ability depending on dimer composition (Hoffmann and Baltimore 2006). The p65, Rel, and cRel proteins possess a transcriptional activation (TA) domain, which is required for target gene transcription. In contrast, the p50 and p52 subunits do not contain a TA domain, and are unable to regulate gene transcription unless dimerized to another NFκB subunit that possesses a TA domain. Dimers exclusively composed of p50 or p52 are therefore often referred to as transcriptional repressors. Finally, the p65/Rel, cRel/Rel, and Rel/Rel dimers are unable to bind to DNA.
2.2.2 Canonical and non-canonical NFκB pathways
Similar to GR, the NFκB protein complex resides sequestered in the cytoplasm in the absence of relevant stimuli such as cytokines, growth hormones, cellular and oxidative stress, and synaptic transmission. Within NFκB signaling pathway, there are two distinct cascades, canonical and non-canonical, that are activated by different stimuli and perform different functions. The canonical pathway is activated by pro-inflammatory stimuli including the archetypal pro-inflammatory cytokines TNF-α and IL-1β as well as microbial products. The canonical pathways mediates rapid immune activation such as that during acute infection. The non-canonical pathway can be activated by both immune (lymphotoxin-β, CD40 ligand, B-cell activating factor) and non-immune stimuli, and is involved in slow and sustained immune regulation such as that which occurs during development of the immune organs. The canonical and non-canonical pathways also differ in the composition of NFκB. The canonical pathway features the most common form of NFκB, the heterodimer which consists of p65 and p50. In contrast, the non-canonical pathway features heterodimers containing p52 and Rel proteins.
2.2.3 Molecular Regulation of NFκB Signaling
In addition to different dimer compositions, the two types of NFκB signaling cascades have both shared and distinct regulatory features. Due to the dual points of regulation, cytosolic and nuclear, for purposes of organization, we subset regulatory processes by compartment.
2.2.3a. Regulation of NFκB signaling prior to nuclear translocation
The main regulatory event in both the canonical and non-canonical cascades is the phosphorylation and subsequent degradation of the inhibitor IκB proteins by the appropriately-named IκB kinase (IKK) proteins, thus freeing the NFκB complex to translocate to the nucleus. In the absence of NFκB-activating stimuli, the IκB proteins serve to inhibit nuclear translocation and DNA-binding of NFκB subunits, and they accomplish this by binding to the Rel-homology domain present on all five NFκB proteins. There are several forms of IκB proteins, including IκBα, IκBβ, IκBε, and IκBγ. In addition, the uncleaved precursor proteins p100 and p105 are also considered a type of IκB proteins because they also contain the Ankyrin repeat domain, a hallmark of IκB proteins. While IκB masks the nuclear localization signal on NFκB when the latter is inactive, IκB does not completely prevent nuclear translocation. Rather, IκB is thought to reduce the rate of nuclear translocation of the NFκB-IκB complex that is constantly shuttling between the cytoplasm and the nucleus.
NFκB-activating stimuli trigger the activation of the IKK, which has been described as the core element of NFκB regulation (Israel 2010). There are two IKK proteins, IKKα and IKKβ, and a third regulatory protein IKKγ (also referred to as NEMO). The canonical IKK complex contains a heterodimer of IKKα and IKKβ, and is regulated by the scaffold protein IKKγ. In the canonical pathway, the IKKβ catalytic subunit phosphorylates IκB, which is then ubiquitinated and proteasomally degraded, thus freeing the NFκB dimer to translocate into the nucleus. In contrast, the non-canonical complex contains only the IKKα homodimer and its upstream kinase NIK (Hoffmann and Baltimore 2006). In the non-canonical pathway, IKKα phosphorylates the p100 or p105 subunit, which are then processed into p50 or p52, respectively, getting rid of the inhibitory Ankyrin repeat domain in the cleaving process. Translocation of the newly released NFκB is mediated via dynein (Mikenberg, Widera et al. 2007) and importin molecules (importin-α) (Fagerlund, Melen et al. 2008) (See Figure 1B).
2.2.3b. Regulation of NFκB following nuclear translocation
Once in the nucleus, NFκB binds κB response elements (κRE) in the promoters of cytokines, chemokines, adhesion molecules, inflammatory enzymes, and receptors. Binding of NFκB to κRE initiates transcription via recruitment of the basal transcription machinery including RNA Pol II as well as co-factors necessary for transcription such as p300 and CREB binding protein (CBP). All dimers of NFκB can bind to the κB response element; however, the expression and activation of each dimer type is highly cell type-, developmental stage- and stimulus-dependent (Hoffmann and Baltimore 2006, Sen and Smale 2010). In addition, NFκB binding to the promoter has been described to trigger the formation of enhanceosomes, which are nucleoprotein complexes consisting of several cooperating transcription factors, that ensure the spatially- and temporally- specific induction of a gene (Tian and Brasier 2003). Due in part to the extensive redundancy and cross-feedback in inflammatory signaling pathways, different inflammatory transcription factors usually act in concert to yield stimulus-specific activation of each target gene. This is accomplished by varying combinations of transcription factors such as NFκB, SP-1, IRFs, STATs, CREB, and AP-1 sequentially binding to their respective response elements within an enhancer region of the target gene. Another key determinant of the rate of target gene induction by NFκB is elements of the promoter architecture such as number and proximity of κRE present (with greater number of κRE clustered closely together corresponding to stronger induction).
While many of the target genes induced by NFκB are pro-inflammatory in nature, and can perpetuate and prolong the NFκB response, other target genes function to suppress and contain the inflammatory response caused by NFκB. For example, among the target genes of NFκB is its negative regulator, IκB. Newly synthesized IκB protein retains the NFκB complex in the cytoplasm and prevents its transcriptional activity, thus creating an ultra-short negative feedback loop on NFκB activity – similar to that discussed above between GR and FKBP5. Following NFκB activation, the quantity of total IκB protein can include both initial IκB protein that was available prior to NFκB activation and newly synthesized IκB post-activation; therefore, researchers distinguish between phosphorylated IκB amount, which indicates IκB about to be degraded, and total IκB protein in the cell.
2.2.3c. Post-translational modifications of the NFκB pathway
Activation of the IKK proteins, which is necessary for NFκB induction, has been described to be accomplished by various post-translational events (reviewed in (Perkins 2006). The most common modifications include the ubiquitination of IKKγ and phosphorylation of IKKα and IKKβ. In addition, the TA domain present on p65, Rel, and cRel may be subject to post-translational modifications such as phosphorylation and acetylation for selective induction and optimal regulation of NFκB. Numerous post-translational modifications of NFκB have been described in a stimulus- and cell type-specific manner (Perkins 2006). For example, lipopolysaccharide (LPS), a bacterial toxin widely used in research to stimulate NFκB, signals via TLR4 and then phosphorylates serine 276 on p65 via the catalytic subunit of PKA, whereas several other kinases have been reported to phosphorylate S536 following LPS. Another common post-translational modification of NFκB is the reversible acetylation of p65 on one of multiple lysine residues by the transcriptional co-activator proteins p300 and CBP, which enhances p65’s binding to DNA (Chen, Mu et al. 2002).
Activity of NFκB can therefore be measured via assessing the 1) availability of IKK proteins that initiate the entire NFκB cascade, 2) amount of phosphorylated IκB proteins, 3) degree of post-translational modification of NFκB subunits specific to stimulus, 4) degree of nuclear translocation or presence of NFκB subunits, most commonly p65, 5) extent of NFκB binding to DNA (κRE), and 6) NFκB-mediated transcription of its downstream target genes including both endogenous target genes and reporter constructs.
3. Interactions between GR and NFκB
The mutual regulation and interaction between GR and NFκB are understood to be highly stimulus-, cell type-, and time course-dependent, and are an ongoing focus of a substantial body of research (see De Bosscher, Vanden Berghe et al. (2003) for review). Under most acute activation scenarios, GR and NFκB display mutually antagonistic effects, and as such the focus of Section 3 will be details of this two-way negative regulation. However, certain combinations of stimuli, or prolonged activation, can cause GR and NFκB to exert synergistic effects on a subset of genes (Rao, McCalman et al. 2011), depending on the architecture of GREs and κREs present on target gene promoters. Furthermore, disruptions to the endocrine-immune balance, such as that which occurs during chronic stress, may also entail synergistic effects between GR and NFκB, and will be discussed in Section 4.
The crosstalk between GR and NFκB primarily occurs following nuclear entry (Section 3.2) via several mechanisms most notably among them transrepression and trans-activation. Additionally, interactions between the two pathways in the cytoplasm prior to nuclear translocation have also been documented as detailed in Section 3.1.
3.1 GR-NFκB interactions at the level of nuclear translocation
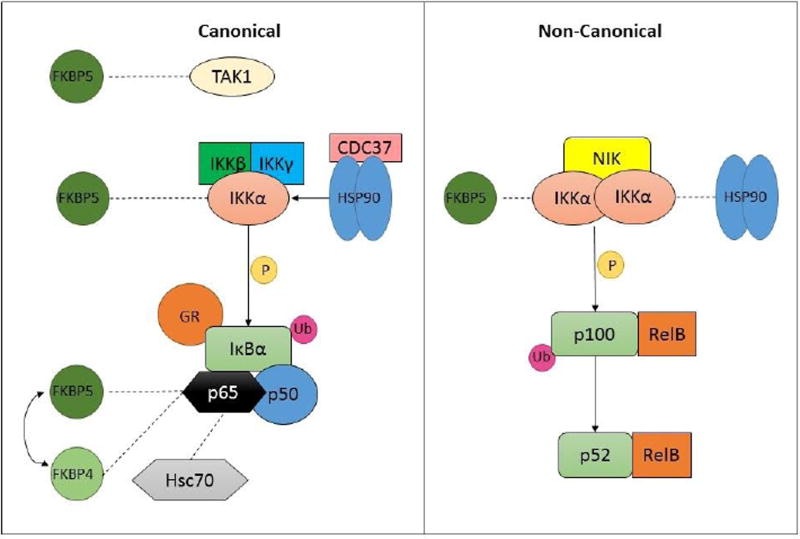
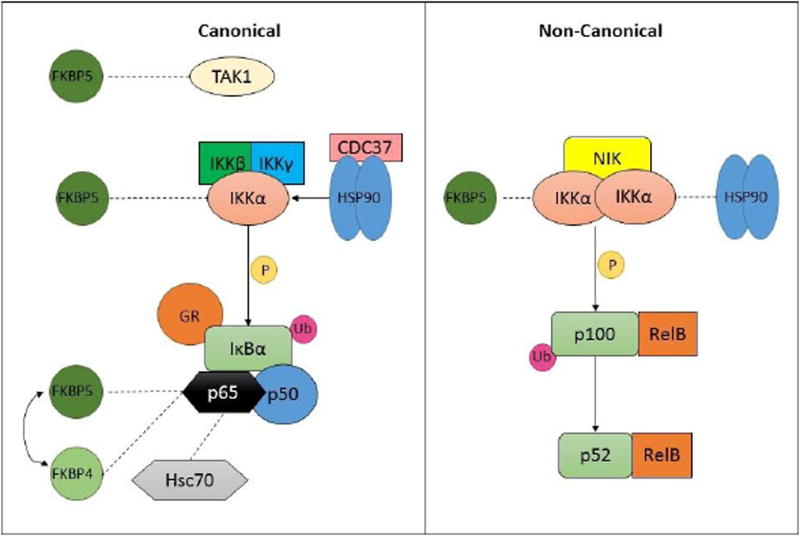
As described above, both GR and NFκB are inducible transcription factors, and their activity is regulated by negative feedback loops involving transcription of their respective inhibitors FΚBP5 and IκB. Both GR and NFκB reside in the cytoplasm tethered by these negative inhibitor proteins, and nuclear translocation is a required step for function of both. Considerable similarities exist in the mechanisms regulating the nuclear translocation of GR and NFκB, and several avenues for protein-protein interactions between the two pathways have been described (see Figure 2).
Figure 2. Cytoplasmic interactions between GR and NFκB cascades.
The co-chaperone FKBP5 has been identified as a binding partner to multiple proteins constituting the NFκB pathway including TAK1, IKKα, and p65. Furthermore, the chaperone proteins CDC37, Hsp90, and Hsc70 also bind IKKα and p65. The functional significance of these protein-protein interactions remain incompletely understood.
3.1.2 GR effects on IκB
As detailed previously, NFκB’s rate of nuclear translocation and DNA-binding activity is suppressed by its binding to IκB proteins. Furthermore, GR increases the binding affinity of IκB to NFκB complex (Scheinman, Cogswell et al. 1995). Even in the absence of corticosteroid signaling, cytosolic GR has been shown to interact with p65, p50, and IκB, and inhibit nuclear translocation of NFκB (Widen, Gustafsson et al. 2003).
3.1.3 Chaperone Influences
Protein-protein interaction partners of NFκB and GR overlap substantially, with several key players having been shown to influence the nuclear translocation of each transcription factor primarily in vitro (Bouwmeester, Bauch et al. 2004, Hinz, Broemer et al. 2007, Rein 2016). GR is tethered in the cytoplasm by the chaperone Hsp90 and a number of co-chaperone proteins including most notably FΚBP5. Emerging evidence indicates that subunits of the NFκB complex as well proteins constituting the cascade that regulates NFκB activation are able to interact with various members of the family of chaperone proteins that regulate GR’s nuclear translocation, yet the functional significance of these associations in vivo remain largely undetermined (Rein 2016). For example, NFκB has been shown to associate with the cellular stress-inducible heat shock protein 70 (Hsp70) in human T-lymphoma cells (Guzhova, Darieva et al. 1997) and heat shock cognate 70 (Hsc-70), the constitutively expressed form of Hsp70, in hippocampal neurons (Klenke, Widera et al. 2013). While heat shock proteins including Hsp90 and Hsp70 are best known for promoting protein folding, trafficking, and degradation, and along with p23 form the cytoplasmic chaperone machinery that enables GR transport, the latter finding was interpreted to suggest a potential role in axonal transport in neurons.
3.1.4 NFκB’s interactions with FKBP5
The co-chaperone FΚBP5, which inhibits the nuclear translocation of GR, and under chronic stress conditions promotes a glucocorticoid-insensitive state (Guidotti, Calabrese et al. 2013), has been reported to interact with many members of the TNF-α-induced NFκB pathway including TAK1, MEKK1, (Bouwmeester, Bauch et al. 2004), TNF receptor-associated factor-2 (TRAF2) (Romano, Xiao et al. 2015), TRAF3, TRAF6 (Akiyama, Shiraishi et al. 2014), IKKα, IKKε (Bouwmeester, Bauch et al. 2004, Romano, Xiao et al. 2015), IKKβ (Romano, Xiao et al. 2015) and p65 (Erlejman, De Leo et al. 2014), with conflicting reports of its impact on overall NFκB activity. Whereas siRNA-mediated knockdown of FΚBP5 was found to decrease the nuclear translocation of p65 in MEF cells (Akiyama, Shiraishi et al. 2014) and suppress NFκB reporter activity in HEK293 cells (Bouwmeester, Bauch et al. 2004), another study found that FΚBP5 downregulation had no impact on either IκB degradation or p65 translocation (Hinz, Broemer et al. 2007). In contrast, one study reported that overexpression of FΚBP5 prevented, whereas that of FΚBP4 facilitated, the nuclear translocation of NFκB in HEK293 cells, resembling the co-chaperones’ well-described regulation of GR (Erlejman, De Leo et al. 2014). Although it is unclear in some of these reports whether the impact of FΚBP5 on NFκB activity is mediated via direct protein-protein interactions or via FKBP5’s regulation of GR, taken together these reports raise the intriguing possibility that co-chaperone-regulated nuclear translocation may represent a previously unexamined checkpoint for GR and NFκB balance. As indicated above, FΚBP5’s involvement in NFκB pathway has been studied in the context of oncology because of its overexpression in some types of cancer (Jiang, Cazacu et al. 2008, Romano, Mallardo et al. 2011); however, despite the abundant knowledge of FΚBP5’s impact on the activity of GR in the context of endocrine disruptions, and the established potent anti-inflammatory properties of GR, little work has investigated FΚBP5’s role in chronic stress-induced inflammation.
3.1.5 Beyond protein-protein interactions
Beyond shared protein-protein interaction partners, several other mechanisms have been described to influence GR and NFκB regulation. The catalytic subunit of protein kinase A (PKAc) which phosphorylates p65 attenuates the mutual antagonism between GR and NFκB (Doucas, Shi et al. 2000). Patients with major depression may be particularly vulnerable to the anti-glucocorticoid effects of inflammatory cytokines due to the decreased PKA signaling activity (a pathway that promotes GR activity that decreases the inhibitory activity of cytokines IFNα on GR) (Shelton, Sanders-Bush et al. 2009, Pace, Hu et al. 2011). Activation of the cAMP/PKA pathway through use of forskolin attenuates IFNα-mediated inhibition on GR, and inhibition of PKA impairs GR transcriptional activity (Pace, Hu et al. 2011). The disinhibition of GR signaling by PKA may be mediated by PKA’s inhibitory effects on NFκB (Gao, Hibi et al. 2010, Pace, Hu et al. 2011). Activation of cAMP/PKA also impairs activity of pSTAT5, an inhibitor of GR, which could contribute to enhanced GR activity (Pace, Hu et al. 2011).
3.2 GR-NFκB interactions at the transcriptional level
Mutual antagonism between GR and NFκB has been described to occur via several well-defined mechanisms including transrepression (e.g. repression of pro-inflammatory NFκB targets by GR), trans-activation (e.g. activation of NFκB inhibitor genes by GR), and competition for co-factors and co-activator proteins necessary for transcriptional activity as detailed below (see Figure 3). It should be noted that considerable redundancies exist between inflammatory signaling pathways, and components of one pathway can often compensate the loss of another pathway’s functional effectiveness, and conversely, glucocorticoid-mediated suppression of parallel pathways such as the MAPK pathway can also diminish NFκB activation (see Clark (2003) for review). As such, the GR signaling pathway not only cross-regulates NFκB but also similar inflammatory pathways including TNF-α (Van Bogaert, De Bosscher et al. 2010).
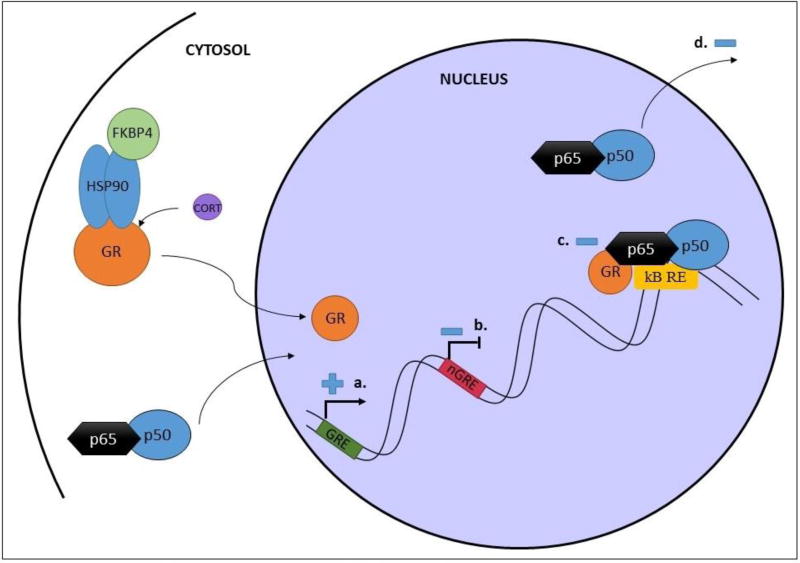
Figure 3. Nuclear interactions between GR and NFκB cascades.
The GR interacts with GREs (a.) and negative GREs (b.) to promote or suppress gene expression at numerous target genes. Interactions with these regulatory elements can be reduced by interactions between GR and NFκB. Interaction of GR with the p65 subunit of NFκB (c.) can also inhibit NFκB transcriptional activity at κBREs. Finally, some reports suggest that GR can promote nuclear export of NFκB, further reducing its transcriptional activity.
3.2.1 Transrepression
The mechanism of interaction between GR and NFκB has been extensively studied, and the GR has been found to interact with NFκB through direct protein-protein interactions that impair NFκB transactivation (De Bosscher, Schmitz et al. 1997). The precise nature of the interaction between GR and NFκB is gene-specific (De Bosscher, Vanden Berghe et al. 2003). GR physically interacts with p65, and decreased transcriptional activity of NFκB at the IL-6 promoter occurs with dexamethasone treatment (Ray and Prefontaine 1994). Dexamethasone also decreases IL-8 transcription through inhibiting NFκB’s transcriptional activity. These studies determined that dexamethasone’s inhibitory activity on IL-8 transcription does not depend on decreased nuclear translocation of NFκB but through blocking the binding of NFκB to the DNA (Mukaida, Morita et al. 1994). However, other studies examining transrepression of additional NFκB genes such as IL-6 do not require impaired DNA binding of NFκB to elicit reduced gene expression but rather argue that GR impairs transactivation of p65 through altered interaction with basal transcriptional machinery (De Bosscher, Schmitz et al. 1997, De Bosscher, Vanden Berghe et al. 2000). The GR can also interact with p50, resulting in impaired transcriptional activity of NFκB. Interactions of the GR with p65 and p50 require the zinc finger domain of GR (Scheinman, Gualberto et al. 1995).
The interaction of GR and NFκB is not one-sided in transrepressive activity. P65 also inhibits GR activity at glucocorticoid response elements (Scheinman, Gualberto et al. 1995), and GR-mediated transcription of specific target genes is dose-dependently decreased through overexpression of p65 (Waltner-Law, Daniels et al. 2000). This inhibition occurs through two amino acids in the C-terminal zinc finger of the DNA binding domain of GR (Liden, Delaunay et al. 1997). Though the earliest understanding of GR-NFκB interactions is that activity of one only reduces activity of the other, more recent studies have identified that GR and NFκB can have synergistic effects or stimulatory effects on transcription of the other at specific genes (Lamirand, Ramauge et al. 2011).
3.2.2 Trans-activation of anti-inflammatory targets and upregulation of IκB by GR
Although much of GR’s suppression of NFκB is mediated by transrepression mechanisms, a substantial body of evidence also suggests that trans-activation of anti-inflammatory genes by GR also contributes to inflammatory suppression (Newton and Holden 2007). For example, transcription of MKP-1, a kinase that negatively regulates the p38 MAPK pathway (and by extension the NFκB pathway), has been demonstrated to be strongly glucocorticoid-responsive (Lasa, Abraham et al. 2002). Interestingly, the expression of anti-inflammatory genes normally transcribed by NFκB as part of a negative feedback regulation (including TNFAIP and NFKBIA) were found to be spared from suppression by GR, unlike many pro-inflammatory target genes of NFκB (Altonsy, Sasse et al. 2014). These studies suggest that GR’s anti-inflammatory effects on NFκB are nuanced and employ highly specific mechanisms depending on the promoter architecture and function of each target gene. Furthermore, early studies indicated that dexamethasone-activated GR can transcribe IκB, resulting in newly synthesized IκB protein that can quickly associate with NFκB and inhibit the latter’s nuclear translocation (Scheinman, Cogswell et al. 1995). However, IκB upregulation by GR is unlikely to be the primary mechanism of glucocorticoid-mediated suppression of inflammation because NFκB suppression by GR still occurs in the absence of trans-activation by GR (De Bosscher, Vanden Berghe et al. 2003).
3.2.3 Competition for co-activators
Upon translocation to the nucleus, both GR and NFκB interact with the co-activators CBP and steroid receptor coactivator-1 (SRC-1). Some studies demonstrate that competition for these coactivators may also contribute to the mutual antagonism between GR and NFκB (Sheppard, Phelps et al. 1998) though other studies disagree with the limiting potential of CBP (De Bosscher, Vanden Berghe et al. 2000, McKay and Cidlowski 2000). And finally, in addition to transrepressing NFκB activity, GR may also increase nuclear export of NFκB (Nelson, Wilde et al. 2003) and specifically by exporting p65 following treatment with dexamethasone (Scheinman, Gualberto et al. 1995).
3.2.4 Influence of GR Receptor Subtype on NFκB
As discussed in Section 2.1.2b, there are two isoforms of GR. Differences between GRα and GRβ may play a role in the interactions between GR and NFκB signaling in the cell. For example, cytokine-induced NFκB transcriptional activity has been demonstrated to increase the expression of GRβ relative to GRα (Webster, Oakley et al. 2001). The increased GRβ protein driven by cytokine signaling may mediate glucocorticoid resistance and correlated with GRβ protein increases (Webster, Oakley et al. 2001). Furthermore disinhibition may occur by interactions between GRs, such that, GRβ can diminish GRα-mediated inhibition of NFκB (Oakley, Jewell et al. 1999).
3.2.5 Influence of non-genomic GR on NFκB
Anti-inflammatory effects of glucocorticoids have been observed rapidly following stimulation. These rapid anti-inflammatory effects following glucocorticoid stimulation have been attributed to the non-genomic activity of glucocorticoids rather than through GR’s genomic actions (Lowenberg, Tuynman et al. 2005, Lowenberg, Verhaar et al. 2006). Non-genomic glucocorticoid effects have been detected in immune cells (Long, Wang et al. 2005, Lowenberg, Verhaar et al. 2006). Furthermore, non-genomic effects of glucocorticoids may also alter multiple downstream signaling pathways including the proteins p38 MAPK, JNK, protein kinase C, protein kinase A, and ERK1/2 (Qiu, Wang et al. 2001, Solito, Mulla et al. 2003, Lowenberg, Tuynman et al. 2005, Qi, Qiu et al. 2005, Verriere, Hynes et al. 2005, Roy and Rai 2009). As discussed in previous sections, signaling through p38 MAPK, JNK, and PKA can have modulatory effects on NFκB, and non-genomic effects of glucocorticoids on these signaling proteins could be another mechanism through which GR signaling impacts NFκB and inflammation.
4. Hallmarks of GR- NFκB Dysfunction?
4.1 Glucocorticoid Resistance
Prolonged exposure to glucocorticoids, such as during chronic stress, has the capacity to lead to glucocorticoid resistance, which broadly refers to decreased glucocorticoid signaling due to impaired activity of GR. Even the terminology, glucocorticoid resistance, has taken on a dual identity reflecting an impact within both the endocrine and immune systems. Within the chronic stress literature, glucocorticoid resistance primarily indicates impaired negative feedback regulation of the stress axis – which is mediated by GR – culminating in increased cortisol availability. In the context of chronic inflammatory disorders, glucocorticoid resistance indicates diminished ability of glucocorticoids to suppress inflammatory processes such as release of cytokines, proliferation of immune cells and receptors, or at high concentrations, to trigger apoptosis (Barnes and Adcock 2009).
We focus here on chronic stress-related mood disorders because they represent a multidimensional sum of the two forms of glucocorticoid resistance with increased glucocorticoid levels and a less transcriptionally active GR (Pace, Hu et al. 2007), coupled with greater activation of innate immunity in the brain and the body that may be further exacerbated by glucocorticoids (Frank, Thompson et al. 2012, Sorrells, Munhoz et al. 2014). Perhaps even more salient than individual alterations in GR or NFκB is the impaired balance and crosstalk between GR and NFκB. In healthy systems, the crosstalk and balance between GR and NFκB activity are precisely coordinated and regulated (Adcock and Barnes 2008). Following acute psychosocial stressors in healthy humans, NFκB activity in peripheral immune cells peaks within 20 minutes (Bierhaus, Wolf et al. 2003, Wolf, Rohleder et al. 2009, Kuebler, Zuccarella-Hackl et al. 2015), and the subsequent activation of the HPA axis – including the release of adrenocorticotropic hormone (ACTH) and cortisol which peaks by 30–40 minutes following onset of stressor (Bierhaus, Wolf et al. 2003, Kuebler, Zuccarella-Hackl et al. 2015) – serves to suppress this NFκB-driven inflammation. However, evidence demonstrates that in disease conditions not only is GR unable to adequately suppress NFκB activity but also the resulting excessive NFκB leads to further impairment of GR’s anti-inflammatory function, thus setting in motion a vicious circle of GR-NFκB imbalance (Pace, Hu et al. 2007). In the context of mood disorders, excessive or inappropriate activation of NFκB within both peripheral immune cells and directly in the brain plays a key role in initiating and sustaining stress-induced neuroinflammation, as well as structural and behavioral deficits (Munhoz, Lepsch et al. 2006, Koo, Russo et al. 2010).
Much of the human evidence implicating concurrent dysregulation of GR and NFκB in psychiatric illnesses is derived from blood cell samples from patients with mood disorders or chronically stressed populations. These studies indicate a transcriptional fingerprint characterized by reduced GR- and increased NFκB-mediated transcription (Miller, Chen et al. 2008, Miller, Murphy et al. 2014). For example, healthy adult parents who were caregivers to pediatric cancer patients displayed reduced morning cortisol levels compared to parents of healthy children, and had a reduced ability of dexamethasone to suppress IL-6, but not TNFα or IL-1β production (Miller, Cohen et al. 2002). Adolescent female subjects with higher chronic interpersonal stress displayed greater increases in NFκB, GRβ, and IκB mRNA in leukocytes over a six month period, and exhibited higher LPS-stimulated IL-6 expression compared to subjects with lower interpersonal stress (Miller, Rohleder et al. 2009), suggesting glucocorticoid resistance. Greater NFκB DNA-binding concurrent with blunted glucocorticoid sensitivity has been reported in women with childhood abuse-related post-traumatic stress disorder (PTSD) (Pace, Wingenfeld et al. 2012). Diminished suppression of inflammation by cortisol in monocytes was associated with increased depressive mood in patients (Cheng, Dimitrov et al. 2016). Conversely, cognitive-behavioral stress management has been found to reverse anxiety-related upregulation of pro-inflammatory transcripts in leukocytes of women undergoing breast cancer treatment (Antoni, Lutgendorf et al. 2012). Specifically, transcripts that were upregulated by cognitive-behavioral stress management were found to have an underrepresentation of the κB response element in the promoter and an overrepresentation of GREs, suggesting decreased transcriptional activity at κRE coupled with increased activity at GREs (Antoni, Lutgendorf et al. 2012). Similarly, practicing yogic meditation was found to reverse NFκB and interferon regulatory factor (IRF)-related transcriptome in leukocytes of caregivers (Black, Cole et al. 2013). Cumulatively, these findings strongly implicate the concurrent dysfunction and imbalance of GR and NFκB in stress-related psychiatric illnesses and associated glucocorticoid resistance.
4.2 Potential Mechanisms of Glucocorticoid Resistance
Several mechanisms by which chronic stress could lead to decreased GR and increased NFκB activity are possible (see Figure 4). First, co-chaperone variations could drive altered interactions between GR and NFκB as suggested by polymorphisms of the FKBP5 gene which have been associated with increased incidence of depressive episodes in humans and altered response to antidepressant treatment (Binder, Salyakina et al. 2004). Altered FΚBP5 protein levels in the frontal cortex, indicative of GR dysregulation, have also been found in the frontal cortex of subjects with major depressive disorder (Tatro, Everall et al. 2009). Interestingly, microRNAs (miRNA) that are predicted to interact with GR and FKBP5 have also been implicated as potential variables in altered glucocorticoid responsivity in rodent models of stress-induced disorders (Jung, Wang et al. 2015). Altered phosphorylation of GR may be another mechanism through which glucocorticoid and inflammatory systems achieve balance. IL-2 and IL-4 decrease GR phosphorylation (Pazdrak, Straub et al. 2016), and phosphorylation of GR impacts its ability to interact and transrepress NFκB (Galliher-Beckley, Williams et al. 2008). To this end, patients with depression exhibited higher cortisol levels that correlated with S226 phosphorylation of GR (Simic, Maric et al. 2013). Nuclear expression of GR phosphorylated at S226 in leukocytes also correlated with self-reported current depression in women, and this effect was absent in men (Simic, Adzic et al. 2013).
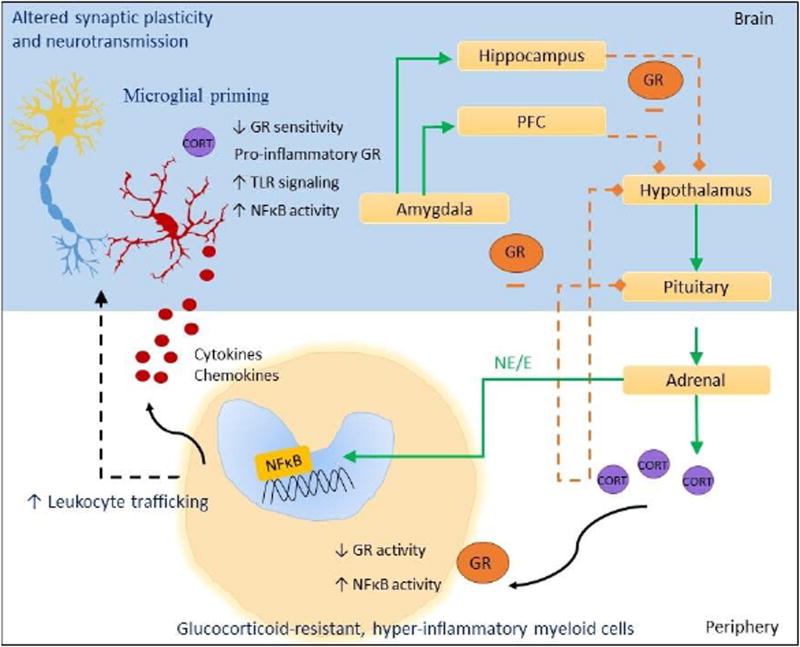
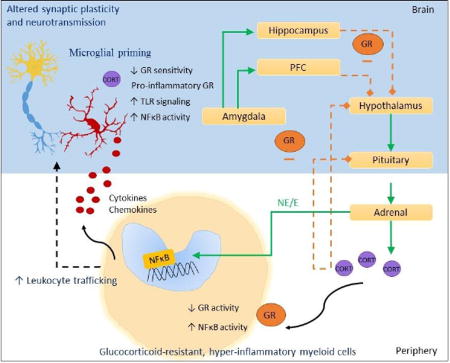
Figure 4. GR and NFκB in glucocorticoid resistance in the brain and immune system.
Under acute stress conditions, release of catecholamines by the sympathetic nervous system activates NFκB within peripheral immune cells, and leads to expression of cytokines and chemokines. Activation of the hypothalamic-pituitary adrenal axis results in the release of glucocorticoids, and GR suppresses NFκB in peripheral immune cells. Under chronic stress conditions, impaired GR activity within peripheral myeloid cells can disinhibit NFκB-mediated inflammation. In turn, glucocorticoid-resistant, hyper-inflammatory immune cells can traffic into the brain, eventually getting recruited to brain regions regulating the stress response. The GR-mediated negative feedback regulation of HPA axis is also impaired in chronic stress pathology, thus further perpetuating stress-induced neuroinflammation. The disruption in endocrine-immune balance appears to be due partly to primed microglia that display decreased GR sensitivity, pro-inflammatory GR, and exaggerated innate immune activation including increased NFκB and Toll-like Receptor signaling. The functional significance of the altered GR-NFκB balance may be manifested in the brain as altered synaptic plasticity and neurotransmission.
Finally, it has recently been proposed that when dysfunctional, glucocorticoids not only signal less effectively, but also exert a pro-inflammatory impact when they do (Horowitz and Zunszain 2015). In particular, glucocorticoid signaling has been demonstrated to enhance inflammation under both stress (Frank, Thompson et al. 2012) and conditions of central nervous system injury (Sorrells, Caso et al. 2009). One potential mechanism through which glucocorticoids may act in a pro-inflammatory fashion is through changes to stimulus specificity of GR and NFκB. For example, when cells from steroid resistant but otherwise healthy individuals were stimulated with dexamethasone, the expected increase in GR DNA-binding was absent and increased NFκB DNA-binding was found to occur (Dawson, Dhanda et al. 2012). Other molecular mechanisms of glucocorticoid-immune imbalance are discussed within the context of specific brain regions in the next section.
5. Traversing the Blood Brain Barrier: from peripheral resistance to central mayhem
5.1 Expression and interaction of GR and NFκB in the brain
In the rat brain, GR is expressed in highest density in the hippocampus (lateral septum and the dentate gyrus), and to a lesser extent in the nucleus tractus solitarii, amygdala (Reul and de Kloet 1985), and hypothalamus. GR is expressed in both neurons and glia. Glial GR is involved in microglial development and proliferation, and is thought to perform crucial anti-inflammatory functions during CNS inflammation and provides neuroprotection (Carrillo-de Sauvage, Maatouk et al. 2013). However, under certain conditions characterized by stress or injury, glucocorticoids can prime hippocampal microglia to assume a more pro-inflammatory profile (Frank, Baratta et al. 2007, Frank, Thompson et al. 2012). This will be further discussed in Section 5.2.
Constitutive NFκB activity has been demonstrated in several brain regions including the hypothalamus, cortex, hippocampus, amygdala, olfactory lobes, and cerebellum (Schmidt-Ullrich, Memet et al. 1996, Bhakar, Tannis et al. 2002). During systemic inflammation, brain regions most accessible to cerebrospinal fluid (CSF) and blood including the circumventricular organs, and neighboring regions as well as elements of the brain microvasculature and blood-brain barrier (BBB) are likely to express the greatest extent of NFκB activation owing to the soluble pro-inflammatory factors present in the CSF and blood (Rivest 2001). NFκB is likely primarily active in the immunocompetent cells of the CNS including microglia due to the widespread expression of immune receptors on these cells. However, NFκB is expressed in neurons, and is thought to play roles in synaptic plasticity, brain development (Boersma, Dresselhaus et al. 2011), synaptic signaling (Meffert, Chang et al. 2003). Conflicting views exist regarding the origin of NFκB activity in neurons. Whereas some groups have found minimal NFκB activity in primary neuronal cultures (Listwak, Rathore et al. 2013, Mao, Phanavanh et al. 2016) others have found constitutive NFκB activity in neurons (Kaltschmidt, Kaltschmidt et al. 1994, Bhakar, Tannis et al. 2002), and the discrepancy is thought by some to result from varying degrees of purity of neuronal cultures due to mixed glial populations (Barger & Mao, 2012). It has been suggested that NFκB signaling in glia mediates pathological pro-inflammatory processes whereas signaling within neurons promotes their survival (Camandola and Mattson 2007).
GR and NFκB interactions have been documented in murine brain cells. Dexamethasone has been shown to suppress the expression of NFκB target cytokines such as IL-1β (Minami, Kuraishi et al. 1990), IL-6 (Benveniste, Sparacio et al. 1990) and TNFα (Brenner, Yamin et al. 1993) in mouse brain cells. Conversely, activation of NFκB in mouse hypothalamic cultures has been demonstrated to suppress dexamethasone-induced genes (Hagimoto, Arima et al. 2013). Though many of the early studies examining transcriptional level interactions of the GR and NFκB were performed in vitro, interactions of GR and NFκB have been confirmed in the brain using rodent models as well. For instance, glucocorticoids have been shown to decrease binding of NFκB to DNA (Unlap and Jope 1997, Chantong, Kratschmar et al. 2012) in the rat brain. Evidence of GR-NFκB interactions in the brain have also been provided by demonstrations of co-immunoprecipitation of the two proteins (Ros-Bernal, Hunot et al. 2011). In addition, adrenalectomy increases NFκB-DNA binding in the cortex and hippocampus, supporting GR’s inhibitory role on NFκB activity (Unlap and Jope 1995).
5.2 Chronic-stress induced neuroinflammation
A convincing body of evidence suggests that stress-induced inflammation contributes to the harmful effects of chronic stress on mood and behavior (Padgett and Glaser 2003). Yet, the means by which chronic psychosocial stress leads to inflammation in the brain remains incompletely understood. Due to its central role in regulating innate immunity, NFκB is positioned to serve critical roles in several mechanisms of stress-induced neuroinflammation as detailed below.
During stress, catecholamine signaling in peripheral immune cells leads to NFκB activation (Bierhaus, Wolf et al. 2003), resulting in secretion of soluble inflammatory molecules such as cytokines, chemokines, and acute phase proteins. Peripherally released inflammatory molecules can access the brain via several routes including the transport across the BBB, activation of perivascular macrophages and endothelial cells, and the efferent vagus nerve (Dantzer, O'Connor et al. 2008). Prolonged inflammation such as that occurs in chronic stress has been documented to alter permeability of the BBB and promote increased extravasation of soluble factors. In addition to the peripherally-originated soluble signaling molecules, immune cells themselves can infiltrate the brain, and traffic to sites expressing increased chemokine receptors following repeated social defeat (Wohleb, Powell et al. 2013).
Subsequent to the sympathetic nervous system (SNS)-triggered activation of the immune response, the HPA axis is activated either via catecholamine signaling or cytokine signaling (Pace, Hu et al. 2007). Although the effects of GR are commonly conceptualized as anti-inflammatory, there are several contexts in which GR has been documented to enhance pro-inflammatory processes. In particular, timing of glucocorticoid release has been proposed as a key determinant of this shift (Dhabhar 2009, Horowitz and Zunszain 2015) in which glucocorticoid presence preceding the onset of immune activation has a priming or stimulatory effect, whereas glucocorticoid release that follows immune activation exerts a suppressing effect on inflammation (Frank, Miguel et al. 2010). In fact, glucocorticoids released by both acute inescapable shock stress (Frank, Thompson et al. 2012) or their acute and chronic exogenous administration (Frank, Miguel et al. 2010, Frank, Hershman et al. 2014) sensitize the central and peripheral inflammatory response. Furthermore, in mice experiencing seizures or ischemia glucocorticoid signaling can promote inflammatory processes, including NFκB activation, in the CNS, and perpetuate tissue damage (Sorrells, Caso et al. 2013, Sorrells, Munhoz et al. 2014).
Stress appears to derail neuroimmune processes by utilizing the immune cells in the brain such as microglia as well as peripheral myeloid cells, and tapping into existing mechanisms that signal cellular stress. In the absence of overt inflammation in the central nervous system (CNS), stress is capable of activating inflammatory signaling pathways via release of danger/damage-associated molecular patterns (DAMPs) and microbe-associated molecular patterns (MAMPs) that bind to immune receptors, a process termed “sterile inflammation” (Fleshner 2013, Slavich and Irwin 2014). High mobility group box 1 (HMGB1), one of the best-characterized DAMPs, resides undetected in the nucleus of a cell when unstimulated but is released upon activation as demonstrated in a rodent model of inescapable footshock stress (Cheng, Pardo et al. 2016). The released HMGB1 protein can signal through toll-like receptors (TLRs) present primarily on microglia to activate these cells and further promote the spread of inflammation in the brain via NFκB signaling (Weber, Frank et al. 2015, Cheng, Pardo et al. 2016). In fact, TLR2 and TLR4 signaling within microglia has been demonstrated to be one mechanism underlying the immune-priming effect of glucocorticoids (Frank, Miguel et al. 2010, Weber, Frank et al. 2013). Activation of NFκB via HMGB1 can also induce the inflammasome-associated protein NLRP3 (nod-like receptor protein 3), which has been implicated in microglial priming by stress and behavioral deficits due to chronic stress (Weber, Frank et al. 2015). In addition to causing the above-mentioned changes in microglia, stress can also prime peripheral immune cells such as splenic monocytes to become glucocorticoid resistant and hyper-inflammatory (Quan 2003). The release and trafficking of these primed cells to the brain have been shown to underlie subsequent anxiety-like behavior both at baseline (Wohleb, Powell et al. 2013) and when re-exposed to a subthreshold stressor (McKim, Patterson et al. 2016).
It should be noted that the majority of the studies examining NFκB activation in the brain use pro-inflammatory stimuli such as LPS that induce the canonical NFκB pathway. However, there is some evidence that the non-canonical NFκB pathway may be involved in affective-like behavior. Kassed and Herkenham (2004) demonstrated that mice deficient in the p50 subunit of the NFκB complex (thus deficient in non-canonical signaling) show reduced anxiety-like behaviors in tests of exploratory drive and anxiety. A number of alterations in GR and NFκB systems have been described in various rodent stress paradigms with a particular focus on the hippocampus and prefrontal cortex (PFC), regions involved in the regulation of the HPA axis. These findings are summarized below in 5.3.
5.3 Brain Region-Specific Changes in GR and NFκB Signaling
5.3.1 Prefrontal Cortex
Several studies to date have reported induction of NFκB in the PFC following psychosocial stress in rodent models. For example, Madrigal, Moro et al. (2001) found that restraint stress lasting four, but not two, hours led to an increase in nuclear translocation of the p50/p65 NFκB complex. Increased TNF-α signaling in the brain following immobilization stress has been demonstrated to induce NFκB-mediated release of inducible nitric oxide synthase (iNOS) which mediates neurotoxic damage (Madrigal, Moro et al. 2001, Madrigal, Hurtado et al. 2002) and accounts for at least one mechanism by which peripheral inflammation can translate into neuronal dysfunction. Furthermore, repeated exposure to combined restraint and acoustic stress led to the activation of the TLR4 pathway in the PFC of mice, and a functional mutation in the TLR4 gene abolished the stress-induced increase in p65 as well as iNOS and COX-2 (Garate, Garcia-Bueno et al. 2013).
Although the functional and behavioral consequences of chronic stress are thought to rest upon the balance between pro- and anti-inflammatory signaling, few studies have examined both GR and NFκB concurrently. The ratio of GR to NFκB signaling may be a mechanism through which environmental stressors impact activity of brain regions involved in the stress response. Hu, Zhang et al. (2016) reported that chronic treatment of mice with dexamethasone for 21 and 28 days decreased the mRNA and protein expression of GR, and increased NFκB expression in the PFC and hippocampus, and that the net result of increased inflammation may drive the decrease in motor and exploratory behavior and neurodegeneration reported in these mice. Another study found that acute immobilization stress, and to a lesser degree, chronic isolation stress, increased the nuclear ratio of GR to NFκB protein expression in the rat PFC, (Djordjevic, Adzic et al. 2010) along with evidence of pro-apoptotic signaling. However, nuclear level of NFκB was unchanged in chronically stressed rats, suggesting that changes to the NFκB signaling system as a result of chronic stress may not be evident at baseline and that it may take an additional immune stressor to reveal any priming effects of chronic stress to NFκB.
Consistent with this idea, evidence suggests that chronic stress primes the NFκB pathway in the brain including in the PFC to a subsequent immune challenge. Rats subjected to chronic unpredictable stress did not differ from non-stressed littermates in basal NFκB DNA binding, but upon a systemic LPS probe were found to display exaggerated NFκB DNA binding in the PFC and hippocampus (Munhoz, Lepsch et al. 2006). Interestingly, this stress-mediated NFκB priming was attenuated by GR inhibition, suggesting an unexpected facilitative role of GR in these regions on the inflammatory consequences of chronic stress exposure (Munhoz, Lepsch et al. 2006). Similar findings were reported by de Pablos, Villaran et al. (2006) who found exaggerated TNF-α induction in the PFC following chronic stress and an intracortical LPS challenge. Likewise, this effect was successfully attenuated with pharmacological inhibition of GR with RU486. Collectively, evidence in the PFC suggests that both baseline and induced expression of NFκB is potentiated by stress, and that these findings are also accompanied by signs of decreased GR signaling and a shift to pro-inflammatory impact of GR.
5.3.2 Hippocampus
The hippocampus has continued to be a primary focus of chronic stress studies given its vulnerability to both stress and inflammation. The intricate mechanisms that regulate both GR and NFκB likely contribute to the variability in outcomes such that timing, type of stress, organism selection, and endpoint tissue likely dictate the specific response. Exposure to 21 days of chronic restraint stress has been shown to increase the expression of NFκB pathway proteins including IκB, p65, p50 both at baseline and also upon a novel acute stressor (Gray, Rubin et al. 2014). Interestingly, the chronic restraint-induced increase in p50 persisted even after a 21-day recovery, potentially suggesting a role for the p50-mediated non-canonical NFκB pathway in mediating the lasting impact of chronic stress. Hippocampal NFκB activity has also been demonstrated to underlie behavioral deficits in a PTSD model where mice experienced a single exposure to predator odor (Cohen, Kozlovsky et al. 2011). The prevalence of extreme behavioral responders was diminished by both a systemic administration of high-dose corticosterone and pyrrolidine dithiocarbamate, a selective NFκB inhibitor (Cohen, Kozlovsky et al. 2011). Many of the findings of altered GR-NFκB dynamics previously discussed with regards to the PFC have also been reported in the hippocampus, including stress-induced NFκB priming (Munhoz, Lepsch et al. 2006), lower GR-to-NFκB nuclear ratio due to chronic isolation stress (Djordjevic, Adzic et al. 2009), and chronic dexamethasone-induced decrease in GR and increase in NFκB (Hu, Zhang et al. 2016). In particular, hippocampal microglia have been demonstrated to underlie the altered endocrine-immune balance in stressed rodents (Frank, Baratta et al. 2007). However, it is important to be mindful of the dramatic variability in chronic stress paradigms which has produced conflicting evidence regarding the impact of chronic stress on NFκB activity and function. In particular, stress paradigms of either considerable severity or long duration may suppress the immune system, thus potentially leading to conflicting findings regarding NFκB activity. By way of example, in a sensory contact model in which subordinate mice bred for long attack latency (LAL) live with highly aggressive, dominant mice bred for short attack latency (SAL), separated by a perforated transparent partition for 25 days, NFκB pathway genes were found to be downregulated in the hippocampus of LAL mice (Feldker, Morsink et al. 2006). Similarly, daily administration of corticosterone to rats for 60 days did not cause hippocampal NFκB activation despite causing behavioral deficits (Plaschke, Feindt et al. 2006).
Although ample evidence of changes in GR and NFκB signaling in chronic stress or immune alteration models exists, the functional implications of such changes remain incompletely understood. Differential activity and balance between these two transcription factors may impact function of brain circuitry important in mood and behavior. To this end, hippocampal increases in NFκB signaling was found to mediate the pro-depressive actions of four weeks of CUS through suppressing adult neurogenesis and altering the ratio of neural stem-like cells (Koo, Russo et al. 2010). Furthermore, rats exposed to chronic social isolation have increased NFκB to GR ratio in the hippocampus and increased cytosolic Bcl-2, an initiation step of apoptosis (Djordjevic, Adzic et al. 2009). The interactions of GR and NFκB may be particularly salient. In addition to effects of glucocorticoids on neurogenesis, hippocampal structure, and cell viability (Behl, Lezoualc'h et al. 1997, Zhang, Pan et al. 2016), synthetic glucocorticoid treatment impairs NFκB DNA binding activity in hippocampal cells and subsequently impairs NFκB mediated transcription. This additional impairment may facilitate a second and compounding pathway of alterations given that impaired NFκB transcriptional activity increases susceptibility to oxidative stress induced cell death (Braun, Liebetrau et al. 2000). However, the effects of impairment and stimulation are not simple and bivalent. For instance, the glucocorticoid-expressed kinase SGK1 has been found to increase the formation of spatial memory (water maze) through increasing phosphorylation of IKKα which increases hippocampal NFκB activity (Tai, Su et al. 2009). Together it is still unclear specifically what balance between GR and NFκB promotes healthy regulation vs disease, but these studies do implicate alterations in signaling of the two proteins both as a consequence of stress or chronic glucocorticoid exposure, but additional study is necessary to fully elucidate the factors that determine the nature and implications of the interactions of these transcription factors in the hippocampus.
5.4 Synaptic Plasticity
Various forms of chronic stress have been demonstrated to alter the structure and plasticity of synapses. In general, chronic stress has been associated with dendritic atrophy and retraction (Conrad 2008), region-specific alterations in dendritic spine density (see Christoffel, Golden et al. (2011) for review) and reduced long term potentiation (LTP) that were reversible by anti-glucocorticoid treatment (Krugers, Goltstein et al. 2006). While the role of stress and glucocorticoid-mediated structural and synaptic changes have been intensely studied, the role of NFκB in synaptic signaling and architecture has only recently been recognized (Kaltschmidt, Widera et al. 2005). It is possible that the effect of chronic stress on plasticity is partially mediated by a stress-induced increase in NFκB activity. In fact, NFκB and IκB were found to localize in the postsynaptic densities and dendritic spines in the rat hippocampus and PFC (Suzuki, Mitake et al. 1997). NFκB is triggered by synaptic activation (Meffert, Chang et al. 2003) and upon termination of synaptic signal is transported from the synapse back to the nucleus of neurons via a dynein/dynactin motor complex (Widera, Klenke et al. 2016). Furthermore, NFκB signaling was found to serve as a link between nerve growth factor (NGF) signaling and Notch-dependent gene transcription to mediate neurite growth in hippocampal neurons (Salama-Cohen, Arevalo et al. 2005).
Altered NFκB activity has been implicated in chronic stress-induced synaptic alterations in the nucleus accumbens (NAc), a region that is involved in anhedonia, one of the core symptoms of depression. However, the functional implications of NFκB induction by stress appears to vary according to stressor type and duration, as well as the sex of the animal. On one hand, NFκB signaling in the NAc has been shown to protect against the pro-depressive effects of six days of chronic unpredictable stress (CUS) in ovariectomized female mice compared to intact females (LaPlant, Chakravarty et al. 2009). Conversely, following social defeat stress in male mice, IKK-mediated NFκB signaling promotes social avoidance behavior (Christoffel, Golden et al. 2012) and led to an increase in the number of stubby dendritic spines on medium spiny neurons in the NAc (Christoffel, Golden et al. 2011). In these studies, activation of IKK signaling pathway was found to be necessary and sufficient to mediate both behavioral and structural alterations. Environmental stressors, including exposure to several drugs of abuse such as cocaine (Ang, Chen et al. 2001, Russo, Wilkinson et al. 2009) and alcohol, also upregulate NFκB signaling in the nucleus accumbens.
NFκB-mediated alterations in synaptic plasticity also appears to be important for encoding and retaining fear memory particularly in the amygdala. A single two-hour session of cold restraint stress upregulated NFκB protein expression in the hippocampus, hypothalamus, amygdala, NAc, and PFC, and the antidepressant citalopram partially restored NFκB levels in the hippocampus and amygdala (Garabadu, Reddy et al. 2015). An increase in NFκB DNA-binding was found to be required in the rat amygdala during fear potentiated startle as well as LTP induction that are necessary for fear memory consolidation (Yeh, Lin et al. 2002). Furthermore, fear conditioning was found to increase acetylation of the p65 subunit of NFκB, and promote long-term retention of fear memory (Yeh, Lin et al. 2004). Collectively, the available data to date demonstrate a plausible role for both GR and NFκB in modulation at the synaptic level, but this does not rule out additional mechanisms by which both transcription factors may modulate neural function and thereby behavior.
6. Influence of Age and Sex on GR-NFκB Interactions
In order to promote clarity, the discussion to this point has focused primarily on adult endpoints and has not attended to sex differences. However, both development and sex differences are key variables to consider in attempts to characterize and understand GR and NFκB.
6.1 Development
Chronic stress paradigms alter inflammatory responsivity differently throughout development. Males and females undergoing prenatal stress exhibit an exaggerated corticosterone response following IL-1β challenge or exposure to acute restraint, and only females exhibit decreased GR mRNA in the CA2 region of the hippocampus (Brunton and Russell 2010). In addition, rats that were maternally separated during development exhibit a prolonged HPA response following acute stress exposure and decreased GR mRNA in the hippocampus (Ladd, Huot et al. 2004). In reference to immune challenge as the primary signal, early life exposure to an immune challenge may alter the balance between glucocorticoid and inflammatory signaling through adulthood. Rats exposed to a neonatal LPS challenge exhibit a decreased febrile response when subsequently challenged with LPS in adulthood, coupled with reduced expression of pro-inflammatory cytokines and reduced phosphorylation of IκBα. In addition, following neonatal immune challenge, rats also exhibit an increased corticosterone response to LPS, and impairing glucocorticoid signaling via adrenalectomy or treatment with the GR inhibitor RU-486, restored normal febrile and cytokine response to LPS challenge (Ellis, Mouihate et al. 2005). These data highlight the importance of inflammatory exposure during development in long-term alterations in the balance between GR and NFκB signaling, effects that occur throughout the lifespan. Furthermore, the impact of stress exposure and immune challenge on behavior and glucocorticoid regulation may pass through generations (Grundwald and Brunton 2015).
6.2 Sex Differences
Implications of developmental exposure to stress and inflammation can be influenced by sex. Though much of the mechanistic research concerning mechanisms of GR and NFκB activity were performed in vitro, recent studies have examined sex differences in the interactions of GR and NFκB activity in the context of stress or inflammatory exposure. For instance, adolescent female rats exposed to chronic stress exhibit impaired nuclear translocation of GR and increased FKBP5 gene expression in the hippocampus following an acute forced-swim stressor (Bourke, Raees et al. 2013), and these effects are coupled with increased depressive-like behaviors in adolescent females (Bourke and Neigh 2011). However, these alterations in GR signaling were absent in adolescent male rats who underwent chronic stress (Bourke, Raees et al. 2013). In addition to impacting GR function and regulation in a sex-specific manner, exposure to chronic adolescent stress may also exert sex-specific influences on inflammatory processes. Males with a history of chronic adolescent stress, while unchanged on baseline metrics, exhibit an exaggerated hippocampal pro-inflammatory response including enhanced expression of TNF-α, IL-1β, and IκB when challenged with LPS in adulthood (Pyter, Kelly et al. 2013). This inflammatory priming effect was not seen in female rats that underwent chronic adolescent stress. Chronic immune activation may also lead to sex-specific alterations in GR-NFκB dynamics. Treatment of rats with LPS for seven days led to increased nuclear accumulation of NFκB and depressive-like behavior in both male and female rats. However, while females displayed elevated nuclear GR, males displayed increased cytosolic GR, suggesting a potentially male-specific effect of impaired GR inhibition of NFκB (Adzic, Djordjevic et al. 2015). Although DNA binding or transcriptional activity was not measured in this study, and thus the relative effects of elevated nuclear GR and NFκB could not be determined (Adzic, Djordjevic et al. 2015), a separate study demonstrated a male-specific effects of LPS on nuclear glucocorticoid receptor expression 24-hours following the final LPS treatment (Brkic, Petrovic et al. 2016).
7. Conclusions
Through this review we have highlighted multiple mechanisms through which GR and NFκB activity can be modulated and provided examples of alterations in their activity and interactions in neuropsychiatric disease and models of chronic stress and chronic inflammation. Although the specific role of GR and NFκB in neuropsychiatric illness and the precise implications of altered balance between GR and NFκB-mediated transcription are unclear, it is evident that the two transcription factors impact activity of each other at multiple levels and treatments aimed at one will likely impact the other.
Understanding of the interactions between these transcription factors may yield therapeutic benefit. It is already known that following cognitive behavioral stress management, upregulated genes exhibit an overrepresentation of GREs and an underrepresentation of κREs, suggesting that with stress management, patients experience a shift to increased GR-mediated signaling and possibly more adaptive function (Antoni, Lutgendorf et al. 2012). Additional therapeutic avenues may be feasible through modulation of the molecular mechanisms that mediate these transcription factors such as interventions at the level of FKBP5 (Hinds, Stechschulte et al. 2014, Gaali, Kirschner et al. 2015, Hartmann, Wagner et al. 2015, Maiaru, Tochiki et al. 2016) which interacts with both GR and NFκB (Rein 2016). Furthermore, several anti-inflammatory and anti-cytokine agents have been considered for the treatment of mood disorders with moderate success. Some of the more successful anti-inflammatory strategies are NSAIDs and target the COX-2 pathway which is downstream of NFκB (Bremner and Heinrich 2002). Given the extensive crosstalk between NFκB and GR, amelioration of NFκB dysfunction is likely to reciprocally facilitate improved function of GR. Through continued study of GR and NFκB with attention to the nuances of the effects of these transcription factors and targeted strategies to modify their intricate interactions, improved intervention strategies for stress-induced and inflammatory disorders of both the central nervous system and other functions subject to the balance between the endocrine and immune systems may be achievable.
Highlights.
The endocrine-immune balance is disturbed by environmental or psychosocial stressors.
Concurrent GR and NFκB dysregulation is seen in stress-linked psychiatric disorders.
Mechanisms of GR-NFκB interactions are discussed in states of health and disease.
Chronic stress shifts GR-NFκB dynamics in brain regions underlying mood and behavior.
Developmental influences and sex differences modify the GR-NFκB balance.
Acknowledgments
The authors receive funding from NIH: NR014886 and MH110364. SAR was supported by NIH training grant T32-GM008602.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of interest
The authors have no conflicting interests.
References
- Adcock IM, Barnes PJ. Molecular mechanisms of corticosteroid resistance. Chest. 2008;134(2):394–401. doi: 10.1378/chest.08-0440. [DOI] [PubMed] [Google Scholar]
- Adzic M, Djordjevic J, Mitic M, Brkic Z, Lukic I, Radojcic M. The contribution of hypothalamic neuroendocrine, neuroplastic and neuroinflammatory processes to lipopolysaccharide-induced depressive-like behaviour in female and male rats: Involvement of glucocorticoid receptor and C/EBP-beta. Behav Brain Res. 2015;291:130–139. doi: 10.1016/j.bbr.2015.05.029. [DOI] [PubMed] [Google Scholar]
- Akiyama T, Shiraishi T, Qin J, Konno H, Akiyama N, Shinzawa M, Miyauchi M, Takizawa N, Yanai H, Ohashi H, Miyamoto-Sato E, Yanagawa H, Yong W, Shou W, Inoue J. Mitochondria-nucleus shuttling FK506-binding protein 51 interacts with TRAF proteins and facilitates the RIG-I-like receptor-mediated expression of type I IFN. PLoS One. 2014;9(5):e95992. doi: 10.1371/journal.pone.0095992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altonsy MO, Sasse SK, Phang TL, Gerber AN. Context-dependent cooperation between nuclear factor kappaB (NF-kappaB) and the glucocorticoid receptor at a TNFAIP3 intronic enhancer: a mechanism to maintain negative feedback control of inflammation. J Biol Chem. 2014;289(12):8231–8239. doi: 10.1074/jbc.M113.545178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ang E, Chen J, Zagouras P, Magna H, Holland J, Schaeffer E, Nestler EJ. Induction of nuclear factor-kappaB in nucleus accumbens by chronic cocaine administration. J Neurochem. 2001;79(1):221–224. doi: 10.1046/j.1471-4159.2001.00563.x. [DOI] [PubMed] [Google Scholar]
- Antoni MH, Lutgendorf SK, Blomberg B, Carver CS, Lechner S, Diaz A, Stagl J, Arevalo JM, Cole SW. Cognitive-behavioral stress management reverses anxiety-related leukocyte transcriptional dynamics. Biol Psychiatry. 2012;71(4):366–372. doi: 10.1016/j.biopsych.2011.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes PJ, Adcock IM. Glucocorticoid resistance in inflammatory diseases. Lancet. 2009;373(9678):1905–1917. doi: 10.1016/S0140-6736(09)60326-3. [DOI] [PubMed] [Google Scholar]
- Bartholome B, Spies CM, Gaber T, Schuchmann S, Berki T, Kunkel D, Bienert M, Radbruch A, Burmester GR, Lauster R, Scheffold A, Buttgereit F. Membrane glucocorticoid receptors (mGCR) are expressed in normal human peripheral blood mononuclear cells and up-regulated after in vitro stimulation and in patients with rheumatoid arthritis. Faseb j. 2004;18(1):70–80. doi: 10.1096/fj.03-0328com. [DOI] [PubMed] [Google Scholar]
- Baxter JD, Rousseau GG, Benson MC, Garcea RL, Ito J, Tomkins GM. Role of DNA and specific cytoplasmic receptors in glucocorticoid action. Proc Natl Acad Sci U S A. 1972;69(7):1892–1896. doi: 10.1073/pnas.69.7.1892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behl C, Lezoualc'h F, Trapp T, Widmann M, Skutella T, Holsboer F. Glucocorticoids enhance oxidative stress-induced cell death in hippocampal neurons in vitro. Endocrinology. 1997;138(1):101–106. doi: 10.1210/endo.138.1.4835. [DOI] [PubMed] [Google Scholar]
- Bell PA, Munck A. Steroid binding and stabilization of glucocorticoid-receptor proteins from rat thymus. Biochem J. 1972;126(3):11p. doi: 10.1042/bj1260011pa. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benveniste EN, Sparacio SM, Norris JG, Grenett HE, Fuller GM. Induction and regulation of interleukin-6 gene expression in rat astrocytes. J Neuroimmunol. 1990;30(2–3):201–212. doi: 10.1016/0165-5728(90)90104-u. [DOI] [PubMed] [Google Scholar]
- Bhakar AL, Tannis LL, Zeindler C, Russo MP, Jobin C, Park DS, MacPherson S, Barker PA. Constitutive nuclear factor-kappa B activity is required for central neuron survival. J Neurosci. 2002;22(19):8466–8475. doi: 10.1523/JNEUROSCI.22-19-08466.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bierhaus A, Wolf J, Andrassy M, Rohleder N, Humpert PM, Petrov D, Ferstl R, von Eynatten M, Wendt T, Rudofsky G, Joswig M, Morcos M, Schwaninger M, McEwen B, Kirschbaum C, Nawroth PP. A mechanism converting psychosocial stress into mononuclear cell activation. Proc Natl Acad Sci U S A. 2003;100(4):1920–1925. doi: 10.1073/pnas.0438019100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Binder EB. The role of FKBP5, a co-chaperone of the glucocorticoid receptor in the pathogenesis and therapy of affective and anxiety disorders. Psychoneuroendocrinology. 2009;34(Suppl 1):S186–195. doi: 10.1016/j.psyneuen.2009.05.021. [DOI] [PubMed] [Google Scholar]
- Binder EB, Salyakina D, Lichtner P, Wochnik GM, Ising M, Putz B, Papiol S, Seaman S, Lucae S, Kohli MA, Nickel T, Kunzel HE, Fuchs B, Majer M, Pfennig A, Kern N, Brunner J, Modell S, Baghai T, Deiml T, Zill P, Bondy B, Rupprecht R, Messer T, Kohnlein O, Dabitz H, Bruckl T, Muller N, Pfister H, Lieb R, Mueller JC, Lohmussaar E, Strom TM, Bettecken T, Meitinger T, Uhr M, Rein T, Holsboer F, Muller-Myhsok B. Polymorphisms in FKBP5 are associated with increased recurrence of depressive episodes and rapid response to antidepressant treatment. Nat Genet. 2004;36(12):1319–1325. doi: 10.1038/ng1479. [DOI] [PubMed] [Google Scholar]
- Black DS, Cole SW, Irwin MR, Breen E, St Cyr NM, Nazarian N, Khalsa DS, Lavretsky H. Yogic meditation reverses NF-kappaB and IRF-related transcriptome dynamics in leukocytes of family dementia caregivers in a randomized controlled trial. Psychoneuroendocrinology. 2013;38(3):348–355. doi: 10.1016/j.psyneuen.2012.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blind RD, Garabedian MJ. Differential recruitment of glucocorticoid receptor phosphoisoforms to glucocorticoid-induced genes. J Steroid Biochem Mol Biol. 2008;109(1–2):150–157. doi: 10.1016/j.jsbmb.2008.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boersma MC, Dresselhaus EC, De Biase LM, Mihalas AB, Bergles DE, Meffert MK. A requirement for nuclear factor-kappaB in developmental and plasticity-associated synaptogenesis. J Neurosci. 2011;31(14):5414–5425. doi: 10.1523/JNEUROSCI.2456-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourke CH, Neigh GN. Behavioral effects of chronic adolescent stress are sustained and sexually dimorphic. Horm Behav. 2011;60(1):112–120. doi: 10.1016/j.yhbeh.2011.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourke CH, Raees MQ, Malviya S, Bradburn CA, Binder EB, Neigh GN. Glucocorticoid sensitizers Bag1 and Ppid are regulated by adolescent stress in a sex-dependent manner. Psychoneuroendocrinology. 2013;38(1):84–93. doi: 10.1016/j.psyneuen.2012.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouwmeester T, Bauch A, Ruffner H, Angrand PO, Bergamini G, Croughton K, Cruciat C, Eberhard D, Gagneur J, Ghidelli S, Hopf C, Huhse B, Mangano R, Michon AM, Schirle M, Schlegl J, Schwab M, Stein MA, Bauer A, Casari G, Drewes G, Gavin AC, Jackson DB, Joberty G, Neubauer G, Rick J, Kuster B, Superti-Furga G. A physical and functional map of the human TNF-alpha/NF-kappa B signal transduction pathway. Nat Cell Biol. 2004;6(2):97–105. doi: 10.1038/ncb1086. [DOI] [PubMed] [Google Scholar]
- Braun S, Liebetrau W, Berning B, Behl C. Dexamethasone-enhanced sensitivity of mouse hippocampal HT22 cells for oxidative stress is associated with the suppression of nuclear factor-kappaB. Neurosci Lett. 2000;295(3):101–104. doi: 10.1016/s0304-3940(00)01603-7. [DOI] [PubMed] [Google Scholar]
- Bremner P, Heinrich M. Natural products as targeted modulators of the nuclear factor-kappaB pathway. J Pharm Pharmacol. 2002;54(4):453–472. doi: 10.1211/0022357021778637. [DOI] [PubMed] [Google Scholar]
- Brenner T, Yamin A, Abramsky O, Gallily R. Stimulation of tumor necrosis factor-alpha production by mycoplasmas and inhibition by dexamethasone in cultured astrocytes. Brain Res. 1993;608(2):273–279. doi: 10.1016/0006-8993(93)91468-8. [DOI] [PubMed] [Google Scholar]
- Bresnick EH, Dalman FC, Sanchez ER, Pratt WB. Evidence that the 90-kDa heat shock protein is necessary for the steroid binding conformation of the L cell glucocorticoid receptor. J Biol Chem. 1989;264(9):4992–4997. [PubMed] [Google Scholar]
- Brkic Z, Petrovic Z, Franic D, Mitic M, Adzic M. Male-specific effects of lipopolysaccharide on glucocorticoid receptor nuclear translocation in the prefrontal cortex of depressive rats. Psychopharmacology (Berl) 2016;233(18):3315–3330. doi: 10.1007/s00213-016-4374-y. [DOI] [PubMed] [Google Scholar]
- Brunton PJ, Russell JA. Prenatal social stress in the rat programmes neuroendocrine and behavioural responses to stress in the adult offspring: sex-specific effects. J Neuroendocrinol. 2010;22(4):258–271. doi: 10.1111/j.1365-2826.2010.01969.x. [DOI] [PubMed] [Google Scholar]
- Camandola S, Mattson MP. NF-kappa B as a therapeutic target in neurodegenerative diseases. Expert Opin Ther Targets. 2007;11(2):123–132. doi: 10.1517/14728222.11.2.123. [DOI] [PubMed] [Google Scholar]
- Carrillo-de Sauvage MA, Maatouk L, Arnoux I, Pasco M, Sanz Diez A, Delahaye M, Herrero MT, Newman TA, Calvo CF, Audinat E, Tronche F, Vyas S. Potent and multiple regulatory actions of microglial glucocorticoid receptors during CNS inflammation. Cell Death Differ. 2013;20(11):1546–1557. doi: 10.1038/cdd.2013.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler VL, Maler BA, Yamamoto KR. DNA sequences bound specifically by glucocorticoid receptor in vitro render a heterologous promoter hormone responsive in vivo. Cell. 1983;33(2):489–499. doi: 10.1016/0092-8674(83)90430-0. [DOI] [PubMed] [Google Scholar]
- Chantong B, Kratschmar DV, Nashev LG, Balazs Z, Odermatt A. Mineralocorticoid and glucocorticoid receptors differentially regulate NF-kappaB activity and pro-inflammatory cytokine production in murine BV-2 microglial cells. J Neuroinflammation. 2012;9:260. doi: 10.1186/1742-2094-9-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen LF, Mu Y, Greene WC. Acetylation of RelA at discrete sites regulates distinct nuclear functions of NF-kappaB. EMBO J. 2002;21(23):6539–6548. doi: 10.1093/emboj/cdf660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W, Dang T, Blind RD, Wang Z, Cavasotto CN, Hittelman AB, Rogatsky I, Logan SK, Garabedian MJ. Glucocorticoid receptor phosphorylation differentially affects target gene expression. Mol Endocrinol. 2008;22(8):1754–1766. doi: 10.1210/me.2007-0219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng T, Dimitrov S, Pruitt C, Hong S. Glucocorticoid mediated regulation of inflammation in human monocytes is associated with depressive mood and obesity. Psychoneuroendocrinology. 2016;66:195–204. doi: 10.1016/j.psyneuen.2016.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Y, Pardo M, Armini Rde S, Martinez A, Mouhsine H, Zagury JF, Jope RS, Beurel E. Stress-induced neuroinflammation is mediated by GSK3-dependent TLR4 signaling that promotes susceptibility to depression-like behavior. Brain Behav Immun. 2016;53:207–222. doi: 10.1016/j.bbi.2015.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung-Flynn J, Roberts PJ, Riggs DL, Smith DF. C-terminal sequences outside the tetratricopeptide repeat domain of FKBP51 and FKBP52 cause differential binding to Hsp90. J Biol Chem. 2003;278(19):17388–17394. doi: 10.1074/jbc.M300955200. [DOI] [PubMed] [Google Scholar]
- Christoffel DJ, Golden SA, Dumitriu D, Robison AJ, Janssen WG, Ahn HF, Krishnan V, Reyes CM, Han MH, Ables JL, Eisch AJ, Dietz DM, Ferguson D, Neve RL, Greengard P, Kim Y, Morrison JH, Russo SJ. IkappaB kinase regulates social defeat stress-induced synaptic and behavioral plasticity. J Neurosci. 2011;31(1):314–321. doi: 10.1523/JNEUROSCI.4763-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christoffel DJ, Golden SA, Heshmati M, Graham A, Birnbaum S, Neve RL, Hodes GE, Russo SJ. Effects of inhibitor of kappaB kinase activity in the nucleus accumbens on emotional behavior. Neuropsychopharmacology. 2012;37(12):2615–2623. doi: 10.1038/npp.2012.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christoffel DJ, Golden SA, Russo SJ. Structural and synaptic plasticity in stress-related disorders. Rev Neurosci. 2011;22(5):535–549. doi: 10.1515/RNS.2011.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cidlowski JA, Cidlowski NB. Regulation of glucocorticoid receptors by glucocorticoids in cultured HeLa S3 cells. Endocrinology. 1981;109(6):1975–1982. doi: 10.1210/endo-109-6-1975. [DOI] [PubMed] [Google Scholar]
- Clark A. Crosstalk between glucocorticoids and mitogen-activated protein kinase signalling pathways. Current Opinion in Pharmacology. 2003;3(4):404–411. doi: 10.1016/s1471-4892(03)00073-0. [DOI] [PubMed] [Google Scholar]
- Cohen H, Kozlovsky N, Matar MA, Zohar J, Kaplan Z. The characteristic long-term upregulation of hippocampal NF-kappaB complex in PTSD-like behavioral stress response is normalized by high-dose corticosterone and pyrrolidine dithiocarbamate administered immediately after exposure. Neuropsychopharmacology. 2011;36(11):2286–2302. doi: 10.1038/npp.2011.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad CD. Chronic stress-induced hippocampal vulnerability: the glucocorticoid vulnerability hypothesis. Rev Neurosci. 2008;19(6):395–411. doi: 10.1515/revneuro.2008.19.6.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalman FC, Sanchez ER, Lin AL, Perini F, Pratt WB. Localization of phosphorylation sites with respect to the functional domains of the mouse L cell glucocorticoid receptor. J Biol Chem. 1988;263(25):12259–12267. [PubMed] [Google Scholar]
- Dantzer R, O'Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci. 2008;9(1):46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies TH, Ning YM, Sanchez ER. A new first step in activation of steroid receptors: hormone-induced switching of FKBP51 and FKBP52 immunophilins. J Biol Chem. 2002;277(7):4597–4600. doi: 10.1074/jbc.C100531200. [DOI] [PubMed] [Google Scholar]
- Dawson C, Dhanda A, Conway-Campbell B, Dimambro A, Lightman S, Dayan C. NFkappaB and glucocorticoid receptor activity in steroid resistance. J Recept Signal Transduct Res. 2012;32(1):29–35. doi: 10.3109/10799893.2011.641977. [DOI] [PubMed] [Google Scholar]
- De Bosscher K, Schmitz ML, Vanden Berghe W, Plaisance S, Fiers W, Haegeman G. Glucocorticoid-mediated repression of nuclear factor-kappaB-dependent transcription involves direct interference with transactivation. Proc Natl Acad Sci U S A. 1997;94(25):13504–13509. doi: 10.1073/pnas.94.25.13504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Bosscher K, Vanden Berghe W, Haegeman G. The interplay between the glucocorticoid receptor and nuclear factor-kappaB or activator protein-1: molecular mechanisms for gene repression. Endocr Rev. 2003;24(4):488–522. doi: 10.1210/er.2002-0006. [DOI] [PubMed] [Google Scholar]
- De Bosscher K, Vanden Berghe W, Vermeulen L, Plaisance S, Boone E, Haegeman G. Glucocorticoids repress NF-kappaB-driven genes by disturbing the interaction of p65 with the basal transcription machinery, irrespective of coactivator levels in the cell. Proc Natl Acad Sci U S A. 2000;97(8):3919–3924. doi: 10.1073/pnas.97.8.3919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Pablos RM, Villaran RF, Arguelles S, Herrera AJ, Venero JL, Ayala A, Cano J, Machado A. Stress increases vulnerability to inflammation in the rat prefrontal cortex. J Neurosci. 2006;26(21):5709–5719. doi: 10.1523/JNEUROSCI.0802-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demand J, Alberti S, Patterson C, Hohfeld J. Cooperation of a ubiquitin domain protein and an E3 ubiquitin ligase during chaperone/proteasome coupling. Curr Biol. 2001;11(20):1569–1577. doi: 10.1016/s0960-9822(01)00487-0. [DOI] [PubMed] [Google Scholar]
- Deng Q, Riquelme D, Trinh L, Low MJ, Tomic M, Stojilkovic S, Aguilera G. Rapid Glucocorticoid Feedback Inhibition of ACTH Secretion Involves Ligand-Dependent Membrane Association of Glucocorticoid Receptors. Endocrinology. 2015;156(9):3215–3227. doi: 10.1210/EN.2015-1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhabhar FS. A hassle a day may keep the pathogens away: The fight-or-flight stress response and the augmentation of immune function. Integr Comp Biol. 2009;49(3):215–236. doi: 10.1093/icb/icp045. [DOI] [PubMed] [Google Scholar]
- Dittmar KD, Pratt WB. Folding of the glucocorticoid receptor by the reconstituted Hsp90-based chaperone machinery. The initial hsp90.p60.hsp70-dependent step is sufficient for creating the steroid binding conformation. J Biol Chem. 1997;272(20):13047–13054. doi: 10.1074/jbc.272.20.13047. [DOI] [PubMed] [Google Scholar]
- Djordjevic A, Adzic M, Djordjevic J, Radojcic MB. Chronic social isolation is related to both upregulation of plasticity genes and initiation of proapoptotic signaling in Wistar rat hippocampus. J Neural Transm (Vienna) 2009;116(12):1579–1589. doi: 10.1007/s00702-009-0286-x. [DOI] [PubMed] [Google Scholar]
- Djordjevic A, Adzic M, Djordjevic J, Radojcic MB. Chronic social isolation suppresses proplastic response and promotes proapoptotic signalling in prefrontal cortex of Wistar rats. J Neurosci Res. 2010;88(11):2524–2533. doi: 10.1002/jnr.22403. [DOI] [PubMed] [Google Scholar]
- Doucas V, Shi Y, Miyamoto S, West A, Verma I, Evans RM. Cytoplasmic catalytic subunit of protein kinase A mediates cross-repression by NF-kappa B and the glucocorticoid receptor. Proc Natl Acad Sci U S A. 2000;97(22):11893–11898. doi: 10.1073/pnas.220413297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis S, Mouihate A, Pittman QJ. Early life immune challenge alters innate immune responses to lipopolysaccharide: implications for host defense as adults. Faseb j. 2005;19(11):1519–1521. doi: 10.1096/fj.04-3569fje. [DOI] [PubMed] [Google Scholar]
- Erlejman AG, De Leo SA, Mazaira GI, Molinari AM, Camisay MF, Fontana V, Cox MB, Piwien-Pilipuk G, Galigniana MD. NF-kappaB transcriptional activity is modulated by FK506-binding proteins FKBP51 and FKBP52: a role for peptidyl-prolyl isomerase activity. J Biol Chem. 2014;289(38):26263–26276. doi: 10.1074/jbc.M114.582882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fagerlund R, Melen K, Cao X, Julkunen I. NF-kappaB p52, RelB and c-Rel are transported into the nucleus via a subset of importin alpha molecules. Cell Signal. 2008;20(8):1442–1451. doi: 10.1016/j.cellsig.2008.03.012. [DOI] [PubMed] [Google Scholar]
- Feldker DE, Morsink MC, Veenema AH, Datson NA, Proutski V, Lathouwers D, de Kloet ER, Vreugdenhil E. The effect of chronic exposure to highly aggressive mice on hippocampal gene expression of non-aggressive subordinates. Brain Res. 2006;1089(1):10–20. doi: 10.1016/j.brainres.2006.02.110. [DOI] [PubMed] [Google Scholar]
- Fleshner M. Stress-evoked sterile inflammation, danger associated molecular patterns (DAMPs), microbial associated molecular patterns (MAMPs) and the inflammasome. Brain Behav Immun. 2013;27(1):1–7. doi: 10.1016/j.bbi.2012.08.012. [DOI] [PubMed] [Google Scholar]
- Frank MG, Baratta MV, Sprunger DB, Watkins LR, Maier SF. Microglia serve as a neuroimmune substrate for stress-induced potentiation of CNS pro-inflammatory cytokine responses. Brain Behav Immun. 2007;21(1):47–59. doi: 10.1016/j.bbi.2006.03.005. [DOI] [PubMed] [Google Scholar]
- Frank MG, Hershman SA, Weber MD, Watkins LR, Maier SF. Chronic exposure to exogenous glucocorticoids primes microglia to pro-inflammatory stimuli and induces NLRP3 mRNA in the hippocampus. Psychoneuroendocrinology. 2014;40:191–200. doi: 10.1016/j.psyneuen.2013.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank MG, Miguel ZD, Watkins LR, Maier SF. Prior exposure to glucocorticoids sensitizes the neuroinflammatory and peripheral inflammatory responses to E. coli lipopolysaccharide. Brain Behav Immun. 2010;24(1):19–30. doi: 10.1016/j.bbi.2009.07.008. [DOI] [PubMed] [Google Scholar]
- Frank MG, Thompson BM, Watkins LR, Maier SF. Glucocorticoids mediate stress-induced priming of microglial pro-inflammatory responses. Brain Behav Immun. 2012;26(2):337–345. doi: 10.1016/j.bbi.2011.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaali S, Kirschner A, Cuboni S, Hartmann J, Kozany C, Balsevich G, Namendorf C, Fernandez-Vizarra P, Sippel C, Zannas AS, Draenert R, Binder EB, Almeida OF, Ruhter G, Uhr M, Schmidt MV, Touma C, Bracher A, Hausch F. Selective inhibitors of the FK506-binding protein 51 by induced fit. Nat Chem Biol. 2015;11(1):33–37. doi: 10.1038/nchembio.1699. [DOI] [PubMed] [Google Scholar]
- Galigniana MD, Harrell JM, Murphy PJ, Chinkers M, Radanyi C, Renoir JM, Zhang M, Pratt WB. Binding of hsp90-associated immunophilins to cytoplasmic dynein: direct binding and in vivo evidence that the peptidylprolyl isomerase domain is a dynein interaction domain. Biochemistry. 2002;41(46):13602–13610. doi: 10.1021/bi020399z. [DOI] [PubMed] [Google Scholar]
- Galigniana MD, Radanyi C, Renoir JM, Housley PR, Pratt WB. Evidence that the peptidylprolyl isomerase domain of the hsp90-binding immunophilin FKBP52 is involved in both dynein interaction and glucocorticoid receptor movement to the nucleus. J Biol Chem. 2001;276(18):14884–14889. doi: 10.1074/jbc.M010809200. [DOI] [PubMed] [Google Scholar]
- Galliher-Beckley AJ, Williams JG, Cidlowski JA. Ligand-independent phosphorylation of the glucocorticoid receptor integrates cellular stress pathways with nuclear receptor signaling. Mol Cell Biol. 2011;31(23):4663–4675. doi: 10.1128/MCB.05866-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galliher-Beckley AJ, Williams JG, Collins JB, Cidlowski JA. Glycogen synthase kinase 3beta-mediated serine phosphorylation of the human glucocorticoid receptor redirects gene expression profiles. Mol Cell Biol. 2008;28(24):7309–7322. doi: 10.1128/MCB.00808-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao N, Hibi Y, Cueno M, Asamitsu K, Okamoto T. A-kinase-interacting protein 1 (AKIP1) acts as a molecular determinant of PKA in NF-kappaB signaling. J Biol Chem. 2010;285(36):28097–28104. doi: 10.1074/jbc.M110.116566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garabadu D, Reddy BC, Krishnamurthy S. Citalopram protects against cold-restraint stress-induced activation of brain-derived neurotrophic factor and expression of nuclear factor kappa-light-chain-enhancer of activated B cells in rats. J Mol Neurosci. 2015;55(2):355–366. doi: 10.1007/s12031-014-0334-3. [DOI] [PubMed] [Google Scholar]
- Garate I, Garcia-Bueno B, Madrigal JL, Caso JR, Alou L, Gomez-Lus ML, Mico JA, Leza JC. Stress-induced neuroinflammation: role of the Toll-like receptor-4 pathway. Biol Psychiatry. 2013;73 doi: 10.1016/j.biopsych.2012.07.005. [DOI] [PubMed] [Google Scholar]
- Giguere V, Hollenberg SM, Rosenfeld MG, Evans RM. Functional domains of the human glucocorticoid receptor. Cell. 1986;46(5):645–652. doi: 10.1016/0092-8674(86)90339-9. [DOI] [PubMed] [Google Scholar]
- Gray JD, Rubin TG, Hunter RG, McEwen BS. Hippocampal gene expression changes underlying stress sensitization and recovery. Mol Psychiatry. 2014;19(11):1171–1178. doi: 10.1038/mp.2013.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grundwald NJ, Brunton PJ. Prenatal stress programs neuroendocrine stress responses and affective behaviors in second generation rats in a sex-dependent manner. Psychoneuroendocrinology. 2015;62:204–216. doi: 10.1016/j.psyneuen.2015.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guidotti G, Calabrese F, Anacker C, Racagni G, Pariante CM, Riva MA. Glucocorticoid receptor and FKBP5 expression is altered following exposure to chronic stress: modulation by antidepressant treatment. Neuropsychopharmacology. 2013;38(4):616–627. doi: 10.1038/npp.2012.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guzhova IV, Darieva ZA, Melo AR, Margulis BA. Major stress protein Hsp70 interacts with NF-kB regulatory complex in human T-lymphoma cells. Cell Stress Chaperones. 1997;2(2):132–139. doi: 10.1379/1466-1268(1997)002<0132:msphiw>2.3.co;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagimoto S, Arima H, Adachi K, Ito Y, Suga H, Sugimura Y, Goto M, Banno R, Oiso Y. Expression of neuropeptide Y and agouti-related protein mRNA stimulated by glucocorticoids is attenuated via NF-kappaB p65 under ER stress in mouse hypothalamic cultures. Neurosci Lett. 2013;553:165–169. doi: 10.1016/j.neulet.2013.08.040. [DOI] [PubMed] [Google Scholar]
- Harrell JM, Murphy PJ, Morishima Y, Chen H, Mansfield JF, Galigniana MD, Pratt WB. Evidence for glucocorticoid receptor transport on microtubules by dynein. J Biol Chem. 2004;279(52):54647–54654. doi: 10.1074/jbc.M406863200. [DOI] [PubMed] [Google Scholar]
- Hartmann J, Wagner KV, Gaali S, Kirschner A, Kozany C, Ruhter G, Dedic N, Hausl AS, Hoeijmakers L, Westerholz S, Namendorf C, Gerlach T, Uhr M, Chen A, Deussing JM, Holsboer F, Hausch F, Schmidt MV. Pharmacological Inhibition of the Psychiatric Risk Factor FKBP51 Has Anxiolytic Properties. J Neurosci. 2015;35(24):9007–9016. doi: 10.1523/JNEUROSCI.4024-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hecht K, Carlstedt-Duke J, Stierna P, Gustafsson J, Bronnegard M, Wikstrom AC. Evidence that the beta-isoform of the human glucocorticoid receptor does not act as a physiologically significant repressor. J Biol Chem. 1997;272(42):26659–26664. doi: 10.1074/jbc.272.42.26659. [DOI] [PubMed] [Google Scholar]
- Hinds TD, Stechschulte LA, Elkhairi F, Sanchez ER. Analysis of FK506, timcodar (VX-853) and FKBP51 and FKBP52 chaperones in control of glucocorticoid receptor activity and phosphorylation. Pharmacol Res Perspect. 2014;2(6):e00076. doi: 10.1002/prp2.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinz M, Broemer M, Arslan SC, Otto A, Mueller EC, Dettmer R, Scheidereit C. Signal responsiveness of IkappaB kinases is determined by Cdc37-assisted transient interaction with Hsp90. J Biol Chem. 2007;282(44):32311–32319. doi: 10.1074/jbc.M705785200. [DOI] [PubMed] [Google Scholar]
- Hoffmann A, Baltimore D. Circuitry of nuclear factor kappaB signaling. Immunol Rev. 2006;210:171–186. doi: 10.1111/j.0105-2896.2006.00375.x. [DOI] [PubMed] [Google Scholar]
- Hollenberg SM, Weinberger C, Ong ES, Cerelli G, Oro A, Lebo R, Thompson EB, Rosenfeld MG, Evans RM. Primary structure and expression of a functional human glucocorticoid receptor cDNA. Nature. 1985;318(6047):635–641. doi: 10.1038/318635a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honda M, Orii F, Ayabe T, Imai S, Ashida T, Obara T, Kohgo Y. Expression of glucocorticoid receptor beta in lymphocytes of patients with glucocorticoid-resistant ulcerative colitis. Gastroenterology. 2000;118(5):859–866. doi: 10.1016/s0016-5085(00)70172-7. [DOI] [PubMed] [Google Scholar]
- Horowitz MA, Zunszain PA. Neuroimmune and neuroendocrine abnormalities in depression: two sides of the same coin. Ann N Y Acad Sci. 2015;1351:68–79. doi: 10.1111/nyas.12781. [DOI] [PubMed] [Google Scholar]
- Hu W, Zhang Y, Wu W, Yin Y, Huang D, Wang Y, Li W, Li W. Chronic glucocorticoids exposure enhances neurodegeneration in the frontal cortex and hippocampus via NLRP-1 inflammasome activation in male mice. Brain Behav Immun. 2016;52:58–70. doi: 10.1016/j.bbi.2015.09.019. [DOI] [PubMed] [Google Scholar]
- Hudson WH, Youn C, Ortlund EA. The structural basis of direct glucocorticoid-mediated transrepression. Nat Struct Mol Biol. 2013;20(1):53–58. doi: 10.1038/nsmb.2456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Israel A. The IKK complex, a central regulator of NF-kappaB activation. Cold Spring Harb Perspect Biol. 2010;2(3):a000158. doi: 10.1101/cshperspect.a000158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh M, Adachi M, Yasui H, Takekawa M, Tanaka H, Imai K. Nuclear export of glucocorticoid receptor is enhanced by c-Jun N-terminal kinase-mediated phosphorylation. Mol Endocrinol. 2002;16(10):2382–2392. doi: 10.1210/me.2002-0144. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Aoki Y, Katahira M, Oiso Y, Saito H. Non-genomic mechanisms of glucocorticoid inhibition of adrenocorticotropin secretion: possible involvement of GTP-binding protein. Biochem Biophys Res Commun. 1997;235(2):295–299. doi: 10.1006/bbrc.1997.6785. [DOI] [PubMed] [Google Scholar]
- Jiang W, Cazacu S, Xiang C, Zenklusen JC, Fine HA, Berens M, Armstrong B, Brodie C, Mikkelsen T. FK506 Binding Protein Mediates Glioma Cell Growth and Sensitivity to Rapamycin Treatment by Regulating NF-κB Signaling Pathway. Neoplasia. 2008;10(3):235–243. doi: 10.1593/neo.07929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung SH, Wang Y, Kim T, Tarr A, Reader B, Powell N, Sheridan JF. Molecular mechanisms of repeated social defeat-induced glucocorticoid resistance: Role of microRNA. Brain Behav Immun. 2015;44:195–206. doi: 10.1016/j.bbi.2014.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaltschmidt B, Kaltschmidt C. NF-kappaB in the nervous system. Cold Spring Harb Perspect Biol. 2009;1(3):a001271. doi: 10.1101/cshperspect.a001271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaltschmidt B, Widera D, Kaltschmidt C. Signaling via NF-kappaB in the nervous system. Biochim Biophys Acta. 2005;1745(3):287–299. doi: 10.1016/j.bbamcr.2005.05.009. [DOI] [PubMed] [Google Scholar]
- Kaltschmidt C, Kaltschmidt B, Neumann H, Wekerle H, Baeuerle PA. Constitutive NF-kappa B activity in neurons. Mol Cell Biol. 1994;14(6):3981–3992. doi: 10.1128/mcb.14.6.3981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassed CA, Herkenham M. NF-kappaB p50-deficient mice show reduced anxiety-like behaviors in tests of exploratory drive and anxiety. Behav Brain Res. 2004;154(2):577–584. doi: 10.1016/j.bbr.2004.03.026. [DOI] [PubMed] [Google Scholar]
- Klenke C, Widera D, Engelen T, Muller J, Noll T, Niehaus K, Schmitz ML, Kaltschmidt B, Kaltschmidt C. Hsc70 is a novel interactor of NF-kappaB p65 in living hippocampal neurons. PLoS One. 2013;8(6):e65280. doi: 10.1371/journal.pone.0065280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo JW, Russo SJ, Ferguson D, Nestler EJ, Duman RS. Nuclear factor-kappaB is a critical mediator of stress-impaired neurogenesis and depressive behavior. Proc Natl Acad Sci U S A. 2010;107(6):2669–2674. doi: 10.1073/pnas.0910658107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozaci DL, Chernajovsky Y, Chikanza IC. The differential expression of corticosteroid receptor isoforms in corticosteroid-resistant and -sensitive patients with rheumatoid arthritis. Rheumatology (Oxford) 2007;46(4):579–585. doi: 10.1093/rheumatology/kel276. [DOI] [PubMed] [Google Scholar]
- Krugers HJ, Goltstein PM, van der Linden S, Joels M. Blockade of glucocorticoid receptors rapidly restores hippocampal CA1 synaptic plasticity after exposure to chronic stress. Eur J Neurosci. 2006;23(11):3051–3055. doi: 10.1111/j.1460-9568.2006.04842.x. [DOI] [PubMed] [Google Scholar]
- Kuebler U, Zuccarella-Hackl C, Arpagaus A, Wolf JM, Farahmand F, von Kanel R, Ehlert U, Wirtz PH. Stress-induced modulation of NF-kappaB activation, inflammation-associated gene expression, and cytokine levels in blood of healthy men. Brain Behav Immun. 2015;46:87–95. doi: 10.1016/j.bbi.2014.12.024. [DOI] [PubMed] [Google Scholar]
- Ladd CO, Huot RL, Thrivikraman KV, Nemeroff CB, Plotsky PM. Long-term adaptations in glucocorticoid receptor and mineralocorticoid receptor mRNA and negative feedback on the hypothalamo-pituitary-adrenal axis following neonatal maternal separation. Biol Psychiatry. 2004;55(4):367–375. doi: 10.1016/j.biopsych.2003.10.007. [DOI] [PubMed] [Google Scholar]
- Lamirand A, Ramauge M, Pierre M, Courtin F. Bacterial lipopolysaccharide induces type 2 deiodinase in cultured rat astrocytes. J Endocrinol. 2011;208(2):183–192. doi: 10.1677/JOE-10-0218. [DOI] [PubMed] [Google Scholar]
- LaPlant Q, Chakravarty S, Vialou V, Mukherjee S, Koo JW, Kalahasti G, Bradbury KR, Taylor SV, Maze I, Kumar A, Graham A, Birnbaum SG, Krishnan V, Truong HT, Neve RL, Nestler EJ, Russo SJ. Role of nuclear factor kappaB in ovarian hormone-mediated stress hypersensitivity in female mice. Biol Psychiatry. 2009;65(10):874–880. doi: 10.1016/j.biopsych.2009.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasa M, Abraham SM, Boucheron C, Saklatvala J, Clark AR. Dexamethasone Causes Sustained Expression of Mitogen-Activated Protein Kinase (MAPK) Phosphatase 1 and Phosphatase-Mediated Inhibition of MAPK p38. Molecular and Cellular Biology. 2002;22(22):7802–7811. doi: 10.1128/MCB.22.22.7802-7811.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung DY, Hamid Q, Vottero A, Szefler SJ, Surs W, Minshall E, Chrousos GP, Klemm DJ. Association of glucocorticoid insensitivity with increased expression of glucocorticoid receptor beta. J Exp Med. 1997;186(9):1567–1574. doi: 10.1084/jem.186.9.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levinson BB, Baxter JD, Rousseau GG, Tomkins GM. Cellular site of glucocorticoid-receptor complex formation. Science. 1972;175(4018):189–190. doi: 10.1126/science.175.4018.189. [DOI] [PubMed] [Google Scholar]
- Liden J, Delaunay F, Rafter I, Gustafsson J, Okret S. A new function for the C-terminal zinc finger of the glucocorticoid receptor. Repression of RelA transactivation. J Biol Chem. 1997;272(34):21467–21472. doi: 10.1074/jbc.272.34.21467. [DOI] [PubMed] [Google Scholar]
- Listwak SJ, Rathore P, Herkenham M. Minimal NF-kappaB activity in neurons. Neuroscience. 2013;250:282–299. doi: 10.1016/j.neuroscience.2013.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long F, Wang YX, Liu L, Zhou J, Cui RY, Jiang CL. Rapid nongenomic inhibitory effects of glucocorticoids on phagocytosis and superoxide anion production by macrophages. Steroids. 2005;70(1):55–61. doi: 10.1016/j.steroids.2004.10.004. [DOI] [PubMed] [Google Scholar]
- Lowenberg M, Tuynman J, Bilderbeek J, Gaber T, Buttgereit F, van Deventer S, Peppelenbosch M, Hommes D. Rapid immunosuppressive effects of glucocorticoids mediated through Lck and Fyn. Blood. 2005;106(5):1703–1710. doi: 10.1182/blood-2004-12-4790. [DOI] [PubMed] [Google Scholar]
- Lowenberg M, Verhaar AP, Bilderbeek J, Marle J, Buttgereit F, Peppelenbosch MP, van Deventer SJ, Hommes DW. Glucocorticoids cause rapid dissociation of a T-cell-receptor-associated protein complex containing LCK and FYN. EMBO Rep. 2006;7(10):1023–1029. doi: 10.1038/sj.embor.7400775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luisi BF, Xu WX, Otwinowski Z, Freedman LP, Yamamoto KR, Sigler PB. Crystallographic analysis of the interaction of the glucocorticoid receptor with DNA. Nature. 1991;352(6335):497–505. doi: 10.1038/352497a0. [DOI] [PubMed] [Google Scholar]
- Madrigal JL, Hurtado O, Moro MA, Lizasoain I, Lorenzo P, Castrillo A, Bosca L, Leza JC. The increase in TNF-alpha levels is implicated in NF-kappaB activation and inducible nitric oxide synthase expression in brain cortex after immobilization stress. Neuropsychopharmacology. 2002;26(2):155–163. doi: 10.1016/S0893-133X(01)00292-5. [DOI] [PubMed] [Google Scholar]
- Madrigal JLM, Moro MA, Lizasoain I, Lorenzo P, Castrillo A, Boscá L, Leza JC. Inducible nitric oxide synthase expression in brain cortex after acute restraint stress is regulated by nuclear factor κB-mediated mechanisms. Journal of Neurochemistry. 2001;76(2):532–538. doi: 10.1046/j.1471-4159.2001.00108.x. [DOI] [PubMed] [Google Scholar]
- Maiaru M, Tochiki KK, Cox MB, Annan LV, Bell CG, Feng X, Hausch F, Geranton SM. The stress regulator FKBP51 drives chronic pain by modulating spinal glucocorticoid signaling. Sci Transl Med. 2016;8(325):325ra319. doi: 10.1126/scitranslmed.aab3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao X, Phanavanh B, Hamdan H, Moerman-Herzog AM, Barger SW. NFkappaB-inducing kinase inhibits NFkappaB activity specifically in neurons of the CNS. J Neurochem. 2016;137(2):154–163. doi: 10.1111/jnc.13526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason BL, Pariante CM, Thomas SA. A revised role for P-glycoprotein in the brain distribution of dexamethasone, cortisol, and corticosterone in wild-type and ABCB1A/B-deficient mice. Endocrinology. 2008;149(10):5244–5253. doi: 10.1210/en.2008-0041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason BL, Pariante CM, Thomas SA. Changes in the brain accumulation of glucocorticoids in abcb1a-deficient CF-1 mice. J Neuroendocrinol. 2012;24(11):1440–1446. doi: 10.1111/j.1365-2826.2012.02353.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mata-Greenwood E, Stewart JM, Steinhorn RH, Pearce WJ. Role of BCL2-associated athanogene 1 in differential sensitivity of human endothelial cells to glucocorticoids. Arterioscler Thromb Vasc Biol. 2013;33(5):1046–1055. doi: 10.1161/ATVBAHA.113.301247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen BS. Glucocorticoid binding sites in rat brain: subcellular and anatomical localizations. Prog Brain Res. 1973;39:87–97. doi: 10.1016/S0079-6123(08)64068-6. [DOI] [PubMed] [Google Scholar]
- McKay LI, Cidlowski JA. CBP (CREB binding protein) integrates NF-kappaB (nuclear factor-kappaB) and glucocorticoid receptor physical interactions and antagonism. Mol Endocrinol. 2000;14(8):1222–1234. doi: 10.1210/mend.14.8.0506. [DOI] [PubMed] [Google Scholar]
- McKim DB, Patterson JM, Wohleb ES, Jarrett BL, Reader BF, Godbout JP, Sheridan JF. Sympathetic Release of Splenic Monocytes Promotes Recurring Anxiety Following Repeated Social Defeat. Biol Psychiatry. 2016;79(10):803–813. doi: 10.1016/j.biopsych.2015.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meffert MK, Chang JM, Wiltgen BJ, Fanselow MS, Baltimore D. NF-kappa B functions in synaptic signaling and behavior. Nat Neurosci. 2003;6(10):1072–1078. doi: 10.1038/nn1110. [DOI] [PubMed] [Google Scholar]
- Mikenberg I, Widera D, Kaus A, Kaltschmidt B, Kaltschmidt C. Transcription factor NF-kappaB is transported to the nucleus via cytoplasmic dynein/dynactin motor complex in hippocampal neurons. PLoS One. 2007;2(7):e589. doi: 10.1371/journal.pone.0000589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller AL, Webb MS, Copik AJ, Wang Y, Johnson BH, Kumar R, Thompson EB. p38 Mitogen-activated protein kinase (MAPK) is a key mediator in glucocorticoid-induced apoptosis of lymphoid cells: correlation between p38 MAPK activation and site-specific phosphorylation of the human glucocorticoid receptor at serine 211. Mol Endocrinol. 2005;19(6):1569–1583. doi: 10.1210/me.2004-0528. [DOI] [PubMed] [Google Scholar]
- Miller GE, Chen E, Sze J, Marin T, Arevalo JM, Doll R, Ma R, Cole SW. A functional genomic fingerprint of chronic stress in humans: blunted glucocorticoid and increased NF-kappaB signaling. Biol Psychiatry. 2008;64(4):266–272. doi: 10.1016/j.biopsych.2008.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller GE, Cohen S, Ritchey AK. Chronic psychological stress and the regulation of proinflammatory cytokines: a glucocorticoid-resistance model. Health Psychol. 2002;21(6):531–541. doi: 10.1037//0278-6133.21.6.531. [DOI] [PubMed] [Google Scholar]
- Miller GE, Murphy ML, Cashman R, Ma R, Ma J, Arevalo JM, Kobor MS, Cole SW. Greater inflammatory activity and blunted glucocorticoid signaling in monocytes of chronically stressed caregivers. Brain Behav Immun. 2014;41:191–199. doi: 10.1016/j.bbi.2014.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller GE, Rohleder N, Cole SW. Chronic interpersonal stress predicts activation of proand anti-inflammatory signaling pathways 6 months later. Psychosom Med. 2009;71(1):57–62. doi: 10.1097/PSY.0b013e318190d7de. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minami M, Kuraishi Y, Yamaguchi T, Nakai S, Hirai Y, Satoh M. Convulsants induce interleukin-1 beta messenger RNA in rat brain. Biochem Biophys Res Commun. 1990;171(2):832–837. doi: 10.1016/0006-291x(90)91221-d. [DOI] [PubMed] [Google Scholar]
- Mukaida N, Morita M, Ishikawa Y, Rice N, Okamoto S, Kasahara T, Matsushima K. Novel mechanism of glucocorticoid-mediated gene repression. Nuclear factor-kappa B is target for glucocorticoid-mediated interleukin 8 gene repression. J Biol Chem. 1994;269(18):13289–13295. [PubMed] [Google Scholar]
- Munhoz CD, Lepsch LB, Kawamoto EM, Malta MB, Lima Lde S, Avellar MC, Sapolsky RM, Scavone C. Chronic unpredictable stress exacerbates lipopolysaccharide-induced activation of nuclear factor-kappaB in the frontal cortex and hippocampus via glucocorticoid secretion. J Neurosci. 2006;26(14):3813–3820. doi: 10.1523/JNEUROSCI.4398-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson G, Wilde GJ, Spiller DG, Kennedy SM, Ray DW, Sullivan E, Unitt JF, White MR. NF-kappaB signalling is inhibited by glucocorticoid receptor and STAT6 via distinct mechanisms. J Cell Sci. 2003;116(Pt 12):2495–2503. doi: 10.1242/jcs.00461. [DOI] [PubMed] [Google Scholar]
- Newton R, Holden NS. Separating transrepression and transactivation: a distressing divorce for the glucocorticoid receptor? Mol Pharmacol. 2007;72(4):799–809. doi: 10.1124/mol.107.038794. [DOI] [PubMed] [Google Scholar]
- Oakley RH, Jewell CM, Yudt MR, Bofetiado DM, Cidlowski JA. The dominant negative activity of the human glucocorticoid receptor beta isoform. Specificity and mechanisms of action. J Biol Chem. 1999;274(39):27857–27866. doi: 10.1074/jbc.274.39.27857. [DOI] [PubMed] [Google Scholar]
- Oakley RH, Sar M, Cidlowski JA. The human glucocorticoid receptor beta isoform. Expression, biochemical properties, and putative function. J Biol Chem. 1996;271(16):9550–9559. doi: 10.1074/jbc.271.16.9550. [DOI] [PubMed] [Google Scholar]
- Oeckinghaus A, Hayden MS, Ghosh S. Crosstalk in NF-kappaB signaling pathways. Nat Immunol. 2011;12(8):695–708. doi: 10.1038/ni.2065. [DOI] [PubMed] [Google Scholar]
- Orti E, Mendel DB, Smith LI, Munck A. Agonist-dependent phosphorylation and nuclear dephosphorylation of glucocorticoid receptors in intact cells. J Biol Chem. 1989;264(17):9728–9731. [PubMed] [Google Scholar]
- Pace TW, Hu F, Miller AH. Cytokine-effects on glucocorticoid receptor function: relevance to glucocorticoid resistance and the pathophysiology and treatment of major depression. Brain Behav Immun. 2007;21(1):9–19. doi: 10.1016/j.bbi.2006.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace TW, Hu F, Miller AH. Activation of cAMP-protein kinase A abrogates STAT5-mediated inhibition of glucocorticoid receptor signaling by interferon-alpha. Brain Behav Immun. 2011;25(8):1716–1724. doi: 10.1016/j.bbi.2011.07.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pace TW, Wingenfeld K, Schmidt I, Meinlschmidt G, Hellhammer DH, Heim CM. Increased peripheral NF-kappaB pathway activity in women with childhood abuse-related posttraumatic stress disorder. Brain Behav Immun. 2012;26(1):13–17. doi: 10.1016/j.bbi.2011.07.232. [DOI] [PubMed] [Google Scholar]
- Padgett DA, Glaser R. How stress influences the immune response. Trends in Immunology. 2003;24(8):444–448. doi: 10.1016/s1471-4906(03)00173-x. [DOI] [PubMed] [Google Scholar]
- Panagiotakopoulos L, Neigh GN. Development of the HPA axis: where and when do sex differences manifest? Front Neuroendocrinol. 2014;35(3):285–302. doi: 10.1016/j.yfrne.2014.03.002. [DOI] [PubMed] [Google Scholar]
- Pariante CM, Kim RB, Makoff A, Kerwin RW. Antidepressant fluoxetine enhances glucocorticoid receptor function in vitro by modulating membrane steroid transporters. Br J Pharmacol. 2003;139(6):1111–1118. doi: 10.1038/sj.bjp.0705357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pazdrak K, Straub C, Maroto R, Stafford S, White WI, Calhoun WJ, Kurosky A. Cytokine-Induced Glucocorticoid Resistance from Eosinophil Activation: Protein Phosphatase 5 Modulation of Glucocorticoid Receptor Phosphorylation and Signaling. J Immunol. 2016;197(10):3782–3791. doi: 10.4049/jimmunol.1601029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perez MH, Cormack J, Mallinson D, Mutungi G. A membrane glucocorticoid receptor mediates the rapid/non-genomic actions of glucocorticoids in mammalian skeletal muscle fibres. J Physiol. 2013;591(20):5171–5185. doi: 10.1113/jphysiol.2013.256586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins ND. Post-translational modifications regulating the activity and function of the nuclear factor kappa B pathway. Oncogene. 2006;25(51):6717–6730. doi: 10.1038/sj.onc.1209937. [DOI] [PubMed] [Google Scholar]
- Plaschke K, Feindt J, Djuric Z, Heiland S, Autschbach F, Lewicka S, Martin E, Bardenheuer HJ, Nawroth PP, Bierhaus A. Chronic corticosterone-induced deterioration in rat behaviour is not paralleled by changes in hippocampal NF-kappaB-activation. Stress. 2006;9(2):97–106. doi: 10.1080/10253890600691551. [DOI] [PubMed] [Google Scholar]
- Pujols L, Mullol J, Roca-Ferrer J, Torrego A, Xaubet A, Cidlowski JA, Picado C. Expression of glucocorticoid receptor alpha- and beta-isoforms in human cells and tissues. Am J Physiol Cell Physiol. 2002;283(4):C1324–1331. doi: 10.1152/ajpcell.00363.2001. [DOI] [PubMed] [Google Scholar]
- Pyter LM, Kelly SD, Harrell CS, Neigh GN. Sex differences in the effects of adolescent stress on adult brain inflammatory markers in rats. Brain Behav Immun. 2013;30:88–94. doi: 10.1016/j.bbi.2013.01.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qi AQ, Qiu J, Xiao L, Chen YZ. Rapid activation of JNK and p38 by glucocorticoids in primary cultured hippocampal cells. J Neurosci Res. 2005;80(4):510–517. doi: 10.1002/jnr.20491. [DOI] [PubMed] [Google Scholar]
- Qiu J, Wang P, Jing Q, Zhang W, Li X, Zhong Y, Sun G, Pei G, Chen Y. Rapid activation of ERK1/2 mitogen-activated protein kinase by corticosterone in PC12 cells. Biochem Biophys Res Commun. 2001;287(4):1017–1024. doi: 10.1006/bbrc.2001.5691. [DOI] [PubMed] [Google Scholar]
- Quan N. Molecular mechanisms of glucocorticoid resistance in splenocytes of socially stressed male mice. Journal of Neuroimmunology. 2003;137(1–2):51–58. doi: 10.1016/s0165-5728(03)00042-0. [DOI] [PubMed] [Google Scholar]
- Rao NA, McCalman MT, Moulos P, Francoijs KJ, Chatziioannou A, Kolisis FN, Alexis MN, Mitsiou DJ, Stunnenberg HG. Coactivation of GR and NFKB alters the repertoire of their binding sites and target genes. Genome Res. 2011;21(9):1404–1416. doi: 10.1101/gr.118042.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray A, Prefontaine KE. Physical association and functional antagonism between the p65 subunit of transcription factor NF-kappa B and the glucocorticoid receptor. Proc Natl Acad Sci U S A. 1994;91(2):752–756. doi: 10.1073/pnas.91.2.752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rein T. FK506 binding protein 51 integrates pathways of adaptation: FKBP51 shapes the reactivity to environmental change. Bioessays. 2016 doi: 10.1002/bies.201600050. [DOI] [PubMed] [Google Scholar]
- Reul JM, de Kloet ER. Two receptor systems for corticosterone in rat brain: microdistribution and differential occupation. Endocrinology. 1985;117(6):2505–2511. doi: 10.1210/endo-117-6-2505. [DOI] [PubMed] [Google Scholar]
- Rivest S. How circulating cytokines trigger the neural circuits that control the hypothalamic-pituitary-adrenal axis. Psychoneuroendocrinology. 2001;26(8):761–788. doi: 10.1016/s0306-4530(01)00064-6. [DOI] [PubMed] [Google Scholar]
- Rogatsky I, Logan SK, Garabedian MJ. Antagonism of glucocorticoid receptor transcriptional activation by the c-Jun N-terminal kinase. Proc Natl Acad Sci U S A. 1998;95(5):2050–2055. doi: 10.1073/pnas.95.5.2050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romano S, Mallardo M, Romano MF. FKBP51 and the NF-kappaB regulatory pathway in cancer. Curr Opin Pharmacol. 2011;11(4):288–293. doi: 10.1016/j.coph.2011.04.011. [DOI] [PubMed] [Google Scholar]
- Romano S, Xiao Y, Nakaya M, D'Angelillo A, Chang M, Jin J, Hausch F, Masullo M, Feng X, Romano MF, Sun S-C. FKBP51 employs both scaffold and isomerase functions to promote NF-κB activation in melanoma. Nucleic Acids Research. 2015;43(14):6983–6993. doi: 10.1093/nar/gkv615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ros-Bernal F, Hunot S, Herrero MT, Parnadeau S, Corvol JC, Lu L, Alvarez-Fischer D, Carrillo-de Sauvage MA, Saurini F, Coussieu C, Kinugawa K, Prigent A, Hoglinger G, Hamon M, Tronche F, Hirsch EC, Vyas S. Microglial glucocorticoid receptors play a pivotal role in regulating dopaminergic neurodegeneration in parkinsonism. Proc Natl Acad Sci U S A. 2011;108(16):6632–6637. doi: 10.1073/pnas.1017820108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rousseau GG, Higgins SJ, Baxter JD, Gelfand D, Tomkins GM. Binding of glucocorticoid receptors to DNA. J Biol Chem. 1975;250(15):6015–6021. [PubMed] [Google Scholar]
- Roy B, Rai U. Genomic and non-genomic effect of cortisol on phagocytosis in freshwater teleost Channa punctatus: an in vitro study. Steroids. 2009;74(4–5):449–455. doi: 10.1016/j.steroids.2008.12.013. [DOI] [PubMed] [Google Scholar]
- Russo SJ, Wilkinson MB, Mazei-Robison MS, Dietz DM, Maze I, Krishnan V, Renthal W, Graham A, Birnbaum SG, Green TA, Robison B, Lesselyong A, Perrotti LI, Bolanos CA, Kumar A, Clark MS, Neumaier JF, Neve RL, Bhakar AL, Barker PA, Nestler EJ. Nuclear factor kappa B signaling regulates neuronal morphology and cocaine reward. J Neurosci. 2009;29(11):3529–3537. doi: 10.1523/JNEUROSCI.6173-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salama-Cohen P, Arevalo MA, Meier J, Grantyn R, Rodriguez-Tebar A. NGF controls dendrite development in hippocampal neurons by binding to p75NTR and modulating the cellular targets of Notch. Mol Biol Cell. 2005;16(1):339–347. doi: 10.1091/mbc.E04-05-0438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheinman RI, Cogswell PC, Lofquist AK, Baldwin AS., Jr Role of transcriptional activation of I kappa B alpha in mediation of immunosuppression by glucocorticoids. Science. 1995;270(5234):283–286. doi: 10.1126/science.270.5234.283. [DOI] [PubMed] [Google Scholar]
- Scheinman RI, Gualberto A, Jewell CM, Cidlowski JA, Baldwin AS., Jr Characterization of mechanisms involved in transrepression of NF-kappa B by activated glucocorticoid receptors. Mol Cell Biol. 1995;15(2):943–953. doi: 10.1128/mcb.15.2.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt-Ullrich R, Memet S, Lilienbaum A, Feuillard J, Raphael M, Israel A. NF-kappaB activity in transgenic mice: developmental regulation and tissue specificity. Development. 1996;122(7):2117–2128. doi: 10.1242/dev.122.7.2117. [DOI] [PubMed] [Google Scholar]
- Sen R, Smale ST. Selectivity of the NF-{kappa}B response. Cold Spring Harb Perspect Biol. 2010;2(4):a000257. doi: 10.1101/cshperspect.a000257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaqura M, Li X, Al-Khrasani M, Shakibaei M, Tafelski S, Furst S, Beyer A, Kawata M, Schafer M, Mousa SA. Membrane-bound glucocorticoid receptors on distinct nociceptive neurons as potential targets for pain control through rapid non-genomic effects. Neuropharmacology. 2016;111:1–13. doi: 10.1016/j.neuropharm.2016.08.019. [DOI] [PubMed] [Google Scholar]
- Shelton RC, Sanders-Bush E, Manier DH, Lewis DA. Elevated 5-HT 2A receptors in postmortem prefrontal cortex in major depression is associated with reduced activity of protein kinase A. Neuroscience. 2009;158(4):1406–1415. doi: 10.1016/j.neuroscience.2008.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheppard KA, Phelps KM, Williams AJ, Thanos D, Glass CK, Rosenfeld MG, Gerritsen ME, Collins T. Nuclear integration of glucocorticoid receptor and nuclear factor-kappaB signaling by CREB-binding protein and steroid receptor coactivator-1. J Biol Chem. 1998;273(45):29291–29294. doi: 10.1074/jbc.273.45.29291. [DOI] [PubMed] [Google Scholar]
- Silva CM, Powell-Oliver FE, Jewell CM, Sar M, Allgood VE, Cidlowski JA. Regulation of the human glucocorticoid receptor by long-term and chronic treatment with glucocorticoid. Steroids. 1994;59(7):436–442. doi: 10.1016/0039-128x(94)90013-2. [DOI] [PubMed] [Google Scholar]
- Silverstein AM, Galigniana MD, Kanelakis KC, Radanyi C, Renoir JM, Pratt WB. Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein. J Biol Chem. 1999;274(52):36980–36986. doi: 10.1074/jbc.274.52.36980. [DOI] [PubMed] [Google Scholar]
- Simic I, Adzic M, Maric N, Savic D, Djordjevic J, Mihaljevic M, Mitic M, Pavlovic Z, Soldatovic I, Krstic-Demonacos M, Jasovic-Gasic M, Radojcic M. A preliminary evaluation of leukocyte phospho-glucocorticoid receptor as a potential biomarker of depressogenic vulnerability in healthy adults. Psychiatry Res. 2013;209(3):658–664. doi: 10.1016/j.psychres.2013.02.002. [DOI] [PubMed] [Google Scholar]
- Simic I, Maric NP, Mitic M, Soldatovic I, Pavlovic Z, Mihaljevic M, Andric S, Radojcic MB, Adzic M. Phosphorylation of leukocyte glucocorticoid receptor in patients with current episode of major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2013;40:281–285. doi: 10.1016/j.pnpbp.2012.10.021. [DOI] [PubMed] [Google Scholar]
- Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol Bull. 2014;140(3):774–815. doi: 10.1037/a0035302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solito E, Mulla A, Morris JF, Christian HC, Flower RJ, Buckingham JC. Dexamethasone induces rapid serine-phosphorylation and membrane translocation of annexin 1 in a human folliculostellate cell line via a novel nongenomic mechanism involving the glucocorticoid receptor, protein kinase C, phosphatidylinositol 3-kinase, and mitogen-activated protein kinase. Endocrinology. 2003;144(4):1164–1174. doi: 10.1210/en.2002-220592. [DOI] [PubMed] [Google Scholar]
- Sorrells SF, Caso JR, Munhoz CD, Hu CK, Tran KV, Miguel ZD, Chien BY, Sapolsky RM. Glucocorticoid signaling in myeloid cells worsens acute CNS injury and inflammation. J Neurosci. 2013;33(18):7877–7889. doi: 10.1523/JNEUROSCI.4705-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorrells SF, Caso JR, Munhoz CD, Sapolsky RM. The stressed CNS: when glucocorticoids aggravate inflammation. Neuron. 2009;64(1):33–39. doi: 10.1016/j.neuron.2009.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorrells SF, Munhoz CD, Manley NC, Yen S, Sapolsky RM. Glucocorticoids increase excitotoxic injury and inflammation in the hippocampus of adult male rats. Neuroendocrinology. 2014;100(2–3):129–140. doi: 10.1159/000367849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa AR, Lane SJ, Cidlowski JA, Staynov DZ, Lee TH. Glucocorticoid resistance in asthma is associated with elevated in vivo expression of the glucocorticoid receptor beta-isoform. J Allergy Clin Immunol. 2000;105(5):943–950. doi: 10.1067/mai.2000.106486. [DOI] [PubMed] [Google Scholar]
- Strahle U, Klock G, Schutz G. A DNA sequence of 15 base pairs is sufficient to mediate both glucocorticoid and progesterone induction of gene expression. Proc Natl Acad Sci U S A. 1987;84(22):7871–7875. doi: 10.1073/pnas.84.22.7871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surjit M, Ganti KP, Mukherji A, Ye T, Hua G, Metzger D, Li M, Chambon P. Widespread negative response elements mediate direct repression by agonist-liganded glucocorticoid receptor. Cell. 2011;145(2):224–241. doi: 10.1016/j.cell.2011.03.027. [DOI] [PubMed] [Google Scholar]
- Suzuki T, Mitake S, Okumura-Noji K, Yang JP, Fujii T, Okamoto T. Presence of NF-kappaB-like and IkappaB-like immunoreactivities in postsynaptic densities. Neuroreport. 1997;8(13):2931–2935. doi: 10.1097/00001756-199709080-00025. [DOI] [PubMed] [Google Scholar]
- Szatmary Z, Garabedian MJ, Vilcek J. Inhibition of glucocorticoid receptor-mediated transcriptional activation by p38 mitogen-activated protein (MAP) kinase. J Biol Chem. 2004;279(42):43708–43715. doi: 10.1074/jbc.M406568200. [DOI] [PubMed] [Google Scholar]
- Tai DJ, Su CC, Ma YL, Lee EH. SGK1 phosphorylation of IkappaB Kinase alpha and p300 Up-regulates NF-kappaB activity and increases N-Methyl-D-aspartate receptor NR2A and NR2B expression. J Biol Chem. 2009;284(7):4073–4089. doi: 10.1074/jbc.M805055200. [DOI] [PubMed] [Google Scholar]
- Tatro ET, Everall IP, Kaul M, Achim CL. Modulation of glucocorticoid receptor nuclear translocation in neurons by immunophilins FKBP51 and FKBP52: implications for major depressive disorder. Brain Res. 2009;1286:1–12. doi: 10.1016/j.brainres.2009.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatro ET, Everall IP, Masliah E, Hult BJ, Lucero G, Chana G, Soontornniyomkij V, Achim CL, H. I. V. N. R. Center Differential expression of immunophilins FKBP51 and FKBP52 in the frontal cortex of HIV-infected patients with major depressive disorder. J Neuroimmune Pharmacol. 2009;4(2):218–226. doi: 10.1007/s11481-009-9146-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian B, Brasier AR. Identification of a nuclear factor kappa B-dependent gene network. Recent Prog Horm Res. 2003;58:95–130. doi: 10.1210/rp.58.1.95. [DOI] [PubMed] [Google Scholar]
- Tryc AB, Spies CM, Schneider U, Kunkel D, Berki T, Sieper J, Burmester GR, Radbruch A, Scheffold A, Buttgereit F. Membrane glucocorticoid receptor expression on peripheral blood mononuclear cells in patients with ankylosing spondylitis. J Rheumatol. 2006;33(11):2249–2253. [PubMed] [Google Scholar]
- Unlap MT, Jope RS. Dexamethasone attenuates NF-kappa B DNA binding activity without inducing I kappa B levels in rat brain in vivo. Brain Res Mol Brain Res. 1997;45(1):83–89. doi: 10.1016/s0169-328x(96)00240-9. [DOI] [PubMed] [Google Scholar]
- Unlap T, Jope RS. Inhibition of NFkB DNA binding activity by glucocorticoids in rat brain. Neurosci Lett. 1995;198(1):41–44. doi: 10.1016/0304-3940(95)11963-w. [DOI] [PubMed] [Google Scholar]
- Van Bogaert T, De Bosscher K, Libert C. Crosstalk between TNF and glucocorticoid receptor signaling pathways. Cytokine Growth Factor Rev. 2010;21(4):275–286. doi: 10.1016/j.cytogfr.2010.04.003. [DOI] [PubMed] [Google Scholar]
- Verriere VA, Hynes D, Faherty S, Devaney J, Bousquet J, Harvey BJ, Urbach V. Rapid effects of dexamethasone on intracellular pH and Na+/H+ exchanger activity in human bronchial epithelial cells. J Biol Chem. 2005;280(43):35807–35814. doi: 10.1074/jbc.M506584200. [DOI] [PubMed] [Google Scholar]
- Wallace AD, Cidlowski JA. Proteasome-mediated glucocorticoid receptor degradation restricts transcriptional signaling by glucocorticoids. J Biol Chem. 2001;276(46):42714–42721. doi: 10.1074/jbc.M106033200. [DOI] [PubMed] [Google Scholar]
- Waltner-Law M, Daniels MC, Sutherland C, Granner DK. NF-kappa B inhibits glucocorticoid and cAMP-mediated expression of the phosphoenolpyruvate carboxykinase gene. J Biol Chem. 2000;275(41):31847–31856. doi: 10.1074/jbc.M003656200. [DOI] [PubMed] [Google Scholar]
- Wang X, Wu H, Lakdawala VS, Hu F, Hanson ND, Miller AH. Inhibition of Jun N-terminal kinase (JNK) enhances glucocorticoid receptor-mediated function in mouse hippocampal HT22 cells. Neuropsychopharmacology. 2005;30(2):242–249. doi: 10.1038/sj.npp.1300606. [DOI] [PubMed] [Google Scholar]
- Wang Z, Frederick J, Garabedian MJ. Deciphering the phosphorylation "code" of the glucocorticoid receptor in vivo. J Biol Chem. 2002;277(29):26573–26580. doi: 10.1074/jbc.M110530200. [DOI] [PubMed] [Google Scholar]
- Weber MD, Frank MG, Sobesky JL, Watkins LR, Maier SF. Blocking toll-like receptor 2 and 4 signaling during a stressor prevents stress-induced priming of neuroinflammatory responses to a subsequent immune challenge. Brain Behav Immun. 2013;32:112–121. doi: 10.1016/j.bbi.2013.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber MD, Frank MG, Tracey KJ, Watkins LR, Maier SF. Stress induces the danger-associated molecular pattern HMGB-1 in the hippocampus of male Sprague Dawley rats: a priming stimulus of microglia and the NLRP3 inflammasome. J Neurosci. 2015;35(1):316–324. doi: 10.1523/JNEUROSCI.3561-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster JC, Oakley RH, Jewell CM, Cidlowski JA. Proinflammatory cytokines regulate human glucocorticoid receptor gene expression and lead to the accumulation of the dominant negative beta isoform: a mechanism for the generation of glucocorticoid resistance. Proc Natl Acad Sci U S A. 2001;98(12):6865–6870. doi: 10.1073/pnas.121455098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitesell L, Cook P. Stable and specific binding of heat shock protein 90 by geldanamycin disrupts glucocorticoid receptor function in intact cells. Mol Endocrinol. 1996;10(6):705–712. doi: 10.1210/mend.10.6.8776730. [DOI] [PubMed] [Google Scholar]
- Widen C, Gustafsson JA, Wikstrom AC. Cytosolic glucocorticoid receptor interaction with nuclear factor-kappa B proteins in rat liver cells. Biochem J. 2003;373(Pt 1):211–220. doi: 10.1042/BJ20030175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Widera D, Klenke C, Nair D, Heidbreder M, Malkusch S, Sibarita JB, Choquet D, Kaltschmidt B, Heilemann M, Kaltschmidt C. Single-particle tracking uncovers dynamics of glutamate-induced retrograde transport of NF-kappaB p65 in living neurons. Neurophotonics. 2016;3(4):041804. doi: 10.1117/1.NPh.3.4.041804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wochnik GM, Ruegg J, Abel GA, Schmidt U, Holsboer F, Rein T. FK506-binding proteins 51 and 52 differentially regulate dynein interaction and nuclear translocation of the glucocorticoid receptor in mammalian cells. J Biol Chem. 2005;280(6):4609–4616. doi: 10.1074/jbc.M407498200. [DOI] [PubMed] [Google Scholar]
- Wohleb ES, Powell ND, Godbout JP, Sheridan JF. Stress-induced recruitment of bone marrow-derived monocytes to the brain promotes anxiety-like behavior. J Neurosci. 2013;33(34):13820–13833. doi: 10.1523/JNEUROSCI.1671-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf JM, Rohleder N, Bierhaus A, Nawroth PP, Kirschbaum C. Determinants of the NF-kappaB response to acute psychosocial stress in humans. Brain Behav Immun. 2009;23(6):742–749. doi: 10.1016/j.bbi.2008.09.009. [DOI] [PubMed] [Google Scholar]
- Yeh SH, Lin CH, Gean PW. Acetylation of nuclear factor-kappaB in rat amygdala improves long-term but not short-term retention of fear memory. Mol Pharmacol. 2004;65(5):1286–1292. doi: 10.1124/mol.65.5.1286. [DOI] [PubMed] [Google Scholar]
- Yeh SH, Lin CH, Lee CF, Gean PW. A requirement of nuclear factor-kappaB activation in fear-potentiated startle. J Biol Chem. 2002;277(48):46720–46729. doi: 10.1074/jbc.M206258200. [DOI] [PubMed] [Google Scholar]
- Zhang K, Pan X, Wang F, Ma J, Su G, Dong Y, Yang J, Wu C. Baicalin promotes hippocampal neurogenesis via SGK1- and FKBP5-mediated glucocorticoid receptor phosphorylation in a neuroendocrine mouse model of anxiety/depression. Sci Rep. 2016;6:30951. doi: 10.1038/srep30951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Clark AF, Yorio T. Heat shock protein 90 is an essential molecular chaperone for nuclear transport of glucocorticoid receptor beta. Invest Ophthalmol Vis Sci. 2006;47(2):700–708. doi: 10.1167/iovs.05-0697. [DOI] [PubMed] [Google Scholar]
- Zhang X, Clark AF, Yorio T. FK506-binding protein 51 regulates nuclear transport of the glucocorticoid receptor beta and glucocorticoid responsiveness. Invest Ophthalmol Vis Sci. 2008;49(3):1037–1047. doi: 10.1167/iovs.07-1279. [DOI] [PMC free article] [PubMed] [Google Scholar]