Abstract
Polycomb group (PcG) and Trithorax group (TrxG) genes encode important regulators of development and differentiation in metazoans. These two groups of genes were discovered in Drosophila by their opposing effects on homeotic gene (Hox) expression. PcG genes collectively behave as genetic repressors of Hox genes, while the TrxG genes are necessary for HOX gene expression or function. Biochemical studies showed that many PcG proteins are present in two protein complexes, Polycomb repressive complexes 1 and 2, which repress transcription via chromatin modifications. TrxG proteins activate transcription via a variety of mechanisms. Here we summarize the large body of genetic and biochemical experiments in Drosophila on these two important groups of genes.
Keywords: Drosophila, Polycomb, Trithorax, FlyBook
THE Polycomb group (PcG) and Trithorax group (TrxG) genes were first identified in Drosophila as trans-acting regulators of bithorax complex (BX-C) and Antennapedia complex (ANT-C) homeotic genes. We will refer to the BX-C and ANT-C homeotic genes, collectively, as the Hox genes. The Drosophila body is divided into segments along the anterior-posterior axis at the embryonic, larval, and adult stages. The Hox genes specify the identities of segments at all stages of development, and either loss of function or ectopic expression can alter segmental identity. In the maintenance of proper Hox gene expression, the PcG and TrxG proteins add epigenetic memory to the regulation of their target genes. This is an important function in metazoans, which must differentiate specialized cells at specific times and locations during development. Single-celled organisms must often rapidly change their specialized functions to take advantage of changing environments, and consequently, their use of epigenetic memory can differ from that in metazoans. A striking example is PcG transcriptional silencing based on trimethylation of lysine 27 (K27me3) of histone H3, which is conserved between Drosophila and vertebrates, but appears to be lacking in both Saccharomyces cerevisiae and Schizosaccharomyces pombe (Lachner et al. 2004; Garcia et al. 2007).
History of PcG and TrxG
In Drosophila, a specialized row of distinctive bristles (the sex comb) is present on the first pair of thoracic legs of adult males (Figure 1A). In 1940, Slifer found a recessive mutation whose phenotype included the presence of partial sex combs on the second and third pairs of legs of adult males. She named this mutation extra sex combs (esc) (Slifer 1942). Several years later, Pam Lewis isolated a dominant mutation with a similar phenotype, Polycomb (Pc) (Lewis 1947). Over the next 30 years, additional dominant and recessive mutations with the extra-sex-combs phenotype were isolated, but they were usually viewed in a leg-specific developmental context, such as affecting some type of pattern gradient from anterior to posterior in the thorax (Slifer 1942), or as a defect in imaginal-disc proliferation causing transdetermination (Gehring 1970; Shearn et al. 1978). The conceptual breakthrough came with Ed Lewis’s description of the phenotype of homozygous Pc mutant larvae (in which the thoracic and first seven abdominal segments were partially transformed toward the identity of the eighth abdominal segment) and his proposal that Pc encodes a global repressor of all of the BX-C genes (Lewis 1978). This was a fundamental shift in thinking that shaped all subsequent investigations of the PcG genes.
Figure 1.
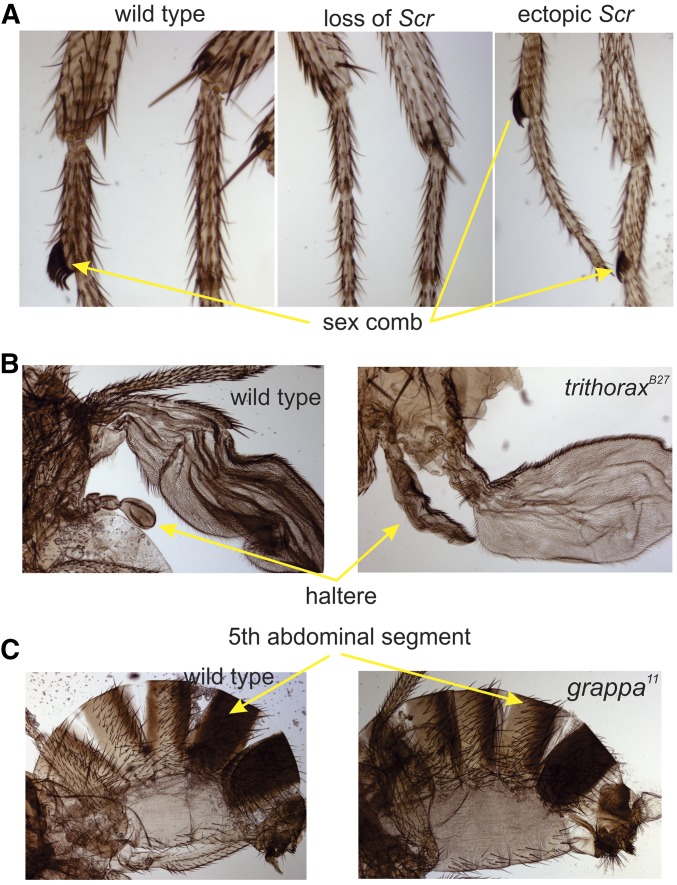
Homeotic transformations are diagnostic phenotypes of PcG and TrxG mutants. (A) Shows the first tarsal segments of the first and second thoracic legs of adult males. The Hox gene Scr is expressed in the cells that form the first leg, causing the cells to differentiate the row of distinctive bristles called a sex comb. At the left of (A) is a wild-type male with a sex comb on the first leg. In the middle of (A) is a male with loss of Scr function and no sex comb on either first or second legs (a TrxG mutant phenotype). At the right of (A) is a male with ectopic expression Scr in the second leg and sex combs on both the first and second legs (a PcG phenotype). (B) Shows the wings and halteres of pharate adult flies. At the left is a wild-type fly and at the right is a homozygous trithoraxB27 mutant fly. Loss of function of the Hox gene Ubx in the third thoracic segment caused the differentiation of anterior wing structures in place of the haltere structures. There are also transformations of the posterior wing to a more anterior identity caused by en loss of function. (C) Shows the abdominal segments of adult males. At the left is a wild-type male and at the right is a grappa11 mutant male. Loss of Abd-B function in the grappa mutant transformed the fifth abdominal segment to a fourth abdominal segment identity.
As mutations that caused the extra-sex-combs phenotype were recovered in an increasing number of genes (Gehring 1970; Shearn et al. 1978; Duncan 1982; Ingham 1984; Dura et al. 1985; Jürgens 1985), it was suggested that those genes in which zygotic mutations have phenotypes that resembled weak Pc mutants in both their dominant adult and recessive embryonic phenotypes be collectively referred to as the PcG (Jürgens 1985). While the original list of five PcG genes [Pc, Additional sex combs (Asx), Polycomblike (Pcl), Posterior sex combs (Psc), and Sex combs on midleg (Scm)] excluded the genes for which the extra-sex-combs phenotype was a recessive rather than dominant mutant phenotype {esc, pleiohomeotic (pho), Enhancer of zeste [E(z)], super sex combs (sxc), and polyhomeotic (ph)}, these latter genes have been included in every subsequent list of PcG genes. Since 1985, mutations that cause an extra-sex-combs phenotype have been characterized in only five additional genes {Sex combs extra (Sce), multi sex combs (mxc), cramped (crm), Suppressor of zeste 12 [Su(z)12], and wings apart-like (wapl)} (Breen and Duncan 1986; Santamaría and Randsholt 1995; Yamamoto et al. 1997; Birve et al. 2001; Cunningham et al. 2012). While the extra-sex-combs phenotype, which is caused by ectopic expression of the Hox gene Sex combs reduced (Scr), was the original phenotype for defining the PcG genes, mutant phenotypes caused by ectopic expression of other Hox genes have also been used to suggest inclusion in the PcG. For example, clones of Scm-related gene containing four mbt domains (Sfmbt) or calypso mutant cells in imaginal discs show ectopic expression of multiple Hox genes (Klymenko et al. 2006; Gaytán et al. 2007).
Once the idea of a global repressor of BX-C genes was proposed, the suggestion of a global activator soon followed. Ed Lewis mentioned a candidate mutation for a positive regulator for the BX-C genes that he had localized to salivary gland chromosome subdivision 88B, but gave no details (Lewis 1968). This mutation, which Lewis first named lethal(3)bithoraxvariegated [l(3)bxv] and then renamed Regulator of bithorax (Rg-bx), was extensively characterized by Capdevila and Garcia-Bellido (Garcia-Bellido and Capdevila 1978; Capdevila and Garcia-Bellido 1981). At about the same time, another allele was characterized under the name trithorax1 (trx1) (Ingham and Whittle 1980; Ingham 1981). The trithorax mutant phenotypes mimic the loss-of-function phenotypes of the Hox genes. Mutants that mimic the Hox gene loss-of-function phenotypes were also identified at several other genes, including female sterile (1) homeotic [fs(1)h]; absent, small, or homeotic discs 1 (ash1); and absent, small, or homeotic discs 2 (ash2) (Forquignon 1981; Digan et al. 1986; Shearn et al. 1987). The positive regulators of the Hox genes are now collectively known as the TrxG genes. Given the complexity of factors required for gene expression, the TrxG was expected to be more heterogeneous than the PcG (Kennison and Tamkun 1988; Kennison 1993, 1995); an expectation fulfilled by both the diverse mutant phenotypes and the biochemical requirements for TrxG proteins at multiple steps in transcriptional activation and elongation (see below).
Genetic Screens Used To Isolate PcG and TrxG Genes
While the first PcG and TrxG mutations were isolated by chance, there have been several forward genetic screens designed to identify new members of both groups. Most screens for PcG genes relied heavily on the extra-sex-combs phenotype (shown in Figure 1A). Screens for dominant or recessive mutations with the extra-sex-combs phenotype, however, identified only a few genes (Gehring 1970; Ingham 1984; Dura et al. 1985; Jürgens 1985). A second approach was to screen for dominant enhancers of PcG mutants (Duncan 1982; Kennison and Tamkun 1988; Fauvarque et al. 2001) or dominant enhancers of cis-regulatory mutations that partially derepressed various Hox genes (Botas et al. 1982; Breen and Duncan 1986). Again, mutations were isolated at only a few genes. A recent phenotypic screen for PcG mutations used mitotic recombination to generate clones of homozygous mutant imaginal wing cells in individuals heterozygous for newly induced mutations. Mutations with a mutant-wing phenotype that resembled the phenotype of Pc mutants were then examined for ectopic expression of Hox genes (Gaytán et al. 2007). Another recent screen isolated dominant suppressors of the pairing-sensitive silencing caused by PcG response elements (Cunningham et al. 2012). Finally, additional PcG mutations were isolated in screens that were not intended to identify regulators of Hox genes, but were designed to isolated dominant modifiers of the eye color mutant z1, a gain-of-function allele of zeste (z) (Kalisch and Rasmuson 1974; Wu et al. 1989; Birve et al. 2001). Many of these dominant modifiers of the z1 mutation are alleles of the PcG genes Suppressor of zeste 2 [Su(z)2], Su(z)12, Scm, and E(z). An interesting quirk of Drosophila nomenclature has caused considerable confusion for those reading the PcG literature for the first time. The E(z) gene was named for the first allele, which is a dominant enhancer of z1 (Kalisch and Rasmuson 1974). This allele, however, is a gain-of-function allele (Jones and Gelbart 1990). Loss-of-function alleles of E(z) are dominant suppressors of z1 (Wu et al. 1989; Jones and Gelbart 1990).
Only a few forward genetic screens were actually designed to identify TrxG genes. One of the reasons for the initial suggestion that trx is a global activator of the Hox genes was the dosage-sensitive genetic interactions observed between trx and Pc mutations (Capdevila and Garcia-Bellido 1981; Capdevila et al. 1986). In flies heterozygous for mutations or deletions of trx, the phenotypes of Pc mutants were suppressed; i.e., trx mutations are dominant suppressors of Pc mutants. Screening for dominant suppressors of Pc identified more than a dozen new genes required for the positive regulation of the Hox genes, including brahma (brm), moira (mor), osa (osa), kismet (kis), kohtalo (kto), and skuld (skd) (Kennison and Tamkun 1988; Fauvarque et al. 2001). Forward genetic screens for dominant enhancers of TrxG mutants have also identified several new TrxG genes (Vázquez et al. 1999; Gildea et al. 2000; Gutiérrez et al. 2003). An extensive genetic screen was conducted in the McGinnis laboratory in the 1990s (Harding et al. 1995; Gellon et al. 1997; Florence and McGinnis 1998) to isolate dominant mutations that reduced the viability of a mutant genotype with reduced function of the Hox gene, Deformed (Dfd). While this was a more general screen designed to isolate mutations with effects on Dfd regulation or function, TrxG mutations were a subgroup of the mutations recovered. In addition to the forward genetic screens to isolate mutations in PcG and TrxG genes, reverse genetic approaches have also been used. In the case of reverse genetics, a candidate gene is first identified based on its protein sequence. Mutations in that candidate gene are then characterized to determine whether the mutant phenotype shows misregulation of Hox gene function or expression. In the absence of clear evidence of Hox gene misregulation, a candidate gene is often then tested for enhancement or suppression of known PcG and TrxG mutant phenotypes (see Kennison 2004 for a description of some of the mutant phenotypes used for such genetic tests).
PcG and TrxG Mutant Phenotypes
There have been extensive characterizations of the mutant phenotypes for both PcG and TrxG genes. These include descriptions of the differentiation of larval and adult cuticular structures, the expression patterns of target genes in embryos and imaginal cells, and the expressions of target genes in tissue culture cells. The mutants examined may result from loss of zygotic and/or maternal functions, or from loss in clones of cells. The loss of function in clones of cells has been generated in heterozygous mutant flies by mitotic recombination, or by expression of RNA interference (RNAi) constructs. For tissue culture cells, the most common approach has been reducing expression using RNAi. We will only describe the results of a few of these studies, primarily to emphasize that there are additional target genes beyond the Hox genes and that the PcG and TrxG proteins are not monolithic in function.
PcG mutants
To determine the complete loss-of-function phenotypes for Drosophila genes, it is necessary not only to remove the zygotic functions, but also the maternal contributions of wild-type gene products deposited in the unfertilized eggs. For at least 70% of Drosophila genes, the maternal contribution can alter the zygotic mutant phenotype (Perrimon and Mahowald 1986). To examine the phenotype after loss of maternal function, both pole cell transplantation and mitotic recombination in the female germ line have been used to block the deposition of maternally encoded gene products. In some cases, the mutant germ cells failed to produce mature eggs, showing that the tested gene is required for normal oogenesis. Thus, for the PcG genes crm, E(z), mxc, Su(z)12, and Sfmbt, the effects of complete loss of the maternal functions cannot be determined, since these gene functions are required for oogenesis (Shannon et al. 1972; Phillips and Shearn 1990; Docquier et al. 1996; Birve et al. 2001; Klymenko et al. 2006; Iovino et al. 2013). However, when fertilized eggs are laid, the effect of maternally encoded gene products can be examined in embryos that also lack zygotic gene products, as well as in embryos that receive a paternal wild-type allele.
Among the PcG genes, the first examined for loss of both maternal and zygotic function in embryos were Pc and esc. Embryos that lacked both maternal and zygotic Pc functions showed the same homeotic phenotype (transformation of all thoracic and abdominal segments to an eighth abdominal segment identity) first described for embryos that lacked only zygotic Pc functions, but with stronger and more consistent transformations (Haynie 1983; Lawrence et al. 1983). Embryos that lacked both maternal and zygotic esc functions had homeotic phenotypes like that of homozygous Pc embryos, while embryos that lacked only maternal or only zygotic esc functions gave rise to some viable adults (Struhl 1981; Lawrence et al. 1983). Neither Pc nor esc is required for oogenesis or for normal segmentation of the embryo. However, the lack of effects on oogenesis or segmentation in embryos that lack esc function may be due to the presence of a second gene, escl, which partially compensates for some esc functions (Wang et al. 2006; Kurzhals et al. 2008; Ohno et al. 2008). Loss of escl function alone does not affect viability, fertility, or visible phenotype, but it strongly enhances many esc mutant phenotypes.
Loss of maternal and zygotic functions for Sce, Scm, or Asx produced embryos with homeotic transformations of most segments, but no segmentation defects (Breen and Duncan 1986; Soto et al. 1995; Fritsch et al. 2003). Loss of both maternal and zygotic functions for either sxc or calypso also produced embryos with no segmentation defects and only weak homeotic transformations of abdominal segments (Ingham 1984; Gaytán et al. 2007). Loss of both maternal and zygotic functions for Pcl produced embryos with not only homeotic transformation, but also with defects in even numbered segments (Breen and Duncan 1986).
The esc gene is not the only PcG gene whose analyses have been complicated by the presence of a related gene in the genome. The PcG genes pho, ph, and Psc also have related genes in the genome. Almost all embryos that lacked both maternal and zygotic functions of pho failed to differentiate the cuticle, but the few that did differentiate the cuticle showed severe defects in segmentation (Breen and Duncan 1986). Again, the lack of effects on oogenesis may be due to a second gene related to pho, pleiohomeotic like (phol), which can partially compensate for some pho functions (Brown et al. 2003). Since its original description, the ph gene has been shown to be two adjacent genes, ph-d and ph-p, which appear to have arisen by tandem duplication and have largely redundant functions at all developmental stages (Dura et al. 1987). Loss of both maternal and zygotic functions for either gene alone had only minor effects on adult phenotypes (the extra-sex-combs phenotype being the most penetrant) (Dura et al. 1985, 1987), but loss of maternal and zygotic functions of both genes (the double mutant) produced embryos that failed to differentiate the cuticle (Dura et al. 1988; Smouse et al. 1988). Finally, the Psc and Su(z)2 genes are also adjacent in the genome and encode related proteins. Loss of both maternal and zygotic Psc functions produced embryos with homeotic defects, HOX gene misexpression, and early developmental defects (Martin and Adler 1993; Soto et al. 1995). Loss of both maternal and zygotic Su(z)2 functions produced embryos with no homeotic or segmentation defects and no HOX gene misexpression (Soto et al. 1995). Simultaneous loss of maternal and zygotic functions for both Psc and Su(z)2 produced embryos slightly more defective in Hox gene regulation than loss of Psc alone, suggesting limited redundancy between Psc and Su(z)2 in HOX gene regulation in embryos which is only observable when Psc function is greatly reduced (Soto et al. 1995). The situation in imaginal wing disc cells, however, is quite different. Clones of cells that were homozygous mutants for either Psc or Su(z)2 showed no derepression of Hox genes, while clones of cells that were homozygous mutants for both genes showed extensive Hox gene derepression (Beuchle et al. 2001). Thus, Psc and Su(z)2 appear completely redundant in HOX gene regulation in wing imaginal cells.
As described above, although PcG genes were first identified by Hox-gene-misexpression phenotypes, the mutant phenotypes suggest that some PcG proteins have additional target genes, such as some of the segmentation genes. For example, embryos that lacked both ph-d and ph-p zygotic functions showed extensive misexpression of the segmentation gene engrailed (en) (Dura and Ingham 1988). In addition, although embryos that lacked both maternal and zygotic functions of either Pc, Scm, or Asx appeared to differentiate the larval cuticle with no segmentation defects as described above, closer examination showed ectopic expression of en in a few cells in mutant embryos that lacked zygotic function (Moazed and O’Farrell 1992). The necessity for PcG functions to maintain repression of en is even more evident in imaginal cells (Busturia and Morata 1988). In addition to en and the Hox genes, many other targets of PcG repression have been identified, including some of the PcG genes themselves (Fauvarque et al. 1995; Bloyer et al. 2003; Ali and Bender 2004; Park et al. 2012). Each target gene may require a different subset of proteins to maintain transcriptional repression, and the subset of proteins required may differ between cell types or at different stages of development. While there is often disagreement on how broadly to define the PcG, there is a consensus that the PcG should include genes that encode proteins directly required for maintaining transcriptional repression of the Hox genes in embryos and/or in imaginal cells.
Some PcG genes also appear to have functions that extend beyond transcriptional repression, such as chromosome condensation, integrity, or behavior. For example, E(z) mutants exhibited both failures of condensation and chromosomal breakage at mitotic metaphase (Gatti and Baker 1989; Phillips and Shearn 1990; O’Dor et al. 2006). Mutants for ph-p (but not mutants for ph-d) exhibited anaphase bridges at mitotic divisions in early embryos (Lupo et al. 2001; O’Dor et al. 2006). This is the clearest example of a nonredundant function between the ph-d and ph-p genes. The PcG gene Psc also appears to have a function in mitosis that is not dependent on its function in transcriptional regulation. Psc mutant embryos exhibited anaphase bridges at mitotic divisions (O’Dor et al. 2006; Mohd-Sarip et al. 2012). This may be due to defects in cell cycle control, since Psc protein physically interacts with Cyc-B protein and Psc mutants showed defects in Cyc-B degradation (Mohd-Sarip et al. 2012).
TrxG mutants
Only a few of the TrxG genes were first identified because their phenotypes mimic the phenotypes of Hox gene loss of function. These include fs(1)h, trx, ash1, and ash2. Although fs(1)h is required for oogenesis (Perrimon et al. 1984), loss of maternal and/or zygotic functions have been extensively studied using the temperature-sensitive allele, fs(1)h1. At more restrictive temperatures, fs(1)h1 mutant females laid fertilized eggs that had defects in the early nuclear divisions (Zalokar et al. 1975). In about half of the embryos, the nuclei were haploid. There were also defects in both the yolk nuclei and the blastoderm nuclei in many embryos, including polyploid mitoses in later blastoderm stages. At more permissive temperatures, the fs(1)h1 mutant females laid fertilized eggs that gave rise to viable adults with homeotic defects, including transformations of anterior metanotum and anterior haltere to anterior mesonotum and anterior wing, respectively. These homeotic transformations mimic loss of function for the Hox gene Ultrabithorax (Ubx) (Forquignon 1981). In addition to the homeotic transformations, mutant progeny were often missing legs, halteres, or tergites.
The trx and ash1 genes have very similar (but not identical) mutant phenotypes. Neither is required for oogenesis, and loss of both maternal and zygotic functions for trx or ash1 gives rise to embryos with no defects in segmentation, and few homeotic transformations (Ingham 1983; Tripoulas et al. 1994; Klymenko and Müller 2004). Loss of trx zygotic function is only slightly more normal than loss of both maternal and zygotic functions, and reduces the embryonic expression of multiple Hox genes (Mazo et al. 1990; Breen and Harte 1993). In contrast, reduction of the expression of Hox genes was only observed in embryos that lacked both maternal and zygotic ash1 functions (Klymenko and Müller 2004). While loss of trx or ash1 causes only minor defects in mutant embryos, the effects on imaginal tissues are striking, with homeotic transformations observed in many segments, including transformations of distal antenna and arista to distal leg structures, proboscis to leg structures, dorsal prothorax to wing, first and third legs to a second leg identity, haltere to wing, and transformations of abdominal and genital structures to more anterior identities (Ingham and Whittle 1980; Ingham 1981, 1985; Shearn et al. 1987; Tripoulas et al. 1994). The transformation of haltere to anterior wing in a trx mutant is shown in Figure 1B. In addition to the phenotypes expected from loss of Hox gene functions, trx and ash1 also have mutant phenotypes that resemble loss of en function in imaginal tissues (Ingham 1985; Shearn et al. 1987). Zygotic loss of ash2 function causes homeotic phenotypes very similar to those of trx and ash1 (Shearn et al. 1987). In contrast to trx and ash1, however, the ash2 mutant phenotypes do not suggest defects in en function in either embryos or imaginal tissues.
The rest of the TrxG genes were identified based on genetic interactions or by reverse genetics. These TrxG genes have diverse mutant phenotypes that suggest not only the regulation of many target genes beyond the Hox genes, but also many differences in the sets of target genes. In this review, we will primarily describe the homeotic and segmentation phenotypes of TrxG genes.
In addition to fs(1)h, several other TrxG genes are required for oogenesis, including brm, mor, Snf5-related 1 (Snr1), and Trithorax-like (Trl) (Brizuela et al. 1994; Bhat et al. 1996; Brizuela and Kennison 1997; Zraly et al. 2003). Loss of zygotic mor function is embryonic lethal with head defects that resemble the defects seen in hypomorphic mutants of the Hox gene Dfd (Harding et al. 1995). Clones of mor mutant cells in imaginal discs caused transformations of metanotum and haltere to mesonotum and anterior wing, respectively, and transformations of posterior wing to anterior wing (Brizuela and Kennison 1997). The transformations of posterior wing to anterior wing were associated with reductions in en expression. Clones of brm and Snr1 mutant cells were small (suggesting defects in cell division) and caused no clear homeotic transformations, but had defects suggesting effects on the adult peripheral nervous system (Elfring et al. 1998; Zraly et al. 2003). Hypomorphic brm mutants that survived to adults did have homeotic phenotypes, including reductions in the numbers of sex comb teeth and transformations of the fifth abdominal segment to a more anterior identity (Tamkun et al. 1992). Loss of zygotic function for Trl is lethal at the third larval instar with no apparent cuticular defects, but hypomorphic mutant males survive and have a few bristles on the sixth sternite; suggesting a partial loss of function of the Hox gene Abdominal B (Abd-B) (Farkas et al. 1994). Clones of Trl mutant cells in imaginal discs caused no misexpression of Hox genes (Brown et al. 2003; Bejarano and Busturia 2004).
The TrxG genes osa, kis, and tonalli (tna) are not required for oogenesis and the phenotypes after loss of both maternal and zygotic functions have been examined (Daubresse et al. 1999; Vázquez et al. 1999; Gutiérrez et al. 2003). For osa or kis, embryos that lacked zygotic function had no obvious homeotic or segmentation defects. Loss of maternal function for either caused segmentation defects in embryos, with loss of maternal osa resembling the phenotypes of gap segmentation mutants and loss of maternal kis resembling the phenotype of pair-rule segmentation mutants. Clones of kis mutant cells in imaginal tissues caused homeotic transformations of the fifth abdominal segment to a more anterior identity (loss of Abd-B function), and some transformations of first leg toward a second leg identity (loss of Scr function). The leg transformations were only observed if the kis mutant clones were induced early in development, at the cellular blastoderm stage. For tna, loss of zygotic function caused lethality over a broad period of development, from the third larval instar to the pharate adult stages. Males that survived to the pharate adult stage had transformations of haltere to wing and reductions in the numbers of sex comb teeth (loss of Ubx and Scr functions, respectively). Loss of maternal tna function was completely rescued paternally, but loss of both maternal and zygotic functions caused lethality primarily at the third larval instar.
Loss of function for either kto or skd causes almost identical phenotypes. Clones of mutant cells in regions of the leg imaginal discs that give rise to distal leg structures showed both homeotic transformations (reductions in the numbers of sex comb teeth in the first leg) and defects in segmentation of the tarsal segments (Loncle et al. 2007). Clones of mutant cells in the wing imaginal disc caused Ubx misexpression in a small subset of clones in the wing pouch, but clones of mutant cells in leg, haltere, wing, or eye-antennal discs caused no misexpression of the Hox genes Scr, abdominal A (abd-A), or Abd-B (Gaytán et al. 2007).
Finally, hypomorphic mutants for grappa (gpp) and modifier of mdg4 [mod(mdg4)] can eclose as adults with homeotic transformations. Hypomorphic gpp mutants showed antenna to leg transformations, reductions in the numbers of sex comb teeth on the first legs of males, and transformations of posterior abdominal segments to more anterior identities (Shanower et al. 2005). A gpp mutant with transformation of the fifth abdominal segment to a more anterior identity is shown in Figure 1C. Hypomorphic mod(mdg4) mutant males also showed some transformation of the fifth abdominal segment to a more anterior identity (Dorn et al. 1993).
Developmental and Genetic Models for PcG and TrxG Functions
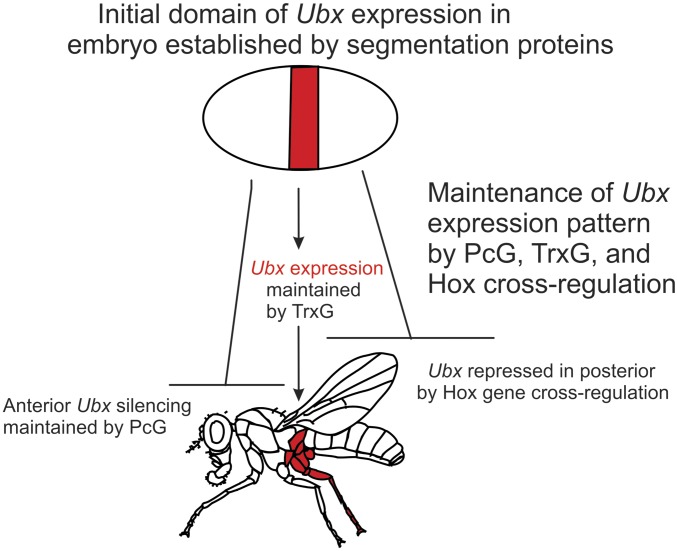
The current developmental model for PcG and TrxG functions is relatively simple, and is illustrated for the Hox gene Ubx in Figure 2. The Hox genes are expressed in restricted spatial domains within the anterior-posterior axis of the Drosophila body plan. For each Hox gene, the domain of expression is determined before the cellular blastoderm stage, and is maintained through many cell divisions until differentiation of either the larval or adult structures. Expression of a Hox gene within its proper domain requires the TrxG gene products. Transcriptional silencing of a Hox gene in cells in which it should not be expressed relies on initial repression by the products of the segmentation genes (the genes that are responsible for dividing the Drosophila body into segments), and subsequent maintenance of repression by the PcG gene products. This model arose fairly early in the studies of Hox gene regulation, as described below.
Figure 2.
Developmental model of the regulation of Hox genes by the PcG and TrxG proteins. The establishment of the initial domain of the Hox gene Ubx by the segmentation proteins is shown at the top, with Ubx repressed anterior to its domain of expression by the Hb gap segmentation protein. The initial domain of Ubx expression is maintained through larval and pupal development by the TrxG proteins. Maintenance of silencing of the Ubx gene anterior to this domain requires both PcG proteins and trimethylation of lysine 27 on histone H3 (H3K27me3). Posterior to its normal domain of expression, Ubx is repressed by the Hox proteins Abd-A and Abd-B, not by the PcG proteins, or by histone H3K27me3.
Based on the cell-autonomous requirement for Pc in the larval imaginal cells and the partial transformations observed in Pc embryos, it was suggested that the role of Pc is in maintenance, rather than initiation, of BX-C gene expression (Struhl 1981; Denell and Frederick 1983). Struhl and Akam (1985) provided the first molecular evidence for this model. They showed that in mutant embryos that lacked both maternal and zygotic esc functions, the Hox gene Ubx was initially expressed in its normal domain at the cellular blastoderm stage, but showed extensive ectopic expression after gastrulation and germ band extension. Similar results were described for embryos that lacked both maternal and zygotic functions of another PcG gene, E(z) (Jones and Gelbart 1990). Around the same time, the initial domains of Hox gene expressions were found to be altered in embryos mutant for various segmentation genes (Duncan 1986; Ingham and Martinez-Arias 1986; White and Lehmann 1986; Akam 1987; Harding and Levine 1988; Ingham 1988). Since many of the segmentation gene products disappear shortly after the initial domains of expression of the Hox genes are set at the cellular blastoderm stage (although many of them are expressed again later during development to determine subsequent cell fates, such as in the nervous system), the need for the PcG and TrxG proteins to maintain the domains of Hox gene expressions quickly became apparent (reviewed in Akam 1987; Duncan 1987; Ingham 1988; Kennison and Tamkun 1992).
How is the switch from the early repression by the segmentation proteins to the PcG maintenance repression accomplished? One clue comes from studies on the Ubx gene. The protein encoded by the hunchback (hb) segmentation gene binds to sites in the Ubx cis-regulatory elements and represses Ubx anterior to the normal domain of expression for Ubx in the early embryo (Qian et al. 1991; Zhang et al. 1991; Zhang and Bienz 1992). Hb protein physically interacts with the Mi-2 subunit of the NURD chromatin-remodeling complex (Kehle et al. 1998), which could modify chromatin to facilitate the recruitment or activation of PcG proteins. Mutants for either of two subunits of the NURD complex, Mi-2 and the histone deacetylase Rpd3, were shown to have defects in Hox gene silencing (Kehle et al. 1998; Chang et al. 2001). While the PcG proteins maintain repression of the Ubx gene anterior to its normal domain of expression, the PcG proteins do not appear to be required for maintenance of Ubx repression posterior to its normal domain of expression. In parasegment 7, the region of the embryo just posterior to the domain of Ubx expression, the Ubx gene does not show enrichment for trimethylation of histone H3K27 (Bowman et al. 2014). Instead, Ubx repression posterior to its normal expression domain appears to be the result of direct repression by the Hox proteins encoded by abd-A and Abd-B (Struhl and White 1985; White and Wilcox 1985).
While TrxG proteins are required for the expression of Hox genes within their normal domain, this requirement appears to be PcG-dependent for some TrxG genes. In the absence of PcG repression, neither trx nor ash1 is required for Hox gene expression in either embryos or in larval imaginal discs. This was first shown for embryos that are mutant for both esc and trx (Ingham 1983). While embryos mutant for esc have homeotic phenotypes that result from missexpression of Hox genes and embryos mutant for trx have homeotic phenotypes that result from failure to express Hox genes, the double mutant embryos have phenotypes that are almost wild type. Even without trx functions, the Hox genes are expressed at levels sufficient for almost normal cuticle differentiation. These observations were confirmed and extended by Klymenko and Müller (2004). They found that in both embryos and in imaginal discs, mutants for either trx or ash1 failed to express Hox genes in the proper spatial domains. However, in combination with PcG mutations, Hox gene expression was now restored in trx or ash1 mutants. These results suggest that at least some of the TrxG proteins function mainly to block establishment of PcG repression.
While these early models still provide the basic framework for transcriptional regulation by the PcG and TrxG genes, considerable progress has been made in understanding the molecular mechanisms behind the models. The remainder of this review will focus on our current understanding of the molecular mechanisms of PcG and TrxG functions in transcriptional regulation.
Mechanism of Action of PcG and TrxG Proteins
The molecular characterization of PcG and TrxG genes and their products provided the first evidence that they might regulate transcription by altering chromatin structure. The fundamental unit of chromatin structure is the nucleosome: an octamer containing histones H2A, H2B, H3, and H4, around which DNA is wrapped like thread around a spool. Nucleosomes and other components of chromatin can repress transcription by blocking the access of regulatory proteins and the general transcription machinery to DNA. Two general mechanisms are used to regulate the repressive effects of nucleosomes on transcription: the covalent modification of nucleosomal histones and ATP-dependent chromatin remodeling. As discussed at length below, PcG and TrxG proteins have been implicated in both of these mechanisms for regulating gene expression.
The covalent modification of nucleosomal histones by methylation, phosphorylation, acetylation, or ubiquitination can alter the binding of structural or regulatory proteins to chromatin. Some histone modifications, including the methylation of lysines 9 and 27 of histone H3 (H3K9 and H3K27), are associated with transcriptional repression; while others, including the methylation of lysines 4 and 36 of histone H3 (H3K4 and H3K36) and the acetylation of lysine 16 of histone H4 (H4K16), are associated with transcriptional activation (Bannister and Kouzarides 2011). Chromatin-remodeling reactions are catalyzed by proteins and protein complexes that use the energy of ATP hydrolysis to alter the assembly, structure, or spacing of nucleosomes (Becker and Workman 2013). By catalyzing ATP-dependent alterations in nucleosome structure or positioning, chromatin-remodeling factors regulate the access of regulatory proteins to DNA in the context of chromatin. Like histone-modifying enzymes, ATP-dependent chromatin-remodeling factors have been implicated in both transcriptional activation and repression.
The first connection between a PcG protein and chromatin was revealed when Paro and Hogness (1991) determined the sequence of the Pc protein and discovered that it contains a 37 aa segment (the chromodomain) that is conserved in HP1, a heterochromatin-associated protein. HP1 is encoded by Su(var)205, a suppressor of position-effect variegation (Eissenberg et al. 1990). Position-effect variegation occurs when a euchromatic gene is juxtaposed to heterochromatin, leading to its hereditable silencing. Because Pc had also been implicated in heritable gene silencing, the presence of chromodomains in the two proteins immediately suggested that Pc might regulate gene expression by altering chromatin structure. This possibility was verified when subsequent studies revealed that chromodomains directly bind methylated histone tails (see below).
Another early connection between PcG proteins and chromatin was suggested by the sequence of the E(z) protein (Jones and Gelbart 1990). E(z) contains a conserved domain (the SET domain) that is present in Trx and Su(var)3-9 (Tschiersch et al. 1994), another suppressor of position-effect variegation implicated in hereditable gene silencing. The SET domain was later shown to be required for the catalytic activity of lysine histone methyltransferases, further strengthening the connection between PcG proteins and chromatin. As described below, the ability of PcG proteins to function as “writers” or “readers” of histone modifications is essential for their ability to maintain heritable gene silencing in Drosophila and other organisms.
Many PcG Proteins Function in Complexes
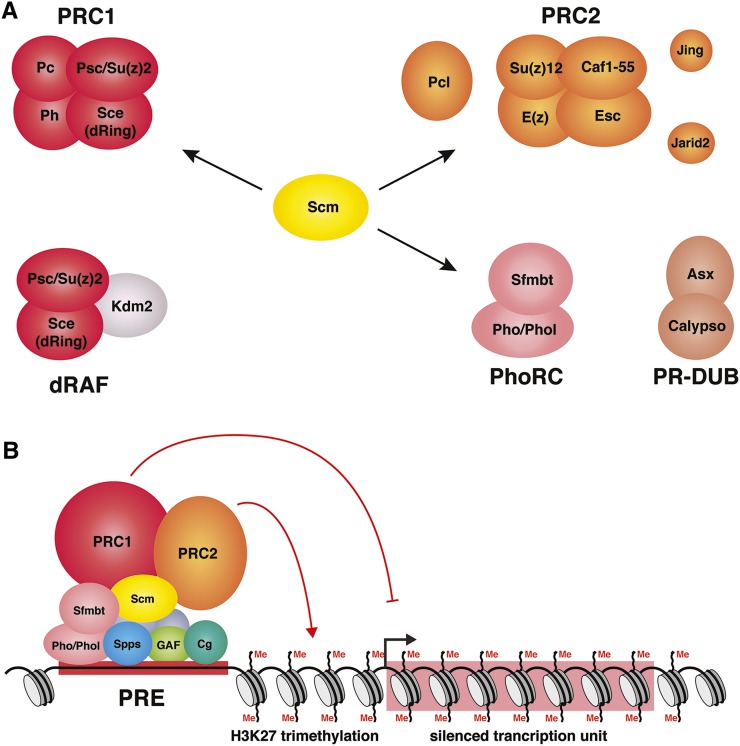
Many of the PcG proteins can be isolated in soluble protein complexes; the best-characterized are Polycomb repressive complex 1 (PRC1) and Polycomb repressive complex 2 (PRC2) (see references below) (Figure 3A). E(z), a core component of PRC2, encodes a histone methyltransferase that trimethylates histone H3 at lysine 27 (H3K27me3); the diagnostic mark of PcG-regulated genes (Figure 3B). “Classical” PcG targets, such as the Hox genes, are coregulated by PRC1 and PRC2 as well as other PcG protein complexes including Polycomb repressive deubiquitinase (PR-DUB), dRing-associated factors (dRAF), and the recruiter complex Pho repressive complex (PhoRC) (Figure 3) (Klymenko et al. 2006; Lagarou et al. 2008; Scheuermann et al. 2010). The combined activities of these protein complexes lead to stable and heritable transcriptional repression of PcG target genes. PRC1 and PRC2 are present in most metazoans and their biochemical properties have been reviewed extensively (Schwartz and Pirrotta 2013; Simon and Kingston 2013; Grossniklaus and Paro 2014). Below we review the Drosophila PcG protein complexes and their activities, as well as highlight a few of the key experiments that made use of the genetic tools available in Drosophila.
Figure 3.
PcG proteins and complexes. (A) PcG protein complexes discussed in this review are shown. Pcl, Jing, and Jarid2 are PRC2-associated proteins that modify the activity of PRC2 (see text). Psc/Su(z)2 and Sce (also known as dRing) are in both PRC1 and dRAF. A recent article provided compelling evidence that Scm interacts closely with PRC1, PRC2, and PhoRC, and suggested that Scm plays a key role in connecting these three complexes (Kang et al. 2015; see text). (B) PcG protein complexes are recruited to DNA by PREs. PREs have binding sites for a large number of DNA-binding proteins; Pho or Phol, Spps, GAF (encoded by the Trl gene), and Cg are shown. PRC2 trimethylates H3K27 and PRC1 inhibits transcription by a variety of mechanisms (see text). Me, methylation.
An Early Model of PcG Protein Recruitment
As soon as antibodies to PcG proteins were made, they were used to detect PcG proteins in embryos and larval tissues. PcG proteins bind at specific bands on larval salivary gland polytene chromosomes, including the locations of the Hox genes (Zink and Paro 1989). This suggested there might be DNA sequences present in Hox genes that could recruit PcG proteins to chromatin. Soon after, specific DNA fragments in transgenes were discovered that could recruit PcG proteins to polytene chromosomes and render reporter gene expression responsive to mutations in PcG genes. These DNA fragments were called “Polycomb group response elements” (PREs) (see below for references and an expanded discussion of PREs) (Figure 3B). While most PcG proteins do not contain DNA-binding domains, one PcG protein, Pho, was found to bind to a specific sequence present in PREs from Hox genes and other PcG targets (Brown et al. 1998). In-vitro experiments showed that Pho could directly interact with E(z) and Esc (L. Wang et al. 2004). This led to the model that PcG recruitment occurred in a sequential order: first, Pho bound to PREs via its DNA-binding domain, and directly recruited PRC2 by protein–protein interactions with E(z) and Esc. PRC2 then acted on flanking nucleosomes to create the H3K27me3 mark. Finally, Pc binding to H3K27me3 via its chromodomain caused the recruitment of PRC1 (L. Wang et al. 2004). This early model was based on recruitment to a single PRE in the Ubx gene in wing discs and other experiments do not support this model (see below).
PRC1
Pc and ph were among the first PcG genes cloned (Paro and Hogness 1991; DeCamillis et al. 1992). Early experiments showed that their proteins co-immunoprecipitated from embryonic nuclear extracts and copurified in a soluble nuclear complex (Franke et al. 1992). To purify Pc and Ph protein complexes, transgenes encoding FLAG-tagged proteins were cloned in P-element vectors, transgenic Drosophila were made, and soluble nuclear protein complexes were purified from embryos (Shao et al. 1999). Mass spectrometry of FLAG-tagged purified protein complexes identified Pc, Ph, Psc, and Sce (also known as dRing) as stoichiometric components of a protein complex, along with substoichiometric amounts of Scm and many other proteins. Further experiments showed that Pc, Ph, Psc, and Sce formed a stable complex when produced in Sf9 insect cells (Francis et al. 2001). This complex is known as PRC1 (Figure 3A). Su(z)2 is a functional homolog of Psc and can replace it in the PRC1 complex (Lo et al. 2009). PRC1 complexes inhibit nucleosome remodeling, transcription, and compact chromatin templates in vitro (Shao et al. 1999; Francis et al. 2001, 2004). Further, each protein has specific domains and activities that give clues to its functions in the PRC1 complex.
Biochemical properties of core PRC1 proteins
As stated above, Pc contains a chromodomain, a 37-amino-acid domain that binds methylated histones. Early experiments expressing truncated and mutated Pc proteins from transgenes showed that the chromodomain was essential for the binding of Pc to chromatin (Messmer et al. 1992). In addition, the chromodomain is sufficient to target Pc to PcG-regulated genes. HP1 and Pc are associated with different chromosomal bands in polytene chromosomes of the larval salivary gland. Replacement of the HP1 chromodomain with the Pc chromodomain created a hybrid protein that bound both Pc and HP1 targets on polytene chromosomes (Platero et al. 1995). This showed that the Pc chromodomain was sufficient to target the hybrid protein to PcG target genes. Subsequent experiments gave a biochemical basis for this result. The HP1 chromodomain binds to H3K9me3 (Bannister et al. 2001; Lachner et al. 2004), a mark of heterochromatin; and the Pc chromodomain binds to H3K27me3, the mark of PcG target genes (Cao et al. 2002; Czermin et al. 2002; Müller et al. 2002). The fact that the Pc chromodomain was sufficient to target a hybrid protein to PcG target genes suggests a hierarchical model for PcG recruitment, i.e., that the H3K27me3 domain created by PRC2 could recruit PRC1 via Pc binding directly to H3K27me3.
Ph proteins are encoded by two adjacent genes ph-d and ph-p (Hodgson et al. 1997), which encode nearly identical proteins. Both Ph-d and Ph-p are present in purified PRC1 protein complexes. We will refer to the Ph-d and Ph-p proteins collectively as Ph. Ph contains a sterile α-motif (SAM) domain, a protein-interaction domain that is also present in the PcG proteins Sfmbt and Scm. tcgqzan almost complete loss hetero- and homo-oligomerization, and the SAM domain of Ph can form a helical polymer (Kim et al. 2002). Deletion of the SAM domain from Ph causes an almost complete loss of Ph activity (Gambetta and Müller 2014). Mutation of a single amino acid in the SAM domain, which disrupts polymerization, also renders Ph unable to repress PcG target genes (Gambetta and Müller 2014). Thus, the SAM domain of Ph is required for the repression activity of PRC1.
The Psc protein contains a homology region (HR) of 200 amino acids also present in Su(z)2 and in the mammalian proteins Bmi-1 and Mel-18 (Brunk et al. 1991; van Lohuizen et al. 1991). This HR includes a cysteine-rich sequence known as a ring finger and a helix-turn-helix motif. Although outside of this region of homology there are no other recognizable domains, the C-termini of Su(z)2 and Psc share similar amino-acid compositions (Brunk et al. 1991; Emmons et al. 2009). In fact, Su(z)2 shares many of the biochemical properties of Psc (Lo et al. 2009), including the ability to inhibit chromatin remodeling and compact chromatin. The HR is required for incorporation of Psc or Su(z)2 into the PRC1 protein complex; the C-terminal nonhomologous region mediates chromatin compaction and inhibition of chromatin remodeling (King et al. 2005; Lo et al. 2009). Functional analysis of Psc mutants showed the requirement of the nonhomologous region of Psc for Hox gene repression in vivo (King et al. 2005). Overexpression of a truncated form of Psc, which contained the HR but lacked the C-terminal repression domain, acted in a dominant-negative fashion in embryos (King et al. 2005). The simplest interpretation of this result is that PRC1 complexes could form with the truncated Psc protein but they could not mediate transcriptional repression and thus interfered with formation of functional PRC1 protein complexes.
Sce (also known as dRing) has H2A ubiquitin-ligase activity and is required for the deposition of the H2AK118ub chromatin mark (H. Wang et al. 2004). Sce is present in at least two other protein complexes and the preponderance of evidence suggests that PRC1 has very low ubiquitin-ligase activity. The protein complex dRAF (Figure 3A) was isolated by immunoprecipitating Sce from Pc-depleted extracts (Lagarou et al. 2008). dRAF contains the core components Psc, Sce, and the demethylase Kdm2, which demethylates H3K36me2, a histone modification catalyzed by the TrxG protein Ash1 (see below). Decreasing the level of Kdm2 via RNAi in cells led to a dramatic decrease in H2AK118ub levels. Purified recombinant dRAF complex ubiquitinated H2AK118, whereas similarly produced PRC1 did not. Recent experiments suggest that an alternative complex involving Sce and the protein L(3)73Ah, a protein that contains homology to Psc and Su(z)2 in the RING domain region, contributes a large amount of the K2AK118 ubiquitination activity in S2 cells (Lee et al. 2015).
Interestingly, the catalytic activity of Sce is not necessary for repression of canonical PcG target genes (Pengelly et al. 2015). Embryos lacking Sce protein contained no H2AK118ub and misexpressed the PcG target genes Ubx, Abd-B, Antp, and en. However, embryos with an enzymatically inactive Sce protein contained no H2AK118ub, but showed no misexpression of these same PcG target genes. Similarly, clones of larval cells that contained mutated H2A and H2Av that cannot be ubiquitinated showed no misexpression of Hox genes (Pengelly et al. 2015). These data show that the H2AK118ub modification is not required for PcG target gene repression. However, the catalytic activity of Sce is necessary for viability. Previous work has shown that H2AK118 monoubiquitination promotes H3K27 methylation by PRC2 (Kalb et al. 2014); consistent with this, embryos with catalytically inactive Sce have lower H3K27me3 levels than wild-type embryos. Thus, it is likely that H2AK118ub contributes to the robustness of PcG repression. Finally, some PcG target genes (including eve, dac, and pros) are not derepressed in embryos lacking both maternal and zygotic Sce protein (Gutiérrez et al. 2012), reinforcing the idea that not all PcG target genes are regulated in the same way.
Sxc modifies the activity of PRC1 by modifying Ph
The PcG gene sxc encodes the glycosyltransferase Ogt that adds O-linked N-Acetyl glucosamine (O-GlcNAc) to nuclear and cytosolic proteins (Gambetta et al. 2009; Sinclair et al. 2009). Ogt is not required for transcriptional repression of all PcG targets; for example, the Hox gene Abd-B is derepressed in sxc mutant embryos, while the segmentation gene eve is not (Gambetta and Müller 2014). Of the PcG proteins, only Ph has been shown to be modified by Ogt (Gambetta et al. 2009). In the absence of the O-GlcNAc modification, Ph protein still forms PRC1 complexes, but also forms aggregates. A serine/threonine (S/T) region of Ph is the target of Ogt. Interestingly, rescue of a ph null mutant with a Ph protein with the S/T region deleted yields embryos with an Ogt phenotype; i.e., Abd-B is misexpressed, but eve repression is still intact. These data suggest that the PcG phenotype of sxc/ogt mutants can be completely explained by the lack of O-GlcNAc on the S/T region of Ph (Gambetta and Müller 2014). In addition, these data again show that different PcG targets have different requirements for PcG repression.
PRC2
As stated above, the function of PRC2 is to trimethylate lysine 27 on histone H3 (H3K27me3). The core PRC2 complex consists of E(z), Esc, Su(z)12, and Caf1-55 (reviewed in O’Meara and Simon 2012) (Figure 3A). In addition to the core components, the PcG protein Pcl is implicated in PRC2 activity (see references below). Finally, the PRC2-associated proteins Jarid2 and Jing/AEBP2 are homologs of proteins originally identified in mammalian PRC2 complexes (Cao et al. 2002; Li et al. 2010).
Biochemical properties of core PRC2 proteins
E(z) contains a SET domain that is essential for enzymatic function, however, the enzymatic activity of E(z) is very low in the absence of the subunits Esc and Su(z)12 (reviewed in O’Meara and Simon 2012). The catalytic activity of E(z) is absolutely required for Hox gene repression, strongly suggesting that the H3K27me3 mark is required for PcG-mediated repression (Cao et al. 2002; Czermin et al. 2002; Müller et al. 2002). Consistent with this, generating clones of cells with H3 mutated at K27 to either arginine (K27R) or alanine (K27A), so it cannot be methylated, caused derepression of Hox genes in a manner entirely consistent with E(z) mutations (Pengelly et al. 2013; McKay et al. 2015). This result strengthens the consensus view that H3K27me3 is the “hallmark” of PcG-mediated repression.
Esc and the related protein Escl are WD-repeat proteins that fold into seven-bladed β-propellers that provide a scaffold for interactions with protein partners and effectors (Ng et al. 1997; Tie et al. 1998; Wang et al. 2006). Esc is present at its highest levels in midembryogenesis and then rapidly declines. In contrast, the highest levels of Escl are from late embryonic development through the pupal period (Wang et al. 2006; Kurzhals et al. 2008; Ohno et al. 2008). Recombinant PRC2 complexes containing either Esc or Escl both have high activities of H3 methyltransferase activity (Wang et al. 2006; Ohno et al. 2008). The phenotypes of esc, escl, and esc-escl double mutants suggest that esc provides activity early in embryogenesis, and escl provides activity later, consistent with the biochemical evidence (Wang et al. 2006; Kurzhals et al. 2008; Ohno et al. 2008). Available evidence suggests that different domains within the Esc protein bind E(z), the histone core, and H3K27me3 (Xu et al. 2010). Further, binding of Esc to H3K27me3 increases PRC2 activity and may facilitate the spreading of the H3K27me3 domain (Margueron et al. 2009; Tie et al. 2007; Xu et al. 2010).
Su(z)12 is also required for PRC2 formation and activity. A recombinant Su(z)12, Esc, E(z) complex (without Caf1-55) has high H3K27me3 activity in vitro (Ketel et al. 2005; Nekrasov et al. 2005). A cell line was made from cells that contain the Su(z)124 mutation, a nonsense mutation that is thought to make no functional Su(z)12 protein. Interestingly, this cell line has no H3K27me2 or H3K27me3, showing that Su(z)12 is absolutely required for the H3K27me3 mark (Lee et al. 2015). Work with recombinant PRC2 complexes showed that a conserved VEFS domain within Su(z)12 is important for PRC2 assembly and stimulates its enzymatic activity in vitro (Ketel et al. 2005). Recent structural studies on crystallized PRC2 subcomplexes from other species confirm the biochemical experiments and provide additional information as to how Su(z)12 and Esc stimulate the enzymatic activity of E(z) (Jiao and Liu 2015; Justin et al. 2016).
It is possible that Su(z)12 and Esc/Escl function solely in the context of the PRC2 protein complex. These proteins have not been found in other protein complexes and the phenotypes of their mutants are consistent with a dedicated role in PcG repression. In contrast, Caf1-55 is present in other chromatin-modifying complexes (Suganuma et al. 2008). Like Esc and Escl, Caf1-55 is a WD-repeat protein that forms a seven-bladed propeller (Song et al. 2008). Unlike Su(z)12 and Esc/Escl, Caf1-55 is not required for PRC2 histone methyltransferase activity in vitro (Ketel et al. 2005; Nekrasov et al. 2005); its role in PRC2 activity in vivo is unclear (Anderson et al. 2011; Wen et al. 2012).
Jarid2 and Jing (Aebp2)
In addition to homologs of E(z), Esc, Su(z)12, and Caf1-55, mammalian PRC2 contains two other subunits, Jarid2 and Aebp2, which are thought to play roles in stabilizing PRC2 and targeting it to chromatin (Li et al. 2010; Ciferri et al. 2012). Drosophila has homologs of Jarid2 and Aebp2; however, their role in Drosophila development or PRC2 activity is unclear. Jarid2 and Jing (the Drosophila homolog of mammalian Aebp2) were identified as proteins highly enriched in the purification of BioTAP-tagged E(z) protein from Drosophila tissue culture cells, embryos, and larvae, along with the known PRC2 components (Kang et al. 2015). Esc, Su(z)12, E(z), Caf1-55, and Jing also copurified with FLAG-HA-labeled Jarid2 isolated from embryos (Herz et al. 2012). Jing and Jarid2 [as well as E(z) and Su(z)12] were affinity purified from embryo extracts on recombinant H2AK118ub oligonucleosomes (Kalb et al. 2014). Genome-wide ChIP-sequencing (ChIP-seq) experiments in larvae showed that Jarid2 colocalizes with the PRC2 core component Su(z)12 at most sites; however, many PREs at Hox genes are not cobound by Jarid2 in larvae (Herz et al. 2012). Similarly, Jing may act at a subset of PcG targets. Although genome-wide ChIP experiments are not yet available for Jing, genetic experiments suggest that some PcG target genes are more sensitive to jing mutations than others. For example, heterozygosity for jing enhances the partial wing to haltere transformations sometimes observed in flies heterozygous for Pc, Psc, Pcl, or Asx mutations. In contrast, jing heterozygosity does not enhance the extra-sex-combs phenotype in these same genetic backgrounds (Culi et al. 2006).
Pcl
The first indication that Pcl associates with PRC2 were the demonstrations that Pcl interacted with E(z) in a yeast two-hybrid system and in GST-pulldown experiments, and that Pcl co-immunoprecipitated with E(z) from embryo extracts (O’Connell et al. 2001; Tie et al. 2003). In biochemical fractionation experiments, Pcl copurified with E(z) and other PRC2 components in embryos and larvae (Tie et al. 2003; Nekrasov et al. 2007; Kang et al. 2015). In addition, Pcl copurified with BioTAP-tagged E(z) but not BioTAP-tagged Pc (Kang et al. 2015). Pcl completely colocalized with E(z) on polytene chromosomes and colocalized with other PcG proteins to PREs in ChIP experiments (Lonie et al. 1994; Papp and Müller 2006; Nekrasov et al. 2007). Thus, there is strong evidence that Pcl represses PcG targets via interactions with PRC2. However, how does Pcl function? In one study, Pcl was found to be required for high levels of H3K27 trimethylation at target genes in embryos and the authors suggested that Pcl stimulates the histone lysine methyltransferase activity of PRC2 (Nekrasov et al. 2007). In another study, Pcl was required for E(z) recruitment both to polytene chromosomes and to the Ubx PRE in wing imaginal discs (Savla et al. 2008). While these studies suggest that Pcl plays different roles at different stages of development, more work needs to be done to fully understand the role of Pcl in PRC2 recruitment and activity.
Is Scm a Link Between PRC1 and PRC2?
Scm contains several functional conserved domains, including two MBT repeats and a SAM domain (Bornemann et al. 1996). Early biochemical experiments showed a close association of Scm with PRC1 and Scm has often been classified as a PRC1 component. This was based on the observations that Scm could bind to the core PRC1 component Ph in a yeast two-hybrid system (Peterson et al. 1997) and was a substoichiometric component of PRC1 (Shao et al. 1999; Saurin et al. 2001). Scm can interact directly with the PRC1 component Ph via its SAM domain (previously called the SPM domain); mutation or overexpression of the Scm SAM domain disrupts PcG silencing (Peterson et al. 2004). The MBT repeats of Scm bind monomethylated lysine residues, an activity that is also required for PcG silencing of Hox genes (Grimm et al. 2007). Thus, Scm plays an important role in PcG repression.
A recent article provides evidence that Scm closely interacts with PRC1, PRC2, and PhoRC. Scm copurified with both BioTAP-tagged Pc and E(z), suggesting that Scm is tightly associated with both PRC1 and PRC2 (Kang et al. 2015). Further, recombinant Scm could interact with recombinant PRC2 produced using the Sf9 baculovirus system. Consistent with this, isolation of BioTAP-tagged Scm identified PRC1 and PRC2 components as well as PhoRC and other repressive complexes (Kang et al. 2015). Further, Scm and Sfmbt interact directly through their SAM domains (Frey et al. 2016). Thus, Scm could serve as an important functional link between PhoRC, PRC1, and PRC2.
Other PcG Proteins and Complexes
PR-DUB complex
The PcG proteins Asx and Calypso form a protein complex that deubiquitinates H2Aub1 at lysine 119 in vertebrates and lysine 118 in Drosophila (Scheuermann et al. 2010) (Figure 3A). Calypso is the catalytic subunit and is a member of the C-terminal hydrolase (UCH) subclass of deubiquitinating enzymes. The catalytic activity of Calypso in vitro is greatly increased by its association with Asx. In vivo, Calypso protein levels were greatly reduced and H2AK118ub1 levels were greatly increased in Asx mutant embryos. ChIP experiments showed that both Calypso and Asx are present at many PREs. Clones mutant for a catalytically inactive Calypso protein derepressed Ubx in wing discs. Thus, PR-DUB is a bona fide PcG complex; it is bound at PREs and required for Hox gene silencing.
It is curious that the PcG protein Sce monoubiquitinates H2AK118 and PR-DUB deubiquitinates the same residue, especially considering the recent observation that there was no misexpression of Hox genes in embryos with mutant H2A that cannot be ubiquitinated (Pengelly et al. 2015). Although one target of PR-DUB is clearly H2AK118ub1, there may be other relevant targets. Finally, it is worth noting that H2A118ub1 levels vary widely at different PcG target genes (Lee et al. 2015; Kahn et al. 2016), further drawing into question the role of H2A118ub1 in PcG repression.
PhoRC
The PcG gene pho encodes a DNA-binding protein homologous to the mammalian multifunctional transcription factor YY1 (Brown et al. 1998). The Drosophila genome also contains another YY1 homolog encoded by the phol gene. Pho and Phol contain four zinc fingers that are 96 and 80% identical to the zinc fingers of YY1, including all of the amino acids involved in making important DNA contacts. As expected, Pho, Phol, and mammalian YY1 all have the same DNA-binding specificity (Brown et al. 2003). In addition, Pho and Phol share a short, conserved “spacer” domain. PhoRC consists of Pho or Phol bound to Sfmbt (Klymenko et al. 2006) (Figure 3A). Structural analysis showed that the spacer domain of Pho/Phol binds to the MBT repeats of Sfmbt (Alfieri et al. 2013). As noted above, Sfmbt also contains a SAM domain that binds to the SAM domain of Scm (Frey et al. 2016), thus providing a link between the DNA-binding protein Pho and Scm recruitment to the PRE. The physical interactions between the SAM domains of Scm and Ph provide a method for recruitment of PRC1 (Peterson et al. 2004; Kim et al. 2005). Other evidence that PhoRC directly recruits PRC1 comes from the observation that PhoRC copurified with biotinylated Pc from Drosophila embryos; PRC2 components did not copurify (nor did Scm) (Strübbe et al. 2011). Similarly, BioTAP-tagged Pc copurified with Sfmbt (and Scm) but not with PRC2 components (Kang et al. 2015). No PhoRC components were among the top interacting proteins with BioTAP-tagged E(z) (Kang et al. 2015). Other evidence suggests that Pho can directly bind to Ph and Pc in vitro (Mohd-Sarip et al. 2002). These data suggest that PhoRC plays a direct role in recruiting PRC1 to DNA.
Pho and Phol also facilitate PRC2 recruitment. Mutation of Pho-binding sites within a transgene that contains a strong PRE from the Ubx gene led to the loss of both PRC1 and PRC2 on the transgene (Frey et al. 2016), showing that Pho-binding sites are absolutely required for PRE activity. Further, PRC1 and PRC2 were both lost from this same PRE in the endogenous Ubx gene in phol; pho double mutant wing imaginal discs (L. Wang et al. 2004). In yeast two-hybrid and GST-pull-down experiments, Pho directly interacts with E(z) and Esc; Phol directly interacts with Esc (L. Wang et al. 2004). Thus, available data suggest that Pho binding is required for recruitment of both PRC1 and PRC2. An early model suggested that Pho first recruits E(z), which then trimethylates H3K27 to form H3K27me3. The H3K27me3 then recruits Pc via its chromodomain (L. Wang et al. 2004). However, other work suggests that this hierarchical model is not correct (Kahn et al. 2014, 2016; Schuettengruber et al. 2014). In fact, as discussed above, there is strong evidence that PRC1 is recruited directly by the PhoRC complex. In addition, experiments in tissue culture cells that lacked PRC1 or PRC2 components strongly argue that PRC1 recruitment can occur independently of PRC2 (Kahn et al. 2016). Recent work on PcG recruitment in mammals showed that H2A ubiquitination by a variant PRC1 complex was required for PRC2 recruitment (Blackledge et al. 2014). However, as discussed above, this modification is not required for PcG silencing in Drosophila (Pengelly et al. 2015). Currently it is clear that multiple protein–protein interactions lead to the recruitment of PRC1 and PRC2 and that the histone modifications catalyzed by these complexes further stabilize the localization and spreading of these two important PcG repressive complexes.
Mxc and Crm
As discussed above, the majority of PcG genes encode subunits of interacting complexes that repress transcription by altering chromatin structure. The PcG proteins Mxc and Crm act outside of these complexes and their roles are less well understood. The Mxc protein is localized to the histone bodies and may regulate PcG repression indirectly through its effects on histone levels (White et al. 2011). Crm is a chromatin-associated protein that is present mainly during S phase and physically interacts with Mus209 (the Drosophila homolog of PCNA) (Yamamoto et al. 1997); its biochemical role in PcG-mediated repression is at present unknown.
PREs
PcG proteins are recruited to their target genes by a special class of cis-regulatory elements termed PREs. PREs were discovered in transgenes by three different assays. First, transgenes that contained PREs formed new PcG protein binding sites in salivary gland polytene chromosomes (Zink et al. 1991). Second, PREs silenced transgene expression in region-specific ways, and this repression was dependent on PcG proteins (Müller and Bienz 1991; Simon et al. 1993; Chan et al. 1994; Chiang et al. 1995). Third, in a phenomenon called “pairing-sensitive silencing,” PREs repressed the expression of the commonly used P-element reporter gene mini-white (w+mC), and this repression was stronger in flies that had two copies of the P{PRE: w+mC} transgene in proximity to each other (Kassis et al. 1991; Kassis 1994). Because of its simplicity, pairing-sensitive silencing is one of the most commonly used assays for PRE activity. Genome-wide ChIP experiments showed that the PREs characterized in transgenes are strong binding sites for PcG proteins in chromatin (Schwartz et al. 2006).
PREs contain binding sites for multiple proteins
Pho was the first PRE-binding protein identified, and is the only known DNA-binding protein encoded by a gene that, when mutated, gives a PcG phenotype (Brown et al. 1998). Pho-binding sites are required for PRE activity in transgenes (Brown et al. 1998; Fritsch et al. 1999; Shimell et al. 2000; Busturia et al. 2001; Mishra et al. 2001; Fujioka et al. 2008), and also for the activity of a PRE in the endogenous Ubx gene (Kozma et al. 2008). As discussed above, Pho (along with Phol) forms a complex with Sfmbt and plays an important role in PcG complex recruitment. However, Pho-binding sites alone are not sufficient to recruit PcG proteins (Americo et al. 2002). PREs contain binding sites for many DNA-binding proteins (reviewed in Kassis and Brown 2013) (Figure 3B). The sequence GAGAG is enriched in PREs and is bound by two proteins, GAGA factor (GAF) [encoded by the Trithorax-like (Trl) gene] (Farkas et al. 1994) and Pipsqueak (Psq) (Schwendemann and Lehmann 2002). In vitro, GAF facilitates Pho binding to a chromatinized template (Mahmoudi et al. 2003). Psq copurified with FLAG-tagged Pc from Drosophila S2 cells and psq mutations enhance Pc mutations (Huang et al. 2002). Other DNA-binding proteins with target sites in PREs include Sp1-like factor for pairing-sensitive silencing (Spps) (Brown and Kassis 2010), Combgap (Cg) (Ray et al. 2016), Dorsal switch protein 1 (Dsp1) (Déjardin et al. 2005), Grainyhead (Grh), Adh transcription factor 1 (Adf1) (Orsi et al. 2014), Zeste (Z) (Hagstrom et al. 1997), and Fs(1)h. Z and Fs(1)h bind the same consensus binding site (Chang et al. 2007). Mutations of binding sites for many of these proteins attenuate or destroy the activity of the PRE in transgenes (Brown and Kassis 2013), but how these proteins function at PREs is unknown. Many of these proteins can act as transcriptional activators in a context-dependent manner, making it more difficult to discern their function at PREs.
Diversity among PREs
Genome-wide ChIP experiments have identified hundreds to thousands of locations in the genome where binding of Pho, Phol, GAF, Dsp1, Adf1, Z, and Cg proteins overlap with components of PRC1 and PRC2 (Kwong et al. 2008; Oktaba et al. 2008; Orsi et al. 2014; Schuettengruber et al. 2014; Ray et al. 2016). Most of these presumptive PREs are bound by Pho, but the presence of the other PRE-binding proteins varies among sites. Sequence analyses of known PREs showed that while PREs shared a number of consensus binding sequences for PRE-binding proteins, the number, spacing, and order of binding sites varied (Brown and Kassis 2013). Given this, it is perhaps not surprising that computational methods to identify PREs have only been marginally effective (for review see Kassis and Brown 2013).
Several factors have complicated the identification and analysis of PREs using reporter genes and other transgenes in vivo. PRE activity in transgenes is highly dependent on the chromosomal insertion site; this is because PRE activity is influenced by the activities of flanking genes (for examples see Americo et al. 2002; Brown et al. 2005; Cunningham et al. 2010). Thus, when using P-element vectors that insert in the genome in a semirandom manner, many lines must be generated to discover the frequency of PRE activity. PREs generate pairing-sensitive silencing of mini-white at frequencies between ∼25 and 80% in P-element-based vectors (for examples see Americo et al. 2002 and Brown and Kassis 2013). Using ϕ-C31 site-specific integration, PRE activity also varies between insertion sites (Okulski et al. 2011). Further, many PREs are adjacent to, or overlapping with, other regulatory elements. Nevertheless, most, if not all, PREs mediate pairing-sensitive silencing, and PREs from the en, Ubx, and eve genes can substitute for each other in transgene assays (Fujioka et al. 2013; Americo et al. 2002; Cunningham et al. 2010). Similarly, PREs from the gap gene giant can substitute for en PREs in an embryonic reporter transgene (Abed et al. 2013). However, not all PREs behave the same in every situation. For example, two PREs from the en gene, PRE1 and PRE2, behave differently in a ϕ-C31-integrated Ubx-reporter gene (Brown and Kassis 2013). There is also the suggestion that PREs from the Psc/Su(z)2 gene complex may be functionally distinct from other PREs. Although the Psc/Su(z)2 gene complex is in a domain of H3K27me3, these genes are not silenced by this repressive mark but are ubiquitously expressed (Park et al. 2012). Results from transgene experiments suggested that some PREs from the Psc/Su(z)2 gene complex decrease the expression of a reporter gene rather than completely silencing it (Park et al. 2012). Whether this reflects a difference in PRE-strength (i.e., how much PcG protein complex is recruited) vs. a difference in the PcG proteins recruited is unknown.
Genome-wide localization of PcG proteins also shows diversity of binding sites with developmental stage, suggesting stage-specific PREs (Négre et al. 2006; De et al. 2016; Lorberbaum et al. 2016). Studies of PcG protein binding in larvae mutant for the DNA-binding protein Cg suggest that some PREs require Cg function, while others do not (Ray et al. 2016). Further evidence for PRE diversity comes from a study on transformed tissue culture cells that lacked either Su(z)12 (and thus PRC2) or Psc and Su(z)2 (thus PRC1). In these transformed cells, two classes of PREs were evident: (1) those that required PRC1 for PRC2 recruitment, and (2) those that recruited PRC2 in the absence of PRC1 (Kahn et al. 2016). We suggest that while PREs share the core function of recruitment of PcG proteins, the exact DNA-binding proteins and mechanisms involved vary among genes. Thus, PREs have evolved to work within the context of the gene(s) they regulate.
Are PREs also TrxG response elements?
Fragments of DNA that contain PREs have also been shown to mediate gene activation in transgenes under certain conditions and at some chromosomal insertion sites (reviewed in Ringrose and Paro 2004, 2007; Kassis and Brown 2013). Some experiments have shown that PRE/TrxG response element (TRE) functions can be subdivided to different fragments (Tillib et al. 1999). In other cases, the activating and repressing sequences appear to be overlapping (Déjardin and Cavalli 2004; Fujioka et al. 2008). Interestingly, ChIP studies show that Trx binds to PREs (Schuettengruber et al. 2009; Schwartz et al. 2010). What recruits Trx to PREs is unknown. Mutation of Pho-binding sites within an Ubx PRE in a transgene totally abrogated PRC1 and PRC2 binding, but left Trx binding intact (Frey et al. 2016). Thus, it is reasonable to conclude that Trx is recruited to PREs independently of Pho and PcG proteins. While it is generally agreed that transcription through a PRE inactivates its silencing activity (Schmitt et al. 2005; Erokhin et al. 2015), the idea that transcription turns a PRE into a TRE is more controversial. The role of PRE/TRE transcription and their RNA products in PcG and TrxG function are areas of ongoing research (Hekimoglu and Ringrose 2009; Herzog et al. 2014).
What Constitutes Epigenetic Memory of the Repressed State?
At Hox genes, PcG proteins maintain transcriptional repression through many rounds of cell division. This is often referred to as epigenetic memory of the “off” state. But what constitutes epigenetic memory? Beuchle et al. (2001) showed that deletion of Su(z)2-Psc or ph in mitotic clones in imaginal discs led to transcriptional activation of Hox genes; however, when these PcG genes were resupplied, Hox genes again became repressed. Thus, the memory of the off state was not lost in Su(z)2-Psc or ph clones. Thus, transcription does not erase the memory of the off state. In agreement with this, transcription through a PRE in a transgene did not abrogate the H3K27me3 mark or binding of PcG proteins to the PRE (Erokhin et al. 2015). Other PcG genes, including Pc and Scm, were more important for transcriptional memory than transcriptional repression (Beuchle et al. 2001). Recent experiments on PRE-containing transgenes showed that the H3K27me3 mark was inherited at repressed loci, but is diluted by unmodified H3 upon DNA replication and cell division after PRE excision (Coleman and Struhl 2017; Laprell et al. 2017). Thus, although H3K27me3 is one aspect of epigenetic memory, the PRE is absolutely required for stable inheritance of this mark through multiple cell divisions.
Mechanisms of Action of TrxG Proteins
Biochemical studies have revealed that TrxG proteins activate transcription via a wide variety of mechanisms, including the covalent modification of nucleosomal histones, ATP-dependent chromatin-remodeling, chromosome cohesion, and the recruitment of RNA polymerase II (RNA Pol II) to promoters. The remainder of this review is focused on the TrxG proteins that activate transcription and counteract PcG repression by altering chromatin structure.
TrxG Proteins That Covalently Modify Nucleosomes
The cloning and molecular characterization of the TrxG gene trx revealed that it encodes an extremely large, chromatin-associated protein with a SET domain related to those found in other known and presumed regulators of chromatin structure (Mazo et al. 1990; Kuzin et al. 1994; Tripoulas et al. 1994, 1996; Stassen et al. 1995). As discussed above, the SET domain is required for the catalytic activity of histone methyltransferases. Trx also contains four PHD fingers, a domain that mediates interactions between histone methyltransferases and their nucleosome substrates. The sequence of the Trx protein therefore suggested that it maintains heritable states of active transcription by methylating histone tails.
One of the best initial candidates for a histone modification catalyzed by Trx was trimethylation of lysine 4 of histone H3 (H3K4). This histone modification is associated with actively transcribed genes and is present at high levels at active promoters in organisms ranging from yeast to humans. Biochemical studies confirmed that Trx has H3K4 methyltransferase activity in vitro and a point mutation that abolishes this activity fails to complement trx mutations, suggesting that the H3K4 methyltransferase activity of Trx is critical for its function in vivo (Smith et al. 2004).
Trx and Ash2 are subunits of a complex with histone methyltransferase activity
The analyses of H3K4 methyltransferases in budding yeast and humans have provided clues to the function of Trx. A single enzyme, SET1, is responsible for all forms of H3K4 methylation in budding yeast; this histone methyltransferase is a subunit of a protein complex known as Set1C or complex of proteins associated with SET1 (COMPASS) (Miller et al. 2001; Roguev et al. 2001). Humans contain multiple H3K4 methyltransferases that function as subunits of complexes related to COMPASS (Shilatifard 2012; Piunti and Shilatifard 2016).
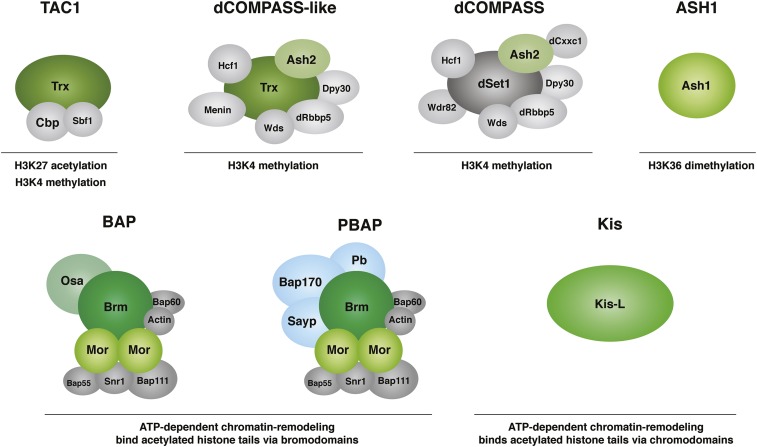
Interestingly, yeast COMPASS and its human counterparts each contain a subunit related to the TrxG protein Ash2. Are Trx and Ash2 subunits of a Drosophila complex related to COMPASS? Trx and two other H3K4 methyltransferases, dSET1 and Trithorax-related (Trr), copurify with Ash2, suggesting the existence of three COMPASS complexes in Drosophila (Mohan et al. 2011). Each of the complexes contains a distinct histone methyltransferase (Trx, dSET1, or TRR), Ash2, and three other common subunits (Rbbp5, Wdr5, and Dpy30) (Figure 4). Studies in other organisms suggest that Ash2 is required for the stability or histone methyltransferase activity of COMPASS (Dehe et al. 2006; Dou et al. 2006).
Figure 4.
TrxG proteins and complexes that affect chromatin structure. (Top panel) TrxG proteins and protein complexes that modify histones. Complexes are shown, along with the histone modification(s) they catalyze. Proteins in green are designated TrxG proteins because mutants have TrxG phenotypes. All other subunits shown were identified as biochemical components of the complexes. (Bottom panel) TrxG proteins and protein complexes involved in chromatin remodeling. Proteins identified as products of genes whose mutants have TrxG phenotypes or that act as suppressors of Pc are in green. Pb, Bap170, and Sayp (blue) are present in PBAP, but not in BAP. All subunits shown in gray are present in both BAP and PBAP.
dSET1 is responsible for the bulk of H3K4 di- and trimethylation in flies (Ardehali et al. 2011; Hallson et al. 2012). Which H3K4 modifications are catalyzed by Trx? Biochemical evidence showed that the SET domain of Trx monomethylates H3K4 in vitro (Tie et al. 2014). Further, these authors show a striking correlation between the genomic distributions of Trx-C (one of the major Trx protein isoforms; see below) and H3K4me1 in S2 cells, located at enhancers and PREs (Tie et al. 2014). A missense mutation that abolishes the histone methyltransferase activity of Trx reduces the level of H3K4me1, but not H3K4me3, in vivo (Tie et al. 2014). Recently another group showed that H3K4me2 (a mark not examined by Tie et al.) is highly correlated with PREs in a genome-wide study in S2 cells (Rickels et al. 2016). Both groups found no correlation between H3K4me3 and PREs. More experiments need to be done to understand the relative importance of H3K4me1 and H3K4me2 and the relationship of these marks to Trx activity.
Trx and dCBP are subunits of a complex with histone acetyltransferase activity
The purification of Trx and associated proteins from Drosophila embryos identified a 1-MDa complex, Trx acetylation complex 1 (TAC1) (Figure 4) that is distinct from COMPASS (Petruk et al. 2001). TAC1 contains three subunits: Trx, dCBP, and the antiphosphatase Sbf1. dCBP, the product of the nejire (nej) gene, is the Drosophila homolog of CREB-binding protein, a mammalian coactivator with histone acetyltransferase activity (Mannervik 2014). Trx, dCBP, and Sbf1 colocalize at many sites on polytene chromosomes (Petruk et al. 2001). TAC1 acetylates nucleosomal histones and nej mutations reduce the expression of the Hox gene Ubx and a trx-dependent reporter gene (Petruk et al. 2001). These findings suggest that Trx activates transcription by promoting both the methylation and acetylation of nucleosomal histones.
Cross talk between histone modifications is relatively common and recent studies have suggested that Trx and dCBP play synergistic roles in transcriptional activation (Tie et al. 2014). dCBP acetylates H3K27 in vitro; this activity is strongly enhanced by the monomethylation of H3K4. Trx, dCBP, H3K4me1, and H3K27Ac colocalize at active enhancers and PREs and the loss of either dCBP or Trx function decreases H3K27 acetylation in vivo. Thus, the monomethylation of H3K4 by Trx appears to directly stimulate the acetylation of H3K27 by dCBP.
We note that TAC1 was purified from Drosophila embryos, and was not found in Drosophila S2 cells (Mohan et al. 2011). In mammalian cells, no similar TAC1 protein complex has been identified, Trx-related proteins are present in COMPASS complexes. Further, TAC1 has not been reconstituted from recombinant proteins. Thus, its existence is somewhat controversial. Nevertheless, Tie et al. (2014) demonstrated that CBP co-immunoprecipitates with Trx from Drosophila embryos. In addition, they identified protein–protein interaction domains in Trx and CBP (Tie et al. 2014). Still, more investigation and characterization of the Trx complexes present in Drosophila embryos is warranted.
The TrxG protein Ash1 methylates lysine 36 of histone H3
The TrxG gene Ash1 encodes a large chromatin-associated protein with a SET domain and a PHD finger (Tripoulas et al. 1996). Early biochemical studies suggested that Ash1, like Trx, methylates H3K4 (Byrd and Shearn 2003) but these findings were challenged by several subsequent observations. Ash1 is not associated with Ash2, a core subunit of H3K4 methyltransferase complexes related to COMPASS (Mohan et al. 2011). Instead, Ash1 (and its human counterparts) dimethylate lysine 36 of histone H3 (H3K36) in vitro (Tanaka et al. 2007; An et al. 2011; Yuan et al. 2011). The replacement of H3K36, but not H3K4, with an alanine blocks histone H3 methylation by Ash1 in vitro (Tanaka et al. 2007), and the loss of ash1 function reduces the level of H3K36me2 on polytene chromosomes (Dorighi and Tamkun 2013). Thus, although both Ash1 and Trx are histone methyltransferases, they modify different lysine residues of histone H3. The functional significance of these modifications is discussed below.
The TrxG gene fs(1)h encodes BET domain proteins that physically interact with Ash1
Biochemical studies recently revealed that Ash1 is physically associated with the products of another TrxG gene, fs(1)h, suggesting that the functions of the two TrxG proteins are intimately related (Kockmann et al. 2013). fs(1)h encodes members of the BET family of transcriptional activators, which contain tandem bromodomains and an extraterminal domain. Bromodomains directly bind acetylated histone tails and are found in a wide variety of transcriptional regulators that interact with chromatin (Ferri et al. 2016). Alternative splicing produces two Fsh isoforms, Fsh-L and Fsh-S, that share a common N-terminal segment containing the bromodomains and extraterminal domain (Haynes et al. 1989). Fsh-L contains a C-terminal extension that is not present in the Fsh-S protein. Both Fsh isoforms copurified with epitope-tagged Ash1, suggesting that the region common to Fsh-S and Fsh-L is required for the interaction with Ash1 (Kockmann et al. 2013). The presence of bromodomains in Fsh-S and Fsh-L suggests that they are readers of histone modifications.
Studies of BRD4, the human counterpart of Fsh, have suggested possible mechanisms by which Fsh activates transcription. BRD4 binds acetylated histones at enhancers and promoters via its bromodomains and promotes the transition to active elongation by recruiting positive transcription elongation factor b (P-TEFb) to promoters (Jang et al. 2005; Yang et al. 2005; Itzen et al. 2014; Jonkers and Lis 2015). BRD4 is also a serine-threonine kinase that can directly phosphorylate the C-terminal domain of the largest subunit of RNA Pol II to promote transcription elongation (Devaiah et al. 2012).
Fsh-S is also a serine-threonine kinase that activates Ubx transcription via a regulatory element located near the promoter (Chang et al. 2007). Using the GAL4-upstream activation sequence system, Chang et al. (2007) found that the expression of elevated levels of Fsh-S, but not Fsh-L, causes homeotic transformations due to the ectopic expression of Hox genes. fs(1)h mutations that cause the loss of both Fsh isoforms lead to homeotic transformations due to the reduced expression of Hox genes. In contrast, individuals homozygous for the fs(1)h17 allele, which specifically reduces the expression of Fsh-L, do not display homeotic transformations (Chang et al. 2007). These findings suggest that Fsh-S, but not Fsh-L, is an activator of Hox transcription. Differences in the genomic distributions of Fsh-S and Fsh-L are consistent with these findings. Fsh-S is associated primarily with enhancers and active promoters, while Fsh-L is associated primarily with insulator elements (Kellner et al. 2013). It is therefore tempting to speculate that Fsh-S, like its human counterpart, binds acetylated histones at enhancers and promoters and activates transcription by promoting the release of paused RNA Pol II.
TrxG Proteins Involved in ATP-Dependent Chromatin Remodeling
Many other TrxG genes encode proteins involved in ATP-dependent chromatin remodeling: one of the main mechanisms used by eukaryotic cells to regulate chromatin structure and gene expression. Chromatin-remodeling reactions (including changes in the structure, assembly, or spacing of nucleosomes) are catalyzed by large proteins or protein complexes that couple ATP hydrolysis to changes in nucleosome structure (Becker and Workman 2013). Dozens of chromatin-remodeling complexes have been identified in many different species, including the yeast SWI/SNF complex and its metazoan counterparts. Each of these complexes contains a catalytic subunit that belongs to the SWI2/SNF2 family of ATPases. By catalyzing ATP-dependent changes in nucleosome structure or positioning, SWI/SNF complexes can regulate the binding of transcription factors and the general transcriptional machinery to DNA.
The TrxG proteins Brm, Mor, and Osa are subunits of SWI/SNF complexes
The cloning and characterization of brm provided the first evidence that TrxG genes activate transcription via ATP-dependent chromatin remodeling (Tamkun et al. 1992). brm encodes a large protein that is highly related to the yeast proteins SWI2/SNF2 and STH1, the ATPase subunits of the SWI/SNF, and RSC chromatin-remodeling complexes, respectively. In addition to a highly conserved ATPase domain, the Brm protein contains the C-terminal bromodomain found in other members of the SWI2/SNF2 subfamily of ATPases. The purification of Brm and associated proteins revealed that Brm is the ATPase subunit of a complex homologous to the yeast SWI/SNF and RSC complexes (Dingwall et al. 1995; Papoulas et al. 1998; Kal et al. 2000).
Multiple complexes related to SWI/SNF with different ATPase subunits are present in most eukaryotes (Kingston and Tamkun 2014). Drosophila initially appeared to be an exception to this rule, since Brm is the only ATPase highly related to SWI2/SNF2 in this organism. Subsequent studies revealed that Brm is the ATPase subunit of two chromatin-remodeling complexes: Brahma-associated protein complex (BAP) and Polybromo-containing BAP complex (PBAP) (Mohrmann et al. 2004) (Figure 4). BAP is more highly related to yeast SWI/SNF, while PBAP is more closely related to yeast RSC. In addition to Brm, the BAP and PBAP complexes contain seven common subunits and a small number of complex-specific subunits (Mohrmann et al. 2004; Chalkley et al. 2008). These findings suggest that BAP and PBAP can be targeted to specific genes via interactions with transcription factors and directly bind acetylated histones via their bromodomain-containing subunits.
Other TrxG genes, including mor and osa, also encode subunits of the Brm complexes (Figure 4). mor encodes a common subunit of BAP and PBAP that is related to the SWI3 subunit of yeast SWI/SNF and RSC (Crosby et al. 1999; Kal et al. 2000). Studies of SWI/SNF complexes in other organisms suggest that Brm and Mor directly interact to form the functional core of the Drosophila Brm complexes (Crosby et al. 1999; Phelan et al. 1999). osa encodes a BAP-specific subunit related to the SWI1 subunit of yeast SWI/SNF (Collins et al. 1999; Vázquez et al. 1999; Kal et al. 2000). Osa contains an ARID domain, a nonspecific DNA-binding domain that may facilitate interactions between BAP and its target genes (Collins et al. 1999). Distinguishing subunits of PBAP include BAP170, Polybromo (Pb), and Sayp (Mohrmann et al. 2004; Chalkley et al. 2008).
Although none of the genes encoding PBAP-specific subunits were identified in genetic screens for TrxG genes, Sayp mutations, like mutations in osa and many other TrxG genes, suppress the homeotic transformations observed in Pc heterozygotes; this finding suggests that both the BAP and PBAP complexes are activators of Hox genes (Chalkley et al. 2008). Both complexes play relatively global roles in transcriptional activation and repression, however; arguing against a restricted role for either complex in Hox regulation or epigenetic inheritance.
The TrxG gene kis encodes a member of the CHD subfamily of ATP-dependent chromatin-remodeling factors
The TrxG gene kis encodes an extremely large protein (Kis-L) that is highly related to human CHD7 and other members of the CHD subfamily of ATP-dependent, chromatin-remodeling factors (Daubresse et al. 1999; Therrien et al. 2000). In addition to highly conserved ATPase domains, Kis-L and CHD7 contain two chromodomains and a BRK domain, a short domain of unknown function that is also found in Brm and its human homologs. CHD7 can increase the accessibility of DNA on the surface of a mononucleosome and catalyze the ATP-dependent sliding of nucleosomes toward the center of a DNA fragment in vitro (Bouazoune and Kingston 2012). These findings strongly suggest that Kis activates the expression of Hox genes by altering the structure or positioning of nucleosomes.
Like Brm, Kis-L is associated with most transcriptionally active regions of polytene chromosomes (Srinivasan et al. 2008). Although Kis-L is not required for the recruitment of RNA Pol II to promoters or promoter clearance, the partial loss of kis function leads to a marked reduction in the level of RNA Pol II and elongation factors associated with polytene chromosomes. These results suggest that Kis promotes an early stage of transcription elongation (Srinivasan et al. 2008). The loss of kis function also causes a significant decrease in the level of Ash1 and Trx associated with polytene chromosomes. This is accompanied by a decrease in H3K36 dimethylation and an increase in repressive H3K27 methylation, suggesting that Kis counteracts PcG repression by promoting the histone modifications catalyzed by Ash1 and Trx (Dorighi and Tamkun 2013).
TrxG Proteins That Activate Transcription via Other Mechanisms
The TrxG genes skd and kto encode mediator subunits
Other TrxG genes, including skd and kto, encode subunits of mediator, a highly conserved 1.5-MDa complex that activates transcription by promoting interactions between gene-specific regulatory proteins, general transcription factors, and RNA Pol II (Treisman 2001; Janody et al. 2003; Allen and Taatjes 2015). Mediator contains >24 subunits organized into head, middle, and tail modules (Plaschka et al. 2016). The head and middle modules directly bind RNA Pol II; the tail module binds a wide variety of gene-specific regulatory proteins. Mediator also contains a dissociable kinase module that can have both positive and negative effects on transcription. The kinase module contains four subunits: CycC, Cdk8, Med12 (Kto), and Med13 (Skd). Although the precise roles of Med12 and Med13 in transcription are not well understood, both proteins are required for the activation of wingless and Notch target genes during Drosophila development (Carrera et al. 2008; Janody and Treisman 2011). Recent studies of Med12 in mammalian cells have suggested that it is required for the formation of cohesin-dependent chromosome loops between enhancers and promoters (Apostolou et al. 2013). These findings suggest that Skd and Kto activate the transcription of Hox genes by promoting interactions between DNA-binding regulatory proteins and RNA Pol II.
A TrxG gene encodes the Rad21 subunit of cohesin
Several recent studies have suggested that a subset of PcG and TrxG proteins activate transcription by altering aspects of chromosome organization above the level of the nucleosome. For example, the TrxG gene verthandi (vtd) was identified in a screen for dominant suppressors of Pc mutations (Kennison and Tamkun 1988) and subsequently shown to encode the Rad21 subunit of cohesin, a ring-shaped protein complex that encircles DNA (Kennison and Tamkun 1988; Hallson et al. 2008). Cohesin is required for proper chromosome segregation during mitosis and meiosis. In interphase cells, cohesin can alter transcription by altering interactions between insulators, enhancers, and promoters, or by other mechanisms (Dorsett and Merkenschlager 2013; Remeseiro and Losada 2013). Cohesin is loaded along chromosomes during interphase by the kollerin complex (Nipped-B and Mau) and removed by the releasin complex (Pds5 and Wapl) (Haering and Gruber 2016). Mutations that lower the level of cohesin on interphase chromosomes give TrxG phenotypes (vtd and Nipped-B) (Hallson et al. 2008). In contrast, a mutation in the wapl gene stabilizes cohesin binding to chromatin and causes a PcG phenotype (Cunningham et al. 2012). Cohesin is bound to many active promoters (Misulovin et al. 2008; Fay et al. 2011). These same promoters are bound by PRC1 (Schaaf et al. 2013). One model posits that cohesin tethers PRC1 to active promoters; increasing cohesin levels causes more PRC1 binding to these active genes, decreasing PRC1 levels at repressed PcG target genes, causing a PcG phenotype. In contrast, reducing cohesin levels reduces the amount of PRC1 bound to active genes, increasing the PRC1 pool available for binding PcG targets, causing a decrease in Hox gene expression and a TrxG phenotype (Schaaf et al. 2013; Dorsett and Kassis 2014). Although an attractive model, more work needs to be done to fully understand the cohesin-PcG-TrxG connection.
Are TrxG Proteins PcG Antirepressors or Global Activators of Transcription?
One of the key issues in the field concerns the roles of TrxG proteins in gene expression and epigenetic inheritance. Do TrxG proteins play relatively global roles in transcription or do they directly antagonize PcG repression to heritably maintain the expression of Hox genes? Many TrxG proteins, including Ash2, Fsh, Brm, other subunits of the BAP and PBAP complexes, and Kis are broadly associated with transcriptionally active regions and their loss of function leads to widespread defects in gene expression (Armstrong et al. 2002; Beltran et al. 2007; Moshkin et al. 2007; Srinivasan et al. 2008; Kellner et al. 2013; Kockmann et al. 2013). Although this does not rule out an important role for these proteins in epigenetic inheritance, their functions are clearly not limited to antagonizing PcG repression.
This situation is more complicated for other TrxG proteins, including Ash1 and Trx. The histone modifications promoted by Ash1 and Trx (including H3K36 dimethylation, H3K4 methylation, and H3K27 acetylation) are also associated with active regions of the genome (Barski et al. 2007; Bell et al. 2007; Heintzman et al. 2007; Wang et al. 2008). However, there is strong evidence that both Ash1 and Trx play specialized roles in antagonizing PcG repression in addition to playing general roles in transcription (O’Meara and Simon 2012). For example, both H3K4 and K36 methylation inhibit the catalytic activity of PRC2 in vitro via an allosteric mechanism (Schmitges et al. 2011; Yuan et al. 2011), and the acetylation of H3K27 directly blocks the methylation of this residue (Tie et al. 2009). These observations are consistent with earlier genetic studies suggesting that Ash1 and Trx counteract Polycomb repression (Klymenko and Müller 2004; Schmitges et al. 2011).
Perspectives
In addition to the Hox genes, there are hundreds of other genes that bind PcG proteins at presumptive PREs and that are covered with the H3K27me3 mark. Location of a gene within an H3K27me3 domain presumably means that PcG proteins regulate their expression, but, in most cases, direct evidence for this is lacking. Available evidence suggests that H3K27me3 “Polycomb domains” contribute to the three-dimensional genome organization of the Drosophila, the subject of an interesting FlyBook review (Schwartz and Cavalli 2017). It is also worth noting that although H3K27me3 is predominantly associated with silent genes, it is also present at rare actively transcribed genes, including the Su(z)2 and Psc gene complex (Park et al. 2012). Thus, H3K27me3, by itself, does not silence transcription.
Here we have reviewed genetic and biochemical studies on PcG and TrxG genes. One thing absolutely clear from genetic, biochemical, and genomic studies is that not all PcG targets are regulated in the same way. The balance of the “on” vs. the off transcriptional states is most likely due to a competition between transcriptional activation (via enhancers and TrxG proteins) and PcG silencing. Removal of PcG proteins from a Drosophila gene will not activate gene expression in the absence of activators. Finally, given the diverse phenotypes caused by different PcG mutations, it is likely that, in addition to working in PRC1 and PRC2, many of these proteins also function independently and/or in other protein complexes. There is still much to learn.
Acknowledgments
J.A. Kassis and J.A. Kennison are supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health.
Footnotes
Communicating editor: E. Furlong
Literature Cited
- Abed J. A., Cheng C. L., Crowell C. R., Madigan L. L., Onwuegbuchu E., et al. , 2013. Mapping Polycomb response element at the Drosophila melanogaster giant locus. G3 (Bethesda) 3: 2297–2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akam M. E., 1987. The molecular basis for metameric pattern in the Drosophila embryo. Development 101: 1–22. [PubMed] [Google Scholar]
- Alfieri C., Gambetta M. C., Matos R., Glatt S., Sehr P., et al. , 2013. Structural basis for targeting the chromatin repressor Sfmbt to Polycomb response elements. Genes Dev. 27: 2367–2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ali J. Y., Bender W., 2004. Cross-regulation among the Polycomb group genes in Drosophila melanogaster. Mol. Cell. Biol. 24: 7737–7747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen B. L., Taatjes D. J., 2015. The mediator complex: a central integrator of transcription. Nat. Rev. Mol. Cell Biol. 16: 155–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Americo J., Whiteley M., Brown J. L., Fujioka M., Jaynes J. B., et al. , 2002. A complex array of DNA-binding proteins required for pairing-sensitive silencing by a Polycomb group response element from the Drosophila engrailed gene. Genetics 160: 1561–1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- An S., Yeo K. J., Jeon Y. H., Song J. J., 2011. Crystal structure of the human histone methyltransferase ASH1L catalytic domain and its implications for the regulatory mechanism. J. Biol. Chem. 286: 8369–8374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson A. E., Karandikar U. C., Pepple K. L., Chen Z. H., Bergmann A., et al. , 2011. The enhancer of Trithorax and Polycomb gene Caf1/p55 is essential for cell survival and patterning in Drosophila development. Development 138: 1957–1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apostolou E., Ferrari F., Walsh R. M., Bar-Nur O., Stadtfeld M., et al. , 2013. Genome-wide chromatin interactions of the Nanog locus in pluripotency, differentiation, and reprogramming. Cell Stem Cell 12: 699–712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ardehali M. B., Mei A., Zobeck K. L., Caron M., Lis J. T., et al. , 2011. Drosophila Set1 is the major histone H3 lysine 4 trimethyltransferase with role in transcription. EMBO J. 30: 2817–2828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armstrong J. A., Papoulas O., Daubresse G., Sperling A. S., Lis J. T., et al. , 2002. The Drosophila BRM complex facilitates global transcription by RNA polymerase II. EMBO J. 21: 5245–5254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bannister A. J., Kouzarides T., 2011. Regulation of chromatin by histone modifications. Cell Res. 21: 381–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bannister A. J., Zegerman P., Partridge J. F., Miska E. A., Thomas J. O., et al. , 2001. Selective recognition of methylated lysine 9 on histon H3 by the HP1 chromo domain. Nature 410: 120–124. [DOI] [PubMed] [Google Scholar]
- Barski A., Cuddapah S., Cui K., Roh T. Y., Schones D. E., et al. , 2007. High-resolution profiling of histone methylations in the human genome. Cell 129: 823–837. [DOI] [PubMed] [Google Scholar]
- Becker P. B., Workman J. L., 2013. Nucleosome remodeling and epigenetics. Cold Spring Harb. Perspect. Biol. 5: a017905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bejarano F., Busturia A., 2004. Function of the Trithorax-like gene during Drosophila development. Dev. Biol. 268: 327–341. [DOI] [PubMed] [Google Scholar]
- Bell O., Wirbelauer C., Hild M., Scharf A. N., Schwaiger M., et al. , 2007. Localized H3K36 methylation states define histone H4K16 acetylation during transcriptional elongation in Drosophila. EMBO J. 26: 4974–4984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beltran S., Angulo M., Pignatelli M., Serras F., Corominas M., 2007. Functional dissection of the ash2 and ash1 transcriptomes provides insights into the transcriptional basis of wing phenotypes and reveals conserved protein interactions. Genome Biol. 8: R67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beuchle D., Struhl G., Müller J., 2001. Polycomb group proteins and heritable silencing of Drosophila Hox genes. Development 128: 993–1004. [DOI] [PubMed] [Google Scholar]
- Bhat K. M., Farkas G., Karch F., Gyurkovics H., Gausz J., et al. , 1996. The GAGA factor is required in the early Drosophila embryo not only for transcriptional regulation but also for nuclear division. Development 122: 1113–1124. [DOI] [PubMed] [Google Scholar]
- Birve A., Sengupta A. K., Beuchle D., Larsson J., Kennison J. A., et al. , 2001. Su(z)12, a novel Drosophila Polycomb group gene that is conserved in vertebrates and plants. Development 128: 3371–3379. [DOI] [PubMed] [Google Scholar]
- Blackledge N. P., Farcas A. M., Kondo T., King H. W., McGouran J. F., et al. , 2014. Variant PRC1 complex-dependent H2A ubiquitylation drives PRC2 recruitment and Polycomb domain formation. Cell 157: 1445–1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloyer S., Cavalli G., Brock H. W., Dura J.-M., 2003. Identification and characterization of polyhomeotic PREs and TREs. Dev. Biol. 261: 426–442. [DOI] [PubMed] [Google Scholar]
- Bornemann D., Miller E., Simon J., 1996. The Drosophila Polycomb group gene Sex comb on midleg (Scm) encodes a zinc finger protein with similarity to polyhomeotic protein. Development 122: 1621–1630. [DOI] [PubMed] [Google Scholar]
- Botas J., Moscoso del Prado J., Garcia-Bellido A., 1982. Gene-dose titration analysis in the search of trans-regulatory genes in Drosophila. EMBO J. 1: 307–310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouazoune K., Kingston R. E., 2012. Chromatin remodeling by the CHD7 protein is impaired by mutations that cause human developmental disorders. Proc. Natl. Acad. Sci. USA 109: 19238–19243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowman S. K., Deaton A. M., Domingues H., Wang P. I., Sadreyev R. I., et al. , 2014. H3K27 modifications define segmental regulatory domains in the Drosophila bithorax complex. Elife 3: e02833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breen T. R., Duncan I. M., 1986. Maternal expression of genes that regulate the bithorax complex of Drosophila melanogaster. Dev. Biol. 118: 442–456. [DOI] [PubMed] [Google Scholar]
- Breen T. R., Harte P. J., 1993. Trithorax regulates multiple homeotic genes in the bithorax and Antennapedia complexes and exerts different tissue-specific, parasegment-specific and promoter-specific effects on each. Development 117: 119–134. [DOI] [PubMed] [Google Scholar]
- Brizuela B. J., Kennison J. A., 1997. The Drosophila homeotic gene moira regulates expression of engrailed and HOM genes in imaginal tissues. Mech. Dev. 65: 209–220. [DOI] [PubMed] [Google Scholar]
- Brizuela B. J., Elfring L., Ballard J., Tamkun J. W., Kennison J. A., 1994. Genetic analysis of the brahma gene of Drosophila melanogaster and polytene chromosome subdivisions 72AB. Genetics 137: 803–813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown J. L., Kassis J. A., 2010. Spps, a Drosophila Sp1/KLF family member, binds to PREs and is required for PRE activity late in development. Development 137: 2597–2602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown J. L., Kassis J. A., 2013. Architectural and functional diversity of Polycomb group response elements in Drosophila. Genetics 195: 407–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown J. L., Mucci D., Whiteley M., Dirksen M. L., Kassis J. A., 1998. The Drosophila Polycomb group gene pleiohomeotic encodes a DNA binding protein with homology to the transcription factor YY1. Mol. Cell 1: 1057–1064. [DOI] [PubMed] [Google Scholar]
- Brown J. L., Fritsch C., Mueller J., Kassis J. A., 2003. The Drosophila pho-like gene encodes a YY1-related DNA binding protein that is redundant with pleiohomeotic in homeotic gene silencing. Development 130: 285–294. [DOI] [PubMed] [Google Scholar]
- Brown J. L., Grau D. J., DeVido S. K., Kassis J. A., 2005. An Sp1/KLF binding site is important for the activity of a Polycomb group response element from the Drosophila engrailed gene. Nucleic Acids Res. 33: 5181–5189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunk B. P., Martin E. C., Adler P. N., 1991. Drosophila genes Posterior sex combs and Suppressor two of zeste encode proteins with homology to the murine bmi-1 oncogene. Nature 353: 351–353. [DOI] [PubMed] [Google Scholar]
- Busturia A., Morata G., 1988. Ectopic expression of homeotic genes caused by the elimination of the Polycomb gene in Drosophila imaginal epidermis. Development 104: 713–720. [DOI] [PubMed] [Google Scholar]
- Busturia A., Lloyd A., Bejarano F., Zavortink M., Xin H., et al. , 2001. The MCP silencer of the Drosophila Abd-B gene requires both Pleohomeotic and GAGA factor for the maintenance of repression. Development 128: 2163–2173. [DOI] [PubMed] [Google Scholar]
- Byrd K. N., Shearn A., 2003. ASH1, a Drosophila trithorax group protein, is required for methylation of lysine 4 residues on histone H3. Proc. Natl. Acad. Sci. USA 100: 11535–11540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao R., Wang L., Wang H., Xia L., Erdjument-Bromage H., et al. , 2002. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298: 1039–1043. [DOI] [PubMed] [Google Scholar]
- Capdevila M. P., Garcia-Bellido A., 1981. Genes involved in the activation of the bithorax complex of Drosophila. Wilhelm Roux’s Arch. Dev. Biol. 190: 339–350. [DOI] [PubMed] [Google Scholar]
- Capdevila M. P., Botas J., Garcia-Bellido A., 1986. Genetic interactions between the Polycomb locus and the Antennapedia and bithorax complexes of Drosophila. Wilhelm Roux’s Arch. Dev. Biol. 195: 417–432. [DOI] [PubMed] [Google Scholar]
- Carrera I., Janody F., Leeds N., Duveau F., Treisman J. E., 2008. Pygopus activates wingless target gene transcription through the mediator complex subunits Med12 and Med13. Proc. Natl. Acad. Sci. USA 105: 6644–6649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalkley G. E., Moshkin Y. M., Langenberg K., Bezstarosti K., Blastyak A., et al. , 2008. The transcriptional coactivator SAYP is a trithorax group signature subunit of the PBAP chromatin remodeling complex. Mol. Cell. Biol. 28: 2920–2929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan C.-S., Rastelli L., Pirrotta V., 1994. A Polycomb response element in the Ubx gene that determines an epigenetically inherited state of repression. EMBO J. 13: 2553–2564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang Y.-L., Peng Y.-H., Pan I.-C., Sun D.-S., King B., et al. , 2001. Essential role of Drosophila Hdac1 in homeotic gene silencing. Proc. Natl. Acad. Sci. USA 98: 9730–9735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang Y. L., King B., Lin S. C., Kennison J. A., Huang D. H., 2007. A double-bromodomain protein, FSH-S, activates the homeotic gene ultrabithorax through a critical promoter-proximal region. Mol. Cell. Biol. 27: 5486–5498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang A., O’Connor M. B., Paro R., Simon J., Bender W., 1995. Discrete Polycomb-binding sites in each parasegmental domain of the bithorax complex. Development 121: 1681–1689. [DOI] [PubMed] [Google Scholar]
- Ciferri C., Lander G. C., Maiolica A., Herzog F., Aebersold R., et al. , 2012. Molecular architecture of human polycomb repressive complex 2. Elife 1: e00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleman R. T., Struhl G., 2017. Causal role for inheritance of H3K27me3 in maintaining the OFF state of a Drosophila HOX gene. Science 356: eaai8236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins R. T., Furukawa T., Tanese N., Treisman J. E., 1999. Osa associates with the brahma chromatin remodeling complex and promotes the activation of some target genes. EMBO J. 18: 7029–7040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crosby M. A., Miller C., Alon T., Watson K. L., Verrijzer C. P., et al. , 1999. The trithorax group gene moira encodes a brahma-associated putative chromatin-remodeling factor in Drosophila melanogaster. Mol. Cell. Biol. 19: 1159–1170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Culi J., Aroca P., Modolell J., Mann R. S., 2006. jing is required for wing development and to establish the proximo-distal axis of the leg in Drosophila melanogaster. Genetics 173: 255–266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham M. D., Brown J. L., Kassis J. A., 2010. Characterization of the Polycomb group response elements of the Drosophila melanogaster invected locus. Mol. Cell Biol. 30: 820–828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham M. D., Gause M., Cheng Y., Noyes A., Dorsett D., et al. , 2012. Wapl antagonizes cohesin binding and promotes Polycomb-group silencing in Drosophila. Development 139: 4172–4179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czermin B., Melfi R., McCabe D., Seitz V., Imhof A., et al. , 2002. Drosophila enhancer of zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal Polycomb sites. Cell 111: 185–196. [DOI] [PubMed] [Google Scholar]
- Daubresse G., Deuring R., Moore L., Papoulas O., Zakrajsek I., et al. , 1999. The Drosophila kismet gene is related to chromatin-remodeling factors and is required for both segmentation and segment identity. Development 126: 1175–1187. [DOI] [PubMed] [Google Scholar]
- De S., Mitra A., Cheng Y., Pfeifer K., Kassis J. A., 2016. Formation of a Polycomb-domain in the absence of strong Polycomb Response Elements. PLoS Genet. 12: e006200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeCamillis M., Cheng N., Pierre D., Brock H. W., 1992. The polyhomeotic gene of Drosophila encodes a chromatin protein that shares polytene chromosome-binding sites with Polycomb. Genes Dev. 6: 223–232. [DOI] [PubMed] [Google Scholar]
- Dehe P. M., Dichtl B., Schaft D., Roguev A., Pamblanco M., et al. , 2006. Protein interactions within the Set1 complex and their roles in the regulation of histone 3 lysine 4 methylation. J. Biol. Chem. 281: 35404–35412. [DOI] [PubMed] [Google Scholar]
- Déjardin J., Cavalli G., 2004. Chromatin inheritance upon zeste-mediated brahma recruitment at a minimal cellular memory module. EMBO J. 23: 857–868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Déjardin J., Rappailles A., Cuvier O., Grimaud C., Decoville M., et al. , 2005. Recruitment of Drosophila Polycomb group proteins to chromatin by DSP1. Nature 434: 533–538. [DOI] [PubMed] [Google Scholar]
- Denell R. E., Frederick R. D., 1983. Homoeosis in Drosophila: a description of the Polycomb lethal syndrome. Dev. Biol. 97: 34–47. [DOI] [PubMed] [Google Scholar]
- Devaiah B. N., Lewis B. A., Cherman N., Hewitt M. C., Albrecht B. K., et al. , 2012. BRD4 is an atypical kinase that phosphorylates serine2 of the RNA polymerase II carboxy-terminal domain. Proc. Natl. Acad. Sci. USA 109: 6927–6932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Digan M. E., Haynes S. R., Mozer B. A., Dawid I. B., Forquignon F., et al. , 1986. Genetic and molecular analysis of fs(1)h, a maternal effect homeotic gene in Drosophila. Dev. Biol. 114: 161–169. [DOI] [PubMed] [Google Scholar]
- Dingwall A. K., Beek S. J., McCallum C. M., Tamkun J. W., Kalpana G. V., et al. , 1995. The Drosophila snr1 and brm proteins are related to yeast SWI/SNF proteins and are components of a large protein complex. Mol. Biol. Cell 6: 777–791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Docquier F., Saget O., Forquignon F., Randsholt N. B., Santamaria P., 1996. The multi sex combs gene of Drosophila melanogaster is required for proliferation of the germline. Wilhelm Roux’s Arch. Dev. Biol. 205: 203–214. [DOI] [PubMed] [Google Scholar]
- Dorighi K. M., Tamkun J. W., 2013. The trithorax group proteins Kismet and ASH1 promote H3K36 dimethylation to counteract Polycomb group repression in Drosophila. Development 140: 4182–4192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorn R., Krauss V., Reuter G., Saumweber H., 1993. The enhancer of position-effect variegation of Drosophila, E(var)3–93D, codes for a chromatin protein containing a conserved domain common to several transcriptional regulators. Proc. Natl. Acad. Sci. USA 90: 11376–11380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorsett D., Kassis J. A., 2014. Checks and balances between cohesin and Polycomb in gene silencing and transcription. Curr. Biol. 24: R535–R539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorsett D., Merkenschlager M., 2013. Cohesin at active genes: a unifying theme for cohesin and gene expression from model organisms to humans. Curr. Opin. Cell Biol. 25: 327–333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dou Y., Milne T. A., Ruthenburg A. J., Lee S., Lee J. W., et al. , 2006. Regulation of MLL1 H3K4 methyltransferase activity by its core components. Nat. Struct. Mol. Biol. 13: 713–719. [DOI] [PubMed] [Google Scholar]
- Duncan I. M., 1982. Polycomblike: a gene that appears to be required for the normal expression of the bithorax and Antennapedia gene complexes of Drosophila melanogaster. Genetics 102: 49–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan I. M., 1986. Control of bithorax complex functions by the segmentation gene fushi tarazu of D. melanogaster. Cell 47: 297–309. [DOI] [PubMed] [Google Scholar]
- Duncan I. M., 1987. The bithorax complex. Annu. Rev. Genet. 21: 285–319. [DOI] [PubMed] [Google Scholar]
- Dura J.-M., Ingham P. W., 1988. Tissue- and stage-specific control of homeotic and segmentation gene expression in Drosophila embryos by the polyhomeotic gene. Development 103: 733–741. [DOI] [PubMed] [Google Scholar]
- Dura J.-M., Brock H. W., Santamaria P., 1985. Polyhomeotic: a gene of Drosophila melanogaster required for correct expression of segmental identity. Mol. Gen. Genet. 198: 213–220. [DOI] [PubMed] [Google Scholar]
- Dura J.-M., Randsholt N. B., Deatrick J., Erk I., Santamaria P., et al. , 1987. A complex genetic locus, polyhomeotic, is required for segmental specification and epidermal development in D. melanogaster. Cell 51: 829–839. [DOI] [PubMed] [Google Scholar]
- Dura J.-M., Deatrick J., Randsholt N. B., Brock H. W., Santamaria P., 1988. Maternal and zygotic requirement for the polyhomeotic complex genetic locus in Drosophila. Wilhelm Roux’s Arch. Dev. Biol. 197: 239–246. [DOI] [PubMed] [Google Scholar]
- Eissenberg J. C., Tharappel J. C., Foster-Hartnett D. M., Harnett T., Ngan V., et al. , 1990. Mutation in a heterochromatin-specific chromosomal protein is associated with suppression of position-effect variegation in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 87: 9923–9927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elfring L. K., Daniel C., Papoulas O., Deuring R., Sarte M., et al. , 1998. Genetic analysis of brahma: the Drosophila homolog of the yeast chromatin remodeling factor SWI2/SNF2. Genetics 148: 251–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emmons R. B., Genetti H., Filandrinos S., Lokere J., Wu C.-t., 2009. Molecular genetic analysis of suppressor 2 of zeste identifies key functional domains. Genetics 182: 999–1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erokhin M., Elizar’ev P., Parshikov A., Schedl P., Georgiev P., et al. , 2015. Transcriptional read-through is not sufficient to induce an epigenetic switch in the silencing activity of Polycomb response elements. Proc. Natl. Acad. Sci. USA 112: 14930–14935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farkas G., Gausz J., Galloni M., Reuter G., Gyurkovics H., et al. , 1994. The Trithorax-like gene encodes the Drosophila GAGA factor. Nature 371: 806–808. [DOI] [PubMed] [Google Scholar]
- Fauvarque M.-O., Zuber V., Dura J.-M., 1995. Regulation of polyhomeotic transcription may involve local changes in chromatin activity in Drosophila. Mech. Dev. 52: 343–355. [DOI] [PubMed] [Google Scholar]
- Fauvarque M.-O., Laurenti P., Boivin A., Bloyer S., Griffin-Shea R., et al. , 2001. Dominant modifiers of the polyhomeotic extra-sex-combs phenotype induced by marked P element insertional mutagenesis in Drosophila. Genet. Res. 78: 137–148. [DOI] [PubMed] [Google Scholar]
- Fay A., Misulovin Z., Li J., Schaaf C. A., Gause M., et al. , 2011. Cohesin selectively binds and regulates genes with paused RNA polymerase. Curr. Biol. 21: 1624–1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferri E., Petosa C., McKenna C. E., 2016. Bromodomains: structure, function and pharmacology of inhibition. Biochem. Pharmacol. 106: 1–18. [DOI] [PubMed] [Google Scholar]
- Florence B., McGinnis W., 1998. A genetic screen of the Drosophila X chromosome for mutations that modify Deformed function. Genetics 150: 1497–1511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forquignon F., 1981. A maternal effect mutation leading to deficiencies of organs and homeotic transformations in the adults of Drosophila. Wilhelm Roux’s Arch. Dev. Biol. 190: 132–138. [DOI] [PubMed] [Google Scholar]
- Francis N. J., Saurin A. J., Shao Z., Kingston R. E., 2001. Reconstitution of a functional core Polycomb repressive complex. Mol. Cell 8: 545–556. [DOI] [PubMed] [Google Scholar]
- Francis N. J., Kingston R. E., Woodcock C. L., 2004. Chromatin compaction by a Polycomb group protein complex. Science 306: 1574–1577. [DOI] [PubMed] [Google Scholar]
- Franke A., DeCamillis M., Zink D., Cheng N., Brock H. W., et al. , 1992. Polycomb and polyhomeotic are constituents of a multimeric protein complex in chromatin of Drosophila melanogaster. EMBO J. 11: 2941–2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frey F., Sheahan T., Finkl K., Stoehr G., Mann M., et al. , 2016. Molecular basis of PRC1 targeting to Polycomb response elements by PhoRC. Genes Dev. 30: 1116–1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritsch C., Brown J. L., Kassis J. A., Müller J., 1999. The DNA-binding Polycomb group protein pleiohomeotic mediates silencing of a Drosophila homeotic gene. Development 126: 3905–3913. [DOI] [PubMed] [Google Scholar]
- Fritsch C., Beuchle D., Müller J., 2003. Molecular and genetic analysis of the Polycomb group gene sex combs extra/ring in Drosophila. Mech. Dev. 120: 949–954. [DOI] [PubMed] [Google Scholar]
- Fujioka M., Yusibova G. L., Zhou J., Jaynes J. B., 2008. The DNA-binding Polycomb-group protein pleiohomeotic maintains both active and repressed transcriptional states through a single site. Development 135: 4131–4139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujioka M., Sun G. Z., Jaynes J. B., 2013. The Drosophila even insulator Homie promotes eve expression and protects the adjacent gene from repression by Polycomb spreading. PLoS Genet. 9: e1003883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gambetta M. C., Müller J., 2014. O-GlcNAcylation prevents aggregation of the Polycomb group repressor polyhomeotic. Dev. Cell 31: 1–11. [DOI] [PubMed] [Google Scholar]
- Gambetta M. C., Oktaba K., Müller J., 2009. Essential role of the glycosyltransferase Sxc/Ogt in Polycomb repression. Science 325: 93–95. [DOI] [PubMed] [Google Scholar]
- Garcia B. A., Hake S. B., Diaz R. L., Kauer M., Morris S. A., et al. , 2007. Organismal differences in post-translational modifications in histones H3 and H4. J. Biol. Chem. 282: 7641–7655. [DOI] [PubMed] [Google Scholar]
- Garcia-Bellido A., Capdevila M. P., 1978. The initiation and maintenance of gene activity in a developmental pathway of Drosophila, pp. 3–21 in Clonal Basis of Development, edited by Subtelny S., Sussex I. M. Academic Press, London. [Google Scholar]
- Gatti M., Baker B. S., 1989. Genes controlling essential cell-cycle functions in Drosophila melanogaster. Genes Dev. 3: 438–453. [DOI] [PubMed] [Google Scholar]
- Gaytán A., Gutiérrez L., Fritsch C., Papp B., Beuchle D., et al. , 2007. A genetic screen identifies novel Polycomb group genes in Drosophila. Genetics 176: 2099–2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gehring W. J., 1970. A recessive lethal (l(4)29) with a homeotic effect in D. melanogaster. Drosoph. Inf. Serv. 45: 103. [Google Scholar]
- Gellon G., Harding K. W., McGinnis N., Martin M. M., McGinnis W., 1997. A genetic screen for modifiers of Deformed homeotic function identifies novel genes required for head development. Development 124: 3321–3331. [DOI] [PubMed] [Google Scholar]
- Gildea J. J., Lopez R., Shearn A., 2000. A screen for new trithorax group genes identified little imaginal discs, the Drosophila melanogaster homologue of human retinoblastoma binding protein 2. Genetics 156: 645–663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm C., Alanso A. G. D., Rybin V., Steuerwald U., Ly-Hartig N., et al. , 2007. Structural and functional analyses of methyl-lysine binding by the malignant brain tumour repeat protein sex comb on midleg. EMBO Rep. 8: 1031–1037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossniklaus U., Paro R., 2014. Transcriptional silencing by Polycomb-group proteins. Cold Spring Harb. Perspect. Biol. 6: a019331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutiérrez L., Zurita M., Kennison J. A., Vázquez M., 2003. The Drosophila trithorax group gene tonalli (tna) interacts genetically with the brahma remodeling complex and encodes an SP-RING finger protein. Development 130: 343–354. [DOI] [PubMed] [Google Scholar]
- Gutiérrez L., Oktaba K., Scheuermann J. C., Gambetta M. C., Ly-Hartig N., et al. , 2012. The role of the histone H2A ubiquitinase Sce in Polycomb repression. Development 139: 117–127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haering C. H., Gruber S., 2016. SnapShot: SMC protein complexes, part II. Cell 164: 818–819. [DOI] [PubMed] [Google Scholar]
- Hagstrom K., Müller M., Schedl P., 1997. A Polycomb and GAGA dependent silencer adjoins the Fab-7 boundary in the Drosophila bithorax complex. Genetics 146: 1365–1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallson G., Syrzycka M., Beck S. A., Kennison J. A., Dorsett D., et al. , 2008. The Drosophila cohesin subunit Rad21 is a trithorax group (trxG) protein. Proc. Natl. Acad. Sci. USA 105: 12405–12410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallson G., Hollebakken R. E., Li T., Syrzycka M., Kim I., et al. , 2012. dSet1 is the main H3K4 di- and tri-methyltransferase throughout Drosophila development. Genetics 190: 91–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harding K., Levine M., 1988. Gap genes define the limits of Antennapedia and bithorax gene expression during early development in Drosophila. EMBO J. 7: 205–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harding K. W., Gellon G., McGinnis N., McGinnis W., 1995. A screen for modifiers of Deformed function in Drosophila. Genetics 140: 1339–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haynes S. R., Mozer B. A., Bhatia-Dey N., Dawid I. B., 1989. The Drosophila fsh locus, a maternal effect homeotic gene, encodes apparent membrane proteins. Dev. Biol. 134: 246–257. [DOI] [PubMed] [Google Scholar]
- Haynie J. L., 1983. The maternal and zygotic roles of the gene Polycomb in embryonic determination in Drosophila melanogaster. Dev. Biol. 100: 399–411. [DOI] [PubMed] [Google Scholar]
- Heintzman N. D., Stuart R. K., Hon G., Fu Y., Ching C. W., et al. , 2007. Distinct and predictive chromatin signatures of transcriptional promoters and enhancers in the human genome. Nat. Genet. 39: 311–318. [DOI] [PubMed] [Google Scholar]
- Hekimoglu B., Ringrose L., 2009. Non-coding RNAs in Polycomb/Trithorax regulation. RNA Biol. 6: 129–137. [DOI] [PubMed] [Google Scholar]
- Herz H.-M., Mohan M., Garrett A. S., Miller C., Castro D., et al. , 2012. Polycomb repressive complex 2-dependent and -independent functions of Jarid2 in transcriptional regulation in Drosophila. Mol. Cell. Biol. 32: 1683–1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herzog V. A., Lempradi A., Trupke J., Okulski H., Altmutter C., et al. , 2014. A strand-specific switch in noncoding transcription switches the function of a Polycomb/Trithorax response element. Nat. Genet. 46: 973–981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodgson J. W., Cheng N. N., Sinclair D. A. R., Kyba M., Randsholt N. B., et al. , 1997. The polyhomeotic locus of Drosophila melanogaster is transcriptionally and post-transcriptionally regulated during embryogenesis. Mech. Dev. 66: 69–81. [DOI] [PubMed] [Google Scholar]
- Huang D. H., Chang Y. L., Yang C. C., Pan I. C., King B., 2002. Pisqueak encodes a factor essential for sequence-specific targeting of a Polycomb group protein complex. Mol. Cell. Biol. 17: 6261–6271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingham P. W., 1981. Trithorax: a new homoeotic mutation of Drosophila melanogaster. II. The role of trx+ after embryogenesis. Wilhelm Roux’s Arch. Dev. Biol. 190: 365–369. [DOI] [PubMed] [Google Scholar]
- Ingham P. W., 1983. Differential expression of bithorax complex genes in the absence of the extra sex combs and trithorax genes. Nature 306: 591–593. [DOI] [PubMed] [Google Scholar]
- Ingham P. W., 1984. A gene that regulates the bithorax complex differentially in larval and adult cells of Drosophila. Cell 37: 815–823. [DOI] [PubMed] [Google Scholar]
- Ingham P. W., 1985. A clonal analysis of the requirement for the trithorax gene in the diversification of segments in Drosophila. J. Embryol. Exp. Morphol. 89: 349–365. [PubMed] [Google Scholar]
- Ingham P. W., 1988. The molecular genetics of embryonic pattern formation in Drosophila. Nature 335: 25–34. [DOI] [PubMed] [Google Scholar]
- Ingham P. W., Martinez-Arias A., 1986. The correct activation of Antennapedia and bithorax complex genes reguires the fushi tarazu gene. Nature 324: 592–597. [DOI] [PubMed] [Google Scholar]
- Ingham P. W., Whittle J. R. S., 1980. Trithorax: a new homoeotic mutation of Drosophila melanogaster causing transformations of abdominal and thoracic imaginal segments. I. Putative role during embryogenesis. Mol. Gen. Genet. 179: 607–614. [Google Scholar]
- Iovino N., Ciabrelli F., Cavalli G., 2013. PRC2 controls Drosophila oocyte cell fate by repressing cell cycle genes. Dev. Cell 26: 431–439. [DOI] [PubMed] [Google Scholar]
- Itzen F., Greifenberg A. K., Bosken C. A., Geyer M., 2014. Brd4 activates P-TEFb for RNA polymerase II CTD phosphorylation. Nucleic Acids Res. 42: 7577–7590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang M. K., Mochizuki K., Zhou M., Jeong H. S., Brady J. N., et al. , 2005. The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol. Cell 19: 523–534. [DOI] [PubMed] [Google Scholar]
- Janody F., Treisman J. E., 2011. Requirements for mediator complex subunits distinguish three classes of notch target genes at the Drosophila wing margin. Dev. Dyn. 240: 2051–2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janody F., Martirosyan Z., Benlali A., Treisman J. E., 2003. Two subunits of the Drosophila mediator complex act together to control cell affinity. Development 130: 3691–3701. [DOI] [PubMed] [Google Scholar]
- Jiao L., Liu X., 2015. Structural basis of histone H3K27 trimethylation by an active polycomb repressive complex 2. Science 350: aac4383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones R. S., Gelbart W. M., 1990. Genetic analysis of the Enhancer of zeste locus and its role in gene regulation in Drosophila melanogaster. Genetics 126: 185–199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jonkers I., Lis J. T., 2015. Getting up to speed with transcription elongation by RNA polymerase II. Nat. Rev. Mol. Cell Biol. 16: 167–177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jürgens G., 1985. A group of genes controlling the spatial expression of the bithorax complex in Drosophila. Nature 316: 153–155. [Google Scholar]
- Justin N., Zhang Y., Tarricone C., Martin S. R., Chen S., et al. , 2016. Structural basis of oncogenic histone H3K27M inhibition of human polycomb repressive complex 2. Nature Com 7: 11316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn T. G., Stenberg P., Pirrotta V., Schwartz Y. B., 2014. Combinatorial interactions are required for the efficient recruitment of Pho repressive complex (PhoRC) to Polycomb response elements. PLoS Genet. 10: e1004495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahn T. G., Dorafshan E., Schultheis D., Zare A., Stenberg P., et al. , 2016. Interdependence of PRC1 and PRC2 for recruitment to Polycomb response elements. Nucleic Acids Res. 44: 10132–10149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kal A. J., Mahmoudi T., Zak N. B., Verrijzer C. P., 2000. The Drosophila brahma complex is an essential coactivator for the trithorax group protein zeste. Genes Dev. 14: 1058–1071. [PMC free article] [PubMed] [Google Scholar]
- Kalb R., Latwiel S., Baymaz H. I., Jansen P. W. T. C., Müller C. W., et al. , 2014. Histone H2A monoubiquitination promotes histone H3 methylation in Polycomb repression. Nat. Struct. Mol. Biol. 21: 569–571. [DOI] [PubMed] [Google Scholar]
- Kalisch W.-E., Rasmuson B., 1974. Changes of zeste phenotype induced by autosomal mutations in Drosophila melanogaster. Hereditas 78: 97–104. [DOI] [PubMed] [Google Scholar]
- Kang H., McElroy K. A., Jung Y. L., Alekseyenko A. A., Zee B. M., et al. , 2015. Sex comb on midleg (Scm) is a functional link between PcG-repressive complexes in Drosophila. Genes Dev. 29: 1136–1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassis J. A., 1994. Unusual properties of regulatory DNA from the Drosophila engrailed gene – 3 pairing-sensitive sites within a 1.6-kb region. Genetics 136: 1025–1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassis J. A., Brown J. L., 2013. Polycomb group response element in Drosophila and vertebrates. Adv. Genet. 81: 83–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassis J. A., VanSickle E. P., Sensabaugh S. M., 1991. A fragment of engrailed regulatory DNA can mediate transvection of the white gene in Drosophila. Genetics 128: 751–761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehle J., Beuchle D., Treuheit S., Christen B., Kennison J. A., et al. , 1998. dMi-2, a hunchback-interacting protein that functions in Polycomb repression. Science 282: 1897–1900. [DOI] [PubMed] [Google Scholar]
- Kellner W. A., Van Bortle K., Li L., Ramos E., Takenaka N., et al. , 2013. Distinct isoforms of the Drosophila Brd4 homologue are present at enhancers, promoters and insulator sites. Nucleic Acids Res. 41: 9274–9283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennison J. A., 1993. Transcriptional activation of Drosophila homeotic genes from distant regulatory elements. Trends Genet. 9: 75–79. [DOI] [PubMed] [Google Scholar]
- Kennison J. A., 1995. The Polycomb and trithorax group proteins of Drosophila: trans- regulators of homeotic gene function. Annu. Rev. Genet. 29: 289–303. [DOI] [PubMed] [Google Scholar]
- Kennison J. A., 2004. Introduction to Trx-G and Pc-G genes. Methods Enzymol. 377: 61–70. [DOI] [PubMed] [Google Scholar]
- Kennison J. A., Tamkun J. W., 1988. Dosage-dependent modifiers of polycomb and antennapedia mutations in Drosophila. Proc. Natl. Acad. Sci. USA 85: 8136–8140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennison J. A., Tamkun J. W., 1992. Trans-regulation of homeotic genes in Drosophila. New Biol. 4: 91–96. [PubMed] [Google Scholar]
- Ketel C. S., Andersen E. F., Vargas M. L., Sug J., Strome S., et al. , 2005. Subunit contributions to histone methyltransferase activity of fly and worm Polycomb group complexes. Mol. Cell. Biol. 25: 6857–6868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim C. A., Gingery M., Pilpa R. M., Bowie J. U., 2002. The SAM domain of polyhomeotic forms a helical polymer. Nat. Struct. Biol. 9: 453–457. [DOI] [PubMed] [Google Scholar]
- Kim C. A., Sawaya M. R., Cascio D., Kim W., Bowie J. U., 2005. Structural organization of a sex-comb-on-midleg/polyhomeotic co-polymer. J. Biol. Chem. 280: 27769–27775. [DOI] [PubMed] [Google Scholar]
- King I. F. G., Emmons R. B., Francis N. J., Wild B., Müller J., et al. , 2005. Analysis of a Polycomb group protein defines regions that link repressive activity on nucleosomal templates to in vivo function. Mol. Cell. Biol. 25: 6578–6591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kingston R. E., Tamkun J. W., 2014. Transcriptional regulation by trithorax-group proteins. Cold Spring Harb. Perspect. Biol. 6: a019349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klymenko T., Müller J., 2004. The histone methyltransferases Trithorax and Ash1 prevent transcriptional silencing by Polycomb group proteins. EMBO Rep. 5: 373–377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klymenko T., Papp B., Fischle W., Kocher T., Schelder M., et al. , 2006. A Polycomb group protein complex with sequence-specific DNA-binding and selective methyl-lysine-binding activities. Genes Dev. 20: 1110–1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kockmann T., Gerstung M., Schlumpf T., Xhinzhou Z., Hess D., et al. , 2013. The BET protein FSH functionally interacts with ASH1 to orchestrate global gene activity in Drosophila. Genome Biol. 14: R18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozma G., Bender W., Sipos L., 2008. Replacement of a Drosophila Polycomb response element core, and in situ analysis of its DNA motifs. Mol. Genet. Genomics 279: 595–603. [DOI] [PubMed] [Google Scholar]
- Kwong C., Adryan B., Bell I., Meadows L., Russell S., et al. , 2008. Stability and dynamics of Polycomb target sites in Drosophila development. PLoS Genet. 4: e1000178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurzhals R. L., Tie F., Stratton C. A., Harte P. J., 2008. Drosophila ESC-like can substitute for ESC and becomes required for Polycomb silencing if ESC is absent. Dev. Biol. 313: 293–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuzin B., Tillib S., Sedkov Y., Mizrokhi L., Mazo A., 1994. The Drosophila trithorax gene encodes a chromosomal protein and directly regulates the region-specific homeotic gene fork head. Genes Dev. 8: 2478–2490. [DOI] [PubMed] [Google Scholar]
- Lachner M., Sengupta R., Schotta G., Jenuwein T., 2004. Trilogies of histone lysine methylation as epigenetic landmarks of the eukaryotic genome. Cold Spring Harb. Symp. Quant. Biol. 69: 209–218. [DOI] [PubMed] [Google Scholar]
- Lagarou A., Mohd-Sarip A., Moshkin Y. M., Chalkley G. E., Bezstarosti K., et al. , 2008. dKDM2 couples histone H2A ubiquitylation to histone H3 demethylation during Polycomb group silencing. Genes Dev. 22: 2799–2810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laprell F., Finkl K., Müller J., 2017. Propagation of Polycomb-repressed chromatin requires sequence-specific recruitment to DNA. Science 356: 85–88. [DOI] [PubMed] [Google Scholar]
- Lawrence P. A., Johnston P., Struhl G., 1983. Different requirements for homeotic genes in the soma and germ line of Drosophila. Cell 35: 27–34. [DOI] [PubMed] [Google Scholar]
- Lee H.-G., Kahn T. G., Simcox A., Schwartz Y. B., Pirrotta V., 2015. Genome-wide activities of Polycomb complexes control pervasive transcription. Genome Res. 25: 1170–1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis E. B., 1968. Genetic control of developmental pathways in Drosophila melanogaster, pp. 96–97 in Proceedings of the Twelfth International Congress of Genetics, Vol. 2, edited by Oshima C. Science Council of Japan, Tokyo. [Google Scholar]
- Lewis E. B., 1978. A gene complex controlling segmentation in Drosophila. Nature 276: 565–570. [DOI] [PubMed] [Google Scholar]
- Lewis P. H., 1947. New mutants. Drosoph. Inf. Serv. 21: 69. [Google Scholar]
- Li G., Margueron R., Ku M., Chambon P., Berstein B. E., et al. , 2010. Jarid2 and PRC2, partners in regulating gene expression. Genes Dev. 24: 368–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo S. M., Ahuja N. K., Francis N. J., 2009. Polycomb group protein suppressor 2 of zeste is a functional homolog of Posterior sex combs. Mol. Cell. Biol. 29: 515–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loncle N., Boube M., Joulia L., Boschiero C., Werner M., et al. , 2007. Distinct roles for mediator Cdk8 module subunits in Drosophila development. EMBO J. 26: 1045–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lonie A., D’Andrea R., Paro R., Saint R., 1994. Molecular characterization of the Polycomblike gene of Drosophila melanogaster, a trans-acting negative regulator of homeotic gene expression. Development 120: 2629–2636. [DOI] [PubMed] [Google Scholar]
- Lorberbaum D. S., Ramos A. I., Peterson K. A., Carpenter B. S., Parker D. S., et al. , 2016. An ancient yet flexible cis-regulatory architecture allows localized Hedgehog tuning by patched/Ptch1. Elife 5: e13550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lupo R., Breiling A., Bianchi M. E., Orlando V., 2001. Drosophila chromosome condensation proteins topoisomerase II and Barren colocalize with Polycomb and maintain Fab-7 PRE silencing. Mol. Cell 7: 127–136. [DOI] [PubMed] [Google Scholar]
- Mahmoudi T., Zuijderduijn L. M. P., Mohd-Sarip A., Verrijzer C. P., 2003. GAGA facilitates binding of pleiohomeotic to a chromatinized Polycomb response element. Nucleic Acids Res. 31: 4147–4156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannervik M., 2014. Control of Drosophila embryo patterning by transcriptional co-regulators. Exp. Cell Res. 321: 47–57. [DOI] [PubMed] [Google Scholar]
- Margueron R., Justin N., Ohno K., Sharpe M. L., Son J., et al. , 2009. Role of the Polycomb protein EED in the propagation of repressive histone marks. Nature 461: 762–767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin E. C., Adler P. N., 1993. The Polycomb group gene posterior sex combs encodes a chromosomal protein. Development 117: 641–655. [DOI] [PubMed] [Google Scholar]
- Mazo A. M., Huang D. H., Mozer B. A., Dawid I. B., 1990. The trithorax gene, a trans-acting regulator of the bithorax complex in Drosophila, encodes a protein with zinc-binding domains. Proc. Natl. Acad. Sci. USA 87: 2112–2116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKay D., Klusza S., Penke T. J. R., Meers M. P., Curry K. P., et al. , 2015. Interrogating the function of metazoan histones using engineered gene clusters. Dev. Cell 32: 373–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messmer S., Franke A., Paro R., 1992. Analysis of the functional role of the Polycomb chromo domain in Drosophila melanogaster. Genes Dev. 6: 1241–1254. [DOI] [PubMed] [Google Scholar]
- Miller T., Krogan N. J., Dover J., Erdjument-Bromage H., Tempst P., et al. , 2001. COMPASS: a complex of proteins associated with a trithorax-related SET domain protein. Proc. Natl. Acad. Sci. USA 98: 12902–12907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra R. K., Mihaly J., Barges S., Spiere A., Karch F., et al. , 2001. The iab-7 Polycomb response element maps to a nucleosome-free region of chromatin and requires both GAGA and pleiohomeotic for silencing activity. Mol. Cell. Biol. 21: 1311–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misulovin Z., Schwartz Y. B., Li X. Y., Kahn T. G., Gause M., et al. , 2008. Association of cohesin and Nipped-B with transcriptionally active regions of the Drosophila melanogaster genome. Chromosoma 117: 89–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moazed D., O’Farrell P. H., 1992. Maintenance of the engrailed expression pattern by Polycomb group genes in Drosophila. Development 116: 805–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohan M., Herz H. M., Smith E. R., Zhang Y., Jackson J., et al. , 2011. The COMPASS family of H3K4 methylases in Drosophila. Mol. Cell. Biol. 31: 4310–4318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohd-Sarip A., Venturini F., Chalkley G. E., Verrijzer C. P., 2002. Pleiohomeotic can link Polycomb to DNA and mediate transcriptional repression. Mol. Cell. Biol. 22: 7473–7483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohd-Sarip A., Lagarou A., Doyen C. M., Van der Knaap J. A., Aslan U., et al. , 2012. Transcription-independent function of Polycomb group protein PSC in cell cycle control. Science 336: 744–747. [DOI] [PubMed] [Google Scholar]
- Mohrmann L., Langenberg K., Krijgsveld J., Kal A. J., Heck A. J., et al. , 2004. Differential targeting of two distinct SWI/SNF-related Drosophila chromatin-remodeling complexes. Mol. Cell. Biol. 24: 3077–3088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moshkin Y. M., Mohrmann L., van Ijcken W. F., Verrijzer C. P., 2007. Functional differentiation of SWI/SNF remodelers in transcription and cell cycle control. Mol. Cell. Biol. 27: 651–661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller J., Bienz M., 1991. Long range repression conferring boundaries of Ultrabithorax expression in the Drosophila embryo. EMBO J. 10: 3147–3155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller J., Hart C. M., Francis N. J., Vargas M. L., Sengupta A., et al. , 2002. Histone methyltransferase activity of a Drosophila Polycomb group repressor complex. Cell 111: 197–208. [DOI] [PubMed] [Google Scholar]
- Négre N., Hennetin J., Sun L. V., Lavrov S., Bellis M., et al. , 2006. Chromosomal distribution of PcG proteins during Drosophila development. PLoS Biol. 4: e170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nekrasov M., Wild B., Müller J., 2005. Nucleosome binding and histone methyltransferase activity of Drosophila PRC2. EMBO Rep. 6: 348–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nekrasov M., Klymenko T., Fraterman S., Papp B., Oktaba K., et al. , 2007. Pcl-PRC2 is needed to generate high levels of H3–K27 trimethylation at Polycomb target genes. EMBO J. 26: 4078–4088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ng J., Li R., Morgan K., Simon J., 1997. Evolutionary conservation and predicted structure of the Drosophila extra sex combs repressor protein. Mol. Cell. Biol. 17: 6663–6672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Connell S., Wang L. J., Robert S., Jones C. A., Saint R., et al. , 2001. Polycomblike PHD fingers mediate conserved interaction with enhancer of zeste protein. J. Biol. Chem. 276: 43065–43073. [DOI] [PubMed] [Google Scholar]
- O’Dor E., Beck S. A., Brock H. W., 2006. Polycomb group mutants exhibit mitotic defects in syncytial cell cycles of Drosophila embryos. Dev. Biol. 290: 312–322. [DOI] [PubMed] [Google Scholar]
- Ohno K., McCabe D., Czermin B., Imhof A., Pirrotta V., 2008. ESC, ESCL and their roles in Polycomb group mechanisms. Mech. Dev. 125: 527–541. [DOI] [PubMed] [Google Scholar]
- Oktaba K., Gutierrez L., Gagneur J., Girardot C., Sengupta A. K., et al. , 2008. Dynamic regulation by Polycomb group protein complexes controls pattern formation and the cell cycle in Drosophila. Dev. Cell 15: 877–889. [DOI] [PubMed] [Google Scholar]
- Okulski H., Druck B., Bhalerao S., Ringrose L., 2011. Quantitative analysis of polycomb response elements (PREs) at identical genomic locations distinguishes contributions of PRE sequences and genomic environment. Epigenetics Chromatin 4: 4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Meara M. M., Simon J. A., 2012. Inner workings and regulatory inputs that control Polycomb repressive complex 2. Chromosoma 121: 221–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orsi G., Dasinathan S., Hughes K. T., Saminadin-Peter S., Henikoff S., et al. , 2014. High-resolution mapping defines the cooperative architecture of Polycomb response elements. Genome Res. 24: 809–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papoulas O., Beek S. J., Moseley S. L., McCallum C. M., Sarte M., et al. , 1998. The Drosophila trithorax group proteins BRM, ASH1 and ASH2 are subunits of distinct protein complexes. Development 125: 3955–3966. [DOI] [PubMed] [Google Scholar]
- Papp B., Müller J., 2006. Histone trimethylation and the maintenance of transcriptional ON and OFF states by trxG and PcG proteins. Genes Dev. 20: 2041–2054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park S. Y., Schwartz Y. B., Kahn T. G., Asker D., Pirrotta V., 2012. Regulation of Polycomb group genes Psc and Su(z)2 in Drosophila melanogaster. Mech. Dev. 128: 536–547. [DOI] [PubMed] [Google Scholar]
- Paro R., Hogness D. S., 1991. The Polycomb protein shares a homologous domain with a heterochromatin-associated protein of Drosophila. Proc. Natl. Acad. Sci. USA 88: 263–267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pengelly A. R., Copur Ö., Jäckle H., Herzig A., Müller J., 2013. A histone mutant reproduces the phenotype caused by loss of histone-modifying factor Polycomb. Science 229: 698–699. [DOI] [PubMed] [Google Scholar]
- Pengelly A. R., Kalb R., Finkl K., Müller J., 2015. Transcriptional repression by PRC1 in the absence of H2A monubiquitylation. Genes Dev. 29: 1487–1492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrimon N., Mahowald A. P., 1986. Maternal role of zygotic lethals during early embryogenesis in Drosophila, pp. 221–235 in Gametogenesis and the Early Embryo, edited by Gall J. Alan R. Liss, New York. [Google Scholar]
- Perrimon N., Engstrom L., Mahowald A. P., 1984. The effects of zygotic lethal mutations on female germ-line functions in Drosophila. Dev. Biol. 105: 404–414. [DOI] [PubMed] [Google Scholar]
- Peterson A. J., Kyba M., Bornemann D., Morgan K., Brock H. W., et al. , 1997. A domain shared by the Polycomb group proteins Scm and Ph mediates heterotypic and homotypic interactions. Mol. Cell. Biol. 17: 6683–6692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson A. J., Mallin D. R., Francis N. J., Ketel C. S., Stamm J., et al. , 2004. Requirement for sex comb on midleg protein interactions in Drosophila Polycomb group repression. Genetics 167: 1225–1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petruk S., Sedkov Y., Smith S., Tillib S., Kraevski V., et al. , 2001. Trithorax and dCBP acting in a complex to maintain expression of a homeotic gene. Science 294: 1331–1334. [DOI] [PubMed] [Google Scholar]
- Phelan M. L., Sif S., Narlikar G. J., Kingston R. E., 1999. Reconstitution of a core chromatin remodeling complex from SWI/SNF subunits. Mol. Cell 3: 247–253. [DOI] [PubMed] [Google Scholar]
- Phillips M. D., Shearn A., 1990. Mutations in polycombeotic, a Drosophila polycomb-group gene, cause a wide range of maternal and zygotic phenotypes. Genetics 125: 91–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piunti A., Shilatifard A., 2016. Epigenetic balance of gene expression by Polycomb and COMPASS families. Science 352: aad9780. [DOI] [PubMed] [Google Scholar]
- Plaschka C., Nozawa K., Cramer P., 2016. Mediator architecture and RNA polymerase II interaction. J. Mol. Biol. 428: 2569–2574. [DOI] [PubMed] [Google Scholar]
- Platero J. S., Hartnett T., Eissenberg J. C., 1995. Functional analysis of the chrome domain of HP1. EMBO J. 14: 3977–3986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian S., Capovilla M., Pirrotta V., 1991. The bx region enhancer, a distant cis-control element of the Drosophila Ubx gene and its regulation by hunchback and other segmentation genes. EMBO J. 10: 1415–1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray P., De S., Mitra A., Bezstarosti K., Demmers J. A. A., et al. , 2016. Combgap contributes to recruitment of Polycomb group proteins in Drosophila. Proc. Natl. Acad. Sci. USA 113: 3826–3831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remeseiro S., Losada A., 2013. Cohesin, a chromatin engagement ring. Curr. Opin. Cell Biol. 25: 63–71. [DOI] [PubMed] [Google Scholar]
- Rickels R., Hu D., Collings C. K., Woodfin A. R., Piunti A., et al. , 2016. An evolutionary conserved epigenetic mark of Polycomb response elements implemented by Trx/MLL/COMPASS. Mol. Cell 63: 318–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ringrose L., Paro R., 2004. Epigenetic regulation of cellular memory by the Polycomb and Trithorax group proteins. Annu. Rev. Genet. 38: 413–443. [DOI] [PubMed] [Google Scholar]
- Ringrose L., Paro R., 2007. Polycomb/Trithorax response elements and epigenetic memory of cell identity. Development 134: 223–232. [DOI] [PubMed] [Google Scholar]
- Roguev A., Schaft D., Shevchenko A., Pijnappel W. W., Wilm M., et al. , 2001. The Saccharomyces cerevisiae Set1 complex includes an Ash2 homologue and methylates histone 3 lysine 4. EMBO J. 20: 7137–7148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santamaría P., Randsholt N. B., 1995. Characterization of a region of the X chromosome of Drosophila including multi sex combs (mxc), a Polycomb group gene which also functions as a tumour suppressor. Mol. Gen. Genet. 246: 282–290. [DOI] [PubMed] [Google Scholar]
- Saurin A. J., Shao Z., Erdjument-Bromage H., Tempst P., Kingston R. E., 2001. A Drosophila Polycomb group complex includes zeste and dTAFII proteins. Nature 412: 655–660. [DOI] [PubMed] [Google Scholar]
- Savla U., Benes J., Zhang J., Jones R. S., 2008. Recruitment of Drosophila Polycomb-group proteins by Polycomblike, a component of a novel protein complex in larvae. Development 135: 813–817. [DOI] [PubMed] [Google Scholar]
- Schaaf C. A., Misulovin Z., Gause M., Koenig A., Gohara D. W., et al. , 2013. Cohesin and Polycomb proteins functionally interact to control transcription at silenced and active genes. PLoS Genet. 9: e1003560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheuermann J. C., Gaytán A., Oktaba K., Ly-Hartig N., McGinty R. K., et al. , 2010. Histone H2A deubiquitinase activity of the Polycomb repressive complex PR-DUB. Nature 465: 243–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitges F. W., Prusty A. B., Faty M., Stutzer A., Lingaraju G. M., et al. , 2011. Histone methylation by PRC2 is inhibited by active chromatin marks. Mol. Cell 42: 330–341. [DOI] [PubMed] [Google Scholar]
- Schmitt S., Prestel M., Paro R., 2005. Intergenic transcription through a Polycomb group response element counteracts silencing. Genes Dev. 19: 697–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuettengruber B., Ganaphathi M., Leblanc B., Portoso M., Jaschek R., et al. , 2009. Functional anatomy of Polycomb and Trithorax chromatin landscapes in Drosophila embryos. PLoS Biol. 7: 146–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schuettengruber B., Elkayam N. O., Sexton T., Entrevan M., Stem S., et al. , 2014. Cooperativity, specificity, and evolutionary stability of Polycomb targeting in Drosopohila. Cell Rep. 9: 219–233. [DOI] [PubMed] [Google Scholar]
- Schwartz Y. B., Cavalli G., 2017. Three-dimensional genome organization and function in Drosophila. Genetics 205: 5–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz Y. B., Pirrotta V., 2013. A new world of Polycombs: unexpected partnerships and emerging functions. Nat. Rev. Genet. 14: 853–864. [DOI] [PubMed] [Google Scholar]
- Schwartz Y. B., Kahn T. G., Niz D. A., Li X. Y., Bourgon R., et al. , 2006. Genome-wide analysis of Polycomb targets in Drosophila melanogaster. Nat. Genet. 38: 700–705. [DOI] [PubMed] [Google Scholar]
- Schwartz Y. G., Kahn T. G., Stenberg P., Ohno K., Bourgon R., et al. , 2010. Alternative epigenetic chromatin states of Polycomb target genes. PLoS Genet. 6: e10000805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwendemann A., Lehmann M., 2002. Pipsqueak and GAGA factor act in concert as partners at homeotic and many other loci. Proc. Natl. Acad. Sci. USA 99: 12883–12888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon M. P., Kaufman T. C., Shen M. W., Judd B. H., 1972. Lethality patterns and morphology of selected lethal and semi-lethal mutations in the zeste-white region of Drosophila melanogaster. Genetics 72: 615–638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanower G. A., Muller M., Blanton J. L., Honti V., Gyurkovics H., et al. , 2005. Characterization of the grappa gene, the Drosophila histone H3 lysine 79 methyltransferase. Genetics 169: 173–184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shao Z. H., Raible F., Mollaaghababa R., Guyon J. R., Wu C. T., et al. , 1999. Stabilization of chromatin structure by PRC1, a Polycomb complex. Cell 98: 37–46. [DOI] [PubMed] [Google Scholar]
- Shearn A., Hersperger G., Hersperger E., 1978. Genetic analysis of two allelic temperature-sensitive mutants of Drosophila melanogaster both of which are zygotic and maternal-effect lethals. Genetics 89: 341–353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shearn A., Hersperger E., Hersperger G., 1987. Genetic studies of mutations at two loci of Drosophila melanogaster which cause a wide variety of homeotic transformations. Wilhelm Roux’s Arch. Dev. Biol. 196: 231–242. [DOI] [PubMed] [Google Scholar]
- Shilatifard A., 2012. The COMPASS family of histone H3K4 methylases: mechanisms of regulation in development and disease pathogenesis. Annu. Rev. Biochem. 81: 65–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimell M. J., Peterson A. J., Burr J., Simon J. A., O’Conner M. B., 2000. Functional analysis of repressor binding sites in the iab-2 regulatory region of the abdominal-A homeotic gene. Dev. Biol. 218: 38–52. [DOI] [PubMed] [Google Scholar]
- Simon J., Chiang A., Bender W., Shimell M. J., O’Conner M., 1993. Elements of the Drosophila Bithorax complex that mediate repression by Polycomb group products. Dev. Biol. 158: 131–144. [DOI] [PubMed] [Google Scholar]
- Simon J. A., Kingston R. E., 2013. Occupying chromatin: polycomb mechanisms for getting to genomic targets, stopping transcriptional traffic, and staying put. Mol. Cell 49: 808–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinclair D. A. R., Syrzycka M., Macauley M. S., Rastgardani R., Komljenovic I., et al. , 2009. Drosophila O-GlcNAc transferase (OGT) is encoded by the Polycomb group (PcG) gene, super sex combs (sxc). Proc. Natl. Acad. Sci. USA 106: 13427–13432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slifer E. H., 1942. A mutant stock of Drosophila with extra sex combs. J. Exp. Zool. 90: 31–40. [Google Scholar]
- Smith S. T., Petruk S., Sedkov Y., Cho E., Tillib S., et al. , 2004. Modulation of heat shock gene expression by the TAC1 chromatin-modifying complex. Nat. Cell Biol. 6: 162–167. [DOI] [PubMed] [Google Scholar]
- Smouse D., Goodman C. S., Mahowald A. P., Perrimon N., 1988. polyhomeotic: a gene required for the embryonic development of axon pathways in the central nervous system of Drosophila. Genes Dev. 2: 830–842. [DOI] [PubMed] [Google Scholar]
- Song J. J., Garlick J. D., Kingston R. E., 2008. Structural basis of histone H4 recognition by p55. Genes Dev. 22: 1313–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soto M. C., Chou T.-B., Bender W., 1995. Comparison of germline mosaics of genes in the polycomb group of Drosophila melanogaster. Genetics 140: 231–243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasan S., Dorighi K. M., Tamkun J. W., 2008. Drosophila kismet regulates histone H3 lysine 27 methylation and early elongation by RNA polymerase II. PLoS Genet. 4: e1000217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stassen M. J., Bailey D., Nelson S., Chinwalla V., Harte P. J., 1995. The Drosophila trithorax proteins contain a novel variant of the nuclear receptor type DNA binding domain and an ancient conserved motif found in other chromosomal proteins. Mech. Dev. 52: 209–223. [DOI] [PubMed] [Google Scholar]
- Strübbe G., Popp C., Schmidt A., Pauli A., Ringrose L., et al. , 2011. Polycomb purification by in vivo biotinylation tagging reveals cohesin and trithorax group proteins as interaction partners. Proc. Natl. Acad. Sci. USA 108: 5572–5577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Struhl G., 1981. A gene product required for correct initiation of segmental determination in Drosophila. Nature 293: 36–41. [DOI] [PubMed] [Google Scholar]
- Struhl G., Akam M. E., 1985. Altered distributions of Ultrabithorax transcripts in extra sex combs mutant embryos of Drosophila. EMBO J. 4: 3259–3264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Struhl G., White R. A. H., 1985. Regulation of the Ultrabithorax gene of Drosophila by other bithorax complex genes. Cell 43: 507–519. [DOI] [PubMed] [Google Scholar]
- Suganuma T., Pattenden S. G., Workman J. L., 2008. Diverse functions of WD40 repeat proteins in histone recognition. Genes Dev. 22: 1265–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamkun J. W., Deuring R., Scott M. P., Kissinger M., Pattatucci A. M., et al. , 1992. brahma: a regulator of Drosophila homeotic genes structurally related to the yeast transcriptional activator SNF2/SWI2. Cell 68: 561–572. [DOI] [PubMed] [Google Scholar]
- Tanaka Y., Katagiri Z., Kawahashi K., Kioussis D., Kitajima S., 2007. Trithorax-group protein ASH1 methylates histone H3 lysine 36. Gene 397: 161–168. [DOI] [PubMed] [Google Scholar]
- Tschiersch B., Hofmann A., Krauss V., Dorn R., Korge G., et al. , 1994. The protein encoded by the Drosophila position-effect variegation suppressor gene Su(var)3-9 combines domains of antagonistic regulators of homeotic gene complexes. EMBO J. 13: 3822–3831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Therrien M., Morrison D. K., Wong A. M., Rubin G. M., 2000. A genetic screen for modifiers of a kinase suppressor of Ras-dependent rough eye phenotype in Drosophila. Genetics 156: 1231–1242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tie F., Furuyama T., Harte P. J., 1998. The Drosophila polycomb group proteins Esc and E(z) bind directly to each other and co-localize at multiple chromosomal sites. Development 125: 3483–3496. [DOI] [PubMed] [Google Scholar]
- Tie F., Prasad-Sinha J., Birve A., Rasmuson-Lestander A., Harte P. J., 2003. A 1-megadalton Esc/E(z) complex from Drosophila that contains Polycomblike and RPD3. Mol. Cell. Biol. 23: 3352–3362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tie F., Stratton C. A., Durzhals R. L., Harte P. J., 2007. The N terminus of Drosophila ESC brinds directly to histone H3 and is required for E(z)-dependent trimethylation of H3 lysine 27. Mol. Cell Biol. 27: 2014–2026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tie F., Banerjee R., Stratton C. A., Prasad-Sinha J., Stepanik V., et al. , 2009. CBP-mediated acetylation of histone H3 lysine 27 antagonizes Drosophila polycomb silencing. Development 136: 3131–3141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tie F., Banerjee R., Saiakhova A. R., Howard B., Monteith K. E., et al. , 2014. Trithorax monomethylates histone H3K4 and interacts directly with CBP to promote H3K27 acetylation and antagonize Polycomb silencing. Development 141: 1129–1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tillib S., Petruk S., Sedkov Y., Kuzin A., Fujioka M., et al. , 1999. Trithorax- and Polycomb-group response elements within an ultrabithorax transcription maintenance unit consist of closely situated but separable sequences. Mol. Cell. Biol. 19: 5189–5202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolhuis B., Blom M., Kerkhoven R. M., Pagie L., Teunissen H., et al. , 2011. Interactions among Polycomb domains are guided by chromosome architecture. PLoS Genet. 7: e1001343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treisman J., 2001. Drosophila homologues of the transcriptional coactivation complex subunits TRAP240 and TRAP230 are required for identical processes in eye-antennal disc development. Development 128: 603–615. [DOI] [PubMed] [Google Scholar]
- Tripoulas N., LaJeunesse D., Gildea J., Shearn A., 1996. The Drosophila ash1 gene product, which is localized at specific sites on polytene chromosomes, contains a SET domain and a PHD finger. Genetics 143: 913–928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tripoulas N. A., Hersperger E., La Jeunesse D., Shearn A., 1994. Molecular genetic analysis of the Drosophila melanogaster gene absent, small or homeotic discs1 (ash1). Genetics 137: 1027–1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Lohuizen M., Frasch M., Wientjens E., Berns A., 1991. Sequence similarity between the mammalian bmi-1 proto-oncogene and the Drosophila regulatory genes Psc and Su(z)2. Nature 353: 353–355. [DOI] [PubMed] [Google Scholar]
- Vázquez M., Moore L., Kennison J. A., 1999. The trithorax group gene osa encodes an ARID-domain protein that genetically interacts with the brahma chromatin-remodeling factor to regulate transcription. Development 126: 733–742. [DOI] [PubMed] [Google Scholar]
- Wang H., Wang L., Erdjument-Bromage H., Vidal M., Tempst P., et al. , 2004. Role of histone H2A ubiquitination in Polycomb silencing. Nature 431: 873–878. [DOI] [PubMed] [Google Scholar]
- Wang L., Brown J. L., Cao R., Zhang Y., Kassis J. A., et al. , 2004. Hierarchical recruitment of Polycomb group silencing complexes. Mol. Cell 14: 637–646. [DOI] [PubMed] [Google Scholar]
- Wang L., Jahren N., Vargas M. L., Anderson E. F., Benes J., et al. , 2006. Alternative ESC and ESC-LIKE subunits of a Polycomb group histone methyltransferase complex are differentially deployed during Drosophila development. Mol. Cell. Biol. 26: 2637–2647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z., Zang C., Rosenfeld J. A., Schones D. E., Barski A., et al. , 2008. Combinatorial patterns of histone acetylations and methylations in the human genome. Nat. Genet. 40: 897–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen P., Quan Z., Xi R., 2012. The biological function of the WD40 repeat-containing protein Caf1/p55 in Drosophila. Dev. Dyn. 241: 455–464. [DOI] [PubMed] [Google Scholar]
- White A. E., Burch B. D., Yang X.-C., Gasdaska P. Y., Dominski Z., et al. , 2011. Drosophila histone locus bodies form by hierarchical recruitment of components. J. Cell Biol. 193: 677–694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White R. A. H., Lehmann R., 1986. A gap gene, hunchback, regulates the spatial expression of ultrabithorax. Cell 47: 311–321. [DOI] [PubMed] [Google Scholar]
- White R. A. H., Wilcox M., 1985. Regulation of the distribution of Ultrabithorax proteins in Drosophila. Nature 318: 563–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C.-T., Jones R. S., Lasko P. F., Gelbart W. M., 1989. Homeosis and the interaction of zeste and white in Drosophila. Mol. Gen. Genet. 218: 559–564. [DOI] [PubMed] [Google Scholar]
- Xu C., Bian C., Yang W., Galka M., Ouyang H., et al. , 2010. Binding of different histone marks differentially regulates the activity and specificity of polycomb repressive complex 2 (PRC2). Proc. Natl. Acad. Sci. USA 107: 19266–19271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto Y., Girard F., Bello B., Affolter M., Gehring W. J., 1997. The cramped gene of Drosophila is a member of the Polycomb- group, and interacts with mus209, the gene encoding proliferating cell nuclear antigen. Development 124: 3385–3394. [DOI] [PubMed] [Google Scholar]
- Yang Z., Yik J. H., Chen R., He N., Jang M. K., et al. , 2005. Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol. Cell 19: 535–545. [DOI] [PubMed] [Google Scholar]
- Yuan W., Xu M., Huang C., Liu N., Chen S., et al. , 2011. H3K36 methylation antagonizes PRC2-mediated H3K27 methylation. J. Biol. Chem. 286: 7983–7989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zalokar M., Audit C., Erk I., 1975. Developmental defects of female-sterile mutants of Drosophila melanogaster. Dev. Biol. 47: 419–432. [DOI] [PubMed] [Google Scholar]
- Zhang C.-C., Bienz M., 1992. Segmental determination in Drosophila conferred by hunchback (hb), a repressor of the homeotic gene Ultrabithorax (Ubx). Proc. Natl. Acad. Sci. USA 89: 7511–7515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C.-C., Müller J., Hoch M., Jäckle H., Bienz M., 1991. Target sequences for hunchback in a control region conferring Ultrabithorax expression boundaries. Development 113: 117–1179. [DOI] [PubMed] [Google Scholar]
- Zink B., Paro R., 1989. In vivo binding pattern of a trans-regulator of homoeotic genes in Drosophila melanogaster. Nature 337: 468–471. [DOI] [PubMed] [Google Scholar]
- Zink B., Engstrom Y., Gehring W. J., Paro R., 1991. Direct interaction of the Polycomb protein with Antennapedia regulatory sequences in polytene chromosomes of Drosophila melanogaster. EMBO J. 10: 153–162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zraly C. B., Marenda D. R., Nanchal R., Cavalli G., Muchardt C., et al. , 2003. SNR1 is an essential subunit in a subset of Drosophila brm complexes, targeting specific functions during development. Dev. Biol. 253: 291–308. [DOI] [PubMed] [Google Scholar]