The thymus medulla prevents T cell–driven autoimmunity via central tolerance. Cosway et al. show that this specialization occurs independently of the topology that classically defines its structure and demonstrate that medulla function requires LTβR-mediated regulation of dendritic cells for negative selection.
Abstract
During αβT cell development, the thymus medulla represents an essential microenvironment for T cell tolerance. This functional specialization is attributed to its typical organized topology consisting of a branching structure that contains medullary thymic epithelial cell (mTEC) networks to support negative selection and Foxp3+ T-regulatory cell (T-reg) development. Here, by performing TEC-specific deletion of the thymus medulla regulator lymphotoxin β receptor (LTβR), we show that thymic tolerance mechanisms operate independently of LTβR-mediated mTEC development and organization. Consistent with this, mTECs continue to express Fezf2 and Aire, regulators of intrathymic self-antigens, and support T-reg development despite loss of LTβR-mediated medulla organogenesis. Moreover, we demonstrate that LTβR controls thymic tolerance by regulating the frequency and makeup of intrathymic dendritic cells (DCs) required for effective thymocyte negative selection. In all, our study demonstrates that thymus medulla specialization for thymic tolerance segregates from medulla organogenesis and instead involves LTβR-mediated regulation of the thymic DC pool.
Introduction
The thymus generates αβT cells that respond to foreign antigens presented by self-MHC molecules (Boehm, 2008). During intrathymic development, thymocytes express a randomly generated αβTCR repertoire that is screened to bias thymus function toward self-tolerant T cell production (Kappler et al., 1987; Jenkinson et al., 1989; Kishimoto and Sprent, 1997). This requires thymic selection mechanisms involving stromal cells in anatomically distinct areas (Takahama et al., 2008). In the cortex, cortical thymic epithelial cells (TECs [cTECs]) trigger positive selection (Anderson et al., 1994; Laufer et al., 1996; Murata et al., 2007). This process also induces expression of CCR4 and CCR7 (Ueno et al., 2004; Ehrlich et al., 2009; Cowan et al., 2014; Hu et al., 2015) to allow newly selected thymocytes access to the medulla. Here, negative selection eliminates thymocytes bearing high-affinity αβTCRs via apoptosis (Daniels et al., 2006; Klein et al., 2014). The medulla also supports Foxp3+ T-regulatory (T-reg) development (Aschenbrenner et al., 2007; Perry et al., 2014; Malhotra et al., 2016), and although mechanisms discriminating these processes are unclear, both medullary TECs (mTECs) and DCs are important (Cowan et al., 2013; Perry et al., 2014; Herbin et al., 2016).
Several features of the medulla may explain its specialization for tolerance. First, formation from clonally derived islets creates a complex 3D topology which, in WT mice, consists of small areas that may be connected to a larger medullary compartment. This process is initiated during organogenesis, is maintained in adulthood (Rodewald et al., 2001; Boehm et al., 2003; Irla et al., 2013), and provides lymphostromal interactions for single-positive thymocytes (Anderson and Takahama, 2012). Second, the medulla houses DCs, with Aire+ mTECs producing XCL1 to control DC positioning for T-reg generation (Lei et al., 2011). Finally, specialized mTEC subsets express key genes that collectively regulate tolerance. Of these, Aire and Fezf2 are the two known regulators of intrathymic expression of tissue-restricted antigens (TRAs). Absence of either Aire (Anderson et al., 2002) or Fezf2 (Takaba et al., 2015) results in tolerance breakdown, which fits well with their ability to regulate differing TRAs. Although Aire is controlled by RANK (Rossi et al., 2007; Akiyama et al., 2008; Hikosaka et al., 2008), lymphotoxin β receptor (LTβR) was reported as an essential regulator of Fezf2 expression in mTECs (Takaba et al., 2015). Indeed, both Rank−/− (Rossi et al., 2007; Akiyama et al., 2008; Hikosaka et al., 2008) and Ltbr−/− (Boehm et al., 2003; Venanzi et al., 2007; Zhu et al., 2007; White et al., 2010) mice demonstrate defective medulla formation and loss of tolerance. Collectively, these findings suggest a dual requirement for RANK/Aire and LTβR/Fezf2 pathways during T cell tolerance. Furthermore, they help form current models in which mTEC organization and development are prerequisites of tolerance induction, with medulla abnormalities being causative factors in tolerance breakdown (Akiyama et al., 2015; Abramson and Anderson, 2017).
Here, we have explored mechanisms that control the thymus medulla and determine its ability to mediate tolerance. Specifically, we examined the relationship between LTβR and coordination of mTECs and DCs for negative selection and T-reg generation. We show that despite a profound perturbation of mTECs caused by TEC-specific deletion of LTβR, T cell tolerance remains intact, challenging the notion that thymic tolerance is determined by medulla organization and development. Rather, we show that an essential feature of medulla function involves LTβR-mediated control of the thymic DC pool for negative selection. In all, our study separates the process of medulla formation from its control of thymic tolerance and identifies a new role for LTβR in the regulation of thymus function.
Results and discussion
TEC-restricted deletion of LTβR dissociates medulla topology from tolerance induction
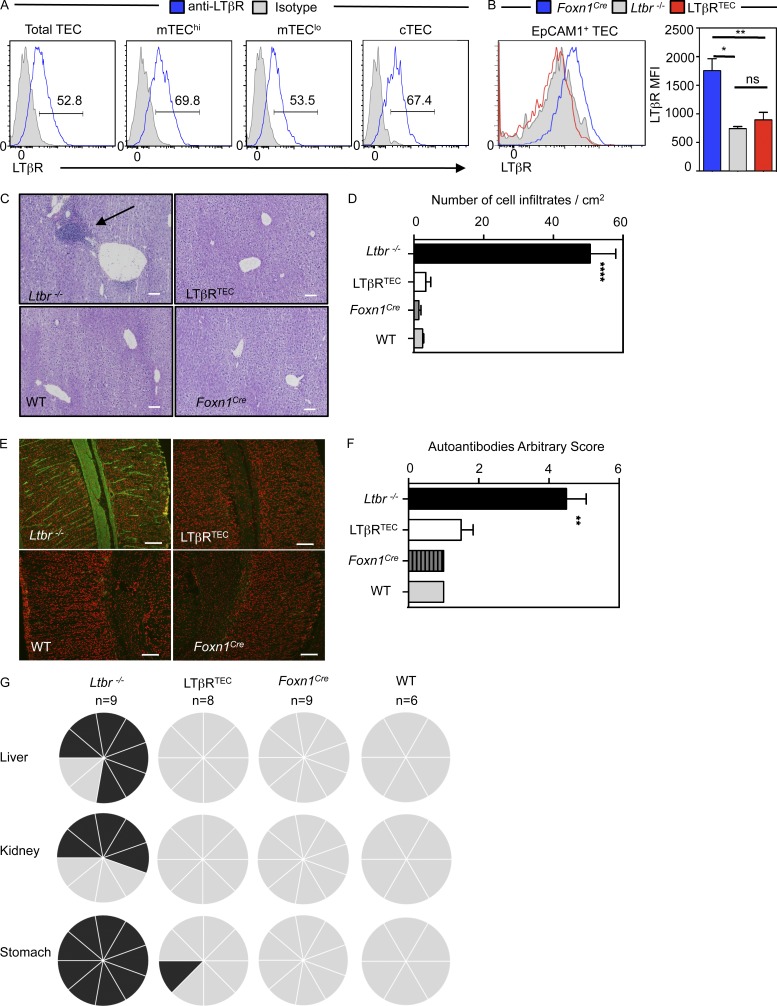
Normal programs of mTEC development and medulla formation are seen to be essential for the specialized function of this site. The TNF receptor superfamily (TNFRSF) member LTβR is a key regulator of thymic microenvironments and intrathymic tolerance, and its expression is readily detectable in multiple TEC subsets (Fig. 1 A). However, in studies using germline Ltbr−/− models, it is unclear whether effects on tolerance are directly attributable to alterations in TEC development or function. To examine this, we crossed Foxn1Cre mice (Gordon et al., 2007) with mice carrying floxed alleles of LTβR (Wang et al., 2010) to create LTβRTEC mice. Importantly, LTβR expression by EpCAM1+ TEC was absent in Ltbr−/− mice and LTβRTEC mice (Fig. 1 B), demonstrating the effectiveness of this model to examine the relationship between medulla function and tolerance.
Figure 1.
LTβR deletion in thymic epithelium corrects the autoimmunity in germline Ltbr−/− mice. (A) LTβR in WT EpCAM1+ TECs, Ly51+ cTECs, Ly51−MHCIIloCD80lo mTEClo cells, and Ly51−MHCIIhiCD80hi mTEChi cells; gray histogram is isotype control staining. (B) Anti-LTβR staining in TECs from indicated strains. Data represent two experiments, n ≥ 4 mice. (C) Liver sections from mice at 8–12 wk of age. Arrow indicates lymphocytic infiltrates. Bars, 100 µm. (D) Quantitation of infiltrates in B. Data from ≥3 mice from two experiments. (E) WT sections incubated with 1/80 sera to detect autoantibodies (green); DAPI in red; staining on stomach shown as an example. Bars, 100 µm. (F) Quantification of autoantibody staining in stomach. Data represent at least two experiments, n ≥ 5 mice. (G) Summary of autoantibody detection in various tissues. Each segment represents one mouse; black denotes positive staining. Error bars indicate SEM. *, P < 0.05; **, P < 0.01; ****, P < 0.0001.
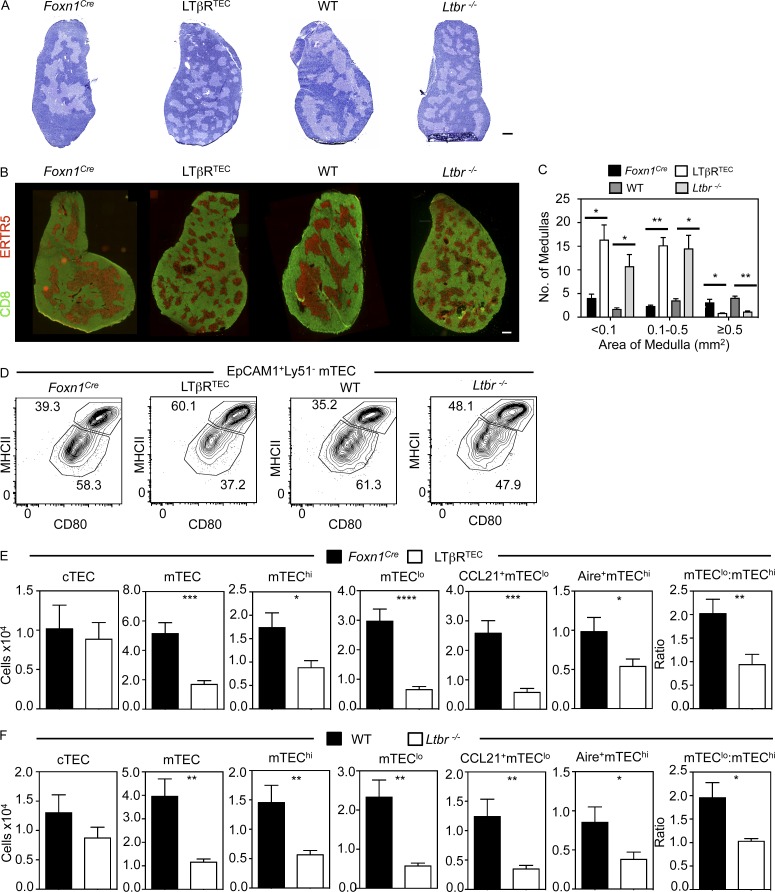
A key feature of Ltbr−/− mice is a breakdown in central tolerance. This manifests as lymphocytic infiltrates in multiple organs and the presence of serum autoantibodies (Boehm et al., 2003; Venanzi et al., 2007; Zhu et al., 2007; Martins et al., 2008). Because disruption of thymic tolerance in Ltbr−/− mice correlates with defective medulla formation and mTEC development (Boehm et al., 2003), we examined thymic tolerance in LTβRTEC mice. Although several tissues (liver, kidney, stomach, salivary gland) from Ltbr−/− mice showed signs of autoimmunity including cell infiltrates, positive autoantibody staining, and presence of activated T cells, these features were markedly absent from LTβRTEC mice (Fig. 1, C–G; and Fig. S1). Notably, the lack of measurable autoimmunity in LTβRTEC mice occurred despite the presence of intact secondary lymphoid tissues that are defective in Ltbr−/− mice (not depicted). We examined LTβRTEC mice up to 5 mo of age and still failed to observe signs of autoimmunity (not depicted). Importantly, compared with medulla organization in WT and Foxn1Cre controls, both Ltbr−/− and LTβRTEC mice showed disruption of the typical 3D medulla architecture (Fig. 2, A and B). Indeed, large ERTR5+ mTEC areas present in control mice were absent, and ERTR5+ areas were smaller and scattered throughout thymic sections (Fig. 2 B). Thus, Ltbr−/− and LTβRTEC mice both showed a significant decrease in the number of large (≥0.5-mm2) medulla areas per thymus section and an increased number of smaller (<0.1-mm2 and 0.1–0.5-mm2) medullas (Fig. 2 C). Interestingly, despite detectable LTβR expression by cTECs (Fig. 1 A), we saw no significant alterations in cTEC numbers in Ltbr−/− and LTβRTEC mice (Fig. 2, E and F). In contrast, both mice had defects in mTECs, including reduced numbers of mTEClow, mTEChi, and subsets of CCL21+ and Aire+ cells (Fig. 2, D–F). Thus, in LTβRTEC mice where TEC specific deletion of LTβR recapitulates the medullary disorganization in Ltbr−/− mice, T cell tolerance is maintained. Collectively, these findings indicate that autoimmunity is not a direct consequence of medulla dysgenesis and is distinct from the impact of LTβR on mTECs.
Figure 2.
Defective mTEC development and medulla topology in LTβRTEC mice. (A) Thymic architecture in indicated mouse strains. Bar, 500 µm. (B) Sections stained with anti-CD8 (green) to detect cortex and ERTR5 (red) to detect medulla. Bar, 500 µm. Images represent ≥4 mice. (C) Quantitation of medulla areas in sections. Data are means of three sections per mouse, n = 3 per strain. Data from three separate experiments. (D) CD80/MHCII in EpCAM1+Ly51− mTECs. (E and F) TEC quantitation in Foxn1Cre, LTβRTEC, WT, and Ltbr−/− mice. Data from four experiments, n = 12. Error bars indicate SEM. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
RANK controls both known intrathymic regulators of promiscuous gene expression
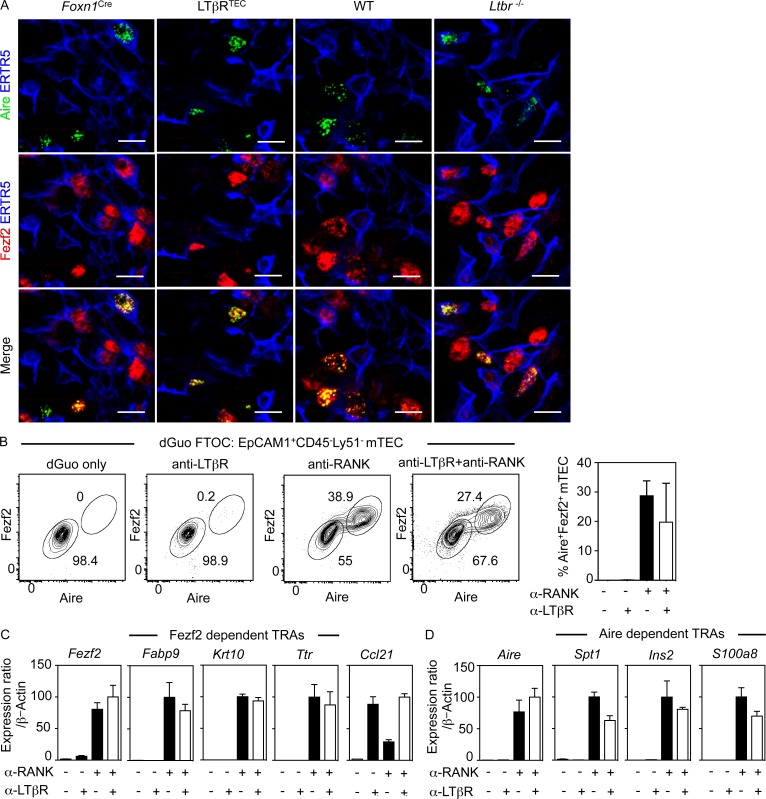
Significant to our findings is a study suggesting that LTβR controls mTEC expression of Fezf2, a transcription factor that regulates intrathymic TRAs (Takaba et al., 2015). Indeed, absence of Fezf2+ mTEC from Ltbr−/− mice was reported as a major factor in loss of tolerance in these mice. Importantly, using the same anti-Fezf2 antibody (Takaba et al., 2015), we detected Fezf2+ mTEC in both Ltbr−/− and LTβRTEC mice (Fig. 3 A). Moreover, anti-LTβR stimulation of dGuo fetal thymus organ culture (FTOC) did not induce expression of Fezf2 (Fig. 3, B and C) nor the previously reported Fezf2-dependent TRAs Fabp9, Krt10, and Ttr (Fig. 3 C; Takaba et al., 2015). Importantly, this failure was not caused by ineffective stimulation, because anti-LTβR induced expression of Ccl21 mRNA (Fig. 3 C). In contrast, anti-RANK stimulation induced high levels of both Fezf2 and Aire in mTECs (Fig. 3, B–D), as well as Aire-dependent and Fezf2-dependent TRAs (Fig. 3, C and D). Expression of Aire, Fezf2, and associated TRAs was not augmented by combined RANK and LTβR stimulation (Fig. 3, B–D). Consistent with the expression pattern of Aire (Gray et al., 2007), Fezf2 was detectable only in mTEChi cells (not depicted). Thus, although LTβR influences mTEC development and organization (Boehm et al., 2003; Lkhagvasuren et al., 2013; Wu et al., 2017), it is not required for generation of Fezf2+ mTECs. Rather, RANK represents a key regulator of both Aire+ and Fezf2+ mTECs. This demonstrates that the requirement for RANK in thymic tolerance is linked to control of mTEC development, including Aire and now Fezf2 expression, and emphasizes that the role of LTβR in central tolerance is distinct from its ability to control intrathymic TRA expression. This is reinforced by our finding that LTβRTEC mice do not show symptoms of autoimmunity and suggests that despite a reduction in the number of TRA-producing cells, the capacity for mTEC-dependent self-antigen production in LTβRTEC mice exceeds any threshold requirement for tolerance induction in the naturally diverse αβTCR repertoire.
Figure 3.
RANK controls intrathymic regulators of promiscuous gene expression. (A) Confocal images of thymus sections stained for Aire (green), ERTR5 (blue), and Fezf2 (red). Bars, 10 µm. Data represent three experiments, n ≥ 6. (B) Aire and Fezf2 in Ly51− mTECs in dGuo FTOCs treated as indicated. Graph shows percentage Aire+Fezf2+ mTECs. Data from at least three separate experiments. (C and D) qPCR of indicated genes in anti-RANK/anti-LTβR stimulated dGuo FTOCs. Data from at least two independent experiments. Error bars represent SEM.
Foxp3+ T-reg production occurs independently of LTβR and medulla organization
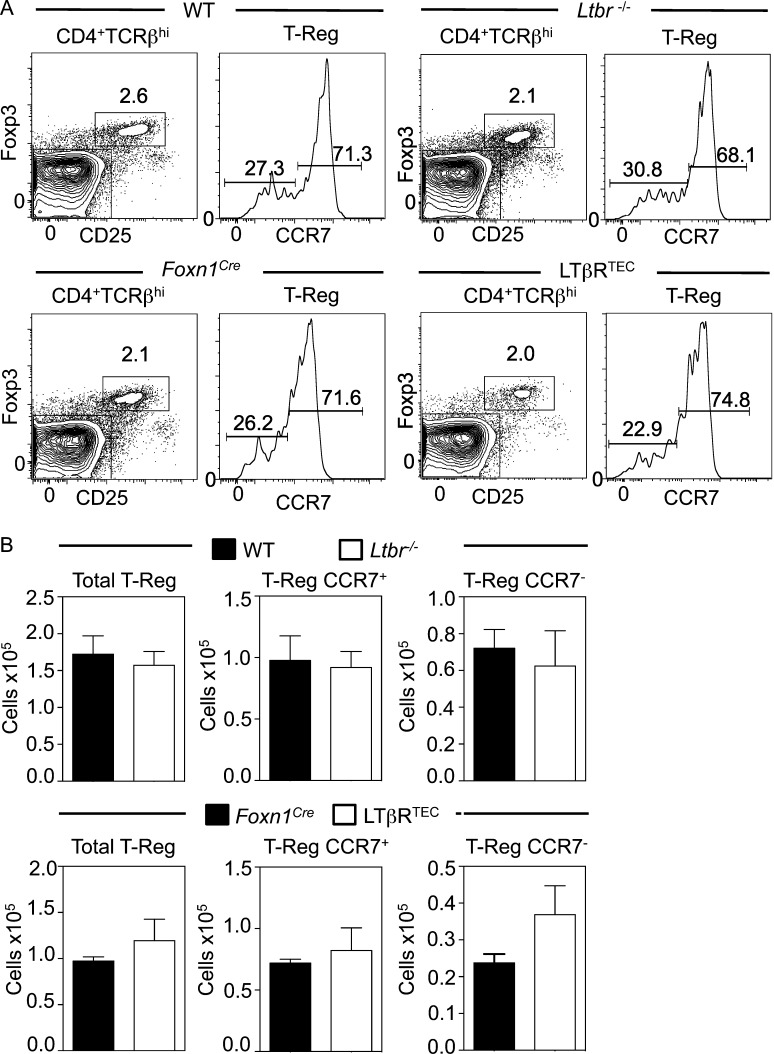
In the medulla, interactions between thymocytes and mTECs/DCs result in Foxp3+ T-reg development (Tai et al., 2013; Perry et al., 2014). Given the essential requirement for mTECs in Foxp3+ T-reg development (Cowan et al., 2013), we analyzed this process in Ltbr−/− and LTβRTEC mice. Importantly, and in contrast to previous studies (Zhu et al., 2007; Martins et al., 2008), we separated total thymic T-reg using CCR7 to discriminate de novo from recirculating T-reg (Cowan et al., 2016). In Ltbr−/− and LTβRTEC mice, both proportions and absolute numbers of newly generated CCR7+Foxp3+ T-reg were comparable to those of control mice (Fig. 4, A and B). Thus, Foxp3+ T cell development is not dependent on LTβR-mediated control of the medulla, suggesting that failure of tolerance in Ltbr−/− mice is not caused by defective Foxp3+ T-reg generation secondary to disruption of medulla structure.
Figure 4.
De novo Foxp3+ T-reg development occurs independently of LTβR. (A) Analysis of CD25+Foxp3+CD4+TCRβ+ thymocytes, with CCR7 expression to identify newly selected CCR7+ Foxp3+ T-reg. (B) Absolute numbers of CCR7+ and CCR7− Foxp3+ T-reg. Data typical of at least seven mice from three separate experiments. Error bars represent SEM.
LTβR controls the thymic DC pool for negative selection
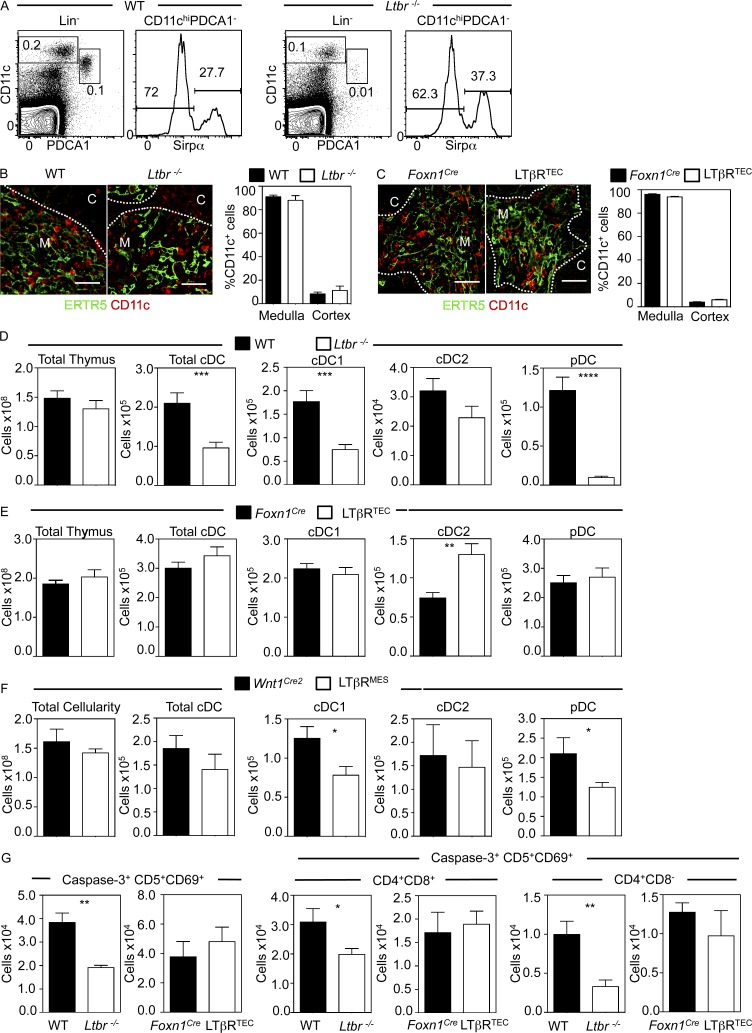
Given the importance of multiple DC subsets in thymic tolerance (Proietto et al., 2008; Hadeiba et al., 2012; Perry et al., 2014), we examined thymic DCs in Ltbr−/− and LTβRTEC mice. We used flow cytometric analysis of digested thymuses to identify PDCA1+CD11clow plasmacytoid DCs (pDCs) and PDCA1−CD11c+ conventional DC (cDC) subsets in the CD3−CD19−NK1.1− (Lin−) fraction. CD11c+ cDCs were further subdivided into SIRPα− cDC1 and SIRPα+ cDC2 cells (Fig. 5 A). Although DCs in both WT and Ltbr−/− mice were predominantly located in the medulla (Fig. 5 B), we saw alterations in the thymic DC pool size of Ltbr−/− mice, with numbers of both pDCs and cDCs significantly reduced compared with WT (Fig. 5 D). This impact on cDCs mapped to a selective reduction in cDC1 cells, with comparable cDC2 numbers in WT and Ltbr−/− mice (Fig. 5 D). Importantly, we saw no differences in BrdU incorporation in thymic DCs from Ltbr−/− and LTβRTEC mice (Fig. S2), suggesting that the diminished numbers were not caused by reduced DC proliferation. Thus, in Ltbr−/− mice where both mTECs and tolerance are defective, LTβR controls the size and makeup of the thymic DC pool. When we analyzed thymic DCs in LTβRTEC mice, where mTEC development is impaired but tolerance is maintained, DCs were located throughout ERTR5+ medullary areas in a manner comparable to control mice (Fig. 5 C). However, and in contrast to Ltbr−/− mice, we found no reduction in thymic DCs in LTβRTEC mice (Fig. 5 E). Indeed, we saw increased cDC2 cells in LTβRTEC mice compared with Foxn1Cre controls. Although the reasons for this are currently unclear, one possibility is that LTβR expression by TECs may act to suppress intrathymic cDC2 numbers as part of its role in controlling the size and makeup of the intrathymic DC pool. Collectively, analysis of intrathymic DCs in Ltbr−/− and LTβRTEC mice shows that the requirement for LTβR in thymic tolerance correlates with a reduction in thymic DC frequency that is unconnected to LTβR-mediated mTEC regulation.
Figure 5.
LTβR controls formation of the thymic DC pool for negative selection. (A) cDC1, cDC2, and pDCs in Lin− thymus preparations. (B and C) CD11c+ DCs in ERTR5+ areas of indicated mice. Bars, 50 µm. Data from three experiments, n ≥ 6. (D and E) Thymic DCs in WT/Ltbr−/− and LTβRTEC/Foxn1Cre mice; n = 11 from three experiments. (F) Thymic DC numbers in Wnt1Cre2 and LTβRMES mice; data from three experiments, n ≥ 6. (G) Numbers of total, DP, and CD4+ CD5+CD69+Caspase3+ thymocytes. Data obtained from at least two experiments where n ≥ 5 for all strains. Error bars indicate SEM. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
LTβR controls splenic DCs in a cell-autonomous manner (Kabashima et al., 2005; Wang et al., 2005). We generated BM chimeras using WT and Ltbr−/− host/donor combinations, and this confirmed the cell-intrinsic requirement for LTβR by splenic DCs (Fig. S3). Importantly, however, thymic DCs were significantly reduced in WT:Ltbr−/− but not Ltbr−/−:WT chimeras (Fig. S3). Thus, the requirement for LTβR by thymic DCs is non–cell autonomous and instead maps to radioresistant stroma, with no reduction in thymic DC numbers in LTβRTEC mice, indicating a role for LTβR expression by non-TEC stroma. As the thymic mesenchyme has been implicated in various aspects of thymus function (Jenkinson et al., 2003, 2007), we investigated whether these cells play a role in controlling thymic DCs. Thus, we crossed Wnt1Cre2 mice, in which Cre is expressed by neural crest derived mesenchymal cells (Lewis et al., 2013), with Ltbrfl/fl mice to generate LTβRMES mice in which thymic deletion of LTβR is limited to the mesenchyme. Interestingly, we saw a significant and selective decrease in both cDC1 cells and pDCs in LTβRMES mice compared with Wnt1Cre2 controls (Fig. 5 F), a pattern that mirrors the thymic DC defect in Ltbr−/− mice. Collectively, comparison of the cellular regulators of thymic tolerance in Ltbr−/− and LTβRTEC mice and analysis of their autoimmune status indicates that LTβR controls formation of the thymic DC pool via a mechanism distinct from its regulation of mTEC development and medulla formation. Because a key role of DCs is clonal deletion of autoreactive thymocytes (Gallegos and Bevan, 2004; Bonasio et al., 2006), we analyzed the frequency of Caspase-3+CD5+CD69+ thymocytes, representing cells undergoing negative selection in the naturally diverse WT αβTCR repertoire (Stritesky et al., 2013). Compared with WT, Ltbr−/− mice contained fewer Caspase-3+CD5+CD69+ thymocytes, indicating a reduction in negative selection (Fig. 5 G). Moreover, combined analysis of CD4, CD8, and Caspase-3 expression showed a greater reduction in the number of CD4+ thymocytes undergoing negative selection compared with double-positive (DP) thymocytes (Fig. 5 G). In contrast, in LTβRTEC mice, in which self-tolerance and thymic DCs are maintained, the frequency of total, DP, or CD4+ Caspase-3+CD5+CD69+ thymocytes was not changed (Fig. 5 G). Thus, breakdown of thymic tolerance in Ltbr−/− but not LTβRTEC mice correlates with reductions in both thymic DCs and the frequency of thymocytes undergoing negative selection.
The thymus medulla is a specialized microenvironment essential for T cell tolerance. This has been attributed to its 3D organization and the presence of multiple mTEC subsets residing within a complex structure consisting of multiple islets that may branch from a larger medullary core (Irla et al., 2013). This anatomical specialization is thought to foster mTEC and DC function and limit autoimmune responses via negative selection and Foxp3+ T-reg development. Here, we show that TEC-specific deletion of LTβR disrupts medulla formation and mTEC development and limits mTEC availability. Although it is currently unclear whether LTβR expression by cTECs plays a functional role in T cell development, it is important to note that absence of LTβR does not alter cTEC numbers, suggesting that it may not play an essential role in cTEC development. Importantly, and despite the alterations in mTECs caused by TEC-specific loss of LTβR, thymus dysgenesis does not alter its ability to impose T cell tolerance mechanisms. Thus, our data suggest that “form ever follows function” models (Sullivan, 1896) do not necessarily apply to the thymus medulla. Although mTECs are an essential requirement for tolerance induction (Cowan et al., 2013), we show that quantitative limitation of their availability, and a loss of typical thymus architecture, still allows the medulla to operate as a tolerizing site. This finding is important in understanding how the medulla imposes tolerance mechanisms. For example, although TCR transgenic T-reg development is limited by intrathymic niche availability (Bautista et al., 2009; DiPaolo and Shevach, 2009; Leung et al., 2009), mTEC loss in LTβRTEC mice does not impair Foxp3+ T-reg development or negative selection. Thus, for the naturally diverse αβTCR repertoire, intrathymic niche availability does not rate-limit the ability of the medulla to support both dominant and recessive tolerance. Furthermore, despite reduced mTECs in LTβRTEC mice, frequencies of mature CD4+ and CD8+ thymocytes are unaltered (unpublished data). Whether this reflects changes in thymocyte motility/dwell time that compensate for reduced mTEC availability is not known. In addition, because LTβR influences mTEC shape (Boehm et al., 2003), alterations in mTEC cell surface area in LTβRTEC mice may alter thymocyte interactions. Interestingly, however, the segmented nature of the medulla in LTβRTEC mice does not lead to tolerance breakdown, which is perhaps consistent with a similar distribution of medullary islands in juvenile mice (Rodewald et al., 2001) and the confinement of thymocytes to tolerance-inducing medulla subunits (Le Borgne et al., 2009).
Perhaps most significant to current understanding of mTEC development is our finding that LTβR does not control expression of Fezf2, a regulator of intrathymic TRAs. This contrasts with a recent study (Takaba et al., 2015), and although the reasons for this difference are not clear, it is important to note that our finding remains compatible with the idea that Fezf2 regulates tolerance via control of TRA expression. Importantly, we find that Fezf2 expression in mTECs is regulated by RANK-mediated signaling events. Thus, RANK signaling controls expression of both Aire and Fezf2, the two known regulators of intrathymic TRAs, which demonstrates the requirement for this TNFRSF member in thymic tolerance maps to its regulation of mTECs. In addition, the effect of LTβR on mTECs is separable from its importance in thymic tolerance, further suggesting that LTβR mediates tolerance by another mechanism. Our observation that LTβR controls thymic cDC1/pDC availability in Ltbr−/− mice provides an explanation for this, and fits well with the autoimmune phenotype of these mice and the need for both intrathymic (cDC1) and extrathymic (pDC) cells in tolerance induction (Proietto et al., 2008; Hadeiba et al., 2012; Perry et al., 2014). Interestingly, although thymic DCs regulate both negative selection and T-reg development, DC defects in Ltbr−/− mice correlate with a selective reduction in thymocytes undergoing negative selection. This emphasizes the importance of DC-mediated negative selection as a mechanism of thymic tolerance, which is in agreement with a quantitative requirement for DCs in thymocyte deletion (Anderson et al., 1998; Kroger et al., 2016). Moreover, although mTECs influence thymic DCs (Lei et al., 2011; Spidale et al., 2014), the role of LTβR in formation of the thymic DC pool maps to non-TEC stroma. Indeed, similar to Ltbr−/− mice, we saw thymic DC defects in LTβRMES mice in which LTβR was deleted in the mesenchyme, demonstrating a role for these cells in the regulation of intrathymic DCs. How mesenchymal cells control thymic DCs is not currently known, although it is interesting that in both mesenchyme and endothelium LTβR regulates expression of chemokines and adhesion molecules (Lkhagvasuren et al., 2013; Lucas et al., 2016) that may aid thymus entry of DCs/DC progenitors.
In summary, we examined properties of the thymus medulla that enable it to act as a highly effective and essential site for T cell tolerance. Our finding that LTβR controls thymic DCs identifies a new role for this TNFRSF member in regulating thymus function and demonstrates the importance of negative selection during tolerance induction. Moreover, that correct medulla formation can be separated from its ability to support thymic tolerance raises the possibility that typical medullary topology is closely associated with other functions of this site. These may include aspects of postselection αβT cell development (Webb et al., 2016; Xing et al., 2016) and the regulation of thymic emigration (Zamora-Pineda et al., 2016), as well the medulla’s role in supporting nonconventional T cell lineages (Roberts et al., 2012; White et al., 2014; Jenkinson et al., 2015).
Materials and methods
Mice
All mice were age 8–12 wk on a C57BL/6 background: WT (CD45.2), WT BoyJ (CD45.1), germline LTβR-deficient (Ltbr−/−; Fütterer et al., 1998), Foxn1Cre (Gordon et al., 2007), Wnt1Cre2 (Lewis et al., 2013), and Ltbrfl/fl (Wang et al., 2010) mice. The latter were crossed with Foxn1Cre mice to obtain LTβRTEC mice and with Wnt1Cre2 mice to generate LTβRMES mice. In all experiments, WT C57/BL6 controls were used for Ltbr−/− mice, and Foxn1Cre or Wnt1Cre2 mice were used as controls for LTβRTEC and LTβRMES mice, respectively. Mice were housed at the University of Birmingham Biomedical Services Unit. All experimental procedures were approved by the Birmingham Animal Welfare and Ethical Review Body and were performed in accordance with UK Home Office regulations.
Antibodies and cell sorting
For stromal analysis, thymus samples were digested in collagenase dispase and DNase I (Sigma-Aldrich). Samples were stained with antibodies to the following (from eBioscience unless stated otherwise): CD45 APC (30-F11), EpCAM-1 PerCp Cy5.5 (G8.8), Ly51 PE (6C3), MHCII IA/IE Pacific Blue (M5/114.15.2), Aire Alexa Fluor 488 (5H12), anti-Fezf2 (F441; IBL), and CD80 BV605 (16-10A1; BioLegend). Rabbit anti-CCL21 (Lifespan Biosciences) was detected using Alexa Fluor 647–conjugated goat anti-rabbit (Life Technologies); biotinylated anti-LTβR (3C8) and biotinylated UEA-1 (Vector Laboratories) were detected using streptavidin PE Cy7. For thymocyte analysis, thymic tissue was mechanically disrupted and stained with antibodies to CD4 BV711 (RM4-5; BioLegend), CD8 BV510 (53-6.7; BioLegend), TCRβ APC-Cy7 (H57-597), CD25 APC (PC61.5), CCR7 PE (4B12), and CD5 Biotin (53-7.3) and detected with streptavidin PE Cy7, CD69 PerCp Cy5.5 (H1.2F3), CD3ε PE (clone 145-2C11), Foxp3 FITC (FJK-16s), and cleaved Caspase-3 PE (Asp175, 5AIE; Cell Signaling Technology). Intracellular staining was performed using the Foxp3/transcription factor staining buffer set (eBioscience) according to the manufacturer’s instructions. To detect activated αβT cells in salivary glands, submandibular salivary glands were digested with 30 µg/ml Liberase (Roche) and stained with antibodies to CD45, CD4, CD8, TCRβ, CD69, and CD44 (IM7; eBioscience). For DC analysis, samples were digested using collagenase D and DNase I and stained with antibodies to the following: CD45.2 BV785 (104; BioLegend), PDCA-1 Pacific Blue (129C1; BioLegend), CD11c PeCy7 (N418), Sirpα PE (P84), and CD45.1 APCCy7 (A20). A lineage cocktail containing FITC-labeled antibodies to CD3 (145-2C11), CD19 (eBio1D3), and NK1.1 (PK136) was also used.
Autoantibody detection
Autoantibodies were detected in serum samples obtained from 8–12-wk-old WT, Ltbr−/−, Foxn1Cre, and Ltbrfl/fl mice using a NovaLite rat liver, kidney, and stomach multicomposite kit (Innova Diagnostics). In brief, tissue sections were incubated with 1/80 sera from the indicated mouse strains at room temperature followed by detection with goat (Fab)2 anti–mouse IgG(H+L) FITC (SouthernBiotech). Images were acquired with a DM6000 microscope (Leica Microsystems). Quantification of autoantibodies was performed by two independent staff members based on positive staining intensity on an arbitrary scale of 1–6.
Histology
Liver and salivary gland samples from WT, Ltbr−/−, Foxn1Cre, and LTβRTEC mice were embedded in OCT compound (Sakura Finetek), snap frozen, and sectioned to a thickness of 7 µm. Sections were fixed in acetone for 10 min at 4°C and stained with hematoxylin and eosin, and images were acquired with a Axio ScanZ1 microscope (Zeiss). Cellular infiltrates were quantified by counting cell foci on 3–5 sections per tissue 30–40 µm apart, with infiltrates scored as more than 25 cells clustered together. Software used for analysis was Zeiss Zen Blue.
Confocal microscopy
Snap-frozen thymus tissues were mounted in OCT, sectioned at 7 µm, and fixed in acetone. The following reagents were used: anti-Aire Alexa Fluor 488 (clone 5H12), anti-Fezf2 (F441, IBL), donkey anti–rabbit IgG Alexa Fluor 594 (Thermo Fisher Scientific), ERTR5 (gift from W. van Ewijk, Leiden University Medical Centre, Leiden, Netherlands), goat anti–rat IgM Alexa Fluor 647 (Thermo Fisher Scientific), goat anti–rat IgM Alexa Fluor 488 (Thermo Fisher Scientific), anti-CD11c Biotin (HL3; BD), anti-CD8 Biotin (53-6.7), and streptavidin Alexa Fluor 555 (Thermo Fisher Scientific). All confocal microscopy was performed on a Zeiss Zen 780 microscope. For quantitation, three to four thymus sections were stained, five images were acquired of medullary and cortical areas, and CD11c+ cells were enumerated. All imaging analysis was conducted using Zeiss Zen Black software.
Quantitation of medullary areas
In frozen thymus sections, boundaries of ERTR5+ medullary areas were identified using Zeiss Zen Blue software. Three sections per thymus were analyzed, with a minimum of three mice per strain. Medullary regions were categorized according to area in square millimeters, and the mean number of medullas within each size category was calculated.
BrdU incorporation in thymic DCs
Adult mice were injected i.p. with 1.5 mg BrdU, and tissues were harvested 18 h later. Thymic DC subsets were identified by flow cytometry, and BrdU incorporation was revealed after cell permeabilization using BrdU flow kit (BD PharMingen) and staining with an APC-conjugated anti-BrdU antibody (MoBU-1).
BM chimeras
BM cells from femurs and tibias of Ltbr−/− (CD45.2), C57BL/6 (CD45.2), or BoyJ (CD45.1) mice were T cell depleted using PE-labeled anti-CD3 and anti-PE microbeads and LS columns (Miltenyi Biotec). Host mice were lethally irradiated (two split doses of 500 rad) and reconstituted on the second day of irradiation with 5 × 106 T cell–depleted BM cells from donor mice. Mice were analyzed 8 wk after reconstitution.
Fetal thymus organ cultures
Embryonic day 15 lobes were cultured with 1.35 mM 2 deoxyguanosine (2dGuo) for 7 d (Cowan et al., 2013). 2dGuo FTOCs were stimulated with 2 µg/ml each of anti-RANK (R&D Systems) or anti-LTβR (Banks et al., 2005) for 4 d. Lobes were digested using 0.25% trypsin/0.02% EDTA (Sigma-Aldrich) and depleted of any remaining CD45+ with Dynabeads (Anderson et al., 1993). Cells were then snap frozen for quantitative PCR (qPCR) or permeabilized and stained with antibodies to CD45, EpCAM1, Ly51, Aire, and Fezf2.
qPCR
qPCR was performed exactly as described (Cowan et al., 2013). mRNA levels were normalized to β-actin, fold levels represent replicate reaction mean (±SEM), and data are typical of at least two independently sorted biological samples. Primer sequences were as follows: β-actin QuantiTect Mm Actb 1SG Primer Assay (QT00095242; Qiagen); Aire forward, 5′-TGCATAGCATCCTGGACGGCTTCC-3′, and reverse, 5′-CCTGGGCTGGAGACGCTCTTTGAG-3′; Fezf2 forward, 5′-ACCCAGCTTCCTATCCCCAT-3′, and reverse, 5′-GAGCATTGAACACCTTGCCG-3′; Ccl21 forward, 5′-ATCCCGGCAATCCTGTTCTC-3′, and reverse, 5′-GGGGCTTTGTTTCCCTGGG-3′; Fabp9 forward, 5′-GAATGTGAGCCCCGGAAAGTC-3′, and reverse, 5′-GGATCATTGACCCACCTTCAAA-3′; Ttr forward, 5′-CACCAAATCGTACTGGAAGACA-3′, and reverse, 5′-GTCGTTGGCTGTGAAAACCAC-3′; Krt10 forward, 5′-CAGCTGGCCCTGAAACAATC-3′, and reverse, 5′-AGTTGTTGGTACTCGGCGTT-3′; Spt1 forward, 5′-TACTGAAACTTCTGGAACTGCTGAT-3′, and reverse, 5′-TCGACTGAATCAGAGGAATCAACT-3′; Ins2 forward, 5′-CACCAGCCCTAAGTGATCCG-3′, and reverse, 5′-GCCATGTTGAAACAATAACCTTCCT-3′; and S100a8 forward, 5′-AAATCACCATGCCCTCTACAAG-3′, and reverse, 5′-CCCACTTTTATCACCATCGCAA-3′.
Statistical analysis
All analyses used GraphPad Prism 6.0. To compare expression levels of LTβR in Foxn1Cre, Ltbr−/−, and LTβRTEC mice, one-way ANOVA test was used. In all other cases, we used unpaired Student’s t test. Only p-values <0.05 were noted as significant. Nonsignificant differences were not specified. In all figures, error bars represent SEM.
Online supplemental material
Fig. S1 shows histological and flow cytometric analysis of lymphocyte infiltrates and activated αβT cells in submandibular salivary glands from Ltbr−/−, LTβRTEC, and control mice. Fig. S2 shows flow cytometric analysis of BrdU incorporation in thymic DC subsets from Ltbr−/−, LTβRTEC, and control mice. Fig. S3 shows flow cytometric analysis of DC populations in thymus and spleen of WT:Ltbr−/− and Ltbr−/−:WT BM chimeric mice, harvested 8 wk after transplant.
Supplementary Material
Acknowledgments
We thank Carl Ware for his gift of agonistic anti-LTβR, Paul Garside and James Brewer for helpful discussions, Eric Jenkinson for critical reading of the manuscript, Andrea Bacon and BMSU for animal husbandry, Matthew MacKenzie for cell sorting, and Gary Reynolds for help with histology.
This work was supported by a Medical Research Council Program Grant to G. Anderson, a Biotechnology and Biological Sciences Research Council project grant to W.E. Jenkinson, a Wellcome Trust Seed Award to A.J. White, and an Arthritis Research UK Rheumatoid Arthritis Pathogenesis Centre of Excellence PhD studentship to E.J. Cosway.
The authors declare no competing financial interests.
Author contributions: E.J. Cosway, B. Lucas, K.D. James, S.M. Parnell, M. Carvalho-Gaspar, and A.J. White designed and performed experiments and analyzed data. A.V. Tumanov provided important experimental materials. W.E. Jenkinson and G. Anderson conceived the study and designed experiments, and E.J. Cosway, W.E. Jenkinson, and G. Anderson wrote the manuscript.
Footnotes
Abbreviations used:
- cDC
- conventional DC
- cTEC
- cortical TEC
- LTβR
- lymphotoxin β receptor
- mTEC
- medullary TEC
- pDC
- plasmacytoid DC
- TEC
- thymic epithelial cell
- TNFRSF
- TNF receptor superfamily
- TRA
- tissue-restricted antigen
References
- Abramson J., and Anderson G.. 2017. Thymic epithelial cells. Annu. Rev. Immunol. 35:85–118. 10.1146/annurev-immunol-051116-052320 [DOI] [PubMed] [Google Scholar]
- Akiyama T., Shimo Y., Yanai H., Qin J., Ohshima D., Maruyama Y., Asaumi Y., Kitazawa J., Takayanagi H., Penninger J.M., et al. 2008. The tumor necrosis factor family receptors RANK and CD40 cooperatively establish the thymic medullary microenvironment and self-tolerance. Immunity. 29:423–437. 10.1016/j.immuni.2008.06.015 [DOI] [PubMed] [Google Scholar]
- Akiyama T., Tateishi R., Akiyama N., Yoshinaga R., and Kobayashi T.J.. 2015. Positive and negative regulatory mechanisms for fine-tuning cellularity and functions of medullary thymic epithelial cells. Front. Immunol. 6:461 10.3389/fimmu.2015.00461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson G., and Takahama Y.. 2012. Thymic epithelial cells: Working class heroes for T cell development and repertoire selection. Trends Immunol. 33:256–263. 10.1016/j.it.2012.03.005 [DOI] [PubMed] [Google Scholar]
- Anderson G., Jenkinson E.J., Moore N.C., and Owen J.J.. 1993. MHC class II-positive epithelium and mesenchyme cells are both required for T-cell development in the thymus. Nature. 362:70–73. 10.1038/362070a0 [DOI] [PubMed] [Google Scholar]
- Anderson G., Owen J.J., Moore N.C., and Jenkinson E.J.. 1994. Thymic epithelial cells provide unique signals for positive selection of CD4+CD8+ thymocytes in vitro. J. Exp. Med. 179:2027–2031. 10.1084/jem.179.6.2027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson G., Partington K.M., and Jenkinson E.J.. 1998. Differential effects of peptide diversity and stromal cell type in positive and negative selection in the thymus. J. Immunol. 161:6599–6603. [PubMed] [Google Scholar]
- Anderson M.S., Venanzi E.S., Klein L., Chen Z., Berzins S.P., Turley S.J., von Boehmer H., Bronson R., Dierich A., Benoist C., and Mathis D.. 2002. Projection of an immunological self shadow within the thymus by the aire protein. Science. 298:1395–1401. 10.1126/science.1075958 [DOI] [PubMed] [Google Scholar]
- Aschenbrenner K., D’Cruz L.M., Vollmann E.H., Hinterberger M., Emmerich J., Swee L.K., Rolink A., and Klein L.. 2007. Selection of Foxp3+ regulatory T cells specific for self antigen expressed and presented by Aire+ medullary thymic epithelial cells. Nat. Immunol. 8:351–358. 10.1038/ni1444 [DOI] [PubMed] [Google Scholar]
- Banks T.A., Rickert S., Benedict C.A., Ma L., Ko M., Meier J., Ha W., Schneider K., Granger S.W., Turovskaya O., et al. 2005. A lymphotoxin-IFN-beta axis essential for lymphocyte survival revealed during cytomegalovirus infection. J. Immunol. 174:7217–7225. 10.4049/jimmunol.174.11.7217 [DOI] [PubMed] [Google Scholar]
- Bautista J.L., Lio C.W., Lathrop S.K., Forbush K., Liang Y., Luo J., Rudensky A.Y., and Hsieh C.S.. 2009. Intraclonal competition limits the fate determination of regulatory T cells in the thymus. Nat. Immunol. 10:610–617. 10.1038/ni.1739 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boehm T. 2008. Thymus development and function. Curr. Opin. Immunol. 20:178–184. 10.1016/j.coi.2008.03.001 [DOI] [PubMed] [Google Scholar]
- Boehm T., Scheu S., Pfeffer K., and Bleul C.C.. 2003. Thymic medullary epithelial cell differentiation, thymocyte emigration, and the control of autoimmunity require lympho-epithelial cross talk via LTbetaR. J. Exp. Med. 198:757–769. 10.1084/jem.20030794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonasio R., Scimone M.L., Schaerli P., Grabie N., Lichtman A.H., and von Andrian U.H.. 2006. Clonal deletion of thymocytes by circulating dendritic cells homing to the thymus. Nat. Immunol. 7:1092–1100. 10.1038/ni1385 [DOI] [PubMed] [Google Scholar]
- Cowan J.E., Parnell S.M., Nakamura K., Caamano J.H., Lane P.J., Jenkinson E.J., Jenkinson W.E., and Anderson G.. 2013. The thymic medulla is required for Foxp3+ regulatory but not conventional CD4+ thymocyte development. J. Exp. Med. 210:675–681. 10.1084/jem.20122070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan J.E., McCarthy N.I., Parnell S.M., White A.J., Bacon A., Serge A., Irla M., Lane P.J., Jenkinson E.J., Jenkinson W.E., and Anderson G.. 2014. Differential requirement for CCR4 and CCR7 during the development of innate and adaptive αβT cells in the adult thymus. J. Immunol. 193:1204–1212. 10.4049/jimmunol.1400993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan J.E., McCarthy N.I., and Anderson G.. 2016. CCR7 controls thymus recirculation, but not production and emigration, of Foxp3(+) T cells. Cell Reports. 14:1041–1048. 10.1016/j.celrep.2016.01.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniels M.A., Teixeiro E., Gill J., Hausmann B., Roubaty D., Holmberg K., Werlen G., Holländer G.A., Gascoigne N.R., and Palmer E.. 2006. Thymic selection threshold defined by compartmentalization of Ras/MAPK signalling. Nature. 444:724–729. 10.1038/nature05269 [DOI] [PubMed] [Google Scholar]
- DiPaolo R.J., and Shevach E.M.. 2009. CD4+ T-cell development in a mouse expressing a transgenic TCR derived from a Treg. Eur. J. Immunol. 39:234–240. 10.1002/eji.200838772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrlich L.I., Oh D.Y., Weissman I.L., and Lewis R.S.. 2009. Differential contribution of chemotaxis and substrate restriction to segregation of immature and mature thymocytes. Immunity. 31:986–998. 10.1016/j.immuni.2009.09.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fütterer A., Mink K., Luz A., Kosco-Vilbois M.H., and Pfeffer K.. 1998. The lymphotoxin beta receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity. 9:59–70. 10.1016/S1074-7613(00)80588-9 [DOI] [PubMed] [Google Scholar]
- Gallegos A.M., and Bevan M.J.. 2004. Central tolerance to tissue-specific antigens mediated by direct and indirect antigen presentation. J. Exp. Med. 200:1039–1049. 10.1084/jem.20041457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon J., Xiao S., Hughes B. III, Su D.M., Navarre S.P., Condie B.G., and Manley N.R.. 2007. Specific expression of lacZ and cre recombinase in fetal thymic epithelial cells by multiplex gene targeting at the Foxn1 locus. BMC Dev. Biol. 7:69 10.1186/1471-213X-7-69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray D., Abramson J., Benoist C., and Mathis D.. 2007. Proliferative arrest and rapid turnover of thymic epithelial cells expressing Aire. J. Exp. Med. 204:2521–2528. 10.1084/jem.20070795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadeiba H., Lahl K., Edalati A., Oderup C., Habtezion A., Pachynski R., Nguyen L., Ghodsi A., Adler S., and Butcher E.C.. 2012. Plasmacytoid dendritic cells transport peripheral antigens to the thymus to promote central tolerance. Immunity. 36:438–450. 10.1016/j.immuni.2012.01.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbin O., Bonito A.J., Jeong S., Weinstein E.G., Rahman A.H., Xiong H., Merad M., and Alexandropoulos K.. 2016. Medullary thymic epithelial cells and CD8α(+) dendritic cells coordinately regulate central tolerance but CD8α(+) cells are dispensable for thymic regulatory T cell production. J. Autoimmun. 75:141–149. 10.1016/j.jaut.2016.08.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hikosaka Y., Nitta T., Ohigashi I., Yano K., Ishimaru N., Hayashi Y., Matsumoto M., Matsuo K., Penninger J.M., Takayanagi H., et al. 2008. The cytokine RANKL produced by positively selected thymocytes fosters medullary thymic epithelial cells that express autoimmune regulator. Immunity. 29:438–450. 10.1016/j.immuni.2008.06.018 [DOI] [PubMed] [Google Scholar]
- Hu Z., Lancaster J.N., Sasiponganan C., and Ehrlich L.I.. 2015. CCR4 promotes medullary entry and thymocyte-dendritic cell interactions required for central tolerance. J. Exp. Med. 212:1947–1965. 10.1084/jem.20150178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irla M., Guenot J., Sealy G., Reith W., Imhof B.A., and Sergé A.. 2013. Three-dimensional visualization of the mouse thymus organization in health and immunodeficiency. J. Immunol. 190:586–596. 10.4049/jimmunol.1200119 [DOI] [PubMed] [Google Scholar]
- Jenkinson E.J., Kingston R., Smith C.A., Williams G.T., and Owen J.J.. 1989. Antigen-induced apoptosis in developing T cells: A mechanism for negative selection of the T cell receptor repertoire. Eur. J. Immunol. 19:2175–2177. 10.1002/eji.1830191132 [DOI] [PubMed] [Google Scholar]
- Jenkinson W.E., Jenkinson E.J., and Anderson G.. 2003. Differential requirement for mesenchyme in the proliferation and maturation of thymic epithelial progenitors. J. Exp. Med. 198:325–332. 10.1084/jem.20022135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson W.E., Rossi S.W., Parnell S.M., Jenkinson E.J., and Anderson G.. 2007. PDGFRα-expressing mesenchyme regulates thymus growth and the availability of intrathymic niches. Blood. 109:954–960. 10.1182/blood-2006-05-023143 [DOI] [PubMed] [Google Scholar]
- Jenkinson W.E., McCarthy N.I., Dutton E.E., Cowan J.E., Parnell S.M., White A.J., and Anderson G.. 2015. Natural Th17 cells are critically regulated by functional medullary thymic microenvironments. J. Autoimmun. 63:13–22. 10.1016/j.jaut.2015.06.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabashima K., Banks T.A., Ansel K.M., Lu T.T., Ware C.F., and Cyster J.G.. 2005. Intrinsic lymphotoxin-beta receptor requirement for homeostasis of lymphoid tissue dendritic cells. Immunity. 22:439–450. 10.1016/j.immuni.2005.02.007 [DOI] [PubMed] [Google Scholar]
- Kappler J.W., Roehm N., and Marrack P.. 1987. T cell tolerance by clonal elimination in the thymus. Cell. 49:273–280. 10.1016/0092-8674(87)90568-X [DOI] [PubMed] [Google Scholar]
- Kishimoto H., and Sprent J.. 1997. Negative selection in the thymus includes semimature T cells. J. Exp. Med. 185:263–272. 10.1084/jem.185.2.263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein L., Kyewski B., Allen P.M., and Hogquist K.A.. 2014. Positive and negative selection of the T cell repertoire: What thymocytes see (and don’t see). Nat. Rev. Immunol. 14:377–391. 10.1038/nri3667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroger C.J., Wang B., and Tisch R.. 2016. Temporal increase in thymocyte negative selection parallels enhanced thymic SIRPα(+) DC function. Eur. J. Immunol. 46:2352–2362. 10.1002/eji.201646354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laufer T.M., DeKoning J., Markowitz J.S., Lo D., and Glimcher L.H.. 1996. Unopposed positive selection and autoreactivity in mice expressing class II MHC only on thymic cortex. Nature. 383:81–85. 10.1038/383081a0 [DOI] [PubMed] [Google Scholar]
- Le Borgne M., Ladi E., Dzhagalov I., Herzmark P., Liao Y.F., Chakraborty A.K., and Robey E.A.. 2009. The impact of negative selection on thymocyte migration in the medulla. Nat. Immunol. 10:823–830. 10.1038/ni.1761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei Y., Ripen A.M., Ishimaru N., Ohigashi I., Nagasawa T., Jeker L.T., Bösl M.R., Holländer G.A., Hayashi Y., Malefyt R.W., et al. 2011. Aire-dependent production of XCL1 mediates medullary accumulation of thymic dendritic cells and contributes to regulatory T cell development. J. Exp. Med. 208:383–394. 10.1084/jem.20102327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung M.W., Shen S., and Lafaille J.J.. 2009. TCR-dependent differentiation of thymic Foxp3+ cells is limited to small clonal sizes. J. Exp. Med. 206:2121–2130. 10.1084/jem.20091033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis A.E., Vasudevan H.N., O’Neill A.K., Soriano P., and Bush J.O.. 2013. The widely used Wnt1-Cre transgene causes developmental phenotypes by ectopic activation of Wnt signaling. Dev. Biol. 379:229–234. 10.1016/j.ydbio.2013.04.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lkhagvasuren E., Sakata M., Ohigashi I., and Takahama Y.. 2013. Lymphotoxin β receptor regulates the development of CCL21-expressing subset of postnatal medullary thymic epithelial cells. J. Immunol. 190:5110–5117. 10.4049/jimmunol.1203203 [DOI] [PubMed] [Google Scholar]
- Lucas B., James K.D., Cosway E.J., Parnell S.M., Tumanov A.V., Ware C.F., Jenkinson W.E., and Anderson G.. 2016. Lymphotoxin β receptor controls T cell progenitor entry to the thymus. J. Immunol. 197:2665–2672. 10.4049/jimmunol.1601189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malhotra D., Linehan J.L., Dileepan T., Lee Y.J., Purtha W.E., Lu J.V., Nelson R.W., Fife B.T., Orr H.T., Anderson M.S., et al. 2016. Tolerance is established in polyclonal CD4(+) T cells by distinct mechanisms, according to self-peptide expression patterns. Nat. Immunol. 17:187–195. 10.1038/ni.3327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins V.C., Boehm T., and Bleul C.C.. 2008. Ltbetar signaling does not regulate Aire-dependent transcripts in medullary thymic epithelial cells. J. Immunol. 181:400–407. 10.4049/jimmunol.181.1.400 [DOI] [PubMed] [Google Scholar]
- Murata S., Sasaki K., Kishimoto T., Niwa S., Hayashi H., Takahama Y., and Tanaka K.. 2007. Regulation of CD8+ T cell development by thymus-specific proteasomes. Science. 316:1349–1353. 10.1126/science.1141915 [DOI] [PubMed] [Google Scholar]
- Perry J.S., Lio C.W., Kau A.L., Nutsch K., Yang Z., Gordon J.I., Murphy K.M., and Hsieh C.S.. 2014. Distinct contributions of Aire and antigen-presenting-cell subsets to the generation of self-tolerance in the thymus. Immunity. 41:414–426. 10.1016/j.immuni.2014.08.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proietto A.I., van Dommelen S., Zhou P., Rizzitelli A., D’Amico A., Steptoe R.J., Naik S.H., Lahoud M.H., Liu Y., Zheng P., et al. 2008. Dendritic cells in the thymus contribute to T-regulatory cell induction. Proc. Natl. Acad. Sci. USA. 105:19869–19874. 10.1073/pnas.0810268105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts N.A., White A.J., Jenkinson W.E., Turchinovich G., Nakamura K., Withers D.R., McConnell F.M., Desanti G.E., Benezech C., Parnell S.M., et al. 2012. Rank signaling links the development of invariant γδ T cell progenitors and Aire(+) medullary epithelium. Immunity. 36:427–437. 10.1016/j.immuni.2012.01.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodewald H.R., Paul S., Haller C., Bluethmann H., and Blum C.. 2001. Thymus medulla consisting of epithelial islets each derived from a single progenitor. Nature. 414:763–768. 10.1038/414763a [DOI] [PubMed] [Google Scholar]
- Rossi S.W., Kim M.Y., Leibbrandt A., Parnell S.M., Jenkinson W.E., Glanville S.H., McConnell F.M., Scott H.S., Penninger J.M., Jenkinson E.J., et al. 2007. RANK signals from CD4(+)3(-) inducer cells regulate development of Aire-expressing epithelial cells in the thymic medulla. J. Exp. Med. 204:1267–1272. 10.1084/jem.20062497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spidale N.A., Wang B., and Tisch R.. 2014. Cutting edge: Antigen-specific thymocyte feedback regulates homeostatic thymic conventional dendritic cell maturation. J. Immunol. 193:21–25. 10.4049/jimmunol.1400321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stritesky G.L., Xing Y., Erickson J.R., Kalekar L.A., Wang X., Mueller D.L., Jameson S.C., and Hogquist K.A.. 2013. Murine thymic selection quantified using a unique method to capture deleted T cells. Proc. Natl. Acad. Sci. USA. 110:4679–4684. 10.1073/pnas.1217532110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan L.H.1896. “The tall office building artistically considered,” Lippincott’s Magazine. (March):403–409.
- Tai X., Erman B., Alag A., Mu J., Kimura M., Katz G., Guinter T., McCaughtry T., Etzensperger R., Feigenbaum L., et al. 2013. Foxp3 transcription factor is proapoptotic and lethal to developing regulatory T cells unless counterbalanced by cytokine survival signals. Immunity. 38:1116–1128. 10.1016/j.immuni.2013.02.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takaba H., Morishita Y., Tomofuji Y., Danks L., Nitta T., Komatsu N., Kodama T., and Takayanagi H.. 2015. Fezf2 orchestrates a thymic program of self-antigen expression for immune tolerance. Cell. 163:975–987. 10.1016/j.cell.2015.10.013 [DOI] [PubMed] [Google Scholar]
- Takahama Y., Tanaka K., and Murata S.. 2008. Modest cortex and promiscuous medulla for thymic repertoire formation. Trends Immunol. 29:251–255. 10.1016/j.it.2008.03.003 [DOI] [PubMed] [Google Scholar]
- Ueno T., Saito F., Gray D.H., Kuse S., Hieshima K., Nakano H., Kakiuchi T., Lipp M., Boyd R.L., and Takahama Y.. 2004. CCR7 signals are essential for cortex-medulla migration of developing thymocytes. J. Exp. Med. 200:493–505. 10.1084/jem.20040643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venanzi E.S., Gray D.H., Benoist C., and Mathis D.. 2007. Lymphotoxin pathway and Aire influences on thymic medullary epithelial cells are unconnected. J. Immunol. 179:5693–5700. 10.4049/jimmunol.179.9.5693 [DOI] [PubMed] [Google Scholar]
- Wang Y.G., Kim K.D., Wang J., Yu P., and Fu Y.X.. 2005. Stimulating lymphotoxin beta receptor on the dendritic cells is critical for their homeostasis and expansion. J. Immunol. 175:6997–7002. 10.4049/jimmunol.175.10.6997 [DOI] [PubMed] [Google Scholar]
- Wang Y., Koroleva E.P., Kruglov A.A., Kuprash D.V., Nedospasov S.A., Fu Y.X., and Tumanov A.V.. 2010. Lymphotoxin beta receptor signaling in intestinal epithelial cells orchestrates innate immune responses against mucosal bacterial infection. Immunity. 32:403–413. 10.1016/j.immuni.2010.02.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webb L.V., Ley S.C., and Seddon B.. 2016. TNF activation of NF-κB is essential for development of single-positive thymocytes. J. Exp. Med. 213:1399–1407. 10.1084/jem.20151604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White A.J., Nakamura K., Jenkinson W.E., Saini M., Sinclair C., Seddon B., Narendran P., Pfeffer K., Nitta T., Takahama Y., et al. 2010. Lymphotoxin signals from positively selected thymocytes regulate the terminal differentiation of medullary thymic epithelial cells. J. Immunol. 185:4769–4776. 10.4049/jimmunol.1002151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- White A.J., Jenkinson W.E., Cowan J.E., Parnell S.M., Bacon A., Jones N.D., Jenkinson E.J., and Anderson G.. 2014. An essential role for medullary thymic epithelial cells during the intrathymic development of invariant NKT cells. J. Immunol. 192:2659–2666. 10.4049/jimmunol.1303057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu W., Shi Y., Xia H., Chai Q., Jin C., Ren B., and Zhu M.. 2017. Epithelial LTβR signaling controls the population size of the progenitors of medullary thymic epithelial cells in neonatal mice. Sci. Rep. 7:44481 10.1038/srep44481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xing Y., Wang X., Jameson S.C., and Hogquist K.A.. 2016. Late stages of T cell maturation in the thymus involve NF-κB and tonic type I interferon signaling. Nat. Immunol. 17:565–573. 10.1038/ni.3419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamora-Pineda J., Kumar A., Suh J.H., Zhang M., and Saba J.D.. 2016. Dendritic cell sphingosine-1-phosphate lyase regulates thymic egress. J. Exp. Med. 213:2773–2791. 10.1084/jem.20160287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu M., Chin R.K., Tumanov A.V., Liu X., and Fu Y.X.. 2007. Lymphotoxin beta receptor is required for the migration and selection of autoreactive T cells in thymic medulla. J. Immunol. 179:8069–8075. 10.4049/jimmunol.179.12.8069 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.