Abstract
Context:
Fibroblast growth factor 21 (FGF21) secretion has been shown to respond directly to carbohydrate consumption, with glucose, fructose, and sucrose all reported to increase plasma levels of FGF21 in rodents and humans. However, carbohydrate consumption also results in secretion of insulin.
Objective:
The aim of this study was to examine the combined and independent effects of hyperglycemia and hyperinsulinemia on total and bioactive FGF21 in the postprandial period in humans, and determine whether this effect is attenuated in conditions of altered insulin secretion and action.
Methods:
Circulating glucose, insulin, total and bioactive FGF21, and fibroblast activation protein were measured in adults with and without type 2 diabetes (T2D) following an oral glucose tolerance test (OGTT), and under a series of insulin and glucose clamp conditions and following high-fat diet in healthy adults.
Results:
Circulating total and bioactive FGF21 levels responded acutely to OGTT, and their ratio was attenuated in T2D patients with reduced postprandial insulin response. The clamp studies revealed that insulin but not glucose accounts for the postprandial rise in FGF21. Finally, there was an attenuated rise in FGF21 in response to a high-fat dietary intervention that is known to alter insulin-stimulated substrate utilization in metabolically active tissues.
Conclusions:
Insulin rather than glucose per se increases total and bioactive FGF21 in the postprandial period in adult humans. Understanding the impact of T2D on bioactive FGF21 will have a significant effect upon the efficacy of therapeutic agents designed to target the FGF21 pathway.
Circulating FGF21 levels respond acutely to insulin but not glucose. A high-fat diet impairs this response. The postprandial rise in the ratio of bioactive to total FGF21 is attenuated in T2D.
Fibroblast growth factor (FGF)21 is a 181-amino-acid (aa) hormone with significant potential for the treatment of type 2 diabetes (T2D) (1, 2). The therapeutic actions of FGF21 require binding of its N terminus to the tyrosine kinase receptor, FGF receptor 1, and its C terminus to the coreceptor βklotho (KLB), forming the FGF21 receptor complex (FGF receptor 1-KLB) (3–5). Circulating FGF21 levels are driven primarily by hepatic production (6, 7), whereas other tissues, including skeletal muscle and fat, are able to contribute in response to specific stimuli (8). In the obese and T2D states, FGF21 is elevated in the plasma, albeit demonstrating significant interindividual variation (9–12). Furthermore, increased circulating concentrations of FGF21 are associated with conditions characterized by increased circulating lipids and abnormal hepatic metabolism (13–15).
Recently, FGF21 secretion has been shown to respond directly to carbohydrate consumption, with glucose, fructose, and sucrose all reported to increase plasma levels of FGF21 in rodents and humans (6, 16–18). Studies in rodents suggested increased hepatic expression of carbohydrate-responsive element-binding protein (ChREBP) as a mechanism for the increased plasma levels of FGF21 following fructose ingestion (19). However, the mechanism by which glucose regulates FGF21 levels in the postprandial period in adult humans has not been explored. Interestingly, in subjects diagnosed with metabolic syndrome (MetS), a greater increase in circulating FGF21 was reported when compared with healthy controls following oral glucose consumption. However, as this was accompanied with a greater blood glucose and insulin response in these subjects (14), it is difficult to disentangle whether the induction of FGF21 following a glucose load is a result of the associated hyperglycemia and/or hyperinsulinemia. Furthermore, FGF21 has a relatively short half-life (1 to 2 hours) and circulates in inactive [3 to 181 aa (16% to 30%), 5 to 181 aa (10% to 25%)] and bioactive [1 to 171 aa (10% to 34%)] forms in healthy participants (3, 20–22). The inactive form of FGF21 is generated via proteolytic cleavage of the C terminus by fibroblast activation protein (FAP)α, a serine dipeptidase and member of the S9 family proteases (23). Numerous studies have examined the impact of T2D on total FGF21 levels (10, 24), although it is not clear what effect T2D has on the bioactive form of FGF21. Understanding the impact of T2D on bioactive rather than total FGF21 will have a significant effect upon the efficacy of therapeutic agents designed to target the FGF21 pathway.
Therefore, the aim of the current study was to investigate the combined and independent effects of hyperglycemia and hyperinsulinemia on FGF21 in the postprandial period in adult humans, and to determine whether this effect is attenuated in conditions of altered insulin secretion and action. Furthermore, we assessed the levels of total and bioactive FGF21, in addition to FAPα in these states. Our findings suggest that FGF21 is an insulin-dependent postprandially regulated hormone in adult humans.
Research Design and Methods
Subjects
Diabetes study
Seven control subjects [age 41.9 ± 4.0 years; body mass index (BMI) 31.2 ± 1.5 kg/m2] and seven patients with T2D (age 48.3 ± 2.3 years; BMI 28.5 ± 1.3 kg/m2) controlling their T2D with diet alone (n = 2) or metformin (n = 5; dose 1300 ± 300 mg/d) participated. Patients were excluded if they were taking antihyperglycemic medication other than metformin or presented with any secondary complications of T2D. Control subjects were excluded at a screening oral glucose tolerance test (OGTT) visit if their fasting blood glucose was >5.6 mmol/L or 2-hour blood glucose was >7.0 mmol/L.
Clamp studies
Six healthy, nonobese male individuals (age 23.2 ± 2.4 years; BMI 23.9 ± 1.0 kg/m2) participated in the hyperglycemic/hyperinsulinemic studies, whereas nine healthy males (age 26.1 ± 2.8 years, and BMI 23.4 ± 1.1 kg/m2) were recruited for the high-fat (HF) study.
In all studies, subjects were informed of all procedures and risks associated with the experiments prior to obtaining written informed consent. All procedures were performed according to the Declaration of Helsinki and approved by the University of Nottingham Medical School Ethics Committee (clamp studies) and the local National Health System Research Ethics Committee (diabetes study).
Experimental protocols
Diabetes study
All subjects underwent an OGTT performed after consumption of an isocaloric diet for 72 hours. They also refrained from strenuous exercise for 48 hours before the visit. Patients with T2D who were using metformin did not take metformin on the morning of the trial. Subjects attended the laboratory after an overnight fast, having consumed a standardized meal the evening before, comprised of 55% carbohydrate, 30% fat, and 15% protein. At the start of the 2-hour OGTT, subjects consumed 75 g dextrose dissolved in 300 mL water prepared on the morning of the trial. Arterialized blood samples were obtained from a dorsal hand vein of one arm (placed in a hot-air box maintained at 50 to 55°C) at baseline and every 30 minutes during the OGTT.
Clamp studies
On three randomized occasions, 2 weeks apart, after an overnight fast, all six subjects underwent the following 4-hour clamps: (1) hyperinsulinemic (78 ± 3 mU/L)-hyperglycemic (10.1 ± 0.1 mmol/L) clamp (HIHG trial); (2) euinsulinemic (7.3 ± 1.1 mU/L)-hyperglycemic (10.4 ± 0.1 mmol/L) clamp (EIHG trial); and (3) hyperinsulinemic (76 ± 2 mU/L)-euglycemic (4.4 ± 0.1 mmol/L) clamp (HIEG trial). On two occasions, infusion of human soluble insulin (Actrapid; Novo, Copenhagen, Denmark) into an antecubital vein on one arm commenced at a rate of 50 mU m−2 min−1 and continued throughout each clamp, with 20% dextrose infused at a variable rate to maintain blood glucose concentrations at either euglycemic (HIEG trial) or hyperglycemic (HIHG trial) levels, respectively. On both occasions, infusion of somatostatin at 500 mg/h (to inhibit endogenous insulin secretion) and replacement infusion of glucagon (0.7 ng kg−1 min−1) started 30 minutes before dextrose infusion. On a third occasion, 20% dextrose was infused at a variable rate to maintain blood glucose concentration at the designated level (EIHG trial). Infusion of somatostatin at 500 mg/h and basal replacement infusions of glucagon (0.7 ng kg−1 min−1) and insulin (5 mU m−2 min−1) started 30 minutes before dextrose infusion. On all occasions, arterialized blood samples were obtained from a dorsal vein from the nondominant hand at baseline and every 5 minutes for the determination of blood glucose concentrations, and every 60 minutes for hormone concentrations.
HF study
All subjects underwent two 7-day trials, at least 2 weeks apart, in a randomized crossover design. On each occasion, subjects consumed for 6 days either a HF (76.7 ± 0.4% energy as fat) or normal diet [(control) 32.3 ± 0.7% fat]. On day 7, after an overnight fast, subjects underwent a 4-hour hyperinsulinemic (control: 71.8 ± 3.5 and HF: 70.0 ± 3.5 mU/L)-euglycemic (4.5 ± 0.2 mmol/L) clamp, as described previously. Arterialized blood samples were obtained at baseline and every 5 minutes for the determination of blood glucose concentration, and before and after each clamp for the determination of hormone concentrations.
Blood analysis.
In all studies, blood glucose concentrations were determined using a Yellow Springs Instrument Analyzer (YSI 2300 STAT PLUS; Yellow Springs Instruments, Yellow Springs, OH). Serum was separated by centrifugation (15 minutes at 3000g) and analyzed for insulin concentrations by radioimmunoassay (Diagnostics Products, Llanberis, Wales, UK), and total FGF21 (Biovendor, Research and Diagnostics Products, Brno, Czech Republic), bioactive FGF21 (Eagle Biosciences, Nashua, NH), and FAPα (Abcam, Cambridge, UK) concentrations by enzyme-linked immunoassays.
Statistics
All data are expressed as means ± standard error of the mean. Data from each study were analyzed via two-way analysis of variance and Tukey’s post hoc test. P < 0.05 was considered significant.
Results
Baseline blood measurements
T2D subjects had higher fasting blood glucose concentrations when compared with nondiabetic controls (6.7 ± 0.4 versus 4.4 ± 0.2 mmol/L; P < 0.01), but similar fasting insulin levels (12.0 ± 1.0 versus 13.9 ± 2.3 mU/L) (Table 1). Despite higher fasting levels of FAPα in T2D patients compared with controls (168.4 ± 12.1 versus 134.3 ± 11.8 ng/mL; P < 0.05), there was no difference in fasting levels of total or bioactive FGF21 (Table 1). No correlation was observed between baseline levels of FAPα and FGF21 (bioactive, total or their ratio).
Table 1.
Anthropometric Characteristics and Baseline Blood Biochemistry Data
| Control | T2D | |
|---|---|---|
| Subjects | n = 7 | n = 7 |
| Age (y) | 41.9 ± 4.0 | 48.3 ± 2.3 |
| BMI (kg/m2) | 31.2 ± 1.5 | 28.5 ± 1.3 |
| Fasting plasma glucose (mmol/L) | 4.4 ± 0.2 | 6.7 ± 0.4a |
| Fasting plasma insulin (mU/L) | 13.9 ± 2.3 | 12.0 ± 1.0 |
| Fasting total FGF21 (pg/mL) | 99.7 ± 16.2 | 117.0 ± 28.1 |
| Fasting bioactive FGF21 (pg/mL) | 68.3 ± 18.5 | 53.5 ± 23.1 |
| Fasting FAPα (ng/mL) | 134.3 ± 11.8 | 168.4 ± 12.1b |
Values are means ± standard error of the mean obtained in the fasted state.
P < 0.01 from Con (n = 7).
P < 0.05 from Con (n = 7).
Blood glucose and insulin responses to OGTT
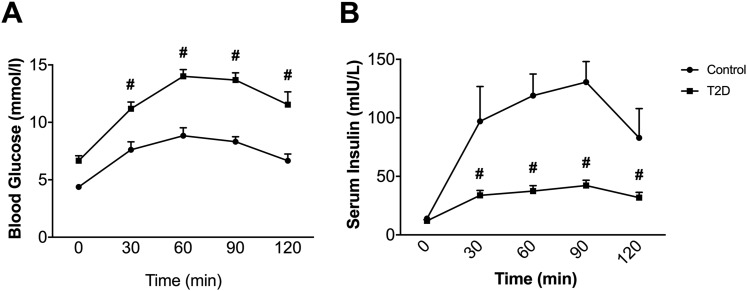
In nondiabetic and T2D subjects, oral administration of a 75 g dextrose solution increased both circulating glucose [Fig. 1(A)] and insulin [Fig. 1(B)]. Despite a higher increase in circulating glucose, the effect on insulin secretion was significantly attenuated in the T2D group (Fig. 1). To determine whether a postprandial rise of FGF21 occurred in response to OGTT, we measured circulating FGF21 levels (total and bioactive) in both groups.
Figure 1.
Effect of oral administration of dextrose on circulating glucose (A) and insulin (B). Data are means ± standard error of the mean; #P < 0.05 from control (n = 7).
FGF21 is a postprandial hormone that is impaired in T2D
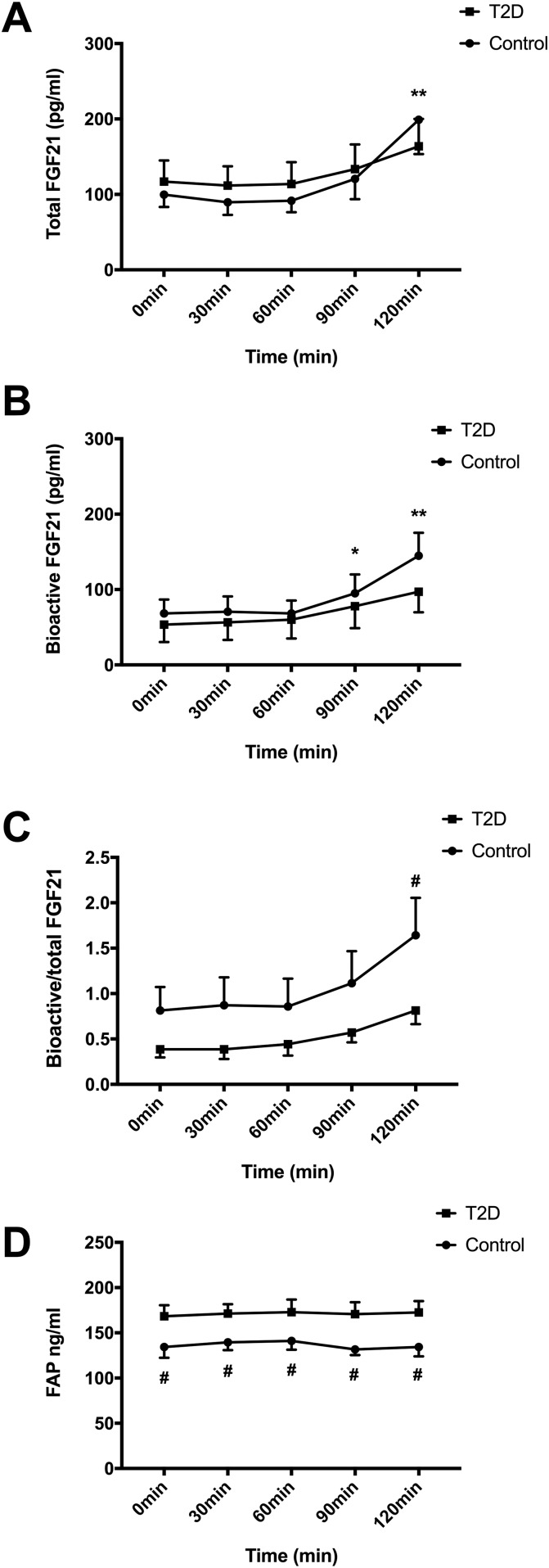
In contrast to the rapid elevation of circulating glucose and insulin, total FGF21 was not increased (P < 0.01) until 120 minutes [Fig. 2(A)]. Notably, this late induction of total FGF21 was mirrored by an increase (P < 0.01) in the bioactive form of FGF21 after 90 and 120 minutes [Fig. 2(B)]. There was no effect of dextrose ingestion on circulating FAPα throughout the OGTT [Fig. 2(D)]. Taken together, these data indicated that the postprandial rise in total FGF21 following dextrose consumption is accompanied by a proportional increase in bioactive FGF21 and elevated glucose and insulin levels.
Figure 2.
Effect of OGTT on circulating total (A) and bioactive (B) FGF21, the ratio of bioactive to total FGF21 (C) and FAPα (D). Data are means ± standard error of the mean; *P < 0.05 from 0 minutes; **P < 0.01 from 0 minutes; #P < 0.05 from T2D (n = 7).
Although there was no significant difference in the postprandial rise in total and bioactive FGF21 between the nondiabetic (control) and T2D groups [Fig. 2(A) and 2(B)], the ratio of bioactive to total FGF21 responded differently over time (P < 0.05) with a significant increase observed in the control subjects that was impaired in patients with T2D [Fig. 2(C)]. In line with the lower ratio of bioactive to total FGF21, circulating FAPα concentrations remained higher (P < 0.05) in T2D subjects throughout the OGTT [Fig. 2(D)]. Thus, in this work we report that the normal postprandial rise in the ratio of bioactive to total FGF21 is attenuated in T2D patients, and this effect is associated with both reduced postprandial insulin concentrations and increased circulating levels of the protease FAPα. However, based on our OGTT data, we could not distinguish whether the induction of FGF21 following dextrose consumption was a result of increased levels of circulating glucose and/or insulin.
FGF21 is an insulin-dependent postprandial hormone
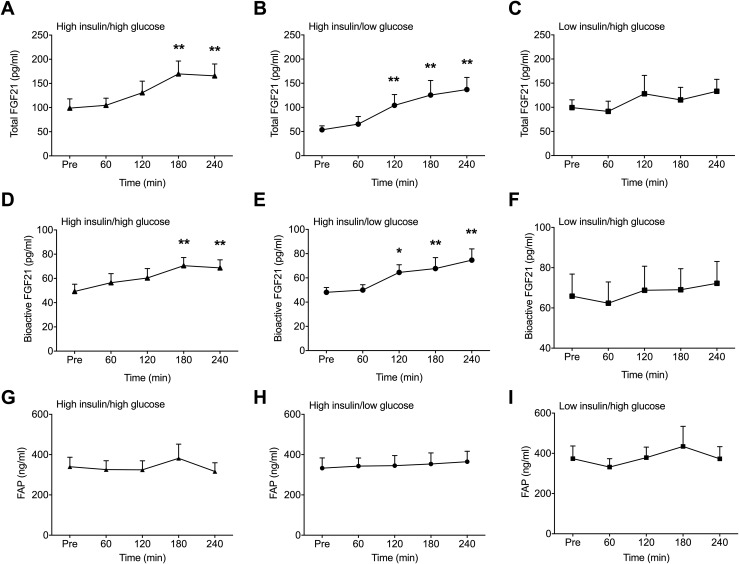
To determine directly whether glucose and/or insulin account for the postprandial rise in circulating FGF21 following dextrose consumption, we assessed FGF21 in blood from healthy volunteers collected during 4 hours of HIHG, HIEG, and EIHG clamps. The magnitude of hyperinsulinemia and hyperglycemia achieved in those clamps was within the range of blood glucose and insulin levels typically seen after oral carbohydrate administration. Consistent with the OGTT data in nondiabetic volunteers, there was a clear time-dependent effect of the HIHG clamp on FGF21. Total circulating levels of FGF21 and its bioactive form were increased (P < 0.01) after 3 hours of infusion [Fig. 3(A) and 3(D)]. There was also an increase (P < 0.01) in total and bioactive FGF21 following the HIEG clamps [Fig. 3(B) and 3(E)]. In contrast, there was no effect of the EIHG clamps on total or bioactive FGF21 levels [Fig. 3(C) and 3(F)]. There was no difference between trials in the bioactive to total FGF21 ratio or circulating FAPα [Fig. 3(G)–3(I)]. Taken together with results collected in nondiabetic and diabetic patients during the OGTT, these data suggest that the rise in total and bioactive FGF21 that occurs following dextrose ingestion is facilitated by the corresponding increase in circulating insulin levels.
Figure 3.
Effect of HIHG, HIEG, and EIHG clamps on circulating total (A–C) and bioactive FGF21 (D–F) and FAPα (G–I). Data are means ± standard error of the mean; *P < 0.05 from Pre; **P < 0.01 from Pre (n = 6).
Insulin-mediated secretion of FGF21 is impaired following HF feeding
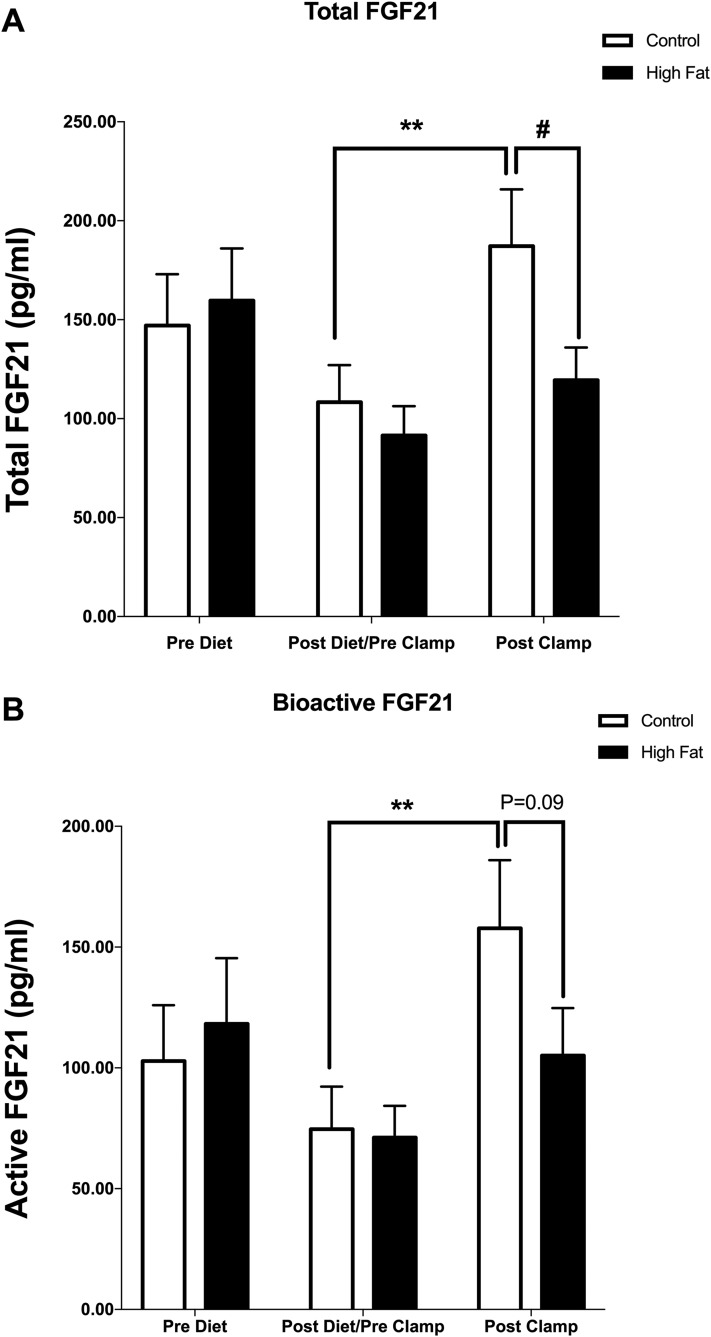
Short-term HF in humans has been shown to alter inulin-stimulated substrate utilization in metabolically active tissues without inducing peripheral insulin resistance (25, 26). Thus, to investigate whether the rise in circulating FGF21 in response to insulin was altered under such conditions, we analyzed the levels of total and bioactive FGF21, and FAPα, before and after a 4-hour HIEG clamp in human subjects following a control or HF for 6 consecutive days. As expected, insulin-stimulated carbohydrate oxidation rate was 20% lower and fat oxidation 60% higher in HF compared with control, and there was no effect of treatment on peripheral glucose uptake. HF feeding per se (prior to performing the insulin clamps) did not alter fasting levels of either total or bioactive FGF21 (Fig. 4) nor FAPα (data not shown). In line with the clamp studies described previously, subjects receiving the control diet demonstrated a robust induction of FGF21 in response to insulin (P < 0.01). However, following the HF diet, the response to insulin clamp was impaired resulting in lower (P < 0.05) total FGF21 levels [Fig. 4(A)] and a tendency (P = 0.09) for lower bioactive form [Fig. 4(B)] when compared with the control diet. The ratio of bioactive to total FGF21 was unaffected.
Figure 4.
Circulating total (A) and bioactive (B) FGF21 before (Pre Diet) and after (Post Diet) 6 days of either a HF or control diet. The postdiet values were obtained in the fasting state on day 7 immediately before a 4-hour insulin clamp and were also used as baseline values (Pre Clamp) to assess the effect of treatment on insulin-stimulated secretion of total and bioactive FGF21 (Post Clamp data). Data are means ± standard error of the mean; **P < 0.01 from Pre; #P < 0.05 from control (n = 9).
Discussion
This study demonstrates that circulating bioactive FGF21 levels respond acutely to changes in insulin per se, rather than glycemia, in the postprandial period. In particular, there were four main findings. First, we found that fasting total and bioactive FGF21 levels are similar in nondiabetic and T2D subjects, despite higher fasting levels of FAPα in T2D patients. Second, the normal postprandial increase in the ratio of bioactive to total FGF21 is impaired in T2D patients with attenuated insulin response to OGTT when compared with nondiabetic individuals. Third, we demonstrate, in a series of clamp studies that are consistent with the OGTT data, that there is a clear time-dependent effect of the HIEG clamp on FGF21 (both total and bioactive). This effect is lost in the EIHG clamp. These data suggest the rise in total and bioactive FGF21 that occurs following dextrose ingestion is a consequence of the increase in insulin secretion. Finally, in response to a HF dietary intervention that is known to alter insulin-stimulated substrate utilization, the response in circulating FGF21 is attenuated. Confirming the mechanism by which insulin regulates secretion of FGF21 and the subsequent tissue-specific actions of FGF21 will require further investigation.
Recently, excess dietary carbohydrate had been shown to increase FGF21 secretion in healthy humans (18). Furthermore, fructose ingestion briefly increased plasma total FGF21 concentration at 2 hours, returning to baseline within 5 hours (6). This increase was correlated with elevated levels of circulating glucose and insulin. Baseline levels of total FGF21 have been previously shown to be elevated in subjects with MetS that also exhibit exaggerated glucose and insulin responses to an oral glucose load (24).
Studies in rodents have also implicated carbohydrates in FGF21 induction; 12-hour sucrose feeding (following a 24-hour fast) was shown to induce insulin and FGF21 messenger RNA in rat liver (27). Although glucose-induced increases in FGF21 gene expression in hepatocytes and mouse liver were mediated via the transcription factor ChREBP (a notion supported by no change in FGF21 content in plasma of ChREBP knockout mice in response to various sugars), it was suggested that glucose-stimulated FGF21 messenger RNA expression may require insulin action (28). In support of this notion, FGF21 expression was upregulated by insulin in a phosphatidylinositol 3-kinase–dependent manner in cultured C2C12 myocytes and 3T3-L1 adipocytes (29). Our data demonstrate the importance of insulin, rather than glucose per se, in regulating secretion of FGF21 in adult humans. Interestingly, the normal postprandial increase in the ratio of bioactive to total FGF21 is impaired in T2D patients with attenuated insulin response to OGTT when compared with nondiabetic individuals. Whether this is a consequence of reduced insulin levels per se or resistance to the action of insulin requires further investigation. Although FGF21 is synthesized in multiple organs and can act on multiple tissues in either a paracrine or endocrine fashion, the major site of FGF21 production is the liver (7). Therefore, it is possible that hepatic insulin resistance in subjects with MetS, nonalcoholic fatty liver disease, or diabetes may play an important role in the regulation of FGF21 by insulin, which may also explain the significant interindividual variation in its levels in those populations.
FAP has an extensive tissue expression profile in addition to circulating in the blood of mice, nonhuman primates, and humans (22, 23, 30, 31). The importance of FAP’s actions was demonstrated in vitro, in which deletion of more than four amino acids from the C terminus of FGF21 significantly attenuates KLB-binding affinity, and in vivo, in which the metabolic actions of FGF21 are diminished in the absence of KLB binding (7, 16, 32–35). Interestingly, FAP is homologous (48% sequence identity) to dipeptidyl peptidase-4 (36), the therapeutic target of the antidiabetic dipeptidyl peptidase-4 inhibitor (37). However, whereas FAPα activity has been shown to increase in liver and plasma from patients with liver disease (23), the impact of MetS on FAPα activity, and hence FGF21 biology, remains largely unknown. In this study, we demonstrate that, in T2D, levels of FAPα are increased when compared with nondiabetic controls. Interestingly, Talabostat (TB), a known FAP inhibitor, reduced body weight and food intake, increased energy expenditure, and improved glucose tolerance and insulin sensitivity in diet-induced obese mice, in which total and bioactive plasma FGF21 were observed to be elevated. Interestingly, these effects were attenuated in FGF21 knockout animals (38). TB was previously pursued as an anticancer treatment and was found to be safe to support repeated dosing in human clinical trials (39). Although TB is not selective to FAP, further studies in humans are required to evaluate the use of FAP inhibitors as relevant treatment strategy in T2D patients, particularly as FGF21 improves MetS in humans (1, 40).
In summary, we used a physiological and dietary strategy in human subjects that revealed a stimulatory effect of insulin on FGF21 secretion. We demonstrate that dextrose ingestion acutely and robustly increases total and bioactive FGF21 in humans. The normal postprandial rise in the ratio of bioactive to total FGF21 is impaired in T2D patients that have attenuated insulin response to OGTT. The effect of insulin, rather than glucose per se, on FGF21 was confirmed in a series of insulin and glucose clamp experiments, in addition to the HF study, suggesting that FGF21 secretion is regulated by insulin and is therefore a postprandial hormone in adult humans.
Acknowledgments
Financial Support: This work was supported by the Biotechnology and Biological Sciences Research Council (UK) [grant numbers BB/M001555/1, BB/M021629/1]; and the Diabetes UK [grant number 13/0004659].
Author Contributions: R.J.S., J.E.L., L.N., C.J.G., F.B.S., and K.T. designed the study, carried out the experiments, and researched data (with additional input by T.B., D.P.S., C.C.C., and J.W.P.). J.E.L., R.J.S., F.J.P.E., and K.T. wrote the manuscript. All of the authors reviewed and edited the manuscript.
Acknowledgments
Disclosure Summary: R.J.S., A.C.A., C.C.C., T.B., D.P.S., and J.W.P. are employees of Eli Lilly and Company. All other authors have nothing to disclose.
Footnotes
- aa
- amino acid
- BMI
- body mass index
- ChREBP
- carbohydrate-responsive element-binding protein
- EIHG
- euinsulinemic-hyperglycemic
- FAP
- fibroblast activation protein
- FGF
- fibroblast growth factor
- HF
- high fat
- HIEG
- hyperinsulinemic-euglycemic
- HIHG
- hyperinsulinemic-hyperglycemic
- KLB
- βklotho
- MetS
- metabolic syndrome
- OGTT
- oral glucose tolerance test
- T2D
- type 2 diabetes
- TB
- Talabostat.
References
- 1.Gaich G, Chien JY, Fu H, Glass LC, Deeg MA, Holland WL, Kharitonenkov A, Bumol T, Schilske HK, Moller DE. The effects of LY2405319, an FGF21 analog, in obese human subjects with type 2 diabetes. Cell Metab. 2013;18(3):333–340. [DOI] [PubMed] [Google Scholar]
- 2.Gimeno RE, Moller DE. FGF21-based pharmacotherapy--potential utility for metabolic disorders. Trends Endocrinol Metab. 2014;25(6):303–311. [DOI] [PubMed] [Google Scholar]
- 3.Kharitonenkov A, Wroblewski VJ, Koester A, Chen YF, Clutinger CK, Tigno XT, Hansen BC, Shanafelt AB, Etgen GJ. The metabolic state of diabetic monkeys is regulated by fibroblast growth factor-21. Endocrinology. 2007;148(2):774–781. [DOI] [PubMed] [Google Scholar]
- 4.Micanovic R, Raches DW, Dunbar JD, Driver DA, Bina HA, Dickinson CD, Kharitonenkov A. Different roles of N- and C- termini in the functional activity of FGF21. J Cell Physiol. 2009;219(2):227–234. [DOI] [PubMed] [Google Scholar]
- 5.Suzuki M, Uehara Y, Motomura-Matsuzaka K, Oki J, Koyama Y, Kimura M, Asada M, Komi-Kuramochi A, Oka S, Imamura T. betaKlotho is required for fibroblast growth factor (FGF) 21 signaling through FGF receptor (FGFR) 1c and FGFR3c. Mol Endocrinol. 2008;22(4):1006–1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dushay JR, Toschi E, Mitten EK, Fisher FM, Herman MA, Maratos-Flier E. Fructose ingestion acutely stimulates circulating FGF21 levels in humans. Mol Metab. 2014;4(1):51–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Markan KR, Naber MC, Ameka MK, Anderegg MD, Mangelsdorf DJ, Kliewer SA, Mohammadi M, Potthoff MJ. Circulating FGF21 is liver derived and enhances glucose uptake during refeeding and overfeeding. Diabetes. 2014;63(12):4057–4063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Luo Y, McKeehan WL. Stressed liver and muscle call on adipocytes with FGF21. Front Endocrinol (Lausanne). 2013;4:194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Angelin B, Larsson TE, Rudling M. Circulating fibroblast growth factors as metabolic regulators--a critical appraisal. Cell Metab. 2012;16(6):693–705. [DOI] [PubMed] [Google Scholar]
- 10.Chen WW, Li L,Yang GY, Li K, Qi XY, Zhu W, Tang Y, Liu H, Boden G. Circulating FGF-21 levels in normal subjects and in newly diagnosed patients with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes. 2008;116(1):65–68. [DOI] [PubMed] [Google Scholar]
- 11.Chavez AO, Molina-Carrion M, Abdul-Ghani MA, Folli F, Defronzo RA, Tripathy D. Circulating fibroblast growth factor-21 is elevated in impaired glucose tolerance and type 2 diabetes and correlates with muscle and hepatic insulin resistance. Diabetes Care. 2009;32(8):1542–1546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gallego-Escuredo JM, Gómez-Ambrosi J, Catalan V, Domingo P, Giralt M, Frühbeck G, Villarroya F. Opposite alterations in FGF21 and FGF19 levels and disturbed expression of the receptor machinery for endocrine FGFs in obese patients. Int J Obes. 2015;39(1):121–129. [DOI] [PubMed] [Google Scholar]
- 13.Inagaki T, Lin VY, Goetz R, Mohammadi M, Mangelsdorf DJ, Kliewer SA. Inhibition of growth hormone signaling by the fasting-induced hormone FGF21. Cell Metab. 2008;8(1):77–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dushay J, Chui PC, Gopalakrishnan GS, Varela-Rey M, Crawley M, Fisher FM, Badman MK, Martinez-Chantar ML, Maratos-Flier E. Increased fibroblast growth factor 21 in obesity and nonalcoholic fatty liver disease. Gastroenterology. 2010;139(2):456–463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fazeli PK, Lun M, Kim SM, Bredella MA, Wright S, Zhang Y, Lee H, Catana C, Klibanski A, Patwari P, Steinhauser ML. FGF21 and the late adaptive response to starvation in humans. J Clin Invest. 2015;125(12):4601–4611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.von Holstein-Rathlou S, BonDurant LD, Peltekian L, Naber MC, Yin TC, Claflin KE, Urizar AI, Madsen AN, Ratner C, Holst B, Karstoft K, Vandenbeuch A, Anderson CB, Cassell MD, Thompson AP, Solomon TP, Rahmouni K, Kinnamon SC, Pieper AA, Gillum MP, Potthoff MJ. FGF21 mediates endocrine control of simple sugar intake and sweet taste preference by the liver. Cell Metab. 2016;23(2):335–343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Talukdar S, Owen BM, Song P, Hernandez G, Zhang Y, Zhou Y, Scott WT, Paratala B, Turner T, Smith A, Bernardo B, Müller CP, Tang H, Mangelsdorf DJ, Goodwin B, Kliewer SA. FGF21 regulates sweet and alcohol preference. Cell Metab. 2016;23(2):344–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lundsgaard AM, Fritzen AM, Sjøberg KA, Myrmel LS, Madsen L, Wojtaszewski JF, Richter EA, Kiens B. Circulating FGF21 in humans is potently induced by short term overfeeding of carbohydrates. Mol Metab. 2016;6(1):22–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fisher FM, Kim M, Doridot L, Cunniff JC, Parker TS, Levine DM, Hellerstein MK, Hudgins LC, Maratos-Flier E, Herman MA. A critical role for ChREBP-mediated FGF21 secretion in hepatic fructose metabolism. Mol Metab. 2016;6(1):14–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu J, Stanislaus S, Chinookoswong N, Lau YY, Hager T, Patel J, Ge H, Weiszmann J, Lu SC, Graham M, Busby J, Hecht R, Li YS, Li Y, Lindberg R, Véniant MM. Acute glucose-lowering and insulin-sensitizing action of FGF21 in insulin-resistant mouse models--association with liver and adipose tissue effects. Am J Physiol Endocrinol Metab. 2009;297(5):E1105–E1114. [DOI] [PubMed] [Google Scholar]
- 21.Hager T, Spahr C, Xu J, Salimi-Moosavi H, Hall M. Differential enzyme-linked immunosorbent assay and ligand-binding mass spectrometry for analysis of biotransformation of protein therapeutics: application to various FGF21 modalities. Anal Chem. 2013;85(5):2731–2738. [DOI] [PubMed] [Google Scholar]
- 22.Zhen EY, Jin Z, Ackermann BL, Thomas MK, Gutierrez JA. Circulating FGF21 proteolytic processing mediated by fibroblast activation protein. Biochem J. 2016;473(5):605–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Keane FM, Yao TW, Seelk S, Gall MG, Chowdhury S, Poplawski SE, Lai JH, Li Y, Wu W, Farrell P, Vieira de Ribeiro AJ, Osborne B, Yu DM, Seth D, Rahman K, Haber P, Topaloglu AK, Wang C, Thomson S, Hennessy A, Prins J, Twigg SM, McLennan SV, McCaughan GW, Bachovchin WW, Gorrell MD. Quantitation of fibroblast activation protein (FAP)-specific protease activity in mouse, baboon and human fluids and organs. FEBS Open Bio. 2013;4:43–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, Wong RL, Chow WS, Tso AW, Lam KS, Xu A. Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes. 2008;57(5):1246–1253. [DOI] [PubMed] [Google Scholar]
- 25.Brøns C, Jensen CB, Storgaard H, Hiscock NJ, White A, Appel JS, Jacobsen S, Nilsson E, Larsen CM, Astrup A, Quistorff B, Vaag A. Impact of short-term high-fat feeding on glucose and insulin metabolism in young healthy men. J Physiol. 2009;587(Pt 10):2387–2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chokkalingam K, Jewell K, Norton L, Littlewood J, van Loon LJ, Mansell P, Macdonald IA, Tsintzas K. High-fat/low-carbohydrate diet reduces insulin-stimulated carbohydrate oxidation but stimulates nonoxidative glucose disposal in humans: an important role for skeletal muscle pyruvate dehydrogenase kinase 4. J Clin Endocrinol Metab. 2007;92(1):284–292. [DOI] [PubMed] [Google Scholar]
- 27.Sánchez J, Palou A, Picó C. Response to carbohydrate and fat refeeding in the expression of genes involved in nutrient partitioning and metabolism: striking effects on fibroblast growth factor-21 induction. Endocrinology. 2009;150(12):5341–5350. [DOI] [PubMed] [Google Scholar]
- 28.Iizuka K, Takeda J, Horikawa Y. Glucose induces FGF21 mRNA expression through ChREBP activation in rat hepatocytes. FEBS Lett. 2009;583(17):2882–2886. [DOI] [PubMed] [Google Scholar]
- 29.Uebanso T, Taketani Y, Yamamoto H, Amo K, Ominami H, Arai H, Takei Y, Masuda M, Tanimura A, Harada N, Yamanaka-Okumura H, Takeda E. Paradoxical regulation of human FGF21 by both fasting and feeding signals: is FGF21 a nutritional adaptation factor? PLoS One. 2011;6(8):e22976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dunshee DR, Bainbridge TW, Kljavin NM, Zavala-Solorio J, Schroeder AC, Chan R, Corpuz R, Wong M, Zhou W, Deshmukh G, Ly J, Sutherlin DP, Ernst JA, Sonoda J. Fibroblast activation protein cleaves and inactivates fibroblast growth factor 21. J Biol Chem. 2016;291(11):5986–5996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Coppage AL, Heard KR, DiMare MT, Liu Y, Wu W, Lai JH, Bachovchin WW. Human FGF-21 is a substrate of fibroblast activation protein. PLoS One. 2016;11(3):e0151269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Adams AC, Cheng CC, Coskun T, Kharitonenkov A. FGF21 requires βklotho to act in vivo. PLoS One. 2012;7(11):e49977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ding X, Boney-Montoya J, Owen BM, Bookout AL, Coate KC, Mangelsdorf DJ, Kliewer SA. βKlotho is required for fibroblast growth factor 21 effects on growth and metabolism. Cell Metab. 2012;16(3):387–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Owen BM, Ding X, Morgan DA, Coate KC, Bookout AL, Rahmouni K, Kliewer SA, Mangelsdorf DJ. FGF21 acts centrally to induce sympathetic nerve activity, energy expenditure, and weight loss. Cell Metab. 2014;20(4):670–677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Coate KC, Hernandez G, Thorne CA, Sun S, Le TD, Vale K, Kliewer SA, Mangelsdorf DJ. FGF21 is an exocrine pancreas secretagogue. Cell Metab. 2017;25(2):472–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mulvihill EE, Drucker DJ. Pharmacology, physiology, and mechanisms of action of dipeptidyl peptidase-4 inhibitors. Endocr Rev. 2014;35(6):992–1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Drucker DJ. Dipeptidyl peptidase-4 inhibition and the treatment of type 2 diabetes: preclinical biology and mechanisms of action. Diabetes Care. 2007;30(6):1335–1343. [DOI] [PubMed] [Google Scholar]
- 38.Sánchez-Garrido MA, Habegger KM, Clemmensen C, Holleman C, Müller TD, Perez-Tilve D, Li P, Agrawal AS, Finan B, Drucker DJ, Tschöp MH, DiMarchi RD, Kharitonenkov A. Fibroblast activation protein (FAP) as a novel metabolic target. Mol Metab. 2016;5(10):1015–1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cunningham CCT. Talabostat. Expert Opin Investig Drugs. 2007;16(9):1459–1465. [DOI] [PubMed] [Google Scholar]
- 40.Ryabtsova O, Jansen K, Van Goethem S, Joossens J, Cheng JD, Lambeir AM, De Meester I, Augustyns K, Van der Veken P. Acylated Gly-(2-cyano)pyrrolidines as inhibitors of fibroblast activation protein (FAP) and the issue of FAP/prolyl oligopeptidase (PREP)-selectivity. Bioorg Med Chem Lett. 2012;22(10):3412–3417. [DOI] [PubMed] [Google Scholar]