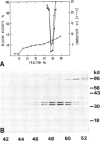
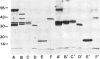
Abstract
Dictyostelium discoideum contains a soluble actin-binding protein that caps actin filaments at their fast growing ends. The purified protein consists of two subunits with 34 kd and 32 kd apparent mol. wts. Like similar proteins from Acanthamoeba and bovine brain the capping protein from D. discoideum acts in a Ca2+ -independent manner. It lacks severing activity as indicated by its inability to disrupt the stress fibers and the microfilament network in detergent-extracted cells. Two actin-binding proteins from a plasma membrane-enriched fraction were labeled with [125I]actin using a gel overlay technique. One of these proteins, with an apparent mol. wt. of 17 kd in SDS-polyacrylamide gels, has been purified from high-salt extracts, the other protein with an apparent mol. wt. of 31 kd has been purified from Triton X-100 extracted membranes. Monoclonal antibodies were raised against D. discoideum severin, α-actinin, the larger subunit of the capping protein, and the 17-kd membrane-associated protein. Immunoblotting of proteins from whole cell lysates showed that all these actin-binding proteins were present in both growth phase and aggregation-competent cells.
Keywords: actin-binding proteins, cytoskeleton, cell motility, Dictyostelium
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blikstad I., Sundkvist I., Eriksson S. Isolation and characterization of profilactin and profilin from calf thymus and brain. Eur J Biochem. 1980 Apr;105(3):425–433. doi: 10.1111/j.1432-1033.1980.tb04517.x. [DOI] [PubMed] [Google Scholar]
- Bolton A. E., Hunter W. M. The labelling of proteins to high specific radioactivities by conjugation to a 125I-containing acylating agent. Biochem J. 1973 Jul;133(3):529–539. doi: 10.1042/bj1330529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner S. L., Korn E. D. Spectrin-actin interaction. Phosphorylated and dephosphorylated spectrin tetramer cross-link F-actin. J Biol Chem. 1979 Sep 10;254(17):8620–8627. [PubMed] [Google Scholar]
- Bretscher A., Weber K. Villin is a major protein of the microvillus cytoskeleton which binds both G and F actin in a calcium-dependent manner. Cell. 1980 Jul;20(3):839–847. doi: 10.1016/0092-8674(80)90330-x. [DOI] [PubMed] [Google Scholar]
- Brier J., Fechheimer M., Swanson J., Taylor D. L. Abundance, relative gelation activity, and distribution of the 95,000-dalton actin-binding protein from Dictyostelium discoideum. J Cell Biol. 1983 Jul;97(1):178–185. doi: 10.1083/jcb.97.1.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown S. S., Yamamoto K., Spudich J. A. A 40,000-dalton protein from Dictyostelium discoideum affects assembly properties of actin in a Ca2+-dependent manner. J Cell Biol. 1982 Apr;93(1):205–210. doi: 10.1083/jcb.93.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlsson L., Nyström L. E., Sundkvist I., Markey F., Lindberg U. Actin polymerizability is influenced by profilin, a low molecular weight protein in non-muscle cells. J Mol Biol. 1977 Sep 25;115(3):465–483. doi: 10.1016/0022-2836(77)90166-8. [DOI] [PubMed] [Google Scholar]
- Claviez M., Pagh K., Maruta H., Baltes W., Fisher P., Gerisch G. Electron microscopic mapping of monoclonal antibodies on the tail region of Dictyostelium myosin. EMBO J. 1982;1(8):1017–1022. doi: 10.1002/j.1460-2075.1982.tb01287.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Condeelis J., Vahey M. A calcium- and pH-regulated protein from Dictyostelium discoideum that cross-links actin filaments. J Cell Biol. 1982 Aug;94(2):466–471. doi: 10.1083/jcb.94.2.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fechheimer M., Taylor D. L. Isolation and characterization of a 30,000-dalton calcium-sensitive actin cross-linking protein from Dictyostelium discoideum. J Biol Chem. 1984 Apr 10;259(7):4514–4520. [PubMed] [Google Scholar]
- Gerisch G. Chemotaxis in Dictyostelium. Annu Rev Physiol. 1982;44:535–552. doi: 10.1146/annurev.ph.44.030182.002535. [DOI] [PubMed] [Google Scholar]
- Gerisch G., Fromm H., Huesgen A., Wick U. Control of cell-contact sites by cyclic AMP pulses in differentiating Dictyostelium cells. Nature. 1975 Jun 12;255(5509):547–549. doi: 10.1038/255547a0. [DOI] [PubMed] [Google Scholar]
- Gerisch G., Keller H. U. Chemotactic reorientation of granulocytes stimulated with micropipettes containing fMet-Leu-Phe. J Cell Sci. 1981 Dec;52:1–10. doi: 10.1242/jcs.52.1.1. [DOI] [PubMed] [Google Scholar]
- Glenney J. R., Jr, Kaulfus P., Matsudaira P., Weber K. F-actin binding and bundling properties of fimbrin, a major cytoskeletal protein of microvillus core filaments. J Biol Chem. 1981 Sep 10;256(17):9283–9288. [PubMed] [Google Scholar]
- Glenney J. R., Jr, Kaulfus P., Weber K. F actin assembly modulated by villin: Ca++-dependent nucleation and capping of the barbed end. Cell. 1981 May;24(2):471–480. doi: 10.1016/0092-8674(81)90338-x. [DOI] [PubMed] [Google Scholar]
- Harris H. E., Weeds A. G. Platelet actin: sub-cellular distribution and association with profilin. FEBS Lett. 1978 Jun 1;90(1):84–88. doi: 10.1016/0014-5793(78)80303-2. [DOI] [PubMed] [Google Scholar]
- Hartwig J. H., Stossel T. P. Structure of macrophage actin-binding protein molecules in solution and interacting with actin filaments. J Mol Biol. 1981 Jan 25;145(3):563–581. doi: 10.1016/0022-2836(81)90545-3. [DOI] [PubMed] [Google Scholar]
- Hasegawa T., Takahashi S., Hayashi H., Hatano S. Fragmin: a calcium ion sensitive regulatory factor on the formation of actin filaments. Biochemistry. 1980 Jun 10;19(12):2677–2683. doi: 10.1021/bi00553a021. [DOI] [PubMed] [Google Scholar]
- Hinssen H. An actin-modulating protein from Physarum polycephalum. I. Isolation and purification. Eur J Cell Biol. 1981 Feb;23(2):225–233. [PubMed] [Google Scholar]
- Isenberg G., Aebi U., Pollard T. D. An actin-binding protein from Acanthamoeba regulates actin filament polymerization and interactions. Nature. 1980 Dec 4;288(5790):455–459. doi: 10.1038/288455a0. [DOI] [PubMed] [Google Scholar]
- Isenberg G., Ohnheiser R., Maruta H. 'Cap 90', a 90-kDa Ca2+-dependent F-actin-capping protein from vertebrate brain. FEBS Lett. 1983 Nov 14;163(2):225–229. doi: 10.1016/0014-5793(83)80824-2. [DOI] [PubMed] [Google Scholar]
- Jockusch B. M., Isenberg G. Interaction of alpha-actinin and vinculin with actin: opposite effects on filament network formation. Proc Natl Acad Sci U S A. 1981 May;78(5):3005–3009. doi: 10.1073/pnas.78.5.3005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilimann M. W., Isenberg G. Actin filament capping protein from bovine brain. EMBO J. 1982;1(7):889–894. doi: 10.1002/j.1460-2075.1982.tb01265.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korn E. D. Actin polymerization and its regulation by proteins from nonmuscle cells. Physiol Rev. 1982 Apr;62(2):672–737. doi: 10.1152/physrev.1982.62.2.672. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- MacLean-Fletcher S. D., Pollard T. D. Viscometric analysis of the gelation of Acanthamoeba extracts and purification of two gelation factors. J Cell Biol. 1980 May;85(2):414–428. doi: 10.1083/jcb.85.2.414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mannherz H. G., Goody R. S., Konrad M., Nowak E. The interaction of bovine pancreatic deoxyribonuclease I and skeletal muscle actin. Eur J Biochem. 1980 Mar;104(2):367–379. doi: 10.1111/j.1432-1033.1980.tb04437.x. [DOI] [PubMed] [Google Scholar]
- Maruta H., Isenberg G. Ca2+-dependent actin-binding phosphoprotein in Physarum polycephalum. II. Ca2+-dependent f-actin-capping activity of subunit a and its regulation by phosphorylation of subunit b. J Biol Chem. 1983 Aug 25;258(16):10151–10158. [PubMed] [Google Scholar]
- Matsudaira P. T., Burgess D. R. SDS microslab linear gradient polyacrylamide gel electrophoresis. Anal Biochem. 1978 Jul 1;87(2):386–396. doi: 10.1016/0003-2697(78)90688-7. [DOI] [PubMed] [Google Scholar]
- Reichstein E., Korn E. D. Acanthamoeba profilin. A protein of low molecular weight from Acanpthamoeba castellanii that inhibits actin nucleation. J Biol Chem. 1979 Jul 10;254(13):6174–6179. [PubMed] [Google Scholar]
- Snabes M. C., Boyd A. E., 3rd, Bryan J. Detection of actin-binding proteins in human platelets by 125I-actin overlay of polyacrylamide gels. J Cell Biol. 1981 Sep;90(3):809–812. doi: 10.1083/jcb.90.3.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snabes M. C., Boyd A. E., 3rd, Bryan J. Identification of G actin-binding proteins in rat tissues using a gel overlay technique. Exp Cell Res. 1983 Jun;146(1):63–70. doi: 10.1016/0014-4827(83)90324-5. [DOI] [PubMed] [Google Scholar]
- Snabes M. C., Boyd A. E., 3rd, Pardue R. L., Bryan J. A DNase I binding/immunoprecipitation assay for actin. J Biol Chem. 1981 Jun 25;256(12):6291–6295. [PubMed] [Google Scholar]
- Southwick F. S., Hartwig J. H. Acumentin, a protein in macrophages which caps the "pointed" end of action filaments. Nature. 1982 May 27;297(5864):303–307. doi: 10.1038/297303a0. [DOI] [PubMed] [Google Scholar]
- Spudich J. A., Watt S. The regulation of rabbit skeletal muscle contraction. I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J Biol Chem. 1971 Aug 10;246(15):4866–4871. [PubMed] [Google Scholar]
- Stadler J., Bordier C., Lottspeich F., Henschen A., Gerisch G. Improved purification and N-terminal amino acid sequence determination of the contact site A glycoprotein of Dictyostelium discoideum. Hoppe Seylers Z Physiol Chem. 1982 Aug;363(8):771–776. doi: 10.1515/bchm2.1982.363.2.771. [DOI] [PubMed] [Google Scholar]
- Swanson J. A., Taylor D. L. Local and spatially coordinated movements in Dictyostelium discoideum amoebae during chemotaxis. Cell. 1982 Feb;28(2):225–232. doi: 10.1016/0092-8674(82)90340-3. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Eldik L. J., Burgess W. H. Analytical subcellular distribution of calmodulin and calmodulin-binding proteins in normal and virus-transformed fibroblasts. J Biol Chem. 1983 Apr 10;258(7):4539–4547. [PubMed] [Google Scholar]
- Wang L. L., Bryan J. Isolation of calcium-dependent platelet proteins that interact with actin. Cell. 1981 Sep;25(3):637–649. doi: 10.1016/0092-8674(81)90171-9. [DOI] [PubMed] [Google Scholar]
- Weeds A. Actin-binding proteins--regulators of cell architecture and motility. Nature. 1982 Apr 29;296(5860):811–816. doi: 10.1038/296811a0. [DOI] [PubMed] [Google Scholar]
- Yamamoto K., Pardee J. D., Reidler J., Stryer L., Spudich J. A. Mechanism of interaction of Dictyostelium severin with actin filaments. J Cell Biol. 1982 Dec;95(3):711–719. doi: 10.1083/jcb.95.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]