Abstract
This study examined the effect of increasing BW ratio (BWR) between rider and horse, in the BWR range common for Icelandic horses (20% to 35%), on heart rate (HR), plasma lactate concentration (Lac), BWR at Lac 4 mmol/l (W4), breathing frequency (BF), rectal temperature (RT) and hematocrit (Hct) in Icelandic horses. In total, eight experienced school-horses were used in an incremental exercise test performed outdoors on an oval riding track and one rider rode all horses. The exercise test consisted of five phases (each 642 m) in tölt, a four-beat symmetrical gait, at a speed of 5.4±0.1 m/s (mean±SD), where BWR between rider (including saddle) and horse started at 20% (BWR20), was increased to 25% (BWR25), 30% (BWR30), and 35% (BWR35) and finally decreased to 20% (BWR20b). Between phases, the horses were stopped (~5.5 min) to add lead weights to specially adjusted saddle bags and a vest on the rider. Heart rate was measured during warm-up, the exercise test and after 5, 15 and 30 min of recovery and blood samples were taken and BF recorded at rest, and at end of each of these aforementioned occasions. Rectal temperature was measured at rest, at end of the exercise test and after a 30-min recovery period. Body size and body condition score (BCS) were registered and a clinical examination performed on the day before the test and for 2 days after. Heart rate and BF increased linearly (P<0.05) and Lac exponentially (P<0.05) with increasing BWR. The W4 was 22.7±4.3% (individual range 17.0% to 27.5%). There was a positive correlation between back BCS and W4 (r=0.75; P=0.032), but no other correlations between body measurements and W4 were found. Hematocrit was not affected by BWR (P>0.05), but negative correlations (P<0.05) existed between body size measurements and Hct. While HR, Hct and BF recovered to values at rest within 30 min, Lac and RT did not. All horses had no clinical remarks on palpation and at walk 1 and 2 days after the test. In conclusion, increasing BWR from 20% to 35% resulted in increased HR, Lac, RT and BF responses in the test group of experienced adult Icelandic riding horses. The horses mainly worked aerobically until BWR reached 22.7%, but considerable individual differences (17.0% to 27.5%) existed that were not linked to horse size, but to back BCS.
Keywords: Icelandic horse, rider weight, weight carrying capacity, heart rate, lactate
Implications
The Icelandic horse is especially known for its tölt (amble) gait and is widely used as a leisure and sport horse. Although it is small as a riding horse, its riders are commonly adults. However, there is a lack of knowledge on the weight carrying capacity of the Icelandic horse. It is important in terms of horse welfare to generate such knowledge and to adjust training and use of the Icelandic horse accordingly.
Introduction
The Icelandic horse is popular and can be found in more than 30 countries (Worldfengur, 2015). It is a naturally gaited riding horse (Andersson et al., 2012) and is known particularly for its tölt (Björnsson and Sveinsson, 2006), which is a four-beat symmetrical gait with one or two legs always on the ground, that is without a suspension (Feldmann and Rostock, 1990). However, the Icelandic horse is rather small (141±3 cm; Stefánsdóttir et al., 2014) compared with for example, Swedish and German Warmblood riding horses (~165 cm; Holmström, 1990; Stock and Distl, 2006), and there is a welfare debate in the industry on how heavy its rider can be and some tourist companies have already put some limitation on the weight of the rider (Margrét Gunnarsdóttir, personal communication). Considering the average weight of Scandinavians (67 to 73 kg for women and 82 to 88 kg for men; Statistiska centralbyrån (SCB), 2005; Þórsson et al., 2009), Icelandic horses can be expected to carry ~20% to 25% of their BW, tack excluded. However, some riders of Icelandic horses weigh over 100 kg, corresponding to ~30% to 35% of BW (Stefánsdóttir et al., 2014). Powell et al. (2008) showed that horses carrying a rider corresponding to 25% or 30% of their BW had higher heart rate (HR), breathing frequency (BF) and rectal temperature (RT) than when carrying a rider corresponding to 15% and 20% of their BW. Therefore it is a welfare issue to create knowledge on how the BW of a rider affects physiological response to exercise in the Icelandic horse.
The aim of the present study was to measure the effect of increasing BW ratio (BWR) between rider and horse, in the BWR range common for Icelandic horses (20% to 35%) on HR, plasma lactate concentration (Lac), BWR at which Lac equals 4 mmol/l (W4), BF and hematocrit (Hct), in Icelandic horses in tölt. Our hypothesis was that horses would show higher HR, Lac, Hct and BF, in response to increased BWR.
Material and methods
The study was approved by the National Animal Research Committee of Iceland.
Horses
In total, eight Icelandic riding horses (four geldings, four mares), aged 17.0±1.2 years (range 15 to 19 years), with height at withers 140±2 cm (range 138 to 144 cm), BW before feeding in the morning 366±15 kg (range 340 to 382 kg; SmartScale 300; Gallagher, New Providence, PA, USA) and in their summer coat were used in the study. From January until mid-May, all horses were kept in the same stable in individual boxes (size 1.95×3.40 m) on permanent sawdust bedding at the Equine Science Department, Hólar University College, Saudarkrokur, Iceland. Grass forage was the main feed and all horses were fed the same feeds, but according to individual requirements (National Research Council, 2007) and BCS target 3.0 (scale 1.0 to 5.0) according to Stefánsdóttir and Björnsdóttir (2001). Actual BCS was 3.2±0.1 (range 3.00 to 3.25). A mineral and vitamin supplement (70 to 90 g/day; Racing Mineral, Trouw, Ghent, Belgium) was fed and the horses had free access to salt block (99% salt). Water was offered from automatic water bowls (flow ~11 l/min). The horses had all been used by university students in the same teaching program for 9 months before the study. For 1 week in May, before starting preparation and training for the experiment, the horses were kept on pasture and with free access to big-bale grass forage. When starting training for the experiment and on the experimental days, the horses were kept inside in the stable, except for 3 to 4 h in the afternoon when they grazed on pasture. In the mornings during the training and experimental days, they were fed ~1 kg of grass forage and had free access to salt block (99% salt) and automatic water bowls.
Preparation of the horses
In the last 2 weeks before the experiment, the horses were prepared for the experimental exercise test. The test was performed in tölt (at 5.4 m/s) and preparations included adjusting the speed and getting both the horses and the rider who later rode them in the test accustomed to carrying the equipment and lead weights. All horses were rested for at least 3 days before the experiment. The shoes and boots of all horses complied with the regulations for international gait competitions with Icelandic horses (International Federation of Icelandic Horse Associations (FEIF), 2014). All horses were shod with 8 mm shoes on all hooves and had 115 g boots on front hooves.
Rider and saddle
One rider rode all horses. The rider was a 25-year-old female, 65 kg of weight and 177 cm in height, and very lean. She was a professional rider and riding teacher for Icelandic horses and had a BSc degree in riding and riding teaching from Hólar University College. The rider was instructed to ride all horses in same manner for all BWRs, that is, to sit fully in the saddle and to use minimal aids to ride the horses in balance in a clear-beated tölt at the intended speed. The same saddle was used on all the horses. It was a saddle-type commonly used and specially designed for Icelandic horses (weight 8.4 kg; Ástund Super; Ástund, Reykjavík, Iceland). The experimental saddle was specially adapted (by the producing company Ástund) with three bags tightly fixed to the saddle tree and sewn to the flaps on both sides, where it was possible to add up to 10 kg lead weight on each side. Three saddle pads (1.3, 2.2 and 13.8 kg) and extra heavy stirrups (4 kg each) were also added when needed to reach the appropriate weight. In addition, the rider could carry added weight of up to 15 kg in a vest designed for scuba diving. Equal amounts of dead weight were added on both sides of the horse and rider.
Experimental design
A clinical examination was performed before and at 24 and 48 h after the exercise test. The examination included palpation/examination of neck, back and legs, and a whole limb flexion test (30 s) with a trot up on a concrete floor and walk and trot in a circle on soft ground. An exercise test with incrementally increased BWR was performed outdoors at Hólar University College in Iceland on 2 to 4 of June 2014, on a 300 m oval gravel riding track accepted for gæðinga competitions (FEIF, 2012). The rider rode on the outer circumference of the track, which corresponded to a distance of 321 m. Each horse performed the exercise test once and all exercise tests were ridden on the left hand. The incremental exercise test was preceded by a warm-up (1.1±0.2 km), consisting of 5 min of walking on a flat gravel surface (1.0 to 1.6 m/s) and 5 min of slow speed (1.8 to 3.1 m/s) in tölt in circles of different sizes in both directions (clockwise and counterclockwise) in the middle of the experimental track area. The exercise test started within 4:18±1:24 min:s (mean±SD) after the warm-up except for one horse, for which it started after 9:25 min:s because of problems with the equipment. The incremental exercise test consisted of five phases each comprising 642 m in tölt at a speed of 5.4±0.1 m/s (mean±SD), where BWR between rider (including saddle) and horse started at 20% (BWR20) and was then increased to 25% (BWR25), 30% (BWR30), 35% (BWR35) and in the final phase decreased to 20% (BWR20b). Between phases, the horses were stopped and the rider dismounted for 5:26±1:13 min:s (mean±SD), to add weight and for collection of a blood sample.
Data collection and handling
During warm-up, the exercise test, and a 30-min recovery, the duration was timed with a stopwatch. During the exercise test, the rider was informed, via wireless communication and an earpiece, after every 107 m (one-third of the length of the track) of the duration for guidance to keep the intended speed. The riding track had clear markings (bars) every 107 m on the 321 m oval track to facilitate adjustment of the speed and identify start and end of the exercise test. The HR of both horse and rider (Polar HR Monitor RS800CX; Polar Electro Oy, Kempele, Finland), speed and distance (Polar G3 GPS sensor, Polar Electro, Kempele, Finland) were recorded. Global positioning system (GPS) data were used for estimation of speed and distance of warm-up, but during the exercise test the stopwatch speed was used. According to the GPS manufacturer’s data, the accuracy of distance measurements was ±2% and of speed measurements ±2 km/h. The HR monitors were set to record in 1 s mode. All HR recordings were input to the software Polar ProTrainer 5 Equine Edition (Polar Electro). Heart rate data from the warm-up, the five phases of the exercise test, and the 30 min recovery were collected for the horses and from the exercise test for the rider. During each phase of the exercise test, the average HR during the last 60 s was used for both the horses and the rider. However, HR data on two horses for the whole exercise test and recovery were missing because of technical failure and for the rider for two exercise tests because of lost connectivity. Resting HR of the horses was taken late at night 2 to 4 days after the exercise tests, and a period of low HR during 30 min was used. Rectal temperature was measured at rest, at end of exercise test, and at end of 30 min recovery using a digital thermometer (Omron; Hoofddorp, the Netherlands). Breathing frequency was counted for at least 15 s at rest, at end of warm-up, at end of each phase of the exercise test, and at 5, 15 and 30 min within the recovery period.
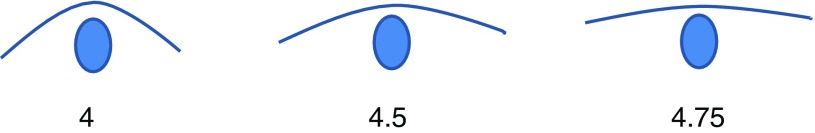
Height at withers, lowest point of the back, and croup and circumference of girth, carpus and cannon bone were measured on the day after finishing the exercise tests. Cannon bone circumference was measured both at the narrowest point and the mid-point between carpus and fetlock, on the left front leg. Body measurements were made using a stick or a tape and recorded to the nearest 0.5 or 1 cm. Back condition was scored according to the Henneke scale (score 1 to 9; Henneke et al., 1983), with the slight modification of using an accuracy of 0.25 of a score (Figure 1).
Figure 1.
Body condition score of the back (place of the saddle), based on the Henneke scale (Henneke et al., 1983) but modified to an accuracy of 0.25 of a score (the spot symbolizes the spine).
The horses in the study were numbered from 1 to 8 and got the same number in both part I and II of the study.
The weather was recorded every 5 min at an automatic weather station (Model WH-1080; Clas Ohlson, Insjön, Sweden) located ~1 km from the riding track. The ambient temperature was 12.1±4°C (daily range 7.6°C to 17.6°C), the relative humidity 66±16.7% (daily range 42% to 84%), the wind speed 0.5±0.5 m/s (daily range 0.3 to 1.0 m/s) and the average strongest wind speed 1.3±0.9 m/s (daily range 1.1 to 2.2 m/s); 2 days were dry, but on 1 day 0.3 to 1.2 mm/h rain fell during 3 h.
Blood sampling and blood analysis
The horses were fitted with a catheter (2.0×105 mm, Intranule; Vygon Sweden AB, Skellefteå, Sweden) in the jugular vein under local anesthesia (20 mg/ml, Xylocaine; AstraZeneca, Södertälje, Sweden) at least 1 h before starting the exercise test in the morning, and it was removed on the same day after the 30-min recovery period. Blood samples were taken at rest, after warm-up, after each phase of the exercise test, and after 5, 15 and 30 min recovery, using chilled lithium heparinized tubes (9 ml; Vacuette®, Greine Bio-One, Kremsmuenster, Austria). Two additional blood samples (only analyzed for Lac, creatine kinase (CK) and aspartate amino transferase (AST)) were taken by vacutainer 1 and 2 days after the test (after 24 and 48 h). Within 30 min, Hct was analyzed after centrifugation in capillary tubes (5 min, 10 062×g; Heraeus Septech (Biofuge A), Heraeus, Hanau, Germany). Duplicate analyses were performed and a mean value used in statistical analyses. The plasma was separated by centrifugation (10 min, 1167×g; Hettich, Tuttlingen, Germany) and then stored at −18°C until analysis of lactate concentration. Plasma lactate concentration was analyzed using an enzymatic (l-lactate dehydrogenase and glutamate-pyruvate transaminase) and spectrophotometric method (Bohehringer Mannheim/R-Biopharm, Darmstadt, Germany) with a CV of 2.3% according to the manufacturer. All plasma samples were analyzed in duplicate (CV of analysis 2.3%). Plasma activity of CK and AST was analyzed for samples before, at the end of the exercise test, and 1 and 2 days after the test using an enzymatic method (spectrophotometer, Architect c4000, Abbott Park, IL, USA). The intra- and inter-assay CV was 0.5% to 1.5% for both CK and AST in equine serum samples.
Statistical analysis and calculations
Statistical analyses were performed using SAS (version 9.4; SAS Institute Inc., Cary NC, USA). Normal distribution of the data was verified with residual plots. The experimental unit was each horse. PROC MIXED model 1, Y i=µ+α i+e i, was used to analyze differences in physiological responses of the horses and HR of the rider between all BWRs (BWR20 to BWR20b), where Y i is the observation/parameter, µ the mean value, α i the fixed effect of BWR and e i the residuals. Correlations between different BWRs for the same horse were estimated using unstructured covariance. PROC MIXED model 2, Y i=µ+α i+e i, was used to analyze the linear relationship between physiological responses (HR, BF, Hct) and BWR20 to BWR35, where Y i is the observation/parameter, µ the mean value, α i the continuous effect of BWR and e i the residuals. Correlations between different BWRs for the same horse were estimated using unstructured covariance. PROC MIXED model 3, Y i=µ+α i+e i, was used to analyze differences in physiological responses between all samples from rest until after 48 h recovery and number of samples ranged from 3 to 12 depending on physiological variable, where Y i is the observation/parameter, µ the mean value, α i the fixed effect of sample and e i the residuals. Correlations between different samples for the same horse were estimated using spatial power covariance structure (sp (pow) (time=0, 14.5, 22, 29.5, 37, 44.5, 52, 57, 67, 82, 1522, 2962 min)) and the two last time points were used for Lac, CK and AST. Data were presented as least square means with root mean standard error (RMSE) from model 1 and 3. RMSE was given for each BWR-group when using model 1. Data for Lac were log-transformed before analysis with model 3, as variances of the samples were different and the residuals were not normally distributed. Back transformed values were presented. For comparison, Tukey’s test was used and level of statistical significance was set to P<0.05 or a tendency if P<0.1. PROC CORR and PROC REG were used to examine the relationship between body measurements and physiological parameters. Microsoft Excel 2010 (Microsoft, Redmond, WA, USA) was used to calculate the exponential regressions between BWR and Lac and to estimate W4 for individual horses and the mean for the group.
Results
Heart rate and hematocrit
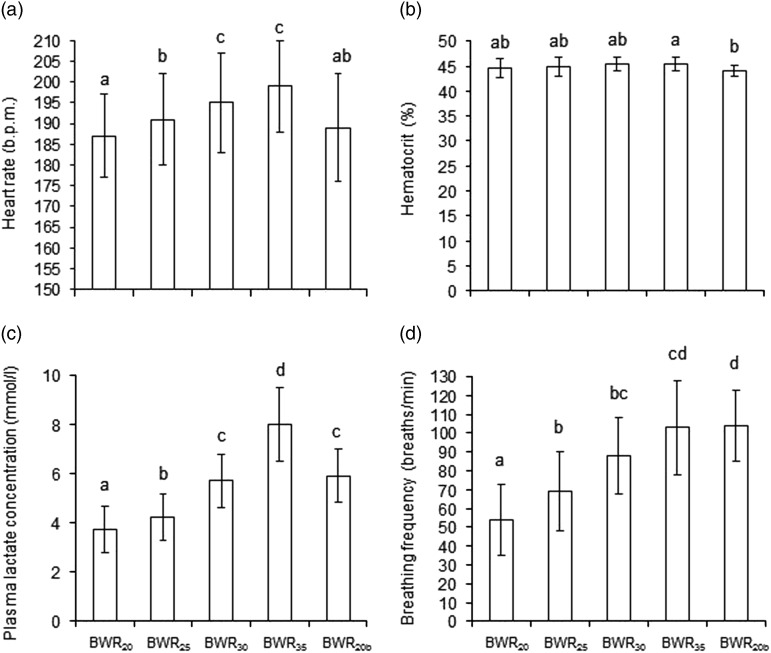
Heart rate increased linearly with increasing BWR (P<0.05; Figure 2a) and, although HR during BWR35 was numerically higher than during BWR30, the difference was not significant (P>0.05). Heart rate during BWR20b was lower than during BWR30 and BWR35 (P<0.05) and not different from during BWR20 and BWR25 (P>0.05). Peak HR increased with increasing BWR (193±8, 195±11, 198±12 and 203±10 beats per minute (b.p.m.) for BWR20, BWR25, BWR30 and BWR35, respectively), and was higher (P<0.05) during BWR35 than BWR20, BWR25 and BWR20b. For individual horses, there was a positive linear relationship between BWR and HR (mean±SD: r 2=0.89±0.10; range 0.77 to 0.99). Both HR and peak HR increased by 7 b.p.m. for every 10% increase in BWR in the range tested (P<0.01). Heart rate was back to resting level after 30 min recovery (P>0.05; Table 1). Hematocrit (%) was not affected by BWR (P>0.05; Figure 2b) apart from being lower after BWR20b than after BWR35 (P<0.05). Hematocrit was back to resting level after 30 min recovery (P>0.05; Table 1).
Figure 2.
(a) Heart rate, (b) hematocrit, (c) plasma lactate concentration and (d) breathing frequency (least square means±root mean standard error) after five phases (each 642 m in tölt) in an incremental exercise test in eight Icelandic horses where the BW ratio between rider and horse started at 20% (BWR20), was increased to 25% (BWR25), 30% (BWR30) and 35% (BWR35), and then decreased to 20% (BWR20b). Blood samples were collected and breathing frequency counted at end of each of the five phases. Heart rate was recorded during the last minute of each phase. a,b,c,dSignificantly different mean value (P<0.05).
Table 1.
Physiological responses before, during and after an incremental exercise test in eight mature Icelandic riding horses 1
| Incremental exercise test | Recovery | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Item | At rest | Warm-up | BWR20 | BWR25 | BWR30 | BW35 | BWR20b | 5 min | 15 min | 30 min | 24 h | 48 h | RMSE |
| HR (b.p.m.) | 36a | 145b | 187c | 191cd | 195cd | 199d | 189c | 78e | 62f | 49a | 8 | ||
| Lac (mmol/l) | 0.9a | 1.4b | 3.6cde | 4.1cd | 5.7gh | 7.9i | 5.8g | 4.5ch | 2.9df | 2.3ef | 0.9a | 0.8a | 1.0 |
| Hct (%) | 34a | 43bc | 45bc | 45b | 46b | 45b | 44b | 42c | 38d | 36a | 2 | ||
| BF (breaths/min) | 15a | 38ab | 54bcd | 69ce | 88ef | 101f | 104f | 58bce | 42ac | 28ad | 19 | ||
| Rectal temperature (°C) | 37.5a | 39.5b | 38.5c | 0.3 | |||||||||
| CK concentration (U/l) | 395a | 404a | 443a | 463a | 67 | ||||||||
| AST concentration (U/l) | 368a | 386b | 379ab | 394ab | 80 | ||||||||
BF=breathing frequency; CK=creatine kinase; AST, aspartate amino transferase.
Values presented as least square means and root mean standard error (RMSE). Samples for plasma lactate concentration (Lac), hematocrit (Hct) and breathing frequency (BF) were collected at rest, at end of warm-up, at end of five phases (each 642 m in tölt) of the exercise test, where the ratio between rider : horse BW started at 20% (BWR20), was increased to 25% (BWR25), 30% (BWR30) and 35% (BWR35), and then decreased to 20% (BWR20b), and at end of 5, 15, 30 min recovery and 24 and 48 h recovery. Measurements of heart rate (HR) were made during the last minute of the same phases except at rest, where HR was measured at night using a 30-min period with a low HR. Means within a row with different letters are significantly different (P<0.05).
Heart rate of the rider was 108±7, 109±6, 111±6, 123±10 and 113±7 b.p.m. for BWR20, BWR25, BWR30, BWR35 and BWR20b, respectively, and did not differ between BWRs, but showed a tendency to be higher for BWR35 than for BWR25 (P=0.0606).
Plasma lactate concentration
Plasma lactate concentration increased exponentially when BWR increased from 20% to 35% (Lac=1.2221×e0.0524×BWR; r 2=0.97; P<0.05; Figure 2c) and was highest after BWR35 (P<0.05; Figure 2c). For individual horses there was also a strong exponential relationship between BWR and Lac (range 0.87 to 0.98). BWR at Lac 4 mmol/l was 22.7±4.3% (individual range 17.0% to 27.5%). Plasma lactate concentration decreased after BWR20b compared with BWR35, but was still higher than after BWR20 and BWR25 (P<0.05). Plasma lactate concentration was not back to resting level after 30 min recovery (P<0.001), but was back after 24 and 48 h (P>0.05; Table 1).
Breathing frequency and rectal temperature
Breathing frequency increased linearly with increasing BWR (P<0.05; Figure 2d) and was not lower after BWR20b than BWR35 (P>0.05). Breathing frequency increased by three breaths per min for every 1% increase in BWR in the range tested (P<0.001). Breathing frequency was back to resting level after 30 min recovery (P>0.05; Table 1). Rectal temperature increased from rest until after end of the exercise test and had decreased after 30 min recovery, but was still higher than the resting level (P<0.001; Table 1).
Correlations between physiological parameters and BW, body condition score and body measurements
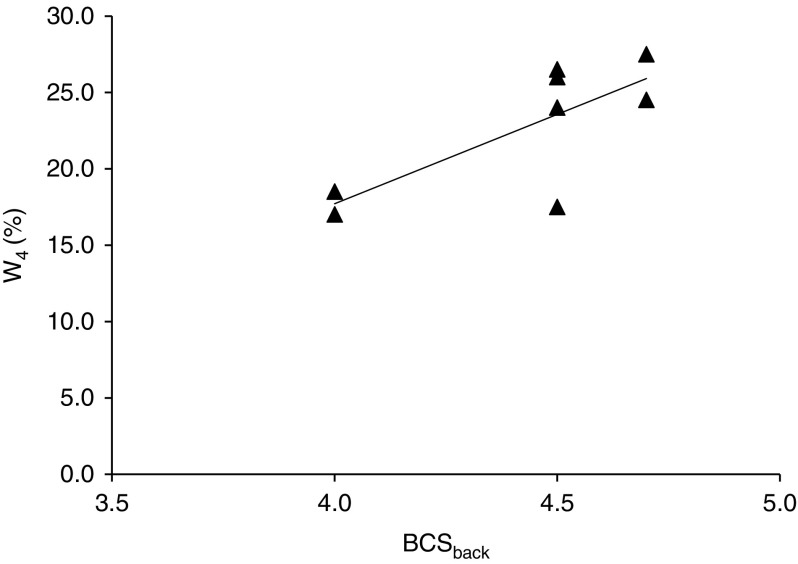
There was a positive correlation between back BCS and W4 (r=0.75; P=0.032; Figure 3), but no other correlations between body measurements and W4 were found. Furthermore, back BCS was negatively correlated with Lac after 15 min recovery (r=−0.91; P=0.0018). There were negative correlations between Hct (after warm-up, after BWR20, BWR25 and BWR30, and after 5 and 15 min recovery) and height at withers and at croup, cannon bone and carpus circumference (range: r=−0.73 to −0.93; P<0.05). There were also negative correlations between BW and Hct (after BWR25 and BWR30) (r=−0.74 and r=−0.81, respectively; P<0.05).
Figure 3.
Relationship between back body condition score of the horses (BCSback) (Henneke scale 1–9; Henneke et al., 1983) and the BW ratio between rider and horse at which the horses reached 4 mmol/l (W4) (y=11.706×−29.11, r 2=0.56).
Health and levels of creatine kinase and aspartate amino transferase
All horses were without clinical remarks during palpation and at walk both before and 1 and 2 days after the test. All horses except one (horse number 2) were also without clinical remarks at trot before and after the test. The one exception appeared ʻshort-strided and earth-boundʼ (although symmetrical) in trot before the test and showed right hind lameness (<1 degree) on the day after and was again short-strided and earth-bound 2 days later. In addition, a temporary tremor was observed in the right quadriceps muscle of this horse immediately after the BWR35 treatment. One other horse (number 5) was slightly asymmetrical on trot at a circle on soft ground 2 days after the test. There were no differences in the plasma activity of CK after exercise compared to with at rest (Table 1), but AST was higher (P<0.01) after 5 min recovery than at rest but after 24 and 48 h it was not different from at rest (Table 1).
Discussion
As hypothesized, the physiological response of the horses increased as BWR increased from 20% to 35%. These results are in accordance with the fundamental laws of physics (Newton’s second law), and also in accordance with results on weight carrying in other riding horses (Thornton et al., 1987; Sloet van Oldruitenborgh-Oosterbaan et al., 1995; Powell et al., 2008). However, to the best of our knowledge this is the first study to measure the physiological response to increased BWR in Icelandic horses and also the first study to assess a weight threshold at which lactate rapidly accumulates (W4) in a group of horses. On the basis of our observations, it is likely that the continuous time at which these horses could have stayed in tölt (at this same speed) would have been limited at BWR >23%. However, there was considerable individual variation in W4 (range 17.5% to 27.5%) despite the use of this uniform group of horses that had been bred, kept and trained for the same purpose at the same place (Hólar University College) all their life. This indicates that individual and genetic factors could have a substantial effect on W4 and weight carrying capacity.
Interestingly, we found no effect of BW or size on W4, but the results indicate that back or Musculus longissimus dorsi conformation could be important for weight carrying ability, since W4 increased with back BCS. This could indicate that horses with more ‘muscular’ backs either had support from more aerobic muscle tissue or that their muscle tissue was larger or better at metabolizing lactate during exercise. It is known that the M. longissimus consists mainly of type IIA and IIX fibers (Gottlieb et al., 1989; Hyytiäinen et al., 2014), and individual variations in muscle fiber proportion might affect the metabolic response to exercise. Dobec et al. (1994) have suggested that the size of the M. longissimus is correlated to performance in Standardbred horses (the larger the better), but this could not be verified by Ringmark et al. (2015). However, the suggestion that back conformation affects weight carrying ability is also supported by observations by Powell et al. (2008), who found that post exercise muscle soreness was worse in horses with narrower loins. It would be of interest to collect more data on the relationship between the muscle conformation of the topline (back, loin and croup), limbs, trunk (sides of the body) and underline, and the weight carrying capacity of horses.
It is known that the amount of red blood cells released from the spleen reflects the intensity of exercise until maximum splenic contraction is reached (Persson, 1967). Despite the significant increase in HR, Lac and BF as BWR increased, there was no increase in Hct in the present study, indicating that the horses were close to their maximum Hct response already after BWR20. However, the negative correlations between exercise Hct and some of the body size measurements indicate that there was an individual response related to horse size, that is, the smaller the horse, the larger the Hct. Interestingly, further analysis showed that by dividing the horses into two groups based on withers height, smaller horses (⩽140 cm, five horses) had higher (P<0.05) Hct values during exercise test and recovery than larger horses (>140 cm, three horses) (data not shown). This indicates that smaller horses had higher aerobic capacity, due perhaps to having been subjected to more intensive training (i.e., carrying comparatively heavier weights) than the larger horses, and accordingly developed a higher Hct. This agrees with findings by Ringmark et al. (2015), who found that horses subjected to more high intensity training developed higher Hct than a group with shorter distances of high intensity training. The lack of correlation between body size and W4 could therefore perhaps also have been due to greater aerobic potential in smaller horses. Nevertheless, the peak Hct levels observed here were low (mean±SD: 46±1%) compared with those reported in other breeds (mean: >60%; Persson, 1983; Evans et al., 1993), but similar to those observed in Icelandic pace horses (mean±SD: 44±3%; Stefánsdóttir et al., unpublished data) and in 180 Icelandic mares (4 to 11 years) after performing an intensive exercise test (mean±SD: 44±2%; Stefánsdóttir et al., 2014). Low levels could partly be due to the samples not being collected during exercise but immediately afterwards, when Hct might have decreased, as shown by Bayly et al. (2006) in a study where Hct decreased by 3% from the time of fatigue until immediately after the end of exercise. However, peak Hct was still considerably lower than that observed in Standardbreds sampled in the same way as in the present study (mean±SE: 60±1%; Jansson and Dahlborn, 1999). One explanation could be breed effects, since spleen weight differs, with racing breeds (Thoroughbred and Standardbred) having significantly (P<0.01) greater relative spleen size than other breeds (stock types, Arabian and draft types) (Kline and Foreman, 1991). The reason and importance of the comparatively low Hct values in Icelandic horses need further investigation.
To evaluate the response to weight carrying, a number of different exercise tests could be performed. Use of an incremental standardized exercise test, like that used in the present study, is a widely accepted method for evaluating physiological responses and fitness in both humans and horses (Couroucé-Malblanc and Hodgson, 2014; Couroucé-Malblanc and van Erck-Westergren, 2014). It usually consists of several bouts of exercise (often 1 to 3 min duration per bout) at increasing speed, after a warm-up, which may or may not be separated by a period of recovery (Couroucé-Malblanc and van Erck-Westergren, 2014). Instead of increasing speed between phases (bouts) in our study, we kept the same speed and gait but added more weight on the horse in each phase, thus increasing the workload. It was not the aim of this study to exercise the horses to fatigue, but rather to evaluate physiological responses to different BWRs. In order to ‘verify’ that the physiological responses measured in each phase of the exercise test mainly reflected the carried weight, and not the accumulated effect of the exercise test, the 20% BWR was repeated at the end. The HR and Hct responses to BWR20b were not different from those to BWR20, which indicates that the physiological response after each BWR mainly reflected the workload of the BWR just performed. Plasma lactate concentration also decreased after BWR20b compared with after BWR35, indicating efficient recovery (significant aerobic metabolism of lactate). However, as for incremental exercise tests in general, some minor cumulative effects could confound the results. For example, BF did not decrease between BWR35 and BWR20b. It is known that BF is to some extent limited by stride frequency during exercise (depending on gait) (Butler et al., 1993) and therefore breathing can do more of a catch-up post exercise. According to our knowledge little is known about how BF is affected by tölt at different speeds. Breathing can also assist in cooling the body during exercise, although vasodilation and sweating are more important (Hodgson, 2014). Nonetheless, the RT of the horses was 2°C higher at the end of the exercise test and still 1°C higher than at rest after 30 min recovery, indicating a substantial need for cooling. However, to be able to truly evaluate any accumulative effects, an additional test must be performed where BWR20 is repeated five times instead of the incremental design.
In our study the aim was to isolate the effect of weight of the rider and to exclude the effect of riding style. Therefore one rider rode all the horses and extra weight was added as lead fixed to the saddle or rider. The extra weight was intended to follow the movement of the horse or the rider closely and therefore behave more as live weight rather than dead weight. Sloet van Oldruitenborgh-Oosterbaan et al. (1995) did not find differences in the physiological responses in horses mounted or lead-loaded with equal weight, but argued that it was important that the dead weight was stable and followed the movement of the horse. The rider in our study only needed to carry extra weight in the vest during BWR35, and although HR of the rider was not different for different BWRs, it was numerically highest and showed the greatest SD during BWR35. This might indicate that the physical intensity of the rider differed most during that BWR. The reason could be that, according to the rider, the horses needed more assistance and helping aids in keeping the correct beat in tölt as the weight increased. However, the rider did not feel that the horses had less willing attitude or needed more driving aids with the increased weight. It has earlier been shown that HR and workload in riders of dressage horses reflected how much the horses needed to be driven forward (Devienne and Guezennec, 2000).
As discussed earlier, exercise at an intensity where plasma lactate levels exceed 4 mmol/l can only be maintained for relatively short periods (minutes) without causing fatigue or reducing performance. This intensity was reached at 25% BWR in the present study, whereas in a study by Powell et al. (2008), horses did not reach 4 mmol/l until after carrying 30% of their BW. This may reflect differences in the intensity of the exercise tests, for example, speed and gait, but also track surface, environmental conditions and fitness of the horses. Moreover, as already mentioned, the present study found considerable individual variation in W4 that was not related to BW or height at withers of the horses. Garlinghouse and Burrill (1999) also showed that completion rate among 360 horses in a 160 km endurance race was not affected by BWR (range 15% to 31%). Altogether, this highlights the problem with providing general recommendations for how much weight a horse can carry. Thus this study cannot provide any general answer to the question asked by practitioners ‘How much weight can an Icelandic horse carry?’, except to say that the size of the horse is not a simple and decisive measure of how much weight it can carry and for how long. Other studies indicate that overall BCS is more important than BWR for performance (Garlinghouse and Burrill, 1999) and also that the rider’s balance and technique (De Cocq et al., 2010) and the coupling between carrier and load might affect energetic cost (De Cocq and van Weeren, 2014).
However, in order to answer the question ‘How much weight can a horse carry?’, it is necessary to decide ‘gold standard’ parameters to estimate weight carrying capacity, for example physiological and/or locomotor parameters, or short-term or long-term assessments. We suggest that long-term health and locomotion symmetry should be used, since short-term deviations from normal/symmetrical conditions might be part of the training adaptation process. However, further research is needed to relate short- and long-term physiological responses to long-term health (e.g., back, legs) and locomotion.
The total distance ridden in our study (4310 m; warm-up plus exercise test) and the speed (5.4 m/s) during the exercise test can reflect a common 1 day exercise for an adult Icelandic riding horse and the speed was selected as the upper limit of medium speed when riding tölt in Icelandic horses by leisure riders. With the exception of Lac and RT, the horses had recovered within 30 min, and CK and AST activity was not significantly increased from rest at 24 and 48 h recovery, indicating that the exercise test did not cause any substantial damage to the muscle tissue (Kingston, 2008). Clinical examination also showed that the test caused no or very limited new clinical findings. However, one horse showed tremor after carrying the heaviest weight and the same individual appeared ‘short-strided and earth-bound’ before the test. The importance and link between these observations are unknown. In the study by Powell et al. (2008), horses which carried 30% of their body weight for a longer distance than in our study (6400 m, ~3.3 m/s) had significantly elevated CK activity immediately post exercise and 48 h after exercise than when carrying 15%, 20% and 25% of their body weight (same exercise test) and a higher score in muscle soreness and stiffness 24 h post exercise. These results are difficult to compare with ours and their observations might also have been affected by the fact that their horses had been rested for 4 months before the study (Powell et al., 2008) and might not have been fit enough for the exercise test.
In the present study we did not include an evaluation where the horses carried no weight at all. This might be of academic interest but our main focus was to evaluate the effect of the rider weight. Another reason for excluding the ‘no weight treatment’ was that it is difficult to have horses in tölt at standard speed without a rider.
It should be noticed that the results from our study are relevant to Icelandic horses but might not be transferred to all riding horse breeds or types without caution.
Without doubt, there is a need for further studies on how BWR affects exercise performance, fatigue, and recovery after different types of exercise and also how age and training background (fitness) of horses affect their weight carrying capacity. We suggest that the type of test used in this study could be used for evaluation and comparison of the physiological and metabolic response to weight carrying in horses (under standardized conditions).
Conclusions
This study showed that increasing the BWR from 20% to 35% increased HR, Lac, RT and BF responses, in the group of experienced adult Icelandic riding horses tested here. The horses mainly worked aerobically until the BWR reached 22.7%, but considerable individual differences (17.0% to 27.5%) existed which were not linked to horse size, but to back BCS.
Acknowledgments
The authors sincerely thank the research team at Hólar in June 2014 for their valuable work and cooperation. Special thanks to Fredrica Fagerlund for training and riding the horses, Ástund Reykjavík for the help with adjustment of the saddle, Claudia von Brömssen for statistical help and Anna-Greta Haglund for analyses of plasma lactate.
The project was funded by the Stock Protection Fund for the Icelandic horse, the Developmental Fund for Icelandic Horse Breeding, the Pálmi Jónsson Natural Conservation Fund and Hólar University College.
References
- Andersson LS, Larhammar M, Memic F, Wootz H, Schwochow D, Rubin CJ, Patra K, Arnason T, Wellbring L, Hjälm G, Imsland F, Petersen JL, McCue ME, Mickelson JR, Cothran G, Ahituv N, Roepstorff L, Mikko S, Vallstedt A, Lindgren G, Andersson L and Kullander K 2012. Mutations in DMRT3 affect locomotion in horses and spinal circuit function in mice. Nature 488, 642–646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayly WM, Kingston JK, Brown JA, Keegan RD, Greene SA and Sides RH 2006. Changes in arterial, mixed venous and intraerythrocytic concentrations of ions in supramaximally exercising horses. Equine Veterinary Journal 38 (suppl. 36), 294–297. [DOI] [PubMed] [Google Scholar]
- Björnsson GB and Sveinsson HJ 2006. The Icelandic horse. Mál og menning, Reykjavík, Iceland. [Google Scholar]
- Butler PJ, Woakes AJ, Smale K, Roberts CA, Hillidge CJ, Snow DH and Marlin DJ 1993. Respiratory and cardiovascular adjustments during exercise of increasing intensity and during recovery in Thoroughbred racehorses. Journal of Experimental Biology 179, 159–180. [DOI] [PubMed] [Google Scholar]
- Couroucé-Malblanc A and Hodgson DR 2014. Clinical exercise testing In: The athletic horse, principles and practice of equine sports medicine, 2nd edition (ed. DR Hodgson, KH McKeever and CM McGowan), pp. 366–378. Elsevier Saunders, St. Louis, MO, USA. [Google Scholar]
- Couroucé-Malblanc A and van Erck-Westergren E 2014. Exercise testing in the field In: Equine sports medicine and surgery, 2nd edition (ed. KW Hinchcliff, AJ Kaneps and RJ Geor), pp. 25–42. Elsevier Saunders, Edinburgh, UK. [Google Scholar]
- De Cocq P, Duncker AM, Clayton HM, Bobbert MF, Muller M and van Leeuwen JL 2010. Vertical forces on the horse’s back in sitting and rising trot. Journal of Biomechanics 43, 627–631. [DOI] [PubMed] [Google Scholar]
- De Cocq P and van Weeren PR 2014. Functional biomechanics: effect of the rider and tack In: The athletic horse, principles and practice of equine sports medicine, 2nd edition (ed. DR Hodgson, KH McKeever and CM McGowan), pp. 293–298. Elsevier Saunders, St. Louis, MO, USA. [Google Scholar]
- Devienne MF and Guezennec CY 2000. Energy expenditure of horse riding. European Journal of Applied Physiology 82, 499–503. [DOI] [PubMed] [Google Scholar]
- Dobec RL, Borger ML and Neal SM 1994. Correlation of real-time ultrasonic measures of fat and longissimus muscle area in Standardbred horses with lifetime racing record and lifetime winnings. Journal of Animal Science 72, 44. [Google Scholar]
- Evans DL, Harris RC and Snow DH 1993. Correlation of racing performance with blood lactate and heart rate after exercise in Thoroughbred horses. Equine Veterinary Journal 25, 441–445. [DOI] [PubMed] [Google Scholar]
- Feldmann W and Rostock AK 1990. Hesturinn og reiðmennskan. Steindórsprent, Reykjavík, Iceland. [Google Scholar]
- Garlinghouse SE and Burrill MJ 1999. Relationship of body condition score to completion rate during 160 km endurance races. Equine Veterinary Journal 31, 591–595. [DOI] [PubMed] [Google Scholar]
- Gottlieb M, Essén-Gustavsson B, Lindholm A and Persson SG 1989. Effects of a draft-loaded interval-training program on skeletal muscle in the horse. Journal of Applied Physiology 67, 570–577. [DOI] [PubMed] [Google Scholar]
- Henneke DR, Potter GD, Kreider JL and Yeates BF 1983. Relationship between condition score, physical measurements and body fat percentage in mares. Equine Veterinary Journal 15, 371–372. [DOI] [PubMed] [Google Scholar]
- Hodgson DR 2014. Thermoregulation In: The athletic horse, principles and practice of equine sports medicine, 2nd edition (ed. DR Hodgson, KH McKeever and CM McGowan), pp. 108–124. Elsevier Saunders, St. Louis, MO, USA. [Google Scholar]
- Holmström M 1990. Variation in conformation of Swedish Warmblood horses and conformational characteristics of élite sport horses. Equine Veterinary Journal 22, 186–193. [DOI] [PubMed] [Google Scholar]
- Hyytiäinen HK, Mykkänen AK, Hielm-Björkman AK, Stubbs NC and McGowan CM 2014. Muscle fibre type distribution of the thoracolumbar and hindlimb regions of horses: relating fibre type and functional role. Acta Veterinaria Scandinavica 56, 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- International Federation of Icelandic Horse Associations (FEIF) 2012. Prefix to the ‘gæðingakeppni’. Retrieved on 13 February 2015 from http://www.feiffengur.com/documents/G%C3%A6%C3%B0ingakeppni%202012.pdf.
- International Federation of Icelandic Horse Associations (FEIF) 2014. Feif rules for Icelandic horse sport events (FIPO). Retrieved on 13 February 2015 from http://www.feiffengur.com/documents/fipo2014.
- Jansson A and Dahlborn K 1999. Effects of feeding frequency and voluntary salt intake on fluid and electrolyte regulation in athletic horses. Journal of Applied Physiology 86, 1610–1616. [DOI] [PubMed] [Google Scholar]
- Kingston JK 2008. Hematologic and serum biochemical responses to exercise and training In: Equine exercise physiology, the science of exercise in the athletic horse (ed. KW Hinchcliff, RJ Geor and AJ Kaneps), pp. 398–409. Elsevier Saunders, Philadelphia, PA, USA. [Google Scholar]
- Kline H and Foreman JH 1991. Heart and spleen weights as a function of breed and somatotype. In Proceedings of the 3rd International Conference on Equine Exercise Physiology (ed. SGB Persson, A Lindholm and LB Jeffcott), 15–19 July 1990, Uppsala Sweden, pp. 17–21. ICEEP Publications, Davis, CA, USA.
- National Research Council 2007. Nutrient requirements of domestic animals. Nutrient requirements of horses, 6th revised edition National Academy Press, Washington, DC, USA. [Google Scholar]
- Persson SGB 1967. On blood volume and working capacity in horses. Acta Veterinaria Scandinavica 19 (suppl.), 1–189. [PubMed] [Google Scholar]
- Persson SGB 1983. The significance of haematological data in the evaluation of soundness and fitness in the horse. In Proceedings of the 1st International Conference on Equine Exercise Physiology (ed. DH Snow, SGB Persson and RJ Rose), 22–24 September 1982, Oxford, England, pp. 324–327. Granta Publications, Cambridge, UK.
- Powell DM, Bennett-Wimbush K, Peeples A and Duthie M 2008. Evaluation of indicators of weight-carrying ability of light riding horses. Journal of Equine Veterinary Science 28, 28–33. [Google Scholar]
- Ringmark S, Lindholm A, Hedenström U, Lindinger M, Dahlborn K, Kvart C and Jansson A 2015. Reduced high intensity training distance had no effect on VLa4 but attenuated heart rate response in 2–3-year-old Standardbred horses. Acta Veterinaria Scandinavica 57, 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Statistiska centralbyrån (SCB) 2005. Pressrelease från statistiska centralbyrån. Retrieved on 8 April 2015 from http://www.scb.se/sv_/Hitta-statistik/Artiklar/Vi-vaxer-pa-bredden/.
- Sloet van Oldruitenborgh-Oosterbaan MM, Barnevald A and Schamhardt HC 1995. Effects of weight and riding on workload and locomotion during treadmill exercise. Equine Veterinary Journal 27, 413–417. [Google Scholar]
- Stefánsdóttir GJ and Björnsdóttir S 2001. Body condition scoring of horses (Mat á holdafari hrossa) special issue. Eiðfaxi-Ræktun 1, 60–65. [Google Scholar]
- Stefánsdóttir GJ, Ragnarsson S, Gunnarsson V and Jansson A 2014. Physiological response to a breed evaluation field test in Icelandic horses. Animal 8, 431–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stock KF and Distl O 2006. Genetic correlations between conformation traits and radiographic findings in the limbs of German Warmblood Riding horses. Genetics Selection Evolution 38, 657–671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornton J, Pagan J and Persson S 1987. The oxygen cost of weight loading and inclined treadmill exercise in the horse. In Proceedings of the 2nd International Conference on Equine Exercise Physiology (ed. JR Gillespie and NE Robinson), 7–11 August 1986, San Diego, CA, USA, pp. 206–215. ICEEP Publications, Davis, CA, USA.
- Þórsson B, Aspelund T, Harris T, Launer LJ and Guðnason V 2009. Trend in body weight and diabetes in forty years in Iceland. Icelandic Medical Journal (Læknablaðið) 95, 259–266. [PubMed] [Google Scholar]
- Worldfengur 2015. The studbook of origin for the Icelandic horse. Retrieved on 28 April 2015 from www.worldfengur.com.