Abstract
Approximately 95% of the viroid RNA which is present in potato spindle tuber viroid (PSTV)-infected tomato plant leaf issue, is associated with the nucleolar fraction obtained from purified nuclei. Viroids were released from the nucleolar fraction by increasing the ionic strength of the medium to 0.66 suggesting that viroid RNA is present in these subnuclear components in a protein-nucleic acid complex. A purification procedure for nuclei from leaf tissue had to be newly developed; it involves two Percoll density centrifugations as final steps. The nuclei were sonicated and the sonicate fractionated into fractions either highly enriched in nucleoli or in broken chromatin and ribonucleoprotein particles. The viroid content in the different samples was determined by gel electrophoresis. Depending upon the progress of the disease, viroid copy numbers between 200 and 10,000 per cell were observed in homogenized tissue, purified nuclei and in the nucleolar fraction. In chloroplasts, practically no viroids were detected. The results are discussed in the light of current hypotheses about the replication, pathogenicity and origin of viroids.
Full text
PDF
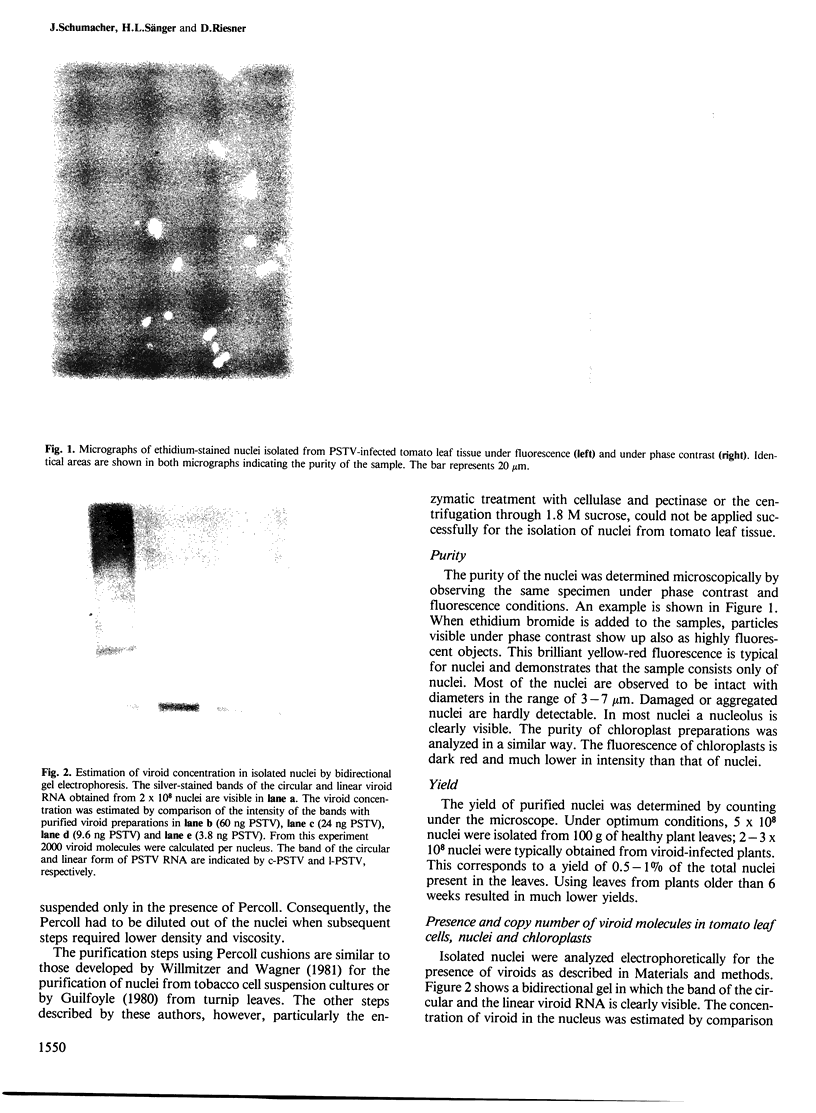





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Branch A. D., Robertson H. D., Dickson E. Longer-than-unit-length viroid minus strands are present in RNA from infected plants. Proc Natl Acad Sci U S A. 1981 Oct;78(10):6381–6385. doi: 10.1073/pnas.78.10.6381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busch H., Reddy R., Rothblum L., Choi Y. C. SnRNAs, SnRNPs, and RNA processing. Annu Rev Biochem. 1982;51:617–654. doi: 10.1146/annurev.bi.51.070182.003153. [DOI] [PubMed] [Google Scholar]
- Chen Y. M., Lin C. Y., Chang H., Guilfoyle T. J., Key J. L. Isolation and Properties of Nuclei from Control and Auxin-treated Soybean Hypocotyl. Plant Physiol. 1975 Jul;56(1):78–82. doi: 10.1104/pp.56.1.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickson E. A model for the involvement of viroids in RNA splicing. Virology. 1981 Nov;115(1):216–221. doi: 10.1016/0042-6822(81)90104-5. [DOI] [PubMed] [Google Scholar]
- Diener T. O. Are viroids escaped introns? Proc Natl Acad Sci U S A. 1981 Aug;78(8):5014–5015. doi: 10.1073/pnas.78.8.5014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flores R., Semancik J. S. Properties of a cell-free system for synthesis of citrus exocortis viroid. Proc Natl Acad Sci U S A. 1982 Oct;79(20):6285–6288. doi: 10.1073/pnas.79.20.6285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grill L. K., Semancik J. S. RNA sequences complementary to citrus exocortis viroid in nucleic acid preparations from infected Gynura aurantiaca. Proc Natl Acad Sci U S A. 1978 Feb;75(2):896–900. doi: 10.1073/pnas.75.2.896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross H. J., Domdey H., Lossow C., Jank P., Raba M., Alberty H., Sänger H. L. Nucleotide sequence and secondary structure of potato spindle tuber viroid. Nature. 1978 May 18;273(5659):203–208. doi: 10.1038/273203a0. [DOI] [PubMed] [Google Scholar]
- Gross H. J., Krupp G., Domdey H., Raba M., Jank P., Lossow C., Alberty H., Ramm K., Sänger H. L. Nucleotide sequence and secondary structure of citrus exocortis and chrysanthemum stunt viroid. Eur J Biochem. 1982 Jan;121(2):249–257. doi: 10.1111/j.1432-1033.1982.tb05779.x. [DOI] [PubMed] [Google Scholar]
- Lerner M. R., Boyle J. A., Mount S. M., Wolin S. L., Steitz J. A. Are snRNPs involved in splicing? Nature. 1980 Jan 10;283(5743):220–224. doi: 10.1038/283220a0. [DOI] [PubMed] [Google Scholar]
- Mühlbach H. P., Sänger H. L. Viroid replication is inhibited by alpha-amanitin. Nature. 1979 Mar 8;278(5700):185–188. doi: 10.1038/278185a0. [DOI] [PubMed] [Google Scholar]
- Pederson T. Gene activation in eukaryotes: are nuclear acidic proteins the cause or the effect? Proc Natl Acad Sci U S A. 1974 Mar;71(3):617–621. doi: 10.1073/pnas.71.3.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rackwitz H. R., Rohde W., Sänger H. L. DNA-dependent RNA polymerase II of plant origin transcribes viroid RNA into full-length copies. Nature. 1981 May 28;291(5813):297–301. doi: 10.1038/291297a0. [DOI] [PubMed] [Google Scholar]
- Reddy R., Henning D., Busch H. Nucleotide sequence of nucleolar U3B RNA. J Biol Chem. 1979 Nov 10;254(21):11097–11105. [PubMed] [Google Scholar]
- Rohde W., Sänger H. L. Detection of complementary RNA intermediates of viroid replication by Northern blot hybridization. Biosci Rep. 1981 Apr;1(4):327–336. doi: 10.1007/BF01114872. [DOI] [PubMed] [Google Scholar]
- Semancik J. S., Tsuruda D., Zaner L., Geelen J. L., Weathers J. G. Exocortis disease: subcellular distribution of pathogenic (viroid) RNA. Virology. 1976 Feb;69(2):669–676. doi: 10.1016/0042-6822(76)90495-5. [DOI] [PubMed] [Google Scholar]
- Stein A. DNA folding by histones: the kinetics of chromatin core particle reassembly and the interaction of nucleosomes with histones. J Mol Biol. 1979 May 15;130(2):103–134. doi: 10.1016/0022-2836(79)90421-2. [DOI] [PubMed] [Google Scholar]
- Takahashi T., Diener T. O. Potato spindle tuber viroid. XIV. Replication in nuclei isolated from infected leaves. Virology. 1975 Mar;64(1):106–114. doi: 10.1016/0042-6822(75)90083-5. [DOI] [PubMed] [Google Scholar]
- Willmitzer L., Wagner K. G. The isolation of nuclei from tissue-cultured plant cells. Exp Cell Res. 1981 Sep;135(1):69–77. doi: 10.1016/0014-4827(81)90300-1. [DOI] [PubMed] [Google Scholar]
- Zieve G., Penman S. Subnuclear particles containing a small nuclear RNA and heterogeneous nuclear RNA. J Mol Biol. 1981 Jan 25;145(3):501–523. doi: 10.1016/0022-2836(81)90542-8. [DOI] [PubMed] [Google Scholar]
- deHaseth P. L., Lohman T. M., Burgess R. R., Record M. T., Jr Nonspecific interactions of Escherichia coli RNA polymerase with native and denatured DNA: differences in the binding behavior of core and holoenzyme. Biochemistry. 1978 May 2;17(9):1612–1622. doi: 10.1021/bi00602a006. [DOI] [PubMed] [Google Scholar]







