Abstract
Objective
To determine the prevalence, correlates and prognostic implications of anaemia and iron deficiency (ID) in patients with heart failure (HF) in Tanzania.
Method
This was a cross-sectional and prospective observational study conducted at Muhimbili National Hospital in Dar es Salaam, Tanzania. Patients were ≥18 years of age, with HF defined according to the Framingham criteria. The primary outcome was anaemia and the secondary outcome was a composite of hospitalisation for HF or all-cause mortality.
Results
A total of 401 HF patients (median age 56 years, IQR 41–67 years; women 51%) were included. The prevalence of anaemia was 57%. The overall prevalence of ID was 49% distributed as 69% versus 21% in subjects with and without anaemia (p<0.001). Normocytic anaemia was seen in 18% of the patients while none had macrocytic anaemia. The risk of having anaemia was positively associated with residency outside Dar es Salaam (OR 1.72 (95% CI 1.02 to 2.89); p=0.038), atrial fibrillation (4.12 (1.60 to 10.61); p=0.003), LVEF <45% (2.70 (1.57 to 4.67); p<0.001) and negatively (ORs per unit decrease) with creatinine clearance (0.98 (0.97 to 0.99); p=0.012) and total cholesterol (0.78 (0.63 to 0.98); p=0.029). One-year survival free from a composite endpoint was 70%. The presence of ID anaemia increased the likelihood for an event (HR 2.67; 95% CI 1.39 to 5.07; p=0.003), while anaemia without ID did not influence the risk.
Conclusions
ID anaemia was common in Tanzanian patients with HF and was independently associated with the risk for hospitalisation or death.
Background
Despite available standardised therapy, successful management of heart failure (HF) remains difficult in the presence of concomitant disorders. Anaemia, one comorbidity associated with refractory HF,1 is associated with increased risk of mortality.2 3 In high-income countries (HIC), the prevalence of anaemia in HF ranges from 4% to 55%4–6 depending on the study population and the level of haemoglobin (Hb) cut-off used to define anaemia. The prevalence in HF populations in Sub-Saharan Africa (SSA) is less well known but has been estimated to range between 12% and 64%.7–11 Further characterisation of its prevalence provides an opportunity for increasing the understanding of the role of anaemia in HF as well as options for treatment.
The relation between iron deficiency (ID) anaemia and HF has been explored in HIC.12 ID, not yet causing anaemia, is associated with a compromised prognosis in HF.12 13 Less is known about the role of ID and other types of anaemia in HF populations from SSA14 and needs to be explored in SSA populations where ID anaemia and chronic infections are prevalent.
The present study aims to determine the prevalence and prognostic implications of anaemia and ID in HF in Tanzania.
Methods
Study participants and investigations
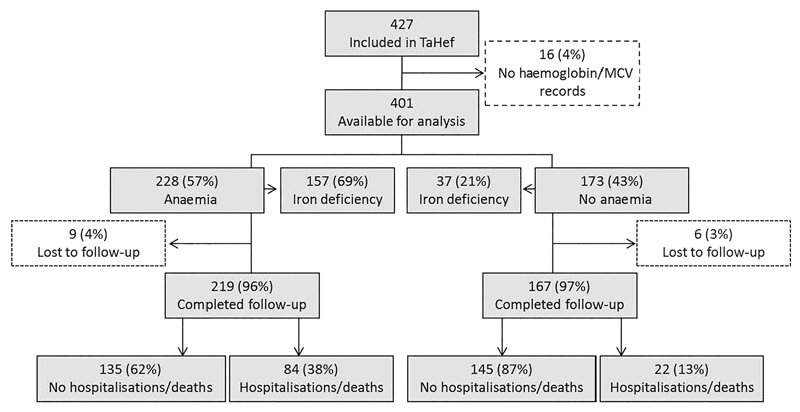
The Tanzania Heart Failure (TaHeF) study was both a cross-sectional and prospective observational study conducted at the Cardiovascular Centre of the Muhimbili National Hospital (MNH), a national referral centre for a population of 45 million people. A detailed description of the study design has been reported.7 In brief, 427 consecutive patients with HF according to the Framingham criteria15 were recruited from February 2012 to August 2013 (figure 1). The exclusion criteria were age <18 years and that a patient declined participation. Data on demographic, social, clinical, comorbidity, laboratory and echocardiographic variables were collected (table 1).
Figure 1.
Flow chart of patients included in The Tanzania Heart Failure (TaHef) study for anaemia and iron deficiency analyses. MCV, mean corpuscular volume.
Table 1.
Baseline characteristics of patients with heart failure (HF) (n=401)
| Characteristics | n | Per cent |
|---|---|---|
| Sex (female) | 206 | 51 |
| Median age, years (IQR) | 55 | 41–67 |
| Residence in Dar es Salaam | 255 | 64 |
| Formal education | 318 | 79 |
| Unemployed | 198 | 49 |
| Location of recruitment (inpatient) | 112 | 30 |
| Median HF duration, months (IQR) | 13 | 4–28 |
| ≥6 months | 270 | 67 |
| New York Heart Association (NYHA) class | ||
| II | 81 | 20 |
| III | 192 | 48 |
| IV | 128 | 32 |
| HF cause | ||
| Cardiomyopathies | 128 | 35 |
| Cardiomyopathies HF | 31 | 9 |
| Hypertensive HF | 162 | 44 |
| Rheumatic heart disease | 44 | 12 |
| Previous cardiovascular admission | 224 | 52 |
| Loss of appetite | 84 | 21 |
| Haematemesis | 9 | 2 |
| Peptic ulcer disease | 5 | 1 |
| Haematuria | 10 | 10 |
| Ever smoked | 53 | 13 |
| Ever took alcohol | 134 | 33 |
| Comorbidities | ||
| Diabetes | 50 | 12 |
| Atrial fibrillation | 67 | 17 |
| Stroke | 15 | 4 |
| Tuberculosis | 11 | 3 |
| Asthma or chronic obstructive lung disease (COPD) | 13 | 3 |
| HIV | 8 | 2 |
| Thyroid disease | 3 | 1 |
| Deep venous thrombosis | 4 | 1 |
| Pulmonary hypertension (N=362) | 73 | 19 |
| Chronic liver disease | 8 | 2 |
| Malignancy | 8 | 2 |
| Pharmacological treatments | ||
| ACE blocker or angiotensin receptor blocker | 393 | 92 |
| β blockers | 177 | 41 |
| Calcium channel blockers | 79 | 19 |
| Loop diuretics | 350 | 82 |
| Aldosterone antagonist | 308 | 72 |
| Digoxin | 165 | 39 |
| Nitrates | 274 | 65 |
| Hydralazine | 19 | 4 |
| Statins | 33 | 8 |
| Aspirin | 109 | 26 |
| Clopidogrel | 21 | 5 |
| Highly reactive antiretroviral therapy (HAART) | 9 | 2.2 |
| Warfarin | 11 | 3 |
| Clinical–physical examination | ||
| Body mass index (kg/m2) | 25.5 | 5 |
| Radial pulse (bpm) | 88.7 | 18 |
| Blood pressure (mm Hg) | ||
| Systolic blood pressure | 132 | 28 |
| Diastolic blood pressure | 84 | 16 |
| Mean arterial pressure | 100 | 19 |
| Laboratory/echocardiography | ||
| Haemoglobin (g/dL) | 12 | 2 |
| Men | 12 | 2 |
| Women | 12 | 2 |
| Mean corpuscular volume (fL) | 80 | 10 |
| Men | 80 | 11 |
| Women | 80 | 8 |
| Serum Na+ (mmol/L) (N=379) | 140 | 8 |
| Serum K+ (mmol/L) (N=379) | 4 | 1 |
| LVEF (%)(N=362) | 41 | 12 |
| LVEF <45% (N=362) | 249 | 68 |
Continuous variables are presented as mean±SD and categorical variables as numbers (n) and per cent (%) if not otherwise stated.
Full blood pictures were obtained by means of a haematology analyser (Beckman Coulter ACT5 DIFF), while total cholesterol, serum Na+, K+ and creatinine were analysed by the use of a chemistry analyser (Roche Cobas Integra 400 plus) at the National Central Pathology Laboratory at MNH. At echocardiography (General Electric Vivid 5 with a 2.5–5 MHz probe), the acquired data included pulmonary pressure and LVEF. The LVEF was measured by the 2D biplane Simpson technique and agreed by consensus between two senior cardiologists (JL and PK).
Patients were followed monthly in the outpatient clinic until August 2013. Those who missed their appointments or were hospitalised outside MNH were contacted through mobile phones. In the case of patients who died, the date of their status was obtained from a next of kin (figure 1).
Outcome measure and definitions
The primary outcome was anaemia and the secondary outcome was a composite of hospitalisation for HF or all-cause mortality (whichever came first). Based on the WHO, anaemia was defined as Hb <13 g/dL for men and <12 g/dL for women16 and graded as mild for levels between 10.0 and 12.9 g/dL (men)/11.9 g/dL (women), moderate for a Hb level between 7.0 and 9.9 g/dL and severe if <7 g/dL.
ID was defined as a mean corpuscular volume (MCV) of <80 femtolitres (fL) in the absence of serum iron markers.16 Subsequently, the ID and anaemia status of each participant was allocated into one of the following four categories: (1) No anaemia, no ID=absence of anaemia+MCV ≥80 fL; (2) No anaemia, yes ID=absence of anaemia+MCV <80 fL; (3) Yes anaemia, yes ID=presence of anaemia+MCV <80 fL and (4) Yes anaemia, no ID=presence of anaemia+MCV ≥80 fL.
Diagnostic criteria for hypertensive HF, rheumatic heart disease and cardiomyopathy were modified after the chronic HF cohort in the Heart of Soweto Study,17 according to the definitions previously published.7
Formal education was defined as primary school or above and unemployment as not on private or governmental official duties with a monthly income. Creatinine clearance was calculated according to the Cockcroft–Gault formula.18 Severe renal dysfunction was defined as creatinine clearance <30 mL/min. Pulmonary hypertension (PH) was defined as an echocardiographically estimated right ventricular systolic pressure >35 mm Hg.17
Statistical analyses
Data were entered and validated using OpenClinica software. Continuous variables were tested for normality distribution and reported as mean±SD or median (IQR) as appropriate. Categorical variables were compared using χ2 test. Selected continuous variables were categorised according to previously established clinically relevant criteria19 (table 3).
Table 3.
Univariate and multivariate analyses of correlates of anaemia in patients with heart failure (HF)
| Univariate | Multivariate | ||||||
|---|---|---|---|---|---|---|---|
| Exposure status | OR | 95% CI | p Value | OR | 95% CI | p Value | |
| Sex (female) | 0.75 | (0.51 to 1.11) | 0.151 | 0.67 | (0.40 to 1.27) | 0.131 | |
| Age (years) | 1.00 | (0.99 to 1.01) | 0.663 | 0.99 | (0.97 to 1.00) | 0.289 | |
| Residence outside Dar es Salaam | 1.55 | (1.03 to 2.34) | 0.036† | 1.72 | (1.02 to 2.89) | 0.043 | |
| Formal education | 0.95 | (0.58 to 1.55) | 0.841 | ||||
| Unemployed | 0.97 | (0.66 to 1.45) | 0.907 | ||||
| Recruitment site (inpatient) | 3.02 | (1.86 to 4.91) | <0.001† | 1.75 | (1.02 to 3.47) | 0.081 | |
| HF duration | |||||||
| <6 months | 1 | 0.332 | |||||
| ≥6 months | 0.81 | (0.53 to 1.24) | |||||
| NYHA class | 0.011† | ||||||
| II | 1 | 1 | |||||
| III | 1.47 | (0.87 to 2.47) | 0.97 | (0.49 to 1.90) | 0.936 | ||
| IV | 2.35 | (1.33 to 4.16) | 0.66 | (0.30 to 1.46) | 0.03 | ||
| HF cause | 0.520 | ||||||
| Cardiomyopathies | 1.30 | (0.80 to 2.06) | 0.96 | (0.49 to 1.90) | 0.913 | ||
| Ischaemic HF | 0.97 | (0.49 to 2.10) | 0.60 | (0.23 to 1.52) | 0.278 | ||
| Hypertensive HF | 1 | 1 | |||||
| Rheumatic heart disease | 1.55 | (0.92 to 3.10) | 1.36 | (0.54 to 3.40) | 0.516 | ||
| Previous cardiovascular admission | 1.31 | (0.88 to 1.95) | 0.183 | ||||
| Loss of appetite | 1.58 | (0.96 to 2.61) | 0.074† | 1.56 | (0.80 to 3.05) | 0.93 | |
| Haematemesis | 0.95 | (0.25 to 3.58) | 0.936 | ||||
| Peptic ulcer disease | 3.07 | (0.34 to 27.72) | 0.318 | ||||
| Haematuria | 1.01 | (0.22 to 4.58) | 0.988 | ||||
| Ever smoked | 1.73 | (0.93 to 3.18) | 0.083† | 1.04 | (0.561.89) | 0.904 | |
| Ever took alcohol | 1.04 | (0.68 to 1.58) | 0.862 | ||||
| Comorbidities | |||||||
| Diabetes | 1.44 | (0.77 to 2.71) | 0.251 | ||||
| Atrial fibrillation | 8.47 | (3.76 to 19.07) | <0.001† | 4.12 | (1.60 to 10.61) | 0.003 | |
| Stroke | 0.49 | (0.17 to 1.41) | 0.187 | ||||
| Tuberculosis | 3.51 | (0.75 to 16.47) | 0.111 | ||||
| Asthma/COPD | 0.88 | (0.29 to 2.67) | 0.824 | ||||
| HIV | 1.53 | (0.38 to 6.21) | 0.551 | ||||
| Pulmonary hypertension | 4.56 | (2.30 to 9.04) | <0.001† | 1.96 | (0.87 to 4.44) | 0.107 | |
| Pharmacological treatments | |||||||
| ACEI/ARB | 0.70 | (0.34 to 1.45) | 0.336 | ||||
| β blockers | 0.92 | (0.62 to 1.38) | 0.690 | ||||
| Loop diuretics | 0.97 | (0.58 to 1.61) | 0.897 | ||||
| Aspirin | 1.11 | (0.71 to 1.76) | 0.643 | ||||
| Warfarin | 0.28 | (0.07 to 1.05) | 0.059† | 0.47 | (0.07 to 3.08) | 0.430 | |
| HAART | 1.27 | (0.30 to 5.39) | 0.745 | ||||
| Clinical–physical examination | |||||||
| Body mass index (kg/m2) | 0.021† | ||||||
| <18.5 | 0.33 | (0.09 to 1.17) | 0.91 | (0.21 to 3.96) | 0.898 | ||
| 18.5–24.9 | 1 | 1 | |||||
| ≥25 | 0.22 | (1.34 to 16.23) | 0.86 | (0.19 to 3.77) | 0.844 | ||
| Mean arterial pressure (mm Hg) | 0.98 | (0.97 to 0.99) | 0.004† | 0.93 | (0.97 to 1.00) | 0.093 | |
| Laboratory/echocardiography | |||||||
| Creatinine clearance (mL/min) | 0.99 | (0.98 to 0.99) | 0.026† | 0.98 | (0.97 to 0.99) | 0.012 | |
| Serum cholesterol (mmol/L) | 0.77 | (0.64 to 0.91) | 0.003† | 0.78 | (0.63 to 0.98) | 0.029 | |
| Serum Na+ (mmol/L) | 0.152 | ||||||
| <135 | 0.66 | (0.35 to 1.25) | |||||
| 135–145 | 1 | ||||||
| >145 | 1.04 | (0.49 to 2.20) | |||||
| Serum K+ (mmol/L) | 0.192 | ||||||
| <3.5 | 1.35 | (0.74 to 2.46) | |||||
| 3.5–5 | 1 | ||||||
| >5 | 2.50 | (0.93 to 6.73) | |||||
| LVEF <45% | 3.40 | (2.15 to 5.37) | <0.001† | 2.70 | (1.57 to 4.67) | <0.001 | |
All variables with p value <0.10+ Age+ Sex+ HF causes were included in the multivariate analysis.
ACEI, ACE inhibitors; ARB, angiotensin receptor blocker; COPD, chronic obstructive lung disease; HAART, highly reactive antiretroviral therapy; NYHA, New York Heart Association.
The influence of selected characteristics on survival free from the secondary outcome (composite of hospitalisation due to HF or all-cause mortality) was assessed by Kaplan–Meier plots. Univariate and multivariate logistic regression analyses were used to determine the correlates of anaemia, while Cox proportional hazard was used to determine the effect of anaemia and ID on the composite endpoint. Additional restricted cubic spline models at three knots were used to determine the linear effect of quantitative variables on composite endpoint. In all multivariate analyses (logistic regression, Cox proportional hazard regression and cubic spline), variables with p value <0.10 and in addition age, sex and HF causes were included. Patients who were lost to follow-up (16 of 401) were excluded from the analyses of the secondary outcome. Interaction terms between exposure categories and potential effect modifiers (sex, LVEF, site of recruitment and New York Heart Association (NYHA) class) were created and assessed for significance in the multivariate models using a likelihood ratio test, estimates of the associations being stratified by the variable of interest. Analyses were performed using Stata V.13 and R statistical packages. A two-sided p value of 0.05 was considered statistically significant.
Results
Patient flow and baseline characteristics
Figure 1 shows the flow of patients recruited into the TaHeF, while baseline characteristics are presented in table 1. A total of 401 patients with HF met the inclusion criteria (women=206; 51%). The median age was 56 years, (IQR 41–67 years). Most patients (70%) were recruited from the outpatient clinic. Twenty per cent were in NYHA class II, 48% in class III and 32% in class IV. Atrial fibrillation was present in 17% of the patients and diabetes in 12%. PH was seen in 19% and severe renal dysfunction was present in 18% of the patients. The mean LVEF was 41±12% and 68% of the patients had a LVEF <45%. ACE inhibitors or angiotensin receptor blockers were used in 92%, loop diuretics in 82% and β blockers in 41%.
Prevalence of anaemia and ID
The total prevalence of anaemia was 57%, mild in 45%, moderate in 10% and severe in 2% of the patients. The overall prevalence of ID was 49%, 69% in subjects with anaemia versus 21% among those without anaemia (p<0.001). Normocytic anaemia was seen in 18% of all study patients while none had macrocytic anaemia. The prevalence distribution of anaemia and ID did not differ significantly in relation to sex (p=0.068) and NYHA class (p=0.101) except for LVEF (p<0.001) in which patients with LVEF <45% had a higher prevalence (table 2).
Table 2.
The prevalence distribution of anaemia and iron deficiency by sex, New York Heart Association (NYHA) class and LVEF
| Variable | No anaemia Iron deficiency |
Anaemia Iron deficiency |
Test statistic, p value | ||
|---|---|---|---|---|---|
| No, n (%) | Yes, n (%) | Yes, n (%) | No, n (%) | ||
| Sex | |||||
| Female | 70 (33) | 26 (12.6) | 78 (37.9) | 32 (15.5) | |
| Male | 66 (33.9) | 11 (5.6) | 79 (40.5) | 39 (20) | χ2=6.60, 0.086 |
| NYHA class | |||||
| II | 35 (43.2) | 9 (11.1) | 23 (28.4) | 14 (17.3) | χ2=10.6, 0.101 |
| III | 69 (35.9) | 17 (8.9) | 73 (38) | 33 (17.2) | |
| IV | 32 (24.9) | 11 (8.6) | 61 (47.7) | 24 (18.8) | |
| LVEF (%) | |||||
| ≥45 | 58 (50) | 13 (11.2) | 33 (28.5) | 12 (10.3) | χ2=28.8, <0.001 |
| <45 | 62 (24.9) | 17 (6.8) | 119 (47.8) | 51 (20.5) | |
Correlates of anaemia in HF
Univariate and multivariate correlates of anaemia in HF are presented in table 3 Age and gender did not correlate with anaemia. Significant independent predictors from multivariate model were residency outside Dar es Salaam, atrial fibrillation, creatinine clearance, total cholesterol and LVEF <45%. NYHA class, being inpatient, PH, body mass index and mean arterial pressure appeared to be risk markers of anaemia in the univariate analyses but did not remain after controlling for potential confounders.
Composite endpoint during follow-up
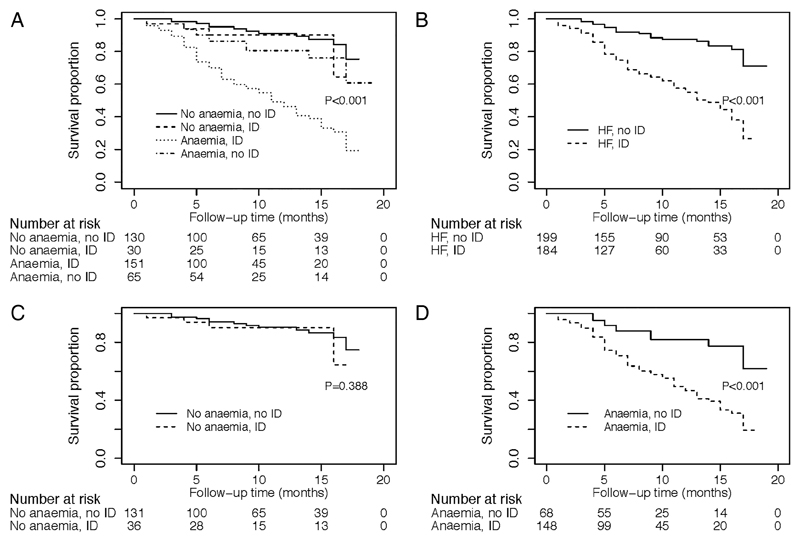
Complete follow-up was obtained on 385 (96%) of the 401 patients during the median follow-up time of 7.7 months. During this period, 106 (26%) of the patients experienced a composite of hospitalisation due to HF (n=40) or all-cause mortality (n=66). The overall 1-year survival free from a composite endpoint was 70%. The distribution of survival rates on the various forms of anaemia and ID is shown in figure 2A–D. The composite endpoint was most frequently experienced by patients with both anaemia and ID (figure 2A, D) and in those with HF and (+) ID versus HF and (−) ID (figure 2B).
Figure 2.
(A–D) Kaplan–Meier survival curves for composite endpoint by four different models of anaemia and iron deficiency.
Associations of anaemia and ID with the composite endpoint
Associations of anaemia and ID with the composite endpoint are presented in table 4. In the multivariate Cox proportional hazard, anaemia and ID were significantly associated with a higher HR (HR 2.67; 95% CI 1.39 to 5.07; p=0.003), but the association was not statistically significant for ID without anaemia or anaemia without ID.
Table 4.
HRs for the impact of anaemia and iron deficiency (ID) on the composite endpoint of hospitalisation due to heart failure (HF) or death in patients
| Exposure status | Univariate HR (95% CI) | p Value | Multivariate HR* (95% CI) | p Value |
|---|---|---|---|---|
| Anaemia and ID | ||||
| No anaemia, no ID | 1 | <0.001 | 1 | 0.001 |
| No anaemia, yes ID | 1.56 (0.61 to 4.03) | 1.68 (0.64 to 4.46) | 0.295 | |
| Yes anaemia, yes ID | 5.90 (3.37 to 10.32) | 2.70 (1.42 to 5.14) | 0.002 | |
| Yes anaemia, no ID | 1.78 (0.82 to 3.80) | 0.94 (1.42 to 2.15) | 0.894 |
Adjusted for sex, age, recruitment site, New York Heart Association class, HF cause, loss of appetite, atrial fibrillation, pulmonary hypertension, body mass index, mean arterial pressure, creatinine clearance and LVEF.
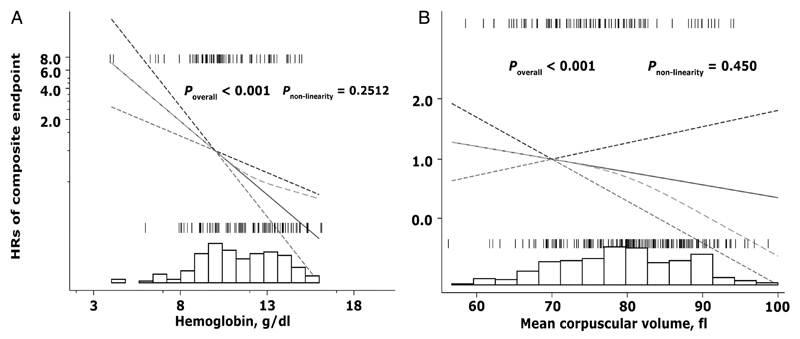
Restricted cubic spline models (figure 3) assessing the effects of change in Hb and MCV as continuous predictors of secondary outcome revealed no significant departures from linearity for Hb (p=0.25) or MCV (p=0.45). Fewer cases of composite endpoint were seen on the extremes values of Hb and MCV.
Figure 3.
Adjusted HRs for (A) haemoglobin (Hb) and (B) mean corpuscular volume (MCV) as independent predictors of composite endpoint among patients with heart failure. Results from Cox regression models showing the linear relationships of Hb and MCV with composite endpoint were overlaid on the plot of the spline model, demonstrating that the simpler parametric model adequately described the covariate effects. Dashed green lines represent the fit of the spline. Solid black lines represent the parametric Cox regression fit, while dotted lines represent pointwise 95% confidence limits for the parametric fits. All models were constrained to cross at a HR of unity. Distribution of patients across Hb and MCV are represented by underlaid histograms. Tick marks above the HR curve represent patients with composite endpoint, whereas tick marks below the curve represent patients without composite endpoint.
The relation of ID and anaemia with composite endpoint was modified by female gender (p for interaction=0.031) and site of recruitment (p for interaction=0.044).
Discussion
This study provides important information on the characterisation of anaemia and ID in HF in a SSA country. The presence of ID with anaemia had a deleterious prognostic impact warranting active detection and managing as a potential way to improve the prognosis of HF in Tanzania.
Anaemia was common in HF although a majority of the patients were relatively young (median age, 56 years), in a stable condition (48% in NYHA III) and ambulatory (70% outpatients). The current findings support a similar investigation from Uganda11 but were, in both studies, higher than in other SSA countries (13–41%).8 10 Compared with HIC (21–33%), anaemia was frequent, especially considering the about two decades higher age in such populations and that the prevalence of acute HF at study recruitment was more common in these countries.4–6 The high prevalence of ID and anaemia in the present study is reasonably explained by underlying malnutrition and chronic infections not commonly seen in HIC. Another possibility maybe a difference in pathophysiology of anaemia and HF in the setting of a mainly ischaemic background to HF as in HIC compared with non-ischaemic HF as in the present population. The current data show that HF was mainly due to hypertension (44%) in the present population. Hypertension in the Tanzanian general population has been estimated to be about 30%.20
Anaemia has a significant impact on mortality in HF in SSA compared with HIC7 and public health interventions are required to identify treatable causes, hence improving the prognosis. In SSA, it is not well documented whether the anaemia seen in HF is mainly due to ID or anaemia related to chronic disease (ACD). The present 69% prevalence of ID in anaemia is higher than previously reported proportions of 21–61%.12 13 21 22 To the best of our knowledge, this is the first study in SSA showing that ID anaemia is common in HF. However, further studies to determine ID anaemia in HF should be done using validated tools applicable to SSA where infections and inflammatory conditions are very common.
The overall prevalence of ID regardless of anaemia was 49%, that is, comparable with findings reported from a pooled analysis13 but is higher than in other studies (22% and 37%) in HIC12 21 and lower than in others5 22 23 in which ID was seen in 58–73% of the patients. Data on the epidemiology and pathophysiology of ID in HF in SSA are scarce. The variations in ID prevalence might be explained by the lack of standard definitions for ID in HF and differences in the population characteristics. It may also be reflected that only MCV was used to define ID. The use of serum ferritin and transferrin saturation might have provided a somewhat more accurate classification. However, in discriminant analysis, serum ferritin alone or in combination with other inflammatory markers provide only marginal improvement in distinguishing ID from ACD compared with MCV alone.24 25
Anaemia is known to be associated with poor LVEF, renal failure, ageing and gender.13 26 27 In our study, atrial fibrillation and high total cholesterol, but not age and gender, were additional independent risk factors for anaemia in HF suggesting a need for more understanding of the pathophysiological factors.
There is limited and partially conflicting information on the impact of ID on prognosis in HF patients, although poor outcome appears predominate. In most available studies,12 13 21 22 ID carried a higher risk of poor outcome in HF irrespective of the presence of anaemia. In contrast, Parikh et al23 found that ID was not associated with all-cause mortality or cardiovascular mortality in patients with self-reported HF. The present data show that ID anaemia but not anaemia alone or ID alone was an independent predictor of secondary outcome in HF and this was more pronounced in women compared with men. Four models were analysed taking into consideration both ID and anaemia and including both patients with preserved and reduced LV systolic function. It appeared that not only anaemia or ID alone, but, more importantly, ID anaemia had the worst prognostic implication. Further studies are needed to explain the prognostic variations contributed by each of the ID, ID anaemia and anaemia in HF.
A strength is that this report is based on a large and well-characterised population with minor loss to follow-up in a resource-limited country in which HF is mainly non-ischaemic. This expands the understanding of anaemia and ID in HF beyond that reported from HIC.
Study limitations
Information on serum iron, ferritin, total iron binding capacity, folate and cobalamin would have given further possibilities to characterise anaemia. The use of MCV to define ID might also overestimate its magnitude. MCV was used as a simple and inexpensive initial tool for identifying ID and should be useful for initiating treatment if resources are limited or biochemical markers are not readily available. As underlined, there are no standard criteria for the diagnosis of ID and its differentiating from ACDs in HF. The absence of natriuretic peptides and invasive ischaemic HF diagnosis/verification may also be seen as limitations, thus potentially underestimating the magnitude of ischaemic heart disease. Although the relatively low number of events may have underpowered the anaemia and ID model, it is still sufficient to identify at least some clinically important factors that may be intervened against. The study also still provides a comprehensive evaluation of ID and anaemia in HF in countries with limited resources.
Conclusion
ID with anaemia was common in the present Sub-Saharan HF population and had a negative prognostic implication. It is recommended that ID and anaemia are actively assessed and managed in HF.
Key messages.
What is already known on this subject?
-
►
The epidemiology and prognosis of anaemia in heart failure (HF) have been extensively studied in high-income countries (HIC) but less in Sub-Saharan Africa.
-
►
In HIC, iron deficiency (ID), not yet causing anaemia, is associated with a compromised prognosis in HF.
What might this add?
-
►
Anaemia and ID are common in HF in Tanzania.
-
►
Atrial fibrillation, creatinine clearance, total cholesterol and LVEF <45% are independent predictors of anaemia in HF in Tanzania.
-
►
ID anaemia, however, not anaemia alone or ID alone is an independent predictor of poor prognosis in HF.
How might this impact on clinical practice?
-
►
The presence of ID with anaemia has a deleterious prognostic impact warranting active detection and managing as a potential way to improve the prognosis of HF in Tanzania.
Acknowledgements
The authors thank MUHAS, Karolinska Institutet and MNH’s staff for collaboration in implementing this study. We would also like to thank all patients who contributed to these findings.
Funding The authors disclosed the receipt of the following financial support for the research, authorship and/or publication of this article: Unit of Cardiology, Karolinska Institutet; The Swedish Heart and Lung Foundation; Stockholm County Council; AM was supported by the Swedish International Development Cooperation Agency (SIDA) through a joint capacity strengthening programme with MUHAS and by the Germany exchange programme for education (DAAD).
Footnotes
Contributors All authors have made significant contributions to the design, execution, analysis and writing of this study and will share responsibility for the published material.
Competing interests None.
Ethics approval TaHeF was approved by the ethical review board of the Muhimbili University of Health and Allied Sciences (MUHAS). All patients provided written informed consent.
Provenance and peer review Not commissioned; externally peer reviewed.
Data sharing statement Patient-level data, full data set and statistical codes are available from the corresponding author. Informed consent for data sharing was not obtained, but the presented data are anonymised and risk of identification is low.
References
- 1.Mahfoud F, Kindermann I, Kindermann M, et al. [Comorbidity: anemia and heart failure] Dtsch Med Wochenschr. 2009;134:825–30. doi: 10.1055/s-0029-1220238. [DOI] [PubMed] [Google Scholar]
- 2.Groenveld HF, Januzzi JL, Damman K, et al. Anemia and mortality in heart failure patients a systematic review and meta-analysis. J Am Coll Cardiol. 2008;52:818–27. doi: 10.1016/j.jacc.2008.04.061. [DOI] [PubMed] [Google Scholar]
- 3.O’Meara E, Clayton T, McEntegart MB, et al. Clinical correlates and consequences of anemia in a broad spectrum of patients with heart failure: results of the Candesartan in Heart Failure: assessment of Reduction in Mortality and Morbidity (CHARM) Program. Circulation. 2006;113:986–94. doi: 10.1161/CIRCULATIONAHA.105.582577. [DOI] [PubMed] [Google Scholar]
- 4.Androne AS. Hemodilution is common in patients with advanced heart failure. Circulation. 2003;107:226–9. doi: 10.1161/01.cir.0000052623.16194.80. [DOI] [PubMed] [Google Scholar]
- 5.Nanas JN, Matsouka C, Karageorgopoulos D, et al. Etiology of anemia in patients with advanced heart failure. J Am Coll Cardiol. 2006;48:2485–9. doi: 10.1016/j.jacc.2006.08.034. [DOI] [PubMed] [Google Scholar]
- 6.Allen LA, Anstrom KJ, Horton JR, et al. Relationship between anemia and health care costs in heart failure. J Card Fail. 2009;15:843–9. doi: 10.1016/j.cardfail.2009.06.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Makubi A, Hage C, Lwakatare J, et al. Contemporary aetiology, clinical characteristics and prognosis of adults with heart failure observed in a tertiary hospital in Tanzania: the prospective Tanzania Heart Failure (TaHeF) study. Heart. 2014;100:1235–41. doi: 10.1136/heartjnl-2014-305599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karaye KM, Sani MU. Factors associated with poor prognosis among patients admitted with heart failure in a Nigerian tertiary medical centre: a cross-sectional study. BMC Cardiovasc Disord. 2008;8:16. doi: 10.1186/1471-2261-8-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Oyoo GO, Ogola EN. Clinical and socio demographic aspects of congestive heart failure patients at Kenyatta National Hospital, Nairobi. East Afr Med J. 1999;76:23–7. [PubMed] [Google Scholar]
- 10.Inglis SC, Stewart S, Papachan A, et al. Anaemia and renal function in heart failure due to idiopathic dilated cardiomyopathy. Eur J Heart Fail. 2007;9:384–90. doi: 10.1016/j.ejheart.2006.10.013. [DOI] [PubMed] [Google Scholar]
- 11.Kuule JK, Seremba E, Freers J. Anaemia among patients with congestive cardiac failure in Uganda—its impact on treatment outcomes. S Afr Med J. 2009;99:876–80. [PubMed] [Google Scholar]
- 12.Jankowska EA, Rozentryt P, Witkowska A, et al. Iron deficiency: an ominous sign in patients with systolic chronic heart failure. Eur Heart J. 2010;31:1872–80. doi: 10.1093/eurheartj/ehq158. [DOI] [PubMed] [Google Scholar]
- 13.Klip IT, Comin-Colet J, Voors AA, et al. Iron deficiency in chronic heart failure: an international pooled analysis. Am Heart J. 2013;165:575–82. doi: 10.1016/j.ahj.2013.01.017. [DOI] [PubMed] [Google Scholar]
- 14.Horwich TB, Fonarow GC, Hamilton MA, et al. Anemia is associated with worse symptoms, greater impairment in functional capacity and a significant increase in mortality in patients with advanced heart failure. J Am Coll Cardiol. 2002;39:1780–6. doi: 10.1016/s0735-1097(02)01854-5. [DOI] [PubMed] [Google Scholar]
- 15.McKee PA, Castelli WP, McNamara PM, et al. The natural history of congestive heart failure: the Framingham study. N Engl J Med. 1971;285:1441–6. doi: 10.1056/NEJM197112232852601. [DOI] [PubMed] [Google Scholar]
- 16.World Health Organisation. Nutritional anaemias: report of a WHO scientific group. WHO Tech Rep Ser. 1968;405:3–37. [PubMed] [Google Scholar]
- 17.Stewart S, Wilkinson D, Hansen C, et al. Predominance of heart failure in the heart of Soweto Study cohort: emerging challenges for urban African communities. Circulation. 2008;118:2360–7. doi: 10.1161/CIRCULATIONAHA.108.786244. [DOI] [PubMed] [Google Scholar]
- 18.Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16:31–41. doi: 10.1159/000180580. [DOI] [PubMed] [Google Scholar]
- 19.Damasceno A, Mayosi BM, Sani M, et al. The causes, treatment, and outcome of acute heart failure in 1006 Africans from 9 countries. Arch Intern Med. 2012;172:1386–94. doi: 10.1001/archinternmed.2012.3310. [DOI] [PubMed] [Google Scholar]
- 20.Edwards R, Unwin N, Mugusi F, et al. Hypertension prevalence and care in an urban and rural area of Tanzania. J Hypertens. 2000;18:145–52. doi: 10.1097/00004872-200018020-00003. [DOI] [PubMed] [Google Scholar]
- 21.Rangel I, Gonçalves A, de Sousa C, et al. Iron deficiency status irrespective of anemia: a predictor of unfavorable outcome in chronic heart failure patients. Cardiology. 2014;128:320–6. doi: 10.1159/000358377. [DOI] [PubMed] [Google Scholar]
- 22.Enjuanes C, Klip IT, Bruguera J, et al. Iron deficiency and health-related quality of life in chronic heart failure: results from a multicenter European study. Int J Cardiol. 2014;174:268–75. doi: 10.1016/j.ijcard.2014.03.169. [DOI] [PubMed] [Google Scholar]
- 23.Parikh A, Natarajan S, Lipsitz SR, et al. Iron deficiency in community-dwelling US adults with self-reported heart failure in the National Health and Nutrition Examination Survey III: prevalence and associations with anemia and inflammation. Circ Heart Fail. 2011;4:599–606. doi: 10.1161/CIRCHEARTFAILURE.111.960906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ahluwalia N, Lammi-Keefe CJ, Bendel RB, et al. Iron deficiency and anemia of chronic disease in elderly women: a discriminant-analysis approach for differentiation. Am J Clin Nutr. 1995;61:590–6. doi: 10.1093/ajcn/61.3.590. [DOI] [PubMed] [Google Scholar]
- 25.Suominen P, Punnonen K, Rajamäki A, et al. Serum transferrin receptor and transferrin receptor-ferritin index identify healthy subjects with subclinical iron deficits. Blood. 1998;92:2934–9. [PubMed] [Google Scholar]
- 26.Ezekowitz JA, McAlister FA, Armstrong PW. Anemia is common in heart failure and is associated with poor outcomes: insights from a cohort of 12 065 patients with new-onset heart failure. Circulation. 2003;107:223–5. doi: 10.1161/01.cir.0000052622.51963.fc. [DOI] [PubMed] [Google Scholar]
- 27.Silverberg DS, Wexler D, Blum M, et al. The use of subcutaneous erythropoietin and intravenous iron for the treatment of the anemia of severe, resistant congestive heart failure improves cardiac and renal function and functional cardiac class, and markedly reduces hospitalizations. J Am Coll Cardiol. 2000;35:1737–44. doi: 10.1016/s0735-1097(00)00613-6. [DOI] [PubMed] [Google Scholar]