Abstract
Introduction
To respond to the threat of dementia to public health and the economy, we need to prioritize research resources on strategies that would be the most effective. In relation to the prevention of dementia, this article considers whether lowering plasma homocysteine by B-vitamin supplementation is one of the top priority and cost-effective treatments.
Method
A decision model was constructed to calculate the lifetime costs and quality-adjusted life years (QALYs) of providing B-vitamin treatment to people in the United Kingdom over 60 years with high levels (>13 μmol/L) of plasma homocysteine, which was compared to the lifetime costs and outcomes of not providing them with the treatment.
Results
Treatment with B-vitamins will save £60,021 per QALY gained and so is highly cost-effective.
Discussion
We anticipate that this provocative finding will be debated by scientists, clinicians, and policy makers and eventually be tested in future clinical trials.
Keywords: B-Vitamins, Homocysteine, Cost-effectiveness, Dementia, UK
Highlights
-
•
We need to prioritize resources on strategies to prevent dementia.
-
•
Treatment with B-vitamins at an early stage appears to be highly cost-effective.
-
•
This provocative finding should be debated and tested.
1. Introduction
Dementia is an increasing threat to population health because of its association with the aging of the population, reduced quality of life, and premature mortality. It also poses substantial financial demands on the economy due to the increasing requirement for health and social care for people with dementia as well as substantial need for informal care. A recent report estimated the overall annual costs of dementia to society in the United Kingdom to be £26.8 billion, of which only £4.3 billion is spent on health care [1]. Considering also that a third of people in the UK born in 2015 will develop dementia in their lifetime [2] and the lack of an effective treatment for dementia [3], focusing on prevention of dementia certainly deserves to be high in health policy agenda [4]. Decision-makers urgently need evidence about the cost-effectiveness of the potential prevention strategies [3].
However, limited mental health budgets and underspending in research on dementia compared with public and private funds for research in cancer and cardiovascular disease [5] limits the possibility of providing evidence for all available prevention strategies in the short term. Therefore, we need to prioritize resources on strategies that would be the most worthwhile to focus future research on.
This article argues that B-vitamins are one of the top priority preventive treatments and aims to stimulate debate on this argument by investigating whether B-vitamins are potentially a cost-effective treatment, taking the situation in the United Kingdom as an example.
1.1. The rationale to focus on B-vitamin–based treatment
Strategies for prevention, with the aim of decreasing the prevalence of dementia, are based on monitoring and controlling risk factors associated with dementia. These risk factors are broadly categorized as age, genetic factors, vascular and metabolic factors, lifestyle factors, diet and nutritional factors, and other factors (e.g., depression, occupational exposure) [3]. Among modifiable risk factors, high-plasma total homocysteine (tHcy) level, lower education attainment, and decreased physical activity were found in a recent meta-analysis to be particularly strong predictors of dementia [6]. Among these three strong predictors, tHcy is the only chemical risk factor biomarker for cognitive impairment and dementia [7]. Plasma tHcy concentrations can be screened relatively easily and are readily lowered by high-dose B vitamin supplementation [8]. Although there are no trials to test the effect of lowering tHcy on the incidence of dementia, a recent literature review showed that trials with high-risk subjects have shown positive results of B-vitamin treatment in modifying the dementia disease process [7]. The FACIT trial showed that folic acid supplementation could lower tHcy levels and delay age-related cognitive decline in people aged 50–70 years with high tHcy [9]. B-vitamin supplementation (a combination of vitamin B6, vitamin B12, and folic acid) in the VITACOG trial has been shown to be effective as a disease-modifying intervention in elderly with mild cognitive impairment by reducing elevated tHcy levels, by slowing whole brain atrophy [10] and regional brain atrophy [11], and by slowing further cognitive decline [12]. The VITAL trial showed a positive effect of B-vitamin treatment in slowing cognitive decline on the Alzheimer Disease Assessment Scale (ADAS-cog) [13] and on the Mini-Mental State Examination (Fig. 7 in ref. [7]) for patients with mild AD. B-vitamin supplements are available over-the-counter at very low cost, in contrast to long-term more expensive strategies that aim to increase education attainment or physical activity. B-vitamins, in the doses required, are considered without harm to health. Hence, we hypothesize that B-vitamin treatment has great potential to be cost-effective, and so, we performed a simulation exercise to investigate the potential cost-effectiveness of screening people above the age of 60 years in the United Kingdom and treating those with high levels of tHcy with B-vitamins. The aim of this exercise was to provoke discussion about cost-effective prevention of dementia and to stimulate further clinical trials on the impact of lowering tHcy levels.
2. Methods
To achieve the aim of the study, an explorative cost-effectiveness analysis was undertaken by following NICE recommendations for modeling and exploring uncertainty in technology appraisals [14]. A lifetime time-horizon was chosen in the analysis.
2.1. Stochastic decision model
A stochastic decision model was constructed in Excel to calculate the lifetime costs and outcomes of providing B-vitamin treatment to people over 60 years with high levels of tHcy, which was compared to the lifetime costs and outcomes of not providing them with the treatment. The model performed 10,000 iterations of all cost and effect parameters using prespecified distributions of input parameters and recording incremental costs and incremental quality-adjusted life years (QALYs) from each iteration. Input parameters were related to the target population, treatment effect, costs, and health outcomes (life years and QALYs). The incremental cost-effectiveness ratios (ICERs) were expressed as costs per QALY per treated person. The ICERs were plotted on cost-effectiveness planes to display the uncertainty in the results.
To populate the model, we used information from the literature about the potential target population, treatment effect and costs of dementia as well as life expectancy and quality of life of people over 60 years with and without dementia. When there was no information available in the literature, we made assumptions based on expert opinions. Furthermore, we used a relative standard error (i.e., standard error as percentage of the mean estimate) of 30% when standard errors of the mean were not available. This is considered to be the maximum relative standard error allowing for the report of a mean estimate. As an exception, for the utility (or else quality of life) parameters (with and without dementia), we used a relative standard error of 5% to restrict the cases where people attach higher utility to having dementia than not to having it, which is hardly realistic. Vitamins were assumed not to have negative impact on health and therefore, incremental QALYs can theoretically never become negative in our analysis. However, we allowed incremental QALYs to take negative values to incorporate extreme, close to unrealistic, cases in which treated patients would value the disutility of taking vitamins every day during the gained dementia-free years higher than having dementia in the same period. Moreover, the model assumed that people would receive B-vitamin treatment until dementia onset or death.
2.2. Parameters used in the model
The parameters used in the decision model are listed in Table 1 and discussed in detail in the subsequent sections. We defined a “high tHcy concentration” as > 13 μmol/L, as used by Pfeiffer et al [15]. The VITACOG trial showed that elderly people with mild cognitive impairment who had concentrations of tHcy above 13 μmol/L responded to B-vitamin treatment with a 53% reduction in the rate of whole brain atrophy [10], a 9-fold slowing of the rate of atrophy of specific brain regions involved in Alzheimer's disease [11], and a measurable clinical improvement as assessed by the Clinical Dementia Rating score [12].
Table 1.
Parameters used in the decision model
| Code | Variable | Value | Standard error | Source |
|---|---|---|---|---|
| Target population | ||||
| A | Population >60 y in the United Kingdom (2014) | 14,918,400 | Office of National Statistics, 2015 | |
| B | % of people >60 y with high level tHcy | 0.09 | 0.02 | Pfeiffer et al. [15] |
| C | Population attributable risk percentage (PAR%) | 0.2170 | 0.05 | Beydoun et al. [6] |
| D | Dementia prevalence over 60 y (2014) | 831,000 | 43,878 | Lewis et al. [16] |
| E | People in UK with high tHcy in UK in 2014 | 1,293,425 | Calculated by authors based on A*B | |
| F | People at risk to develop dementia due to high tHcy | 280,673 | Calculated by authors based on C*D | |
| G | People over 60 y who will develop dementia due to high tHcy | 60,906 | Calculated by authors based on C*F | |
| Treatment | ||||
| H | Delay to develop dementia due to treatment, y | 0.91 | 0.38 | Oulhaj et al. [17] |
| I | Adherence to vitamin B treatment | 0.78 | 0.23 | Smith et al. [10] |
| J | Elapsed years from tHcy screening to dementia | 10.16 | 3.05 | Calculated based on Prince et al. [1] and Statistics OoN [18] |
| K | Vitamin effect on delaying dementia, y | 0.71 | ||
| Health outcomes | ||||
| L | Mean life years with dementia >60 y | 5.96 | Calculated based on Lewis [2] and Statistics OoN [18], [19] | |
| M | Mean life years without dementia >60 y | 14.91 | Calculated based on Lewis [2] and Statistics OoN [18], [19] | |
| N | Mean QALYs with dementia >60 y | 3.19 | Calculated based on Lewis [2], Statistics OoN [18], [19], and Knott [20] | |
| O | Mean QALYs without dementia >60 y | 12.52 | Calculated based on Lewis [2], Statistics OoN [18], [19], Knott [20], and Mesterton [21] | |
| P | Mean utility with dementia >60 y | 0.54 | 0.03 | N/L |
| Q | Mean utility without dementia >60 y | 0.84 | 0.04 | O/M |
| Costs | ||||
| R | Costs of dementia per person per year, £ | 28,507 | 8552 | Luengo-Fernandez et al. [5] |
| S | Cost of vitamins per day, £ | 0.03 | 0.01 | Retail price, from Lindens [22] |
| T | Screening costs of tHcy per person, £ | 10.00 | 3.00 | Cost of screening kit |
Abbreviations: QALYs, quality-adjusted life years; tHcy, total homocysteine.
2.2.1. Target population
In 2014, there were 14.9 million people over 60 years in the United Kingdom, of which approximately 1.3 million had high level of tHcy. Because there was no available UK estimate, we calculate the latter figure based on a US proportion (i.e., 8.67%) of women over 60 years with high levels (>13 μmol/L) of tHcy [15]. This proportion was almost 5 percentage points lower than the proportion of US males with high tHcy [15]. This was a conservative assumption because the levels in the UK are likely to be higher than in the United States [7] (see Sensitivity analysis, below). To estimate the proportion of people with high tHcy who will develop dementia, we used a population attributable risk percentage (PAR%) for high tHcy of 21.7% [6] and calculated that 280,673 (=1.3 million × 21.7%) people will be at risk of developing dementia due to tHcy and that 60,906 (280,673 × 21.7%) of these will eventually develop dementia in the course of their life.
2.2.2. Treatment effect
A study showed that lowering tHcy by 4 μmol/L (achievable by B-vitamin treatment) would delay cognitive decline by 10.9 months in 70-year-old patients with Alzheimer's disease [17]. We used this estimate as the treatment effect of B-vitamins in delaying dementia onset. In the VITACOG trial, adherence to B-vitamin treatment was 78% [10]. Furthermore, we approximated that the mean age of people over 60 years in the United Kingdom was 72 years in 2014 [18], [19], whereas the mean age of people over 60 years with dementia was 82 years in the same year [16]. Based on the difference in these mean ages, we assumed that it takes on average 10 years from screening tHcy in people over 60 years to dementia onset. Taking into account the above parameters, we estimated the delay in dementia onset due to B-vitamin treatment by multiplying the B-vitamin effect on cognitive decline, treatment adherence, and years to dementia.
2.2.3. Health outcomes
We calculated life expectancy of people >60 years and total life years (LYs) based on UK Life Table 2012–2014 [19] and population estimates [18] for the general population and based on estimates from Lewis et al. for people with dementia [16]. QALYs for the general population were calculated based on the EQ5D utilities reported in the Health Survey England [20]. QALYs for people with dementia were calculated based on the number of people with mild, moderate, or severe dementia [1] and the EQ5D utilities in each of the three levels of dementia severity [21]. Using the estimated LYs and QALYs, we calculated the mean EQ5D utility of people over 60 years with and without dementia.
2.2.4. Costs
The costs of dementia per patient per year were £28,507 based on a recent study [5], whereas the costs of a daily dose of B-vitamins per patient (one tablet) were set at £0.03 based on retail prices. Furthermore, the screening costs of tHcy per patient were estimated to be £10, which includes the purchase cost of a tHcy kit for typical clinical chemistry analyzers.
2.3. Sensitivity analysis
Five univariate sensitivity analyses were performed to investigate further the uncertainty in the cost-effectiveness results (Table 2). In the first univariate sensitivity analysis, we used 19.8%, instead of 8.67% in the main analysis, as the proportion of people over 60 years with high levels of tHcy. This proportion derives from the same study [15] and may more realistically reflect the UK situation without mandatory folic acid fortification, because it was based on a 1991–1994 estimates which preceded the introduction of mandatory folic acid fortification in the United States. Furthermore, a delay of 18 months in global cognitive decline (instead of 10.9 months in the main analysis) was used in the second-sensitivity analysis based on the results of the FACIT trial [9]. In the third-sensitivity analysis, we assumed the screening costs to be £20 in case the tHcy test would take place in GP practices without the use of automated kits. We also assumed the tHcy screening costs to be £5 instead of £10 in the main analysis in a fourth sensitivity analysis. This assumption seems realistic as tHcy tests can be included in routinely ordered blood tests. Moreover, assuming that health authorities would be able to support the treatment for only half of the targeted population due to budget constraints, we performed a fifth univariate sensitivity analysis by changing the treated population from 1.3 million to 648,000 people.
Table 2.
Results from the univariate sensitivity analyses
| Variable | Incremental costs | Incremental QALYs | ICER | % More costly & more effective | % Less costly & more effective |
|---|---|---|---|---|---|
| People with high tHcy level: 19.8% instead of 8.67% | −581 | 0.008 | −£69,026 | 14 | 86 |
| Treatment effect: 18 months delay in cognitive decline instead of 10.9 months | −1277 | 0.017 | −£76,514 | 8 | 92 |
| Screening costs: £20 instead of £10 | −372 | 0.008 | −£44,299 | 33 | 67 |
| Screening costs: £5 instead of £10 | −548 | 0.008 | −£66,169 | 15 | 85 |
| Treated population: 648,000 instead of 1.3 million | −367 | 0.008 | −£44,304 | 34 | 66 |
Abbreviations: QALYs, quality-adjusted life years; tHcy, total homocysteine.
3. Results
3.1. Main results
The results of the cost-effectiveness analysis are listed in Table 3. The total costs per treated person was £7179 and consisted of £126 screening costs, £162 treatment costs, and £6891 dementia-related costs. If the target group was not treated with B-vitamins, the dementia-related costs would be £7681. Hence, B-vitamins save on average £502 per treated person. Moreover, B-vitamins lead to 0.008 QALY gain per treated person (= 0.023 QALYs with treatment −0.015 QALYs without treatment). As a result, providing B-vitamin treatment dominates (i.e., more QALYs and less costs) the strategy of not providing the treatment to people over 60 years with high levels of tHcy. The estimated ICER denotes that B-vitamins save £60,021 for an additional QALY gained by the treated population. They are, therefore, more effective and less costly than not providing treatment.
Table 3.
Cost-effectiveness results per treated patient (n = approximately 1.3 million)
| Variable | Mean | Standard deviation | Lower 95% CI | Upper 95% CI |
|---|---|---|---|---|
| Intervention costs | ||||
| Screening costs | £126 | 48 | 54 | 240 |
| Treatment costs | £162 | 48 | 82 | 270 |
| Total lifetime costs | ||||
| Dementia lifetime costs | £6891 | 3820 | 1928 | 16,602 |
| Total costs with vitamins per treated person | £7179 | 3820 | 2217 | 16,871 |
| Total costs without vitamins per (un)treated person | £7681 | 4200 | 2129 | 18,290 |
| Incremental lifetime costs | −£502 | 683 | −2302 | 245 |
| Total lifetime QALYs | ||||
| Total QALYs with vitamins per treated person | 0.023 | 0.018 | 0.002 | 0.070 |
| Total QALYs without vitamins per (un)treated person | 0.015 | 0.012 | 0.001 | 0.045 |
| Incremental QALYs per treated person | 0.008 | 0.007 | 0.001 | 0.026 |
| Incremental cost-effectiveness ratio | −£60,021 |
Abbreviation: QALYs, quality-adjusted life years.
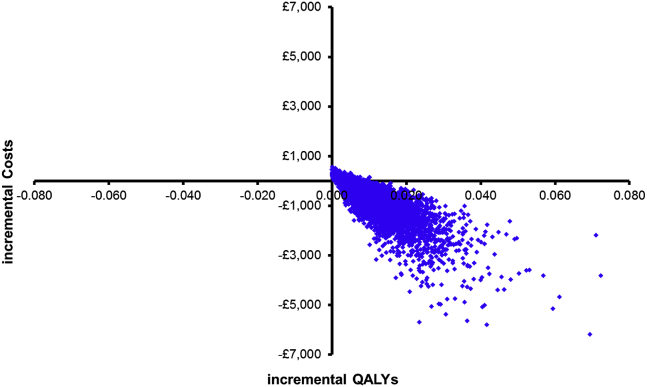
Fig. 1 illustrates the 10,000 ICERs plotted on the cost-effectiveness plane and shows that 79% of them were located in the South-East quadrant indicating QALY gains and cost savings, whereas the remaining 21% was located in the North-East quadrant indicating QALY gains but at higher costs.
Fig. 1.
Cost-effectiveness plane.
3.2. Univariate sensitivity analyses
The results from the univariate sensitivity analyses show that B-vitamin treatment was very likely to be the dominant strategy (i.e., more effective and less costly) in all cases. The treatment appeared to lead to the highest QALY gains and cost savings in the case of having a 18-month delay in global cognitive decline due to B-vitamins. The costs savings were lower when the screening costs were assumed to be £20 per screened person and when only 640,000 eligible people were assumed to be treated with B-vitamins.
4. Discussion
4.1. Stimulating debate and inspiring future research
Most trials of B-vitamin treatment against cognitive decline were poorly designed by including subjects unlikely to benefit during the trial period. In contrast, trials in high-risk subjects, which have taken into account the baseline B vitamin status, show a slowing of cognitive decline and of atrophy in critical brain regions [7]. Therefore, our hypothesis was that B-vitamins treatment is potentially a cost-effective health population strategy for people with elevated levels of tHcy. The results of this explorative analysis are consistent with this hypothesis.
This argument may spark discussions on whether to invest public resources to provide evidence about B-vitamin treatment assuming that it is more cost-effective (or more cost saving) than other prevention strategies [23], [24], early diagnosis strategies [25], [26], disease modification treatments [27], and currently used medication [28]. Although the results of this explorative study support our postulate, we will not be able to know unless we have more robust evidence. This evidence could only derive from future studies with adequate study design and individual patient data. Appropriate dementia disease progression models could also be used in these studies to provide robust estimates about the lifetime costs and QALYs of treating patients with B-vitamins [29]. It should be also noted that B-vitamins may be even more cost-effective if we look outside dementia and incorporate the effect of these vitamins on reducing cerebral atrophy [11], and consider the full costs of mild cognitive impairment [30]. Establishing a screening program for tHcy may have positive spill-over effects on the prevention and treatment of other diseases, such as psychiatric disorders [31], age-related macular degeneration [32], and cardiovascular diseases, notably first stroke [33]. This could increase the overall welfare gain of initiating tHcy screening. Moreover, if tHcy screening became part of ordinary blood testing ordered in GP practices and outpatient departments for people over 60 years, the marginal screening costs would be much lower than £10 assumed in this study. This would increase the cost-effectiveness of B-vitamin treatment. Perhaps, the most relevant question from the perspective of public health authorities is who would bear the costs of the B-vitamins. If the public health budget is limited, individuals over 60 years with high levels of tHcy would be asked to bear these costs. This may not be as contrary to the universal nature of NHS England as it sounds considering that the lifetime treatment costs are only £162 per treated person. If public health authorities take up these costs, the cost-effectiveness of B-vitamin treatment would increase, assuming lower treatment costs due to strong purchasing power.
We have followed a conservative approach in selecting the input parameters in our model to explore the lowest potential of B-vitamin treatment to be cost-effective. This was the reason for using a treatment effect in delaying dementia onset from an observational study in the main analysis and a RCT (FACIT) on cognitive decline in the sensitivity analysis. Using the treatment, effect from other RCTs (e.g., the VITAL trial) would have also shown B-vitamins to be more cost-effective than our main analysis did.
4.2. Conclusions
The results from the explorative cost-effectiveness analysis support the view that B-vitamins are potentially a cost-saving prevention treatment for dementia to be used at population level. We anticipate that this provocative statement will be debated by scientists, clinicians, and policy makers and, we hope, eventually tested by the results of future clinical trials.
Acknowledgments
Author contributions: A.D.S. conceived the study and critically revised the article; A.T. conceived the study, developed the methods, carried out the analyses, interpreted the data, drafted, and critically revised the article.
Footnotes
AT received financial support from the Department of Pharmacology, University Of Oxford for this work. ADS is named as an inventor on four patents held by the University of Oxford on the use of B-vitamins to treat Alzheimer disease or mild cognitive impairment (US6008221, US6127370, US9364497 and PCT/GB2010/051557). This research was supported in part by a benefaction from the Critchley Legacy to OPTIMA.
References
- 1.Prince M., Knapp M., Guerchet M., McGrone P., Prina M., Comas-Herrera A. 2014. Dementia UK: Update. Available at: http://www.cfas.ac.uk/files/2015/07/P326_AS_Dementia_Report_WEB2.pdf. Accessed August 12, 2016. [Google Scholar]
- 2.Lewis F. Office of Health Economics; London: 2015. Estimation of future cases of dementia from those born in 2015. [Google Scholar]
- 3.Winblad B., Amouyel P., Andrieu S., Ballard C., Brayne C., Brodaty H. Defeating Alzheimer's disease and other dementias: a priority for European science and society. Lancet Neurol. 2016;15:455–532. doi: 10.1016/S1474-4422(16)00062-4. [DOI] [PubMed] [Google Scholar]
- 4.Smith A.D., Yaffe K. Dementia (including Alzheimer's Disease) can be prevented: Statement supported by international experts. J Alzheimers Dis. 2014;38:699–703. doi: 10.3233/JAD-132372. [DOI] [PubMed] [Google Scholar]
- 5.Luengo-Fernandez R., Leal J., Gray A. UK research spend in 2008 and 2012: comparing stroke, cancer, coronary heart disease and dementia. BMJ Open. 2015;5:e006648. doi: 10.1136/bmjopen-2014-006648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beydoun M.A., Beydoun H.A., Gamaldo A.A., Teel A., Zonderman A.B., Wang Y. Epidemiologic studies of modifiable factors associated with cognition and dementia: systematic review and meta-analysis. BMC Public Health. 2014;14:643. doi: 10.1186/1471-2458-14-643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Smith A.D., Refsum H. Homocysteine, B vitamins and cognitive impairment. Annu Rev Nutr. 2016;36:211–239. doi: 10.1146/annurev-nutr-071715-050947. [DOI] [PubMed] [Google Scholar]
- 8.Refsum H., Smith A.D., Ueland P.M., Nexo E., Clarke R., McPartlin J. Facts and recommendations about total homocysteine determinations: an expert opinion. Clin Chem. 2004;50:3–32. doi: 10.1373/clinchem.2003.021634. [DOI] [PubMed] [Google Scholar]
- 9.Durga J., van Boxtel M.P., Schouten E.G., Kok F.J., Jolles J., Katan M.B. Effect of 3-year folic acid supplementation on cognitive function in older adults in the FACIT trial: a randomised, double blind, controlled trial. Lancet. 2007;369:208–216. doi: 10.1016/S0140-6736(07)60109-3. [DOI] [PubMed] [Google Scholar]
- 10.Smith A.D., Smith S.M., de Jager C.A., Whitbread P., Johnston C., Agacinski G. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: a randomized controlled trial. PLoS One. 2010;5:e12244. doi: 10.1371/journal.pone.0012244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Douaud G., Refsum H., de Jager C.A., Jacoby R., Nichols T.E., Smith S.M. Preventing Alzheimer's disease-related gray matter atrophy by B-vitamin treatment. Proc Natl Acad Sci U S A. 2013;110:9523–9528. doi: 10.1073/pnas.1301816110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.de Jager C.A., Oulhaj A., Jacoby R., Refsum H., Smith A.D. Cognitive and clinical outcomes of homocysteine-lowering B-vitamin treatment in mild cognitive impairment: a randomized controlled trial. Int J Geriatr Psychiatry. 2012;27:592–600. doi: 10.1002/gps.2758. [DOI] [PubMed] [Google Scholar]
- 13.Aisen P.S., Schneider L.S., Sano M., Diaz-Arrastia R., van Dyck C.H., Weiner M.F. High-dose B vitamin supplementation and cognitive decline in Alzheimer disease: a randomized controlled trial. JAMA. 2008;300:1774–1783. doi: 10.1001/jama.300.15.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.NICE . National Institute for Health and Care Excellence; London, UK: 2013. Guide to the methods of technology appraisal 2013. [PubMed] [Google Scholar]
- 15.Pfeiffer C.M., Osterloh J.D., Kennedy-Stephenson J., Picciano M.F., Yetley E.A., Rader J.I. Trends in circulating concentrations of total homocysteine among US adolescents and adults: findings from the 1991-1994 and 1999-2004 National Health and Nutrition Examination Surveys. Clin Chem. 2008;54:801–813. doi: 10.1373/clinchem.2007.100214. [DOI] [PubMed] [Google Scholar]
- 16.Lewis F., Karlsberg-Schaffer S., Sussex J., O'Neil P., Cockroft L. Office of Health Economics; London: 2014. The trajectory of dementia in the UK- making a difference. [Google Scholar]
- 17.Oulhaj A., Refsum H., Beaumont H., Williams J., King E., Jacoby R. Homocysteine as a predictor of cognitive decline in Alzheimer's disease. Int J Geriatr Psychiatry. 2010;25:82–90. doi: 10.1002/gps.2303. [DOI] [PubMed] [Google Scholar]
- 18.Statistics OoN . Office for National Statistics; London, UK: 2015. Annual Mid-Year Population Estimates for the UK. [Google Scholar]
- 19.Statistics OoN . Office for National Statistics; London, UK: 2015. National life tables, United Kingdom, 2012-2014. [Google Scholar]
- 20.Knott C. Health and Social Care Information Centre; 2012. General mental and physical health. [Google Scholar]
- 21.Mesterton J., Wimo A., By A., Langworth S., Winblad B., Jonsson L. Cross sectional observational study on the societal costs of Alzheimer's disease. Curr Alzheimer Res. 2010;7:358–367. doi: 10.2174/156720510791162430. [DOI] [PubMed] [Google Scholar]
- 22.Lindens.co.uk [cited 2016 May 09]. Available at: https://www.lindens.co.uk/product/480/neurovits_plus__b1,_b6,_b12,_folic_acid__tablets/view.html. Accessed August 12, 2016.
- 23.Zhang Y., Kivipelto M., Solomon A., Wimo A. Cost-effectiveness of a health intervention program with risk reductions for getting demented: results of a Markov model in a Swedish/Finnish setting. J Alzheimers Dis. 2011;26:735–744. doi: 10.3233/JAD-2011-110065. [DOI] [PubMed] [Google Scholar]
- 24.Baal P.H., Hoogendoorn M., Fischer A. Preventing dementia by promoting physical activity and the long-term impact on health and social care expenditures. Prev Med. 2016;85:78–83. doi: 10.1016/j.ypmed.2016.01.013. [DOI] [PubMed] [Google Scholar]
- 25.Wimo A., Ballard C., Brayne C., Gauthier S., Handels R., Jones R.W. Health economic evaluation of treatments for Alzheimer's disease: impact of new diagnostic criteria. J Intern Med. 2014;275:304–316. doi: 10.1111/joim.12167. [DOI] [PubMed] [Google Scholar]
- 26.Banerjee S., Wittenberg R. Clinical and cost effectiveness of services for early diagnosis and intervention in dementia. Int J Geriatr Psychiatry. 2009;24:748–754. doi: 10.1002/gps.2191. [DOI] [PubMed] [Google Scholar]
- 27.Budd D., Burns L.C., Guo Z., L'Italien G., Lapuerta P. Impact of early intervention and disease modification in patients with predementia Alzheimer's disease: a Markov model simulation. Clinicoecon Outcomes Res. 2011;3:189–195. doi: 10.2147/CEOR.S22265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Clegg A., Bryant J., Nicholson T., McIntyre L., De Broe S., Gerard K. Clinical and cost-effectiveness of donepezil, rivastigmine and galantamine for Alzheimer's disease: a rapid and systematic review. Health Technol Assess. 2001;5:1–137. doi: 10.3310/hta5010. [DOI] [PubMed] [Google Scholar]
- 29.Green C., Shearer J., Ritchie C.W., Zajicek J.P. Model-based economic evaluation in Alzheimer's disease: a review of the methods available to model Alzheimer's disease progression. Value Health. 2011;14:621–630. doi: 10.1016/j.jval.2010.12.008. [DOI] [PubMed] [Google Scholar]
- 30.Lin P.J., Neumann P.J. The economics of mild cognitive impairment. Alzheimers Dement. 2013;9:58–62. doi: 10.1016/j.jalz.2012.05.2117. [DOI] [PubMed] [Google Scholar]
- 31.Folstein M., Liu T., Peter I., Buell J., Arsenault L., Scott T. The homocysteine hypothesis of depression. Am J Psychiatry. 2007;164:861–867. doi: 10.1176/ajp.2007.164.6.861. [DOI] [PubMed] [Google Scholar]
- 32.Huang P., Wang F., Kumar Sah B., Jiang J., Ni Z., Wang J. Homocysteine and the risk of age-related macular degeneration: a systematic review and meta-analysis. Sci Rep. 2015;5:10585. doi: 10.1038/srep10585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Huo Y., Li J., Qin X., Huang Y., Wang X., Gottesman R.F. Efficacy of folic acid therapy in primary prevention of stroke among adults with hypertension in China: the CSPPT randomized clinical trial. JAMA. 2015;313:1325–1335. doi: 10.1001/jama.2015.2274. [DOI] [PubMed] [Google Scholar]