Abstract
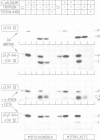
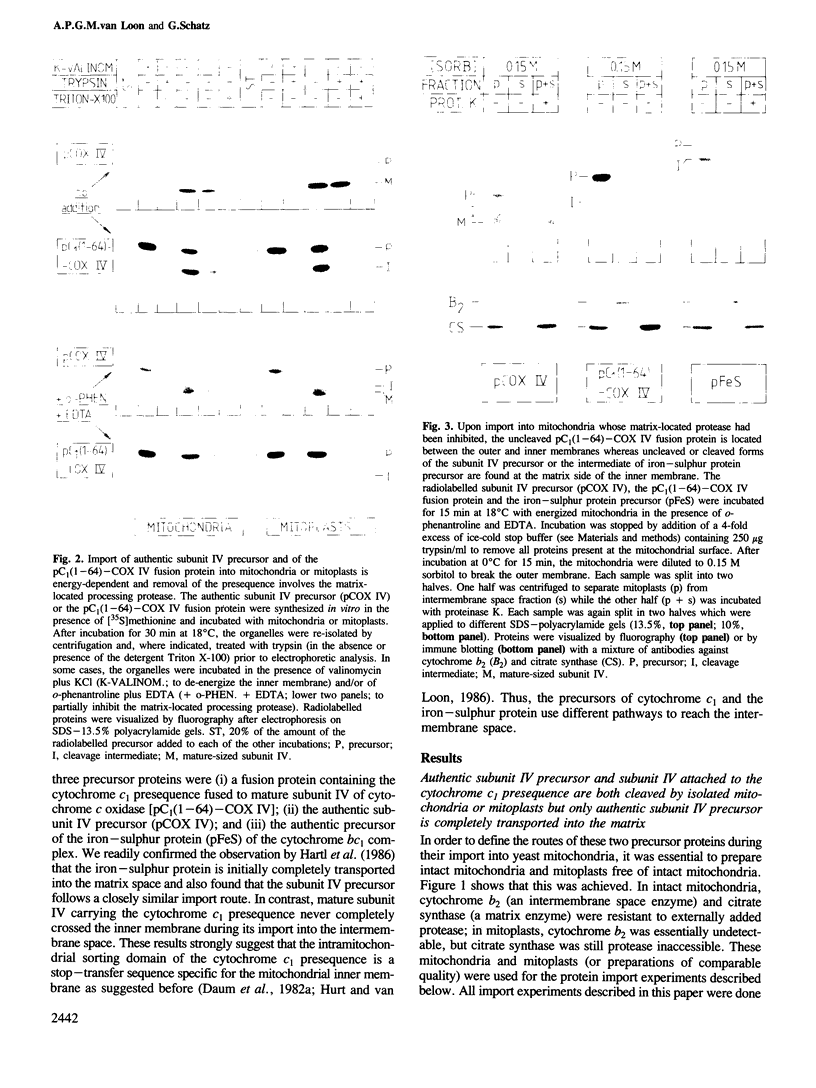
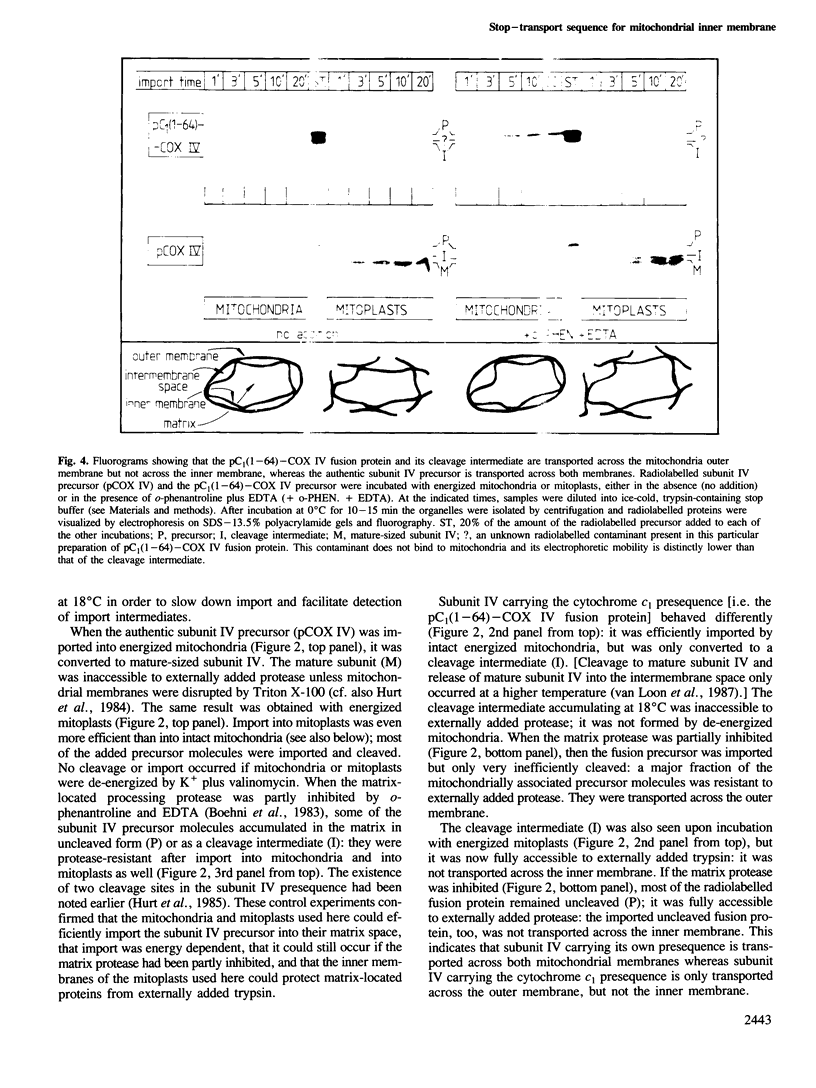
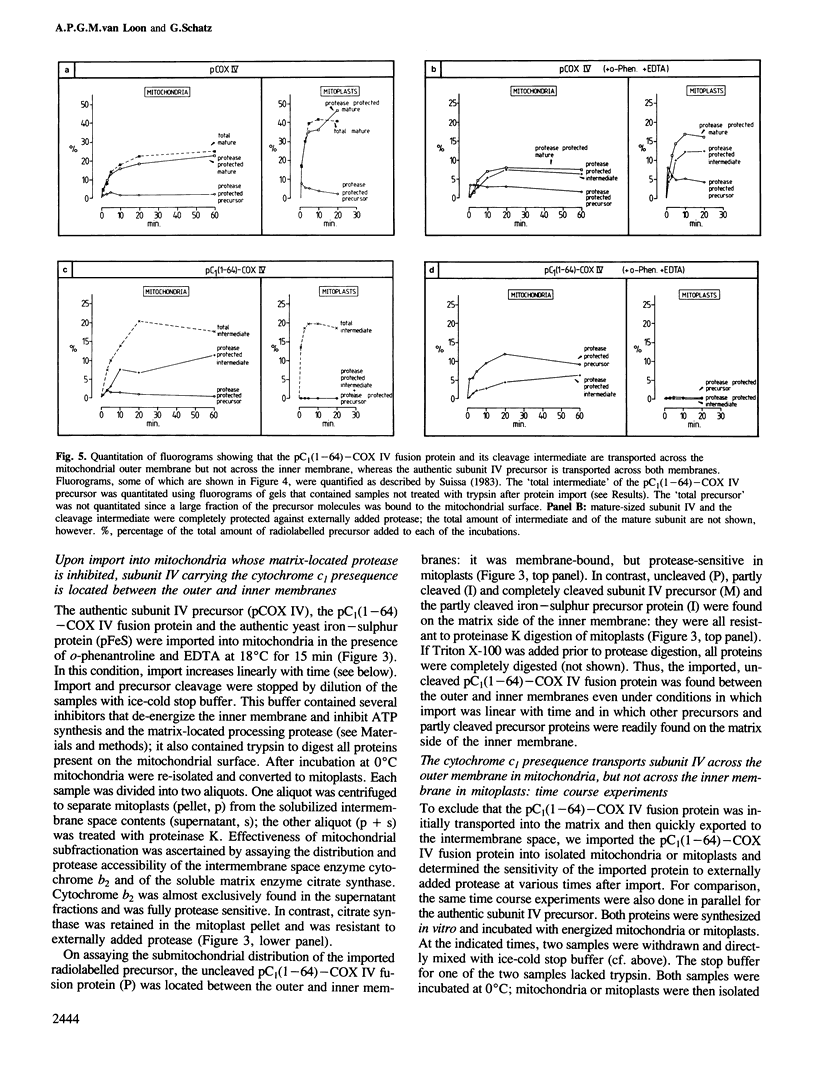
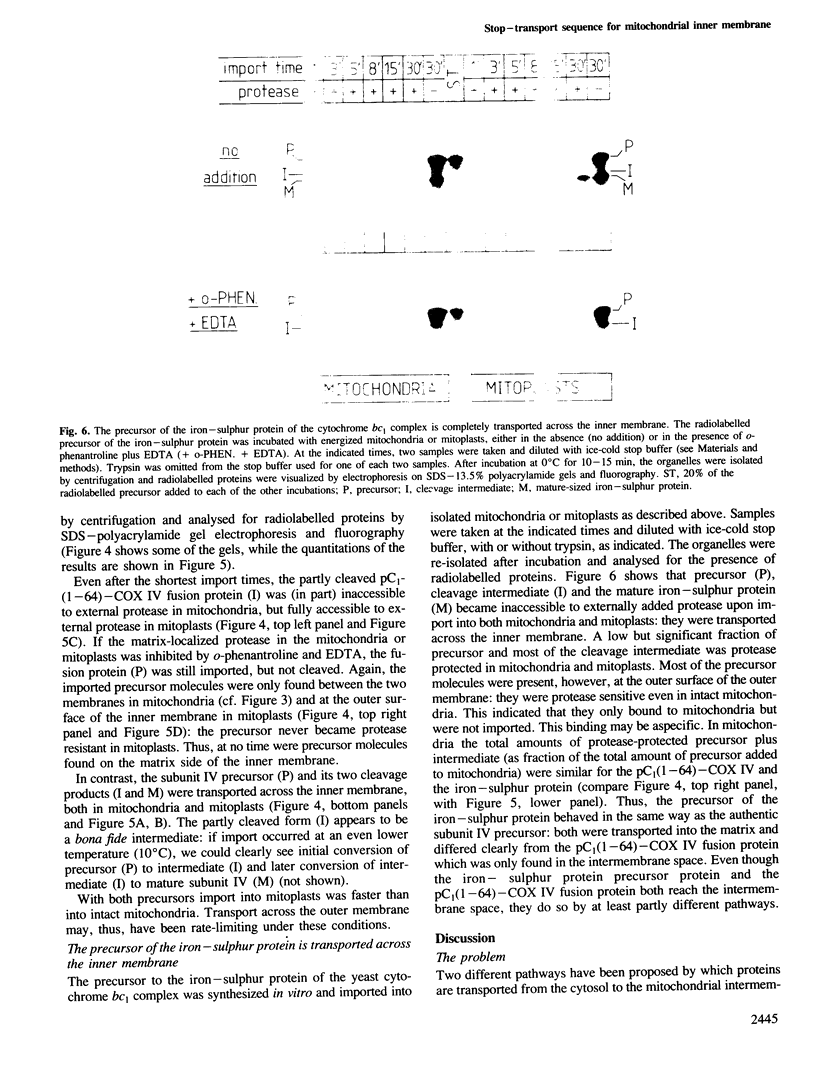
The presequence of yeast cytochrome c1 (an inner membrane protein protruding into the intermembrane space) contains a matrix-targeting domain and an intramitochondrial sorting domain. This presequence transports attached subunit IV of cytochrome c oxidase into the intermembrane space (van Loon et al. (1987) EMBO J., 6, 2433-2439). In order to determine how this fusion protein reaches the intermembrane space, we studied the kinetics of its import into isolated mitochondria or mitoplasts and its accumulation in the various submitochondrial compartments. The imported, uncleaved fusion precursor and a cleavage intermediate were bound to the inner membrane and were always exposed to the intermembrane space; they were never found at the matrix side of the inner membrane. In contrast, analogous import experiments with the authentic subunit IV precursor, or the precursor of the iron-sulphur protein of the cytochrome bc1 complex also an inner membrane protein exposed to the intermembrane space), readily showed that these precursors were initially transported across both mitochondrial membranes. We conclude that the intramitochondrial sorting domain within the cytochrome c1 presequence prevents transport of attached proteins across the inner, but not the outer membrane: it is a stop-transfer sequence for the inner membrane. Since the presequence of the iron-sulphur protein lacks such 'stop-transfer' domain, it acts by a different mechanism.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bell R. L., Sweetland J., Ludwig B., Capaldi R. A. Labeling of complex III, with [35S]diazobenzenesulfonate: orientation of this electron transfer segment in the mitochondrial inner membrane. Proc Natl Acad Sci U S A. 1979 Feb;76(2):741–745. doi: 10.1073/pnas.76.2.741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Böhni P. C., Daum G., Schatz G. Import of proteins into mitochondria. Partial purification of a matrix-located protease involved in cleavage of mitochondrial precursor polypeptides. J Biol Chem. 1983 Apr 25;258(8):4937–4943. [PubMed] [Google Scholar]
- Daum G., Böhni P. C., Schatz G. Import of proteins into mitochondria. Cytochrome b2 and cytochrome c peroxidase are located in the intermembrane space of yeast mitochondria. J Biol Chem. 1982 Nov 10;257(21):13028–13033. [PubMed] [Google Scholar]
- Daum G., Gasser S. M., Schatz G. Import of proteins into mitochondria. Energy-dependent, two-step processing of the intermembrane space enzyme cytochrome b2 by isolated yeast mitochondria. J Biol Chem. 1982 Nov 10;257(21):13075–13080. [PubMed] [Google Scholar]
- Fujiki Y., Hubbard A. L., Fowler S., Lazarow P. B. Isolation of intracellular membranes by means of sodium carbonate treatment: application to endoplasmic reticulum. J Cell Biol. 1982 Apr;93(1):97–102. doi: 10.1083/jcb.93.1.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasser S. M., Daum G., Schatz G. Import of proteins into mitochondria. Energy-dependent uptake of precursors by isolated mitochondria. J Biol Chem. 1982 Nov 10;257(21):13034–13041. [PubMed] [Google Scholar]
- Haid A., Suissa M. Immunochemical identification of membrane proteins after sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Methods Enzymol. 1983;96:192–205. doi: 10.1016/s0076-6879(83)96017-2. [DOI] [PubMed] [Google Scholar]
- Hartl F. U., Schmidt B., Wachter E., Weiss H., Neupert W. Transport into mitochondria and intramitochondrial sorting of the Fe/S protein of ubiquinol-cytochrome c reductase. Cell. 1986 Dec 26;47(6):939–951. doi: 10.1016/0092-8674(86)90809-3. [DOI] [PubMed] [Google Scholar]
- Hurt E. C., Pesold-Hurt B., Schatz G. The amino-terminal region of an imported mitochondrial precursor polypeptide can direct cytoplasmic dihydrofolate reductase into the mitochondrial matrix. EMBO J. 1984 Dec 20;3(13):3149–3156. doi: 10.1002/j.1460-2075.1984.tb02272.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurt E. C., Pesold-Hurt B., Suda K., Oppliger W., Schatz G. The first twelve amino acids (less than half of the pre-sequence) of an imported mitochondrial protein can direct mouse cytosolic dihydrofolate reductase into the yeast mitochondrial matrix. EMBO J. 1985 Aug;4(8):2061–2068. doi: 10.1002/j.1460-2075.1985.tb03892.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaput J., Goltz S., Blobel G. Nucleotide sequence of the yeast nuclear gene for cytochrome c peroxidase precursor. Functional implications of the pre sequence for protein transport into mitochondria. J Biol Chem. 1982 Dec 25;257(24):15054–15058. [PubMed] [Google Scholar]
- Karlsson B., Hovmöller S., Weiss H., Leonard K. Structural studies of cytochrome reductase. Subunit topography determined by electron microscopy of membrane crystals of a subcomplex. J Mol Biol. 1983 Apr 5;165(2):287–302. doi: 10.1016/s0022-2836(83)80258-7. [DOI] [PubMed] [Google Scholar]
- Li Y., De Vries S., Leonard K., Weiss H. Topography of the iron-sulphur subunit in mitochondrial ubiquinol:cytochrome c reductase. FEBS Lett. 1981 Dec 7;135(2):277–280. doi: 10.1016/0014-5793(81)80800-9. [DOI] [PubMed] [Google Scholar]
- Li Y., Leonard K., Weiss H. Membrane-bound and water-soluble cytochrome c1 from Neurospora mitochondria. Eur J Biochem. 1981 May;116(1):199–205. doi: 10.1111/j.1432-1033.1981.tb05319.x. [DOI] [PubMed] [Google Scholar]
- Maarse A. C., Van Loon A. P., Riezman H., Gregor I., Schatz G., Grivell L. A. Subunit IV of yeast cytochrome c oxidase: cloning and nucleotide sequencing of the gene and partial amino acid sequencing of the mature protein. EMBO J. 1984 Dec 1;3(12):2831–2837. doi: 10.1002/j.1460-2075.1984.tb02216.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohashi A., Gibson J., Gregor I., Schatz G. Import of proteins into mitochondria. The precursor of cytochrome c1 is processed in two steps, one of them heme-dependent. J Biol Chem. 1982 Nov 10;257(21):13042–13047. [PubMed] [Google Scholar]
- Riezman H., Hay R., Witte C., Nelson N., Schatz G. Yeast mitochondrial outer membrane specifically binds cytoplasmically-synthesized precursors of mitochondrial proteins. EMBO J. 1983;2(7):1113–1118. doi: 10.1002/j.1460-2075.1983.tb01554.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadler I., Suda K., Schatz G., Kaudewitz F., Haid A. Sequencing of the nuclear gene for the yeast cytochrome c1 precursor reveals an unusually complex amino-terminal presequence. EMBO J. 1984 Sep;3(9):2137–2143. doi: 10.1002/j.1460-2075.1984.tb02103.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stueber D., Ibrahimi I., Cutler D., Dobberstein B., Bujard H. A novel in vitro transcription-translation system: accurate and efficient synthesis of single proteins from cloned DNA sequences. EMBO J. 1984 Dec 20;3(13):3143–3148. doi: 10.1002/j.1460-2075.1984.tb02271.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suissa M. Spectrophotometric quantitation of silver grains eluted from autoradiograms. Anal Biochem. 1983 Sep;133(2):511–514. doi: 10.1016/0003-2697(83)90117-3. [DOI] [PubMed] [Google Scholar]
- Wakabayashi S., Matsubara H., Kim C. H., Kawai K., King T. E. The complete amino acid sequence of bovine heart cytochrome C1. Biochem Biophys Res Commun. 1980 Dec 31;97(4):1548–1554. doi: 10.1016/s0006-291x(80)80042-8. [DOI] [PubMed] [Google Scholar]
- van Loon A. P., Brändli A. W., Pesold-Hurt B., Blank D., Schatz G. Transport of proteins to the mitochondrial intermembrane space: the 'matrix-targeting' and the 'sorting' domains in the cytochrome c1 presequence. EMBO J. 1987 Aug;6(8):2433–2439. doi: 10.1002/j.1460-2075.1987.tb02522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Loon A. P., Brändli A. W., Schatz G. The presequences of two imported mitochondrial proteins contain information for intracellular and intramitochondrial sorting. Cell. 1986 Mar 14;44(5):801–812. doi: 10.1016/0092-8674(86)90846-9. [DOI] [PubMed] [Google Scholar]
- van Loon A. P., Young E. T. Intracellular sorting of alcohol dehydrogenase isoenzymes in yeast: a cytosolic location reflects absence of an amino-terminal targeting sequence for the mitochondrion. EMBO J. 1986 Jan;5(1):161–165. doi: 10.1002/j.1460-2075.1986.tb04191.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Jagow G., Sebald W. b-Type cytochromes. Annu Rev Biochem. 1980;49:281–314. doi: 10.1146/annurev.bi.49.070180.001433. [DOI] [PubMed] [Google Scholar]